Abstract
This is a safety assessment of 6 Eucalyptus globulus (eucalyptus)-derived ingredients as used in cosmetics. The reported functions of the Eucalyptus globulus (eucalyptus)-derived ingredients include abrasive, fragrance ingredient, and skin-conditioning agent (miscellaneous and occlusive). The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the relevant data on these ingredients. Because final product formulations may contain multiple botanicals, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. Industry should use good manufacturing practices to limit impurities. The Panel concluded that Eucalyptus globulus (eucalyptus)-derived ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be non-sensitizing.
Keywords
Introduction
This is a safety assessment of 6 Eucalyptus globulus (eucalyptus)-derived ingredients as used in cosmetics. According to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), the reported functions of the Eucalyptus globulus (eucalyptus)-derived ingredients listed below include abrasive, fragrance ingredient, and skin-conditioning agent (miscellaneous and occlusive; Table 1).
1
Eucalyptus Globulus Leaf Eucalyptus Globulus Leaf Extract Eucalyptus Globulus Leaf Oil Eucalyptus Globulus Leaf Powder Eucalyptus Globulus Leaf/Twig Oil Eucalyptus Globulus Leaf Water Definitions and Reported Functions of Eucalyptus globulus-Derived Ingredients in this Safety Assessment.
1

To avoid redundancy of effort, the Cosmetic Ingredient Review (CIR) may exclude from review ingredients that are known to exclusively function as fragrance ingredients when the ingredient has been or will be evaluated by the Research Institute for Fragrance Materials (RIFM). According to the Dictionary, Eucalyptus Globulus Leaf/Twig Oil and Eucalyptus Globulus Leaf Water are only reported to function as fragrance ingredients. 1 However, communications with RIFM in November 2017 revealed that these ingredients have neither been assessed for safety by the RIFM Expert Panel, nor are these ingredients on RIFM’s prioritized agenda to be reviewed in the foreseeable future. Thus, the Panel is reviewing the safety of these ingredients as part of this assessment.
Plant-derived cosmetic ingredients, such as Eucalyptus globulus (eucalyptus)-derived ingredients, may contain hundreds of constituents, some of which have the potential to cause toxic effects. For example, geraniol is reported to be a potential dermal senisitzer.2-6 In this safety assessment, the Panel is reviewing information available to evaluate the potential toxicity of each of the Eucalyptus globulus-derived ingredients as whole, complex mixtures. Except for specific constituents of concern, the Panel is not reviewing information that may be available to assess the potential toxicity of the individual constituents derived from Eucalyptus globulus. However, Eucalyptus Globulus Leaf Oil consists of not less than 70% (w/w) eucalyptol (also known as cineol, cineole, or 1,8-cineole), a cosmetic ingredient that has not been reviewed by the Panel.1,7 Since the content of eucalyptol is so high, it is appropriate to include relevant toxicity data on eucalyptol as supporting information for the Eucalyptus globulus (eucalyptus)-derived ingredients. Representative data are summarized in the relevant sections in this safety assessment. While the data are being considered in evaluating the safety of Eucalyptus globulus (eucalyptus)-derived ingredients, the safety of eucalyptol as used in cosmetics is not being assessed in this report.
The Panel has reported on related ingredients that can be used to support the safety of the Eucalyptus globulus-derived ingredients. Phytosterols were found in chloroform and methanol extracts of Eucalyptus globulus leaves. 8 The Panel reviewed the safety of phytosterols, which are plant-derived sterols, in 2013 and concluded that the phytosterol cosmetic ingredients are safe as used. 9
The names of the cosmetic ingredients in this report are written in accordance with the International Nomenclature Cosmetic Ingredient (INCI) naming conventions as shown above, i.e., capitalized without italics and without abbreviations. When referring to the plant from which these ingredients are derived, the standard taxonomic practice of using italics is followed (e.g., Eucalyptus globulus). Often in the published literature, the information provided is not sufficient to determine how well the tested substance represents the cosmetic ingredient. Therefore, the taxonomic name is used or it is noted that the similarity could not be determined, unless it is clear that the test substance is similar to cosmetic ingredients. If the tested substance is a cosmetic ingredient, then the INCI name is used.
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics and chemicals industries, as well as by other interested parties.
Some of the data included in this safety assessment were found on the European Chemicals Agency (ECHA) 10 and the International Program of Chemical Safety (INCHEM) 11 websites. In this safety assessment, ECHA and INCHEM are cited as the references for summaries of information obtained from these websites. Also referenced in this safety assessment are summary data found in reports made publicly available by the World Health Organization (WHO) 7 and the European Medicines Agency (EMA) Products Committee on Herbal Medicinal Products (HMPC). 12
Chemistry
Definition
The definitions of the ingredients in this safety assessment are provided in Table 1. The genus Eucalyptus contains more than 750 species (i.e., Eucalyptus cordata, Eucalyptus diversifolia, Eucalyptus gigantea, Eucalyptus glauca, and Eucalyptus pulverulenta, etc.) and the term “eucalyptus” in the literature can refer to any or all of these. 13 There are four subspecies of Eucalyptus globulus: bicostata, globulus, maidenii, and pseudoglobulus. 14 It is not known if only one or all of these are used in cosmetics. This review cites studies where it can be reasonably certain that the test substance is Eucalyptus globulus. The Dictionary defines Eucalyptus Globulus Leaf Oil as the volatile oil obtained from the leaves of Eucalyptus globulus and other species of Eucalyptus. “Eucalyptus oil” may be extracted from any Eucalyptus species that is rich in eucalyptol. 7 The other main species that Eucalyptus essential oil is extracted from are Eucalyptus polybractea and Eucalyptus smithii, which contain a minimum of 70% eucalyptol. 15
In addition, according to the Dictionary, the CAS number that is associated with Eucalyptus Globulus Leaf Oil (defined above) is 8000-48-4. However, according to the Chemical Abstracts Service (CAS) database, the substance associated with CAS number 8000-48-4 is defined as “extractives and their physically modified derivatives of Eucalyptus, Myrtaceae.” Also, according to the Dictionary, CAS number 84625-32-1 is associated with Eucalyptus Globulus Leaf Extract, which is defined as the extract of the leaves of Eucalyptus globulus. However, according to the CAS database, the substance associated with this CAS number is defined as “extractives and their physically modified derivatives such as tinctures, concretes, absolutes, essential oils, oleoresins, terpenes, terpene-free fractions, distillates, residues, etc., obtained from Eucalyptus globulus, Myrtaceae.”
Plant Identification
Eucalyptus globulus, also referred to as blue gum or Tasmanian blue gum tree, is a member of the Myrtaceae family. These plants are evergreens that are indigenous to Tasmania and southeastern Australia, and are cultivated in subtropical regions of the world including Africa, South America, Asia, southern Europe (Spain and the Black Sea region) and the U.S.7,12
Eucalyptus globulus is a large tree with smooth, very pale or ash-grey bark, which grows up to 20 m high.7,12,16-20 The bark types vary with plant age, and include: stringy bark, ironbark, tessellated bark, box, and ribbon. The bark cells are able to photosynthesize in the absence of foliage, giving the plant an increased ability to re-fix internal carbon dioxide following partial defoliation. This allows the tree to grow in less-than-ideal climates. Branchlets are quadrangular or glaucous. Eucalyptus leaves are ensiform (shaped like a sword blade; long and narrow with sharp edges and a pointed tip), usually ranging from 15 to 30 cm, and possibly up to 40 cm, long and 5 cm wide. The leaves, which are bluish-green in hue, alternate and are vertical. The leaves are studded with brown lenticels and colorless glands containing fragrant volatile oil. Younger leaves tend to have higher oil content than mature ones; however, eucalyptol content is higher in mature leaves. The flowers, which are present most of the year, have very short pedicels, mostly umbellate, sometimes 2 to 3 in a fascicle. The flowers consist of several white fluffy stamens (12 mm long), which are numerous, threadlike, white anthers opening in broad slits with round gland. The fruit has numerous small seeds and is enclosed by a cup-shaped receptacle. The root system grows rapidly and uses large quantities of water; it consists of a strong taproot, at least 6 ft (1.8 m) in length, and lateral roots that can spread up to 100 ft (30.5 m).
Physical and Chemical Properties
Chemical Properties of Eucalyptus globulus-Derived Ingredients.

Method of Manufacture
The definitions of several of the Eucalyptus globulus-derived ingredients in this safety assessment give insight into possible methods of manufacture. For example, the definition of Eucalyptus Globulus Leaf Water states that this ingredient is an aqueous solution of the steam distillate obtained from the leaves of Eucalyptus globulus. 1
Methods of Manufacture Reported in the Literature.

Composition/Constituents
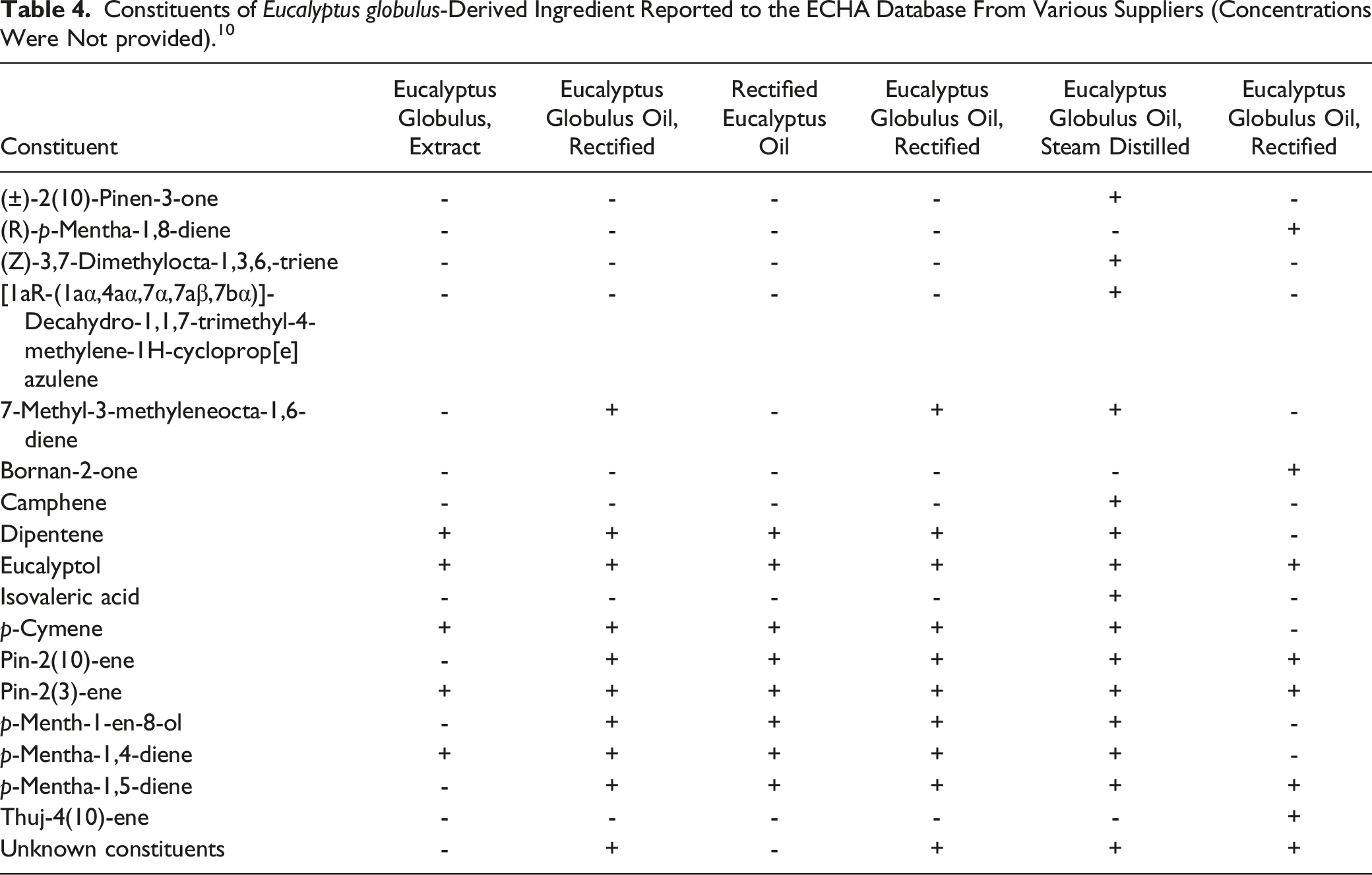
Constituents of Eucalyptus globulus-Derived Ingredient Reported to the ECHA Database From Various Suppliers (Concentrations Were Not provided). 10
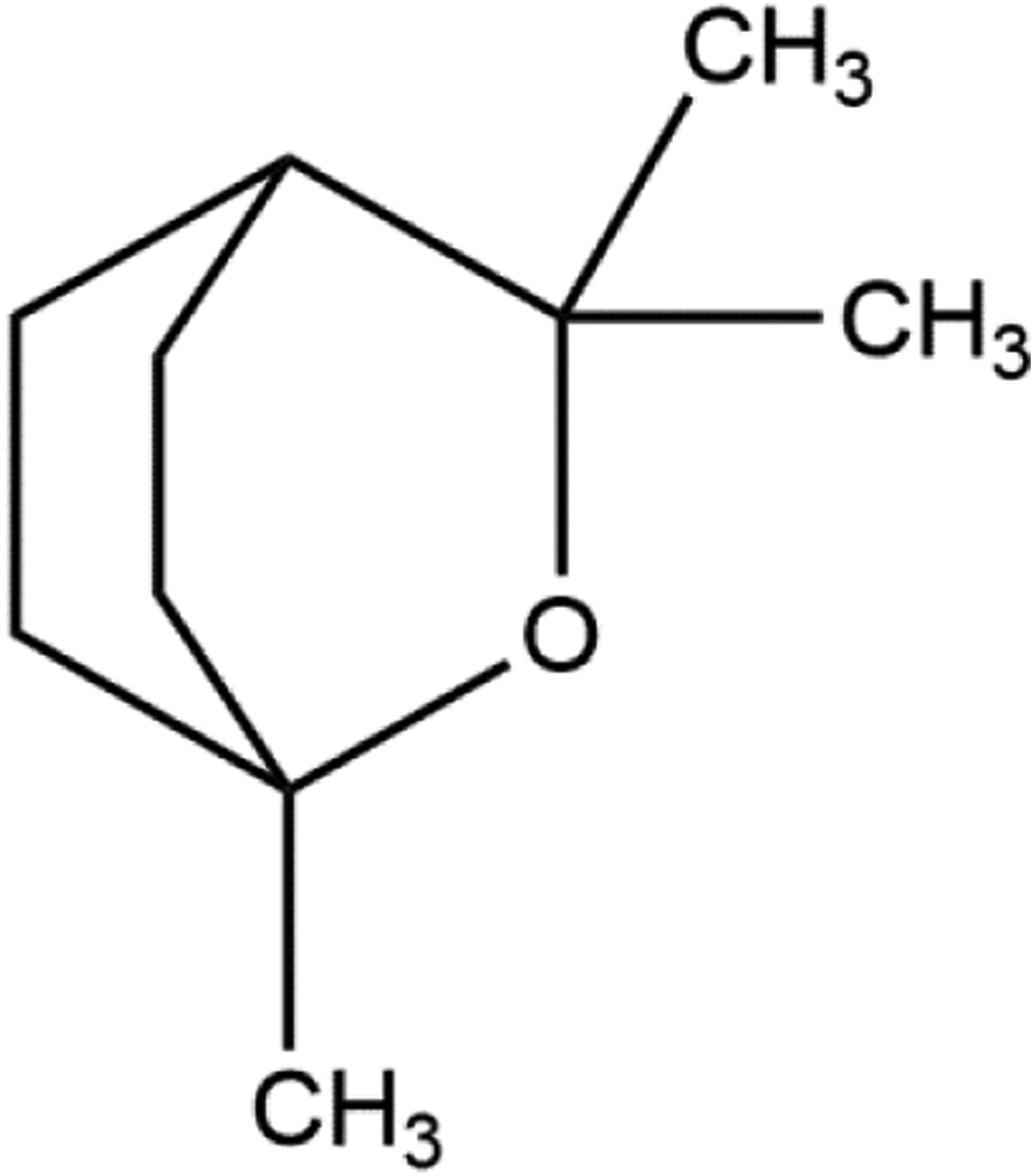
Reported primary component of Eucalyptus, eucalyptol.
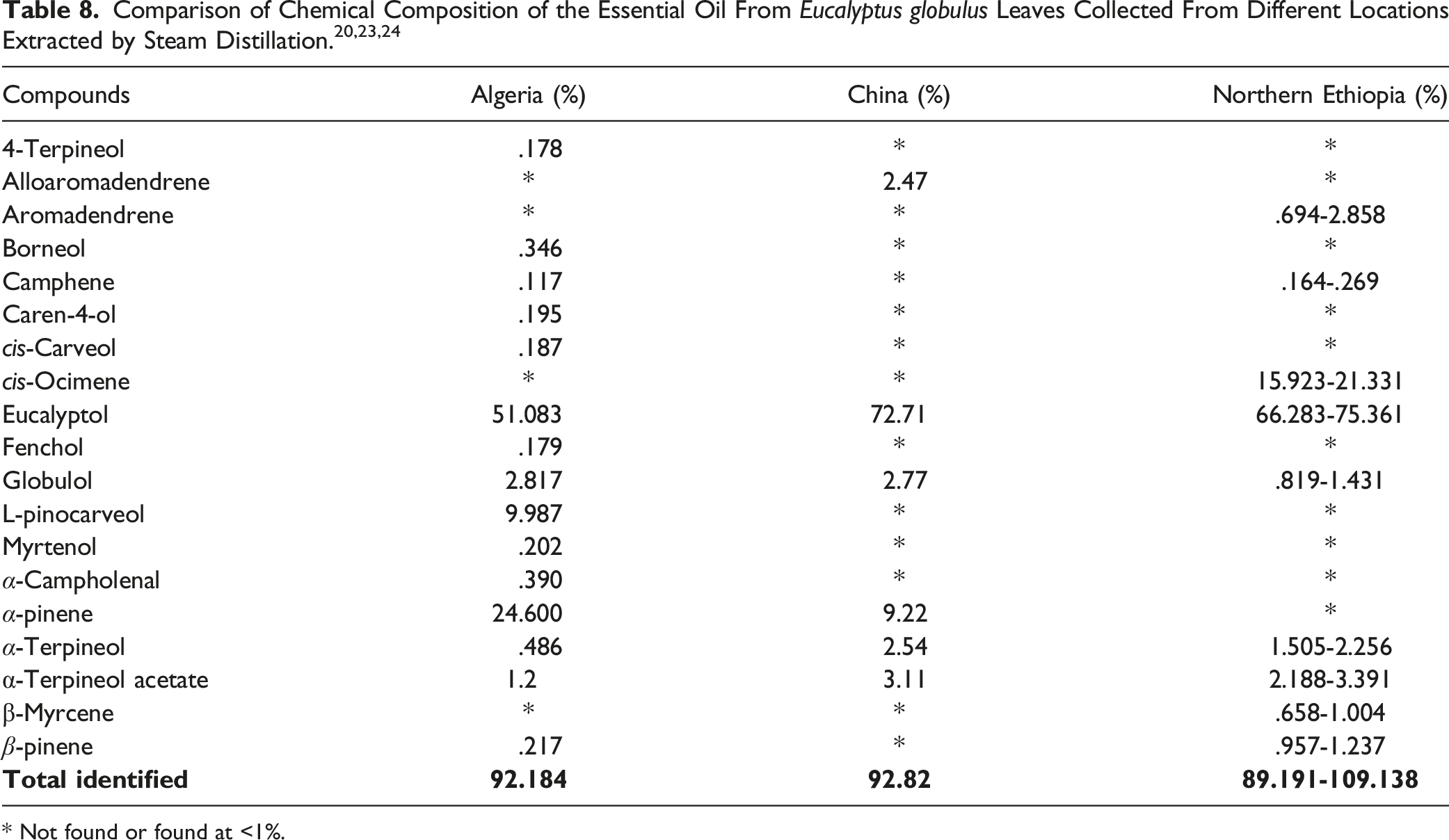
Reported concentrations of Eucalyptus globulus essential oil and its constituents vary in the literature. Eucalyptus globulus leaves contain not less than 2% (v/w) essential oil, consisting of not less than 70% (w/w) eucalyptol. 7 Another report states that fresh leaves of Eucalyptus globulus contained 54% to 61% eucalyptol, 19.5% to 24.3% α-pinene, 6.7% to 9.1% limonene, 2.1% to 5.4% α-terpinyl acetate, and 3.6% to 7.7% sesquiterpenes. 12 The author attributed the differences observed among the different preparation methods to potential hydrolyses during steam distillation. Another author reported that fresh leaves of Eucalyptus globulus contain only 1.87% volatile oil with 35.7% eucalyptol. 12
Constituent Groups Found in Eucalyptus globulus Leaf Extracts Using Different Extract Mediums. 8

Eucalyptus Globulus Leaf Extract
A supplier reports that the aqueous Eucalyptus Globulus Leaf Extract contains no eucalyptol. 22
Eucalyptus Globulus Leaf Oil
The Constituents of Eucalyptus Globulus Leaf Oil Reported by a Supplier. 91

The Variability of Constituent Percentages of Eucalyptus Globulus Leaf Oil (Essential Oil) at 1% or Greater. 6

* Not found or found at <1%.
Eucalyptus Globulus Leaf/Twig Oil
In general, the major constituent of Eucalyptus Globulus Leaf/Twig Oil is eucalyptol (54% to 95%). 7 In addition, there are reported to be moderate amounts of α-pinene (2.6%), p-cymene (2.7%), aromadendrene, cuminaldehyde, globulol and pinocarveol. Eucalyptus Globulus Leaf/Twig Oil for medicinal use contains not less than 70% (w/w) eucalyptol. Eucalyptus Globulus Leaf/Twig Oil also contains monoterpenes such as β-pinene, limonene, geraniol and camphene. 12
Constituents of Concern
Constituents of the Eucalyptus globulus Plant That May be of Concern.

Potential sensitizers include geraniol2-6 (found in the essential oil) and the hydroperoxides of limonene2,6,25 (leaf essential oil) and linalool2,26 (leaf and leaf essential oil). 27
Other constituents of concern found in the Eucalyptus globulus plant are myrcene (leaf essential oil), pinene (essential oil, leaf, and leaf essential oil) and quercetin (leaf and stem bark). 27 These constituents are potential carcinogens or are genotoxic.28-30,30-33
Constituents of Eucalyptus globulus Leaves and Oil That Have IFRA Standards. 34

IFRA - International Fragrance Association.
* Use categories are based on types of contact (e.g., skin, lips), length of contact (e.g., leave-on, rinse-off), or type of use (e.g., mouthwash).
Impurities
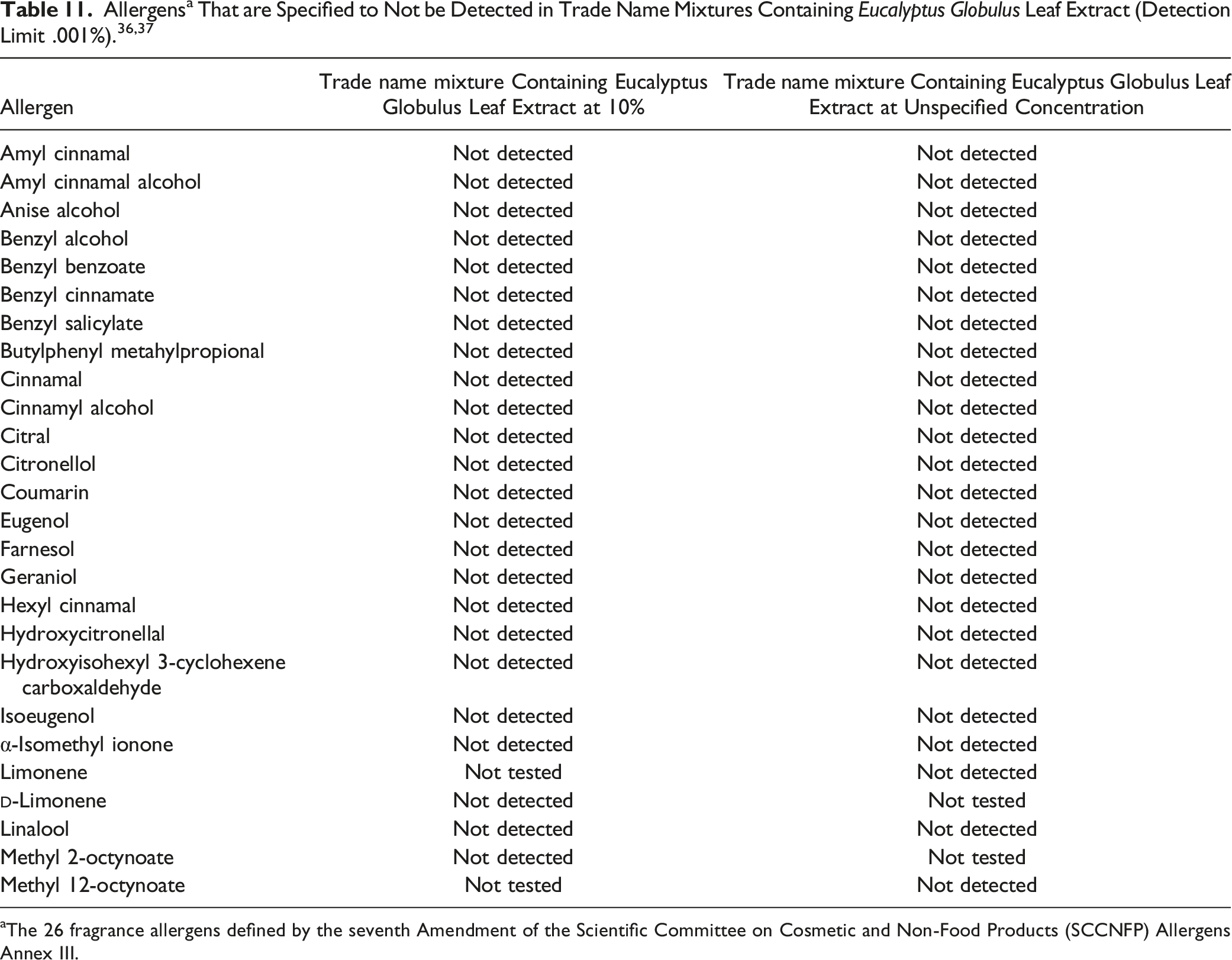
aThe 26 fragrance allergens defined by the seventh Amendment of the Scientific Committee on Cosmetic and Non-Food Products (SCCNFP) Allergens Annex III.
Another supplier reported specifications for another trade name mixture containing Eucalyptus Globulus Leaf Extract (concentration not specified; Table 11). 37 This mixture was also reported to not contain allergens and not to contain antimony (detection limit .021 gm/L), arsenic (.055 mg/L), cadmium (.004 mg/L), chromium (.010 mg/L) iron (.087 mg/L), lead (.015 mg/L), mercury (.0002 mg/L) or nickel (.016 mg/L). There were no residual pesticides detected and a total bacterial count limit of < 100 organisms/g (opg).
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentration by product category.
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
NR = Not Reported; NS = Not Surveyed; Totals = Rinse-off + Leave-on + Diluted for Bath Product Uses.
aIt is possible these products
bNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
cIt is possible these products
d“Eucalyptus” and “eucalyptus extract” are not INCI names but were reported in the VCRP. It is not known if these correspond to cosmetic ingredients in this report.
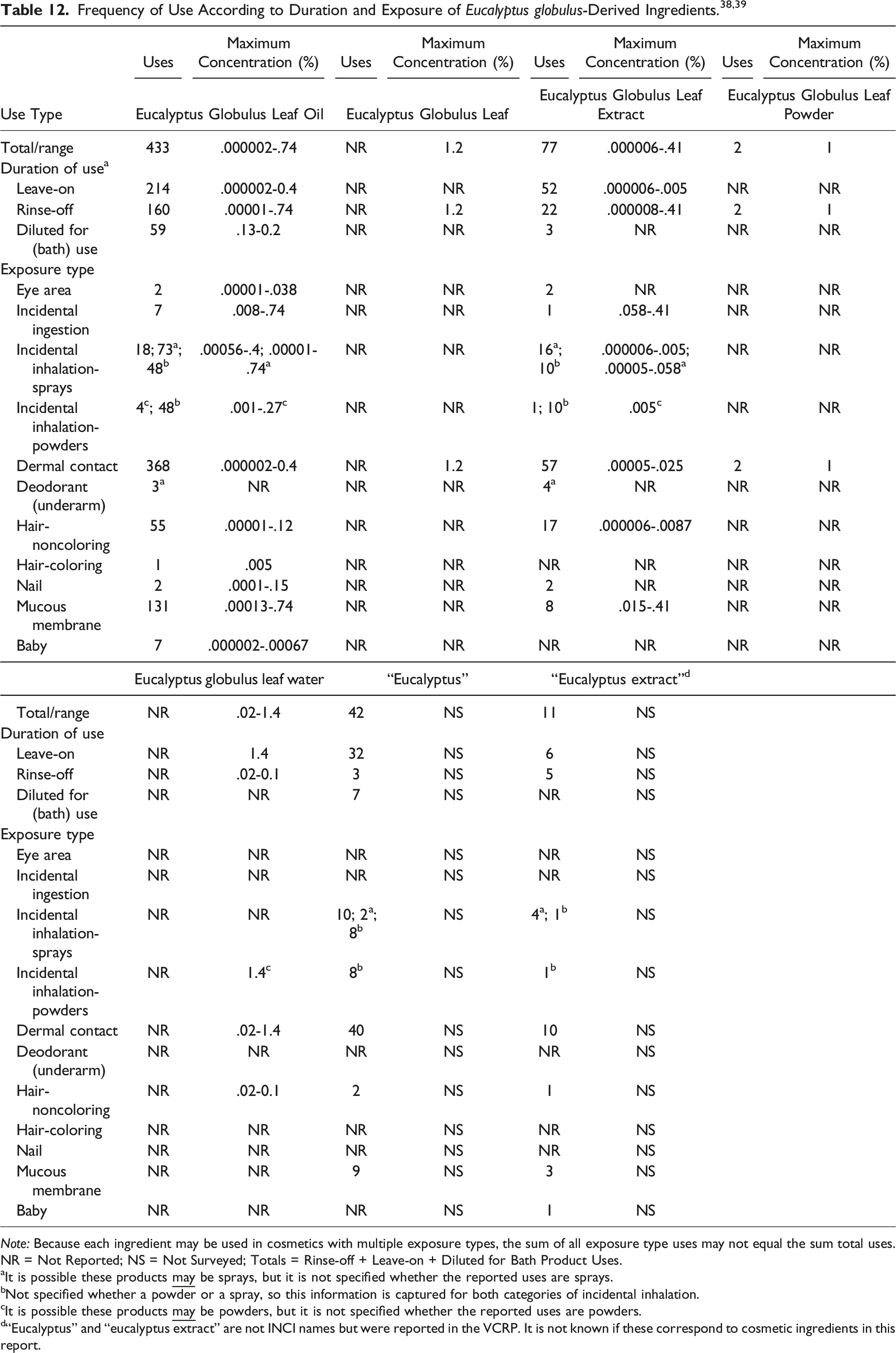
The results of a concentration of use survey conducted by the Council in 2017, and revised in 2018, indicate Eucalyptus Globulus Leaf Water is used at up to 1.4%, with the maximum use reported in face and neck products. 39 The rest of the ingredients with reported concentrations of use are used at a maximum of .4% in leave-on products or 1.2% in rinse-off products.
In some cases, no uses were reported in the VCRP, but concentration of use data were received from industry. For instance, although Eucalyptus Globulus Leaf Water has concentrations of use reported in several categories, there are no reported uses in the VCRP. It should be presumed there is at least one use in every category for which a concentration is reported.
One Eucalyptus globulus-derived ingredient, Eucalyptus Globulus Leaf/Twig Oil, had no uses reported in the VCRP or industry survey.
Eucalyptus Globulus Leaf Oil and Eucalyptus Globulus Leaf Extract are reported to be used in products that are used near the eyes (e.g., eye lotions at up to .038% Eucalyptus Globulus Leaf Oil), and in products that may be ingested and come in contact with mucus membranes (e.g., mouthwashes and breath fresheners at up to .74% Eucalyptus Globulus Leaf Oil). Eucalyptus Globulus Leaf Oil is reported to be used in baby products at up to .00067%.
Additionally, some of the Eucalyptus globulus-derived ingredients are used in cosmetic sprays and could possibly be inhaled; for example, Eucalyptus Globulus Leaf Oil is reported to be used in fragrance products at up to .4% and hair sprays at up to .002%. In practice, most droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles < 10 µm compared with pump sprays.40,41 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.42,43
The cosmetic ingredient SD alcohol 38-B may be denatured with any of several essential oils, including Eucalyptus globulus oil. 1 Essential oils used as a denaturant must meet National Formulary (NF) specifications. [27 CFR 21.92] Specifications for eucalyptol (there are no specific standards for Eucalyptus globulus oil) include a specific gravity between .921 and .924, congealing temperature ≥ 0, distilling temperature range between 174 and 177°C, and no detectable phenols. 44
The FDA lists “Eucalyptus globulus” as a non-traditional preservative for cosmetics in its Compliance Program Guidance Manual. 45
None of the Eucalyptus globulus-derived ingredients named in the report are restricted from use in any way under the rules governing cosmetic products in the European Union. 46
Non-Cosmetic
Food
In the US, Eucalyptus globulus is not generally used for human food, but as an additive. Eucalyptus globulus (Eucalyptus globulus Labill) leaves are food additives permitted for direct addition to food for human consumption as a flavoring agent. [21 CFR 172.510] Australian Aborigines use the roots as a source of water, and cook and eat the roots. Dried Eucalyptus globulus leaves are fed to horses, cattle, and sheep. 17
As a chemical residue in food, an exemption from the requirement of tolerance is established for residues of Eucalyptus globulus oil in or on honey, honeycomb, and honeycomb when used at 2 g or less Eucalyptus globulus oil per hive, where the eucalyptus oil contains 80% or more eucalyptol. [40 CFR 180.1271]
Eucalyptol (for inference to Eucalyptus Globulus Leaf Oil)
The European Commission Scientific Committee on Food (SCF) concluded that the available toxicological studies of eucalyptol are limited and inadequate to derive an acceptable daily intake (ADI). 47 However, the available animal data do not indicate a cause of concern associated with the daily intake from food, estimated from the small amount of information available.
Drugs
Eucalyptus globulus oil may be used in over-the-counter (OTC) products that treat nasal decongestion (in a lozenge or mouthwash), sinusitis, dermal irritation, fever blisters/cold sores, and poison ivy, oak and sumac, and in astringent and external analgesic drug products. However, based on evidence currently available, there are inadequate data to establish general recognition of the safety and effectiveness of these ingredients for the specified uses. [21 CFR 310.545]
Eucalyptus globulus oil (1.2 to 1.3%) is permitted in combinations containing nasal decongestant and an analgesic-antipyretic. [21 CFR 341.40]
Combinations containing camphor, menthol, and Eucalyptus globulus oil are permitted for use as active ingredients in cold, cough, allergy, bronchodilator, and anti-asthmatic OTC drugs when so labeled. [21 CFR 341.85]
Eucalyptus globulus oil has been used in OTC smoking deterrents, but there is a lack of adequate data to establish general recognition of the safety and effectiveness. [21 CFR 310.544]
Eucalyptus Globulus Leaf/Twig Oil is used orally to treat catarrh and coughs, and dermally as a rubefacient for treatment of rheumatic complaints in traditional medicine. 7 Other traditional medicinal uses that are not supported by experimentation or clinical data are treatment of cystitis, diabetes, gastritis, kidney disease (unspecified), neuralgia, laryngitis, leucorrhoea, malaria, pimples, ringworm, sinusitis, wounds, ulcers of the skin, urethritis and vaginitis.
Daily oral dosages of eucalyptus oil obtained by steam distillation range from .3 to .6 mL essential oil or equivalent preparations. 7 Examples of oral dosages include: one capsule of 100 to 200 mg, 2 to 5 times daily; one lozenge of .2 to 15.0 mg dissolved slowly in the mouth, every 30 to 60 min; and a mouthwash as 20 mL of a .91 mg/mL solution, gargled twice daily. The dose for administration by inhalation is 12 drops Eucalyptus globulus oil/150 mL boiling water. For dermal use, daily dosage consists of several drops or 30 mL of the essential oil in 500 mL lukewarm water rubbed into the skin; 5% to 20% of the essential oil in liquid and semisolid preparations; or 5% to 10% in hydroalcoholic preparations. Since there are no sufficient clinical data on children, the EMA states that oral use should be restricted to adolescents over 12 yr of age and the cutaneous use should be limited to children over 4 yr of age. 12
It is recommended that the maximum adult daily oral dose is 600 mg and the maximum dermal use level is 20%. 6 It is noted that essential oils high in eucalyptol can cause central nervous system (CNS) and breathing problems in young children and recommend that the essential oil not be applied to or near the face of infants or children under 10 years of age.
Health Canada restricts the use of Eucalyptus globulus leaf essential oil to 1% to 5% for use as a massage oil (covering more than 10% of the body surface), but may be used up to 25% for a local (less than 10% of the body surface) use. 48 The oil may also be used in aromatherapy to help relieve joint/muscle pain associated with sprain/strain/rheumatoid arthritis, to help relieve headache, and to help relieve colds/cough.
Eucalyptol (for inference to Eucalyptus Globulus Leaf Oil)
Eucalyptol may be used in lozenges and mouthwash that act as nasal decongestants, expectorants, dandruff/seborrheic dermatitis/psoriasis drug products, and oral care products. Based on evidence currently available, there are inadequate data to establish general recognition of the safety and effectiveness of these preparations for the specified uses. [21 CFR 310.545]
Other
Eucalyptus globulus oil may be used in the manufacture of denatured alcohol, rum, and other denatured spirits. [27 CFR 21.65; 27 CFR 21.151]
Toxicokinetic Studies
Obtaining data on the toxicokinetics of unknown, complex mixtures, such as botanicals, would be impractical. However, if the compositions are well understood, including the concentrations of constituents, such studies may be useful.
Penetration Enhancement
In Vitro
Dermal Penetration Enhancement Studies of Eucalyptus Globulus Leaf Oil.

5-FU = 5-Fluorouracil; CHG = chlorhexidine digluconate; DMSO = dimethyl sulfoxide; ER = enhancement ratio; HPLC = high-performance liquid chromatography; PBS = phosphate buffered saline; TMP = 2,3,5,6-tetramethylpyrazine.
Generally, dermal penetration of chlorhexidine digluconate (CHG) through human skin samples over 24 h increased in a manner dependent on the concentration of Eucalyptus Globulus Leaf Oil. 49 Eucalyptus Globulus Leaf Oil (82.9% eucalyptol) at 5% facilitated greater CHG skin penetration to the deeper layers of the skin (below 300 μm) and 10% (v/v) Eucalyptus Globulus Leaf Oil enhanced CHG skin penetration in the upper 900 μm. CHG, with and without 50% Eucalyptus Globulus Leaf Oil, was detected at negligible levels in the receptor compartment over 24 h, suggesting that CHG did not permeate through the full skin thickness, and was retained within the tissue.
When the dermal penetration enhancement of Eucalyptus Globulus Leaf Oil (2.5, 5, or 7.5%) was tested with 2,3,5,6-tetramethylpyrazine (TMP), the enhancement ratios for human skin were 3.38, 4.47, and 4.64, respectively. 50 The TMP flux across the human chest skin with 5% Eucalyptus Globulus Leaf Oil was 17-fold greater (346.0 mg/cm2/h) than the flux (20.1 mg/cm2/h) of a saturated solution of TMP without the oil. The receptor fluid was water. When the ability of Eucalyptus Globulus Leaf Oil (80% to 85% eucalyptol) to enhance the dermal penetration of ketorolac was evaluated using a dermal patch across abdominal rat skin, the enhancement ratios were 1.80, 3.04, and 3.68 for 5, 7.5, and 10%, respectively. 51 Eucalyptus Globulus Leaf Oil increased the dermal penetration of 5-fluorouracil (5-FU) through rat skin when using 2-chamber diffusion cells; the enhancement ratios ranged from 58.49 to 82.55, depending on temperature (100 - 140°C). 52
Absorption, Distribution, Metabolism, and Excretion
Animal
Oral
Eucalyptol (for inference to Eucalyptus Globulus Leaf Oil)
Eucalyptol undergoes oxidation in vivo with the formation of hydroxycineole which is excreted as a glucuronide.53,54 In rats, 2-hydroxycineole, 3-hydroxycineole, and 1,8-dihydroxycineol-9-oic acid were identified as main urinary metabolites. After oral administration to brushtail possums (Trichosurus vulpecula), p-cresol, 9-hydroxycineole, and cineol-9-oic acid were found in urine. Rabbits given eucalyptol by gavage excreted 2-exo- and 2-endo-hydroxycineole as well as 3-exo- and 3-endo-hydroxycineole in the urine.
A gavage study on the metabolism of eucalyptol from rosemary oil (4, 20, or 40 μL rosemary oil containing 39% of eucalyptol (approximately equivalent to 52, 260, and 520 mg/kg eucalyptol, respectively) was conducted in NMRI mice (n = 5).55,56 The rosemary oil was administered in an oil/water emulsion with 10% Tween 80 (.3 mL). Controls were administered water and Tween 80. Blood samples were collected at intervals over 90 min. There was rapid absorption and metabolism; blood concentrations of eucalyptol reached a peak 5 min. At the 52 mg/kg dose, the blood concentration of eucalyptol peaked at approximately 4.5 nL/g and then dropped to close to undetectable over 90 min. At 260 mg/kg, blood concentrations remained constant (between 7.0 and 10.1 nL/g) over the next 90 min, while at 520 mg/kg, the peak blood concentration peaked at 18.0 nL/g and then dropped to 60% of the maximum value after 10 min and remained in that range (9.1 to 12.2 nL/g) for the following 80 min. The slowing of the metabolism of eucalyptol at higher doses suggests saturation.
Inhalation
Eucalyptol (for inference to Eucalyptus Globulus Leaf Oil)
An inhalation study on the metabolism of eucalyptol from rosemary oil (.1, .3, .4, .5, or .6 mL rosemary oil containing 39% of eucalyptol; approximately equivalent to .039, .117, .156, .195, or .234 mL eucalyptol) was conducted in NMRI mice (n = 20 to 30). 56 The mice (5/cage) were placed into air-tight cages in which rosemary oil was applied to filter paper and allowed to evaporate over an hour. The concentrations of rosemary oil and eucalyptol in the breathing air were 35 to 40 nL/mL and 13.7 to 15.6 nL/mL, respectively. Soda lime was used to remove the carbon dioxide, and calcium chloride was used to remove humidity. Oxygen was replaced through an opening in the cage. After an hour, the mice were killed and blood samples were collected. Immediately after exposure, eucalyptol blood concentration was approximately 4.5, 10, 10, 12, and 16 nL/g after doses of .039, .117, .156, .195, or .234 mL eucalyptol/cage, respectively. In a second experiment, mice (n = 5 to 10) were exposed to .195 mL eucalyptol. Blood samples were collected at intervals over 120 min. The eucalyptol blood concentration peaked at 16.2 nL/g and the elimination of the eucalyptol was biphasic. The concentration of eucalyptol dropped to approximately 5 nL/g in 30 min and approximately 1.5 nL/g at 120 min; there was a short half-life of 6 min during the first 10 min and a half-life of about 45 min during a second phase.
Toxicological Studies
Acute Dose Toxicity
Acute Dermal and Oral Toxicity.

Animal
Dermal
The dermal LD50 of Eucalyptus Globulus Leaf Oil was > 5000 mg/kg in rabbits. 10 There were no mortalities or signs of toxicity. The dermal LD50 of eucalyptol was > 5000 g/kg in rabbits. 54
Oral
Eucalyptus Globulus Leaf Oil administered to mice (n = 10) by gavage had an oral LD50 of 3320 mg/kg. 10 The LD50 of an aqueous emulsion comprising 5% Eucalyptus Globulus Leaf Oil in mice was between 3 and 3.5 mL/kg. 57 In the 3.0 and 3.5 mL/kg groups, 1 of 6 and 4 of 6 mice died within 24 h of dosing, respectively. The oral LD50s of Eucalyptus Globulus Leaf Oil were 3811.5 mg/kg, 58 2334.3 mg/kg, 59 and 4400 mg/kg10 in three different studies in rats. Mice orally administered a single dose of eucalyptol (500 mg/kg) had an increase in liver enzyme activity. 53 Reported oral LD50s of eucalyptol in rats were 2480 mg/kg and 1560 mg/kg.53,54
Inhalation
Eucalyptus Globulus Leaf Oil and Eucalyptus Globulus Leaf/Twig Oil
Male and female rabbits (n = 8 to 14) were lightly anesthetized and cannulated through the trachea. 60 A second collecting tube was also installed. Water vapor from a boiling water bath, mixed with ambient air and cooled to the body temperature of the rabbits, was inhaled directly into the rabbit’s trachea. Respiratory tract fluid was collected for a control period of 2 to 4 h. The collecting tracheal tube was then replaced by a new empty tube, and Eucalyptus globulus oil (.4 to 19 683 mg/kg body weight in ethyl alcohol; not known if leaf or leaf/twig oil) was added to the boiling water bath; respiratory tract fluid was collected from each rabbit for a subsequent 4 to 6 h or until the rabbit died. The highest dose caused deaths and significantly augmented the output of respiratory tract fluid; lower doses had no effect on the volume of respiratory tract fluid. Doses of 729 to 19 683 mg/kg produced increasingly lower values for the specific gravity of collected respiratory tract fluid and the two highest doses augmented the concentration of total solids and insoluble mucus. Doses, which are considered to be in the therapeutic range for humans (3 to 243 mg/kg), were repeated once each year in 2 successive years, and in each instance produced no significant change in any parameter measured. Local irritation of the respiratory tract appeared after administration of the two highest doses.
Eucalyptol (for inference to Eucalyptus Globulus Leaf Oil)
Ovalbumin (OVA)-sensitized guinea pigs were exposed to aerosolized eucalyptol for 15 min. The eucalyptol (1 mg/mL) was aerosolized using a nebulizer into a box (21 × 20 × 30 cm3). 61 Approximately 3 min later, the guinea pigs were exposed to aerosolized saline for 15 min. The control group was exposed to aerosolized saline for both exposures. The guinea pigs were killed 24 h later and inflammatory parameters such as tracheal responsiveness to carbachol, cytokine levels and myeloperoxidase activity on bronchoalveolar lavage fluid, as well as mucociliary clearance were evaluated. There were no differences in the numbers of eosinophils, neutrophils, lymphocytes and macrophages in the treatment group compared to controls. Cytokine levels (IL-1, TNFα, and IL-10) were also similar between the two groups.
Human
Oral
Eucalyptus Globulus Leaf Oil
The literature on the oral toxicity in humans of Eucalyptus Globulus Leaf Oil is generally old (years 1900 – 1965). The following is a summary of this information. The substances are referred to as eucalyptus, eucalyptus oil, and similar names, and little or no information on source plant parts, method of manufacture, or concentration/purity is provided.
The probable oral lethal dose for adult humans is .05 to .5 mL/kg. 11 The oral ingestion of Eucalyptus Globulus Leaf Oil may initially result in a burning sensation in the mouth, vomiting, diarrhea, and epigastric pain. Vomiting may be delayed for periods varying from minutes up to 4 h. Permanent sequelae following recovery from the acute phase have not been reported, although symptoms such as drowsiness, ataxia, and fatigue may occasionally persist for 1 to 2 wk. Those subjects who suffered severe gastric irritation who promptly vomited recovered better but almost all made an uneventful recovery within 24 h. Recovery may be interrupted or reversed by bronchopneumonia. Death has occurred from within 15 min to 15 h after ingestion. One patient died 40 h after taking the oil, relapsing after apparent recovery.
Oral ingestion of Eucalyptus Globulus Leaf Oil, as low as approximately 5 mL, can affect the CNS (e.g., loss of consciousness, hypoventilation, depression of reflexes and convulsions), the gastrointestinal system (e.g., abdominal pain, vomiting and diarrhea), and the respiratory system (respiratory depression, dyspnea, pneumonitis, and bronchospasm). Gastrointestinal effects are frequently the initial effects, although drowsiness may occur in a few min and coma within 10 min. Urinary tract symptoms are only occasionally mentioned, and there is little evidence of direct nephrotoxicity following doses of up to 30 mL in an adult or older child; nephritis is rare but has been recorded. The subject may vomit while drowsy or unconscious and aspiration is a major risk. Tachycardia and a weak irregular pulse have been noted. Muscle weakness and ataxia may occur. Both mydriasis and miosis (more commonly) have occurred. CNS depression or vomiting has been delayed up to 4 h. Recovery is often within 24 h. 11
Inhalation
Eucalyptus Globulus Leaf Oil
The literature on the inhalation toxicity in humans of Eucalyptus Globulus Leaf Oil is scarce, and the following is a summary of this information. The substances are referred to in the literature as eucalyptus, eucalyptus oil, and similar names with little or no information on source plant parts, method of manufacture, or concentration/purity.
Inhalation of eucalyptus oil either as liquid or aerosol may result in pneumonitis. 11 Inhalation of vapor may be used medicinally and there are no data available on toxicity by this route. However, respiratory problems include bronchospasm, tachypnea, pulmonary edema, respiratory depression, and pneumonitis following aspiration of the oil. Eucalyptus oil inadvertently given intranasally has caused irritated nasal mucous membranes.
Short-Term Toxicity Studies
No published short-term dermal or inhalation toxicity studies were discovered and no unpublished data were submitted.
Oral
Short-Term Oral Studies.

ACP = acid phosphatase; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; CAT = catalase; GPX = glutathione peroxidase; IU = International units; LOAEL = lowest-observed-adverse-effect level; MDA = malondialdehyde; NOAEL = no-observed-adverse-effect level; OECD GL = Organisation of Economic Co-operation and Development Guidelines; PVC = packed cell volume; RBC = red blood cell count; SGOT = serum glutamic oxalacetic transaminase; SGPT = glutamic pyruvic transaminase; SOD = superoxide dismutase; WBC = white blood cell count.
Eucalyptus Globulus Leaf Extract
Aqueous Eucalyptus Globulus Leaf Extract (2000 mg/kg/d) orally administered (route not specified) to mice for 10 d caused no mortalities, but did cause general weakness and decrease in physical activity. 62 There were significant neurodegenerative changes, including a decrease in size and number of neurons in the cerebral cortex.
Aqueous Eucalyptus Globulus Leaf Extract (0, 80, 100, or 120 mg/kg) administered by gavage to rats for 7 d caused a significant increase in the level of malondialdehyde (MDA) in the liver of all treatment groups and in the serum of the 120 mg/kg group. 63 In a 42-d drinking water study in rats of an aqueous/ethanol Eucalyptus Globulus Leaf Extract (1 g/L), the rats consumed the equivalent of 130 mg dry leaves/d. Consuming the extract caused no changes in creatinine, urea, protein, or uric acid in the blood. 64
Eucalyptus Globulus Leaf Oil
Eucalyptus Globulus Leaf Oil (0, 1.5, or 2.0 mL/kg/d), administered by gavage to mice for 12 wk, caused no signs of toxicity and no mortalities for either treatment group; however, kidney effects (pyknosis of renal tubular epithelial cells and widening of tubular lumen) and liver effects (pyknosis, vacuolations of hepatocytes, and focal necrosis) were observed in the high-dose group at necropsy. 57
In a combined repeated dose and reproductive/developmental study, Eucalyptus Globulus Leaf Oil (0, 100, 300, or 1000 mg/kg/d) orally administered by gavage to rats caused transient signs of reduced activity and unsteady muscle reactions, multiple changes in blood chemistry, hyaline droplet nephropathy in the kidneys of male rats, and centrilobular hepatocytic hypertrophy in the livers of male rats and an increase in glycogenic vacuolation in the livers of female rats. 10 Males were treated starting from 2 wk before mating for at least 5 wk; females were treated from 2 wk before mating until lactation day 6. The no-observed-adverse-effect level (NOAEL) for males was 1000 mg/kg/d based on hyaline droplet nephropathy at all dose levels; however this response is considered to be rat specific and to have no counterpart in humans. The NOAEL for females was 300 mg/kg/d based on effects on body weight and feed consumption.
Eucalyptus Globulus Leaf Oil (100, 300, or 1000 mg/kg/d) orally administered by gavage to rats for 2 wk caused no mortalities. 10 The lowest-observed-adverse-effect level (LOAEL) and NOAEL in female rats could be considered as 300 and 100 mg/kg/d, respectively, based on the clinical signs at 300 and 1000 mg/kg/d and increased liver weight at 1000 mg/kg/d. Since dose-related increases in liver and kidney weights were observed in males at all doses, no NOAEL could be identified for the male rats in this study. The LOAEL in male rats could be considered as 100 mg/kg/d.
Eucalyptus Globulus Leaf Oil (0 or 233 mg/kg/dose in corn oil) administered by gavage every 3 d for 30 d caused an increase in white blood cell (WBC) counts and a decrease in hemoglobin concentration and platelets count in both blood samples, and relatively moderate pathological changes in the liver as congestion of the blood vessels in the portal area associated with inflammatory infiltration. 59
Eucalyptus Globulus Leaf Oil (0, 396, 792, and 1188 mg/kg/d) administered by gavage for 30 d caused no clinical signs, but changes to aspartate transaminase (increased), creatinine (increased), and glucose (decreased) in serum chemistry in the mid- and high-dose groups were observed. 58 In the livers of the experimental groups, the central veins were distended with hyperemia and varying degrees of vacuolar degeneration of hepatocytes were observed.
Eucalyptol (for inference to Eucalyptus Globulus Leaf Oil)
Eucalyptol (150, 300, 600, and 1200 mg/kg/d) administered by stomach tube or in encapsulated form in feed (600 to 5607 mg/kg/d for males and 705 to 6777 mg/kg/d for females) to mice for 28 d caused increased relative liver weights in all but the lowest dose in feed and a minimal hypertrophy of centrilobular hepatocytes in both sexes, especially in the two highest dose levels. 53
Eucalyptol (150, 300, 600, and 1200 mg/kg) administered by stomach tube or in encapsulated form in feed (381 to 3342 mg/kg/d for males and 353 to 3516 mg/kg/d for females) to rats for 28 d caused a dose-dependent decrease of body weight gain starting at 600 mg/kg and an absence of a normal degree of hepatic centrilobular cytoplasmic vacuolization was observed in male rats. 53 Eucalyptol (0, 500, or 1000 mg/kg/d) orally administered by gavage to male rats for 28 d caused no changes in the brain, but minor focal infiltration of mononuclear cells in liver was observed in both treatment groups and a dose-related accumulation of eosinophilic protein droplets containing α2u-globulin in the cytoplasm of proximal tubular epithelial cells was observed in kidneys. 53
Subchronic Toxicity Studies
No published subchronic toxicity studies were discovered and no unpublished data were submitted.
Chronic Toxicity Studies
Oral
Eucalyptol (for inference to Eucalyptus Globulus Leaf Oil)
A toothpaste containing eucalyptol (0, 8, and 32 mg eucalyptol/kg/d; 1 mL toothpaste) was administered by gavage to pathogen-free CFLP mice (n = 52) 6 d/wk for 80 wk, followed by 16 and 24 wk of rest. 53 No treatment-related effects on body weights, feed consumption, survival, weight of adrenals, kidneys, liver, lungs or spleen, on the microscopic appearance of brain, lungs, liver and kidneys, and on the tumor incidence were observed.
Developmental and Reproductive Toxicity (Dart) Studies
Oral
Eucalyptus Globulus Leaf Oil
In a combined repeated dose and reproduction/developmental study, Eucalyptus Globulus Leaf Oil (100, 300, or 1000 mg/kg/d; in corn oil) was administered by gavage to Crl:CD(SD) rats (n = 10/sex). 10 The study was conducted in accordance with Organisation of Economic Co-operation and Development Test Guideline (OECD TG) 422. Males were treated starting from 2 wk before mating for at least 5 wk; females were treated from 2 wk before mating until lactation day 6. The adults and offspring rats were then killed and necropsied. [For results related to short-term toxicity, see Short-Term Toxicity Studies]
There were no adverse effects detected in reproductive assessments on estrous cycles, mating performance and fertility, gestation length and parturition observations, and reproductive performance. There were no significant effects of the Eucalyptus Globulus Leaf Oil on litter size, offspring survival indices or sex ratio. The body weights of offspring of the treatment groups at birth were similar to that of the control group. However, body weight gains of male and female offspring in the 1000 mg/kg/d group were low (approximately 27% to 28% lower than the control group), and by day 4 after parturition, absolute body weights of this group were also significantly lower than that of the control group. A slightly increased incidence of “cold to touch” was observed in litters in the 1000 mg/kg/d group. At microscopic examination, there were no findings attributed to treatment for offspring that died and were examined during the experiment or were killed and examined at the end of the experiment.
Under the test condition, the NOAEL for the females was considered to be 300 mg/kg/d for systemic toxicity, based on lower body weight gain and feed consumption during gestation. The authors stated that both findings appeared to be associated with pregnancy status. It was not possible to link this effect to the taste of the substance since females had shown a significant duration of normal body weight and feed performance prior to day 6 of gestation and after birth of the pups. These latter observations appeared to indicate recovery in females. The NOAEL for developmental toxicity was 300 mg/kg/d, which was based on lower offspring body weight gain, and clinical signs (pups cold to touch) that were only observed in the 1000 mg/kg/d group. This effect may be associated with test material entering the milk. The authors note that fat soluble test materials have a higher chance of becoming incorporated in the milk and Eucalyptus Globulus Leaf Oil is fat soluble. A NOAEL at 300 mg/kg/d was determined for systemic effects in the offspring based on the magnitude of the weight reduction, which was quite high. The effects on offspring body weight were not selective and have been observed at a dose producing maternal toxicity, and therefore the substance was not considered to be a selective reproductive toxicant. The NOAEL for reproductive toxicity was 1000 mg/kg/d, since no adverse effects were observed for any reproductive parameters.
Genotoxicity Studies
In Vitro
In Vitro Genotoxicity Studies.

CHO = Chinese hamster ovary; MSO = Dimethyl sulfoxide; OECD GL = Organisation for Economic Co-operation and Development Guideline.
Eucalyptus Globulus Leaf Extract was not mutagenic, with and without metabolic activation, at up to 5000 µg/plate in an in vitro mammalian cell gene mutation test using mouse lymphoma cells. 10 Eucalyptus Globulus Leaf Oil was not genotoxic in a bacterial reverse mutation assay using Salmonella typhimurium and Escherichia coli at up to 5000 µg/plate, with and without metabolic activation. 10 Eucalyptus Globulus Leaf Oil was not genotoxic in an in vitro mammalian chromosome aberration test using human lymphocytes and an in vitro mammalian cell gene mutation test using mouse lymphoma L5178Y cells. 10 Eucalyptus Globulus Leaf Oil at .12 and .25 μL/mL was found to increase the mitotic instability of the original diploid strain and the number of diploid mitotic recombinants of Aspergillus nidulans. 12 The genotoxicity of the oil was associated with the induction of mitotic crossing-over or with oil-broken chromosomes.
Eucalyptol (concentration not specified) was not mutagenic in Ames assays, with and without metabolic activation. Eucalyptol (concentrations not specified) was not mutagenic to Chinese hamster ovary (CHO) cells in a chromosome aberration assay and a sister chromatid exchange assay, with and without metabolic activation. Eucalyptol (concentration not specified) was not mutagenic in rec assays using Bacillus subtilis. 53
Carcinogenicity Studies
No published carcinogenicity studies were discovered and no unpublished data were submitted on the Eucalyptus globulus (eucalyptus)-derived ingredients in this safety assessment.
Eucalyptol (for Inference to Eucalyptus Globulus Leaf Oil)
A toothpaste containing eucalyptol (0, 8, or 32 mg/kg/d) was administered by gavage to male pathogen-free CFLP mice (n = 52) for 80 wk. 65 The controls were administered nothing (n = 52) or the toothpaste base (n = 260). The mice were observed daily, and were weighed weekly for the first 6 wk of the study then every 2 wk. Mice found dead were necropsied. At week 80, the mice were killed and organ weights for the kidneys, adrenals, lungs, liver, and spleen were examined. All macroscopically identified tumors were examined histopathologically. Tissues from the kidneys, liver, lungs, and brain were also examined histopathologically. All of the mice in the low-dose group and 47 of the mice in the high-dose group were necropsied. There were no differences between the test groups and the control and vehicle control groups in the incidence or severity of tumors of the organs or the presence of malignant lymphoma.
Tumor Promotion
Dermal
Eucalyptus Globulus Leaf Oil
Eucalyptus Globulus Leaf Oil was tested for tumor promotion in mice. 29 A single application of 9,10-dimethyl-1,2-benzanthracene (DMBA) was administered to the clipped backs of 8-wk-old mice (n = 14). The dose of DMBA (225 µg; 2 mL in acetone) was described as being sufficient to initiate skin tumor formation but, generally, inadequate for complete carcinogenesis. After three weeks, Eucalyptus Globulus Leaf Oil (.25 mL/wk) was administered to the backs of the mice once per week for 33 wk. Dorsal hair was removed as necessary. The control group (n = 13) received the DMBA treatment alone. Papillomas were observed on 4 of 14 mice in the treatment group and 0 of 13 in the control group.
Dermal Irritation and Sensitization Studies
Dermal Irritation and Sensitization.
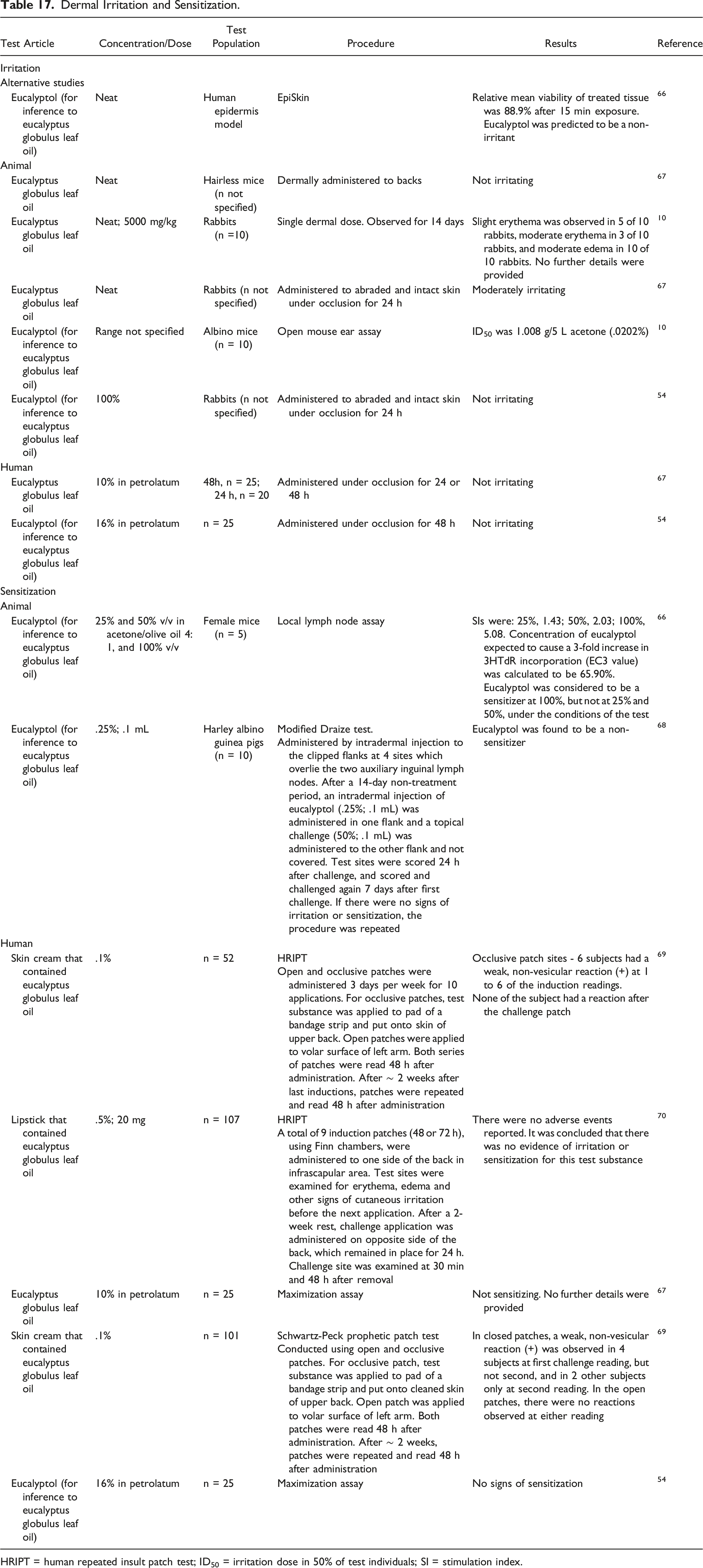
HRIPT = human repeated insult patch test; ID50 = irritation dose in 50% of test individuals; SI = stimulation index.
Irritation
Eucalyptol (100%) was predicted to be a dermal non-irritant in an EpiSkin™ assay. 66 Eucalyptus Globulus Leaf Oil (neat) was not dermally irritating to hairless mice, 67 but was slightly (neat; 5000 mg/kg) or moderately (intact and abraded skin) irritating to rabbits.10,67 In an open mouse ear assay of eucalyptol using albino mice (n = 10), the irritant dose in 50% of test individuals (ID50) was 1.008 g/5 L acetone (.0202%). 10 Eucalyptol (100%) administered to intact and abraded skin of rabbits for 24 h under occlusion was not irritating. 54 Eucalyptus Globulus Leaf Oil (10% in petrolatum) 67 and eucalyptol (16% in petrolatum) were not irritating to human subjects (n = 25). 54
Sensitization
Eucalyptol (25% and 50% v/v) tested in a local lymph node assay (LLNA) using female mice (n = 5) was considered to be a sensitizer at 100%, but not at 25% and 50%. 66 In a Draize test using Harley albino guinea pigs (n = 10), eucalyptol (.25%; .1 mL) was not irritating or sensitizing. 68
In multiple human repeated insult patch tests (HRIPT), cosmetic formulations containing Eucalyptus Globulus Leaf Oil (up to .5%) were found to be non-irritating.69,70 In a maximization assay in human subjects (n = 25), Eucalyptus Globulus Leaf Oil (10% in petrolatum) produced no sensitization reactions. 67
Photosensitization/Phototoxicity
In Vitro
Eucalyptus Globulus Leaf Oil
An in vitro photohemolysis test (human erythrocyte suspensions) was used to evaluate the phototoxicity of Eucalyptus Globulus Leaf Oil (.001, .01, or .1% in alcohol). 71 The ultraviolet A (UVA; long-wave)-rich light source was a UVASUN 5000 lamp (320 to 460 nm; 42 mW/cm2) and the ultraviolet B (UVB; mid-wavelength)-rich light source was a lamp with TL 20 W/12 light bulbs (between 275 and 365 nm; 1 mW/cm2 (UVB) and .4 mW/cm2 (UVA)). There was no hemolysis observed under the test conditions. The authors concluded that the test substance is not expected to be photosensitizing.
Human
In a Draize-Shelanski HRIPT, a skin cream that contained Eucalyptus Globulus Leaf Oil (.1%; no dose specified), was tested on the backs of 52 subjects and exposed to UV light at a distance of 12” (30.48 cm) for 1 min. These exposures were after the first, fourth, seventh, and 10th induction patches and the challenge patch were read. 69 The test sites were read 48 h after each application. Induction applications were rotated between three sites on the back of each subject; therefore, irradiation was administered to the same test site every third patch. The challenge was administered to a naïve site. There were no signs of photosensitization in any subject at any reading. [See Sensitization section for sensitization data.]
In a Schwartz-Peck prophetic patch test, photosensitization potential of a skin cream containing Eucalyptus Globulus Leaf Oil (.1%) (n = 101) was evaluated at the occlusive patch sites. 69 The test sites were exposed to an UV light (at a distance of 12” (30.48 cm) for 1 min) 48 h after the second patch was administered. The test sites were read 48 h after the UV exposure. There were no signs of photosensitization in any subject. [See Sensitization section for sensitization data.]
Ocular Irritation Studies
In Vitro
Eucalyptol (for Inference to Eucalyptus Globulus Leaf Oil)
Eucalyptol (100%; .75 mL) was tested in a bovine corneal opacity and permeability assay. 66 Eucalyptol was not considered to be an ocular corrosive or severe irritant.
Animal
Eucalyptus Globulus Leaf Oil
In an eye irritation study performed in accordance with OECD TG 405 (acute eye irritation/corrosion), undiluted Eucalyptus Globulus Leaf Oil (.1 mL) was instilled into the right eye of a single New Zealand White (Hsdlf:NZW) rabbit. 10 After consideration of the ocular responses produced in the first treated animal, two additional animals were treated. The eyes were not rinsed after administration. The left eye of each rabbit served as control. Animals were observed 1, 24, 48, and 72 h after dosing under a light source from a standard ophthalmoscope. The reactions in the conjunctiva (redness, chemosis and discharge), the iris and the cornea (opacity and area involved) were scored according to the Draize scale. No corneal or iridial effects were observed during the study. Moderate conjunctival irritation was noted in all treated eyes 1 h after treatment with minimal conjunctival irritation noted at the 24- and 48-h observations. All treated eyes appeared normal at 72 h. Mean scores calculated for each rabbit over 24, 48, and 72 h were .0/0.0/0.0 for cornea opacity, .0/0.0/0.0 for iris lesions, .7/1.0/0.7 for redness of the conjunctivae, and .7/0.7/0.7 for chemosis. One rabbit had no body weight gain and two rabbits showed expected gain in body weight during the study.
Clinical Studies
Retrospective and Multicenter Studies
Dermal
Retrospective and Multicenter Studies of Eucalyptus Globulus Leaf Oil.
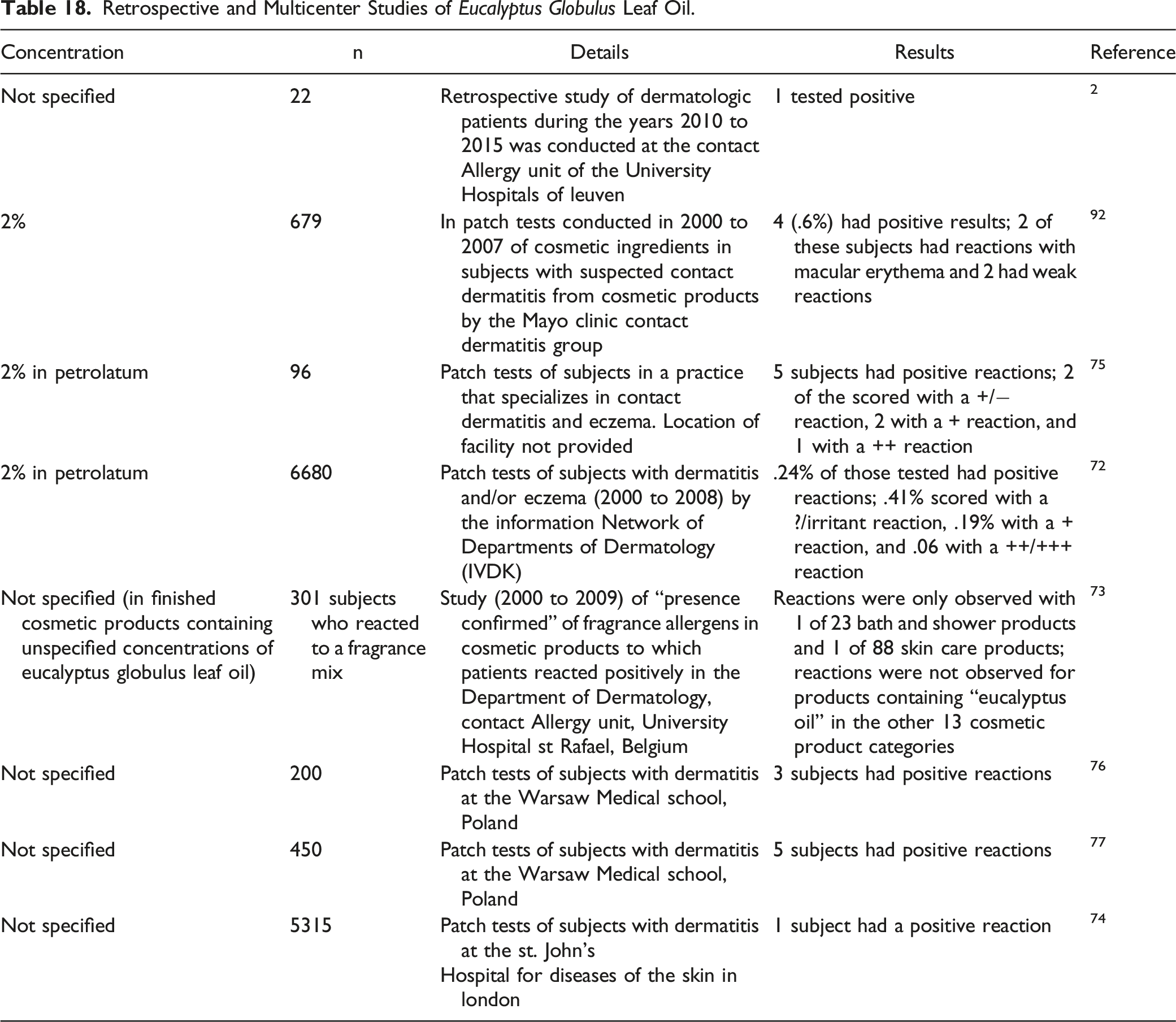
In sensitization tests (method not reported) of patients (n = 5315) in London with dermatitis, 1 subject had a positive reaction to Eucalyptus Globulus Leaf Oil (concentration not specified). 74 In patch tests of subjects (n = 96) with dermatitis and/or eczema, 5 subjects had positive reactions (2 scored with a +/− reaction, 2 with a + reaction, and 1 with a ++ reaction) to Eucalyptus Globulus Leaf Oil (2% in petrolatum). 75
In sensitization tests (method not clear) of patients (n = 200) in Poland with dermatitis, 3 subjects had positive reactions to Eucalyptus Globulus Leaf Oil (concentration not specified). 76 When this study was continued on additional patients (n = 450) with dermatitis, 5 subjects had a positive reaction to Eucalyptus Globulus Leaf Oil (concentration not specified). 77
Oral
In a respective study of accidental ingestion of Eucalyptus Globulus Leaf Oil by children in Australia (n = 109), 41% had no effects, 30% resulted in minor poisoning, 25% resulted in moderate poisoning, and 3% resulted in severe and life threatening poisoning. 78 Of those where the volume was known, 17 ingested 100% oil and 10 ingested inhalation preparations. All of those children who had been given Eucalyptus Globulus Leaf Oil by a parent or guardian by mistake ingested a mean volume of 2.2 mL (range, .2 - 7.5 mL). There were no deaths. Adverse effects included vomiting, depression of conscious state, ataxia, pulmonary disease, miosis, and abdominal pain.
Case Reports
Case reports of adverse reactions to dermal, oral, and inhalation exposure to Eucalyptus Globulus Leaf Oil are presented in Table 19. Dermal effects ranged from none to eczema, erythematous macular lesions, papules and vesicles, and/or pruritus.79-84 Oral effects included esophageal pain, gasping for breath, restlessness, dyspnea, weak pulse, vomiting, drowsiness, and convulsions.11,83,85-88 Inhalation effects included strong characteristic smell on the breath, coughing, chest tightness, dyspnea, hoarseness, and wheezing.83,89 In children, inhalation effects included nasal and epigastric burning, nausea, vomiting, dizziness, muscular weakness, miosis, tachycardia, and a feeling of suffocation. Cyanosis, delirium, and convulsions may be exhibited, especially in infants.
90
Case Reports of Eucalyptus globulus Exposure.

BP – blood pressure; CNS – central nervous system; EEG = electroencephalogram; PVC - premature ventricular contractions; VCD - vocal cord dysfunction.
Summary
This is a review of the safety of 6 Eucalyptus globulus-derived ingredients as used in cosmetics. According to the Dictionary, the reported functions of the Eucalyptus globulus-derived ingredients include abrasive, fragrance ingredient, and skin-conditioning agent (miscellaneous and occlusive). Eucalyptus Globulus Leaf/Twig Oil and Eucalyptus Globulus Leaf Water are reported to function only as fragrance ingredients.
Because Eucalyptus Globulus Leaf Oil consists of not less than 70% (w/w) eucalyptol, relevant toxicity data on eucalyptol are included as supporting information in this safety assessment. Other reported main components of Eucalyptus Globulus Leaf Oil include α-pinene (9.22 - 24.6%), globulol (.819 - 2.817%), and β-pinene (.217 - 1.237%), depending on the origin of the plant.
According to VCRP survey data received in 2018, Eucalyptus Globulus Leaf Oil is reported to be used in 433 formulations (214 leave-on formulations, 160 rinse-off formulations, and 59 formulations that are diluted for the bath). Eucalyptus Globulus Leaf Extract is reported to be used in 77 formulations and Eucalyptus Globulus Leaf Powder is reported to be used in 2 formulations. The VCRP included ingredients with the non-INCI name “eucalyptus” (42 reported uses) and “eucalyptus extract” (11 reported uses). The results of the concentration of use survey conducted by the Council in 2017 and 2018 indicate Eucalyptus Globulus Leaf Water is used at up to 1.4% in face and neck products. The rest of the ingredients with reported concentrations of use are used at 1.2% or less. There were no uses reported to the VCRP or industry survey for Eucalyptus Globulus Leaf/Twig Oil.
In in vitro studies, Eucalyptus Globulus Leaf Oil has been shown to increase the dermal penetration of CHG, TMP, ketorolac, and 5-FU.
Orally administered eucalyptol undergoes oxidation in vivo with the formation of hydroxycineole which is excreted as a glucuronide. In rats, urinary metabolites were 2-hydroxycineole, 3-hydroxycineole, and 1,8-dihydroxycineol-9-oic acid. In mice, eucalyptol is rapidly absorbed and metabolized; when inhaled, elimination was biphasic.
The dermal LD50 for Eucalyptus Globulus Leaf Oil and eucalyptol was > 5000 mg/kg in rabbits. The oral LD50 for Eucalyptus Globulus Leaf Oil was 3320 mg/kg in male mice. At doses at and above 2.5 mL/kg, toxic effects were observed; the clinical signs disappeared in surviving mice. In rats, the oral LD50 for Eucalyptus Globulus Leaf Oil was reported as 3811.5 mg/kg in one study and 4400 mg/kg in another study. Reported oral LD50s of eucalyptol in rats were 2480 mg/kg and 1560 mg/kg.
Rabbits inhaling water vapor containing Eucalyptus Globulus Leaf Oil died; the output of respiratory tract fluid was significantly augmented at 19 683 mg/kg. OVA-sensitized guinea pigs exposed to aerosolized eucalyptol for 15 min showed no pulmonary effects.
The probable oral lethal dose of Eucalyptus Globulus Leaf Oil for adult humans is .05 mL to .5 mL/kg. The oral ingestion of Eucalyptus Globulus Leaf Oil may initially result in a burning sensation in the mouth, vomiting, diarrhea, and epigastric pain. In humans, inhalation of Eucalyptus Globulus Leaf Oil, either as liquid or aerosol, may result in pneumonitis.
An aqueous Eucalyptus Globulus Leaf Extract (2000 mg/kg/d) orally administered to mice for 10 d caused no adverse effects. In short-term oral toxicity studies, Eucalyptus Globulus Leaf Extract administered to rats showed hepatic effects in some studies (starting at 100 mg/kg) and none in another (1 g/L in drinking water for 42 d).
In short-term oral toxicity studies, Eucalyptus Globulus Leaf Oil and eucalyptol caused hepatic effects in both mice and rats. Oral administration of Eucalyptus Globulus Leaf Oil for 12 wk caused renal and hepatic effects at 2.0 mL/kg/d in mice, but not at 1.5 mL/kg/d. In rats, hepatic effects were observed starting at 233 mg/kg Eucalyptus Globulus Leaf Oil. Eucalyptol (150 to 1200 mg/kg/d) administered by stomach tube or in feed (600 to 5607 mg/kg/d for males and 705 to 6777 mg/kg/d for females) to mice for 28 d caused increased relative liver weights in all but the lowest dose in feed. Eucalyptol (500 or 1000 mg/kg/d) orally administered for 28 d to rats caused minor focal infiltration of mononuclear cells in livers in both treatment groups. There were no treatment-related effects in mice orally administered a toothpaste containing up to 32 mg eucalyptol/kg/d for 80 wk.
In a reproduction/developmental study of Eucalyptus Globulus Leaf Extract administered to rats, the NOAEL for the females was 300 mg/kg/d for systemic toxicity, based on lower body weight gain and feed consumption during gestation. The NOAEL for developmental toxicity was 300 mg/kg/d, which was based on lower offspring body weight gain, and clinical signs that were only observed in the 1000 mg/kg/d group. However, a NOAEL at 300 mg/kg/d was determined for systemic effect in the offspring based on the magnitude of the weight reduction. The NOAEL for reproductive toxicity was 1000 mg/kg/d, since no adverse effects were observed.
Eucalyptus Globulus Leaf Extract was not mutagenic, with and without metabolic activation, at up to 5000 µg/plate in an in vitro mammalian cell gene mutation test. Eucalyptus Globulus Leaf Oil was not genotoxic in a bacterial reverse mutation assay (up to 5000 µg/plate), in an in vitro mammalian chromosome aberration test (up to1000 µg/ml) and an in vitro mammalian cell gene mutation test using mouse lymphoma L5178Y cells (up to 300 μg/mL). However, Eucalyptus Globulus Leaf Oil at .12 μL/mL was found to increase the mitotic instability of the original diploid strain and the number of diploid mitotic recombinants of A. nidulans. Eucalyptol was not mutagenic in Ames assays, to CHO cells in a chromosome aberration assay, a sister chromatid exchange assay, and in rec assays using B. subtilis.
There were no differences between the test groups and the untreated control and vehicle control groups in the incidence or severity of tumors of the organs or the presence of malignant lymphoma in male mice administered a toothpaste containing eucalyptol (0, 8, or 32 mg/kg/d) for 80 d. In mice first treated with a single dermal dose of DMBA then dermally administered .25 mL Eucalyptus Globulus Leaf Oil (neat) weekly for 33 wk, papillomas were observed in 4 of 14 mice. None of the 13 control mice had papillomas.
Eucalyptol (100%) was predicted to be a non-irritant in an EpiSkin™ assay. Eucalyptus Globulus Leaf Oil (neat) was not dermally irritating to hairless mice, but was slightly (neat; 5000 mg/kg) or moderately (intact and abraded skin) irritating to rabbits. In an open mouse ear assay of eucalyptol using albino mice (n = 10), the ID50 was 1.008 g/5 L acetone (.0202%). Eucalyptol (100%) administered to intact and abraded skin of rabbits for 24 h under occlusion was not irritating. Eucalyptus Globulus Leaf Oil (10% in petrolatum) and eucalyptol (16% in petrolatum) were not irritating to human subjects.
Eucalyptol tested in an LLNA using female mice (n = 5) had an EC3 of 65.90%, thus was predicted to be a sensitizer at 100%, but not at 50%. In a Draize test using Harley albino guinea pigs (n = 10), eucalyptol (.25%; .1 mL) was not irritating or sensitizing. No dermal irritation or sensitization was reported in HRIPTs of cosmetic formulations containing Eucalyptus Globulus Leaf Oil up to .5%. In a maximization assay in human subjects (n = 25), Eucalyptus Globulus Leaf Oil (10% in petrolatum) produced no sensitization reactions.
In an in vitro photohemolysis test, Eucalyptus Globulus Leaf Oil (neat) was not predicted to be photosensitizing. In combined sensitization and photosensitization tests, there were no signs of photosensitization in two photosensitization patch tests of a skin cream that contained Eucalyptus Globulus Leaf Oil (.1%).
Eucalyptol (100%) was not considered to be an ocular corrosive or severe irritant when tested in a bovine corneal opacity and permeability assay. In rabbits, Eucalyptus Globulus Leaf Oil (1 mL) caused moderate conjunctival irritation in all treated eyes 1 h after treatment. Conjunctival irritation was minimal at 24- and 48-h and all treated eyes appeared normal at 72 h.
In a retrospective study of dermatologic patients during the years 2010 to 2015, of the 22 subjects that were tested for sensitization to Eucalyptus Globulus Leaf Oil, 1 tested positive. In a retrospective study of dermatologic patients during the years 2000 to 2007, 4 of 679 (.6%) had positive results for Eucalyptus Globulus Leaf Oil (2%). In a retrospective study of dermatologic patients during the years 2000 to 2009, of the 6680 subjects that were tested for sensitization to Eucalyptus Globulus Leaf Oil, .24% had positive reactions. In a cross-sectional study conducted in Belgium (2000 to 2009) of 301 subjects having had reactions to fragrance mixes, a reaction was confirmed to “eucalyptus oil” in 1 of 23 bath and shower products and 1 in 88 skin care products. In sensitization tests of patients (n = 5315) in London with dermatitis, 1 subject had a positive reaction to Eucalyptus Globulus Leaf Oil (concentration not specified).
In a respective study of accidental ingestion of Eucalyptus Globulus Leaf Oil by children, adverse effects included vomiting, depression of conscious state, ataxia, pulmonary disease, miosis, and abdominal pain. In case reports of exposure to Eucalyptus Globulus Leaf Oil, dermal effects ranged from none to eczema, erythematous macular lesions, papules and vesicles, and/or pruritus. Oral effects included esophageal pain, gasping for breath, dyspnea, weak pulse, vomiting, drowsiness, and convulsions. Inhalation effects included coughing, chest tightness, dyspnea, hoarseness, and wheezing. In children, inhalation effects included nasal and epigastric burning, nausea, vomiting, dizziness, muscular weakness, miosis, tachycardia, and a feeling of suffocation. Cyanosis, delirium, and convulsions may be exhibited, especially in infants.
Discussion
The Panel examined the data on oral, dermal and inhalation toxicity, ocular and dermal irritation, sensitization, reproduction, genotoxicity, and phototoxicity. The Panel also considered toxicity data on eucalyptol, a major component of Eucalyptus Globulus Leaf Oil and Eucalyptus Globulus Leaf/Twig Oil. The Panel noted the lack of toxicity and the lack of irritation and sensitization at relevant concentrations of these ingredients. The genotoxicity studies and the carcinogenicity study on eucalyptol were negative and gave no cause for concern.
Case studies described adverse effects following both oral and dermal administration. Instances were reported in which persons who consumed Eucalyptus globulus oil became very ill following oral ingestion, with respiratory difficulties (e.g., pneumonitis, pulmonary edema, and bronchopneumonia) reported. The Panel noted that these incidents resulted from bolus doses and exposure was much greater than that expected with cosmetic use. Also, the Panel believes that the cause of the respiratory difficulties was aspiration of vomitus and not directly caused by Eucalyptus globulus oil. The adverse effects in the dermal case studies resulted from administration of Eucalyptus globulus oil at far greater concentrations than that found in cosmetics. Oral ingestion, and the circumstances of the dermal administration of Eucalyptus globulus oil, would lead to a rapidly increased concentration of the oil in the blood that would far exceed what would result from the use of cosmetic formulations containing Eucalyptus Globulus Leaf Oil. These high concentrations in the blood could not be obtained through cosmetic use.
The composition data are robust for Eucalyptus Globulus Leaf Extract and Eucalyptus Globulus Leaf Oil. The data on these two ingredients provide substantial insight into the other ingredients for which composition data are not as robust, and enable consideration of the entire group.
The sensitization data on Eucalyptus Globulus Leaf Oil at 10% and eucalyptol at 16% in humans provided evidence that sensitization at reported concentrations (maximum of 1.4%) of use should not be expected. This is supported by a LLNA and a Draize test in guinea pigs.
There is the possibility of the presence of potential sensitizers in the Eucalyptus globulus-derived ingredients because these constituents exist in the plant. However, if these constituents were to be present in a cosmetic formulation, the concentrations would be far below the level of toxicological concern. The impurity specifications of trade name mixtures containing Eucalyptus Globulus Leaf Extract, the lack of dermal irritation in human patch testing, the lack of sensitization in HRIPTs, and the small proportion of positive results in retrospective studies involving relatively large numbers of individuals assured the Panel that dermal irritation and sensitization from these constituents is not a significant concern in the cosmetic use of Eucalyptus globulus-derived ingredients. Relevant data on eucalyptol also contributed to support for this supposition. However, because final product formulations may contain multiple botanicals, each possibly containing similar constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. For Eucalyptus globulus-derived ingredients, the Panel was concerned about the presence of geraniol, limonene, and linalool in cosmetics, which could result in sensitization. Therefore, when formulating products, manufacturers should avoid reaching levels of plant constituents that may cause sensitization or other adverse health effects. The Panel noted that IFRA standards have been developed, and published, so as to prevent adverse effects for several such constituents (Table 10).
The Panel expressed concern about pesticide residues, heavy metals, and substances from plants of other species (weeds) that may be present in botanical ingredients. To address these concerns, the cosmetics industry should continue to use current good manufacturing practices (cGMP) to limit impurities.
The Panel recognized that Eucalyptus Globulus Leaf Oil can enhance the penetration of other ingredients through the skin (e.g., chlorhexidine). The Panel cautioned that care should be taken in formulating cosmetic products that may contain this ingredient in combination with any ingredients whose safety was based on their lack of dermal absorption data, or when dermal absorption was a concern.
The Panel discussed the issue of incidental inhalation exposure from formulations that may be aerosolized (e.g., colognes and toilet waters at up to .4%). The acute inhalation data and historic case studies available suggest potential respiratory effects only at doses much greater than would be used in cosmetics. The Panel believes that the sizes of a substantial majority of the particles of aerosol and other spray products are larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation, and thus, droplets/particles from cosmetic products would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for Eucalyptus globulus-derived ingredients to cause systemic toxicity, irritation, sensitization, reproductive and developmental toxicity, and genotoxicity. They noted the lack of systemic toxicity at high doses in acute oral exposure studies (in contrast to old case reports), minimal or no irritation or sensitization in tests of dermal exposure at relevant concentrations, and the absence of genotoxicity in multiple assays. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be non-sensitizing: Eucalyptus Globulus Leaf Eucalyptus Globulus Leaf Extract Eucalyptus Globulus Leaf Oil Eucalyptus Globulus Leaf Powder Eucalyptus Globulus Leaf/Twig Oil* Eucalyptus Globulus Leaf Water
* Not reported to be in current use. If this ingredient were to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.