Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 11 Cocos nucifera (coconut)-derived ingredients, most of which are reported to function as skin-conditioning agents in cosmetic products. The Panel reviewed the available data to determine the safety of these ingredients. The Panel concluded that 10 ingredients, derived from coconut flower, fruit, and liquid endosperm, are safe in cosmetics in the present practices of use and concentration described in this safety assessment, and that the available data are insufficient to make a determination of safety for Cocos Nucifera (Coconut) Shell Powder under the intended conditions of use in cosmetic formulations.
Introduction
The assessment of the safety of the following 11 Cocos nucifera (coconut)-derived ingredients is based on the data contained in this report: Coconut Flower Sugar Cocos Nucifera (Coconut) Flower Extract Cocos Nucifera (Coconut) Flower Nectar Extract Cocos Nucifera (Coconut) Fruit Cocos Nucifera (Coconut) Fruit Extract Cocos Nucifera (Coconut) Fruit/Fruit Juice Extract Cocos Nucifera (Coconut) Fruit Juice Cocos Nucifera (Coconut) Fruit Powder Cocos Nucifera (Coconut) Fruit Water Cocos Nucifera (Coconut) Liquid Endosperm Cocos Nucifera (Coconut) Shell Powder
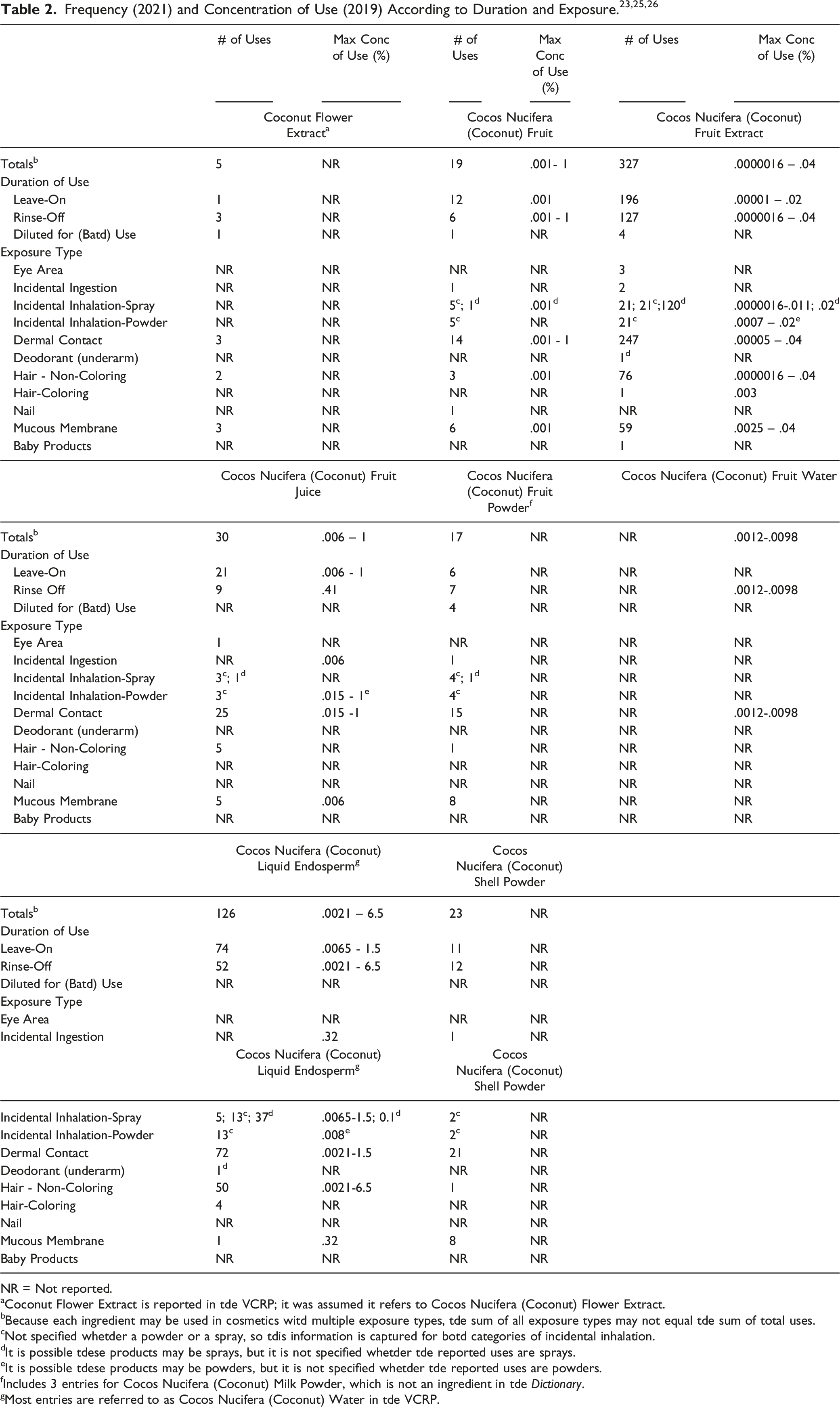
Definitions and Reported Functions of the Ingredients in This Safety Assessment. 1
The Expert Panel for Cosmetic Ingredient Safety (Panel) has previously reviewed the safety of some related Cocos nucifera (coconut)-derived ingredients. In 1986, the Panel first issued a final report on the safety of Cocos Nucifera (Coconut) Oil, Coconut Acid, Hydrogenated Coconut Acid, and Hydrogenated Coconut Oil; the Panel concluded that the ingredients are safe for use as cosmetic ingredients. 2 In 2011, the Panel published a report on the safety of Cocos Nucifera (Coconut) Oil and related ingredients with a conclusion that the ingredients are safe for use as cosmetic ingredients. 3 Finally, in a report published in 2017, the Panel reviewed the safety of plant-derived fatty acid oils and concluded that the 244 plant-derived fatty acid oils, including Cocos Nucifera (Coconut) Oil and Cocos Nucifera (Coconut) Seed Butter, are safe in present practices of use and concentration. 4
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. Due to the paucity of published safety and toxicity data on these ingredients, this report includes summary information included in technical dossiers, when appropriate. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, are provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
These ingredients in this report are all derived from the same species, Cocos nucifera. As such, there is likely some overlap of constituents and/or potential impurities. Botanicals, such as Cocos nucifera-derived ingredients, may contain hundreds of constituents, some of which may have the potential to cause toxic effects. In this assessment, the Panel is reviewing the potential toxicity of each of the botanical ingredients as a whole, complex substance; potential toxicity from exposures to mixtures of different chemical compounds may not replicate the biological activity of the individual components. Additionally, some of the ingredients reviewed in this safety assessment are consumed as food, and daily exposure from food use would result in much larger systemic exposures than possible from use in cosmetic products. Therefore, the primary focus of the safety assessment of these ingredients as used in cosmetics is on the potential for local effects (i.e. from topical exposure).
Chemistry
Definition and Plant Identification
The definitions of the Cocos nucifera-derived ingredients included in this review are provided in Table 1. 1 Cocos nucifera L. is native to Indonesia, Papua, New Guinea, Philippines, Australia, and Vanuatu. 5 Cocos nucifera is an important member of the family Arecaceae (palm family), and is more commonly known as coconut. 6 The plant is an arborescent monocotyledonous tree of approximately 25 m in height (giant coconut) with a dense canopy. 6 The giant adult coconut produces 12-14 groups of flowers that emanate from the main stem (inflorescence spikes) yearly, while the adult dwarf coconut can emanate 18 spikes in the same period.
While the Dictionary definition states Cocos Nucifera (Coconut) Fruit is the fruit (or hardened endosperm) of Cocos nucifera, botanical resources define the coconut fruit as a fibrous drupe with a smooth outside skin (exocarp), which may vary from green to red brown, or even ivory in color.7-9 The exocarp and mesocarp make up the “husk” of the coconut. The mesocarp is the hard, fibrous reddish-brown husk from which coir or fiber is obtained. Between the shell and the kernel is a thin brown seed coat or testa, which adheres to the hard endosperm or kernel (also called meat or jelly), about 12 mm thick in a mature nut. The edible part of the coconut fruit is the endosperm tissue. In young fruit, the endosperm is a liquid called “coconut water,” while in mature fruit, the endosperm is called coconut meat. The kernel (called copra when dried to 60% moisture) constitutes 40%-70% of the weight of the husked nut and contains about 50% water and 30% - 40% oil. “Coconut water” is not the same as “coconut milk;” the latter is obtained by grating coconut meat, with or without the addition of water. 8
Chemical Properties
Cocos Nucifera (Coconut) Fruit Extract
A Supplier Reported that Cocos Nucifera (Coconut) Fruit Extract Prepared in water Is a Colorless to light Yellow liquid With a Characteristic odor. 10 at 25°C, the pH Is 6.7 and the Specific Gravity Is .99 - 1.00. This Ingredient Is Soluble in any Proportion in water
No chemical properties on these cosmetic ingredients were discovered in the published literature, and no other unpublished data were submitted.
Method of Manufacture
Coconut Flower Sugar
Coconut flower sugar, also called coconut sap sugar, is produced from the watery sap from cut flower buds or inflorescence of the coconut tree.11 The sap is usually collected twice a day and boiled in huge woks until only the coconut sugar remains.
Cocos Nucifera (Coconut) Flower Extract
The method described herein is general to the processing of a coconut flower extract, and it is unknown if it applies to cosmetic ingredient manufacture
Cocos Nucifera (Coconut) Flower Nectar Extract
A supplier has reported the Cocos Nucifera (Coconut) Flower Nectar Extract is produced by evaporating nectar from harvested organic flowers at low temperature (60°C) in a closed system. 13 The vapor is trapped and condensed.
Cocos Nucifera (Coconut) Fruit Extract
A supplier reported that Cocos Nucifera (Coconut) Fruit Extract is produced by extracting the fruit with specified eluent(s) under appropriate temperature conditions to yield a concentrate. 10 Typical eluents include water, butylene glycol, glycerin, Carthamus tinctorius (safflower) seed oil, and propylene glycol. The concentrate containing phytochemical constituents is then blended with the desired diluent(s) and preservation system to produce the final ingredient.
Cocos Nucifera (Coconut) Fruit Water
In general, botanical waters are prepared from the leaves, stems, flowers, bark, roots, or other parts of a plant or the whole plant. 1 The condensate from steam distillation produces 2 distinct fractions that contain the volatile ingredients from the plant. The water insoluble fraction contains the “oil.” The water-soluble fraction contains ingredients from the plant that are water soluble, and is identified by the term “Water” in the INCI name.
Cocos Nucifera (Coconut) Liquid Endosperm
A supplier reported that Cocos Nucifera (Coconut) Liquid Endosperm is produced from collecting coconut water from tender coconuts under sterile conditions and filtering the water through membrane filters. 14 The resulting product is then freeze-dried.
Cocos Nucifera (Coconut) Shell Powder
In a study of the composition of coconut shell, the outer pericarp of the Cocos nucifera shell was collected, air-dried, ground into a fine powder, and then sieved with 2 mm mesh pore size. 15
Composition/Impurities
Coconut Flower Sugar
Coconut flower sugar contains vitamin C (16-44 mg/100 g), potassium (957-1075 mg/100 g), sodium (99-112 mg/100 g), calcium (8 mg/100 g), iron (0.5-0.6 mg/100 g), zinc (0.1 mg/100 g), and inulin (4.57 g/100 g). 11 Short chain fatty acids present after fermentation were acetic acid (7-18 mg/g), propionic acid (135 - 187 mg/g), and butyric acid (7-19 mg/g). Additional fatty acids and other volatile present may include lauric acid, 1,4-dimethyl-6-1 butyl-acetate, cyclohexiloctane, 2-butanol, N,N-dimethyl 2-(diphenylmetoxi)-ethylamine, and methylpyrazine. 16 Sugars include sucrose, fructose, glucose, and mannose. 11
Coconut flower sugar may contain microbiological impurities such as mold, yeast, and Salmonella sp. and coliform bacteria. 11
Cocos Nucifera (Coconut) Flower Extract
Cocos nucifera flower extracts that were prepared as described above, were used in phytochemical analysis. 12 The chloroform extract showed the presence of alkaloids, flavonoids, tannins and carbohydrates. The methanol extract showed the presence of alkaloids, flavonoids, phenols, carbohydrates, and amino acids. The ethanol extract contains phytosterols and tannins. The hydroalcoholic extract showed the presence of flavonoids, phenols, tannins, and carbohydrates. The aqueous extract contains alkaloids, flavonoids, phenols, tannins, and carbohydrates. The various extracts of Cocos nucifera flowers showed the absence of saponins and anthraquinones.
Cocos Nucifera (Coconut) Flower Nectar Extract
A supplier has reported that Cocos Nucifera (Coconut) Flower Nectar Extract with pH 3.97 is primarily water containing mixed organic acids (1090 µg/g acetic acid, 53 µg/g malic acid, 7 µg/g formic acid, and 1 µg/g succinic acid) and some minerals (.05 mg/100 ml calcium, .43 g/ml sodium, and 1.09 g/ml potassium).13,17,18 Aerobic bacterial plate counts/ml were less than 1 colony-forming units (cfu) and the most probable number of coliform/100 ml was less than 1.1. 18 Yeast and mold/ml was less than 1 cfu.
Cocos Nucifera (Coconut) Fruit Extract
A supplier reported that an extract of coconut fruit is prepared with 90% water and 10% butylene glycol. 19 The composition of the dry matter is 73% sugars (disaccharides), 22% mineral ashes (calcium, magnesium, sodium, potassium, phosphorus, sulfur, and chloride), and 5% protein.
A supplier reported that Cocos Nucifera (Coconut) Fruit Extract in butylene glycol had the following heavy metals profile via inductively coupled plasma-optical emission spectrometry: .363 ppm antimony, .091 ppm arsenic, .046 ppm nickel, and .054 ppm vanadium. 19 Cadmium, chromium, cobalt, mercury, and lead were not quantifiable. No phytosanitary (pesticide) substances or aflatoxins were detected via high performance liquid chromatography and or gas chromatography.
Another supplier reported that Cocos Nucifera (Coconut) Fruit Extract concentrate in a glycerin and water base had .115 mg/l arsenic and .083 mg/l nickel. 10 Antimony, cadmium, chromium, iron, lead, and mercury were below levels of detection. No residual pesticides were detected. The 26 allergens defined by the European Union Cosmetic Regulations were below threshold levels in a concentrate of Cocos Nucifera (Coconut) Fruit Extract concentrate in an alcohol base.
Cocos Nucifera (Coconut) Liquid Endosperm
The aqueous part of the coconut endosperm contains mainly water (∼94 - 95 g/100 g). 8 Components found in liquid endosperm are dependent on coconut type (young, young green, mature green), and the average weight, age, and source of the coconut. Components that may be present include sugars (sucrose, glucose, fructose), sugar alcohols (mannitol, sorbitol, myo-inositol, scyllo-inositol), lipids, fatty acids, amino acids (alanine, β-alanine, γ-aminobutyric acid, arginine, asparagine and glutamine, aspartic acid, asparagine, cystine, glutamic acid, glutamine, glycine, homoserine, histidine, isoleucine, leucine, lysine, methionine, ornithine, phenylalanine, pipecolic acid, proline, serine, tyrosine, tryptophan, threonine, valine, dihydroxyphenylaline, hydroxyproline and pipecolic acid), nitrogenous compounds (ethanolamine and ammonia), organic acids (malic acid and pyridinoline), and enzymes. Furthermore, naturally-occurring phytohormones were identified in the liquid endosperm from young green and mature coconuts, including indole-3-acetic acid, various cytokinins, gibberellins and abscisic acid, and salicylic acid. The liquid endosperm of tender coconut water contains vitamin B, especially nicotinic acid (vitamin B3), pantothenic acid (vitamin B5), biotin, riboflavin (vitamin B2), folic acid, trace amounts of thiamine (vitamin B1), and pyridoxine (vitamin B6). 20
A supplier reported that Cocos Nucifera (Coconut) Liquid Endosperm is composed of 85% to 100% freeze dried tender coconut water (organic and inorganic materials), 0% to 15% water, no excipients or carriers, and has an extract ratio of 50:1. 21
A supplier reported that the levels of elemental impurities (lead, arsenic, cadmium, and mercury) for Cocos Nucifera (Coconut) Liquid Endosperm comply with the United States (US) Pharmacopeial Convention general chapter on elemental contaminants in dietary supplements. 22 The supplier also reported that the product did not contain volatile organic compounds or any substances prohibited by European Union cosmetics regulations or considered to be substances of very high concern in the European Chemicals Agency (ECHA) database. 22
Cocos Nucifera (Coconut) Shell Powder
The proximate analysis of the composition of coconut shell pericarp indicated that it is composed of carbohydrates (52.63%), crude fiber (32.39%), moisture (10.10%), ash (2.28%), crude fat/oil (2.14%), and protein (.46%). 15 The mineral composition of the coconut shell was reported to be 11.64 mg/100 g phosphorus, 16.02 mg/100 g calcium, 1.22 mg/100 g magnesium, .76 mg/100 g sodium, 3.30 mg/100 g potassium, 618.00 mg/100 g iron, 1.20 mg/100 g zinc, and of 6.00 mg/100 g manganese.
Use
Cosmetic
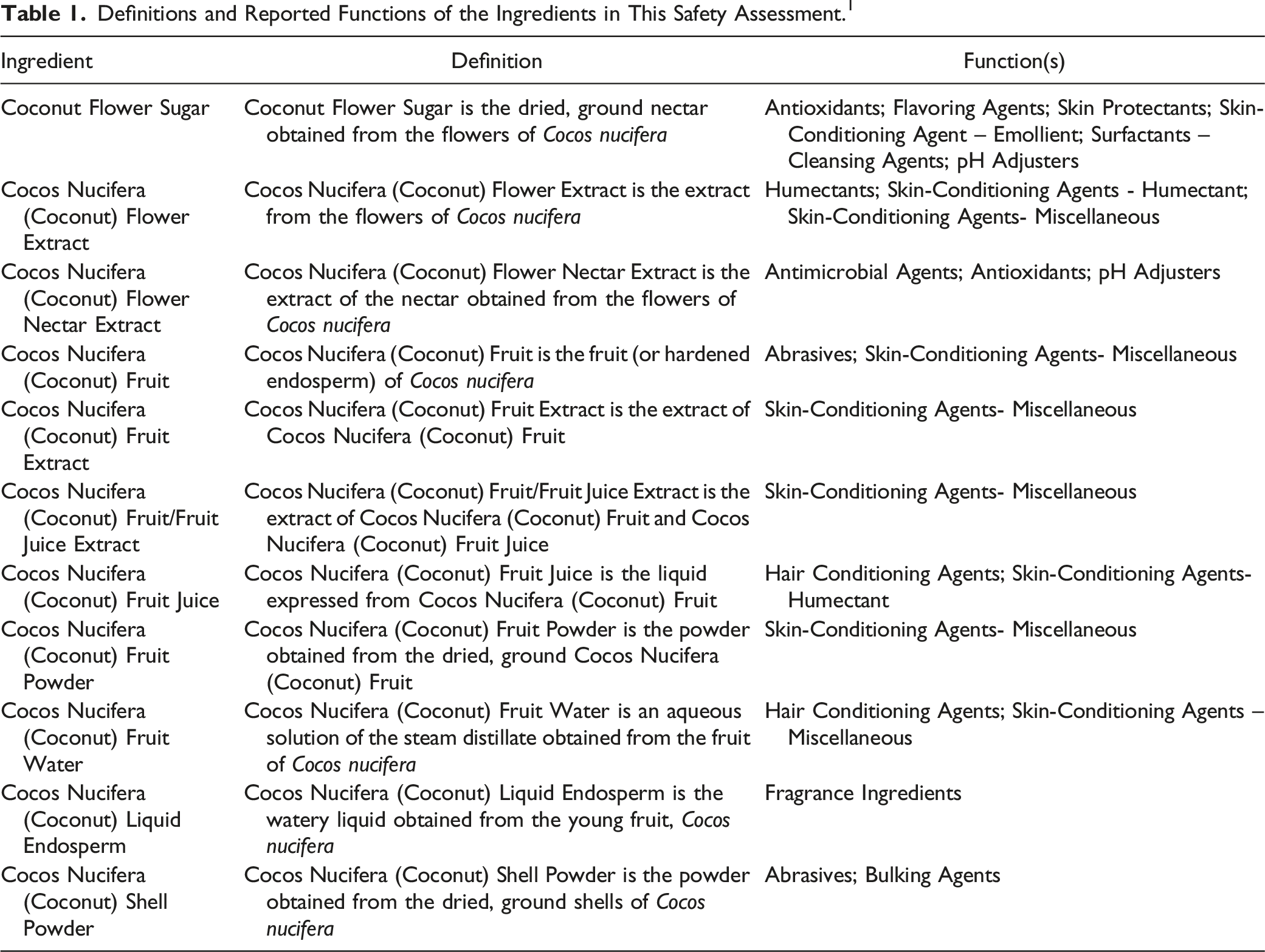
The safety of the cosmetic ingredients addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
NR = Not reported.
aCoconut Flower Extract is reported in tde VCRP; it was assumed it refers to Cocos Nucifera (Coconut) Flower Extract.
bBecause each ingredient may be used in cosmetics witd multiple exposure types, tde sum of all exposure types may not equal tde sum of total uses.
cNot specified whetder a powder or a spray, so tdis information is captured for botd categories of incidental inhalation.
dIt is possible tdese products may be sprays, but it is not specified whetder tde reported uses are sprays.
eIt is possible tdese products may be powders, but it is not specified whetder tde reported uses are powders.
fIncludes 3 entries for Cocos Nucifera (Coconut) Milk Powder, which is not an ingredient in tde Dictionary.
gMost entries are referred to as Cocos Nucifera (Coconut) Water in tde VCRP.
Cocos nucifera (coconut)-derived ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes; for example
The coconut-derived ingredients described in this report are not restricted from use in any way under the rules governing cosmetic products in the European Union. 32
Non-Cosmetic
Coconut fruit can be made into a variety of foods and beverages. 8 Coconut water is served directly as a beverage, while coconut milk is usually used as a food ingredient in various traditional cooking recipes.
The FDA requires allergen labeling when major allergens, such as tree nuts like coconut, are included in food. 33 A major food allergen is an ingredient from a food or food group, such as coconut, that contains protein derived from the food.
The juice and shell of Cocos nucifera have been researched for use as antifungal and antibacterial treatments.20,34-36 Various parts of the coconut including coconut liquid endosperm, husk fiber, and coconut juice, have also been researched for use as alternative or therapeutic treatments (as herbal medicines or dietary supplements) for dermal wounds, health promotion, and cardiovascular protection.37-41 The electrolyte composition of coconut water (e.g., potassium) can cause vasodilation in blood vessels; therefore, coconut water has been used as intravenous fluid for hydration and as a resuscitation fluid. 42 Coconut flower clusters (also called inflorescence) and coconut juice are reportedly used in traditional folk medicine in Southeast Asia to treat menstrual cycle disorders.43,44
A supplier in Thailand reports that coconut flower nectar has been used in food and beverages in South East Asia for centuries. 13 Coconut flower sugar is the main food ingredient that is obtained from coconut flower nectar, and is used in India, Indonesia, and Thailand primarily as confectionery sugar for making sweets and desserts.11,13,16 Coconut flower sugar is being researched as a natural and cheaper alternative for synthetic sweeteners. 11
Toxicokinetic Studies
No relevant toxicokinetics studies on coconut-derived ingredients were found in the public literature, and unpublished data were not submitted. In general, toxicokinetics data are not expected to be found on botanical ingredients because each botanical ingredient is a complex mixture of constituents.
Toxicological Studies
Acute Toxicity Studies
Oral
Cocos Nucifera (Coconut) Fruit Extract
An alcoholic extract of Cocos nucifera fruit was administered orally to male albino mice (6/group). 45 Mice received a single dose of Cocos nucifera fruit extract at doses of .25, .5, 1, 1.5, 2, 2.5, or 3 g/kg body weight via gavage. The control group received 1 ml of saline. Mice were observed for toxicity, morbidity, or mortality for 24 h. No deaths, significant decreases in body weight, or gross pathological abnormalities were observed.
Developmental and Reproductive Toxicity (dart) Studies
Cocos Nucifera (Coconut) Fruit Extract
The potential reproductive toxicity of an alcoholic extract of Cocos nucifera fruit was tested in 2 groups of male albino mice (8/group). 45 A negative control group was administered saline, and test groups 1 and 2 were given 125 mg/kg and 200 mg/kg of the alcoholic extract, respectively, of coconut fruit for 15 d. The left and right testis with epididymides were removed and weighed after removing the fat and surrounding tissues. Sperm counts were made from a homogenized solution of saline and caudal epididymides. There was a significant (P < .01) increase in relative testicular weight/body weight in test group 1 compared to the control group. However, test group 2 showed a significant decrease in testis weight/body weight compared to the control group and test group 1. Also, an increase in sperm concentration was noted in male mice administered 125 mg/kg of coconut fruit extract.
Cocos Nucifera (Coconut) Liquid Endosperm
The conceptive and anti-abortive effects of coconut water was studied using groups of 5 gravid female albino rats. 46 The rats received coconut water starting the day after the rats were mated. The animals were dosed for 21 d (gestation period) as follows: group 1 (control group) was given 1.0 ml/100 g of distilled water; group 2 was given .5 ml/100 g of coconut water; group 3 received 1.0 ml/100 g coconut water; and group 4 was administered 2.0 ml/100 g coconut water. The rats were weighed on days 1, 7, 14, and 21. Rats in groups 1, 2, and 3 underwent laparotomy on day 10 of pregnancy, while group 4 delivered on day 21. (No additional details were readily available.) Upon delivery, the number of offspring from each female was counted. An increase in body weight was noted in pregnant female rats of all dose groups compared to the control. No significant differences in the implantation site and number of litters between treated groups and the control group were observed. The authors determined that low doses of coconut water protracted the gestation period, but not significantly.
The reproductive effects of coconut water were studied in male Wistar rats (5/group). 47 The first group was given 20 ml/kg of distilled water and served as the control. Group 2 was orally administered 20 ml/kg of corn oil, group 3 was given 20 ml/kg of coconut water, group 4 was administered 200 mg/kg of danazol (a synthetic androgen), and group 5 was given 200 mg/kg of danazol and 20 ml/kg coconut water. The rats received the test materials daily for 6 wk. The rats showed a significant (P < .05) increase in sperm count, sperm motility, and sperm viability in groups 3 and 5 while a significant reduction in these variables was observed in group 4. A significant increase in luteinizing hormone (LH), follicle-stimulating hormone (FSH) and testosterone levels were observed in groups 3 and 5 when compared with group 4. All test groups showed a significant increase in reduced glutathione, total protein, and a significant reduction in malondialdehyde in testicular homogenates, when compared with the control and corn oil groups.
Genotoxicity
In Vitro
Cocos Nucifera (Coconut) Liquid Endosperm
The genotoxicity potential of a trade name mixture containing 98% Cocos Nucifera (Coconut) Liquid Endosperm and 2% Leuconostoc/radish root ferment filtrate was evaluated in a bacterial reverse mutation assay using S. typhimurium strains: TA98, TA100, TA1537, and TA1535, and Escherichia coli strain WP2uvrA. 48 The test article dissolved in sterile distilled water was administered at concentrations of 1.5, 5.0, 15, 50, 150, 500, 1500, and 5000 μg/plate, with and without metabolic activation. Appropriate positive and negative controls were used. The trade name mixture containing Cocos Nucifera (Coconut) Liquid Endosperm was non-genotoxic.
In Vivo
No in vivo genotoxicity studies were discovered in the published literature, and no unpublished data were submitted.
Carcinogenicity
No carcinogenicity studies were discovered in the published literature, and no unpublished data were submitted.
Other Relevant Studies
Hormone Replacement
Cocos Nucifera (Coconut) Fruit Juice
A study was performed to analyze the estrogenic effects of young coconut fruit juice using 4 groups of female Wistar rats (10/group). 41 The test material was prepared from reconstituted young coconut juice powder daily for oral administration. The first group consisted of ovariectomized rats, the second group consisted of sham-operated rats (placebo surgery), the third group consisted of ovariectomized rats injected intraperitoneally with exogenous estrogen (2.5 µg/kg of estradiol benzoate) twice a week for 4 wk, and the fourth group consisted of ovariectomized rats that were orally administered young coconut fruit juice (100 ml/kg/d) for 4 wk. The rats were killed on the first day of the sixth week of the study, and their serum estradiol (E2) level was measured by chemiluminescent immunoassay. The ovariectomized rat group administered young coconut juice had significantly higher serum E2 levels (P < .05) than the control ovariectomized group. A significant increase (P < .05) was also observed when compared with the sham-operated group. A significant reduction (P < .05) in neuronal cell death was observed in ovariectomized rats administered young coconut fruit juice compared to the control ovariectomized group. In contrast, circulating E2 was lowest in the ovariectomized rats fed with 100 ml/kg young coconut fruit juice plus 2.5 μg/kg E2. In addition, wound healing was significantly accelerated in ovariectomized rats receiving 100 ml/kg young coconut juice compared with any other groups. The authors presumed that young coconut juice contains estrogen-like hormones which showed an antagonist effect with endogenous E2 by competing for estrogen receptors. However, chemical structure data of presumable phytoestrogens were not provided.
Cytotoxicity
Cocos Nucifera (Coconut) Shell Powder
The cytotoxic properties of coconut shell crude extract (extracted by 3 h in boiling distilled water; concentrations of crude extract ranging from .1 mg/ml to 5 mg/ml) was evaluated in a DNA fragmentation analysis involving immortalized human epithelioid (HeLa) cells. 49 After a 48-h incubation period, cell viability was measured using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. At concentrations ranging from .1 mg/ml to 5 mg/ml, the percentage of inhibition gradually increased from 60% to 85%. The median inhibitory concentration (IC50) was 1.77 mg/ml. The authors concluded that the cytotoxic activity was found due to the apoptosis induced in HeLa cells, which was demonstrated by DNA fragmentation analysis.
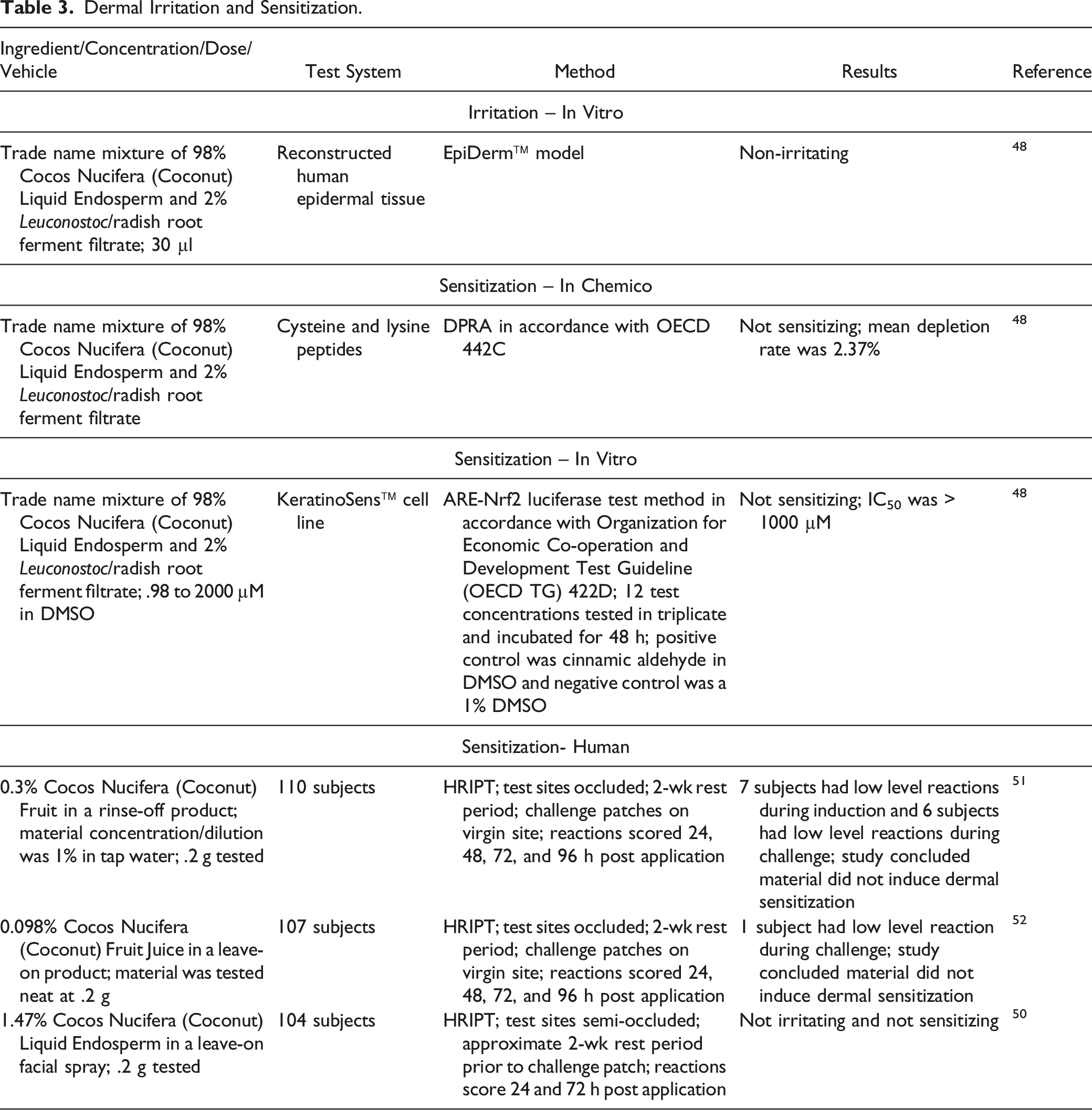
Dermal Irritation and Sensitization
Dermal Irritation and Sensitization.
Ocular Irritation Studies
In Vitro
Cocos Nucifera (Coconut) Liquid Endosperm
The EpiOcularTM model (human corneal epithelial model) assay was used to evaluate the irritation potential of a trade name material containing 98% Cocos Nucifera (Coconut) Liquid Endosperm and 2% Leuconostoc/radish root ferment filtrate. 48 Each tissue was dosed with 20 µl Dulbecco’s phosphate buffered saline (DPBS) prior to administering the test article. Fifty microliters of the undiluted test article were then applied to 2 tissue inserts which were incubated for 30 min for liquid substances and 90 min for solid substances. Sterile DPBS and sterile deionized water were used as negative controls and 5% sodium dodecyl sulfate solution and methyl acetate were used as positive controls. The test substance was considered to be non-irritating.
Summary
According to the Dictionary, most of the 11 Cocos nucifera (coconut)-derived ingredients detailed in this safety assessment are reported to function in cosmetics as skin-conditioning agents, while some are reported to have other functions, such as humectants, abrasives, and hair conditioning agents. Additionally, some of the ingredients reviewed in this safety assessment may be consumed in food, and daily exposure from food use would result in much larger systemic exposures than those from use in cosmetic products. Therefore, the primary focus of the safety assessment of these ingredients as used in cosmetics is on the potential for local effects (i.e. from topical exposure).
According to 2021 VCRP data, Cocos Nucifera (Coconut) Fruit Extract is reported to be used in 327 formulations, 196 of which are leave-on formulations. All other in-use ingredients are reported to be used in 126 formulations or less. The results of the concentration of use survey conducted by the Council in 2019 indicate that Cocos Nucifera (Coconut) Liquid Endosperm has the highest maximum concentration of use; it is used at up to 6.5% in shampoos (non-coloring). Cocos Nucifera (Coconut) Liquid Endosperm also has the highest maximum concentration of use in dermal leave-on formulations; it is used at up to 1.5% in face and neck products. Cocos Nucifera (Coconut) Fruit Extract is reported to be used at up to .02% in leave-on hair products. No uses were reported in the VCRP or by the Council for Coconut Flower Sugar, Cocos Nucifera (Coconut) Flower Nectar Extract, or Cocos Nucifera (Coconut) Fruit/Fruit Juice Extract.
Coconut fruit and coconut water (i.e. coconut liquid endosperm) can be made into a variety of foods and beverages. The juice and shell have been researched for use in alternative or therapeutic treatments. Coconut flower nectar and coconut flower sugar are reported to have been used in food and beverage in South East Asia for centuries.
In an acute oral toxicity study, an alcoholic extract of Cocos nucifera fruit was administered orally to male mice at doses up to 3 g/kg body weight via gavage. No deaths, significant decreases in body weight, or gross pathological abnormalities were observed.
An alcoholic extract of Cocos nucifera fruit increased relative testicular weight/body weight at 125 mg/kg, but significantly decreased testis weight/body weight at 200 mg/kg in male mice. An increase in sperm concentration was noted in male mice that received 125 mg/kg of the test material. In a study of female rats, coconut water at up to 2.0 ml/100 g caused an increase in body weight in pregnant rats when compared to the control. No significant differences in the implantation site or number of litters between treated groups and the control group were observed. In a study of coconut water in male rats, significant increases in sperm count, sperm motility, and sperm viability in groups that received coconut water were observed. A significant increase in LH, FSH, and testosterone levels were also observed in these groups.
A trade name mixture containing Cocos Nucifera (Coconut) Liquid Endosperm (98%) and 2% Leuconostoc/radish root ferment filtrate was not genotoxic in an Ames assay, with or without metabolic activation.
A study performed to analyze the estrogenic effects of coconut fruit juice in rats found that ovariectomized rats that received young coconut juice had significantly higher serum E2 levels than the control ovariectomized rats and sham-operated rats. In contrast, circulating E2 was lowest in the ovariectomized rats fed with both young coconut fruit juice and E2. A significant reduction in neuronal cell death as well as accelerated wound healing was observed in ovariectomized rats administered young coconut fruit juice when compared to the control ovariectomized group.
The cytotoxic properties of coconut shell crude extract (concentrations of crude extract ranging from .1 mg/ml to 5 mg/ml) was evaluated in a DNA fragmentation analysis involving immortalized HeLa cells. The percentage of inhibition gradually increased from 60% to 85%. The median IC50 was 1.77 mg/ml.
The results of an in vitro dermal irritation study of a trade name mixture of 98% Cocos Nucifera (Coconut) Liquid Endosperm and 2% Leuconostoc/radish root ferment filtrate found these substances non-irritating. A trade name mixture containing 98% Cocos Nucifera (Coconut) Liquid Endosperm and 2% Leuconostoc/radish root ferment filtrate was predicted to be non-sensitizing by DPRA. A rinse-off product containing .3% Cocos Nucifera (Coconut) Fruit (diluted 1% in tap water), a leave-on product containing .098% Cocos Nucifera (Coconut) Fruit Juice (undiluted), and a leave-on product containing 1.47% Cocos Nucifera (Coconut) Liquid Endosperm were not irritating and not sensitizing in HRIPTs.
The results of an in vitro ocular irritation study of a trade name mixture containing 98% Cocos Nucifera (Coconut) Liquid Endosperm and 2% Leuconostoc/radish root ferment filtrate found this mixture non-irritating.
No relevant in vivo genotoxicity studies or carcinogenicity studies were discovered in the published literature, and no unpublished data were submitted. No relevant toxicokinetics studies were found in the published literature; however, in general, toxicokinetics data are not expected to be found on botanical ingredients because each botanical ingredient is a complex mixture of constituents.
Discussion
The Panel reviewed the safety of 11 botanical ingredients derived from the plant, Cocos nucifera, and concluded that the data are sufficient for determining safety of 10 ingredients, i.e., those derived from coconut flower, fruit, and liquid endosperm, as reportedly used in cosmetics. The Panel noted the lack of toxicity and carcinogenicity data on the coconut flower, fruit, and liquid endosperm; however, these ingredients are consumed as food, and daily exposure from food use would result in much larger systemic exposures than possible from use in cosmetic products. Additionally, the Panel noted the study of estrogen-like property in young coconut juice; however, the DART studies on coconut liquid endosperm do not implicate any reproductive effects. This, coupled with the very weak estrogenic effects noted in the study that used a concentration greater than that used in cosmetic products, helped mitigate concern.
Some Cocos nucifera-derived ingredients are reported to be used in spray products that could possibly be inhaled; for example, Cocos Nucifera (Coconut) Liquid Endosperm is used in a face and neck spray at 1.5%. There were no inhalation toxicity data available; however, the Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The Panel expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients. Therefore, they stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
However, the Panel also concluded that the data for are insufficient for determining safety of Cocos Nucifera (Coconut) Shell Powder as reportedly used in cosmetics. Specifically, the Panel stated that the following data are needed: • Composition and impurities data • Concentration of use • Dermal irritation and sensitization data
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following 10 Cocos nucifera (coconut)-derived ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment: Coconut Flower Sugar* Cocos Nucifera (Coconut) Flower Extract Cocos Nucifera (Coconut) Flower Nectar Extract* Cocos Nucifera (Coconut) Fruit Cocos Nucifera (Coconut) Fruit Extract Cocos Nucifera (Coconut) Fruit/Fruit Juice Extract* Cocos Nucifera (Coconut) Fruit Juice Cocos Nucifera (Coconut) Fruit Powder Cocos Nucifera (Coconut) Fruit Water Cocos Nucifera (Coconut) Liquid Endosperm
*Not reported to be in current use. Were this ingredient not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
The Panel also concluded that the available data are insufficient to make a determination of safety for Cocos Nucifera (Coconut) Shell Powder under the intended conditions of use in cosmetic formulations.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.