Abstract
The NOD/SCID/IL2Rγnull (NSG) mouse is a relevant model for toxicology and tumorigenicity studies evaluating human cell therapies. Data was compiled from toxicology study control NSG mice exposed to gamma irradiation (0 or 200 cGy) or busulfan. Retrospective data evaluation included mortality, clinical observations, body weights, hematology, and external and internal macroscopic observations. There was no mortality in any of the 129 toxicology control (irradiated and non-irradiated) mice up to the 20-week observation period. Mortalities occurred between Days 1 and 25 among animals given busulfan ≥25 mg/kg/day at 1 or 2 doses via intraperitoneal (i.p.) injection. There were 4/10, 6/10 and 4/10 deaths at 25, 30 and 35 mg/kg/day busulfan, respectively. Busulfan-treated mice presented with dose-dependent clinical signs including signs of anemia in some individuals. Hematology, including white blood cell (WBC) and neutrophil (NEUT) counts, from irradiated mice at Weeks 12 and 20 revealed comparable values to non-irradiated animals. In contrast, irradiated mice treated with a positive control (HL-60) were euthanized prior to Week 12. There were no irradiation-related differences in macroscopic observations with lymphoid atrophy identified comparably in irradiated and non-irradiated groups. These results suggest that irradiation was suitable for conditioning to enable cell engraftment in NSG mice in the context of regulatory toxicology and tumorigenicity studies. Busulfan administered at 20 mg/kg/day for 2 days, i.p. was also well-tolerated, and it could be considered for toxicology studies of genetically modified human cells.
Introduction
Humanized mice that have been developed to partially reconstitute the human immune system have greatly facilitated the study of human diseases. These mice models have become feasible as a result of the development of immunocompromised strains of mice by which human cells can be successfully engraft. 1 The NOD/SCID/IL2Rγnull (NSG) mouse model, for example, is a double homozygous for the combined immunodeficiency (SCID) mutation and interleukin-2Rγ (IL-2Rγ) allelic mutation (γ nullc),2,3 resulting in a highly immunodeficient mouse strain deficient in mature lymphocytes and NK cells. 4 In order to improve engraftment levels of human cells in murine models such as the NSG mouse, Total Body Irradiation (TBI) is a frequent method used for myeloablation or conditioning. The main purpose of TBI is to ablate lymphocyte and myeloid populations prior to immune system reconstitution with human donor grafts 5 or prior to human cell therapy in the case of toxicology studies.
Although there are benefits to irradiation-based conditioning, there can also be risks and complications associated. TBI is not selective in cell targeting and high doses can lead to damage to rapidly replicating cell types, for example, cells of the gastrointestinal crypts resulting in increased susceptibility to opportunistic infections (eg., Pseudomonas aeruginosa, E coli, Clostridium sp., and Klebsiella). Some of the risks and complications which have been associated following gamma irradiation have included weight loss, anemia, infection, gastrointestinal hemorrhage, secondary neoplasia (long-term studies).6,7
As an alternative, myelosuppressive conditioning can be performed with a chemotherapeutic agent such as busulfan. Busulfan is a potent cytotoxic drug and a bifunctional alkylating agent that can result in profound myelosuppression. 8 An advantage of using busulfan over irradiation is that, unlike irradiation which requires specialized facilities, busulfan conditioning only requires i. p. injections and is generally well-tolerated at appropriate doses. 9 It has also been suggested that busulfan may be a more suitable technique to generate bone marrow chimerism in young, old, or diseased mice that are more susceptible to myeloablative irradiation. 9 As busulfan has little effect on lymphocytes, it can be administered in combination with cyclophosphamide (CY) or fludarabine (FLU) in preparative regimens for allogeneic stem cell transplantation when lymphodepletion is required. 10
The purpose of this study was to evaluate retrospective control data from studies previously conducted at Charles River Laboratories Laval. Data were compiled from NOD/SCID/IL2Rγnull (NSG) mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min) (N = 41 males, 68 females) or 0 cGy (N = 10 males, 10 females) which had then been dosed with a vehicle (PBS or PBS/0.1% Human Serum Albumin) or positive control (Human Leukemia-60) via a single intravenous injection and identify differences observed in irradiated vs non-irradiated group. In addition, from a separate cohort of animals, results from irradiation vs busulfan-treated NSG mice were compared for conditioning using gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min) (N = 4 males, 4 females) or busulfan administered either once or twice via i. p. injections at dose levels of 20, 25, 30 and 35 mg/kg/day (N = 5 males/group, 5 females/group). The data evaluated across these studies include toxicology endpoints such as mortality, clinical observations, body weights, hematology, external and internal macroscopic observations.
Methods and Materials
A retrospective analysis was done across studies performed at Charles River Laboratories Laval on irradiated or non-irradiated control NOD/SCID/IL2Rγnull (NSG) mice. During these studies, care and use of animals were conducted in accordance with principles outlined in the current Guide to the Care and Use of Experimental Animals published by the Canadian Council on Animal Care and the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 2010). The Charles River Laval facility is AAALAC accredited. All procedures were conducted as per Standard Operating Procedures (SOPs).
Evaluations Performed and Samples Sizes of Non-irradiated and Irradiated NSG Mice.
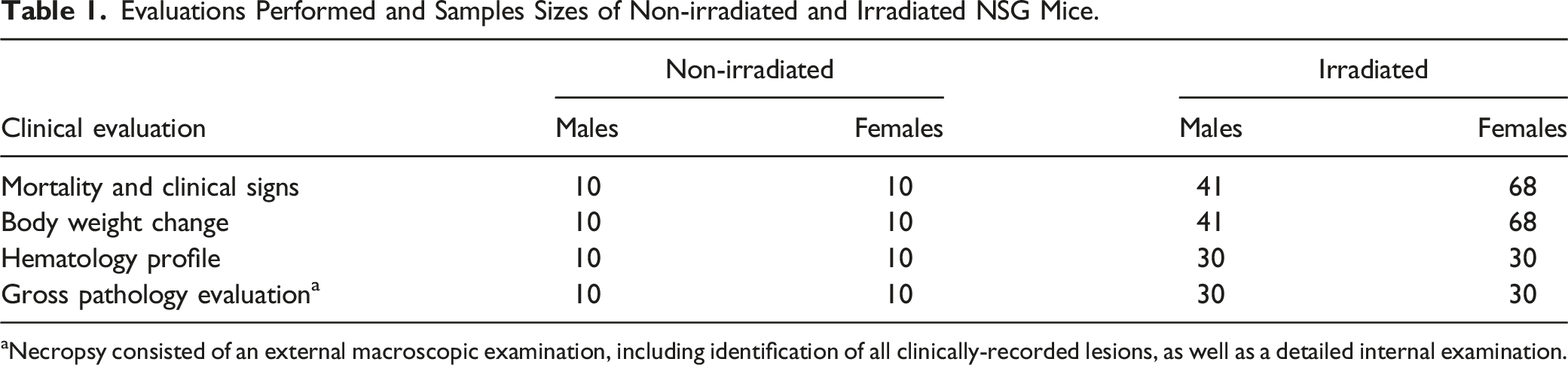
aNecropsy consisted of an external macroscopic examination, including identification of all clinically-recorded lesions, as well as a detailed internal examination.
In some studies, a positive irradiated control (human promyelocytic leukemic cells, HL-60) group was included into the study as a concurrent control under the same experimental conditions. As the purpose of this current evaluation was to compare control irradiated non-transplanted mice vs control non-irradiated non-transplanted mice, results from the positive control group were not compiled nor included in our evaluation. In these studies, animals were either found dead or had to be euthanized prior to Week 12 due to deteriorating clinical signs, which met euthanasia criteria.
In a second cohort of animals, a study was performed to compare irradiated vs busulfan-treated NSG mice at different dose levels. Mice were provided by Jackson Laboratories (Bar Harbor, Maine 04 609 USA) and were 9 weeks old at the onset of dosing and weighed between 15 to 25 g. Irradiated mice (Group 2) were exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min) (N = 4 males and 4 females). Treated animals were administered Bulsulfex® (Otsuka Pharmaceutical) at dose-levels of either 20 (Group 3), 25 (Group 4), or 30 (Group 5) mg/kg/day via i. p. injections twice over 2 consecutive days or 35 (Group 6) mg/kg/day once. The number of animals for each dose level consisted of 5 males and 5 females. Non-irradiated control animals (N = 4 males and 4 females) received 0.9% sodium chloride for injection, USP (saline) alone. Clinical evaluations presented here also included common toxicology endpoints such as mortality and clinical signs, bodyweight change, hematology profile and gross pathology evaluation.
Mortality and Clinical Signs
Clinical signs were recorded on all surviving animals across all studies at least twice daily for at least 1 week prior to irradiation and throughout the observation period. In addition, detailed clinical examinations were performed on each animal prior to animal assignment, 1 day prior to irradiation, and then every 3 days throughout the observation period and prior to necropsy. The incidence of clinical signs for negative control irradiated and non-irradiated mice (males and females combined) during the course of the studies was collected and the number of animals presenting signs on 1 or more occasions (days) was tabulated.
For the busulfan-treated vs irradiated and non-irradiated mice study, clinical signs were recorded twice daily on all animals for at least 1 week prior to irradiation and throughout the observation period and a detailed clinical examination was performed on each animal prior to group assignment, then weekly starting on Day −8, throughout the observation period and including the day prior to irradiation (Group 2), the day prior to dosing (Groups 1, 3 to 6) and prior to euthanasia. The number of animals presenting signs on 1 or more occasions (days) was tabulated.
Body Weights
Across all studies, body weights were recorded for all animals at least once prior to animal assignment, on Day −7 or −8, on the day prior to irradiation (Day −1), and then either every 3 or 6 days throughout the observation period, and prior to necropsy (non-fasted). Body weight changes were calculated for males and females separately (irradiated vs non-irradiated).
For the busulfan-treated vs irradiated or non-irradiated mice comparison, body weights were recorded for all animals at least once prior to animal assignment, on Day −8, on the day prior to irradiation (Group 2), the day prior to busulfan dosing (Groups 1, 3 to 8), then every 3 days throughout the observation period, and prior to unscheduled or scheduled euthanasia. Body weight changes were calculated for males and females separately.
Hematology
At scheduled necropsies, blood samples were collected into tubes containing K3-EDTA as an anticoagulant and were analyzed for their hematology parameters. As only 40% of the studies had terminal necropsies at Week 12, it was only possible to compile hematology parameters for these studies and Week 40 data, for which limited data were available, were separately included. Hematology parameters presented here include red blood cell (RBC), platelet, white blood cell (WBC), neutrophil, lymphocyte, monocyte, eosinophil, basophile and reticulocyte counts, and hematocrit.
Gross Pathology Evaluation
For all animals euthanized as well as animals found dead across all studies, necropsy consisted of an external macroscopic examination, including identification of all clinically-recorded lesions, as well as a detailed internal examination and with a special focus on signs of Graft vs Host Disease (GvHD). For 40% of the studies, necropsy was performed at Week 12 following irradiation and dosing therefore we were able to compare the gross pathology findings between non-irradiated vs irradiated controls using data compiled from these studies.
For the irradiated vs busulfan comparison, all animals euthanized (as well as animals found dead) were subjected to a necropsy, which consisted of an external macroscopic examination, including identification of all clinically-recorded lesions, as well as a detailed internal examination.
Results
Mortality and Clinical Signs
Incidence of Clinical Signs During the Course of all Studies (males and females Combined) for Non-irradiated Versus Irradiated Controls.
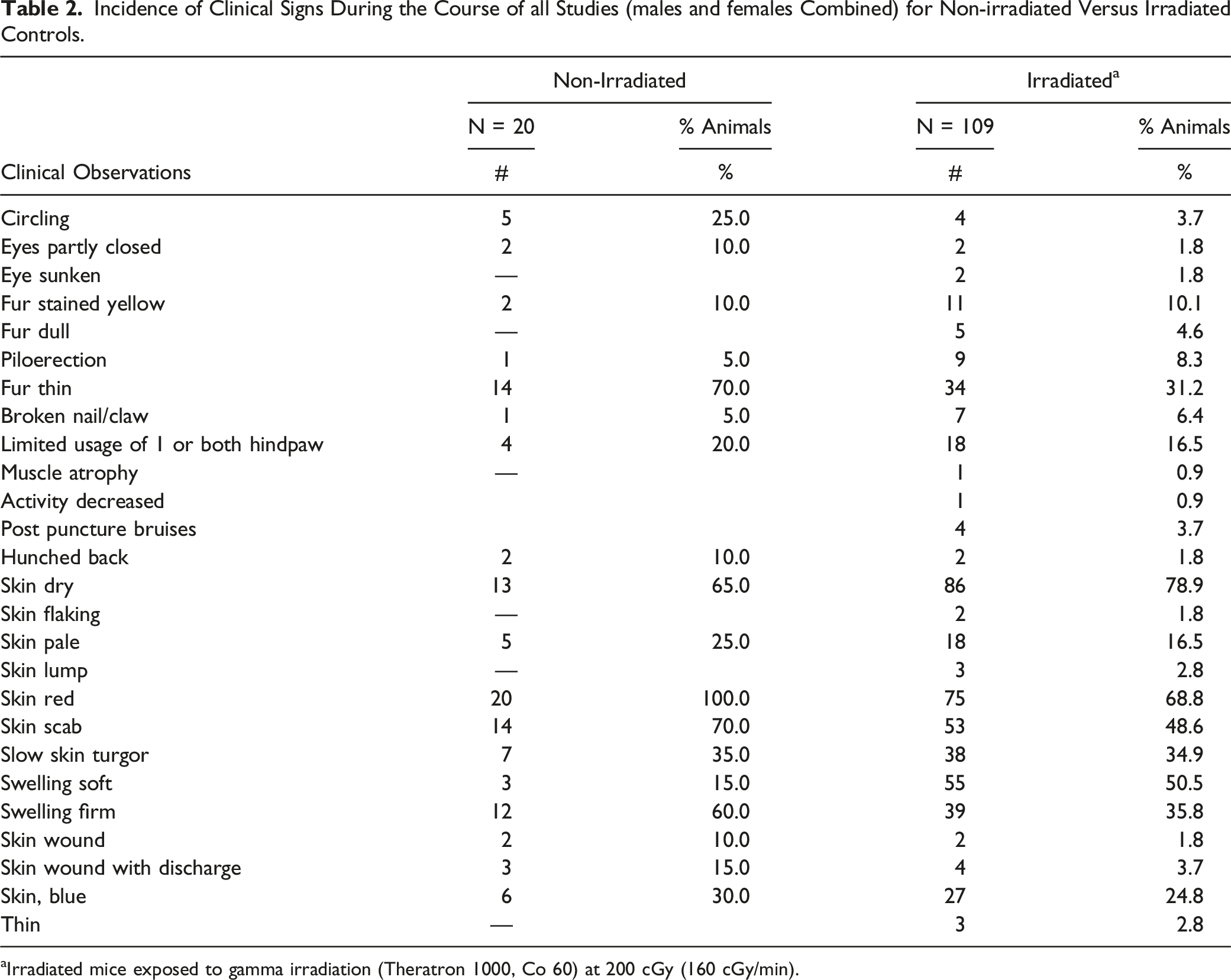
aIrradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
Incidence of Clinical Signs Comparing Busulfan-Treated NSG Mice Versus Irradiated and Non-irradiated NSG Mice.
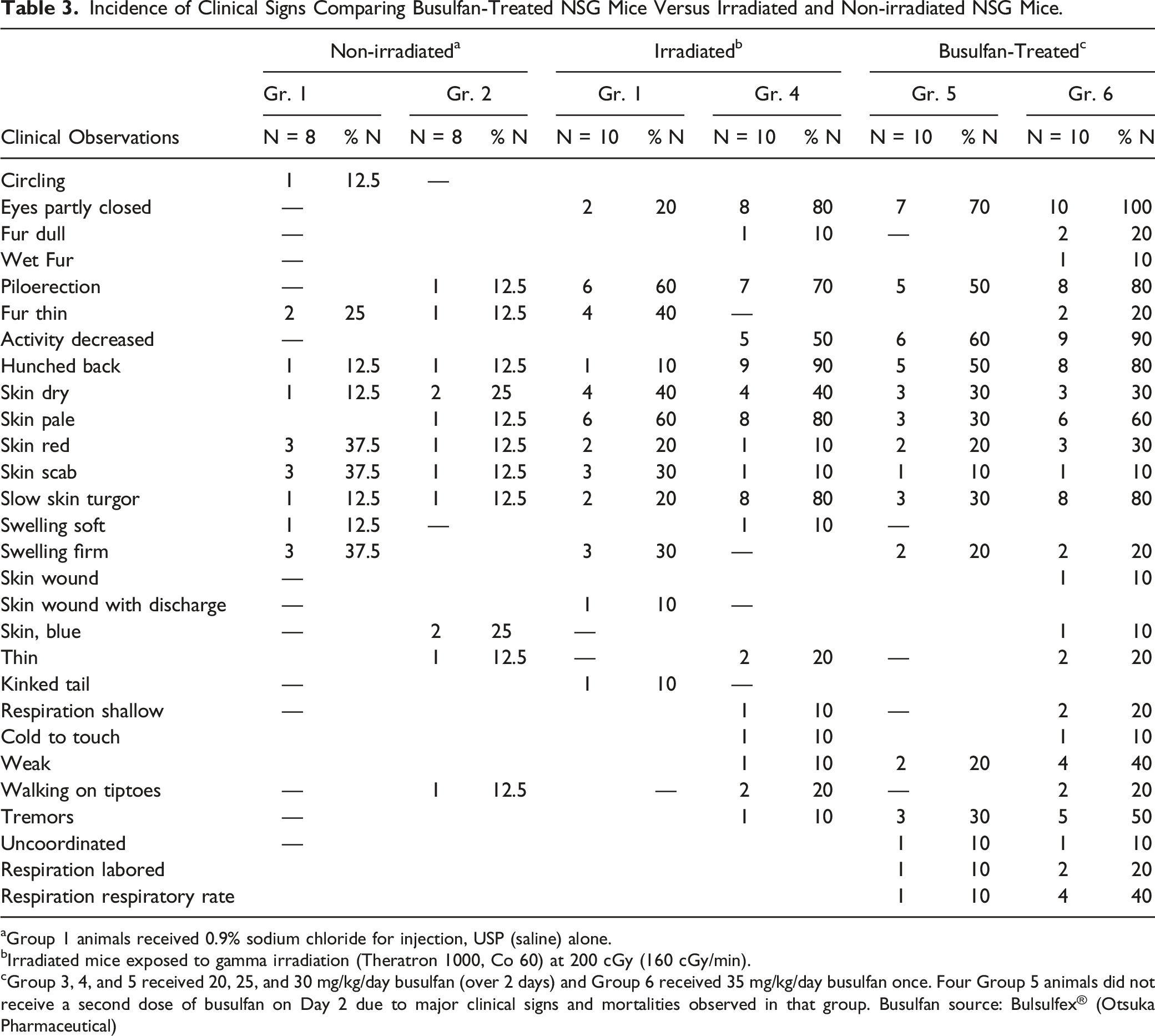
aGroup 1 animals received 0.9% sodium chloride for injection, USP (saline) alone.
bIrradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
cGroup 3, 4, and 5 received 20, 25, and 30 mg/kg/day busulfan (over 2 days) and Group 6 received 35 mg/kg/day busulfan once. Four Group 5 animals did not receive a second dose of busulfan on Day 2 due to major clinical signs and mortalities observed in that group. Busulfan source: Bulsulfex® (Otsuka Pharmaceutical)
Body Weights
Body weight percentage (%) from baseline was compiled from all studies to compare non-irradiated control mice vs irradiated control mice. Figure 1 and Figure 2 show the results for males and females, respectively. The irradiated group showed a minor decrease in body weight; however, the body weights at 21 days post-irradiation were increased and similar to that of non-irradiated controls. Body weight percent (%) change from baseline in gamma-irradiated (Theratron 1000, Co 60) at 200 cGy (160 cGy/min) vs non-irradiated males (A) and females (B). Body weight percent (%) change from baseline in busulfan-treated animals vs gamma-irradiated (Theratron 1000, Co 60) at 200 cGy (160 cGy/min) and non-irradiated controls in males (A) and females (B). Bulsulfex® (Otsuka Pharmaceutical).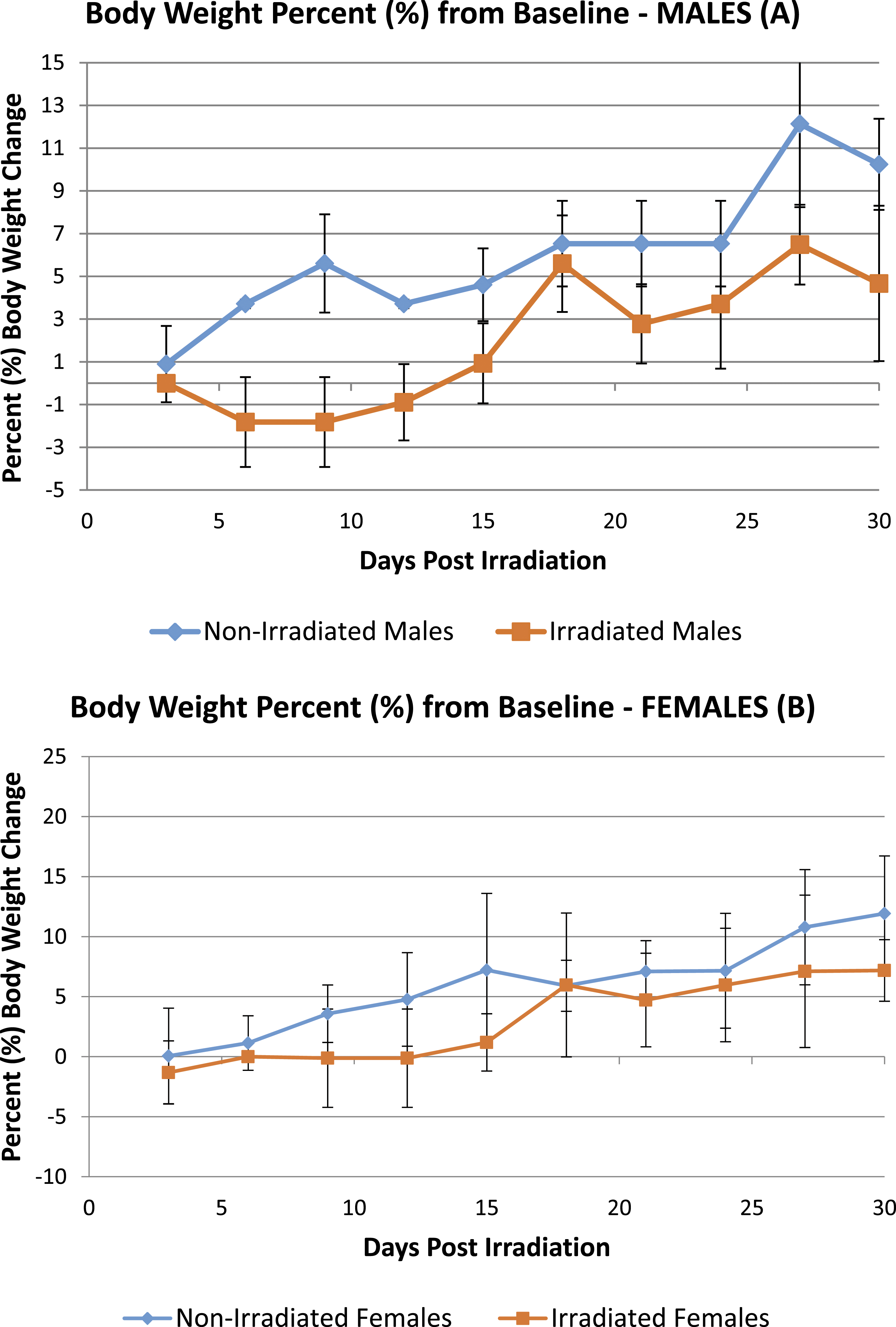
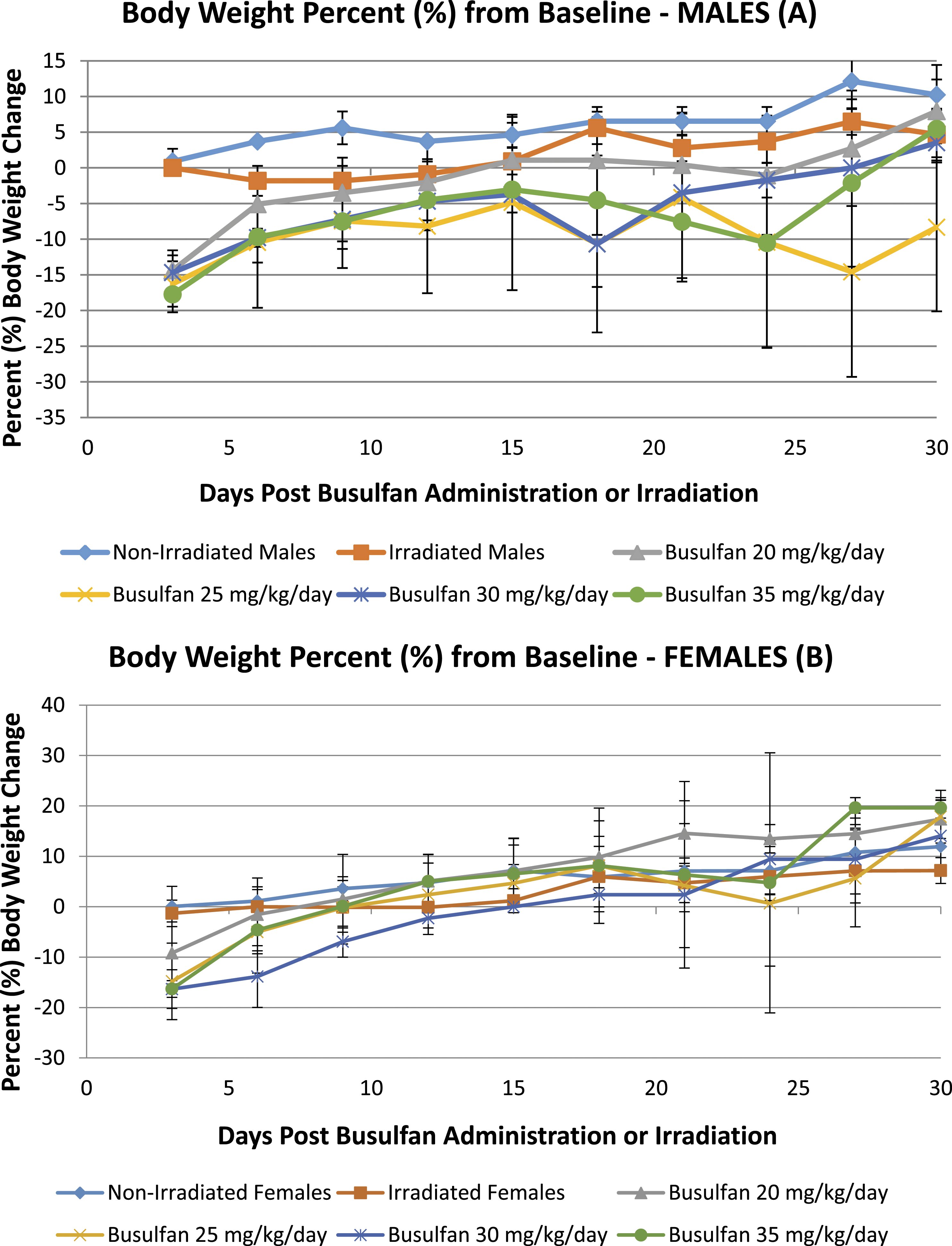
Body weight percentage (%) from baseline was calculated to compare busulfan-treated mice at 20, 25 and 30 mg/kg/day (over 2 days) and 35 mg/kg/day (1 day) vs gamma-irradiated mice and non-irradiated controls. Results showed body weight reductions following gamma-irradiation or busulfan administration on Day 3 compared to non-irradiated controls; however, decrease in body weight was more pronounced in busulfan-treated animals when compared to gamma-irradiated animals. Busulfan-related changes in body weight percentages were observed in both males and females at ≥ 20 mg/kg/day. Decreases were noted to be more severe in males and females, which received 2 doses of 25 mg/kg.
Hematology Evaluation
Hematology Profile on Day of Scheduled Necropsy for Non-irradiated and Irradiated Controls (Week 12) for 40% of the Studies and (Week 20) for 20% of the Studies.
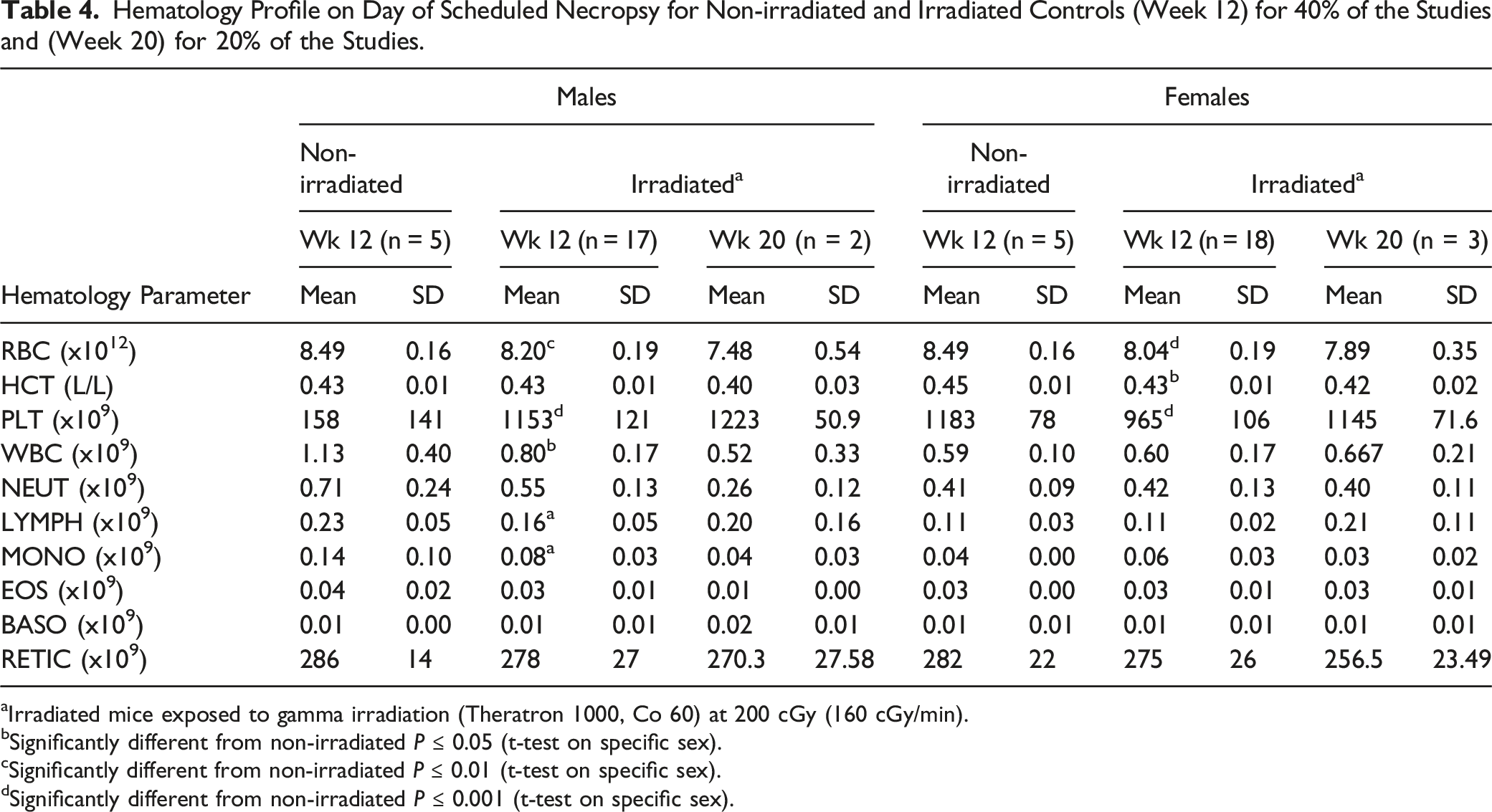
aIrradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
bSignificantly different from non-irradiated P ≤ 0.05 (t-test on specific sex).
cSignificantly different from non-irradiated P ≤ 0.01 (t-test on specific sex).
dSignificantly different from non-irradiated P ≤ 0.001 (t-test on specific sex).
Hematology Profile on Day of Scheduled Necropsy (Day 37) for Non-irradiated and Irradiated Controls and Busulfan-Treated males.
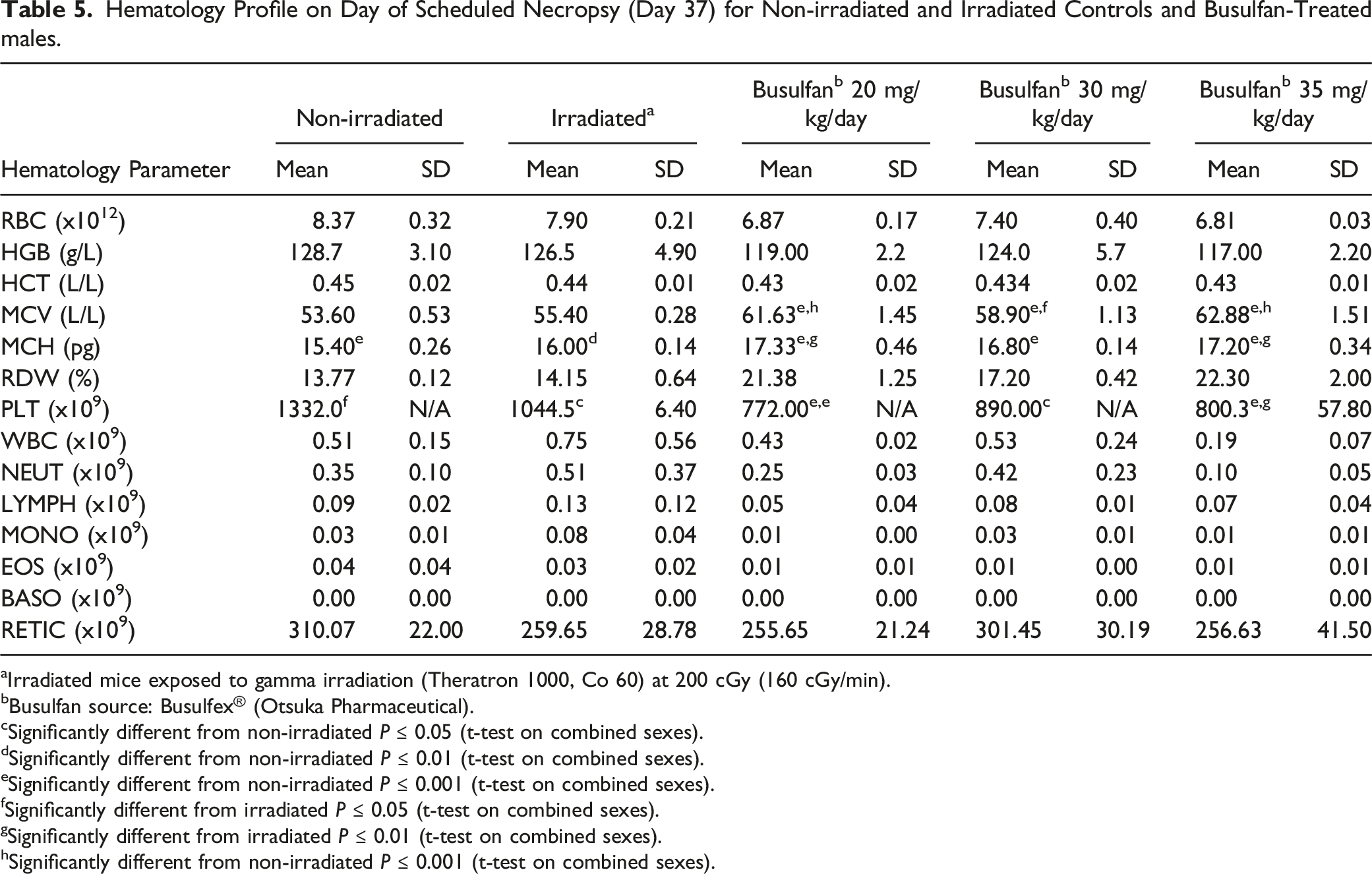
aIrradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
bBusulfan source: Busulfex® (Otsuka Pharmaceutical).
cSignificantly different from non-irradiated P ≤ 0.05 (t-test on combined sexes).
dSignificantly different from non-irradiated P ≤ 0.01 (t-test on combined sexes).
eSignificantly different from non-irradiated P ≤ 0.001 (t-test on combined sexes).
fSignificantly different from irradiated P ≤ 0.05 (t-test on combined sexes).
gSignificantly different from irradiated P ≤ 0.01 (t-test on combined sexes).
hSignificantly different from non-irradiated P ≤ 0.001 (t-test on combined sexes).
Hematology Profile on Day of Scheduled Necropsy (Day 37) for Non-irradiated and Irradiated Controls and Busulfan-Treated females.
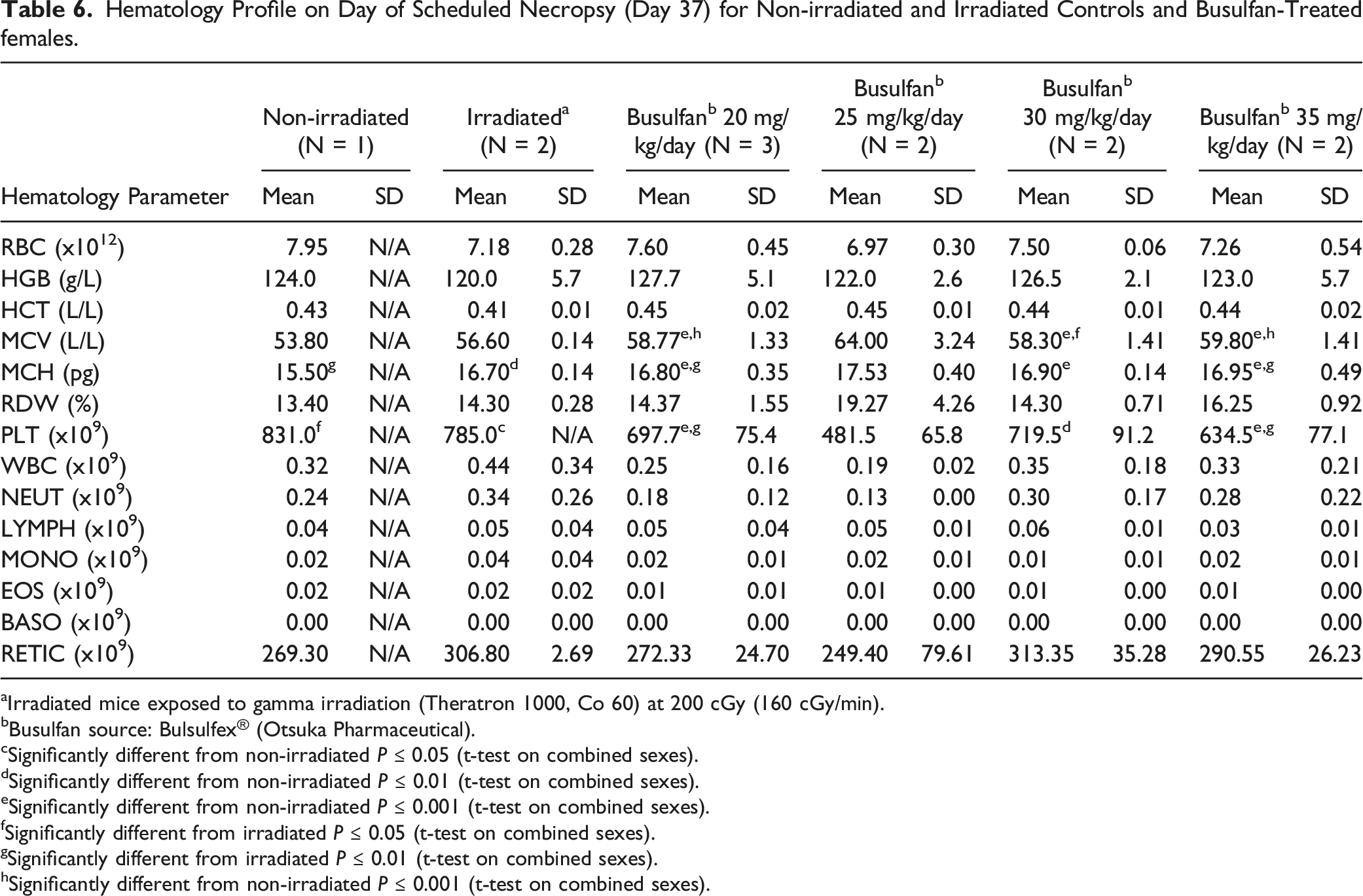
aIrradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
bBusulfan source: Bulsulfex® (Otsuka Pharmaceutical).
cSignificantly different from non-irradiated P ≤ 0.05 (t-test on combined sexes).
dSignificantly different from non-irradiated P ≤ 0.01 (t-test on combined sexes).
eSignificantly different from non-irradiated P ≤ 0.001 (t-test on combined sexes).
fSignificantly different from irradiated P ≤ 0.05 (t-test on combined sexes).
gSignificantly different from irradiated P ≤ 0.01 (t-test on combined sexes).
hSignificantly different from non-irradiated P ≤ 0.001 (t-test on combined sexes).
Gross Pathology Evaluation
Comparison of Gross Pathology Findings Between Non-irradiated and Irradiated Controls at Week 12 Scheduled Termination for 40% of the Studies.
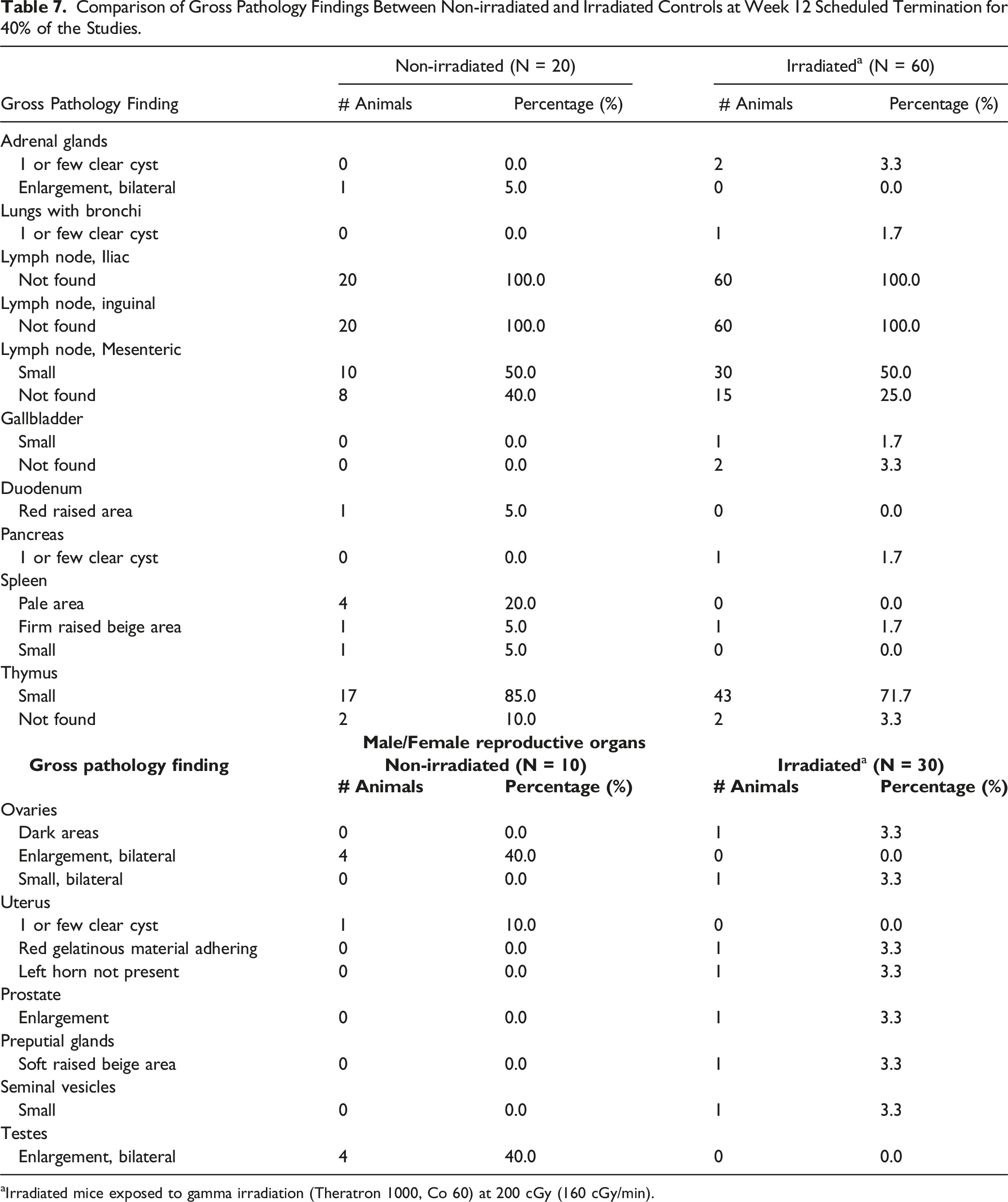
aIrradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
Comparison of Gross Pathology Findings Between Non-irradiated and Irradiated Control Animals Versus Busulfan-Treated Animals at Scheduled Termination.
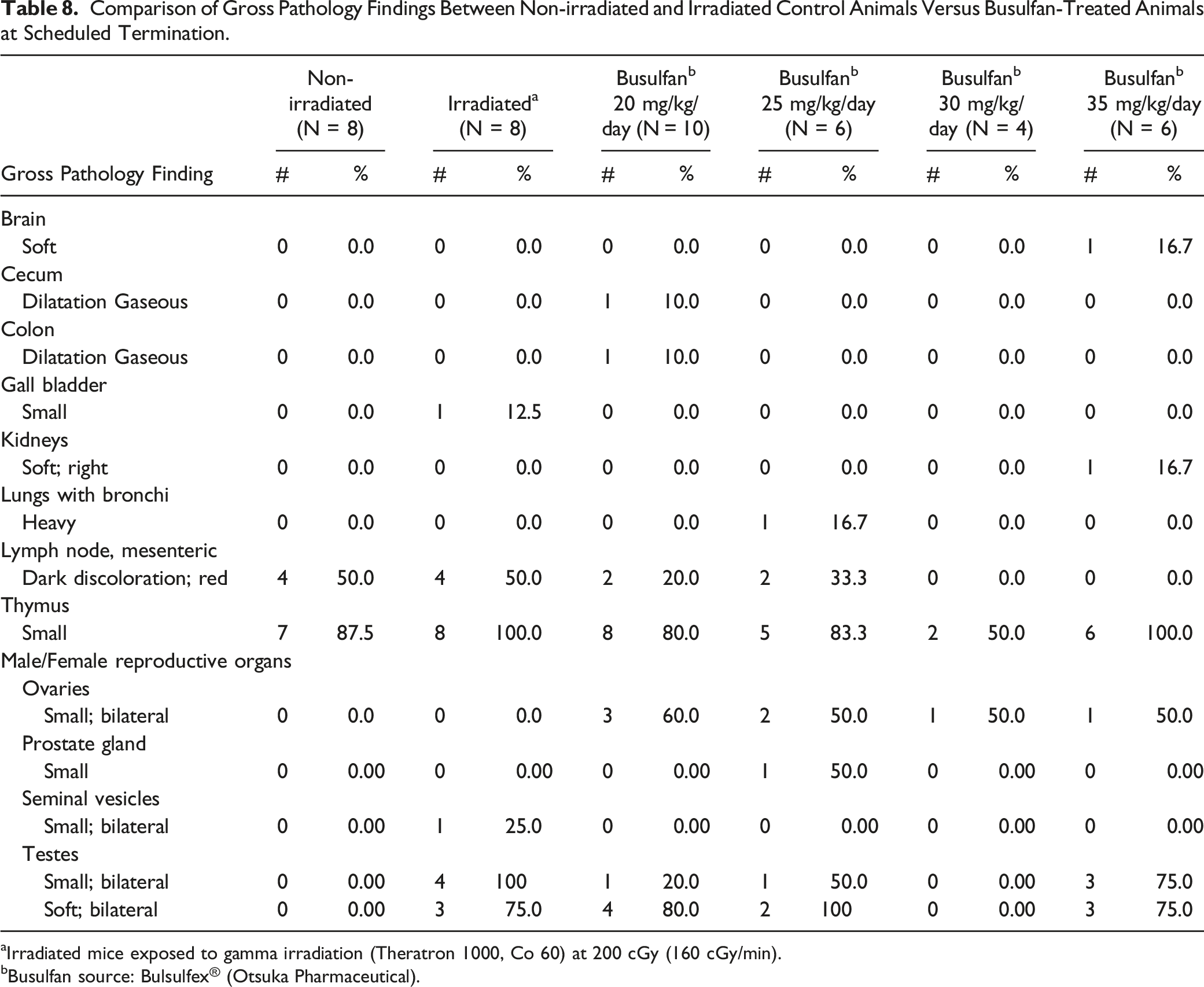
aIrradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
bBusulfan source: Bulsulfex® (Otsuka Pharmaceutical).
Discussion
Total body irradiation has been a valuable tool used for myeloablation (ie, significant reduction in lymphocyte populations in order to generate niche space within the bone marrow compartment of recipient mice for donor cell engraftment). 11 Ionizing radiation damages mitotically active cells (such as haematopoietic cells and those lining the intestinal crypts). The destruction of haematopoietic cells results in a severe suppression of the immune system and therefore a sufficient amount of haematopoietic cells must be provided by a non-irradiated donor in order to reconstitute the immune system. Chemotherapeutic agents such as busulfan have been considered as a potential alternative to total body irradiation (TBI). Busulfan is a myeloablative alkylating agent and functions similarly to TBI in depleting non-cycling primitive stem cells. 12 Unlike other alkylating agents, busulfan does not readily bind to plasma proteins in the blood, and therefore can cross the blood brain barrier, achieving concentrations in the cerebrospinal fluid approximately equal to those in plasma. A previous study in rats using radiolabeled busulfan showed that radioactivity was evenly distributed to the brain by a ratio (brain/plasma) of 0.95 ± 0.12. 13
Here we aimed to retrospectively compare data generated at 1 institution from gamma-irradiated and non-irradiated control NSG mice as well as busulfan-treated NSG mice in order to evaluate differences in overall tolerability of these conditioning treatments. Evaluations were performed by comparing relevant toxicology parameters such as clinical signs, body weights, hematology, and gross pathology findings at termination.
Mortality and Clinical Signs
One of the risks following exposure to ionizing radiation can be the development of Acute Radiation Syndrome (ARS) characterized by radiation-induced multiorgan dysfunction/failure that refers to progressive dysfunction of 2 or more organ systems. 14 This dysfunction and failure is due to radiation damage to cells and tissues over time and can classically be subdivided into 3 different sub-syndromes: hematopoietic (Heme), gastrointestinal (GI) or neurovascular (NV). TBI mouse models have been used to study pathophysiology and clinical signs associated with ARS. 14 Some clinical signs which have been considered to be signs of morbidity or moribundity in irradiated mice have included signs describing appearance (ie, hunched up, absence of grooming, piloerection, ocular discharge and cyanosis), respiratory rate (ie, increased and/or abdominal breathing), general behavior (ie, inability to stand, vocalization, no peer interaction) and provoked behavior (ie, unresponsive to prodding). The incidence of any of these clinical signs in data which was collected from irradiated and non-irradiated mice over the course of the studies evaluated here was very low and/or also present in control animals indicating that irradiation at a dose of 200 cGy (160 cGy/min) in NSG mice was well-tolerated when compared to non-irradiated. In addition, no mortalities occurred in irradiated negative control NSG mice. However, with busulfan-treated NSG mice, results clearly showed an increase and severity of signs associated with increasing dose levels ≥25 mg/kg/day and mortalities which occurred between Days 1 and 25 among animals given busulfan ≥25 mg/kg/day. However, mice administered 2 doses of busulfan at a dose level of 20 mg/kg/day demonstrated clinical signs comparable to irradiated mice exposed to gamma irradiation (Theratron 1000, Co 60) at 200 cGy (160 cGy/min).
Body Weights
Following total body irradiation and busulfan treatment, weight loss can occur due to inappetence and diarrhea, which can be a result of intestinal damage. Results showed a slight decrease in body weight change following irradiation; however, compiled results from all studies showed that body weight percentage when compared to baseline, increased over time for both irradiated males and females when compared to non-irradiated controls and were similar at 21 days post-irradiation to non-irradiated controls. In busulfan-treated mice, a decrease in group mean body weights and body weight loss from baseline was observed in all treated groups by Day 3 following dosing in both males and females. Similarly, in a previous published study, the weights of NSG mice which were administered busulfan at 20, 25 and 30 mg/kg decreased significantly 24 hours after the first injection regardless of dose used and 24 hours after a second injection, the weight loss further decreased in a dose-dependent manner [(9.4% for 20 mg/kg (8 mice), 11.2% for 25 mg/kg (6 mice) and 16% for 30 mg/kg (4 mice)]. Interestingly, males in this study were more sensitive than females and their weight loss was more pronounced even at a dose of 20 mg/kg in males compared to 25 mg/kg in females (up to 13.9% vs 10.4% for males and females, respectively). This sex-specific effect is similar to results obtained by other groups that evaluated such changes in NSG mice after busulfan administration. 15
Hematology
One of the risks which has previously been associated with radiation and the use of busulfan includes bone marrow failure and resulting in aplastic anemia and pancytopenia (associated with severe anemia, neutropenia, and thrombocytopenia). 16 Previously, the effects of dose and dose rates of whole body gamma irradiation on hematological variables and cytokines in young adult C57BL/6 mice (N = 75) were evaluated. 17 Ionizing radiation mainly disrupts the integrity of DNA. Cellular components that are sensitive to γ-radiation include lymphocytes, which have been shown to decline significantly after exposure to the different doses of γ-irradiation due to irradiation-induced DNA damage. 18 In 1 study, when radiation was delivered as a single dose to mice at doses ranging from 2 Gy to 6 Gy, white blood cell (WBC) counts were shown to decrease in a dose-dependent manner when compared to non-irradiated controls. 19 In the current retrospective comparison, hematology, including white blood cell (WBC) and neutrophil (NEUT) counts, from irradiated mice at Weeks 12 and 20, revealed comparable values to non-irradiated animals. This was also observed when irradiated vs non-irradiated controls were compared at Day 37 in the busulfan-treated animal study. In animals administered busulfan at ≥ 20 mg/kg/day, differences in hematology parameters were observed, although not in a dose–dependent manner. These changes included moderate to severe decreases in parameters included RBC counts, hemoglobin, hematocrit, mean corpuscular hemoglobin and volume and platelet counts. These results suggested that, in terms of hematology parameters, busulfan administered at dose levels ≥25 mg/kg/day was not well-tolerated. However, at a dose level of 20 mg/kg/day, results were comparable to irradiated controls.
Gross Pathology
Gross pathology findings attributed to busulfan treatment in scheduled euthanized mice mainly occurred in the testes and ovaries. Findings included males presenting with soft and small testes (including irradiated males) and bilateral small ovaries in busulfan-treated females only (no macroscopic observations in the ovaries of irradiated mice).
In a previous study, testicular weights in the busulfan-treated groups were also shown to be significantly less than the ones in the control group with no significant difference between animals treated with busulfan at 20, 30 and 40 mg/kg. 20
In the current retrospective analysis, 50% of irradiated males were observed to have small testes at Day 37 compared to non-irradiated, yet this macroscopic finding was not observed in 40% of the animals at Week 12 euthanasia.
Conclusion
A comparison of conditioning regimens in NSG mice indicates that overall tolerability was comparable between those administered busulfan via i. p. injection twice at 20 mg/kg/day and those irradiated with 200 cGy gamma irradiation, and that both regimens were well-tolerated. However, busulfan administered in 1 or 2 i. p. injections at levels of ≥25 mg/kg/day resulted in increased mortalities and clinical signs of toxicity in NSG mice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
