Abstract
The alkylating agent busulfan is used in conditioning treatment of chronic myelogenous or granulocytic leukemia prior to stem cell transplantations. Its cytotoxic activity results in primary damage or destruction of hematopoietic cells. While the toxicity of busulfan is well investigated, little is known about the toxic effects of its impurities. In this study, the effect of 4-day intravenous infusion (3 h/d) of 4.8 mg/kg/d busulfan and 0.49, 4.9, and 49 mg/kg/d busulfan impurity 5 (4-((methylsulfonyl)oxy)butyl acetate) was investigated in rats. Whereas busulfan elicited myelotoxic and hepatotoxic effects, no toxic effects were observed in animals receiving the impurity at dosages up to 10 times higher than busulfan. The highest impurity dose of 49 mg/kg/d is therefore considered the no-observed-adverse-effect level of busulfan impurity 5.
Introduction
The alkylating agent busulfan is the drug of preference in conditioning treatment of chronic myelogenous or granulocytic leukemia prior to stem cell transplantations. Its cytotoxic activity results in primary damage or destruction of hematopoietic cells. In clinical trials submitted for market approval, adverse events were mainly mild to moderate and included nausea, stomatitis, leukopenia, thrombocytopenia, vomiting, anemia, fever, anorexia, diarrhea, and insomnia. 1 The serious toxicities observed in these studies mainly concerned the liver, lungs, and nervous system. Liver function tests revealed increased levels of lactate dehydrogenase, serum bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT). 2
Such adverse events may not only result from the drug itself but also be related to its vehicle or impurities. The vehicle of intravenous busulfan solution contains 33% dimethylacetamide (DMA). The target organ of DMA is the liver. 3 It has been shown that single as well as repeated exposure to DMA leads to increases in serum enzymes like AST and ALT followed by histopathological changes including single cell necrosis, hepatic focal cystic degeneration, and biliary hyperplasia. 4 The presence of DMA in the vehicle of busulfan solutions for intravenous administration is therefore considered partially responsible for the hepatotoxicity of busulfan.
However, little is known about the toxic effects of busulfan impurities. 2 In one study, single doses of 1, 5, 10, 20, or 40 mg/kg/bw of 2 lots of a busulfan formulation were administered intravenously to rats, one lot representing a standard batch and one lot that underwent forced degradation. The degraded busulfan formulation was less toxic than the standard batch. 2 The absolute amount of each impurity in this forced degradation batch is, however, not publicly available. We hypothesized that the busulfan impurities are less toxic than busulfan at similar or higher levels. To specifically address the toxicity of 4-((methylsulfonyl)oxy)butyl acetate (busulfan impurity 5), an intravenous repeated dose toxicity study in rats comparing the toxicity of busulfan impurity 5 to busulfan was therefore performed.
Materials and Methods
Animals
One hundred Wistar (aged 9 weeks): Crl:WI (Han) rats were obtained from Charles River Laboratories (Domain des Oncins, France). The rats had an acclimatization period of at least 14 days before start of the experiments.
During the acclimatization period, a polyurethane catheter was implanted into the posterior vena cava via the left femoral vein. Following implantation, the animals were maintained on continuous infusion with physiological saline prior to the start of treatment. All animals were housed singly in a temperature-controlled room with a 12-hour light/dark cycle. The room temperature and relative humidity were maintained in the ranges of 22°C ± 3°C and >35% humidity. All animals had access to food (diet reference A04C-10, obtained from Safe diets, Augy, France) and water ad libitum. The body weights of the animals at the start of infusion ranged from 164 to 294 g. The study was approved by the ethical committee of Charles River and authorized by the French authorities (Ministère de l'Enseignement supérieur, de la Recherche et de l'Innovation).
Chemicals
The structures of busulfan and busulfan impurity 5 are given in Figure 1. Busulfan (batch BUS/FDP0057/043/I0006_Initial) was supplied by Fresenius Oncology Ltd. (Gurgaon, India). Busulfan impurity 5 (batch BUS/CRD0515/093/busulfan monoacetate) was synthetized by Fresenius Oncology Ltd. The vehicle for preparation of impurity infusion solution composed of DMA/polyethylene glycol 400; PEG400 (batch BUS/FDP0057/041/I0000-P) was supplied by Fresenius Oncology Ltd.
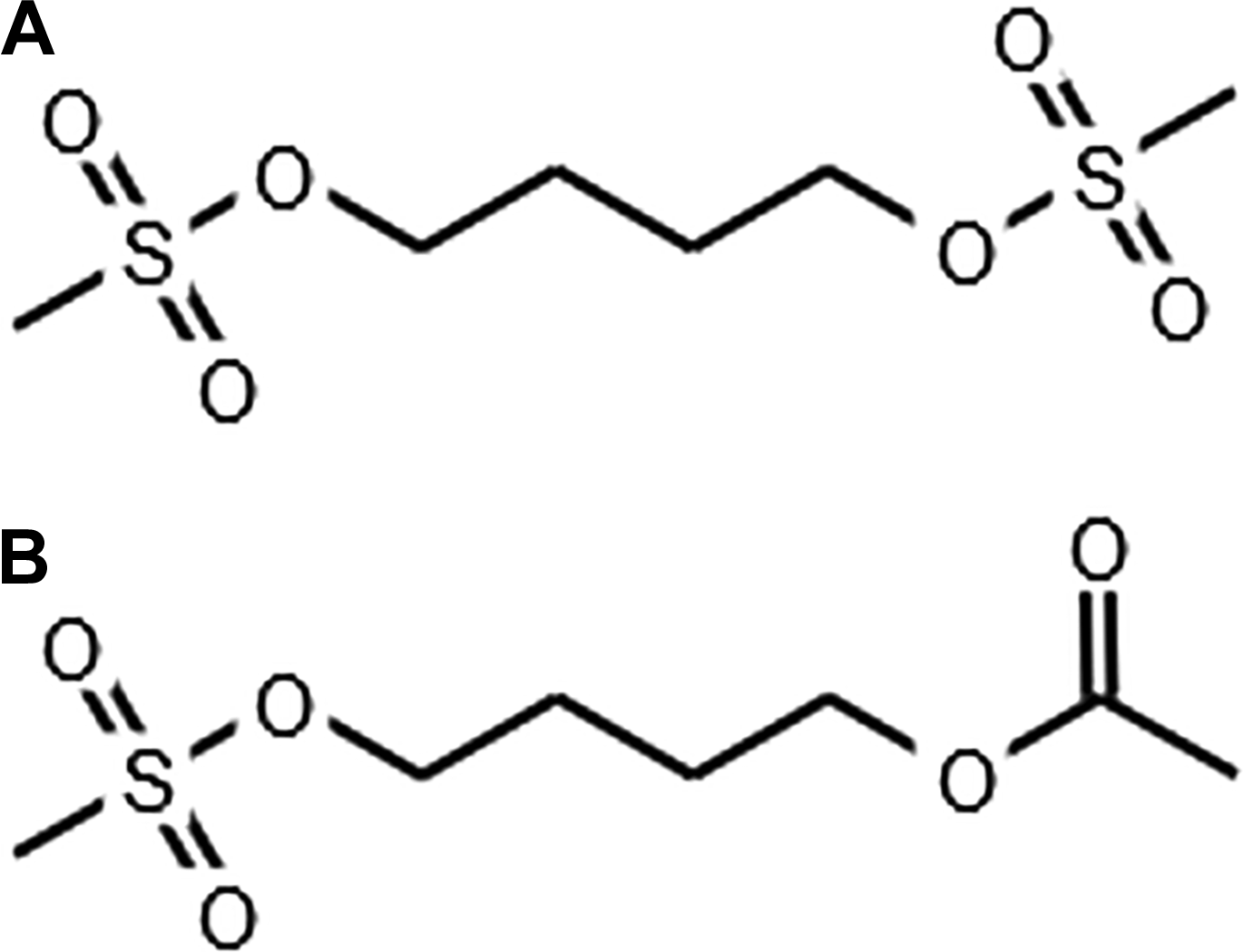
Chemical structure of (A) busulfan and (B) busulfan impurity 5. Structures were created using http://biomodel.uah.es/en/DIY/JSME/draw.en.htm.
Experimental Design
Fifty animals per sex were randomized to 5 experimental groups. Group 1 was treated with the vehicle (33% DMA/67% PEG400). Group 2 received busulfan, while groups 3 to 5 received the busulfan impurity 5 (“impurity”) in different dosages. Every group consisted of 10 animals per sex. A saline control group consisted of 10 animals per sex receiving the same volume of saline to account for any vehicle (33% DMA/67% PEG400) toxicity. All animals were treated for 4 days as described in the section “Dose Administration” and then 5 animals per sex and group were sacrificed and analyzed at day 8 and day 28 after start of treatment.
Dosing
The dosages administered were as follows: busulfan = 4.8 mg/kg/d, busulfan impurity 5 = 0.49, 4.9, and 49 mg/kg/d. Busulfan was prepared according to the package leaflet as a 1/12 solution in 0.9% NaCl at a final concentration of 0.5 mg/mL. Busulfan impurity 5 was prepared as a solution in vehicle as a 10× stock concentration of the final concentration followed by a 1/10 dilution in 0.9% NaCl. Vehicle control group animals received the vehicle diluted 1/10 in 0.9% NaCl. The test items were administered via a 3-hour intravenous infusion once daily for 4 days. The infusion rate was 5 mL/kg/h for the impurity dose groups and 3.2 mL/kg/h for the busulfan group.
Rationale for the treatment schedule selection
There are 2 therapeutic treatment schedules of busulfan. On the one hand, it can be administered as a 2-hour infusion every 6 hours over 4 consecutive days for a total of 16 doses prior to cyclophosphamide or melphalan and hematopoietic progenitor cell transplantation. On the other hand, it can be administered as a single daily 3-hour infusion immediately after fludarabine for 2 or 3 consecutive days. As busulfan is a chemotherapeutic drug, the authors decided to choose a treatment schedule according to the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) S9 guideline. According to the ICH S9 guideline, treatment of up to 5 days every 3 weeks requires a dosing schedule in a nonclinical study of 5 days. As busulfan is administered once for 4 days, the authors considered a single treatment cycle of 4 days as appropriate. Therefore, in this study, the maximum treatment period (4 days) was combined with single daily infusion of 3 hours due to less potential for error than 2 hours of infusion every 6 hours.
Rationale for the dose selection
Busulfan is administered to both adults and pediatrics. The dose administered per kg body weight is higher in pediatrics and was therefore used as a worst-case scenario in this study. In pediatrics (>9 and <16 years), the maximum daily dose of busulfan is 4.8 mg/kg/d in 9.6 mL/kg/d. Busulfan impurity 5 was tested in this study at the same dose level and at dose levels of 0.1 and 10 times the dose of busulfan.
Observations
Animals were observed twice daily. During the treatment period, the animals were observed before and at least 3 times after the start of dosing to detect any clinical signs or reactions to treatment. During the observation period, they were examined twice daily. Ophthalmological analysis was performed in the vehicle-treated animals and in the highest impurity dose group at day 7.
Plasma and Tissue Preparation
Blood samples were taken from the sublingual vein. The following hematology parameters were determined using the ADVIA (120 or 2120, Siemens) system: red blood cell count, hemoglobin, packed cell volume, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, reticulocyte count, platelet count, total white blood cell count, and differential white blood cell count. In addition, a blood smear was prepared for each animal.
The following coagulation parameters were determined using the STA R Max (Stago, Asnières sur Seine, France) system: prothrombin time, activated partial thromboplastin time, and fibrinogen. The following clinical chemistry parameters were determined using the AU680 (Beckman Coulter, Brea, California) system: Na+, K+, Cl−, Ca2+, glucose, urea, creatinine, cholesterol, triglycerides, total protein, albumin, globulin, total bilirubin, AST, alkaline phosphatase (ALP), ALT, and gamma-glutamyl-transferase (GGT).
Urine was collected after placing the animals in metabolic cages for 16 hours. Urine parameters determined were volume, specific gravity, appearance, as well as (using the Clinitek 500 system; Siemens, Saint-Denis Cedex, France) pH, bilirubin, urobilinogen, ketones, blood, glucose, and protein. At the end of the observation periods (day 8 or day 28), all designated animals were necropsied. Histopathological analysis was performed for all animals in the vehicle group as well as in the busulfan and the high-dose impurity group.
Statistical Analyses
Statistical analyses were performed by the Provantis data acquisition system as follows: The best transformation for the data (none, log, or rank) was determined depending upon the kurtosis of the data, the probability of the Bartlett test for homogeneity of the variances, and the similarity of the group sizes. Non- or log-transformed data were analyzed by parametric methods. Rank transformed data were analyzed using nonparametric methods. The homogeneity of means was assessed by analysis of variance.
Data were then analyzed to test for a dose-related trend to detect the lowest dose at which there was a significant effect, based on the Williams test for parametric data or Shirley test for nonparametric data. If no trend was found but the means were not homogeneous, the data were analyzed by a stepwise parametric or nonparametric Dunnett test to look for significant differences from the control group. Data are presented as mean ± standard deviation in the figures.
Results
Clinical Signs and Mortality
On day 15, one of the female rats receiving busulfan 4.8 mg/kg/d was found dead. No particular clinical sign was noted the days before. On the same day, another female rat of this group was sacrificed moribund, followed by 2 animals (1 male and 1 female) sacrificed moribund on day 17. The clinical signs observed were as follows: decreased activity, pale to very pale whole body, partly closed eyes, piloerection, subdued behavior, sometimes associated with marked body weight loss, decreased mean food consumption, chromodacryorrhea, coldness to touch, persistent skin fold, red-stained fur, hunched gait, irregular breathing, and purple-colored skin areas (in males only scrotum or ear). The same clinical signs were observed on remaining animals of this group; thus on day 18, they were sacrificed moribund. No mortality or clinical signs were observed in the animals treated with busulfan impurity 5 at any dose group.
Body Weight
Body weight increased similarly in males and females of the vehicle and impurity-treated groups. During the treatment and observation period (day 1 to day 28), the body weight increased in the vehicle control group from 262.08 ± 14.64 g to 347.98 ± 17.07 g. A similar increase was observed in the animals treated with different dosages of busulfan impurity (low dose: 262.43 ± 11.13 to 347.80 ± 20.73, mid dose: 262.43 ± 11.13 to 347.80 ± 20.73, high dose: 263.97 ± 15.65 to 336.18 ± 20.31 g). Animals treated with busulfan missed this increase in body weight and showed a mean body weight of 267.49 ± 15.47 on day 1 and a mean body weight of 274.55 ± 14.77 at the time of sacrifice. On day 18, the animals in this group were sacrificed moribund. No differences were observed between the vehicle and the saline control group.
Serum Hematology
On day 18 after start of treatment, a marked decrease in red blood cell count, hemoglobin concentration, packed cell volume, reticulocyte count, platelet count, and white blood cell count was observed for both sexes of the busulfan group when compared with the vehicle control group (Figure 2). No changes occurred in animals treated with busulfan impurity 5 regardless of the dose. All other serum hematology parameters tested were within historical control data ranges for all animals. No differences were observed between the vehicle and the saline control group (data of the saline control group not shown).
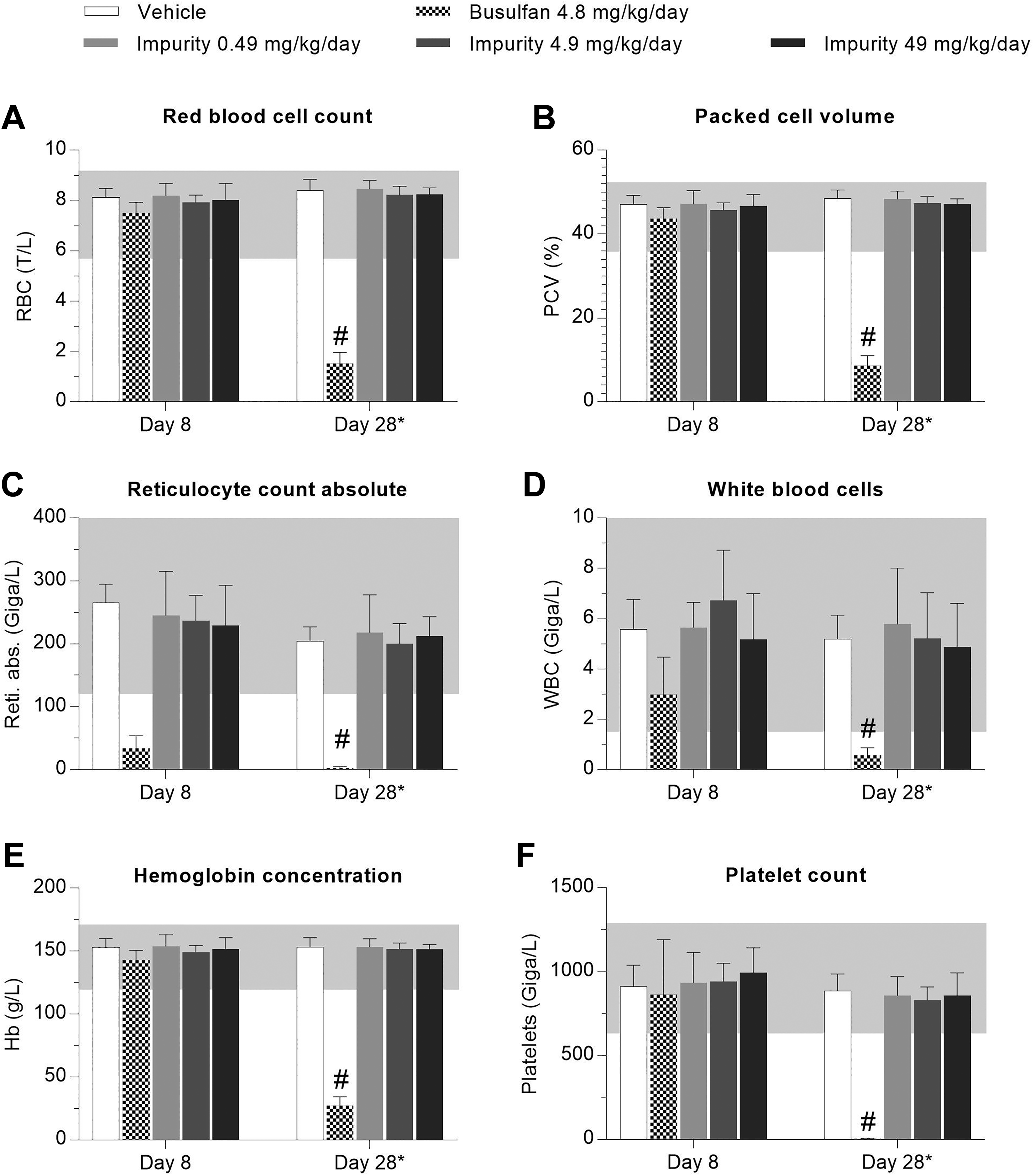
Red blood cell count (A), packed cell volume (B), absolute reticulocyte count (C), white blood cells (D), hemoglobin concentration (E), and platelet count (F) are decreased in animals receiving 4.8 mg/kg/d busulfan on day 28 (18) after start of treatment, but not in animals receiving up to 49 mg/kg/d of busulfan impurity 5. Gray-horizontal bar = range of historical control data, * = day 18 in animals receiving busulfan, #P < 0.05.
Serum Clinical Chemistry
In animals treated with busulfan, total bilirubin and ALT exceeded the upper level of the historical control data range. The levels of animals treated with busulfan impurity 5 were not increased regardless of the dose administered in both sexes. The levels of serum clinical chemistry (Na+, K+, Cl−, Ca2+), glucose, urea, creatinine, cholesterol, triglycerides, total protein, albumin, globulin, alkaline phosphatase, AST, and GGT were within the historical control data ranges in all animals. No differences were observed between the vehicle and saline control group.
Urine Analyses
All parameters of the urine analysis were within the historical control data range in all animals. No sex differences were observed.
Ophthalmology
There were no findings in the ophthalmologic examination in any of the animals. No sex differences were observed.
Bone Marrow Evaluation
Treatment with busulfan impurity 5 at 49 mg/kg/d elicited mild changes in M:E ratios as follows: The M:E ratios of animals administered busulfan impurity at 49 mg/kg/d were compared with the control group values (0.85-1.53). At day 8, minimal decrease in the M:E ratio was observed in 2 males (individual value: 0.79 and 0.78) and minimal increase in the M:E ratio was noted in 1 male (1.75). Mild increases in the M:E ratio were noted in 2 females (individual values: 1.61 and 1.98, respectively). At day 28, minimal decrease in the M:E ratio was noted in 1 male (0.74). Mild to moderate decreases in the M:E ratio were observed in 2 males (individual values: 0.62 and 0.39, respectively). As cells from erythroid, myeloid, and megakaryocytic cell lines presented normal maturation sequences and exhibited normal morphology in all treated animals, these signs were considered nonadverse. The myelotoxic effect of busulfan has already been described in the study by McManus and Weiss 5 and was not analyzed in this study.
Histopathology
Under the study conditions, there was no evidence of macroscopic and microscopic treatment-related findings associated with 4 consecutive daily intravenous administrations of busulfan impurity 5 at any dose level after a 4-day and 24-day observation periods. In animals given busulfan at 4.8 mg/kg/d, adverse treatment-related findings were mainly observed in the bone marrow and lymphoid organs on day 8. These changes were also considered cause of death or poor clinical condition on days 15 to 18. Treatment-related microscopic findings were noted in a wide range of tissues at interim sacrifice (day 8) in animals from both sexes. They mostly consisted of marked to severe decreased cellularity (all cell lines) in the sternal bone marrow, moderate to marked diffuse thymic atrophy, minimal to moderate decreased lymphoid development of lymph nodes, and slight to marked splenic depletion. No differences were observed between the vehicle and saline control group.
Discussion
The main finding of this study is the lack of bone marrow and liver toxicity of busulfan impurity 5 in comparison to busulfan. While busulfan administration to rats led to statistically significantly decreased packed cell volume, hemoglobin concentration, red blood cell, reticulocyte, white blood cell, and platelet count as well as increased levels of total bilirubin and ALT, none of these changes were observed in any of the animals receiving busulfan impurity 5. Furthermore, none of the impurity-treated animals showed the characteristic signs of busulfan-induced bone marrow changes. 5
The toxic effects observed with busulfan in our study are in line with the literature with regard to intravenously administered busulfan. 1,2 Toxicological studies in rats and dogs submitted for market approval report bone marrow depletion and decreased mean white blood cell counts and platelet levels, as well as liver injury characterized by hepatocellular necrosis, hyperbilirubinemia, and increases in ALP, ALT, and GGT. 2 In our study, the increase in ALP and AST was marginal, and the animals did not show hepatocellular necrosis. This is most probably due to the short treatment duration; histopathological changes may have developed later.
A current focus of research is the effect of busulfan on testes and semen quality. 6-16 In our study, however, no effect on the male and female reproductive organs was observed in the busulfan-treated animals. This may be due to the fact that studies investigating the effect of busulfan on reproductive organs either administer high dosages like 10 to 20 mg/kg/bw busulfan intraperitoneally or rely on prolonged administration of oral dosages of busulfan for 2 weeks or more. 5 -19 Even though busulfan is known to cause sterility, 5 these effects may therefore not be present in rat studies not administering high dosages of busulfan. No effect on reproductive organs was observed in animals receiving busulfan impurity 5 at dosages 10 times higher than the busulfan dose administered in this study. The highest nontoxic dose of busulfan impurity 5 was even 5 times higher than dosages of busulfan commonly inducing testicular changes in other studies.
The hepatotoxic effect of busulfan formulations for intravenous administration is considered to be based among others on the presence of DMA. Repeated doses of DMA produce signs of liver toxicity, the first being increases in serum clinical enzymes followed by histopathological changes in the hepatocytes. 2 In our study, all animals received the same nontoxic dose of DMA. Liver injury is still present in the animals in the busulfan-treated group, indicating a DMA-independent hepatotoxic effect of busulfan. This effect, however, was not observed in animals receiving the impurity, even at high dosages. “In busulfan impurity 5, one of the 2 active methane sulfonate groups is replaced by an acetate group. This change in structure may have led to at least partial inactivation of the molecule.”
A limitation of our study is the short treatment duration of 4 days. This is, however, in line with the ICH S9 guideline “Nonclinical Evaluation for Anticancer Pharmaceuticals” and therefore considered appropriate for qualification of the impurity.
In summary, the busulfan impurity 5 neither displayed the busulfan-related toxicity nor any additional signs of toxicity, even at 10 times higher dosages. The dose of 49 mg/kg/d is therefore considered the no-observed-adverse-effect level of busulfan impurity 5. According to the ICH Q3B guideline “Impurities in new drug products” toxicological qualification is required for impurity levels in the drug product exceeding 0.2% of the active pharmaceutical ingredient. In the case of the busulfan impurity, 0.2% of the highest clinical dose of busulfan (4.8 mg/kg/d) would be 9.6 µg/kg/d. Based on the results of our study, busulfan impurity 5 is not associated with any observed toxicity at doses up to 49 mg/kg/d.
Footnotes
Acknowledgments
The authors thank the following employees of Charles River SAS, Lyon, France, for their participation in the study: Hélène Voute for ophthalmology analysis, David Ravelo for histopathological examination, Myriam Defontis for bone marrow analysis, and Fabien Marguerite for formulation analysis of the test item.
Author Contributions
Melanie Kathrin Bothe and Dennis Franckenstein contributed equally to this publication. Melanie Kathrin Bothe contributed to conception, design, analysis, and interpretation and drafted the manuscript. Dennis Franckenstein contributed to conception, design, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Didier Cade contributed to acquisition and analysis and critically revised the manuscript. Emilie Quatresous contributed to acquisition and analysis and critically revised the manuscript. Martin Westphal contributed to conception, design, and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy. Didier Cade and Emilie Quatresous conducted the animal experiments. Didier Cade, Emilie Quatresous, and Martin Westphal finally approved the version to be submitted.
Declaration of Conflicting Interests
The author(s) declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Melanie Kathrin Bothe, Dennis Franckenstein, and Martin Westphal are employees of Fresenius Kabi Deutschland GmbH, the sponsor of this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
