Abstract
Metabolic activation of indirect-acting carcinogens in target organs is a recognized mechanism of carcinogenesis. This study aimed to determine the role of benzo[a]pyrene (BaP) metabolism enzymes lipoxygenase (LOX), cytochrome P4501A1 (CYP1A1), and prostaglandin synthetase (PGS) in the cytotoxicity and DNA damage induced by BaP in the human tracheobronchial epithelial cells (HBECs) using RNA interference strategy and metabolic enzyme inhibitors. Our results showed that in three epithelial cell lines (HBE, HTR-8/SVneo, and HaCat), BaP significantly upregulated 5-LOX protein expression. 15-LOX-2 expression also increased with increasing BaP concentration, but the change was less pronounced than that of 5-LOX. BaP caused significant cytotoxicity, DNA strand breaks, and 8-hydroxy-2'-deoxyguanosine formation in HBE, which was inhibited by 5-LOXshRNA, a specific inhibitor of 5-LOX (AA861), the CYP1A1 inhibitor α-naphthoflavone, and the PGS inhibitor naproxen. The protective effects of 5-LOXshRNA were stronger than AA861, naproxen and α-naphthoflavone. We conclude that BaP may be activated more by 5-LOX than by CYP1A1 and PGS to produce cytotoxicity and DNA damage in HBE.
Introduction
Benzo[a]pyrene (BaP), a known Group 1 human carcinogen classified by the International Agency for Research on Cancer,
1
is the most widespread environmental pollutant among the polycyclic aromatic hydrocarbons formed mainly by incomplete combustion of organic material. Epidemiological investigations reveal that BaP increases the risk of lung cancer,
2
hepatocellular carcinoma
3
and various tumors. As a pro-carcinogen, BaP requires metabolic activation to reactive metabolites (eg, benzo[a]pyrene-7,8-diol-9,10-epoxide (BPDE)) to induce its mutagenic and carcinogenic effects. DNA damage induced by covalent binding of BPDE to nucleophilic centers in DNA and oxidative damage induced by reactive oxygen species (ROS) and other free radicals produced during metabolic activation of BaP4,5 are thought to be a critical event in the initiation of B[a]P-induced tumorigenesis.
6
Cytochrome P450s (CYPs), especially cytochrome P4501A1 (CYP1A1) and CYP1B1, are considered the key enzymes in the metabolic activation.
7
However, research with knockout animals, including aryl hydrocarbon receptor (AhR) knockout mice,
Lipoxygenases (LOXs) are a family of non-heme-iron-containing enzymes that universally exist in mammals and plants. In addition to catalyzing the dioxygenation of polyunsaturated fatty acids (PUFA) in lipids into cell signaling agents, LOXs can co-oxidize xenobiotics to form free radical intermediates and activation products in the presence of PUFA or some other chemicals. 11 In some extrahepatic tissues with little or no CYPs (eg, lung, brain, heart, skin, uterus, placenta, fetal tissues, and blood cells), LOXs generally show relatively high activity and are regarded as a likely alternate oxidative metabolic pathway for endogenous and exogenous compounds.11,12 LOX-mediated oxidation of xenobiotics to active metabolite may be related to these compounds’ carcinogenicity and other toxicity.
5-LOX is a vital human LOX-isoenzyme. The tumor promoting activity of 5-LOX has been confirmed by the overexpression of 5-LOX in a number of cancer tissues compared to normal tissues, and the cancer chemopreventive activity of a 5-LOX inhibitor against lung, 13 prostate, 14 bladder, 15 oral, 16 colorectal, 17 and other tumours has been demonstrated. 18 The chemopreventive effects of 5-LOX inhibitors have also been demonstrated in 7,12-dimethylbenz(a)anthracene (DMBA)-induced breast cancer, 19 vinyl carbamate-induced lung tumor, 20 and N-nitrosobis-2-oxopropylamine (BOP)-induced pancreatic cancer. 21 These results indicate that 5-LOX plays an important role in chemically induced carcinogenesis. However, its mechanism is not fully understood.
In
Oxidative stress is one of the mechanisms of BaP-induced toxicity and carcinogenesis. ROS formed during the metabolic activation process of BaP ultimately interact with biomacromolecules and cause oxidative DNA damage linked to carcinogenesis.4,26 5-LOX is involved in the generation of ROS. 27 LOXs oxidize PUFA and bioactivate xenobiotics to free radical intermediates that initiate ROS formation, resulting in oxidation of proteins, lipids and DNA.11,28 These results suggest LOX may be involved in BaP-induced oxidative DNA damage via catalyzing the metabolic activation of BaP.
In this study, BaP-induced cell proliferation inhibition, DNA strand breaks and 8-hydroxy-2′-deoxyguanosine (8-OHdG) formation were measured. The lentivirus-mediated small interfering RNA targeting 5-LOX was used and the protective effects of 5-LOX, CYP4501A1 and prostaglandin synthase inhibitors against BaP-induced toxic effects were evaluated and compared to further determine the effects of 5-LOX on BaP activation, cytotoxicity and DNA damage.
Materials and Methods
Chemicals and Reagents
Roswell Park Memorial Institute (RPMI) 1640 medium and fetal bovine serum (FBS) were supplied by Hyclone (Logan, Utah). Dulbecco’s modified Eagle’s medium (DMEM) was supplied by Gibco (Grand Island, NY, USA). Penicillin and streptomycin were obtained from Dingguo Changsheng Biotechnology Co. Ltd (Beijing, China). T4 DNA ligase and SYBR Premix Ex Taq were obtained from Takara (Ohtsu, Japan). Small hairpin RNA (shRNA) oligonucleotides, primers of polymerase chain reaction (PCR), and Trizol reagent were supplied by Invitrogen Biotechnology Co. Ltd (Shanghai, China). QIAGEN Plasmid Maxi Kit was purchased from QIAGEN Co. (Hilden, Germany). The psPAX2 packaging plasmid and pMD2.G membrane-protein shell plasmid were purchased from Addgene (Cambridge, Massachusetts). RIPA lysis buffer, horseradish peroxidase-conjugated goat anti-rabbit and goat anti-mouse IgG secondary antibodies were obtained from Beyotime Institute of Biotechnology (Haimen, China). Phenylmethylsulfonyl fluoride, mouse anti-β-actin monoclonal antibody, BaP, dimethyl sulfoxide (DMSO), AA861, α-naphthoflavone, naproxen, 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and propidium iodide (PI) were purchased from Sigma-Aldrich Co. (St Louis, Missouri, USA). DC protein assay kit was purchased from Bio-Rad Laboratories (Hercules, California, USA). Polyvinylidene difluoride (PVDF) membranes were obtained from Millipore (Billerica, Massachusetts, USA). Bovine serum albumin (BSA) and human 8-OHdG enzyme-linked immunosorbent assay (ELISA) kits were obtained from Cusabio (Wuhan, China). Rabbit anti-human 5-LOX and 12-LOX monoclonal antibodies were purchased from Epitomics Inc. (Burlingame, California), and rabbit anti-human 15-LOX-2 monoclonal antibody was obtained from Santa Cruz Biotechnology (Santa Cruz, California). Amersham ECL Plus Western Blotting Detection Reagent was obtained from GE Healthcare (Pittsburgh, PA, USA). M-MLV reverse transcriptase and low melting point agarose were purchased from Promega (Beijing) Biotech Co., Ltd (Beijing, China). Normal melting point agarose was obtained from Biowest (Nuaill′e, France).
Cell Culture and Treatment
HBE cells (Shanghai Fuxiang Biotech, Shanghai, China) and the trophoblast cells HTR-8/SVneo (kindly provided by Dr. Charles H. Graham, Queen’s University, Kingston, ON, Canada) were grown in RPMI 1640 medium. The human keratinocyte cells HaCat (Cell Biology Research Laboratory, the Modern Analysis and Testing Center, Central South University, Changsha, China) and the human embryonic kidney 293T cells (American Type Culture Collection, Manassas, Virginia) were cultured in DMEM. All cells were supplemented with 10% FBS and 1% double antibiotic (100 IU/mL penicillin and 100 μg/mL streptomycin) in Shellab CO2 incubator (Sheldon Manufacturing, Inc., Cornelius, OR, USA) containing 5% CO2 at 37°C. DH5α competent cells (Takara, Ohtsu, Japan) were grown in SOC Medium and grown on LB-agar plate containing ampicillin at 37°C after transformation.
Cells were treated with different concentrations of BaP (0-128 μmol/L) for 24 hours. The control group was treated with the same volume of DMSO. The inhibitor groups were preincubated with 10 μmol/L of 5-lipoxygenase inhibitor AA861, CYP1A1 inhibitor α-naphthoflavone, or prostaglandin synthetase (PGS) inhibitor naproxen for 4 hours before exposure to BaP.
Establishing Lentivirus-Mediated 5-LOX Knockdown HBE Cells
Sequences of Designed 5-LOX-shRNA.

HBE cells were transfected with recombinant lentiviral particles including lentivirus-NC-shRNA and lentivirus-5-LOX-shRNA at the optimum multiplicity of infection (MOI) of 20 to form HBE-NC cells and HBE-5-LOX-shRNA cells respectively. Transfection efficiency was detected by testing the expression ratio of green fluorescence under a BA400EF-UPR fluorescence microscope (Motic, Wuhan, China). Interference efficiency was determined using real-time fluorescence quantitative PCRs (QPCR) and western blot.
Western Blot Analysis
The cells were lysed in RIPA lysis buffer containing phenylmethylsulfonyl fluoride. Total proteins were quantified with a DC protein assay kit. The proteins were separated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred to PVDF membranes at 100V for about 2 hours. After blocking with 5% BSA in Tris-buffered saline and Tween 20 (TBST) for 2 hours, membranes were incubated with rabbit anti-human 5-LOX, 12-LOX monoclonal antibodies (1:1000 dilution), rabbit anti-human 15-LOX-2 monoclonal antibody (1:100 dilution), or mouse anti-β-actin monoclonal antibody (1:2000 dilution) at room temperature for 2 hours, followed by horseradish peroxidase-conjugated goat anti-rabbit or goat anti-mouse IgG secondary antibodies (1:5000 dilution) at room temperature for 1 hour. The protein bands were visualized using Amersham ECL Plus Western Blotting Detection Reagent. The film images were scanned and the gray scale of images was analyzed using Image-Pro Plus 6.0 software (Media Cybernetics, Inc., Silver Spring, Maryland).
Quantitative Polymerase Chain Reaction Assay
Primers of 5-LOX and β-actin.

3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Cell Viability Assay
The HBE, HBE-NC, and HBE-5-LOX-shRNA cells were seeded into 96-well plates at a density of 1 × 105 cells/mL (100 μL/well). Then, cells were treated with enzyme inhibitors and BaP as described in
Comet Assay for Detecting DNA Damage
The comet assay was carried out under minimal illumination, according to the method of Singh et al 31 The cells were resuspended at a density of 2 to 5 × 105 cells/mL in phosphate-buffered saline (PBS). Then, 10 μL cell suspensions were lightly mixed with 65 μL of 0.5% low melting point agarose. The mixture was superimposed on a slide precoated with 100 μL 1% normal melting point agarose and immediately covered with a coverslip. After solidification of the gel in a moist chamber at 4°C for 10 minutes, coverslips were removed and the slides were immersed in lysis solution (2.5 mol/L NaCl, 100 mmol/L Na2EDTA, 10 mmol/L Tris, 1% Sodium Sarcosinate, pH 10; 1% Triton X-100 and 10% DMSO added just before use) at 4°C for 1.5-2 hours. After washing with 0.4 mol/L Tris-HCl (pH 7.5) for 15 minutes, the slides were submerged in a fresh electrophoresis solution (1 mmol/L Na2EDTA and 300 mmol/L NaOH) for 20 minutes, and then electrophoresis was conducted at 300 mA, 25 V, for 30 minutes. The slides were neutralized in 0.4 mol/L Tris-HCl (pH 7.5), kept at room temperature to dry and stained with 50 μL of 5 μg/mL PI. Then, the photographs were taken under a BA400EF-UPR fluorescence microscope (Motic, Wuhan, China). From each slide, 50 cells were randomly selected and measured using CASP 1.2.2 software (CaspLab, Germany). Percentage of DNA in the tail (Tail DNA%) and Olive Tail Moment (OTM) were used to quantify the DNA damage.
8-Hydroxy-2′-Deoxyguanosine (8-OHdG) Analysis
The cells were seeded into 6-well plates at a density of 1 × 106 cells/well. After treatment with enzyme inhibitors and BaP as described in
Statistical Analysis
Statistical analyses were performed using SPSS 18.0 software (SPSS Inc., Chicago, IL, USA). The results were expressed as mean ± standard deviation (SD). Multisample means comparison was analyzed with a one-way analysis of variance (ANOVA), and the multiple comparisons were determined using the SNK-q and Dunnett-t test. The chi-squared (
Results
BaP Modulates LOX Isozyme Protein Expression in HBE, HTR-8/SVneo and HaCat Cells
In all three epithelial cells tested (HBE, HTR-8/SVneo, and HaCat), BaP upregulated 5-LOX protein (ca. 78 kDa) expression. When the BaP concentration reached 32 and 64 μmol/L, the difference was statistically significant. 15-LOX-2 (ca. 75 kDa) expression also increased with increasing BaP concentration, but the change was less pronounced than that of 5-LOX. No 12-LOX (ca. 72 kDa) protein expression was detected in any of the three epithelial cells (Figure 1). Effects of BaP on 5-LOX, 12-LOX, and 15-LOX-2 protein expression in HBE, HaCat, and HTR-8/SVneo cells. Representative Western blot and relative grey intensity of 5-LOX, 12-LOX, and 15-LOX-2 protein in HBE (A), HaCat (B), and HTR-8/SVneo cells (C) treated by 16, 32, 64 μmol/L BaP for 24 hours. β-actin (43 kDa) was used as an internal control and the protein samples extracted from A431 human skin cancer (epidermoid carcinoma) cells were used as positive control of 12-LOX protein expression. Data are presented as mean ± SD of three experiments. *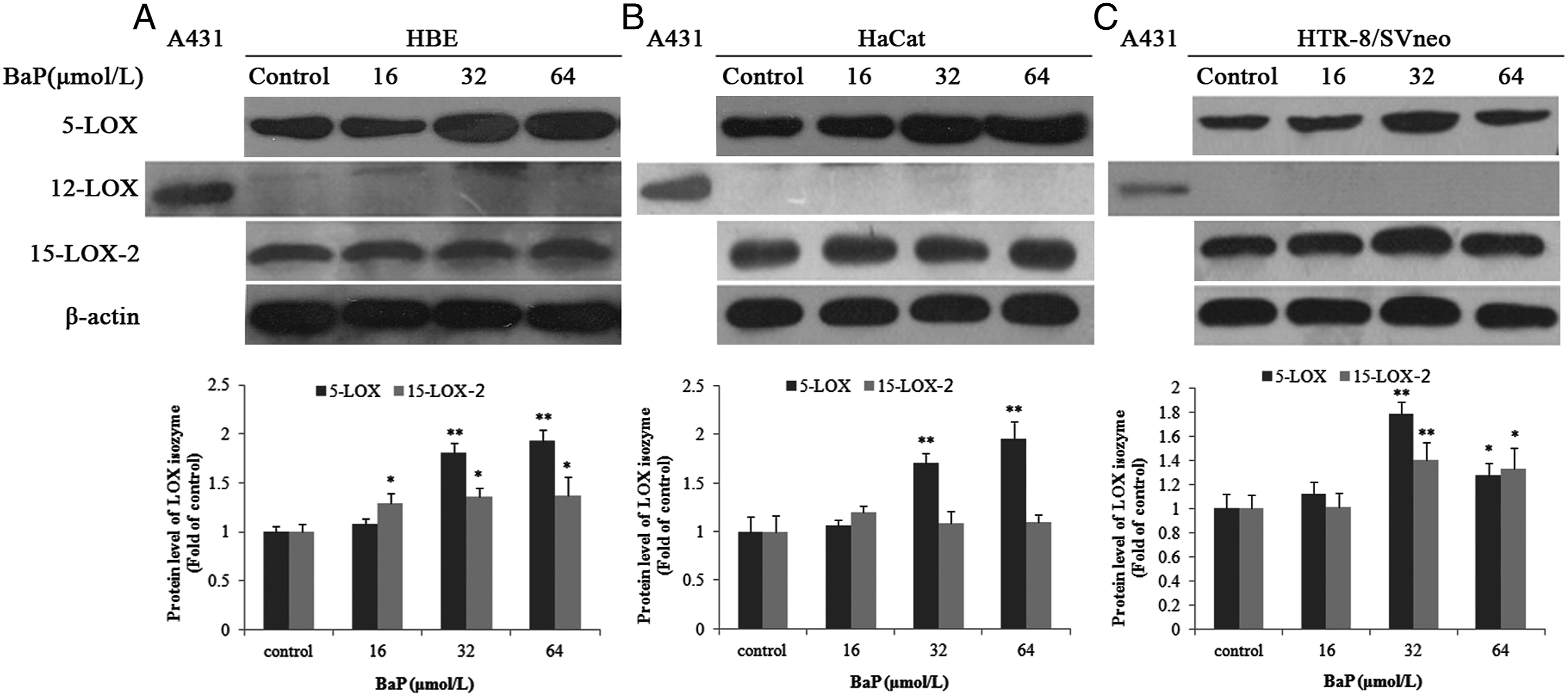
Lentivirus-5-LOX-shRNA, AA861, Naproxen, and α-Naphthoflavone Protect HBE Cells From BaP-Mediated Cell Proliferation Inhibition
Similar extent of cell proliferation inhibition was observed in HBE, HTR-8/SVneo, and HaCat cells (Figure 2A). The effect of Lentivirus-5-LOX-shRNA, AA861 (10 μmol/L, a 5-LOX specific inhibitor), naproxen (10 μmol/L, a PGS inhibitor) and α-naphthoflavone (10 μmol/L, a CYP1A1 inhibitor) on cell viability after BaP treatment was assessed using MTT assay (Figure 2B). No significant differences were observed among HBE, HBE-NC and HBE-5-LOX-shRNA control groups, and single enzyme inhibitor treatment group of HBE. BaP significantly reduced the percentage survival of HBE cells in a dose-dependent manner. Compared with the uninfected and empty vector control, the lentivirus-5-LOX-shRNA infection significantly inhibited the decreasing of cell viability induced by BaP. AA861, naproxen, and α-naphthoflavone also reversed BaP-induced cell proliferation inhibition. The protective effect of lentivirus-5-LOX-shRNA infection was higher than that of AA861, and the protective effect of AA861 was higher than that of naproxen and α-naphthoflavone. BaP-induced cell viability inhibition and the protective effect of lentivirus-5-LOX-shRNA, AA861, naproxen, and α-naphthoflavone. The cells were treated with BaP (0-128 μmol/L) for 24 hours. For inhibitor groups, the cells were preincubated with 10 μmol/L AA861, or α-naphthoflavone, or naproxen for 4 hours before BaP exposure. *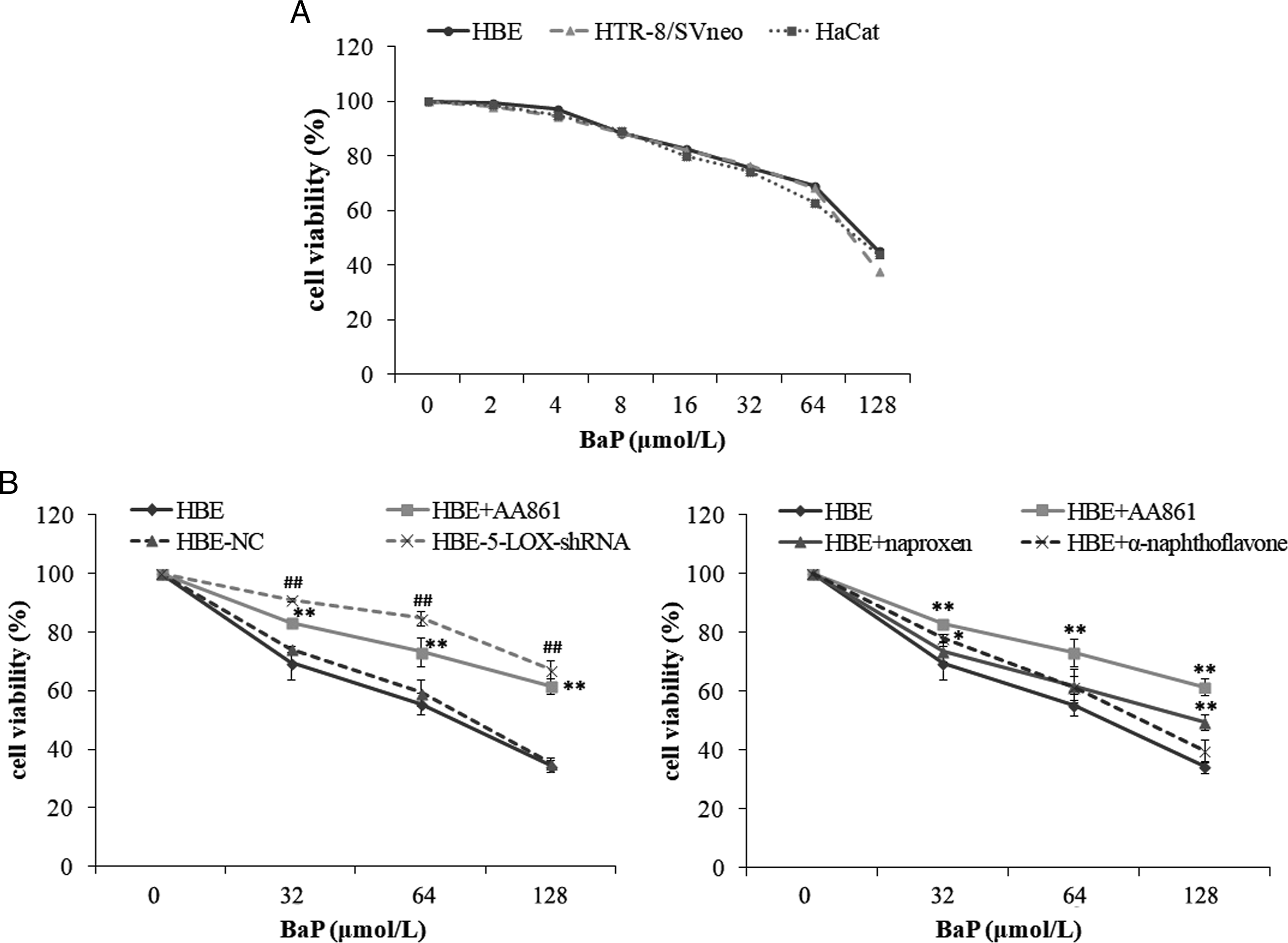
The knockdown efficiency of lentivirus-5-LOX-shRNA against 5-LOX was investigated using QPCR and western blot with β-actin as an inner reference, as shown in Figure 3. The 5-LOX mRNA and protein expression had no significant difference between the HBE-NC cells and the HBE cells. BaP treatment (64 μmol/L) enhanced the 5-LOX mRNA and protein expression in HBE, HBE-NC, and HBE-5-LOX-shRNA cells. The lentivirus-5-LOX-shRNA infection significantly suppressed the 5-LOX mRNA and protein expression in HBE cells compared with the lentivirus-NC-infected cells (mRNA: −87.0%; protein: −57.9%). A similar pattern was also observed in 64 μmol/L BaP treatment groups (mRNA: −82.5%; protein: −66.7%). Lentivirus-5-LOX-shRNA downregulated the expression of 5-LOX mRNA and protein in HBE cells. The BaP-treated groups were treated with 64 μmol/L BaP for 24 hours. A, 5-LOX mRNA expression. B, western blotting and gray analysis of 5-LOX. **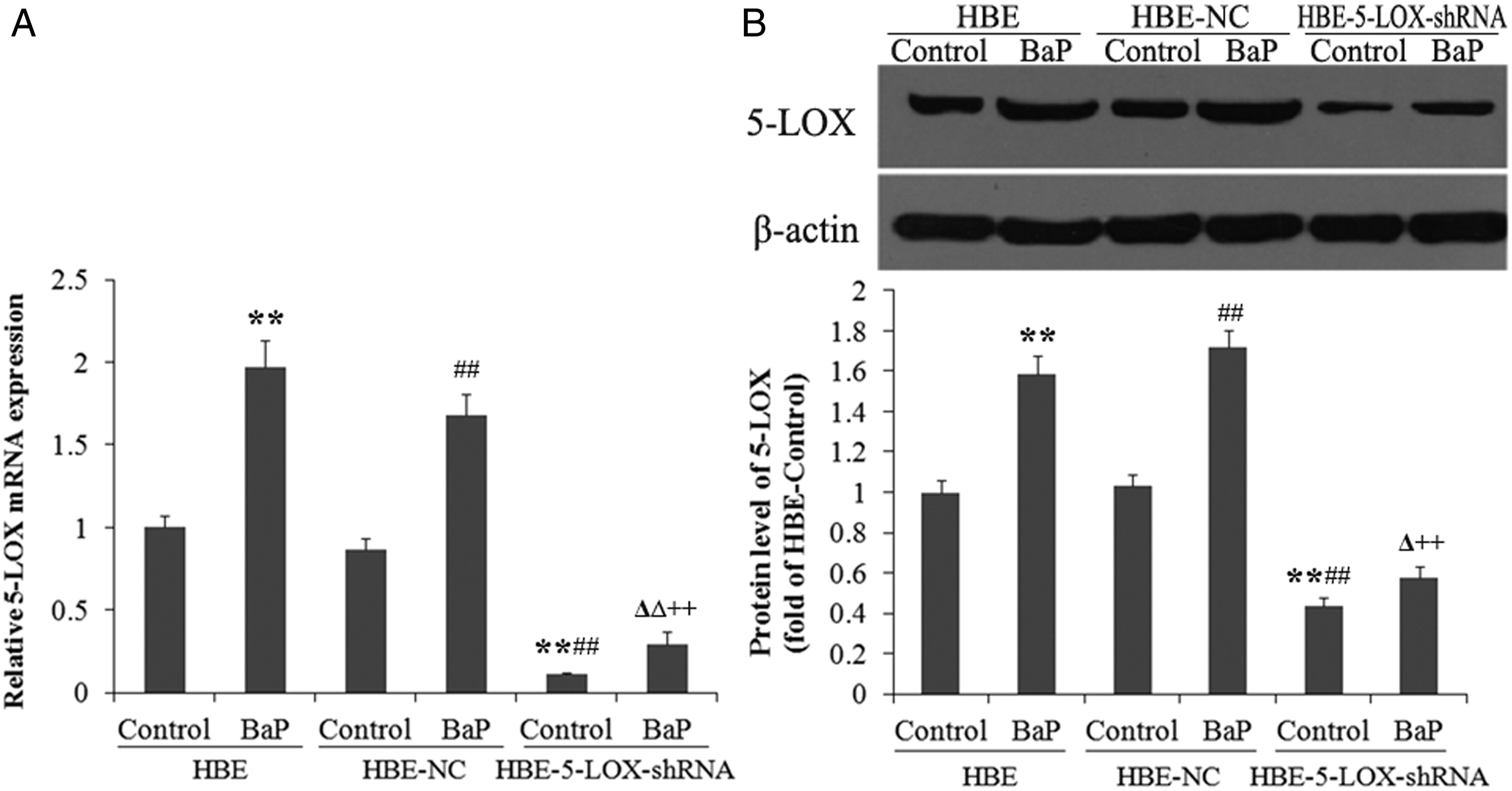
Lentivirus-5-LOX-shRNA, AA861, Naproxen, and α-Naphthoflavone Protect HBE Cells From BaP-Mediated DNA Damage
Comet assay was used to assess the extent of DNA strand breaks. The tail DNA% and Olive Tail Moment in BaP-treated group (64 μmol/L) were significantly higher than those in the corresponding control group, indicating that BaP induced DNA damage of HBE cells. Both the knockdown of 5-LOX by lentivirus-5-LOX-shRNA and 5-LOX inhibitor AA861 significantly suppressed BaP-induced increase in two DNA damage indices (inhibition rate: 46.6% and 57.5% vs 22.7% and 36.4% respectively). Naproxen only significantly inhibited the changes of Olive Tail Moment induced by BaP. No significant difference was observed between the inhibitor pretreatment group of α-naphthoflavone and BaP-treated group (Figure 4). Effects of metabolic enzyme inhibitors and lentivirus-5-LOX-shRNA on BaP-induced DNA damage in HBE cells. The cells were treated with 64 μmol/L BaP for 24 hours with or without 4-hour 10 μmol/L metabolic enzyme inhibitor pretreatment. DNA damage was expressed as (A) tail DNA%; (B) olive tail moment. NC: HBE-NC cells; shRNA: HBE-5-LOX-shRNA cells; NP: naproxen; α-NF: α-naphthoflavone. *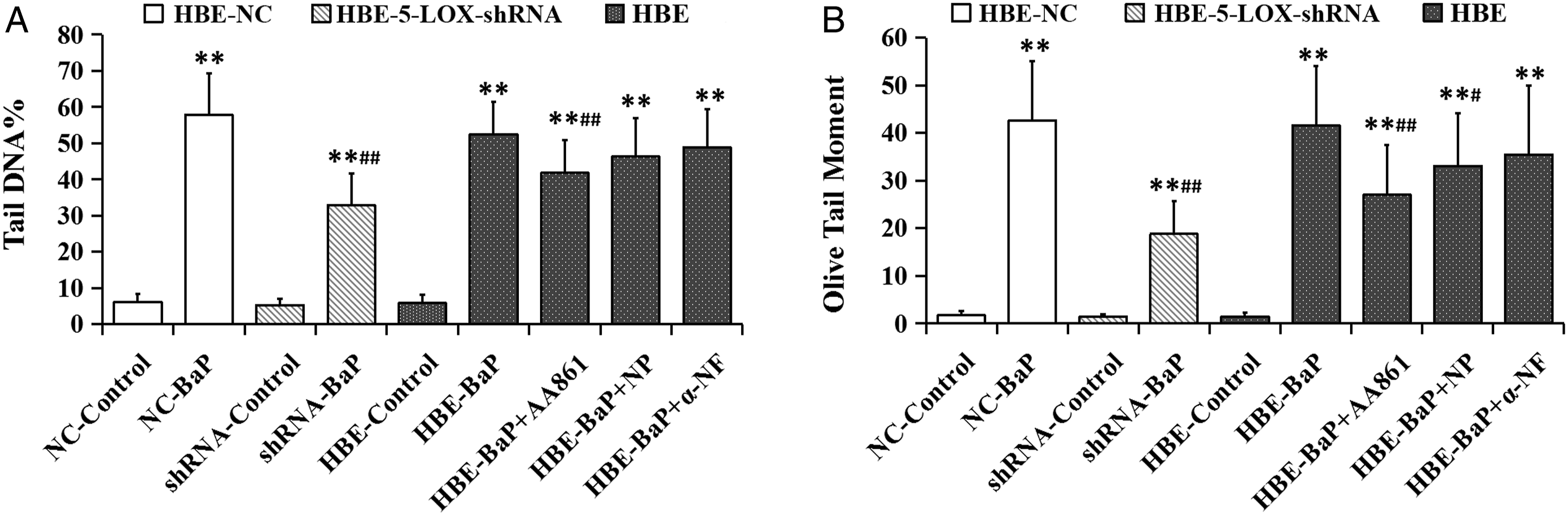
DNA oxidative damage was measured by detecting 8-OHdG formation. As shown in Figure 5, compared with the solvent control group, BaP increased the 8-OHdG content in HBE cells with the increase of exposure dose, and the difference was statistically significant when the concentration of BaP reached 32 and 64 μmol/L. In all BaP treated concentrations, the level of 8-OHdG in HBE-5-LOX-shRNA cells was significantly lower than that in HBE-NC cells and HBE cells. The pretreatment with AA861 or naproxen significantly reduced BaP-induced increasing of 8-OHdG in HBE cells when BaP concentration reached 32 and 64 μmol/L respectively. The order of inhibitory effects was lentivirus-5-LOX-shRNA infection > AA861 > naproxen. No significant changes were observed in α-naphthoflavone pretreatment groups. Effects of metabolic enzyme inhibitors and lentivirus-5-LOX-shRNA on BaP-induced 8-OHdG formation in HBE cells. The cells were treated with BaP (16, 32, 64 μmol/L) for 24 hours with or without 4-hour 10 μmol/L metabolic enzyme inhibitor pretreatment. HBE-shRNA: HBE-5-LOX-shRNA cells; NP: naproxen; α-NF: α-naphthoflavone. *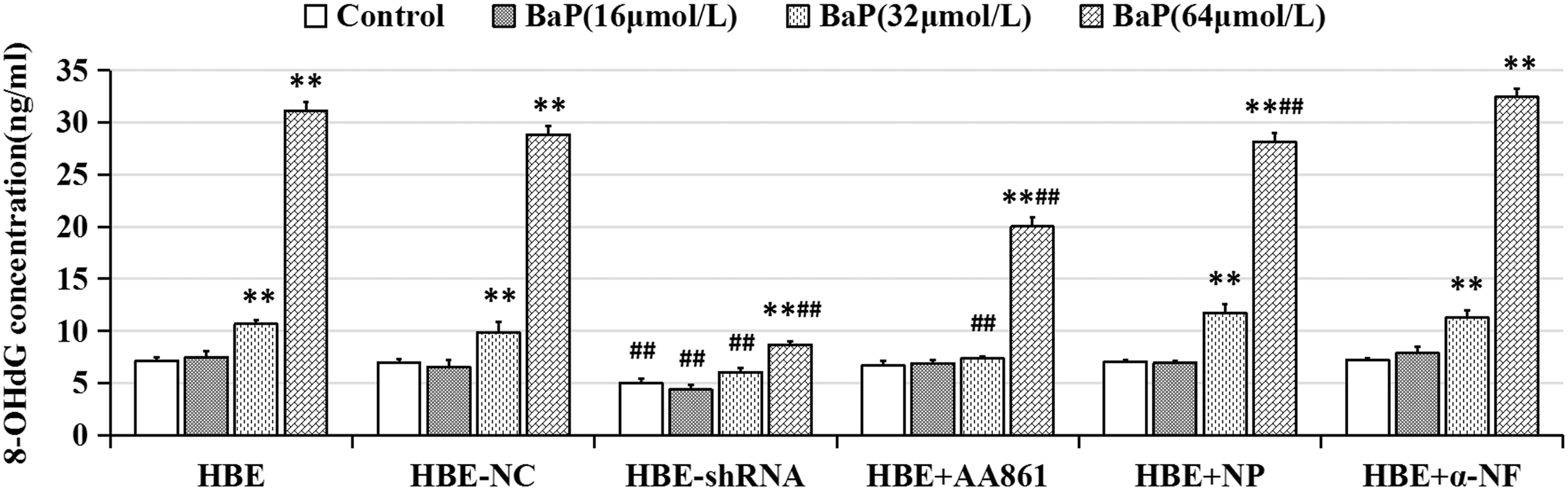
Discussion
In this study, we used the lentivirus-5-LOX-shRNA, 5-LOX-specific inhibitor AA861, PGS inhibitor naproxen, and CYP1A1 inhibitor α-naphthoflavone to investigate the effects of BaP metabolic enzymes, especially 5-LOX, on BaP-induced cytotoxicity, DNA strand breaks and oxidative DNA damage in HBE cells. Our data showed that in three epithelial cells (HBE, HTR-8/SVneo, and HaCat), 5-LOX and 15-LOX-2 proteins were expressed. BaP significantly upregulated 5-LOX expression; inhibited the proliferation of HBE; and significantly increased DNA strand breaks and 8-OHdG levels. Furthermore, the inhibition of 5-LOX, PGS and CYP1A1 by corresponding inhibitors pretreatment reversed BaP-induced anti-proliferation and DNA damage. The effect of 5-LOX inhibitor was greater than PGS and CYP1A1 inhibitor. The strongest protective effect was observed in 5-LOXshRNA.
Numerous references have shown that 5-LOX and 5-LOX activation protein (FLAP) expression was significantly increased in pathological tissues and cells of 5-LOX-related diseases, such as cancer tissues and cell lines,
32
and atherosclerotic tissue,
33
compared to normal tissues and cells. These data indicate that 5-LOX mediates the occurrence of related diseases through regulating its protein expression. In our experiment, the LOX isozyme expression in three epithelial cells, was detected by Western-blot to select the target cells and LOX isozyme for subsequent experiments. After exposure to different concentrations of BaP for 24 h, the expression of the major isozymes of LOX, 5-LOX, 12-LOX, and 15-LOX-2, were detected. There was 5-LOX and 15-LOX-2 protein expression in all three epithelial cells tested. BaP significantly upregulated 5-LOX and 15-LOX-2 protein expression with increasing exposure concentration, which indicated that these 2 isozymes might be responsible for the physiological and pathophysiological roles of LOX in BaP induced toxicity. In our previous study, similar results were observed in benzidine-treated SV-HUC-1 cells.
34
There is also literature supporting the expression of 5-LOX and 15-LOX-2 in HBE
35
and HaCat cells.36,37 These studies further confirmed those results. However, in our study, 12-LOX was not detected in tested three epithelial cells. This is inconsistent with literature reports which show 12-LOX expression in respiratory epithelial cells
38
and human keratinocytes.
39
We also failed to detect 12-LOX expression in our previous study of benzidine-treated SV-HUC-1 cells.
34
12-LOX has no or lower expression levels in other normal cells except predominantly in blood platelets and leukocytes.
40
The selected experimental conditions might be not optimal to detect lower levels of 12-LOX expression. 5-LOX and 15-LOX, which were shown to be upregulated in BaP-treated cells in this study, have been reported to epoxidize 7,8-dihydroxy-7,8-dihydrobenzo[a]pyrene to the ultimate carcinogen BPDE in the presence of arachidonic acid, γ-linolenic acid, or 15-hydroperoxyeicosatetraenoic acid (15-HPETE) in
BaP is an indirect carcinogen. It is generally believed that BaP-7,8-diol-9,10-epoxide (BPDE), the ultimate carcinogen of BaP mediated by bioactive metabolic enzymes, can covalently bind to nucleophilic sites of DNA to form the BPDE-DNA adduct, which is responsible for its carcinogenic and mutagenic effects.
8
In addition, oxidative DNA damage induced by ROS, which is formed during the metabolic activation process of BaP, is also a mechanism of BaP induced genotoxicity.
47
8-OHdG is a biomarker of oxidative DNA damage.
48
In this study, we used MTT assay, comet assay, and 8-OHdG analysis to measure the cytotoxicity and DNA damage respectively. The results showed BaP exposure significantly inhibited cell proliferation and induced DNA damage including DNA strand break and oxidative DNA damage of HBE cells with the increasing concentrations of BaP. Similar effects were reported in human bronchoalveolar H358 cells exposed to the metabolite of BaP BaP-7,8-dione,
49
and in human bronchial epithelial cells (BEAS-2B) treated with the metabolite of BaP BPDE.
50
It has also been reported that BaP induced cytotoxicity and DNA lesions are consistent with the elevated concentrations of BaP metabolites, which indicates that biotransformation contributes to the toxic effects of BaP.
51
These findings suggest that BaP is activated in HBE cells. In addition to the generally accepted BaP activating enzyme cytochrome P450, the BaP metabolic activities of LOX,22,23 PGS
52
and other metabolic enzymes existing in extrahepatic tissues have also been demonstrated
RNA interference (RNAi) is a gene silencing method with the advantages of rapid, reliable and convenient operation. This technology not only plays a fundamental role in plant and animal experiments, but also avoids some shortcomings of antisense nucleic acid technology and gene knockout in gene function research. 53 The gene silencing methods induced by siRNA greatly improve the specificity and efficiency of the inhibition method and provide advanced techniques for studying the specific functions of oxidase. In this study, the 5-LOX-shRNA infection demonstrated a stronger inhibition of the BaP-induced cell proliferation inhibition and DNA damage than brought about by AA861. This provides stronger evidence for the role of 5-LOX in BaP bioactivation. Although there are still many obstacles in the practical application of RNAi, such as the problems of off-target effect and safety after introduction into cells, siRNA is a powerful tool for the study of LOX function and may be a new way for the treatment of LOX-related diseases.
Footnotes
Acknowledgments
We sincerely thank Dr. Charles H. Graham (Queen’s University, Kingston, ON, Canada) and Professor Yanling Wang (Institute of Zoology, Chinese Academy of Sciences, Beijing, China) for generous providing and delivery of HTR-8/SVneo cells respectively.
Author Contributions
YH contributed to conception and design, analysis and interpretation of data, drafted manuscript, and critically revised manuscript. JW contributed to acquisition, analysis and interpretation of data, drafted manuscript, and critically revised manuscript. XZ and SH contributed to acquisition and analysis of data and critically revised manuscript. JH contributed to conception and design, interpretation of data, drafted manuscript, and critically revised manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hunan Provincial Natural Science Foundation of China under grant numbers 2020JJ4775.
