Abstract
Recommendations on study designs that adequately evaluate the in-life effects leading to juvenile bone toxicity, the various imaging modalities that can aid interpretation of the bone effects, biomarkers that may be useful, and regulatory issues were presented in this 2020 ACT symposium. The pathologies encountered in past studies were briefly mentioned. The first speaker covered study design and the numbers of juveniles that may be necessary to power the evaluation. Changes in the International Council for Harmonisation (IHC) guidelines were reviewed. The second speaker launched the rest of the symposium by describing the tools that may help assess juvenile bone toxicity, specifically those used to monitor bone toxicity, healing, and remodeling as they relate or drive the study design including model, species selection, and age. The third speaker addressed in more depth the micro-Computed Tomography (CT) applications in juvenile toxicology for evaluation of skeletal elements and bone growth in both embryo-fetal development (EFD) and pre and postnatal development (PPND) studies. Lastly, a regulatory perspective on strategies to assess juvenile bone toxicity and the concerns of the regulatory agency with respect to these potential changes in the juvenile population was addressed.
Nonclinical Juvenile Toxicity Studies: Design, Interpretation, and Risk Communication Considerations
LaRonda Morford led off by describing the complexity of juvenile bone toxicity studies due to the number of subsets and endpoints, some of which are unique to this study type and not seen in general toxicity studies, that are needed. To meet the objectives of pediatric risk assessment, the juvenile bone toxicity study must include bone-specific endpoints, proper interpretation of these endpoints, and appropriate communication of the results. Ronee Baracani authored the summary of this presentation.
Introduction
Juvenile animal studies (JAS) are complex studies due to the number of subsets and endpoints, some of which are unique to this study type and not often included in general toxicity studies. Designing an effective JAS to meet the objectives of this study type, including bone-specific endpoints, as well as interpreting endpoints on the JAS, and communicating the results of these studies, were highlighted as part of the pediatric risk assessment.
Regulatory Guidance
Regulatory guidance for the conduct of JAS is provided via the ICH guideline entitled, “Nonclinical Safety Testing in Support of Development of Paediatric Pharmaceuticals,” otherwise known as ICH S11. 1 The ICH S11 1 guidance was formally adopted in early 2020 and provides a regulatory framework for nonclinical support of pediatric drug development. Although this guidance pertains specifically to the conduct of JAS, other guidances including the ICH M3(R2), 2 ICH S5 (R3), 3 ICH S6(R1), 4 and ICH S9 5 should also be considered when planning a JAS. The development of the ICH S11 1 guidance was developed to address different regional guidelines pertaining to nonclinical testing in support of pediatric development, including the FDA 6 and EMA 7 nonclinical juvenile animal guidances. A key factor when considering JAS is that the need for, and design of, a JAS should be considered on a case by case basis with regards to necessary endpoints, dosing period, and timing. Each regional guidance, although supported, often in practice did not implement a more standardized study design approach to ensure that all regional preferences were incorporated. The ICH S11 1 provided a harmonized approach on the need for and design of a JAS which was intended to: 1) Reduce the likelihood of substantial differences among regions, 2) Facilitate conduct of pediatric clinical trials due to more meaningful and high-quality JAS data, and 3) Minimize animal use through the spirit of 3Rs.
Is a JAS Required? The Weight of Evidence Approach
An important strategy that came out of the ICH S11 1 is the weight of evidence (WoE) approach, which is an integrated assessment that would aid in determining whether a JAS is indeed necessary. The WoE approach takes into consideration numerous aspects of pediatric clinical development such as pediatric age and indication. The key question to ask when considering the WoE approach is: Would additional nonclinical data add value? The JAS would only be needed if animal and human data are insufficient to support pediatric patients as the JAS identifies safety concerns that cannot be adequately addressed in other nonclinical studies or pediatric clinical trials. Conversely, a JAS would not be warranted if identified safety concerns can be clinically monitored and/or managed in a pediatric clinical trial.
Major Factors When Determining the Need for a Juvenile Animal Study (JAS).

Another point to consider when determining the need for a JAS, is that a drug may have more than one pediatric indication. A WoE should be considered for each pediatric clinical use or trial. A JAS is designed to specifically support and address the pediatric clinical plan, thus if the clinical plans including intended age of patients and indications differ, would one approach cover everything? Therefore, a JAS may need to be planned to support all uses or a separate JAS may need to be conducted for each indication.
Pediatric Development Plans: Pediatric Investigation Plans (PIP) and Pediatric Study Plans (PSPs)
The overall clinical development plan needs to be fully understood to develop an appropriate nonclinical strategy. Historically, the pharmaceutical industry was reluctant to include children in clinical trials due to concerns regarding unknown toxicity in children and the challenges associated with organizing pediatric clinical trials. This led to high rates of off-label use of drugs in pediatric patients. As a result, regulations (e.g., 2012 FDA Safety & Innovation Act [FDASIA] 8 ) were enacted that mandated pediatric drug development, with the requirement that pharmaceutical companies submit pediatric plans to the respective agencies that outline the types and timing of pediatric clinical trials. Therefore, the specific timing of the nonclinical study would be outlined in the PSP for the FDA and/or PIP for the EMA. It is therefore important to understand those timing obligations to ensure compliance with those agreements made with the appropriate agency.6,7
These PSP and PIP agreements would contain an outline of the pediatric study, or studies, that a Sponsor would plan to conduct, including clinical and nonclinical studies. Early discussions are warranted regarding the timing and general design of the JAS. The PIP and PSP documents typically include: 1) Information on the disease in pediatric populations, 2) A request for deferral or waiver (if applicable), 3) The outline of studies including the study objectives and basic design, including the JAS, 4) Timeline for submission of the studies, 5) Information on plans for age-appropriate formulations, and 6) A detailed description of supporting studies and agreements with other health authorities.
PIPs are binding to the Sponsor, and a compliance check does take place by the EMA. Therefore, it is vital that what is included in a PIP document aligns with the JAS plan, as well as fulfillment of promised submission dates, otherwise the Sponsor will be out of compliance. Every single aspect of the agreed upon JAS design will be on the compliance checklist. As a result, it is important to be aware of any PIP changes to ensure the JAS remains in compliance.
Regulatory agencies may also request changes to the clinical plans as detailed in these PIP and PSP documents. For example, the pediatric age group to be included in the clinical trials may be requested to be lowered by the agency. As a result, the JAS design and plan would need to change accordingly to support that younger intended pediatric patient age, such as now dosing younger animals. Likewise, based on the initial WoE assessment that was conducted, it may have been determined that a JAS was not required initially, but with the request to support younger pediatric patients, there now may be a need to conduct a JAS. Therefore, it is crucial to allow sufficient time to obtain and incorporate regulatory feedback before the start of any JAS study.
Designing the JAS
Designing the Juvenile Animal Study.
JAS Endpoint Considerations
Another key aspect that came out of the ICH S11 1 guidance is the establishment of “core” endpoints as part of the JAS. These core endpoints are generally expected to be on every JAS and include mortality and clinical observations, measures of growth including body weights and long bone length, food consumption, sexual development, clinical pathology, anatomic pathology, and toxicokinetics (beginning and end of dosing period). 1 While most of these would be basic to any toxicology study, the long bone length is more specific to the JAS and typically one long bone (e.g., femur) measured for length at necropsy is sufficient. Sexual development is typically determined by documenting the balanopreputial separation in males and vaginal patency for females during the postweaning period (if the study design encompasses this relevant developmental window).
Depending on the safety concerns identified in the WoE assessment, additional endpoints would also be included such as more extensive clinical pathology, biomarker, coagulation, and/or urine collections. 1 Due to the limitations in obtaining sufficient sample volumes from smaller juvenile animals, any additional samples or sample volume will often require the use of more animals. Other additional endpoints might include standard ophthalmic examinations that are not routinely included in a JAS, immunological assessments such as functional assays including T-Cell-Dependent Antibody Response (TDAR), and additional tissues and organs beyond the standard list included in the anatomic pathology evaluation and potentially use of special stains, electron microscopy, and immunochemistry. Likewise, reproductive assessments can also be included on a JAS which would include expanded histopathology examinations and organ weights of the reproductive tissues/organs. Other reproductive assessments may include estrous cycling, sperm analysis (e.g., sperm counts, motility, and morphology), and mating assessments. If the compound has a central nervous system (CNS) target and/or developmental neurotoxicity is a concern based on the WoE assessment, CNS assessments should also be included. These assessments may include detailed behavioral clinical observations, behavioral tests (e.g., locomotor activity), learning and memory tests (e.g., complex maze assays), and expanded CNS histopathology. Finally, other growth and development assays such as crown rump length, body length, and/or withers height as well as serial non-invasive measures of long bone length (e.g., using X-ray) can be included, as well as bone assessments. When there is a concern specific to bone metabolism or structure, then additional assessments of bone mass and geometry using densitometric techniques, biomarkers of bone formation and resorption, and bone histomorphometry should be employed. 1
When designing JAS, study designs can quickly encompass multiple subsets of animals, such as cohorts for toxicokinetics, neurobehavioral assays, and reproductive assessments. Thus, these studies are large and complex, often with many litters and pups. Based on the species utilized in conjunction with the age of the animals, there may be :1) Limitations in blood sampling and tissue collection volume, 2) Routes of administration that are not feasible (e.g., intravenous infusion), 3) Species selection challenges (e.g., NHP typically not an option), and 4) Certain endpoints that may not be practical or feasible depending on the age of the animal.
Example Juvenile Animal Study Design.

Total number of animals on study = 574
*TK: Toxicokinetics.
Interpretation of JAS and Pediatric Labeling
Scientific challenges related to data interpretation on JAS studies need to be considered. Many endpoints in a JAS are interconnected and potentially related to systemic toxicity. For example, body weight is highly correlated with the onset of sexual maturation (e.g., vaginal patency) and animals with reduced body weights will often have a delayed onset. 9 Thus, it is crucial to have a complete understanding of the data to delineate what is test-article related. Additionally, it is key to understand the normal development response in the species selected to ensure that natural processes, such as when unscheduled rodent pup deaths due to poor maternal care or inter-individual variability in growth rates during development occur, are not interpreted as test-article related.
The results of a JAS need to take into consideration all the known information, including nonclinical data in adult animals, and any potential clinical data. Evaluation of: 1) Type and severity of effects, 2) Reversibility, 3) Dose level and exposure where effects occurred, and 4) Age of animals at which effects occurred all need to be related to the intended pediatric use. This integrated assessment provides information for clinical trials and labeling. Results of the nonclinical JAS can be used to set clinical exposure limits, identify biomarkers or endpoints for clinical monitoring, and identify age groups in which the drug should not be used or where special warnings are required.
Juvenile animal data will need to be incorporated into the drug label. Per the final 2019 FDA guidance, “Pediatric Information Incorporated Into Human Prescription Drug and Biological Product Labeling,” the label, under the Pediatric Use subsection, should include a concise summary of clinically relevant nonclinical JAS data under the Juvenile Animal Toxicity Data heading. There would be no need to include JAS data under the Nonclinical Toxicology section. Generally, JAS data is not included in the label when the data do not suggest an adverse signal. When the JAS data suggest adverse signals not previously assessed in a pediatric clinical study, only the information that may have clinical relevance should be summarized. Additionally, a key consideration is that the summary should be discussed in clinically relevant terms including human equivalent dose exposures, the ages of the JAS animals in relation to the approximate human ages, impacted organ systems, the duration of the treatment of animals and the relationship to clinical use, reversibility of adverse effects, and any developmental delay (if any). When pediatric clinical data and nonclinical data suggest similar risk, the clinical data would be used to provide key details and clinical implications of risk and the nonclinical JAS data would be briefly summarized, if appropriate. 10
Example Labels With JAS Data
As previously noted, the JAS data will need to be incorporated into the drug label under the Juvenile Animal Toxicity Data heading including use in specific populations, warnings and precautions for use in pediatric patients, recommendations for use in pediatric patients, and/or contraindications of specific populations (e.g., patients younger than 12 years old). 10
For example, the package for Prezista® (darunavir), a human immunodeficiency virus (HIV-1) protease inhibitor, included a JAS in rats. In the label, it is noted that there were deaths in rats from postnatal day (PND) 5 and toxicity and mortality up to PND 23 to 26. A 4-week repeat dose toxicology study in rats that initiated dosing on PND 23 had no deaths noted. As a result, the outcome on the label was an age limitation for dosing in pediatric patients, specifically limited for use in pediatric patients 3 years of age and older. 11 Another example was provided for Linzess® (linaclotide), which due to the results of the JAS in neonatal mice, the label contains a black box warning which contraindicates pediatric patients less than 6 years of age due to the risk of serious dehydration. 12
Summary
Nonclinical support of pediatric clinical use requires forethought and significant planning. A WoE approach should be undertaken to ensure the need for, and the specific design of, the JAS, as each JAS should be handled case-by-case, including the need for and type of any bone toxicity assessment. The conduct of a JAS is not straightforward, and it is essential to understand the dynamic animal models, endpoints, and the relationships that exist between endpoints. The objective is to develop a scientifically sound, yet technically feasible, strategy to collect meaningful and relevant data that can help ensure that the medicines provided to pediatric patients are as safe as possible.
Specific Study Design and Tools to Evaluate Bone Changes in Juvenile Studies
Aurore Varela presented principles, requirements, and practices for juvenile toxicology studies as well as the utility of different models, emphasis on study design pitfalls, and the most useful assessments and their impact and interpretation. The “Bone Toolbox” was outlined, including biomarkers, imaging, biomechanics, and histomorphometry.
Introduction
Assessment of bone in juvenile toxicity studies is an important endpoint in evaluating drug effects on postnatal bone development and growth. Regulatory guidance for juvenile toxicology studies from the US FDA, the EMA, the Japanese regulatory agency, and ICH S11 1 indicate that the skeleton is an “organ system of concern” and that growth should be assessed by body weights in conjunction with long bone length. When there is an identified concern about bone metabolism or structure, the measurement of bone related biomarkers and/or expanded histopathology (e.g. histomorphometry) should be considered. Assessment of bone mineral density (e.g. micro-CT, Dual-energy X-ray Absorptiometry (DXA), or Peripheral Quantitative Computed Tomography (pQCT)) or bone structure (e.g. micro-CT) can also be conducted as appropriate. Identifying bone as a target tissue in drug development to address any potential for liability is important as data emerges to show that several signaling pathways may positively or negatively affect the skeleton.
Therapies Affecting Skeleton, Skeletal Growth or Development
Risk assessment in developing animals can include comparatively simple measurements of growth that are easily incorporated into standard toxicology study designs. Where there are concerns for off-target effects or when pharmacological endpoints are required to assess postnatal musculoskeletal development, as well as bone quality, more comprehensive evaluations are required. There are several pharmaceuticals with known effects on skeletal maintenance or development such as antidiabetics (e.g. PPAR-gamma agonist), antivirals (e.g. Tenofovir), beta2-adrenergic agonists (e.g. Clenbuterol), bone anabolic agents (e.g. parathormone and antisclerostin antibodies), bone antiresorptives (e.g. bisphosphonates and denosumab), corticosteroids, fibroblast growth factor agonists, fibroblast growth factor receptor 3 antagonists, Janus-activated kinase inhibitors, proton pump inhibitors (e.g. omeprazole), retinoids, and Wnt inhibitors.13-25
Skeletal Postnatal Development
Ossification occurs by two processes: intramembranous and endochondral ossification. Intramembranous ossification is the major process that supports growth of flat bones and foci of woven bone in primary ossification centers formed during the embryonic and early fetal period. Endochondral ossification is the major process that supports longitudinal growth of the long bones and starts during fetal development. The growth plate separating the primary and secondary ossification centers is responsible for continued bone elongation by progressive remodeling of cartilage to bone. Growth in diameter is related to increases in bone width at the periosteum.26,27 Longitudinal growth, radial growth, and cortical and cancellous bone drifts require bone modeling, the main process during growth and development. This is the process where the formation and resorption do not occur on the same surface leading to changes in the shape of bone or the bone mass in response to growth or adaptation to mechanical strain (bone geometry changes can be independent of bone growth). Reshaping of bone by modeling drifts occurs during growth. Resorption can enlarge the marrow cavity, while formation adds bone periosteally to maintain the mechanical competence during bone elongation, thereby maintaining the overall shape of the bone as it grows at other sites.
Adaptation of the mature skeleton and its turnover is based on bone remodelling, which is based on a sequential process of cellular activation, osteoclastic resorption, reversal, and osteoblastic formation at the same location in which resorption and formation are usually coupled. Both bone modeling and remodeling occur on periosteal, endocortical, and trabecular bone surfaces. 26
Growth and development of the skeleton can be assessed through important postnatal milestones including the appearance/fusion of secondary ossification centers, longitudinal bone growth, diametric bone growth (bone expansion), and bone accrual. Growth is completed when adult longitudinal size is attained, while the skeletal maturity is achieved by the closure of the epiphyses. Peak bone mass assessed by bone mineral content (BMC) and/or bone mineral density (BMD) is a determinant of bone strength when associated with bone geometry and bone material properties. In many non-rodent species including humans, skeletal maturity occurs after the growth plate closure and the end of longitudinal bone growth. Generally, peak bone mass occurs after the closure of the epiphyseal growth plates and varies by sex and skeletal site. These sex differences for the timing of growth and development of the skeleton are also encountered in animals and marked differences are present among species. The sequence of growth plate closure is generally similar in mammals.
Skeletal End-Points in Juvenile Toxicology Studies
Investigating the developing skeleton leads to specific challenges in preclinical studies. The tools are available to assess the potential for a compound to affect bone and to characterize those effects. The selection of measurements for bone evaluation can be critical for assessing the effect of a drug and these bone measurements can be specific based on the pharmacological or toxicological target. Integration of these data into toxicology studies, including in the pre and postnatal study, should be routinely performed as part of risk assessment in preclinical drug development. Study duration might vary depending on the species and its duration of the bone turnover cycle. In rodent juvenile studies, the study duration should at least cover the rapid phase of growth. If there is no specific concern for a bone liability, simple physical measurements can be performed as minimal requirements (included long bone length measurements along with body weight and crown to rump). Species selection is also another important point to consider; rodents are a frequently accepted and recommended species for testing bone compounds. Rodent models do not completely reflect human bone responses, but they provide a quick detection of a signal in bone with well powered studies and well validated bone endpoints. Dog and NHP bone is similar to human with respect to intracortical remodeling but with a more rapid turnover. Males are often more sensitive than females as they have a higher growth rate on a longer timeframe.
Qualitative histopathology has sufficient sensitivity to detect pathological changes in most cases. However, the standard microscopic evaluation of decalcified hematoxylin- and eosin-stained bone section is not a very sensitive technique to evaluate changes in bone mass and geometry. There are several established imaging modalities that can be used to evaluate bone end points, and each modality has its own advantages and disadvantages. In vivo evaluations of the musculoskeletal system include radiology to identify abnormalities (focal lesions, abnormal shape, and other radiological findings including growth plate assessment), biochemical markers of bone turnover, measures of bone mass, density, and geometry, and can extend to ex vivo measurements of bone architecture, bone dynamics, and bone strength, as needed.
Radiology remains an essential tool of skeleton evaluation, and provides a simple and commonly available technology, although it requires a skilled and experienced veterinarian or veterinary radiologist. It is a straightforward means for examining the size, shape, skeletal abnormalities, and overall density of the entire bony skeleton or large subdivisions thereof, in a stereotypical orientation. Longitudinal evaluations on skeletal growth and development and growth plate assessment are also key in juvenile toxicology studies. Radiological diagnosis can effectively support histological diagnosis and often is helpful in deciding which bone locations should be examined histologically to best characterize the disease process or treatment effect.
Bone densitometry techniques (DXA, pQCT, and micro-CT) provide information on bone mineral content, bone mineral density, and bone geometry. Data derived from markers and bone densitometry can be used to trigger more in-depth analyses using histomorphometry to derive quantitative data on bone cell function. If appropriate, bone strength can also be measured. In vivo body composition assessments of lean mass and/or fat mass can be captured simultaneously and further characterized histologically or quantitatively using histomorphometry techniques. These studies can be run using all standard and specialized routes of administration in the major rodent and non-rodent laboratory species. DXA limitations are important to understand especially in the context of juvenile studies and include: two-dimensional (2D) projection of a three-dimensional (3D) structure affect bone size and shape, inability to make geometrical evaluations, inability to distinguish effects on trabecular and cortical bone density, and inability to determine dimensions, e.g., the effects of body size and composition on the BMD measurement. BMD data are influenced by bone size and the composition of surrounding soft tissue. Therefore, DXA data can be influenced by treatment effects on body weight where growth may affect bone size, or treatment may affect body composition (lean mass vs fat mass). However, DXA provides bone density data from non-clinical animal studies which are clinically relevant.
Other techniques, primarily pQCT, can compensate for DXA limitations and are recommended for use in addition to, or instead of, DXA. pQCT scans provide information on BMC, volumetric BMD, bone size and geometry (total, trabecular and cortical bone areas, cortical thickness, periosteal and endosteal circumferences), and cross-sectional moment of inertia (CSMI, e.g. bone mass and its distribution in the bone used to derive surrogate measures of bone strength). It allows differentiation between trabecular and cortical bone. pQCT allows assessment of changes in size and geometry along with bone mass and density, which is critical to evaluate and understand changes in relation to all aspects of bone growth and development. Bone assessments are not only a question of BMD.
As with any technique, standardization procedures are key for data reproducibility including technical training, animal or specimen positioning, and anatomical landmarks (particularly critical in growing skeleton where morphology can vary rapidly). Site selections are also critical due to differential growth rate and sequences at different sites and even at proximal vs distal aspects within the same bone which may affect assessments. More specifically for imaging, it is important to highlight the scanner calibration and maintenance, scan settings, image processing, and procedure precision, along with GLP validation of technologies, software, and procedures, definition of the raw data, and standard operating procedures (SOPs).
Bone strength is a critical end point in skeletal assessment, serving as the gold standard of bone quality. 28 Biomechanical testing of bone constitutes the unique opportunity to evaluate the functional impact of alterations in bone turnover, bone mass, and bone geometry. Correlations between biomechanical measurements and bone densitometry parameters provide important insights into the effects of drug treatment on bone quality. Bones are scanned (DXA, pQCT) to obtain information on bone geometry to normalize strength parameters for bone size. Bone density and biomechanics parameters are used to assess the relationship between bone mass and strength. If an “apparent” effect on the skeleton is only related to body weight, normalization of data could help. pQCT will reliably provide measures of BMC and BMD as well as bone geometry which will reflect size and biomechanical testing. This demonstrates if parameters are unaffected when corrected for bone size and will support the conclusion that skeletal changes were related to body weight and did not affect bone quality.
Bone histomorphometry is an important tool for assessing changes in bone tissue, such as bone formation, mineralization, and resorption, and to characterize the mechanisms by which a test article acts on bone. Bone histomorphometry provides valuable information about bone structure, bone formation, bone resorption, bone mineralization, and bone modeling and remodeling activity. Given the widespread use of this technology, bone histomorphometry nomenclature has been standardized.29,30 Fluorochrome labeling with calcium chelators can be administered during a study if effects upon bone formation are suspected. Dynamic bone histomorphometry is critical to assess bone turnover and mineralization in trabecular and/or cortical bone. Injections of fluorochrome label are usually performed on at least two occasions prior to bone harvesting; intervals will depend on species and its bone cycle but also the age of the animals, as bone cycle can be shorter in juvenile animals.
The choice of end points will depend on the mechanism of action of the drug, type of changes expected, objectives/questions asked, and duration and type of study. It is important to understand the techniques, their applications, and limitations, to fully integrate the generated end points in the interpretation of preclinical studies.
Imaging Modalities to Assess Juvenile Bone Toxicity
Arun Tatiparthi addressed the various imaging techniques used in these analyses.
Introduction
Juvenile animals undergo rapid anatomical and physiological changes. Thus, postnatal toxicity in the developing tissues of juvenile animals is of concern and hence the effects of drugs on these developing organs is of great interest. Since bone is one of the primary organs in the skeletal system, imaging is used as a bone biomarker for assessing drug effects on bone in juvenile animal studies (JAS).
There are advantages of using imaging in JAS. First there is high translatability of the methodology from non-clinical to clinical studies since there is a widespread use of these imaging modalities in clinic. Second, the use of longitudinal evaluation for assessing temporal changes helps to use minimal number of animals for assessing drug effects addressing one of the 3R’s.
The most recent ICH S11 1 guideline recommends measurement of femur bone lengths in juvenile animal studies as one of the core (primary) end-points for growth assessment. This can be either a terminal measurement at necropsy or longitudinal measurement via imaging to monitor changes overtime. The guidelines also recommend considering assessment of additional bone endpoints when there is a concern about bone metabolism. These additional end points are bone mass and geometry via densitometric techniques. The ICH S11 1 guidelines though doesn’t recommend any specific imaging modality for use in bone assessment. Other guidelines, such as the “FDA Guidelines for Osteoporosis” details more about using imaging modalities for both densitometric and morphometric assessment and these can be considered in conjunction with ICH S11 1 for designing JAS.
Drugs and Bone Metabolism
Many drugs, including glucocorticoids, various inhibitors, anticonvulsants, and others are known to affect bone metabolism. 31 In mammals, longitudinal skeletal growth occurs rapidly in embryonic and early postnatal life where endochondral ossification is tightly regulated by a number of growth factors (PTHrp, Ihh, IGF-I, BMP/TGFβ, Wnt, VEGF), and transcription factors (Sox9, Runx2, Osterix, β-catenin) that play an important role during skeletal development and maintenance of bone mass. Thus, any drug interactions with these signaling pathways can affect skeletal development and hence bone end points are of importance and advised in JAS studies. For example, thyroid hormones are known to have effects on the secondary ossification centers (SOC), since the time of appearance of SOCs in several species coincides with the time when peak levels of thyroid hormones are attained. 32
Imaging Modalities in JAS
Various imaging modalities, including DXA, pQCT and micro-CT are used in JAS. The choice of the imaging modality depends on factors, such as species of interest, bone end points including densitometry and morphometry, qualitative or quantitative measures and finally longitudinal or terminal evaluation. These imaging techniques may be employed in clinical studies as needed. Some clinical scanners are used in non-clinical studies and the researchers should know the limitations of each of these modalities for making a rational decision on the choice of enrolling a particular imaging modality for JAS.
Two-Dimensional (2D) Radiography
Radiography is a 2D methodology used for both quantitative (like bone lengths) and/or qualitative assessments in JAS. Quantitative measures such as bone lengths and qualitative assessments, including visualizing suspected findings can be achieved with 2D radiography. Both rodents and large animals can be imaged with this modality in either in-vivo or ex-vivo settings. Since radiography is a 2D methodology, animal positioning is of utmost importance. Improper positioning hinders both quantitative measurements and/or qualitative comparisons.
2D radiography is a quick imaging methodology and hence can be utilized to visualize and assess findings like lesions, ectopic calcifications, improper or delayed formation of secondary ossification sites, or to follow through the formation and closure of growth plates. Differences in radio opacity can also be visualized and assessed qualitatively. For example, the qualitative rating scale for the growth plate visualization can comprise appearance of GP, partial union or complete closure of GP. All of these findings can be assessed longitudinally in the animals using the 2D radiography methodology.
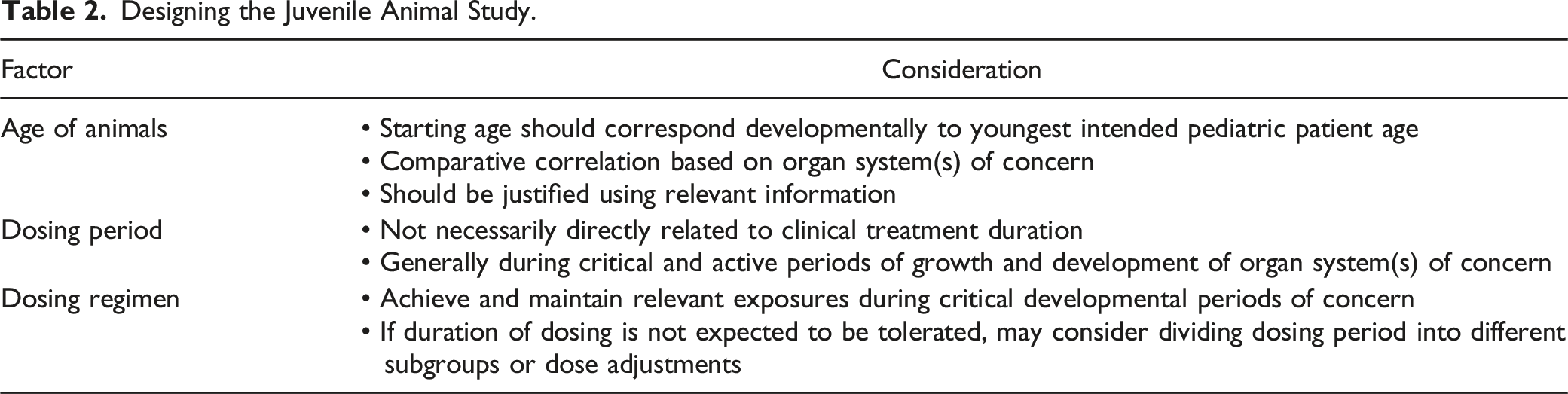
The following sections include examples of 2D radiography applications in JAS for both rodent and large animals. Figure 1 shows bone length measurement from a 7-week old Wistar rat; the same images can be used for assessing growth plates or suspected findings. Figure 2 radiographs from a 3-month old and a 2-year old cynomolgus monkey shows bone length measurements demonstrating how the longitudinal evaluation can be used for measurement and evaluation temporally. Radiographs of a 7-week of Wistar rat. Radiographs of a 3-month old (left) and 2-year old (middle and right) cynomolgus monkey.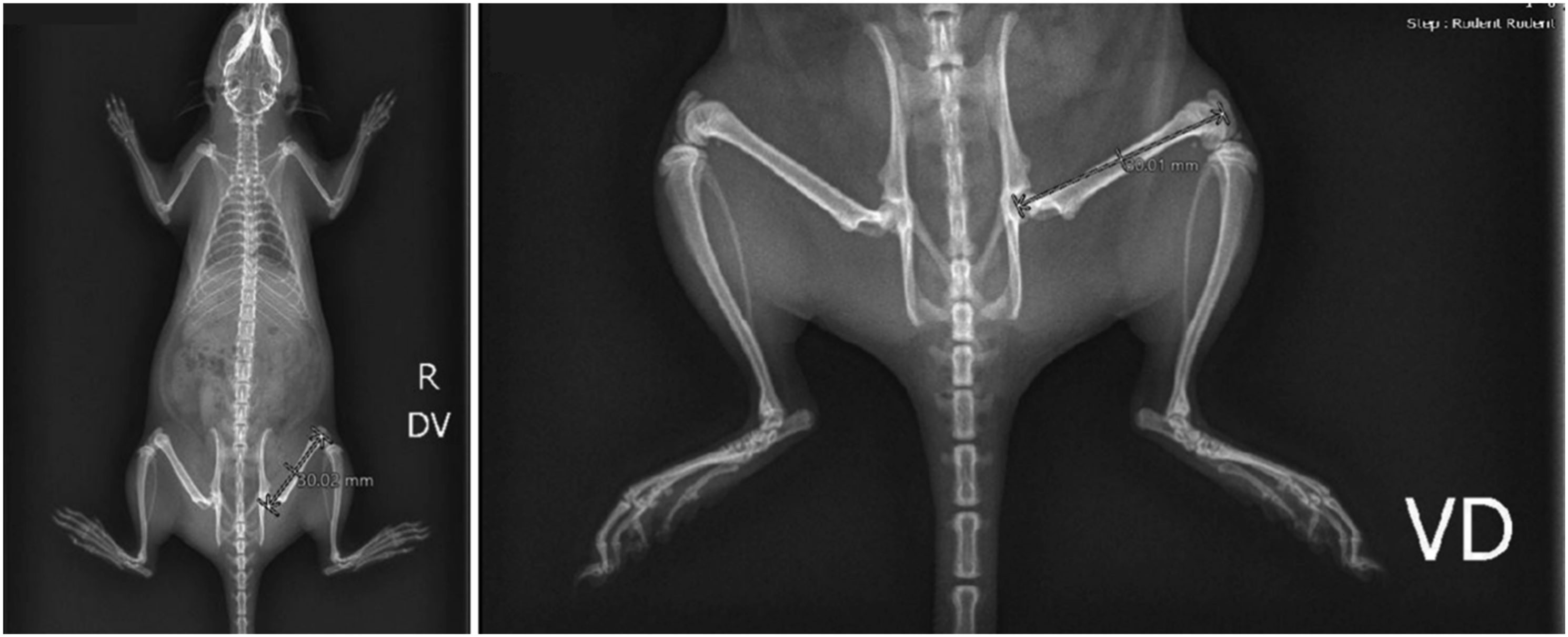
Dual-Energy X-Ray Absorptiometry (DXA)
DXA is a widely used imaging modality and is considered a gold standard for measuring patient BMD in the clinic. DXA is used for BMD measurements in JAS for both longitudinal and terminal evaluations. This imaging modality can accommodate both rodents and large animals depending on the type of clinical or non-clinical scanners.
DXA is a 2-dimensional imaging methodology and hence subject orientation impacts measurements, similar to the radiography measurements. For example, BMD measurements via DXA are influenced by bone area (which is required to be calculated) and the bone area can be affected by subject placement. Also, the size of the animal can influence the BMD measure. Lower BMD values are reported on obese clinical subjects due to greater increase in area. This is because the reported BMD is an areal-BMD and not a true volumetric BMD. 33
There are differences in mechanisms of action of various drugs and this can cause changes in bone structure, such as increases in cortex by anabolic drugs (teriparatide and abaloparatide) that are known not to increase bone density but shows an increase in bone area via DXA that results in a decrease in BMD. 34 This is can be a limitation for the use of this 2D imaging method. Another limitation is that the clinical scanners are not suitable for rodent imaging due to technical limitations on animal body weight (∼250 g). However, rodent DXA scanners are available in smaller footprints to fill this gap. 35
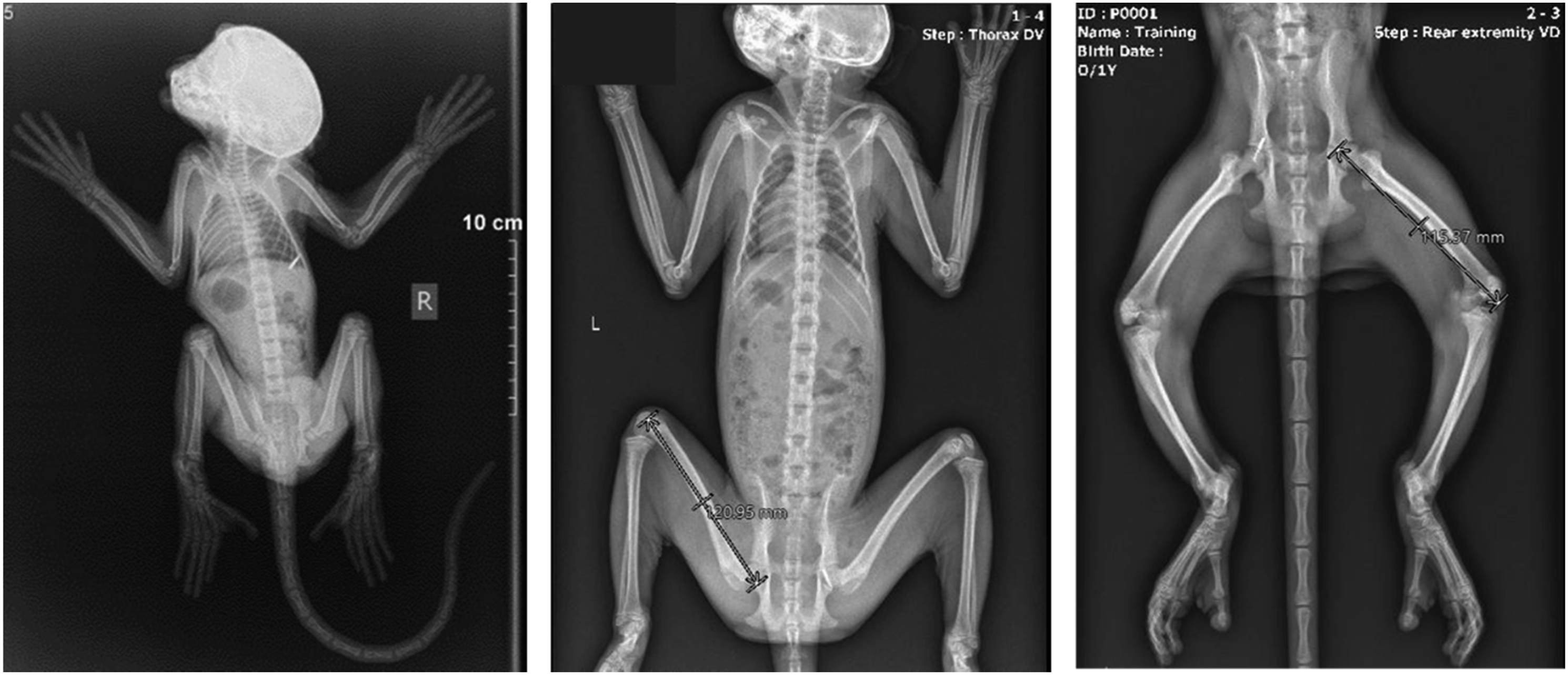
Figure 3 is a DXA image of an excised rat femur scanned post necropsy in a JAS for assessing bone densitometry (total femur bone density, content and bone area). DXA can also be used for longitudinal measurements of bone parameters and this is shown in Figures 4 and 5 for a Sprague Dawley rat and a Cynomolgus monkey JAS study, respectively. The yellow regions of interest shown here represent the areas of analysis like left and right femur, spine, pelvis, and others. Densitometry using Dual-energy X-ray Absorptiometry from an excised femur of an SD rat. In-vivo measurement of densitometry using Dual-energy X-ray Absorptiometry in a SD rat. In-vivo measurement of densitometry using Dual-energy X-ray Absorptiometry in a cynomolgus monkey.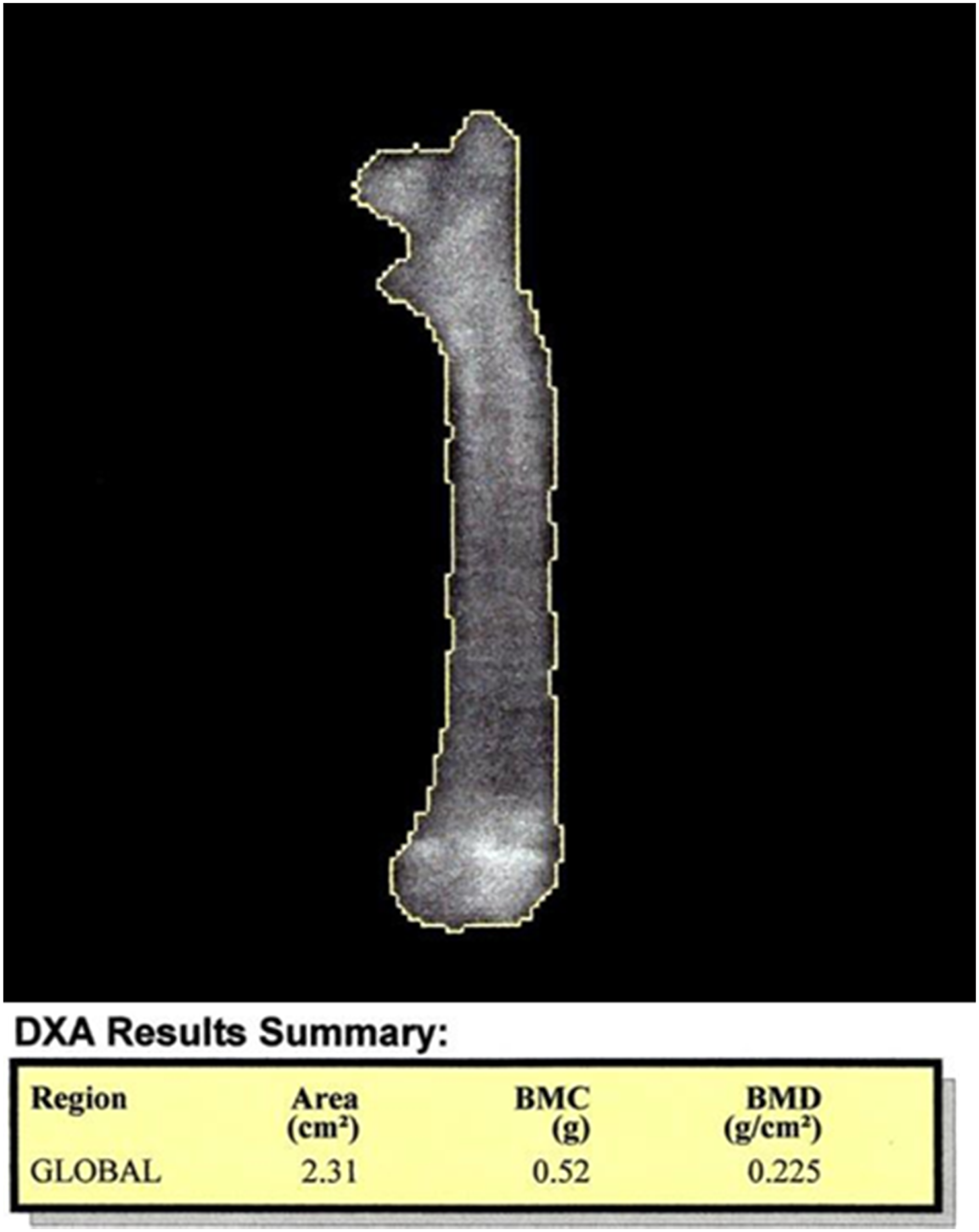
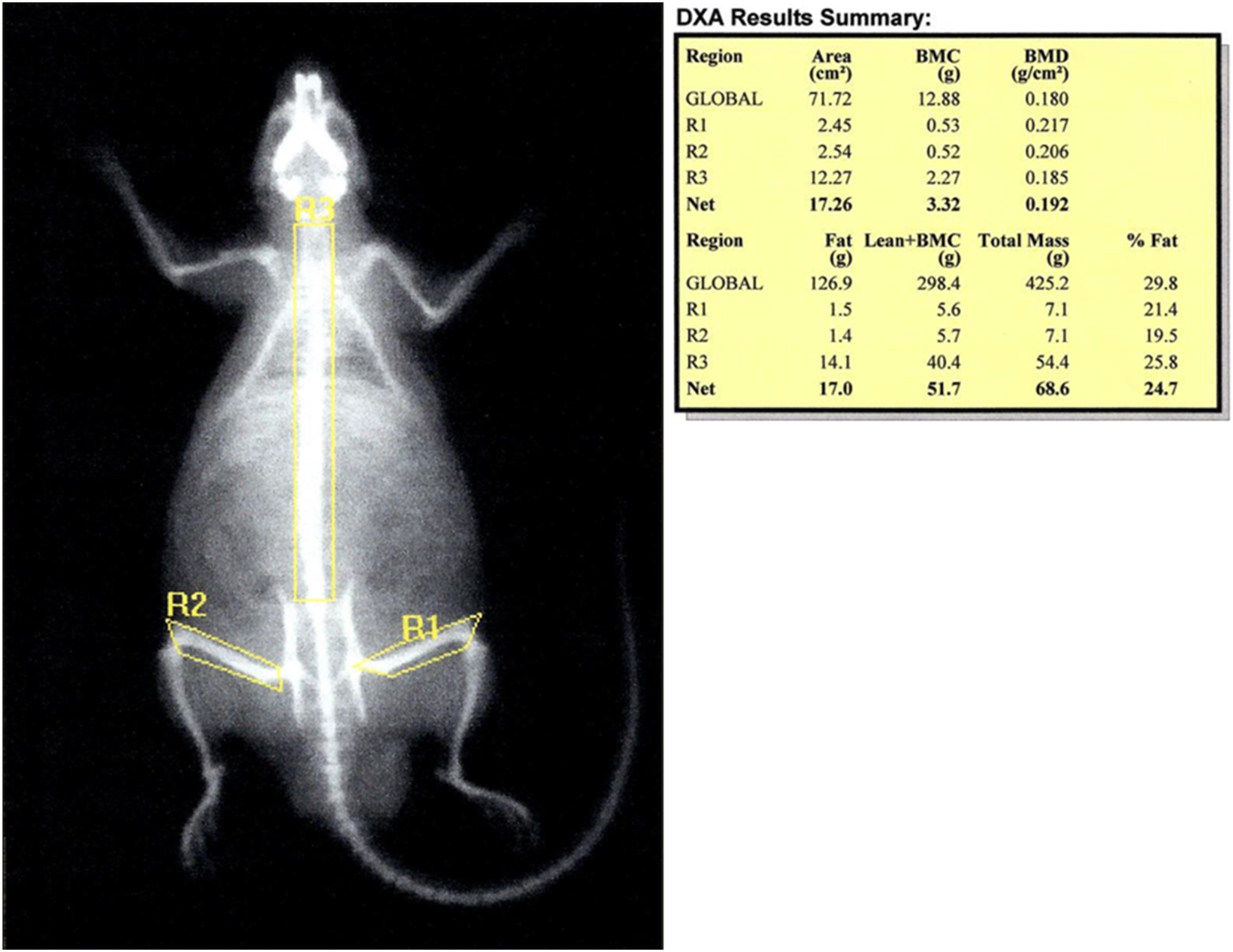
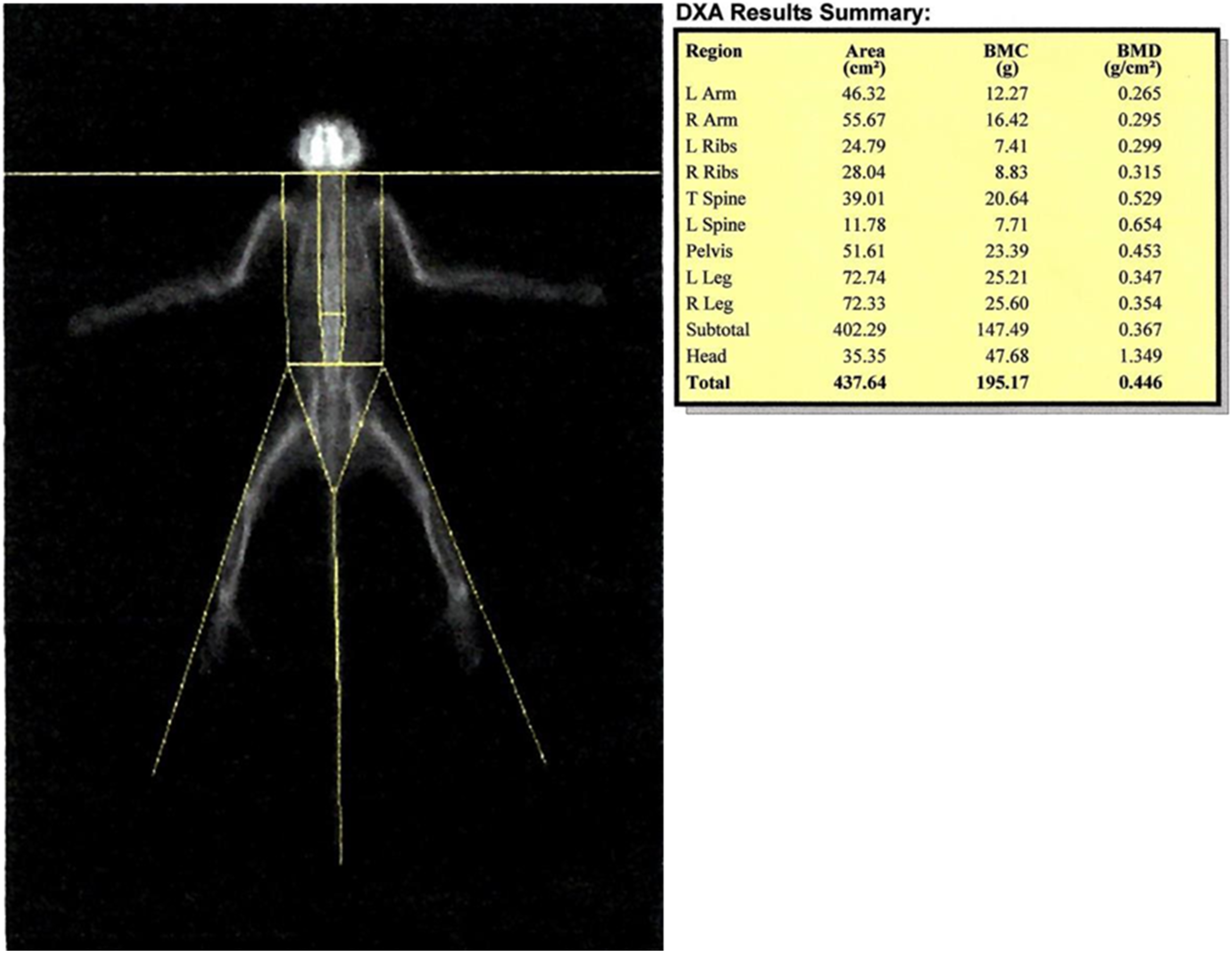
Peripheral Quantitative Computed Tomography (pQCT)
pQCT is a 3-dimensional (3D) imaging methodology used for bone densitometric and morphometric measurements. pQCT provides volumetric BMD measurements and not areal-BMD measures as provided by DXA. This is a high-resolution imaging methodology that can also measure subtle and early bone changes in trabecular and cortical regions of the bone.
pQCT systems are available for both clinical and non-clinical research and they are used particularly for measuring trabecular and cortical bone parameters. These trabecular and cortical bone measures are of interest when the drug in evaluation is known to have primary effects on bone metabolism.
pQCT measurements are based on reference lines and regions of interest and this is shown by Gasser & Willnecker on the left tibia of a 3-month old rat. 36 pQCT is limited for measurement of extremity bones only and in clinic, it is used for measuring mostly the radius and ulna or tibia and fibula. pQCT can analyze only one slice per region, one each in trabecular and cortical regions, not the entire bone or specific region of bone BMD as measured in DXA or Micro-CT.
Micro Computed Tomography (Micro-CT)
Micro-CT is another 3D imaging modality similar to the pQCT but at a higher resolution with the ability to scan the entire length of the bone or the entire animal. Micro-CT can be used for quantitative measurements like bone densitometry, morphometry and even radiography that can be performed with this imaging modality. The CT imaging modality is widely used in clinic and micro-CT is a non-clinical version of this clinical system. Total bone BMD measurements from micro-CT correlate well with DXA measurements and this imaging modality is suitable for imaging both rodents and large animals in a JAS.
Since micro-CT is a 3D imaging methodology, subject orientation will not impact the measurements unlike 2D methods of radiography and DXA. The bone area issues for DXA are not a concern for micro-CT since the BMD measurements are calculated based on bone volume and not bone area. The one limitation for micro-CT imaging is that it cannot perform in-vivo imaging for large animals like dogs and NHP’s due to the bore size limitation. However, this can be achieved with clinical CT scanners. Micro-CT can be used to perform ex-vivo bone measurements from mouse to NHP. Along with quantitative measurements, qualitative assessments can also be performed for confirming any findings like lesions or skeletal abnormalities.
The following examples show the use of Micro-CT imaging for JAS in both ex-vivo and in-vivo measurements. Figure 6 is an example of micro-CT 3d image of an excised left femur of a SD rat from a JAS that was scanned ex-vivo post necropsy. Femur length, total femur densitometry (BMD and BMC) and regional trabecular and cortical bone densitometry and morphometry were calculated. In-vivo longitudinal evaluation of rat pups was studied in a PPND study where skeletal growth was assessed from PND0 to PND21 via measurements of long bones – femur, tibia, radius, and humerus.
37
This example shows micro-CT applications in rodent imaging for longitudinal assessment of drug effects on bone in JAS. The same in-vivo images can be used for qualitative assessments similar to radiography. Micro-CT rendered 3D image of left femur from SD rat.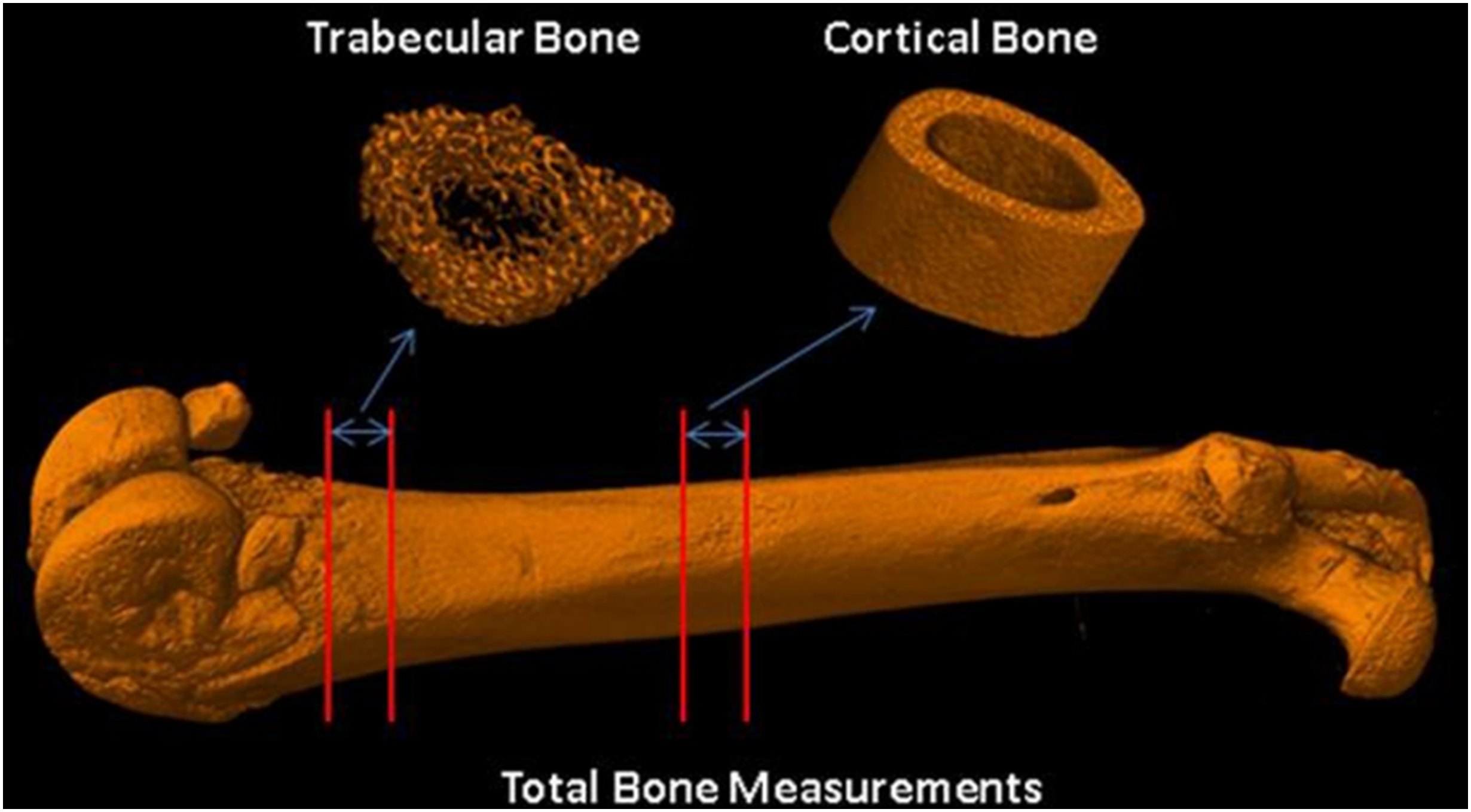
Comparison of Imaging Modalities
As we have seen, each imaging modality comes with its advantages and limitations. A comparison based on various criteria is shown below and in Table 4. ➢ Radiography and DXA are 2D imaging modalities and pQCT and Micro-CT are 3D methodologies ➢ DXA and Micro-CT can perform whole bone BMD measures ➢ pQCT and Micro-CT can perform region trabecular and cortical bone measures as well ➢ Micro-CT is the only methodology that can perform bone lengths, radiography and total and regional bone densitometry – all 3 in one Comparison of Imaging Modalities.

How to Choose the Modality?
The answer to this question depends on the answer to the subsequent question – “is bone metabolism involved as a primary or a secondary response to the drug interactions?”. The following can be used to determine the modality of usage: ➢ Species and the number of imaging time points ➢ Qualitative monitoring or quantitative assessment ➢ Number of imaging time points and the pathology of interest
How to Interpret the Data?
This is an important final step after choosing imaging modality and the end points to read. Here you may measure just the bone length or a combination of bone length, bone densitometry or even morphometry but all these measurements must consider perspectives in growth and bone quality
Juvenile animals have exponential postnatal growth. For example, femur length and BMD grow exponentially up to 75 days in Sprague Dawley rats and then plateaus thereafter.
38
Bone modelling is the predominant process during the early development where bones are shaped by the independent actions of osteoblasts and osteoclasts, and defines skeletal development and growth. Therefore, any changes to skeletal growth due to drug interactions can be detected. ➢ Bone lengths are attributed with growth and should be considered in conjunction with body weights for evaluating any effects. ➢ BMD and BMC should be used as a surrogate measure for bone strength.
39
➢ BMD is attributed to mass-normalized bone strength by measuring mass per unit volume. ➢ BMC reflects bone stiffness by measuring total bone matrix mineralization, hence whole bone BMC is of particular importance.
40
➢ The strength of bone as a tissue is determined by the amount of mineral that is present. Higher BMC indicates higher bone matrix mineralization and higher bone stiffness.
Summary
There are various imaging modalities that are available for use in JAS and it’s important to understand the advantages and limitation of these systems to make an informed decision on what modality to enroll for your JAS.
The choice of the bone end points depends on: ➢ If there are any known drug influences on bone metabolism and the need for measurement of additional bone end points along with the core bone length measurement. ➢ The choice of the imaging modality depends on species of interest, longitudinal or terminal assessment and qualitative or quantitative evaluation. ➢ The suggested bone end points are bone length, bone densitometry and bone geometry/morphometry.
Regulatory Approaches to Drug-Related Juvenile Bone Toxicity
Finally, Mukesh Summan from US FDA addresses the regulatory approaches to testing for and managing drug-related juvenile bone toxicities.
Introduction
From a regulatory point of view, pediatric data gaps exist: a false belief that children are ‘little adults’ and dosing should be based on weight alone; lack of age appropriate formulations; and a limited market for children compared to the market for adult indications lead to fewer studies. These issues are compounded by ethical concerns regarding testing children who are not able to fully consent. Thus, study designs may have difficulty setting acceptable endpoints for children and there may be a lack of validated pediatric assessment tools. Children’s organ developmental stages may affect drug PK and PD or the drug may affect the organ development, leading to safety concerns. One solution offered to overcome pediatric issues includes incentive programs granting a 6-month market exclusivity. Additionally, legislation and implementation of pediatric regulations has helped clarify how studies in children should be conducted.
Regulations
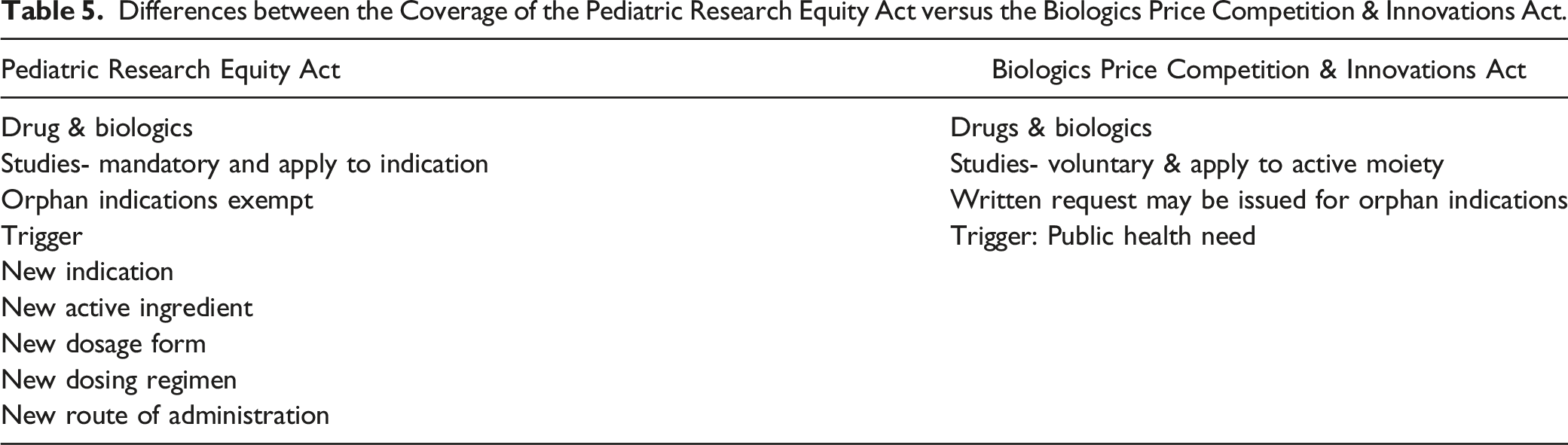
The main regulations covering juvenile bone toxicity testing are the 1998 Pediatric Rule, 41 2002 Best Pharmaceuticals for Children ACT (BPCA), 42 2003 Pediatric Research Equity Act (PREA), 43 2007 FDA Amendments Act (FDAAA), 44 2010 Biologics Price Competition & Innovations Act (BPCI), 45 and 2012 FDA Safety & Innovation Act (FDASIA). 8 The 1998 Pediatric Rule 41 required drugs and biologics to be evaluated for safety and efficacy in children if the product was used in a substantial number of children and the product provides a meaningful therapeutic benefit over existing treatment. The BPCA9,42, provided pediatric exclusivity reauthorization for on-patent products and referred off-patent drugs to the National Institutes of Health (NIH) to study. The PREA 43 replaced the Pediatric Rule and was retroactive for all applications from April 1999. This Act established the Pediatric Advisory Committee. The FDAAA 44 reauthorized the initiatives of the BPCA and PREA and established a Pediatric Review Committee (PeRC). The BPCI 45 allowed biologics to be eligible for a written request review.
Differences between the Coverage of the Pediatric Research Equity Act versus the Biologics Price Competition & Innovations Act.
The BPCA Written Request (WR) 46 is reviewed by the FDA division and the PeRC for the public health benefit; whether the study designs are feasible, sufficient to support the dosing, safety and efficacy; whether all populations and conditions have been addressed; whether there is a PREA requirement, and whether other products are approved for the condition. WRs may include multiple indications, both approved and unapproved indications and may require nonclinical studies.
To develop the PSP, an overview of disease in the pediatric population for the product under development must be included. Other items to include: potential plans and justification for use of extrapolation; plans and justification for full and partial waiver; plans for pediatric-specific formulation development; nonclinical date, both completed and planned, to support studies in children; synopsis of all the planned clinical studies, timeline foe the PSP; and provide any agreements with other Health Authorities (e.g., Pediatric Investigation Plan (PIP) or FDA WR under BPCA).
The agreed PSP includes plans to request any waivers and deferrals of pediatric studies with a clear justification and supporting data that may include sponsor data, literature, consensus documents, etc. Waivers and deferrals are not officially granted until the product is approved. The agreed PSP and any amendments are included with the application submission.
Pediatric drug development is global with occasional exchange of nonclinical and clinical scientific information between FDA and European Medicines Agency (EMA). These interactions include monthly teleconferences to discuss product specific pediatric development and to review PIPs, WR, PREA requirements, pending regulatory actions and safety issues. The point of the interactions is to avoid exposing children to unnecessary or inadequate trials, share safety data to reduce risk, and to share trial data to inform future studies.
For juvenile toxicity study nonclinical data, regulatory expectations are that the PSP and BCPA WR will include this data as appropriate. This is on an individual basis with no class or disease exclusions. Additionally, it is expected that safety endpoints of excipients and salt forms, if part of the active moiety, will be addressed. Nonclinical study relevant data should support the dose and duration of treatment and if no additional studies are planned, a rationale must be given. The review and discussion of the PSP is coordinated with EMA and involves the engagement of the PeRC. Attention is given to the youngest patients and a focus is given to neonates. The Guidance for Industry Nonclinical Safety Evaluation of Pediatric Drug Products (2006) 6 may help the sponsor. 6
The PeRC is an internal review committee with members drawn from across the FDA with expertise in pediatrics (including neonatology), clinical pharmacology, chemistry, safety, statistics, toxicology, and ethics. PREA reviews include the PSPs, waiver and deferral requests, pediatric plans, and pediatric assessments and the BPCA reviews pediatric written requests.
With respect to juvenile bone toxicity, considerations for what may or should be studied are bone size (length, width), longitudinal and appositional bone growth, bone biomarkers, densitometry (DXA and/or pQCT), and/or mechanical strength. Changes in bone size may be bone specific or secondary to body weight effects and may reflect treatment effects on bone cells and/or growth plate chondrocytes. Changes in DXA/pQCT parameters may be related to changes in bone size, e.g., bone mineral content. 47 The agency also considers the mechanism of action (MOA) as to whether the skeletal effect is biologically plausible, whether there is a class effect, and if the findings are clinically significant. Some effects may be species specific. 48 It is important to test a dose range nonclinically that may predict human effects. The agency also considers what should be monitored to identify adverse bone effects in clinical trial subjects.
Case Study
A case study was described for an ongoing clinical trial for a kinase inhibitor in which the trial data reported increased fractures in 5% of adults and 23% of pediatric patients. The nonclinical standard rat and dog studies had no histological bone abnormalities. In the juvenile 13-week rat toxicity study, terminal body weights were decreased and femur length at PND 98 and 128 was decreased. 49 The questions were: was the bone effect secondary to growth effects and were appropriate tests conducted? It was acknowledged that histopathology may not identify effects on bone quality or growth plate activity. Published literature showed evidence of involvement of the drug target in chondrogenesis and osteogenesis. 50 Related drugs showed decreased bone marker activity (P1NP), decreased BMC of the tibia, decreased bone length (femur and spine), and decreased body weight. 51 It was concluded that there was some evidence of a class effect. Nonclinical studies were recommended and included the Schenck assay for bone growth and mineralization and a juvenile rat study with extended bone evaluation that included bone densitometry, histomorphometry, mechanical strength, QCT, and BMC. 52 Monitoring within the pediatric trial subjects included densitometry (DXA, ca. 6-12 months of the lumbar spine, femoral neck, total hip), bone biomarkers (P1NP, BSAP, CTX-1, osteocalcin), calcium metabolism biomarkers (Vitamin D, PTH), linear growth (ca. 6 months), X-ray radiographic evaluation of epiphyses (wrist and knee) at 6-month intervals, and included fractures as an adverse event of interest in the trial protocol.53,54
Footnotes
Acknowledgments
Our thanks to summer interns, C. Pope and A. York, for organizing this manuscript for publication.
Author Contributions
LaRonda Morford, Aurore Varela, Arun Tatiparthi, and Mukesh Summan were the speakers providing the content for this summary article. Ronee Baracani wrote the summary of LaRonda Morford’s presentation. Kathleen Funk wrote the summary of Mukesh Summan’s presentation. Maralee McVean and Kathleen Funk were co-chairs for the organization of this symposium presentation and manuscript submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
