Abstract
In animal studies, light microscopic examination remains an important tool in the detection and characterization of effects on the skeleton. In the case of both anticipated and unanticipated effects on bone histology, pathologists must carefully select terminology that accurately conveys skeletal morphology without overstating what information can be derived from a standard decalcified paraffin-embedded section. The International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) project issued standardized nomenclature for toxicologic pathologists to use with respect to the skeleton. Consistent with general INHAND principles, the nomenclature emphasizes a descriptive approach to classifying lesions, rather than using terms that imply a specific pathogenesis or disease process. This article, which is based on a presentation at the Society of Toxicologic Pathology’s 36th Annual Symposium, will discuss the role of anatomic pathology evaluation in the context of skeletal evaluation in toxicity studies. The integration of anatomic pathology data with quantitative bone end points is reviewed. An overview of the INHAND diagnostic scheme is provided and accompanied by case examples in which the INHAND terminology has been applied. In addition, this review summarizes key considerations for toxicologists and pathologists assigning adversity designations when considering bone end points.
The basis of this article is a presentation from the Society of Toxicologic Pathology’s 36th Annual Symposium that focused on the musculoskeletal system. The primary focus of the presentation was understanding how to apply the previously published International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) bone nomenclature scheme which was finalized and published in 2016 (Fossey et al. 2016). This review presentation also highlighted the important role of microscopic examination in detecting both pharmacologic and toxic effects on the skeleton and emphasized the complementary roles of routine qualitative light microscopic with more quantitative bone end points such as densitometry, histomorphometry, and biomechanics. Finally, the topic of assigning adversity for bone findings was discussed, and the challenges were illustrated with case examples. Because this presentation was not intended to review the complete diagnostic scheme established by INHAND, the reader is referred to the information available in the publication (Fossey et al. 2016) or available on the goRENI website (http://www.goreni.org/).
Anatomic Pathology: An Important Tool in the Bone Toolbox
Various methods are available for the evaluation of the skeleton. As summarized by Dr. Aurore Varela in the preceding presentation in the symposium, important bone end points can include biochemical markers of bone formation and resorption, assessments of mineral ions and hormones relevant to bone metabolism (vitamin D, parathyroid hormone, and fibroblast growth factor), densitometric assessments of bone geometry and mass, biomechanical testing, and static and dynamic histomorphometry. In light of these quantitative and often highly sensitive techniques, it is important to understand the role of qualitative light microscopic examination in preclinical safety evaluation of the skeleton (Figure 1). Standard repeat-dose toxicity studies are screening studies that attempt to detect effects across a wide range of tissues and organs utilizing a relatively standard set of end points that include live-phase observations and clinical and anatomic pathology assessments. In the vast majority of repeat-dose toxicity studies for biopharmaceutical or environmental agents, specialized quantitative examination of the skeleton is not performed. Skeletal effects in these standard studies could be detected by live-phase observations such as fractures and limb deformities or suggested by changes in certain standard clinical pathology assessments such as calcium, phosphorus, or alkaline phosphatase determinations. In reality, only a relatively small fraction of skeletal effects presents as clinically observable findings, which means that the careful evaluation of bone histology is the key method of detecting skeletal effects in standard toxicity evaluations. Because these studies allow the comparison of site-matched, age-matched specimens between the control and treated animals, the study pathologist can assess both qualitative and semiquantitative changes in bone morphology. Examples of qualitative changes or lesions include bone necrosis, cysts, fractures, and neoplasia. Examples of semiquantitative changes include assessments of the relative quantity of cortical or trabecular bone in a particular region, changes in the thickness of physeal or articular cartilage, or changes in the relative numbers of osteoblasts and osteoclasts.
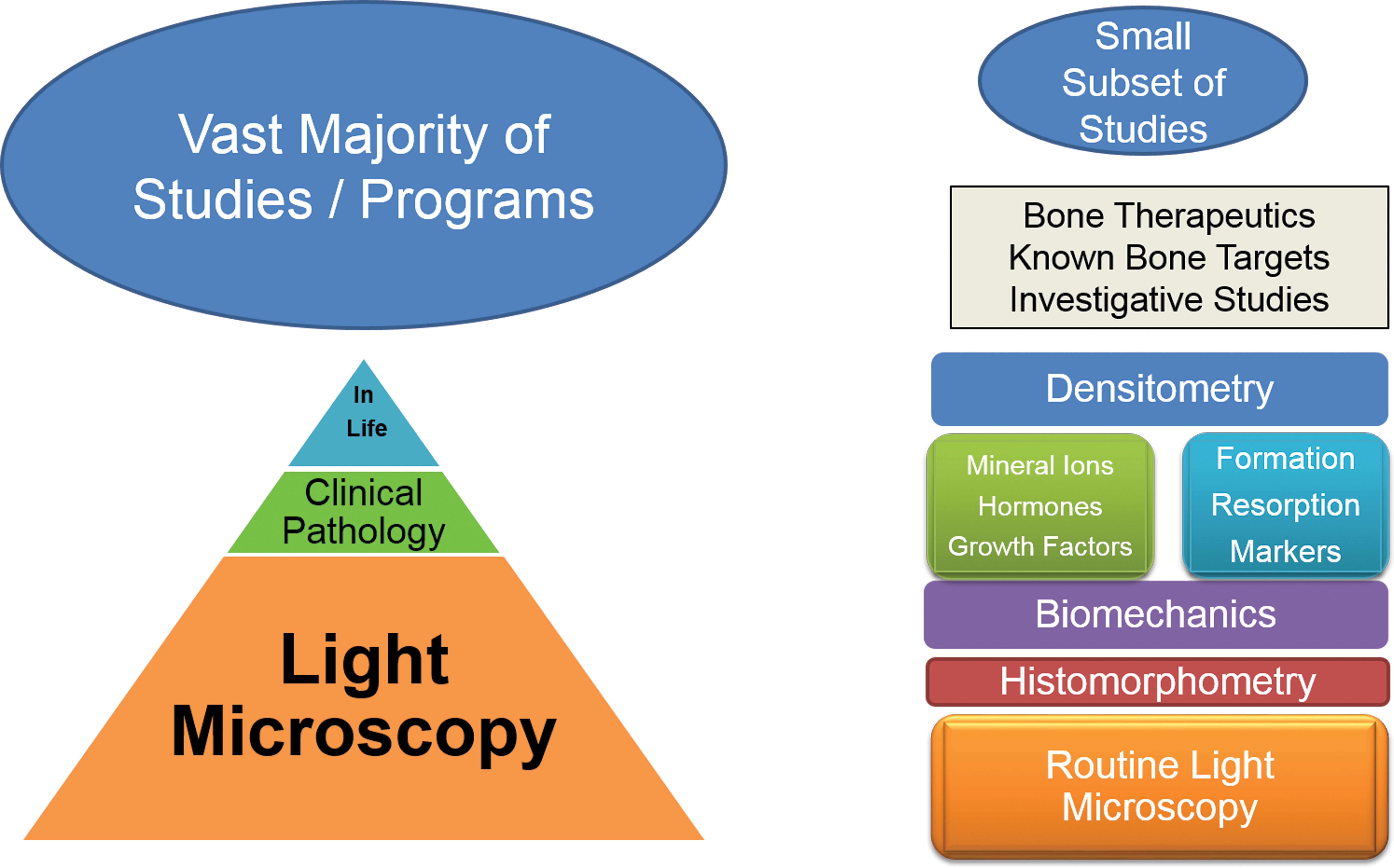
This pictorial was used in the presentation to describe the fact that in the vast majority of standard toxicity studies (left-hand side of diagram), light microscopy is the primary means of detecting skeletal alterations. It is relatively infrequent for live-phase findings to identify a clear skeletal effect and clinical pathology data typically only signal a potential skeletal effect if there are alterations in alkaline phosphatase activity or calcium and phosphorus concentrations. In contrast, there is a smaller, but important, subset of studies (right-hand side of diagram) in which an array of sensitive and quantitative bone end points can be used in conjunction with light microscopic examination to determine the effect of a compound on the skeleton.
In contrast to standard screening repeat-dose toxicity studies, there are occasions when a more in-depth assessment of the skeleton is warranted. Although routine histology can detect a broad array of skeletal effects, it remains a relatively insensitive assessment of overall bone mass and, in general, does not provide information on temporal effects or potential mechanisms. For agents that are intended to be bone therapeutic agents, it is often useful to incorporate quantitative bone analyses to characterize the pharmacodynamic effects of a test article and ensure there are no detrimental effects on bone. For agents that are not intended to be bone therapeutics, but for which either prior class experiences or theoretical skeletal effects are suggested by literature on target biology, addition of quantitative end points may be warranted on a case-by-case basis. In addition, detailed bone evaluations are key components of investigative studies conducted to follow up on bone findings in prior toxicity studies. Across all of these types of studies, the toxicologist and pathologist must carefully consider their experimental objectives and select end points based on those specific objectives. In most cases, it will not be necessary and/or practical to deploy the full range of bone analyses.
In both standard and investigative toxicity studies, there is not a standardized approach to the selection of bones for microscopic examination. This is in contrast to histomorphometry where there is significant standardization of both the specimen evaluated and the end points that are measured (Dempster et al. 2013; Erben and Glosmann 2012). In the majority of repeat-dose toxicity study protocols, bone specimens include a section of sternum as well as a section of a long bone. In many laboratories, the long bone selected is a section through the stifle joint to include distal femur, knee (stifle) joint, and proximal tibia. In other laboratories, the knee joint is disarticulated and section of a single bone (e.g., distal femur) is provided for examination. Optimally, the sectioning approach should allow the study pathologist to assess major anatomic regions such as articular surface, epiphysis, physis, metaphysis, and diaphysis. One challenge that arises in the assessment of nonrodent studies (dog, nonhuman primate) is that in some trimming approaches little to no diaphysis is provided, meaning that assessment of diaphyseal cortical bone is not possible. In addition to standard approaches, pathologists should consider whether specialized trimming or bone collection is warranted based on study-specific objectives. For example, frontal sections of tibia are highly useful for critical evaluations of the physis and primary and secondary spongiosa, as they provide consistent orientation of these structures. In other cases, it may be warranted to trim in a wider array of bones to allow a more systematic survey of the skeleton or to alter specimen collection to ensure any gross and/or radiologic abnormalities are assessed. When evaluating histologic specimens to assess abnormalities identified during macroscopic and/or radiologic examination, it is important to sample the same site from an appropriate number of control animals to allow accurate comparisons to be made.
INHAND Skeletal Nomenclature
The INHAND project (INHAND for Lesions in Rats and Mice, www.toxpath.org/inhand.asp) is a joint initiative of the Societies of Toxicologic Pathology (STPs) from Europe, Great Britain, Japan, and North America to develop an internationally accepted nomenclature for nonproliferative and proliferative lesions in rodents. A working group composed of pathologists from the various societies came together to develop a standardized nomenclature for classifying lesions in the skeletal system (bones, joints, and teeth) of laboratory rodents. This nomenclature and associated diagnostic criteria were published (Fossey et al. 2016) and are available in a database format at the goRENI website (http://www.goreni.org/). The INHAND scheme reflects feedback received from broad STP membership during an open comment period. The focus of the current article is on bone, rather than on joint and tooth abnormalities. The goal of this symposium session was not to review each entity within the INHAND scheme but rather to provide an overview of the approach taken, an outline of the specific diagnosis, and a few representative case examples.
In developing a nomenclature scheme, the INHAND project in general, and the bone section specifically, adopted a generally descriptive, rather than diagnostic approach. In contrast to diagnostic pathology which focuses on a single or small group of cases that present with a clinical syndrome and associated lesions, toxicologic pathology requires the study pathologist to record morphologic alterations across a study population in a consistent and systematic manner so that treatment-related effects can be identified and characterized. In many cases, a test article may induce a morphologic change; however, little is known about the pathogenesis, functional impact, or clinical relevance of the change. As such, it is often more appropriate to use terminology that describes the lesion observed at light microscopy but does not imply a specific pathogenesis or disease process. The presentation used an example of a test article that causes a diffuse increase in trabecular and cortical bone as part of the mechanism of action. In a diagnostic pathology approach, a variety of diagnoses such as hyperostosis or osteopetrosis might seem appropriate. One shortcoming of this approach is that some readers will infer that the use of terminology such as osteopetrosis implies a similar mechanism (genetic dysfunction of osteoclasts) and an associated functional impact (increased fracture risk). In the more descriptive approach recommended by INHAND, a diagnosis of “increased bone” with appropriate modifiers to indicate the bone compartment or region affected provides an objective description of what was observed at light microscopy. An additional consideration is that the terminology used in the pathology tables needs to be augmented and expanded upon in the pathology narrative report. It is in the pathology narrative that the study pathologist can provide additional description of the morphologic features and context as to potential significance.
The descriptive approach detailed above is most relevant to the nonproliferative diagnoses. To conceptualize the types of diagnoses, Table 1 is the author’s separation of those diagnoses that are discrete qualitative abnormalities and those that are semiquantitative and represent a change in the relative size or abundance of either a specific structure, matrix, or cell type. For a complete description of the diagnostic criteria, differential diagnoses, and representative figures, please refer to Fossey et al. (2016) or the goRENI website (http://www.goreni.org/). A few representative examples were discussed in the session and are summarized below. An example of a discrete qualitative lesion is that of bone necrosis. As shown in Figure 2, this entity is characterized by a large number of empty osteocyte lacunae, associated marrow changes (in this example, increased stromal cells and production of reactive new bone). As an example of a semiquantitative change, examples of increased trabecular bone were reviewed during the session. In one example, the increase in bone was present in a routine section of distal femur and in the other example an increase in bone was noted within the bones of the nasal turbinate. In both cases, the INHAND diagnosis of “increased bone, trabeculae” or “increased bone, trabeculae and cortex” was used to record these observations. One entity that provided challenges for the INHAND group in the development of the nomenclature scheme were those situations where routine, decalcified, and hematoxylin and eosin–stained sections demonstrate an increased amount of pale staining matrix. In many cases, this material is thought to represent poorly mineralized osteoid; however, the use of undecalcified sections and special stains (i.e., modified trichrome) is needed to determine the mineralization state of the matrix. Despite the fact that the INHAND nomenclature emphasizes those diagnoses that can be derived from routine sections, the INHAND group included as a diagnostic option “increased osteoid” and indicated that in those situations a provisional diagnosis can be made on the basis of routine decalcified sections; however, in follow-up studies, confirmation with undecalcified sections is warranted. Figure 3 demonstrates an example of increased amounts of unmineralized osteoid in comparing the appearance on a routine versus undecalcified section.
Overview of Classification Scheme of Nonproliferative Bone Lesions.

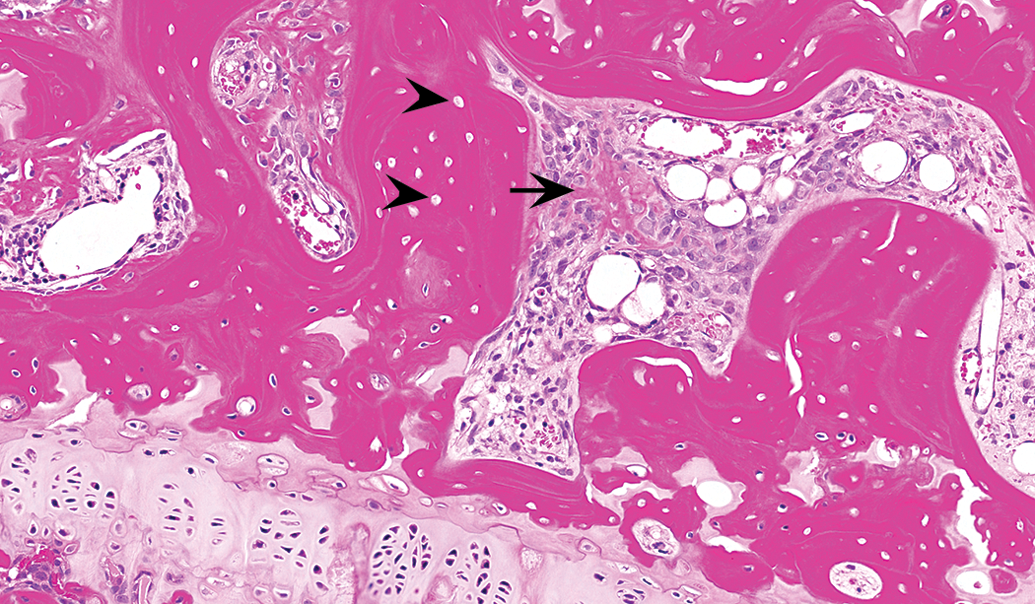
In this section of the epiphysis of the proximal femur of an approximately 15-week-old male Sprague-Dawley rat, bone necrosis is evidenced by empty osteocyte lacunae (arrowheads). In the adjacent marrow space, there is an associated stromal response that includes deposition of woven (“reactive”) bone formation (arrow). Source: Reproduced with permission of the Japanese Society of Toxicologic Pathology from Fossey et al. (2016).
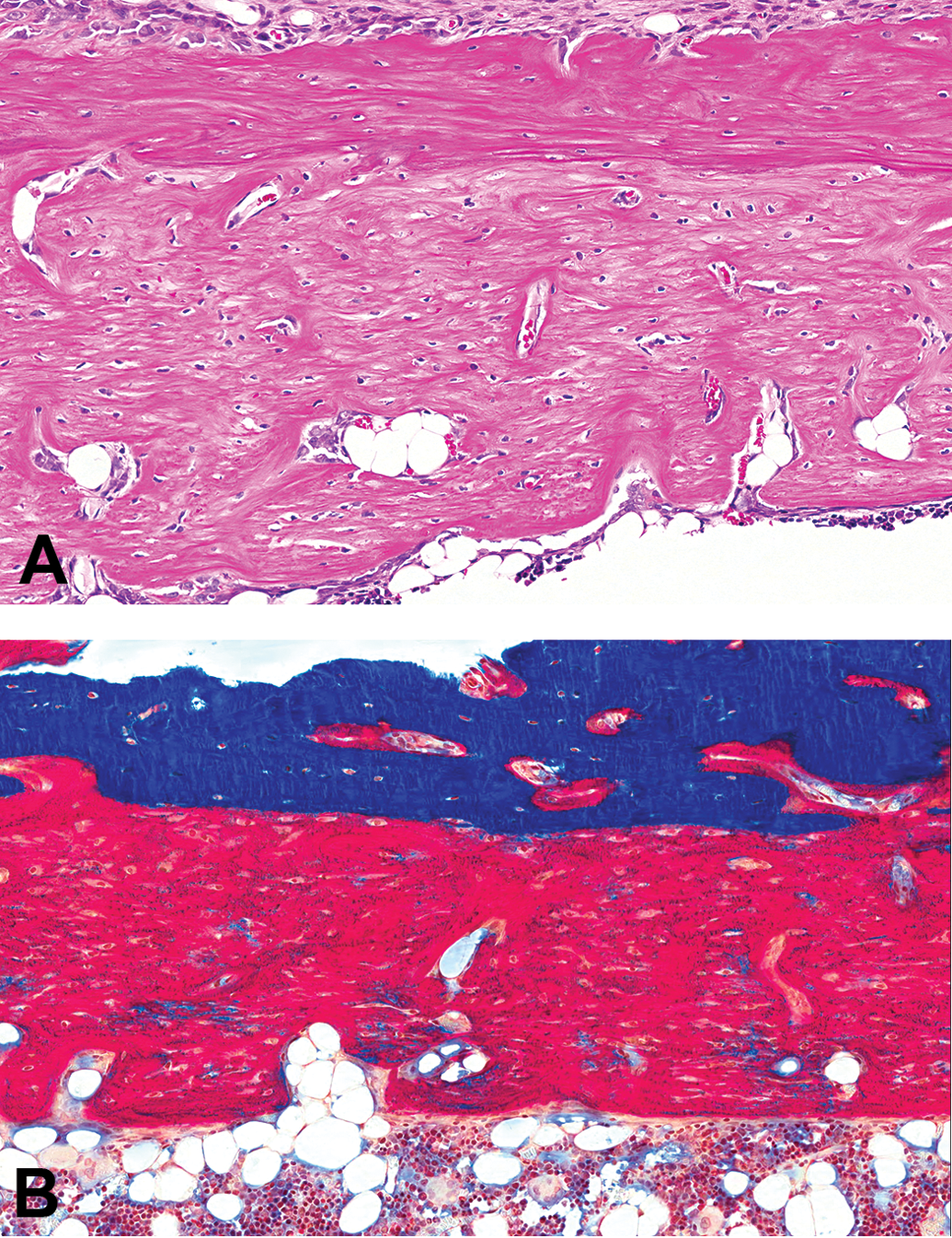
In these sections of tibial cortex of a Sprague-Dawley rat, increased amounts of osteoid are present. The increase is evident as a thick plaque of pale eosinophilic woven bone in the hematoxylin and eosin–stained section of decalcified bone on the top and as bright red matrix in the section of undecalcified bone stained with modified trichrome on the bottom. Source: Reproduced with permission of the Japanese Society of Toxicologic Pathology from Fossey et al. (2016).
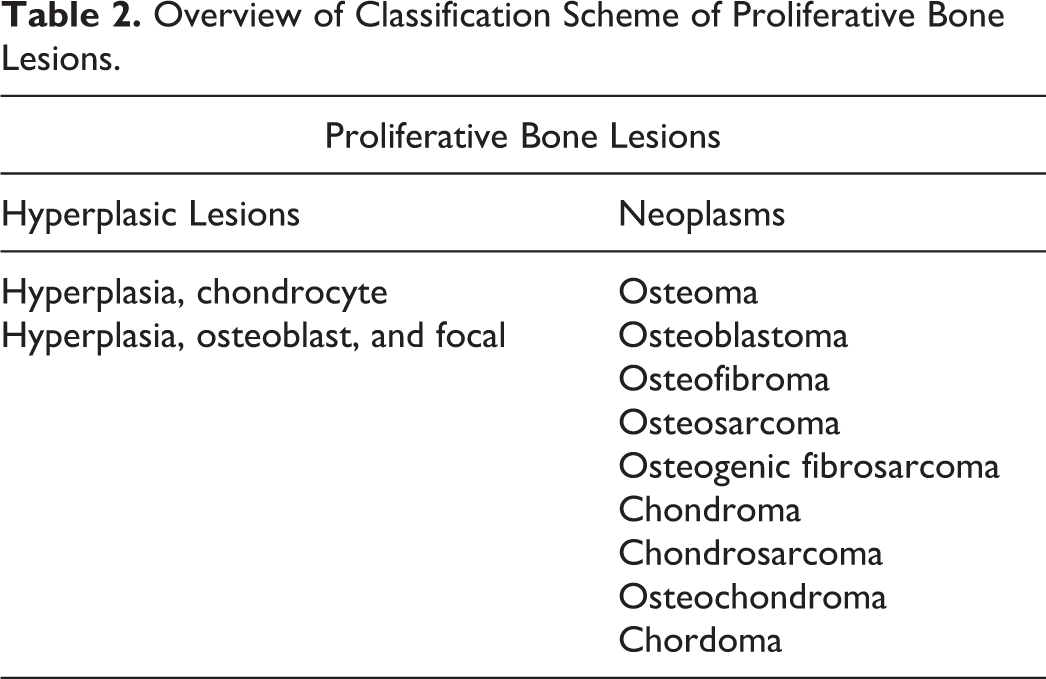
The bone proliferative lesions in the INHAND scheme are listed in Table 2. There were no substantial changes to these diagnoses from prior published diagnostic schemes. It was noted that in routine use, it is not necessary nor warranted to subclassify osteosarcoma into various subtypes as is done in human and veterinary diagnostic pathology. If there is a study-specific need to subclassify osteosarcomas, the study pathologist should refer to and reference the diagnostic scheme they are employing to subclassify the tumors in that study.
Overview of Classification Scheme of Proliferative Bone Lesions.
The presentation also acknowledged that there are situations where, particularly for novel lesions induced by test articles, morphologically complex lesions arise for which the existing nomenclature does not appear to adequately characterize the change. An example was shared in which a test article induced a complex lesion in the sternum of rats following 4 daily oral doses. The lesion included both a relative increase in the amount of bone and a notable increase in osteoblast number, increases in bone marrow stromal cells, and a mild increase in the amount of lightly eosinophilic matrix on trabecular surfaces. The INHAND scheme was consulted to assess options to diagnosis this particular change. It was determined that the increased amount of trabecular bone was readily recorded with the “increased bone, trabeculae” diagnosis; however, there was less certainty regarding a single term to describe the cellular elements of the lesion. Consultation within a subgroup of the INHAND working group yielded various approaches including “osteoblast hyperplasia,” “increased osteoblasts,” and “increased osteoblastic surface.” This example highlighted that even highly experienced bone pathologists might select different terminology. For each of the diagnostic options, there were clear limitations. For example, as per the current INHAND scheme, osteoblast hyperplasia is reserved for a focal proliferative change that is thought to be part of a neoplastic continuum. The descriptive terms of increased osteoblasts or increased osteoblastic surface did not seem to capture the extent of the cellular and matrix changes noted. One of the points raised by this case is the critical importance of the pathology narrative. While the pathologists consulted would have used different primary diagnostic terms, their description of the key features was nearly identical and suggested that the pathology narrative reports provide would have conveyed the same message, despite the fact that they used different primary diagnostic terms.
Bone Changes and Impact on Adversity Assessment
Similar to other organ systems, toxicologists and pathologists often face the challenge of assigning adverse effect levels in toxicity studies in which bone findings occur. As such, the presentation reviewed some basic principles of assigning adverse effect levels and illustrated the challenges using a pair of case studies. There are a number of published review papers and commentaries that have attempted to define adverse effects. In addition, the STP published best practices related to some general principles for determining and communicating adverse effect levels within nonclinical safety studies (Kerlin et al. 2016). The presentation did not attempt to review all aspects of the article; however, there were 2 concepts in the article that are particularly useful when considering skeletal effects. The 2 concepts are related and are stated in the Kerlin et al.’s paper as follows: Adversity is a term indicating “harm” to the test animal within the constraints of the study design (dose, duration, etc.; p. 153). Neither the therapeutic indication nor the patient population should influence adversity decisions in the test species (p. 154).
An additional case challenge scenario that was reviewed involved a rat toxicity study where effects at the high dose included an increase in distal tibial fractures accompanied by histologic changes characterized by markedly increased amounts of lightly eosinophilic osteoid on endosteal surfaces of the bones that were examined (tibia and sternum). At the low dose, there were no fractures and the amount of lightly staining osteoid was only marginally greater than that seen in the concurrent controls in 2 of the 10 animals. Specialized bone end points (e.g., biomechanical testing) and special stains/preparations were not available. Again, 2 schools of thought were presented. One is that all dose levels would be adverse because at its most profound manifestation, the histologic change was associated with altered biomechanical competence (i.e., fracture) and therefore any change from control with respect to altered bone matrix, no matter how small, would be considered adverse. A contrasting interpretation would be that at levels where the histologic change is barely detectable, the change would not be considered adverse but would be adverse at higher incidence/severity grades that approach those which resulted in increased fracture rates. In the study from which this case scenario was derived, all dose levels were considered adverse; however, in this author’s perspective, either approach presented could have been taken as long as the study authors clearly articulated their rationale in assigning the adverse effect level. With the context provided by such a report, both sponsors and regulators can then perform the overall safety assessment and consider appropriate risk management steps.
Concluding Remarks
In the majority of toxicity studies, it is important for the study pathologist to understand that histologic examination of bone is one of the key methods to detect either unexpected pharmacologic effects or overt toxic effects on bone. It is also important for toxicologic pathologists to be aware that our standard histologic assessment of bone is relatively insensitive to detect changes in bone geometry and mass and in no way assesses bone strength. As such, it is useful to have an understanding of when and how to employ quantitative bone end points, tailored to the specific causes for concern. When recording histologic observations, pathologists are strongly encouraged to adopt the terminology provided by the INHAND system; however, it is recognized that novel xenobiotic-induced changes may not fit well within the existing nomenclature scheme. In these cases, the pathologist should adapt or modify the terminology as appropriate and then provide a full and complete morphologic description in the study narrative report. Finally, just as with other organ systems and tissues, determining when an effect in a toxicity study is “adverse” can be quite challenging in the skeletal system. It is recommended to keep adversity decisions focused on the species being studied and to keep in mind that both primary and secondary effects (including exaggerated pharmacologic effect) need to be considered in the overall adversity assessment. Study teams, sponsors, and regulatory reviewers all need to keep in mind that some findings, which may not be adverse in the context of a toxicity study, may still represent an important pharmacologic effect and be communicated clearly in study reports.
Footnotes
Acknowledgment
The author is grateful for the assistance of the INHAND Skeletal Working Group, which developed standardized nomenclature for the toxicologic pathology evaluation of bone and provided advice and consultation on this presentation and subsequent manuscript.
Author Contribution
The author (JV) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; critically revising the manuscript; gave final approval; and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
