Abstract
Hesperetin glucosides such as hesperidin and hesperetin-7-glucoside are abundantly present in citrus fruits and have various pharmacological properties. However, the potential toxicity of hesperetin glucosides remains unclear. An initial assessment of the safety of hesperetin-7-glucoside–β-cyclodextrin inclusion complex (HPTGCD) as a functional food ingredient was undertaken to assess toxicity and mutagenic potential. A bacterial reverse mutation assay (Ames test) using Salmonella typhimurium (strains TA98, TA1535, TA100, and TA1537) and Escherichia coli (strain WP2 uvrA) with HPTGCD (up to 5000 µg/plate) in the absence and presence of metabolic activation was negative. In a single oral (gavage) toxicity study in male and female rats, HPTGCD at dose up to 2000 mg/kg did not produce mortality nor clinical signs of toxicity or change in body weight. In a subchronic oral (dietary admix) toxicity study in rats receiving 0, 1.5, 3, and 5% HPTGCD for 13 weeks, no adverse effects were noted and the no-observed-adverse-effect level (NOAEL) was 5% in the diet (equivalent to 3267.7 mg/kg/day for males and to 3652.4 mg/kg/day for females). These results provide initial evidence of the safety of HPTGCD.
Keywords
Introduction
Hesperidin (HSP: hesperetin-7-rutinoside; rutinoside includes glucose and rhamnose: CAS:520-26-3 [Figure 1]), an insoluble flavonoid, is found abundantly in citrus fruits1,2 and as a functional food component has been associated with anti-inflammatory,
3
and anti-obesity4,5 activity, and improving endothelial (vasomotor) function.
6
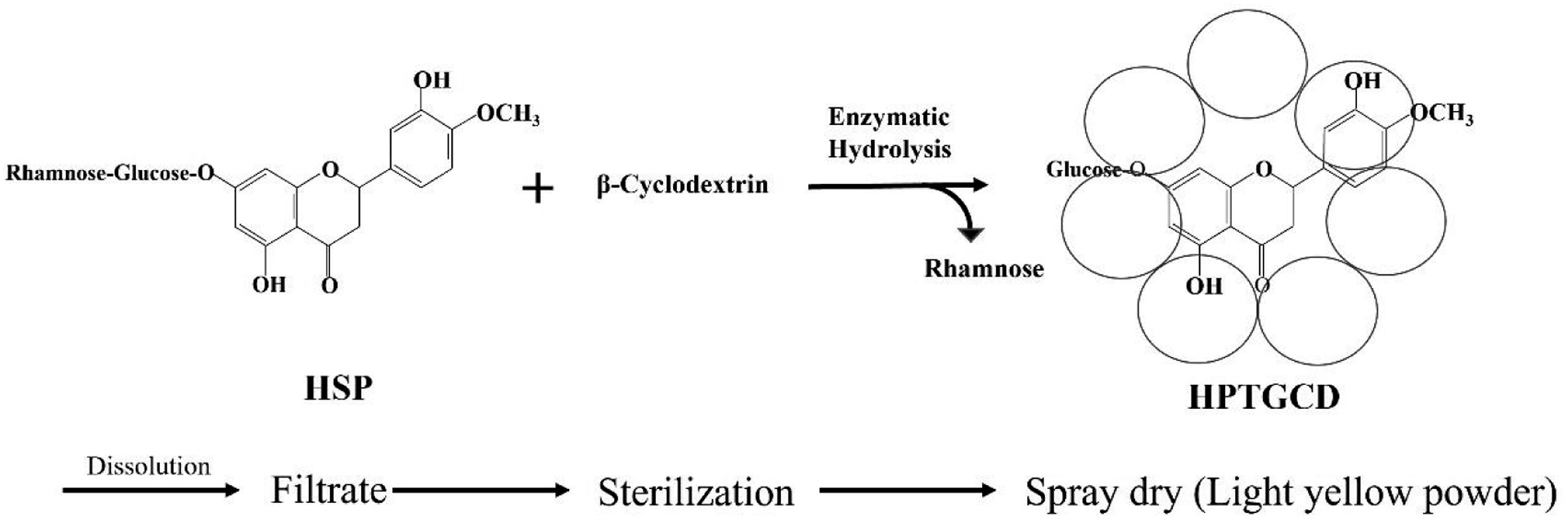
Industrial production method of hesperetin-7-glucoside–β-cyclodextrin inclusion complex (HPTGCD). HPTGCD was produced using a patented method to treat hesperidin (Citrus aurantium extract [hesperidin (HSP) purity: 90.3%] with naringinase [160 units/g, Amano Enzyme, Inc., Nagoya, Japan] and β-cyclodextrin [Cyclo Chem Co., Ltd., Kobe, Japan]) with the following steps
24
: (1) HSP (33 mM) and β-cyclodextrin (βCD, 50 mM) were mixed with water in a tank, (2) The temperature and pH of the solution were adjusted to 73°C and pH4.5 using NaOH and H2SO4., (3) Naringinase digestion (30 units/DSN g) was conducted for 24 h at 73°C, pH 4.5, (4) After confirming a > 96% conversion rate to HPTG*, the solution including the product was filtrated and turned into powder using a spray dryer machine after sterilization. *The conversion rate (%) was performed by nearly 98% using high-performance liquid chromatography (HPLC) analysis.
30
Hesperetin (HPT: CAS 69097-99-0) glucosides, such as HSP and glucosyl HSP (GH, CAS 161713-86-6, FDA GRN 000901) have been investigated in several toxicological studies. Hardigree et al
7
reported that HPT and HSP with nonplanar structure were non-mutagenic in an Ames test, and Ishidate et al
8
showed methyl HSP was not mutagenic in the Ames test. In addition, Ahmadi et al
9
observed an increase in micronucleated polychromatic erythrocytes in male NMRI mice treated with 400 mg/kg/day of HSP for 5 days compared to the control, which was not statistically significant. Besides, Matsumoto et al
10
reported no clastogenic potential in mammalian cells in an in vitro chromosomal aberration assay at 5000 μg/mL GH and no increase in micronuclei in male mice treated with up to 2000 mg/kg GH. Thus, the available data on HPT-related compound
Matsumoto et al
10
recently reported OECD-compliant toxicity studies of GH showing that a subchronic oral (dietary admix) toxicity study in rats showed no deaths and no treatment-related adverse effects at 0.45%, 1.5%, and 5.0% GH, resulted in a NOEL of 5.0% GH in the diet, which corresponds to 3084 mg/kg/day for male rats and 3428 mg/kg/day for female rats. Mayumi et al
15
performed a subchronic oral (dietary admix) toxicity study of methyl HSP in B6C3F mice and concluded that methyl HSP exerts no obvious toxic effects at a dose as high as 5.0%. Kurata et al
16
performed a 96-weeks oral (dietary admix) carcinogenicity study in B6C3F1 mice with methyl HSP up to 5.0% (7500 mg/kg/day in males, and 8600 mg/kg/day in females) and reported no treatment-related findings of oncogenicity. Oral administration of HSP and GH demonstrated physiological effects such as improvement of high triglyceride levels, high
Cyclodextrins are used to improve the stability and water solubility of insoluble flavonoids. Several studies on the improved bioavailability of flavonoid inclusion complexes with cyclodextrins have been reported.26-28 Commercial production of a HPTG/β-cyclodextrin inclusion complex (HPTGCD) was achieved by reacting HSP with naringinase in the presence of cyclodextrins using a patented method29,30 (Figure 1). The HPTGCD solubility is > 1000-fold higher than HSPD and the area under the curve (AUC (0-24)) of the total plasma HPT concentration after oral administration with HPTGCD (1.05 g) was approximately 100 times higher than after treatment with hesperidin/dextrin (HSPD, 1.05 g) in healthy adult human males. 31 Physical parameters (pulse, blood pressure, and heart rate) did not significantly change from before to after ingestion of HPTGCD and control foods. Changes in blood biochemical and hematological markers, and urine examination results before and after ingestion of HPTGCD and control foods also did not appear to be of any clinical significance. 31 Because of the higher bioavailability after HPTGCD consumption in humans, greater physiological responses would be expected relative to HSP; hence safety studies with HPTGCD should be conducted prior to support its safe use in functional foods and alternative medicines. Therefore, we performed in vitro mutagenicity tests using bacterial reverse mutation assays, and acute and repeated-dose toxicity studies with HPTGCD to provide an initial assessment of the toxicity profile and establish a NOAEL.
Materials and Methods
All the studies were conducted in compliance with the Organization for Economic Co-operation and Development (OECD) Principles of Good Laboratory Practice (GLP)
Test Substance
Hesperetin-7-glucoside–β-cyclodextrin inclusion complex (SUNACTIVE HCD: Lot 91122; HPTG content: 15%, β-cyclodextrin [βCD; CAS 7585-39-9, 1135 kDa] content: 60%, saccharides et al content: 22.6%, water content: 2.4% for mutagenicity tests and for the acute oral toxicity study; Lot 00331; HPTG content: 14.2‐14.5%, βCD content: 57.5%, saccharides et al content: 24%, water: 3.8% content for the subchronic dietary toxicity study; manufactured by Taiyo Kagaku, Mie, Japan (Figure 1). The test dietary feeds were composed of HPTGCD (lots 91122 and 00331) and were produced on an industrial scale for the test dietary feeds using our patented method).
30
Hesperetin-7-glucoside–β-cyclodextrin inclusion complex was suspended in water (Japanese Pharmacopoeia, Otsuka Pharmaceutical Factory, Inc., Tokushima, Japan) for use in Ames tests and acute toxicity study. Hesperetin-7-glucoside–β-cyclodextrin inclusion complex and powdered basal diet CR-LPF (γ-Irradiated, Oriental Yeast Co., Ltd., Kanagawa, Japan) were mixed by hand to prepare the test dietary feeds for the subchronic toxicity study. The test/dose formulations and test dietary feeds were analyzed for the HPTGCD concentrations using high-performance liquid chromatography under GLP conditions. The proportions of the nominal concentration of all test/dose formulations and test dietary feeds were 101.6% to 104.2% in the mutagenicity study, 97.5% to 101.8% in the acute toxicity test, and 97.0% to 100.0% in the subchronic toxicity test. The respective coefficients of variation (CVs) were 0.2% to 0.3%, 0.2% to 0.6%, and 0.3% to 3.4%. The concentration and homogeneity in all tests met the acceptance criteria (proportion of the nominal concentration: within 100% ± 10%, CV: 10% or less). Furthermore, we verified that the test/dose formulations at 0.01 and 200 mg/mL were stable and homogeneous at 1°C to 30°C for 24 h after storage in a refrigerator for 4 d, and that the test dietary feeds of lots 91122 and 00331 at 1.5% and 5.0%, respectively, were stable and homogeneous for 15 d at 1 to 30°C. The prepared test/dose formulations were placed in airtight light-resistant hermetic containers, stored in a refrigerator at 1 to 10°C
Mutagenicity Study
Chemicals and Reagents
Water was used as the negative control (vehicle only). The mutagens 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide (AF-2), sodium azide (SAZ), 2-aminoanthracene (2AA), and benzo
Bacterial Reverse Mutation Assay (Ames Test)
The Salmonella typhimurium tester strains TA100 and TA1535, and Escherichia coli WP2 uvrA were used to detect base-pair substitution events, S typhimurium strains TA98 and TA1537 were used to detect frameshift-type reversion events.34-41
Aliquots (0.1 mL) of the test formulation, negative (water), and positive control solution were separately placed in sterilized test tubes. Then, 0.5 mL of the S9 mix or 0.5 mL of phosphate buffer, and 0.1 mL of the bacterial culture suspension were added. The concentrations of the test substance, and the positive control substances with or without S9 metabolic activation are listed in Table S1. The S9 mix was prepared by mixing the S9 microsomal fraction extracted from phenobarbital/benzoflavone-treated male Sprague–Dawley (SD) rats (S9 for Ames Test, BoZo Research Center Inc., Tokyo, Japan) and the cofactor (Cofactor FA for Ames Test, BoZo Research Center Inc.) in a 9:1 ratio. The mixtures were incubated for 20 minutes at 37°C with continuous shaking. After incubation, 2 mL molten top agar was added to each test tube, mixed, uniformly overlaid onto the surface of a minimal glucose agar plate, and allowed to solidify.
Duplicate tests were conducted using triplicate culture plates for each test group, according to the OECD 471 test guideline. After incubation for 48 hours at 37°C, the number of revertant colonies per plate was counted using an automatic colony counter (Colony Analyzer CA-11D, System Science Co., Ltd., Tokyo, Japan). Solubility was visually assessed by the presence or absence of precipitated test substances. The presence/absence of bacterial growth inhibition was visually evaluated using a stereomicroscope (SZ61, Olympus, Tokyo, Japan).
Acute Toxicity Test
Administration
Twenty-five male and twenty-five female five-week-old SD rats (SPF; Crl:CD (SD)) were purchased from Charles River Laboratories Japan Inc. (Kanagawa, Japan) and 20 healthy rats of each sex were assigned to 4 experimental groups (n = 5) to ensure uniformity of the mean body weight of the group. The animals were fasted for approximately 16 hours before HPTGCD administration. The next day, the rats were administered either 500, 1,000, or 2000 mg/kg HPTGCD, or vehicle by oral gavage using a flexible stomach tube at a dose volume of 10 mL/kg body weight. The food was resupplied 4 hours after administration.
Clinical Observations, Body Weights, and Anatomic Pathology: Necropsy
All rats were visually observed for mortality, morbidity, and clinical signs on the day of administration (at 5, 15, and 30 min, and 1, 2, 4, and 6 h after administration) and once daily for the next 14 days after administration. Body weights were measured in the morning on the day of dosing and on days 1, 2, 3, 7, 10, and 14.
On day 14 post-administration, all rats were euthanized and complete necropsies were performed.
Subchronic Toxicity Test
Forty-eight male and forty-eight female four-week-old SD rats (SPF; Crl:CD (SD)) were purchased from Charles River Laboratories Japan Inc. (Kanagawa, Japan) and forty healthy rats of each sex were assigned to 4 experimental groups (n = 10) by stratified randomization procedure to ensure the uniform mean body weight of the group. During the dosing period, rats in the test groups were given the test dietary feeds and those in the control group were given powdered basal CR-LPF diet. Three doses of HPTGCD were used (1.5%, 3.0%, and 5.0%) based on the results of a 2-weeks dietary toxicity study of HPTGCD conducted under non-GLP conditions: In that study, no deaths occurred, nor abnormal clinical signs, or changes in, body weight, food consumption, hematological parameters, blood chemistry indices, organ weights of the liver or kidney, gross pathology, or histopathology of the liver or kidney at 1.5%, 3.0%, and 5.0% HPTGCD in either sex. The NOAEL of HPTGCD was 5.0% in the diet; therefore, a 5.0% dietary admixture was estimated to be well tolerated without toxicity for a subchronic toxicity test.
Clinical Observations
All rats were visually observed for mortality, morbidity, and clinical signs twice daily on weekdays and once daily on weekends and national holidays. Detailed clinical observations were made once before the dosing period and then once a week during the dosing period.
Behavioral Tests, Grip Strength, and Motor Activity
At week 12 of test substance administration, all rats were subjected to behavioral tests, grip strength measurements, and motor activity tests included in a functional observational battery. The behavioral tests were performed by blinded observers and consisted of auditory response, approach response, pain response, pupillary reflex, aerial righting reflex, and landing foot splay. Grip strength measurements of the forelimbs and hind limbs were taken using a CPU gauge Model RX-5 (Aikoh Engineering Co., Ltd., Osaka, Japan). For motor activity assessments, the number of ambulations for 3 minutes in a circular open field (1000 mm in diameter, 300 mm in height, and 25 sections) was visually counted.
Body Weights, Food Intake, and Water Consumption
Body weights were recorded on the first day of administration, and once per week thereafter. Food intake was recorded once per week. Water consumption was recorded once during urinalysis. In addition, HPTGCD intake (mg/kg body weight/day) was calculated from the diet admixture consumption and body weight.
Ophthalmological Evaluation
An ophthalmic evaluation was performed before administration and in the last study week for all rats. A mydriatic agent (Mydrin-P ophthalmic solution, Santen Pharmaceutical Co., Ltd., Osaka, Japan) was applied to the right and left eyes to dilate the pupil, and the anterior portion, optic media, and fundus oculi, were examined using a binocular indirect ophthalmoscope (Omega 200, HEINE Optotechnik GmbH & Co. KG, Bavaria, Germany).
Urinalysis
Urinalysis was performed in the last week of the study period using a metabolic cage with a tray attached for urine collection. Urine samples were collected for 4 hours under fasting conditions with free access to drinking water. Then, urine samples were collected for 20 hours with free access to food and water. The total urine volume (24 h) was calculated using collection samples of 0-4 hours and 4-20 hours. Urinalysis parameters were analyzed using the first (0-4 h) urine sample. Urine pH, protein, ketone bodies, glucose, occult blood, bilirubin, urobilinogen, and specific gravity were analyzed using Multistix SG test strips with a CLINITEK 500 Automatic Urinary Chemical Analyzer (Siemens Healthcare Diagnostics, Inc., PA, USA). The color of the urine was examined macroscopically, whereas urinary sediments, epithelial cells, blood cells, and crystals, were observed by microscopy (BX41, Olympus).
Hematology, Coagulation Tests, and Clinical Blood Biochemistry
At the time of necropsy, on the day after the end of the dosing period, the rats were fasted for 16-21 hours. Blood samples were taken from the abdominal aorta under anesthesia by isoflurane inhalation, for hematology, coagulation tests, and clinical biochemistry. Approximately 1 mL of whole blood was collected in blood collection tubes containing EDTA (BD Vacutainer, Becton, Dickinson, and Company, Tokyo, Japan) and processed immediately for hematological analysis. Routine hematological parameters were measured using an automatic hematology analyzer (Hematology System, ADVIA 2120i, Siemens Healthcare Diagnostics Inc., NY, USA), including red blood cell count, hemoglobin concentration (HGB), hematocrit (HCT), mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular HGB, red blood cell distribution width, reticulocyte count (RTC), platelet count (PLT), white blood cell count; and differential leukocyte count, including lymphocyte count (LYMP), neutrophil count (NEUT), eosinophil (EOS), basophil (BASO), monocyte (MONO), and large unstained cells.
Blood samples (approximately 4 mL) were collected in blood collection tubes containing heparin (Venoject II-Heparin Sodium (5 mL), Terumo Corp., Japan). They were centrifuged twice at 1600 x g for 10 minutes to separate the plasma for blood chemistry. Plasma samples were analyzed using an automated biochemistry analyzer (Automated Clinical Laboratory System TBA-120FR, Canon Medical Systems Corporation, Tochigi, Japan) for aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), alkaline phosphatase (ALP), total bile acid (TBA), total cholesterol (T-CHO), high-density lipoprotein, low-density lipoprotein (LDL), triglyceride (TG), phospholipid (PL), total bilirubin (T-BIL), glucose (GLU), blood urea nitrogen, urea, creatinine
Serum Thyroid Hormone Assay
At the time of blood collection for hematology, coagulation tests, and blood chemistry, additional blood samples (approximately 3 mL) were collected into blood collection tubes containing a blood coagulation accelerator (Venoject II-Autosep (4 mL), Terumo Corp.). Blood samples were centrifuged at 1600 x g for 10 minutes to obtain serum. Serum samples were stored at 80°C and serum levels of triiodothyronine (T3), thyroxine (T4), and thyroid-stimulating hormone (TSH) were determined with the RIA method using RIA-gnost®T3 Kit (Cisbio Bioassays, Budapest, Hungary), RIAgnost®T4 Kit (Cisbio Bioassays), and rTSH [125 I] RIA Kit (Izotop; Institute of Isotopes Co. Ltd.), respectively.
Necropsy and Organ Weights
After collecting blood samples, rats were euthanized by exsanguination via the abdominal aorta and subjected to necropsy by macroscopic observation of the external appearance of all internal organs/tissues.
Histopathology
Portions of the major organs/tissues (heart, kidney, lung, liver, ovary, and testis) and other organs (spleen, femur, seminal vesicle, prostate) were preserved in 10% neutral buffered formalin (NBF; pH 7.4) for histopathological inspections. The eyes were fixed in a medium containing 3.0% glutaraldehyde and 2.5% formalin in phosphate buffer, the testes and epididymides were fixed in Bouin’s solution and then preserved in NBF. After fixation, organs/tissues of the control and high-dose groups were embedded in paraffin, sectioned at 5 μm thickness, and stained with hematoxylin and eosin. The following tissues/organs were microscopically examined: adrenal glands, cecum, cerebellum, cerebrum, colon, duodenum, epididymides, esophagus, eyes, femoral bone marrow, femur, Harderian glands, heart, ileum, jejunum, kidneys, liver, lungs (including bronchi), mammary glands, medulla oblongata, mesenteric lymph nodes, ovaries, pancreas, parathyroid glands, Peyer’s patches (ileal), pituitary gland, pons, prostate, quadriceps femoris muscle, rectum, sciatic nerve, seminal vesicles (including coagulating gland), skin, spinal cord (cervical, thoracic, and lumbar), spleen, sternal bone marrow, sternum, stomach, sublingual glands, submandibular glands, submandibular lymph nodes, testes, thoracic aorta, thymus, thyroid glands, tongue, trachea, urinary bladder, uterus, and vagina.
Statistical Analysis
Statistical analyses were performed for the acute and subchronic toxicity studies. All the numerical data, body weight in the acute and subchronic toxicity tests and quantitative data of behavioral tests and urinalysis, grip strength, motor activity, food intake, water consumption, hematology, coagulation test, clinical blood biochemistry, serum thyroid hormone assay, and organ weights in the subchronic toxicity test, were analyzed using the Statistical Analysis System version 9.1.3 (SAS Institute Inc., Cary, NC, USA). Data are represented as means ± standard deviation. Variance was analyzed using Bartlett’s test at a significance level of 0.01. When the variances were homogeneous, Dunnett’s test was used to compare the test article/HPTGCD intake group and the control group. When the variances were heterogeneous, Steel’s test was performed to compare the test article/HPTGCD intake group and the control groups. Two-tailed Dunnett’s tests and Steel’s tests were performed at significance levels of 0.05 and 0.01, respectively.
Results
Bacterial Reverse Mutation Tests
The effects of HPTGCD (313-5000 μg/plate) on base-pair substitution (TA100, TA1535, and WP2 uvrA) and frameshifts (TA98 and TA1537) with or without metabolic activation with the S9 mix are shown in Table S1. The negative control (vehicle) and positive controls for all experiments confirmed the acceptance criteria. Exposure to varying doses of HPTGCD induced less than twice the number of revertant colonies per plate relative to the negative (vehicle) control group in all tester strains with and without metabolic activation with the S9 mix. No precipitation or growth inhibition was observed at any dose in any tester strain with or without metabolic activation. The results showed no mutagenic activity of HPTGCD up to a dose of 5000 μg/plate.
Acute Oral Toxicity Results
No deaths occurred, nor were there any abnormal clinical signs at any dose in either sex. Therefore, the minimum lethal dose was estimated to be greater than 2000 mg/kg. There were no significant differences in body weight between the vehicle and test article groups in either sex (Table S2). Furthermore, no abnormalities in the external appearance or in the organs/tissues of cranial, thoracic, or abdominal cavities were observed in any animal in either sex (data not shown).
Subchronic Toxicity Results
All animals survived the study duration, with no signs of toxicity or morbidity observed at any dose up to the maximum of 5.0% HPTGCD during the 13-weeks repeated oral (dietary admix) treatment. No abnormalities were noted in daily or weekly clinical observations, including home cage observations, in-the-hand observations, open-field observations, and behavioral test scores (Table S3). Moreover, there were no HPTGCD-related effects on motor activity or any other measured values (Table S4).
Body Weight and Food/Water Consumption
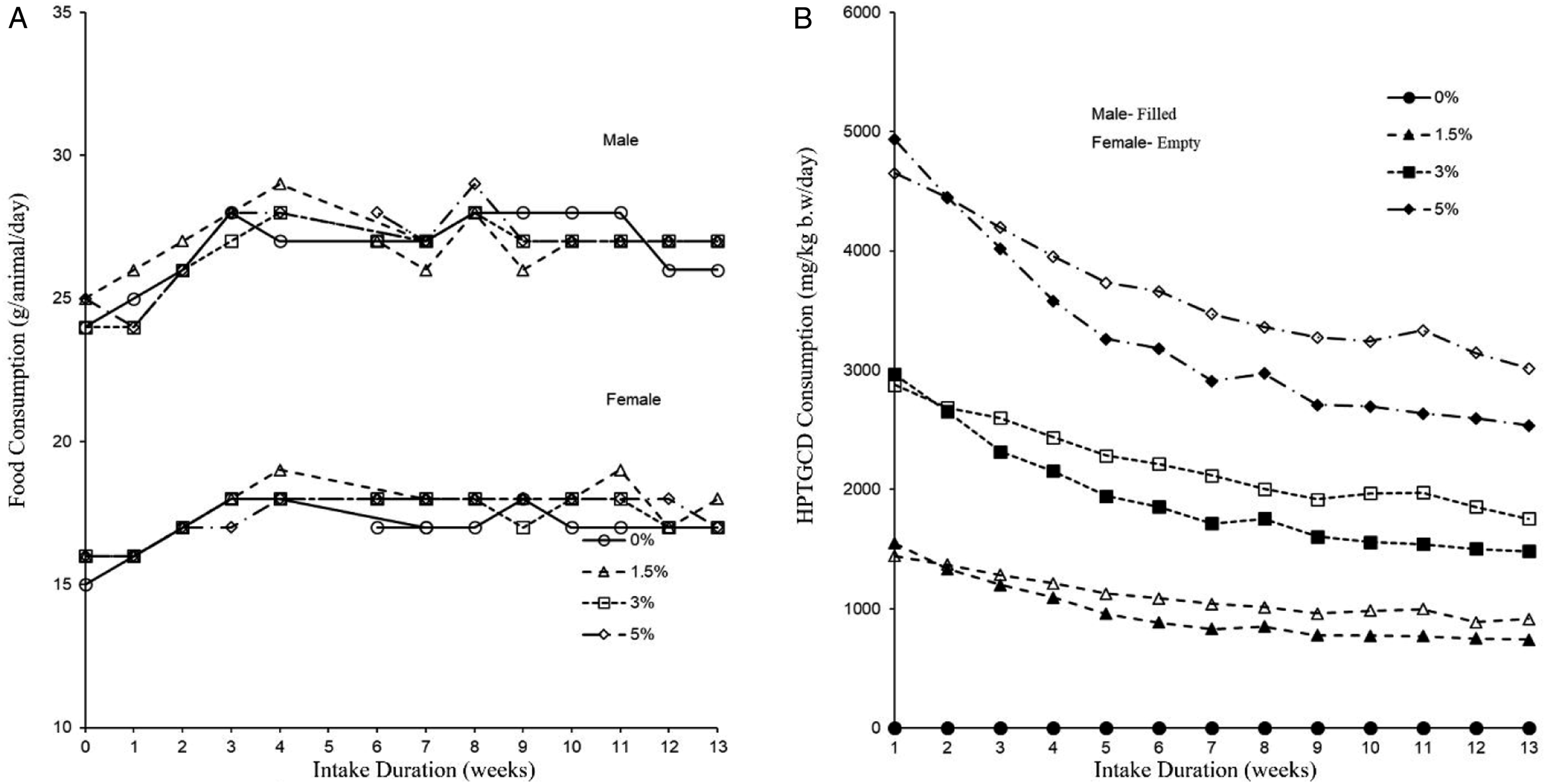
The average body weight, weight gain, and food consumption in the control and HPTGCD diet groups are presented in Table S5. No statistical differences (P > 0.05) in body weight were noted between the control and HPTGCD groups for male or female rats throughout the 13-weeks study period (Figure 2). Furthermore, no significant changes in food consumption were noted between the control and experimental groups throughout the 13-weeks study period (Figure 3(a); Table S5). The general mean daily HPTGCD intake in the 1.5%, 3.0%, and 5.0% dose groups was 962.0, 1926.9, and 3267.7 mg/kg body weight/d for male rats and 1101.0, 2206.5, and 3652.4 mg/kg body weight/d for female rats, respectively (Figure 3b; Table S5). Water intake was not significantly different between the control and HPTGCD groups (P > 0.05; Table S5). Changes in mean body weight in male and female Sprague–Dawley (SD) rats administered a HPTGCD-containing diet in the 13-week subchronic toxicity study. Each point represents the mean value of the animal groups fed with the indicated concentration (0%, 1.5%, 3.0%, and 5.0%) of HPTGCD in the diet. (a) Food consumption per animal/day in male and female SD rats administered an HPTGCD-containing diet in the 13-week subchronic toxicity study. (b) HPTGCD intake based on body weight of SD rats in each study group during the investigation period of 13 weeks for the subchronic toxicity study. Each point represents the mean intake per animal group fed with the respective dose (0%, 1.5%, 3.0%, and 5.0%) of HPTGCD in the daily diet.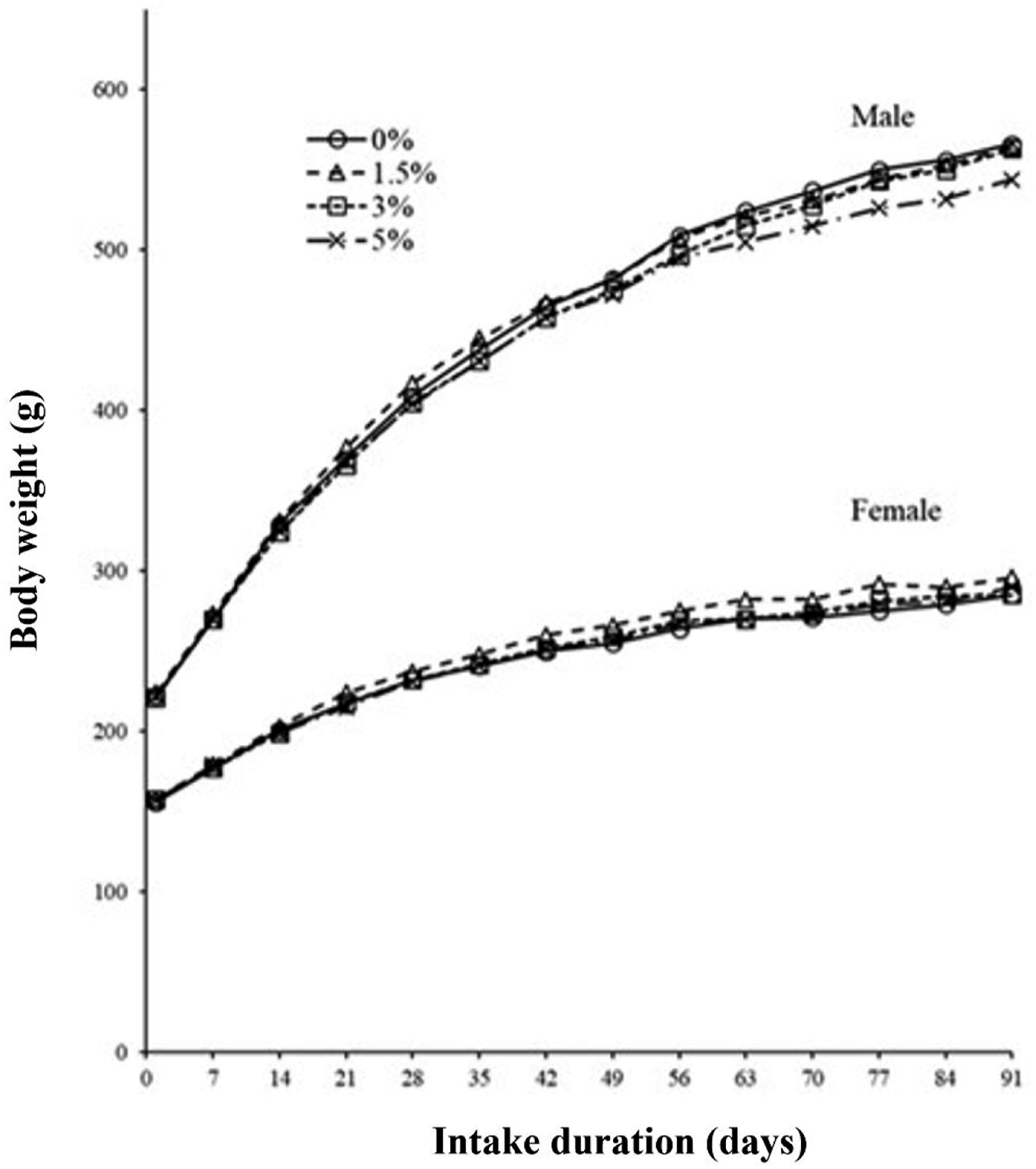
Ophthalmoscopy
Ophthalmoscopy results are summarized in Table S6. Unilateral retinal folds were observed in 1 rat in the 1.5% (male) group and unilateral and bilateral folds were observed in rats in the 5.0% dose group (male); unilateral retinal folds were observed in 1 female rat in the 3.0% dose group. However, retinal folds are occasionally observed in normal SD rats and did not have a causal relationship with HPTGCD.
Urinalysis
The urinalysis parameters of male and female SD rats are summarized in Table S7. There were no significant changes in any of the urinalysis parameters in the HPTGCD intake groups of either sex compared with that in the control group.
Hematological Parameters
Hematological Tests of Male and Female SD Rats Fed HPTGCD for 13 Weeks.
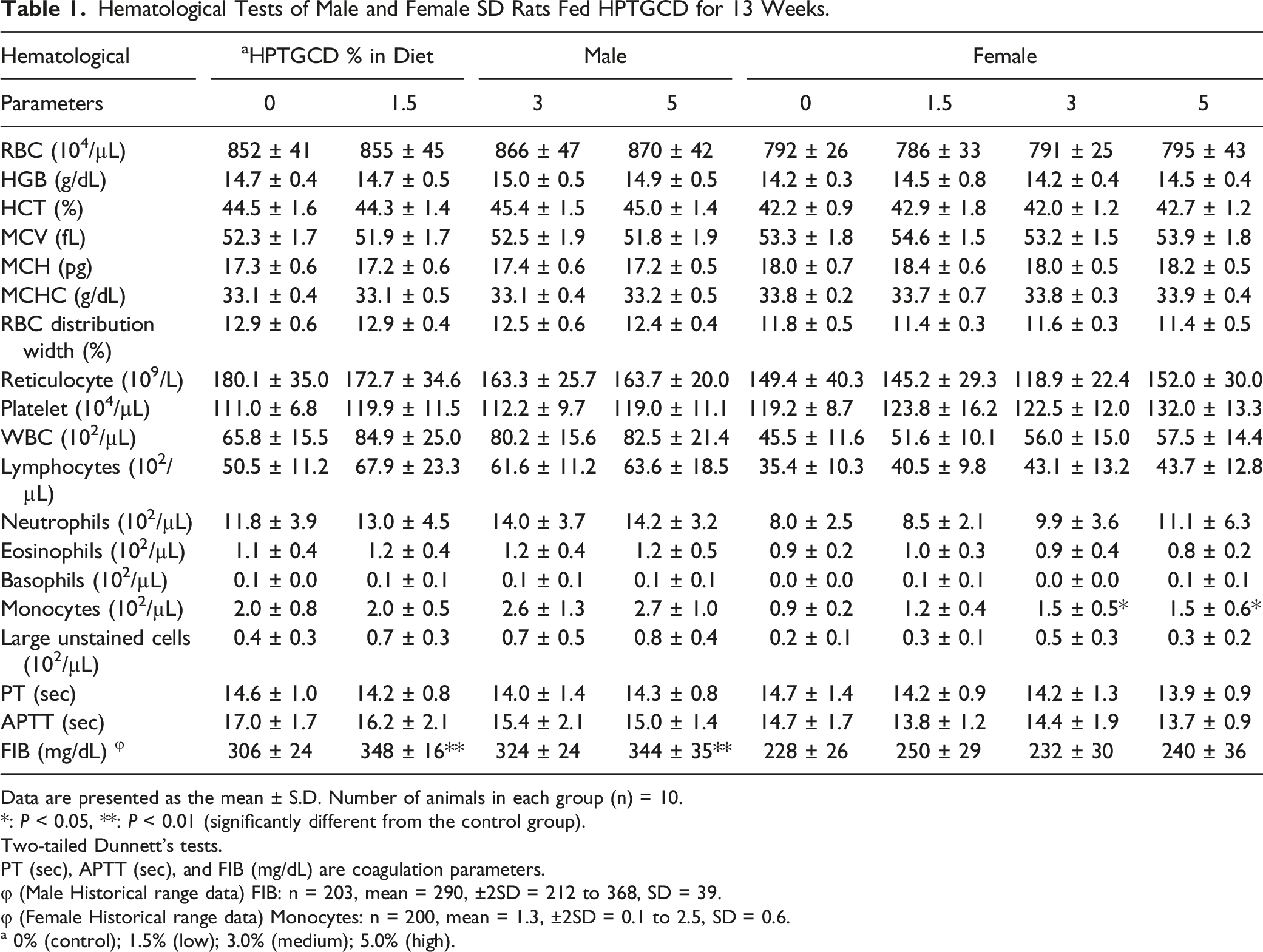
Data are presented as the mean ± S.D. Number of animals in each group (n) = 10.
*: P < 0.05, **: P < 0.01 (significantly different from the control group).
Two-tailed Dunnett’s tests.
PT (sec), APTT (sec), and FIB (mg/dL) are coagulation parameters.
φ (Male Historical range data) FIB: n = 203, mean = 290, ±2SD = 212 to 368, SD = 39.
φ (Female Historical range data) Monocytes: n = 200, mean = 1.3, ±2SD = 0.1 to 2.5, SD = 0.6.
a 0% (control); 1.5% (low); 3.0% (medium); 5.0% (high).
Clinical Biochemistry and Thyroid Hormone Parameters
Plasma Biochemical Tests of Male and Female SD Rats Fed HPTGCD for 13 Weeks.
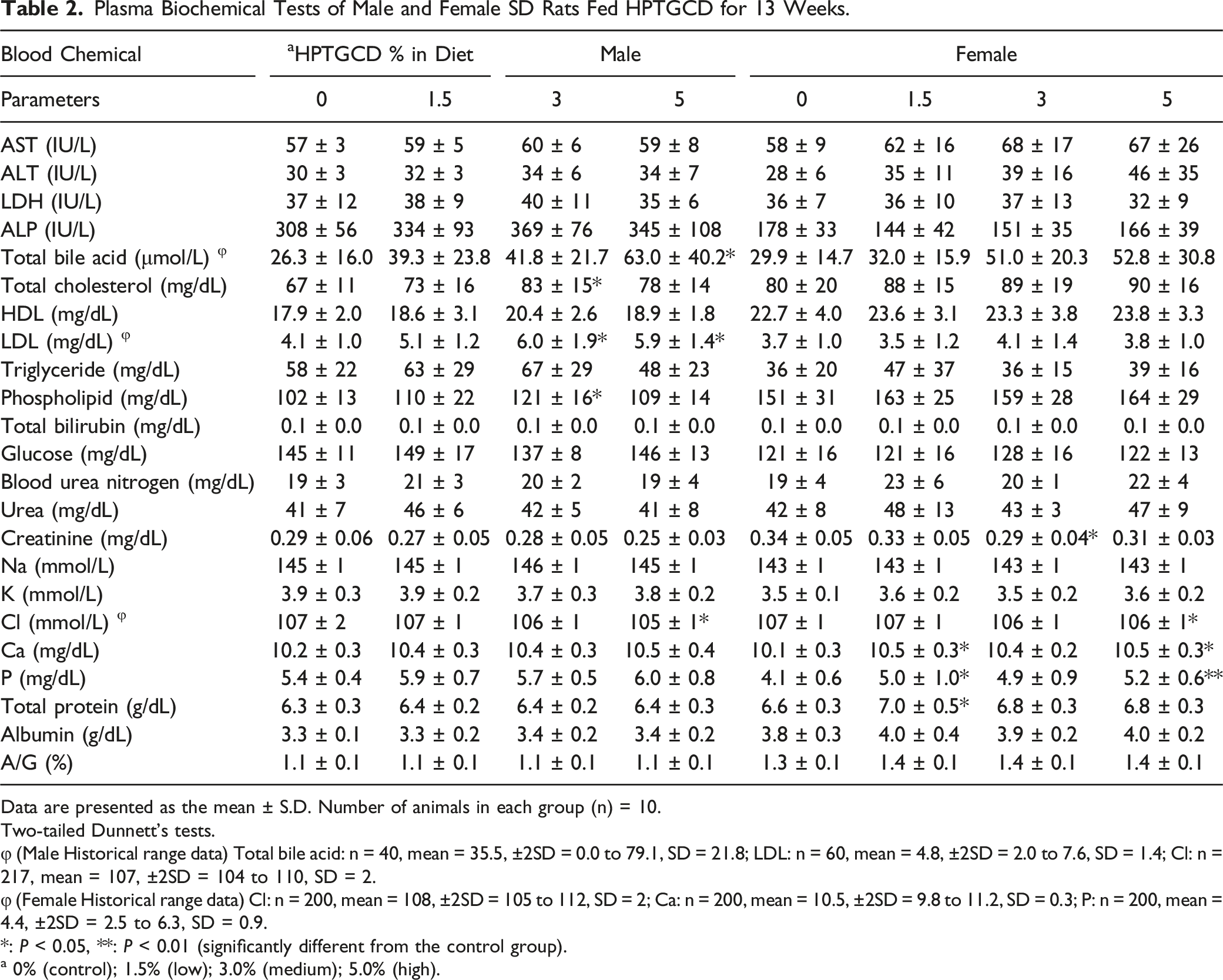
Data are presented as the mean ± S.D. Number of animals in each group (n) = 10.
Two-tailed Dunnett’s tests.
φ (Male Historical range data) Total bile acid: n = 40, mean = 35.5, ±2SD = 0.0 to 79.1, SD = 21.8; LDL: n = 60, mean = 4.8, ±2SD = 2.0 to 7.6, SD = 1.4; Cl: n = 217, mean = 107, ±2SD = 104 to 110, SD = 2.
φ (Female Historical range data) Cl: n = 200, mean = 108, ±2SD = 105 to 112, SD = 2; Ca: n = 200, mean = 10.5, ±2SD = 9.8 to 11.2, SD = 0.3; P: n = 200, mean = 4.4, ±2SD = 2.5 to 6.3, SD = 0.9.
*: P < 0.05, **: P < 0.01 (significantly different from the control group).
a 0% (control); 1.5% (low); 3.0% (medium); 5.0% (high).
Serum thyroid hormone concentrations are presented in Table S8. No significant differences were observed for T3, T4, or TSH levels in the HPTGCD intake groups of either sex compared to those of the control animals. Clinical biochemistry and hormone parameters revealed no abnormalities.
Organ Weight
The absolute and relative weights of the male and female organs after necropsy are summarized in Tables 3. A significant increase in relative liver weight in male rats of the 1.5%, 3.0%, and 5.0% dose group, and increased absolute and relative liver and spleen weights in female rats of the 1.5% and 5.0% dose groups (P < 0.01 or 0.05) were observed. These changes were within the historical control data of the test facility and there were no histopathological correlates (Figure S1, Table S9)
Histopathological Survey and Gross Pathological Analysis
There were no gross pathological findings other than a dark red focus in the glandular stomach in 1 male and a raised focus in the stomach in 1 female in the 3.0% dose group. These findings were considered incidental, because they were not dose-related (Table S10).
The histopathology results are summarized in Table S9. Minimal changes were observed in the heart, kidney, liver, and prostate (male), but these findings were considered incidental because they occur spontaneously in this strain of rats and there was no dose-related pattern to their incidence. There were no other HPTGCD-related pathological changes compared to those in the control groups. Images of the organs of representative males and females in the control and 5.0% HPTGCD diet groups are shown in Figures S1 and S2. In the absence of any HPTGCD-related finding in the high-dose group, organs/tissues of the middle-and low-dose groups were not further evaluated microscopically.
Discussion
To assess the initial safety of HPTGCD, the present study evaluated in vitro mutagenicity, acute toxicity, and subchronic toxicity in SD rats. The in vitro bacterial reverse mutagenicity results showed no activity of HPTGCD at concentrations of up to 5000 μg/plate for induction of point mutations with or without metabolic activation. This is consistent with the finding that Bjeldanes et al 42 for nonplanar flavonoids, with reduced pyrone ring such as HPT, did not exhibit any mutagenic activity. Furthermore, carcinogenicity (such as cell transformation assay), and genotoxicity (such as micronucleus, comet, and chromosomal aberration tests) tests will be conducted to confirm the lack of genotoxicity of HPTGCD as the next step.
The acute toxicity study showed the minimum lethal dose of HPTGCD of > 2000 mg/kg, indicative of a substance with very low potential for toxicity. Further, the 2-week study showed a well-tolerated 5.0% dietary admixture of HPTGCD without toxicity. However, as noted by Li et al. 12 Hesperidin ingestion at much higher levels (5000 mg/kg) in an acute toxicity test showed 10% mortality compared to the control, with a significant increase in absolute liver and spleen weight of HSP-treated rats. A subchronic toxicity study of HSP (1000 mg/kg) using rats reported that weights of the liver and spleen increased significantly (P < 0.05) in male rats compared to that in the controls. Similarly, the weight of the liver (P< 0.01 and P < 0.05) and the spleen (P < 0.05) increased significantly in female rats compared to controls. Moreover, mild inflammatory infiltration and necrosis in the colon and minimal focal to multifocal lymphocytic infiltration in the liver were observed in both male and female rats.
Considering these results, a subchronic toxicity study with HPTGCD, which has a higher bioavailability, was performed. In the 13-week subchronic dietary admix toxicity study at 0%, 1.5%, 3.0%, and 5.0% HPTGCD, there were no mortalities, clinical signs of toxicity, changes in body weights or food consumption, ophthalmic abnormalities, or clinical or anatomic pathology.
Statistically significant increases in the mean relative liver weights of male rats in the 1.5%, 3.0%, and 5.0% dose groups, and in the mean absolute and relative liver weights of female rats were noted in the 1.5% and 5.0% dose groups. Statistically significant increases were noted in several biochemical and hematology parameters: TBA (% group), T-Cho and PL (3% group), LDL (3.0% and 5.0% groups), and FIB (1.5% and 5.0% groups) in male rats, would suggest the possibility of hypertrophy or liver damage in rats.43-48
However, there was no evidence of hepatotoxicity based on the following: (1) Levels of AST, ALT, LDH, ALP, T-BIL, HDL, TG, ALB, and BUN associated with liver function damage in plasma biochemical tests were not significant at every dose in male and female rats compared to the control group (Table 2) (AST, ALT, and ALP,
49
T-BIL albumin,
50
HDL, LDH, TG, and ALB
45
). (2) The levels of PT and APTT coagulation parameters in the hematological tests associated with liver damage were not significant in every dose in male and female rats compared to the control group (Table 1).
43
(3) The liver histopathological revealed no abnormalities in male or female rats compared to the control groups (Figure S1, Table S9). (4) The statistically significant increases were within historical control values at the facility, and for the most part, did not show a dose-response pattern.
Statistically significant increases in Ca, P (females in the 1.5% and -5.0% groups), Cl (males and females in the 5.0% group) and decreases of creatinine (females in the 3.0% group) were noted in the plasma biochemical analyses. However, there were no changes observed in BUN, nor in mean absolute and relative kidney weights, urinalysis parameters, or histopathology correlates (Figure S1, Tables S7, 2, 3, and S9).51-54
Absolute and Relative Organ Weights of Male and Female SD Rats Fed HPTGCD for 13 Weeks.
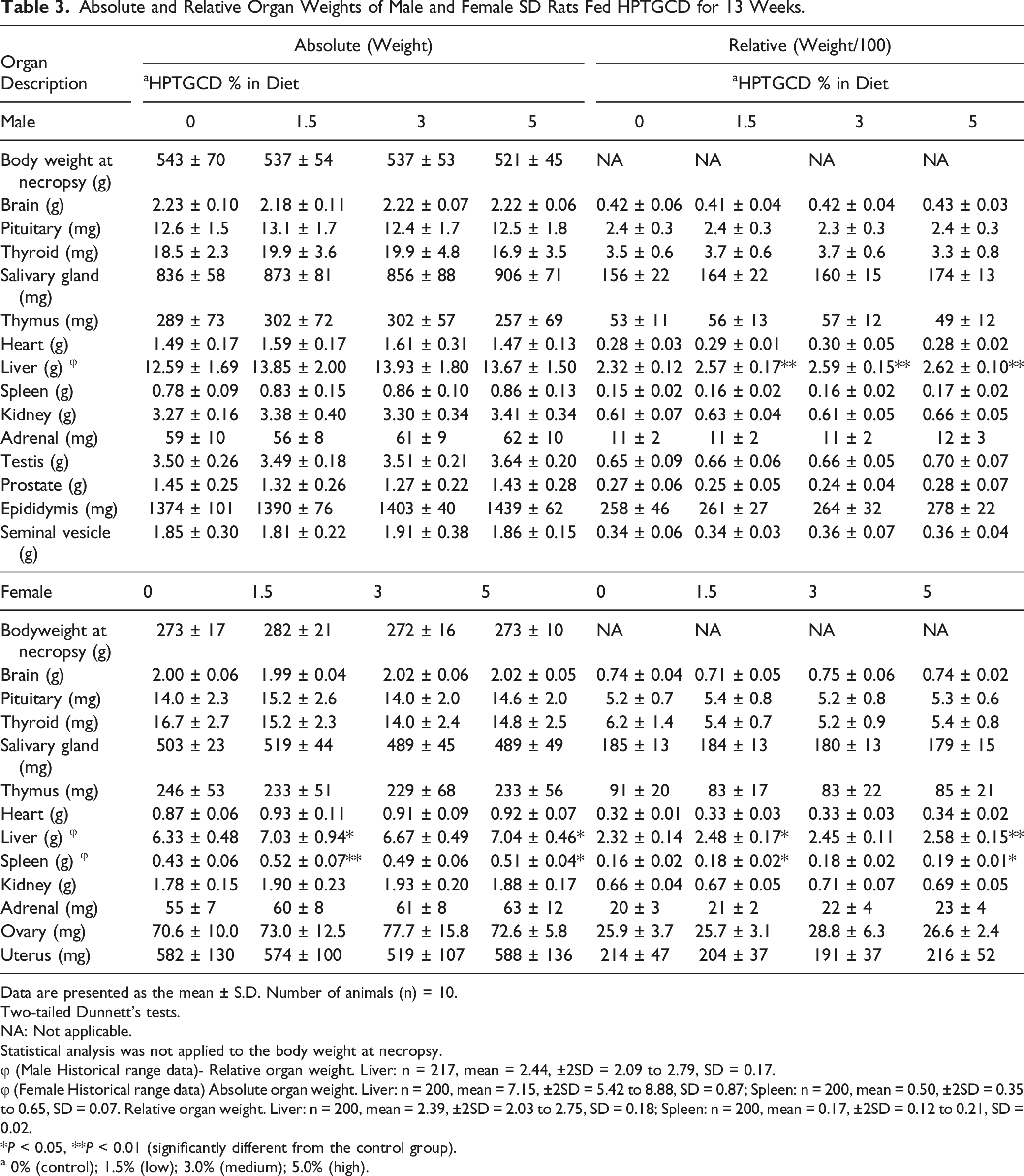
Data are presented as the mean ± S.D. Number of animals (n) = 10.
Two-tailed Dunnett’s tests.
NA: Not applicable.
Statistical analysis was not applied to the body weight at necropsy.
φ (Male Historical range data)- Relative organ weight. Liver: n = 217, mean = 2.44, ±2SD = 2.09 to 2.79, SD = 0.17.
φ (Female Historical range data) Absolute organ weight. Liver: n = 200, mean = 7.15, ±2SD = 5.42 to 8.88, SD = 0.87; Spleen: n = 200, mean = 0.50, ±2SD = 0.35 to 0.65, SD = 0.07. Relative organ weight. Liver: n = 200, mean = 2.39, ±2SD = 2.03 to 2.75, SD = 0.18; Spleen: n = 200, mean = 0.17, ±2SD = 0.12 to 0.21, SD = 0.02.
*P < 0.05, **P < 0.01 (significantly different from the control group).
a 0% (control); 1.5% (low); 3.0% (medium); 5.0% (high).
Taken together, in the subchronic toxicity study, there were no clinical abnormalities or significant changes in body weight, food consumption, survival rates, clinical biochemistry, and microscopic features of the organs. The NOAEL of HPTGCD in the 90-days subchronic toxicity study was 5.0% for both sexes (equivalent to 3267.7 (464 as HPTG conversion) mg/kg/day for males and 3652.4 (519 as HPTG conversion) mg/kg/day for females). Further studies such as carcinogenicity tests, genotoxicity tests, reproductive/developmental tests, and ADME (absorption, distribution, metabolism, and excretion) are warranted to confirm our findings.
Supplemental Material
Supplemental Material - Mutagenic, Acute, and Subchronic Toxicity Studies of the Hesperetin-7- Glucoside –β- Cyclodextrin Inclusion Complex
Supplemental Material for Mutagenic, Acute, and Subchronic Toxicity Studies of the Hesperetin-7-Glucoside–β-Cyclodextrin Inclusion Complex by Masamitsu Moriwaki, Kento Kito1, Ryo Nakagawa, Etsuko Tominaga, Mahendra P. Kapoor, Yoshiki Matsumiya, Tomohisa Fukuhara, Hiroshi Yamagata, Toyohisa Katsumata, and Kazuyuki Minegawa in International Journal of Toxicology
Footnotes
Acknowledgments
The authors acknowledge the contributions of staff members at BoZo Research Center, Inc., who provided technical, animal care, formulation, dosing, necropsy, analytical chemistry, and quality assurance services in support of these studies. The authors would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Author Contributions
Masamitsu Moriwaki contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Kento Kito contributed to design, contributed to analysis, and critically revised manuscript; Ryo Nakagawa contributed to design, contributed to analysis, and critically revised manuscript; Etsuko Tominaga contributed to design, contributed to analysis, and critically revised manuscript; Mahendra P. Kapoor contributed to conception, contributed to acquisition and interpretation, drafted manuscript, and critically revised manuscript; Yoshiki Matsumiya contributed to design, contributed to analysis, and critically revised manuscript; Tomohisa Fukuhara contributed to design, contributed to analysis, and critically revised manuscript; Hiroshi Yamagata contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Toyohisa Katsumata contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Kazuyuki Minegawa contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author (MM) declares patent ownership.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was conducted at the BoZo Research Center Inc. and was funded by Taiyo Kagaku Co., Ltd., a manufacturer of HPTGCD. Taiyo Kagaku Co., Ltd. and BoZo Research Center Inc. were responsible for the study design, the collection, analysis, and interpretation of data, and the writing of the final study reports and manuscript. The decision to submit the article for publication with the support of BoZo Research Center Inc. was made by Taiyo Kagaku Co., Ltd.
CORRECTION (May 2023):
The last sentence before the Acknowledgment section was published with some incorrect results. The online version has been corrected to reflect the correct results.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
