Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Panthenol, Pantothenic Acid, and 5 derivatives as used in cosmetics. These ingredients named in this report are reported to function in cosmetics as hair conditioning agents, and Panthenol also is reported to function as a skin-conditioning agent-humectant and a solvent. The Panel reviewed relevant data for these ingredients, and concluded that these 7 ingredients are safe in cosmetics in the present practices of use concentration described in this safety assessment.
Introduction

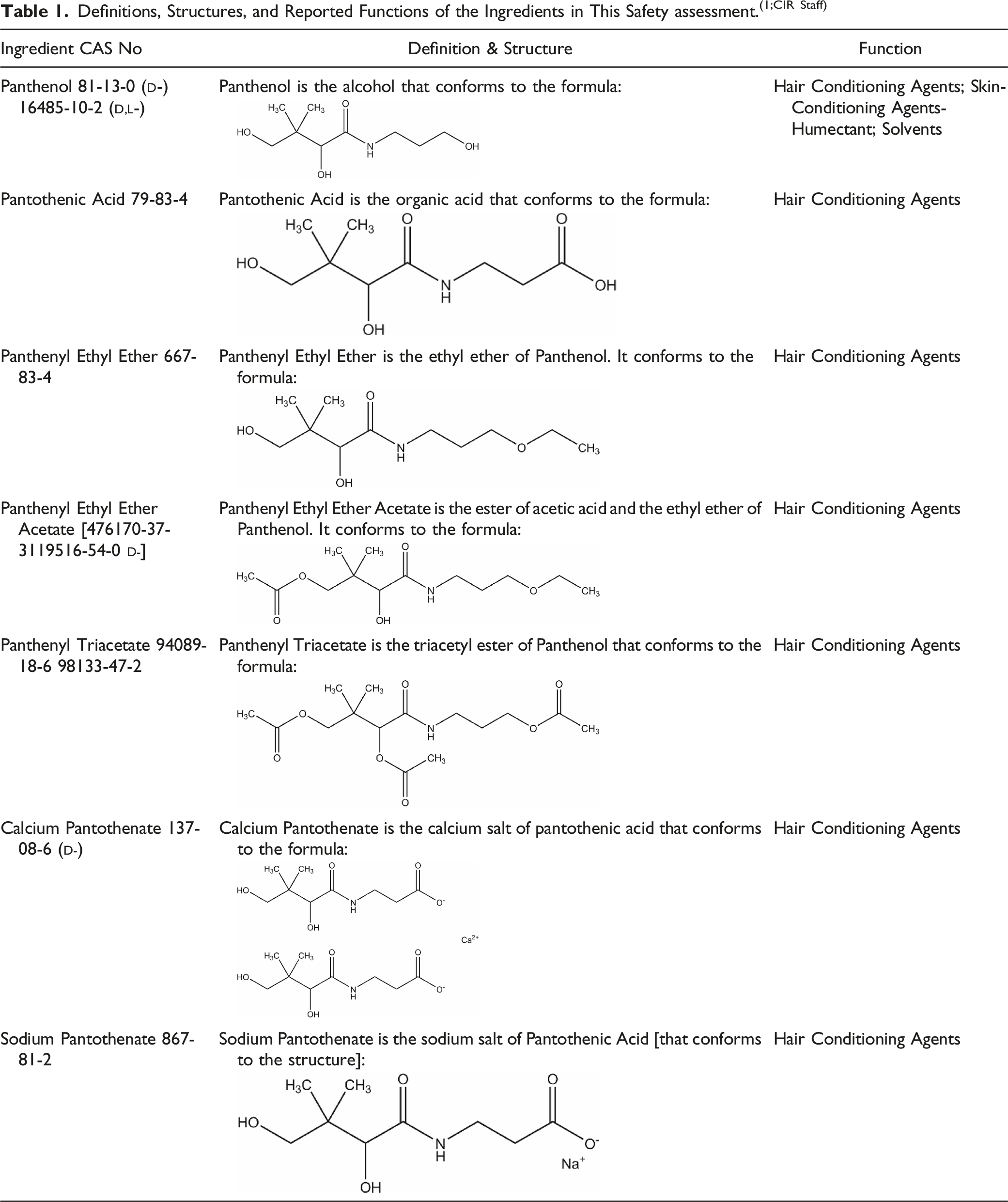
Definitions, Structures, and Reported Functions of the Ingredients in This Safety assessment.(1;CIR Staff)
Although this safety assessment includes two ingredients that have been reviewed previously (i.e., Panthenol and Pantothenic Acid), this report is not a re-review. This report was initiated because of the high frequency of use of Panthenyl Ethyl Ether (382 uses) in cosmetic formulations, as reported by the US Food and Drug Administration (FDA) Voluntary Cosmetic Registration Program (VCRP). 2 Pantothenic Acid, the water-soluble vitamin B5, 3 and its alcohol analogue, Panthenol, are closely related to the five derivatives above and, therefore, are included in this safety assessment. In 1987, the Panel reviewed Panthenol and Pantothenic Acid and concluded that these ingredients are safe for use in cosmetics. 4 In accordance with the Cosmetic Ingredient Review (CIR) Procedures (https://www.cir-safety.org/supplementaldoc/cir-procedures) these ingredients were re-reviewed after 15 years, and the Panel reaffirmed the original conclusion. 5
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
A current search of published literature revealed new data for Panthenol and Pantothenic Acid, which are summarized in this safety assessment as appropriate including in tables and the summary section. Additionally, updated frequency of use and concentration of use data for Panthenol and Pantothenic Acid are included in this safety assessment.
Some of the data included in this safety assessment were found on the European Chemicals Agency (ECHA) website.6,7 In this safety assessment, ECHA is cited as the reference for summaries of information from industry obtained from the ECHA website. Also referenced in this safety assessment are summary data found in reports made publically available by the FDA8-15 and the National Technical Information Service (NTIS). 16
Chemistry
Definition and Structure
The derivative ingredients in this report are related to Panthenol and Pantothenic Acid, sharing the same structural core. Each ingredient is an ethyl ether, acetyl ester, or simple salt of either Panthenol or Pantothenic Acid (Figure 1). The dextrorotary ( Panthenol, pantothenic acid, and derivatives.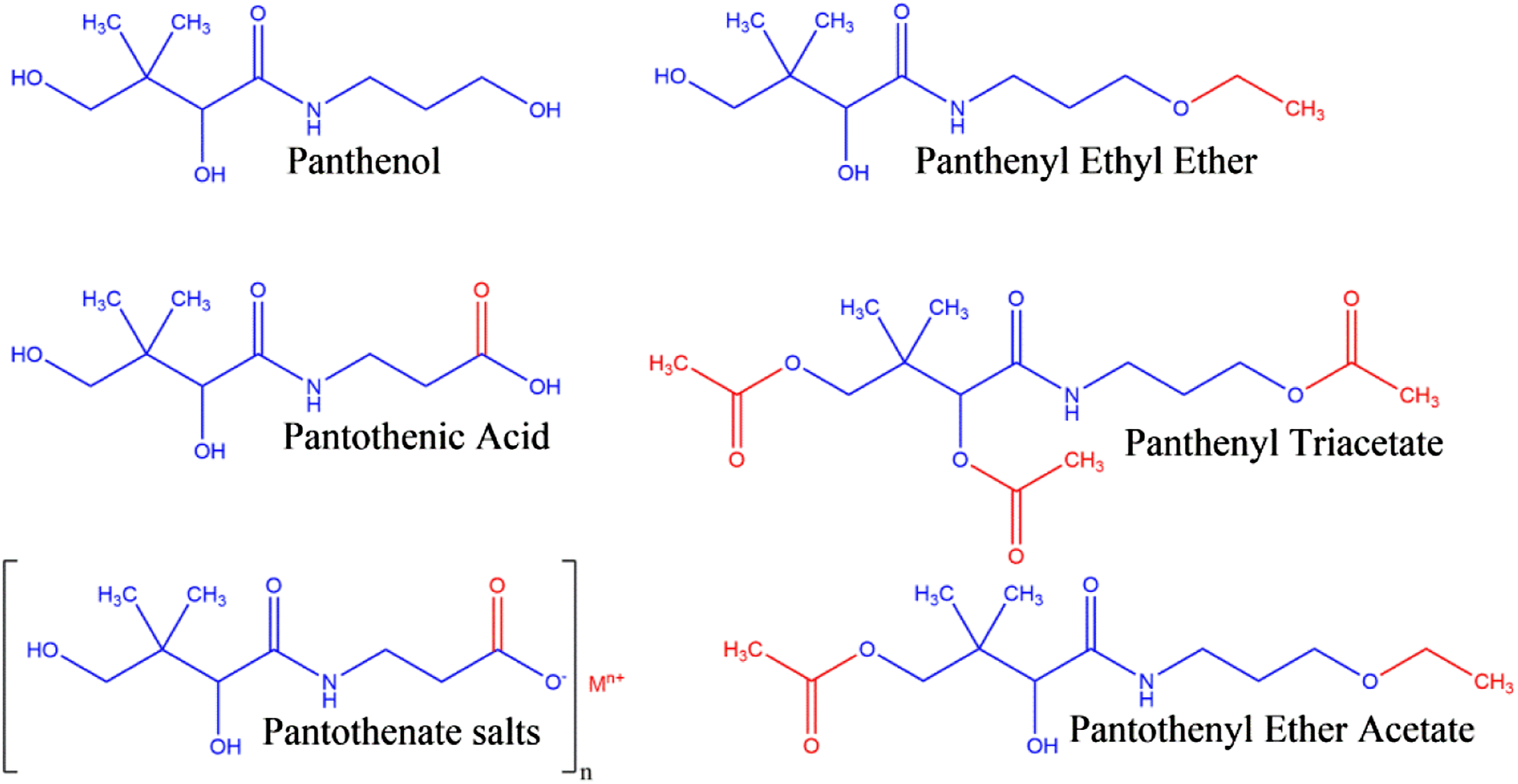
Physical and Chemical Properties
Physical and Chemical Properties.

Calcium Pantothenate is more chemically stable than free Pantothenic Acid and Sodium Pantothenate, which are chemically unstable.
28
Pantothenic Acid has been reported to be stable to heat in neutral or slightly acidic environments, but less stable under alkaline conditions.
29
Calcium pantothenate
When used as a nutritional additive in animal feed,
Method of Manufacture
Panthenol
Pantothenic acid
Pantothenic Acid can be synthesized via saponification of sodium β-alaninate with sodium hydroxide, followed by reaction with
Panthenyl ethyl ether
A condensation reaction of
Panthenyl triacetate
Calcium pantothenate
Sodium pantothenate
Sodium Pantothenate may be prepared by reacting (R)-pantolactone and sodium beta-alaninate in ethanol or methanol. 38
Impurities
Panthenol
According to the Food Chemicals Codex (FCC), food grade specifications limit lead impurities in
A manufacturer reported specifications from a
Specifications reported from a
Panthenyl ethyl ether
Reported specifications from a
Panthenyl triacetate
A certificate of analysis indicated that a sample of
Calcium pantothenate
The FCC specifies that
Natural Occurrence
Pantothenic acid
Jelly from queen bees, rice bran, molasses, and liver are all sources of Pantothenic Acid. 17 Additional sources are meat, whole grains, legumes, eggs, milk, fruits, and vegetables. 41
Use
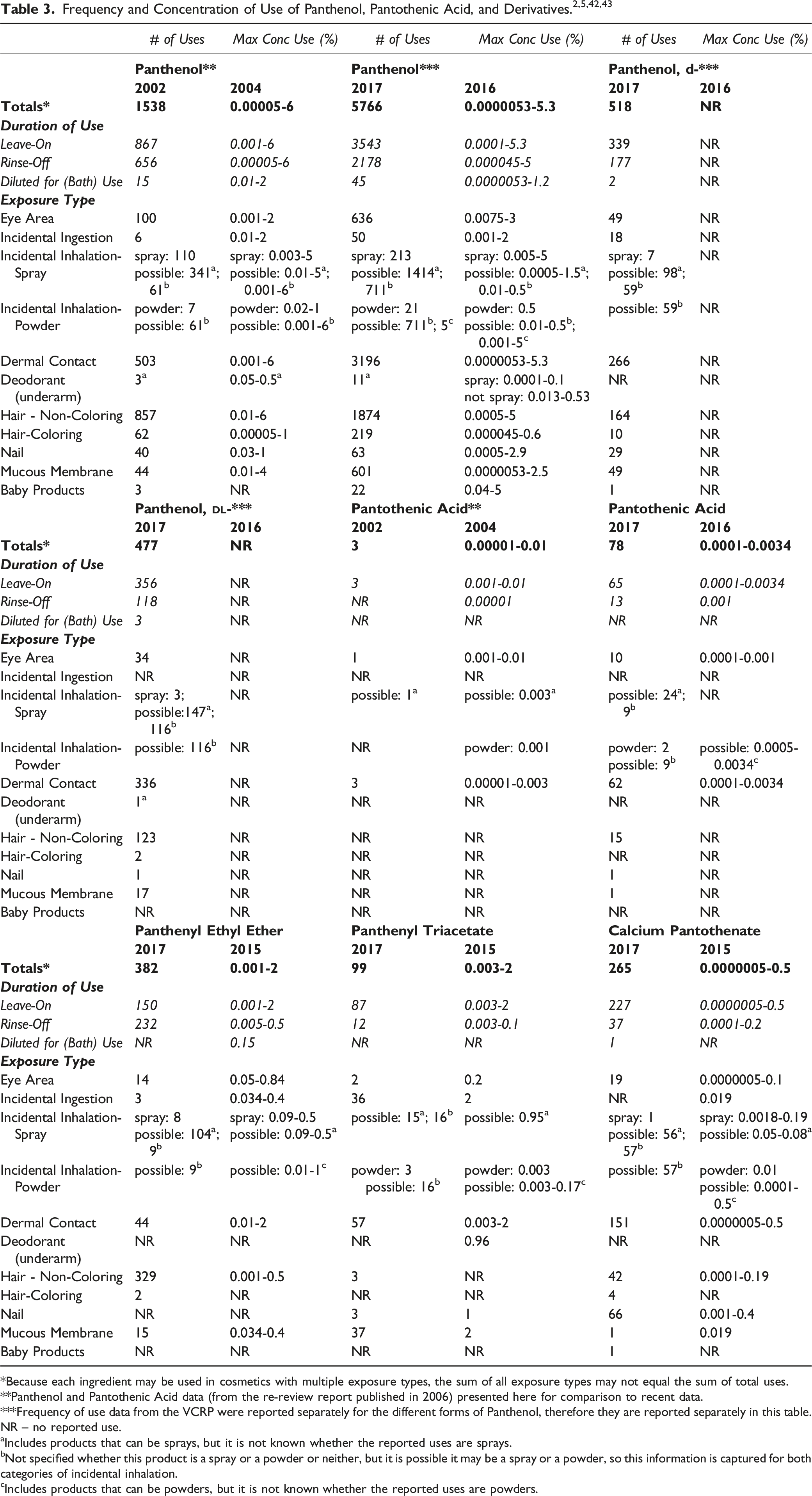
Cosmetic
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
**Panthenol and Pantothenic Acid data (from the re-review report published in 2006) presented here for comparison to recent data.
***Frequency of use data from the VCRP were reported separately for the different forms of Panthenol, therefore they are reported separately in this table.
NR – no reported use.
aIncludes products that can be sprays, but it is not known whether the reported uses are sprays.
bNot specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation.
cIncludes products that can be powders, but it is not known whether the reported uses are powders.
There are no frequency of use or concentration of use reported for Panthenyl Ethyl Ether Acetate and Sodium Pantothenate.2,42
The ingredients in this safety assessment are reported to be used in cosmetic sprays, including hair sprays, body and hand sprays, and fragrances, and could possibly be incidentally inhaled. For example, Panthenol, Panthenyl Ethyl Ether and Calcium Pantothenate are reportedly used in aerosol and pump hair sprays at concentrations up to 0.6%, 0.5%, and 0.19%, respectively.42,43 Panthenol and Panthenyl Ethyl Ether are used in body and hand sprays at concentrations up to 5% and 0.5%, respectively.42,43 Panthenol is used in colognes up to 0.5% and in deodorant sprays up to 0.1%. 43 In practice, most droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm compared with pump sprays.44-47 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.44,46 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 46 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Panthenol, Panthenyl Triacetate, and Calcium Pantothenate are reportedly used in face powders at concentrations up to 0.5%, 0.003%, and 0.01%, respectively, and could possibly be inhaled.42,43 VCRP data indicate that Panthenol and Pantothenic Acid are reportedly used in face powders and Panthenol is used in powders. 2 Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.48-50
Panthenol (3% in eye lotions), Pantothenic Acid (0.001% in eye shadows), and Panthenyl Ethyl Ether (0.84% in eye shadows) are reported to be used in cosmetic formulations indicative of potential eye exposure.42,43 Panthenol (2.5% in other personal cleanliness products; 2% in lipstick) and Panthenyl Triacetate (2% in lipstick) are reported to be used in formulations with possible mucous membrane exposure and/or ingestion.42,43
Panthenol, Pantothenic Acid, and the five derivatives included in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 51
Non-Cosmetic
Appearance of Ingredients in Code of Federal Regulations.
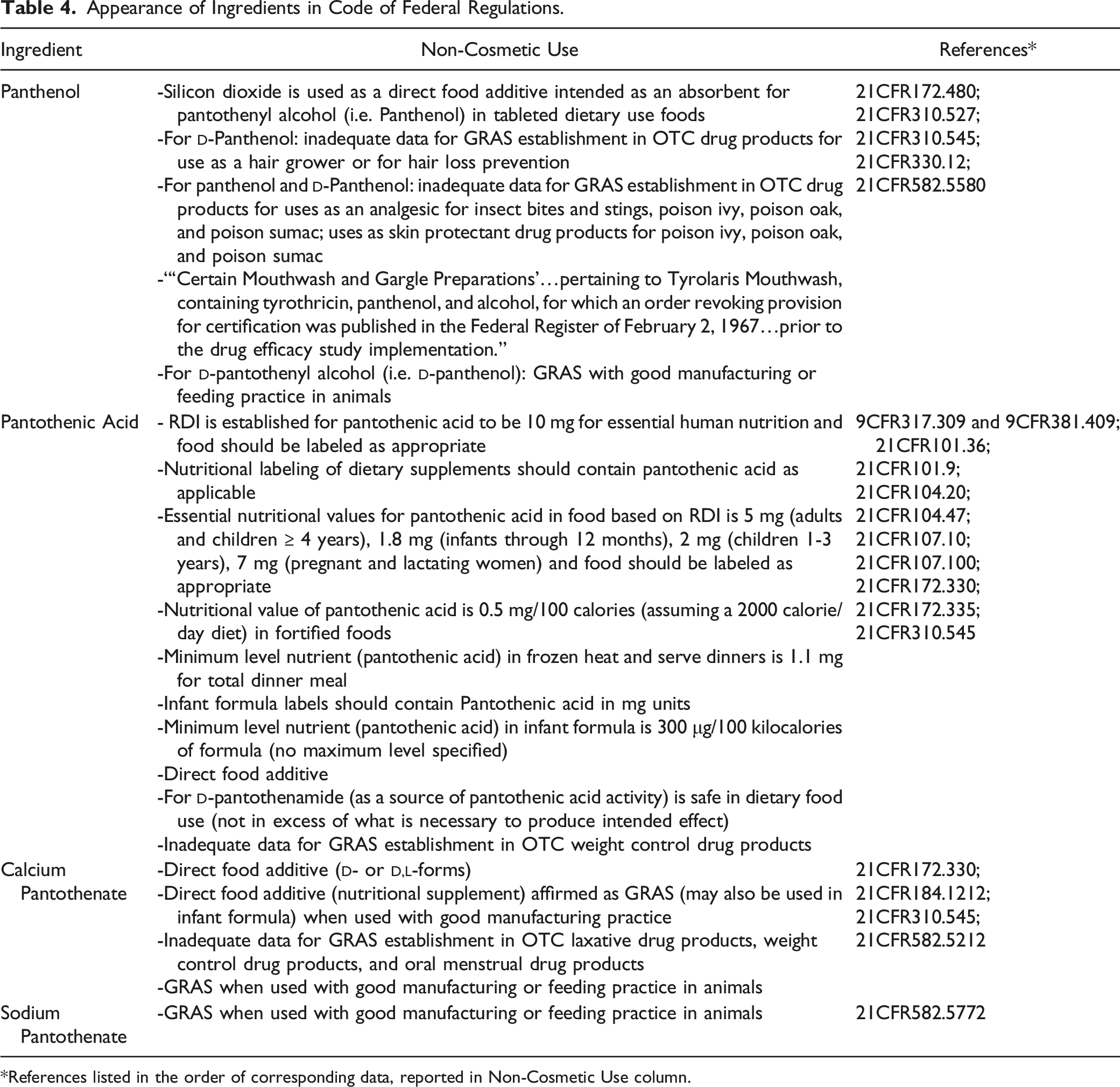
*References listed in the order of corresponding data, reported in Non-Cosmetic Use column.
There was inadequate safety data to establish generally-recognized-as-safe-and-effective status in various over-the-counter (OTC) drug products (including weight control products) for Panthenol, Pantothenic Acid, and Calcium Pantothenate (21CFR310.527, 21CFR310.545).
Panthenol
Calcium pantothenate
The FDA permitted a 510 (k) premarket notification for a medical device marketed for human oocyte in vitro fertilization, which listed Calcium Pantothenate (concentration not specified) as an ingredient. 13
Toxicokinetic Studies
Panthenyl Triacetate
Panthenyl Triacetate has been reported to convert to Panthenol and Pantothenic Acid upon dermal application to human skin.53,54 Panthenyl Triacetate has also been reported to penetrate underarm skin. 54
Dermal and Nail Penetration Studies.
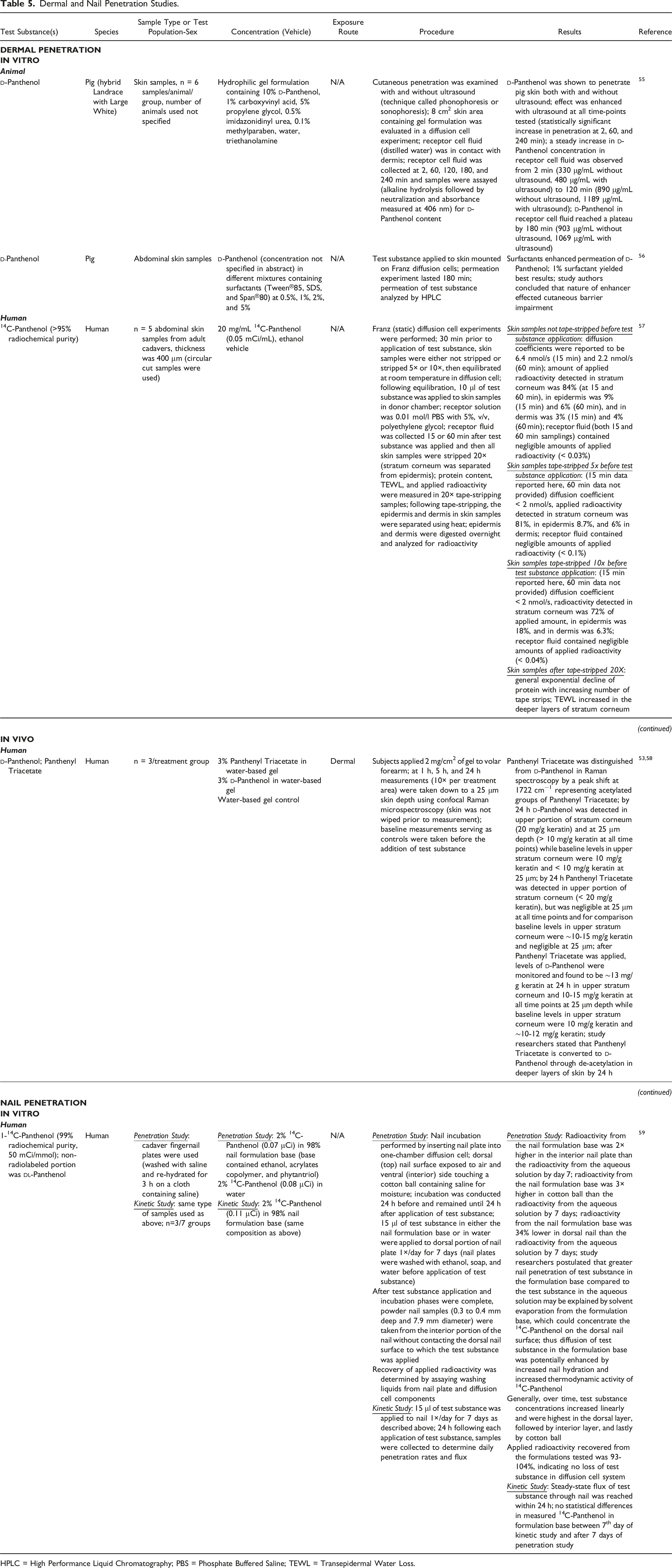
HPLC = High Performance Liquid Chromatography; PBS = Phosphate Buffered Saline; TEWL = Transepidermal Water Loss.
Dermal Penetration
In vitro
Animal
The cutaneous penetration of
Human
The dermal penetration of [14C]Panthenol (20 mg/mL in ethanol, 0.05 mCi/mL) through human abdominal skin samples was evaluated in a Franz (static) diffusion cell experiment. 57 Skin samples were either not stripped or stripped 5× or 10× prior to the application of 10 μl test substance. The receptor solution (0.01 mol/l phosphate buffered saline with 5% polyethylene glycol (v/v)) was collected up to 60 min post-application, and then all skin samples were stripped 20× before analysis. In the skin samples not stripped prior to test substance application, the amount of applied radioactivity detected after 601 min was 84% in the stratum corneum, 6% in the epidermis, and 4% in the dermis; radioactivity in the receptor fluid was negligible (< 0.03%). For the 5× stripped samples, the radioactivity detected 15 min post-application was 81%, 8.7%, and 6% in the stratum corneum, epidermis, and dermis, respectively; radioactivity in the receptor fluid was negligible (< 0.1%). For the 10× stripped samples, the radioactivity detected 15 min post-application was 72, 18, and 6.3% in the stratum corneum, epidermis, and dermis, respectively; radioactivity in the receptor fluid was negligible (< 0.04%).
In Vivo
Human
Nail Penetration
In vitro
Human
An experiment examined the penetration of 1-[14C]Panthenol through human fingernails. 59 Nail incubation was conducted by inserting the nail plate into one-chamber of a diffusion cell with the dorsal nail surface exposed to air and the ventral side touching a cotton ball containing saline for moisture. Fifteen microliters of 2% [14C]Panthenol (0.07 - 0.08 μCi) in either a 98% nail formulation (containing ethanol, acrylates copolymer, and phytantriol) or water, was applied to the dorsal nail daily for 1 wk. Results showed that, by day 7, the applied radioactivity from the formulation was 2 times higher in the interior nail plate and 3 times higher in the cotton ball compared to the radioactivity in the applied aqueous solutions. The radioactivity was 34% lower in the dorsal nail by day 7 when the formulation was used, compared to the aqueous solution. The researchers speculated that solvent evaporation of the formulation may have concentrated the [14C]Panthenol on the dorsal nail, and that diffusion of the test substance may have been enhanced by a formulation-induced increase in nail hydration and increased thermodynamic activity of [14C]Panthenol in the formulation.
Penetration Enhancement
In vitro
Animal
The effect of
Additional experiments evaluating the release of progesterone from the polymer formulations were conducted. The polymer matrix formulations (200 μm total thickness) described above were placed in a diffusion cell without rat skin. The receptor cell conditions and fluid analysis were as described above. The PMA formulations (6% and 20%
Absorption, Distribution, Metabolism, Excretion (ADME)
Panthenol can be oxidized in the skin to Pantothenic Acid.
4
The reactions in which Pantothenic Acid plays a role are the synthesis and metabolism of steroid hormones, sterols, and fatty acids, the synthesis of acetylcholine and porphyrins, and carbohydrate metabolism. A toxicokinetics study in rats fed 20 mg/kg/d
d -panthenol
Pantothenic acid
Pantothenic Acid naturally occurs in all animal and plant tissues. 17 As a vitamin in the B complex, it is vital for coenzyme A synthesis in mammalian cells. The Pantothenic Acid Reference Daily Intake (RDI) for essential human nutrition is 5 to 10 mg (Table 4).
Toxicokinetics Studies-Absorption, Distribution, Metabolism, Excretion (ADME).
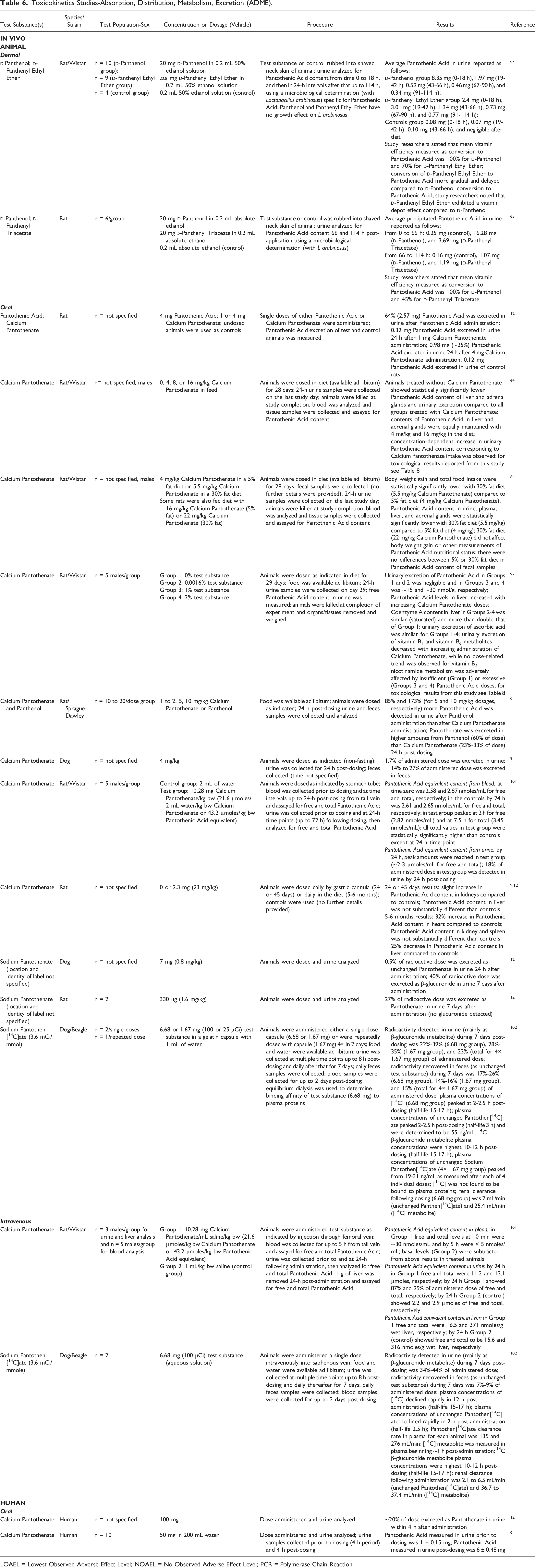
LOAEL = Lowest Observed Adverse Effect Level; NOAEL = No Observed Adverse Effect Level; PCR = Polymerase Chain Reaction.
Animal
A dermal exposure experiment in rats treated with
Single doses of either Pantothenic Acid (4 mg) or Calcium Pantothenate (4 mg) were orally administered to rats; 64% of Pantothenic Acid was detected in the urine 24 h after Pantothenic Acid administration and ∼25% of Pantothenic Acid was found in the urine 24 h following Calcium Pantothenate dosing. 12 In another experiment, rats were dosed daily in the diet with 0, 4, 8, or 16 mg/kg Calcium Pantothenate for 28 d. 64 In the control group (vitamin deficient group), Pantothenic Acid content of the liver and adrenal glands and urinary excretion were statistically significantly lower than all the treated groups. A dose-dependent increase in urinary Pantothenic Acid content corresponding to Calcium Pantothenate intake was observed. A study was conducted in rats fed 0 (vitamin deficient group), 0.0016, 1, or 3% Calcium Pantothenate daily in the diet for 29 d. 65 Notable results included an increase in liver Pantothenic Acid levels and a decrease in urinary excretion of vitamins B1 and B6 metabolites with increasing Calcium Pantothenate doses, and an adverse effect on nicotinamide metabolism in the vitamin deficient group and in the animals exposed to 1% and 3% concentrations. Rats were orally dosed with 1, 2, 5, or 10 mg/kg Calcium Pantothenate or Panthenol; 24 h urine and feces samples were collected and analyzed. 9 Results showed that 85% (from 5 mg/kg dosage) and 173% (from 10 mg/kg dosage) more Pantothenic Acid was detected in urine after Panthenol administration than following Calcium Pantothenate dosing. Pantothenate was excreted in greater amounts after Panthenol exposure (60% of dose) than after Calcium Pantothenate exposure (23 - 33% of dose). In rats orally exposed to 23 mg/kg Calcium Pantothenate daily in the diet for 5 - 6 mo a 32% increase in Pantothenic Acid content in the heart and a 25% decrease in Pantothenic Acid content in the liver were observed.9,12 Radiolabeled Sodium Pantothenate (location and identity of label was not specified) was orally administered to dogs (0.8 mg/kg) and rats (1.6 mg/kg) and urine was analyzed. 12 In dogs, 0.5% of the dosed radioactivity was excreted as unchanged Pantothenate in the urine 24 h post-dosing and 40% was excreted as the β-glucuronide within 7 d. In rats, no glucuronide was detected and 27% of the radioactivity was excreted as Pantothenate in the urine within 7 d of administration.
Human
Human subjects were orally dosed with 100 mg of Calcium Pantothenate (no additional details were provided) and by 4 h post-administration ∼20% of the dose was excreted as Pantothenate in the urine. 12 Following oral administration (dosage not specified) in human subjects, Pantothenic Acid was absorbed from the gastrointestinal tract; urinary excretion of unchanged Pantothenic Acid was approximately 70% and in feces about 30%. 41
Toxicological Studies
Human subjects received 10 - 20 g/d Pantothenic Acid orally for an unspecified period of time; water retention and occasional diarrhea were noted. 4
Acute Toxicity
In acute studies, there were no deaths in mice orally dosed with 10 g/kg
Acute Toxicity Studies.
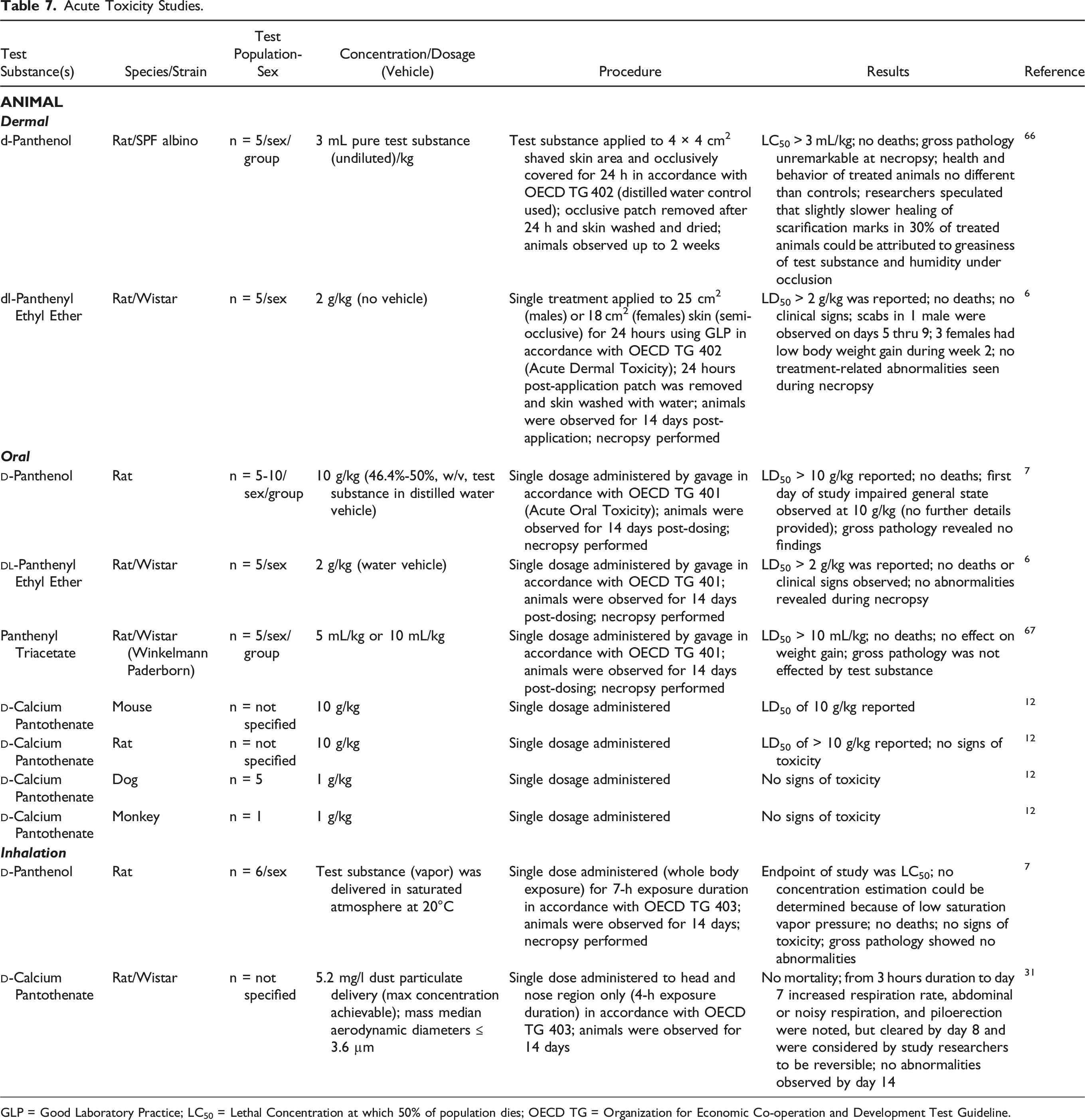
GLP = Good Laboratory Practice; LC50 = Lethal Concentration at which 50% of population dies; OECD TG = Organization for Economic Co-operation and Development Test Guideline.
Dermal
In a 24-h occlusive patch test, 3 mL/kg
Oral
In separate experiments, rats were orally exposed to single dosages of 10 g/kg
Inhalation
A single dose inhalation study in rats exposed to 5.2 mg/l
Short-Term Toxicity
Short-Term, Subchronic, and Chronic Toxicity Studies.
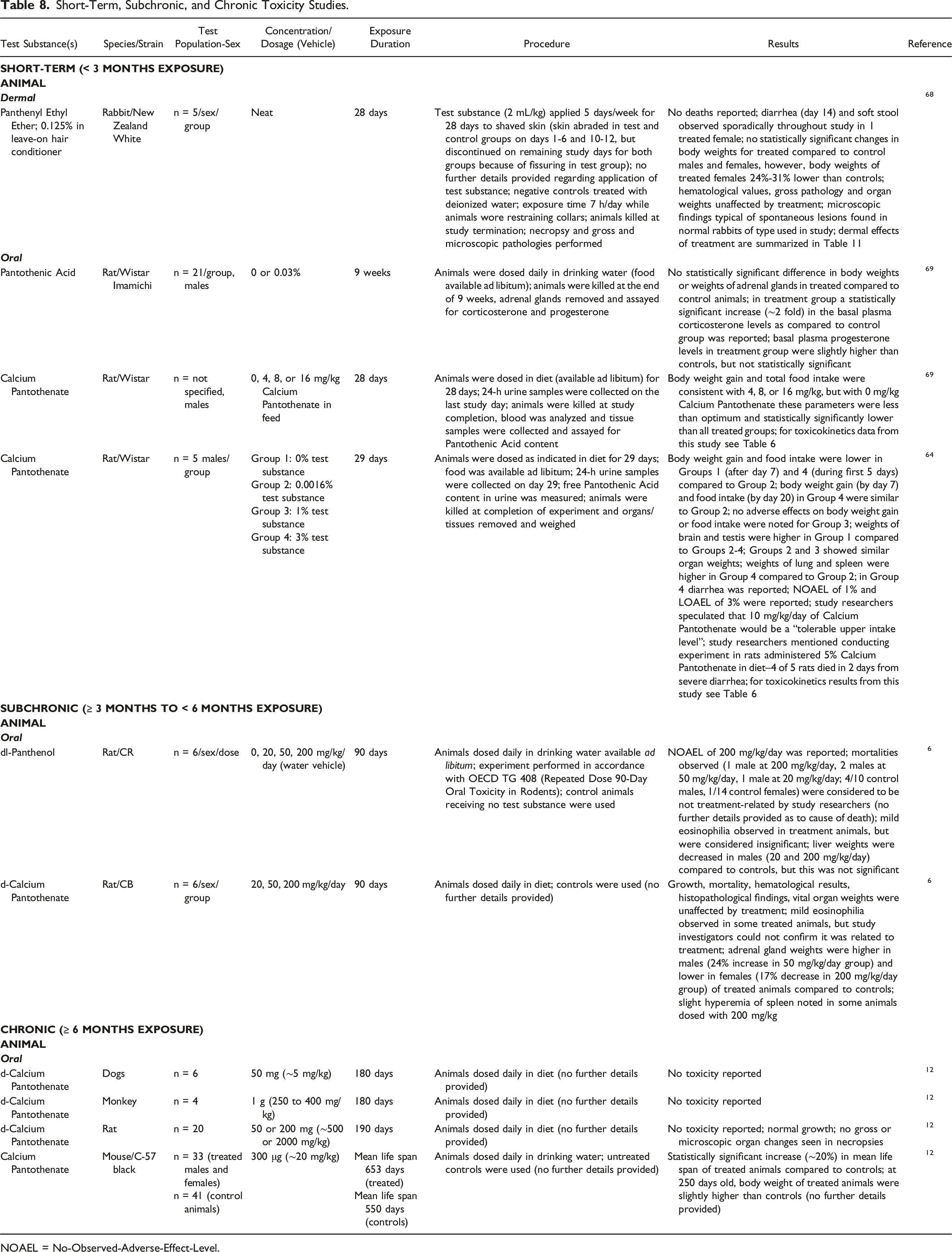
NOAEL = No-Observed-Adverse-Effect-Level.
Dermal
Panthenyl Ethyl Ether (0.125%) in a leave-on hair conditioner was applied (restraint collars used for 7 h after administration of test substance; further details not provided) to the shaved skin of New Zealand White rabbits for 5 d/wk for 28 d. 68 No mortality was reported; diarrhea and soft stools were observed in 1 treated female rabbit periodically throughout the study.
Oral
Rats were administered 0 or 0.03% Pantothenic Acid daily in their drinking water for 9 wk; the only statistically significant finding was a ∼2-fold increase in basal plasma corticosterone levels in the Pantothenic Acid group, compared to the control group. 69 In another experiment, rats were dosed daily in the diet with 0, 4, 8, or 16 mg/kg Calcium Pantothenate for 28 d. 64 In the control group (vitamin deficient group), body weight gain and total food intake were statistically significantly lower than in all the treated groups. A study was conducted in rats fed 0 (vitamin deficient group), 0.0016%, 1%, or 3% Calcium Pantothenate daily in the diet for 29 d. 65 Notable results included a decrease in body weight gain and food intake in the vitamin deficient group, an increase in brain and testis weights in the vitamin deficient group, an increase in lung and spleen weights in the animals exposed to 3%, and diarrhea at 3% concentration. A no-observed-adverse-effect-level (NOAEL) of 1% and a lowest-observed-adverse-effect-level (LOAEL) of 3% Calcium Pantothenate were reported. The same researchers performed a test of 5% Calcium Pantothenate in the diet; 4 of the 5 rats died within 2 d from severe diarrhea.
Subchronic Toxicity
In 3-mo subchronic toxicity studies there were no deaths reported from dermal exposure in rabbits (6 mg/cm2 of 0.5% Panthenol) and rats (227 to 680 mg/kg of 0.2% Panthenol).
4
The rabbits exhibited slight to moderate erythema, edema, and cutaneous desquamation. The rats displayed minimal hyperkeratosis in the subcutis and skin, but no systemic toxicity was observed. There were no toxicological effects reported in rats orally administered up to 200 mg/d
Oral
A NOAEL of 200 mg/kg/d was reported for rats dosed daily in drinking water, available ad libitum, with 20, 50, or 200 mg/kg bw/d
Chronic Toxicity
In rats orally administered 2 mg/d Panthenol for 6 mo there were no histopathological changes. 4
Oral
Developmental and Reproductive Toxicity (DART) Studies
Two different groups of female albino rats were supplemented with the same vitamin mixture and either 100 μg or 1 mg Calcium Pantothenate after giving birth to their first litter (stock diet for all female rats during first pregnancies) and through the birth of young from their second pregnancies (gestation period not provided). 4 The young born from both the first and second pregnancies were normal. No teratogenicity or fetotoxicity were reported.
Developmental and Reproductive Toxicity (DART) Studies.

GLP = Good Laboratory Practice; LOAEL = Lowest-Observed-Adverse-Effect-Level; NOAEL = No-Observed-Adverse-Effect-Level; OECD TG = Organization for Economic Co-operation and Development Test Guideline.
In different experiments examining the effects of orally administered Calcium Pantothenate (up to 2000 mg/kg) on pregnant rats (details on gestation were not provided) no toxicity, teratogenicity, or fetotoxicity was reported; Calcium Pantothenate was found to cross the placenta. 12
Genotoxicity
Genotoxicity Studies.
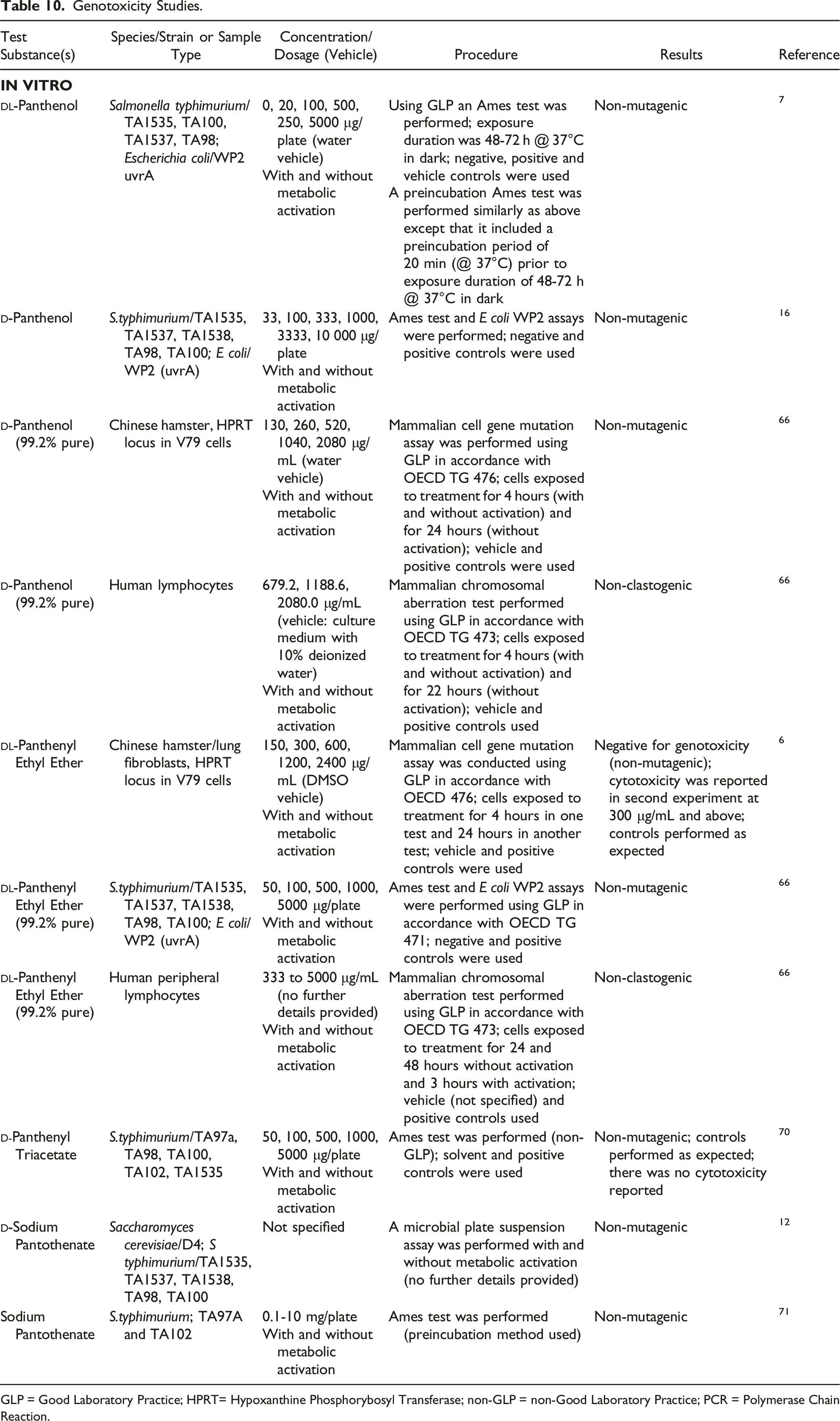
GLP = Good Laboratory Practice; HPRT= Hypoxanthine Phosphorybosyl Transferase; non-GLP = non-Good Laboratory Practice; PCR = Polymerase Chain Reaction.
In Vitro
Carcinogenicity
There were no carcinogenicity studies identified in the literature for the ingredients presented in this report, and unpublished data were not submitted.
Other Relevant Studies
Data summaries included therapeutic uses of
Transformation
Calcium pantothenate
Calcium Pantothenate was evaluated in experiments performed with the BALB/c-3T3 cell neoplastic transformation system, known to produce a tumor-promoting response to phorbol esters. 72 As part of the protocol for the transformation assay, 0.1 μg/mL of 3-methylcholanthrene (carcinogen) was used to initiate the 1-13 cell line of BALB/c-3T3 cells; controls without 3-methylcholanthrene were also used in the experiment. 72 The culture plates were treated with fresh medium (no carcinogen present) 72 h following treatment with 3-methylcholanthrene. On day 7 and twice weekly for 28 d, Calcium Pantothenate (50 μg/mL initiated concentration; 500 μg/mL uninitiated concentration) or control medium were added to dishes treated with 3-methylcholanthrene (0.1 μg/mL) and to dishes not treated with the carcinogen. After 4 wk, 3-methylcholanthrene was removed from the plates. The plates were scanned for Type III foci after staining with Giemsa. Results indicated that Calcium Pantothenate had a promoting effect on Type III transformed foci; a repeat experiment showed this effect to be marginal.
Cytotoxicity
Panthenyl triacetate
The Skin
2
ZK 1200 Model was used in a study evaluating the cytotoxic potential of
Effect on Metabolism
Panthenol and panthenyl triacetate
The epidermis of human abdominal skin samples was treated with 2%
Effect on Human Skin Fibroblasts
Calcium pantothenate, panthenol, and pantothenic acid
RNA from proliferating human dermal fibroblasts was incubated with Calcium Pantothenate (20 μg/mL) or without Calcium Pantothenate for 8 - 12 h in a 2% fetal calf serum medium, then exon array analysis and quantitative polymerase chain reaction were performed. 74 Results indicated that Calcium Pantothenate caused substantial upregulation of mRNA encoding 7 genes in dermal fibroblasts. Human skin fibroblasts were incubated in a medium with Panthenol (up to 20 mM), Pantothenic Acid (up to 1000 μM), or in a control medium for 24 h and analyzed for protein. Heme oxygenase-1 protein inductions were observed in cells treated with Panthenol and Pantothenic Acid. Human skin fibroblasts were treated with Panthenol (up to 20 mM) for 24 h and assayed using chemiluminescence to determine the formation of reactive oxygen species; results showed that Panthenol inhibited the formation of reactive oxygen species.
Wound Healing
In vitro
Calcium pantothenate
In vitro experiments performed in human dermal fibroblast monolayers showed that 20 μg/mL of Calcium Pantothenate accelerated wound healing compared to controls when applied to artificially induced monolayer scrape wounds for 24 h at 37°C. 74 By 20 hours, 80% closure of the wound was observed in treated samples compared to 21% in controls. Further experiments indicated that cell migration also aided in wound closure. Cell culture experiments evaluating cell proliferation, in which 20 or 40 μg/mL of Calcium Pantothenate were incubated with human dermal fibroblasts for up to 16 h, resulted in higher cell counts in treated (effect was more pronounced with 20 than 40 μg/mL) compared to untreated control samples.
Human
d -panthenol and d -panthenyl triacetate
In a double-blind, wound-healing study, suction blisters were formed on the volar forearms of human subjects (n = 40) using a vacuum and then treated (occlusively) with different emulsions containing 3%
Therapeutic Effect
d -panthenol
The therapeutic effect of
Radioprotective Effect
Calcium pantothenate
Calcium Pantothenate (180 mg/day >administered in the diet for 42 d) had radioprotective effects in the skin of partially hepatectomized rats that were exposed to irradiation (Sr 90 -Y 90 beta radiation, 3.6 repetitions/second for 2.48 min), and it facilitated normal metabolic function of hepatocytes. 78 Hepatectomized and irradiated animals that had not been treated with Calcium Pantothenate exhibited both skin changes and hepatocyte dysfunction.
Dermal Irritation and Sensitization Studies
In rabbit skin treated with 100%
A product containing 0.5% Panthenol was applied to the skin of human subjects for 4 d, in a cumulative irritation test (procedures were not provided); results indicated that the test substance was non-irritating. 4 In a different study, a lotion containing 0.5% Panthenol was applied (occlusively) to the backs of 10 subjects. After 23 h the patch was removed and skin washed prior to evaluation. This process was repeated for 21 d. Eight subjects exhibited minimal erythema during the test; study researchers determined that the test substance was mildly irritating.
Panthenol, in various products, was applied to the skin of human subjects and occlusively covered for 24 - 48 hours during the induction and challenge phases of different experiments. 4 In one test, erythema and papules were observed in 3 out of 200 subjects during induction and challenge phases (0.5% Panthenol). Erythema and edema were seen in 3 out of 206 subjects during the induction and challenge phases (0.5% Panthenol) of another test. Erythema was reported in 1 out of 238 subjects during the induction phase (0.5% Panthenol) of an experiment. There were no signs of irritation or sensitization in another study with 200 subjects (0.5% Panthenol) or in a smaller test with 25 subjects (0.5% Panthenol). In other experiments, products containing 0.1% to 0.5% Panthenol were applied to the skin of human subjects and occlusively covered for 24 - 72 h during induction and challenge phases; the test substance was non-sensitizing.
Dermal Irritation and Sensitization Studies.
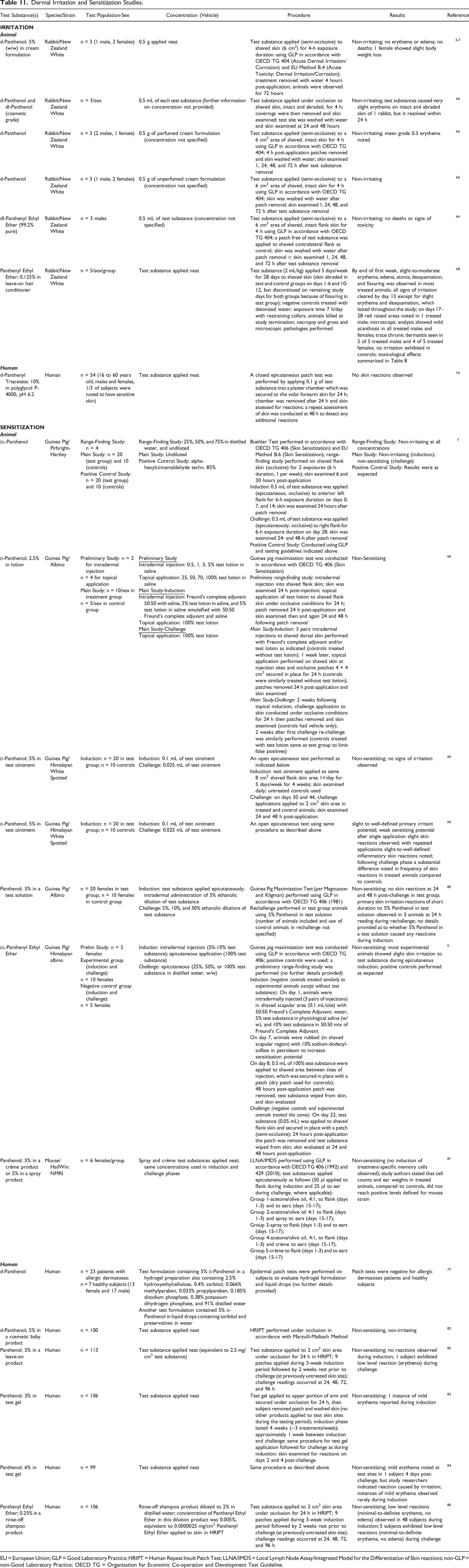
EU = European Union; GLP = Good Laboratory Practice; HRIPT = Human Repeat Insult Patch Test; LLNA/IMDS = Local Lymph Node Assay/Integrated Model for the Differentiation of Skin reactions; non-GLP = non-Good Laboratory Practice; OECD TG = Organization for Economic Co-operation and Development Test Guideline.
Irritation
Animal
An irritation test in rabbits revealed that 0.5 g of 5% (w/w)
Human
Sensitization
Animal
A Buehler test was performed on the shaved flank skin of guinea pigs in accordance with OECD TG 406 (Skin Sensitization) to evaluate the sensitization potential of
Human
Photoirritation/Photosensitization
The structures of these ingredients lack conjugated unsaturations, or other chromophore core moieties. Accordingly, there is no reason to suspect that these would be positive for photoirritation or photosensitization.
Animal
d -panthenol
In an EFSA article,
Ocular Irritation
Rabbits treated with 100%
Ocular Irritation.
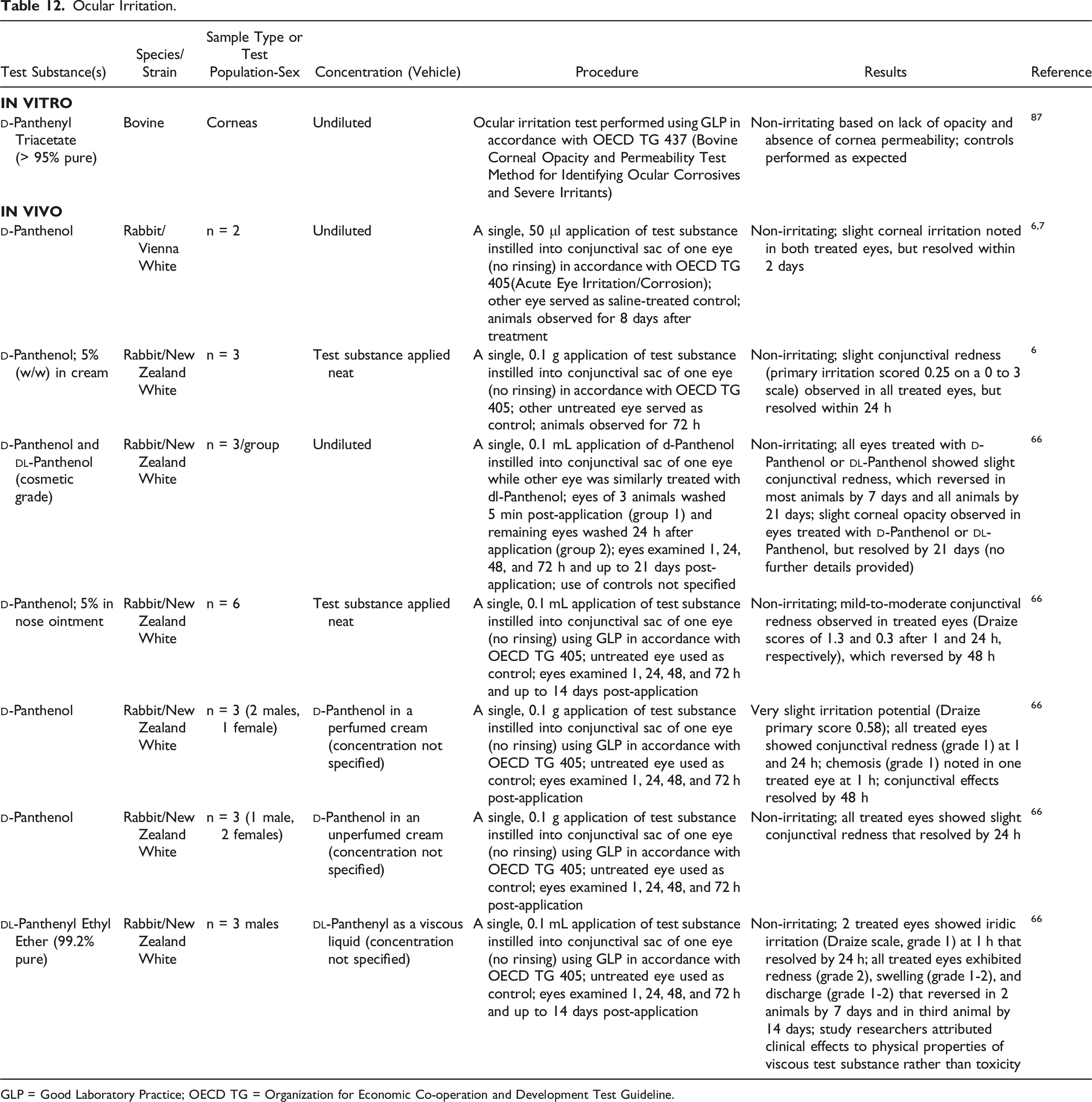
GLP = Good Laboratory Practice; OECD TG = Organization for Economic Co-operation and Development Test Guideline.
In Vitro
An in vitro test was performed using GLP in accordance with OECD TG 437 (Bovine Corneal Opacity and Permeability Test Method for Identifying Ocular Corrosives and Severe Irritants).
87
Animal
In several experiments, single applications of
Clinical Studies
Panthenol
Human subjects in a dermatitis clinic were patch tested with a standard diagnostic series that included a 50%
Retrospective and Multicenter Studies
Panthenol
A European Union report cited data from the Information Network of Departments of Dermatology from 2000 to 2009, documenting 137 positive allergic reactions from a large population (> 96 000 patients) to
In a different study, a total of 3301 patients were patch tested for
Another retrospective study, conducted from 2010 to 2015, included patients who developed cosmetic dermatitis (iatrogenic dermatitis not included) as a result of using cosmetic products. 92 Panthenol was identified as an individual allergen from a cosmetic product (type not defined). It was reported that of the 311 patients patch tested for Panthenol (concentration not specified), 3 (0.96%) exhibited positive reactions. Patch tests were conducted under occlusion for 2 d and test sites read on days 2, 4, and sometimes 7 in accordance with the European Society of Contact Dermatitis guidelines.
Case Reports
Patients with stasis dermatitis and multiple allergies experienced contact allergy to
Case Reports.
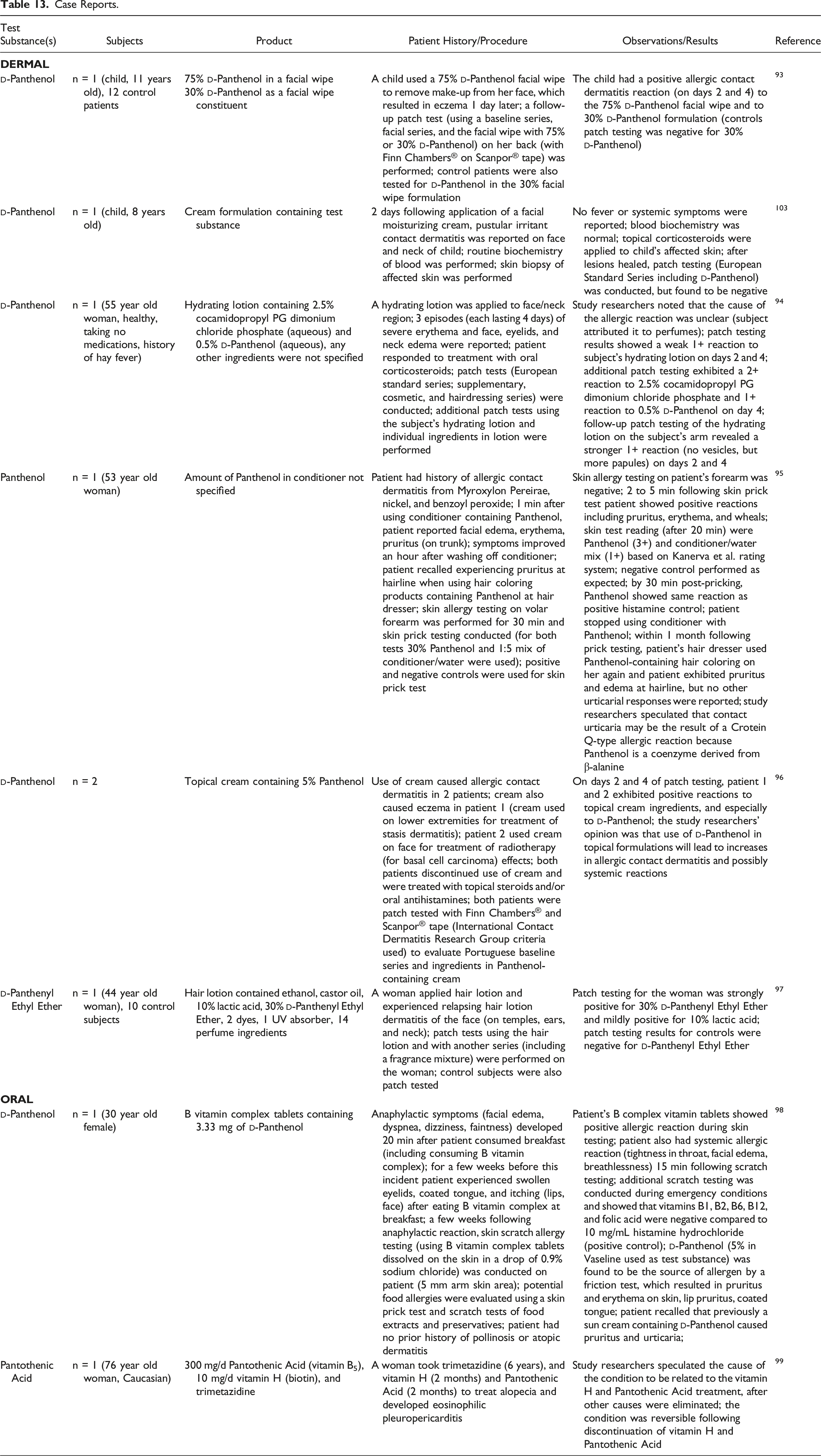
The case reports involving human dermal exposure to Panthenol in lotions or creams include allergic contact dermatitis in a child, caused by the use of a 75%
Included are 2 case reports related to oral exposure. One describes a woman who experienced an anaphylactic reaction attributable to 3.33 mg
Summary
The 7 ingredients included in this safety assessment reportedly function in cosmetics as hair conditioning agents. VCRP data obtained from the FDA in 2017 indicate that the highest reported use frequencies are for Panthenol (5766 uses),
Non-cosmetic uses of Panthenol, Pantothenic Acid, Calcium Pantothenate, and Sodium Pantothenate include nutritional food additives. Panthenol, Calcium Pantothenate, and Sodium Pantothenate are GRAS when used in animal feeds. Calcium Pantothenate is GRAS as a direct food additive for human consumption and is also used in infant formulas.
An in vitro diffusion cell experiment evaluated the penetration of
In human skin, the dermal penetration of [14C]Panthenol (20 mg/mL in ethanol) was evaluated in a Franz (static) diffusion cell experiment. Skin samples were either not stripped or stripped up to 10 times before the addition of test substance. The receptor solution (0.01 mol/l phosphate buffered saline with 5%, polyethylene glycol (v/v)) was collected for up to 60 min post-application. The amount of applied radioactivity measured (after 60 min) in the stratum corneum of skin that was not stripped before application was 84%; 6% and 4% were found in the epidermis and dermis, respectively. For the samples stripped 10 times before application of the test material, the applied radioactivity detected (after 15 min) in the stratum corneum was 72%; 18% and 6.3% were found in the epidermis and dermis, respectively. The receptor fluid for all samples contained negligible amounts of the radioactivity applied.
The penetration of 1-[14C]Panthenol through human fingernails was examined in a nail plate diffusion experiment in vitro. Results indicated that the radioactivity of the formulation base (2% [14C]Panthenol in a 98% nail formulation) was 2 times higher in the interior nail plate and 3 times higher in the cotton ball compared to the radioactivity in the applied aqueous solution (2% [14C]Panthenol in water) after application to the dorsal side of the nail daily for 1 wk.
The in vivo dermal penetration of
The effect of
A dermal exposure experiment in rats treated with
In vivo oral exposure toxicokinetics studies in animals resulted in the following observations: a dose-dependent increase in Pantothenic Acid content in the urine with increasing Calcium Pantothenate dosages (up to 16 mg/kg daily in rat diet for 28 d); by 24 h post-dosing in rats, 85% (5 mg/kg dosage) and 173% (10 mg/kg dosage) more Pantothenic Acid was excreted in urine following Panthenol administration than after Calcium Pantothenate dosing; after radioactive Sodium Pantothenate (location of label not specified) administration in rats (1.6 mg/kg), 27% of radioactivity was detected as urinary Pantothenate by 7 d post-dosing; in dogs, radioactive Sodium Pantothenate (0.8 mg/kg) was found in urine at 24 h post-dosing to be 0.5% of the administered radioactivity and by 7 d 40% of the radioactivity was excreted in urine as the β-glucuronide. In rats dosed daily in the diet for 29 d with up to 3% Calcium Pantothenate, the results indicated the following: a decrease in urinary excretion of vitamins B1 and B6 metabolites; an increase in liver Pantothenic Acid levels with increasing Calcium Pantothenate doses; diarrhea (3% concentration); an adverse effect on nicotinamide metabolism (0%, 1%, and 3% concentrations); and a 1% NOAEL and a 3% LOAEL. An additional test with 5% Calcium Pantothenate (oral administration) caused death in 4 of 5 rats because of severe diarrhea. In rats orally exposed to 23 mg/kg Calcium Pantothenate daily in the diet for 5 - 6 mo a 32% increase in Pantothenic Acid content in the heart and a 25% decrease in Pantothenic Acid content in the liver were observed. In humans, ∼20% of a 100 mg Calcium Pantothenate oral dose was excreted in the urine within 4 h post-administration. In the body,
In acute dermal exposure experiments an LD50 > 3 mL/kg
In a short-term, dermal exposure study, Panthenyl Ethyl Ether (0.125%) in a leave-on hair conditioner was applied (further details regarding application not provided) to the shaved skin of New Zealand White rabbits for 5 d/wk for 28 d. No mortality was reported; diarrhea and soft stools were observed in 1 treated female rabbit periodically throughout the study. In an oral exposure study in rats, the only statistically significant finding was a ∼2-fold increase in basal plasma corticosterone levels in the Pantothenic Acid treated group (0.03% in the diet for 9 wk) as compared to the control group.
A NOAEL of 200 mg/kg/d for
No toxicities were reported when
A maternal and developmental NOAEL ≥ 1000 mg/kg/d for
At concentrations up to 5000 - 10 000 μg/plate,
Other relevant studies included a BALB/c-3T3 cell neoplastic transformation system to which Calcium Pantothenate (50-500 μg/mL) was added several times in a 28-d period to a culture medium either with or without 3-methylcholanthrene (known carcinogen). Results showed that Calcium Pantothenate induced Type III transformed foci, however these effects were considered marginal upon repeat experimentation.
An in vitro test performed using GLP in accordance with OECD TG 437 (Bovine Corneal Opacity and Permeability Test Method for Identifying Ocular Corrosives and Severe Irritants) indicated that
In rabbit eyes,
In clinical studies, positive reactions to a 50%
The case reports associated with dermal exposure to Panthenol or Panthenyl Ethyl Ether include allergic contact dermatitis in a child (75%
Case reports related to oral exposure involve a woman who had an anaphylactic reaction attributable to 3.33 mg
Discussion
The Panel reviewed this safety assessment of Panthenol, Pantothenic Acid and derivatives, and determined that these 7 ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment. The Panel also noted that these ingredients may contain residual, potentially N-nitrosatable, amines as impurities; and, thus cautioned that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed.
The Panel recognized that
Panthenol, Panthenyl Ethyl Ether, Panthenyl Ethyl Ether Acetate, and Panthenyl Triacetate can be metabolized to Pantothenic Acid, an essential nutrient. The Panel recognized that exposures from absorbed quantities of these ingredients are below what would be typical from dietary intake, thereby underscoring the systemic safety of the ingredients. The safety profile is consistent with that of a common dietary constituent and an essential nutrient. Also, the data indicate that Panthenol and it ethers and esters are metabolically converted to Panthenol and Pantothenic Acid, which support the favorable safety profile of the group.
The Panel considered other data available to characterize the potential for Panthenol, Pantothenic Acid, and derivatives to cause systemic toxicity, irritation, sensitization, reproductive and developmental toxicity, and genotoxicity. They noted the low potential for systemic toxicity at high doses in several acute dermal, oral, and inhalation exposure studies, in short-term dermal and oral exposure studies, and in subchronic oral exposure studies. In 6-mo oral exposure studies in multiple animal species, no toxicity was reported. The ingredients were non-toxic in developmental and reproductive toxicity studies. Although no carcinogenicity studies were located in the literature for the ingredients presented in this report, many in vitro studies evaluating genotoxicity were available. Negative results were obtained of multiple Ames tests and mammalian cell gene mutation assays and in chromosomal aberration tests.
The Panel noted that there was minimal potential for the ingredients to cause sensitization and irritation. In dermal exposure studies conducted in animals, Panthenol and Panthenyl Ethyl Ether were non-to-mildly irritating and non-sensitizing. In human subjects, Panthenyl Triacetate was non-irritating and Panthenol was non-to-mildly irritating and non-sensitizing in dermal exposure studies. An in vitro ocular irritation study showed Panthenyl Triacetate to be a non-irritant; in ocular irritation studies conducted in rabbits, Panthenyl Ethyl Ether and Calcium Pantothenate were non-irritating and Panthenol was non-to-slightly irritating. Based on their collective clinical experience, the Panel did not expect these ingredients to be sensitizers or irritants.
The Panel discussed the issue of incidental inhalation exposure from hair sprays, body and hand sprays, fragrances, deodorant sprays, and face powders. These ingredients are reportedly used at concentrations up to 5% in cosmetic products that may be aerosolized and up to 0.5% in other products that may become airborne. The limited data available from animal inhalation studies, including acute exposure data, suggest little potential for respiratory effects at relevant doses. Although particles appear to have reached the lungs in these animal studies, the sizes of the particles used were either clearly within the respirable range (i.e., ≤ 10 μm) or were not reported. The Panel believes that the sizes of a substantial majority of the particles of these ingredients, as manufactured, are larger than the respirable range and/or aggregate and agglomerate to form much larger particles of formulation. Thus, the adverse effects reported using high doses of respirable particles in the inhalation studies do not indicate risks posed by use in cosmetics. The Panel noted that most droplets/particles would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs; in principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, and the overall favorable safety profile of the ingredients in this family, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion

*Not reported to be in current use. Were the ingredients in this group not currently in use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author's Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.