Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 51 triglycerides; 25 of these ingredients were previously reviewed by the Panel, and 26 are reviewed herein for the first time. The majority of the ingredients named in this assessment have several functions, with most reported to function as skin conditioning agents (occlusive or emollient) and/or viscosity increasing agents in cosmetics; some are also reported to function as a fragrance or solvent. The Panel reviewed relevant new data, including frequency and concentration of use, and considered the data from previous reports. The Panel concluded the 51 triglycerides reviewed in this report are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Keywords
INTRODUCTION
The Panel published the Final Report on the Safety Assessment of Trihydroxystearin in 2000. 1 Based on the available animal and clinical data, which included summary data from the safety assessments of Hydroxystearic Acid and Glyceryl Stearate and Glyceryl Stearate SE, the Panel concluded that Trihydroxystearin is safe as used in cosmetics. In 2015, the Panel re-evaluated the safety of Hydroxystearic Acid and Glyceryl Stearate and Glyceryl Stearate SE, reaffirming that Hydroxystearic Acid is safe as a cosmetic ingredient in the present practices of use and concluding that Glyceryl Stearate and Glyceryl Stearate SE are safe in the present practices of use and concentration. 2 (In 1982, the conclusion issued for Glyceryl Stearate and Glyceryl Stearate SE was safe for topical application to humans. 3 )
In accordance with its procedures, the Panel evaluates the conclusions of previously-issued reports every 15 years, and it has been at least 15 years since these assessments have been issued. Because each report was reviewed 15+ years ago and they all comprise triglycerides, i.e., fatty acid triesters of glycerin, and because the collection of these ingredients in one report enables the assembly of reinforcing and complementary test data, the Panel determined these reports should be re-reviewed together in one document; this family is referred to as the triglycerides.

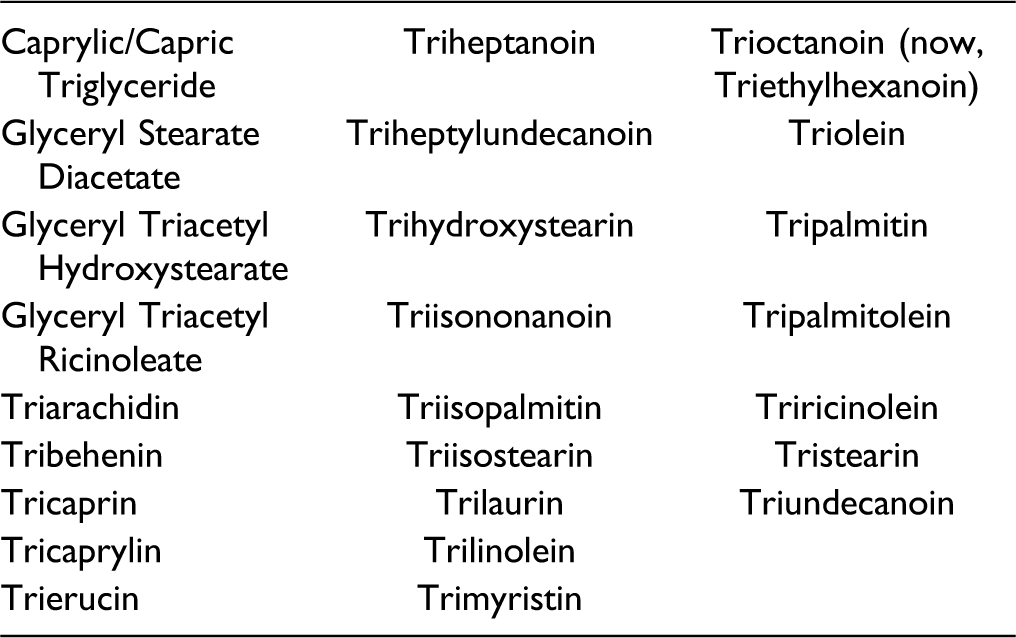
Triglycerides Included in this Report.
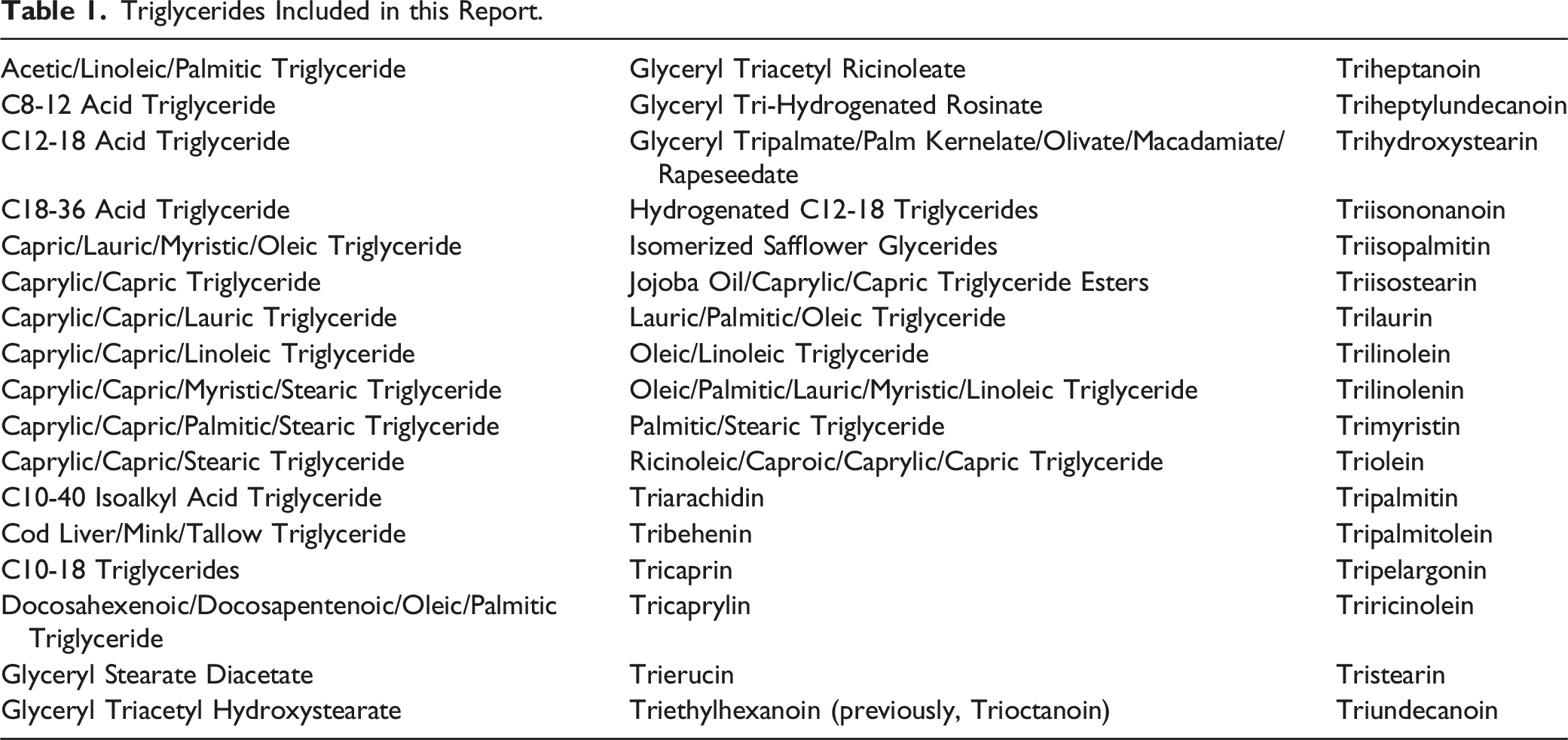
Definitions, Idealized Structures, and Reported Functions of the Ingredients in this Safety Assessment. (7; CIR Staff).
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties. For complete and detailed information, please refer to the original documents, which are available on the CIR website (http://www.cir-safety.org/ingredients). Additionally, the Discussions from the Trihydroxystearin (2000) and Trilaurin (2001) assessments are also included in this document.
Previously Reviewed Components and Related Glyceryl Esters.

Finally, much of the new data included in this safety assessment was found on the European Chemicals Agency (ECHA) website. 10 Please note that the ECHA website provides summaries of information generated by industry, and it is those summary data that are reported in this safety assessment when ECHA is cited.
Chemistry
Definition and Structure
The definitions and structures of the ingredients included in this triglyceride group are provided in Table 2 and are presented in order of increasing chain length, subdivided by chain type. (Toxicity data are presented in the same order.)
Each of the ingredients in this report is a triglyceride; triglycerides are the fatty acid triesters of glycerin. Subsequently, each of the ingredient structures in this report contains a glycerin core, tri-substituted with fatty acid residues (Figures 1 and 2). Triglycerides, wherein each “RC(O)-” is a fatty acid residue. Tricaprylin (the triester of caprylic acid with glycerin).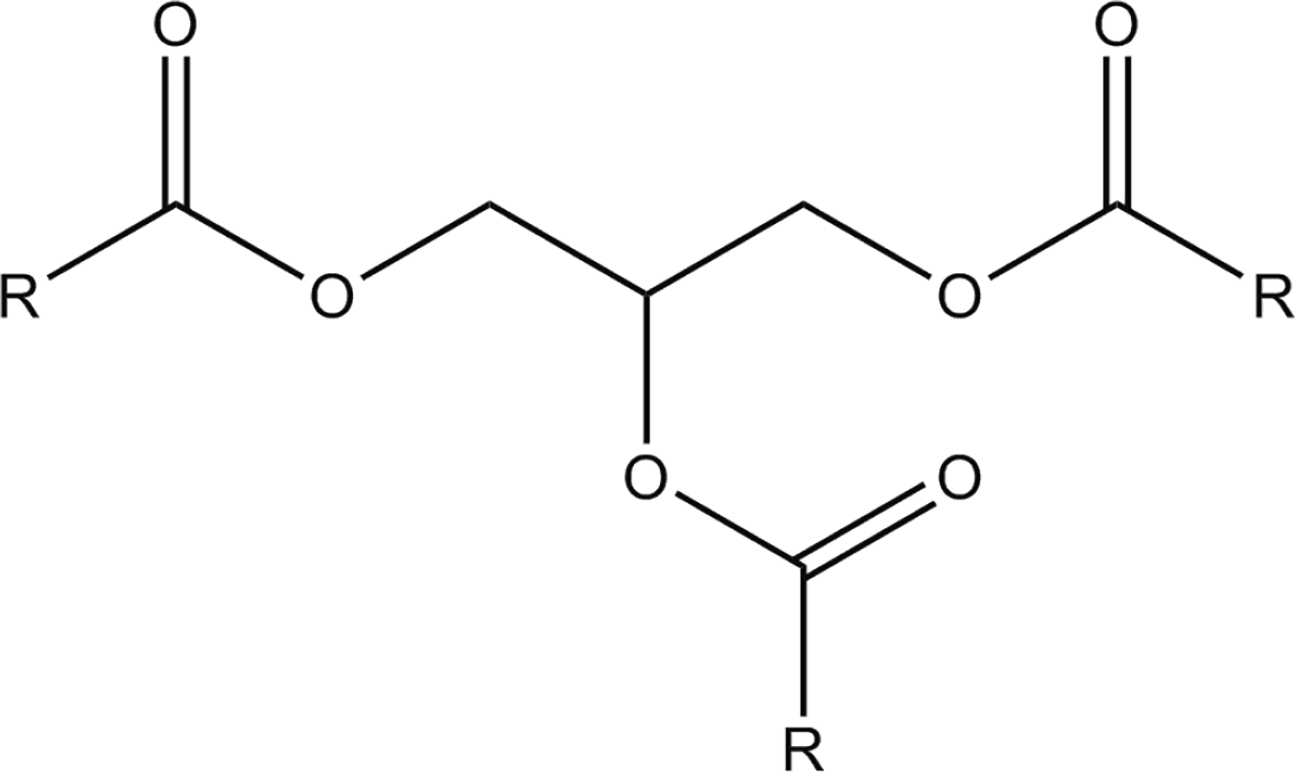
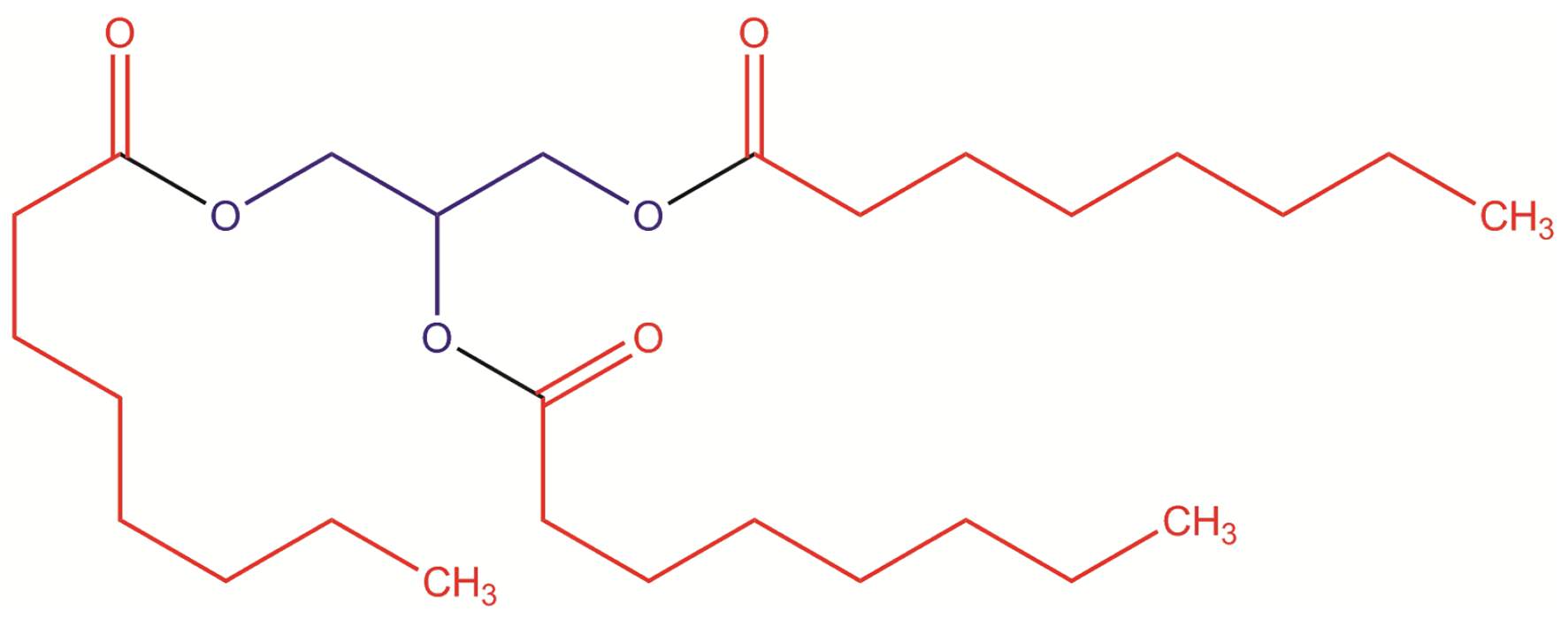
For example, Tricaprylin is the triester of caprylic acid with glycerin.
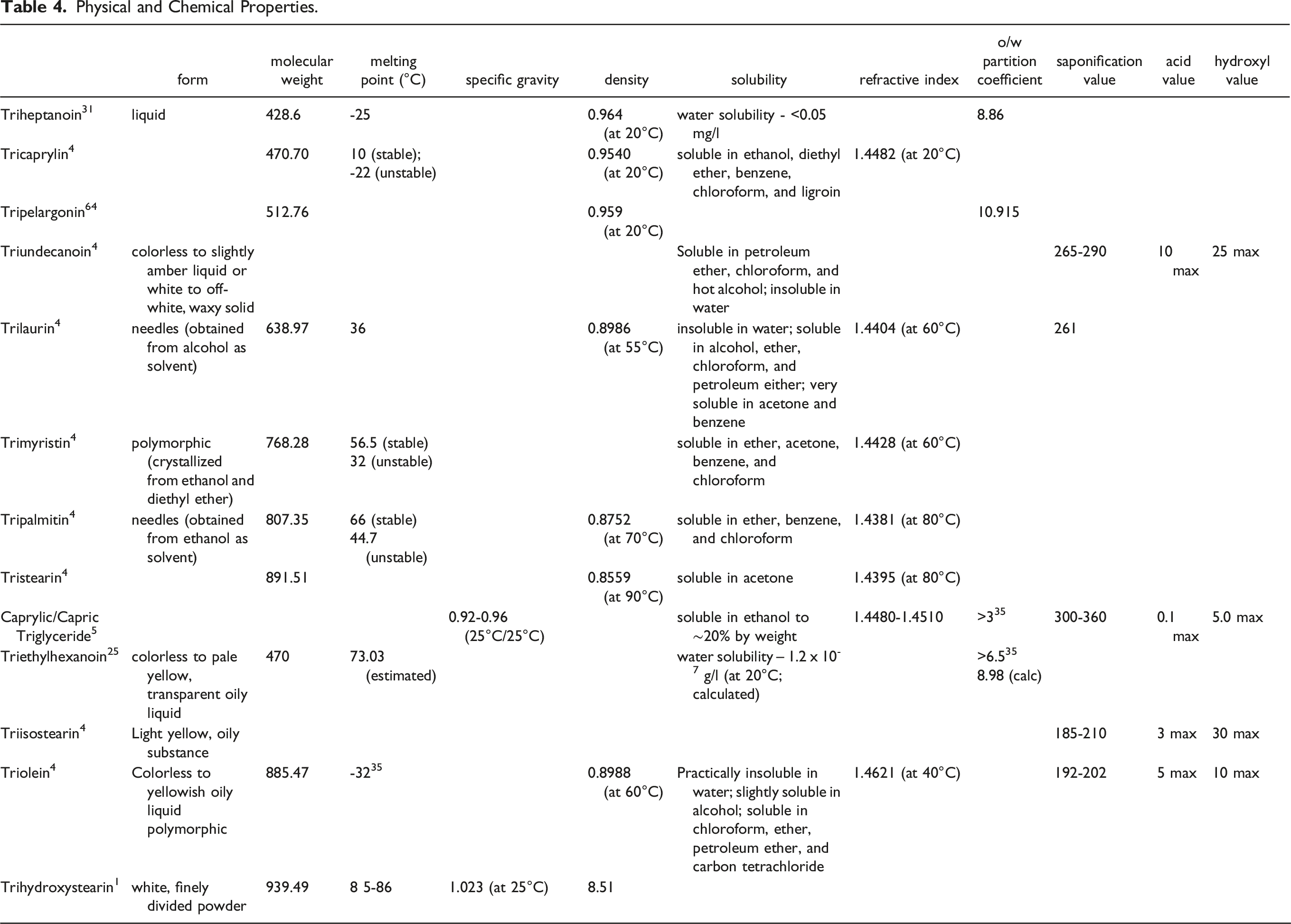
Physical and Chemical Properties
Physical and Chemical Properties.
Methods of Manufacture
One method of production of Trihydroxystearin involves the hydrogenation of castor oil, in the presence of the reagent nickel, at a temperature of 200°C. Another method of production is the reduction of triricinolein. 1
Trilaurin may be produced by reacting glycerin with lauric acid or glycerin with lauroyl chloride (reagent: pyridine or quinoline). 4 The reaction of lauric acid with glycerin is another method of production. Triolein may be prepared by the esterification of oleic acid. Tripalmitin can be prepared from glycerin and palmitic acid in the presence of either Twitchell reagent or trifluoroacetic anhydride. Tristearin may be prepared from stearic acid and glycerin in the presence of Al2O3. Triundecanoin is produced by esterification of undecanoic acid and glycerin. The undecanoic acid is produced from castor oil, which is hydrolyzed to fatty acids and subjected to thermal degradation and fractionation. The resulting undecenoic acid is transformed to undecanoic acid and re-esterified to the glycerin moiety. Deodorization, the final step, is accomplished using steam to remove components that give rise to unwanted flavors and odors.
Caprylic/Capric Triglyceride is manufactured by hydrolyzing coconut oil, removing the free glycerin, and separating the medium chain length fatty acids by fractional distillation. 5 The acids are then blended in the proper ratio and re-esterified with glycerin.
Triglycerides (general)
Some of the triglycerides are produced synthetically via classical Fischer type esterification methods (i.e., reaction of carboxylic acids with glycerin to produce carboxylic esters), although the reaction may be promoted by acid or base catalysis, or by the use of an acid chloride. However, some of these ingredients may be natural sourced and produced by transesterification (i.e., exchange of acid moieties to create a different ester product). For example, the triglycerides in natural oils can be reacted with intended length fatty acids to produce new triglycerides.
The following are method of manufacture schemes for Caprylic/Capric Triglyceride (medium-chain triglycerides (MCT); terminology used in a FDA foods Generally Recognized as Safe (GRAS) notification, defined as triglycerides with alkyl chain lengths from 8 to 10 carbons long) (Figure 3)
11
and medium- and long-chain triacylglycerol (MLCT)-oil (terminology used in a FDA foods GRAS notification, defined as triglycerides with alkyl chain lengths from 8 to 24 carbons long) (Figure 4):
12
Caprylic/Capric Triglyceride (MCT) production scheme. MLCT production scheme.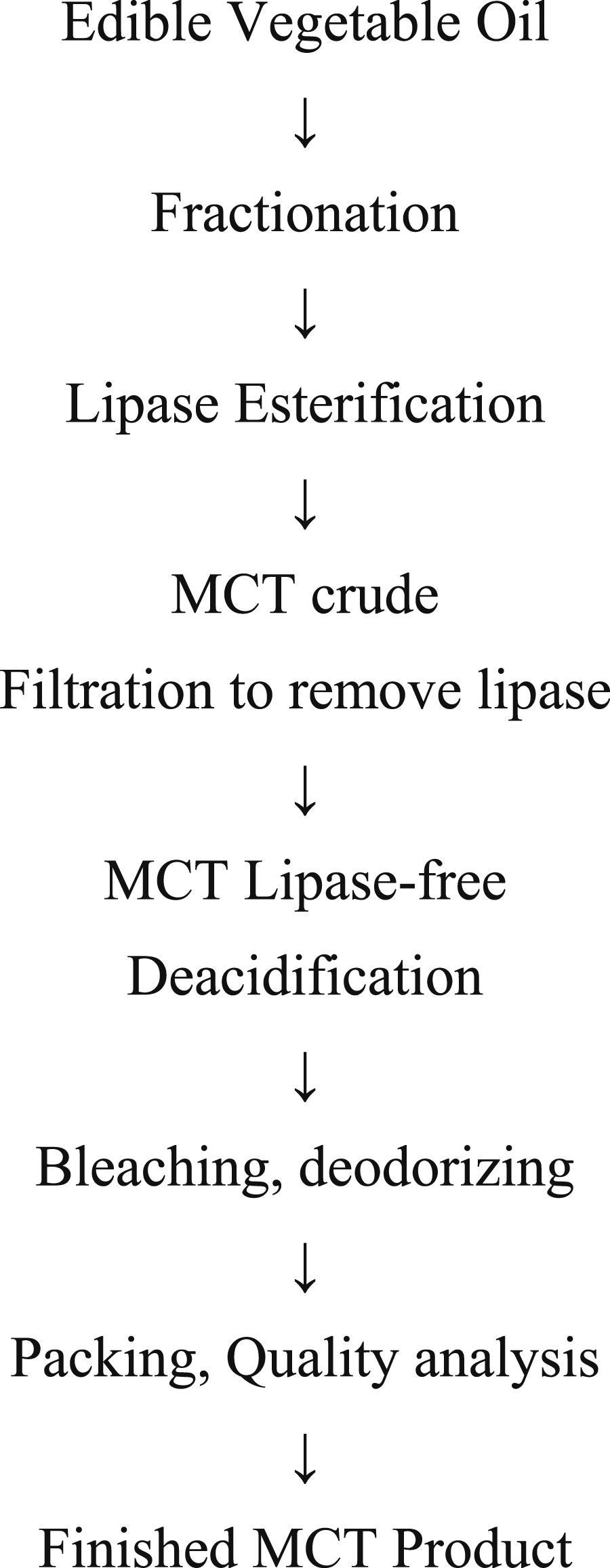

Impurities
Triundecanoin contains no impurities or residues of catalysts or solvents. 4 1,4-Dioxane, ethylene oxide, free amines, and nitrosamines are not added or formed during the production process. Furthermore, volatile compounds are effectively removed, by the deodorization process, below detection limits (0.1 ppm). The deodorization process also has removed any organochlorine or organophosphorus pesticides that may be present in the crude oil used in the production process. It is also important to note that the total content of polycyclic aromatic hydrocarbons (PAHs), if present in the crude oil, is reduced below 10 ppb. Additionally, aflatoxins, if present in the raw materials, are reduced below detection limits (0.5 ppb) by neutralization and bleaching.
The only known impurities of Caprylic/Capric Triglyceride are approximately 300 ppm free fatty acids and as much as 0.2% glycerin. 5 The relatively low iodine number 5, which is determined in an arbitrary but standard method, indicates very little unsaturated material present.
Use
Cosmetic
The safety of the cosmetic ingredients addressed in this safety assessment is evaluated based on data received from the US FDA and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA VCRP database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
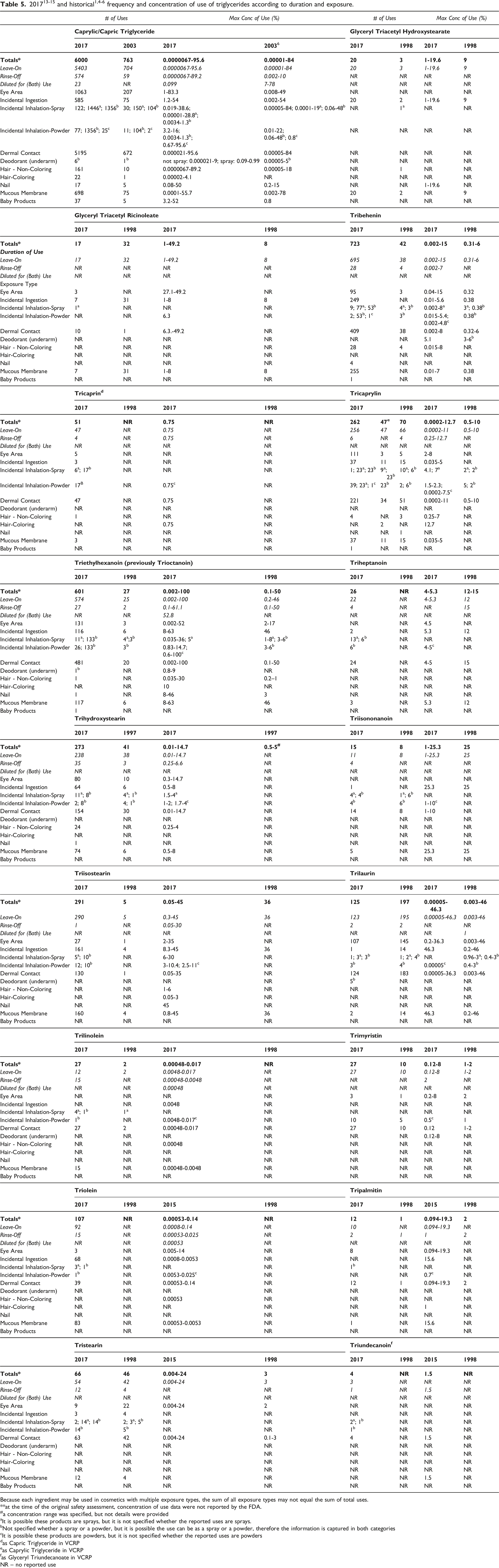
201713-15 and historical1,4-6 frequency and concentration of use of triglycerides according to duration and exposure.
Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
at the time of the original safety assessment, concentration of use data were not reported by the FDA.
#a concentration range was specified, but not details were provided
aIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories
cIt is possible these products are powders, but it is not specified whether the reported uses are powders
das Capric Triglyceride in VCRP
eas Caprylic Triglyceride in VCRP
fas Glyceryl Triundecanoate in VCRP
NR – no reported use
Frequency 13 and concentration of use14,15 previously unreviewed triglycerides.
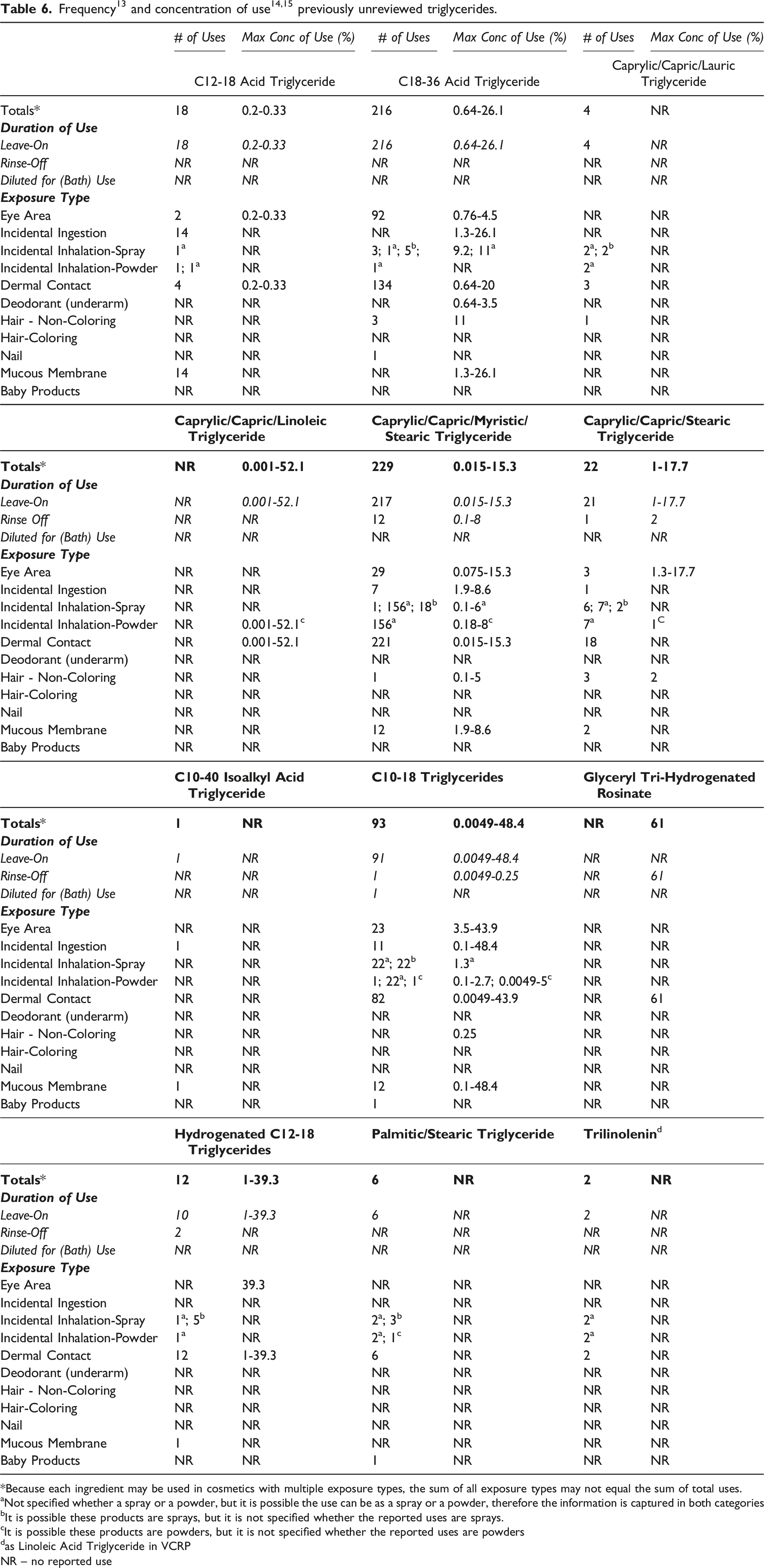
Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories
bIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
cIt is possible these products are powders, but it is not specified whether the reported uses are powders
das Linoleic Acid Triglyceride in VCRP
NR – no reported use
Use concentration survey data were collected in 2015/2016 (and updated in 2017) for some of ingredients, 14 and in 2017 for the remaining ingredients. 15 The results indicate that Triethylhexanoin has the highest maximum use concentration in leave-on formulations, with concentrations of 100% reported for face and neck formulations and 63% in lipstick formulations (Table 5). Caprylic/Capric Triglyceride has the next highest maximum use concentration in leave-ons, with concentrations of 95.6% in face and neck products.
Approximately half of the ingredients included in this safety assessment have been reviewed previously by the Panel. The frequency and maximum concentrations of use for the majority of these ingredients has increased when compared to the previous review. The most remarkable increase is in the frequency of use of Caprylic/Capric Triglyceride; in 2003, this ingredient was reported to be used in 763 formulations and in 2017, it is reported to be used in 6000 formulations.5,6 Concentrations of use have also increased.6,14 In 2003, the maximum leave-on concentration of use for this ingredients was 84%, it is now reported to 95.6%; maximum concentrations of use increased for eye area, non-coloring hair, hair coloring, nail, and baby product formulations. The increase in baby products was quite notable, increasing from 0.8% to 52%.
In some cases, reports of use were received from the VCRP, but no concentration of use data were provided. For example, Trilinolenin is reported to be used in 2 formulations, 16 but no use concentration data were provided. In other cases, no uses were reported to the VCRP, but a maximum use concentration was provided in the industry survey. For example, Caprylic/Capric/Linoleic Triglyceride was not reported in the VCRP database to be in use, but the industry survey indicated that it is used at concentrations up to 52.1% in body and hand product formulations. 15 It should be presumed that Caprylic/ Capric/Linoleic Triglyceride is used in at least one cosmetic formulation for each category for which it is reported to be used.
Some of the triglycerides are used at relatively high concentrations in products that can be used near the eye, can possibly be ingested, or come in contact with mucous membranes; for example, Caprylic/Capric Triglyceride is used at up to 83.3% in eye lotions, and Triethylhexanoin is used at up to 63% in lipstick formulations. Additionally, some of these ingredients are used in cosmetic sprays and powders and could possibly be inhaled; for example, Caprylic/Capric Triglyceride and Triethylhexanoin are reported to be used at maximum concentrations of 38% in spray body and hand formulations and 36% in perfumes, respectively, and 16% and 14.7%, respectively, in face powders. In practice, most of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles < 10 µm compared with pump sprays.17,18 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.19,20 Conservative estimates of inhalation exposures to respirable particles during the use of loose-powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.21-23 Caprylic/Capric Triglyceride is used at up to 0.99% in spray deodorant formulations. There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 20 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
All the triglycerides described in this safety assessment (and listed in the
Non-Cosmetic
Trihydroxystearin has been used as a thickening agent for peanut butter. 1 FDA has listed the following indirect food additive uses in the Code of Federal Regulations (CFR): components of adhesives (21CFR 175.1 05), components of resinous and polymeric coatings (21 CFR 175.300), components of paper and paperboard in contact with aqueous and fatty foods (21 CFR 176.170), components of paper and paperboard in contact with dry food (21 CFR 176.180), defoaming agents used in the manufacture of paper and paperboard (21 CFR 176.21 0), cellophane (21CFR 177.1200), closures with sealing gaskets for food containers (21 CFR 177.1210), polyester resins cross-linked (21 CFR177.2420), and textiles and textile fibers (21 CFR 177.2800).
Trihydroxystearin is among the inert ingredients that are exempt from the requirement of a tolerance under the Federal Food, Drug, and Cosmetic Act when used in pesticide formulations that are applied to crops. 1
Trilaurin has been detected in pharmaceutical excipients. 4 Tricaprylin has been used as an energy source for burn patients and for patients having difficulty digesting long-chain fatty acids. Tristearin has been approved for use as a direct food additive (21 CFR 172.811). Additionally, the following glyceryl triesters have been approved for use as components of articles intended for use in producing, manufacturing, packing, processing, preparing, treating, packaging, transporting, or holding food (i.e., use as indirect food additives): Trilaurin, Trimyristin, Triolein, Tripalmitin, Tristearin (21 CFR 177.2800), and Glyceryl Triacetyl Hydroxystearate (21 CFR 178.3505).
The following non-cosmetic uses of Tristearin have been reported: soap, candles, candies, adhesive pastes, metal polishes, waterproofing paper, textile sizing, leather stuffing, and manufacture of stearic acid. 4
Letters issued by the FDA have attested to the safety of Caprylic/Capric Triglyceride when used as a food additive. 5 In addition, it has also been marketed for consumption since 1962 as a nutritional supplement and blood lipid lowering agent. It has also been suggested for use in enteric drugs and rectal suppositories and as a vehicle for topically applied pharmaceuticals.
C10-18 Triglycerides is approved for use as direct multipurpose food additives (21CFR172.861).
Caprylic/Capric Triglyceride and Triglycerides (general)
The FDA received a GRAS notification request for triglycerides (C8-C24) for use as a food ingredient, such that the total daily consumption would not exceed 31 g/d. 12 The FDA responded that the tailored triglycerides ingredient (12% Caprylic/Capric Triglyceride) is GRAS under the intended conditions of use as an oil in home cooking, salad dressings, vegetable-oil spreads, and frozen dinners (including meat and poultry). 26 The agency has not, however, made its own determination regarding the GRAS status of the subject use of the tailored triglycerides (12% Caprylic/Capric Triglyceride) ingredient.
Caprylic/Capric Triglyceride, is a component of a homogenous lipid emulsion approved for intravenous (i.v.) infusions indicated for use in adults as a source of calories and essential fatty acids for parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. 27 The lipid content of the infusion is 0.20 g/ml, and comprises a mixture of soybean oil, Caprylic/Capric Triglyceride, olive oil, and fish oil; recommended dosing is 1 to 2 g/kg/d, not exceeding 2.5 g/kg/d.
Toxicokinetic Studies
Dermal Penetration
In mice and guinea pigs, little skin penetration was observed.
4
In the mice, [14C]Triolein (undiluted or in hydrophilic ointment) did not penetrate into the body organs of mice, and the oil remained localized at the application site at 48 h post application
Penetration Enhancement
Tricaprylin enhanced the skin penetration of drugs in vivo (Wistar rats) and in full-thickness skin from hairless mice. 4 The skin penetration enhancement of drugs in the presence of Triolein has been reported.
Absorption, Distribution, Metabolism, and Excretion
Metabolism data indicate that most triglycerides (or glyceryl triesters) are split into monoglycerides, free fatty acids, and glycerin in the small intestine and absorbed by the intestinal mucosa 4
When absorbed from the digestive tract, Caprylic/Capric Triglyceride is hydrolyzed, and the fatty acids are catabolized to C2-fragments which may be further metabolized either to CO2 or to form long-chain fatty acids. 5 Caprylic/Capric Triglyceride can undergo hydrolysis by enzymatic or chemical means to produce free fatty acids, partial glycerides, and glycerin. The free fatty acids may, in turn, undergo enzymatic β-oxidation. β-Oxidation of caprylic acid forms β-ketocaprylic acid and can be further oxidized to yield acetic acid and C6-acid.
Caprylic/Capric Triglyceride and Triglycerides (general)
Oral absorption and metabolism of foods containing long-chain triglycerides (LCT) mixtures (alkyl chain lengths greater than 12 (C>12)) differ from those containing Caprylic/Capric Triglyceride. 28 C>12 are degraded by salivary, intestinal and pancreatic lipases into two fatty acids and a monoacyl glycerol, whereas, Caprylic/Capric Triglyceride is degraded by the same enzymes into three fatty acids and the simple glycerol backbone. Caprylic/Capric Triglyceride is readily absorbed from the small intestine directly into the bloodstream and transported to the liver for hepatic metabolism, while C>12 are incorporated into chylomicrons and enter the lymphatic system. Caprylic/Capric Triglyceride is readily broken down to carbon dioxide and two-carbon fragments, while C>12 are re-esterified to triacylglycerols and either metabolized for energy or stored in adipose tissue.
Tripelargonin and Triethylhexanoin
The primary metabolite of Triethylhexanoin, along with glycerol and monoglycerides, is 2-ethylhexanoic acid. 25
Groups of 5 newborn Rhesus (
Groups of 8 New Zealand male rabbits were given a Tripelargonin/LCT (7/3 wt/wt) emulsion via a total parenteral nutrition (TPN) infusion regimen 7 h/d for 11 d. 30 The 3HB concentrations were significantly decreased, and plasma concentrations of propionic, acetic, butyric, and valeric acids were significantly increased. Following overnight fasting on days 9 and 12, fatty glycerol concentrations were statistically significantly increased compared to controls that were fed a standard diet, and on day 12, fasted rabbits were found to have increased triglyceride and non-esterified fatty acid levels.
Toxicological Studies
Acute Toxicity Studies
In acute oral toxicity studies in which Trihydroxystearin was tested using albino rats, the LD50 was not achieved at a dose of 5 g/kg and no deaths were reported. 1
Acute oral LD50 values range from 5 g/kg in mice (Tribehenin) to > 20 g/kg in rats (Tristearin). 4 In other acute oral toxicity studies, Triethylhexanoin was not toxic following oral administration to male mice at a dose of 50 ml/kg, and Triisostearin did not induce toxicity in rats at a dose of 2 g/kg.
Acute oral LD50 values for Caprylic/Capric Triglyceride were > 25 ml/kg in mice and > 5 g/kg in rats. 5 Male rats and guinea pigs in groups of ten each were exposed for 6 h in a 40 l chamber containing an aerosol of Caprylic/Capric Triglyceride at a nominal concentration of 28.1 µl/l of air. The fraction of the aerosol with particles small enough to be inhaled into the lung, i.e., with a diameter of 5 µm or less, represented 1.97 µl/l of the test substance. No adverse effects were observed.
Acute Toxicity Studies.
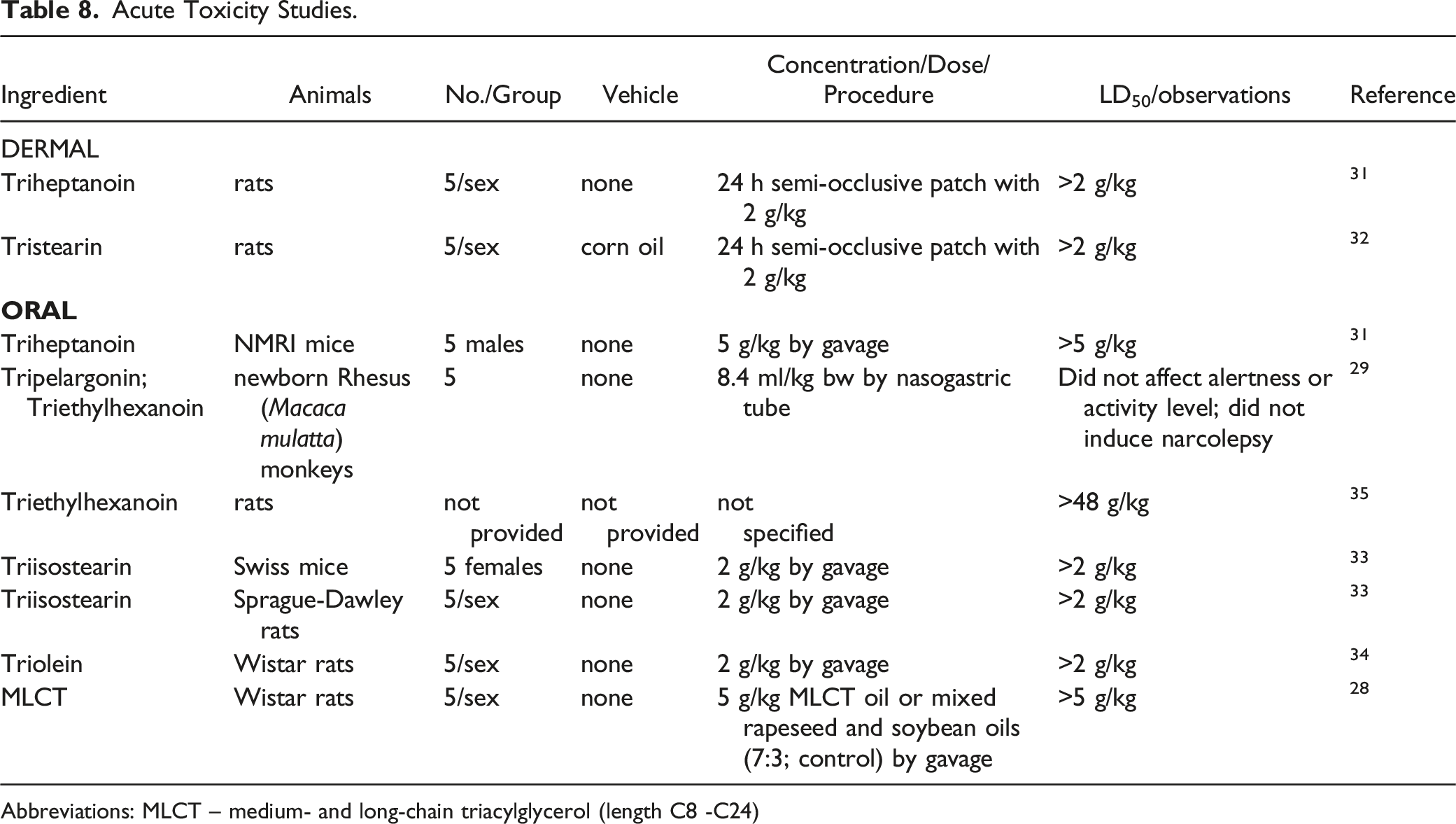
Abbreviations: MLCT – medium- and long-chain triacylglycerol (length C8 -C24)
The dermal LD50 in rats was > 2 g/kg (the highest dose tested) for both Triheptanoin
31
and Tristearin.
32
The oral LD50 was > 2 g/kg for Triisostearin in mice and rats,
33
> 2 g/kg Triolein in mice,
34
> 5 g/kg Triheptanoin in mice, and > 48 g/kg Triethylhexanoin
35
in rats. The oral LD50 of an MLCT oil was > 5 g/kg in rats.
28
A single dose of 8.4 ml/kg bw Tripelargonin and Triethylhexanoin, administered via NG tube, did not affect activity level or induce narcolepsy in newborn Rhesus (
Short-Term Toxicity Studies
The short-term oral administration of Trilaurin, Tristearin, or Triolein to weanling rats did not result in gross or microscopic lesions. 4 However, in another short-term study, significant differences in hematological and clinical chemistry parameters as well as organ weights were noted after administration of Tricaprylin to male and female Wistar rats.
No signs of toxicity were observed in rabbits following 4 wk of applications of a tanning butter formulation containing 22% Caprylic/Capric Triglyceride at a dose of 2 g/kg, five times/wk for 4 wk, to intact and abraded skin. 5 Two groups of 10 rats were dosed by gavage with 7.6 or 21.3 ml/kg undiluted Caprylic/Capric Triglyceride daily for 30 d. 5 With the exception of a few gross observations made in the high-dose group in the first week of the study, no adverse effects were observed.
Short-Term, Subchronic, and Chronic Oral Toxicity Studies.
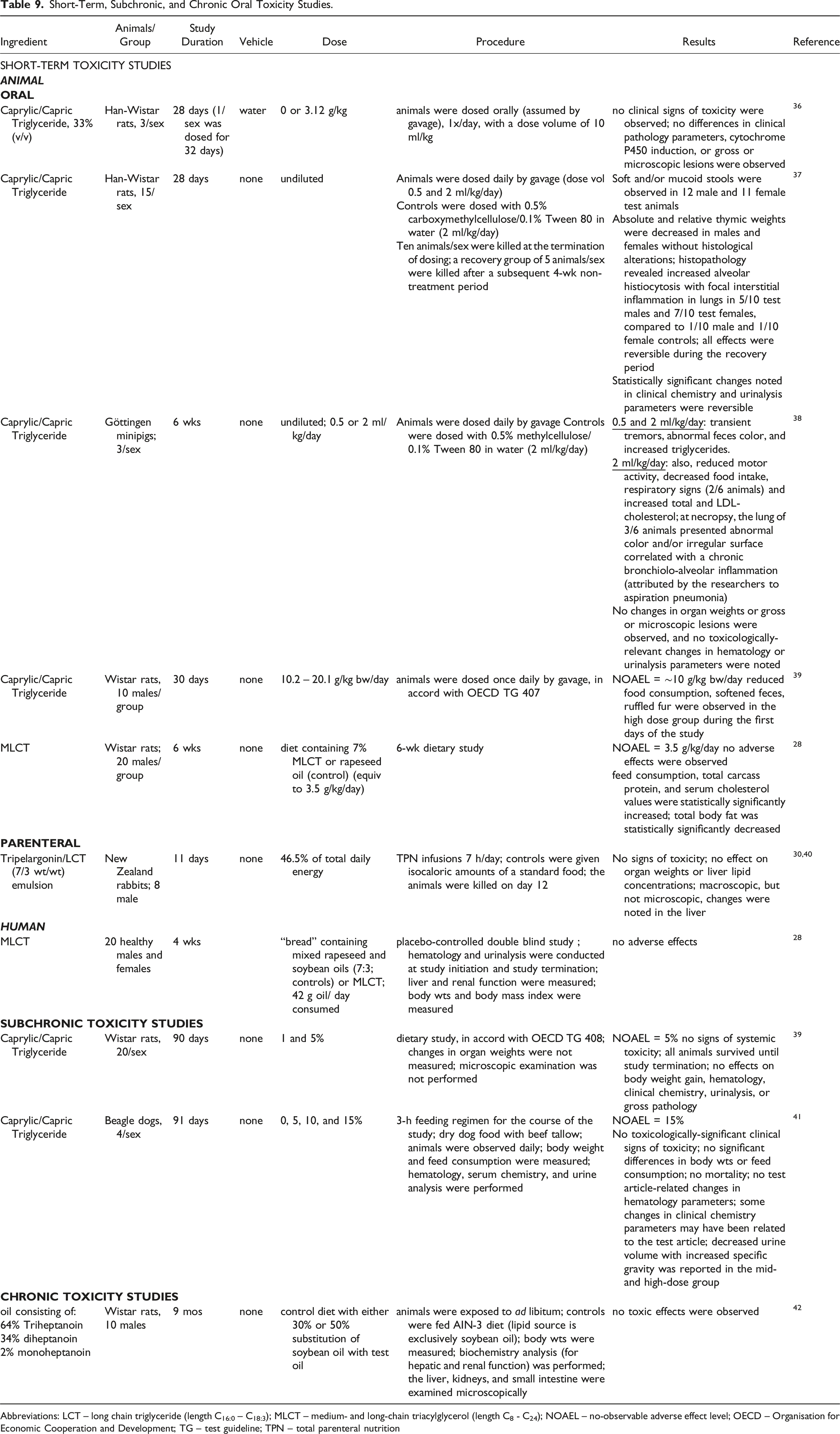
Abbreviations: LCT – long chain triglyceride (length C16:0 – C18:3); MLCT – medium- and long-chain triacylglycerol (length C8 - C24); NOAEL – no-observable adverse effect level; OECD – Organisation for Economic Cooperation and Development; TG – test guideline; TPN – total parenteral nutrition
In 28-d gavage studies in Han-Wistar rats, dosing with 3.12 g/kg of 33% Caprylic/Capric Triglyceride did not produce any signs of toxicity, 36 but undiluted test material produced some gastrointestinal effects, decreased thymic weight, caused inflammation in the lungs, and resulted in changes in some clinical pathology parameters. 37 These changes were reversible. In Göttingen minipigs, clinical signs of toxicity were observed with 0.5 and 2 ml/kg/d Caprylic/Capric Triglyceride administered by gavage; no changes in organ weights or gross or microscopic lesions were observed. 38 In rats, a no-observed-adverse-effect-level (NOAEL) of 10 mg/kg bw/d was reported in a 30 d study with Caprylic/Capric Triglyceride, 39 and a NOAEL of 3500 mg/kg/d was reported with MLCT. 28 In a human study, no adverse effects were observed in a placebo-controlled double-blind study in which healthy subjects ingested 42 g/d MLCT. 28
No adverse effects were observed in a study in which rabbits were given a Tripelargonin/LCT emulsion via a TPN infusion regimen for 7 h/d for 11 d.30,40
Subchronic Toxicity Studies
Application of a perfumed skin softener formulation containing 4% Caprylic/Capric Triglyceride to the shaved skin of female rats at a dose of 2 ml/kg 5 d/wk for 13 wk did not produce any toxic effects. 5 No toxic effects were noted in a 3-mo feeding study of 1 and 5% Caprylic/Capric Triglyceride in the diet of rats.
The subchronic toxicity studies summarized below are described in Table 9.
Three-month feeding studies were performed with Caprylic/Capric Triglyceride in rats 39 and dogs. 41 The NOAELs were 5% and 15%, respectively, and no toxicologically-relevant signs of toxicity were observed at the highest doses.
Chronic Toxicity Studies
No significant differences were found in growth rate or the incidence of lesions between groups of rats fed a mixture containing 0.0002% Trilaurin for 2 yr and controls. 4 In another chronic study, cardiac lipidosis and/or focal fibrosis was observed in rats fed a basal diet consisting of 30 cal% Trierucin for 24 wk. Renal tubular dilatation, proteinaceous casts, or fibrosis were also reported. When the chronic oral toxicity of Tricaprylin was evaluated using groups of male rats, significant reductions in hematological/clinical chemistry parameters (10 ml/kg group) and significant increases in the liver (2 ml/kg) and adrenal gland weights (2 and 10 ml/kg) were noted after 26 wk of dosing. Few lesions in the kidneys, myocardium, and aorta were noted when Tricaprylin was tested in another chronic oral toxicity study. 4
In studies in which rats were fed a diet containing 19.6% of a MCT composed of about 75% caprylic acid and 25% capric acid for 47 wk or an MCT at 20% in the diet, for 1 yr, nutritional effects resulting from long-term consumption of this ingredient were observed, but no effects were interpreted as adverse or toxic. 5
In a 9-mo feeding study, an oil containing 64% Triheptanoin was not toxic in rats Table 9. 42
DEVELOPMENTAL AND REPRODUCTIVE TOXICITY (DART) STUDIES
Tricaprylin (10 ml/kg) was not teratogenic in mice or rats when administered orally 4 In another study on reproductive effects, a uterine injection on 0.1 ml Tricaprylin was effective in producing fusion of the endometrial epithelium (symplasma formation) and decidualization of the stroma in pseudopregnant New Zealand white rabbits. The oral administration of 4750 mg/kg/d Triethylhexanoin on days 6 - 13 of gestation to mice did not result in any significant differences in indices of potential developmental toxicity (i.e., litter size, birth weight, and neonatal growth and survival to postnatal day 3) between test and control groups. Test results for 291 fetuses from various strains of mice injected intraperitoneally with 0.5 mmol/kg bw Triethylhexanoin; was the vehicle control in a teratogenicity study] on days 8 and 12 of gestation indicated various kinds of eye abnormalities in 6.2% of the fetuses.
In a reproduction study, young adult male and female rats were fed a balanced diet containing 19.6% of a triglyceride of 75% caprylic and 25% capric acid for 3 wk before mating. 5 Litter size and birth weight of the test animals were similar to those of rats on conventional or low fat diets, but mortality during lactation was somewhat higher, and there was less weight gain due to a smaller volume of milk secreted. After weaning, the F1 generation was fed as the F0 generation had been and showed a weight gain comparable to that of control rats.
Tricaprylin
In a study evaluating the developmental toxicity of trichloroacetonitrile in which Tricaprylin was used as a vehicle, a possibly biologically significant effect (increased resorptions, reduced fetal weight, and anomalies) was observed in the Tricaprylin control group when compared to the water control group. 43 Therefore, the developmental toxicity of trichloroacetonitrile was reexamined using Tricaprylin and corn oil as vehicles. Groups of 20 gravid Long-Evans rats were dosed by gavage on days 6-18 of gestation with 15 mg/kg/day trichloroacetonitrile in Tricaprylin and 15 - 75 mg/kg/day trichloroacetonitrile in corn oil; vehicle control groups were dosed with Tricaprylin, corn oil only, and water. The dosing volume was 10 ml/kg. All dams were killed on day 20 of gestation.
Statistically significant difference in some parameters was observed in the Tricaprylin control group compared to the water and/or corn oil control groups. There was a statistically significant increase in the percent implantation loss in the Tricaprylin (only) group as compared to both the water and corn oil controls, and the total implants/litter was statistically significantly less when compared to the corn oil, but not the water, control group. Also, there were statistically significant decreases in fetal body weights and crown-rump length in the Tricaprylin control group as compared to the water and corn oil control groups. There was no statistically significant difference in the incidence of fetal anomalies among the three control groups. In the dams, the maternal average kidney weight was statistically significantly increased in the Tricaprylin controls when compared to the water and corn oil controls; no effect on liver or spleen weight was reported.
The study authors postulated that the differences observed between the Tricaprylin group and the other two control groups may be attributable to potential changes in nutritional status. Dams of the Tricaprylin group gained significantly less weight than those of the corn oil group during days 15 - 18 of gestation. However, food and water consumption were not monitored. The study authors also stated that the differences in reproductive parameters could be due to normal variation for Long-Evans rats.
Additionally, the developmental toxicity of trichloroacetonitrile appeared to be vehicle-dependent; developmental effects caused by trichloroacetonitrile were seen at higher doses when administered in corn oil compared to those seen when Tricaprylin was used as the vehicle. The study authors suggested that trichloroacetonitrile and Tricaprylin “appear to interact in some way to potentiate effects of the cardiovascular system.” The adverse effects of trichloroacetonitrile in Tricaprylin were seen at doses as low as 1 mg/kg/d and included a number of different kinds of heart defects.
GENOTOXICITY STUDIES
In Vitro
Ames test results indicated that Trihydroxystearin was not mutagenic to the following
In the Ames test, Tricaprylin was mutagenic in one of four
Genotoxicity studies.
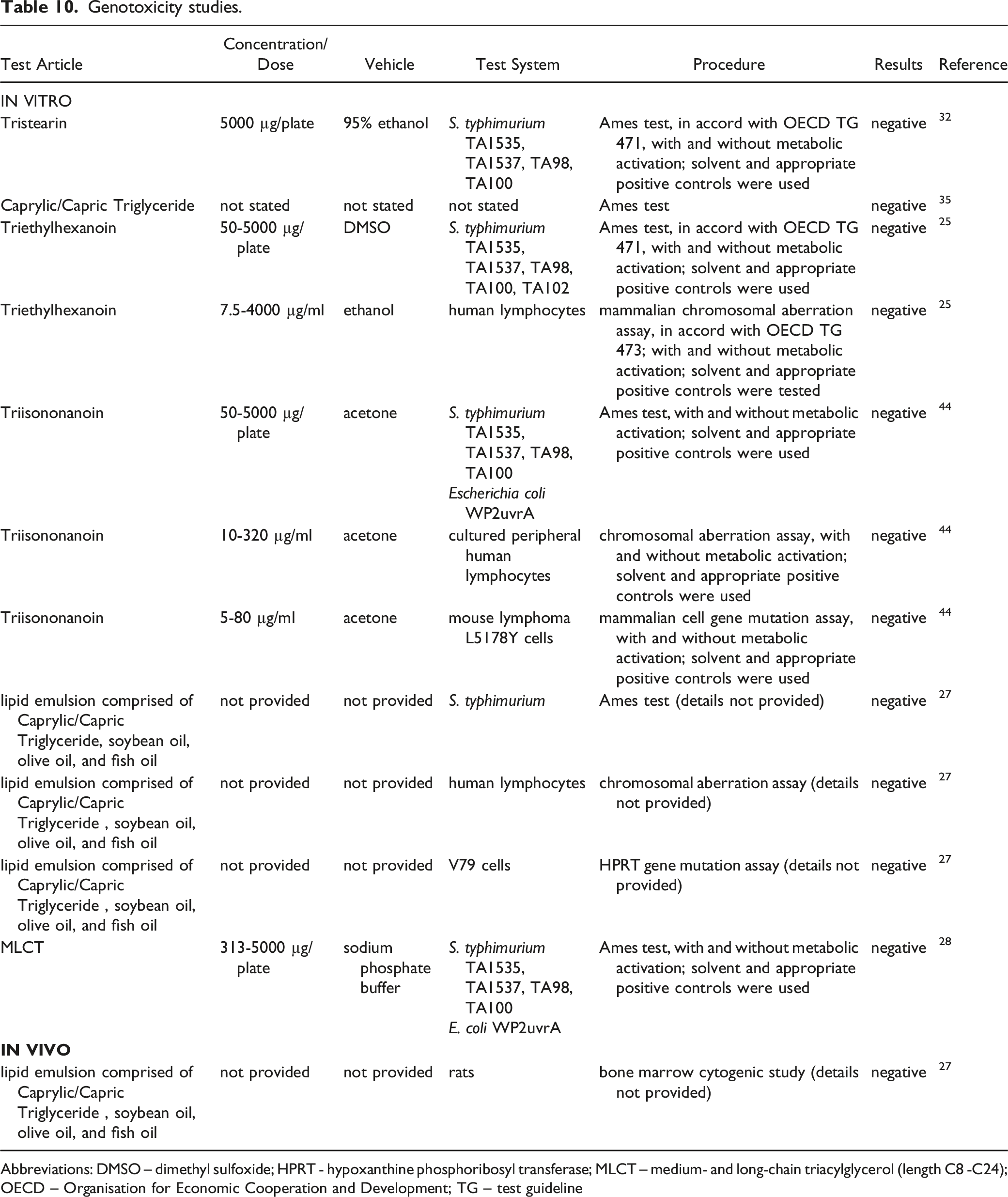
Abbreviations: DMSO – dimethyl sulfoxide; HPRT - hypoxanthine phosphoribosyl transferase; MLCT – medium- and long-chain triacylglycerol (length C8 -C24); OECD – Organisation for Economic Cooperation and Development; TG – test guideline
Tristearin (5000 µg/plate) 32 and Tricaprylin (concentration not stated) 35 were not mutagenic in the Ames test, Triethylhexanoin was not genotoxic in an Ames test (50 - 5000 µg/plate) or a mammalian chromosomal aberration assay (7.5 - 4000 µg/ml), 25 and Triisononanoin was not genotoxic in an Ames test (50 - 5000 µg/plate), chromosomal aberration assay (10 - 320 µg/ml), or a mammalian cell gene mutation assay (5 - 80 µg/ml). 44
A lipid emulsion that comprises a mixture of soybean oil, Caprylic/Capric Triglyceride, olive oil, and fish oil (test concentrations not provided) was not genotoxic in an Ames test, a chromosomal aberration assay, or a hypoxanthine phosphoribosyl transferase (HPRT) gene mutation assay. 27 In vivo, the emulsion was not genotoxic in a bone marrow cytogenic study in rats.
CARCINOGENICITY STUDIES
Following intraperitoneal injection of 0.25 ml Tricaprylin into 30 A/J mice in a tumorigenicity study, lung tumors were observed in 37% of the animals. 4 In the untreated-control group of 30 mice, the lung tumor incidence was 23%. The results of an oral carcinogenicity study by the National Toxicology Program (NTP) indicated that Tricaprylin caused a statistically significant dose-related increase in the incidence of pancreatic acinar cell hyperplasia and adenoma in rats. Tricaprylin did not induce acinar cell carcinomas. Additionally, the incidence of squamous cell papilloma in the squamous portion of the stomach of rats in the highest dose group (10 ml/kg Tricaprylin) was significantly greater when compared to controls.
ANTI-CARCINOGENICITY STUDIES
Trilaurin (dose not specified) completely inhibited the formation of neoplasms initiated by 7,12-dimethylbenz[a]anthracene (DMBA) and promoted by croton oil. 4 Additionally, extensive damage to tumor cells (lymphoma implants in the liver) was noted in rats after oral dosing with Tricaprylin.
DERMAL IRRITATION AND SENSITIZATION STUDIES
Trihydroxystearin was not irritating to the skin of albino rabbits in 24-h occlusive patch tests. 1 In 48-h occlusive patch tests, Trihydroxystearin did not induce skin irritation in any of the 103 subjects tested.
Undiluted Triisostearin and a 20% solution of Tribehenin (0.5 ml) in liquid paraffin were, at most, mildly irritating when applied to the skin of rabbits. 4 Undiluted Triethylhexanoin and an eyeliner containing 36.3% Trilaurin did not induce cutaneous irritation in rabbits. Neither Tribehenin (test concentration not provided) nor Triethylhexanoin; 1% intradermal induction, 100% occlusive topical induction; 25% occlusive challenge] induced sensitization in the Magnusson-Kligman guinea pig maximization test. Triisostearin (0.02 ml/cm2) did not induce significant cutaneous reactions in a study evaluating the phototoxicity and photoallergenicity potential of this ingredient in guinea pigs.
An eyeliner containing 36.3% Trilaurin did not induce skin irritation reactions in 91 test subjects. 4 Triethylhexanoin; details not provided] did not induce skin irritation in 25 subjects. A lip enhancer cream containing 0.38% Tribehenin was not comedogenic and did not induce clinically significant skin irritation in any of the 18 subjects evaluated in a 28-d test. Repeated insult patch test (RIPT) results (occlusive patches) for the following products were negative: eye enhancer cream containing 0.32% Tribehenin (198 subjects), hand cream containing 0.38% Tribehenin (at least 200 subjects), lip cream containing 0.38% Tribehenin (at least 200 subjects), and an eye defining pencil containing 1.68% Tristearin. None of these products induced clinically significant irritant or allergic contact dermatitis. In a skin sensitization test involving 91 subjects, there was no evidence of delayed contact hypersensitivity after repeated applications (occlusive patches) of an eyebrow pencil containing 40% Trilaurin. Also, Triethylhexanoin; details not provided] did not induce sensitization in a contact allergy test.
Application of a perfumed skin softener formulation containing 4% Caprylic/Capric Triglyceride to the shaved skin of female rats at a dose of 2 ml/kg 5 d/wk for 13 wk did not result in any localized skin effects. Caprylic/Capric Triglyceride was not a sensitizer in guinea pigs. Undiluted Caprylic/Capric Triglyceride was not irritating when tested using groups of 12 (21-d patch test), or 40 (test methods not described), and it was not an irritant or sensitizer in 128, subjects (Draize repeated insult patch test).
Dermal irritation and sensitization studies.
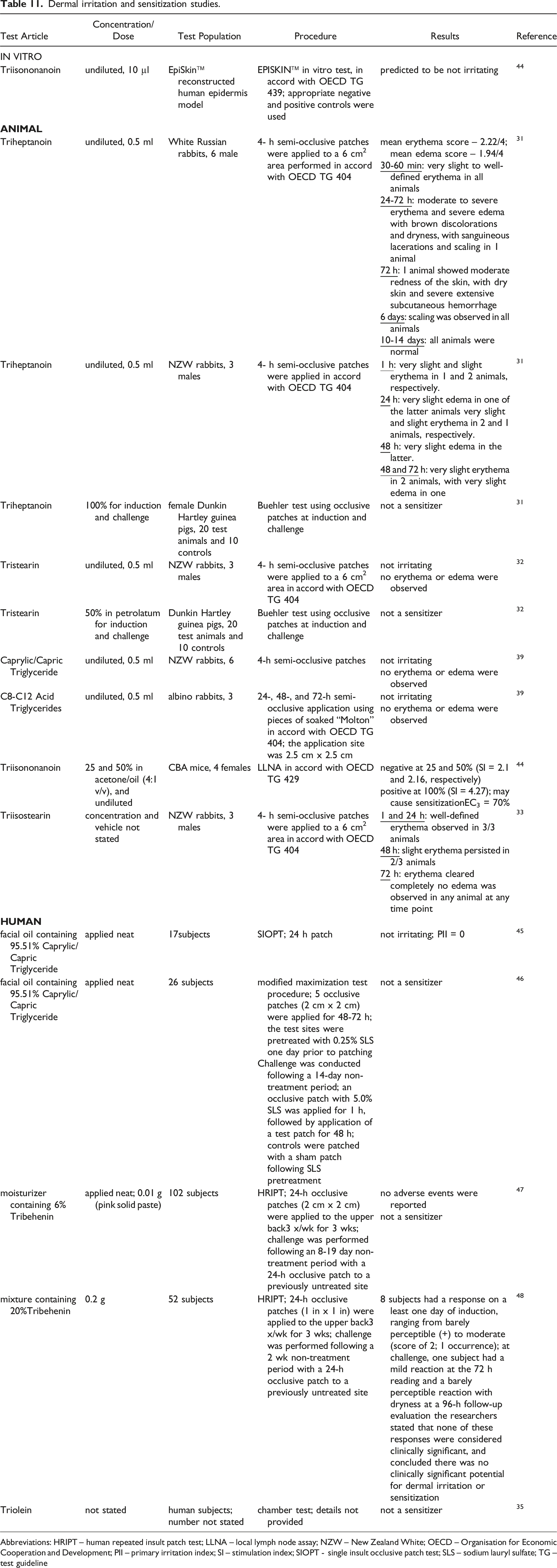
Abbreviations: HRIPT – human repeated insult patch test; LLNA – local lymph node assay; NZW – New Zealand White; OECD – Organisation for Economic Cooperation and Development; PII – primary irritation index; SI – stimulation index; SIOPT - single insult occlusive patch test; SLS – sodium lauryl sulfate; TG – test guideline
Dermal effects were observed in 4-h semi-occlusive patch tests in rabbits with undiluted Triheptanoin; very slight to slight erythema was reported in 1 - 2 of 3 animals in one study, but in the other study, very slight to well-defined erythema was observed in all 6 animals 30 - 60 min after patch removal, moderate to severe erythema and severe edema, discoloration, and dryness with sanguineous lacerations and scaling was observed in 1 animal 24 -7 2 h after dosing, and scaling was observed in all animals at day 6. 31 Triisostearin (test concentration not provided) produced well-defined erythema in all 3 rabbits at 1 and 24 h; all erythema was resolved by 72 h. 33 No irritation was observed in 4-h patch tests with undiluted Tristearin, 32 Caprylic/ Capric Triglyceride, 39 or C8-C12 Acid Triglycerides. 39 Triheptanoin (100%) 31 and Tristearin (50%) 32 were not sensitizers in guinea pigs. Triisononanoin was predicted to be non-irritating in an EpiSkin™ in vitro test. 44 However, in a mouse local lymph node assay (LLNA), it was predicted that Triisononanoin may cause sensitization; results were negative with 25% and 50% Triisononanoin, but positive when tested at 100%. 44
A facial oil containing 95.51% Caprylic/Capric Triglyceride was not an irritant is a 24-h single insult occlusive patch test in 17 human subjects, 45 and it was not a sensitizer in a human modified maximization patch test with 26 subjects. 46 In human repeated insult patch tests, a moisturizer containing 6% Tribehenin was not a sensitizer (102 subjects), 47 and a mixture containing 20% Tribehenin had no clinically significant potential for dermal irritation or sensitization (52 subjects). 48 Triolein was not a sensitizer in a chamber test; details were not provided. 35
PHOTOSENSITIZATION
Caprylic/Capric Triglyceride
The photosensitization potential of a facial cream oil containing 95.51% Caprylic/Capric Triglyceride was evaluated in a RIPT photocontact allergenicity assay completed in 27 subjects. 49 For induction, an occlusive patch consisting of 40 mg of the test material spread uniformly onto a 2 cm x 2 cm (10 mg/cm2) cotton cloth was applied to the lower back of each subject for 24 h; immediately following patch removal, the test site was exposed to two minimal erythema doses (MEDs) from a xenon arc solar simulator. This procedure was repeated 2x/wk for 3 wk, for a total of 6 induction applications. The light source was a 150 W compact xenon arc solar simulator (Solar Light Company) equipped with a UV-reflecting dichroic mirror and a 1 mm thick Schott WG320 filter; a 1 mm thick UG11 filter was also used. The solar spectrum (SSR waveband) was used to determine the individual MED. The size of the irradiated field at skin level was approximately a 1cm diameter circle. Total irradiance at skin level was 90.0 mW/cm2. The UVA intensity was 52.5 mW/cm2.
Following a 10-d non-treatment period, a challenge patch was applied for 24 h to a previously untreated site on the opposite side of the back, followed by exposure to ½ MED of solar simulated radiation plus 4 J/cm2 of UVA. (For the challenge, a 1 mm thick Schott WG-345 filter was added to eliminate the UVB component (290 - 320 nm) and to produce a continuous broadband UVA extending from 320 to 410nm.) An unirradiated site treated with the test product served as a "dark" control. The sites were examined at 48 and 72 h after irradiation for evidence of photocontact sensitization. The facial oil containing 95.51% Caprylic/Capric Triglyceride did not possess a detectable photocontact-sensitizing potential in human skin.
OCULAR IRRITATION STUDIES
Trihydroxystearin was classified as a mild, transient ocular irritant in albino rabbits. 1
An eye enhancer cream containing 0.32% Tribehenin and a hand cream containing 0.38% Tribehenin were classified as non-irritants in an in vitro chorioallantoic membrane vascular assay for determining the ocular irritation potential of chemicals. 4 An eyeliner containing 36.3% Trilaurin and a 20% solution of Tribehenin in liquid paraffin were, at most, mildly irritating to the eyes of rabbits. Triethylhexanoin [Trioctanoin] and Triisostearin did not induce ocular irritation in rabbits.
An eye enhancer cream containing 0.32% Tribehenin induced reactions ranging from mild to moderate ocular irritation in a group of 20 subjects, which resolved to either mild irritation or no irritation reactions at 2 hours post exposure. 4 In a clinical in-use safety test of two eye enhancer creams containing 0.32% Tribehenin, neither ocular irritation nor clinically relevant alterations in visual acuity were observed after 4 consecutive weeks of daily product use. Similar results were reported after testing of another eye enhancer cream containing 0.32% Tribehenin and an eye defining pencil containing 1.68% Tristearin in separate studies according to the same procedure. All of the subjects tested in these studies were contact lens wearers.
Caprylic/Capric Triglyceride was non-irritating, to at most very mildly irritating, to rabbit eyes. 5
Ocular irritation studies.

Abbreviations: HCE - Human Corneal Epithelium; NZW - New Zealand White; OECD – Organisation for Economic Cooperation and Development; TG – test guideline
Undiluted Triheptanoin, 31 Tristearin, 32 Caprylic/Capric Triglyceride, 39 and C8-12 Acid Triglyceride, 39 as well as Triisostearin at an unspecified concentration, 33 were not irritating in rabbit eyes. Triisononanoin was predicted to be non-irritating in an in vitro eye irritation test using the SkinEthic™ reconstructed model. 44
SUMMARY
In 2000, the Panel assessed the safety of Trihydroxystearin and concluded that, based on the available animal and clinical data, which included summary data from the safety assessments of Hydroxystearic Acid and Glyceryl Stearate and Glyceryl Stearate SE, Trihydroxystearin is safe as used in cosmetics. The Panel published two additional reports on related ingredients; the Panel concluded that Caprylic/Capric Triglyceride (1980) and Trilaurin and 22 additional glyceryl triesters (2001) are safe as used in cosmetics. An additional 29 triglycerides that are cosmetic ingredients and have not been reviewed by the Panel have also been identified. This safety assessment is a compilation of these 51 triglycerides, most of which (but not all) function as skin conditioning agents and/or viscosity increasing agents in cosmetics.
Some of these triglycerides are produced synthetically via classical Fischer type esterification methods, although the reaction may be promoted by acid or base catalysis, or by the use of an acid chloride. Additionally, some of these ingredients may be natural sourced and produced by transesterification.
Thirty-one of the 51 ingredients included in this safety assessment are in use, and Caprylic/Capric Triglyceride has the highest frequency of use (6000 formulations). According to the results of a concentration of use survey conducted by the Council, Triethylhexanoin has the highest maximum use concentration, with concentrations of 100% reported for face and neck formulations and 63% in lipstick formulations.
Approximately half of the ingredients included in this safety assessment have been reviewed previously by the Panel. The frequency and maximum concentrations of use for the majority of these ingredients have generally increased since these ingredients were originally reviewed.
Many of the triglycerides are approved by the FDA for use as direct or indirect food additives.
Oral absorption and metabolism of foods containing LCT mixtures differ from those containing Caprylic/Capric Triglyceride. C>12 are degraded by salivary, intestinal and pancreatic lipases into two fatty acids and a monoacyl glycerol, whereas, Caprylic/Capric Triglyceride is degraded by the same enzymes into three fatty acids and the simple glycerol backbone.
In newborn Rhesus monkeys administered a single dose of Tripelargonin or Triethylhexanoin via NG tube, free fatty acid concentrations and ketone 3HB increased with time. In New Zealand male rabbits given a Tripelargonin/LCT emulsion via a TPN infusion regimen 7 h/d for 11 d, 3HB concentrations were significantly decreased and plasma concentrations of short-chain fatty acids were significantly increased.
In acute toxicity testing, the dermal LD50 in rats was > 2 g/kg (the highest dose tested) for both Triheptanoin and Tristearin. The oral LD50 was > 2 g/kg for Triisostearin in mice and rats, > 2 g/kg Triolein in mice, > 5 g/kg Triheptanoin in mice, and > 48 g/kg Triethylhexanoin in rats. The oral LD50 of a MLCT oil was > 5 g/kg in rats. A single dose of 8.4 ml/kg bw Tripelargonin and Triethylhexanoin, administered via NG tube, did not affect activity level or induce narcolepsy in newborn Rhesus monkeys.
In 28-d gavage studies in Han-Wistar rats, dosing with 33% Caprylic/Capric Triglyceride did not produce any signs of toxicity, but undiluted test material produced some gastrointestinal effects, decreased thymic weight, caused inflammation in the lungs, and resulted in changes in some clinical pathology parameters. These changes were reversible. In Göttingen minipigs, clinical signs of toxicity were observed with 0.5 and 2 ml/kg/d Caprylic/Capric Triglyceride administered by gavage; no changes in organ weights or gross or microscopic lesions were observed. No adverse effects were observed in a study in which rabbits were given a Tripelargonin/LCT emulsion via a TPN infusion regimen for 7 h/d for 11 d.
Short-term and subchronic feeding studies were conducted with Caprylic/Capric Triglyceride. In rats, a NOAEL of 10 mg/kg bw/d was reported in a 30 d study with Caprylic/Capric Triglyceride, and a NOAEL of 3500 mg/kg/d was reported with mixture of triglycerides with alkyl chain lengths C8-C24. In a human study, no adverse effects were observed in a placebo-controlled double-blind study in which healthy subjects ingested 42 g/d of the C8-C24 mixture. Three-month feeding studies were performed with Caprylic/Capric Triglyceride in rats and dogs, and the NOAELs were 5% and 15%, respectively; no toxicologically-relevant signs of toxicity were observed at the highest doses.
In a chronic (9-mo) feeding study, an oil containing 64% Triheptanoin was not toxic in rats.
Tricaprylin was used as a vehicle in an oral (gavage) DART study of trichloroacetonitrile, and its effect on the test results was compared to other vehicles. Additionally, the potential developmental toxicity of Tricaprylin was evaluated in comparison to the two other vehicles (water and corn oil). There was a statistically significant increase in the percent implantation loss in the Tricaprylin group as compared to both the water and corn oil controls, and the total implants/litter was statistically significantly less when compared to the corn oil, but not the water, control group. Also, there were statistically significant decreases in fetal body weights and crown-rump length in the Tricaprylin control group as compared to the water and corn oil control groups. The study authors postulated that the differences observed between the Tricaprylin group and the other two control groups may be attributable to potential changes in nutritional status.
Additionally, the developmental toxicity of trichloroacetonitrile appeared to be vehicle-dependent; developmental effects caused by trichloroacetonitrile were seen at higher doses when administered in corn oil compared to those seen when Tricaprylin was used as the vehicle. The study authors suggested that trichloroacetonitrile and Tricaprylin “appear to interact in some way to potentiate effects of the cardiovascular system.”
The genotoxicity of several triglycerides was evaluated, and all the results were negative. Tristearin (5000 µg/plate) and Tricaprylin (concentration not stated) were not mutagenic in the Ames test, Triethylhexanoin was not genotoxic in an Ames test (50 - 5000 µg/plate) or a mammalian chromosomal aberration assay (7.5 - 4000 µg/ml), and Triisononanoin was not genotoxic in an Ames test (50 - 5000 µg/plate), chromosomal aberration assay (10 - 320 µg/ml), or a mammalian cell gene mutation assay (5 - 80 µg/ml).
A lipid emulsion that comprises a mixture of soybean oil, Caprylic/Capric Triglyceride, olive oil, and fish oil (test concentrations not provided) was not genotoxic in an Ames test, a chromosomal aberration assay, or an HPRT gene mutation assay. In vivo, the emulsion was not genotoxic in a bone marrow cytogenic study in rats.
Mixed results were obtained in dermal irritation and sensitization studies. Dermal effects were observed in 4-h semi-occlusive patch tests in rabbits with undiluted Triheptanoin; very slight to slight erythema was reported in 1 - 2 of 3 animals in one study, but in the other, very slight to well-defined erythema was observed in all 6 animals 30 - 60 min after patch removal, moderate to severe erythema and severe edema, discoloration, and dryness with sanguineous lacerations and scaling was observed in 1 animal 24 - 72 h after dosing, and scaling was observed in all animals at day 6. Triisostearin (test concentration not provided) produced well-defined erythema in all 3 rabbits at 1 and 24 h; all erythema was resolved by 72 h. No irritation was observed in 4-h patch tests with undiluted Tristearin, Caprylic/Capric Triglyceride, or C8-C12 Acid Triglycerides. Triheptanoin (100%) and Tristearin (50%) were not sensitizers in guinea pigs. Triisononanoin was predicted to be non-irritating in an EpiSkin™ in vitro test. However, in a mouse LLNA, it was predicted that Triisononanoin may cause sensitization; results were negative with 25% and 50% Triisononanoin but positive when tested at 100%.
In human testing, a facial oil containing 95.51% Caprylic/Capric Triglyceride was not an irritant is a 24-h single insult occlusive patch test in 17 subjects, was not a sensitizer in a human modified maximization patch test with 26 subjects, and was not a photosensitizer. In HRIPTs, a moisturizer containing 6% Tribehenin was not a sensitizer (102 subjects), and a mixture containing 20% Tribehenin had no clinically significant potential for dermal irritation or sensitization (52 subjects). Triolein was not a sensitizer in a chamber test; details were not provided.
Several triglycerides were evaluated and found not to be ocular irritants. Undiluted Triheptanoin, Tristearin, Caprylic/Capric Triglyceride, and C8-12 Acid Triglyceride, as well as Triisostearin at an unspecified concentration, were not irritating in rabbit eyes. Triisononanoin was predicted to be non-irritating in an in vitro eye irritation test using the SkinEthic™ reconstructed model.
No new carcinogenicity data were discovered in an extensive search of the published literature.
DISCUSSION
In accordance with its procedures, the Panel evaluates the conclusions of previously-issued reports every 15 years. The Panel has issued three final reports on the safety of 25 triglycerides (i.e., fatty acid triesters of glycerin) in the past 15+ years. The Panel previously concluded that Trihydroxystearin (2000), Caprylic/Capric Triglyceride (1980; reaffirmed in 2003), and a family of ingredients that included Trilaurin and 22 additional glyceryl triesters (2001) are safe as used in cosmetics. Additionally, the Panel determined that it was appropriate to include 26 triglycerides that have not yet been reviewed. The collection of these ingredients in one report enables the assembly of reinforcing and complementary test data. Safety profiles of these ingredients are consistent with roles of most constituents as dietary components and safe conclusions in previous reports.
Approximately half of the ingredients in this safety assessment have been reviewed previously by the Panel. The frequency and maximum concentrations of use for the majority of these ingredients has increased when compared to the original review. The most remarkable increase is in the frequency of use of Caprylic/Capric Triglyceride; in 2003, this ingredient was reported to be used in 763 formulations and in 2017, it is reported to be used in 6000 formulations. Also, in 2003, the maximum leave-on concentration of use for Caprylic/Capric Triglyceride was 84%, it is now reported to 95.6% in face and neck products.
One reported possible function of Docosahexenoic/Docosapentenoic/Oleic/Palmitic Triglyceride is skin bleaching agent. In the United States, skin bleaching agent is not considered a cosmetic function, and therefore use in that manner is not being assessed in this report.
During its original review of Trilaurin and other glyceryl triesters, the Panel noted that, as part of an effort to evaluate vehicles used in carcinogenicity studies, the NTP conducted a 2-yr carcinogenicity study in rats given Tricaprylin by gavage. This treatment was associated with a statistically significant dose-related increase in pancreatic acinar cell hyperplasia and adenoma, but there were no acinar carcinomas, the incidence of mononuclear leukemia was less, and nephropathy findings were reduced, compared to corn oil controls. In a tumor inhibition study, Trilaurin was found to inhibit the formation of neoplasms initiated by DMBA and promoted by croton oil. However, the Panel stated that no restrictions were warranted for any of these ingredients.
High purity is needed for the triglycerides. In 2007, the Panel published a final report on a diglycerides, and concluded that the ingredients in the diglyceride family are safe in the present practices of use and concentration provided the content of 1,2-diesters is not high enough to induce epidermal hyperplasia. The Panel discussed that there was an increased level of concern because of data regarding the induction of protein kinase C (PKC) and the tumor promotion potential of 1,2-diacylglycerols. The Panel noted that, nominally, glyceryl-1,3-diesters contain 1,2-diesters, raising the concern that 1,2-diesters could potentially induce hyperplasia. The Panel did note that these compounds are more likely to cause these effects when the fatty acid chain length is ≤ 14 carbons, when one fatty acid is saturated and one is not, and when used at high doses, repeatedly.
Based on existing information from a previous safety assessment, minimal percutaneous absorption of Triolein has been demonstrated in vivo using guinea pigs (but not hairless mice), and in vitro using full-thickness skin from hairless mice. However, the Panel recognized that, reportedly, Triolein and Tricaprylin can enhance the skin penetration of other chemicals, and the Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data, or when dermal absorption was a concern.
The Panel acknowledged that some of the triglycerides may be formed from plant-derived or animal-derived constituents. The Panel thus expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to sufficiently limit amounts of such impurities in an ingredient before blending them into cosmetic formulations. Additionally, the Panel considered the risks inherent in using animal-derived ingredients, namely the transmission of infectious agents. Although tallow may be used in the manufacture of Cod Liver/Mink/Tallow Triglyceride and is clearly animal-derived, the Panel notes that tallow is highly processed, and tallow derivatives even more so. The Panel agrees with determinations by the FDA that tallow derivatives are not risk materials for transmission of infectious agents.
Finally, the Panel discussed the issue of incidental inhalation exposure, as some of the triglycerides are used in cosmetic sprays and could possibly be inhaled. For example, Triethylhexanoin and Triisostearin are reported to be used at maximum concentrations of 36% and 30%, respectively, in perfumes, and 14.7% and 10.4%, respectively, in face powders. The Panel noted that in aerosol products, most droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings
CONCLUSION

Footnotes
Author Contributions
Fiume, M. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. Heldreth, B. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.

