Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 19 skin and connective tissue-derived proteins and peptides, which are reported to function mainly as skin and/or hair conditioning agents in cosmetics. The Panel reviewed the relevant data provided and concluded that these ingredients are safe in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
The skin and connective tissue-derived proteins and peptides detailed in this report are described in the International Cosmetic Ingredient Dictionary and Handbook (Dictionary) to function mainly as skin and hair conditioning agents in cosmetics. 1 This report assesses the safety of the following 19 skin and connective tissue-derived ingredients:
Ammonium Hydrolyzed Collagen
Atelocollagen
Calcium Hydrolyzed Collagen
Collagen
Elastin
Fibronectin
Gelatin
Hydrolyzed Actin
Hydrolyzed Collagen
Hydrolyzed Collagen Extract
Hydrolyzed Elastin
Hydrolyzed Fibronectin
Hydrolyzed Gelatin
Hydrolyzed Reticulin
Hydrolyzed Spongin
MEA-Hydrolyzed Collagen
Soluble Collagen
Soluble Elastin
Zinc Hydrolyzed Collagen
The Panel previously reviewed the ingredient Hydrolyzed Collagen, and concluded that it is safe for use in cosmetics; the report was published in 1985 and the conclusion was reaffirmed in a re-review that was published in 2006.2,3 This ingredient was included in this safety assessment because of the relevance of the information in regards to reviewing the safety of the other ingredients in the report.
Additionally, the safety of several other hydrolyzed proteins as used in cosmetics has been reviewed by the Panel in several previous assessments. The Panel concluded that Hydrolyzed Keratin (finalized in 2016), Hydrolyzed Soy Protein (finalized in 2015), Hydrolyzed Silk (finalized in 2015), Hydrolyzed Rice Protein (published in 2006), and Hydrolyzed Corn Protein (published in 2011) are safe for use in cosmetics.4-8 The Panel concluded that Hydrolyzed Wheat Gluten and Hydrolyzed Wheat Protein are safe for use in cosmetics when formulated to restrict peptides to a weight-average MW of 3500 Da or less. 9 The Panel concurrently reviewed the safety of plant-derived and bovine milk-derived proteins, which had, at that time, tentative conclusions of safe as used, in separate reports. In addition to the review of these other protein-derived ingredients, the Panel has assessed the safety of Ethanolamine (also known as monoethanolamine or MEA) and Ethanolamine Salts and concluded these ingredients are safe when formulated to be nonirritating (rinse-off products only) but should not be used in cosmetic products in which N-nitroso compounds may be formed. 10
Actin, Collagen, Elastin, Fibronectin, Gelatin, and reticulin all are derived from essential components in animal tissues. Much of the available published literature evaluated the effects of pharmaceutical or other agents on these proteins in their naturally occurring tissues. These studies were not considered relevant for assessing the safety of the skin and connective tissue-derived ingredients as used in cosmetics and are not included in this assessment.
The sources for these cosmetic ingredients may be from many different land or marine animals. These differing sources could potentially produce or result in skin and connective tissue-derived proteins with unique properties, which may result in varying compositions and impurities within a single ingredient (eg, Hydrolyzed Collagen from animals such as cows may have some impurities that are different from Hydrolyzed Collagen obtained from fish).
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition
Definitions and Functions of the Ingredients in this Safety Assessment. 1
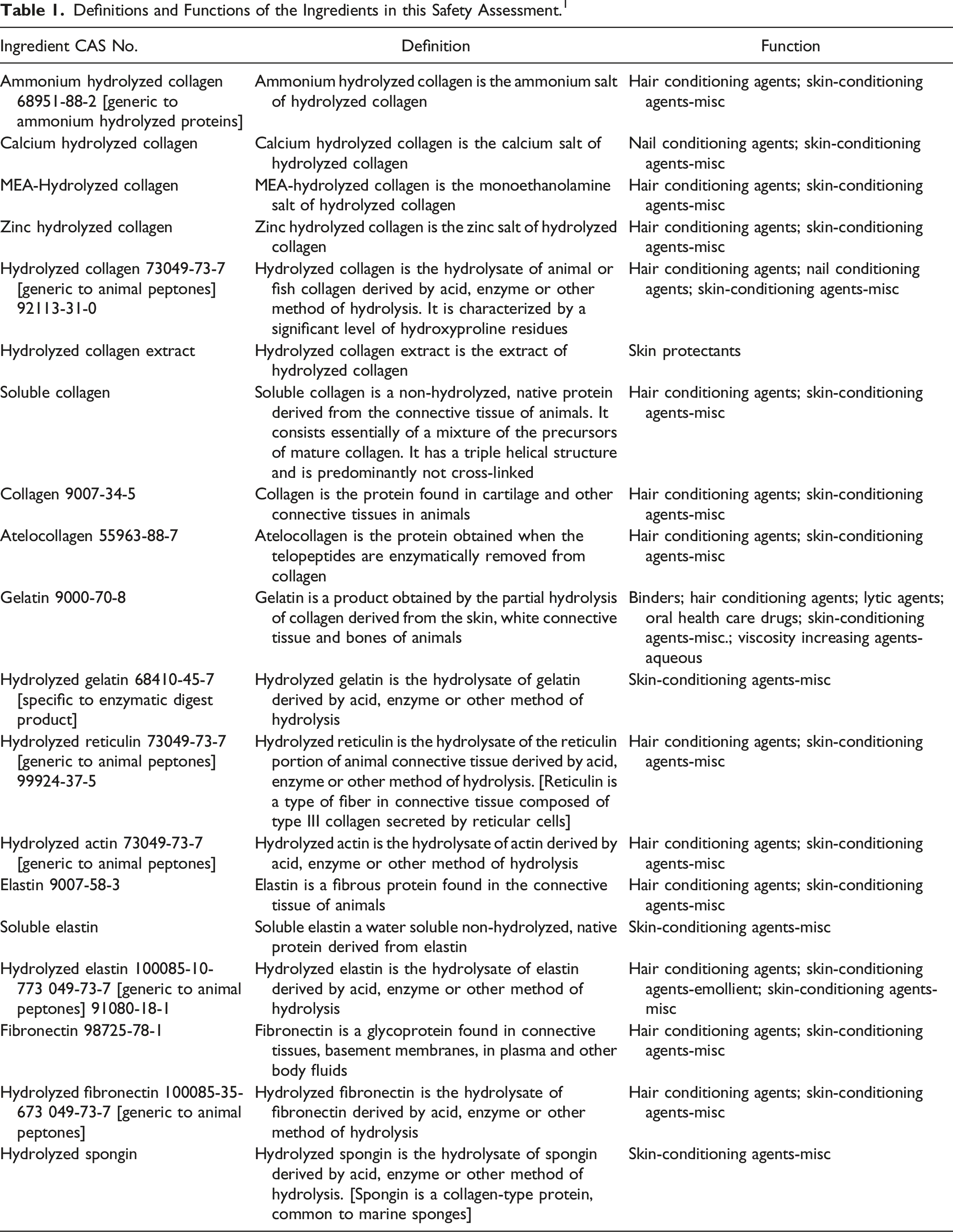
Skin and connective tissue protein derivatives form a broad category of materials that are prepared by extraction from animal tissue and partial hydrolysis to yield cosmetic ingredients. Proteins and protein hydrolysates, including those of animal tissue, are used as conditioning agents in hair and skin products. These proteins are present in many types of tissue, including skin.
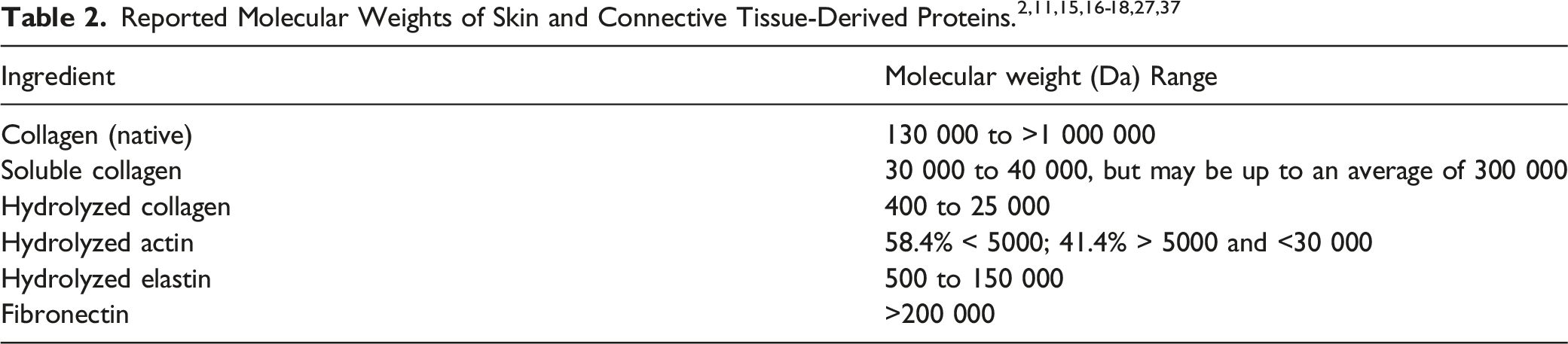
The most abundant protein in mammals is collagen, making up approximately 30% of all proteins by mass.11,12 The collagen family is comprised of 28 members (named collagen I to collagen XXVIII) that all have at least 1 triple helix in their structure at varying degrees (see further description below). 12 The most common are mainly the fibril-forming collagens (types I, II, III, and V) that are found in skin, cartilage, reticulate, and cell surfaces. Most of the other proteins addressed in this report are derivatives of collagen, are co-located with collagen in tissues, or are both. Gelatin, for example, is a product obtained by the partial hydrolysis of collagen derived from the skin, white connective tissue, and bones of animals. 11 Reticulin is a type of fiber in connective tissue composed of type III collagen secreted by reticular cells. Actin, elastin, and fibronectin are discrete in structure from collagens, but are commonly co-located with collagen in tissue (eg, fibronectin commonly provides rigidity on the edges of primarily collagen-based tissues). Spongin, however, is a collagen-like protein found only in marine sponges (constituting the small skeletal elements, or spicules, in the animal).
The preparation of protein hydrolysates can be accomplished via acid, enzyme, or other methodologies. These methodologies, and the degree to which they are utilized, may profoundly affect the size and biological activity of such hydrolysates. In most ingredients in this report, even in ingredients without “hydrolyzed” in the name, the proteins are at least hydrolyzed to some degree as a necessary part of extraction or solubilization. Further steps towards solubilization of these macromolecules are commonly achieved via reaction with an alkaline substance to generate a protein salt (eg, Calcium Hydrolyzed Collagen).
Actin
Actin is a major protein of muscle and an important component of all eukaryotic cells. 11 α-Actin is found in differentiated muscle cells, while β-actin and γ-actin are in all non-muscle cell types.
Collagen
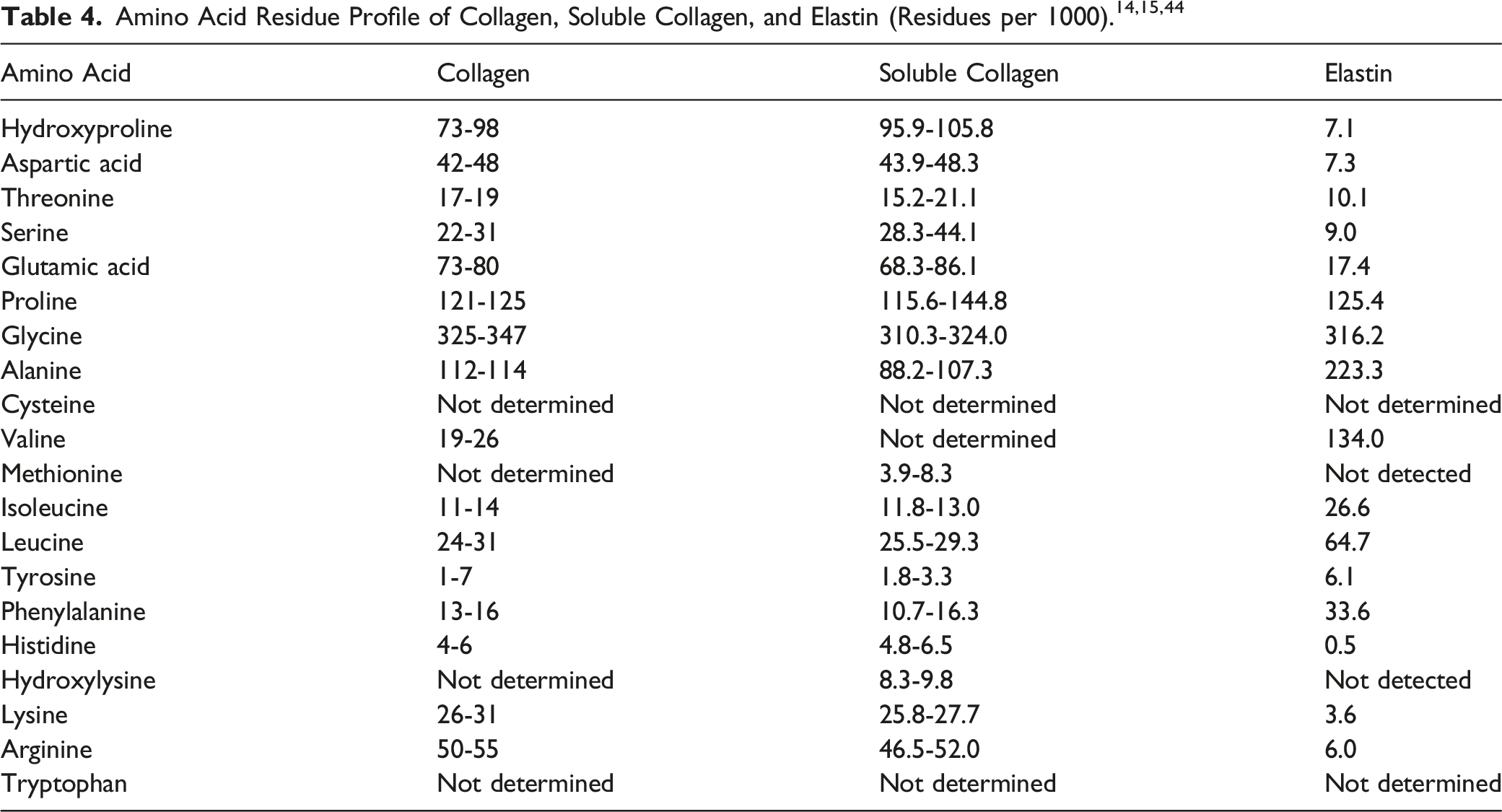
Collagen is the main constituent of skin (comprising 70% to 80% dry weight of the dermis) and connective tissue, and is the organic substance of bones and teeth.11,13 Collagen is primarily responsible for the skin’s tensile strength. One Collagen molecule consists of 3 polypeptide chains, each containing approximately 1000 amino acids in a primary sequence that is rich in proline, hydroxyproline, and hydroxylysine. Collagen is not just 1 discrete, ubiquitous protein sequence, but is a protein superfamily that is diversified across different tissue/function types and source species, including cattle, chicken, and fish.12,14 The common structural feature of collagen proteins is the presence of a triple helix. However, the percentage of each protein that this helix makes up can vary across different members of the collagen superfamily from as little as 10% to nearly 100%. The diversity of the Collagen superfamily is further increased by the presence or absence of several α-chains, the existence of several molecular isoforms and supramolecular structures of specific Collagen types, and the use of different methods of extraction/hydrolysis.
Elastin
Elastin is the primary component of the elastic, load-bearing fibers of animal connective tissue. 11 It is an insoluble, highly cross-linked hydrophobic protein that is rich in nonpolar amino acid residues, such as valine, leucine, isoleucine, and phenylalanine. There are 2 types of elastin: Type 1 is derived from bovine neck ligaments, aorta (as reported in 1987), skin, and related tissues; Type 2 is derived from cartilage and its derivatives. 15 In skin, Elastin is the intact elastic fiber network that comprises approximately 2% to 4% of the dermis by volume. 13
Fibronectin
Fibronectin is a multifunctional glycoprotein found on cell surfaces, in body fluids (especially plasma), in soft connective tissue matrices, and in most basement membranes. 11
Gelatin
Gelatin is a heterogeneous mixture of water-soluble proteins of high average molecular weight that are derived from the denaturation and hydrolysis of Collagen. 11 Glycine or alanine accounts for 1 third to 1 half of the amino acid residues, while another quarter is composed of proline or hydroxyproline.
Reticulin
Reticulin is a connective tissue protein that occurs wherever connective tissue forms a boundary 11
Physical and Chemical Properties
Collagen
Solutions of Collagen for cosmetic use have a pH range of 3.8 to 4.7 14
Hydrolyzed Collagen
Hydrolyzed Collagen may be a powder or solution. 2 A 10% aqueous solution has a pH of 4.0-6.5.
Elastin
Purified Elastin is a pale yellow color and exhibits a bluish fluorescence in UV light. 11 It resists acid and alkaline hydrolysis. It is practically insoluble even in hydrogen-bond-breaking solvents at temperatures up to 100°C, and is nearly impossible to bring into solution except by using reagents capable of hydrolyzing peptide bonds. Unprocessed or native elastin is reported to be too insoluble for use in cosmetic formulations. 15
Fibronectin
Fibronectin can be provided in a solution or as a lyophilized powder. 19
Gelatin
Gelatin is a vitreous, brittle solid that is colorless to faintly yellow.11,20 It is practically odorless and tasteless. When Gelatin granules are immersed in cold water, they hydrate into discrete, swollen particles. When warmed, Gelatin disperses into water. Warm-blooded animal sourced Gelatin has a gel point of 30 to 35°C, while cold-water ocean fish sourced Gelatin has a gel point between 5 and 10°C. Gelatin is soluble in aqueous solution of polyhydric alcohols like glycerin and acetic acid and is insoluble in alcohol, chloroform, ether, and most other organic solvents.
Soluble Elastin
Soluble Elastin is reported to be a cream-colored powder that is soluble in water and ethanol. 15
Method of Manufacturing
Method of Manufacturing.

Hydrolyzed Collagen
A representative manufacturing flow chart for Hydrolyzed Collagen is found in Scheme 1. This process may vary slightly between specific products with the elimination of the use of preservatives after hydrolysis and the addition of filtration and concentration of solution before the first quality control.
26
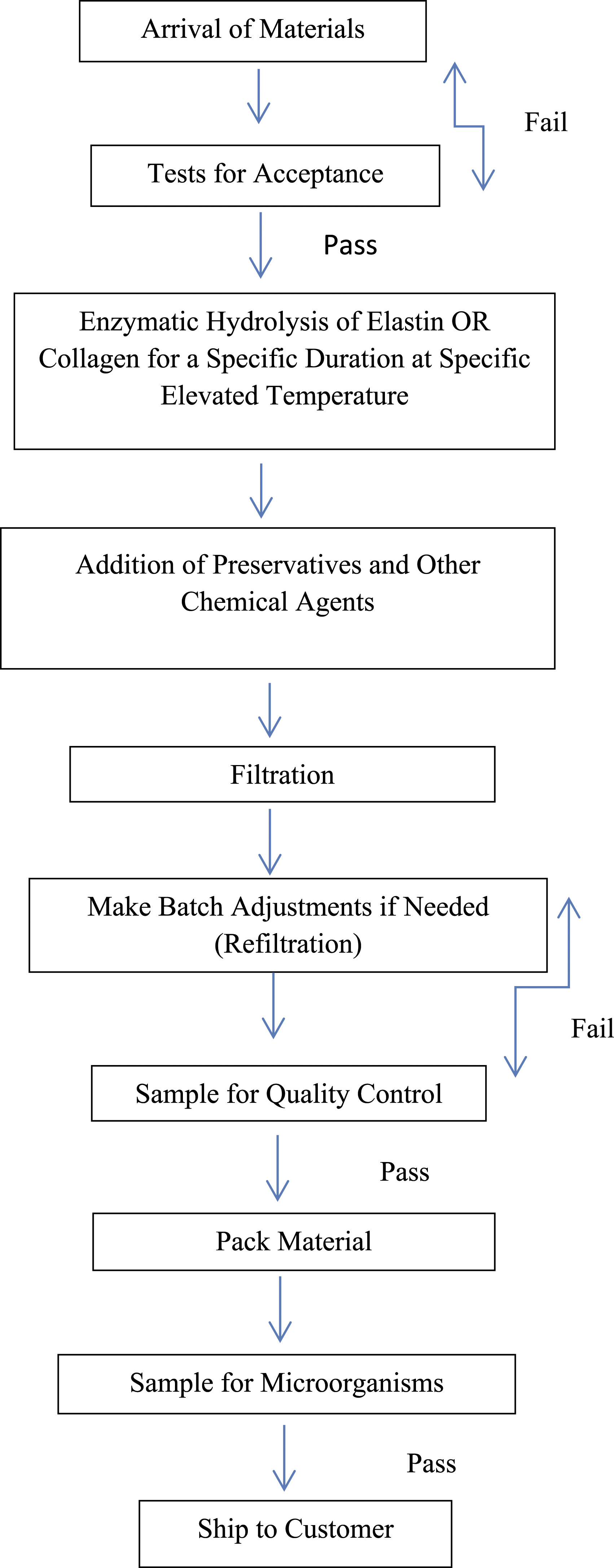
A supplier has reported that their Hydrolyzed Collagen products (6 products with MW ranges of 400 to 2000 Da, concentration up to 50% in water) are prepared by acidic, alkalic, and/or enzymatic hydrolysis of bovine gelatin, swine gelatin, or fish scale until the molecular weight reaches the target range. 27
Soluble collagen
A representative manufacturing flow chart for Soluble Collagen is found in Scheme 2. Manufacturing flow chart for Soluble Collagen (bovine sourced).
41
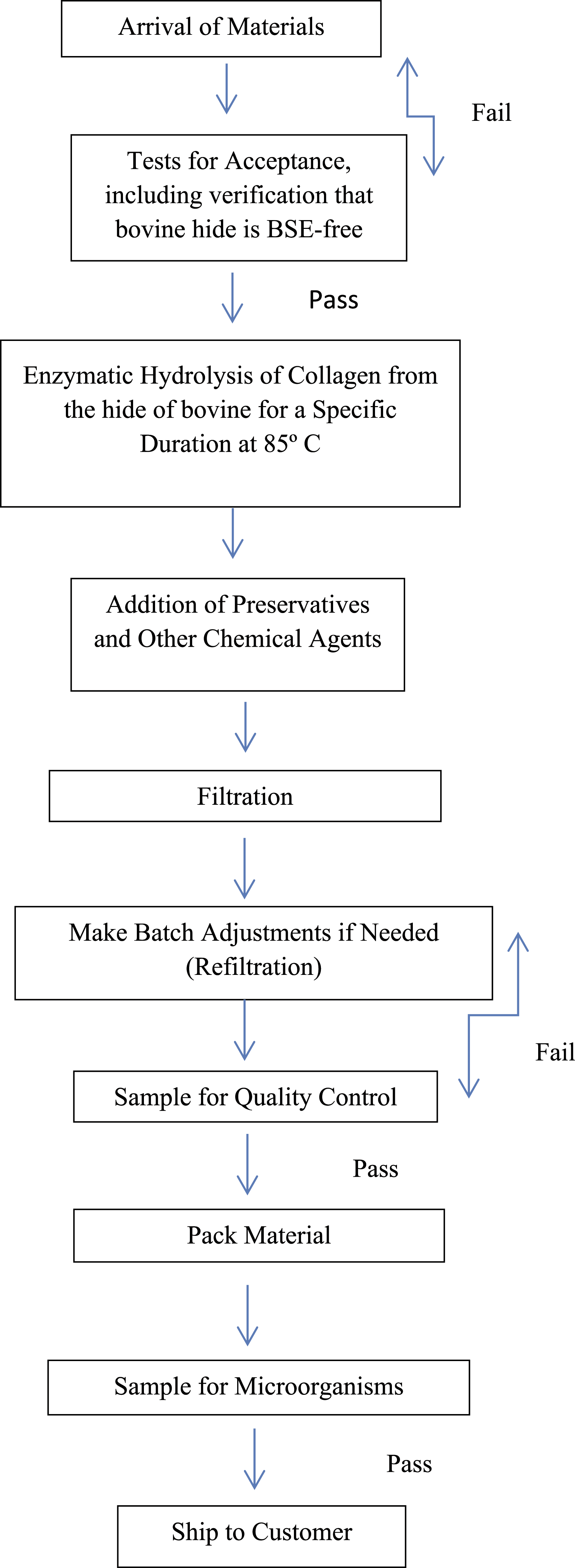
Hydrolyzed elastin
A representative manufacturing flow chart for Hydrolyzed Elastin is found in Scheme 1. This process may vary slightly between specific products with the use of pH adjustment during hydrolysis.
Gelatin
According to 21 CFR§700.27, Gelatin is “… a product that has been obtained by the partial hydrolysis of collagen derived from hides, connective tissue, and/or bones of cattle and swine. Gelatin may be either Type A (derived from an acid-treated precursor) or Type B (derived from an alkali-treated precursor) that has gone through processing steps that include filtration and sterilization or an equivalent process in terms of infectivity reduction.”
Composition
Impurities
Several of the ingredients in this safety assessment, including Hydrolyzed Collagen, Hydrolyzed Elastin, and Gelatin, may be bovine sourced. Some bovine materials may be considered risk materials for transmission of infectious agents (eg, bovine spongiform encephalopathy (BSE) prions). According to 21 CFR§700.27, “no cosmetic shall be manufactured from, processed with, or otherwise contain, prohibited cattle materials.” Prohibited cattle materials “mean specified risk materials, small intestine of all cattle …, material from non-ambulatory disabled cattle, material from cattle not inspected and passed, or mechanically separated.” Gelatin or hides and hide-derived products are not prohibited cattle materials. Cosmetic manufacturers must follow record keeping requirements that “demonstrate that the cosmetic is not manufactured from, processed with, or does not otherwise contain prohibited cattle materials.”
The World Organization for Animal Health (OIE) recommends that “when authorizing import or transit of … gelatin and collagen prepared exclusively from hides and skins … and any products made from these commodities and containing no other tissues from cattle, veterinary authorities should not require any BSE related conditions [ie restrictions], regardless of the BSE risk status of the cattle population of the exporting country, zone, or compartment.” 33
Collagen
An analysis for 3 different Collagen products found the level of arsenic to be less than 1 ppm. 14
Hydrolyzed collagen
The maximum concentrations of iron and heavy metals reported in Hydrolyzed Collagen were 3 ppm and 25 ppm, respectively. 2
A supplier reported that their Hydrolyzed Collagen products (6 products with MW ranges of 400 to 2000 Da, concentration up to 50% in water) sourced from bovine gelatin, swine gelatin, and fish scales contain not more than 10 ppm heavy metals and not more than 1 ppm arsenic. 27
A supplier reported that their Hydrolyzed Collagen products are BSE-free.34,35
Soluble collagen
A supplier reported that their Soluble Collagen product is BSE-free. 36
Elastin and hydrolyzed elastin
Impurities in commercial Elastin-based preparations include contamination by lipoid substances from the raw materials and products of Collagen degradations. 37
A supplier certified that their Hydrolyzed Elastin products are BSE-free.38-40
Gelatin
According to the Food Chemicals Codex, Gelatin must contain no more than 0.0005% sulfur dioxide, 10 mg/kg chromium, 1.5 mg/kg lead, and 0.3 mg/kg pentachlorophenol. 20
A supplier certified that their Gelatin product is BSE-free.
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
NR = not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b It is possible these products may be powders, but it is not specified whether the reported uses are powders.
cIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
dNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
eIncludes 25 uses listed in the VCRP as “soluble animal collagen”.
fNot listed in the Dictionary, possibly the same as Collagen Extract.
NR = not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIt is possible theseproducts may be sprays, but it is not specified whether the reported uses are sprays.
cNot specified whethe a powder or a spray, so this information is captured for both categories of incidental inhalation.
dIt is possible theseproducts may be powders, but it is not specified whether the reported uses are powders
eMajority of the useswere categorized as “Hydrolyzed Animal Protein” in the VCRP database.
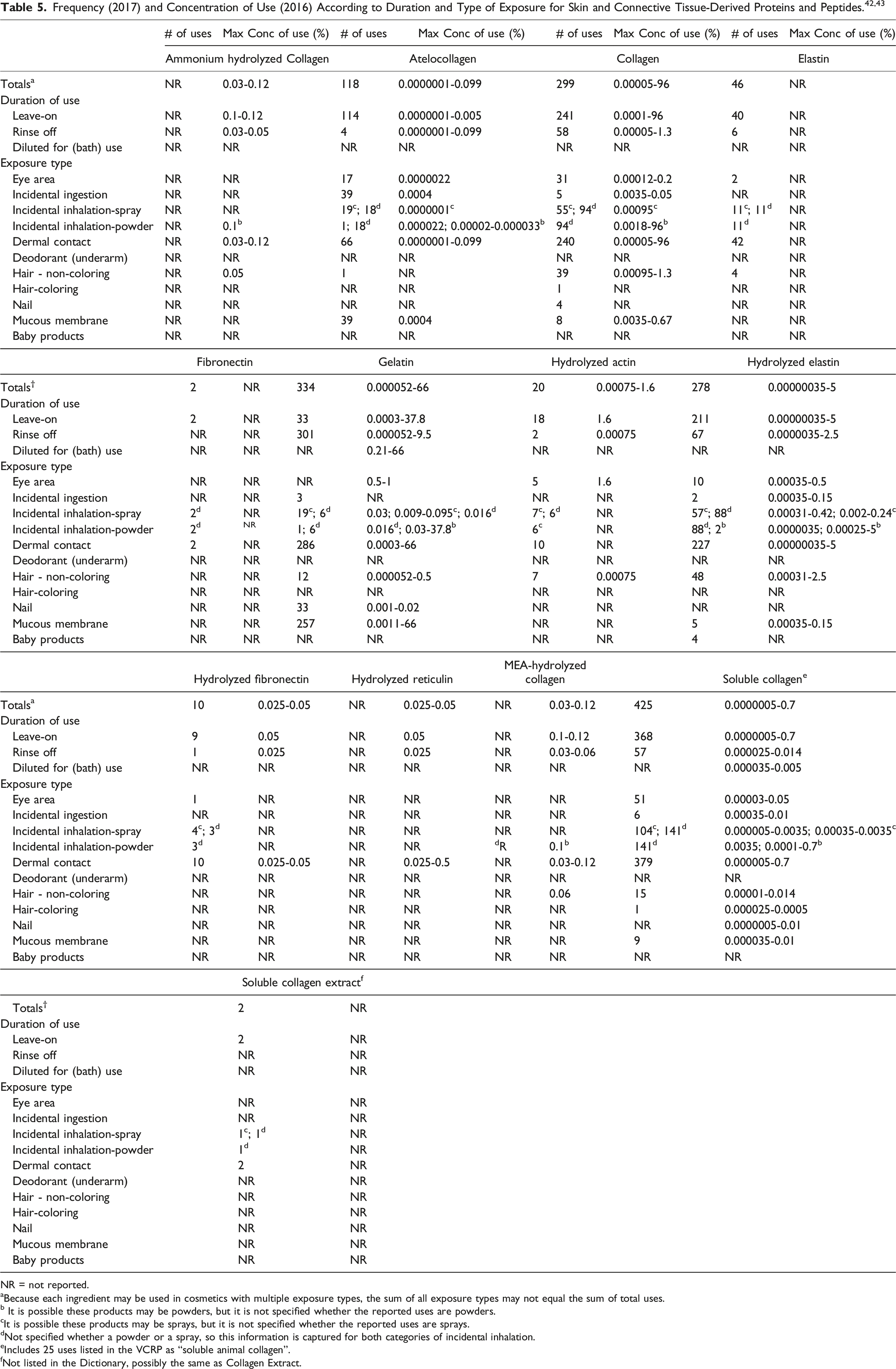
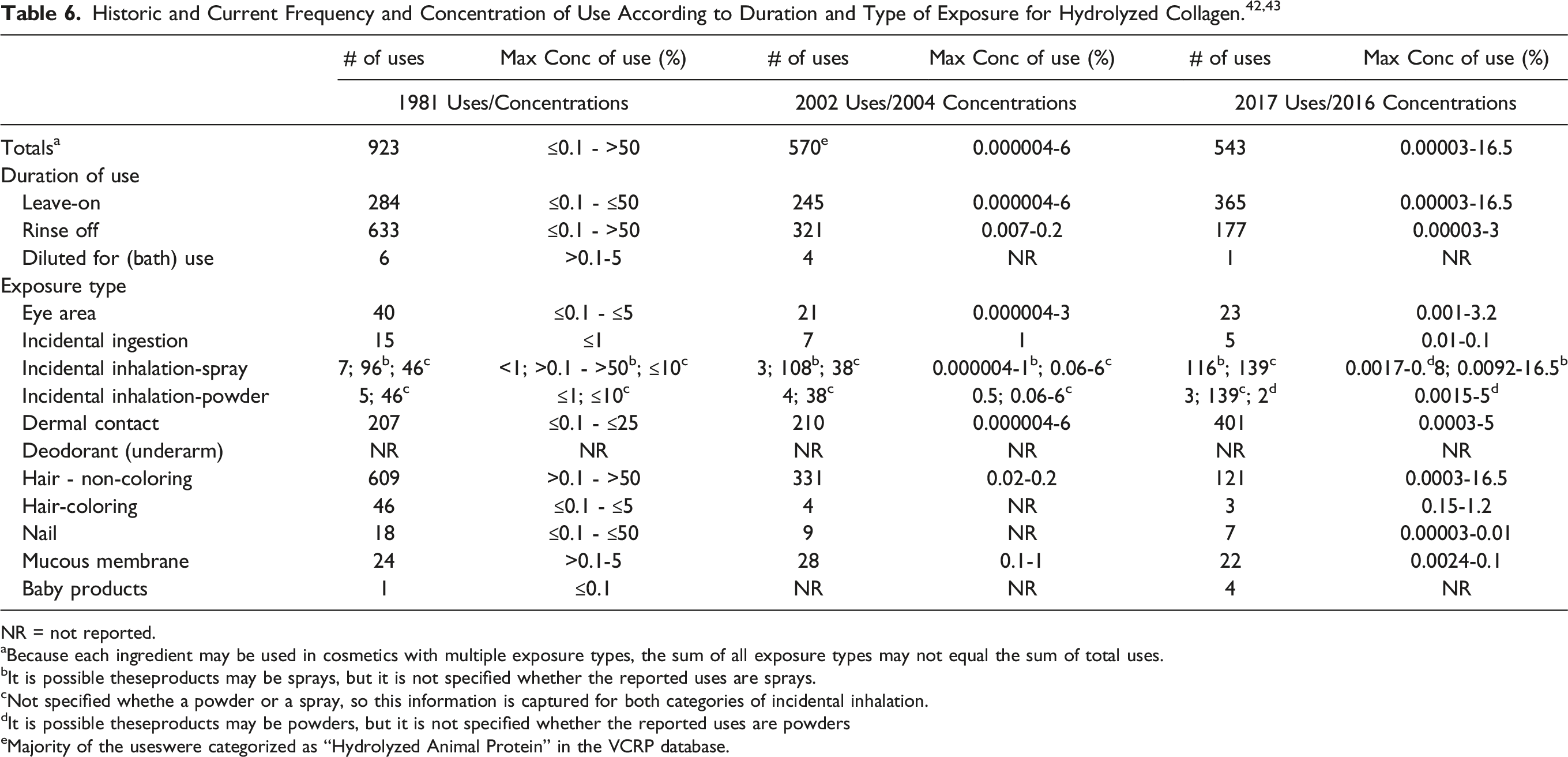
Historic and current use data for Hydrolyzed Collagen is reported in Table 6. The number of uses of Hydrolyzed Collagen has declined since the initial safety assessment in 1981 and the re-review in 2002 (923 and 570 uses, respectively2,3). The maximum use concentration of Hydrolyzed Collagen was reported to be 16.5% in hair tonics and dressings in 2016; it was previously reported to be used at concentrations greater than 50% (in rinse-off formulations).2,43
Ingredients Not Reported in Use.

In some cases, reports of uses were received from the VCRP, but no concentration of use data were provided. For example, Elastin is reported to be used in 46 formulations, but no use concentration data were provided. In other cases, no uses were reported to the VCRP, but a maximum use concentration was provided in the industry survey. For example, Ammonium Hydrolyzed Collagen was not reported in the VCRP database to be in use, but the industry survey indicated that it is used in several formulations at concentrations up to 0.12%.
Some of these ingredients may be used in products that can come into contact with mucous membranes and the eyes. For example, Gelatin is used in bath oils, tablets and salts at up to 66% and Hydrolyzed Collagen is used in an eyeliner at up to 3.2%. 43 Additionally, some of these ingredients were reported to be used in hair care products, skin care preparations, face powders, and fragrances and could possibly be inhaled. For example, Hydrolyzed Collagen was reported to be used in hair spray at a maximum concentration of 0.28% and Soluble Collagen was reported to be used in face powders at a maximum concentration of 0.0035%. In practice, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.45,46 Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.47-49 More information may be found in the Panel’s Respiratory Exposure Resource Document here https://www.cir-safety.org/sites/default/files/report_InhalationDocument_122021.pdf.
The skin and connective tissue-derived protein and peptide ingredients described in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union; however, monoalkanol-amine ingredients must not have a secondary amine content that exceeds 0.5%, and water-soluble zinc salt ingredients must not have more than 1% zinc in ready for use preparations. 50
Non-Cosmetic
The FDA determined that the use of peptones as direct food substances is generally recognized as safe (GRAS). These GRAS peptones are defined as “the variable mixture of polypeptides, oligopeptides, and amino acids that are produced by partial hydrolysis of … animal tissue or gelatin …” (21 CFR §184.1553). The FDA requires allergen labeling when 1 or more of the 8 major food allergens, which includes fish, are included in food. 51
Collagen
Non-cosmetic uses of Collagen include fibers in sutures, leather substitutes, coatings, as a gel in photographic emulsions, and food casings. 11
Gelatin
Non-cosmetic uses of Gelatin include uses in food as a stabilizer, thickener, texturizer, firming agent, surface-active agent, or surface-finishing agent.11,20 Gelatin is also used in the manufacturing of rubber substitute, adhesives, cements, lithographic and printing inks, plastic compounds, artificial silk, photographic plates and films, matches, and light filters for mercury lamps. 11 It is also used as a clarifying agent, in hectographic masters, sizing paper and textiles, and for inhibiting crystallization in culture preparations in bacteriology. In pharmaceuticals, Gelatin is a suspending agent, an encapsulating agent, a tablet binder, and a tablet and coating agent.
Gelatin is a category I active ingredient in ophthalmic demulcent over-the-counter (OTC) drug products at up to 0.01% (21CFR §349.12).
Toxicokinetics
Gelatin
The bioavailability of Gelatin derived from Nile tilapia scales was determined in an oral pharmacokinetic study in rats. 52 Five groups of 6 female Sprague-Dawley rats received 4000 mg/kg body weight Gelatin intragastrically (i.g.), 400 mg/kg hydroxyproline i.g., 400 mg/kg hydroxyproline intravenously (i.v.), normal saline i.g., or normal saline i.v. Blood plasma was then drawn from the rats at different times over 24 h to determine the hydroxyproline concentration. The bioavailability of the Gelatin was indirectly measured by the bioavailability of hydroxyproline in Gelatin. The relative and absolute bioavailability of Gelatin was 74.12% and 85.97%, respectively. The amino acid profile of plasma showed 41.91% of the digested Gelatin was absorbed from the intestine in di- and tri-peptide form. The authors of this study concluded that Gelatin had high oral bioavailability.
Toxicological Studies
Acute
Animal –Dermal
Hydrolyzed collagen
Hydrolyzed Collagen at up to 2% in formulation was practically nontoxic when administered dermally in acute toxicity studies in rabbits. 2
Animal - Oral
Collagen
The safety of a product containing approximately 60% Collagen (type II from chicken sternal cartilage), 20% chondroitin sulfate, and 10% hyaluronic acid was investigated in 5 male and 5 female Sprague-Dawley rats. 53 The rats received a single oral dose of 5000 mg/kg body weight and were observed for clinical signs of toxicity for 14 d. All rats survived the observation period and had normal body weight gains. On the 15th day of the study, the rats were killed and underwent macroscopic necropsy: no gross pathological lesions were observed in any of the animals.
Hydrolyzed collagen
Hydrolyzed Collagen was practically nontoxic when administered orally (up to 100%) in acute toxicity studies of mice and rats. 2
The oral LD50 of Hydrolyzed Collagen (30% solution in water; fish scale sourced; MW ∼ 400 Da) was estimated to be greater than 2500 mg/kg body weight in Sprague-Dawley CD rats. 27 This acute toxicity test was performed in accordance to Organization for Economic Co-operation and Development test guideline (OECD TG) 423. A group of 3 female rats were treated orally with the test material at a dose level of 2000 mg/kg body weight, with another group of 3 fasted female rats receiving also receiving the material at the same dose level. No deaths or signs of systemic toxicity were observed during the 14 d of monitoring post-dosing. All animals exhibited expected gains in body weight. No abnormalities were observed at necropsy.
Short-Term Toxicity Studies
Animal – Oral
Gelatin
In a rat study of the ability of shark skin Gelatin to increase bone mineral density, no adverse effects were reported. 54 The female Wistar rats (n = 40) were ovariectomized approximately a week after the start of receiving a low-protein diet and then received shark Gelatin as oral doses of 10, 20, or 40 mg/100 g body weight/d for 2 week. Control animals were given ovalbumin at 20 mg/100 g body weight/d. No significant differences between experimental groups and the controls were observed in final body weight, feed intake, femoral bone weight, or femoral bone length.
Subchronic Toxicity Studies
Animal – Dermal
Hydrolyzed collagen
Subchronic dermal studies in rabbits and pigs on 2 cosmetic formulations containing 2% Hydrolyzed Collagen were negative for systemic toxicity. 2
Animal - Oral
Collagen
The safety of a product containing approximately 60% Collagen (type II from chicken sternal cartilage), 20% chondroitin sulfate, and 10% hyaluronic acid was investigated in 40 male and 40 female Sprague-Dawley rats. 53 The rats were divided into groups of 10 animals/sex and received the test material in distilled water at 0, 30, 300, or 1000 mg/kg body weight once daily via gavage for 90 d. Animals were observed twice daily for mortality and detailed observations for clinical signs of toxicity were performed once weekly. Body weight and feed consumption were measured weekly. Hematology samples were collected a week before the end of dosing and the animals were killed at the end of the dosing period. A gross necropsy was performed on all animals and tissues were preserved for histopathological examination.
All animals survived until the end of the dosing period and no adverse effects or clinical signs of toxicity were observed during treatment. No significant findings were observed in changes in average body weights, average body weight gain, or hematology parameters. A small but statistically significant decrease in alkaline phosphatase activity in the 1000 mg/kg/d males was observed, but was not considered adverse. Minimal but statistically significant increases in albumin in 300 mg/kg/d males and in globulin in 1000 mg/kg/d females were not considered to be toxicologically significant since these were not dose-related. Statistically significant, but minimal, changes in average brain weight in the low dose females (higher than controls) and spleen to brain weight ratios in the intermediate dose group males (lower than controls) were also not considered to be toxicologically significant. No treatment-related histopathologic changes or gross abnormalities were observed. The researchers concluded that the test material containing Collagen was tolerated well in this rat study. 53
Human - Oral
Hydrolyzed Collagen/Gelatin
In a 4-mo dietary intake study of Hydrolyzed Collagen (interchangeably reported as Gelatin) for the potential role in enhancing bone remodeling in children, no adverse effects were observed. 55 The randomized double-blind study divided the children (ages 6-11 yr) in to 3 groups that received placebo (n = 18), Hydrolyzed Collagen (n = 20), or Hydrolyzed Collagen + calcium (n = 22) daily 250 mL dose.
Developmental and Reproductive Toxicity (Dart) Studies
No published DART studies on skin and connective tissue-derived proteins and peptides were discovered and no unpublished data were submitted.
Genotoxicity
In Vitro
Hydrolyzed collagen
No mutagenicity was observed in an Ames test of Hydrolyzed Collagen (30% solution with water; sourced from fish scales; MW ∼400 Da). 21 Salmonella typhimurium strains TA1535, TA1537, TA98, and TA100 and Escherichia coli strain WP2uvrA were used in this test, which was performed in accordance to OECD TG 471. The dose range was 50 to 5000 μg/plate, with and without metabolic activation.
In another Ames test performed in accordance to OECD TG 471, Hydrolyzed Collagen (20% solution with water; source from fish scales; MW ∼400 Da) was not mutagenic in S. typhimurium strains TA1535, TA1537, TA98, and TA100 and E. coli strain WP2uvrA, with or without metabolic activation. 27 The dose range was 50 to 5000 μg/plate.
Hydrolyzed Collagen (30% solution with water; sourced from fish scales; MW ∼400 Da) was not clastogenic in a Chinese hamster lung (CHL) cell line chromosome aberration test. 27 The cells were tested with and without metabolic activation.
Carcinogenicity
No published carcinogenicity studies on skin and connective tissue-derived proteins and peptides were discovered and no unpublished data were submitted.
Other Relevant Studies
Type 1 Hypersensitivity
Type 1 (ie, immediate) hypersensitivity reactions can occur in individuals allergic to certain proteins, such as those found in fish. An allergen must have at least 2 IgE-binding epitopes, and each epitope must be at least 15 amino-acid residues long, to trigger a Type 1 hypersensitivity reaction. 56 Type 1 responses can be elicited in sensitized patients when pairs of IgE molecules against a specific allergen are bound to receptors on the surface of mast cells and other cells that mediate immune reactions. The binding of an allergen molecule to 2 receptor-bound IgE molecules results in the crosslinking of the pair of IgE molecules. The crosslinking of sufficient numbers of IgE pairs bound to the receptors on the surface of a mast cell results in degranulation of the mast cell and the release of vasoactive amines, which are responsible for the Type 1 reaction. For some hydrolyzed proteins, the minimum number of amino acids (or weight-average MW) to elicit Type 1 hypersensitivity has been demonstrated with experimental data. For example, studies on hydrolyzed wheat protein show that hydrolysates with MWs less than 3500 Da do not have the properties required to induce Type 1 hypersensitivity. 9 Conclusive studies that detail the number of amino acids needed to trigger mast cell degranulation for hydrolyzed fish proteins, however, were not identified.
Skin prick tests and histamine release tests of fish Gelatin and codfish were completed in 30 fish-allergic patients (diagnosed in accordance with European Academy of Allergy and Clinical Immunology Guidelines). 57 Codfish-specific IgE was also measured in the patients and they underwent double-blinded, placebo-controlled food challenges with fish Gelatin. The fish Gelatin used for the study was made through acid extraction of codfish skins and had an average molecular weight of 60 000 Da. All 30 patients had positive skin prick tests, histamine release tests, and specific IgE to codfish. Skin prick tests and histamine release tests with fish Gelatin were positive in 3/30 and 7/30 patients, respectively. Oral challenge resulted in 2 patients reporting mild subjective reactions. One patient had a mild reaction to the placebo but not the fish Gelatin. The proportion of truly sensitive patients was estimated to be 0.03. The study authors concluded that the fish Gelatin in the study presented no risk to fish-allergic patients at doses typically used in foods (3.61 g).
The potential for tuna skin-derived Gelatin to induce allergic reaction in patients with fish allergy or sensitization was investigated using the serum samples of 100 consecutive allergic patients. 58 Serum IgE antibodies were tested against Gelatin and Hydrolyzed Gelatin extracted from yellowfin tuna skin and compared to extracts of yellowfin tuna flesh and skin and bovine or porcine gelatins. Of the 100 samples tested, only 3 exhibited reactivity to tuna skin-derived Gelatin (1 hydrolyzed, 2 non-hydrolyzed). No cross-reactivity was observed between bovine/porcine Gelatin and fish Gelatin.
Dermal Irritation and Sensitization Studies
Irritation
Dermal Irritation Studies of Hydrolyzed Collagen, Soluble Collagen, and Hydrolyzed Elastin.
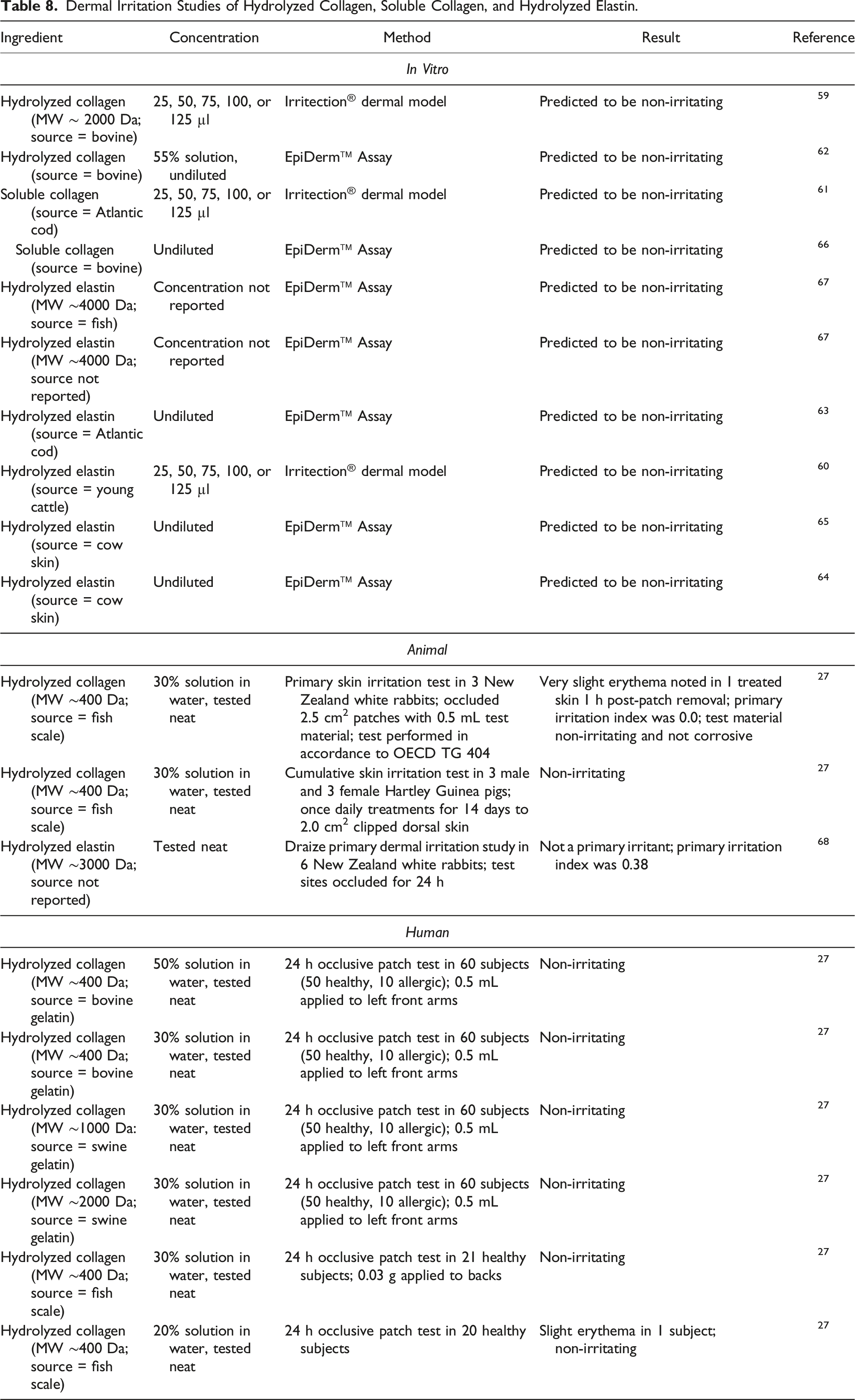
Animal
Hydrolyzed collagen
Primary skin irritation tests in rabbits indicated that Hydrolyzed Collagen was nonirritating or minimally irritating when tested at up to 100%. 2
Human
Hydrolyzed collagen
Irritation was not observed in human volunteers with healthy skin at concentrations up to 28%, but moderate irritation was observed in volunteers with dermatitis. 2
Sensitization
Animal
Hydrolyzed collagen
Hydrolyzed Collagen was nonsensitizing in guinea pig studies at up to 2%. 2
Hydrolyzed Collagen (30% solution in water; fish scale sourced; MW ∼400 Da) was considered to be non-sensitizing in a guinea pig maximization test using 15 Hartley guinea pigs and performed in accordance to OECD TG 406. 27 The animals received 7.5% active ingredient intradermally with Freund’s complete adjuvant during the first induction, while undiluted test material was applied to clipped dorsal skin under occlusive 48 h patch during the second induction. The animals were challenged with undiluted test material on clipped flank skin under occlusive 24 h patch. No reactions were observed in any animal 24 h and 48 h post-challenge patching.
Human
Hydrolyzed collagen
Formulations containing 0.5% to 28% Hydrolyzed Collagen produced some irritation but no sensitization in human repeated insult patch tests (HRIPTs). 2
In a HRIPT with 50 subjects, Hydrolyzed Collagen (20% solution in water; fish scale sourced; MW ∼400 Da) was not sensitizing. 27 The test material (0.2 mL) was applied to infrascapular skin with occlusive patches.
A study of sensitization to protein hydrolysates in hair care products was performed in 3 groups of patients. 69 The first group, which comprised 11 hairdressers with hand dermatitis, submitted to scratch and prick tests with 22 trademarked protein hydrolysates, including Soluble Collagen and Hydrolyzed Collagen, as well as quaternized hydrolyzed proteins. The second test group comprised 1260 consecutive adults with suspected allergic respiratory disease; they were subjected to skin prick tests with 1 to 3 of the protein hydrolysates. The third group of patients comprised 28 adults with atopic dermatitis and was also tested with a protein hydrolysate via a skin prick test.
Positive reactions were seen in a total of 12 patients (all female with atopic dermatitis) from 3 of the 22 protein hydrolysates. All 12 had reactions to hydroxypropyl trimonium hydrolyzed collagen. Three of the 12 also had a reaction to 1 trademarked version of Hydrolyzed Collagen (1% solution), while 1 other had a reaction to hydroxypropyl trimonium hydrolyzed milk protein. 69
Hydrolyzed Elastin
In an HRIPT with 52 subjects, Hydrolyzed Elastin (25% w/v in corn oil; MW = 3000 Da) did not produce dermal irritation or dermal sensitization. 70 The test patches were occlusive.
Phototoxicity
Hydrolyzed collagen
Hydrolyzed Collagen at up to 2% was not phototoxic to guinea pigs and rabbits, nor was it phototoxic or photosensitizing to humans at up to 0.5%. 2 UV-induced erythema was decreased after application of 10% solution of Hydrolyzed Collagen (MW = 1500 Da) onto the skin after irradiation.
Ocular Irritation Studies
Ocular Irritation Studies of Hydrolyzed Collagen, Soluble Collagen, and Hydrolyzed Elastin.
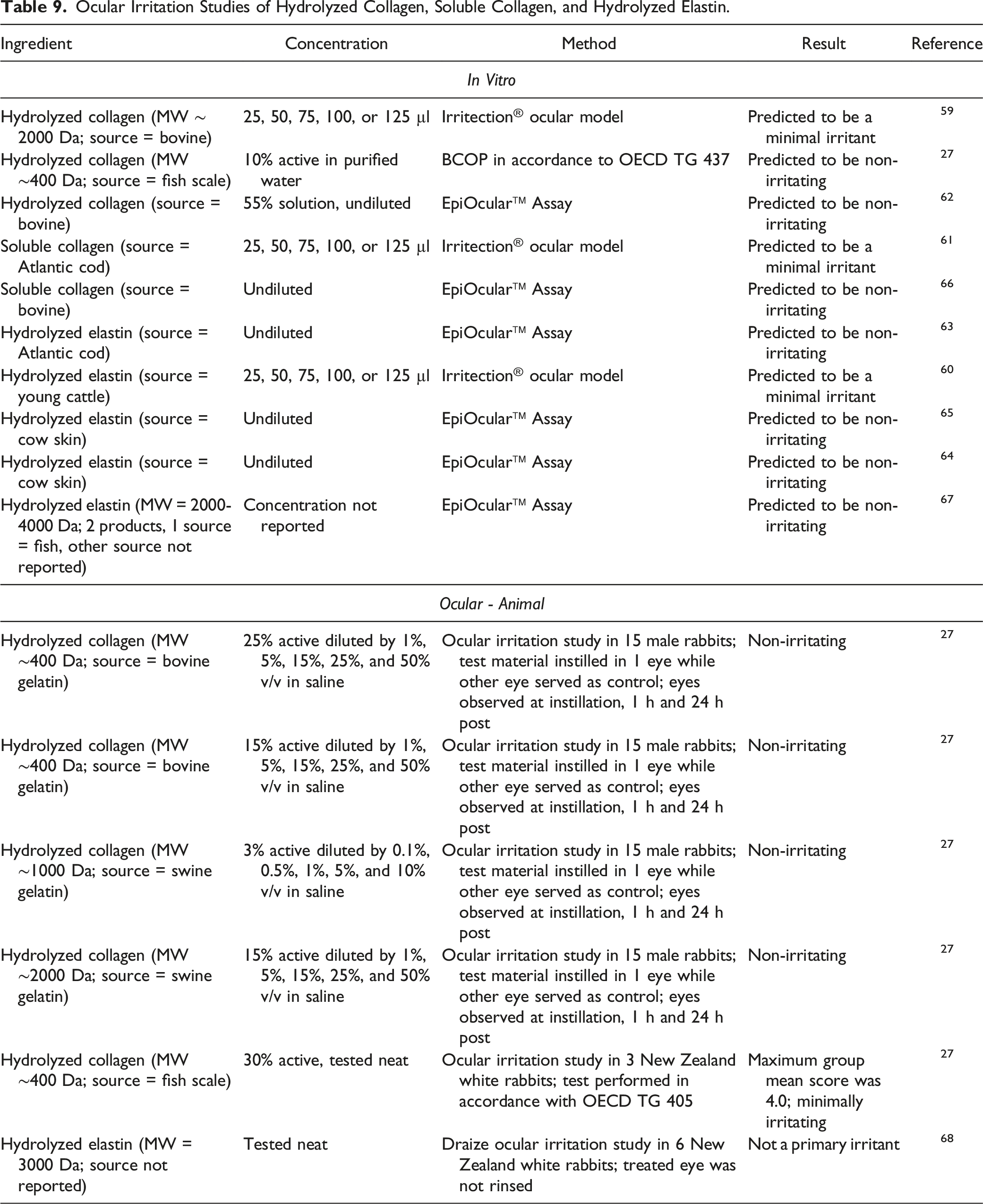
Animal
Hydrolyzed collagen
Hydrolyzed Collagen was minimally irritating to rabbit eyes when tested full-strength. 2
Clinical Studies
Case Reports
Elastin
A 26-yr-old woman with a history of fish allergy experienced urticarial eruptions following use of a cosmetic cream containing codfish-derived Elastin. 71 The patient’s serum total IgE level was 442 kU/l, and strong radioallergosorbent test (RAST) scores for specific IgE were observed for tuna, salmon, mackerel, flatfish, codfish, horsemackerel, sardine, and salmon roe. No prick-tests were performed because of the patient’s history of severe symptoms. Immunoblot analysis revealed that the patient had IgE antibodies against codfish Elastin, parvalbumin, Collagen, and transferrin. The molecular weight range of the proteins that the patient’s serum reacted with was 10 000 to 20 000 Da, which corresponded to the range of codfish Elastin. The company that produced the cosmetic cream reported that the Elastin in the cosmetic cream was derived from the skin and soft tissue of codfish.
Atelocollagen and hydrolyzed collagen
A 30-yr-old woman with a history of atopic dermatitis experienced anaphylaxis twice on separate occasions, once after consuming a fortified yogurt containing fish-sourced Hydrolyzed Collagen and once after consuming a gummy candy containing fish-sourced Hydrolyzed Collagen. 72 Fifteen months prior to the anaphylactic episodes, the patient had been applying a moisturizer containing Atelocollagen derived from fish to her impaired facial skin. The Atelocollagen in the product has a molecular weight of 350 000 Da. Skin prick tests on the patient were positive for fish-sourced Hydrolyzed Collagen in the food products, the moisturizer, Atelocollagen, and fish Gelatin. The tests were negative for Gelatin derived from porcine skin or bovine bone. The patient denied anaphylactic reactions following ingestion of raw or cooked fish. Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and IgE western blot analyses showed that the patient’s serum reacted with an approximately 140 000 Da protein of Atelocollagen and a 120 000 Da protein of Gelatin from fish Collagen. Weak reactions were observed with bovine bone Gelatin protein and no reactions were observed to porcine skin Gelatin protein or fish-sourced Hydrolyzed Collagen protein. The researchers of this case study speculated that the Atelocollagen (350 000 Da) was degraded on the skin surface by proteases into smaller peptides and induced sensitization, but did not rule out the possibility that intact Collagen or degradation products with greater than 4500 Da were antigens because of the patient’s impaired skin.
Hydrolyzed collagen
A 22-yr-old female reported contact urticaria following use of a hair conditioner that contained steartrimonium hydrolyzed animal protein. 73 She had a similar, less severe reaction the year before to another hair conditioner that also contained this ingredient. The patient also had a history of hay fever and recurrent hand dermatitis. Prick testing elicited strongly positive wheal and flare response to both hair conditioners, steartrimonium hydrolyzed animal protein, and other hair conditioners that contained protein, including Hydrolyzed Collagen in some products. Negative reactions were observed when the patient was tested with protein-free hair products. Prick tests with the standard series of allergens yielded positive results for grass mix, rye, English plantain, dust mite, cow’s milk, soybean, baker’s yeast, and wholegrain wheat. Tests with raw meat were negative. The patient’s total IgE was 221 kU/l. RASTs were negative to pork, beef, chicken, and mutton.
Summary
This report assesses the safety of 19 skin and connective tissue-derived ingredients, including Hydrolyzed Collagen, which has been previously reviewed by the Panel. Summary information presented in this safety assessment from the previous report is not repeated below.
Ingredients with the greatest number of reported uses in 2017 are Hydrolyzed Collagen (543 formulations) and Soluble Collagen (425 formulations); the majority of uses are in leave-on skin care products. Gelatin is used in a total of 334 formulations; the majority of the uses are in rinse-off bath soaps and detergents. The results of the concentration of use survey conducted in 2016 by the Council indicate Collagen has the highest reported maximum concentration of use; it is used at up to 96% in face and neck skin care products. Gelatin is used at up to 66% in bath oils, tablets, and salts. The other in-use ingredients are used at much lower concentrations.
A toxicokinetics study of fish-derived Gelatin (4000 mg/kg) in rats found that Gelatin has a high oral bioavailability.
A product containing 60% chicken-derived Collagen did not produce acute toxic effects in rats that were given a single oral dose of 5000 mg/kg.
No adverse effects were reported in a 2 wk oral study of shark skin-derived Gelatin in ovariectomized rats that received the test material at up to 40 mg/100 g daily.
In subchronic toxicity studies, rats tolerated daily oral dosing of a test material containing 60% Collagen. No adverse effects were reported in a 4-mo study of a dietary supplement containing a 250 mL dose of Hydrolyzed Collagen in human children
Gelatin and other skin and connective tissue-derived proteins may be sourced from fish, which is a major food allergen that can produce Type 1 hypersensitivity reactions in sensitized individuals. Researchers have reported a low risk of IgE-mediated reactions to fish Gelatin in individuals with fish allergies.
No dermal irritation was predicted based on in vitro studies of Hydrolyzed Collagen (bovine sourced; up to 55%), Soluble Collagen (fish and bovine sourced; undiluted), and Hydrolyzed Elastin (fish and bovine sourced; undiluted). No dermal irritation was observed in rabbits or guinea pigs treated with Hydrolyzed Collagen (fish sourced; 30% solution) and Hydrolyzed Elastin (source not reported; tested neat). Hydrolyzed Collagen (bovine, swine, and fish sourced) was not irritating in human dermal studies at concentrations up to 50% in water solution.
A guinea pig maximization test found Hydrolyzed Collagen (fish sourced; 30% solution in water) to be non-sensitizing. In HRIPT studies, Hydrolyzed Elastin (25% w/v in corn oil) and Hydrolyzed Collagen (fish sourced; 20% solution in water) did not produce dermal irritation or dermal sensitization. Hydrolyzed Collagen produced positive results in skin prick tests of dermatitic patients.
Hydrolyzed Collagen (fish and bovine sourced; up to 55%), Soluble Collagen (fish and bovine sourced; undiluted), and Hydrolyzed Elastin (fish and bovine sourced; undiluted) were predicted to be minimally or non-irritating in ocular in vitro studies. Hydrolyzed Collagen (fish, swine, and bovine sourced; up to 30%) and Hydrolyzed Elastin (source not reported; tested neat) were not irritating in rabbit ocular studies.
Case reports of dermal sensitization to cosmetics containing Elastin, Atelocollagen, and Collagen derived from fish have been described in the published literature. Reactions to Hydrolyzed Collagen have been reported as well.
No relevant published DART or carcinogenicity studies on skin and connective tissue-derived proteins and peptides were identified in a literature search for these ingredients, and no unpublished data were submitted.
Discussion
The Panel noted that there was a lack of systemic toxicity data (ie reproductive and developmental toxicity, genotoxicity, and carcinogenicity data); however, the Panel was not concerned that these proteins and peptides would cause adverse systemic effects in the general population. These proteins and peptides, similar to the other proteins and peptides reviewed by the Panel, are found in food, and daily exposures from the consumption of food can be expected to yield much larger systemic exposures to these ingredients than those from use in cosmetic products. The Panel also found that the earlier assessments of Hydrolyzed Collagen supported the safety of these ingredients in cosmetic products.
The Panel noted that fish proteins are known food allergens that can elicit Type 1 immediate hypersensitivity reactions when ingested by sensitized individuals. The Panel expressed concern that sensitized individuals would not easily recognize cosmetic products containing fish-derived collagen based on the current naming conventions used in the ingredient lists on product labels (eg, Collagen and Hydrolyzed Collagen may be sourced from fish, though “fish” is not in the ingredient name). In the absence of negative Type 1 immediate hypersensitivity data for fish-derived protein ingredients (or other information supporting an inability of the supplied ingredient to elicit such sensitization (eg, a maximum peptide length that is shorter than the minimum IgE-binding epitopes)), the Panel advised manufacturers to label products containing these fish-derived ingredients to inform individuals sensitized to fish proteins.
The Panel was also concerned about the inherent risks of using animal-derived ingredients in cosmetic products, namely the potential for transmission of infectious agents. While Gelatin and Collagen prepared exclusively from hides and skins do not have the propensity to carry disease, the Panel stressed that these ingredients must be free of detectible infectious pathogens (ie, BSE) if these materials are derived from other bovine materials. Raw material suppliers and formulators of these ingredients must assure that these ingredients are free from pathogenic viruses and other infectious agents.
The Panel discussed the issue of incidental inhalation exposure from hair care products, skin care preparations, face powders, and fragrances. There were no inhalation toxicity data available. Although the Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the 19 skin and connective tissue-derived proteins and peptides listed below are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Ammonium Hydrolyzed Collagen
Atelocollagen
Calcium Hydrolyzed Collagen*
Collagen
Elastin
Fibronectin
Gelatin
Hydrolyzed Actin
Hydrolyzed Collagen
Hydrolyzed Collagen Extract*
Hydrolyzed Elastin
Hydrolyzed Fibronectin
Hydrolyzed Gelatin*
Hydrolyzed Reticulin
Hydrolyzed Spongin*
MEA-Hydrolyzed Collagen
Soluble Collagen
Soluble Elastin*
Zinc Hydrolyzed Collagen*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author Contributions
Burnett, C. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.