Abstract
Colorectal cancer (CRC) is the second cause of cancer-associated death globally. Recently, herbal medicinal products and, in particular, zerumbone have been widely studied and used for cancer treatment as they induce significant anti-cancer effects. However, there is limited information about the anti-cancer effects of zerumbone in CRC. Therefore, we aimed to investigate the in vitro anti-cancer effects of the zerumbone in CRC, focusing on cell apoptosis and migration. Anti-proliferative and anti-migratory effects of zerumbone on HT-29 cells were evaluated using MTT and scratch wound healing assay, respectively. Quantitative real-time PCR (qRT-PCR) was performed to determine the mRNA expression levels of migration and apoptosis-related genes. Apoptosis and cell cycle distribution were evaluated by flow cytometry. The intracellular level of reactive oxygen species (ROS) was measured using a ROS assay kit. Additionally, matrix metalloproteinase-2/-9 (MMP-2/-9) activity was determined using gelatin zymography. Zerumbone suppressed the viability of the HT-29 cells dose-dependently while having less cytotoxicity on normal NIH/3T3 cells. Zerumbone induced apoptosis in HT-29 cells and arrested the cell cycle in the G2/M phase. These effects were associated with alteration in the expression of apoptosis-related genes (up-regulation of Bax and down-regulation of Bcl-2 genes). Zerumbone also enhanced the generation of ROS in HT-29 cells. Furthermore, zerumbone significantly inhibited the migration of HT-29 cells and decreased MMP-2/-9 mRNA expression and activity. Our findings provide a potential use for zerumbone to induce apoptosis and suppress metastasis in HT-29 cells; thus, it could be developed as a promising natural agent for future CRC therapy.
Introduction
There is a sharp rise in colorectal cancer (CRC) globally, particularly in developing countries, making it the second major cause of cancer mortality and the third most commonly diagnosed cancer. 1 Obesity, red meat and processed meat intake, smoking, alcohol consumption, a history of abdominal radiation, and a family history of cancer are well-known risk factors for CRC.2,3 The underlying cause of the majority of CRC treatment failures is primarily metastatic CRC cells and the subsequent development of metastatic cancer. Further, partially suppressing apoptosis is a hallmark of CRC, giving tumors a survival advantage that makes conventional treatment ineffective. 4 Currently, the main therapeutic approaches used for CRC therapy are surgery, chemotherapy, and radiotherapy. 5 However, due to the unwanted side effects of chemotherapeutic agents and poor prognosis after treatment with these conventional methods, there is an urgent need to find out new potential anti-cancer compounds with low adverse effects and high effectiveness.6,7
In recent years, herbal medicines and natural products have received more attention in the treatment of many cancers due to their low toxicity, high efficacy, and broad-spectrum properties.8-10 One of these natural compounds that have shown capability in the treatment of cancer is zerumbone. 11 Due to its minimal toxicity and excellent effectiveness, zerumbone, as a promising phytochemical, has been considered a viable alternative treatment.8,12,13 Zerumbone, a crystalline sesquiterpene extracted from subtropical ginger (Zingiber officinale), has been recognized as an effective herbal product and has various functions, including anti-pyretic, anti-nociceptive, anti-hypersensitive, anti-microbial, anti-inflammatory, hepatoprotective, and anti-diabetic with the minimum undesirable effect on natural cells. 11 Additionally, zerumbone may also be used as an anti-cancer agent, displaying a variety of effects on proliferation, angiogenesis, and apoptosis in a wide range of cancers, which has piqued the interest of several researchers. 14 It has been shown that zerumbone could inhibit the invasion of various tumor cells via the suppression of matrix metalloproteinases such as MMP-2 and MMP-9 enzymes.11,15,16 MMP-2 and MMP-9 are considered as the main factors of proteolytic degradation of extracellular matrix (ECM) that play an important role in metastasis and cancer progression.17,18 In addition, according to recent studies, zerumbone can induce apoptosis in cancer cells by increasing the expression of pro-apoptotic Bax and decreasing the expression of anti-apoptotic Bcl-2 genes in glioma cells. 19 As a consequence, it would be interesting to determine the anti-proliferative and anti-migration impact of zerumbone in CRC and to offer it as a possible alternative candidate agent in CRC treatment. Therefore, this study was conducted to assess the anti-migration and tumor-inhibitory effects of zerumbone on HT-29 human colorectal cells.
Materials and Methods
Materials
High-glucose Dulbecco’s Modified Eagle’s Medium (DMEM) and fetal bovine serum (FBS) were purchased from Gibco (UK). MTT [3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide], penicillin-streptomycin, dimethyl sulfoxide (DMSO), propidium iodide (PI), and trypsin-EDTA were obtained from Sigma-Aldrich (St Louis, MO, USA). The zerumbone (extracted from Zingiber officinale) and dichloro-dihydro-fluorescein diacetate (DCFDA)/H2DCFDA-cellular reactive oxygen species (ROS) detection assay kit were supplied from Abcam (Cambridge, United Kingdom). The Annexin V-FITC/PI apoptosis assay kit was procured from BioLegend (San Diego, CA, USA). The protein assay kit, bicinchoninic acid (BCA), was purchased from Pierce Co. (Pierce, Rockford, IL, USA).
Cell Culture
The human HT-29 colorectal cancer cells and NIH/3T3 murine embryonic fibroblast cells as a control normal cell line, (National Cell Bank of Iran, Pasteur Institute, Iran) were cultured in DMEM supplemented 10% FBS and 1% penicillin-streptomycin. These cell lines were maintained in a humidified incubator at 37°C with 5% CO2.
MTT Assay
The cytotoxicity of zerumbone was assessed by MTT assay. Briefly, HT-29 and NIH/3T3 cells were cultured overnight in 96 wells (1 × 104 cells/well). The cells were exposed to various concentrations of zerumbone (0-800 μM) for 24 h. After that, the media was removed and the cells were incubated with 100 μL FBS-fresh medium containing 0.5 mg/mL MTT, and after 3 h, 200 μL DMSO was added to each well to dissolve the formed formazan crystals. Finally, the optical density was measured at 570 nm using a Stat FAX303 plate reader. All the samples were tested in triplicate.
Scratch Wound Healing Assay
The scratch wound healing assay was performed to evaluate the anti-migration effects of zerumbone on the HT-29 cell line. For this, 7 × 105 cells/well were plated into a 6-well plate then incubated to reach about 95% confluence. Next, a scratch was created in the monolayer utilizing a pipette tip, and cellular debris was washed with PBS. After that, the cells were treated with the desired concentration of zerumbone, and the images of the wounded gap were recorded at 2, 72, and 120 h and analyzed by Image J software.
RNA Extraction and Quantitative Real-Time PCR
The sequences of specific primers for qRT-PCR.
Intracellular Reactive Oxygen Species Assay
In this method, we measured the intracellular accumulation of ROS with a common commercial ROS detection kit as previously described. 19 Briefly, HT-29 cells (25 × 103) were cultured into 6-well plates and incubated overnight. Then, the cells were washed and incubated for 30 minutes with H2DCFDA solution (25 μM) in the dark place. After rewashing, the cells were treated with N-acetyl-cysteine (NAC) (10 mM) or zerumbone (40 and 80 μM) and NAC in combination with zerumbone for 4 h (triplicates for each sample). Tert-butyl hydroperoxide (TBHP, 150 μM) was used as a positive control. Finally, the fluorescence was measured (Excitation/Emission: 485/535 nm) by the fluorescence plate reader FACScan (Becton Dickinson, San Jose, USA).
Cell Cycle and Apoptosis Flow Cytometry Analyses
The effect of zerumbone on the cell cycle distribution was determined using flow cytometry. For this purpose, 7 × 105 HT-29 cells were treated with zerumbone (0, 40, and 80 μM) for 24 h. Afterward, the cells were trypsinized and resuspended in cold PBS and fixed with 70% ethanol at 4°C for 2 h. Subsequently, the cells were incubated with PBS containing 100 μg/mL RNase A and 0.1% Triton X-100 at 37°C for 20 min. Finally, all samples were stained with 400 μl of 1 mg/mL PI solution and incubated in the dark for 15 min and analyzed by flow cytometry.
Apoptosis of HT-29 cells in response to zerumbone (0, 40, and 80 μM) was measured by FITC Annexin V Apoptosis Detection Kit with PI (BioLegend, CA, USA) according to the manufacturer’s protocol as previously described. 21 Flow cytometric analysis was performed using a BD FACS Calibur™ flow cytometer (Becton Dickinson, Mountain View, CA, USA), and the data were analyzed by FlowJo software (v10, FlowJo, Ashland, USA).
Gelatin Zymography
Gelatin zymography was carried out to detect the enzymatic activity of MMP-2 and MMP-9 in the HT-29 cell line. 22 Briefly, the cells were treated with different concentrations of zerumbone (0, 12.5, and 25 μM) and incubated for 24 h. The supernatant of culture media was collected, and protein content was measured utilizing the BCA method. Equal amounts of samples (50 μg/lane) were loaded onto a 12% SDS–PAGE gel containing gelatin (0.1% w/v). After electrophoresis, the separating gel was washed twice for 30 minutes in 2.5% Triton X 100 and incubated for 24 h at 37°C in the developing buffer (containing 2.5% Triton X-100 in 50 mM Tris pH 7.4, 5 mM CaCl2, and 1 μM ZnCl2). Next, the gel was stained with Coomassie brilliant blue R250 and was de-stained in 10% acetic acid until white bands appeared. The gel was scanned by the GS-800TMcalibrated densitometer (Bio-RAD, USA), and zones of gelatinase activity were quantified using Image J software.
Statistical Analysis
All results were analyzed utilizing GraphPad Prism®8.2.1 (GraphPad Software, San Diego, CA, USA). The quantitative ratios of different groups were compared using the one-way ANOVA followed by the Dunnett test. All experiments were performed in triplicate and the results were reported as mean ± standard deviation (SD). The statistical significance was considered for values P ≤ 0.05.
Results
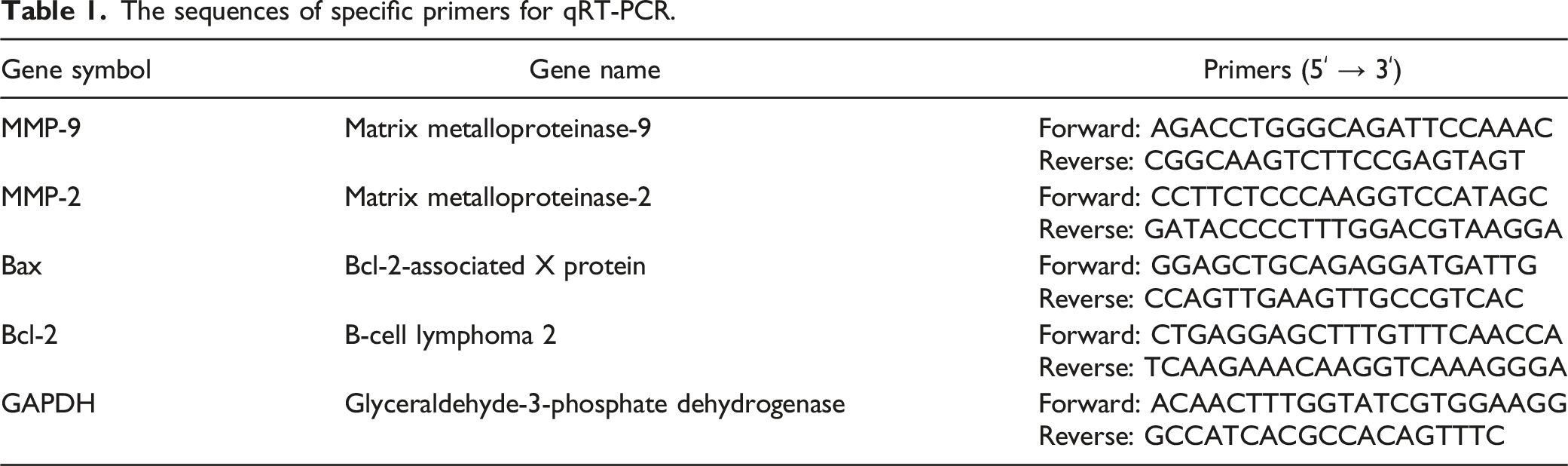
Zerumbone Suppresses HT-29 Cells Proliferation
The HT-29 cells were exposed to the desired concentrations of zerumbone for 24 h, and cell viability was analyzed compared to the control group. As shown in Figure 1, zerumbone exerted a significant inhibitory effect on HT-29 cells in a dose-dependent manner. The IC50 value of zerumbone in NIH/3T3 was higher than IC50 in HT-29 cells after 24 h incubation. In subsequent experiments, we have chosen concentrations lower than the IC50. Cell viability and IC50 values in both HT-29 colorectal cancer cells (A) and NIH/3T3 normal fibroblast cells (B) in the presence of zerumbone.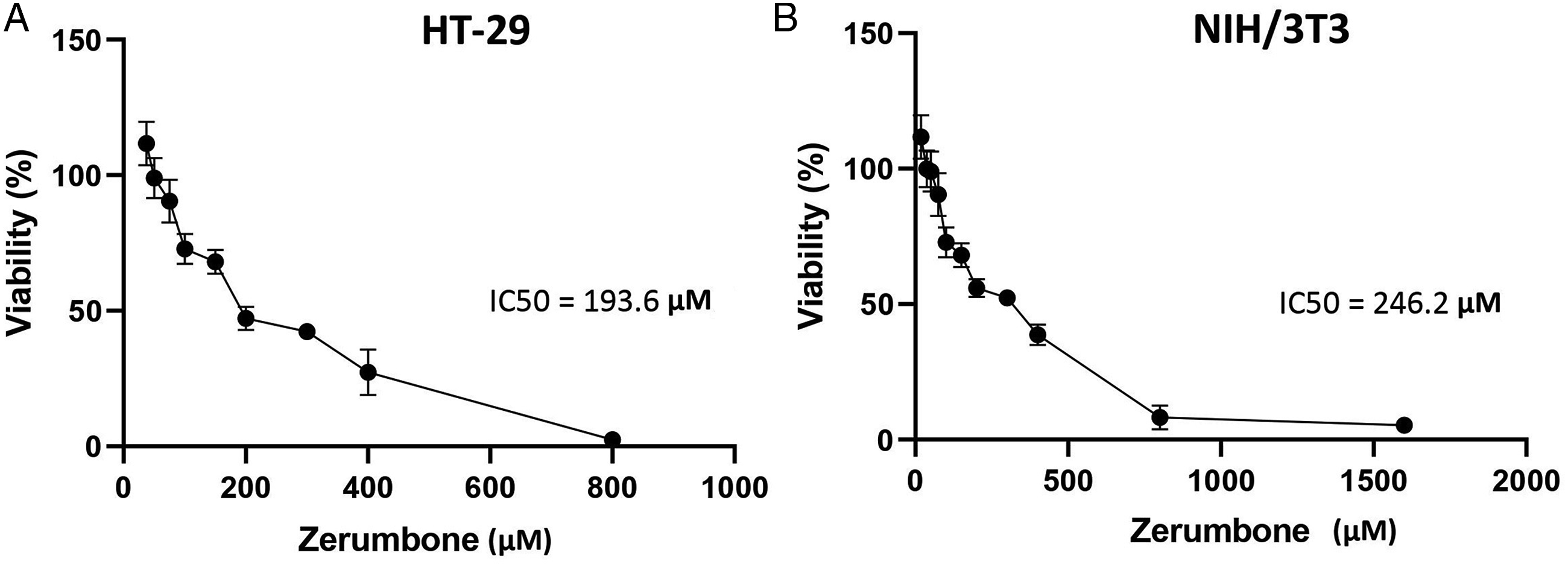
Zerumbone Inhibits the Migration of HT-29 Cells
We performed the scratch wound healing assay to evaluate the anti-migration effects of zerumbone on HT-29 cell line. The results showed that zerumbone significantly prevented the migration of HT-29 cells compared with control after 120 h (Figure 2). The effect of zerumbone on the HT-29 cells migration. HT-29 cells were treated with 12.5 μM (1/16 IC50) and 25 μM (1/8 IC50) of zerumbone, and the migrating ability of cells was investigated using the wound healing scratch assay. The scratch gap was photographed after 2, 72, and 120 h treatment with zerumbone and the wound area was quantified using the Image J software. The data are shown as the mean ± SD (** P ≤ 0.01 and *** P ≤ 0.001 in comparison with the control group).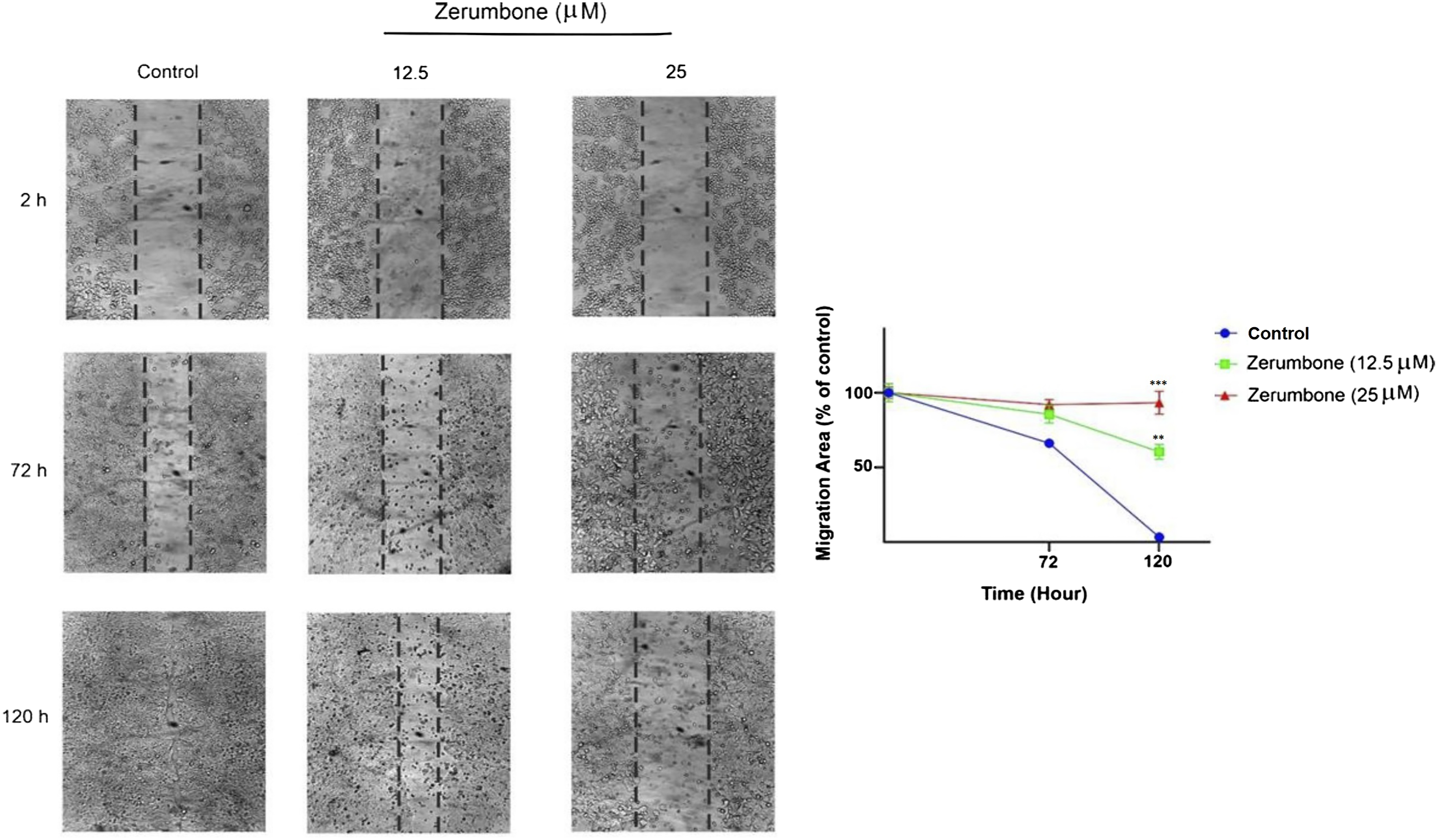
Zerumbone Inhibits the mRNA Expression of Migration- and Apoptosis-Related Genes
We evaluated the effect of zerumbone on mRNA expression of genes implicated in migration (MMP-2 and MMP-9) and apoptosis (Bax and Bcl-2) using the qRT-PCR method. As shown in Figure 3, zerumbone causes a significant reduction in mRNA levels of MMP-2 and MMP-9 compared to the control group. In addition, treatment with zerumbone raised the mRNA levels of the pro-apoptotic Bax and reduced the mRNA levels of the antiapoptotic Bcl-2 at 40 μM (P < 0.05). The mRNA expression of migration- and apoptosis-related genes in HT-29 cells treated with zerumbone. Zerumbone decreased the MMP-2/-9 and Bcl-2 mRNA expression and increased the expression levels of Bax mRNA at the concentration of 40 μM compared to the control group. The data are expressed as the mean ± SD. * P ≤ 0.05 and indicates statistical significance.
Zerumbone Induces ROS Levels in HT-29 Cells
In order to investigate the role of ROS in zerumbone-induced cytotoxicity, we evaluated the intracellular ROS levels in the HT-29 cells. As shown in Figure 4(a), zerumbone caused a significant increase (P < 0.05) of ROS levels in HT-29 cells compared to the control group in a dose-dependent manner, leading to oxidative damage to HT-29 cells. Also, results revealed that treatment with 10 mM N-acetyl-cysteine (NAC) as a ROS inhibitor, significantly reduced intracellular ROS generation by zerombone in HT-29 cells. Moreover, the NAC increased the cell viability after 24 h treatment with zerumbone (Figure 4(b)). The effect of zerumbone on the ROS levels of HT-29 cells. Assessment of ROS levels was carried out using a fluorimeter. (A) HT-29 cells were treated by zerumbone (40 and 80 μM) for 4 h, which led to an increase in ROS levels compared to the control group. Besides, N-acetyl cysteine (NAC, 10 mM) reduced the zerumbone-induced ROS production after 4 h treatment. (B) N-acetyl cysteine (NAC, 10 mM) combined with zerumbone increased the viability of the HT-29 cells at concentrations of 40 and 80 μM after 24 h treatment compared with the same concentrations. The data are shown as the mean ± SD (## P ≤ 0.01 and ### P ≤ 0.001 in comparison with the same concentration, * P ≤ 0.05, ** P ≤ 0.01, and *** P ≤ 0.001 in comparison with the control group).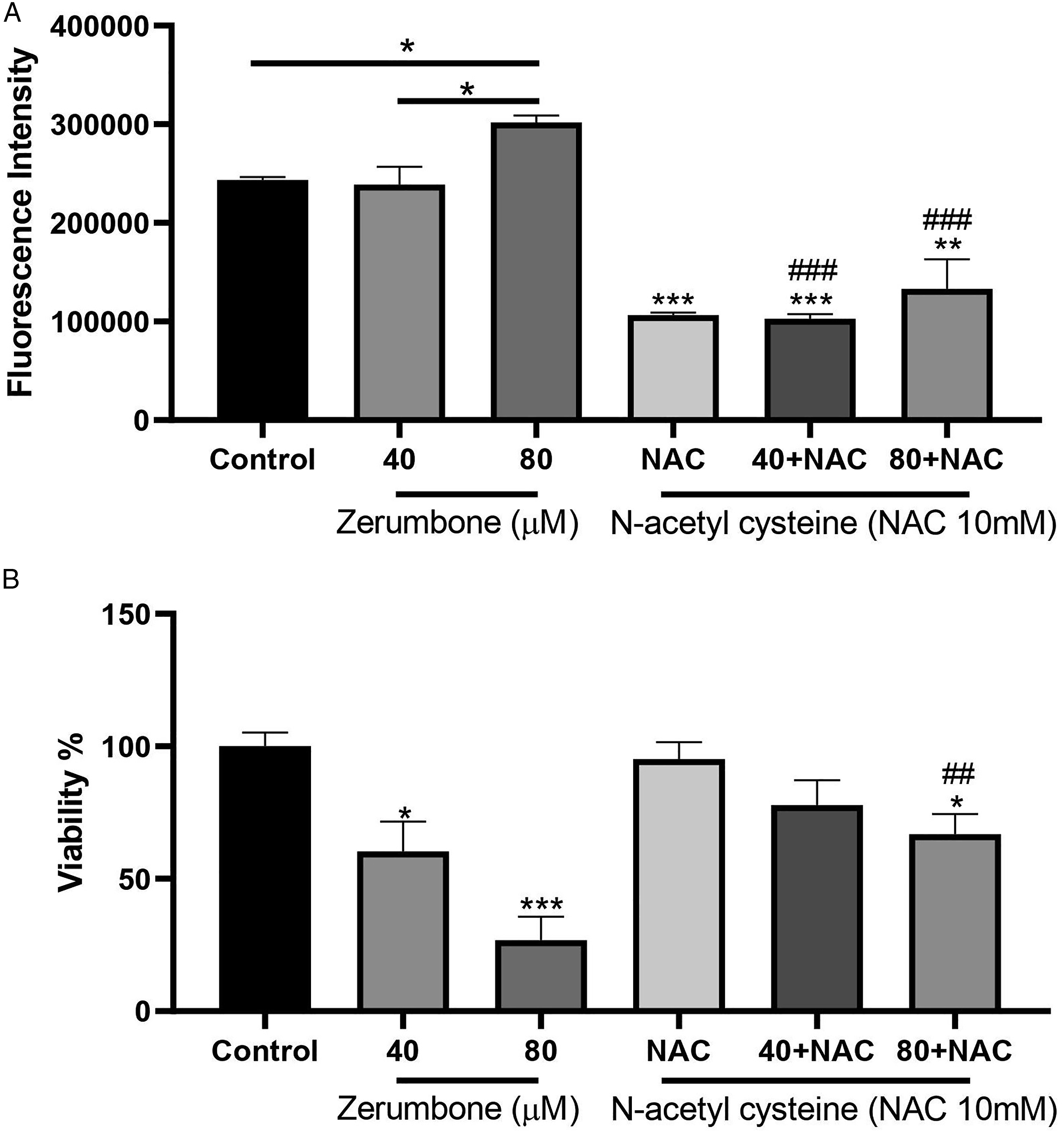
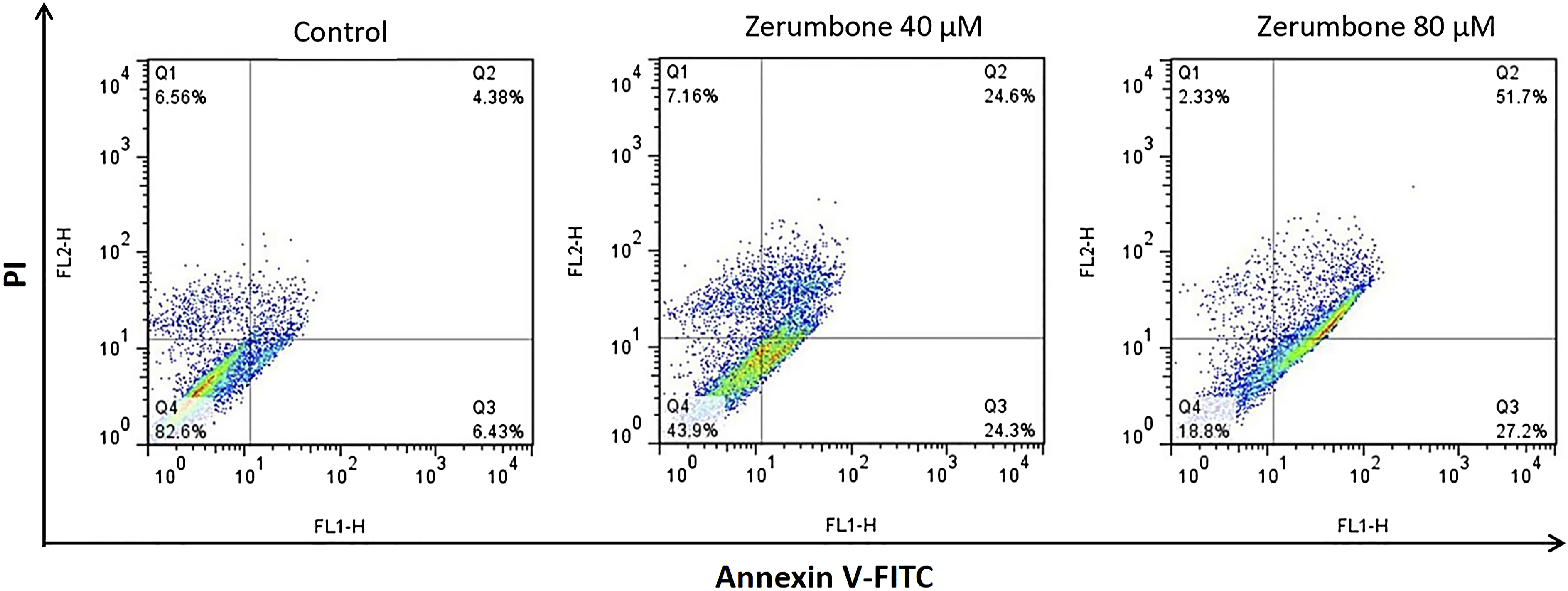
Zerumbone Induces Apoptosis and Cell Cycle Arrest at the G2/M Phase in the HT-29 Cells
The results of cell cycle analysis using flow cytometry indicated that zerumbone dramatically increased the sub-G1 area and induced cell cycle arrest by increasing the number of apoptotic cells at the G2/M phase (Figure 5). In the next step, we investigated the effect of zerumbone on HT-29 cell apoptosis using the Annexin V-FITC/PI staining method. As indicated in Figure 6, treatment with 40 and 80 μM of zerumbone for 24 h showed a markedly increased proportion of apoptotic and necrotic cells compared to the control group. The effect of zerumbone on the cell cycle distribution. The HT-29 cells were treated with zerumbone (40 and 80 μM), which led to an increase in the sub-G1 area and the number of apoptotic cells in the G2/M phase and subsequently arrested the cell cycle at the G2/M phase. Zerumbone induces apoptosis in HT-29 cells. The HT-29 cells were treated with zerumbone (40 and 80 μM) and stained by Annexin V-FITC/PI. Q1, Q2, Q3, and Q4 quadrants represent necrosis, late apoptosis, early apoptosis, and viable cells, respectively.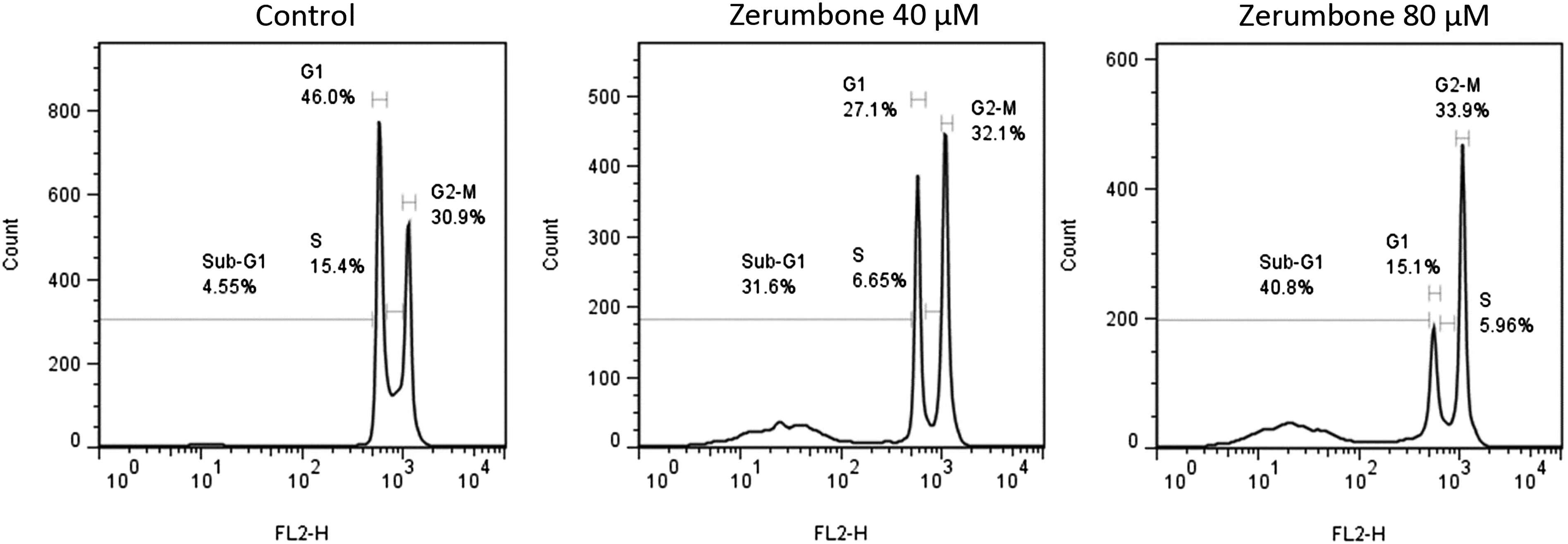
Zerumbone Inhibits the Activity of MMP-2 and MMP-9 in HT-29 Cells
To investigate the effect of zerumbone on the enzymatic activity of MMP-2 and MMP-9, we used the zymography method. The quantitative analysis of the gelatinolytic zones revealed that the zerumbone at the concentration of 25 μM significantly reduced the enzymatic activity of MMP-2 and MMP-9 in HT-29 cells (Figure 7). The effect of zerumbone on the enzymatic activity of MMP-2 and MMP-9 in HT-29 cells. Cells were treated with 12.5 μM and 25 μM of zerumbone for 24 h, and enzymatic activity of MMP-2 and MMP-9 were assessed by gelatin zymography. Gelatinase zymogram (A) and densitometry analysis of MMP-2 (B) and MMP-9 (C) in HT-29 cells. The data are shown as mean ± SD (** P ≤ 0.01).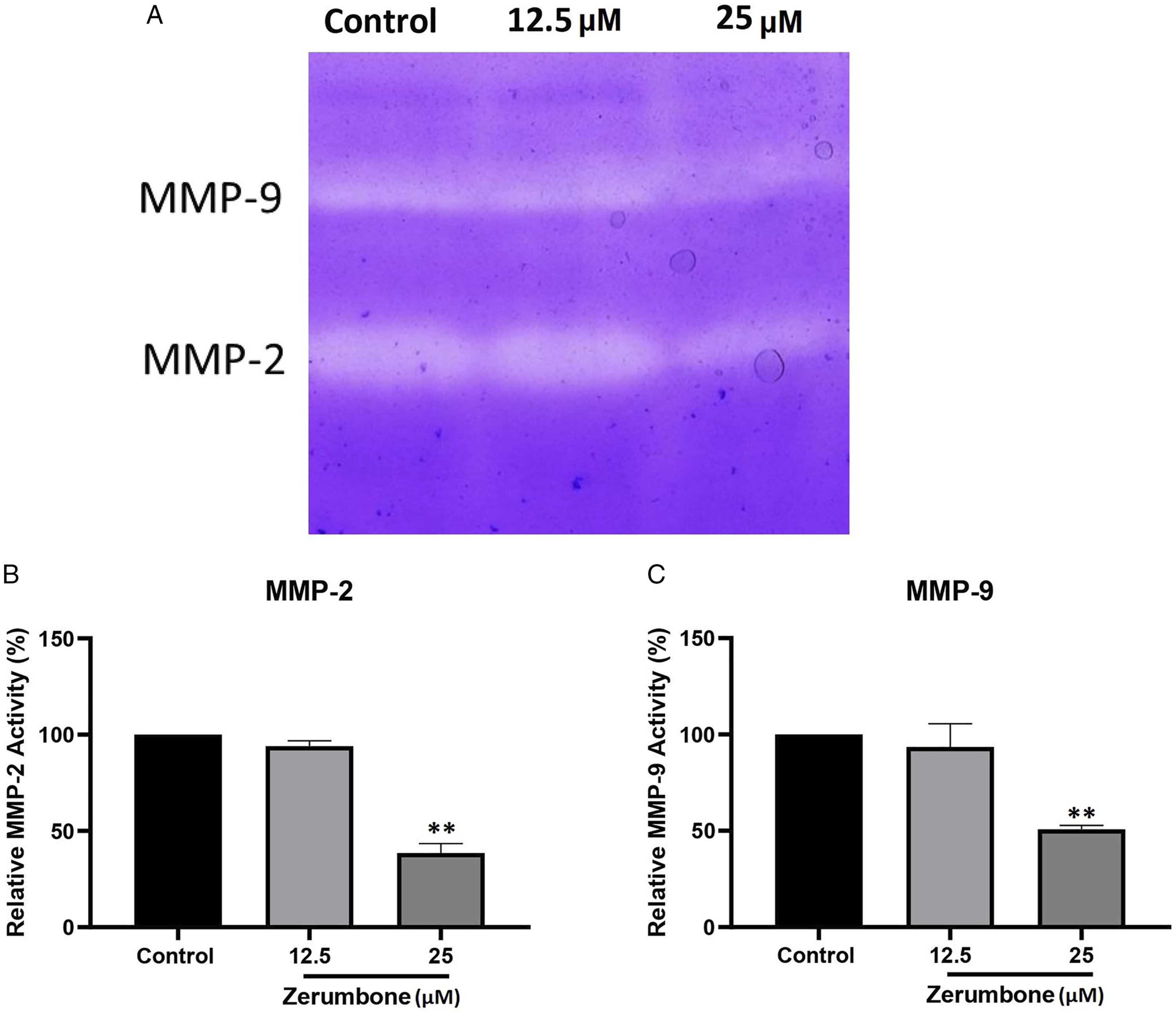
Discussion
In this study, we investigated the impact of zerumbone on metastasis and apoptosis of HT-29 cells in vitro. We found that zerumbone could significantly inhibit the proliferation and migration of CRC cells. Relative expression analysis of genes involved in metastasis (MMP-2 and MMP-9) and apoptosis (Bax and Bcl-2) demonstrated that zerumbone inhibits metastasis and enhances apoptosis in CRC HT-29 cell line. In addition, we showed that zerumbone induced apoptosis through intracellular ROS elevation and cell cycle disruption. According to these findings, zerumbone can inhibit migration and induce apoptosis in HT-29 cells. The viability and proliferation of cells are necessary for developing a tumor and subsequently metastasis. 23 Several studies have revealed that zerumbone has potent anti-proliferative activity against various cancer cells.11,24,25 Hoffmann et al have shown that zerumbone could inhibit cancer cell proliferation in vitro via regulation of intracellular redox in colon cancer, unlike the normal cells. 26 In line with the antiproliferative effect of zerumbone, we found that zerumbone decreases the viability and proliferation of HT-29 cells with less cytotoxicity on normal NIH/3T3 cells. Consistently, Murakami et al 27 reported that zerumbone has a selective cytotoxic effect on human colonic adenocarcinoma cell lines as it inhibited the proliferation of human colonic adenocarcinoma cell lines, while the growth of normal cell lines was less affected. Therefore, these findings imply the particular inhibitory role of zerumbone on HT-29 cancer cells.
Many studies have shown that matrix metalloproteinases such as MMP-2 and MMP-9 play an important role in the metastasis process.28,29 Jalili Nik et al 15 found that zerumbone significantly reduced the enzymatic activity of MMP-2/-9 in glioblastoma multiforme and inhibited the migration and metastasis of U-87 MG cells. Also, zerumbone suppressed migration mediated by decreasing MMP-2/-9 in HCC1806 breast and HepG2 hepatoma cancer cells.30,31 Therefore, we investigated the anti-migratory and anti-metastatic effects of zerumbone against HT-29 colorectal cancer cells. The wound healing scratch assay analysis showed that treating with zerumbone abolished CRC cells migration. This result was confirmed by decreasing expression of MMP-2 and MMP-9 in response to zerumbone, as evaluated by qRT-PCR. Besides, the enzymatic activity of MMP-2 and MMP-9 of HT-29 cells in the presence of zerumbone was significantly suppressed, indicating the anti-metastatic activity of zerumbone in CRC cells.
It is currently widely accepted that cancer develops due to the deregulation of several biological pathways that evading from apoptosis and cell cycle alteration is closely associated with uncontrolled cell growth and subsequent tumor development. 32 Therefore, we investigated the effects of zerumbone on cell cycle progression and apoptosis induction in HT-29 cell line. In the present study, we found that zerumbone arrested the cell cycle of HT-29 at the G2/M phase dose-dependently, causing cell proliferation inhibition. Increasing evidence suggests that different processes like regulated cyclin complexes, tubulin polymerization disruption, and spindle assembly interruption play a key role in G2/M phase arrest, which is mediated by various anticancer compounds.33,34 Studies have shown that zerumbone could cause cell cycle arrest in the G2/M phase; therefore, zerumbone could be suggested as a potential anti-tumor agent in oral cancer and hepatoma cells.31,35 A study also demonstrated that zerumbone mediated apoptosis with a cell cycle arrest in the G2/M phase via Bax/Bak related to apoptosis and downregulation of Cdc25C, Cdc25B, cyclin B1, and Ddk1 in breast cancer. 36
In the next step, we examined the externalization of phosphatidylserine during apoptosis via Annexin V-FITC staining for further confirmation. The induction of apoptosis has been proven to be associated with regulating the expression of genes such as Bax pro-apoptotic and Bcl-2 anti-apoptotic. 37 In this regard, S D Wang et al 38 found that zerumbone downregulated Bcl-2 gene and protein levels, while upregulated gene and protein levels of Bax and activated caspase-3 in A375 melanoma cells. Also, Jegannathan et al 39 have indicated that treatment with zerumbone caused significant growth inhibition and apoptosis induction via upregulation of Bax/Bcl-2 ratio in laryngeal carcinoma. Consistent with these results, our study showed that the zerumbone could increase the expression of Bax and decrease the expression of Bcl-2 in HT-29 CRC cells, confirming the induction of apoptosis through these genes.
Previous studies have suggested that ROS are one of the crucial factors in apoptosis induction, and the majority of anti-cancer agents act by increasing ROS production. 40 ROS are generated by regular oxygen metabolism. However, when ROS levels are above a certain threshold, they may cause cell damage in tumor cells. On the other hand, in different types of cancers, high levels of ROS have been indicated associated with tumor growth and development.41,42 In a study, treatment with zerumbone increased ROS levels and decreased ROS-induced cell viability in U-87 MG cells. 19 In the current study, evaluation of intracellular ROS generation was performed to demonstrate zerumbone-induced cytotoxicity and its association with ROS levels. Results showed that zerumbone caused increased intracellular ROS after 4 h treatment. It is noteworthy that these cytotoxic effects of zerumbone were reversed in the presence of NAC (an antioxidant agent and ROS scavenger). Therefore, it can be suggested that zerumbone-induced cytotoxicity may be due to the production of intracellular ROS.
Taken together, our findings suggest that zerumbone effectively regulates the proliferative and migrative phenotypes of HT-29 cells via altering the levels of apoptosis-related genes and ROS, promoting cell migration, and increasing MMP-2 and MMP-9 expression and activity. Thus, zerumbone can be developed as an effective anti-cancer agent for future of CRC therapy, but more research is required to establish this. Although these findings are promising, however, certain limitations need to be considered. In this study, we only examined the in vitro anti-tumor effects of zerumbone in HT-29 cell line; thus, it would be conceivably beneficial to analyze in multiple cell lines and CRC animal models to verify our findings. The pathogenesis of CRC is complex, so there is much to be learned concerning the anti-tumor mechanisms of zerumbone. Therefore, the therapeutic potentials of zerumbone by targeting different oncogenic pathways should be precisely identified in the future both in vitro and in vivo. Importantly, there is little information regarding the synergistic therapeutic potential of zerumbone and chemotherapeutic drugs, thus examining the combination of zerumbone with CRC chemotherapeutic drugs would provide directions to promote future therapeutic strategies in CRC. It is important to note that the poor aqueous solubility and bioavailability of zerumbone are still the major limitations hindering its clinical applications. Therefore, it is important to follow up with further studies on the characterization of the active compound of zerumbone and developing novel formulations of zerumbone to overcome zerumbone biopharmaceutical challenges.
Footnotes
Author Contributions
Fezzeh Memari contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Farshad Mirzavi contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; Mohammad Jalili-Nik contributed to analysis and critically revised manuscript; Amir R. Afshari contributed to interpretation, drafted manuscript, and critically revised manuscript; Ahmad Ghorbani contributed to acquisition, drafted manuscript, and critically revised manuscript; Mohammad Soukhtanloo contributed to conception and design, contributed to acquisition and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge Mashhad University of Medical Sciences (MUMS) for providing a financial grant (Grant number: 991535).
