Abstract
Apoptosis induction in cancer cells is one of the most efficient ways to treat cancer and find anticancer compounds. The aim of this study was to evaluate the cytotoxic effects of heat-killed indigenous probiotic bacteria and apoptosis induction in the HT-29 human colon adenocarcinoma cell line. The growth-inhibitory effects of probiotic heat-killed Lactobacillus brevis and Lactobacillus paracasei isolated from the traditional Iranian food “Terxine” on the HT-29 cell line were determined by 3-(4,5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide (MTT) assay. Flow cytometry by Annexin-FITC kit, DNA fragmentation assay, 4,6-diamidino-2-phenylindole staining, and the expression of Bax, Bcl2, caspase-3, and caspase-9 were used to analyze apoptosis. MTT results demonstrated that the heat-killed bacteria inhibited the proliferation of HT-29 cells and induced apoptosis in a time-, dose-, and strain-dependent manner. The results demonstrated that both bacteria could induce apoptosis in the HT-29 cell line. Heat-killed probiotic bacteria increased the expression of Bax, caspase-3, and caspase-9 mRNA levels in HT-29 cell lines. Also, heat-killed probiotic bacteria reduced the expression of Bcl2 in HT-29 cells. The heat-killed probiotic bacteria in this study exhibited potent growth inhibitory effects on cancer cells. The results demonstrated that L. brevis has a greater ability to inhibit the growth of HT-29 cells and induce apoptosis, compared with L. paracasei. It is proposed that these bacteria can be used as biological products for the treatment and prevention of cancer, pending further investigation.
Introduction
Cancer is the second leading cause of death in the world after cardiovascular diseases. It is caused by mutations in the genes that control the vital pathways of cells, including the pathways for growth and apoptosis. Colorectal cancer is the third most common form of cancer in Western nations. 1 As for Iran, this disease ranks as the third most common among men and the fourth most common among women. 2 Colon cancer is associated with a number of genetic disorders as well as environmental factors, 3,4 meaning that lifestyle and nutrition can play an important role in its development. 5 Probiotics are living, nonpathogenic microorganisms in certain foods, which, when adequately ingested, exhibit a positive influence on the host’s health and appear to have antitumor effects as well. 6 –8 According to certain in vitro and in vivo studies, probiotics act by preventing the conversion of procarcinogens to carcinogens, binding with and deactivating the mitogenic compounds, reducing the growth of pro-carcinogenic bacteria, reducing the absorption of mitogens, and improving the performance of the immune system against cancer cells. 9
In 2011, Taverniti and Guglielmetti suggested the term “Parabiotic” as a new term besides Probiotic. This term indicates that nonliving probiotic bacteria can have a positive health impact on humans, as many properties linked to the efficiency and therapeutic characteristics of probiotic bacteria are independent of their survival. 10
Recent studies have indicated that nonliving probiotic bacteria, including the inactive complete cells (heat-killed bacteria), exopolysaccharide extracts, peptidoglycans, cellular extracts, and various other cellular components exhibit potentially curative, antitumor and anticancer activities, and also stimulate the immune system. 11 –14
The problem with living probiotics cells is that they must be able to adapt to the gastrointestinal tract to survive and must be administered in sufficient amounts to induce biological responses. Living probiotic cells affect the gastrointestinal microflora and have immunomodulatory effects, while dead cells components can have anti-inflammatory effects. Producing anti-inflammatory, immunological, and biological responses is possible with various ranges of the killed bacteria. Also, using killed probiotic cells, it is possible to use wider ranges of microorganisms for research. On the other hand, standardization based on the killed probiotic cells is relatively much easier than live probiotics, and the usage of dead probiotic bacteria is safer and reduces the risk of pathology in immunocompromised individuals, as well as the transmission rate of infections among individuals. The other benefits of dead probiotic bacteria are the ease of their usage, higher sustainability, and higher self-life. 15 –17
The cytotoxic effects and induced apoptosis by heat-killed probiotic bacteria have been reported in various studies. 7,14 –16,18 –20
The immunomodulatory effects of probiotic bacteria have been shown to prevent tumor growth by modulation of cytokine production, signaling pathways associated with carcinogenesis and epithelial cell growth, leading to a reduction in tumor size in different types of cancers. 21 –27
Since the antiproliferative and anticancer effects of probiotic bacteria are strain-dependent, the study on heat-killed probiotic bacteria requires further investigation before they could be used as supplementary medications that offer higher safety and stability than their living counterparts. The use of potent probiotics in dietary supplements, especially heat-killed probiotics, can help in the formulation of food products that inhibit the growth of cancer cells and ultimately achieve cancer prevention. The products based on inactivated cells are relatively more homogeneous and last longer than the ones from living cells. Proactive steps need to be taken in order to prevent and treat colon cancer.
The aim of this study was to evaluate the antiproliferative and apoptosis-inducing effects of the heat-killed indigenous probiotic bacteria, Lactobacillus brevis and Lactobacillus paracasei, on the HT-29 cell line, using a noncancerous cell line as a comparative control. In addition, due to the importance and involvement of the Bax, Bcl2, caspase-3, and caspase-9 genes in the intrinsic mitochondrial pathway and the induction of apoptosis, the expression levels of these genes were also studied.
Materials and methods
Bacterial strains
In this study, we used L. paracasei and L. brevis, with access numbers IBRC_M10784 and IBRC_M1079, respectively. These strains were isolated by Tajabadi Ebrahimi and his colleagues from the traditional and indigenous Iranian food “Terxine” and were registered with the Iranian Biological Resource Center (IBRC). The probiotic capacity of these bacteria, including biliary salt tolerance, cholesterol absorption level, the resistance of isolates in acidic conditions, and the ability of the isolates to attach to the Caco-2 cell line, has been confirmed in an earlier study. 28
Preparation of heat-killed bacteria
In order to prepare the heat-killed bacteria, 1010 CFU/mL of bacterial cells were inoculated in a medium (MRS-Broth de Man Rogosa and Sharp) and incubated for 48 h at 37°C in anaerobic conditions. The culture was then centrifuged for 20 min at 4000 r/min and the supernatant was discarded. The precipitate was washed with phosphate buffer (pH 7.2), following which 10 mL of phosphate buffer was added to the precipitate and the resulting suspension was heated at 80°C for 30 min to kill the bacteria. In order to ensure the inactivation of the bacteria, the heated bacterial suspension was recultured, and after ensuring that the bacteria did not grow in the culture medium, the sediment was freeze-dried. The production of heat-killed bacteria in this study was based on a modification of the Choi method. 14
Cell culture
The HT-29 (human colorectal adenocarcinoma, IBRC C10097) and noncancerous human embryonic kidney (HEK)-293 (IBRC C10139) cell lines were provided by the IBRC. The HT-29 and HEK-293 cell lines were cultured in an Roswell Park Memorial institute 1640 medium containing 10% fetal bovine serum and 1% penicillin–streptomycin, at 37°C in a humidified atmosphere with 5% carbon dioxide (CO2). The morphology, health, and the number of cells were studied using an inverted microscope. When cell growth reached a minimum of 70%, the cells were detached from the bottom of the flask by 0.05% trypsin and centrifuged at 1500 r/min for 5 min. The resulting precipitate was prepared into a suspension, and the percentage of living cells in this suspension was determined using a Neubauer chamber and Trypan blue dye in optical microscopy. After ensuring no contamination, cells with a viability exceeding 90% were used for the experiments.
MTT assay
The MTT test was used to evaluate the cytotoxic effects of heat-killed bacteria on HT-29 and HEK-293 cells. Of the culture medium, a 100-μL aliquot containing 10,000 cells (HT-29 and HEK-293 cells, separately cultured) was seeded into 96 well plates and incubated for 24 h.
Following this, the different concentrations of heat-killed bacteria, L. paracasei and L. brevis (0.01, 0.1, 1, 10, 100, and 1000 μg/mL), were added to the wells separately, and the plates were incubated for 24, 48, and 72 h. Then, 100 μL of MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; Sigma, Germany) were added to each well at 0.5 mg/mL concentration, and the plates were incubated for 4 h at 37°C in a humidified atmosphere with 5% CO2. 29 Purple formazan crystals formed in the cell cytoplasm were dissolved by adding a pure dimethyl sulfoxide (DMSO) solution to the wells. Optical absorbance was determined using an enzyme-linked immunosorbent assay reader at 570 nm. The results were reported as the viability percentage and the half maximal inhibitory concentration (IC50) values.
Evaluation of apoptosis by Annexin V-FITC
The evaluation of apoptosis induction in the cells was performed using an Annexin V-FITC kit (eBioscience, Affymetrix, Santa Clara, California, USA), in accordance with the manufacturer’s instructions. The cells were treated with different concentrations of the heat-killed bacteria for 48 and 72 h. The cells were treated with 10, 185 (IC50), and 1000 µg/mL of heat-killed L. paracasei for 48 h; 0.1, 3.8 (IC50), and 10 µg/mL of heat-killed L. paracasei for 72 h; 0.1, 1.5 (IC50), and 10 µg/mL of heat-killed L. brevis for 48 h; and 1, 0.1, and 0.01 µg/mL of heat-killed L. brevis for 72 h. IC50 concentration and a concentration lower and higher than the IC50 value were selected. Cellular analysis was performed using a flow cytometry device (Biocompare, San Francisco, California, USA).
Gene expression using real-time polymerase chain reaction
At first, the HT-29 cells were treated with IC50 concentrations of heat-killed L. paracasei and L. brevis for 48 and 72 h. After that, RNA extraction and cDNA synthesis were performed. In order to extract the RNA (according to CinnaGen protocol), the cells were first lysed by RNX solution (Trizol reagent, CinnaGen Company, Tehran, Iran; 200 μL chloroform was added to 1 mL of RNX solution). The lysed samples were centrifuged at 12,000 r/min. Each sample was separated into three phases: RNA molecules in the upper phase, and protein and DNA in the middle and lower phases, respectively. The topmost transparent phase was transferred to a new vial, and an added equal volume of isopropanol, followed by centrifugation for 15 min at 12,000 r/min. The supernatant was discarded and the obtained pellet was dissolved in diethyl pyrocarbonate (DEPC) water.
After evaluating the quality and quantity of the obtained RNA, cDNA synthesis was performed according to the Revert AidTM First Strand cDNA Synthesis Kit’s (Thermo Scientific Fermentas, USA) protocol. Briefly, 1–2 μg of the RNA, 1 μL of DNAse, and 1 μL of 10× buffer were mixed and incubated at 37°C for 30 min. After that, 1 μL of ethylene diamineteraacetic acid (EDTA) was added and incubated at 65°C for 10 min to inactivate the DNAse enzyme. Then, 4 μL of 5× buffer, 0.5 μL of RNasin (40 Unit/μL), 2 μL of dNTP mixture (10 mM), 2 μL of oligo (dT), 1.5 μL of magnesium chloride, and 1 μL of reverse transcriptase enzymes were added, and the reaction mixture was placed in the thermocycler at 42°C for 60 min. The samples were put in the thermocycler again for 10 min, this time at 70°C to deactivate the reveres transcription enzyme. The reaction’s product was stored at −20°C.
The SYBER Green method was used to determine the expression of Bax and Bcl2 genes in the HT-29 cells treated with the two heat-killed bacteria for 48 and 72 h. The β-actin gene was used as an internal control. The final volume of each reaction mixture was 25 μL, comprised of 12.5 μL of Master mix (Bioneer, Korea), 1 μL of synthesized cDNA (0.1–1μg of β-actin, Bax, Bcl2, caspase-3, and caspase-9 genes), 1 μL (10 mM) of forward and reverse primers for each gene (Takapuzist company, Tehran, Iran), and 9.5 μL of DEPC water. The sequence of primers is presented in Table 1. The polymerase chain reaction (PCR) for each gene was performed separately and repeated three times. The real-time PCR reaction was performed with a Bioneer Exicycler 96 system Republic of Korea. The temperature program was optimized as follows: pre-denaturation at 95°C for 10 min; 40 cycles of denaturation at 95°C for 20 s; annealing at 57°C for 40 s; and extension at 72°C for 40 s.
The sequence of primers used in real-time polymerase chain reaction.

After the reaction, the data were obtained from the device as Ct (threshold cycle), and gene expression was measured by the ΔΔCt method using Rest software, and then plotted using SPSS 19 software (SPSS Inc., Chicago, Illinois, USA) and GraphPad software (Version 6).
DNA fragmentation assay
HT-29 cells were treated with IC50 concentrations of heat-killed probiotic bacteria. After 72 h, the cells were washed with phosphate-buffered saline (PBS) solution and the enzymatic disaggregation was carried out with trypsin. The cell suspension was centrifuged at 4000 × g and resuspended in 600 μL of lysis buffer (mM Tris-hydrogen chloride (HCl) pH 7.5, 400-mM sodium chloride (NaCl), 100-mM EDTA, 0.6% Sodium dodecyl sulfate (SDS)) and 10-μL RNAase (4 mg/mL). The solution was incubated for 5 min at 37°C, and then 200 μL of 6 M NaCl was added for protein precipitation. This prepared solution was incubated for 5 min on ice and then centrifuged at 18000 × g at room temperature for 10 min. The supernatant was collected and added to 600 μL of isopropanol and incubated for 15 min on ice. Again, it was centrifuged at 18000 × g for 20 min and the solution was washed with 600 μL of 70% ethanol, dried at room temperature, and dissolved in 200 μL of buffer (10-mM Tris-HCl, Tris-EDTA, 1 mM EDTA (pH 8.0)).
4,6-Diamidino-2-phenylindole staining
To analyze the morphology of the nucleus of the apoptotic cells, the 4,6-diamidino-2-phenylindole (DAPI) method was performed. The HT-29 cells were seeded onto the six-well plate and incubated for 72 h. Afterward, the cells were fixed with 4% paraformaldehyde for 10 min, and then washed with PBS and permeabilized with 0.1% Triton X-100 for 10 min. They were then washed again with PBS, and then stained with DAPI for 10 min. Finally, the morphology of the cells was analyzed by an invert fluorescence microscope, an Olympus IX81equiped with a DP72 digital camera (Olympus, Hamburg, Germany).
Statistical analysis
Statistical analysis was performed using SPSS 19 software (SPSS Inc., Chicago, Illinois, USA), and the results were analyzed by one-way analysis of variance. The expression levels of the target genes in the treated samples and the control group were compared by Tukey’s honestly significant difference (HSD) post hoc test. The data are presented as mean ± standard deviation, and p < 0.05 was considered statistically significant.
Results
The inhibitory effects of heat-killed L. paracasei and L. brevis on the growth of the HT-29 cell line and normal cells
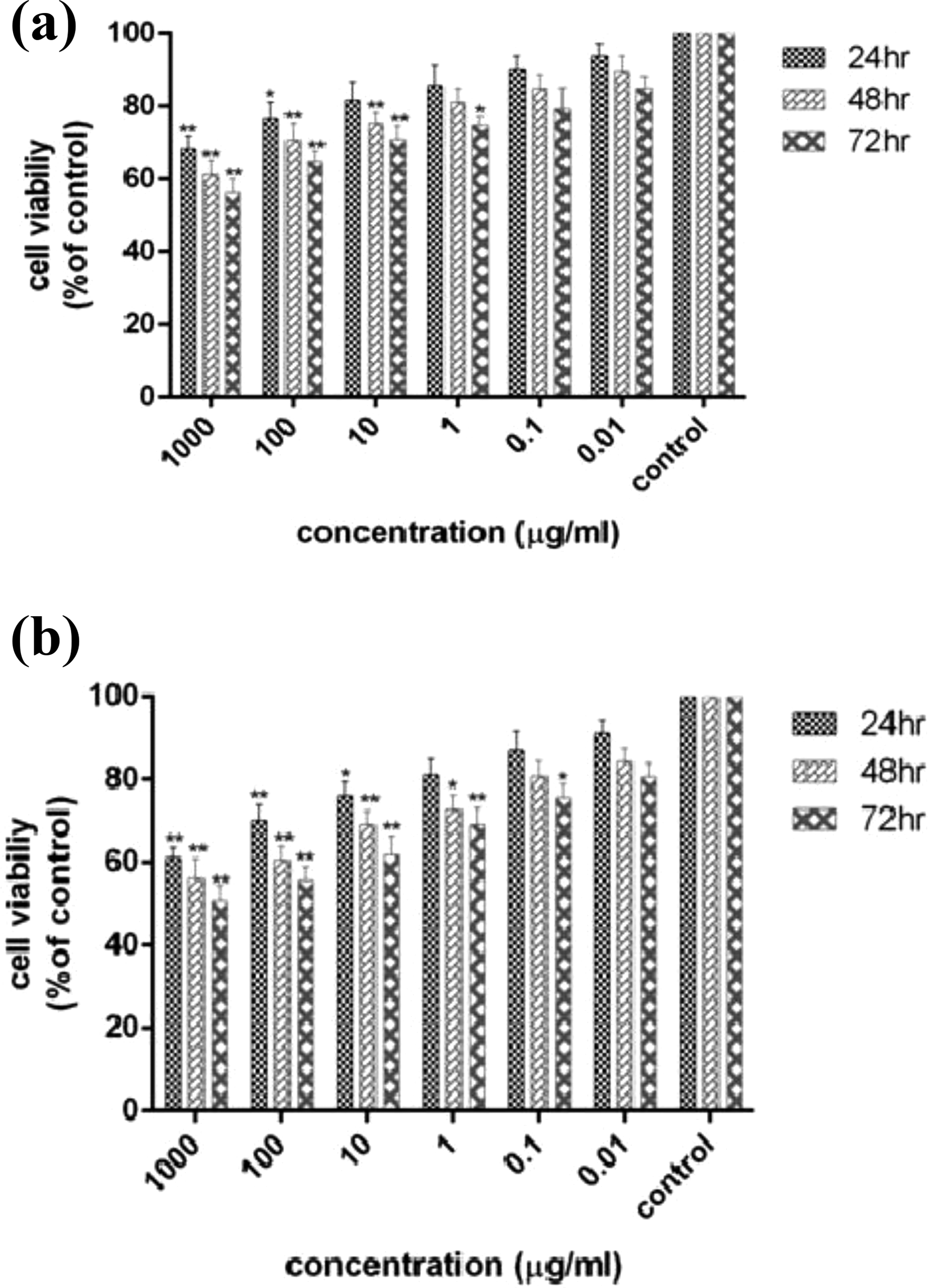
The viability percentage of HT-29 cells treated with various concentrations of heat-killed L. paracasei and L. brevis is summarized in Figure 1. As treatment duration progressed, a decrease in the viability of HT-29 cells was observed along with an increase in the concentrations of both heat-killed bacteria. The results demonstrated that heat-killed probiotic bacteria significantly inhibited the growth of HT-29 tumor cells in a dose- and time-dependent manner (Figure 1).

The viability of HT-29 cell line treated with heat-killed Lactobacillus paracasei (a) and Lactobacillus brevis (b) at 24, 48, and 72 h using MTT assay. The results are reported as survival percentages compared to the controls in three independent experiments (n = 3; *p < 0.05, **p < 0.01, ***p < 0.001).
MTT results indicated that L. brevis had greater antiproliferative and cytotoxic effects on HT-29 cells compared to L. paracasei.
To determine the inhibitory effects of heat-killed probiotic bacteria on the growth of normal cells, a similar experiment was carried out using the HEK-293 cell line. The growth-inhibitory effects of heat-killed L. paracasei and L. brevis on the HEK-293 cell line are presented in Figure 2. The lower cytotoxic effects were observed in normal HEK-293 cells compared to HT-29 cancer cells. The results demonstrated that the cell viability of HEK-293 cells that were treated with heat-killed probiotic bacteria was higher than HT-29 cells.
The viability of HEK-293 cell line treated with heat-killed Lactobacillus paracasei (a) and Lactobacillus brevis (b) at 24, 48, and 72 h using MTT assay. The results are reported as survival percentages compared to the controls in three independent experiments (n = 3; *p < 0.05, **p < 0.01, ***p < 0.001). HEK: human embryonic kidney.
The IC50 values of the heat-killed L. paracasei and L. brevis for HT-29 and HEK-293 cell lines after 24, 48, and 72 h are presented in Table 2.
IC50 values of heat-killed Lactobacillus paracasei and Lactobacillus brevis in HT-29 and HEK-293 cell lines in 24, 48, and 72 h.

HEK: human embryonic kidney.
Apoptosis induction by heat-killed L. paracasei and L. brevis in HT-29 cells
The results of apoptosis induction in the HT-29 cell line upon treatment with heat-killed L. paracasei at 10, 185, and 1000 μg/mL for 48 h are presented in Figure 3. With all the concentrations, an increase in early apoptosis was observed compared to the control group (p < 0.001). The highest rate of early apoptosis was observed at 1000 μg/mL (32.37 ± 0.78%), which was significantly higher than that of the untreated cells (p < 0.001). After 72 h of treatment with 0.1, 3.8, and 10 μg/mL concentrations, the induction of apoptosis was investigated, and the results are presented in Figure 3. As the concentration of bacteria increased, there was an increase in early apoptosis.

Flow cytometry chart for evaluating apoptosis induced by heat-killed Lactobacillus paracasei at 48 and 72 h. (a) control cells (untreated cells); (b) to (d) 10 µg/mL, IC50 (185 µg/mL), and 1000 µg/mL of heat-killed L. paracasei at 48 h, respectively. (e) to (g) 0.1 µg/mL, IC50 (3.8 µg/mL), and 10 µg/mL of heat-killed L. paracasei at 72 h, respectively. Q1 represents early apoptotic cells with Annexin-FITC+ and PI− staining index, Q2 represents late apoptotic cells with Annexin-FITC+ and PI+, Q3 represents healthy cells with Annexin-FITC− and PI− staining index, and Q4 represents necrotic cells with Annexin-FITC− and PI+ staining index.
In order to investigate apoptosis induction by heat-killed L. brevis, 0.1, 1.5, and 10 μg/mL concentrations were used for 48 h, and 0.01, 0.1, and 1 μg/mL concentrations were used for 72 h. The results are presented in Figure 4. After 48 h, an increase in the bacterial concentration from 0.1 μg/mL to 1.5 μg/mL resulted in an increase in apoptosis (p < 0.001). However, increasing the concentration to 10 μg/mL resulted in a decrease in apoptosis by 8%. It appears that 1.5 μg/mL is a more suitable concentration to induce apoptosis in 48 h. The highest level of early apoptosis after 48 and 72 h was observed at 1.5 and 0.1 μg/mL concentrations (IC50 concentrations for 48 and 72 h, respectively). Also, the lowest level of necrotic cells was observed at these concentrations compared to other concentrations (5.66% necrotic cells for 48 h and 2.80% for 72 h, respectively). In other words, the highest levels of apoptosis induction and the lowest levels of necrosis were observed at IC50 concentrations for 48 and 72 h. According to these results, the dependency of apoptosis on dose and time was confirmed.

Flow cytometry chart for evaluating apoptosis induced by heat-killed Lactobacillus brevis in 48 and 72 h. (a) to (c) 0.1 µg/mL, IC50 (1.5 µg/mL), and 10 µg/mL of heat-killed L. brevis in 48 h, respectively. (d) to (f) 0.01µg/mL, IC50 (0.1 µg/mL), and 1 µg/mL of heat-killed L. brevis in 72 h, respectively. Q1 represents early apoptotic cells with Annexin-FITC+ and PI− staining index, Q2 represents late apoptotic cells with Annexin-FITC+ and PI+, Q3 represents healthy cells with Annexin-FITC− and PI− staining index, and Q4 is necrotic cells with Annexin-FITC− and PI+ staining index.
Expression of Bax, Bcl2, caspase-3, and caspase-9
The β-actin gene was chosen as the control as it is expressed in all cells. There was a significant increase in the Bax mRNA level in HT-29 cells treated with heat-killed L. paracasei and L. brevis after 48 and 72 h (p < 0.001) in comparison with the untreated control cells (Figures 5 and 6).

Comparison between the expression of Bax, Bcl2, caspase-3, and caspase-9 mRNA in HT-29 cells treated with heat-killed Lactobacillus paracasei at 48 and 72 h. The results are expressed as mean ± SD of three independent experiments (n = 3). SD: standard deviation.

Comparison between the expression of Bax, Bcl2, caspase-3, and caspase-9 in HT-29 cells treated with heat-killed Lactobacillus brevis in 48 and 72 h. Results are expressed as mean ± SD of three dependent experiments (n = 3). SD: standard deviation.
A downregulation of Bcl2 mRNA expression was observed in the HT-29 cells treated with heat-killed L. brevis and L. paracasei after 48 and 72 h compared to control cells. The expression ratio of Bax/Bcl2, which is the overall estimate of apoptosis induction, exhibited a statistically significant increase after 72 h compared to 48 h, as well as in comparison with the untreated cells (p < 0.05).
A significant increase in caspase-3 and caspase-9 mRNA levels was observed in the HT-29 cells treated with both bacteria for 48 and 72 h compared to the untreated cells (Figures 5 and 6).
Detection of DNA fragmentation and DAPI staining
The ladder pattern of DNA fragmentation in cells treated with heat-killed bacteria after 72 h confirmed the induction of apoptosis in the HT-29 cell line (Figure 7).

DNA fragmentation assay. M: ladder, 1: untreated cells, 2: HT-29 cells treated with heat-killed Lactobacillus brevis, and 3: HT-29 cells treated with heat-killed Lactobacillus paracasei.
In addition, to investigate the morphology of the nucleus of apoptotic cells, DAPI staining was performed. As shown in Figure 8, nucleus fragmentations were observed in the treated cells (Figure 8).

The effect of Lactobacillus brevis on HT-29 cells on the integrity of cell and nuclear morphology and induction of apoptosis following DAPI staining. (a) The untreated HT-29 cells. (b) The HT-29 cells treated with heat-killed L. brevis for 72 h. DAPI: 4,6-diamidino-2-phenylindole.
Discussion
Colorectal cancer is one of the four most common cancers in the world. 30 The main feature of cancer cells is uncontrolled cell proliferation and resistance to apoptosis; therefore, any factor that can induce apoptosis in the cancer cells can be considered as an anticancer agent.
Probiotics are introduced as dietary supplements and include nonpathogenic microorganisms that produce health benefits for the host. 31 They are also considered as chemopreventive agents that can reduce the risk of cancer in different ways, including the attachment and degradation of potential carcinogens, induction of change in intestinal microflora, production of antitumorigenic and antimutagenic compounds, increased immune responses, and cell cycle control. 32 –34
Data extracted from the literature show the effective and protective roles of probiotic bacteria against cancer in various in vivo, in vitro, and epidemiological studies. 15,35,36
It should be noted that the bacteria used in this study (L. brevis and L. paracasei) were isolated from Terxine Doug (a traditional and native food of Iran), and their antiproliferative, apoptosis inducing, and anticancer effects have been investigated for the first time.
The comparison of the IC50 values of the two bacteria revealed that a much higher concentration of L. paracasei, compared to L. brevis, was required to inhibit the proliferation of HT-29 cells. The concentrations of L. paracasei required for antiproliferative activity against HT-29 cells after 24, 48, and 72 h were 123-fold, 42-fold, and 10-fold higher, respectively, compared to the required concentrations of L. brevis. Also, when comparing the IC50 values for cancerous and normal cells, the required concentrations of both the bacteria to induce antiproliferative and cytotoxic effects were much higher for the normal cells than for the cancerous cells, that is, cytotoxicity is induced in cancerous cells at much lower concentrations of bacteria than in normal cells. An increase in the antiproliferative and cytotoxic effects on HT-29 cells treated with heat-killed bacteria had a direct association with dosage (concentration of bacteria) and time. Moreover, higher antiproliferative and cytotoxic effects were observed upon treatment with L. brevis compared to L. paracasei. On the whole, these probiotic bacteria inhibited the proliferation of HT-29 cells in a dose-, time-, and strain-dependent manner.
Also, the induction of apoptosis occurred in a dose-, time-, and bacterial strain-dependent manner. While examining the expression of Bax, caspase-3, caspase-9, and Bcl2 in the cells treated with L. paracasei, the results exhibited an increase in the expression of proapoptotic genes after 48 h as well as 72 h and a more effective apoptosis induction after 72 h compared to 48 h. There was a sevenfold increase in the expression of Bax mRNA after 72 h compared to the control cells and a twofold increase in the Bax mRNA level after 72 h compared to 48 h.
The expression of Bax mRNA was increased 9-fold and 11-fold after 48 and 72 h in HT-29 cells treated with L. brevis, respectively (p < 0.01). Effective apoptosis induction by both bacteria occurs with an increase in the treatment time from 48 h to 72 h. In comparison with the control cells, the increase in the Bax mRNA level after 72 h was 7-fold in the cells treated with L. paracasei (p < 0.001) and 11-fold in the cells treated with L. brevis (p < 0.001).
The treatment of HT-29 cells with both heat-killed bacteria for 48 and 72 h resulted in the upregulation of caspase-3 and caspase-9 mRNA expression compared to the untreated cells. Caspase-3 mRNA levels were increased 3.6-fold, whereas caspase-9 mRNA levels increased 2.9-fold compared to the untreated cells in 72 h in HT-29 cells treated with L. paracasei. A 4.1-fold increase in caspase-3 and 3.3-fold increase in caspase-9 mRNA expression was observed at 72 h compared to the control group in HT-29 cells treated with L. brevis. Heat-killed probiotic bacteria reduced the expression of Bcl2 in HT-29 cells. It appears from these results that both bacteria can increase the expression of apoptosis-related genes in the HT-29 cell line.
The results of the present study suggest that heat-killed L. paracasei and L. brevis exert antiproliferative activity through the activation of cell death via the caspase-dependent intracellular pathway, as evidenced by the increased activity of caspase-3 and caspase-9. The results of gene expression in both 48 and 72 h confirm the results of apoptosis testing by way of flow cytometry, MTT assay, and DNA fragmentation assay. In a comparative study, L. brevis was able to induce more apoptosis and less necrosis than L. paracasei. The antiproliferative and proapoptotic effects against HT-29 cells in the present study are completely dependent on the dose of the heat-killed bacteria, as well as bacterial strain, and treatment time, similar to the patterns observed in the other studies.
Ma et al. showed that the antiproliferative effects of Bacillus polyfermenticus on colon cancer cells are probably due to the presence of proteins of heat-resistant bacteria. 7 Choi et al. demonstrated that both the heat-killed cells of Lactobacillus acidophilus 606 and the soluble polysaccharide components of this strain have antiproliferative potency against all types of cancer cells. 14 L. paracasei IMPC 2.1 and L. GG, not only in living form but also in heat-killed cells, decreased cell proliferation in colon and stomach cancer cells significantly and induced apoptosis. Anti-proliferation and apoptosis induction were similar for both the bacteria and in both cell lines, and no difference was observed between the effects of the living and the inactive forms of bacteria. 15
Madempudi and Kalle showed that heat-treated Bacillus coagulans unique IS2 possess antiproliferative properties and induce apoptosis in colon cancer cells. They can be used as adjuvant therapy in the treatment of colon cancer. The HEK-293 cells were used as normal control cells. Liu and Pan demonstrated that heat-killed cells of Lactobacillus plantarum and L. paracasei have antiproliferative and antioxidant activities in colon and breast cancer cell lines. 18 The antiproliferative activity of heat-killed L. plantarum A7 and Lactobacillus rhamnosus was shown by Sadeghi-Aliabadi and colleagues. 19 Ghoneum and Felo showed that heat-killed Lactobacillus kefiri (PFT) reduced Bcl2 and induced apoptosis in the AGS cell line. 20
Tiptiri-Kouperti and colleagues have proved that the heat-killed cells of Lactobacillus casei prevent the proliferation of HT-29 cells and decrease the size of the tumor in the BALB/c mice. 24
Tuo et al. used the DNA of heat-killed bacteria and cell walls of seven different strains of Lactobacillus on the HT-29 cell line to evaluate their cytotoxic effects. The growth of the cancer cells was inhibited in a strain- and concentration-dependent manner. 37
The antiproliferative effect of L. plantarum kp894100 and L. acidophilus kp942831 on the U-87 and Mcf-7 cells, both in live and heat-killed conditions, was demonstrated by Bharti and colleagues. 38
The cell extract of heat-killed L. casei reduced cell proliferation, increased Bax expression, decreased Bcl2, and induced apoptosis in the HT-29 cell line. 39
Fang et al. showed that heat-killed L. rhamnosus caused the upregulation of the pro-inflammatory cytokine gene expression in Caco-2 cells. 40
In the study on apoptosis and gene expression parameters involved in the pathway of apoptosis, Altonsy et al. showed that L. rhamnosus and Bifidobacterium lactis Bb12 were able to induce apoptosis, increase Bax, release cytochrome C, and activate caspase-9 and caspase-3 in the Caco-2 cell line. 41
Chen et al. demonstrated that the oral administration of L. acidophilus NCFM in mice increased apoptosis in Ct-26 murine colon adenocarcinoma cells in segmental orthotopic colon cancer. It also increased the expression of caspases-3 and 9 and decreased tumor growth. 42 Wang et al. reported that the extract of Lactobacillus cells is capable of inducing apoptosis in HT-29 cells through the mitochondrial membrane pathway. 43 Wan et al. proved that treating SW620 colon cancer cells with Lactobacillus delbrueckii induced apoptosis through the intrinsic pathway and caspase-3 expression. A decrease in Bcl2 gene expression and an increase in apoptosis were observed. 44 Song et al. demonstrated that the proteins isolated from heat-killed L. plantarum L67 were able to induce apoptosis in the HT-29 cell line by stimulating the mitochondrial pathway and caspase-3 activity. 45
In some studies, the roles of probiotics in cancer prevention (especially colon cancer) have been reported. 46,47 The decrease in the size of the tumor and the increase in tumor cell apoptosis have been noted. 42,48
It is noteworthy that the same effects have been reported with the administration of heat-killed probiotics. Recently, Motevaseli et al. reported that the administration of heat-killed Lactobacillus crispatus caused a decrease in the number and the size of tumors in mice bearing breast tumors. The protective effect of L. crispatus on the survival of tumor-bearing mice was observed. 25
Reduced tumor size when using heat-killed L. casei was observed. 24 Jafari et al. showed that heat-killed L. casei reduced breast tumor growth. Also, an increase in interferon (IFN)-γ and a decrease in interleukin (IL)-4, Interleukin (IL)-10, and tranaforming growth factor beta (TGF)-B were observed. 26 The probiotic VSL#3, which is a combination of several heat-killed probiotics, showed significant anti-inflammatory effects through the repression of the STAT3 pathway in the rat colon. 27
Significant inhibitory effects were observed on the carcinogenesis induced by 4-Nitroquioline1-oxide (4NQO) in Lactobacillus salivarius and its secretions. These effects are probably related to 4NQO metabolism, DNA protection against oxide damage, the suppression of cell proliferation, the induction of apoptosis, and downregulation of cox2 expression. 49
Following the administration of L. plantarum AS1, decrements were observed in cell proliferation, tumor size, and the number of tumors. 50 In the case of immune system stimulation by heat-killed probiotics, it has also been shown that their peptidoglycans can cause the induction of different cytokine profiles. 51
Also, heat-killed L. casei and L. plantarum L-137 stimulate the secretion of IL-12 by macrophages and induce Th1 responses. 52 The oral administration of heat-killed Lactobacillus gasseri TMCo356 resulted in elevated levels of ILs 1 and 2 and INFs α and β receptors in mice. 53 Murosaki et al. showed that heat-killed L. plantarum bacteria were able to reduce tumor growth and increase the IL-12 in tumor-bearing mice. 54 The antitumor effects of heat-killed L. casei strain Shirota on the murine orthotopic bladder tumor MBt-2 have been proved. 55
Conclusion
In the present study, the induction of apoptosis was observed along with an increase in Bax, caspase-3, and caspase-9 expression and Bcl2 reduction, which indicates that the probiotics studied in this investigation could be contributing to the release of cytochrome c by activating the mitochondrial pathway and increasing the expression of Bax/Bcl2, thereby promoting apoptosis. The induction of apoptosis in the cells treated with the bacteria increased significantly compared to the untreated cells, and L. brevis exhibited a greater potential for apoptosis induction in comparison with L. paracasei. Considering the percentage of cell survival in the HEK-293 cells treated with a concentration of heat-killed bacteria similar to that used on the HT-29 cells, it appears that the indigenous heat-killed L. paracasei and L. brevis have a lower toxic effect on the HEK-293 cell line compared to the HT-29 cell line. This indicates that the effect of these bacteria differs between normal and cancerous cells. This difference can serve as a positive aspect of the control and treatment of cancer. One of the most important properties of an efficient anticancer drug is its ability to suppress cancerous cells while having minimal toxic effect on normal healthy cells. Given that the antiproliferative and anticancer effects of probiotic bacteria are genus- and strain-dependent, it is proposed that after sufficient complementary testing, these heat-killed indigenous bacteria can be used as supplementary drugs (providing high immunity and having more stability than their living counterparts) for inhibiting the growth of cancer cells. Since the production of heat-killed bacteria is relatively easier and has more stability, it is recommended for use in food formulations.
Footnotes
Acknowledgement
The authors would like to acknowledge the laboratory of Islamic Azad University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
