Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 19 plant-derived proteins and peptides, which function mainly as skin and/or hair conditioning agents in personal care products. The Panel concluded that 18 plant-derived proteins and peptides are safe as used in the present practices of use and concentration as described in this safety assessment, while the data on Hydrolyzed Maple Sycamore Protein are insufficient to determine safety.
Keywords
Introduction
The safety of several hydrolyzed proteins as used in cosmetics has been reviewed by the Panel in several previously published assessments. The Panel concluded that Hydrolyzed Keratin (finalized in 2016), Hydrolyzed Collagen (published in 1985, re-review published in 2006) Hydrolyzed Soy Protein (finalized in 2015), Hydrolyzed Silk (finalized in 2015), Hydrolyzed Rice Protein (published in 2006), and Hydrolyzed Corn Protein (published in 2011) are safe for use in cosmetics.2-8 Additionally, the Panel concluded that Hydrolyzed Wheat Gluten and Hydrolyzed Wheat Protein are safe for use in cosmetics when formulated to restrict peptides to a weight-average molecular weight (MW) of 3500 Da or less. 9
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (http://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; http://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition
Definitions and Functions of the Ingredients in This Safety Assessment 1 (Italicized Text Below Represents Additions Made by CIR).

The preparation of protein hydrolysates can be accomplished via acid (e.g., hydrochloric acid), enzyme (e.g., papain hydrolysis), or other methodologies (e.g., steam). The degree of hydrolysis (i.e., how much the proteins are broken down into smaller polypeptides) may profoundly affect the size and reactivity of such hydrolysates. The degree of hydrolysis can be attenuated by altering the reaction conditions (e.g., changing the temperature or concentration of the hydrolyzing agents). The ingredients in this report, even those ingredients without “hydrolyzed” in the name, may be hydrolyzed to at least some degree in the processes of extraction or solubilization.
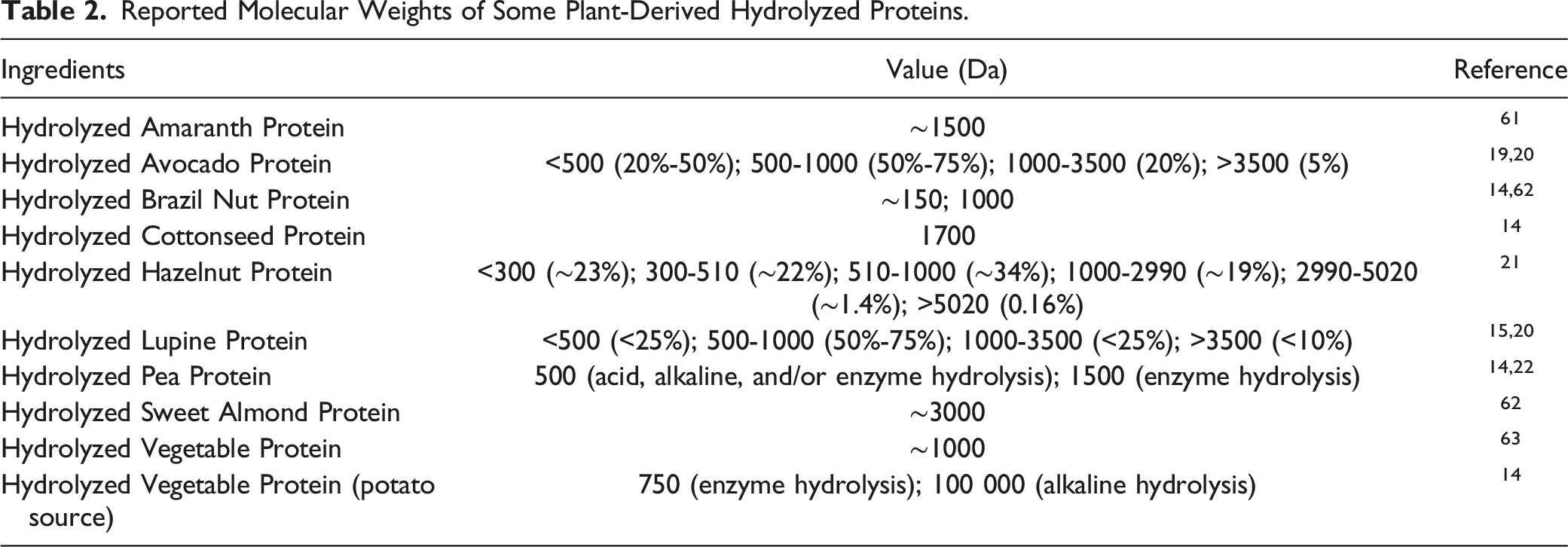
Physical and Chemical Properties
Reported Molecular Weights of Some Plant-Derived Hydrolyzed Proteins.
Method of Manufacturing
Method of Manufacturing.

Composition
Hydrolyzed amaranth protein
Unprocessed and extruded amaranth flours were hydrolyzed sequentially, first with pepsin (for 180 min) and then with pancreatin (for 180 min), and aliquots were collected for analysis at 10, 25, 60, 90, 120, and 180 min after initiating hydrolysis with each of these enzymes. 13 As the duration of the hydrolysis increased, the yield of polypeptides with lower molecular masses also increased (e.g., hydrolysis of unprocessed amaranth yielded molecular masses around 2064 Da at 10 min, 802 Da after 120 min, and 567 Da after 180 min). Extrusion of the amaranth flour yielded more peptides with a lower molecular mass (<1000 Da) immediately after 10 min of hydrolysis. The enzymatic hydrolysis of amaranth flour in this study produced peptides with biological activity, including an angiotensin converting enzyme inhibitor (ACE-inhibitor) and a dipeptidyl peptidase 4 inhibitor (DPP-IV inhibitor).
Hydrolyzed Brazil nut protein
A supplier reported that a formulation containing 10% to 25% Hydrolyzed Brazil Nut Protein (MW = 1000 Da) is also composed of greater than 50% water, 0 to 7% ash (post-pyrolysis; mostly sodium chloride), 0.5% sodium benzoate, and 0.3% potassium sorbate. 14
Hydrolyzed cottonseed protein
A supplier reported that a formulation containing 10% to 25% Hydrolyzed Cottonseed Protein (MW = 1700 Da) is also composed of greater than 50% water, 0 to 6% ash (post pyrolysis; mostly sodium chloride), 0.2% disodium EDTA, 1% phenoxyethanol, and 0.3% potassium sorbate. 14
Hydrolyzed lupine protein
A supplier reported that Hydrolyzed Lupine Protein is comprised of >90% peptides (w/w) and <4% carbohydrates (w/w). 15
Another supplier of three formulations containing Hydrolyzed Lupine Protein reported that two of its formulations (one contained the hydrolyzed protein at up to 26.7% and the other at up to 5.5%) also contained 73% to nearly 81% water, approximately 0.3% to 0.4% phenoxyethanol, and 0.1% parabens. 16 A third formulation (containing up to 24% of the hydrolyzed protein) did not contain parabens and instead had approximately 1.4% to 1.7% phenoxyethanol. The formulation that contained up to 26.7% of the hydrolyzed protein was reported to have phenolic compounds consisting of flavones (∼100%) that represented less than 0.5% of the formulation.
Hydrolyzed pea protein
A supplier reported that a product containing 10% to 25% Hydrolyzed Pea Protein product (MW = 1500 Da) is also composed of greater than 50% water, 0 to 6% ash (post pyrolysis; mostly sodium chloride), 1% phenoxyethanol, and 0.3% potassium sorbate. 14
Hydrolyzed sweet almond protein
A supplier reported that a formulation containing 2.3% to 3.3% Hydrolyzed Sweet Almond Protein also contains 96.15% to 97.25% water, 0.324% to 0.396% phenoxyethanol, and 0.126% to 0.154% parabens. 16
Hydrolyzed vegetable protein
Monosodium glutamate (MSG) occurs naturally in ingredients such as hydrolyzed vegetable protein (generic) and some protein isolates. 17 Hydrolyzed vegetable protein may contain 10 - 30% MSG. 18
A supplier reported that a formulation containing 10% to 25% Hydrolyzed Vegetable Protein (potato source; MW = 750 Da) is also composed of greater than 50% water, 0 to 6.5% ash (post pyrolysis; mostly sodium chloride), 0.2% disodium EDTA; 1% phenoxyethanol, and 0.3% potassium sorbate. 14 The same supplier reported that another formulation containing 7.5% to 15% Hydrolyzed Vegetable Protein (potato source; MW = 100 000 Da) was also composed of greater than 50% water, 0 to 2.5% ash (post pyrolysis; mostly sodium chloride), 0.1% ethylhexylglycerin, 0.9% phenoxyethanol, 0.5% sodium benzoate, and 0.3% potassium sorbate.
Impurities
Hydrolyzed avocado protein
A supplier reported that Hydrolyzed Avocado Protein contains <0.042 μg/kg benzo[a]pyrene; <0.10 ppm arsenic; 0.70 ppm barium; <0.10 ppm cadmium; <0.75 ppm chromium; 0.40 ppm cobalt; <0.05 ppm lead; and <0.10 ppm mercury. 19 Aflatoxins B and G combined were <1.86 μg/kg. Organochlorinated, organophosphorylated, and organosulfur compounds from pesticides were not detected. The 26 allergenic compounds regulated by the European Union were not detected. 20
Hydrolyzed hazelnut protein
A supplier reported that Hydrolyzed Hazelnut Protein has less than 20 ppm heavy metals and less than 2 ppm arsenic. 21
Hydrolyzed lupine protein
A supplier reported that Hydrolyzed Lupine Protein contained <0.042 μg/kg benzo[a]pyrene. 15 Aflatoxins B and G combined were <1.86 μg/kg. Organo-pesticide residues were not detected. When tested on a product with an active concentration of 10%, heavy metals (arsenic, cadmium, mercury, lead, cobalt, nickel, barium, and chromium) were below the quantification limit of 1 ppm. 20 The 26 allergenic compounds regulated by the European Union were not detected.
A supplier of three Hydrolyzed Lupine Protein products reported that one product composed of 4.5% to 5.5% Hydrolyzed Lupine Protein had 25 ppm p-anisyl alcohol and less than 0.5 ppm heavy metals. 16 Pesticides were not detected. Another product composed of 18% to 24% Hydrolyzed Lupine Protein did not contain the 26 allergenic compounds regulated by the European Union, but trace amounts of pesticides (lindane, chlorpyrifos ethyl malathion) were detected. Heavy metals were less than 0.5 ppm. The third product composed of 19.2% to 26.7% Hydrolyzed Lupine Protein had less than 0.6 ppm heavy metals. Alkaloids and pesticides were not detected in this third product.
Hydrolyzed pea protein
A supplier reported that a Hydrolyzed Pea Protein product (25% solution in water; MW = 500 Da) contains no more than 10 ppm heavy metals and no more than 1 ppm arsenic. 22
Hydrolyzed sweet almond protein
A supplier reported that the 26 allergenic compounds regulated by the European Union were not detected in a Hydrolyzed Sweet Almond Protein product (2.3% to 3.3%). 16
Hydrolyzed vegetable protein
Free and esterified forms of 3-monochloro-1,2-propanediol (3-MCPD) and 1,3-dichloro-2-propanol (1,3-DCP) are reported to be found in acid-hydrolyzed vegetable proteins (generic).23,24 3-MCPD is formed from the reaction of triglycerides in the vegetable protein and hydrochloric acid. 25 These are Group 2B compounds (possibly carcinogenic to humans) according to the International Agency for Research on Cancer (IARC). 26 The European Food Safety Authority (EFSA) established a tolerable daily intake (TDI) for 3-MCPD and its fatty acid esters to be 0.8 μg/kg/d with wide margins of exposure for food intake. 27
Use
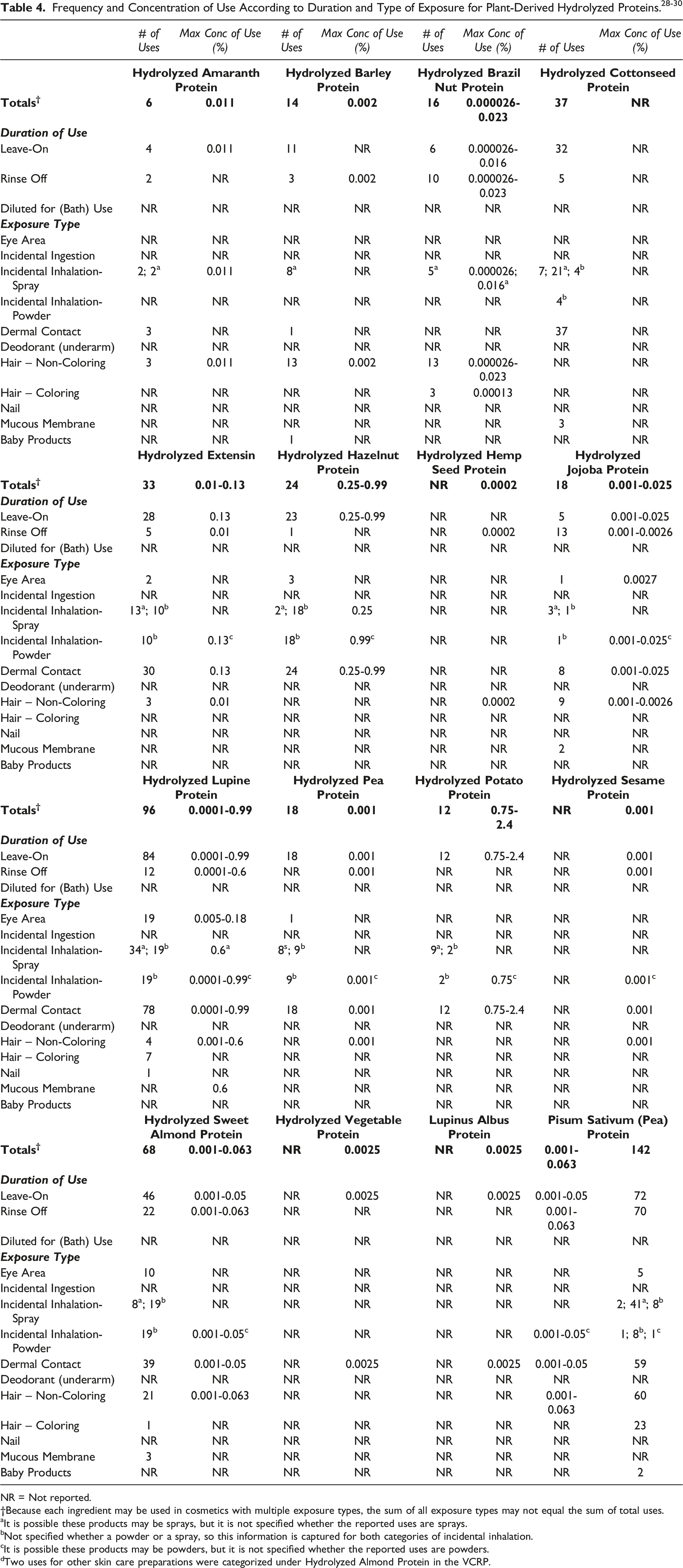
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US FDA and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
NR = Not reported.
†Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
cIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
dTwo uses for other skin care preparations were categorized under Hydrolyzed Almond Protein in the VCRP.
In some cases, reports of uses were received from the VCRP, but no concentration of use data were provided. For example, Hydrolyzed Cottonseed Protein is reported to be used in 37 formulations, but no use concentration data were provided. In other cases, no uses were reported to the VCRP, but a maximum use concentration was provided in the industry survey. For example, Hydrolyzed Hemp Seed Protein was not reported in the VCRP database to be in use, but the industry survey indicated that it is used at concentrations up to 0.0002% in hair conditioners. It should be presumed that Hydrolyzed Hemp Seed Protein is used in at least one cosmetic formulation.
Some of these ingredients may be used in products that can come into contact with mucous membranes and the eyes. For example, Hydrolyzed Lupine Protein is used in bath soaps and detergents at up to 0.6% 30 and Hydrolyzed Vegetable Protein is used in eye lotions at up to 0.3%. 29 Additionally, some of these ingredients were reported to be used in hair sprays, face powders, and fragrances and could possibly be inhaled. For example, Hydrolyzed Hazelnut Protein was reported to be used in perfume at a maximum concentration of 0.25% and Hydrolyzed Vegetable Protein was reported to be used in a face powder (concentration of use not reported). In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm compared with pump sprays.31-34 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.31,33 Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.35-37
The plant-derived protein and peptide ingredients described in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. 38
Non-Cosmetic
Hydrolyzed vegetable proteins (generic) are widely used as seasonings and as ingredients in processed savory food products and range in concentration of use in foods from 0.1% to 40%.18,25 Generally, hydrolyzed proteins (acid hydrolyzed or enzymatically hydrolyzed) do not pose a hazard to humans at levels at which they are used as flavoring agents in foods. 39 Plant protein products are approved food additives according to the FDA (21CFR§170.3).
The FDA requires allergen labeling when major allergens, such as tree nuts, are included in food. 40 A major food allergen is an ingredient from a food or food group, such as tree nuts, that contains protein derived from the food.
Toxicokinetics
No published toxicokinetics studies on plant-derived hydrolyzed proteins and peptides were discovered and no unpublished data were submitted.
Toxicological Studies
Subchronic Toxicity Studies
Lupinus albus protein
The toxicity of Lupinus albus was studied in a 112-d dietary protein study in Charles River rats.
41
Diet consisting of 20% dietary protein from Lupinus albus, L. luteus, or casein (the control) was fed to groups of 12 animals (sex not reported) ad libitum. The lupine diets were supplemented with
Genotoxicity
In Vitro
Carcinogenicity
No published carcinogenicity studies on plant-derived hydrolyzed proteins and peptides were discovered and no unpublished data were submitted.
Other Relevant Studies
Antioxidant Effects
Hydrolyzed hemp seed protein
No adverse effects were mentioned in a study of rats fed hydrolyzed hemp seed meal protein. 44 This study investigated the antioxidant effects of hydrolyzed hemp seed meal protein in spontaneously hypertensive rats. Groups of 8 male rats were fed diets containing 0%, 0.5%, or 1.0% (w/w) hydrolyzed hemp seed meal protein for 8 wk. Half of the rats were killed for blood collection while the remaining half underwent a 4-wk washout, during which they were all fed the diet without hydrolyzed hemp seed meal protein added, and then fed the experimental diets an additional 4 wk before terminal blood collection. Plasma total antioxidant capacity (TAC), superoxide dismutase (SOD), and catalase (CAT) levels were decreased in the rats in the recovery group, when compared to those killed prior. Significant (p < 0.05) increases in plasma SOD and CAT levels accompanied by decreases in total peroxide levels were observed in both the pre- and post-wash-out rats. The hemp seed meal protein in this study was hydrolyzed by pepsin and pancreatin, consecutively.
Type 1 Hypersensitivity
Hydrolyzed Brazil nut protein, hydrolyzed hazelnut protein, hydrolyzed sweet almond protein
As is commonly known, tree nuts, including Bertholletia excelsa (Brazil nut), Corylus spp. (hazelnut), and Prunus dulcis (sweet almond) are major food allergens that produce Type 1 (immediate) reactions in sensitized individuals. A review article reports that the prevalence of “probable” tree nut allergy in the population ranges from 0.05% to 4.9%, with the prevalence of allergies to specific tree nuts varying among the main regions where cases were reported (i.e., Europe, the United States, and the United Kingdom). 45 Walnut and cashew allergies are the most common tree nut allergies in the United States, while hazelnut and almond and walnut are the most common tree nut allergies in Europe and the United Kingdom, respectively.
Lupinus albus protein and pisum sativum (pea) protein
A clinical study examined 3 patients with a history of anaphylactic reactions to peas who subsequently developed signs of sensitization after ingesting peanuts. 46 All 3 patients had positive skin prick tests, as well as elevated serum levels of IgE antibodies against protein extracts of both peas and peanuts. IgE-binding experiments revealed strong binding mainly to vicilin in pea extracts and exclusively to Ara h 1 in a crude peanut extract. IgE binding to the purified Ara h 1 of peanuts was definitively inhibited by purified pea vicilin, but the IgE binding to the pea vicilin was not inhibited to any significant extent by peanut Ara h 1. The authors concluded that clinically-relevant cross-reactivity can occur between vicilin homologs in peanuts (i.e., Ara h1) and in peas. They noted that the allergic reactions to peanuts were attributable to cross-reactive IgE antibodies raised previously against pea allergens in these patients, based on the course of the development of allergic reactions, skin prick test results, specific IgE levels, and the potent inhibition by pea vicilin of IgE binding to Ara h1, compared with the lack of inhibition by Ara h 1 of IgE binding to pea vicilin.
Immunological cross-reactivity was studied among the seeds of widely different species (lupine and pea, as well as peanut, lentil, kidney bean and soybean) using the sera of 12 peanut-sensitive children. 47 IgE binding to the seed proteins of these plant species varied widely among the subjects. IgE binding to peanut polypeptides was prevalent among the subjects, as expected, but binding to the polypeptides of other legumes was also observed. Often the binding was to the basic subunits of 11S globulins. In this study, the subjects exhibited skin prick test results that generally paralleled the results of the IgE binding studies. The most sensitive subjects had strong reactions to peanut, pea, and lentil protein extracts.
Skin prick tests were performed using a panel of protein extracts from the seeds of several legume species on patients (n = 36) with allergies to peanuts and/or other legumes. 48 The plant species tested included lupine and green pea, as well as the dun pea, chickpea, lentil, soybean, white bean and broad bean. The patients were divided into 2 groups. Group 1 included 6 subjects who were not allergic to peanuts but were allergic to lentils (4), dun pea (3), green pea (3), soybean (2), broad bean (2), lupine (1), and/or chickpeas (1). Each of these patients had positive skin prick tests to at least 4 of the legume extracts tested. Group 2 included 30 patients with peanut allergy and was subdivided into 3 subgroups. Group 2a included 13 patients who were not allergic to other legumes, all of whom exhibited negative skin prick tests to the protein extracts of legumes other than peanuts or ate all legumes other than peanuts without reactions. Group 2b included 8 patients who were sensitized to legumes, in addition to peanuts, without having previously experienced clinical reactions to legumes other than peanuts. These patients exhibited positive skin prick tests to the proteins of at least 1 and up to 5 legumes, in addition to peanut proteins. Group 2c included 9 patients with allergies to peanuts and to other legumes, including green peas (4), dun peas (3), lentils (3), soybeans (2), and lupine (1), and positive skin prick tests to the proteins of at least 1 and up to 5 legumes. In this study, 96% (22/23) of the patients who were sensitized or allergic to legumes other than peanuts (whether or not they were also allergic to peanuts) and 100% (17/17) of the patients allergic to peanuts and other legumes had specific IgE against Ara h 1. Only 54% (7/13) of the patients with peanut allergy who were not also sensitized to other legumes had specific IgE against Ara h 1. Further, peanut protein extracts inhibited the binding of dun pea specific IgE to dun pea proteins. The authors concluded, based on the overall results of their study that peanut-allergic patients sensitized to Ara h 1 are at greater risk of becoming sensitized or developing allergies to other legumes, compared with those not sensitized to Ara h 1.
Dermal irritation and sensitization studies
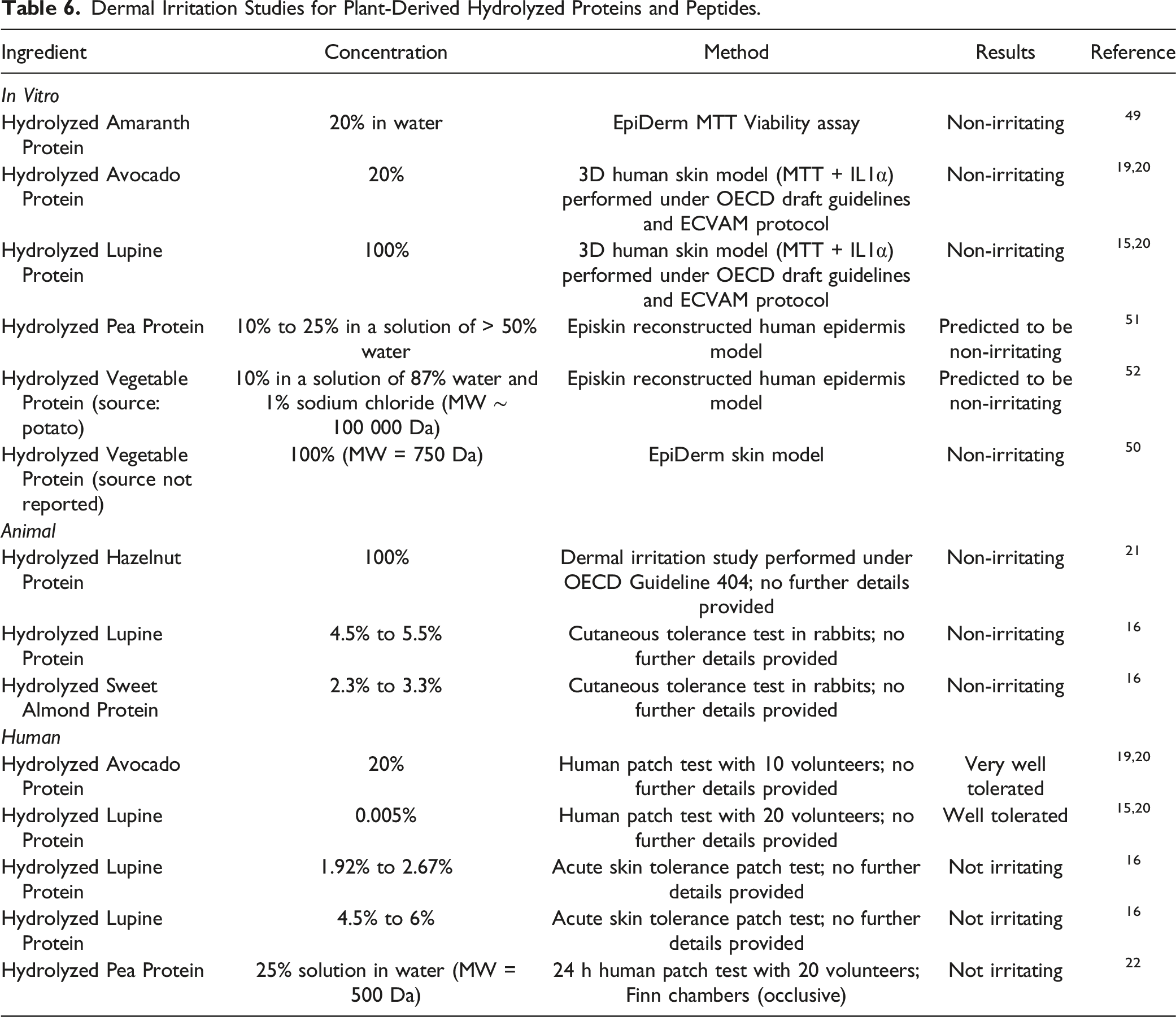
Irritation
Dermal Irritation Studies for Plant-Derived Hydrolyzed Proteins and Peptides.
Sensitization
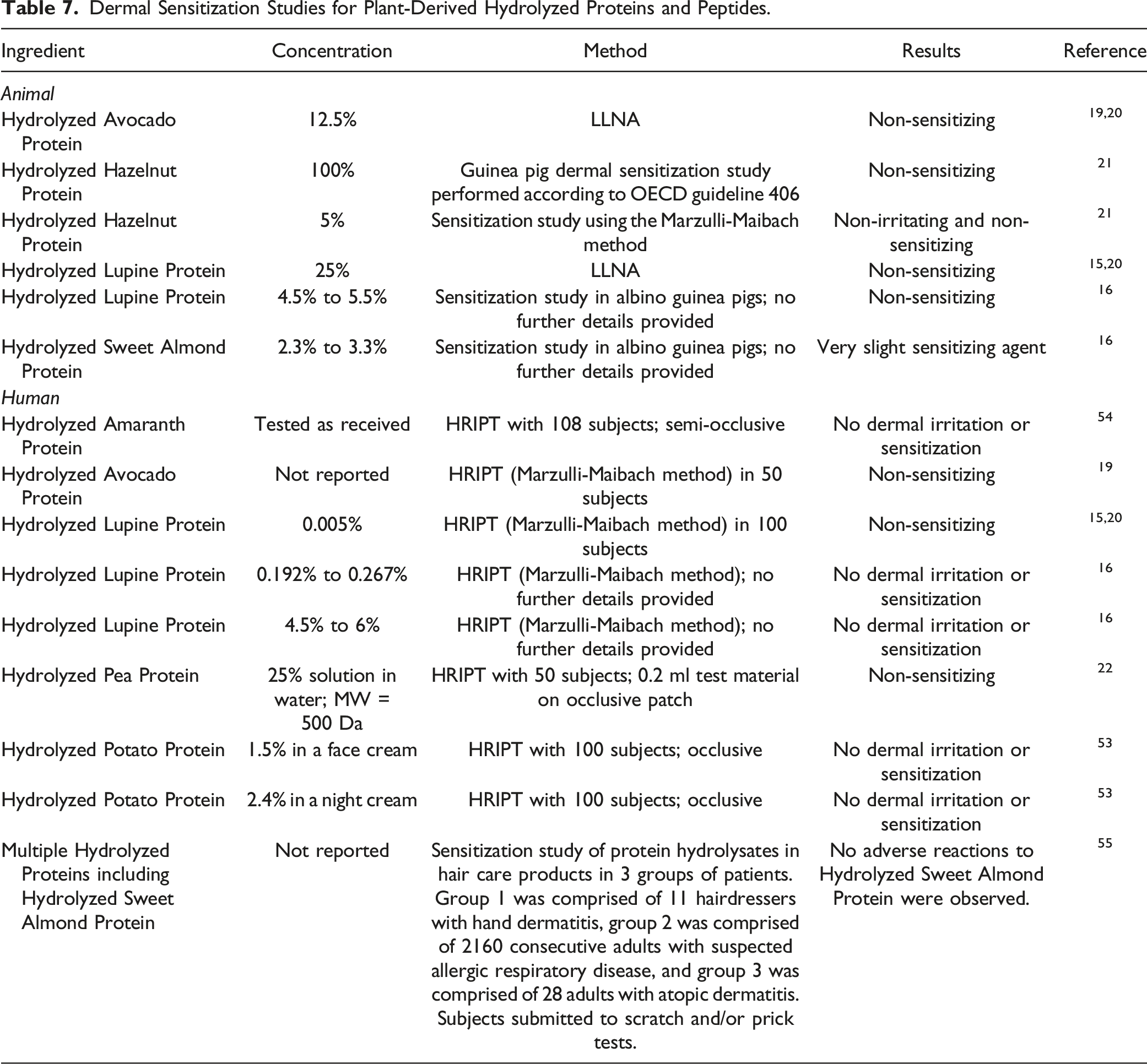
Dermal Sensitization Studies for Plant-Derived Hydrolyzed Proteins and Peptides.
Phototoxicity
In Vitro Phototoxicity Studies for Plant-Derived Hydrolyzed Proteins and Peptides.

Ocular Irritation Studies
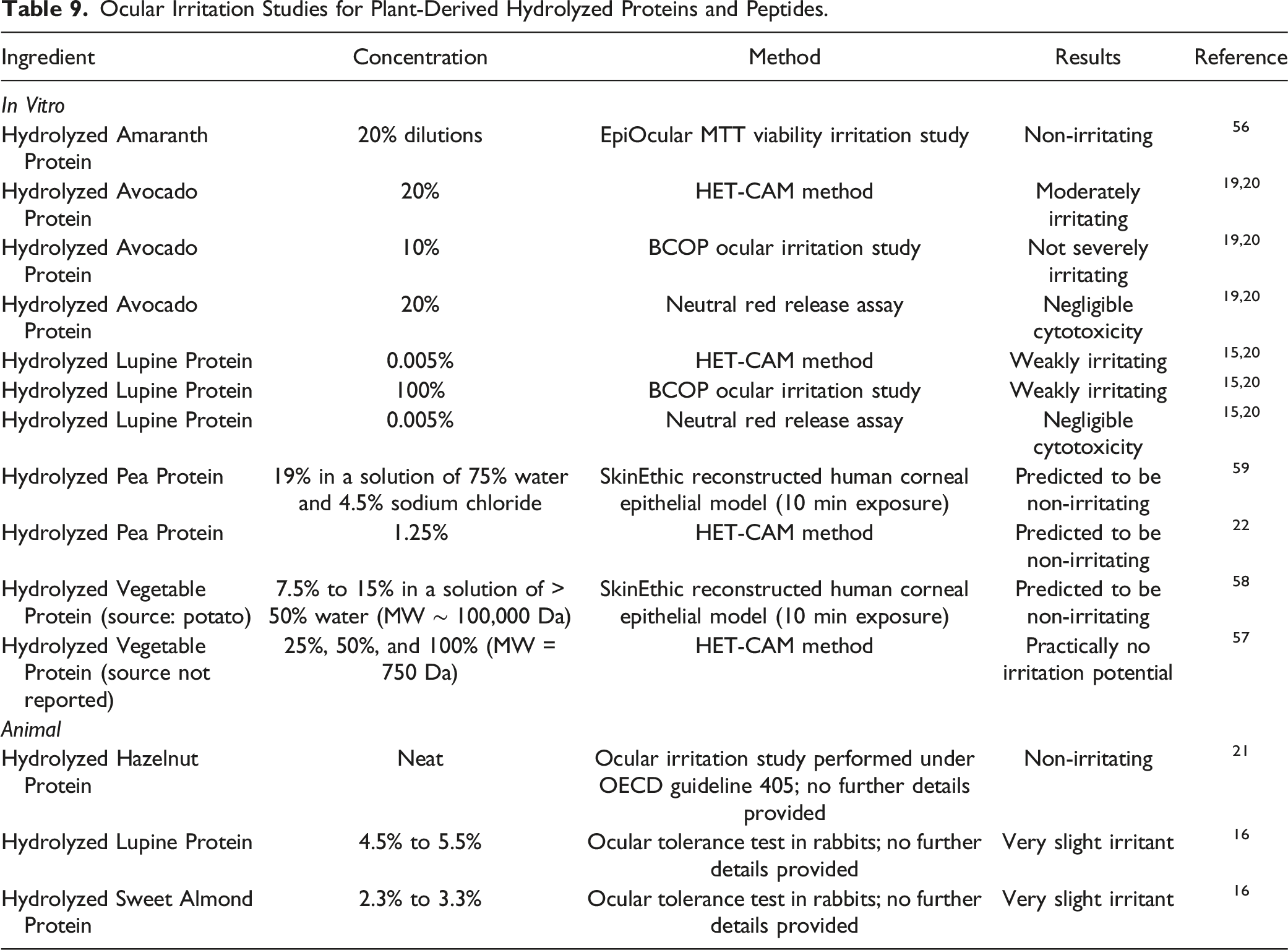
Ocular Irritation Studies for Plant-Derived Hydrolyzed Proteins and Peptides.
Clinical Studies
Case Studies
Hydrolyzed vegetable protein
MSG symptom complex has been reported in sensitive people who have consumed foods containing hydrolyzed vegetable protein (generic). 18 No adverse effects from cosmetic use were discovered in the published literature.
Summary
Plant-derived proteins and peptides function primarily as skin and/or hair conditioning agents in personal care products. These protein derivatives are prepared by subjecting vegetable proteins to hydrolysis via enzymes, acid, or other methodologies such as steam.
Hydrolyzed Vegetable Protein has the most reported uses in personal care products, with a total of 142 formulations; approximately half of the uses are in leave-on products. Hydrolyzed Lupine Protein has the second greatest number of overall uses reported, with a total of 96; the majority of the uses are in leave-on formulations.
Hydrolyzed Potato Protein is used at up to 2.4% in nighttime skin care products. Hydrolyzed Hazelnut Protein and Hydrolyzed Lupine Protein have maximum use concentrations of 0.99% in body and hand skin care preparations.
No uses were reported for Hydrolyzed Avocado Protein, Hydrolyzed Maple Sycamore Protein, or Hydrolyzed Zein in the VCRP or by Council.
Hydrolyzed vegetable proteins (generic) are widely used as seasonings and as ingredients in processed savory food products and range in concentration of use in foods from 0.1% to 40%. Generally, hydrolyzed proteins (acid hydrolyzed or enzymatically hydrolyzed) at levels used as flavoring agents in foods do not pose a hazard to humans. Plant protein products are approved food additives according to the FDA. The FDA requires allergen labeling when major allergens, such as tree nuts, are included in food.
Relevant to the ingredient, Lupinus Albus Protein, the toxicity of Lupinus albus was studied in a 112-d study in rats where diets comprised 20% dietary protein from L. albus, L. luteus, or casein (the control). The rats fed the L. albus diets gained weight at a slightly lower rate than those fed L. luteus and casein. There were no differences in the feed intakes and feed efficiencies of both lupine groups during week 1-6. There were no differences observed in organ-to-body weight ratios of liver, spleen, heart, and adrenals of rats fed either lupines or casein. No adverse effects were reported. No significant differences were observed in the gross necropsy findings or the microscopic examinations.
Relevant to the ingredient, Hydrolyzed Hemp Seed Protein, no adverse effects were reported in rats fed hydrolyzed hemp seed meal protein. The rats were fed diets containing 0%, 0.5%, or 1.0% (w/w) hydrolyzed hemp seed meal protein for 8 wk.
Hydrolyzed Lupine Protein (up to 26.7%), Hydrolyzed Pea Protein (up to 25%), Hydrolyzed Sweet Almond Protein (up to 3.3%) and Hydrolyzed Vegetable Protein (10.9%) were not mutagenic in Ames tests.
Relevant to the ingredients, Hydrolyzed Brazil Nut Protein, Hydrolyzed Hazelnut Protein, and Hydrolyzed Sweet Almond Protein, tree nuts, including Bertholletia excelsa (Brazil nut), Corylus spp. (hazelnut), and Prunus dulcis (sweet almond) are well known major food allergens that produce Type 1 (immediate) reactions in sensitive individuals. Type 1 allergic responses also have been reported following the consumption of legumes such as peanut, lupine, and pea, which is relevant to the ingredients Hydrolyzed Lupine Protein, Hydrolyzed Pea Protein, Lupinus Albus Protein, and Pisum Sativum (Pea) Protein.
The results of in vitro and animal dermal irritation studied indicated that Hydrolyzed Amaranth Protein (20% in water), Hydrolyzed Avocado Protein (concentration not reported), Hydrolyzed Hazelnut Protein (100%), Hydrolyzed Lupine Protein (concentration not reported), and Hydrolyzed Vegetable Protein (100%) were not irritants. No irritation was observed in human dermal studies for Hydrolyzed Avocado Protein (20%), Hydrolyzed Lupine Protein (up to 6%), and Hydrolyzed Pea Protein (25% solution in water).
No sensitization was observed in animal studies of Hydrolyzed Avocado Protein (concentration not reported), Hydrolyzed Hazelnut Protein (up to 100%), and Hydrolyzed Lupine Protein (concentration not reported). No sensitization was observed in human studies of the following hydrolyzed protein ingredients: amaranth (concentration not reported), avocado (concentration not reported), lupine (concentration not reported), potato (up to 2.4% in formulation), and sweet almond (concentration not reported).
Hydrolyzed Avocado Protein (50%), Hydrolyzed Hazelnut Protein (concentration not reported), and Hydrolyzed Lupine Protein (100%) were not phototoxic in in vitro studies.
The results of in vitro ocular studies indicated that Hydrolyzed Amaranth Protein (20%) and Hydrolyzed Vegetable Protein (up to 100%) were not irritating. In vitro studies predicted Hydrolyzed Avocado Protein (tested at 20% in a HET-CAM assay; 10% in a BCOP test) may be an eye irritant. Hydrolyzed Lupine Protein (concentration not reported) was weakly irritating in both the HET-CAM and BCOP tests. In animal studies, Hydrolyzed Hazelnut Protein was not irritating when tested neat, while Hydrolyzed Lupine Protein (up to 5.5%) and Hydrolyzed Sweet Almond Protein (up to 3.3%) were very slight irritants in rabbit eyes.
Discussion
The Panel noted that these plant-derived protein and peptide ingredients are processed extensively during production, which substantially reduces or eliminates any constituents of concern that may be present in the plant material from which they are derived. The Panel expressed concern about aflatoxins, pesticide residues, heavy metals, and other chemical species that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Most of the protein-derived ingredients in this assessment are found in foods, and daily exposures from the consumption of foods can be expected to yield much larger systemic exposures to these ingredients than those from use in cosmetic products. Plant proteins are approved food additives. The Panel did acknowledge that Type I immediate hypersensitivity reactions could possibly occur following exposure to a protein-derived ingredient by sensitized individuals, especially via incidental inhalation. Human Repeat Insult Patch Tests (HRIPTs) and related test data do not detect Type I reactions. Thus, the Panel recommends that people with known allergies to tree nut, seed, and avocado proteins avoid using personal care products that contain these ingredients.
The Panel discussed the issue of incidental inhalation exposure from hair sprays, fragrance preparations, and face powders. There were no inhalation toxicity data available. The Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount; however, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects in users without known allergies to tree nut, seed, and avocado proteins. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel determined that the data were sufficient to support safety of 18 plant-derived protein and peptide ingredients in the present practices of use and concentration. The Panel found the data were insufficient to determine safety of Hydrolyzed Maple Sycamore Protein and issued an insufficient data announcement in December 2016. The data needs were not met. The additional data needed to evaluate the safety of Hydrolyzed Maple Sycamore Protein are: • Method of manufacturing • Chemical composition and impurities • Clarification on food safety status, specifically whether this ingredient is generally recognized as safe (GRAS) • If this ingredient is not GRAS, then studies of systemic endpoints such as a 28-d dermal toxicity, reproductive and developmental toxicity, and genotoxicity are needed, as well as UV absorption spectra
Conclusion
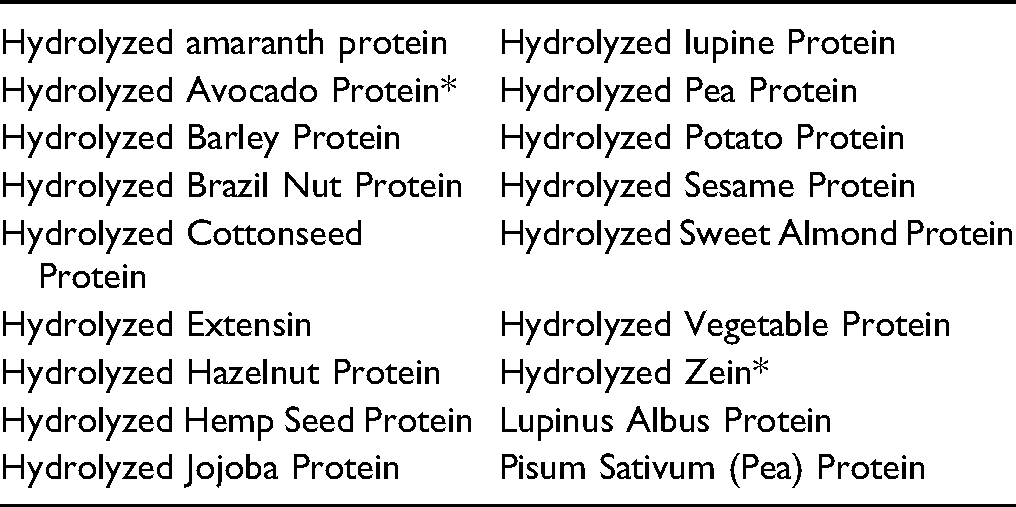
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
However, the Panel concluded that the data on Hydrolyzed Maple Sycamore Protein are insufficient to determine safety. This ingredient is not reported to be in use.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Burnett, C.L. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Boyer, I.J. contributed to analysis and interpretation; Bergfeld, W.F., Belsito, D.V., Hill, R.A., Klaassen, C.D., Liebler, D.C., Marks, J.G., Shank, R.C., Slaga, T.J., and Snyder, P.W. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.