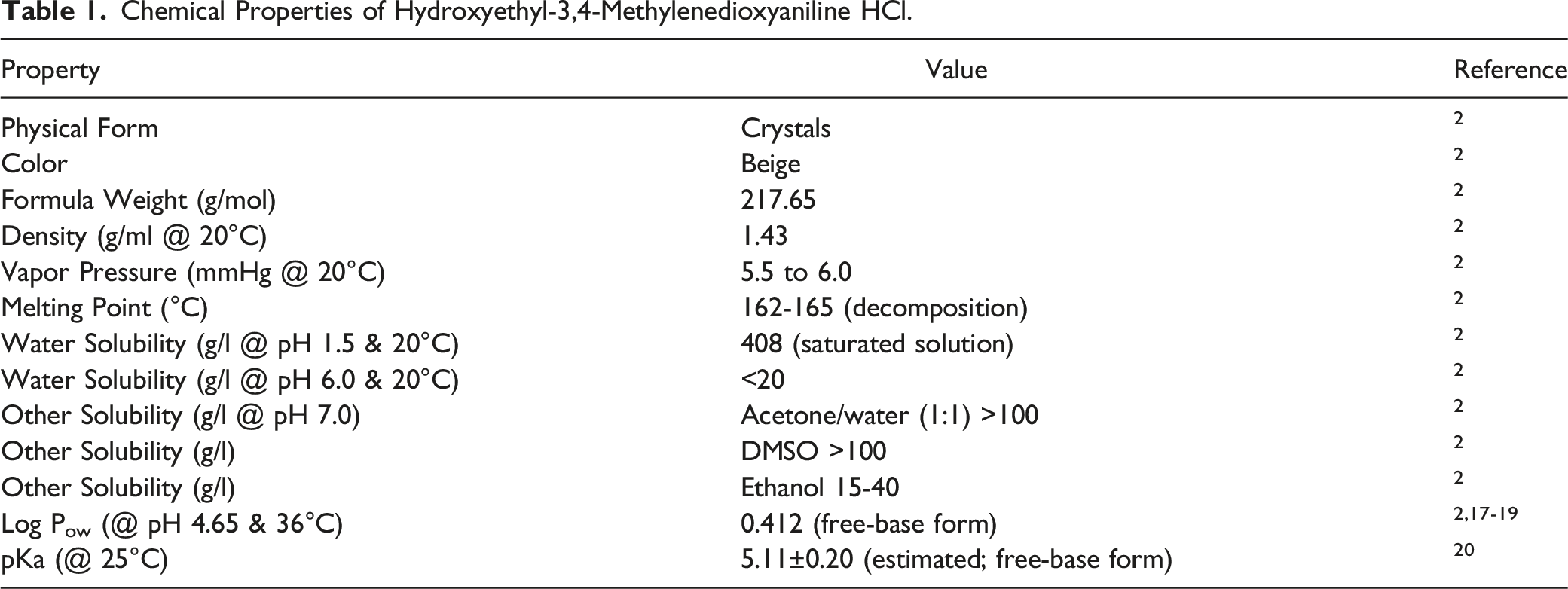
Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of Hydroxyethyl-3,4-Methylenedioxyaniline HCl, which is reported to function as a hair dye ingredient. The Panel reviewed relevant data provided in this safety assessment, and concluded that Hydroxyethyl-3,4-Methylenedioxyaniline HCl is safe for use as a hair dye ingredient in the present practices of use and concentrations described in this report. The Panel cautions that this ingredient should not be used in cosmetic products in which N-nitroso compounds can be formed.
Introduction
This assessment addresses the safety of Hydroxyethyl-3,4-Methylenedioxyaniline HCl as used in cosmetics. According to the International Cosmetic Ingredient Dictionary and Handbook (Dictionary), this cosmetic ingredient functions as a hair colorant. 1
The majority of the data included in this safety assessment were gathered from a report titled “European Commission Scientific Committee on Consumer Safety (SCCS) Opinion on Hydroxyethyl-3,4-Methylenedioxyaniline HCl”, 2 the European Union’s website (EUROPA),2-4 and the Australian Government Department of Health National Industrial Chemicals Notification and Assessment Scheme (NICNAS) website. 5 The above references provide summaries of data generated by industry, and it is those summary data that are reported in this safety assessment when the references above are cited.
Chemistry
Definition and Structure
Hydroxyethyl-3,4-Methylenedioxyaniline HCl (CAS No. 94158-14-2) is the bicyclic, ammonium salt that conforms to the structure in Figure 1.
1
Hydroxyethyl-3,4-Methylenedioxyaniline HCl.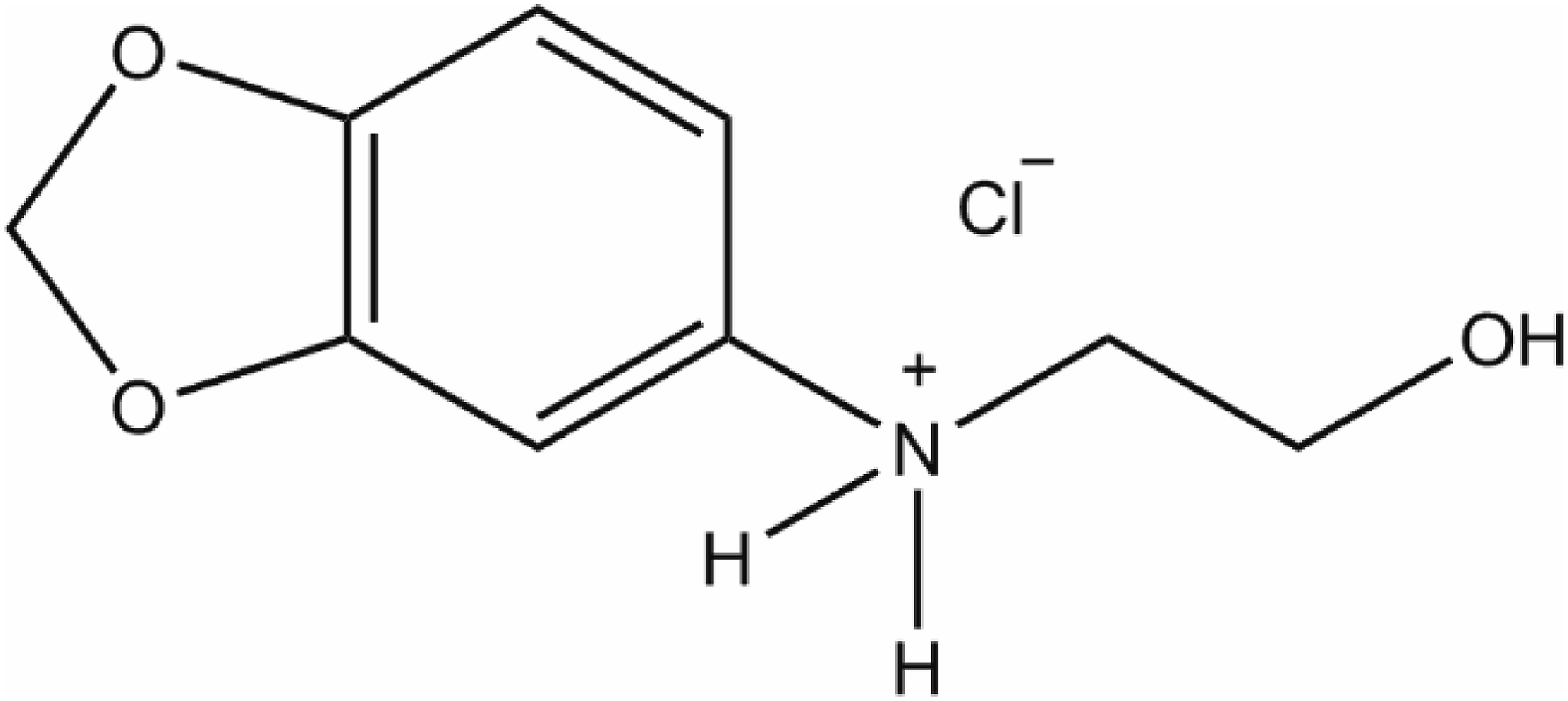
Hydroxyethyl-3,4-Methylenedioxyaniline HCl is used as an oxidative hair coloring “precursor” ingredient.2,6 In a 2009 SCCS report, an industry submission indicated that this oxidative coloring precursor and a developer (hydrogen peroxide) were mixed at ratios ranging from 1:1 to 1:3. 2 However, Hydroxyethyl-3,4-Methylenedioxyaniline HCl was also listed as a “coupler” in a different SCCS report from 2010. 3 No reaction scheme was found in the literature. In a typical formulation, a precursor is activated via an oxidant, such as hydrogen peroxide. The resultant activated precursor proceeds to react with a coupler to form in-situ a product that is purported to be the actual dye that colors hair, in this type of oxidative hair dye product.
Physical and Chemical Properties
Method of Manufacture
There were no method of manufacture data identified in the literature for Hydroxyethyl-3,4-Methylenedioxyaniline HCl.
Impurities and Nitrosation
Impurities that may be present in Hydroxyethyl-3,4-Methylenedioxyaniline HCl include 3,4-methylenedioxy-aniline (114 to 1097 ppm in 5 batches tested), 1,3-benzodioxol (below limit of detection [35 ppm] in 5 batches tested), and 1,2-methylenedioxy-4-nitrobenzene (below limit of detection [8 ppm] in 5 batches tested). 2 The following residual solvents were not identified at a 100 ppm detection limit: methanol, ethanol, isopropanol, acetone, ethyl acetate, cyclohexane, methyl ethyl ketone, and monochlorobenzene.
Hydroxyethyl-3,4-Methylenedioxyaniline HCl contains a free, secondary aromatic substituted amine group (aniline derivative), thus raising a concern about potential N-nitrosation. A concern in cosmetics is the conversion (nitrosation) of nitrogen-containing ingredients, such as hair dyes, into N-nitroso chemicals that may be carcinogenic. In one study, 85% of the approximately 209 nitrosamines tested were shown to produce cancer in laboratory animals. 7 Nitrosation can occur under physiologic conditions. 8 Depending on the nitrosating agent and the substrate, nitrosation can occur under acidic, neutral, or alkaline conditions. Atmospheric NO2 may also participate in nitrosation in aqueous solution. 9 Consequently, hair dye formulations containing Hydroxyethyl-3,4-Methylenedioxyaniline HCl, and formulations intended for admixture with this ingredient (e.g., to generate an active hair dye), should be free of nitrosating agents.
Furthermore, nitrosamines may be present in a cosmetic as an impurity of an ingredient. The Panel advised that manufacturers may avoid these issues by formulating ingredients in a way that reduces the formation of nitrosamines, and by eliminating the presence of impurities that are N-nitrosated or contain nitrosating agents.
In an SCCS report, concern was raised about nitrosation of the impurity 3,4-methylenedioxy-aniline (a primary aromatic amine) that may occur in Hydroxyethyl-3,4-Methylenedioxyaniline HCl. 2 The total content of N-nitroso compounds for Hydroxyethyl-3,4-Methylenedioxyaniline HCl was determined to be <10 μg/kg (ppb) in 3 batches tested; the SCCS concluded that nitrosamine content should be limited to <50 ppb. The SCCS also determined that Hydroxyethyl-3,4-Methylenedioxyaniline HCl should “not be used in combination with nitrosating substances.” 2
Use
Cosmetic
The Panel evaluates the safety of the cosmetic ingredient included in this assessment based on the expected use of, and potential exposure to, the ingredient in cosmetics. The data received from the United States (US) Food and Drug Administration (FDA) are collected from manufacturers through the FDA Voluntary Cosmetic Registration Program (VCRP), and include the use of individual ingredients in cosmetics by cosmetic product category. The data received from the cosmetic industry are collected by the Personal Care Products Council (Council) in response to a survey of the maximum reported use concentrations by product category. VCRP data obtained from the FDA in 2017 indicate that Hydroxyethyl-3,4-Methylenedioxyaniline HCl has 67 reported uses in hair dyes and colors (all requiring caution statements and patch tests). 10 The 2016 Council industry survey indicated that Hydroxyethyl-3,4-Methylenedioxyaniline HCl was reported to be used at maximum concentrations of 0.52% to 0.75% (diluted or “on head” concentrations) in hair dyes and colors. 11
Hydroxyethyl-3,4-Methylenedioxyaniline HCl is considered to be a coal tar hair dye for which regulations require caution statements and instructions for patch testing to exempt the dye from adulteration and color additive provisions of the US Federal Food, Drug, and Cosmetics Act. To be exempt, the following caution statement must be displayed on all coal tar hair dye products:
Caution-this product contains ingredients which may cause skin irritation on certain individuals and a preliminary test according to accompanying directions should be made. This product must not be used for dyeing the eyelashes or eyebrows; to do so may cause blindness.
Product labels shall also bear patch test instructions for determining whether the product causes skin irritation. The Panel recommends, for products containing this type of ingredient, that an open patch test be applied and evaluated by the beautician and/or consumer for sensitization 48 h after application of the test material and prior to the use of a hair dye formulation.
In 2012, a report was published about such self-testing for contact sensitization to hair dyes. 12 The authors concluded that, in its present form, the hair dye self-test has severe limitations. The authors issued the warning that, if the use of a hair dye self-test to predict contact sensitization becomes widespread, there is severe risk that a tool has been marketed that may cause morbidity in European consumers. An accompanying editorial performed on behalf of the European Society of Contact Dermatitis (ESCD) asserted that industry is focusing on predicting the risks from exposure to hair dyes by having millions of European consumers perform a self-test prior to each hair dying and stated that it is the opinion of the ESCD that attention must be given to reducing the risks of serious allergic reactions by improving the safety of the products themselves. 13
The opinion issued by the European Commission SCCS on Hydroxyethyl-3,4-Methylenedioxyaniline HCl in 2009 expressed that Hydroxyethyl-3,4-Methylenedioxyaniline HCl is not considered a health risk to the consumer when the hair dye is used at a maximum concentration of 1.5% on the head after mixing under oxidative conditions.2,6 The SCCS also concluded that Hydroxyethyl-3,4-Methylenedioxyaniline HCl has the potential to be a strong sensitizer. Additionally, the SCCS determined that genotoxicity/mutagenicity studies in finished hair dyes should be conducted in accordance with Scientific Committee on Consumer Products and Non-Food Products Intended for Consumers (SCCNFP/SCCP) opinions and Notes of Guidance.
According to the European Commission, Hydroxyethyl-3,4-Methylenedioxyaniline HCl is allowed for restricted cosmetic use as regulated in the Annex III List of Substances which Cosmetic Products Must Not Contain Except Subject to the Restrictions Laid Down (European Union Reference #246), as stated above.6 The European Commission stipulates that oxidative hair dye ingredients, including Hydroxyethyl-3,4-Methylenedioxyaniline HCl, must be labeled as such:
“The mixing ratio must be printed on the label. Hair colorants can cause severe allergic reactions. Read and follow instructions. This product is not intended for use on persons under the age of 16. Temporary ‘black henna’ tattoos may increase your risk of allergy. Do not color your hair if you have a rash on your face or sensitive, irritated and damaged scalp, you have ever experienced any reaction after coloring your hair, you have ever experienced any reaction to a temporary ‘black henna’ tattoo in the past.“6
A Swedish market analysis was conducted on 122 oxidative hair dye products sold at typical retailers (ie, grocery stores, beauty shops, hairdressing salons). 14 The hair dye products evaluated were marketed in Europe and internationally. Shades from light to dark were represented in this evaluation. Information about the ingredients comprising the hair dye formulations was gathered from the product label and the European Commission cosmetic ingredient database (COSING); no chemical testing was performed in this study. Of the 122 hair dye formulations examined, 120 contained ingredients known to be potent skin sensitizers. Notably, more than 80% of the hair dye products contained at least 4 potent skin sensitizers. The range in the numbers of potent skin sensitizers was 0 to 8 in light blonde shades, 2 to 12 in dark brown colors, and 3 to 7 in black dyes. Hydroxyethyl-3,4-Methylenedioxyaniline HCl was one of the 37 target hair dyes identified by the researchers conducting the analysis to be a potent skin sensitizer. It was reported to be used at the maximum authorized concentration of 3% or, when combined with hydrogen peroxide, at 1.5%, as determined by the Cosmetics Directive 76/768/EEC, consolidated version 2008-04-24. No additional information specific to Hydroxyethyl-3,4-Methylenedioxyaniline HCl was provided in this analysis.
In Australia, there are no known restrictions for Hydroxyethyl-3,4-Methylenedioxyaniline HCl as reported by NICNAS, although it is recommended that consumers use products containing the ingredient as instructed on the label; the assessment suggested control measures for industry to minimize risk associated with dermal exposure to Hydroxyethyl-3,4-Methylenedioxyaniline HCl.5 The NICNAS assessment mentions a recommendation, to be included in Schedule 6 of the Poisons Standard 2015-Standard for the Uniform Scheduling of Medicines and Poisons, that the concentration of use should be appropriately limited for use in hair dye products.
Non-Cosmetic
There was no relevant, non-cosmetic use information found in the literature.
Toxicokinetic Studies
Dermal Penetration
In Vitro
A percutaneous absorption study was conducted in accordance with Organization for Economic Co-operation and Development Test Guideline (OECD TG) 428 and Good Laboratory Practice (GLP). 2 Four-hundred mg of an oxidative hair dye formulation that consisted of 1.5% Hydroxyethyl-3,4-Methylenedioxyaniline HCl, 1.75 mg of an unspecified “reaction partner”, and 3% hydrogen peroxide were applied to a 4 cm2 sample of Schweizer Edelschwein porcine skin in a diffusion Teflon-chamber. This resulted in a dermal exposure of 1.5 mg/cm2 to 1.5% Hydroxyethyl-3,4-Methylenedioxyaniline HCl. A phosphate buffer containing sodium chloride and antibiotics was pumped through the receptor compartment at 5 mL/h. At 60 min post-application, the test site was washed twice with water (4 mL) followed by a shampoo formulation (4 mL) and then twice again with water. The portion of dye in the washing solutions was quantitatively determined by high-performance liquid chromatography (HPLC). At 16 and 24 h post-application, fractions of receptor compartment fluid were collected, concentrated and analyzed. When the experiment was completed, the skin above the basal layer was mechanically separated from the basal layer skin down to the upper dermis by heat treatment. These skin compartments were analyzed and the amount of dye present was quantitated by HPLC.
Test results concerning the integrity of the skin (using tritiated water) indicated that penetration of the applied amount ranged from 0.9% to 1.4%. 2 The total recovery of the applied amount of Hydroxyethyl-3,4-Methylenedioxyaniline HCl was 19.8%. The SCCS report states that the low recovery may have been caused in part by the reaction partner. The amount of Hydroxyethyl-3,4-Methylenedioxyaniline HCl that remained on the skin surface was 19.5% of the applied amount. By 24 h the applied amount recovered was 0.047% in the upper skin, 0.0067% in the lower skin, and 0.3% in the receptor compartment fluid. The skin penetration rate (i.e., the bioavailable amount, calculated by adding the portions in the skin compartments and the receptor fluid) was determined to be 5.8 μg/cm2 during the 24-h exposure duration.
Animal
Toxicokinetics Studies (ADME) for Hydroxyethyl-3,4-Methylenedioxyaniline HCl.
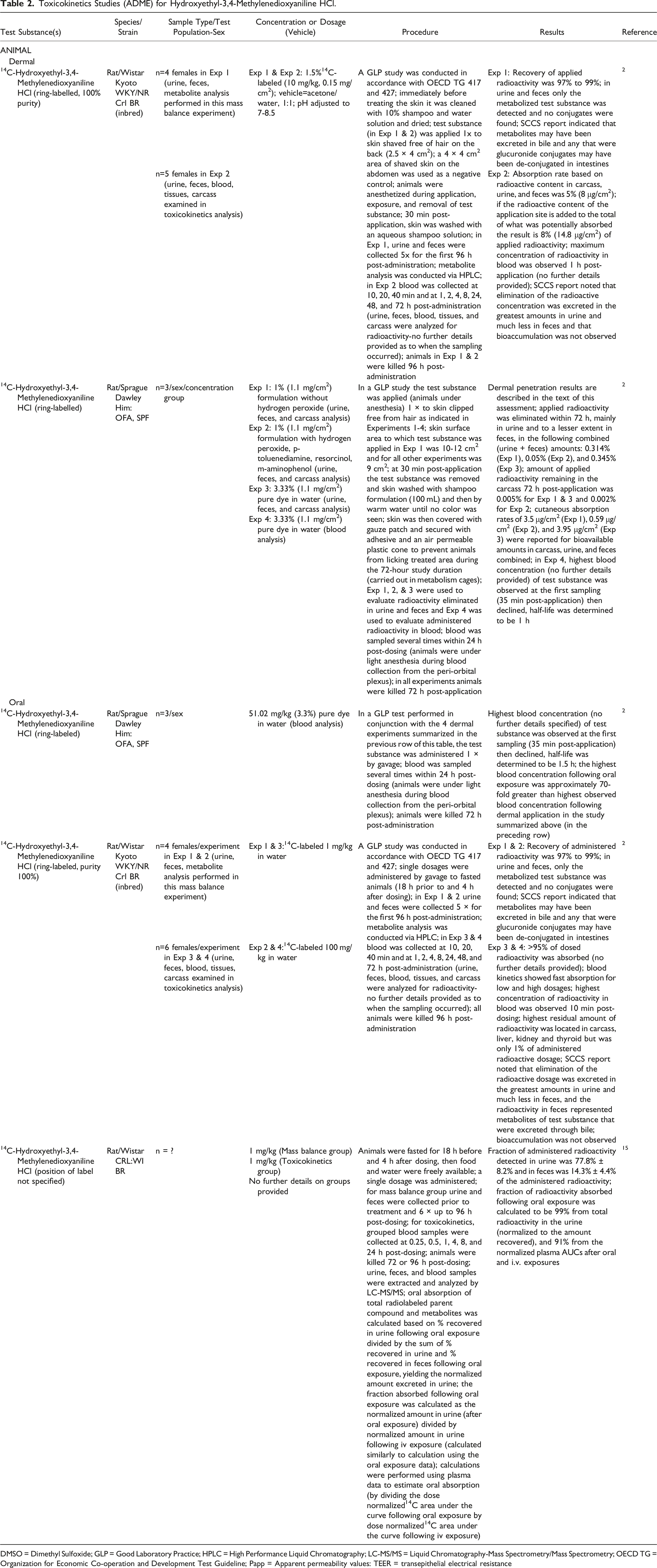
DMSO = Dimethyl Sulfoxide; GLP = Good Laboratory Practice; HPLC = High Performance Liquid Chromatography; LC-MS/MS = Liquid Chromatography-Mass Spectrometry/Mass Spectrometry; OECD TG = Organization for Economic Co-operation and Development Test Guideline; Papp = Apparent permeability values: TEER = transepithelial electrical resistance
Absorption, Distribution, Metabolism, Excretion
Absorption, distribution, metabolism, and excretion (ADME) studies are summarized below; details are presented in Table 2.
Studies were conducted to evaluate the ADME of Hydroxyethyl-3,4-Methylenedioxyaniline HCl via dermal and oral exposure routes. Experiments were performed in which 1% to 3% [14C]Hydroxyethyl-3,4-Methylenedioxyaniline HCl (ring-labeled) was applied to rat skin (clipped free of hair) for 30 min and then removed by washing. 2 The applied radioactivity was eliminated mainly in urine and to a lesser extent in feces; 0.05% to 0.345% of the applied radiolabel was recovered in urine and feces (combined) by 72 h post-application. Dermal absorption ranged from 0.59 μg/cm2 to 14.8 μg/cm2 and no substantial bioaccumulation was observed. The biological half-life of [14C]Hydroxyethyl-3,4-Methylenedioxyaniline HCl was determined from the blood analysis to be 1 h.
Experiments evaluating the effects of [14C]-Hydroxyethyl-3,4-Methylenedioxyaniline HCl (ring-labeled) in orally exposed rats (1 to 100 mg/kg single dosage) yielded results indicating that >95% of the administered radioactivity was absorbed.2,15 Blood, urine, and feces samples were collected up to 24 to 96 h post-dosing. The radioactivity detected in urine and feces were 78% and 14%, respectively, of the administered radioactivity 15 ; bioaccumulation was not observed. 2 Blood analysis showed the half-life of the test substance to be 1.5 h. 2 The highest blood concentration detected following oral exposure was 70-fold greater than the highest blood concentration after dermal exposure.
Toxicological Studies
Subchronic Toxicity
Animal
Oral
A 90-d study conducted in accordance with OECD TG 408 and GLP in Wistar HanBrl:WIST (SPF) rats (n=10/sex/dosage) was performed to evaluate the effects of Hydroxyethyl-3,4-Methylenedioxyaniline HCl (99.8% purity) following daily oral exposure, by gavage, at 0, 20, 100, and 350 mg/kg/d using a purified water vehicle. 2 One male died (350 mg/kg/d) on day 85; the SCCS report, in which the study was presented, indicated that the study researchers thought the death was unlikely to be treatment-related because no clinical symptoms were reported. In the 100 and 350 mg/kg/d groups, both sexes showed reduced locomotion. Treatment-related increases in mean absolute and relative reticulocyte counts were noted in males at 350 mg/kg/d after 13 wk, and this effect was associated with splenic extramedullary hematopoiesis found during histopathology. Reduced red blood cell count and hematocrit and hemoglobin measurements, as well as elevated mean absolute and relative reticulocyte counts, were observed in females (350 mg/kg/d). There was a decrease in thymus weights, an increase in ovary and spleen weights, extramedullary hematopoiesis in the spleen, and corpus luteum hypertrophy in the ovaries of females (350 mg/kg/d). Elevated clinical biochemistry parameters, increased liver and kidney weights, increased urine volume, hepatocellular hypertrophy, and renal tubular damage were reported in both sexes (350 mg/kg/d). At the 100 mg/kg/d dosage rate, bilirubin and phospholipid levels (clinical biochemistry parameters) were elevated in females, increases in absolute and relative liver weights and hepatocellular hypertrophy were noted in males, and increases in blood cholesterol and urinary volume occurred in both sexes. A no-observed-adverse-effect level (NOAEL) of 20 mg/kg/d was reported.
Developmental and Reproductive Toxicity (DART) Studies
A teratogenicity study was conducted in accordance with OECD TG 414 and GLP to evaluate Hydroxyethyl-3,4-Methylenedioxyaniline HCl (purity 99.8%) in rats (HanBrl: WIST, SPF quality). 2 The test substance in bi-distilled water was administered by gavage to pregnant rats (n=22/group) on days 6 to 20 of gestation at 0, 50, 250, and 1000 mg/kg/d. All animals were killed on day 21 of gestation and necropsies were performed.
Instability of the test solution was reported in this study (assumed to have been caused by “incorrect sample handling” contributing to oxidation of the test substance), although previous experiments showed Hydroxyethyl-3,4-Methylenedioxyaniline HCl to be stable in water at room temperature for 7 d. 2 Therefore, dosage rates were re-calculated based on analytical results to be 26, 152, and 702 mg/kg/day.
Following the first 3 d of dosing (day 9 to day 20 of gestation) the high dosage rate of 702 mg/kg/d was reduced to 688 mg/kg/d because of severe toxicity and mortality (5 animals died or were killed because of moribund conditions). 2 In the 688 mg/kg/d group, hypoactivity, hunched posture, lacrimation, and behavioral indications of discomfort were observed. Discomfort was also seen at 152 mg/kg/d. With 152 mg/kg/d and 688 mg/kg/d, reduced food consumption and body weight were noted. At the same dosage rates, severe stomach abnormalities and effects on adrenals, spleen, and kidneys were observed. An increase in fetal resorptions occurred with the 688 mg/kg/d group only, compared to controls. The 688 mg/kg/d treatment resulted in effects on litter size and fetal body weight (no further details provided), but not on the sex ratio of the fetuses. In the 152 and 688 mg/kg/d groups, skeletal and visceral abnormalities were seen (at 688 mg/kg/d: hemorrhages, dilated lateral brain ventricles, thymus, cranial displacement and/or elongation; at 688 and 152 mg/kg/d: heart, aorta and pulmonary effects). At 152 mg/kg/d and 688 mg/kg/d, there was an increase in non-ossified and incompletely ossified bones of the cranium, vertebrae, ribs, sternebrae, and also of extremities and supernumerary ribs. Treatment-related effects were seen on cervical and thoracic vertebrae and on costal cartilage in the 152 mg/kg/d and 688 mg/kg/d groups.
The SCCS report states that the severe reproductive and developmental effects at the 688 mg/kg/d and 152 mg/kg/d dosage rates were caused by maternal toxicity. 2 A reproductive and developmental NOAEL of 26 mg/kg/d was reported.
Genotoxicity Studies
Genotoxicity Studies for Hydroxyethyl-3,4-Methylenedioxyaniline HCl.
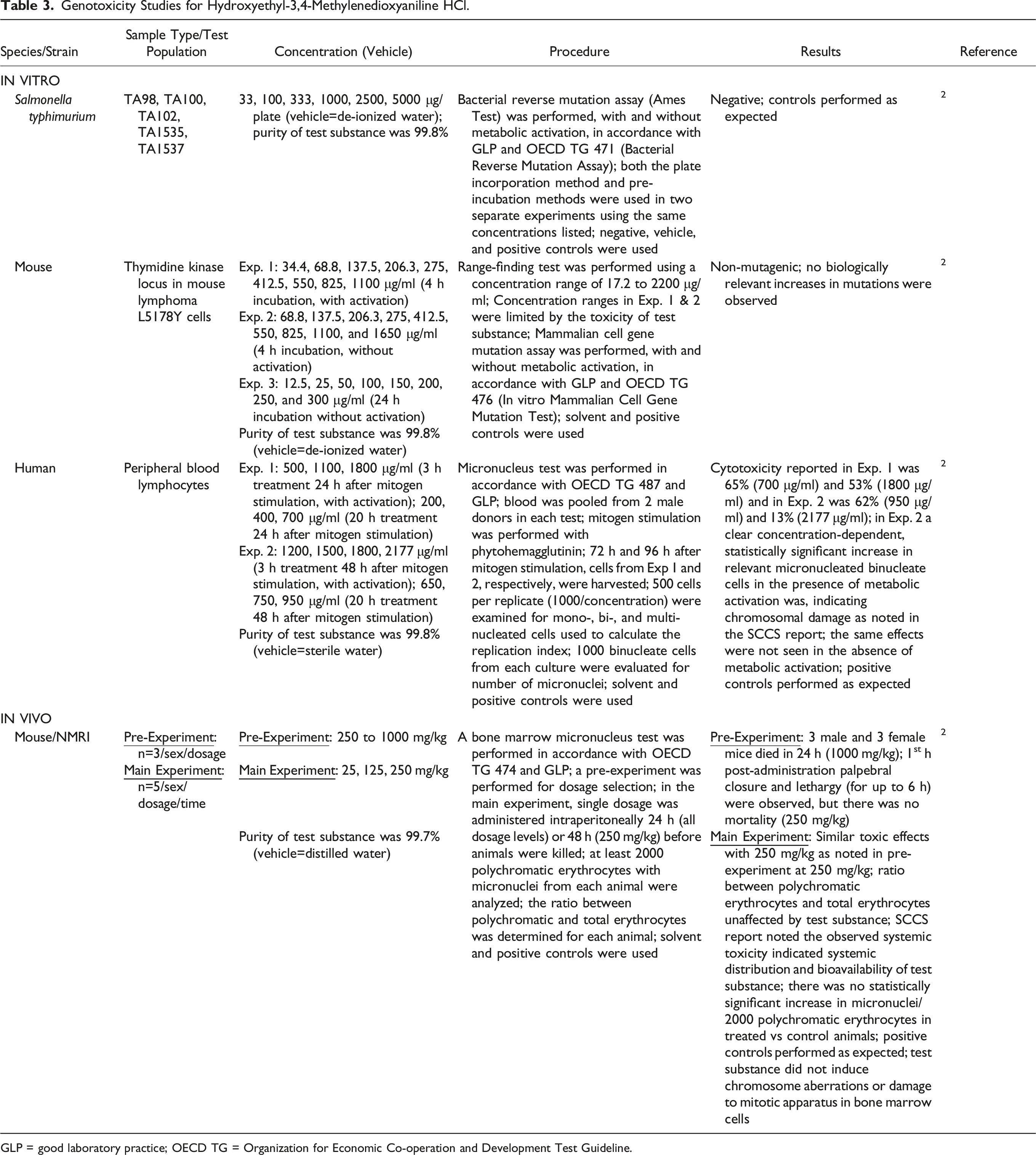
GLP = good laboratory practice; OECD TG = Organization for Economic Co-operation and Development Test Guideline.
Experiments examining Hydroxyethyl-3,4-Methylenedioxyaniline HCl were conducted in vitro. An Ames test using Salmonella typhimurium was negative for genotoxicity up to 5000 μg/plate. 2 A mammalian cell gene mutation assay using mouse lymphoma cells was non-mutagenic during a 4-h incubation with concentrations up to 1100 μg/ml with metabolic activation and up to 1650 μg/ml without activation. In an in vitro micronucleus test performed in human peripheral blood lymphocytes, there was a statistically significant, concentration-dependent increase in relevant micronucleated binucleate cells in the presence of metabolic activation. This indication of chromosomal damage resulted from a 3-h treatment (at concentrations up to 2177 μg/ml) performed 48 h after mitogen stimulation (cells were harvested 96 h after mitogen stimulation); cytotoxicity (13% to 65%) was noted at concentrations ranging from 700 to 2177 μg/ml in several of the micronucleus experiments.
Palpebral closure and lethargy were observed 1 h post-administration in an in vivo bone marrow micronucleus test conducted in mice that were intraperitoneally exposed to Hydroxyethyl-3,4-Methylenedioxyaniline HCl (up to 250 mg/kg). 2 No chromosomal aberrations or damage to the mitotic apparatus in bone marrow cells were observed from treatment with the test substance.
Carcinogenicity Studies
There were no carcinogenicity studies for Hydroxyethyl-3,4-Methylenedioxyaniline HCl found in the literature, and unpublished data were not provided.
Dermal Irritation and Sensitization Studies
Irritation
Animal
A non-GLP study was conducted in female, Pirbright White (SPF) guinea pigs (n=15) to evaluate the effect of Hydroxyethyl-3,4-Methylenedioxyaniline HCl from dermal exposure. 2 A 5% aqueous solution of Hydroxyethyl-3,4-Methylenedioxyaniline HCl (purity not specified) was applied (non-occlusive) to a 12 cm2 area of clipped flank skin one time per day for 5 consecutive days. The skin was examined for signs of irritation 5 h following each application and on the third day following the final application. The results indicated that the test substance was non-irritating.
Sensitization
Animal
A local lymph node assay (LLNA) was performed in accordance with OECD TG 429 and GLP. 2 To examine skin sensitization potential, the cell proliferation in the draining lymph nodes was measured in CBA/J mice (n=5 females/concentration) following dermal exposure to Hydroxyethyl-3,4-Methylenedioxyaniline HCl (99.7% purity). Twenty-five microliters of 0 (vehicle only), 0.5, 1.5, 5, and 10% Hydroxyethyl-3,4-Methylenedioxyaniline HCl (in DMSO, or in a 3:1 mixture of water/acetone (1:1) and olive oil) were applied to the ear skin of mice for 3 consecutive days. A hair dryer was used to dry the ears after application.
At the 1.5% test concentration (DMSO vehicle), slight-to-severe skin desquamation on the ears was observed in all animals by day 5, and erythema was noted in one animal. The SCCS report indicated that these effects were attributed to the low pH of the test solution. 2 The stimulation indices for all the test concentrations in either vehicle were greater than 3 (dose-dependent with DMSO vehicle), and an EC3 (“equal to the concentration inducing a stimulation index of 3”) was not estimated. Positive controls performed as expected. The SCCS report, in which this study was presented, indicated that the results using either vehicle indicated that dermally applied Hydroxyethyl-3,4-Methylenedioxyaniline HCl has strong sensitization potential in mice.
Quantitative Structure-Activity Relationship (QSAR) Analysis
A study conducted in 2004 applied a previously published, quantitative structure-activity relationship (QSAR) model to estimate the skin sensitization potential of all European registered hair dye substances, including Hydroxyethyl-3,4-Methylenedioxyaniline HCl. 16 Topological sub-structural molecular descriptors (TOPS-MODE) were correlated with LLNA experimental data (based on LLNAs of 93 chemicals) to construct a general QSAR model, unrestrictive to chemical class. This QSAR model was not specifically designed for hair dye substances, but deemed by the researchers conducting the study to be suitable for this application. The allergic contact dermatitis that may develop from the use of skin-sensitizing compounds involves the formation of an antigenic complex, consisting of a protein covalently bonded to a hapten (low-molecular-weight molecule). This type of biochemical reaction is amenable to developing predictive tools, such as QSAR models. A literature review was also conducted as part of this study to retrieve relevant LLNA data (up to July 2003) and human evidence (up to August 2003) to help validate the QSAR predictions or elucidate areas in need of improvement. Hair dyes that were studied in this analysis were classified as strong/moderate, weak, or extremely weak sensitizers or non-sensitizing. Values indicating rank were assigned to each chemical studied, reflecting the predicted relative sensitization potential within each classification group. Approximately 75% of the 229 hair dye substances assessed were classified as strong/moderate sensitizers based on the QSAR model prediction. Hydroxyethyl-3,4-Methylenedioxyaniline HCl was predicted to be a moderate or strong sensitizer with an assigned numerical value of 0.2. For comparison, the highest value assigned to the hair dye substances categorized as strong/moderate sensitizers was 16.3 for Direct Red 80.
Ocular Irritation Studies
A non-GLP study was conducted in Pirbright White (SPF) guinea pigs (n=10 females) evaluating 2% (water vehicle) Hydroxyethyl-3,4-Methylenedioxyaniline HCl. 2 A single, 0.1 mL application of the test substance was instilled into the conjunctival sac of the right eye, and the left eye served as the control; the eyes were not rinsed following application. Eyes were examined 0.5, 1, 2, 3, 4, 6, and 7 h after the instillation of the test substance. At 24 h following treatment, a fluorescein-instillation reading was performed. Edema, slight conjunctival redness, and corneal opacity were observed in 2 animals 3 h post-instillation. The SCCS report indicated that 2% Hydroxyethyl-3,4-Methylenedioxyaniline HCl caused transient irritation to guinea pig eyes.
Epidemiology Studies
Hydryoxyethyl-3,4-Methylenedioxyaniline HCl is used as an oxidative (permanent) hair dye ingredient. While the safety of individual hair dye ingredients is not addressed in epidemiology studies that seek to determine links, if any, between hair-dye use and disease, such studies do provide broad information. Currently available epidemiology studies provided insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers.
A detailed summary of the available hair dye epidemiology data is available at http://www.cir-safety.org/cir-findings.
Summary
Hydroxyethyl-3,4-Methylenedioxyaniline HCl functions in cosmetics as a hair colorant. VCRP data received in 2017 indicate that there are 67 reported uses of Hydroxyethyl-3,4-Methylenedioxyaniline HCl in hair dyes and colors; a Council industry survey received in 2016 reported maximum use concentrations to be 0.52% to 0.75% (diluted or “on head” concentrations) in hair dyes and colors.
Hydroxyethyl-3,4-Methylenedioxyaniline HCl contains a free, secondary aromatic substituted amine (aniline derivative), which raises a concern about possible N-nitrosation. The SCCS report specified a limit of <50 ppb for nitrosating content in hair dye formulations containing Hydroxyethyl-3,4-Methylenedioxyaniline HCl.
An in vitro percutaneous absorption study evaluated 1.5 mg/cm2 (1.5%) Hydroxyethyl-3,4-Methylenedioxyaniline HCl as part of an oxidative hair dye formulation that was applied to 4 cm2 porcine skin mounted on a diffusion-chamber. The test substance was washed off the skin 60 min post-application. At 16 and 24 h post-application, fractions of receptor compartment fluid were collected, concentrated, and analyzed. By 24 h the applied amount recovered was 0.047% in the upper skin (above the basal layer), 0.0067% in the lower skin (basal layer skin down to upper dermis), and 0.3% in the receptor compartment fluid. The skin penetration rate was determined to be 5.8 μg/cm2.
An in vivo percutaneous absorption experiment in rat skin was conducted to evaluate [14C]Hydroxyethyl-3,4-Methylenedioxyaniline HCl (ring-labeled) in the following formulations: 3.33% pure dye in water; 1% formulation with hydrogen peroxide (also containing p-toluenediamine, resorcinol, and m-aminophenol in this application); 1% formulation without hydrogen peroxide. At 30 min post-application the test substance was removed and the skin washed. Results indicated that the washing solutions contained 95.9% to 97.1% of the applied radiolabeled concentration. The skin contained 1.68% of the applied radioactivity for the formulation containing water, 0.56% for the formulation containing hydrogen peroxide, and 0.34% for the formulation without hydrogen peroxide.
In dermal ADME studies (in vivo), 1% to 3% [14C]Hydroxyethyl-3,4-Methylenedioxyaniline HCl (ring-labeled) was applied to rat skin for 30 min and then removed by washing. The applied radioactive concentration detected in urine and feces (combined percentages) by 72 h post-application were 0.05% to 0.345%. Dermal absorption rates ranged from 0.59 μg/cm2 to 14.8 μg/cm2 and no substantial bioaccumulation was observed.
Oral ADME studies were performed in rats administered single dosages by gavage of 1 to 100 mg/kg [14C]Hydroxyethyl-3,4-Methylenedioxyaniline HCl (ring-labeled). Results indicated that >95% of the radioactivity was absorbed. Blood, urine, and feces samples were collected up to 24 to 96 h post-dosing. The radioactivity detected in urine and feces were 78% and 14%, respectively; bioaccumulation was not observed. The highest blood concentration detected following oral exposure was 70-fold greater than the highest blood concentration after dermal exposure.
A subchronic (90-d) oral exposure study was conducted in rats to evaluate the effects of Hydroxyethyl-3,4-Methylenedioxyaniline HCl following administration by gavage of 0, 20, 100, and 350 mg/kg/d in purified water. In the 100 and 350 mg/kg/d groups, both sexes showed reduced locomotion. Treatment-related abnormalities in red blood cells were observed in males at 350 mg/kg/d after 13 wk, and were associated with splenic extramedullary hematopoiesis found during histopathology. Abnormalities in red blood cells and organs (thymus, ovary, spleen) were observed in females (350 mg/kg/d). In both sexes elevated clinical biochemistry parameters and liver and kidney abnormalities were reported (350 mg/kg/d). At 100 mg/kg/d elevated bilirubin levels and phospholipids were seen in females, liver abnormalities were noted in males, and increased cholesterol and urinary volume occurred in both sexes. A NOAEL of 20 mg/kg/d was reported.
A teratogenicity study was conducted to evaluate the effects of Hydroxyethyl-3,4-Methylenedioxyaniline HCl in pregnant rats that were exposed by gavage to the following dosage rates (on days 6 to day 20 of gestation): 0, 26, 152, and 702 mg/kg/d. Following the first 3 d of dosing (days 9 to day 20 of gestation), 702 mg/kg/d was reduced to 688 mg/kg/d because of severe toxicity and mortality. In the 688 mg/kg/d group, hypoactivity, hunched posture, lacrimation, and behavioral indications of discomfort (discomfort also seen with 152 mg/kg/d) were observed. At 152 and 688 mg/kg/d reduced food consumption and body weight were noted. At the same dosage rates, stomach abnormalities and effects on adrenals, spleen, and kidneys were observed. An increase in fetal resorptions occurred in the 688 mg/kg/d group only. The 688 mg/kg/d treatment resulted in effects on litter size and fetal body weight, but not on sex ratio of fetuses. For the 152 and 688 mg/kg/d groups, skeletal and visceral abnormalities were seen. The SCCS report states the severe reproductive and developmental effects observed with the 152 and 688 mg/kg/d dosage rates were caused by maternal toxicity. A reproductive and developmental NOAEL of 26 mg/kg/d was reported.
Hydroxyethyl-3,4-Methylenedioxyaniline HCl was negative in an Ames test (using Salmonella typhimurium) up to 5000 μg/plate. Results from a mammalian cell gene mutation assay using mouse lymphoma cells indicated that the ingredient was non-mutagenic during a 4-hour incubation at concentrations up to 1100 μg/ml with metabolic activation and up to 1650 μg/ml without activation. An in vitro micronucleus test performed in human peripheral blood lymphocytes showed a statistically significant, concentration-dependent increase in relevant micronucleated binucleate cells in the presence of metabolic activation. This indication of chromosomal damage resulted from a 3-h treatment (at concentrations up to 2177 μg/ml) performed 48 h after stimulation with a mitogen. No chromosomal aberrations or damage to the mitotic apparatus in bone marrow cells were observed in an in vivo bone marrow micronucleus test conducted in mice intraperitoneally exposed to Hydroxyethyl-3,4-Methylenedioxyaniline HCl (up to 250 mg/kg).
There were no carcinogenicity studies for Hydroxyethyl-3,4-Methylenedioxyaniline HCl found in the literature.
Hydroxyethyl-3,4-Methylenedioxyaniline HCl (5% aqueous solution) was not irritating when applied (non-occlusive) one time per day for 5 consecutive days to guinea pig skin clipped free of hair.
Hydroxyethyl-3,4-Methylenedioxyaniline HCl (up to 10% in DMSO or in a 3:1 mixture of aqua/acetone (1:1) and olive oil) was shown to have strong sensitization potential in mouse skin in an LLNA test. In a Swedish market analysis (no chemical testing was conducted in this study) Hydroxyethyl-3,4-Methylenedioxyaniline HCl was one of 37 target hair dyes in 122 products examined. Hydroxyethyl-3,4-Methylenedioxyaniline HCl was identified by the researchers conducting the analysis to be a potent skin sensitizer. It was reported to be used at the maximum authorized concentration of 3% or when combined with hydrogen peroxide at 1.5%.
A study was conducted applying a previously published QSAR model (based on TOPS-MODE and LLNA experimental data) to estimate the skin sensitization potential of all European registered hair dye substances in 2004, including Hydroxyethyl-3,4-Methylenedioxyaniline HCl. Hydroxyethyl-3,4-Methylenedioxyaniline HCl was predicted to be a moderate or strong sensitizer.
A study was conducted in guinea pigs to evaluate the ocular irritation potential of 2% (water vehicle) Hydroxyethyl-3,4-Methylenedioxyaniline HCl. A single, 0.1 mL application of the test substance was instilled into the conjunctival sac of the right eye while the left eye served as the control; the eyes were not rinsed following application. Edema, slight conjunctival redness, and corneal opacity were observed in 2 animals, 3 h after treatment. The SCCS report indicated that 2% Hydroxyethyl-3,4-Methylenedioxyaniline HCl caused transient irritation to guinea pig eyes.
The most recent, comprehensive review of available epidemiology studies concluded that there is insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers. A summary of the available hair dye epidemiology data is available at http://www.cir-safety.org/findings.html.
Discussion
The Panel reviewed the data presented in this assessment to determine the safety of Hydroxyethyl-3,4-Methylenedioxyaniline HCl as used in hair dyes. Although no carcinogenicity data were found in the literature, the Panel concluded that the animal studies evaluating percutaneous dermal absorption, toxicokinetics (dermal and oral exposure), subchronic oral exposure, teratogenicity, and genotoxicity adequately support a low toxicity profile for this hair dye ingredient when used according to the present practices of use and concentration described in this safety assessment. Hydroxyethyl-3,4-Methylenedioxyaniline HCl was non-irritating to guinea pig skin and transiently irritating to guinea pig eyes at concentrations greater than those expected in human use. Animal tests and QSAR analysis, predictive of potential human response, indicated that Hydroxyethyl-3,4-Methylenedioxyaniline HCl may be a skin sensitizer. Hair dye ingredients are known to be potential skin irritants/sensitizers. The Panel emphasized that labeling instructions included for the use of Hydroxyethyl-3,4-Methylenedioxyanline HCl in hair dye formulations should be carefully followed.
Hair dyes containing Hydroxyethyl-3,4-Methylenedioxyaniline HCl, as a coal tar hair dye ingredient, are exempt from certain adulteration and color additive provisions of the Federal Food, Drug, and Cosmetic Act, when the label includes a caution statement and consumer patch test instructions for determining whether the product causes skin irritation. The Expert Panel expects that following this procedure will identify prospective individuals who would have an irritation/sensitization reaction and allow them to avoid significant exposures. The Panel considered concerns that such self-testing might induce sensitization, but agreed that there was not a sufficient basis for changing this advice to consumers at this time.
The Panel concluded that hair dye formulations containing Hydroxyethyl-3,4-Methylenedioxyaniline HCl should not be used with nitrosating agents because of the potential for N-nitrosation of the free, secondary aromatic substituted amine in the ingredient and the possible presence of nitrosating impurities (i.e., 3,4-methylenedioxy-aniline).
In considering hair dye epidemiology data, the Panel concluded that the available epidemiology studies are insufficient to scientifically support a causal relationship between hair dye use and cancer or other toxicologic endpoints, based on lack of strength of the associations and inconsistency of findings.
The Panel noted that the use of oxidative hair dye formulations involves exposure to precursors and coupling agents, as well as to their reaction products. While reaction intermediates are formed, human exposure is primarily to the precursors, coupling agents, and reaction products, not the reaction intermediates. Exposures to the precursors and couplers are low, because they are consumed in the color forming reaction, and exposures to reaction products are even lower, because they are adsorbed onto and physically retained on the hair shaft. Therefore, it was the consensus of the Panel that safety assessments of oxidative hair dyes are primarily determined by the toxicological evaluation of the ingredients (i.e., precursors and coupling agents), and not of the reaction intermediates or products formed during use.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Hydroxyethyl-3,4-Methylenedioxyaniline HCl is safe for use as a hair dye ingredient in the present practices of use and concentrations described in this safety assessment.
Footnotes
Author’s Contribuion
Scott, L.N. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W.F., Belsito, D.V., Hill, R.A., Klaassen, C.D., Liebler, D.C., Marks, J.G., Shank, R.C., Slaga, T.J., and Snyder, P.W. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.