Abstract
The aim of this study was to investigate the mechanism of isoflurane in proliferation, invasion, and migration in prostate cancer (PC) cells in vitro by regulating BACH1 and miR-375. The effect of different concentrations of isoflurane (0%, 0.5%, 1%, and 2%) on PC cell proliferation (PC3 and 22RV1) was measured. After PC cells and normal human prostate stromal immortalized WPMY-1 cells were treated with isoflurane, BACH1 and miR-375 expression was measured. Subsequently, PC3 and 22RV1 cells underwent gain- and loss-of-function assays with or without 4-h 2% isoflurane pretreatment. The levels of miR-375, BACH1, and PTEN were assessed. The binding of BACH1 to miR-375 promoter was detected by ChIP assay. Dual-luciferase reporter assay detected the targeting relationship of miR-375 with BACH1 and PTEN. Isoflurane promoted PC3 and 22RV1 cell proliferation. In addition, isoflurane elevated the levels of BACH1 and miR-375 in a dosage-dependent manner in PC cells. Transfection with miR-375 inhibitor or sh-BACH1 repressed PC cell proliferation, invasion, and migration, while exposure to 2% isoflurane for 4 h before transfection counteracted the inhibitory effects of sh-BACH1 or miR-375 inhibitor on PC cells. PTEN expression was suppressed after 2% isoflurane treatment, but the transfection with miR-375 inhibitor partly abrogated this suppressive effect in PC cells. Moreover, BACH1 bound to miR-375 and miR-375 negatively targeted PTEN. miR-375 mimic could partially reverse the inhibitory effects of sh-BACH1 on the proliferation, invasion, and migration of isoflurane-treated PC cells. Isoflurane facilitated PC cell proliferation, migration, and invasion by activating BACH1 to upregulate miR-375.
Introduction
Prostate cancer (PC) is the most common cancer in developed countries and the second-leading cause of cancer-related deaths in men worldwide. 1 Over the past few decades, the risk factors for PC have been discussed in the areas of androgen-independent growth and genetic risks.2-5 The global cases and deaths of PC was 1.4 million and 0.38 million, respectively, in 2016, during which the disability-adjusted life-years reached 6.1 million. 6 A recent study has shown that the 5-year survival rate of patients with localized PC is 100% but the survival rate of late staged cancer patients is only 28%. 7 Recently, there have been discussions that the use of anesthetic techniques may affect the outcomes of PC. 8 Hence, further researches are warranted to dissect the exact relationship between anesthetic techniques and PC development.
Isoflurane is one type of inhalational anesthetic, which rapidly induces anesthesia and emergence. 9 It has been reported that isoflurane has a cytoprotective effect in vivo and in vitro. 10 However, a growing volume of literature suggests that isoflurane promotes cell proliferation and aggravates malignancy in squamous cervical cancer cells, renal cell carcinoma cells, ovarian cancer cells as well as glioblastoma stem cells.11,12,10,13 Intriguingly, isoflurane may have promoting effects on PC cells, 14 but few studies have focused on the mechanisms of isoflurane on PC malignant behaviors.
microRNAs (miRNAs) are a class of small non-coding RNAs that are located in open chromatin and have a length of 18–25 nucleotides.15,16 Mounting evidence reported that miRNAs are widely involved in the proliferation, growth, apoptosis, and differentiation of tumor cells, suggesting that miRNAs could be acted as potential predictive parameters for PC diagnosis and prognosis.17,18 miR-375, located on the genetic regions of CCDC108 and CRYBA2 on 2q35, is considered as an islet-specific miRNA in regulating insulin secretion. 19 Also, miR-375 can be involved in many types of tumors via targeting important genes.20-23 The correlation of miR-375 expression with PC metastasis, development, and other clinicopathological characteristics has been widely reported.24-27,22,28,29 Of which, a growing list of studies revealed that miR-375 was highly expressed in PC samples in contrast to that in normal prostate samples.30-35 However, others hold the opposite view that miR-375 was effectively down-regulated in PC patients. 25 Moreover, some negative signals for the relationship between miR-375 and PC were also reported. 36 Thus, data published on the expression of miR-375 for PC remains controversial, which needs deeper validation studies.
Thus, the work was to explore the function of isoflurane on cell proliferation, migration, and invasion of PC in vitro and its underlying mechanism. The present study revealed a novel mechanism, showing that isoflurane could activate BACH1 to increase miR-375 expression, thus promoting PC cell proliferation, invasion, and migration.
Materials and Methods
Cell Culture
Normal human prostate stromal immortalized cells WPMY-1, human PC cell lines (PC3 and 22RV1), and human embryonic kidney 293T (HEK-293T) cells were acquired from the Cell Bank of the American Type Culture Collection. The PC cell lines were maintained in RPMI-1640 (Thermo Fisher Scientific, MA, USA) supplemented with 100 U/mL penicillin, 100 µg/mL streptomycin, and 10% fetal bovine serum (FBS). HEK-293T cells were cultured in DMEM (Thermo Fisher Scientific) containing 100 U/mL penicillin, 100 µg/mL streptomycin, and 10% FBS. Cells were cultured at 37°C in a 5% CO2 atmosphere.
Isoflurane Exposure
PC3 or 22RV1 cells were cultured at a density of 1 × 106 cells/mL on Petri dishes for 24 h. Upon reaching 80% confluence, the cells were placed in a closed plexiglass chamber. The air intake was connected to the anesthetic evaporator and the air outlet to the gas analyzer. Thereafter, the evaporator (containing 5% CO2, 21% O2, and 74% N2) was opened before the gas flow was adjusted to 3 L/min and maintained for 3 min. Then, the gas flow was changed to 0.5 L/min, after which the evaporator was opened and the concentrations of inhalation anesthetics reached the corresponding concentrations of 0%, 0.5%, 1%, and 2% (these were labeled as 0% group, 0.5% group, 1% group, and 2% group) and were maintained for 5 min. Afterward, the air inlet and outlet were closed and the closed plexiglass chamber was placed for 4 h in a constant incubator at 37°C, followed by incubation at 37°C in a 5% CO2 incubator for 24 h. Meanwhile, cells utilized as the treatment control (the blank group) were positioned in an identical gas chamber with 21% O2 and 5% CO2 balanced with nitrogen at 37°C.
Cell Transfection
Logarithmic phase PC3 and 22RV1 cells were transfected with 2 µg of short hairpin (sh)-negative control (NC), sh-BACH1, 100 nM of miR-375 inhibitor -negative control (inhibitor -NC), miR-375 inhibitor, miR-589-5p mimic-negative control (mimic NC), or miR-589-5p mimic (RiboBio, Guangzhou, China), which were separately assigned into the following groups: a sh-NC group, a sh-BACH1 group, an inhibitor NC group, a miR-375 inhibitor group, a mimic NC group, a miR-375 mimic group, and a control group (without treatment). In addition, PC3 and 22RV1 cells in logarithmic phase treated with 2% isoflurane for 4 h were transfected with 2 μg of sh-NC or sh-BACH1, and/or 100 nM of miR-375 inhibitor, mimic NC or miR-375 mimic (RiboBio), after which the cells were assigned to the Iso + sh-NC group, the Iso + sh-BACH1 group, the Iso + miR-375 inhibitor group, the Iso + sh-BACH1 + mimic NC group, the Iso + sh-BACH1 + miR-375 mimic group, and the Iso group (untransfected cells). All transfections were performed strictly following the protocols using the lipofectamine 2000 kit (Thermo Fisher Scientific). The transfected cells were cultured in RPMI-1640 and stored in a 37°C incubator with 5% CO2.
MTT Assay
Cells were counted after corresponding treatment at the indicated time (24 h, 48 h, 72 h, and 96 h). Thereafter, 100 μL of cell suspension (104 to 105 cells) was inoculated into each well of 96-well plates and each group was arranged three duplicate wells before the cells were incubated with 5% CO2 at 37°C. Afterward, 20 μL of MTT solution (5 mg/mL, Sigma-Aldrich, St. Louis, MI, USA) was added into each well and cultured for 4 h before the culture medium was discarded. Then, DMSO was added at a dosage of 150 μL and shaken gently for 10 min to facilitate the dissolution of crystal. The absorbance value for each well was determined using an enzyme-linked immunometric meter at a wavelength of 450 nm (OD450). Time was set as the abscissa and absorbance value as the ordinate to draw MTT curves. The mean value was obtained after the absorbance value of each group was determined thrice.
Transwell Assay
Firstly, 0.1 mL Matrigel (200 μg/mL; BD Bioscience, Franklin Lakes, NJ, USA) was plated into Transwell plates (Corning Costar, Cambridge, MA) overnight at 37°C. Then, the transfected PC3 and 22RV1 cells were resuspended in apical Transwell chambers and inoculated at a density of 5 × 104 cells/well. Meanwhile, RPMI-1640 medium containing 10% FBS was added into the basolateral chambers and cultured in a constant incubator in 5% CO2 (37°C, 24 h). Thereafter, the migrated cells were fixed with 4% paraformaldehyde for 10 min and stained with 0.5% crystal violet (10 min, room temperature). Then, the number of invasive cells was counted under a light microscope. The number of invasive cells in each group was counted from five randomly selected fields and analyses were independently repeated thrice.
Cell Scratch Test
Cell scratch assays were performed as described previously. 37 Briefly, cells in the experiment and control groups were plated in six-well plates. After cells grew to 90% confluence in six-well plates, three scratches were drawn in the plates with a 100 μL pipette tip. Thereafter, cells were washed with PBS and replaced with serum-free medium. After incubation for 24 h, the gap between cells was observed under a low-magnification phase-contrast microscope (Olympus MK, Tokyo, Japan). Migration rate = (0 h scratch distance − 24 h scratch distance)/0 h scratch distance.
qRT-PCR
Primer Sequences for qRT-PCR to Determine the Expression Levels of BACH1, miR-375, PTEN, GAPDH, and U6.
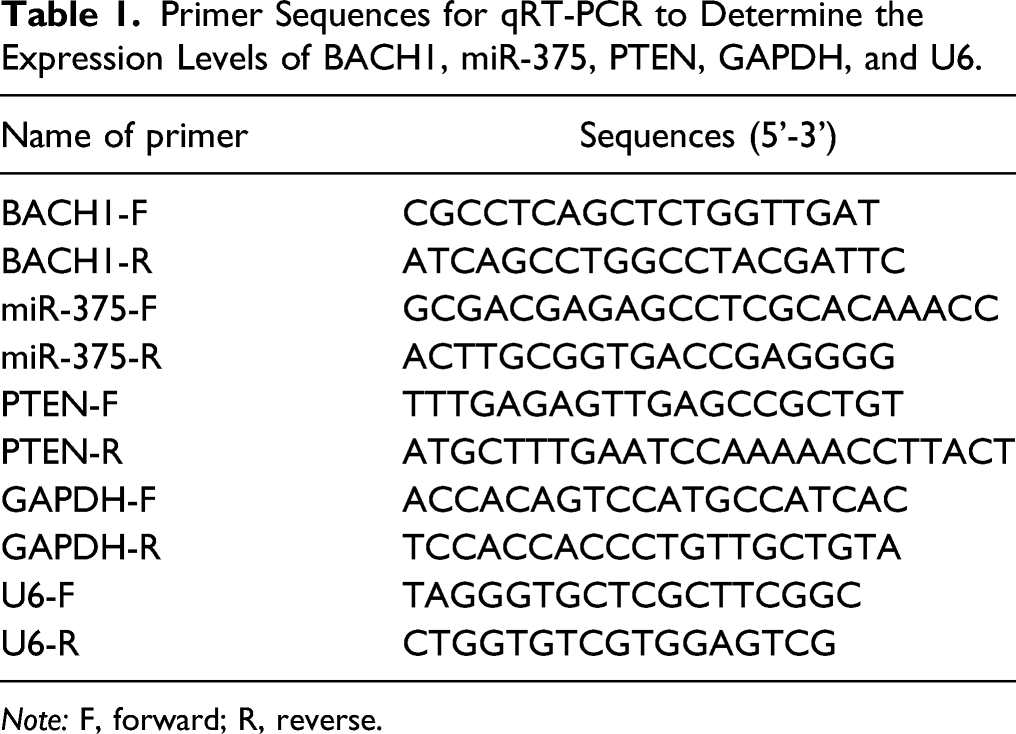
Note: F, forward; R, reverse.
Western Blot Analysis
PC3 and 22RV1 cells were rinsed 3 times with pre-cooled PBS buffer, followed by the addition of protein extraction lysis buffer (100 μL/50 mL) on ice (30 min). After centrifugation at 4°C and 12,000 rpm for 10 min, the supernatant was transferred to 0.5-mL centrifuge tubes and stored at −20°C before protein quantification using BCA kits (Sigma-Aldrich) and protein denaturation with 6 × SDS loading buffer at 100°C. Thereafter, the proteins were separated by SDS electrophoresis and then transferred into PDVF membranes in a pre-cooled transfer buffer (1.5 h, 4°C), followed by membrane blocking with nonfat milk (5%) in TBST buffer (1 h). TBST-prepared primary rabbit antibodies against BACH1 (1:2000, ab180853), GAPDH (1:10000, ab181602), and PTEN (1:1000, ab267787) (Abcam, Cambridge, USA) were separately incubated with the membranes overnight at 4°C before 3 TBST washes (10 min each). Then, the membranes were incubated with goat anti-mouse immunoglobulin G (IgG) or goat anti-rabbit IgG (1:5000, Beijing ComWin Biotech Co., Ltd, Beijing, China) (2 h, room temperature). After TBST washing, the levels of proteins were detected after development.
Chromatin Immunoprecipitation
The binding of BACH1 to miR-375 promoter was analyzed by ChIP assay. All steps were performed utilizing a ChIP Kit (Millipore, Billerica, MA, USA) following the manufacturer’s instructions. In brief, the crosslink between proteins and DNA was fixed with formaldehyde for 30 min. Then, DNA was isolated from PC cells and fragmented into 200 ∼ 1000 bp with sonication. Afterward, the DNA fragments were incubated with BACH1 or IgG antibody before qRT-PCR.
Dual-Luciferase Reporter Assay
The binding sites of BACH1 and miR-375 were predicted using a bioinformatics method (TransmiR v2.0 database). Online software Starbase was employed to predict the binding sites of miR-375 to PTEN. In light of the prediction results, the wild-type (WT) and mutant-type (MUT) sequences of the binding sites of BACH1 to miR-375 or of PTEN to miR-375 were separately designed. Then, the WT and MUT sequences of BACH1 and miR-375 were cloned and conjugated to psiCHECK2 vectors (Hymed, Guangzhou, China), and those of PTEN and miR-375 to pmirGLO vectors (Promega, WI, USA), which were designated as WT-miR-375, MUT-miR-375, WT-PTEN, and MUT-PTEN, respectively. Then, WT-miR-375 or MUT-miR-375 was co-transfected with sh-NC or sh-BACH1 into HEK-293T cells. WT-PTEN or MUT-PTEN was co-transfected with miR-589-5p mimic, miR-589-5p inhibitor, or corresponding NCs into HEK-293T cells. Afterward, the HEK-293T cells were further cultured in RPMI-1640 medium at constant temperature (37°C) with 5% CO2 for 48 h. Finally, the luciferase activity was assessed using a dual-luciferase reporter kit (Promega).
Statistical Analysis
Data were analyzed by GraphPad Prism 7.0 (GraphPad Software Inc.) and SPSS 18.0 (IBM Corp., Armonk, NY, USA). All data were represented as mean ± standard deviation. A T-test was applied to compare two groups, and one-way analysis of variance (ANOVA) was adopted to compare multiple groups. P < 0.05 was deemed as statistically significant.
Results
Isoflurane Promoted PC Cell Proliferation
In this study, the effects of isoflurane at different concentrations (0%, 0.5%, 1%, and 2%) on the proliferation of PC cell lines (PC3 and 22RV1) were detected. Untreated cells were utilized as an empty control (the blank group). MTT assay denoted that isoflurane exposure for 4 h increased PC3 and 22RV1 cell proliferation in a dose-dependent manner (P < 0.05), but there was no statistical difference between the 0% group and the blank group (Figure 1A-B). Normal human prostate stromal immortalized cells WPMY-1 underwent the same treatments. MTT assay revealed that treatment with different concentrations of isoflurane exerted no notable effect on WPMY-1 cell proliferation (Figure 1C). These results implicated the facilitation of PC cell proliferation by isoflurane treatment. Isoflurane potentiates PC cell proliferation.Note: After PC cell lines (PC3 and 22RV1) and normal human prostate stromal immortalized cells WPMY-1 were treated with different concentrations of isoflurane (0%, 0.5%, 1%, and 2%), and untreated cells were arranged as the control, cell viability of PC3 cells (A), 22RV1 cells (B), and normal human prostate stromal immortalized cells WPMY-1 (C) were tested by MTT assay. Values were expressed as mean ± standard deviation. *P < 0.05, **P < 0.01, vs the 0% group; PC, prostate cancer.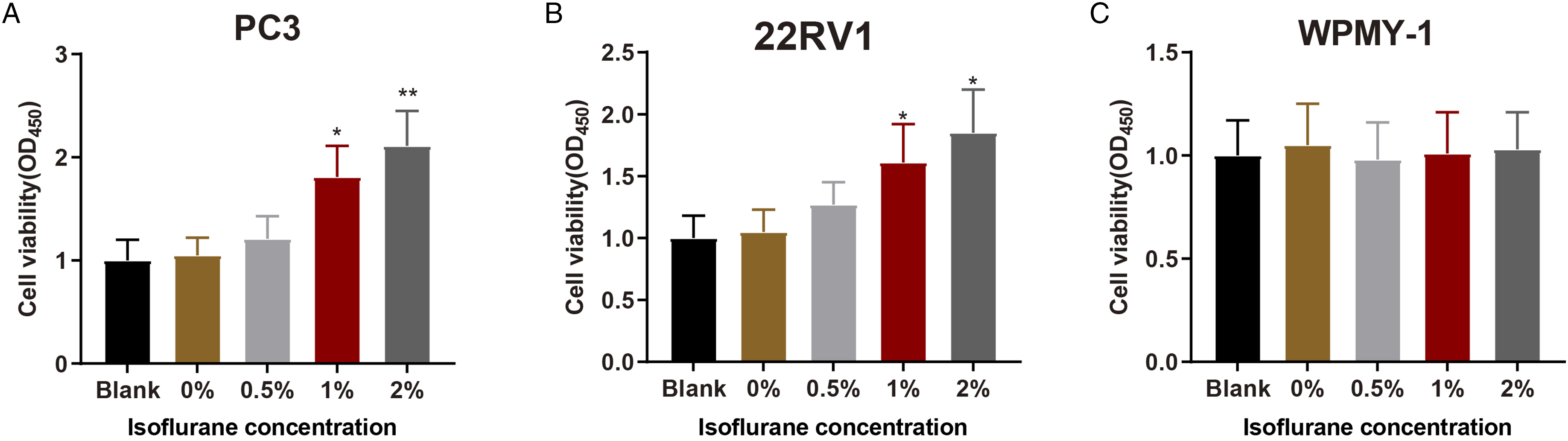
Isoflurane Upregulated BACH1 and miR-375 Expression in PC Cells
Four h after PC3 and 22RV1 cells were exposed to different concentrations of isoflurane (0%, 0.5%, 1%, 2%), qRT-PCR and western blot analysis revealed that BACH1 expression was increased (Figure 2A-D, P < 0.05), and miR-375 expression was also elevated in a dose-dependent manner, while there was insignificant difference between the 0% group and the blank group (Figure 2E-F, P < 0.01). Additionally, 2% isoflurane treatment significantly elevated BACH1 and miR-375 expression. However, in WPMY-1 cell with the same treatment, there was insignificant difference in BACH1 and miR-375 expression among all groups (Figure 2G-I). Therefore, 2% isoflurane was selected for the subsequent treatment of PC cell lines (PC3 and 22RV1) to investigate the impact of isoflurane on PC cell behaviors. Isoflurane increases BACH1 and miR-375 expression in PC cells. RT-qPCR and western blot analysis were employed for respectively detecting mRNA and protein expressions of BACH1 in PC3 (A-B); RT-qPCR and western blot analysis were utilized for respectively detecting mRNA and protein expressions of BACH1 in 22RV1 (C-D); the expression of miR-375 in PC3 and 22RV1 was detected by RT-qPCR (E-F); mRNA and protein expressions of BACH1 in WPMY-1 were detected by RT-qPCR and western blot analysis (G-H); miR-375 expression was tested by RT-qPCR (I). Values are expressed as mean ± standard deviation. *P < 0.05, **P < 0.01, vs the 0% group; PC, prostate cancer.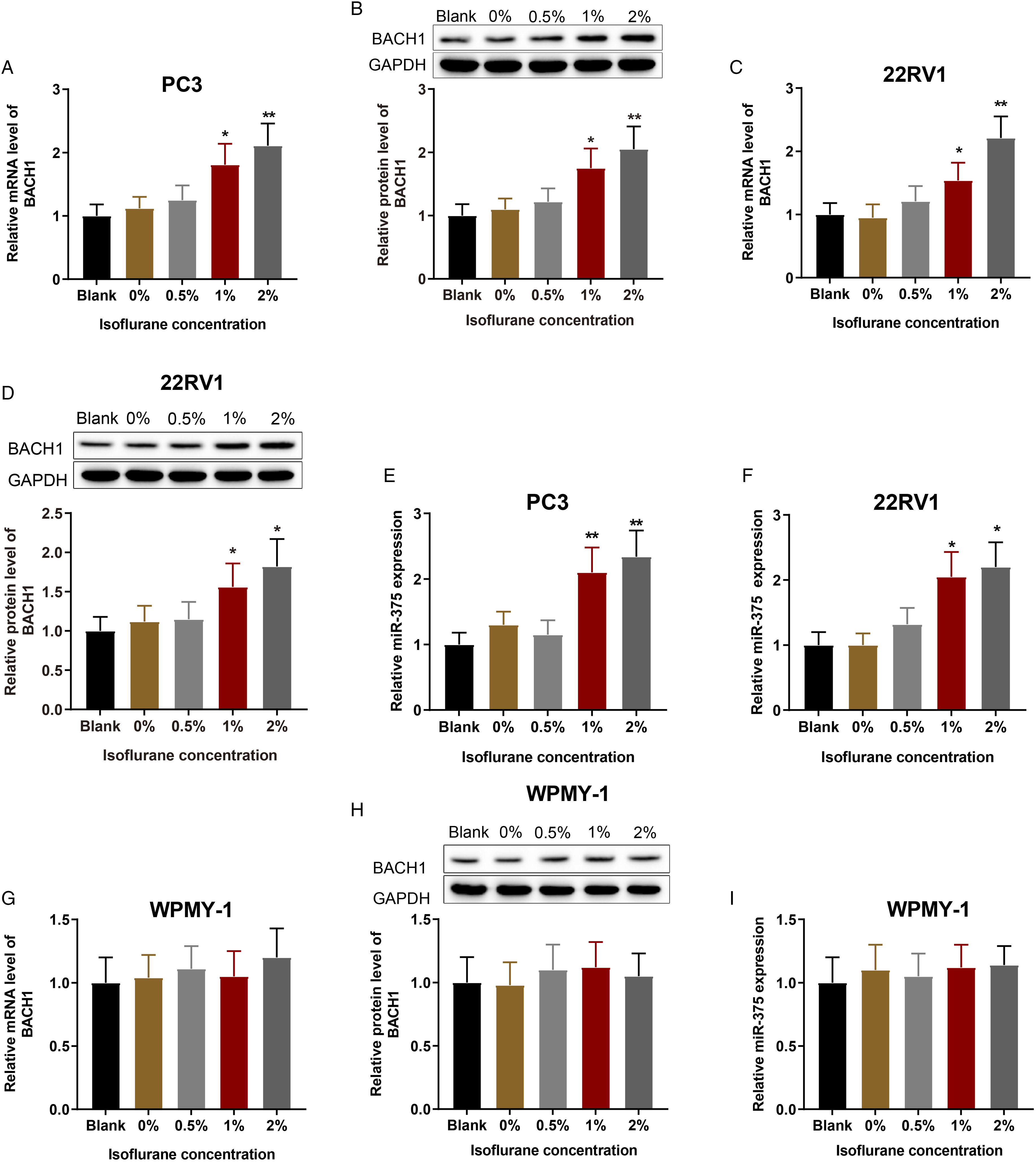
Isoflurane Upregulated BACH1 to Promote PC Cell Proliferation, Invasion, and Migration
To investigate the effects of BACH1 on PC cells, PC3 and 22RV1 cells were transfected with si-BACH1 and/or exposed to 2% isoflurane for 4 h. qRT-PCR and western blot analysis revealed that miR-375 and BACH1 expression were inhibited in the sh-BACH1 group (vs. the sh-NC group) but elevated in the Iso group (vs. the control group) (Figure 3A-C, P < 0.01). Moreover, miR-375 and BACH1 expression was effectively constrained in the Iso + sh-BACH1 group (vs. the Iso group) (Figure 3A-C, P < 0.01). These data suggested that isoflurane enhanced BACH1 expression and that inhibition of BACH1 diminished miR-375 expression. Isoflurane upregulates BACH1 to facilitate PC cell proliferation, invasion, and migration. After PC3 and 22RV1 cells were treated with sh-BACH1 or treated with 2% isoflurane, qRT-PCR was operated to determine BACH1 mRNA level (A); Western bot analysis was employed to measure BACH1 protein level (B); miR-375 expression was assessed by qRT-PCR (C); the proliferation abilities of PC3 cells (D) and 22RV1 cells (E) were probed with MTT assay; Transwell assay and cell scratch test were performed to measure PC3 and 22RV1 cell invasion (F-G) and migration (H-I). Values are expressed as mean ± standard deviation. *P < 0.05, **P < 0.01; PC, prostate cancer.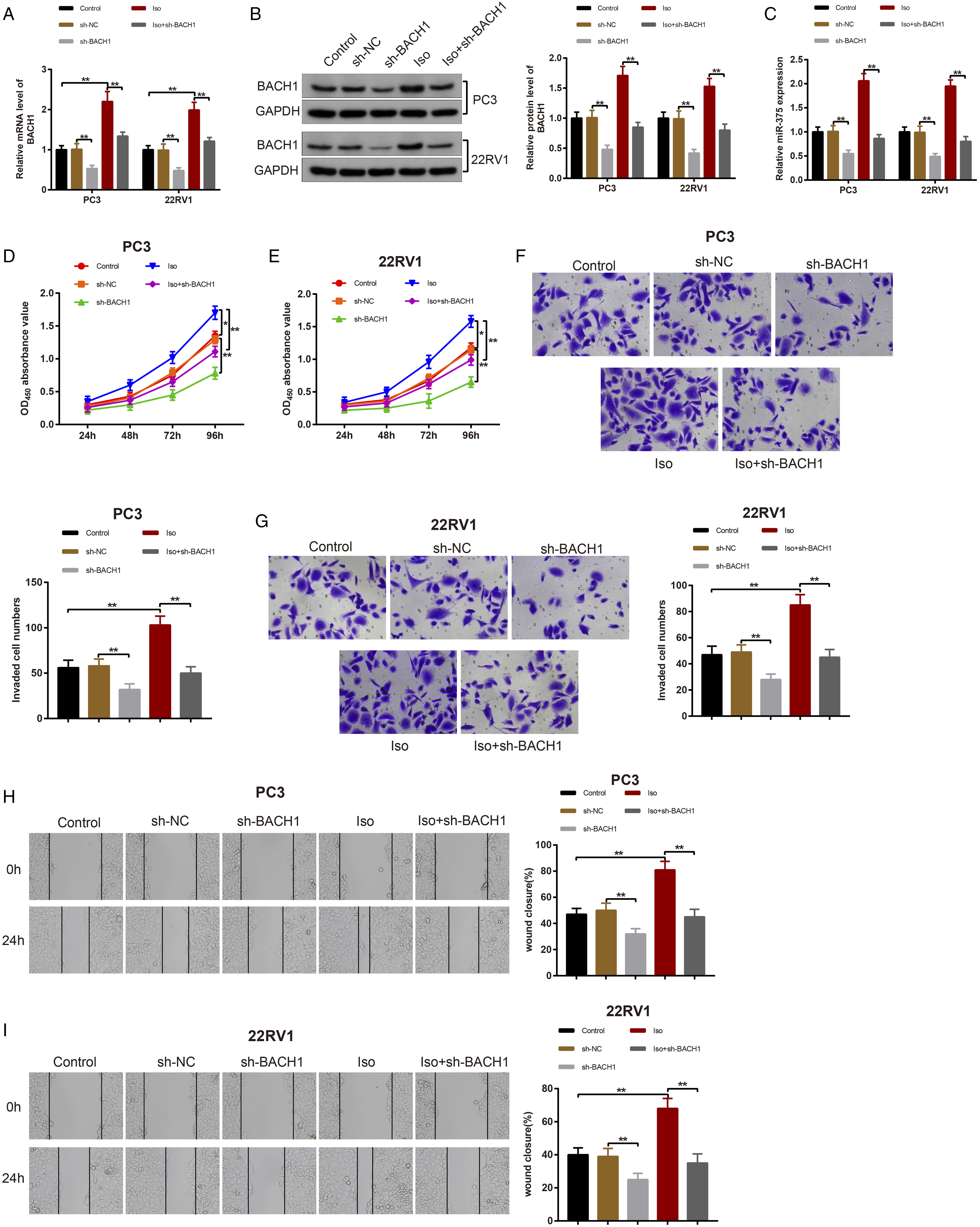
In addition, MTT assay showed that, in contrast to the sh-NC group, the sh-BACH1 group had decreased cell proliferation, while the Iso group had elevated cell proliferation (vs. the control group) (Figure 3D-E, P < 0.05). Importantly, the Iso + sh-BACH1 group had repressed cell proliferation in contrast to the Iso group (Figure 3D-E, P < 0.05). These results suggested that isoflurane increased PC3 and 22RV1 cell proliferation.
Furthermore, Transwell assay and cell scratch test revealed that cell invasive and migratory abilities were dramatically lowered in the sh-BACH1 group (vs. the sh-NC group), while there were significant increases of cell invasive and migratory abilities in the Iso group (vs. the control group) (Figure 3F-I, P < 0.01). However, the Iso + sh-BACH1 group showed obviously reduced cell invasive and migratory abilities (vs. the Iso group) (Figure 3F-I, P < 0.01). The aforementioned data manifested that isoflurane upregulated BACH1 expression to potentiate PC cell proliferative, invasive, and migratory capabilities.
Isoflurane upregulated miR-375 to stimulate PC cell proliferative, invasive, and migratory properties
To further ascertain the impacts of miR-375 on PC cells, miR-375 expression was inhibited in PC3 and 22RV1 cells, or PC3 and 22RV1 cells were treated with 2% isoflurane for 4 h. qRT-PCR results documented low expression of miR-375 in the miR-375 inhibitor group in contrast to the inhibitor NC group. Meantime, there was no considerable difference of BACH1 expression between the miR-375 inhibitor and inhibitor NC groups (Figure 4A-B, P < 0.05). In contrast to the control group, miR-375 and BACH1 expression was increased in the Iso group. However, miR-375 expression was repressed, whereas the mRNA level of BACH1 remained unchanged in the Iso + miR-375 inhibitor group (vs. the Iso group) (Figure 4A-B, P < 0.05), which revealed that isoflurane elevated miR-375 expression and that miR-375 inhibition had no obvious impact on BACH1 mRNA expression. Isoflurane upregulates miR-375 to accelerate PC cell proliferation, invasion, and migration. Note. After PC3 and 22RV1 cells were transfected with miR-375 inhibitor or treated with 2% isoflurane, miR-375 and BACH1 expression was detected by qRT-PCR (A-B); cell proliferation, invasion, and migration abilities were monitored by MTT (C-D), Transwell assay (E-F), and cell scratch test (G-H); the mRNA level of PTEN in PC3 cells (I) and 22RV1 cells (K) was measured by qRT-PCR; the protein level of PTEN in PC3 cells (J) and 22RV1 cells (L) was evaluated by western blot analysis. Values are expressed as mean ± standard deviation. ns, P > 0.05, *P < 0.05, **P < 0.01; PC, prostate cancer.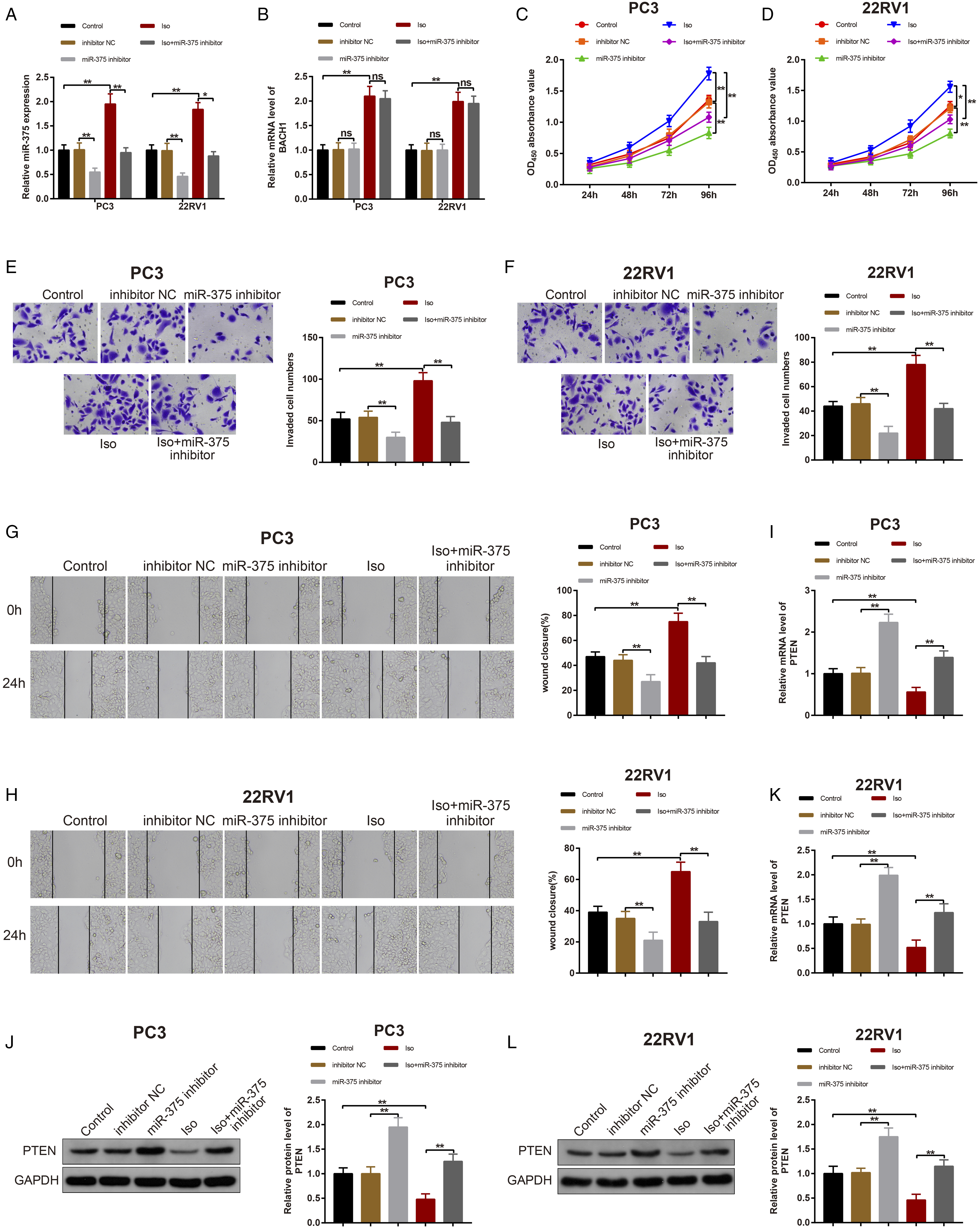
Next, MTT assay showed that cell proliferation was reduced in the miR-375 inhibitor group (vs. the inhibitor NC group) but increased in the Iso group (vs. the control group). However, cell proliferation was reduced in the Iso + miR-375 inhibitor group (vs. the Iso group) (Figure 4C-D, P < 0.05).
Additionally, Transwell assay and cell scratch test showed the decline of cell invasive and migratory abilities in the miR-375 inhibitor group (vs. the inhibitor NC group), but the enhancement of cell invasive and migratory abilities in the Iso group (vs. the control group) (Figure 4E-H, P < 0.01). Compared with the Iso group, cell invasive and migratory abilities were substantially reduced in the Iso + miR-375 inhibitor group (Figure 4E-H, P < 0.01).
qRT-PCR and western blot analysis revealed that PTEN expression was elevated in the miR-375 inhibitor group (vs. the inhibitor NC group) but reduced in the Iso group (vs. the control group). In addition, PTEN expression was increased in the Iso + miR-375 inhibitor group in contrast to the Iso group (Figure 4I-L, P < 0.01). All of these findings suggested that isoflurane accelerated PC cell proliferation, invasion, and migration by upregulating miR-375.
BACH1 Targeted miR-375 and miR-375 Negatively Regulate PTEN Expression
To determine the potential mechanism underlying miR-375 upregulation in PC cells, a bioinformatics method (TransmiR v2.0 database) was used to predict transcription factors regulating miR-375. The results showed a binding site between BACH1 and miR-375) (Figure 5A-B). Meanwhile, the binding of BACH1 and miR-375 was verified by ChIP assay, which showed that the complexes pulled down by BACH1 antibody were enriched miR-375 promoter (Figure 5C, P < 0.001). In addition, the designed mutation sites and the predicted binding sites between BACH1 and miR-375 are listed in Figure 5D. To verify their targeting relationship, the luciferase reporter vectors of WT-miR-375 and MUT-miR-375 were constructed. The detection revealed that sh-BACH1 obviously decreased the luciferase activity in HEK-293T cells transfected with WT-miR-375 (vs. the sh-NC group) (Figure 5E, P < 0.01). However, sh-BACH1 failed to affect the luciferase activity in HEK-293T cells transfected with MUT-miR-375 (Figure 5E). These findings corroborated the targeting relationship between BACH1 and miR-375. BACH1 targets miR-375 and miR-375 inversely correlates to PTEN. TransmiR v2.0 predicted that BACH1 was one of the transcription factors of miR-375 (A); the binding sites of miR-375 and BACH1 were predicted (B); the binding of BACH1 to miR-375 was verified by ChIP assay (C); TransmiR v2.0 predicted the binding sites between miR-375 and BACH1 in addition to the designed mutation sites of miR-375 (D). After HEK-293T cells were co-transfected with sh-NC/sh-BACH1 and WT-miR-375/MUT-miR-375, dual-luciferase reporter assay was used to measure the luciferase activity (E). After PC3 and 22RV1 cells were transfected with mimic NC, miR-375 mimic, inhibitor NC, or miR-375 inhibitor, the expression of miR-375 was quantified by qRT-PCR (F); the mRNA and protein levels of PTEN were observed by qRT-PCR and western blot analysis (G-H); the binding sites between miR-375 and PTEN as well as the designed mutation sites of PTEN were predicted by Starbase (I). After HEK-293T cells were co-transfected with mimic NC, miR-375 mimic, inhibitor NC, or miR-375 inhibitor and with WT-PTEN/MUT-PTEN, the luciferase activity was detected with dual-luciferase reporter assay (J). Values are expressed as mean ± standard deviation. ns, P > 0.05, **P < 0.01, ***P < 0.001.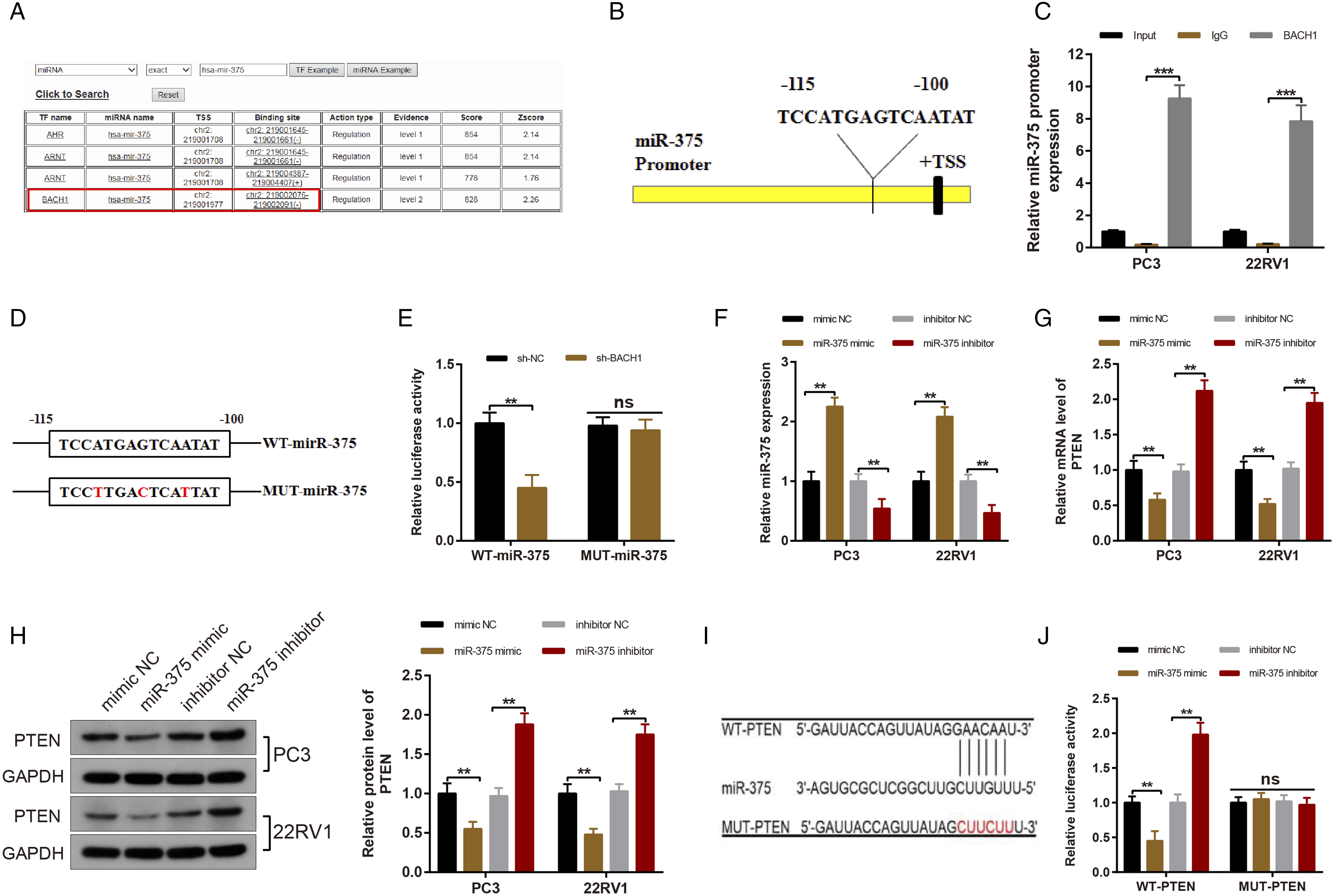
The online software Starbase predicted that there were binding sites between miR-375 and PTEN. To validate whether miR-375 negatively targeted PTEN, miR-375 activity was mimicked or inhibited in PC3 and 22RV1 cells. qRT-PCR and western blot analysis evidenced that PTEN expression prominently diminished in the miR-375 mimic group (vs. the mimic NC group), but effectively increased in the miR-375 inhibitor group (vs. the inhibitor NC group), indicating that miR-375 reversely manipulated PTEN expression (Figure 5F-H, P < 0.01). Moreover, the designed mutation sites and the binding sites of miR-375 in the 3’untranslated region of PTEN were predicted via the online software Starbase, as depicted in Figure 5I. To confirm their targeting relationship, the luciferase reporter vectors containing the WT PTEN 3’UTR (WT-PTEN) and MUT PTEN 3’UTR (MUT-PTEN, containing 6 mutant sites of binding sites to miR-375) were constructed. The luciferase activity was clearly reduced after co-transfection with WT-PTEN and miR-375 mimic (vs. the mimic NC group) but appreciably elevated after co-transfection with WT-PTEN and miR-375 inhibitor (vs. the inhibitor NC group) (Figure 5J, P < 0.01). However, the luciferase activity remained unaffected after HEK-293T cells were co-transfected with MUT-PTEN and miR-375 mimic or miR-375 inhibitor (Figure 5J). These findings illustrated that miR-375 inversely targeted PTEN.
Isoflurane Enhanced PC Cell Proliferative, Invasive, and Migratory Properties by Upregulating BACH1 and miR-375
To determine whether isoflurane-induced PC cell proliferation, invasion, and migration by activating BACH1 and upregulating miR-375, PC3 cells were treated with 2% isoflurane for 4 h before transfection or co-transfection with sh-NC, sh-BACH1, mimic NC, or miR-375 mimic. qRT-PCR and western blot analysis exhibited that BACH1 and miR-375 expression was intensively augmented in the Iso group (vs. the control group), whereas BACH1 and miR-375 expression was evidently lowered in the Iso + sh-BACH1 group (vs. the Iso + sh-NC group) (Figure 6A-C, P < 0.01). Compared with the Iso + sh-BACH1 + mimic NC group, there was no notable change with respect to BACH1 expression miR-375 expression was enhanced dramatically in the Iso + sh-BACH1 + miR-375 mimic group (Figure 6A-C, P < 0.01). There was no detectable difference in miR-375 expression between the Iso and Iso + sh-NC groups and between the Iso + sh-BACH1 and Iso + sh-BACH1 + mimic NC groups (Figure 6A-C, P < 0.01). Isoflurane facilitates PC cell proliferation, invasion, and migration through upregulation of BACH1 and miR-375. After PC3 cells were treated with 2% isoflurane 4 h followed by transfection/co-transfection with sh-NC, sh-BACH1, mimic NC or miR-375 mimic, BACH1 expression was monitored by qRT-PCR and western blot analysis (A-B). The expression of miR-375 was assessed by qRT-PCR (C). The proliferation, invasion, and migration abilities were probed with MTT, Transwell assay, and cell scratch test (D-F). Values are expressed as mean ± standard deviation. ns, P > 0.05, *P < 0.05, **P < 0.01; PC, prostate cancer.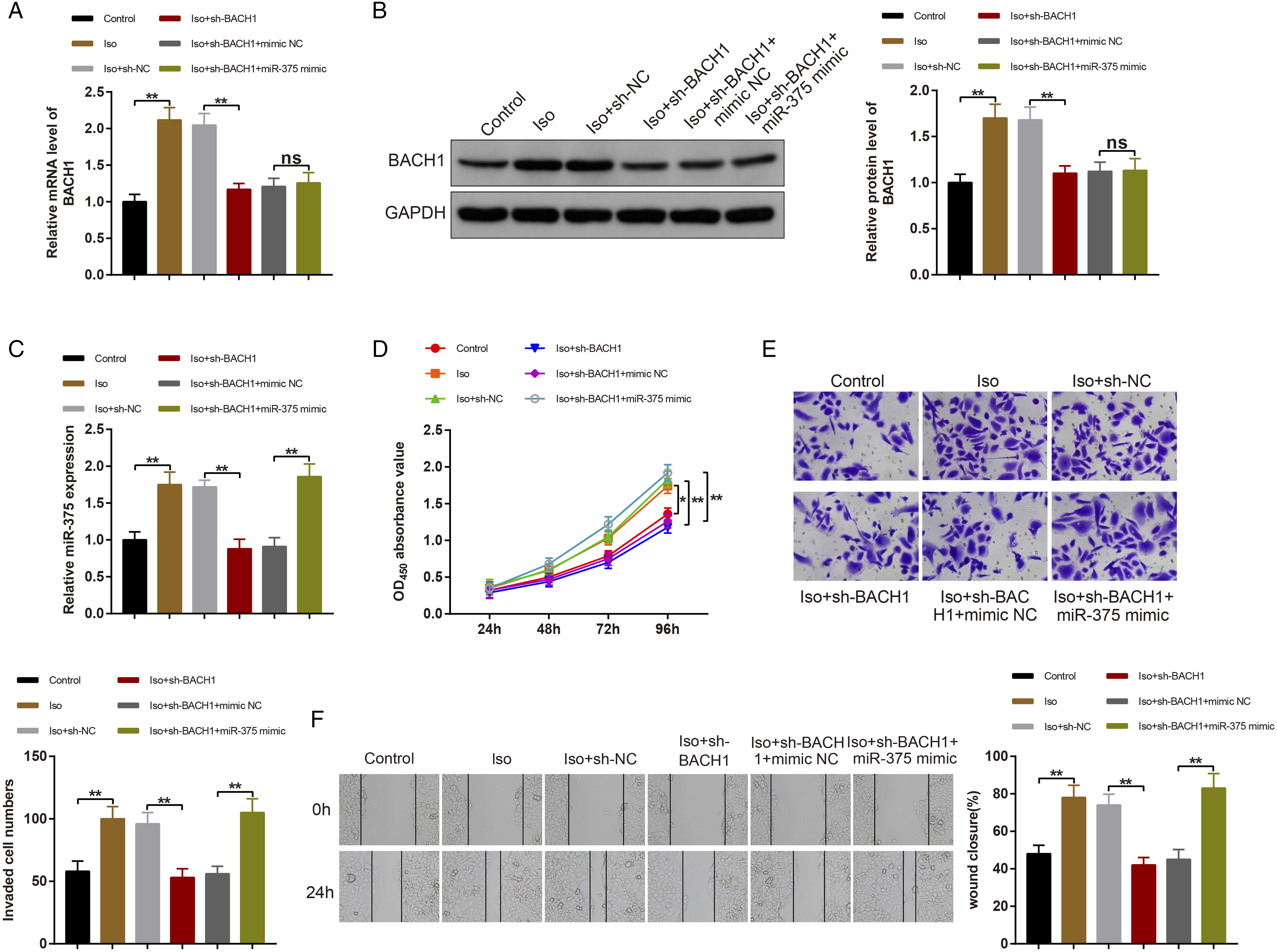
MTT assay revealed that cell proliferation ability was greatly increased in the Iso group (vs. the control group) and the Iso + sh-BACH1 + miR-375 mimic group (vs. the Iso + sh-BACH1 + mimic NC group) while decreased in the Iso + sh-BACH1 group (vs. the Iso + sh-NC group) (Figure 6D, P < 0.01). These data indicated that silencing BACH1 impeded PC cell proliferation caused by isoflurane, while overexpression of miR-375 could partially attenuate the inhibitory impacts of BACH1 downregulation on PC cell proliferation.
Additionally, Transwell assay and cell scratch test showed that cell invasive and migratory properties were distinctly enhanced in the Iso group (vs. the control group) and the Iso + sh-BACH1 + miR-375 mimic group (vs. the Iso + sh-BACH1 + mimic NC group), which was contrary in the Iso + sh-BACH1 group (vs. the Iso + sh-NC group) (Figure 6E-F, P < 0.01). These data showed that BACH1 silencing suppressed invasion and migration of PC cells treated by isoflurane, while these effects were partially blocked by miR-375 overexpression. Altogether, isoflurane activated BACH1 to upregulate miR-375, thus accelerating PC cell proliferation, invasion, and migration.
Discussion
The present study provided evidence that isoflurane promoted the proliferation, migration, and invasion of PC cells in vitro, which was mediated by upregulating BACH1 and miR-375. In addition, we revealed that isoflurane activated BACH1 to mediate miR-375/PTEN axis, thus promoting the proliferation, migration, and invasion in PC.
The effects of isoflurane on cancer cell proliferation are less well characterized. Isoflurane was reported to significantly inhibit the proliferation and survival of cervical cancer cells. 39 However, isoflurane was reported to accelerate cell proliferation in the study of bladder cancer cells. 40 In the present study, we revealed that isoflurane promoted PC cell proliferation. However, the underlying mechanism by which isoflurane regulates PC progression remains unclear.
BACH1, a transcription regulatory protein, is widely expressed in various tissues of mice and humans. 41 Reportedly, silencing of BACH1 in PC cells can suppress migration and invasion by altering the expression of metastasis-related genes. 42 However, the interaction between BACH1 and isoflurane in PC needs to be further elucidated. Here, we first described that the BACH1 is expressed more commonly in isoflurane-treated PC cells, suggesting that isoflurane exposure caused a marked increase of BACH1 expression in PC cells. It is well known that the abnormal proliferative ability is one of the characteristics distinguishing tumor cells from normal ones. 11 BACH1 is associated with tumor metastasis and progression, 43 but the potential mechanisms contributing to the tumor effects of BACH1 are largely undefined. Here, MTT assay showed that BACH1 silencing could reduce cell proliferation, and knockdown of BACH1 abrogated the proliferative effects of isoflurane on PC cells, supporting this as a mechanism. Migration and invasion are two main processes of malignant tumors, usually leading to tumor-related death. 44 Thereafter, Transwell and cell scratch assays showed that BACH1 silencing could reduce cell invasion and migration abilities. Moreover, downregulation of BACH1 counteracted the effects of isoflurane on PC cells. Together, isoflurane increases BACH1 expression to promote cell proliferative, invasive, and migratory properties in PC.
Next, we investigated the mechanism underlying the effects of isoflurane and BACH1 in modulating PC progression. Previous work disclosed that miR-375 is downregulated in a variety of cancers, 45 and therefore, miR-375 is considered to be a key tumor suppressor by targeting important oncogenes and regulating multiple biological activities including cancer cell invasion, migration, proliferation, autophagy, metastasis, and apoptosis.46-49 However, miR-375 is also reported to be upregulated in some cancer types including PC and higher expression of miR-375 is associated with advanced PC stage. 50 Moreover, isoflurane could enhance the invasion and proliferation of cervical carcinoma cells by downregulating miR-375 expression. 51 Our study firstly recognized the binding sites of BACH1 on the miR-375 promoter, which uncovered a new BACH1 effect during PC tumorigenesis. Here, miR-375 and BACH1 expression were elevated in isoflurane-treated PC cells, which showed that isoflurane could increase miR-375 expression. Moreover, the levels of miR-375 were inhibited after isoflurane and sh-BACH1 co-treatment, suggesting the synergic effects of isoflurane and BACH1 silencing could repress miR-375 expression. Previously, uncontrolled cell proliferation is the primary feature of cancer. 52 Mounting studies corroborated that miR-375 could stimulate PC cell invasion and proliferation.53,24 Interestingly, a series of experiments showed that silencing miR-375 expression decreased PC cell proliferation, invasion, and migration abilities. Moreover, miR-375 knockdown restricted isoflurane-promoted cell proliferation, invasion, and migration abilities. These results suggested that isoflurane could increase miR-375 expression and play a critical role in PC cell proliferation, invasion, and migration.
To deeper ascertain the relationship between BACH1 and miR-375 in PC cells, our findings revealed the targeting relationship between BACH1 and miR-375. Next, we deciphered the mechanism of BACH1 and miR-375 regarding isoflurane in regulating PC cell proliferative, invasive, and migratory abilities. PTEN, a tumor suppressor, has been found to control cell proliferation, survival, and apoptosis.54,55 In patients with nasopharyngeal carcinoma, downregulation of PTEN leads to tumor cell metastasis and invasion. 56 The association between miR-375 and PTEN has also been revealed. 57 It was confirmed in our study that BACH1 targeted miR-375 to regulate PTEN expression. Thereafter, we ascertained whether isoflurane potentiated PC cell proliferative, invasive, and migratory capabilities by upregulating BACH1 and miR-375 expressions. The detection unraveled that isoflurane increased BACH1 and miR-375 expressions. Moreover, BACH1 knockdown significantly perturbed the promotion effects of isoflurane on cell proliferative, invasive, and migratory properties. Furthermore, overexpression of miR-375 mitigated the inhibition of BACH1 knockdown on PC cell proliferative, invasive, and migratory abilities. Based on these results, our results highlighted that isoflurane aggravated PC cell proliferative, invasive, and migratory capabilities by increasing BACH1 and miR-375 expression.
Conclusions
In conclusion, our study suggests that BACH1 could be a novel target that may aid with the future development of therapies for aggressive PC. Importantly, the BACH1/miR-375/PTEN axis as a novel mechanism has a vital role in studying the tumor-promoting effects of isoflurane on PC growth.
Footnotes
Author Contributions
Zhang, J. substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, drafted manuscript, and critically revised manuscript; Chen, G. contributed to acquisition, analysis, or interpretation of data, drafted manuscript, and critically revised manuscript; Li, T. contributed to acquisition, analysis, or interpretation of data and critically revised manuscript; He, X. contributed to acquisition, analysis, or interpretation of data and drafted manuscript; Luo, Y. contributed to acquisition, analysis, or interpretation of data and drafted manuscript; Ke, Y. substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Foundation of Education Bureau of Hunan Province (Grant No.20C1154), the Scientific Research Foundation of Administration of Traditional Chinese Medicine of Hunan Province (Grant No.2021041) and the Guiding Project of Clinical Medical Technology Innovation in Hunan Province (Grant No.2020SK50901).
