Abstract
Objective
To investigate the diagnostic efficiency of miR-222–3p in plasma exosomes (Exos) and plasma for preeclampsia (PE) and the effect of miR-222–3p targeting STMN1 in PE
Methods
MiR-222–3p levels in total plasma and plasma Exos were detected in PE patients and healthy controls. A bioinformatics database and dual-luciferase reporter assay were employed to verify the targeting relationship between miR-222–3p and STMN1. Trophoblast HTR-8/Svneo cells were transfected with miR-222–3p inhibitors with/without STMN1 shRNA, followed by MTT, wound healing and Transwell invasion assays. The mRNA and protein expressions were measured by qRT‒PCR and Western blotting, respectively.
Results
MiR-222–3p levels in total plasma and plasma Exos were higher in PE patients than in healthy controls, particularly in severe PE patients. In addition, miR-222–3p levels in total plasma and plasma Exos from PE patients were positively correlated with diastolic and systolic blood pressure. The area under the curve (AUC) of miR-222–3p in total plasma for PE diagnostic efficiency was 0.798, with a sensitivity of 76.67% and specificity of 71.93%, while the AUC of miR-222–3p in plasma Exos was 0.708 (sensitivity: 61.67%; specificity: 78.95%). In vitro, miR-222–3p targeted STMN1 in HTR-8/Svneo cells. Low miR-222–3p expression reversed the inhibitory effect of STMN1 shRNA on the proliferation, invasion and migration of HTR/SVneo cells.
Conclusion
PE patients had increased miR-222–3p expression in total plasma and plasma Exos, which both have high diagnostic efficiency for PE. MiR-222–3p can target STMN1 to promote the proliferation, invasion and migration of HTR-8/Svneo cells and is a potential therapeutic target of PE.
Introduction
Preeclampsia (PE) is a pregnancy-specific complication characterized by arterial hypertension with organ diseases, such as proteinuria, thrombocytopenia, renal and liver failure, and in severe cases, it can lead to eclampsia with brain complications. 1 The incidence of PE is approximately 3–8% among all pregnancies, and PE is the main cause of perinatal morbidity and mortality, accounting for approximately 16–18% of all maternal deaths and approximately 40% of fetal and neonatal deaths. 2 Usually, in early pregnancy, extravillous trophoblasts participate in the physiological expansion of the uterine spiral artery. Spiral artery remodeling and the impairment of their invasive ability can lead to spiral artery remodeling disorder and placental ischemia and hypoxia, ultimately causing PE. 3 In recent years, great progress has been made in understanding PE, but its pathogenesis and mechanism are still unknown. Thus, fully understanding the pathogenesis of PE is of great importance for the diagnosis, prevention, treatment and early intervention of PE and the reduction of perinatal morbidity and mortality. 4
Exosomes are a class of small extracellular vesicles (sEVs) secreted by cells, with diameters of 30–200 nm, and they are rich in proteins, DNA, mRNA and microRNAs. 5 Recently, investigating the prevalence, expression and possible clinical use of miRNA-containing Exos during human pregnancy has become a hot spot. 6 MiRNAs are a class of RNAs consisting of 22 nucleotides and are widely distributed in plasma and other body fluids. Because miRNAs are very stable and easy to detect, they have been recognized for their potential as novel biomarkers in diseases, including PE.1,6 For instance, the levels of miR-210, miR-136, miR-494 and miR-495 were significantly upregulated in exosomes of peripheral blood from patients with PE compared to normal pregnancies, and these miRNAs showed significant, moderate to strong, positive correlations with the clinical parameters of PE, including SBP, DBP, cholesterol level and HDL level, indicating that such expression patterns could serve as important noninvasive biomarkers for the early detection of PE. 7 Eric J. Devor et al. reported statistically significant differential expression of 14 miRNAs in plasma Exos between women with PE and those without PE. 6 The plasma exosomal miRNA hsa-miR-222-3p has been found to be abnormally expressed in many diseases, including primary immune thrombocytopenia, 8 adriamycin-resistant breast cancer, 9 and lymph node metastasis in papillary thyroid carcinomas. 10 Here, we investigated the diagnostic efficiency of miR-222-3p in total plasma and plasma Exos.
miRNAs can act on the 3’ UTR of target mRNA to induce the degradation to translation inhibition of mRNA, thus regulating gene expression at a posttranscriptional level. 1 Based on the analysis of many online bioprediction websites, miR-222-3p contains a binding site for the Stathmin 1 (STMN1) 3’ UTR. As a member of the stathmin family, STMN1 can encode a ubiquitous cytosolic phosphoprotein and is regarded as an intracellular relay integrating regulatory signals of the cellular environment. 11 STMN1 is related to recurrent miscarriage 12 and pregnancy outcomes. 13
In this study, human trophoblast HTR-8/Svneo cells in vitro were transfected with miR-222-3p inhibitors with/without STMN1 shRNA to investigate whether miR-222-3p can target STMN1 to play a regulatory role in PE.
Materials and methods
Ethics statement
All participants were well informed of the experiment and signed the consent form prior to the study. The study was approved by the Ethics Committee of our hospital.
Recruited participants
Inclusion criteria for PE patients (n = 60) were based on concomitant symptoms, including high blood pressure with two or more entries of diastolic blood pressure (DBP) of ≥90 mmHg taken ≥4 h apart and proteinuria with the secretion of ≥300 mg of protein over 24 h after 20 weeks of gestation. 14 Exclusion criteria included chronic hypertension, (gestational) diabetes mellitus, polycyesis, fetal anomaly, hypercholesterolemia, chronic kidney disease, autoimmune disorder, connective tissue disease or use of acetylsalicylic acid. According to the guidelines of the American College of Obstetricians and Gynecologists (ACOG) in 2020, 15 PE patients were divided into a mild PE group and a severe PE group. Severe PE was defined as severe gestational hypertension, with systolic blood pressure (SBP) ≥160 mmHg or DBP ≥110 mmHg on two occasions at least 4 hours apart. Proteinuria was detected and quantified on a 24-h urine collection. Healthy pregnant women (n = 57) were recruited as healthy controls, without any pregnancy complications, such as artificial insemination, threatened abortion, premature rupture of membranes and/or premature birth, placenta previa and macrosomia.
Plasma sample collection and Exo isolation
We collected 10 mL of blood in Na-EDTA tubes during routine blood testing, which was performed when each participant was admitted for labor and delivery at our hospital. Blood samples were centrifuged for 10 min at 1500 g at 4°C, and the supernatant was centrifuged again under the same conditions. Plasma was preserved at −80°C. An exosome isolation kit was used to separate human plasma Exo (4,484,450, Thermo Fisher Scientific Inc., China) following the manufacturer’s protocol. In brief, the blood sample was bathed in 37°C water until it was completely liquid and placed on ice. The plasma sample was centrifuged for 20 min at 2000 g, and the supernatant was centrifuged again for 20 min at room temperature, followed by Exo isolation with proteinase treatment. In this study, flow cytometry, electron microscopy or other technology was not used to check the quality of isolated Exos because we were not interested in analyzing the exosome subpopulations present in the samples or studying exosome function.
Dual-luciferase reporter assay
Human trophoblast HTR-8/Svneo cells (Catalog No. CRL-3271) were purchased from ATCC (USA) and cultured in RPMI-1640 medium (Catalog No. 30–2001, ATCC, USA) supplemented with fetal bovine serum (FBS) at a final concentration of 5%. A sequence fragment that contains binding sites for both wild-type (WT) and mutant (MUT) STMN1 3’UTR was designed and inserted into luciferase expression vectors to construct the STMN1-WT plasmid and STMN1-MUT plasmid. Lipofectamine 3000 was used to mix miR-NC (Catalog No. 4,464,058, ThermoFisher Scientific, USA), miR-222-3p mimics (Catalog No. 4,464,066, ThermoFisher Scientific, USA) and miR-222-3p inhibitors (Catalog No. 4,464,084, ThermoFisher Scientific, USA) with STMN1-WT plasmid or STMN1-MUT plasmid, which were transfected into HTR-8/Svneo cells. After transfection for 48 h, luciferase activity was detected following the instructions of the kit, and the ratio of firefly luciferase activity to Renilla luciferase activity was used for statistical analysis.
Cell grouping
HTR-8/Svneo cells were divided into six groups: a mock group (cells without transfection), miR-NC group (cells transfected with miRNA negative control), miR-222-3p inhibitor group (cells transfected with miR-222-3p inhibitors), NC shRNA group (cells transfected with NC shRNA, Catalog No. sc-108,060, Santa Cruz Biotechnology, Inc., USA), STMN1 shRNA group (cells transfected with STMN1 shRNA, Catalog No. sc-36,127, Santa Cruz Biotechnology, Inc., USA), and miR-222-3p inhibitor + STMN1 shRNA group (cells cotransfected with miR-222-3p inhibitors and STMN1 shRNA). The liposome method was used for transfection according to the instructions of Lipofectamine 3000 Transfection Reagent (Catalog No. L3000015, Invitrogen, USA). Subsequent experiments were conducted after transfection for 24 h.
Real-time quantitative polymerase chain reaction (qRT‒PCR)
After Exos were isolated, total RNA was extracted using a Total Exosome RNA Isolation Kit (4,478,545, Thermo Fisher Scientific Inc., China). The TRIzol method was used to extract total RNA from HTR-8/Svneo cells and plasma total RNA. SuperScript IV reverse transcriptase (Catalog # 18,090,010, Invitrogen, USA) was used for reverse transcription of RNA into cDNA. Appropriate amounts of cDNA were employed as templates for qRT‒PCR with a SuperScript III Platinum SYBR Green One-Step qRT‒PCR Kit (Catalog # 11,736,059, Invitrogen, USA) using U6 and GAPDH as loading controls.
Western blotting
Total proteins were extracted from cells, and the protein concentration was determined with the BCA method. Next, 20 μg of protein sample was loaded for separation via SDS‒PAGE. Proteins were transferred to PVDF membranes and blocked for 2 h in 5% defatted milk at room temperature. Primary antibodies were added overnight reaction at 4°C, including anti-STMN1 antibody (ab119991) at 1/5000 dilution, anti-MMP2 antibody (ab92536) at 1/1000 dilution, anti-MMP9 antibody (ab76003) at 1/200 dilution, and anti-β-actin antibody (ab8227) at 1 µg/ml. Then, the membrane was washed with TBST, followed by the addition of goat anti-rabbit IgG H&L (HRP) at a 1/1000 dilution for 2 h of incubation at room temperature. After another round of membrane washing, ECL was performed in a dark room for development. ImageJ software was used to analyze the gray values of the protein bands. All antibodies were purchased from Abcam (USA).
MTT Assay
Cells (1 × 104) were seeded into 96-well plates. After 24 h, MTT (Sigma‒Aldrich, St. Louis, MO, USA) (final concentration 5 mg/mL) was added for incubation at 37°C with 5% CO2 for 3 h and removed. MTT formazan crystals were dissolved in 150 μL of DMSO. Absorbance at 570 nm was determined with an automated microplate reader (BioTekSynergy HT, Watertown, MA, USA).
Wound healing
Cells in good condition were inoculated into 6-well plates and cultured in serum-free and antibody-free medium for 24 h until cell confluence reached approximately 80%. A pipette (200 μL) tip was used to evenly draw horizontal lines across the middle of the 6-well plate. The plate was rinsed three times with PBS buffer to remove broken cells. Markers were made at the bottom of the plate for subsequent observation. Next, serum-free and antibody-free medium was added for continued culture, and photos were taken at 0 h and 24 h. Software was used to analyze the migration of cells.
Transwell invasion assay
Matrigel was thawed at 4°C and diluted with PBS 1:8 on ice. Transwell chambers were placed in 24-well plates, which were covered with 50 μL of diluted Matrigel and cultured in an incubator for 2–3 h until Matrigel solidification. In the upper chamber of the Transwell system, 4 groups of cell suspensions (density 1 × 105 cells/mL) (200 μL) were added, while 10% FBS-containing RPMI 1640 medium was added to the lower chamber for 24 h of incubation at 37°C with 5% CO2. Next, the membrane was washed, and the cells were fixed in formaldehyde for 30 min and stained with crystal violet for 5 min before observation under a microscope. Six visual fields (×200) were randomly selected for observation, and migrated cells were counted. The experiment was repeated three times to calculate the mean value of cell numbers in each chamber.
Statistical methods
The statistical software SPSS 19.0 and GraphPad Prism were used for data analysis. Measurement data are presented as the mean ± standard deviation. Intergroup comparisons were tested by Student’s t test, while multiple-group comparisons were analyzed by one-way ANOVA and tested by Tukey’s test. Enumeration data are presented as counts and rates, with intergroup comparisons tested by the chi-square test (χ2). Correlation analysis was performed with Pearson correlation coefficients. Receiver operating characteristic (ROC) curves were used to analyze the sensitivity and specificity of disease diagnosis. p < .05 indicates a statistically significant difference.
Results
Effect of miR-222-3p in total plasma and plasma Exos on the diagnostic efficiency of PE
General clinical data comparison between PE patients and healthy controls.


Effect of miR-222-3p in total plasma and plasma Exos on the diagnostic efficiency of PE.
Correlation of miR-222-3p levels in total plasma and plasma Exos with the severity and blood pressure of PE patients
Compared with severe PE patients, mild PE patients had decreased miR-222-3p levels in total plasma and plasma Exos (both p < .05, Figures 2(A) and (B)). We analyzed the correlation of miR-222-3p expression in total plasma and plasma Exos with SBP and DBP in PE patients (Figures 2(C) and (D)). Plasma miR-222-3p levels were positively correlated with SBP (r = 0.325, p = .011) and DBP (r = 0.351, p = .006), and miR-222-3p expression in plasma Exos was also positively correlated with SBP (r = 0.525, p < .001) and DBP (r = 0.273, p = .035) in PE patients. Correlation of miR-222-3p levels in total plasma and plasma Exos with the severity and blood pressure of PE patients. 
miR-222-3p can target STMN1 to regulate its expression
Multiple databases, including TargetScan,
16
miRDB.,
17
ENCORI
18
and DIANA tools-Tarbasev8,
19
were used to analyze the target gene of miR-222-3p. Based on the analysis, STMN1 (predicted relative KD in TargetScan: −5.119, predictive score in DIANA tools-TarBase v8: 0.905) was selected (Figure 3(A)). The binding site between miR-222-3p and the STMN1 3’ UTR is shown in Figure 3(B). According to the results of the dual-luciferase reporter assay (Figure 3(C)), compared with transfection with NC mimics, cotransfection of miR-222-3p mimics and STMN1-WT significantly reduced the luciferase activity of HTR-8/Svneo cells, while cotransfection of miR-222-3p inhibitors and STMN1-WT led to an apparent increase in the luciferase activity of HTR-8/Svneo cells (both p < .05), which indicates that miR-222-3p can target STMN1 to regulate its expression. Additionally, miR-222-3p inhibitors substantially downregulated miR-222-3p expression in HTR/SVneo cells but enhanced STMN1 gene and protein expression (all p < .05). Moreover, the inhibitory effect of STMN1 shRNA on STMN1 in HTR/SVneo cells was reversed by miR-222-3p inhibitors (p < .05), which indicates that miR-222-3p can negatively regulate STMN1 expression in HTR/SVneo cells (Figures 3(D) to (F)). miR-222-3p can target STMN1 to regulate its expression 
miR-222-3p targets STMN1 to inhibit the proliferation, invasion and migration of HTR/SVneo cells
Through the MTT assay (Figure 4(A)), we found that low miR-222-3p expression significantly promoted the proliferation of HTR/SVneo cells (p < .05). The inhibitory effect of low STMN1 expression on HTR/SVneo cell proliferation could be reversed by low miR-222-3p expression. In addition, decreased proliferation rates due to STMN1 downregulation may lead to decreased migration/invasion of trophoblast cells. Here, we used wound healing (Figures 4(B) and (C)) and Transwell assays (Figures 4(D) and (E)) to detect the migration and invasion of cells. According to the results, compared with the miR-NC group, HTR/SVneo cells in the miR-222-3p inhibitor group showed dramatically increased migration and invasion (all p < .05). Additionally, compared with the STMN1 shRNA group, HTR/SVneo cells in the miR-222-3p inhibitor+STMN1 shRNA group showed significantly increased invasion and migration abilities (all p < .05). miR-222-3p targets STMN1 to inhibit the proliferation, invasion and migration of HTR/SVneo cells 
miR-222-3p targets STMN1 to affect MMP-2/9 expression in HTR/SVneo cells
Furthermore, we analyzed the expression levels of MMP-2/9 involved in the promotion and inhibition of cell migration and invasion (Figure 5). According to the qRT‒PCR results, low STMN1 expression could inhibit the expression of MMP-2/9 in HTR/SVneo cells. Compared with the STMN1 shRNA group, HTR/SVneo cells in the miR-222-3p inhibitor+STMN1 shRNA group had significantly increased MMP-2/9 expression. Subsequent Western blotting revealed that MMP-2/9 protein expression was the same as MMP-2/9 mRNA expression. miR-222-3p targets STMN1 to affect MMP-2/9 expression in HTR/SVneo cells Note: A-B, MMP-2 (A) and MMP-9 (B) expression in HTR/SVneo cells measured by qRT‒PCR; C-E, Protein expression of MMP-2/9 in HTR/SVneo cells detected by Western blotting. * Compared with the mock and miR-NC groups, p < .05; # compared with the miR-222-3p inhibitor group, p < .05; & compared with the NC shRNA group, p < .05; @ compared with the STMN1 shRNA group, p < .05.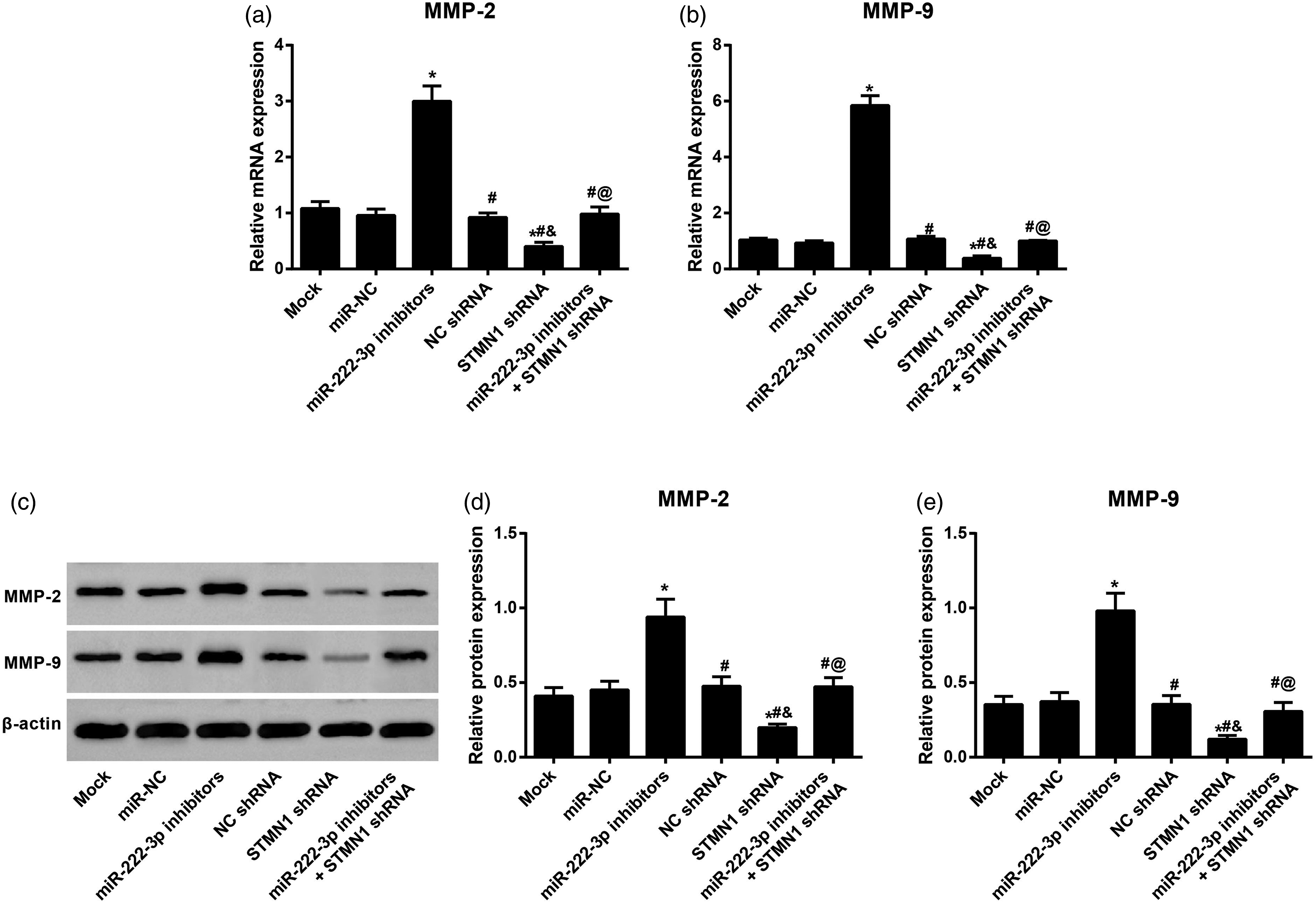
Discussion
In the current study, miR-222-3p expression was significantly upregulated in the plasma of PE patients, especially in the plasma of severe PE patients. In a previous study, miR-222-3p was found to be upregulated in the placentas of PE patients and in primary placental mesenchymal stem cells (MSCs) under hypoxia. 20 Women with severe PE showed an appreciable increase in miR-222-3p in their placentas. 21 Héctor Ojeda-Casares et al. 22 and Malia Su-Qin Murphy et al. 23 also reported that compared with the control group in the second and third trimesters of pregnancy, patients in the PE period had overexpression of miR-222-3p in plasma. In addition, we found that the sensitivity and specificity of plasma miR-222-3p for PE diagnosis were 78.57% and 70%, respectively, which suggests that miR-222-3p has a pathogenic effect in PE and that plasma miR-222-3p levels have a high diagnostic efficiency for PE. As an important medium of intercellular communication, extracellular vesicles (EVs) are continuously secreted by cells as smaller (30–200 nm) or larger (micron size) particles. 24 Although these vesicles are all called EVs, smaller vesicles are usually known as Exos and are able to promote directional and random cell movement, invasion and serum-independent growth.24,25 Small EVs (sEVs) contain miRNAs and are widely used as reliable biomarkers. 26 In a recent study, compared with healthy controls, colorectal cancer patients showed significantly increased miR-222-3p levels in plasma, which decreased effectively after surgical tumor removal. 27 Circulating sEV-derived miR-222-3p can serve as a promising noninvasive diagnostic biomarker for inflammatory breast cancer. 28 Consistent with a previous study, our findings revealed that miR-222-3p expression in plasma Exos of PE patients was also upregulated. 29 By further analyzing the diagnostic efficiency of miR-222-3p, we found that miR-222-3p in plasma Exos had an AUC of 0.711, sensitivity of 71.43% and specificity of 75% for PE diagnosis. Additionally, miR-222-3p can regulate the function of vascular endothelial cells and smooth muscle cells, 30 while some antihypertensive drugs can regulate miR-222-3p expression to play a regulatory role in hypertensive patients. 31 Compared with normal rats, rats with pulmonary arterial hypertension were unregulated regarding miR-222-3p expression in the right ventricle. 32 A miR-222-3p inhibitor can attenuate the blood pressure of PE rats. 33 Given the above, we speculate that miR-222-3p expression is related to blood pressure. Correlation analysis also showed that miR-222-3p in total plasma and plasma Exos were both positively correlated with SBP and DBP in PE patients, which suggests that miR-222-3p can regulate blood pressure to play a therapeutic role in PE treatment.
Considering the potential pathogenic effect of miR-222-3p on PE, we selected human villous trophoblast HTR-8/Svneo cells for transfection with miR-222-3p inhibitors and observed an increase in the proliferation, innovation and migration of HTR-8/Svneo cells, which is consistent with the findings of a previous study. 33 Since miRNAs can regulate their target genes to participate in the occurrence and progression of diseases, we used four target gene prediction websites to analyze the target gene of miR-222-3p and found that STMN1 is the target of miR-222-3p. This finding was further verified by dual-luciferase reporter assay, qRT‒PCR and Western blotting. In the uterus of rodents, STMN1 (also known as OP18) is upregulated in implantation sites, thus playing a part in the early stages of pregnancy. 34 As reported by another study, STMN1 expression in uNK cells at the maternal-fetal interface may be helpful for the regulation of uNK cell function and beneficial for a successful pregnancy. 35 In a recent study, STMN1 was found to be expressed differently in human endometrial and placental cells, and it may participate in decidualization. Specifically, STMN1 was remarkably downregulated in placental villi tissues and trophoblasts of patients with recurrent spontaneous abortion (RSA), and it is a microtubule regulatory protein related to trophoblast proliferation and invasion and thus involved in the pathogenesis of RSA.12,13 siRNA-induced STMN1 silencing remarkably inhibited the migration and proliferation of trophoblast cells, 12 which is consistent with our findings. The decrease in MMP-2 and MMP-9 could induce decreased vasodilation, increased vasoconstriction, hypertensive pregnancy and PE. 36 In addition, overexpression of miR-222-3p inhibited the expression of MMP2 and MMP9 in HTR-8/SVneo cells. 33 STMN1 may mediate the secretion and activation of MMP2 and MMP9 proteins to promote the growth and invasion of endometrial cancer cells. 37 However, the effect of low STMN1 expression on the biological characteristics of HTR-8/Svneo cells can be reversed by low miR-222-3p expression, which suggests that miR-222-3p can target STMN1 to inhibit the proliferation, invasion and migration of human villous trophoblasts. However, the sample size calculation was not performed in the clinical experiment, which may play a crucial role in influencing the reliability of the result. Moreover, miR-222-3p was related to high SBP and DBP in PE patients. The etiology of PE is gestational hypertension, suggesting that the level of miR-222-3p might be different in general hypertension patients. Therefore, a control group of essential hypertension will be added to a future study, and this is the main limitation in our study.
In summary, PE patients showed upregulated miR-222-3p expression in total plasma and plasma Exos, which was positively correlated with the SBP and DBP of PE patients. In addition, miR-222-3p has a high diagnostic efficiency for PE and can target STMN1 to promote the proliferation, invasion and migration of HTR-8/Svneo cells.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was surpported by the Innovation Capacity Improvement Plan of Hebei Province (NO. 20577705D).
