Abstract
To investigate the effects of EH domain containing protein 2 (EHD2) on clear cell renal cell carcinoma (ccRCC) and provide new insights for the clinical treatment of rental cancer. Forty patients (26 males and 14 females, 62.4 ± 5.7 years old) with ccRCC were selected from January 2015 to December 2016 to serve as research subjects in this study. The EHD2 protein expression in the tumor tissues and adjacent healthy tissues of ccRCC patients were detected by Western Blot assay. The cells of ccRCC cell lines RLC-310 and 786-O were divided into normal control group (control), no-load control group (pLV), EHD2 overexpression group (pLV-EHD2), and EHD2 interference group (pLV-siEHD2). The expression levels of EHD2 protein in each group of cells were detected by western blot. The cell proliferation was detected by Cell Counting Kit-8 (CCK-8) assay. Wound healing assay was performed to check the cell migration ability. Transwell invasion assay was used to detect the cell invasion ability. Cell apoptosis was detected by flow cytometry. The expression level of EHD2 was significantly increased in pLV-EHD2 group and decreased in pLV-siEHD2 group compared with control group and pLV-siEHD2 group, indicating the successfully established EHD2 overexpression cell line and EHD2 RNA interference cell line. EHD2 overexpression enhanced the proliferation, invasion, and migration but inhibited the apoptosis of ccRCC cells, while EHD2 interference showed opposite functions. EHD2 interference can inhibit the development of ccRCC by inhibiting the proliferation, invasion, and migration, and EHD2 can potentially serve as a molecular target for the clinical treatment of ccRCC.
Introduction
As the most common type of rental malignant tumors, renal cell carcinoma (RCC) is responsible for about 3% of all the deaths in men worldwide every year. 1,2 Previous studies indicated that various populations were in poor health, suffering from RCC. 3,4 RCC is developed from the lining of the proximal convoluted tubule, which is a part of the very small tubes that is responsible for the transportation of waste from the blood circulatory system to urine. 5 Based on the histological features, genetic characteristics and clinical phenotypes, RCC can be divided into four different types including clear cell renal cell carcinoma (ccRCC), renal oncocytoma (RO), papillary RCC (pRCC), and chromophobe RCC (chRCC). 6 As the dominant type of RCC, ccRCC accounts for the 75–85% of all cases of RCC. 2,7 Different from many other types of malignant tumors that are mainly related to the mutations happened in oncogenes, 8,9 ccRCC is mainly caused by the mutations happened in suppressor genes including PBRM1, VHL, SETD2, and BAP1. 10,11 Traditional chemotherapy and radiotherapy are the two common methods used in the treatment of ccRCC, however, the treatment efficacy was usually not satisfied due to the resistance of ccRCC to radiation and the drug tolerance developed during the drug treatment. 12 Although previous studies have shown that targeted therapies can extend the progression-free survival of the patients with ccRCC, the application is still limited by the variable individual responses. The onset and development of ccRCC is a very complex process with a variety of environmental as well as internal factors involved. So, it will be with significant clinical values to identify molecular targets that participate in this process to serve as the target of the treatment of ccRCC.
EH domain containing protein 2, which is also called EHD2, is a member of EHD protein family. EHD2 is a membrane-remodeling ATPase that is responsible for the membrane trafficking between the endosomes and plasma membrane. 13,14 As a tumor suppressor gene, EHD2 is usually overexpressed in a variety of cancer tumors including esophageal squamous cell carcinoma and ovarian cancer. 15 The EHD2 mRNA expression in papillary thyroid carcinoma (PTC) was a novel prognostic marker for PTC patients. 16 However, previous studies also showed that the expression of EHD2 was downregulated in some other types of cancer tissues, such as human breast carcinoma. 17 In the study of hepatocellular carcinoma, EHD2 was found to be able to inhibit the invasion and migration of hepatocellular carcinoma possibly through the interaction with E-cadherin, and the inhibited EHD2 expression promoted metastasis of hepatocellular carcinoma and leaded to poor prognosis. 18 EHD2 is associated with migration and invasion of tumor cells as a possible prognostic marker in esophageal cancers. 19 However, the role of EHD2 in ccRCC remains unclear. In view of the different important functions of EHD2 in the development of various cancers, it is reasonable to speculate that EHD2 may also play a role in the onset and development of ccRCC, and EHD2 is possible to serve as a new target for the treatment of ccRCC.
In order to test the above hypothesis, the EHD2 overexpression and interference cell lines were established. The proliferation, invasion, migration, and apoptosis of normal ccRCC cells as well as the cells with EHD2 overexpression and interference were detected and compared.
Materials and methods
Subjects
A total of 40 patients (26 males and 14 females, 62.4 ± 5.7 years old) with ccRCC were selected from January 2015 to December 2016 to serve as research subjects in this study. None of the patients received radiotherapy, chemotherapy, or other special treatment before admission. All patients were required to undergo surgical excision. Tumor tissues and adjacent healthy tissues were collected during the operation. All procedures strict adherence to the principle of asepsis, all samples were immediately transferred to a −80°C liquid nitrogen refrigerator for freezing after they were taken. During radical nephrectomy, the kidneys were sampled and frozen immediately after ex vivo (cancer group). Normal tissue samples were taken from the edge of the tumor more than 5 cm, and the size of specimens cut out not less than 0.5 × 0.5 × 0.5 cm3. This study has been approved by the ethic committee of the Affiliated Yantai Yuhuangding Hospital of Qingdao University and all patients signed informed consent. The experiments were performed according to declaration of Helsinki (DoH).
Cell culture
ccRCC cell lines RLC-310 and 786-O, and human embryonic kidney epithelial cell line 239FT were purchased from by the Center Laboratory of the First Affiliated Hospital of Xi’an Jiao Tong University medical college and Cell Bank, Chinese Academy of Sciences (Shanghai, China). The cells were cultured in DMEM medium (Hyclone, Logan, Utah, USA) containing 10% FBS (Gibco, Carlsbad, California, USA) and 1% double antibiotics (penicillin and streptomycin) (Gibco, Carlsbad, California, USA). The cell culture was carried at in cell incubator (Forma, Waltham, MA, USA) containing 5% CO2 at 37°C.
Lentiviral packaging, collection, concentration, and transfection
The 239FT cells at logarithmic growth phase were collected, digested, and resuspended. After that, the 239FT cells (1.5 × 106) were inoculated in 75 cm flasks 2 days in advance, then the no-load plasmid pLV-EF1A-RFP (Cyagen Biosciences, USA), overexpression plasmid pLV-EF1A-EHD2-RFP (Cyagen Biosciences, USA), and the interference plasmid pLV-EF1A-siEHD2-RFP (Cyagen Biosciences, USA) were packaged using the lentivirus packaging kit (GeneCopoeia, USA) according to the instructions. After 48 h, the supernatant was collected, centrifuged, and filtered. The filtrate was then concentrated by centrifugation (50,000 × g) at 4°C for 4 h, stored in −80°C. The collected viruses were named pLV-RFP, pLV-EHD2-RFP, and pLV-siEHD2-RFP, respectively. Based on transfection methods in some previous reports, 20,21 1 day before transfection, cells (5 × 105) of ccRCC cell line RLC-310 and 786-O were seeded in six-well plates. The cells in the control group did not perform any operations and 200 μl of containing pLV-RFP (pLV group), pLV-EHD2-RFP (pLV-EHD2 group), and pLV-siEHD2-RFP virus solutions (pLV-siEHD2 group) were used to infect those cells, respectively. After 48 h, the cells were subcultured for another 24 h.
Detection of the protein expression in each group of cells
Western blot kit was purchased from Bio-Rad (Hercules, California, USA). The total protein was extracted using the conventional methods and the protein concentration was measured by BCA Protein Assay Kit (Pierce, Waltham, MA, USA). The total protein (40 μg) from each sample was subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis, followed by transmembrane to polyvinylidene fluoride (PVDF) membrane. The membrane was blocked by 5% skim for 1 h. After that, the primary antibodies including rabbit anti-human EHD2 monoclonal antibody, anti-human MMP2 antibody, anti-human MMP9 antibody, anti-human Bcl-2, antibody and anti-human Bax antibody and rabbit anti-human GAPDH polyclonal antibody (Abcam, Cambridge, UK) was diluted (1: 1000) using 50 g/l BSA solution. The diluted primary antibodies were incubated with the membrane overnight at 4°C. The membrane was then washed three times with TBST (TBS containing 1 ml/l Tween-20), 5 min for time. After washing, the goat anti-rabbit secondary antibody (1:10,000, Abcam, Cambridge, UK) was added and incubated at room temperature for 2 h. Then the membrane was washed three times with TBST, 10 min for each time. Enhanced chemo-luminescence (ECL) was performed in dark room. The grayscale was scanned and quantified using Image J 2.1 software. The expression level of EHD2 protein was normalized according to GAPDH.
Detection of cell proliferation
Cell Counting Kit-8 (CCK-8) assay was performed to detect the cell proliferation. 22 The cells at logarithmic growth phase were collected and used to prepare single cell suspensions. After adjusting the density to 1 × 106 cells/ml, the single cell suspension was inoculated in 96-well plates with 100 μl/well (1 × 105 cells/well). Five replicate wells were set for each group of cells. The cells were cultured in incubator (37°C, 5% CO2), and 10 μl of CCK-8 solution (Beyotime Biotechnology, Shanghai, China) was added at 24 h, 48 h, 72 h, and 96 h after the beginning of cell culture. After adding CCK-8 solution, the cells were cultured at 37°C for another 1h to 4 h. The optical density (OD) value of each well was measured at 450 nm using a microplate reader (Bio-Rad, USA). The mean OD value and standard deviation were calculated. With the time as abscissa and the OD value as ordinate, the cell proliferation curve was plotted.
Detection of cell invasion ability
The Transwell invasion assay was performed to detect the cell invasion ability. The invasion chamber (Corning, New York, USA) was first transferred from −20°C to room temperature, and then 500 μl of serum-free DMEM medium preheated at 37°C was added to the invasion chamber. The cells were collected at 24 h after transfection. After digestion, the cells were resuspended in serum free DMEM medium. The cell density was adjusted to 25 × 104/ml, and 200 μl cell suspension was added to the chamber. DMEM medium (600 μl) containing 10% FBS was added to the 24-well plate in the lower part of the chamber. After 24 h incubation, the cells that penetrated into the lower chamber were fixed with 4% paraformaldehyde, stained with 0.5% crystal violet for 20 min, followed by 0.1% crystal violet staining for 15 min. After washing, the membrane was air-dried and observed under an inverted microscope (Olympus, Japan). Five high power fields (100×) were randomly selected and the invasion ability was represented by the number of the cells passed through the membrane. The experiment was repeated 3 times for each group of cells.
Detection of cell migration ability
The migration ability of each group of cells was detected by wound healing assay. The cells were collected at logarithmic growth phase, after routine digestion and centrifugation (Beckman, USA), the cells were resuspended and the cell density was adjusted to 5 × 104/ml. The cells were inoculated in a six-well plate (Corning, New York, USA). After the cells were adherent and the density reached about 80%, a 10 μl tip was used to scratch the cells. The cell culture medium was removed and the cells were washed with phosphate buffer saline (PBS). The cell migration was recorded at 0 h and 48 h after scratching. Image-ProPlus 6.0 software was used to measure the width of the wound to calculate cell migration rate.
Detection of apoptosis
The cells were collected from each group. After centrifugation (Beckman, USA), the cells were resuspended and the cell density was adjusted to 5 × 106/ml. The cell solution (100 μl) was transferred to a 5 ml test tube, and 5 μl FITC Annexin V and 5 μl PI were added. The mixture was gently mixed and kept at room temperature for 15 min. After that, 400 μl 1× Annexin V binding buffer was added and flow cytometry (Beckman, USA) was used to detect the apoptosis.
Statistical analysis
Statistical analysis was performed using SPSS19.0 statistical software. The measurement data were expressed as mean ± standard deviation (SD). The comparison among groups was performed using one-way analysis of variance, least-significant difference (LSD) test for subsequent analysis, and p < 0.05 was considered to be statistically significant.
Results
The expression of EHD2 protein in tissues
The EHD2 protein expression in the ccRCC patients were detected by Western Blot assay. The two representative protein bands, that EHD2 expressed the lowest (sample 1) and the highest (sample 2) were selected in the forty patients. As shown in Figure 1, the expression of EHD2 in sample 1 and sample 2 cancer tissues were significant increased compared with the control (p < 0.01).

The expression levels of EHD2 protein. (a) The expression levels of EHD2 protein in ccRCC patients; (b) quantitative analysis of EHD2 relative changes. *Compared with the control, p < 0.01. EHD2: EH domain containing protein 2.
The expression of EHD2 protein in each group after transfection
After transfection, EHD2 protein showed similar expression patterns in RLC-310 and 786-O cells. No significant difference was found between control group and pLV group. Compared with control group and pLV group, the expression level of EHD2 in RLC-310 pLV-EHD2 group and 786-O pLV-EHD2 group were increased to 2.81 eq and 2.95 eq, respectively (p < 0.05), while the expression level of EHD2 in RLC-310 pLV-siEHD2 group and 786-O pLV-siEHD2 group were decreased to 0.23 eq and 0.17 eq, respectively (p < 0.05). Significant differences were found between pLV-EHD2 groups and pLV-siEHD2 groups (p < 0.05) (Figure 2). Those results showed that the EHD2 overexpression and interference cell lines were successfully established.

(a, c) The expression levels of EHD2 protein in RLC-310 cells after different treatments; (b, d) the expression levels of EHD2 protein in 786-O cells after different treatments. *Compared with the control group, p < 0.05; #compared with pLV group, p < 0.05; ⋆compared with pLV-EHD2 group, p < 0.05. EHD2: EH domain containing protein 2.
The effects of EHD2 expression on cell proliferation
The cell proliferation abilities of RLC-310 and 786-O cells with different treatments were detected by CCK-8 assay at 24 h, 48 h, 72 h, and 96 h after the beginning of cell culture. As shown in Figure 3, similar proliferation patterns were observed in RLC-310 and 786-O cells after different treatments. The cell proliferation of each group was increased along with the time. The highest increase in cell counts was observed in pLV-EHD2 group (0.24–0.88), and the lowest increase in cell counts was observed in pLV-siEHD2 group (0.22–0.48), control group and pLV group were in the middle (0.23–0.60).
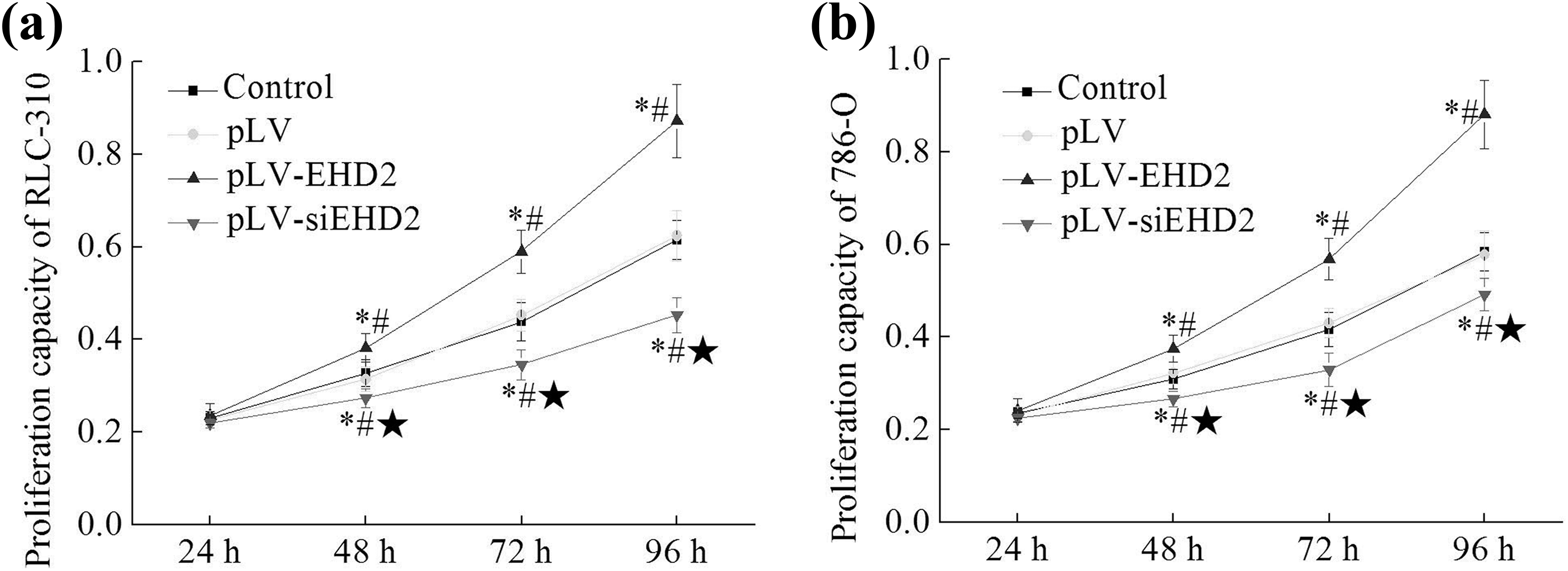
(a) Effect of EHD2 on the proliferation of RLC-310 cells with different treatments; (b) effect of EHD2 on the proliferation of 786-O cells with different treatments. *Compared with the control group, p < 0.05; #compared with pLV group, p < 0.05; ⋆compared with pLV-EHD2 group, p < 0.05. EHD2: EH domain containing protein 2.
Taking RLC-310 cells as an example, compared with control group and pLV group, the cell numbers of pLV-EHD2 group were significantly increased from 48 h (1.17 eq, 1.35 eq, and 1.42 eq at 48 h, 72 h, and 96 h, respectively) (p < 0.05), while the cell numbers of pLV-siEHD2 group were significantly decreased (0.83 eq, 0.79 eq, and 0.74 eq at 48 h, 72 h, and 96 h, respectively) (p < 0.05). So the cell proliferation ability of cells in pLV-siEHD2 group was significantly lower than that of pLV-EHD2 group (p < 0.05). These results suggest that the inhibited EHD2 expression can inhibit the proliferation of ccRCC cells.
Effects of EHD2 on cell invasion
As shown in Figure 4, similar results were got from RLC-310 cells and 786-O cells. Taking RLC-310 cells as an example, no significant difference in cell invasion ability was found between control group and pLV group. Compared with control group and pLV group, the cell invasion ability of pLV-EHD2 group was significantly increased (2.78 eq, p < 0.05) and the cell invasion ability of pLV-siEHD2 group was significantly decreased (0.32 eq, p < 0.05). Compared with pLV-EHD2 group, the cell invasion ability of pLV-siEHD2 group was significantly decreased (p < 0.05). Those results indicate that the inhibited EHD2 expression can inhibit the invasion of ccRCC cells.

(a) Effect of EHD2 on the invasion ability of RLC-310 cells; (b) effect of EHD2 on the invasion ability of 786-O cells (10 × 10). *Compared with the control group, p < 0.05; #compared with pLV group, p < 0.05; ⋆compared with pLV-EHD2 group, p < 0.05. EHD2: EH domain containing protein 2.
The effect of EHD2 on cell migration ability
As shown in Figure 5, similar results were got from RLC-310 cells and 786-O cells. Taking 786-O cells as an example, no significant difference was found in the cell migration ability between control group and pLV group. The cell migration ability of pLV-EHD2 group was significantly higher than that of control group and pLV group (1.99 eq, p < 0.05), while the cell migration ability of pLV-siEHD2 group was significantly lower than that of control group and pLV group (0.72 eq, p < 0.05). Compared with pLV-EHD2 group, the cell migration ability of pLV-siEHD2 group was significantly decreased (p < 0.05). Those data indicate that the EHD2 interference can inhibit the migration of ccRCC cells.

(a) Effect of EHD2 on the migration of RLC-310 cells; (b) effect of EHD2 on the migration of 786-O cells. *Compared with the control group, p < 0.05; #compared with pLV group, p < 0.05; ⋆compared with pLV-EHD2 group, p < 0.05. EHD2: EH domain containing protein 2.
Effects of EHD2 on cell apoptosis
As shown in Figure 6, similar results were got from RLC-310 cells and 786-O cells. Taking 786-O cells as an example, no significant difference was found in apoptotic rate between control group and pLV group. Compared with control group and pLV group, the apoptotic rate of pLV-EHD2 group was significantly decreased (0.47 eq, p < 0.05), while the apoptotic rate of pLV-siEHD2 group was significantly increased (2.78 eq, p < 0.05). The apoptotic rate of pLV-siEHD2 group was significantly higher than that of pLV-EHD2 group (p < 0.05). The results showed that the EHD2 interference can promote the apoptosis of ccRCC cells.

(a) Effect of EHD2 on the apoptosis of RLC-310 cells; (b) effect of EHD2 on the apoptosis of 786-O cells. *Compared with the control group, p < 0.05; #compared with pLV group, p < 0.05; ⋆compared with pLV-EHD2 group, p < 0.05. EHD2: EH domain containing protein 2.
Effects of EHD2 on MMP2, MMP9, Bcl-2, and Bax protein expression
As shown in Figure 7, compared with control group and pLV group, the expression level of MMP2, MMP9, and Bcl-2 in RLC-310 pLV-EHD2 group and 786-O pLV-EHD2 group were significantly increased (p < 0.05), while the expression levels in RLC-310 pLV-siEHD2 group and 786-O pLV-siEHD2 group were decreased (p < 0.05). Significant differences were found between pLV-EHD2 groups and pLV-siEHD2 groups (p < 0.05) (Figure 7). In contrast, the expression level of Bax protein in RLC-310 pLV-EHD2 group and 786-O pLV-EHD2 group was significantly decreased, and the expression level in RLC-310 pLV-siEHD2 group and 786-O pLV-siEHD2 group was increased (p < 0.05). Those results further showed that inhibited EHD2 expression can promoted the apoptosis of ccRCC cells.

(a) The expression of MMP2, MMP9, Bcl-2, and Bax in RLC-310 cells; (b) the expression of MMP2, MMP9, Bcl-2, and Bax in 786-O cells. *Compared with the control group, p < 0.05; #compared with pLV group, p < 0.05; ⋆compared with pLV-EHD2 group, p < 0.05. EHD2: EH domain containing protein 2.
Discussion
Recent studies have shown that the onset and development of ccRCC was closely related to the genetic changes, especially the mutations happened in tumor suppressor genes. 23 The mechanism of ccRCC is still unknown, current experimental and clinical studies of ccRCC are mainly focused on the identification of molecular biomarkers, which can be used to predict the onset, development, and metastasis of ccRCC and the outcomes after treatment. Due to the unsatisfied outcomes from traditional therapies in the treatment of ccRCC, targeted therapies, which can target ccRCC-related pathways (such as VEGF pathway) and individual genes (such as tumor suppressor genes) have been developed. 24 Previous studies have shown that the application of anti-vascular epithelial growth factor (VEGF) agents, mammalian target of rapamycin (mTOR) inhibitors, and epidermal growth factor receptor inhibitors can significant inhibit the progression of ccRCC in patients and the combined applications of those agents can even achieve better outcomes. 24 Von Hippel-Lindau (VHL) is a tumor suppressor gene that plays important roles in inhibiting ccRCC progression. Although VHL is not an optimal target for the treatment of ccRCC, the phosphorylation status of VHL can be used as an indicator in the target selections of target therapies. 25 However, the application of target therapies in the treatment of ccRCC is still facing the problem that the treatment efficacy is variable in different patients due to the variable individual response. The limited optimal targets were not sufficient to cover all or the majority of the ccRCC patients.
The main function of MMPs family is to degrade the extracellular mechanism, which is closely related to the invasion and metastasis of tumors. MMP2 and MMP9 are two very important molecules in the MMPs family. Their activity characterizes the migration and invasion of tumor cells. 26 In this study, the expression of MMP2 and MMP9 protein was detected to investigate the cell invasive ability. Bcl-2 and Bax were used to assess the cell apoptosis. The EHD2 overexpression promoted the expression of MMP2, MMP9, and Bcl-2, and inhibited the expression of Bax, demonstrated the role of EHD2 at the molecular level in ccRCC.
EHD2, as a tumor suppressor gene, has long time be used as an indicator for the progression evaluation and treatment outcome prediction of a variety of cancers. A recent study found that the overexpression of EHD2 can inhibit the invasion and migration of breast cancer cells possibly by interacting with Ras-related C3 botulinum toxin substrate 1 to inhibit its expression, while reduced EHD2 expression promoted the invasion and migration of breast cancer cells probably by abrogating Rac1 expression. 15 It was also found that the low expression level of EHD2 might be able to promote the pathogenesis of human esophageal squamous cell carcinoma. 19
In the study, EHD2 was selected as a target to investigate the pathogenesis of ccRCC. In contrast with the findings in previous studies, opposite role of EHD2 has been found in the development of ccRCC. The EHD2 protein expression in the ccRCC patients were detected by Western Blot assay. The expression level of EHD2 in 40 ccRCC patients’ cancer tissues were obviously increased compared with normal tissue (p < 0.01). Although EHD2 is a tumor suppressor gene, the overexpression of EHD2 was found to be able to promote the proliferation, invasion, and migration of the cells of two ccRCC cell lines—RLC-310 and 786-O, at the same time, the reduced expression of EHD2 significantly inhibited the apoptosis of ccRCC cells in this study. In contrast, the reduced expression level of EHD2 can possibly slow down the progression of ccRCC by cell proliferation, invasion and migration, and promote cell apoptosis.
At present, there is no consistent study with us that EHD2 is related to the development of ccRCC, but there is a report that the inhibited expression of EHD2 is correlated with good cancer prognosis. 16 Though only selected 40 cases were selected, and these cases were randomly selected. However, there still needs to collect a large number of clinical samples in the later period to study the relationship between the expression of EHD2 and the clinical characteristics and prognosis of ccRCC in detail. The opposite functions of EHD2 in tumor development identified in the study indicate the complex regulatory network of different cancers.
Conclusion
EHD2 can promote the proliferation, invasion, and migration and inhibit the apoptosis of ccRCC cells, so EHD2 interference can significantly inhibit the development of ccRCC, and EHD2 can potentially serve as a molecular target for the clinical treatment of ccRCC.
Footnotes
Authors’ note
This work was supported by Projects of medical and health technology development program in Shandong province “The study of Molecular mechanism of EHD2 as a new drug target for renal clear cell carcinoma treatment” (No. 2016WS0716).
Author Contributions
C Liu and S Liu contributed equally to this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
