Abstract
A series of studies was conducted to assess the genetic toxicity of a novel ketone ester, bis hexanoyl (R)-1,3-butanediol (herein referred to as BH-BD), according to Organization for Economic Co-operation and Development testing guidelines under the standards of Good Laboratory Practices. In bacterial reverse mutation tests, there was no evidence of mutagenic activity in any of the Salmonella typhimurium strains tested or in Escherichia coli strain WP2uvrA, at dose levels up to 5,000 μg/plate in the presence or absence of Aroclor 1254-induced rat liver (S9 mix) for metabolic activation. In the in vitro micronucleus test using human TK6 cells, BH-BD did not show a statistically significant increase in the number of cells containing micronuclei when compared with concurrent control cultures at all time points and at any of the concentrations analyzed (up to 100 μg/mL, final concentration in culture medium), with and without S9 mix activation. In the in vivo micronucleus test using Sprague Dawley rats, BH-BD did not show a statistically significant increase in the incidence of micronucleated polychromatic erythrocytes relative to the vehicle control group. Therefore, BH-BD was concluded to be negative in all 3 tests. These results support the safety assessment of BH-BD for potential use in food.
Introduction
Ketone bodies, or “ketones” (β-hydroxybutyrate, acetoacetate, and acetone), act as both fuel substrates and signaling metabolites. Natural elevation of blood ketone concentrations occurs as a result of a strict ketogenic diet, which is low in carbohydrate, moderate in protein, and high in fat,
1
and during periods of starvation or voluntary caloric restriction.
2
In these conditions, low glucose availability and low insulin concentrations increase fatty acid mobilization, and these fatty acids undergo hepatic conversion into ketones. A healthy human of ∼70 kg can produce ∼150 g of ketones per day during a period of prolonged fasting,
3
reaching blood
Another method to moderately elevate BHB concentrations and achieve physiological ketosis without dietary changes is to ingest an exogenous ketone compound. One class of exogenous ketones are “ketone esters,” (KE = ketone ester) comprising of 2 or more ketone and/or ketogenic precursor moieties, joined by one or more ester bonds. Following ingestion, these KEs are hydrolyzed by ubiquitously expressed esterase enzymes, and the breakdown products are either absorbed directly into the blood as ketones or undergo metabolic conversion in the liver to ketones. We recently synthesized a novel KE compound; bis hexanoyl (R)-1,3-butanediol (BH-BD; Figure 1) and reported that BH-BD undergoes hydrolysis in several sites of the gastrointestinal tract, in liver microsomes and in plasma. Oral administration of BH-BD raises blood BHB concentrations to between 3 and 5 mM in rats and mice. 7 This led us to investigate the opportunity to use BH-BD as a novel food ingredient to elevate blood ketone levels in humans.
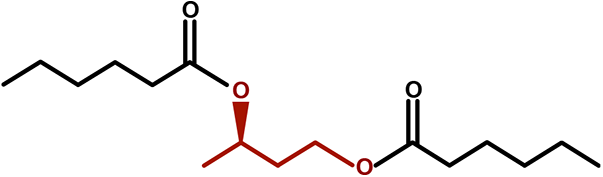
Structure of bis hexanoyl (R)-1,3-butanediol.
In preparation for its use as a food ingredient, we investigated the potential for genetic toxicity of BH-BD through consideration of its known hydrolysis products ((R)-1,3-butanediol and hexanoic acid), the major downstream metabolite (BHB) and the intact KE. (R)-1,3-butanediol is an approved food additive; the available evidence suggests that (R)-1,3-butanediol is unlikely to be genotoxic as it is not carcinogenic or developmentally toxic. 8 -10 Hexanoic acid is commonly consumed as an ingredient and flavor in foods 11 and was negative in a battery of in vitro genetic toxicity assays. 11,12 The ketone body BHB is an endogenous metabolite and no studies that have chronically elevated plasma concentrations through a ketogenic diet or using an exogenous source have found any evidence of mutagenic effects. Finally, as BH-BD is a novel compound there was no literature available to inform its potential for genetic toxicity. Therefore, Quantitative Structure Activity Relationship analysis was performed to screen BH-BD for structural alerts and hazard classification, which demonstrated a low hazard classification by Cramer et al 13 (class I) and did not trigger structural alerts for genetic toxicity or mutagenicity. Although our in vitro and in vivo experiments to date do indicate complete breakdown of BH-BD into its nongenotoxic hydrolysis products likely occurs prior to absorption, 7 in order to definitively confirm the safety of BH-BD for use in food, the genetic toxicity of the intact KE should be directly tested according to the US Food and Drug Administration (FDA) recommended toxicological testing for food ingredients. 14
Therefore, the 3 studies reported herein were conducted to evaluate the genetic toxicity potential of BH-BD using in vitro bacterial or mammalian cell-based tests and an in vivo test. Firstly, the bacterial reverse mutation test, which evaluate the mutagenic potential of the test article by measuring its ability to induce reverse mutations at selected loci of several strains of Salmonella typhimurium and at the tryptophan locus of Escherichia coli WP2uvrA in the presence and absence of an exogenous metabolic activation system. 15 -17 Secondly, the in vitro mammalian cell micronucleus (MN) test in TK6 cells (a human lymphoblastoid cell line), which detects clastogenic or aneugenic activity of a test article in the presence and absence of exogenous metabolic activation by assessing the presence of micronuclei in the cytoplasm of postmitotic cells. 18 -20 Finally, the in vivo mammalian erythrocyte MN test, which evaluates the test article for in vivo clastogenic activity and/or disruption of the mitotic apparatus by detecting micronuclei in polychromatic erythrocytes (PCE) in bone marrow following oral administration to male Sprague Dawley rats. 21,22
All studies were conducted at BioReliance Corporation between May and August 2020. All studies were performed in compliance with the US FDA Good Laboratory Practice Regulations (21 CFR Part 58) (FDA, 1987). The study protocols were designed in general accordance with the relevant testing guidelines of Organization for Economic Co-operation and Development (OECD), that is, OECD Testing Guideline No. 487 “In Vitro Mammalian Cell Micronucleus Test, 23 OECD Testing Guideline No. 474 “Mammalian Erythrocyte Micronucleus Test,” 24 and OECD Testing Guideline No. 471 “Genetic Toxicology: Bacterial Reverse Mutation Test,” 25 which are consistent with respective US FDA Redbook testing guidelines. 26
Methods
Test Article
Bis hexanoyl (R) 1,3-butanediol is a ketone ester that is synthesized through esterification of the ketogenic precursors hexanoic acid and (R)-1,3 – butanediol. The BH-BD test sample used for these studies was supplied by BHB Therapeutics Ltd as a clear oil with 98.7% of BH-BD and the remaining 1.3% accounted for by monosubstituted esters, as verified by gas chromatography linked to mass spectrometry analysis. The test article was confirmed to comply with established specifications for material composition and identity (eg, 98% minimum ketone esters containing ≥95% BH-BD). For both the bacterial reverse mutation test and in vitro mammalian cell MN test, the test article was dissolved in a vehicle of dimethyl sulfoxide (DMSO, Sigma-Aldrich). For the in vivo mammalian cell MN test, prior to administration the test article was stirred into a vial of food-grade canola oil vehicle (Gordon’s Choice Salad Oil) to give the required dose concentrations. Formulation vials were restirred prior to and during dosing.
Test System, Culture Medium, and S9 Activation System
Bacterial reverse mutation test
Tester strains were Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 as described by Ames and McCann, 15 which were obtained from Dr. Bruce Ames, University of California, Berkeley, and the Escherichia coli strain WP2uvrA as described by Green and Muriel, 27 which was from the National Collection of Industrial and Marine Bacteria, Aberdeen, Scotland (United Kingdom). On the day of use in each test, all tester strain cultures were checked for the appropriate genetic markers. The S9 liver homogenate was purchased from MolTox. It was prepared from Aroclor 1254-induced male Sprague Dawley rats and prepared at the testing facility according to Ames and McCann 15 and Maron and Ames. 28 Each bulk preparation of S9 was assayed for its ability to metabolize benzo(a)pyrene and 2 aminoanthracene to forms mutagenic to Salmonella typhimurium TA100. On the day of use, aliquots of the S9 liver homogenate were prepared and mixed with a nicotinamide adenine dinucleotide phosphate generating system. The final concentrations were MgCl2 8 mM, KCl 33 mM, Glucose-6-phosphate (G-6-P) 5 mM, NADP 4 mM, sodium phosphate 100 mM (pH 7.4), and S9 10% (vol/vol).
In vitro mammalian cell MN test
The TK6 cell line was obtained from the American Type Culture Collection (Repository number CRL-8015) and maintained as a master stock in log phase. Prior to treatment with BH-BD, cells were set up in T-75 cm2 flasks in a culture medium that consisted of Roswell Park Memorial Institute 1640 medium (with
The S9 liver homogenate was obtained and prepared as described above for the bacterial reverse mutation test. For the in vitro MN test, the S9 mix consisted of MgCl2 2 mM, KCl 6 mM, G-6-P 1 mM, NADP 1 mM, and S9 20 µL/mL.
In vivo mammalian erythrocyte MN test
Healthy male Sprague-Dawley rats (n = 24) aged 6 weeks, weighing 150 to 174 g were purchased from Envigo RMS, Inc and acclimated for 6 days prior to initiation of the study. Animals were housed in a controlled environment at 72 ± 3 °F and 50% ± 20% relative humidity with a 12-hour light/dark cycle. Animals were maintained in accordance with The Guide for the Care and Use of Laboratory Animals (National Academy Press, 2011) under a study protocol approved by the BioReliance Institutional Animal Care and Use Committee. A certified laboratory rodent chow (Envigo 2018C Teklad Global 18% Protein Rodent Diet) was provided ad libitum. Animals were assigned to groups using a randomization procedure within a dedicated laboratory software program (Provantis). At the time of randomization, the weight variation of animals did not exceed ±20% of the mean weight.
Treatment, Sampling, and Analysis
Bacterial reverse mutation test
The test article was dissolved in DMSO at 100 mg/mL based on a purity of 98.7%. TA98, TA100, TA1535, TA1537, and WP2uvrA were exposed to the vehicle alone, positive controls and 6 to 8 dose levels of test article, in triplicate, in the presence and absence of Aroclor induced rat liver S9. Positive controls were 2-aminoanthracene (in DMSO), 2-nitrofluorene (in DMSO), sodium azide (in water), 9-aminoacridine (in DMSO), and methyl methanesulfonate (in DMSO); these were obtained from MolTox. Based on a preliminary toxicity assay, the dose range selected for the mutagenicity test tested ranged from 3.33 to 5,000 μg/plate.
The test system was exposed to the test article via the plate incorporation methodology originally described by Ames and McCann 15 and updated by Maron and Ames. 28 The plates were incubated at ca. 37 °C for approximately 48 to 72 hours. After the incubation, the plates were assessed for the presence of precipitate (as detected by the naked eye per OECD 471). Subsequently, the his + and trp + revertants were counted either manually or by machine. The condition of the bacterial background lawn was evaluated for evidence of test article toxicity and precipitate. Evidence of toxicity was scored relative to the vehicle control plate and recorded along with the revertant count for that plate. Toxicity was evaluated as a decrease (by at least 50%) in the number of revertant colonies per plate and/or a thinning or disappearance of the bacterial background lawn. Precipitation was evaluated after the incubation period by visual examination without magnification. As appropriate, colonies were enumerated either by hand or by machine.
In vitro mammalian cell MN test
Treatment occurred by incorporation of the test article vehicle mixture into the treatment medium. At the end of the recovery period, the cells in each culture were harvested and processed as per standard procedure. Cells on slides were swollen with hypotonic solution (KCl, 1:1 vol/vol), washed with fixative (methanol: glacial acetic acid, 25:1 vol/vol), centrifuged, and then resuspended in fixative. The cell suspension was applied to glass microscope slides, air dried, and then stained with acridine orange for microscopic evaluation. Cells were evaluated by fluorescent microscopy by a reader who was blinded to test conditions. A minimum of 2,000 mononucleated cells from each dose level were scored for the presence of micronuclei using established criteria. 29
For the short-term exposure (4 hours), TK6 cells (2.5 × 105 cells/mL) were treated with BH-BD in the absence or presence of S9. Following 4 hours exposure, cultures were washed, the treatment medium was replaced by CCM and cells were incubated for an additional 23 hours. For the long-term exposure (27 hours), cells were treated with BH-BD in the absence of S9 continuously. Mitomycin C (MMC, 0.08 and 0.12 µg/mL, Sigma-Aldrich), vinblastine (VB, 10 and 12 ng/mL, Sigma-Aldrich), and cyclophosphamide (CP, 2.5, 3.0, and 4.0 µg/mL, Sigma-Aldrich) were incorporated as positive controls, and DMSO was used as the vehicle control throughout.
Statistical analysis was performed using the Fisher exact test (P ≤ .05) for a pairwise comparison of the percentage of micronucleated cells in each treatment group with that of the vehicle control. The Cochran-Armitage trend test was used to assess dose-responsiveness.
In vivo mammalian erythrocyte MN test
Dose selection was based upon toxicity information from a previous 14-day toxicity study (BHB Therapeutics Internal Data), in which an oral gavage dose of BH-BD at 15,000 mg/kg/d was well tolerated in Sprague Dawley rats, supporting use of the standard limit dose for the MN test (2,000 mg/kg/d). Doses used were 500, 1,000, and 2,000 mg/kg/d (n = 6 animals per dose group). All dose formulations were administered once per day on 2 consecutive days at a volume of 10 mL/kg via oral gavage. The second dose occurred approximately 24 hours after the first dose. The vehicle control for this study was food-grade canola oil (n = 6 animals). Positive control slides generated by the testing facility were included to verify scoring. These slides were generated from a positive control group of animals treated once with cyclophosphamide monohydrate at 40 mg/kg (n = 5 animals).
Animals were euthanized by carbon dioxide inhalation at approximately 24 hours after the final dose administration. Immediately following euthanasia, the femurs were exposed, cut just above the knee, and the bone marrow was aspirated into a syringe containing fetal bovine serum. The cell suspension was concentrated by centrifugation, and the supernatant was discarded. The bone marrow was transferred to a centrifuge tube containing 2 mL fetal bovine serum, the cells were pelleted by centrifugation, and the supernatant was drawn off leaving a small amount of fetal bovine serum with the pellet. Cells were resuspended and a small drop of the bone marrow suspension was spread onto a clean glass slide, air dried, fixed by dipping in methanol, and stained with acridine orange for microscopic evaluation. Bone marrow was evaluated by fluorescent microscopy by a blinded reader. A total of 4,000 PCEs/animal were scored for the presence of micronuclei (MnPCEs) using an established criteria (Schmid, 1975). In addition, at least 500 total erythrocytes (PCEs + normochromatic erythrocytes) were scored per animal to determine the proportion of PCEs as an index of bone marrow cytotoxicity.
Statistical analysis was performed on the MN frequency (%MnPCE) and %PCE using the animal as the unit. The mean and standard deviation of %MnPCE and %PCE were presented for each treatment group. The group variances for MN frequency for the vehicle and test article groups at the respective sampling time were compared using Levene’s test (significance level of P ≤ 0.05). Since the variation between groups was found not to be significant, a parametric 1-way analysis of variance (ANOVA) was performed followed by a Dunnett’s post hoc analysis to compare each dose group to the concurrent vehicle control. A linear regression analysis was conducted to assess dose responsiveness in the test article–treated groups (P ≤ 0.01 and R 2 ≥ 70%). A pair-wise comparison (Student t test; P ≤ 0.05) was used to compare the positive control group to the concurrent vehicle control group.
Results
Bacterial Reverse Mutation Test
The results of the mutagenicity test are presented in Table 1. No toxicity was observed. Precipitate was observed beginning at 3,333 µg/plate in the absence of S9 activation and at 5,000 µg/plate in the repeat test with TA100 in the presence of S9 activation. The vehicle controls for TA100 in the absence of S9 activation and WP2uvrA in the presence and absence of S9 activation were outside of the respective 95% control limits but within the upper 99% control limits. The vehicle control for TA100 in the presence of S9 activation was outside the 99% control limits and did not meet the criteria for test acceptance. Therefore, this experimental condition was repeated. No positive mutagenic responses were observed with any of the tester strains in either the presence or absence of S9 activation.
Bacterial Reverse Mutation Test of BH-BD.
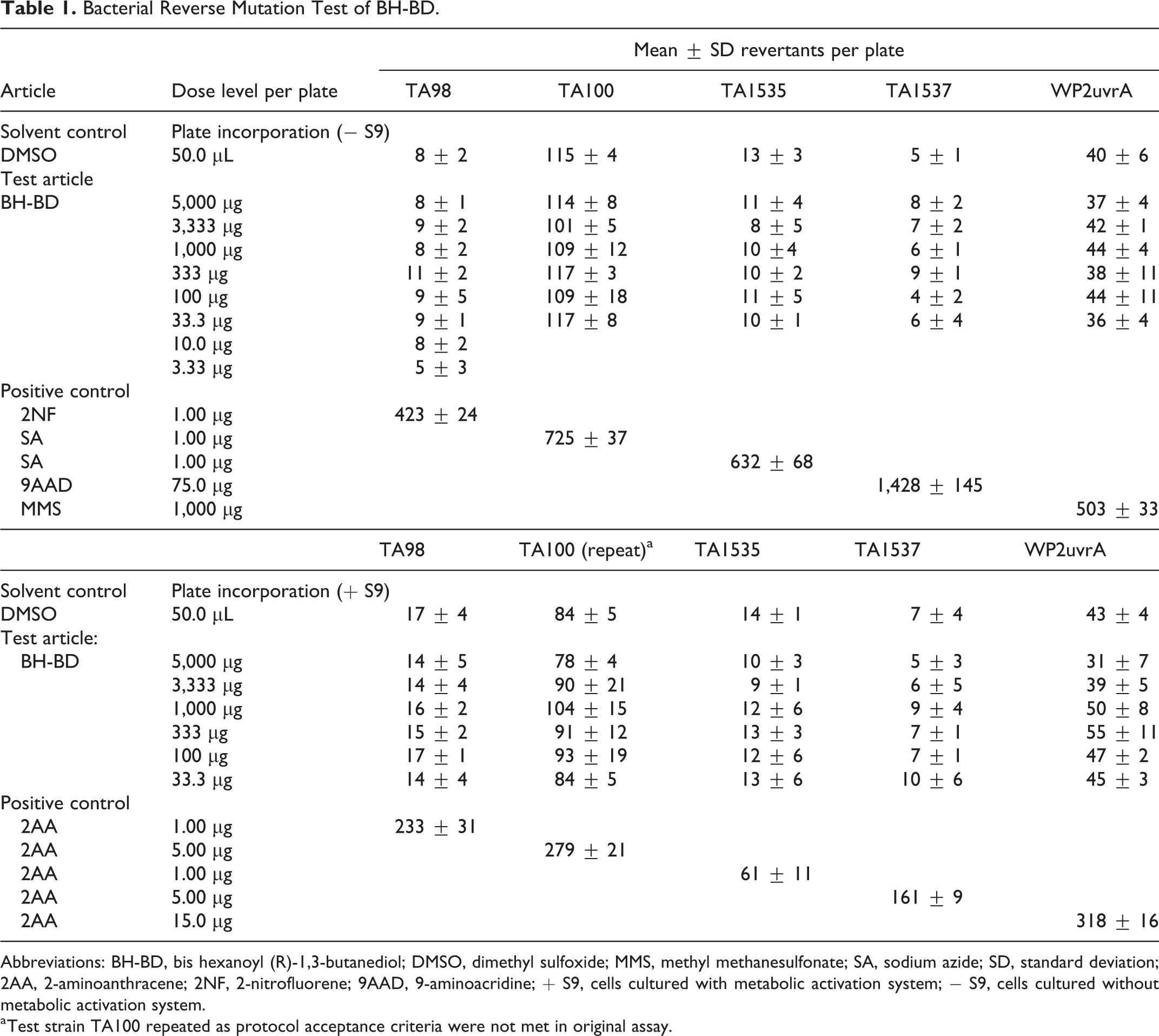
Abbreviations: BH-BD, bis hexanoyl (R)-1,3-butanediol; DMSO, dimethyl sulfoxide; MMS, methyl methanesulfonate; SA, sodium azide; SD, standard deviation; 2AA, 2-aminoanthracene; 2NF, 2-nitrofluorene; 9AAD, 9-aminoacridine; + S9, cells cultured with metabolic activation system; − S9, cells cultured without metabolic activation system.
a Test strain TA100 repeated as protocol acceptance criteria were not met in original assay.
The results indicate that, under the conditions of this study, BH-BD did not cause a positive mutagenic response with any of the tester strains in either the presence or absence of Aroclor induced rat liver S9.
In Vitro Mammalian Cell MN Test
Treatment with BH-BD did not lead to a statistically significant or dose-dependent increase in micronuclei induction at any dose compared to control cultures, with or without metabolic activation by S9 (P > 0.05; Fisher exact and Cochran-Armitage tests; Table 2). The results were within the 95% control limit of the historical negative control data of the laboratory and results observed for the negative and positive control treated cultures demonstrated that all criteria for a valid test were met. Cytotoxicity (≥50% decrease in population doubling relative to the vehicle control) was not observed at any dose in the nonactivated and S9-activated 4-hour exposure groups. Cytotoxicity was observed at doses ≥150 µg/mL in the nonactivated 27 hour exposure group. The highest dose evaluated in the MN test in the nonactivated 27 hour exposure group was 100 µg/mL.
In Vitro Micronucleus Test Using BH-BD in the Presence and Absence of Exogenous Metabolic Activation (4 Hours) and Absence of Metabolic Activation (27 Hours).
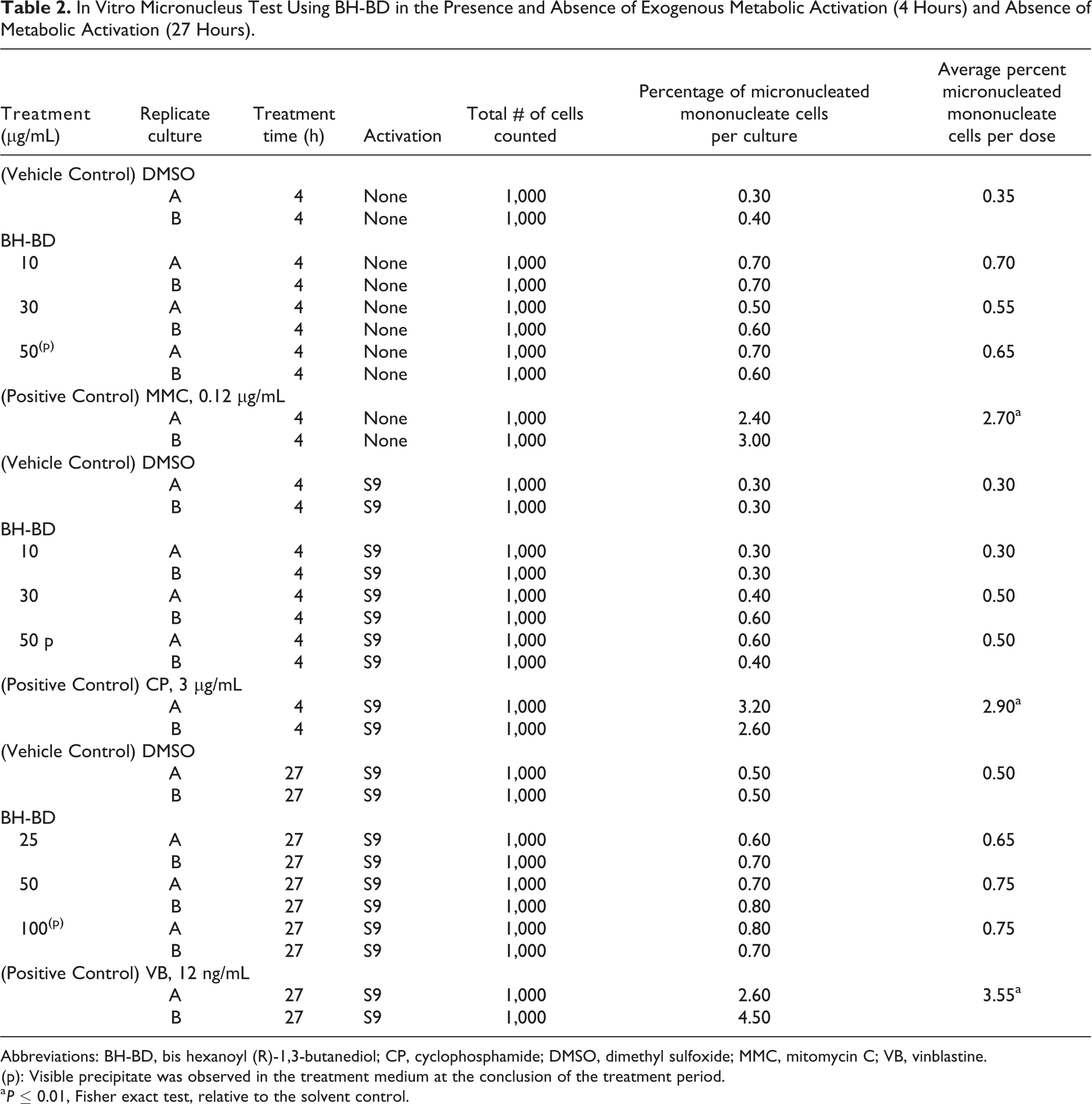
Abbreviations: BH-BD, bis hexanoyl (R)-1,3-butanediol; CP, cyclophosphamide; DMSO, dimethyl sulfoxide; MMC, mitomycin C; VB, vinblastine.
(p): Visible precipitate was observed in the treatment medium at the conclusion of the treatment period.
a P ≤ 0.01, Fisher exact test, relative to the solvent control.
From the results obtained, it was concluded that, under these experimental conditions, BH-BD was nongenotoxic in the nonactivated and S9-activated treatment condition in the in vitro mammalian MN test using TK6 cells.
In Vivo Mammalian Erythrocyte MN Test
No mortality occurred at any dose level during the course of the study. All animals appeared normal throughout the observation period.
The incidence of MnPCEs per 20,000 to 24,000 PCEs scored (4,000 PCEs/animal) and the proportion of PCE per total erythrocytes are summarized and presented for each treatment group by sacrifice time in Table 3. No appreciable reductions in the PCEs/EC ratio was observed in the test article groups compared to the vehicle control group, indicating the test article did not induce cytotoxicity. No statistically significant increase in the incidence of MnPCEs was observed in the test article–treated groups relative to the vehicle control group (ANOVA followed by Dunnett’s post hoc analysis, P > 0.05). The positive control, cyclophosphamide monohydrate, induced a statistically significant increase in the incidence of MnPCEs (Student t test, P ≤ 0.05).
Summary of Bone Marrow Micronucleus Analysis.
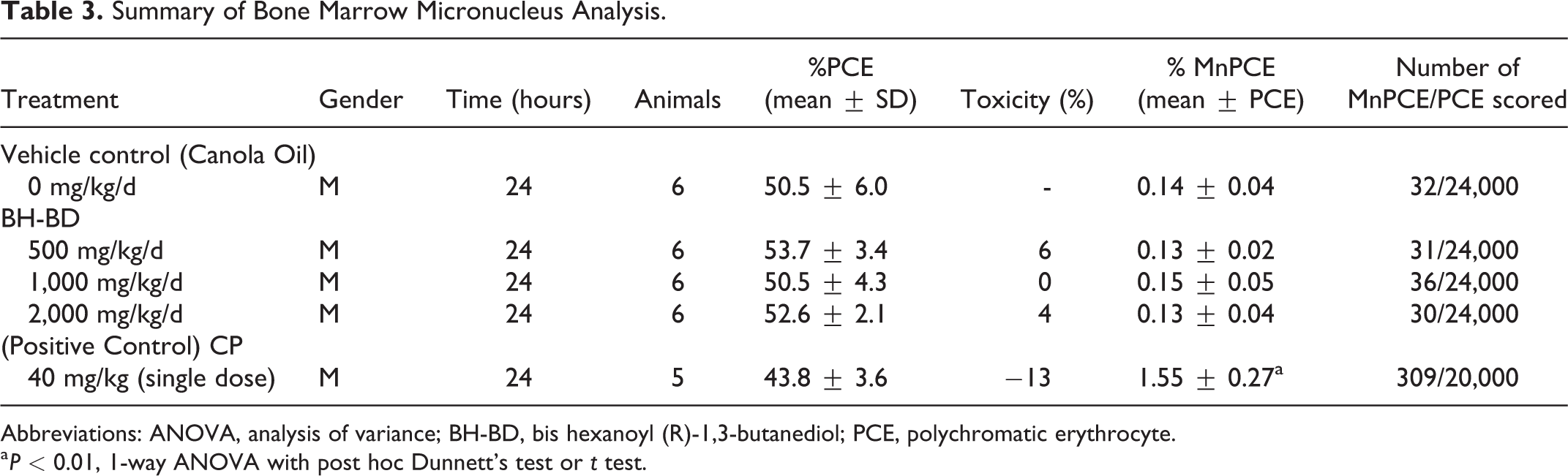
Abbreviations: ANOVA, analysis of variance; BH-BD, bis hexanoyl (R)-1,3-butanediol; PCE, polychromatic erythrocyte.
a P < 0.01, 1-way ANOVA with post hoc Dunnett’s test or t test.
From the results obtained from the in vivo mammalian cell MN test, it was concluded that, under these experimental conditions, BH-BD was negative for the induction of micronucleated PCE.
Discussion and Conclusions
The objective of this work was to obtain information on the genetic toxicity potential of the novel ketone ester, BH-BD, in 3 test systems (2 in vitro and 1 in vivo), including the bacterial reverse mutation test, the in vitro MN test using TK6 cells, and the in vivo mammalian erythrocyte MN test in rats. Negative results in all 3 studies demonstrate that BH-BD is without any significant genotoxic activity under the conditions of the tests performed. The hydrolysis products of BH-BD, (R)-1,3-butandeiol and hexanoic acid, and the downstream endogenous metabolite, BHB, have not demonstrated genotoxic activity. The results reported herein demonstrate no genetic toxicity of intact BH-BD should any of the compound reach the circulation prior to hydrolysis. This adds to the safety database under development for BH-BD, supporting its potential use in food.
Footnotes
Acknowledgments
The authors thank Dr Megan Young for her support in the role of Study Director for the bacterial reverse mutation study.
Author Contributions
Stubbs, B. contributed to conception and design, contributed to interpretation, and drafted manuscript; Nikiforov, A. contributed to conception and design, contributed to interpretation, and critically revised manuscript; Rihner, M. contributed to conception, contributed to interpretation, and critically revised manuscript; Weston, S. contributed to design, contributed to analysis, and critically revised manuscript; Higley, N. contributed to conception and critically revised manuscript; Roy, S. contributed to design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Dakoulas, E. contributed to design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Verdin, E. contributed to conception and critically revised manuscript; Newman, J. contributed to conception and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.C.N and E.V. are cofounders with equity interest of BHB Therapeutics Ltd, which is developing products related to ketone bodies. B.J.S. has an equity interest in HVMN, Inc, which markets products related to ketone bodies, and stock options in BHB Therapeutics Ltd. J.C.N., E.V., and B.J.S. are inventors on patents related to the use of ketone bodies. N.H., A.N., M.R., and S.W. were scientific consultants in the design, review, and audit of these studies. They derive no direct financial benefit from the results of this research. S.R. and E.D. are employees of BioReliance Corporation which received funding to conduct these studies. BioReliance Corporation derives no direct financial benefit from the results of this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by BHB Therapeutics Ltd.
