Abstract
Introduction
Bis-octanoyl (R)-1,3-butanediol (BO-BD) is a novel, palatable ketone ester that, when consumed, is hydrolyzed in the gastrointestinal tract into octanoic acid (OCT) and (R)-1,3-butanediol (BDO) which are subsequently metabolized into beta-hydroxybutyrate (BHB). Metabolism of BO-BD is hypothesized to be similar to bis-hexanoyl (R)-1,3-butanediol (BH-BD), apart from release of octanoic acid instead of hexanoic acid (HEX).
Methods
As part of the safety assessment for BO-BD a randomized, cross-over, open-label study in middle-aged, healthy adults (
Results
All study products increased plasma BHB and BDO up to 4 h post-consumption. BH-BD increased HEX, whereas BO-BD increased OCT. All kinetic parameters for BHB and BDO were similar between 12.5
Discussion
The two novel ketone ester molecules exhibit similar metabolic breakdown to BHB and BDO and result in transiently higher concentrations of the plasma fatty acids, OCT and HEX, in vivo.
Conclusions
Given the similar ketone delivery with greater acceptability, BO-BD may offer a more broadly translatable tool to induce physiologic ketosis than BH-BD.
Introduction
Ketone bodies, beta-hydroxybutyrate (BHB), acetoacetate, and acetone, are endogenous metabolites derived from lipids with two significant functions. First, ketones provide a critical alternative metabolic fuel during starvation,
1
second, ketones act as a signaling metabolites, linking the nutrient-environment to cellular function. ‘Ketosis’ is usually clinically defined by capillary blood BHB concentrations
Consuming exogenous sources of ketones can elevate blood ketone concentrations without other dietary changes. Ketones from exogenous sources can act as a fuel and signal, through the same pathways as endogenously derived ketones, and therefore may benefit health and performance. One class of exogenous ketones, called ketone esters, has been investigated for their impact on multiple endpoints, including physical and cognitive performance,7–13 blood glucose regulation14–17 and cardiac function.18,19
There is a previously developed novel ketone di-ester that induces nutritional ketosis within 30 min: bis-hexanoyl (R)-1,3-butanediol (BH-BD, common name: C6 ketone di-ester). Based on a series of in vitro Schematic illustrating the metabolism of bis-hexanoyl (R)-1,3-butanediol (BH-BD) and bis-octanoyl (R)-1,3-butanediol (BO-BD). Both ketone di-esters are hydrolyzed into butanediol and either hexanoic or octanoic acid, which are largely metabolized into ketone bodies such as beta-hydroxybutyrate (BHB) in the liver but may be released into the systemic circulation. Figure created using BioRender.com.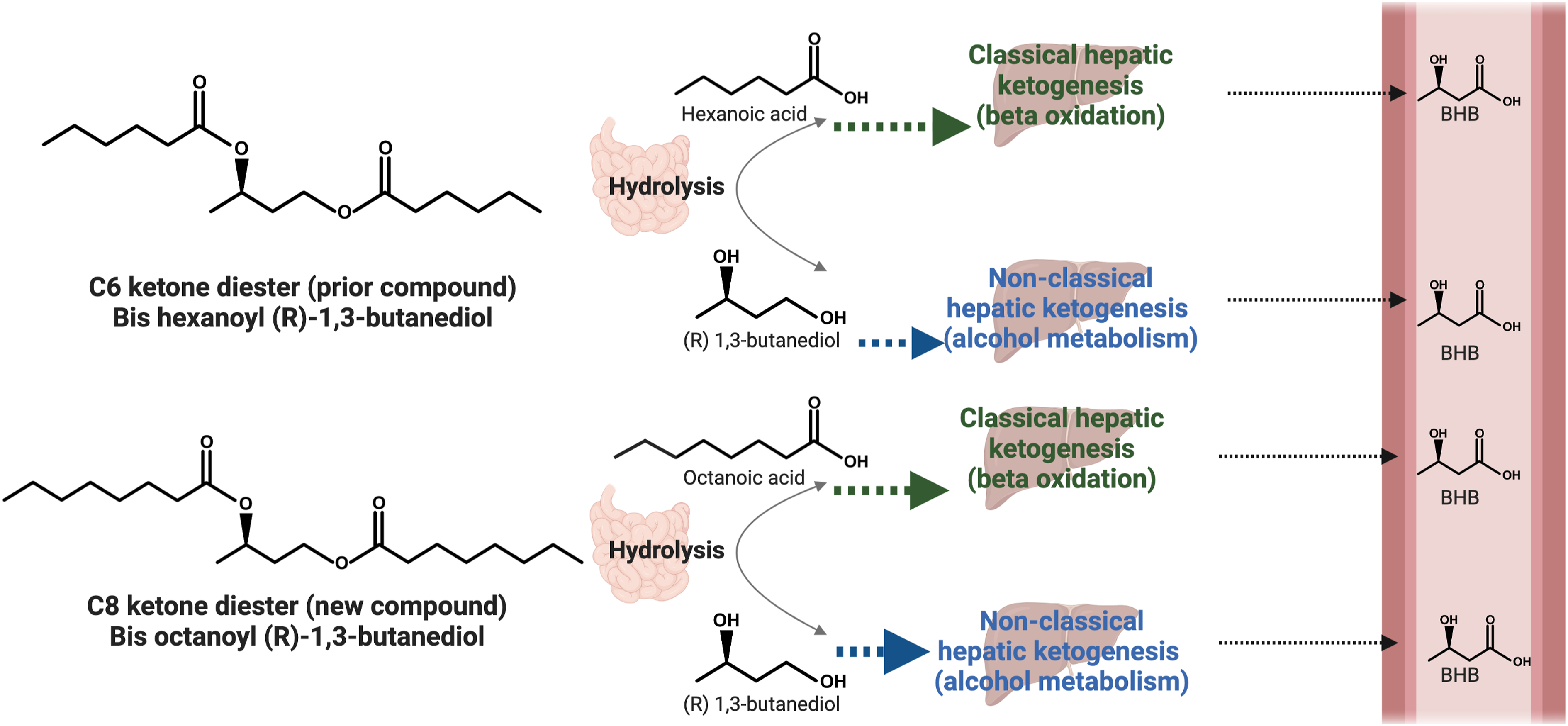
Consumption of BH-BD results in dose-dependent increases in circulating BHB in both humans22–24 and rodents
29
that last for several hours, as well as small, transient increases in plasma concentrations of HEX and BDO.
23
Although both BH-BD and BO-BD are expected to have a similar metabolic fate, a qualitative comparison is required to allow for extrapolation from the existing safety literature on BH-BD and to support a safety assessment of BO-BD. We hypothesized that similar to BH-BD, BO-BD will undergo hydrolysis into a medium chain fatty acid (OCT) and BDO, followed by metabolic conversion to BHB, resulting in similar plasma concentrations of the hydrolysis and metabolic products. To test this hypothesis, a randomized, open-label, cross-over pilot study in healthy adults was undertaken with the objective of determining blood BHB, HEX, OCT and BDO pharmacokinetics after ingestion of 12.5 or 25
Materials and methods
Clinical study overview
Healthy adults (n = 12) underwent screening (Visit 1, Day -7) and took part in a randomized, open label, cross-over pilot study that evaluated the plasma kinetic parameters of BHB, BDO, OCT and HEX for 8 h following ingestion of one or two servings of BO-BD (12.5 and 25 Schematic showing overall study design, study product and servings, and schedule for pharmacokinetics (PK) visits. Figure created using BioRender.com.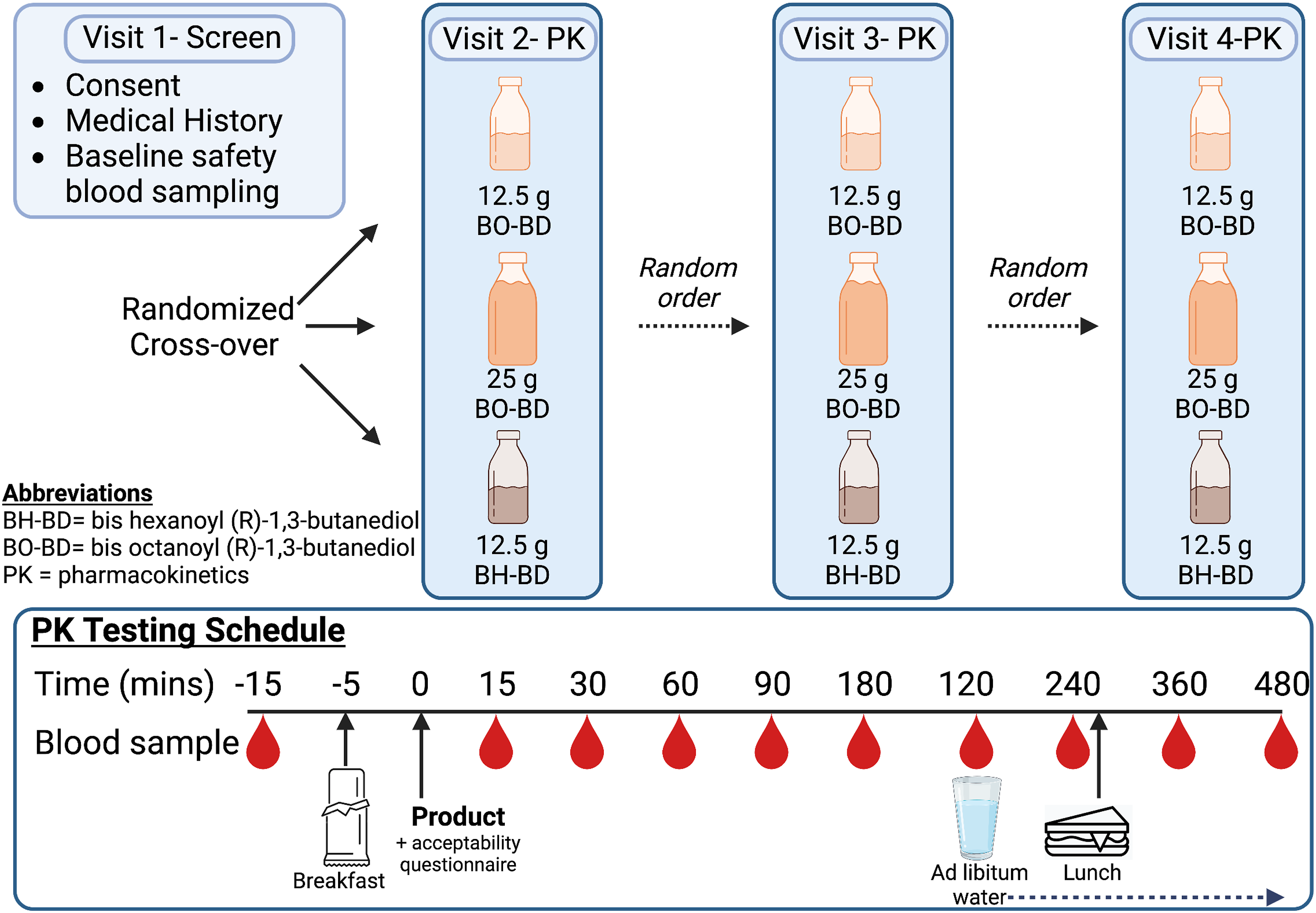
An institutional review board (Sterling IRB, Atlanta, GA, January 6th 2023, BIO2302) approved all study related material including the protocol and informed consent documents prior to initiation of the study. Signed informed consent and authorization for use of protected health information was provided by the subjects prior to implementation of any protocol specific procedures. The study was registered in the clinicaltrials.gov database (NCT05686083). The study was conducted in accordance with the Declaration of Helsinki and the United Sates Code of Federal Regulation Title 21. Studies took place at Biofortis Research (Addison, IL) between January 2023 and March 2023. Recruitment of subjects began on 17 January 2023 and the last subject follow up took place on 28 February 2023. CONSORT reporting guidelines were used for reporting this study. 30
Subjects and screening
Participants were healthy, aged 18 – 65 years, BMI 18.5–34.9 kg/m2. Major exclusion criteria included – no history of major illness, no clinically important gastrointestinal conditions, not pregnant and using contraception to prevent pregnancy (females only), no recent infection, no extreme dietary habits (i.e. intermittent fasting, ketogenic diet, vegan), no unstable medication use, no recent use of weight loss supplements or medications known to influence gastrointestinal function, no recent use of ketone supplements, and no known allergies to any of the study beverage ingredients (including soy and milk protein). Pregnant or lactating women were excluded from the study and women of childbearing potential (<60 years old) were required to use appropriate contraceptive methods to avoid pregnancy. Documentation of contraception method was recorded in the source chart. There were no other controls on menstrual cycle phase or type of contraceptive used. Full inclusion and exclusion criteria are included in Supplemental Information (Table S1).
At the screening visit (Visit 1, Day -7), participants completed a medical history questionnaire in addition to assessment of height, weight, BMI, vital signs, last menses (females only), current medication/supplement use, and review of inclusion/exclusion criteria to determine eligibility. Subjects were assessed for the suitability for use of an intravenous catheter by study phlebotomists. In addition, fasting (10 –14 h) blood samples were collected for analysis of clinical chemistry and hematology. Females under the age of 60 years completed a urine pregnancy test. Subjects had the opportunity to taste the study beverages (19 mL) to evaluate palatability.
Study products and standard meals
BO-BD (common name, C8 ketone di-ester) was provided as an ingredient in a prototype tropical flavored beverage (supplied by BHB Therapeutics). BH-BD (common name, C6 ketone di-ester) was provided as an ingredient in a chocolate flavored beverage product, “
Subjects kept a diet log for all foods consumed after 5 p.m. the day prior to Visit 2 (Day 0) and were asked to replicate intake for each of the subsequent testing visits. At all visits, subjects consumed a light standard breakfast (Nutri-Grain® Soft Baked Breakfast Bar) 5 min prior to study product consumption and a standard lunch (offerings included: Smart Ones® slow roasted turkey breast, baby carrots, lowfat Yoplait® yogurt, Mott’s® apple sauce, Quaker Oats® lowfat oatmeal raisin bar, decaffeinated coffee, decaffeinated tea, diet soda, water) following the 240 min post-product blood sample. Lunch was consumed within 30 min and subjects were instructed to eat until comfortably full on Day 0; intake was replicated on Days 7 and 14.
Study procedures
For in-clinic testing days (Day 0, 7 and 14), subjects reported to the test facility having fasted for 10 – 14 h (water only) and having avoided exercise for 24 h. Subjects were asked to maintain their habitual diet and exercise habits during the study. Subjects were queried to ensure compliance with these instructions at the start of each clinic visit. A screening capillary blood glucose measurement was done to confirm compliance. Subjects were considered compliant to fasting if blood glucose was ≤126 mg/dL, which is the clinical threshold fasting glucose value for diabetes. 31 Body weight and vital signs were measured, concomitant medication and supplement use was queried, females completed a last menses query and inclusion and exclusion criteria were reviewed at the start of each visit. At Day 0, subjects were randomly allocated to one of the study groups using a 1:1:1 sequence generated by a study statistician. An intravenous catheter was inserted into the antecubital fossa at least 5 min prior to the first blood sampling time point. The catheter was flushed with saline solution at least hourly during the study to maintain patency. The first blood sample was collected at t = −5 to – 15 min, where t = 0 was the time of study product consumption. Subjects consumed a light breakfast at t = −5 min (see details above), followed by their allocated study product (see details above). They then completed a product acceptability questionnaire to assess their liking of the study product. Briefly, five domains were ranked on a 1 – seven scale where low scores indicated low liking, and higher scores indicated high liking. Following study product consumption (t = 0), blood samples were collected at 15, 30, 60, 90, 120, 180, 240, 360 and 480 (sample collection was permitted up to 5 min before or after scheduled time). Ad libitum water intake was allowed after the 120 min blood draw and at Day 0, intake was recorded and replicated at Day 7 and 14. Immediately following the 240 min blood sample a standard lunch was consumed (see details above). At the end of each visit, the venous catheter was removed, and adverse events were assessed by an open-ended question. The investigator graded the adverse events by severity (mild, moderate, severe) and judged the likelihood they were related to the study product (definitely, probably, possibly, unlikely, not related).
In vitro BO-BD and BH-BD plasma hydrolysis
In vitro plasma incubations were conducted at Keystone Bioanalytical Inc (North Wales, PA, USA). Human plasma (fresh, unfrozen) was obtained from BioIVT (Westbury, NY, USA). Plasma (5 mL) was incubated with BO-BD or BH-BD (20 μg/mL) in a water bath at 37°C, and 50 μL aliquots were removed at 0, 15, 30, 60 and 90 min after the start of incubation (Figure 3). Aliquots were mixed with 200 μL of ice-cold internal standard (BH-BD-d3; Lot no. EW35732-10-P1 synthesized by Wuxi App Tec with 99.2% purity, 2 μg/mL) mixed for ∼2 min and centrifuged at 14,000 r/min at 4°C for 5 min. 100 μL of the upper phase was removed and transferred to a HPLC injection vial with 100 μL of (de-ionized) water. Analytical methods are described below. Schematic for in vitro plasma hydrolysis study design. Figure created using BioRender.com.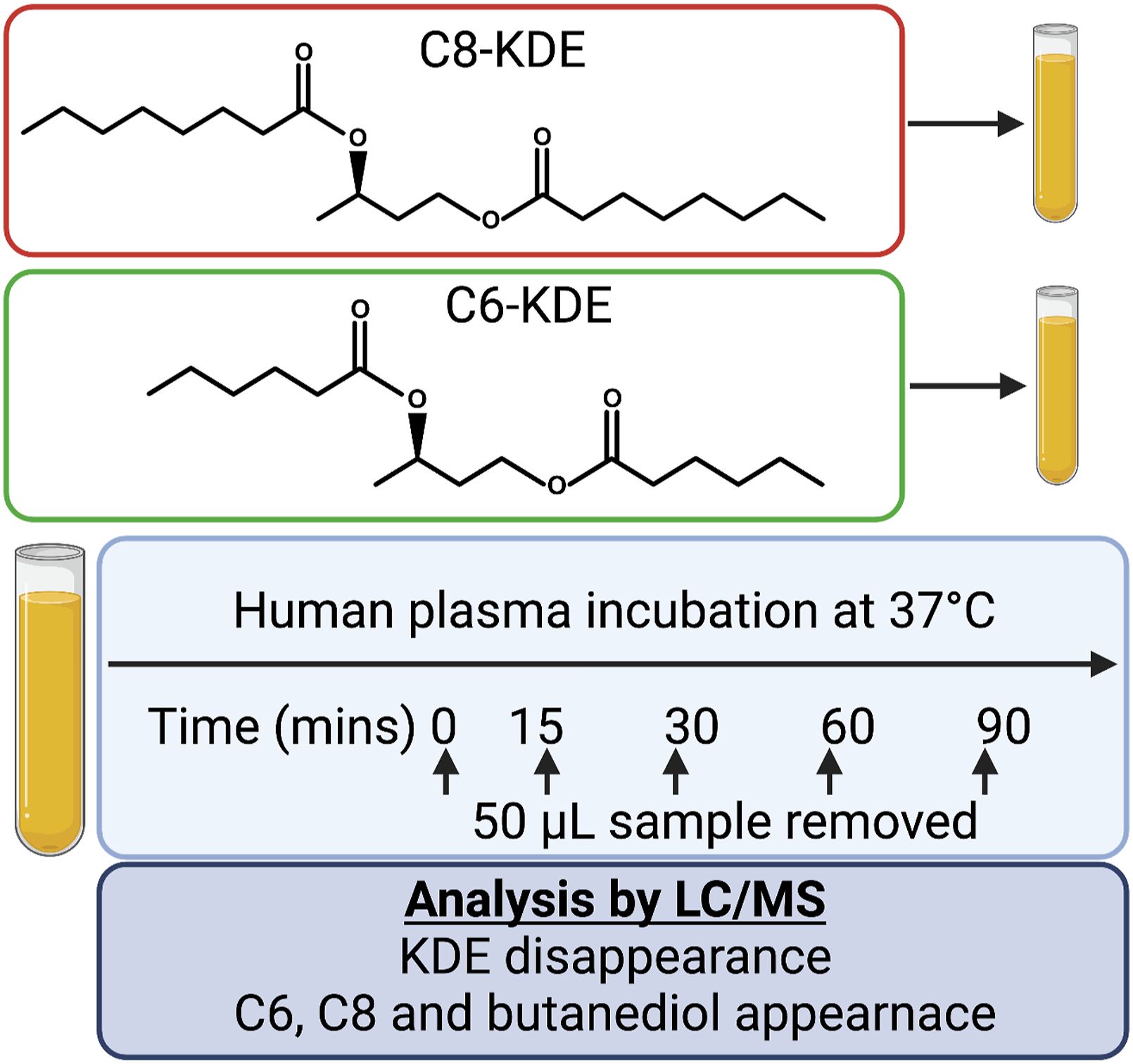
Sample analysis
Analysis of blood samples collected during screening (Visit 1, Day -7) took place at Elmhurst Memorial Reference Laboratory (Elmhurst, IL). Analysis included standard clinical chemistry and hematology panels, to ensure subject suitability for enrollment.
Blood samples from PK visits at Day 0, 7 and 14 were frozen until they were analyzed for BHB, BDO, OCT and HEX at Keystone Bioanalytical Inc (North Wales, PA, USA). BH-BD, BHB, BDO, OCT and HEX were determined using high performance liquid chromatography/mass spectrometry (HPLC/MS) using gradient elution. For the in vitro plasma hydrolysis experiments, a Shimadzu LC-20AD binary HPLC connected with a Phenomenex Kinetex XB-C18 column, 50x2.1 mm, 2.6 µm and Sciex API5500 LC/MS/MS detector were used for the determination of BO-BD and BH-BD. Mass transitions under the positive with ESI (electrospray ionization) are m/z 287.2->171 and m/z 290.2 ->174 were used to monitor BH-BD, BO-BD and BH-BD -d3. Area ratio of the BH-BD and BO-BD to BH-BD -d3 was used for the quantitative determination of BH-BD and BO-BD with a calibration range 312.5 to 20,000 ng/mL. The HPLC flow was 0.5 mL/min using 0.1% formic acid in water, and 0.1% formic acid in acetonitrile (ACN) as the mobile phase with a gradient program; run time was 5 min. The BH-BD concentration at baseline (0 mins) was used as 100% to calculate the rate of hydrolysis. Samples were analyzed in triplicate. For the clinical study, PBS (Phosphate Buffered Saline, pH 7.4) was used as surrogate matrix for blanks, calibration standards and QCs preparation. Calibration standards and QCs were prepared by spiking the target analyte (BHB, BDO, OCT, HEX) into PBS. Standards used for calibration were as follows. For BHB, the reference standard was (±)-β-hydroxybutyrate (Cayman Chemical; product number 14148) and the internal standard was (±)-β-hydroxybutyrate-d4 (Cayman Chemical; product number 14158). For BDO, the reference standard was (±)-1,3-butanediol (Sigma-Aldrich; product number 9068) and the internal standard was (±)-1,3-butanediol-1,3- 13 C2 (Sigma-Aldrich; product number 900065). For HEX, the reference standard was hexanoic acid (Sigma-Aldrich; product number 21529) and the internal standard was hexanoic acid-d11 (Cayman Chemical; product number 28082). For OCT, the reference standard was octanoic acid (Cayman Chemical; product number 33674) and the internal standard was octanoic acid-d3 (Cayman Chemical; product number 28869). PBS was spiked with the appropriate concentrations of BHB, BDO, OCT or HEX to give target concentrations yielding a lower limit of quantitation (LLOQ) and upper limit of quantitation (ULOQ) of 0.5 μg/mL and 350 μg/mL, respectively, for BHB; 0.2 μg/mL and 150 μg/mL, respectively, for BDO; 0.1 μg/mL and 50 μg/mL, respectively, for OCT and HEX. Plasma samples were thawed and mixed by vortex. For BHB and BDO, 50 μL of plasma was combined with 50 μL of 50% ACN/50% water. To this, 50 μL internal standard solution (for BHB: 5 μg/mL of BHB-d4 in 50%ACN/50%water, for BDO: 5 μg/mL of BDO-d2 in 50%ACN/50%Water) was added. Finally, 0.15 mL ACN was added, the mixture vortexed, and centrifuged at 14,000 r/min and 4°C for 5 min. For analysis by HPLC, 25 μL of supernatant was combined with 225 μL nanopure water prior to injection. Sample preparation for OCT and HEX was similar. Internal standard solution (5 μg/mL of HEX-d11 or OCT-d3 in 50%ACN/50% water) was added to the 50:50 plasma: ACN/water and well-mixed. Centrifugation under the same conditions as used for BHB and BDO was followed by transfer of 50 μL supernatant into a 1.5 mL conical plastic tube. To this, 50 μL of 3-NPH (3-nitrophenylhydrazine), 50 μL EDAC (1-ethyl-3-3(3-dimethylaminopropyl)carbodiimide), and 50 μL of 10% pyridine solution was added, vortexed. Tubes were heated at 65°C for 15 min and then prepared for HPLC injection as described for BHB and BDO.
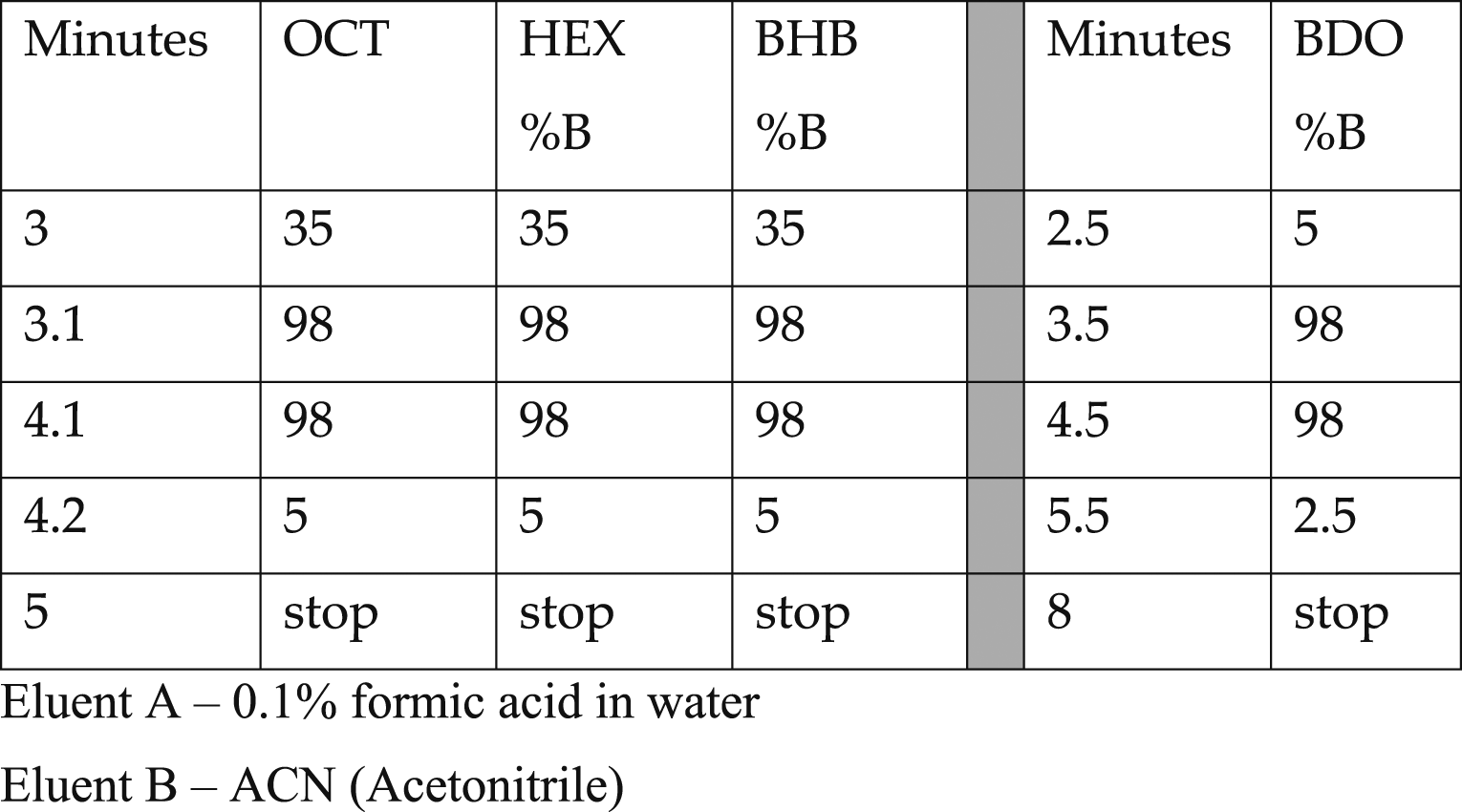
Gradient elution programs for systems A and B.
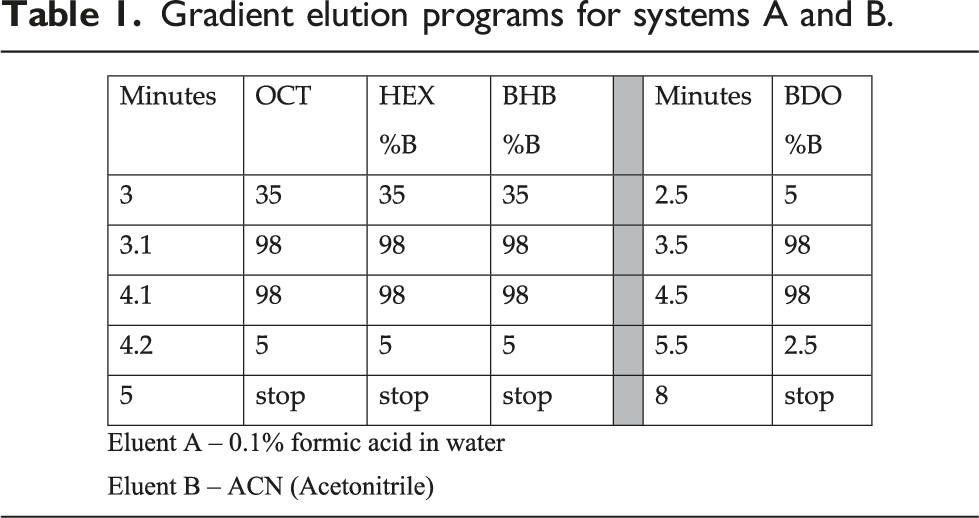
Concentrations at or below the quantification limit (QL) were observed for OCT, HEX and BDO and were replaced with the QL divided by the square root of 2. The LLOQ for BDO, OCT and HEX was 0.2, 0.1 and 0.1 μg/mL, respectively.
Statistical methods
Sample size
In a previous study of BH-BD, total β-hydroxybutyrate AUC for 12.5
Outcome analysis
R version 4.2.0 with the package “NonCompart” 32 was used to calculate the non-compartmental analysis parameters for OCT, HEX, BDO, and BHB for each subject. The following parameters were defined: Total AUC from pre-product consumption (t = −0.25 h) to 8 h (AUC0-8h) where the time prior to product consumption (t= −15 min) was used as time 0. The AUC was calculated by the linear-log trapezoidal method (“linear-up log-down”) where the linear trapezoidal method was used when concentrations were increasing (“absorption phase”) and the logarithmic trapezoidal method used when the concentrations were decreasing (“elimination phase”).
Maximal concentration (Cmax) was defined as the maximum recorded concentration over the 8 h collection period and time of maximal concentration (Tmax) was defined as the time of the maximum concentration. Half-life (t1/2) was calculated as the natural log of two divided by the estimate of the terminal elimination rate constant (λZ) where λZ was estimated by negative slope of the linear regression of the terminal data for the time versus log concentration curve.
For each of HEX (BH-BD test condition only), OCT (12.5
To explore the relationship between the body weight adjusted dose and the PK parameters AUC and Cmax, the linear mixed model was used including an interaction term for product and body weight adjusted dose.
Results
Clinical study subject demographics and disposition
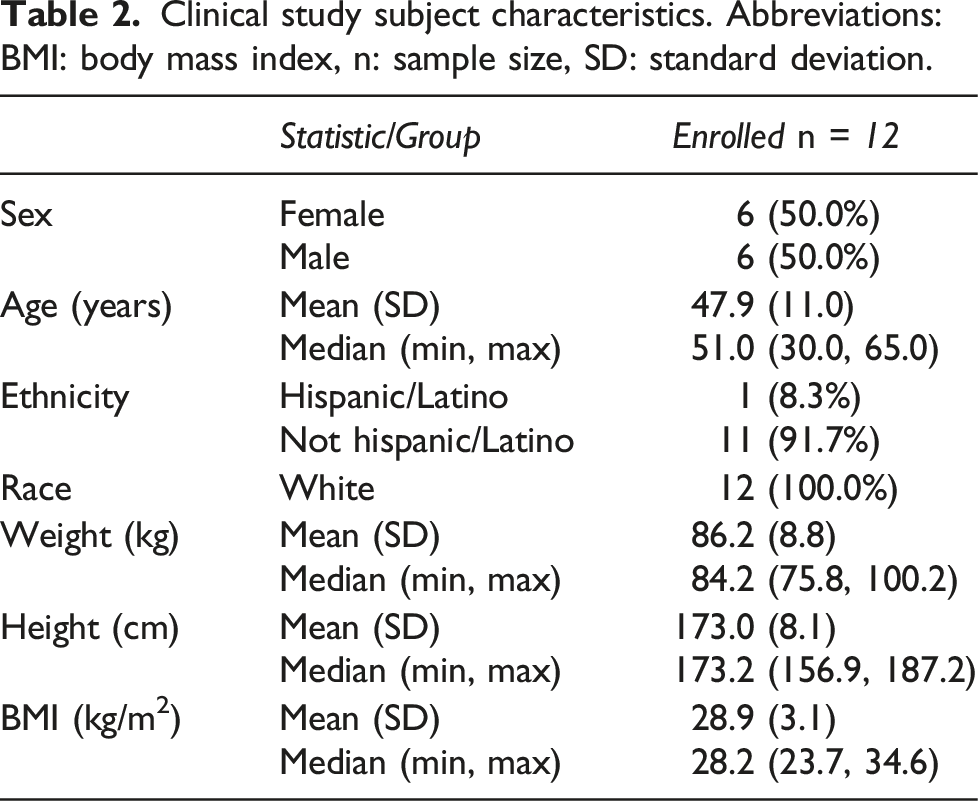
Clinical study subject characteristics. Abbreviations: BMI: body mass index, n: sample size, SD: standard deviation.
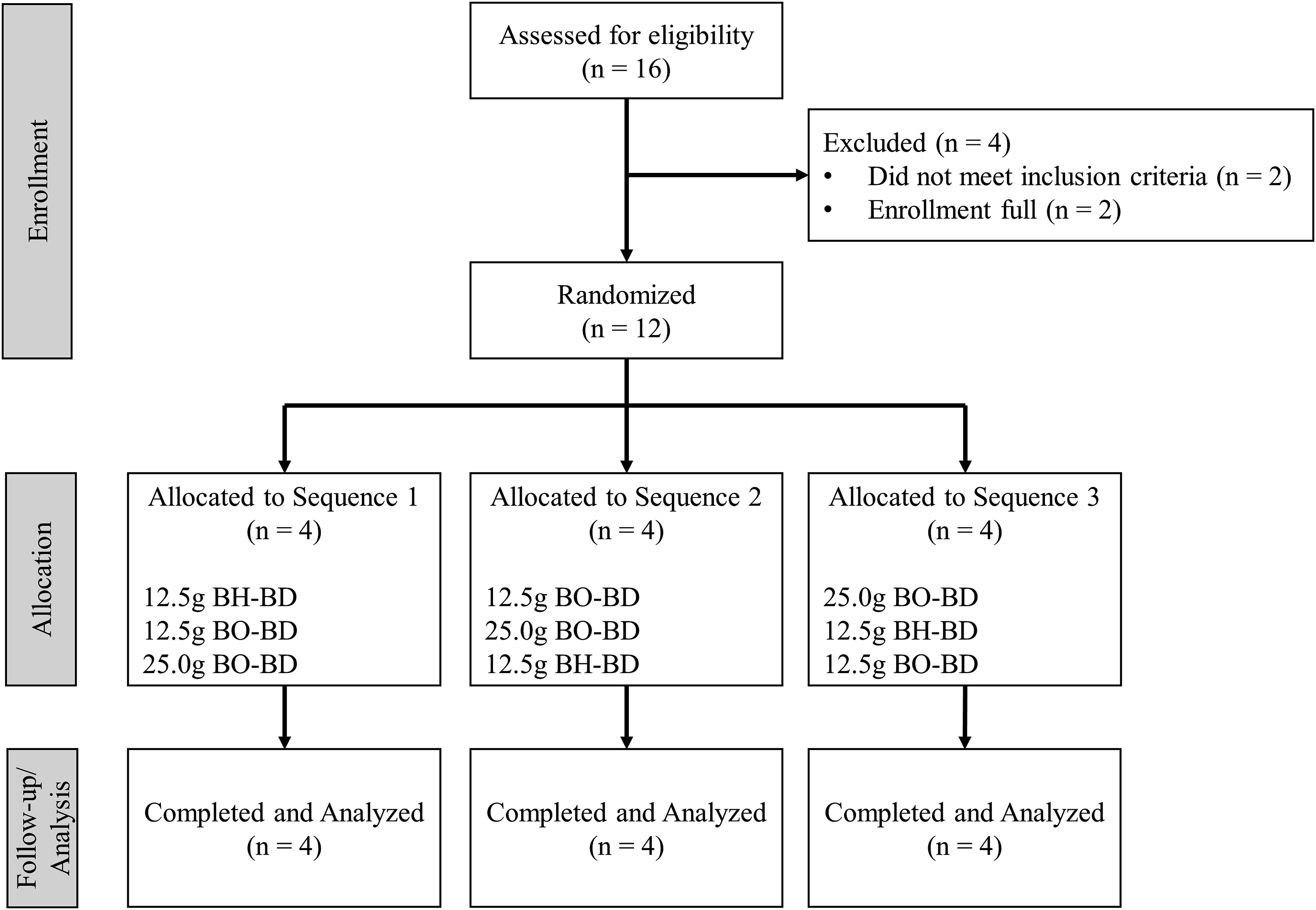
Study flow chart showing the disposition of subjects by study group.
Adverse events
A total of two subjects reported adverse events (AEs) at least once during the study. One subject reported menstrual cramps during one visit. One subject reported a vasovagal reaction and bruising immediately following the IV catheter insertion. All AEs were judged as mild in severity, none were related to the study product, and all were confirmed to have resolved during follow-up.
Plasma beta hydroxybutyrate
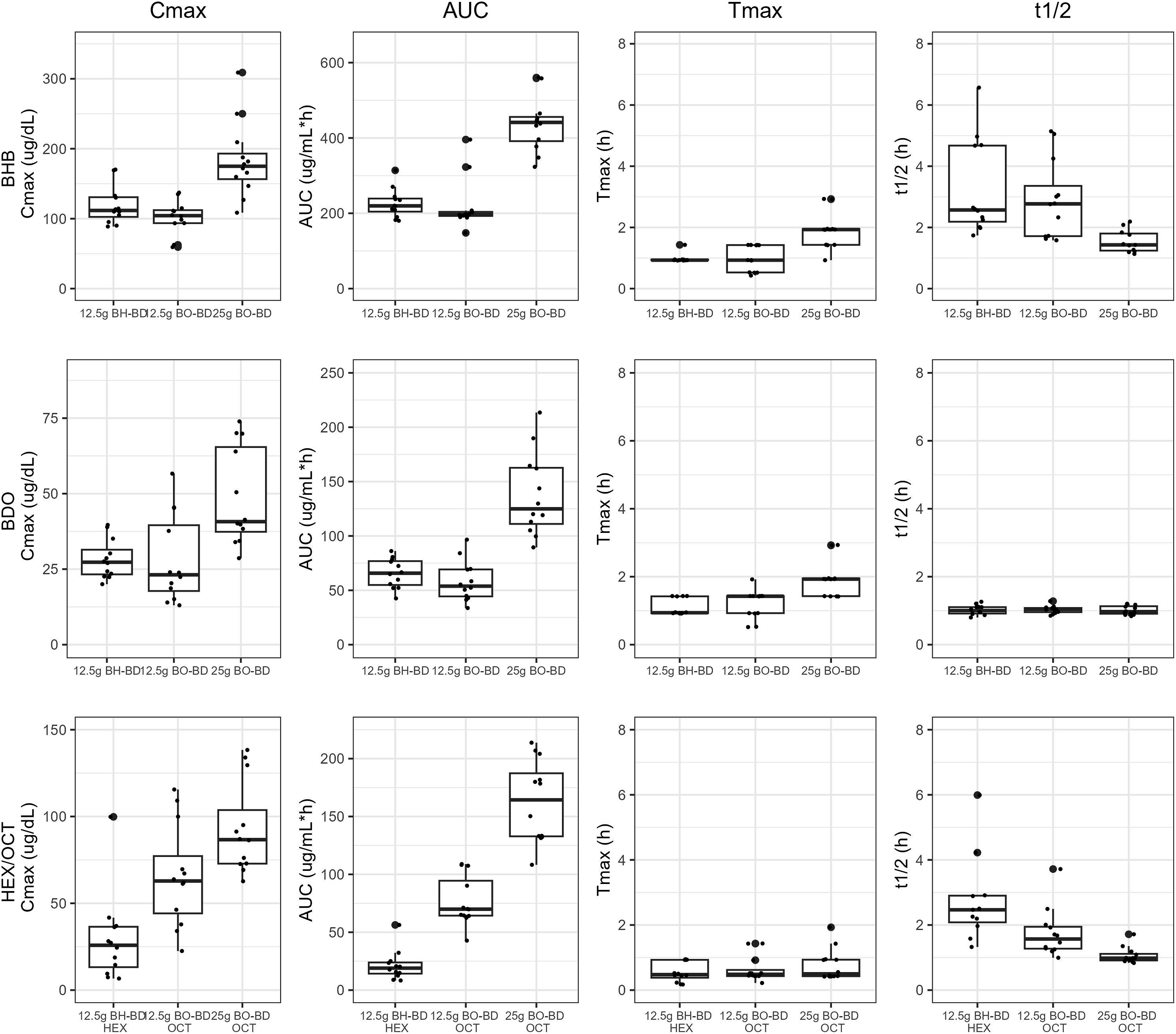
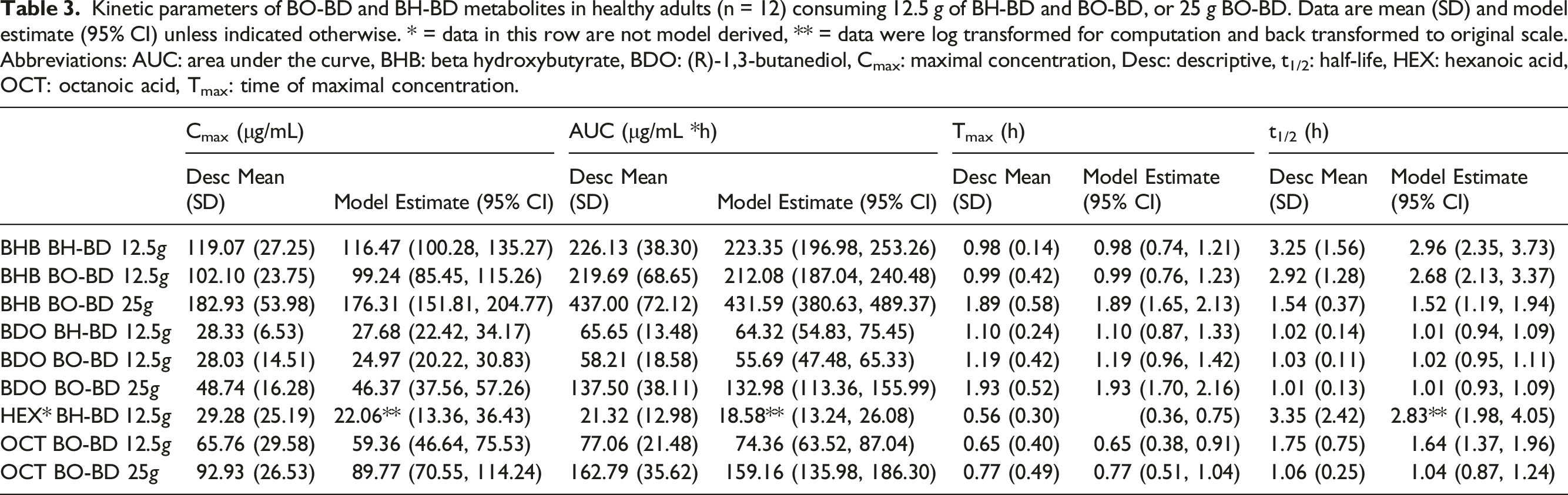
Plasma BHB concentration increased immediately following consumption of all study products and consistently returned to baseline between 4 and 6 h after ingestion (Figure 5). BHB concentration was consistently higher than BDO and OCT or HEX at all time points within each study condition. BHB concentrations in μg/mL were converted to the common unit (mM), data are provided in the Supplemental Information (Table S2, Figure S1). The threshold for nutritional ketosis (BHB 0.5 mM, 52 μg/mL) was reached between 15 and 30 min for all study groups. There was an overall significant effect of study condition on all BHB kinetic parameters (AUC, Time course of BO-BD and BH-BD metabolites, BHB, BDO, OCT and HEX in healthy adults (n = 12) consuming 12.5 Comparison of BO-BD and BH-BD metabolites in healthy adults (n = 12) consuming 12.5 Kinetic parameters of BO-BD and BH-BD metabolites in healthy adults (n = 12) consuming 12.5 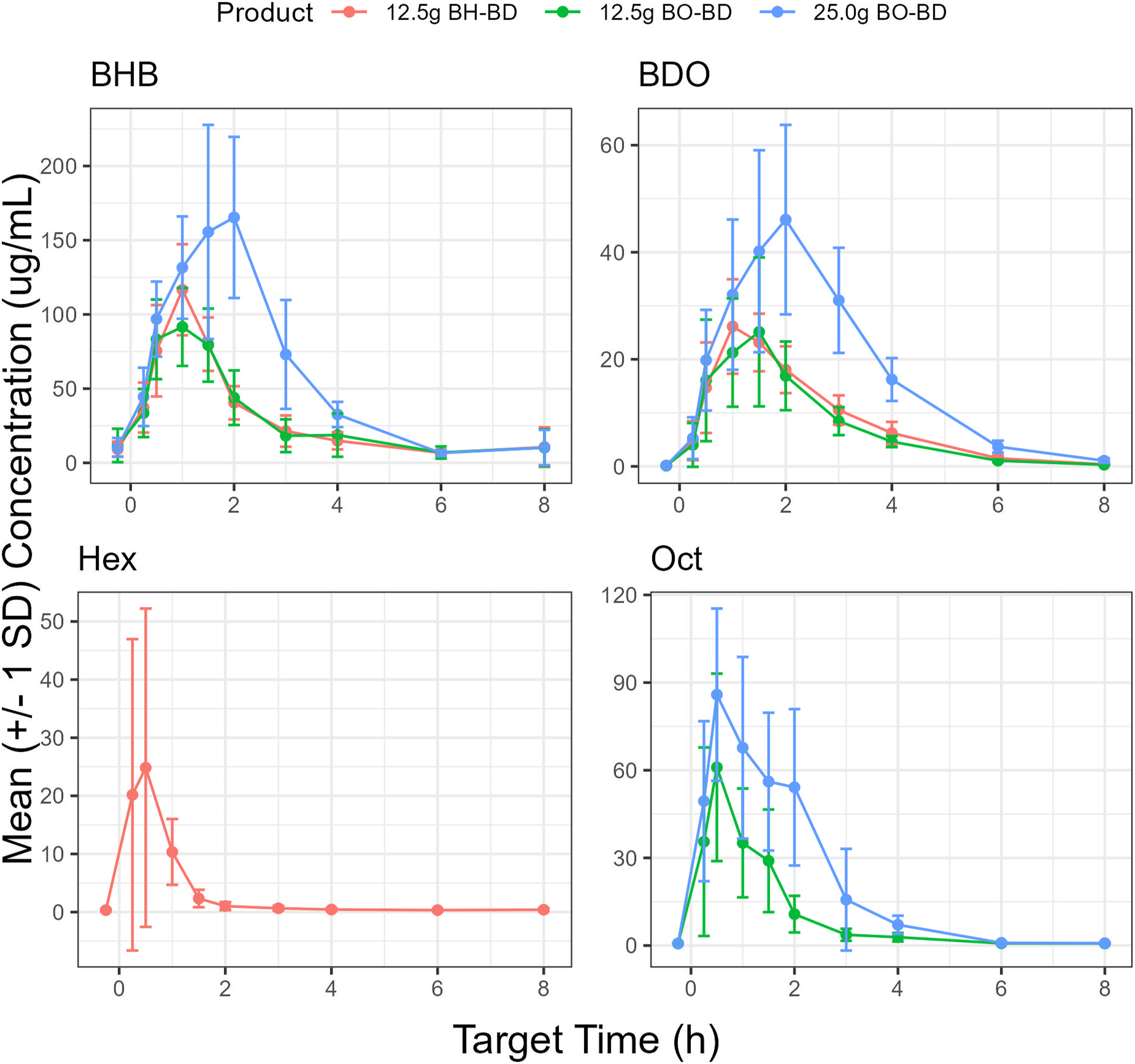
Product acceptability survey scored with a Likert scale between 1 (lowest) and 7 (highest). Results presented are mean score (min, max) for each question and model derived 95% CI. The composite score was calculated as the sum of mean scores for each question. * =

Plasma (R)-1,3-butanediol
Plasma BDO concentrations increased immediately following consumption of all study products and consistently returned to baseline by the end of the study (Figure 5). On average, BDO concentrations were consistently lower than BHB, although they reached maximum concentrations and returned to baseline with comparable kinetics as BHB. BDO and BHB concentrations also tended to remain elevated for longer than OCT or HEX within each dose condition. For both serving sizes of BO-BD study products, BDO had a similar AUC, but lower Cmax than OCT (Figure 6, Table 3, Table S2). For BH-BD, BDO had a higher AUC but similar Cmax to HEX. There was an overall significant effect of condition on all BDO kinetic parameters except for t1/2 (AUC,
In an exploratory analysis, we divided BDO AUC and Cmax by serving size (Table S3) and found that the confidence intervals for Cmax in all intervention groups overlapped indicating a lack of a statistical difference and supported by the overall F-test (
Plasma octanoic and hexanoic acid
Blood OCT and HEX concentrations increased immediately following consumption of BO-BD and BH-BD respectively, and consistently returned to baseline – between 2 and 6 h after study product ingestion (Figure 5). As expected, BH-BD consumption increased plasma HEX concentration with no change in OCT, whereas BO-BD consumption increased plasma OCT concentration, with no change in HEX. Matched serving sizes of BO-BD and BH-BD resulted in similar Tmax, a higher AUC and Cmax, and shorter t1/2 for OCT versus HEX, based on CIs (Figure 6, Table 3, Table S2). Increasing serving size of BO-BD from 12.5 to 25
Product acceptability
BO-BD was rated as more acceptable than BH-BD across all the attributes that were rated (Table 4). In the 12.5
In vitro BO-BD and BH-BD plasma hydrolysis
Following oral ingestion, non-specific lipases and esterases that are secreted into the small intestine rapidly hydrolyze the ester bonds of ketone esters such as BH-BD 29 and BO-BD. Although it is unlikely that intact BO-BD and BH-BD could escape the ubiquitous hydrolase activity in the gut, in vitro hydrolysis experiments were conducted to demonstrate that human plasma esterases also possess the ability to hydrolyze BO-BD and BH-BD.
When mixed and incubated with human plasma at 37°C, BH-BD disappeared rapidly, reaching ∼50% after 15 min of incubation and being almost completely absent in the final sample after 90 min (Figure 7(a)), as seen previously.
29
By contrast, BO-BD was slower to disappear, reaching 58% of the orignal concentration after 90 min of incubation. The concentration of detected fatty acid, HEX and OCT respectively for BH-BD and BO-BD, increased more rapidly in the BH-BD versus BO-BD incubations (Figure 7(b)). A similar pattern was seen with BDO appearance (Figure 7(c)) where concentration increased more quickly in the BH-BD incubation compared to the BO-BD. Metabolism of BO-BD and BH-BD in human plasma in vitro. (a) Percentage of intact BH-BD and BO-BD remaining (b) Concentration of fatty acid (hexanoic - HEX –r octanoic - OCT) in plasma; (c) Concentration of butanediol in plasma. Results of analysis of samples collected from n=2 independent in vitro incubations in human plasma at 37°C.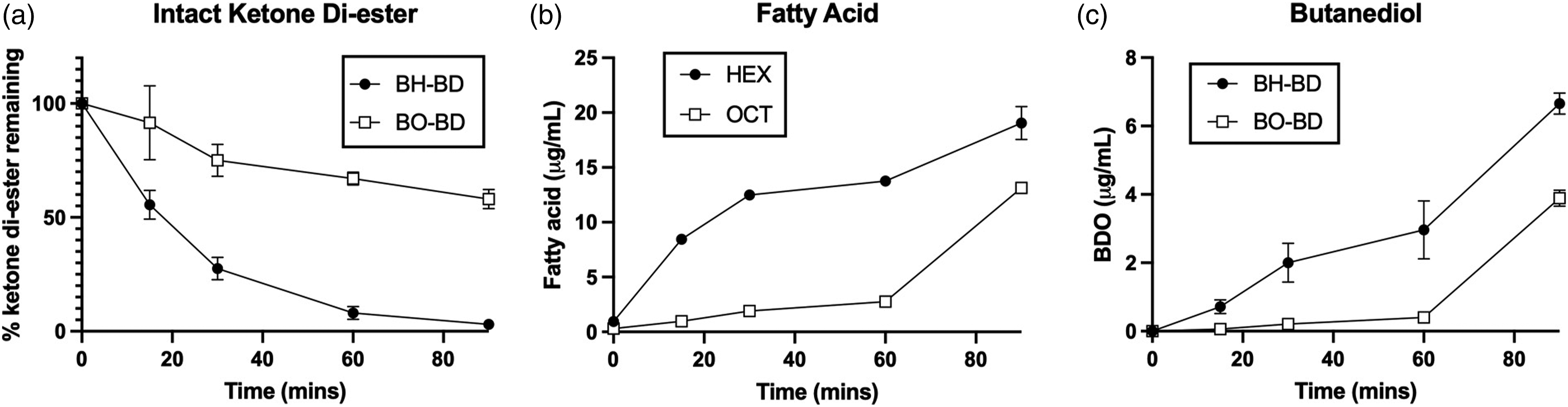
Discussion
This study aimed to investigate the fate of the novel palatable ketone ester BO-BD and the PK of its hydrolysis and metabolic products, BHB, BDO and OCT in healthy adults. To facilitate the safety assessment of the novel ketone ester, BO-BD, we included BH-BD as a qualitative comparator, as it has extensively characterised PK parameters, and is structurally similar. The main findings were firstly, like what was previously demonstrated for BH-BD, 25 BO-BD intake resulted in serving size dependant elevations in blood concentrations of BHB, with smaller increases in BDO and OCT or HEX (for BO-BD and BH-BD, respectively). Secondly, the kinetic parameters of BO-BD metabolites BHB and BDO were similar to a matched serving size of BH-BD, although the BO-BD fatty acid metabolite, OCT, was present at higher concentrations than the BH-BD metabolite, HEX. In a plasma in vitro hydrolysis experiment, it was noted that BO-BD was hydrolysed relatively slower than BH-BD. Finally, a major finding of this study was that acceptability scores for BO-BD were higher than for BH-BD and no adverse events judged to be related to study product were reported. These data indicate BO-BD and BH-BD have similar metabolism with respect to BHB and BDO, therefore given its improved acceptability, BO-BD is a promising nutritional strategy to induce exogenous ketosis for several hours after consumption.
The kinetic parameters of BO-BD observed in this study were similar to the BH-BD comparator in this study, as well as those from previous published work using BH-BD. Crabtree
Given the range of subject body mass for this study (76 – 100 kg) and that other studies of ketone esters have found body mass to be an important co-variate in determining BHB response, 33 we also adjusted BHB AUC and Cmax parameters for serving size adjusted by body mass, and this did not change results or decrease the variability, as we have seen previously in a study of BH-BD. 23 Despite the variability in blood BHB responses, all subjects remained in a state of ‘physiological’ ketosis and did not enter a state of ‘pathological ketoacidosis’ (BHB >10 mM).
A curious disparity between the metabolism of BO-BD and BH-BD is the relatively higher concentration of OCT following BO-BD compared to HEX following BH-BD. The difference persisted when the concentration unit was converted to a molar unit (Table S2), despite the larger molar mass of OCT versus HEX. Both medium chain fatty acids are expected to be rapidly liberated by hydrolysis in the gastrointestinal tract.
29
One possible fate of HEX is conversion to BHB, but as BHB AUCs were similar between matched servings of the two study products, this does not support that HEX was converted to BHB more readily than OCT. The reduced systemic concentration of HEX compared to OCT could potentially be caused by higher gut oxidation, lower gut uptake or lower hepatic oxidation of the HEX medium chain fatty acids. This study was not designed to address the metabolic fate of the hydrolysis products. Importantly, the greater systemic exposure to OCT versus HEX does not affect the safety of BO-BD at its intended use level (two 12.5
The finding that BO-BD exhibited a slower in vitro hydrolysis rate in plasma compared to BH-BD was unexpected, however it likely can be explained by physical differences between the compounds. Firstly, BO-BD is more viscous than BH-BD (BO-BD = 10 cP; BH-BD = 7.6 cP) which may have affected mixing and thus decreased enzyme access to ester bonds. Furthermore, small esters are fairly soluble in water but solubility decreases with increasing chain length, as seen with ethyl methanoate (solubility = 10.5
A major finding of this study was the improvement in sensory acceptance for the novel BO-BD product. The utility of previous exogenous ketone products has been severely limited by their bitter taste and low acceptability.20,21 The greater acceptability of BO-BD compared to BH-BD, therefore, represents a substantial development in this field. The magnitude of improvement seems surprising given the relatively small structural difference between the ketone esters. Bitter taste receptor binding pockets are known to have wide tuning breadth allowing them to interact with many structurally diverse bitter substances. 38 Comparison of the physical properties of OCT and HEX listed on PubChem indicates that OCT has a ‘mild’ or ‘slight’ odour and a ‘slightly sour’ or ‘slightly unpleasant rancid’ taste, 39 whereas HEX odour is described as ‘cheese,’ ‘sweat-like,’ ‘unpleasant’ and a ‘stench’. 40 HEX is also implicated in rancid odours of low-quality Japanese sake products 41 and in the ‘putrid’ odour of Ginko Bilboa fruits. 42 Therefore, these observations support that a HEX-forming ketone ester could have a stronger odour and flavour profile that an OCT-containing ketone ester.
This study had some limitations which should be considered when interpreting the results. Firstly, as this was a pilot study, the sample size was small, the population middle-aged with BMI ranging from normal weight to obese. However, the cross-over design reduces the effect of inter-individual variability and given the replication of previous studies of BH-BD, these results are likely to be generalizable. Secondly, we did not measure plasma concentration of intact ketone ester in the clinical study, as our previous work with BH-BD demonstrated rapid hydrolysis in intestinal and liver homogenates, as well as plasma. 29 Additionally, only trace BH-BD concentrations were detected in rats that ingested the ketone di-ester. 29 We also did not measure the secondary blood ketone body, acetoacetate due to its known lack of stability in stored samples. 43
In conclusion, we found that whilst BO-BD has a lower plasma hydrolysis rate in vitro, as compared to BH-BD, metabolite kinetics in our clinical study were very similar and the acceptability of the beverage was significantly improved. The data from this study supports the appropriateness of extrapolating between existing data for BH-BD to inform the safety assessment of BO-BD and its incorporation into ketosis-inducing nutritional products.
Supplemental Material
Supplemental Material - A randomized, open-label, cross-over pilot study investigating metabolic product kinetics of the palatable novel ketone ester, bis-octanoyl (R)-1,3-butanediol, and bis-hexanoyl (R)-1,3-butanediol ingestion in healthy adults
Supplemental Material for A randomized, open-label, cross-over pilot study investigating metabolic product kinetics of the palatable novel ketone ester, bis-octanoyl (R)-1,3-butanediol, and bis-hexanoyl (R)-1,3-butanediol ingestion in healthy adults by Brianna J Stubbs, Chad Cook, Traci M Blonquist, Kristen Taggart, Dawn Beckman, Claire Kruger, Dietrich Conze, and Amy C Boileau in Toxicology Research and Application
Footnotes
Acknowledgements
The authors wish to acknowledge Alan Xu, PhD of Keystone Bioanalytics for exceptional bioanalytic services for this study.
Author contributions
Conceptualization, C.K., D.C., A.B., B.J.S.; methodology, A.B., B.J.S.; formal analysis, T.B.; investigation, C.C., K.T., D.B.; data curation, T.B., B.J.S., writing—original draft preparation, B.J.S.; writing—review and editing, C.K., D.C., A.B., C.C., K.T., T.B.; visualization, T.B., B.J.S.; project administration, B.J.S., A.B C.C., K.T.; funding acquisition, A.B, B.J.S. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.J.S. has stock options in BHB Therapeutics Ltd., and Juvenescence Ltd. B.J.S. is an inventor on patents related to the use of ketone bodies. A.B. is an employee of Juvenescence US Corp., and has stock options in Juvenescence Ltd and Juvenescence Life Sciences Ltd. All other authors have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by BHB Therapeutics (Ireland) Limited (“BHB Therapeutics”), which is licensed to develop products related to ketone bodies. BHB Therapeutics provided the study beverages used in this work.
Ethical approval statement
Data Availability Statement
The data presented here may be available upon reasonable request from the corresponding author and in accordance with intellectual property considerations ![]() .
.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.