Abstract
Diabetic ketoacidosis (DKA) is a life-threatening complication of diabetes mellitus (DM). The standard method of detection of ketone bodies is the dipstick method, which detects semiquantitatively acetoacetate, but not β-hydroxybutyrate (β-HB). The objectives of the current study were to assess the diagnostic utility of β-HB to diagnose diabetic ketosis (DK) and DKA in cats and to establish a cut-off value for the diagnosis of DKA. Sixty-two cats were included in the study. Eleven cats were healthy (group 1); in the remainder of cats (51), a diagnosis of DM was based on hyperglycemia, glucosuria, and increased fructosamine concentrations. Nineteen of 51 cats suffered from nonketotic diabetes mellitus (group 2). In 11 cats, plasma ketone bodies were detected with the dipstick method (diabetic ketosis, group 3). In 21 cats, plasma ketone bodies and metabolic acidosis were present (DKA, group 4). Plasma β-HB was measured in all cats by an enzymatic method (spectrophotometry). A cut-off value for the diagnosis of DKA was calculated based on the receiver operating characteristic curve. In healthy cats, the β-HB concentration ranged from 0 to 0.1 mmol/l; in cats of group 2, from 0 to 0.9 mmol/l (median: 0.1 mmol/l); in cats of group 3, from 0.6 to 6.8 mmol/l (median: 1.7 mmol/l); and in cats of group 4, from 3.8 to 12.2 mmol/l (median: 7.9 mmol/l). A cut-off value of 2.4 mmol/l revealed 100% sensitivity and 87% specificity to diagnose DKA. Beta-hydroxybutyrate is a useful parameter for the diagnosis of diabetic ketosis and DKA in cats.
Introduction
Ketone bodies are produced by the liver and are used as an energy source when glucose is not available. The 2 main ketone bodies are acetoacetate and β-hydroxybutyrate (β-HB), while acetone is the least abundant ketone body. 13 Beta-hydroxybutyrate is derived from the reduction of acetoacetate in the mitochondria of the liver, and acetone is generated by spontaneous decarboxylation of acetoacetate. Patients suffering from diabetic ketoacidosis (DKA) display hyperglycemia, glucosuria, ketonuria, ketonemia, and metabolic acidosis. 9 The most popular method for the detection of ketone bodies is the urine dipstick method. Commercial ketone tests for urine are based on the Legal reaction, in which acetoacetate reacts with nitroprusside to produce a purple-colored complex on the test strip. If glycine is added to the test reagent, the Legal reaction can also detect acetone. However, β-HB cannot be detected by using this method. 13 Measurement of ketones in blood rather than in urine helps eliminate the risk of false negatives due to insensitivity and false positives due to drug interference. 24 In urine samples, the nitroprusside reaction has been reported to give false-positive results in the presence of drugs containing sulfhydryl groups. False-negative readings have been reported when the test strips have been exposed to air for an extended time, after large intakes of vitamin C, or when bacteria in the urine metabolize acetoacetate. 17 Moreover, the measurement of ketone bodies is influenced by renal function. 8 Another difficulty is the subjective evaluation of the color change of the urine stick. In human beings, the ketone body ratio in DKA is initially 3:1 (β-HB:acetoacetate) and rises to as high as 10–20:1, leading to an underdiagnosis of hyperketonemia when testing for ketone bodies is performed by an urine stick only.10,15 During ketosis, β-HB levels increase more than levels of acetone and acetoacetate, making β-HB a more sensitive marker of ketosis. 24 Moreover, β-HB is a more stable ketone body than acetone or acetoacetate. 14 In response to insulin therapy, β-HB levels commonly decrease long before acetoacetate levels. 13 In human medicine, quantitative measurement of capillary β-HB is performed routinely.4,11,12 In feline medicine, measurement of β-HB using serum, plasma, or capillary blood has been described only in a few studies to date.5,22–24 The objectives of the current study were to assess the diagnostic utility of β-HB to diagnose diabetic ketosis (DK) and DKA, employing the hypothesis that cats with DKA have higher β-HB concentrations compared to cats with DK, and to establish a cut-off value for the diagnosis of DKA.
Material and methods
Animals
Blood samples were collected from 11 healthy cats and from 51 cats with diabetes mellitus (DM) presented to the Clinic of Small Animals, Faculty of Veterinary Medicine, Freie Universität (Berlin, Germany). All cats admitted to the hospital between March 2009 and July 2010 with the diagnosis of DM were included in the study. Diabetes mellitus was diagnosed based on clinical signs, hyperglycemia in association with glucosuria, and increased fructosamine concentrations. 9 Cats were excluded in cases of insufficient blood samples. In cats with DK and DKA but not in cats with nonketotic diabetes mellitus (NKD), plasma ketone bodies were also detected with the urine dipstick method. In addition to the detection of ketone bodies, cats with DKA were acidotic (HCO3- < 15 mmol/l; anion gap ≥ 25 mmol/l). 9 Healthy cats that were presented for blood donation were used as controls, as they were considered healthy based on history and results of physical and laboratory examinations.
Blood collection and handling
Blood was collected from the cephalic, saphena, or external jugular vein by use of a 24-gauge needle and was placed in a heparinized tube. a Plasma was obtained by centrifugation (5 min at 8,000 × g). b The heparinized plasma was stored at –45°C for a maximum of 5 months until β-HB was determined. The study protocol was not approved by an animal use and care committee because cats provided samples as a part of their routine blood donor health check or as part of their diagnostic evaluation at the hospital. The rest of the plasma samples were used for the analysis of β-HB.
Beta-hydroxybutyrate measurement
Beta-hydroxybutyrate was measured by an enzymatic spectrophotometric methodc,d by an accredited veterinary laboratory. e Beta-hydroxybutyrate, in the presence of hydroxybutyrate dehydrogenase, is oxidized to acetoacetate with the concomitant reduction of NAD+ to NADH. The NADH is re-oxidized to NAD+ by a redox mediator, which was directly proportional to the β-HB concentration. The coefficient of variation of the reference method was 3.7% between runs and 8.8% within runs. e
Blood gas analysis
Venous blood was drawn from all cats by special blood gas syringes f ; pH, pCO2, pO2, and lactate were measured in all cats within 15 min. The base excess and HCO3- concentrations were calculated in consideration of the the rectal temperature. g The chloride concentration was measured by an automatic analyzer. h The anion gap was calculated according to the following formula: (Na++ K+) – (Cl-+ HCO3-). 7 Blood gas parameters, glucose, and lactate were measured with an automated analyzer using ion selective electrodes. g
Ketone body measurement by the dipstick method
Ketone bodies (acetoacetate, acetone) were semiquantitatively assessed in plasma by the use of urine dipsticks.i,24 According to the package insert, color changes of 1+, 2+, or 3+ correspond to concentrations of 1 mmol/l, 5 mmol/l, or 15 mmol/l, respectively. A color change of (+) was interpreted as 0.5 mmol/l.
Statistical analysis
Descriptive statistics were used to report the results of the β-HB concentration, glucose, pH, HCO3-, base excess, lactate, anion gap, and ketone bodies measured with the urine dipstick. SPSS 17 was used as statistical software. j A Mann–Whitney test was performed to compare the β-HB concentrations between the healthy cats and the cats with DM. Moreover, the values of DKA cats were compared to the values of the cats with DK. P-values ≤ 0.01 were considered significant. A receiver operating characteristic (ROC) curve was constructed from β-HB values measured with the reference method. Sensitivity and specificity were calculated with MedCalc statistical software. k The cut-off value for the diagnosis of DKA and the area under the curve were calculated based on the ROC curve.
Results
Animals
Nineteen cats suffered from NKD (group 2), 11 from DK (group 3), and 21 from DKA (group 4). The results of the blood gas analysis, glucose, and ketone measurements are presented in Table 1.
Plasma glucose, pH, HCO3-, base excess, lactate, anion gap, β-hydroxybutyrate (β-HB) concentrations, and plasma ketone bodies (dipstick method) in 11 healthy cats and 51 cats with diabetes mellitus.
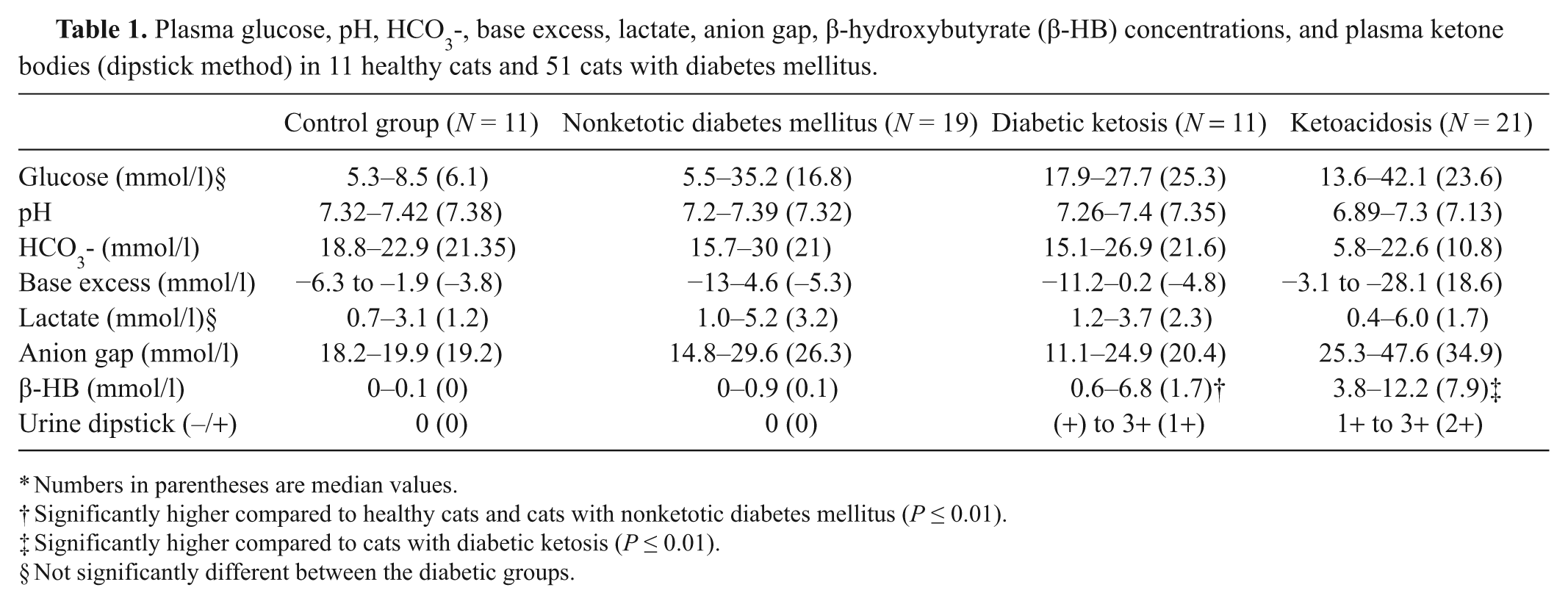
Numbers in parentheses are median values.
Significantly higher compared to healthy cats and cats with nonketotic diabetes mellitus (P ≤ 0.01).
Significantly higher compared to cats with diabetic ketosis (P ≤ 0.01).
Not significantly different between the diabetic groups.
Control group (group 1)
Nine of the cats were Domestic Shorthair cats, 1 was a Norwegian Forest Cat, and 1 a Maine Coon. The ages ranged from 2 to 11 years (median: 5 years). Two cats were female (both spayed), and 9 were males (8 neutered). The β-HB concentration in the 11 healthy cats ranged from 0 to 0.1 mmol/l (median: 0 mmol/l). The urine stick was negative for plasma ketone bodies in all cats in group 1.
Group 2 (nonketotic diabetes mellitus)
Group 2 was composed of Domestic Shorthair cats, and their ages ranged from 9 to 17 years (median: 14 years). Ten cats were female (all spayed), and 9 cats were male (all neutered). Six of 19 cats had chronic renal failure, 2 cats had hypertrophic cardiomyopathy (HCMP), 3 cats had pancreatitis, and 4 cats were diagnosed with pemphigus foliaceus, inflammatory bowel disease, cat flu, and hyperthyroidism, respectively. In 4 cats, there was no concurrent disease present. Twelve cats were on insulin treatment. Cats with NKD had β-HB concentrations ranging from 0 to 0.9 mmol/l (median: 0.1 mmol/l). The dipstick method revealed no ketone bodies in plasma; however, in 8 cats, β-HB concentrations were increased (0.2–0.9 mmol/l, median: 0.4 mmol/l) compared with healthy cats. One cat suffering from severe pancreatitis and thoracic effusion died. This cat had a β-HB concentration of 0.5 mmol/l and an increased lactate concentration (5 mmol/l, pH 7.19).
Group 3 (diabetic ketosis)
Group 3 included 9 Domestic Shorthair cats, 1 British Shorthair, and 1 mixed-breed cat. The ages ranged from 7 to 15 years (median: 12 years). Five cats were female (all spayed), and 6 cats were male (all neutered). In 5 cats with DK, the following additional diseases were detected: HCMP (n = 2), pancreatitis (n = 2), and cachexia with severe icterus (n = 1). Six of the 11 cats were already on insulin treatment. The cats in group 3 had β-HB values ranging from 0.6 and 6.8 mmol/l (median: 1.7 mmol/l). All cats had ketone bodies in their plasma when tested by the urine stick ((+) to 3+, median: 1+). Seven cats with a dipstick reaction of (+) to 1+ had β-HB values ranging from 0.6 to 6.8 mmol/l (median: 1.2 mmol/l). In 2 cats, the dipstick reaction was 2+ (β-HB: 2.4 and 5.6 mmol/l) and 3+ (β-HB: 5.2 and 6.5 mmol/l), respectively. Nine cats survived. Two cats were euthanized for financial reasons.
Group 4 (diabetic ketoacidosis)
Group 4 was composed of 20 Domestic Shorthair cats and 1 Chartreux. The ages ranged from 6 to 13 years (median: 9 years). Six cats were female (all spayed), and 15 cats were male (all neutered). Sixteen of the 21 cats had additional diseases: pancreatitis (n = 8), HCMP (n = 3), hepatopathy (n = 2), atopy and glucocorticoid therapy (n = 1), and IBD, glucocorticoid therapy, and chronic renal failure (n = 1). Eight of the 21 cats were on insulin therapy at the time of presentation. The cats in group 4 had β-HB-concentrations ranging from 3.8 to 12.2 mmol/l (median: 7.9 mmol/l). In all 21 cats, plasma ketone bodies were also detected with the urine stick (1+ to 3+, median: 2+). Four cats had β-HB values ranging from 3.8 to 7.6 mmol/l (median: 6.2 mmol/l), and the dipstick reaction was weak (1+). In 11 cats with a dipstick reaction of 2+, the β-HB concentrations ranged from 4.2 to 12.2 mmol/l (median: 8 mmol/l). In 6 cats with dipstick reactions of 3+, the β-HB concentrations ranged from 4.9 to 10.4 mmol/l (median: 8.9 mmol/l). Ten cats of this group survived. Seven cats were euthanized as requested by their owners. Four cats died. These 4 cats had β-HB values of 5.7, 6.3, 6.7, and 6.8 mmol/l, respectively. The β-HB concentrations were significantly higher in DKA cats compared to DK cats (P < 0.001).
Receiver operating characteristic analysis
All cats with DKA (group 4) had β-HB values ≥ 3.8 mmol/l. In 26 of 30 cats (86.7%) with NKD or DK, β-HB values were <2.4 mmol/l, the other 4 cats had values of 5.1, 5.2, 6.5, and 6.8 mmol/l, respectively. An area under the curve of 0.97 was determined by ROC analysis (Fig. 1). At a cut-off value of 2.4 mmol/l, the sensitivity and specificity was 100% and 87%, respectively, for the diagnosis of DKA (Table 2).
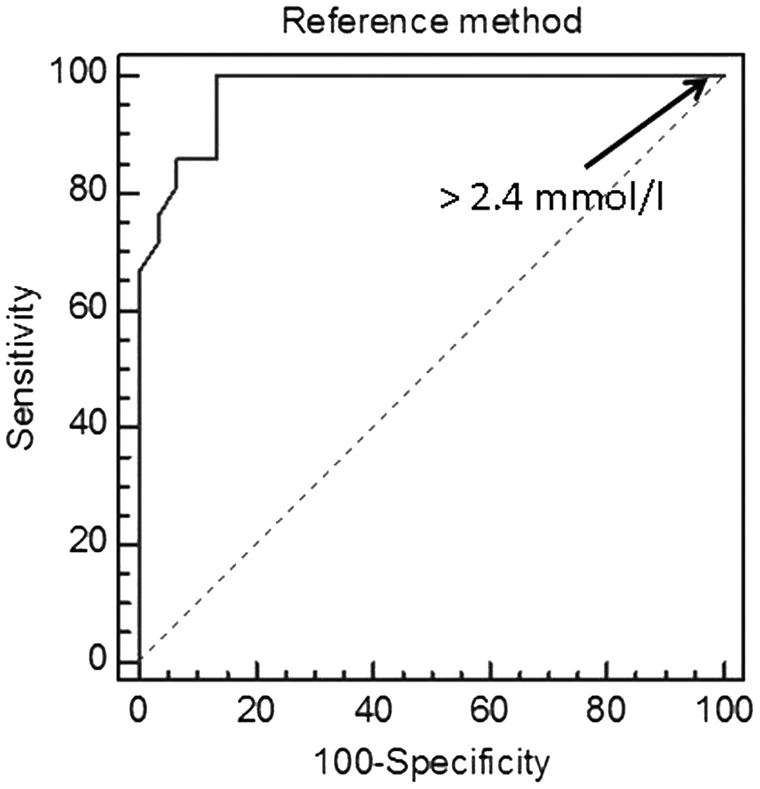
Receiver operating characteristic curve depicting the diagnostic value of the spectrophotometric method to detect increased β-hydroxybutyrate concentrations in cats. The area under the curve is 0.97.
Criterion values and coordinates of the receiver operating characteristic curve.*
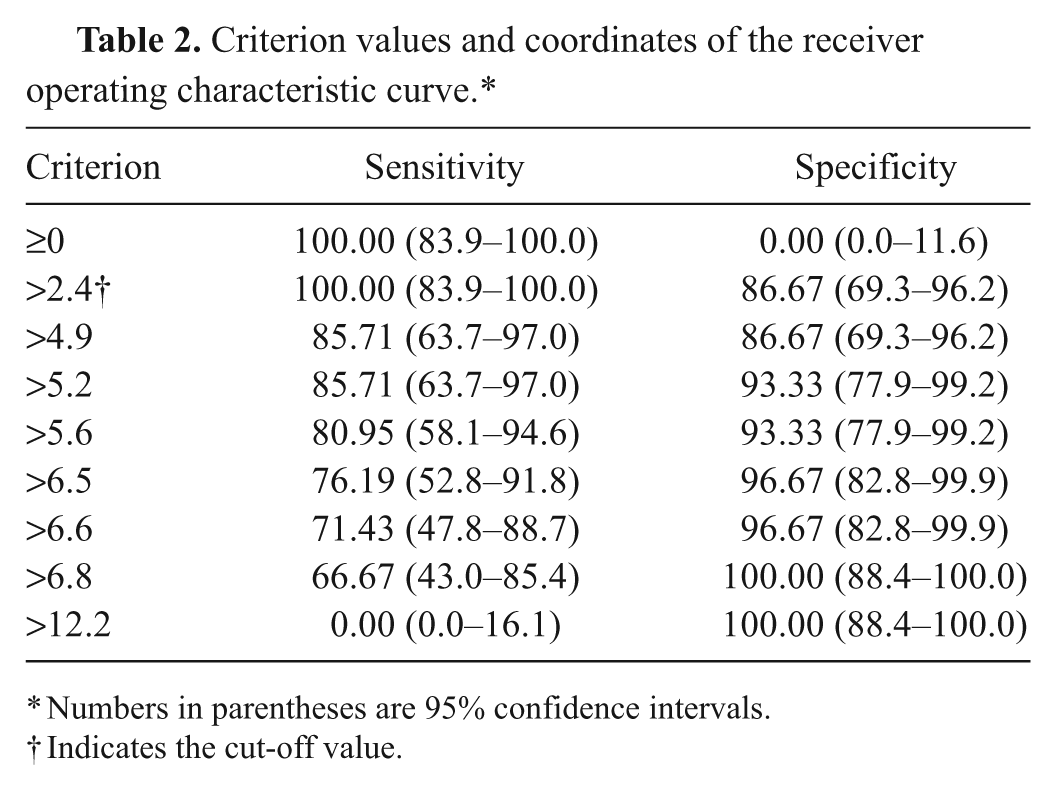
Numbers in parentheses are 95% confidence intervals.
Indicates the cut-off value.
Discussion
In human medicine, monitoring of ketone bodies is recommended in the guidelines of the American Diabetes Association and the International Diabetes Federation, and the determination of β-HB in blood is widely preferred over urine testing. 21 In feline medicine, the traditional measurement of acetoacetate predominates, although determination of β-HB in blood has been published.5,22–24 Healthy cats have significantly lower β-HB concentrations compared to diabetic cats. Therefore, β-HB can be used as a marker for DM. 23 In a study published in 2009, 24 β-HB concentration was measured in the plasma of 54 cats with the hydroxybutyrate esterase assay. Cats with NKD (n = 3) had β-HB concentrations of 0.02, 0.04, and 0.05 mmol/l, while those suffering from DK (n = 40) had concentrations between 0.06 and 8.5 mmol/l (median: 0.72 mmol/l) and those suffering from DKA (n = 11) had β-HB values ranging from 6.5 to 17.6 mmol/l (median: 11 mmol/l). 24 The data of NKD and DK cats were similar compared to the results of the current study. However, the β-HB values of the cats with DKA were higher, which may be explained by differences in the assay.
The plasma samples in the present study were stored for up to 5 months at −45°C. The influence of storage was not evaluated. Human EDTA plasma samples were stored at room temperature, 4°C, −17°C, and ∓80°C. Within 21 days, a significant decrease in β-HB was detected after storage at room temperature or at 4°C, but not at −17°C or below. 14 To the authors’ knowledge, data from veterinary medicine are not available.
The results of the present study revealed that the determination of urine ketone bodies using only the nitroprusside reaction can underestimate the number of cats with ketosis. In 8 cats with a β-HB concentration of 0.2–0.9 mmol/l, the dipstick reaction was negative. The dipstick reaction was lower in 5 (1+/β-HB: 5.1–7.6 mmol/l; 2+/β-HB: 12 mmol/l) and higher in 3 (3+/β-HB: 4.9-6.5 mmol/l) cats compared to the β-HB concentration. The quantitative measurement of β-HB is more useful for follow-ups of cats with DK or DKA compared to the dipstick method.
Similar to human patients, β-HB may be the predominant ketone body in cats, and the measurement of β-HB is an important parameter for the diagnosis of ketonemia. In human medicine, treatment guidelines based on the level of β-HB concentration exist.6,12,18–20 Beta-hydroxybutyrate levels do not normally exceed 1 mmol/l in patients who have no evidence of metabolic decompensation, regardless of the prevailing glucose concentration. Human beings with β-HB levels higher than 3 mmol/l are at risk of DKA, and immediate medical advice is recommended. 19 In another human study, a cut-off value of 3 mmol/l revealed 100% sensitivity and 88% specificity to diagnose DKA. 12 In human medicine, the change in β-HB during treatment of DKA is also interpreted. Optimal treatment would reduce β-HB by 1 mmol/l per hour; failure to do so suggests inadequate treatment. 20
In the present study, a cut-off value of 2.4 mmol/l revealed 100% sensitivity and 87% specificity for the diagnosis of DKA in cats, which is similar to the cut-off value in human beings. This cut-off value led to false-positive results in 13% of the cases and to no false-negative results. This means that in 4 cats, DKA was suspected when only DK was present. The suspicion of DKA would lead to a more intensive monitoring and treatment, which is not harmful for the cat. In the case of a false-negative result, however, a life-threatening situation may be missed. In cats with a β-HB concentration < 2.4 mmol/l, DKA is very unlikely.
In 4 out of 30 cats with DK, β-HB values were higher than 2.4 mmol/l. One of these cats suffered from severe HCMP, 2 cats from acute pancreatitis, and the fourth cat was cachectic and severely icteric. An increased catabolism due to diseases such as pancreatitis, hepatic lipidosis, or other metabolic disturbances might have caused increased concentrations of β-HB in these 4 cats, even though there was no acidosis present (Aroch I, Shechter M, Segev G: 2009, A retrospective case-controlled study of serum betahydroxybutyric acid in 215 ill cats: clinical and clinical-pathological findings, diagnosis, morbidity and mortality. Proceeding of the 19th Annual European College of Veterinary Internal Medicine Congress, September 10–12, Porto, Portugal). In a prospective study, it was shown that the β-HB concentration increased (up to 1.57 mmol/l) in 8 obese cats after a fasting period of 5–6 weeks. 1 Moreover, the elimination of ketone bodies is regulated by renal function and may be increased in cases of renal failure.2,6 However, none of these 4 cats was azotemic. Another cat in group 2 (NKD) had metabolic acidosis (pH 7.19), but a negative dipstick test. This cat had lactic acidosis and was classified as NKD, although the β-HB concentration was 0.5 mmol/l.
Limitations of the current study are the criteria to define DKA. Although DKA is normally defined as a triad composed of hyperglycemia, acidosis, and ketosis, specific values for glucose, bicarbonate, and blood pH vary in the veterinary literature. 16 This lack of standardization could have contributed to the overlap in β-HB concentrations among the groups. Furthermore, cats with DKA frequently are presented with conditions such as protracted vomiting, diarrhea, and dehydration that may cause mixed acid-base disturbances. In human medicine, it is assumed that anion gap, HCO3-, and pH are relatively nonspecific for DKA because they can be affected by the degree of respiratory compensation or the presence of a separate acid disturbance. Ketone body production on the other hand is accompanied by equimolar production of hydrogen ions. 18 Another limitation of the study is the usage of venous blood samples for the determination of the acid-base status. Technical difficulties in drawing arterial blood samples from cats were the cause for this approach. A study in human medicine, however, concluded that venous blood gas measurements accurately demonstrate the degree of acidosis of patients with DKA. 3
Blood ketone testing methods that quantify β-HB are not immediately available for every practitioner. The measurement of β-HB in an external laboratory is time-consuming. Portable devices used in human medicine can also be used in veterinary medicine. 22 In a previous study, it was shown that in cats a portable ketone meter l was accurate for the quantitative measurement of β-HB concentrations <4 mmol/l. 22 Values higher than 4 mmol/l β-HB concentrations were underestimated. However, data from the portable ketone meter tested cannot be extrapolated to other instruments; each portable device has to be evaluated individually.
The measurement of β-HB is a useful parameter for the diagnosis of DKA. A cut-off value of 2.4 mmol/l revealed 100% sensitivity and 87% specificity to diagnose DKA. If a blood gas analysis cannot be performed, measuring the β-HB concentration allows for a diagnosis of DKA if β-HB concentrations are beyond the cut-off value. But it is important to note that there is no single test that can be used for a diagnosis of DKA on its own.
Footnotes
Acknowledgements
The authors would like to thank Laboklin, Bad Kissingen, Germany, for performing the β-HB measurements with the spectrophotometric method.
a.
Microtube Li-Heparin LH/1.3, Sarstedt AG & Co, Nümbrecht, Germany.
b.
Heraeus Pico 21 centrifuge, Fisher Scientific GmbH, Schwerte, Germany.
c.
Ranbut, Randox Laboratories Ltd, Crumlin, County Antrim, United Kingdom.
d.
Modular P 800, Roche Diagnostics GmbH, Mannheim, Germany.
e.
Laboklin GmbH & Co.KG, Bad Kissingen, Germany.
f.
Blood gas syringes, Instrumentation Laboratory, SC Sanuis Counting GmbH, Nümbrecht, Germany.
g.
GemPremier3000, Instrumentation Laboratory, Kirchheim, Germany.
h.
Konelab 30 i, Fisher Scientific GmbH, Schwerte, Germany.
i.
Combur9 Test, Roche Diagnostics GmbH, Mannheim, Germany.
j.
SPSS, IBM Deutschland GmbH, Ehningen, Germany.
k.
MedCalc Software, Mariakerke, Belgium.
l.
Precision Xceed, Abbott GmbH & Co. KG, Ludwigshafen, Germany.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
