Abstract
Polysorbate 80 is a non-ionic detergent derived from polyethoxylated sorbitan and oleic acid. It is widely used in pharmaceuticals, foods, and cosmetics as an emulsifier. Nav1.7 is a peripheral sodium channel that is highly expressed in sympathetic and sensory neurons, and it plays a critical role in determining the threshold of action potentials (APs). We found that 10 μg/mL polysorbate 80 either abolished APs or increased the threshold of the APs of dorsal root ganglions. We thus investigated whether polysorbate 80 inhibits Nav1.7 sodium current using a whole-cell patch-clamp recording technique. Polysorbate 80 decreased the Nav1.7 current in a concentration-dependent manner with a half-maximal inhibitory concentration (IC50) of 250.4 μg/mL at a holding potential of −120 mV. However, the IC50 was 1.1 μg/mL at a holding potential of −90 mV and was estimated to be 0.9 μg/mL at the resting potentials of neurons, where most channels are inactivated. The activation rate and the voltage dependency of activation of Nav1.7 were not changed by polysorbate 80. However, polysorbate 80 caused hyperpolarizing shifts in the voltage dependency of the steady-state fast inactivation curve. The blocking of Nav1.7 currents by polysorbate 80 was not reversible at a holding potential of −90 mV but was completely reversible at −120 mV, where the channels were mostly in the closed state. Polysorbate 80 also slowed recovery from inactivation and induced robust use-dependent inhibition, indicating that it is likely to bind to and stabilize the inactivated state. Our results indicate that polysorbate 80 inhibits Nav1.7 current in concentration-, state-, and use-dependent manners when used even below commercial concentrations. This suggests that polysorbate 80 may be helpful in pain medicine as an excipient. In addition, in vitro experiments using polysorbate 80 with neurons should be conducted with caution.
Introduction
Polysorbate 80 (polyoxyethylene sorbitan monooleate), also known as Tween 80, is a synthetic non-ionic surfactant that is widely and commercially used in biochemical applications, such as solubilizing proteins and emulsifying and dispersing substances in medical and food products.1–4 Polysorbate 80 inhibits aggregation and minimizes surface absorption at the air-water interface and upon dilution into intravenous fluids. The concentration of polysorbate 80 as an excipient ranges from 0.01–2 mg/mL, corresponding to 10–2000 ppm. 4 Although polysorbate 80 has been associated with several systemic reactions, such as hypersensitivity, nonallergic anaphylaxis and rash, and injection- and infusion-site adverse events such as pain, erythema, and thrombophlebitis,5–9 whether polysorbate 80 affects the excitability or ion channel activity of neurons has not been investigated.
Voltage-gated sodium channels are essential for electrogenesis in excitable cells. In humans, they exist as nine sodium channel isoforms and display different kinetics and voltage-dependent properties, and their different deployment in different types of neurons endows these cells with distinct firing properties. 10 Among these channels, Nav1.7, Nav1.8, and Nav1.9 are preferentially and selectively expressed in the peripheral nervous system, particularly nociceptive dorsal root ganglions (DRG).11,12 The expression of Nav1.7 was first identified in sympathetic and somatosensory ganglion neurons and has since been found in myenteric neurons, olfactory sensory neurons, visceral sensory neurons, and smooth myocytes. 13 Activated by small, slow ramp stimuli, Nav1.7 amplifies generator potentials toward the action potential (AP) threshold and acts as a threshold channel in nociceptive neurons. Nav1.8 generates slow TTX-resistance sodium currents 11 and carries the largest inward charge during the upstroke of the AP.14,15 Nav1.9 produces persistent sodium current in DRG neurons and appears to modulate resting membrane potential. 16
We unexpectedly found that APs in many small-diameter nociceptive DRG neurons disappeared after perfusion with 10 μg/mL polysorbate 80 as a control condition for evaluating drug efficacy. As polysorbate is a common excipient used in therapeutic protein formulas, we investigated how polysorbate 80 at the commercially used concentration could diminish neuronal excitability.
Materials and methods
Primary dorsal root ganglions culture
Adult male Sprague Dawley rats (6-7 weeks old) were used according to the Catholic University of Korea Institutional Animal Care and Use Committee guidelines. L4-L6 DRG neurons were isolated and incubated in a complete saline solution (in mM: 137 NaCl, 5.3 KCl, 1 MgCl2, 25 D-sorbitol, 3 CaCl2, and 10 HEPES at a final pH of 7.2, adjusted with NaOH), as described previously by Rizzo et al. 17 and enzymatically digested with collagenase A (1 mg/mL; Roche, Indianapolis, IN) and then with collagenase D (1 mg/mL; Roche) and papain (30 U/mL; Worthington, Lakewood, NJ) for 20 min at 37°C, and then gently centrifuged (900 rpm for 3 min). Pellets were triturated in 1:1 DMEM/F-12 media (Thermo Fisher Scientific, Waltham, MA) with 10% FBS and 100X Penicillin-Streptomycin (Thermo Fisher Scientific) containing bovine serum albumin (1.5 mg/mL; Sigma, S Louis, MO) and trypsin inhibitor (1.5 mg/mL; Sigma). Cells were plated on 12 mm Poly-D-Lysine/Laminin coated glass coverslips (Corning, Corning, NY). The electrophysiological recording was started after 2 hours when DRG cells were stabilized and attached well on coverslips.
Cell culture and preparation of Nav1.7
Human embryonic kidney (HEK293) cells stably expressing human Nav1.7 purchased from Millipore (CYL3011; Billerica, MA) were cultured in Dulbecco’s modified Eagle’s medium (Thermo Fisher Scientific) supplemented with 10% fetal bovine serum (Thermo Fisher Scientific) in an incubator. Cells were maintained in a humidified atmosphere at 37°C with 5% CO2-enriched air. Cells were passaged every 2-3 days by brief trypsinization. Cells treated with 0.25% trypsin-EDTA (Thermo Fisher Scientific) were plated on 12 mm glass coverslips (Fisher Scientific, Pittsburgh, PA) in 35 mm cell culture dishes. The electrophysiological recording was conducted after 24 hours.
Whole-cell patch-clamp recordings
Whole-cell patch-clamp recordings were performed with Multiclamp 700B amplifiers (Molecular Devices, Sunnyvale, CA) for the current-clamp experiments at room temperature (22–25°C). The internal solution contained (in mM) 140 KCl, 1 EGTA, 10 NaCl, 10 HEPES, and 2 ATP-Mg at a final pH of 7.3 using KOH (osmolarity was adjusted to 310 mOsmol/L with sucrose). The external solution contained (in mM) 140 NaCl, 3 KCl, 1 MgCl2, 1 CaCl2, 10 glucose, and 10 HEPES at a final pH of 7.3 using NaOH (osmolarity was adjusted to 320 mOsmol/L with sucrose). Small-diameter DRG neurons (≤30 μm in diameter) were selected as most small-diameter neurons are known to be nociceptive neurons.
Voltage-clamp recordings of Nav1.7 were obtained using an Axopatch 200B amplifier (Molecular Devices) using the following solutions: the internal solution contained (in mM) 140 CsF, 10 NaCl, 1 EGTA, and 10 HEPES at a final pH of 7.3 using CsOH (osmolarity was adjusted with sucrose); the external solution for the Nav1.7 sodium currents contained (in mM) 140 NaCl, 3 KCl, 1 MgCl2, 1 CaCl2, and 10 HEPES at a final pH of 7.3 using NaOH (osmolarity was adjusted with sucrose).
Pipettes were pulled from soft glass capillaries (World Precision Instruments, Sarasota, FL) using a programmable horizontal microelectrode puller (Sutter Instrument Co., Novato, CA) and were polished using a microforge. The resistances of pipettes were 1.5–2.0 MΩ and 0.9–1.2 MΩ when filled with the internal solution for current-clamp and voltage-clamp recordings, respectively. During the perfusion of the external bath solution, pipettes were wrapped with parafilm to reduce the capacitance. The coverslips containing adherent cells were on the glass bottom of the chamber and maintained at 22°C using a temperature controller (Dagan Corporation, Minneapolis, MN). Cells were not considered for analysis if they had a high leakage current (holding current > 500 pA at a holding potential of −90 mV) or an access resistance greater than 2.0 MΩ.
The current-clamp recordings were started when the resting membrane potential was stabilized, and then the cells were continuously perfused with either the external solution or 10 μg/mL polysorbate 80 for 5 min before collecting data. Voltage-clamp data were recorded after 7 min perfusion with either the external solution or 10 μg/mL polysorbate 80. The currents were filtered at 5 kHz and the sampling rate was 100 kHz. Voltage errors were minimized using a 90% series resistance compensation in all recordings. Voltage-clamp pulses and data acquisition were controlled using pClamp 10.7 Software (Molecular Devices) and Digidata 1440A acquisition board (Molecular Devices).
Compound application
Tween™ 80 Surfact-Amps™ Detergent Solution (10% (w/v) solution) was purchased from Thermo Fisher Scientific and diluted with the standard external bath solution to obtain the desired concentration. Polysorbate 80 solutions were made fresh before patch-clamp recordings and applied through a gravity-driven system with a perfusion pencil (Automate Scientific, Berkeley, CA), allowing rapid perfusion of the recording chamber. During the recording, the cells were continuously perfused with the test solution.
Protocols and data analysis
For current-clamp recordings, APs were elicited by three types of current injections: 500 ms long-step current, 50 ms short-step current, and 4 pA/ms ramp current. Long-step current injections were used to measure the current threshold and the number of APs at 500 pA current injection. Short-step current injections were used to analyze the shape of APs and the voltage threshold.
For voltage-clamp recordings, the concentration-dependent inhibition of currents by polysorbate 80 was elicited using a 100 ms depolarizing pulse of 0 mV from various holding potentials (−120, −110, −105, −100, and −90 mV) for Nav1.7 at 5 s intervals. Data were best fitted to the logistic equation Y = 1/[1 + IC50/D]p using OriginPro Software, where IC50 is the 50% inhibition of polysorbate 80 concentration, D is the concentration of polysorbate 80, and p is the Hill coefficient.
Whole-cell Nav1.7 currents were elicited by using a 50 ms depolarizing pulse at potentials between −70 mV and +40 mV in steps of +5 mV with a holding potential of −90 mV at 5 s intervals. Peak currents at each voltage step were normalized into conductance (G) according to the relationship V = IR, I = G(V/Vrev), where Vrev is the reversal potential of sodium current. This was used to analyze the voltage dependency of activation. Steady-state inactivation was determined using a test pulse of +10 mV following 500 ms conditioning pulses between −170 mV and −30 mV with a holding potential of −90 mV. The voltage-dependent activation curves and steady-state inactivation curves were fitted by the Boltzman equation: G/Gmax (or I/Imax) = 1/[1 + exp(Vm−V1/2)/k]], where Vm is the test (or conditioning) potential, V1/2 is the midpoint of activation (or inactivation) curve, and k is the slope factor of the curve. Recovery from inactivation was measured at each peak current in response to a step of 0 mV, which was preceded by a 50 ms pulse of 0 mV and a recovery period with variable durations and potentials. Use-dependent inhibition was determined using 20 repetitive 50 ms depolarizing pulses of −10 mV with a holding potential of −90 mV at 0.5, 3, and 10 Hz.
Statistics
Student’s t-tests were used for statistical analysis. All averaged and normalized data are presented as the mean ± SE. A value of p < 0.05 was considered statistically significant. Data acquisition and analysis were performed using pClamp 10.7 software (Molecular Devices) and OriginPro 8.5 Software (Origin Lab Corp., Northampton, MA).
Results
Alteration of action potential characteristics by polysorbate 80 in rat dorsal root ganglions neurons
Properties of AP in rat DRG neurons in the absence (n = 7) and presence (n = 7) of 10 μg/mL polysorbate 80.
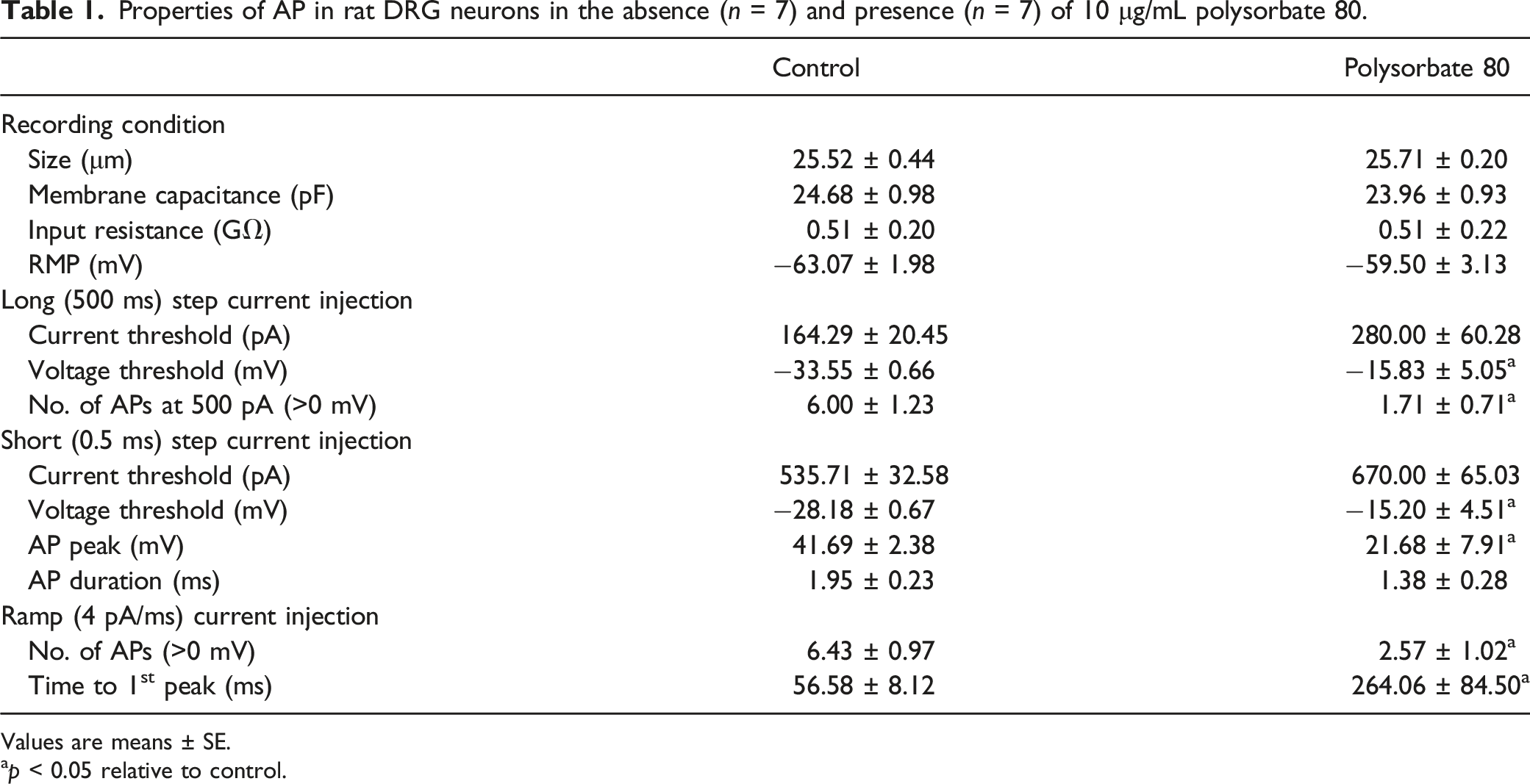
Values are means ± SE.
ap < 0.05 relative to control.

APs recorded by the current clamp experiments in the absence (left) and presence (right) of 10 μg/mL polysorbate 80. AP characteristics are summarized in Table 1. Representative traces for 500 pA of 500 ms long step current (a), 0.5 ms short step current (b), and 4 pA/ms ramp current (c).
To identify whether polysorbate 80 affects the characteristics of AP shape (Figure 1(b)), we injected a short (0.5 ms) minimum suprathreshold current to generate a single AP while also minimizing the effect of the injected stimulus current. The current threshold was not significantly changed and the voltage threshold was increased by polysorbate 80, similar to those by long current injection. While the duration of AP was not changed by polysorbate 80, the peak amplitude of AP was significantly lowered, indicating that polysorbate 80 may primarily inhibit sodium channels but not potassium channels.
We also analyzed the APs generated by slow ramp current injections, which mimic physiological stimulation (Figure 1(c)). Due to the adaptation of APs, we only counted overshooting APs (AP peaks >0 mV). When polysorbate 80 was present, the number of APs significantly decreased, and it took longer to generate the first AP in instances where they were produced. Moreover, the cells which did not produce any APs by long injection current, never showed any AP, even by the most robust ramp current injection. Therefore, these results suggest that polysorbate 80 decreases neuronal excitability by increasing current and voltage thresholds.
Inhibition of Nav1.7 currents by polysorbate 80 is concentration-dependent and increases at more depolarized holding potentials
Nav1.7 is known as a threshold sodium channel for pain perception in human beings.11,18 Therefore, we investigated whether polysorbate 80 inhibits human Nav1.7 current. Polysorbate 80 decreased peak currents of Nav1.7 in a concentration-dependent manner at a holding potential of −120 mV with a half-maximal inhibition concentration (IC50) of 250.4 ± 61.3 μg/mL and a Hill coefficient of 0.49 ± 0.04 (Figure 2(a), n = 7). As the IC50 was higher than we expected and our previous report
19
showed that some sodium channel blockers produced a potent blocking effect at the depolarized membrane potentials where sodium channels exist at more inactivated state, we also tested at a holding potential of −90 mV (Figure 2(b)). The IC50 for polysorbate 80 was 1.1 ± 0.1 μg/mL (n = 8) and the Hill coefficient was 1.02 ± 0.04. Interestingly, depolarized membrane potential not only increased the efficacy of polysorbate 80 but also brought the Hill coefficiency close to 1. To estimate the IC50 at the most depolarized holding potential, we obtained IC50 at various membrane potentials. The IC50 at holding potentials of −110, −105, and −100 mV were 70.8 ± 14.2 (n = 10), 17.2 ± 1.9 (n = 5), and 4.3 ± 0.5 (n = 7), respectively (Figure 2(c)). The Hill coefficients at holding potentials of −110, −105, and −100 mV were 0.59 ± 0.02, 0.70 ± 0.02, and 0.82 ± 0.06, respectively. When the holding potential was closer to the average resting membrane potential of DRG neurons (i.e. approximately −60 mV, Table 1), the value of IC50 was lower, and the slope of the graph was steeper. We assumed that polysorbate 80 may bind with Nav1.7 at a 1:1 ratio with a holding potential of −90 mV, but when the holding potential is hyperpolarized and closer to the resting state for ion channels, there are more binding sites which also show negative cooperativity. Based on a previous report,
20
we measured the maximal and minimal IC50 of polysorbate 80 to the closed state at rest (1/Kr) and the inactivated state (1/Ki). Means of IC50 values calculated from Figure 2(c) at each holding potential were plotted and fitted with Kapp = 1/[h/Kr + (1 − h)/Ki)], where h and 1-h are the fraction of channels in the resting and inactivated states, respectively. The IC50 values for Kr and Ki were 207.40 μg/mL and 0.95 μg/mL, indicating that IC50 values at the resting membrane potentials were approximately 1 μg/mL. Concentration- and voltage-dependence of polysorbate 80. Representative whole-cell Nav1.7 currents in the absence and presence of polysorbate 80 at holding potentials of −120 mV (a) and −90 mV (b). (c) The normalized peaks of Nav1.7 currents are plotted as a function of polysorbate 80 concentration. Dose-response curves at holding potentials of −120 mV (■, n = 8), −110 mV (□, n = 7), −105 mV (●, n = 5), −100 mV (○, n = 10), and −90 mV (▲, n = 7) were fitted with a logistic equation. (d) The values of IC50 were calculated from Figure 2(c); the fitted line is the best fit to the equation Kapp = 1/[h/Kr + (1 − h)/Ki)] where h and 1 − h are the fractions of channels in the resting and inactivated states, respectively.
Polysorbate 80 has a robust binding affinity in the inactivated state of Nav1.7
We tested whether the effects of polysorbate 80 on Nav1.7 were reversible. To normalize the blocking effect of polysorbate 80, Nav1.7 currents continuously perfused with the control solution were initially recorded at a holding potential of −120 mV to measure the maximal peak current (Figure 3(a), ■) and then changed to a holding potential of −90 mV (★). After stabilizing the sodium current at a holding potential of −90 mV, 10 μg/mL polysorbate 80 was applied until the current reduction was stable in at least three consequent traces. The sodium current was inhibited by 82.9 ± 4.3% (n = 4) of peak current (•) with two exponential phases. A double exponential function well fitted the fast and slow components of inhibition, of which the time constants were 19.0 ± 14.1 and 120.2 ± 31.2 s, respectively. The block of Nav1.7 currents was partially recovered after washout with a time constant of 28.9 ± 1.6 s at a holding potential of −90 mV (▲). However, the time constant was much faster (3.8 ± 0.4 s) when the holding potential was changed to −120 mV (◆), and 94.1 ± 0.4% of currents were recovered. These results indicate that polysorbate 80 preferentially binds to the inactivated state of Nav1.7 and readily dissociates from the resting state of it. Reversible inhibition of Nav1.7 currents by polysorbate 80. (a) Time course of Nav1.7 current inhibition by 10 μg/mL polysorbate 80 at a holding potential of −90 mV (●) after applying the same test pulse from holding potentials of −120 mV (■) and −90 mV (★) in the absence of polysorbate 80 and washout effect at holding potentials of −90 mV (▲) and −120 mV (◆). (b) representative Nav1.7 current traces for blocking and washout by polysorbate 80.
Polysorbate 80 hyperpolarized the voltage-dependency of steady-state fast inactivation of Nav1.7
We investigated the effect of polysorbate 80 on the voltage dependence of activation and steady-state fast inactivation. As the membrane potential of neuronal cells is approximately −60 mV, we used 1 μg/mL polysorbate 80 at a holding potential of −90 mV. Figure 4(a) shows representative traces of Nav1.7 in the absence (top) and presence (bottom) of 1 μg/mL polysorbate 80. When the voltage-dependent activation curves (Figure 4(b)) were fitted to the Boltzmann distribution equation, the midpoint of activation (V1/2) and slope (k) values were −26.88 ± 0.94 mV and 6.51 ± 0.09 mV in the absence of polysorbate 80 (n = 9), respectively, and −26.77 ± 0.75 mV and 6.54 ± 0.28 mV in the presence of 1 μg/mL polysorbate 80 (n = 11), respectively. Therefore, the voltage dependency of the activation was not changed by polysorbate 80. We also examined the rates of activation (Figure 4(c)) and inactivation (Figure 4(d)) kinetics, indicating closed-to-open and open-to-inactivated transitions, respectively. At all test potentials, activation and inactivation kinetics were similar in the absence and presence of polysorbate 80. Contrary to the voltage-dependency of activation, the voltage-dependency of steady-state fast inactivation was more hyperpolarized than in the absence of polysorbate 80 (Figure 4(e)). The value of V1/2 in the presence of 1 μg/mL polysorbate 80 (−100.66 ± 3.06 mV) was significantly more hyperpolarized than in the absence of polysorbate 80 (−80.72 ± 0.93 mV). In addition, the k of the steady-state fast inactivation curve in the presence of polysorbate 80 (10.41 ± 0.29 mV) was shallower than in its absence (5.19 ± 0.18 mV). Effect of polysorbate 80 on channel kinetics at a holding potential of −90 mV. Whole-cell Nav1.7 currents were elicited by a 50 ms depolarizing pulse to potentials between −70 and +40 mV in steps of +5 mV at 5 s intervals. (a) The representative whole-cell Nav1.7 channel current trace in the absence (upper) and presence (lower) of 1 μg/mL polysorbate 80. (b) The activation curves were fitted to the Boltzmann distribution equation under control conditions (□, n = 9) and in the presence of 1 μg/mL polysorbate 80 (●, n = 11). Time-to-peak (c) and inactivation kinetics (d) as a function of test potentials in control conditions (□, n = 9) and in the presence of 1 μg/mL polysorbate 80 (●, n = 11). (e) Steady-state inactivation curves were determined using a test pulse at +10 mV after 500 ms conditioning pulses to voltages between −140 and −30 mV (for the control solution) and −170 and −30 mV (for 1 μg/mL polysorbate 80). Data were fitted to the Boltzmann distribution equation under control conditions (□, n = 7) and in the presence of 1 μg/mL polysorbate 80 (●, n = 7).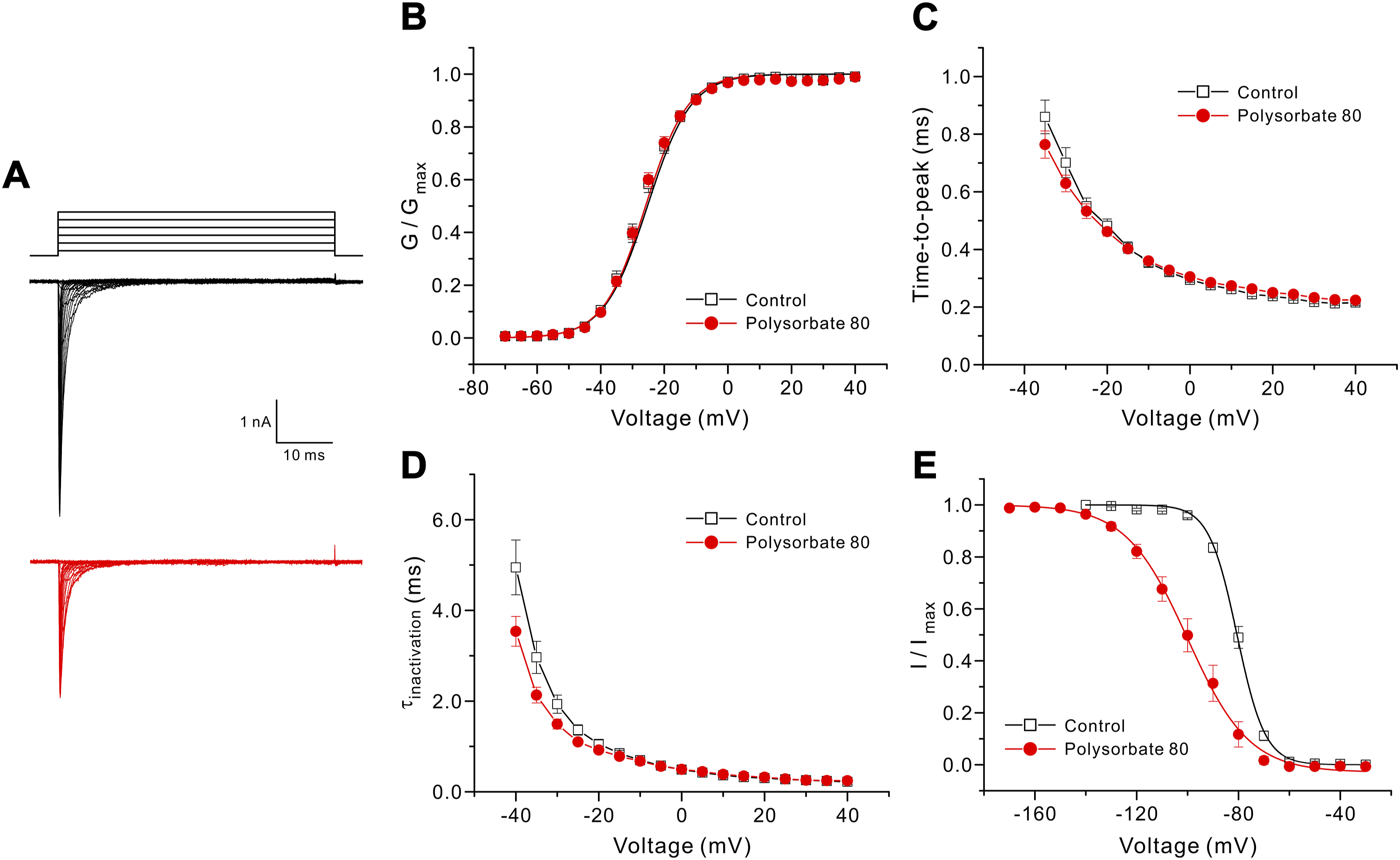
Nav1.7 channels in the presence of polysorbate 80 recover slowly from inactivation
The high potency at depolarized holding potentials and the hyperpolarized shifts in the steady-state fast inactivation curve suggested that Nav1.7 may slowly recover from inactivation and that channels remain inactivated for longer time periods. To confirm this hypothesis, we used a double-pulse protocol that measured the peak current in response to a step to 0 mV, which was preceded by a 50 ms pulse to 0 mV and a recovery period of 2.5, 5, 10, 20, 100, 500, 1,000, or 3000 ms at −90 mV (Figure 5(a)) or −120 mV (Figure 5(b)). The kinetics of recovery from inactivation in the absence of polysorbate 80 was fitted to a monoexponential function and the presence of polysorbate 80 was well fitted to a biexponential function. As expected, the time constant of recovery from fast (τfast) and slow (τslow) components of Nav1.7 current in the presence of polysorbate 80 at −90 mV (τfast: 49.7 ± 12.1 s, τslow: 1201.1 ± 248.0 s, n = 5) and −120 mV (τfast: 46.4 ± 20.6 s, τslow: 717.5 ± 129.1 s, n = 6) were significantly slower than in the absence of polysorbate 80 (42.2 ± 5.1 s at −90 mV and 5.7 ± 0.6 s at −120 mV, n = 7). Interestingly, the recovery of the Nav1.7 current reaches almost four times the normalized response of the control (Figure 5(b)), implying that long recovery periods at −120 mV induced the unbinding of polysorbate 80, which is shown in Figure 3, in addition to the recovery from inactivation of Nav1.7. Recovery from inactivation and use-dependent inhibition of Nav1.7. Recovery from inactivation was measured at peak current in response to a step to 0 mV, which was preceded by a 50 ms pulse at 0 mV and a recovery period with variable duration and potential. Recovery from inactivation at −90 mV (a) and −120 mV (b) under control conditions (□, n = 7) and in the presence of 1 μg/mL polysorbate 80 (●, n = 5–6). The data were fitted using monoexponential (control) and bi-exponential (polysorbate 80) functions. Use-dependent inhibition of Nav1.7 at different frequencies in the presence and absence of 1 μg/mL polysorbate 80. Use-dependent inhibition was determined using 20 repetitive 50 ms depolarizing pulses to −10 mV with a holding potential of −90 mV at 0.5, 3, and 10 Hz. (c) Use-dependent fall-off of peak currents under control conditions (open symbols, n = 6–8) and in the presence of 1 μg/mL polysorbate 80 (closed symbols, n = 4–5) at 0.5 (squares), 3 (circles), and 10 (triangles) Hz. (d) The percentage decrease in the peak current between the 1st and 20th pulse of each frequency stimulation protocol under control conditions and in the presence of 1 μg/mL polysorbate 80 (*, p < 0.05).
Polysorbate 80 produced strong use-dependent inhibition of Nav1.7
As polysorbate 80 slowed recovery from inactivation, we assumed that it might promote the accumulation of inactivated Nav1.7 at higher frequencies of depolarization pulses in the presence of polysorbate 80. We investigated the use-dependent inhibition of polysorbate 80 at frequencies of 0.5, 3, and 10 Hz in the absence and presence of 1 μg/mL polysorbate 80 (Figure 5(c)). In the absence of polysorbate 80, the peak amplitude of Nav1.7 was decreased by 6.2, 22.7, and 43.1% at 0.5, 3, and 10 Hz, respectively (Figure 5(d)). However, in the presence of polysorbate 80, the peak amplitude of Nav1.7 decreased by 14.6, 66.7, and 91.6% at 0.5, 3, and 10 Hz, respectively. These results indicate that polysorbate 80 produced strong use-dependent inhibition in proportion to the stimulation frequency.
Discussion
Small molecule drugs have been developed as major therapeutics for nearly a century because most can be administered orally and can pass through cell membranes to reach intracellular targets. 21 Recently, biological therapeutics such as antibodies and mRNA vaccines, including traditional vaccines, have been highlighted due to their high specificity, and many are now commercially available in the pharmaceutical market.2,4,22 They are designed to engage biological targets by various modes of action, but need adjuvants, solubilizers, or stabilizers of emulsions to prevent surface adsorption while also stabilizing against protein aggregation. Polysorbate 80 is the most common surfactant used in antibody formulations and is widely used in pharmaceuticals, foods, and cosmetics as an emulsifier.2,22 Its concentration for drug formulation ranges from 10–2000 μg/mL, while for antibody formulations, the contents must be higher than the critical micelle concentration, which is approximately 17 μg/mL. 4 Therefore, most commercial contents of polysorbate 80 that require micelle formation must be higher than the IC50 (0.9 μg/mL) we observed in this study (Figure 2).
Polysorbate 80 is a mixture of isomeric organic compounds composed of sorbitan derivatives containing one oleic acid (>58%) or other fatty acids. 4 Oleic acid is an unsaturated fatty acid with one double bond and occurs naturally in animal and vegetable fats and oils. It has been reported that polyunsaturated fatty acids act on many voltage-gated ion channels. 23 Monounsaturated fatty acids such as oleic acid also act on voltage-gated sodium channels by reducing their current and hyperpolarizing the voltage dependency of steady-state inactivation,23–26 which is similar to our findings. It has been reported that there are five binding sites of polyunsaturated fatty acids in ion channels. 23 Among these, site 1 is located in the intracellular cavity of sodium channels, and the binding of polyunsaturated fatty acids reduces the sodium current by inducing hyperpolarization of the steady-state inactivation curve. All other binding sites (site 2–5) are found in potassium or calcium channels, and the effects are not similar to our results. The Hill coefficiencies in this study were smaller than 1 at holding potentials lower than −100 mV, suggesting that the binding sites in the Nav1.7 channel for polysorbate 80 are likely multiple and have negative cooperativity. This is unsurprising as polysorbate 80 is not a pure compound; thus, more than one unsaturated fatty acid residue of polysorbate 80 may bind to multiple sites and produce multiple blocking effects. The on-rate of blocking Nav1.7 current by polysorbate 80 at a holding potential of −90 mV showed biphasic decay (Figure 3). However, the off-rate was fitted well by monoexponential functions individually at each holding potential, indicating that there are at least two binding sites: one in the closed state with a low affinity and the other in the inactivated state with high affinity. This hypothesis is consistent with a lower IC50 at more depolarized holding potentials, suggesting that polysorbate 80 prefers to bind to the inactivated state and block Nav1.7 current by stabilizing it. Taken together, the portion of unsaturated fatty acid in polysorbate 80 may be the primary component that accounts for our results. However, even though our hypothesis is well fitted with the reduction of nociceptive neuronal excitability, we cannot exclude the potential effect of polysorbate 80 on other nociceptive sodium channels such as Nav1.8 and Nav1.9. Among these, Nav1.8 is especially known to contribute to most of the current underlying the depolarizing phase of the APs generated by nociceptive neurons, 27 which is why we can hypothesize that polysorbate 80 may also block this channel. However, it might be challenging to identify the blocking mechanism in detail for Nav1.8 in a matter similar to our present study due to the difficulty of its heterologous expression.
In this study, although we found that polysorbate 80 has dramatic effects on the neuronal excitability of nociceptive DRG neurons at a widely-used concentration (10 μg/mL = 10 ppm = 0.001% = 7.63 μM), there is no obvious evidence yet that polysorbate 80 induced abnormalities of the sympathetic and somatosensory system.6–9 It might be that the bulky hydrophilic sorbitan derivatives prevent rapid diffusion, not allowing for a systemic effect. However, polysorbate 80 kept isolated neurons from generating APs and directly blocked the threshold sodium channel of Nav1.7 that is expressed in the HEK293 cell line in a concentration-, state-, and use-dependent manner. Thus, in vitro experiments using polysorbate 80 with neurons must be cautiously conducted. As Nav1.7 is preferentially expressed in the sympathetic and somatosensory ganglion neurons and the channels are mostly inactivated at resting membrane potentials, polysorbate 80 might be useful as an excipient for pain medicine.
Footnotes
Author contributions
REK performed the electrophysiology, analyzed the data, and prepared the figures and tables. JSC designed the experiment and evaluated the data. REK and JSC wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2016R1A2B4011333, NRF-2018R1A6A1A03025108).
