Abstract
Since the possible roles of surface modifications in determining multi-walled carbon nanotube (MWCNT)–promoted endoplasmic reticulum (ER) stress-mediated lipid-laden macrophage foam cell formation are still in debate, we compared unmodified and carboxylated MWCNT-induced cytotoxicity, lipid profile changes, and expression of ER stress genes in THP-1 macrophages. Particularly, we focused on lipid profile changes by using lipidomics approaches. We found that unmodified and carboxylated MWCNTs significantly decreased cellular viability and appeared to damage the cellular membrane to a similar extent. Likewise, the results from Oil Red O staining showed that both types of MWCNTs slightly but significantly induced lipid accumulation. In keeping with Oil Red O staining results, lipidomics data showed that both types of MWCNTs up-regulated most of the lipid classes. Interestingly, almost all lipid classes were relatively higher in carboxylated MWCNT-exposed THP-1 macrophages compared with unmodified MWCNT-exposed cells, indicating that carboxylated MWCNTs more effectively changed lipid profiles. But in contrast to our expectation, none of the MWCNTs significantly induced the expression of ER stress genes. Even, compared with carboxylated MWCNTs, unmodified MWCNTs induced higher expression of lipid genes, including macrophage scavenger receptor 1 and fatty acid synthase. Combined, our results suggested that even though carboxylation did not significantly affect MWCNT-induced lipid accumulation, carboxylated MWCNTs were more potent to alter lipid profiles in THP-1 macrophages, indicating the need to use omics techniques to understand the exact nanotoxicological effects of MWCNTs. However, the differential effects of unmodified and carboxylated MWCNTs on lipid profiles might not be related with the induction of ER stress.
Keywords
Introduction
Multi-walled carbon nanotubes (MWCNTs) were the firstly discovered carbon-based tubular nanomaterials (NMs). 1 Since their discovery in 1991, MWCNTs have been widely used in many important areas, such as environmental protection (eg, pollutant removal),2,3 sensors (eg, immunosensors),4,5 and medicine (eg, drug delivery),6,7 leading to the direct contact of humans with MWCNTs. However, the potential toxicity of MWCNTs raised health concerns.8,9 In particular, it is necessary to evaluate the interactions between MWCNTs and immune cells, such as macrophages, because NMs including MWCNTs entering human bodies will interact with macrophages and consequently induce immunotoxicity (reviewed in the references 10, 11). Indeed, it has been suggested that exposure to NMs including MWCNTs could induce macrophage foam cell formation to promote cardiovascular disease development (reviewed in the references 12, 13), although the mechanisms remain poorly understood.
It is well-accepted that the toxicological effects of MWCNTs are heavily dependent on the physicochemical properties of MWCNTs (reviewed in the references 14, 15). Interestingly, it seems that the physicochemical properties also played an important role to determine MWCNT-macrophage interactions. For example, Cao et al reported that MWCNTs with larger hydrodynamic sizes were more potent than the ones with smaller hydrodynamic sizes to induce macrophage foam cell formation both in vitro and in atherosclerotic mice. 16 Yang et al found that decreasing diameters or increasing surface areas increased MWCNT-induced cytotoxicity and lipid accumulation in THP-1 macrophages. 17 Fujita et al reported that longer MWCNTs were generally more cytotoxic to macrophages compared with shorter ones. 18 Cohignac et al reported that only tubular-structured MWCNTs but not spherical carbon nanoparticles (NPs) induced autophagic dysfunction in macrophages. 19 While previous studies generally showed a role of shape of MWCNTs, the role of surface modifications in determining MWCNT-macrophage interactions is unclear. Zhang et al reported that surface modifications increased the bioactivity of MWCNTs to murine macrophages. 20 In contrast, Long et al recently reported that unmodified, carboxylated, and hydroxylated MWCNTs induced cytotoxicity and lipid accumulation in THP-1 macrophages to similar levels. 21 However, since MWCNTs used in that study only carried low levels of carboxyl/hydroxyl groups, it may be difficult to investigate the exact roles of surface modifications. 21
Carboxyl modification is commonly used to improve the stability and biocompatibility of MWCNTs.14,15 The present study compared unmodified and carboxylated MWCNT-induced cytotoxicity, lipid profile changes, and expression of endoplasmic reticulum (ER) stress genes in THP-1 macrophages. To this end, THP-1 macrophages were exposed to various concentrations of unmodified or carboxylated MWCNTs. Then, cytotoxicity was evaluated based on cell counting kit-8 (CCK-8) assay, and the morphologies of MWCNT-exposed macrophages were observed by using transmission electron microscope (TEM). The changes of lipid profiles were measured based on Oil Red O staining as well as lipidomics. To investigate the possible mechanisms, the expression of genes associated with ER stress was measured by real-time RT-PCR.
Materials and Methods
THP-1 Cell Culture
THP-1 cell line was purchased from the American Type Culture Collection (Manassas, VA, USA) and were cultured in RPMI 1640 medium (Gibco, NY, USA) supplemented with 10% fetal bovine serum (FBS; GIBCO, Australia) and 1% penicillin/streptomycin (P/S solution; Beyotime, Nantong, China). The cells were differentiated into macrophages by overnight treatment with 10 ng/mL phorbol 12-myristate 13-acetate (PMA; Sigma, St Louis, MO, USA). After overnight differentiation, THP-1 macrophages were immediately used for the following experiments. After the differentiation, THP-1 cells became adherent and did not proliferate anymore.
Multi-Walled Carbon Nanotube Characterization and Exposure
Unmodified MWCNTs (code XFM04, diameter 5-15 nm, length 0.5-2 μm; Website: https://en.xfnano.com/Product/pro68.aspx) and carboxylated MWCNTs (code XFM06, diameter 5-15 nm, length 0.5-2 μm, carboxyl group content 3.86 wt%; Website: https://en.xfnano.com/Product/pro70.aspx) were purchased from Nanjing XFNANO Materials Tech Co, Ltd To characterize the MWCNTs, the morphology and structure of MWCNTs were observed under a TEM (FEI TECNAI G20, Hillsboro, OR, USA). To prepare the suspensions of MWCNTs, 1.28 mg/mL MWCNTs (prepared in Milli-Q water containing 2% FBS) was continuously sonicated for totally 16 min (with cooling on ice) using an ultrasonic processor FS-250N (Shanghai Shengxi, Shanghai, China). Then, the suspension was immediately diluted in cell culture medium for exposure.
Cytotoxicity Assay
Cytotoxicity was evaluated by CCK-8 assay, a widely used assay to determine the activities of mitochondria. Briefly, THP-1 macrophages (2.4 × 105 per well in 24-well plates) were exposed to 0, 2, 4, 8, 16, and 32 μg/mL of XFM04 or XFM06 for 24 h. The concentrations of MWCNTs were selected according to previous in vitro studies. 15 After 24 h exposure, the cells were rinsed once, and CCK-8 assay was done by using commercial kits following manufacturer’s instructions (Beyotime, Nantong, China). Briefly, the cells were carefully rinsed and then incubated with 10% CCK-8 reagents (diluted in cell culture medium). Then, the OD value was read by using an ELISA reader.
MWCNT Internalization
THP-1 macrophages (seeding density 3 × 106 in 60 mm diameter cell culture Petri dishes) were incubated with 16 μg/mL XFM04 or XFM06. After 24 h exposure, the cells were removed from cell culture dishes by trypsin and then fixed with 2.5% glutaraldehyde in PBS overnight. After that, the samples were post-fixed with 1% OsO4 for 3 h, dehydrated in a graded series of ethanol, and finally embedded in epoxy resin (Epon 812). The samples were sectioned using an ultramicrotome at 70 nm, placed on carbon film supported by copper grids, stained with uranyl acetate and lead citrate, and observed under a TEM (JEM-1230, JEOL Ltd, Tokyo, Japan) operated at 80 kV.
Quantification of Lipid Profiles
For Oil Red O staining, THP-1 macrophages (density 1.2 × 106 per well in 6-well plates) were incubated with cell culture medium (control), 16 μg/mL XFM04 or XFM06 for 24 h. After exposure, the cells were rinsed, fixed by 4% paraformaldehyde for 10 min, treated with 60% isopropanol for 5 min, and stained by 2.1 mg/mL oil red (prepared in 60% isopropanol; Sigma-Aldrich, USA) for 10 min. After rinsing again, the cells were incubated with isopropanol on a shaker for 10 min. The OD value was then determined at 500 nm by an ELISA reader.
For lipidomics experiments, THP-1 macrophages were treated with MWCNTs as indicated above. After 24 h exposure, the cells were rinsed, removed from cell culture plates by trypsin, and collected by centrifuge. The lipid was extracted by using chloroform/methanol (2:1; the lipid extraction was repeated twice), and the organic phase was transferred to a new tube. This procedure was repeated twice, and the samples were dried by a stream of nitrogen. Then, the lipid products were dissolved in chloroform/methanol (2:1) and were vortexed (Eyela Cute Mixer CM-1000, Tokyo Rikakikai Co, Tokyo, Japan) for 10 min. The solution was filtered through a 13 mm nylon filter with a pore size of 0.22 μm (Thermo Fisher Scientific, San Jose, CA, USA), and finally mass spectrometer measurement was done by using a UPLC-Q-Exactive Orbitrap mass spectrometer (Thermo Fisher Scientific, CA, USA) with a heated electrospray ionization probe. The samples were measured by using the parameters as previously described. 22
Real-Time RT-PCR
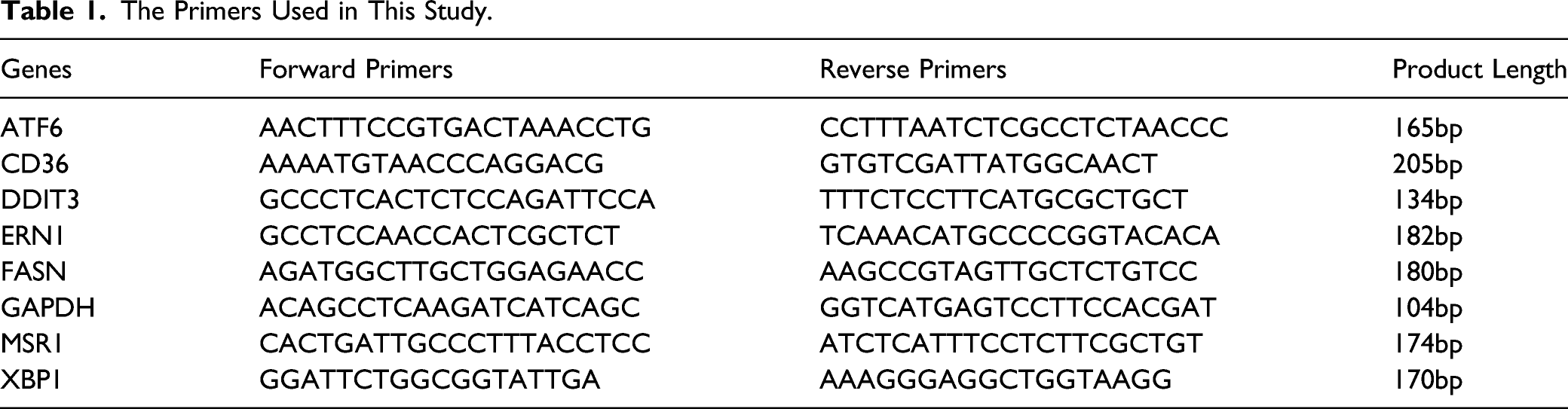
The Primers Used in This Study.
Statistics
The data are expressed as mean ± SD of 3 to 5 independent experiments (n = 3). Two-way ANOVA was used to analyze the effects of diameters and concentrations of MWCNTs. Tukey’s HSD test was then used to compare the difference by using R 3.3.3. A value of P < .05 was considered as statistically significant difference.
Results
Characteristics of MWCNTs
The TEM images clearly showed the fibrous structures of XFM04 (Figures 1A and B) and XFM06 (Figures 1C and D). Both types of MWCNTs appeared to have similar morphologies (comparable in length and diameters). XFM06 contained relatively high level of carboxyl groups (3.86 wt%) as measured by Boehm titration. The representative TEM images of XFM04 (1A and 1B) and XFM06 (1C and 1D).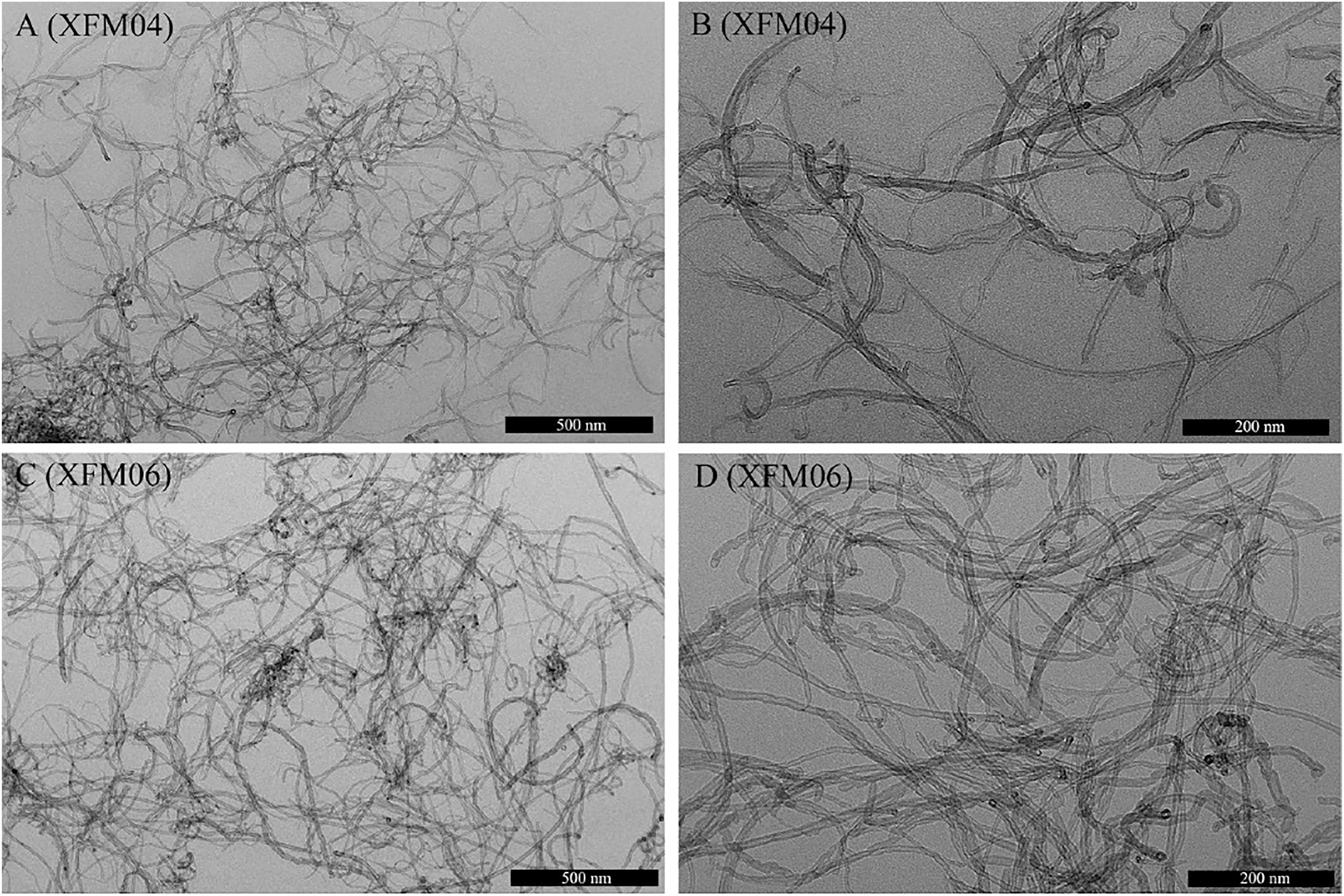
Cytotoxicity
As shown in Figure 2, significantly decreased cellular viability was observed in THP-1 macrophages following exposure to 16 (P < .05) or 32 μg/mL XFM04 (P < .01) or 32 μg/mL XFM06 (P < .01; compared with control). Exposure to XFM04 was associated with relatively lower levels of cellular viability compared with the exposure of XFM06, but there was no statistical difference between XFM04 and XFM06 (P > .05). Cytotoxicity. THP-1 macrophages were exposed to various concentrations of XFM04 or XFM06 for 24 h, and then the cellular viability was measured by CCK-8 assay. *, P < .05, compared with control.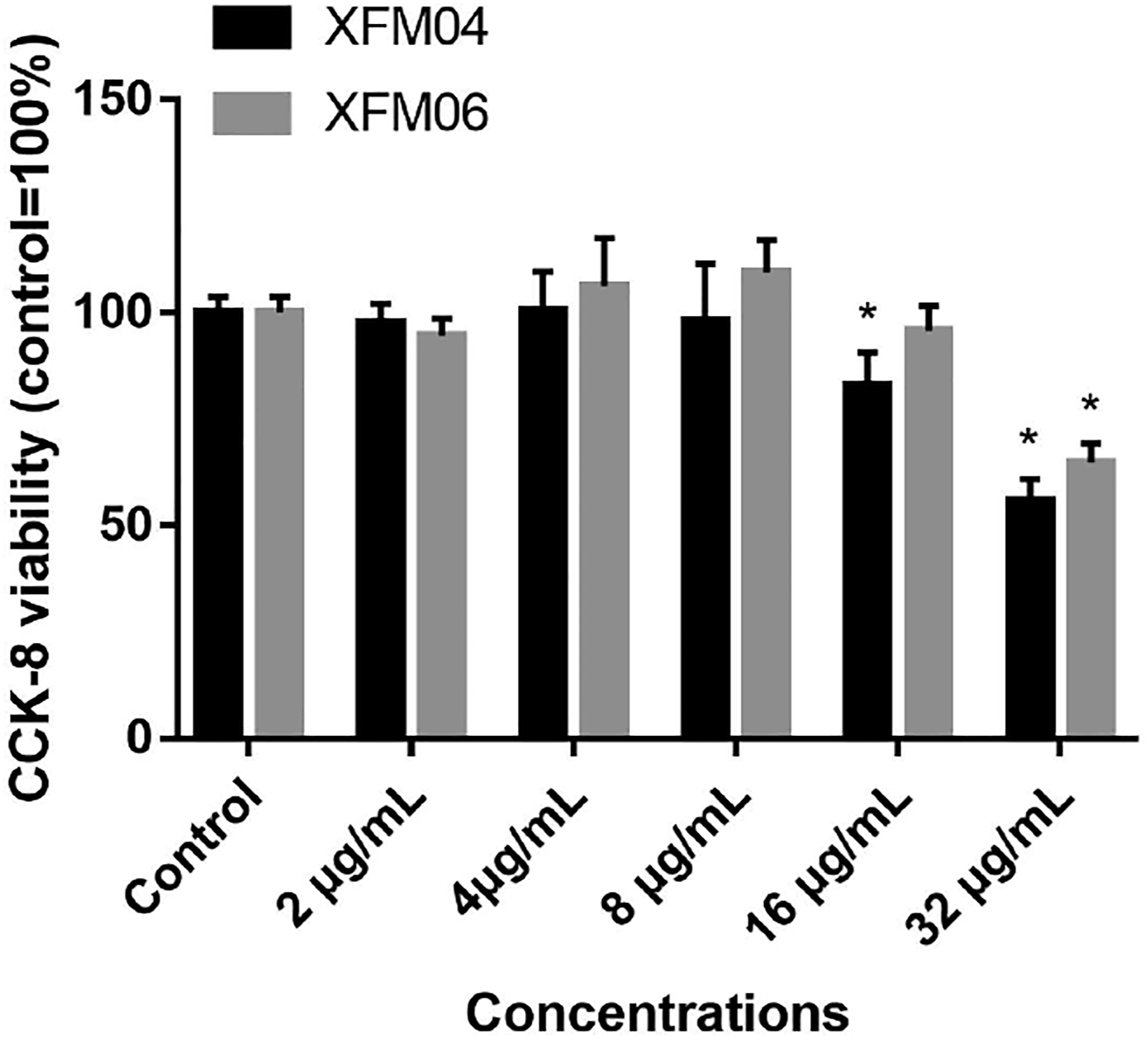
Multi-Walled Carbon Nanotube Internalization
For the cells we observed under TEM, no clear cellular membrane structure was observed, although the nuclear membranes are clear in XFM04- (Figures 3A and B) and XFM06- (Figures 3C and D) exposed cells. However, due to the limitation of TEM, only few cells were observed, and we cannot exclude the possibility that only few cells had damaged cellular membranes after XFM04 or XFM06 exposure. The internalized MWCNTs appeared to contact with or be entrapped into organelles, for example, mitochondria and lysosomes (Figure 3). TEM observation of MWCNT-exposed macrophages. THP-1 macrophages were incubated with 16 μg/mL XFM04 (3A and 3B) or XFM06 (3C and 3D) for 24 h, and the cells were observed under a TEM.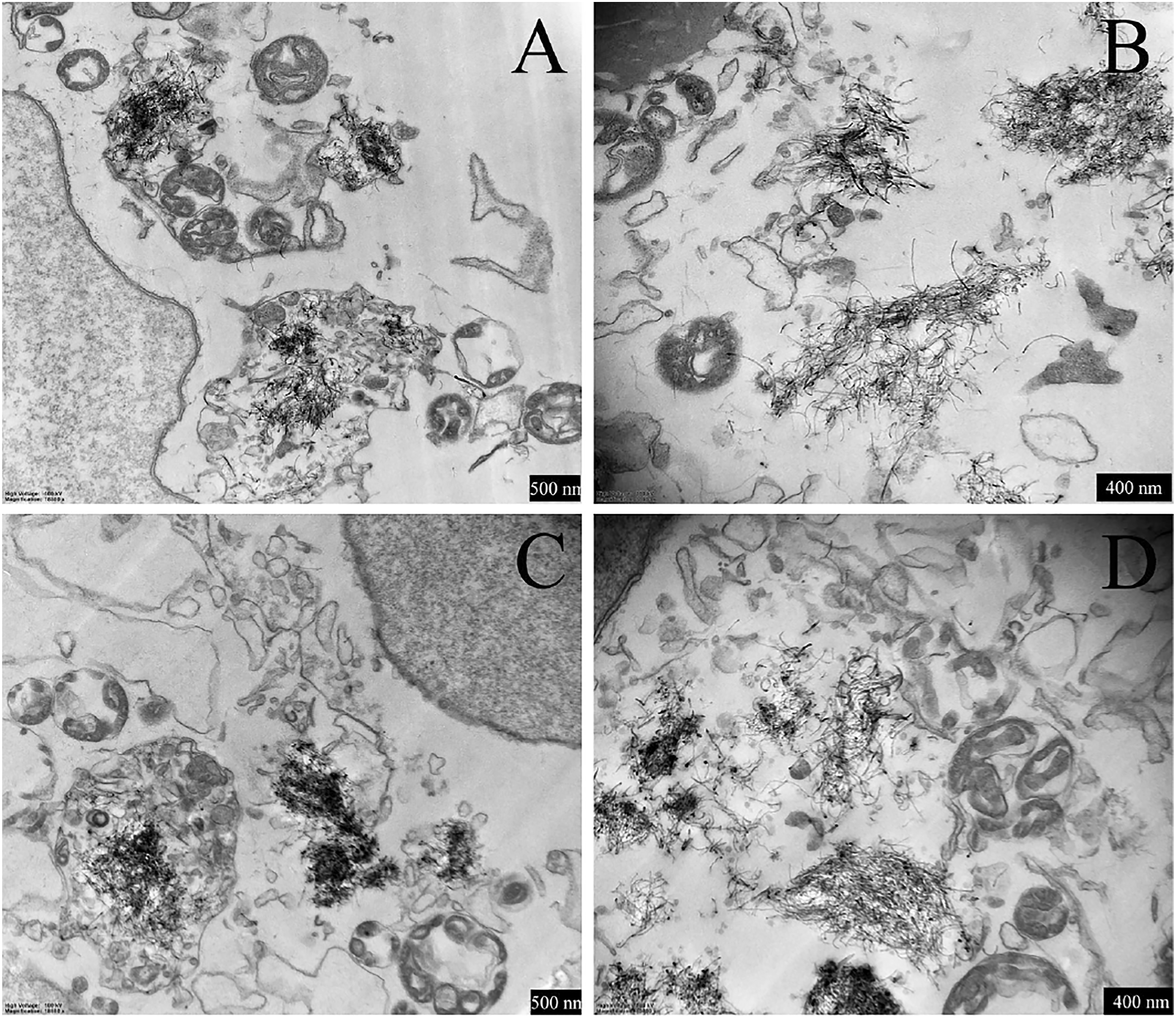
The Changes of Lipid Profiles
As shown in Figure 4, exposure to XFM04 or XFM06 caused a small but statistically significantly increased Oil Red O staining in THP-1 macrophages (P < .01, compared with control). There was no statistically significant difference between XFM04 and XFM06 (P > .05), although XFM06 led to slightly higher level of Oil Red O staining compared with XFM04. The accumulation of lipids. THP-1 macrophages were incubated with cell culture medium (control) or 16 μg/mL XFM04 or XFM06 for 24 h, and then lipid accumulation was quantified by using Oil Red O staining. *, P < .01, compared with control.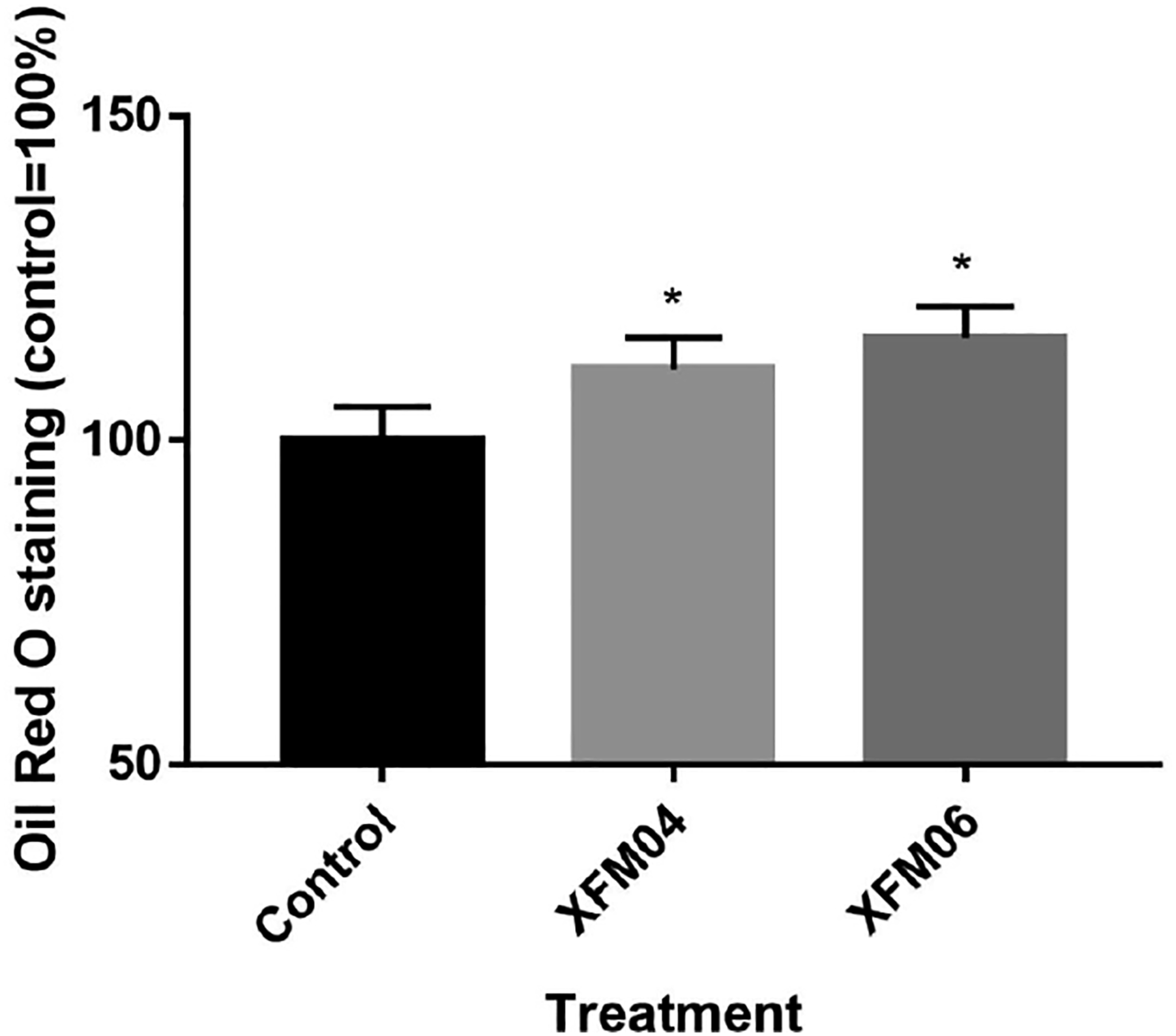
The changes of lipid profiles as measured under negative or positive ion modes are shown in Figures 5A and B, respectively. In keeping with Oil Red O staining results, most of the lipid classes were up-regulated by XFM04 or XFM06, and only few lipid classes, such as FA (fatty acid), were down-regulated by MWCNTs (Figure 5A). Interestingly, not revealed by Oil Red O staining, we found by using lipidomics that many lipid classes, such as LPG (lysophosphatidylglycerol) as measured under negative ion mode (Figure 5A), as well as CL (cardiolipin) as measured under positive ion mode (Figure 5B), were more obviously up-regulated by XFM06 compared with XFM04. The changes of lipid profiles. THP-1 macrophages were incubated with cell culture medium (control) or 16 μg/mL XFM04 or XFM06 for 24 h, and the changes of lipid classes were measured by mass spectrometer under either negative (5A) or positive (5B) ion modes. Note: the abbreviations for all lipid classes: AcCa, acyl carnitine; AcHexSiE, AcylGlcSitosterol ester; BisMePA, bis-methyl phosphatidic acid; CL, cardiolipin; Cer, ceramide; Co, coenzyme; DG, diglyceride; FA, fatty acid; GM2, GM3, gangliosides; Hex1Cer, Hex2Cer, Hex3Cer, CerG3GNAc1, simple Glc series; LPC, lysophosphatidylcholine; LPE, lysophosphatidylethanolamine; LPG, lysophosphatidylglycerol; LPI, lysophosphatidylinositol; LPS, lysophosphatidylserine; LdMePE, lysodimethylphosphatidylethanolamine; OAHFA, OAcyl-(gamma-hydroxy)FA; MePC, methyl phosphatidylcholine; PA, phosphatidic acid; PC, phosphatidylcholine; PE, phosphatidylethanolamine; PEt, phosphatidylethanol; PG, phosphatidylglycerol; PI, phosphatidylinositol; PMe, phosphatidylmethanol; PS, phosphatidylserine; SM, sphingomyelin; SPH, sphingosine; TG, triglyceride; WE, wax esters; dMePE, dimethylphosphatidylethanolamine.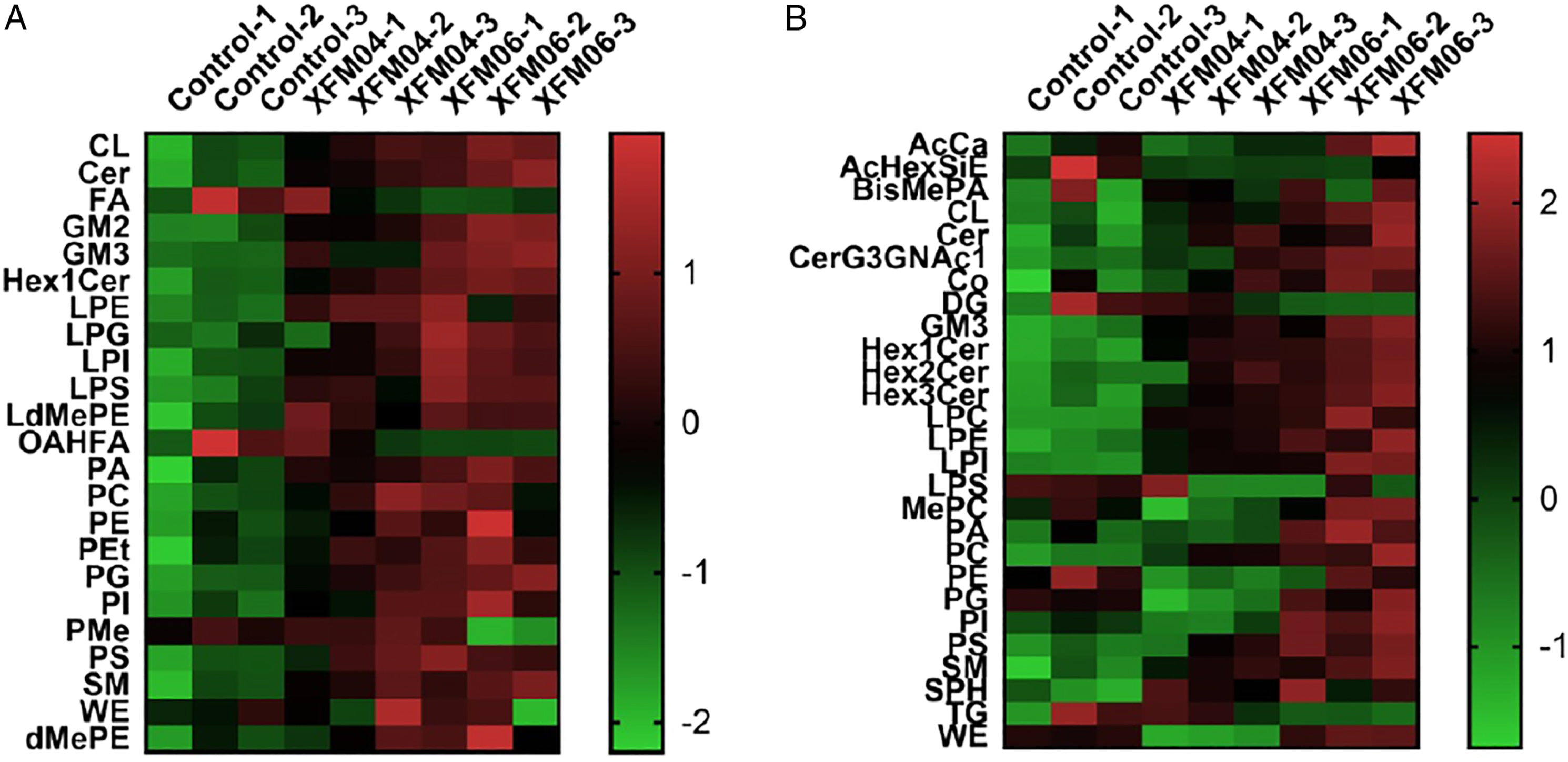
The Expression of ER Stress Genes
The influence of MWCNTs on ER stress expression is shown in Figure 6. Neither XFM04 nor XFM06 significantly induced the expression of ER stress genes. Indeed, XFM06 even significantly decreased the expression of ERN1 (P < .01; Figure 6B), and the expression of DDIT3 was significantly decreased by both XFM04 and XFM06 (P < .01; Figure 6C). The expression of ATF6 (6A) or XBP-1 (6D) was not significantly affected by XFM04 or XFM06 (P > .05). The expression of ER stress genes. THP-1 macrophages were incubated with cell culture medium (control) or 16 μg/mL XFM04 or XFM06 for 24 h, and the expression of ATF6 (6A), ERN1 (6B), DDIT3 (6C), and XBP-1 (6D) was measured by real-time RT-PCR. *, P < .01, compared with control.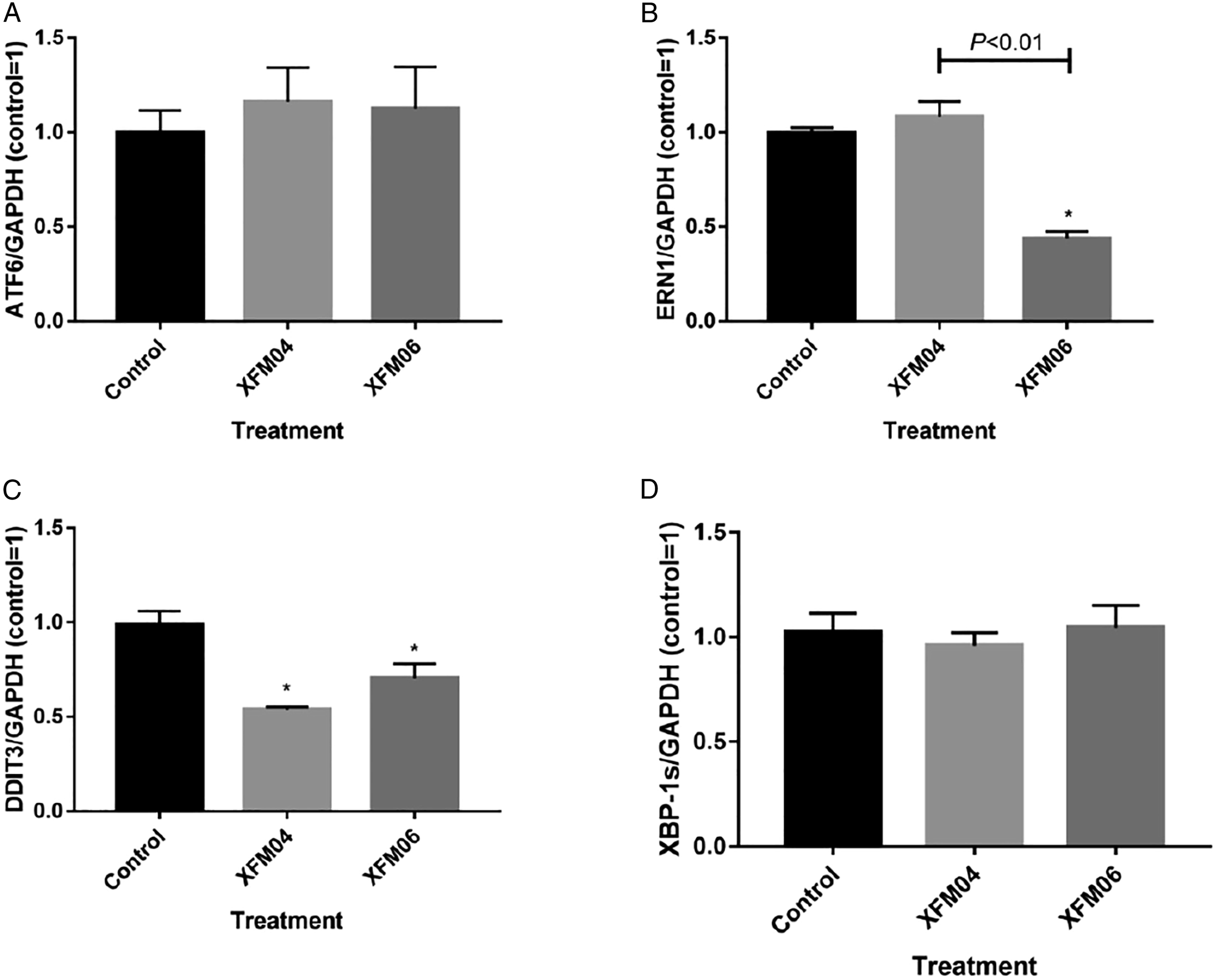
The Expression of Scavenger Receptor and Lipogenesis Genes
Exposure to XFM04 was associated with significantly increased expression of MSR1 (P < 0.05; Figure 7A) and FASN (P<.01; Figure 7C), and the expression of these genes were significantly higher in XFM04-exposed cells compared with XFM06-exposed cells (P < .01). The expression of CD36 was significantly decreased by XFM06 (P < .05) but not XFM04 (P > .05; Figure 7(B)). The expression of MSR1 (7A), CD36 (7B), or FASN (7C). THP-1 macrophages were incubated with cell culture medium (control) or 16 μg/mL XFM04 or XFM06 for 24 h, and the expression of targeted genes was measured by real-time RT-PCR. *, P < .05, compared with control.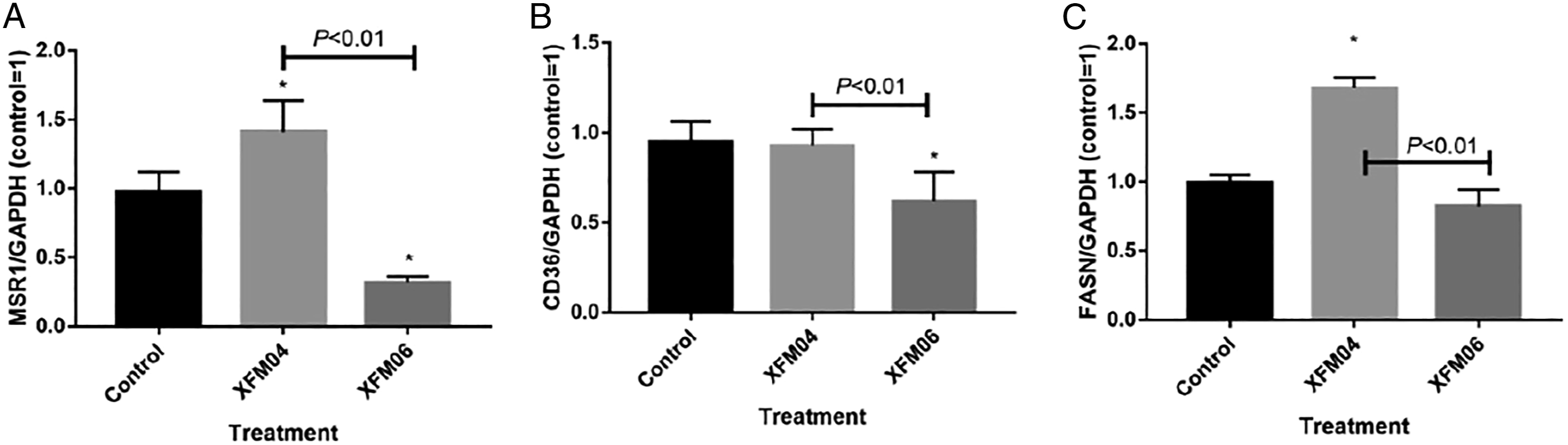
Discussion
In this study, we compared the different toxicity of unmodified (XFM04) and carboxylated (XFM06) MWCNTs to THP-1 macrophages to understand the possible influence of carboxyl modifications. By using CCK-8 assay, we found that both types of MWCNTs significantly decreased cellular viability to a similar extent (Figure 2). In addition, in MWCNT-exposed THP-1 macrophages, no clear cellular membrane was observed (Figure 3). This could further indicate the membrane damages induced by both types of MWCNTs because recent studies showed that MWCNTs of similar diameter (XFM04) induced membrane damage in THP-1 macrophages, whereas the unexposed cells showed clear structures in cellular membrane.17,23 These results indicated that unmodified and carboxylated MWCNTs were equally cytotoxic to THP-1 macrophages. Previous studies also reported that unmodified and carboxylated MWCNTs were equally cytotoxic to human endothelial cells, 24 THP-1 macrophages, 21 vascular smooth muscle cells, 25 and HepG2 cells, 26 although MWCNTs used in previous studies contained a much lower level of carboxyl groups compared with the MWCNTs used in this study (1.23 wt% vs 3.86 wt%). Hereby, our results indicated that even a relatively high degree of carboxylation did not change the cytotoxic potential of MWCNTs to THP-1 macrophages. However, a recent study reported that HepG2 cells were more sensitive to hydroxylated MWCNT, whereas Beas2B cells were more sensitive to carboxylated MWCNTs, 27 and the authors suggested that the sensitivity to MWCNT-induced toxicity is also dependent on the cell types. However, in this study, we did not attempt to use different types of cells as this study focused on the toxicity of MWCNTs to macrophages.
We also determined lipid accumulation induced by unmodified or carboxylated MWCNTs. Very similar to the trend of cytotoxicity, hereby we observed that both types of MWCNTs promoted lipid accumulation to similar extent (Figure 4). It has been shown before that unmodified and carboxylated MWCNTs induced lipid accumulation in THP-1 macrophages 21 and human vascular smooth muscle cells 25 to a similar extent, but unmodified MWCNTs were more potent than carboxylated MWCNTs to induce lipid accumulation in HepG2 hepatocytes, 26 indicating that the influence of carboxylation on MWCNT-induced lipid accumulation is likely to be cell type dependent. Previous studies showed that SiO2 NPs,28,29 ZnO NPs, 30 and carbon black NPs 31 induced lipid accumulation in macrophages, but in contrast, graphene oxide 32 and Ag NPs 33 reduced lipid species in macrophages. These results indicated that NMs altered lipid levels in macrophages in a manner dependent on the compositions of NMs. Hereby, our results suggested that carboxylation is not important to change MWCNT-induced lipid accumulation.
In keeping with Oil Red O staining results, the lipidomics data suggested that both types of MWCNTs up-regulated most lipid classes in THP-1 macrophages. Interestingly, it should be noticed that almost all lipid classes were relatively higher in carboxylated MWCNT-exposed THP-1 macrophages compared with the unmodified MWCNT-exposed cells, indicating that carboxylated MWCNTs were more potent to alter lipid profiles compared with unmodified MWCNTs (Figure 5). A recent study showed that pulmonary surfactant lipids did not influence MWCNT-induced lipid accumulation but pronouncedly altered lipid compositions in THP-1 macrophages. 34 Liu et al recently reported that carboxylated MWCNTs significantly promoted lipid accumulation in endothelial cells, but only 2 lipid classes were up-regulated as revealed by lipidomics. 35 Xie et al found that although MoS2 nanosheets and bulk materials all decreased lipid contents in 3D Caco-2 spheroids, MoS2 nanosheets were more potent to decrease lipid classes as revealed by lipidomics data. 36 Hence, omics-based lipidomics technique is needed to better reveal the lipid profile changes besides the conventional Oil Red O staining.
We then investigated ER stress-mediated lipid metabolism because it has been suggested that the toxicity of NMs is related with the induction of ER stress.37,38 In macrophages, MWCNTs21,39 and SiO2 NPs29,40 have been shown to induce lipid accumulation by inducing ER stress leading to the up-regulation of scavenger receptors and/or de novo lipogenesis. Meanwhile, van der Valk et al reported that liposomal NPs loaded with prednisolone induced lipotoxicity to macrophages through ER stress. 41 In contrast to our expectation, no types of MWCNTs induced the expression of ER stress genes (Figure 6). In addition, only unmodified MWCNTs significantly induced the expression of macrophage scavenger receptor gene MSR1 and de novo lipogenesis gene FASN (Figure 7). But carboxylated MWCNTs were indeed more potent to up-regulate lipid classes compared with unmodified ones (Figure 5). Although we did not further verify the changes of proteins, it is unlikely, based on the presented experimental results, that unmodified and carboxylated MWCNTs altered lipid profiles by inducing ER stress in THP-1 macrophages. It is possible that the types of MWCNTs altered lipid profiles in THP-1 macrophages through other mechanisms, which may need further studies. One possibility is that NM-induced ER stress only interferes lipid synthesis and lipid droplet induction,37,38 whereas the MWCNTs used in this study altered lipid profiles by changing lipid metabolism.
In summary, in this study, we found that unmodified and carboxylated MWCNTs induced cytotoxicity and lipid accumulation to a similar extent in THP-1 macrophages, but carboxylated MWCNTs were more potent to up-regulate the lipid classes compared with unmodified MWCNTs. However, none of the MWCNTs induced the expression of ER stress genes, and unmodified MWCNTs were even more potent to induce scavenger receptor and lipogenesis genes compared with carboxylated MWCNTs. Hence, it is possible that MWCNTs induced cytotoxicity and lipid accumulation in THP-1 macrophages independently of ER stress, and it may be necessary to investigate the influences of MWCNTs on lipid metabolism to understand how unmodified and carboxylated MWCNTs altered lipid profiles in THP-1 macrophages. In addition, the results from this study also indicated that surface modifications other than carboxylation should be considered to alleviate the toxicity of MWCNTs to THP-1 macrophages.
Footnotes
Acknowledgments
We appreciate the scientific assistance from Mr Hao Guo from Chongqing Institute of Forensic Science and Dr Youqi Xian from Thermo-Fisher Scientific for lipidomics experiments.
Author Contributions
Pei, L. contributed to design, contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; Yang, W. contributed to conception, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Cao, Y. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
