Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) assessed the safety of 33 Citrus flower- and leaf-derived ingredients, which are most frequently reported to function in cosmetics as fragrances and/or skin-conditioning agents. The Panel reviewed the available data to determine the safety of these ingredients. Because final product formulations may contain multiple botanicals, each containing similar constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. With Citrus flower- and leaf-derived ingredients, the Panel was concerned about the presence of the hydroperoxides of limonene and linalool in cosmetics. Industry should use good manufacturing practices to limit impurities that could be present in botanical ingredients. The Panel concluded that these ingredients are safe in the present practices of use and concentration when formulated to be non-irritating and non-sensitizing.
Introduction

Definitions and Functions of Citrus-Derived Ingredients. 1

*Accepted or alternate scientific names for these Citrus ingredients are found in Table 3.
The Panel has previously reviewed the safety of Citrus-derived peel oils, Citrus peel-derived ingredients, and Citrus fruit-derived ingredients in separate assessments and concluded that 14 Citrus-derived peel oil ingredients, 47 Citrus peel-derived ingredients, and 80 Citrus fruit-derived ingredients are safe for use in both rinse-off and leave-on cosmetic products when formulated to be non-sensitizing and non-irritating, provided that leave-on products do not contain more than 0.0015% (15 ppm) 5-methoxypsoralen (5-MOP).2-4 The Panel also assessed the safety of 30 Citrus plant- and seed-derived ingredients and concluded that 18 of these ingredients are safe in the present practices of use and concentration when formulated to be non-irritating and non-sensitizing, while the data for the remaining 12 ingredients are insufficient to determine safety. 5
Some of the Citrus flowers and leaves that are used to derive the ingredients described in this safety assessment are food ingredients. The US Food and Drug Administration (FDA) determined that the use of some Citrus flowers and leaves as direct food additives is generally recognized as safe (GRAS). Additionally, essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from bitter orange flowers, sweet orange flowers and leaves, lemon leaves, mandarin and/or tangerine leaves, and possibly other Citrus flowers and leaves are GRAS for their intended use in foods for human and animal consumption. Daily consumption of these GRAS foods would result in much larger systemic exposures than what is expected from use in cosmetic products, even if there was 100% absorption from cosmetics. Thus, the systemic toxicity potential of Citrus flower- and leaf-derived ingredients via oral exposure is not addressed further in this report. The primary focus of the safety assessment is the review of the safety of topical exposure to the ingredients used as foods.
Citrus Ingredients that Potentially Function Exclusively as Fragrance Ingredients.

Botanicals such as Citrus-derived ingredients contain numerous constituents, some of which have the potential to be toxic. In this assessment, CIR is reviewing the potential toxicity of each Citrus flower- or leaf-derived ingredient as a whole, complex substance. Except for specific constituents of concern that the Panel has identified, CIR is not reviewing the potential toxicity of the individual constituents of the Citrus flowers and leaves from which the ingredients in this report are derived.
Review of Citrus Genus Species Names. 6

Chemistry
Definition and General Characterization
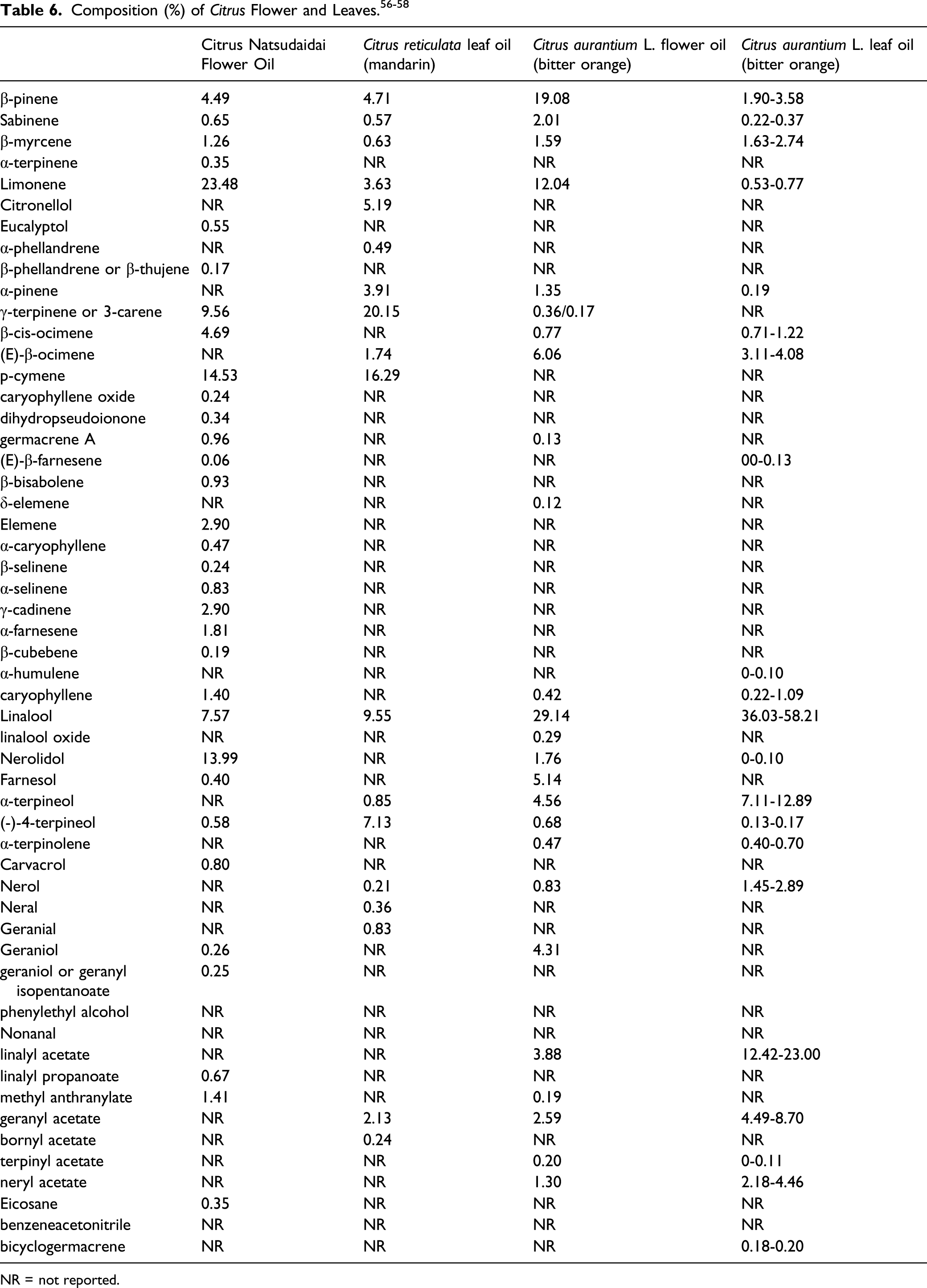
The definitions and functions of the Citrus flower- and leaf-derived ingredients included in this report are provided in Table 1. The definition indicates what part(s) of the plant from which an ingredient is derived. In some cases, the definition provides insight on the method(s) of manufacture. Essential oils are the hydrophobic, liquid, volatile aroma compounds in the insoluble condensate fraction. The essential oils are typically mixtures of small molecules, but their chemical structures can vary widely. Fixed oils, on the other hand, are hydrophobic, nonvolatile, fatty compounds from plants, animals, or algae. These are primarily composed of glycerides, and to some extent, free fatty acids. Constituents of these Citrus-derived ingredients may include both oil types. The volatile nature of essential oils makes them more likely to be useful as fragrances, but that does not mean that fragrance is their only function.
Physical and Chemical Properties
Citrus Aurantium Amara (Bitter Orange) Flower Extract
A supplier reported that Citrus Aurantium Amara (Bitter Orange) Flower Extract in sunflower seed oil has an ultraviolet (UV) absorption wavelength <250 nm, with the maximum absorption value at 220 nm. 7
Citrus Aurantium Dulcis (Orange) Flower Extract
As reported by a supplier, Citrus Aurantium Dulcis (Orange) Flower Extract is a medium-to-dark amber liquid with a characteristic odor. 8 At 25°C, the pH range is 4.0 to 6.5 (actual 4.1). Specific gravity range is 0.99 to 1.01 (1.01 actual) at 25°C. Citrus Aurantium Dulcis (Orange) Flower Extract is soluble in any proportion of water, has less than 100 organisms/g, and has a refractive index range of 1.3250 to 1.3450 (1.3385 actual) at 25°C.
Citrus Aurantium Dulcis (Orange) Flower Wax
A supplier reported that Citrus Aurantium Dulcis (Orange) Flower Wax has a melting point of 60.0°C and a UV absorption value of 1250 at 280 nm. 9 No further details provided.
Method of Manufacturing
According to the Dictionary, essential oils and waters are prepared from leaves, stems, flowers, bark, roots, or other parts of a plant or the whole plant. 1 Essential oils are prepared by a number of processes including, but not limited to, steam or dry distillation, flash pasteurization, and mechanical processes such as cold-pressing; however, the most widely used method for preparing essential oils from plants is steam distillation. The condensate from steam distillation produces two distinct fractions that contain the volatile ingredients from the plant. The water insoluble fraction contains the "oil." The water soluble fraction contains constituents of the plant that are water soluble. The name assigned to the water insoluble fraction from steam distilled plant materials includes the term "oil" in the INCI name. The water soluble fraction from the steam distilled plant material includes the term "water" in the INCI name.
Citrus Aurantium Amara (Bitter Orange) Flower Extract
A supplier reported that Citrus Aurantium (Bitter Orange) Flower Extract is produced by macerating the flowers in hot water followed by clarification, addition of glycerin and preservative, and filtration. 10 This supplier also reported that the flowers may be extracted in sunflower seed oil before clarification and decontamination. 11
Another supplier reported that Citrus Aurantium Amara (Bitter Orange) Flower Extract was extracted by hydrocarbons (not specified) from Citrus aurantium amara flowers grown in Morocco. 12 The resultant mixture was treated with ethanol, filtered, and then concentrated and purified by distillation.
Citrus Aurantium Amara (Bitter Orange) Flower Water
As reported by a supplier, Citrus Aurantium Amara (Bitter Orange) Flower Water is produced through distillation of the flowers followed by acidification, addition of preservative, and decontamination. 13
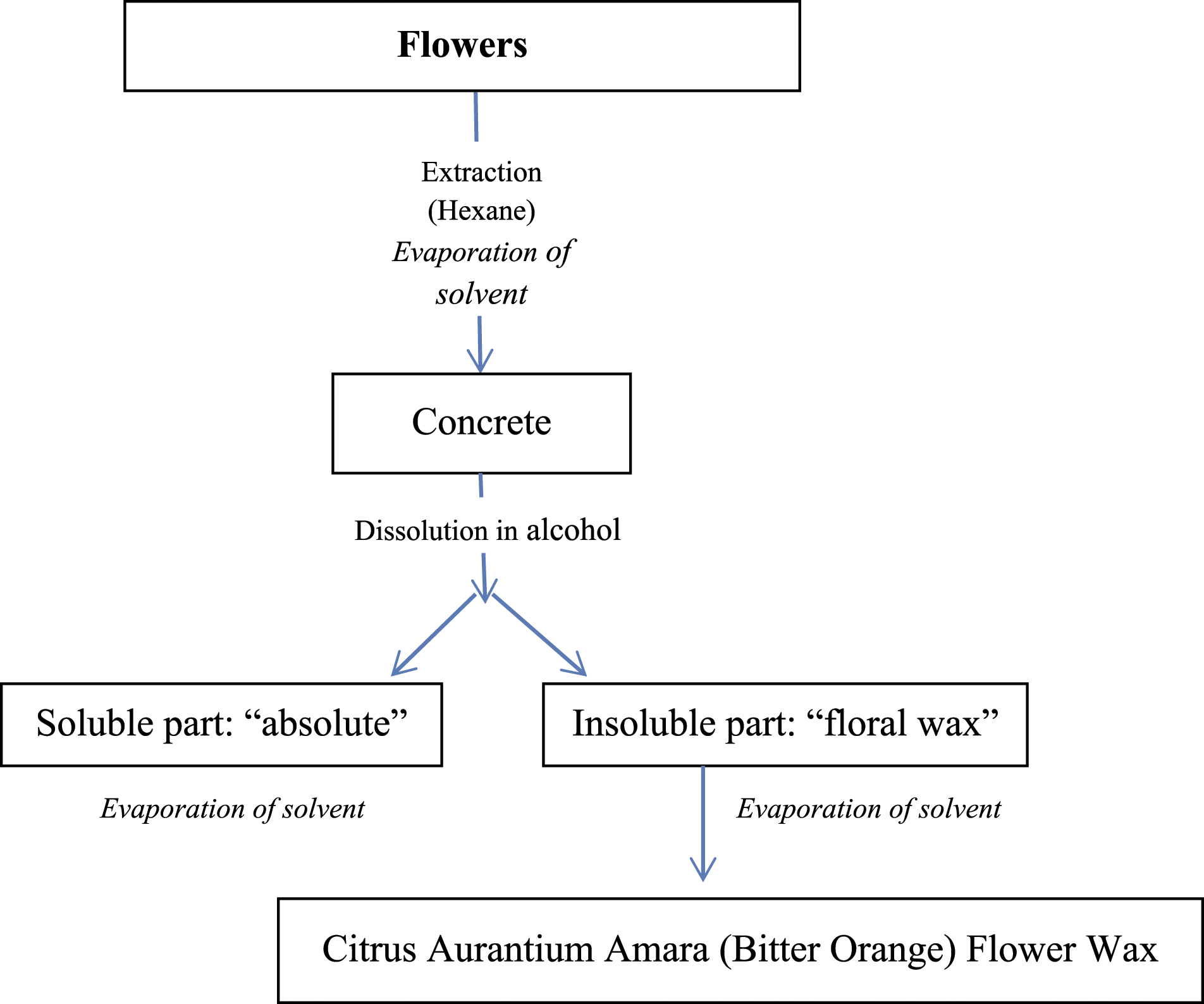
Citrus Aurantium Amara (Bitter Orange) Flower Wax
Figure 1 is a generic representation of the method of manufacturing for Citrus Aurantium Amara (Bitter Orange) Flower Wax. In the preparation of this ingredient, Citrus aurantium amara flowers undergo extraction with an organic solvent to form a “concrete,” which is then dissolved in alcohol. The insoluble portion is the floral wax, which is further refined. Manufacturing flow chart of Citrus Aurantium Amara (Bitter Orange) Flower Wax.
51
Citrus Aurantium Dulcis (Orange) Flower Extract
According to a supplier, fresh or dried flowers of Citrus aurantium dulcis are extracted with specified eluent under appropriate temperatures to yield a concentrate. 8 The concentrate is then blended with the desired diluent and preservation systems to produce Citrus Aurantium Dulcis (Orange) Flower Extract.
Citrus Hystrix Leaf Extract
A supplier has reported that Citrus Hystrix Leaf Extract is produced by extracting dried leaves with 80% ethanolic solution, and the extract is filtered and concentrated before the addition of 70% 1,3-butylene glycolic solution. 14 The material then undergoes sedimentation, filtration, and adjustment before packaging.
Citrus Natsudaidai Flower Water and Oil
In the preparation of Citrus Natsudaidai Flower Water and Citrus Natsudaidai Flower Oil, Citrus natsudaidai flowers were handpicked and then refrigerated. 15 Approximately 4 to 8 kg of flowers were then distilled with 10 to 20 L of purified water. The water and oil were then separated and the resulting products were analyzed for heavy metals and bacteria content.
Citrus Reticulata (Tangerine) Leaf Oil
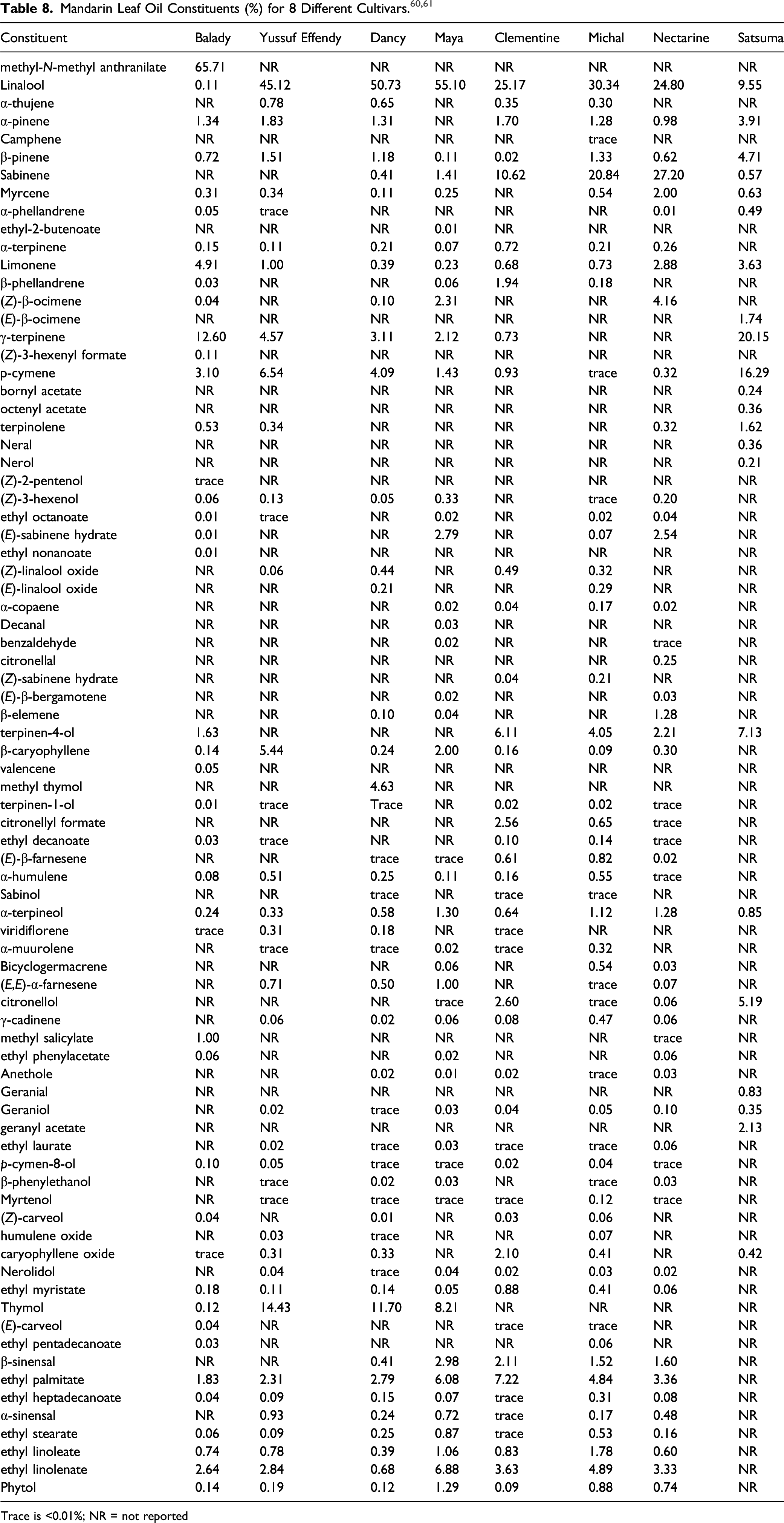
A supplier has reported that Citrus Reticulata (Tangerine) Leaf Oil is produced through the steam distillation of the dried leaves of Citrus reticulata. 16
Constituents/Composition/Impurities
Unless noted, detection limit was 2 ppm.
Detection limit was 20 ppm.
Detection limit was 5 ppm
Detection limit was 1 ppm for all allergens
ND = unable to be detected by GCSM.
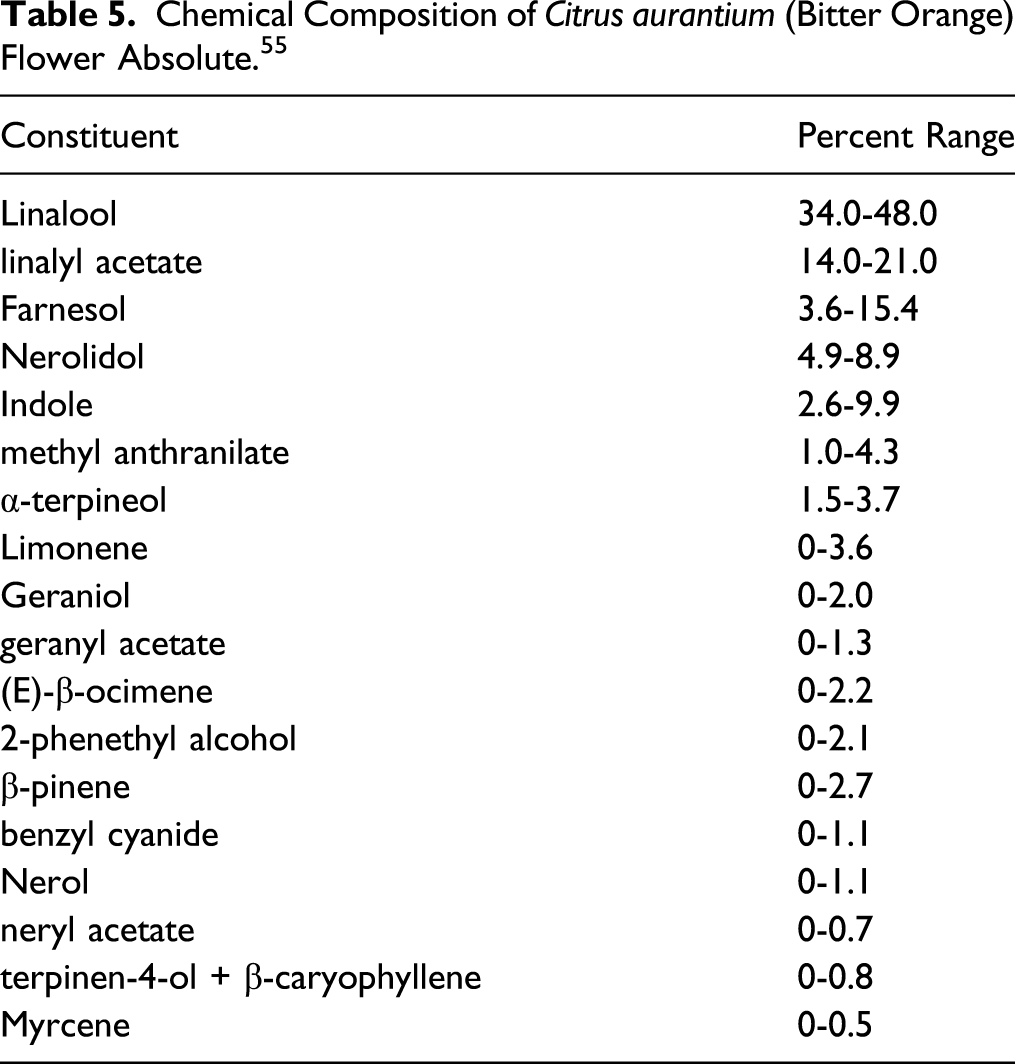
Chemical Composition of Citrus aurantium (Bitter Orange) Flower Absolute. 55
NR = not reported.
Composition reported down to the level of 1%, or lower for known toxic constituents.
NR = not reported.
Trace is <0.01%; NR = not reported
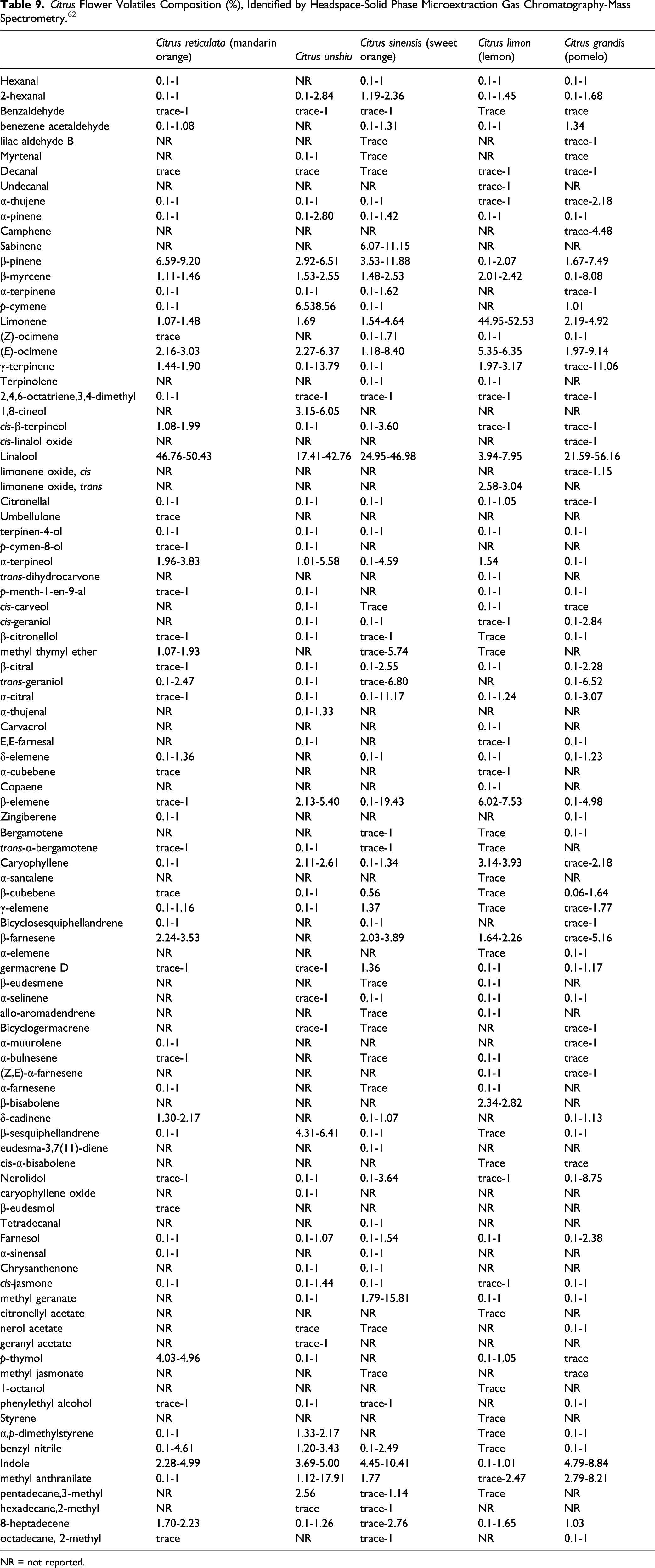
Citrus Flower Volatiles Composition (%), Identified by Headspace-Solid Phase Microextraction Gas Chromatography-Mass Spectrometry. 62
NR = not reported.
Volatile Organic Compounds (%) in Citrus Leaves, Identified by Gas Chromatography-Mass Spectroscopy. 63
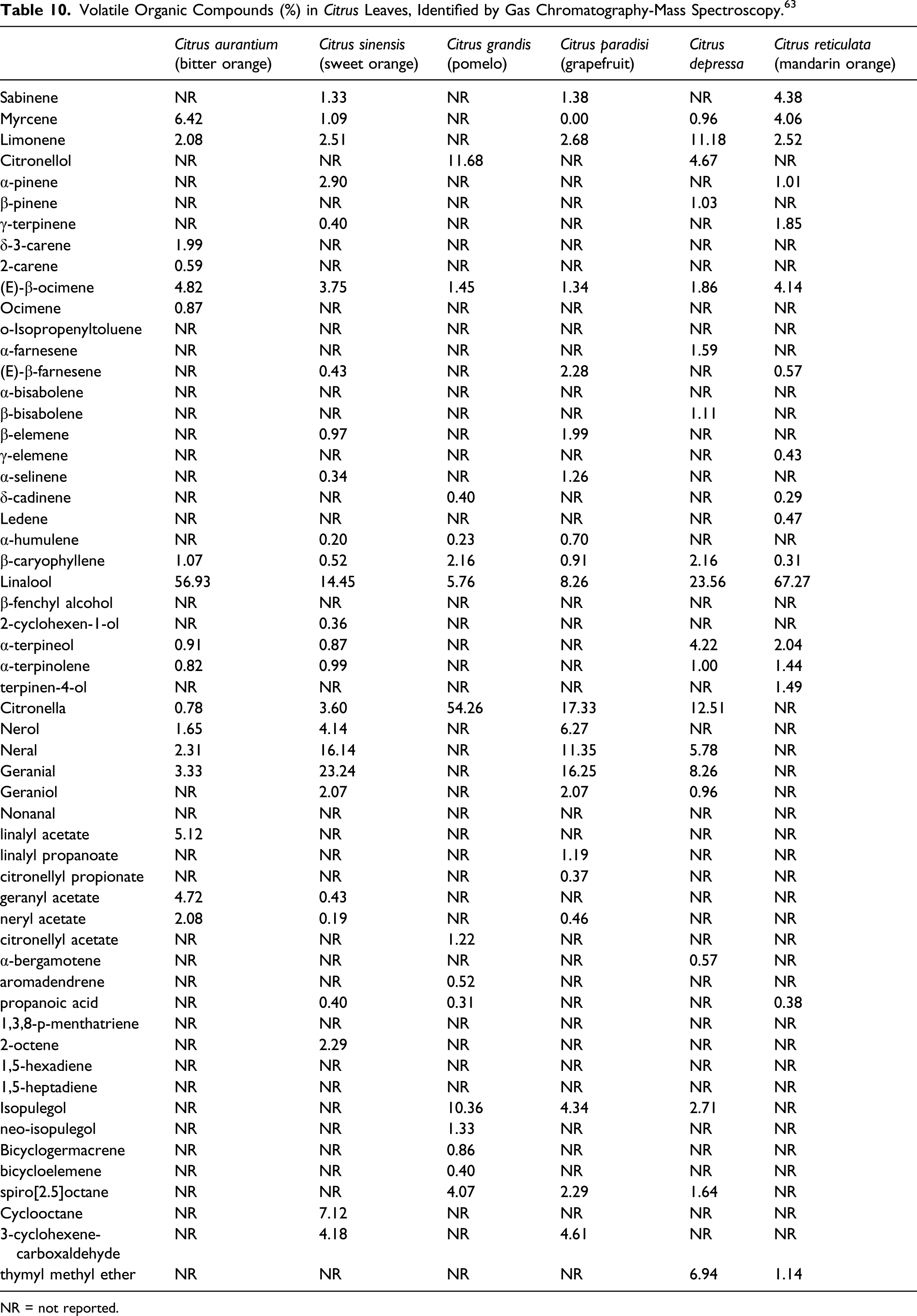
NR = not reported.
The International Fragrance Association (IFRA) has issued standards for limonene and linalool in natural products, stating that these constituents “should only be used when the level of peroxides is kept to the lowest practical level, for instance by adding antioxidants at the time of production.”17,18
International Fragrance Association has also issued a standard for methyl-N-methylanthranilate (or dimethyl anthranilate), stating that this substance should be restricted to 0.1% in leave-on products. 19 No restrictions were set for rinse-off or non-skin contact products.
The European Commission’s Scientific Committee on Consumer Safety (SCCS) issued an opinion on the phototoxicity of methyl-N-methylanthranilate with the conclusion that this substance is phototoxic and may be safe for use at up to 0.1% in leave-on products, except for sunscreen/sun care products and products used on areas exposed to light. 20 There was no safety concern on the use of this substance at concentrations up to 0.2% in rinse-off products. The SCCS noted that this substance is a secondary amine and may be prone to N-nitrosation. The presence of methyl-N-methylantrhanilate in essential oils like petitgrain mandarin oil was considered in the SCCS opinion.
Citrus Aurantium Amara (Bitter Orange) Flower Extract
A supplier reported that their raw material contains 1.5%–2.5% Citrus Aurantium (Bitter Orange) Flower Extract, 73.05%–74.05% water, >23% glycerin, <1.0% citric acid, 0.3% sodium benzoate, and 0.15% potassium sorbate. 21 This product was certified to contain 27 ppm of acetaldehyde (detection limit was 10 ppm). 22 This product was certified to not contain the furocoumarins bergapten and 8-methoxypsoralen (detection limit was 10 ppm). 23
Another raw material of this supplier was reported to contain 0.1%–0.5% Citrus Aurantium Amara (Bitter Orange) Flower Extract in 99.5%–99.9% sunflower seed oil. 24
A raw material containing 0.15% Citrus Aurantium Amara (Bitter Orange) Flower Extract is reported to have 0.0016% (16 ppm) furanocoumarins with the following (calculated) break-out: 0.95 ppm psoralen, 0.3 ppm bergapten, 0.88 ppm oxypeudecanin, 0.08 ppm imperatorin, 0.18 ppm isoimperatorin, and 13.62 ppm epoxybergamottin. 12 This material also contains approximately 630 ppm linalool, 185 ppm linalyl acetate, 6 ppm β-pinene, and 2 ppm ocimene. Total polyphenolic content is approximately 29 ppm.
Citrus Aurantium Amara (Bitter Orange) Flower Water
A supplier reported that their raw material contains >98% Citrus Aurantium Amara Flower Water, <1% citric acid, 0.55% water, 0.3% sodium benzoate, and 0.15% potassium sorbate. 25 This product was certified to not contain furocoumarin, bergapten, bergaptol, bergamothine, citropten, coumarine, imperatorine, isoemperatorine, isopimpinelline, umbelliferone, or scopoletine (detection limit was 10 ppm). 26
Citrus Aurantium Amara (Bitter Orange) Flower Wax
In data provided by a supplier, Citrus Aurantium Amara (Bitter Orange) Flower Wax had less than 0.1 mg/kg heavy metals (arsenic, cadmium, and lead) and no detectable pesticides (<0.005 mg/kg) or polycyclic aromatic hydrocarbons (<0.25 µg/kg). 27 Concentrations of aflatoxins (B1, B2, G1, G2) were less than 0.1 µg/kg, with the total aflatoxins concentration less than 0.4 µg/kg, and dioxins were less than 0.6 pg/g.
Citrus Aurantium Dulcis (Orange) Flower Extract
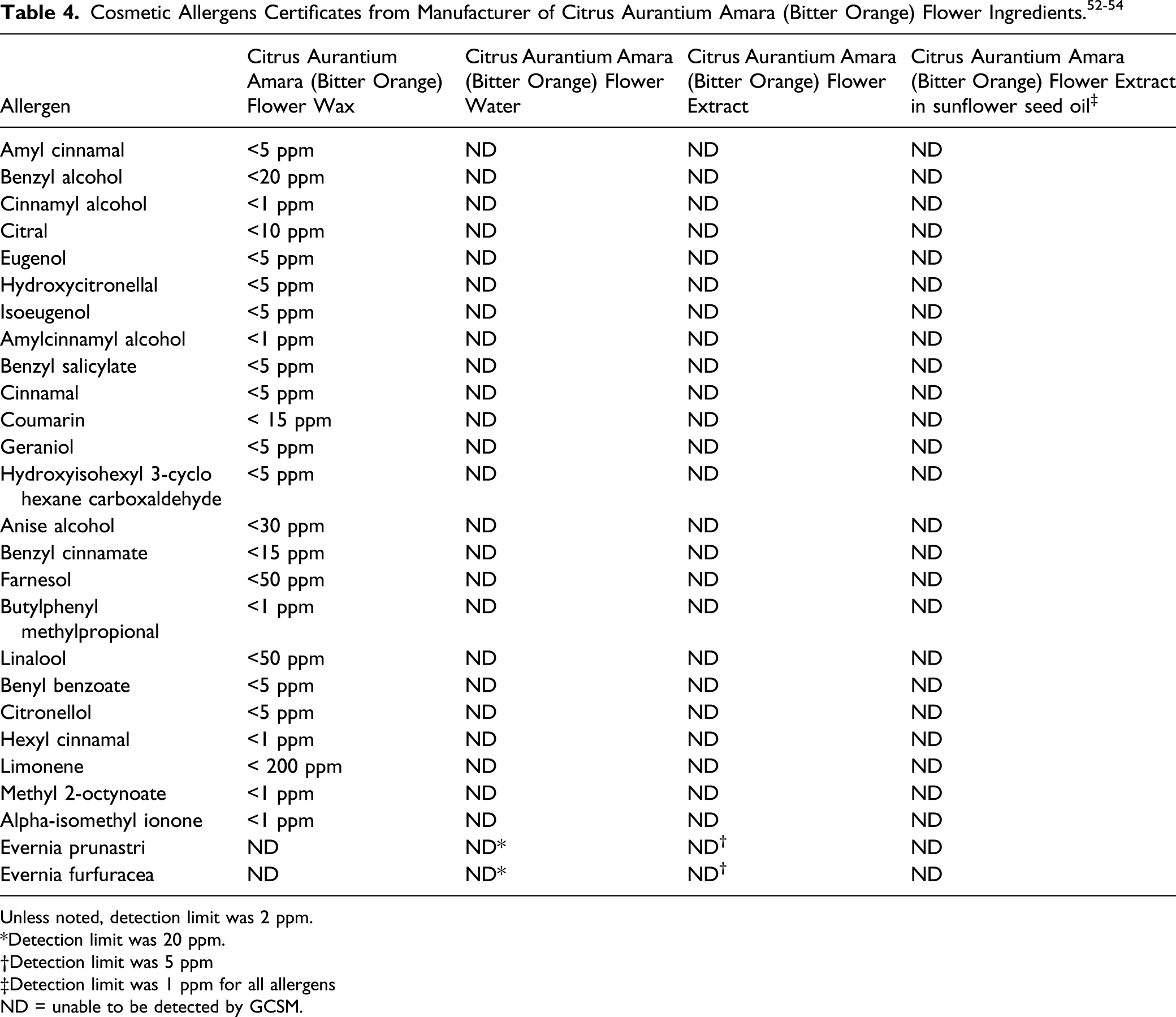
According to a supplier, impurities testing on Citrus Aurantium Dulcis (Orange) Flower Extract is performed on the concentrate in alcohol base. 8 No residual pesticides or heavy metals (including arsenic, lead, and mercury) were detected. In addition, none of the 26 cosmetic allergens listed in Table 4 were detected (detection limit <1 ppm).
Citrus Aurantium Dulcis (Orange) Flower Wax
A supplier reported that Citrus Aurantium Dulcis (Orange) Flower Wax contains 35%–50% hydrocarbons (C21–C35), 12%–20% polycosanols (C24–C36), 25%–40% esters (C40–C60), and 0.5%–5.0% essential oils and bioflavonids. 9
Citrus Hystrix Leaf Extract
A supplier reports that Citrus Hystrix Leaf Extract is composed of tannin and sugar. 14 Impurities of heavy metals are not more than 20 ppm and arsenic is not more than 2 ppm.
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US FDA and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s VCRP database. Use concentration data are submitted by Industry in response to surveys, conducted by the Council, of maximum reported use concentrations by product category.
NR = not reported.
Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
.Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
.It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
It is possible these products may be powders, but it is not specified whether the reported uses are powders.
dIncludes generic lime blossom extract in the VCRP database.
eListed as Citrus Aurantium (Bitter Orange) in the VCRP database.
fListed as Citrus Bergamia (Bergamot Orange) in the VCRP database.
gIncludes uses listed under Orange Blossom in the VCRP database.
hIncludes uses listed under Citrus Sinensis (Sweet Orange) Flower Extract in the VCRP database.
iIncludes uses under Citrus Sinensis (Sweet Orange) Flower Oil; Oil of Orange Flowers; and Orange Flower Oil, Sweet in the VCRP database.
jIncludes uses listed under Orange Blossom Wax in the VCRP database.
kIncludes uses listed under Citrus Reticulata (Mandarin Orange) Leaf Oil in the VCRP database.
Only listed in the VCRP database, not an INCI ingredient. Included because of similarity.
Ingredients that are not Reported to be in Use.

Some of these ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes. For example, Citrus Aurantium Dulcis (Orange) Flower Wax is used at 0.12% in lipstick. Additionally, some of these ingredients were reported to be used in hair sprays, fragrance preparations, and face powders and could possibly be inhaled. For example, Citrus Aurantium Dulcis (Orange) Flower Oil was reported to be used in hair spray at a maximum concentration of 0.015%, and Citrus Aurantium Amara (Bitter Orange) Flower Oil was reported to be used in face powders at a maximum concentration of 0.01%. In practice, 95%–99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.30-33 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.31,32 Conservative estimates of inhalation exposures to respirable particles during the use of loose-powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.34-36
The Citrus flower- and leaf-derived ingredients in this safety assessment are not restricted from use under the rules governing cosmetic products in the European Union. 37
Non-Cosmetic
The essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from the following Citrus plant sources are GRAS for their intended use in foods for human consumption: Citrus aurantifolia (lime); Citrus aurantium (bergamot); Citrus aurantium (bitter orange: the flowers and peel); Citrus limon (lemon: the leaves); Citrus paradisi (grapefruit); Citrus reticulata (tangerine: the leaves); Citrus reticulata blanco (mandarin: the leaves); Citrus sinensis (orange: the leaf, flowers, and peel); and Citrus peels (species not specified) (21CFR182.20). These essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of these Citrus plant sources are GRAS for their intended use in animal drugs, feeds, and related products (21CFR582.20).
While not specifically listed as GRAS, Citrus hystrix (kaffir lime) leaves are recognized as a seasoning ingredient commonly used in Southeast Asian cuisine. 38
Toxicokinetics
No relevant published toxicokinetics studies on Citrus flower- and leaf-derived ingredients were identified in a literature search for these ingredients and no unpublished data were submitted. Toxicokinetics data were not expected to be found because each botanical ingredient is a mixture of hundreds of constituents.
Toxicological Studies
Some of the Citrus ingredients in this assessment are found in foods, and daily exposures from food use would result in much greater systemic doses than those resulting from use in cosmetic products. Also, as noted earlier, essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from bitter orange flowers, sweet orange flowers and leaves, lemon leaves, mandarin and/or tangerine leaves, and possibly other Citrus flowers and leaves are GRAS for their intended use in foods for human and animal consumption according to the FDA. Consequently, the systemic toxicity potential is not addressed further in this report. The safety assessment focuses on the potential for irritation and sensitization from topical exposure to these Citrus ingredients.
Acute and Repeated Dose Toxicity
No relevant published acute or repeated dose toxicity studies on Citrus flower- and leaf-derived ingredients were identified in a literature search for these ingredients, and no unpublished data were submitted.
Reproductive and Developmental Toxicity
No relevant published reproductive and developmental studies on Citrus flower- and leaf-derived ingredients were identified in a literature search for these ingredients, and no unpublished data were submitted.
Genotoxicity
No relevant published genotoxicity studies on Citrus flower- and leaf-derived ingredients were identified in a literature search for these ingredients, and no unpublished data were submitted.
Carcinogenicity
No relevant published carcinogenicity studies on Citrus flower- and leaf-derived ingredients were identified in a literature search for these ingredients, and no unpublished data were submitted.
Irritation and Sensitization
Dermal Irritation
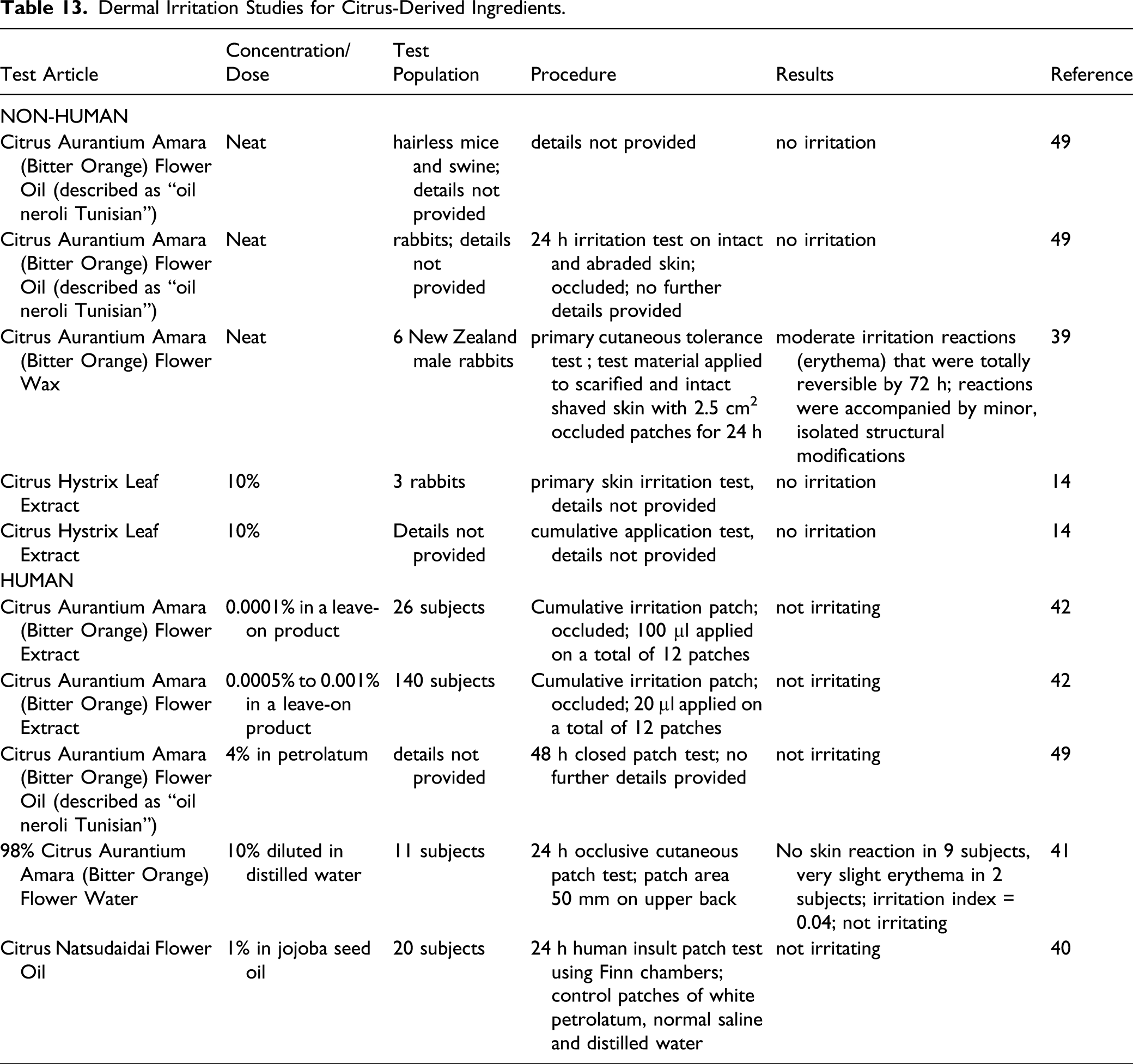
Dermal Irritation Studies for Citrus-Derived Ingredients.
Ocular Irritation
Citrus Aurantium Amara (Bitter Orange) Flower Water
The irritancy potential of a raw material product containing >98% Citrus Aurantium Amara (Bitter Orange) Flower Water was tested in a hen’s egg test-chorioallantoic membrane (HET-CAM) assay and a cytotoxicity in vitro (CFIO) method. 41 The test material was studied diluted at 10% in saline or in Hank’s balanced salt solution. While the HET-CAM assay concluded the test material was practically not irritating, the CFIO method concluded that the test material must be considered slightly irritating to the eye.
Citrus Aurantium Amara (Bitter Orange) Flower Wax
The eye tolerance of Citrus Aurantium Amara (Bitter Orange) Flower Wax (>50%) was tested in vitro using the Statens Seruminstitut rabbit cornea (SIRC) cell model. 43 Tolerance was evaluated by measuring cytotoxicity. Negative control solutions were physiological serum or sample diluent and positive control solutions were 0.01% to 0.2% sodium dodecyl sulfate. Negligible cytotoxicity was observed.
Sensitization
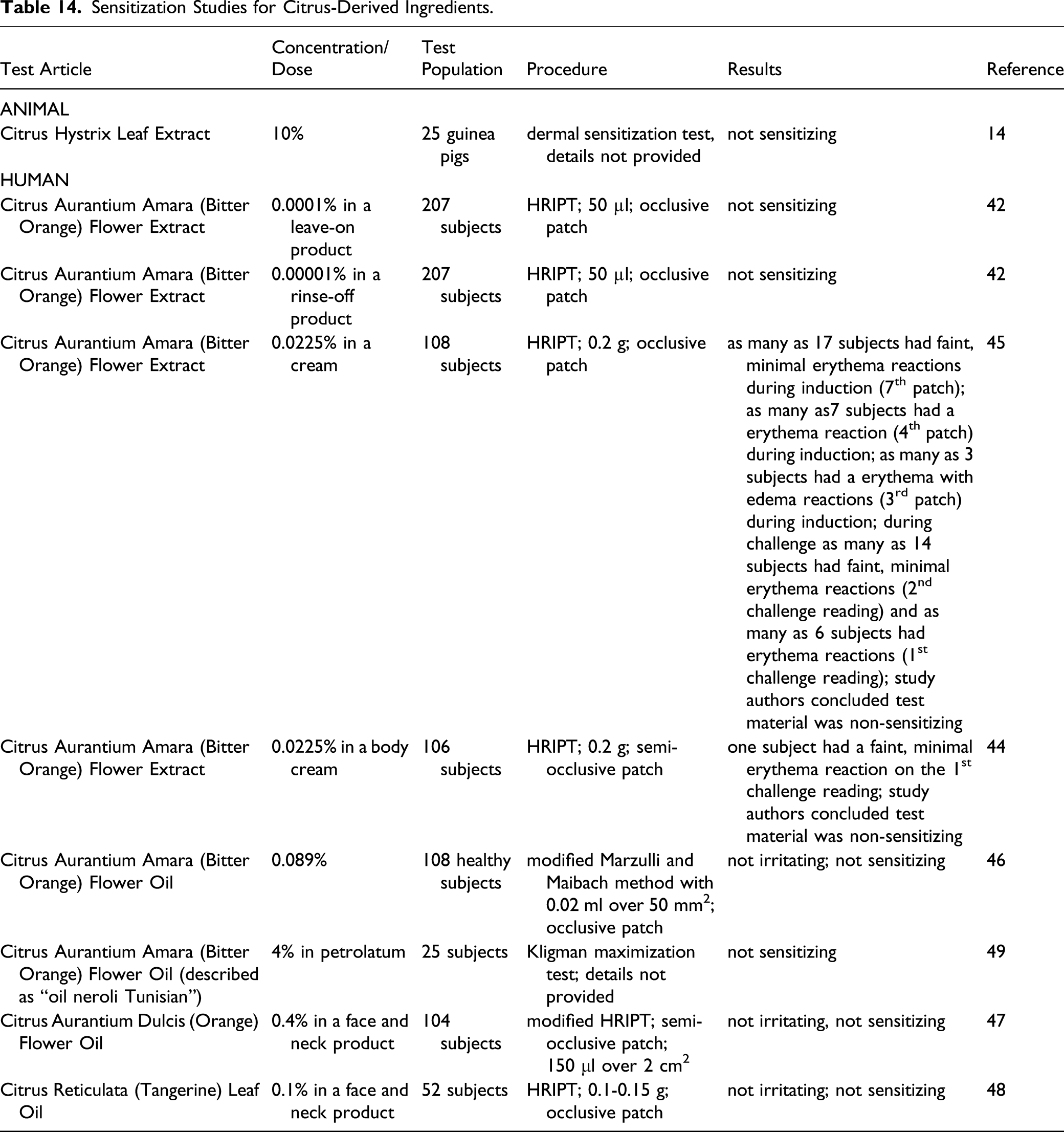
Sensitization Studies for Citrus-Derived Ingredients.
Photosensitization
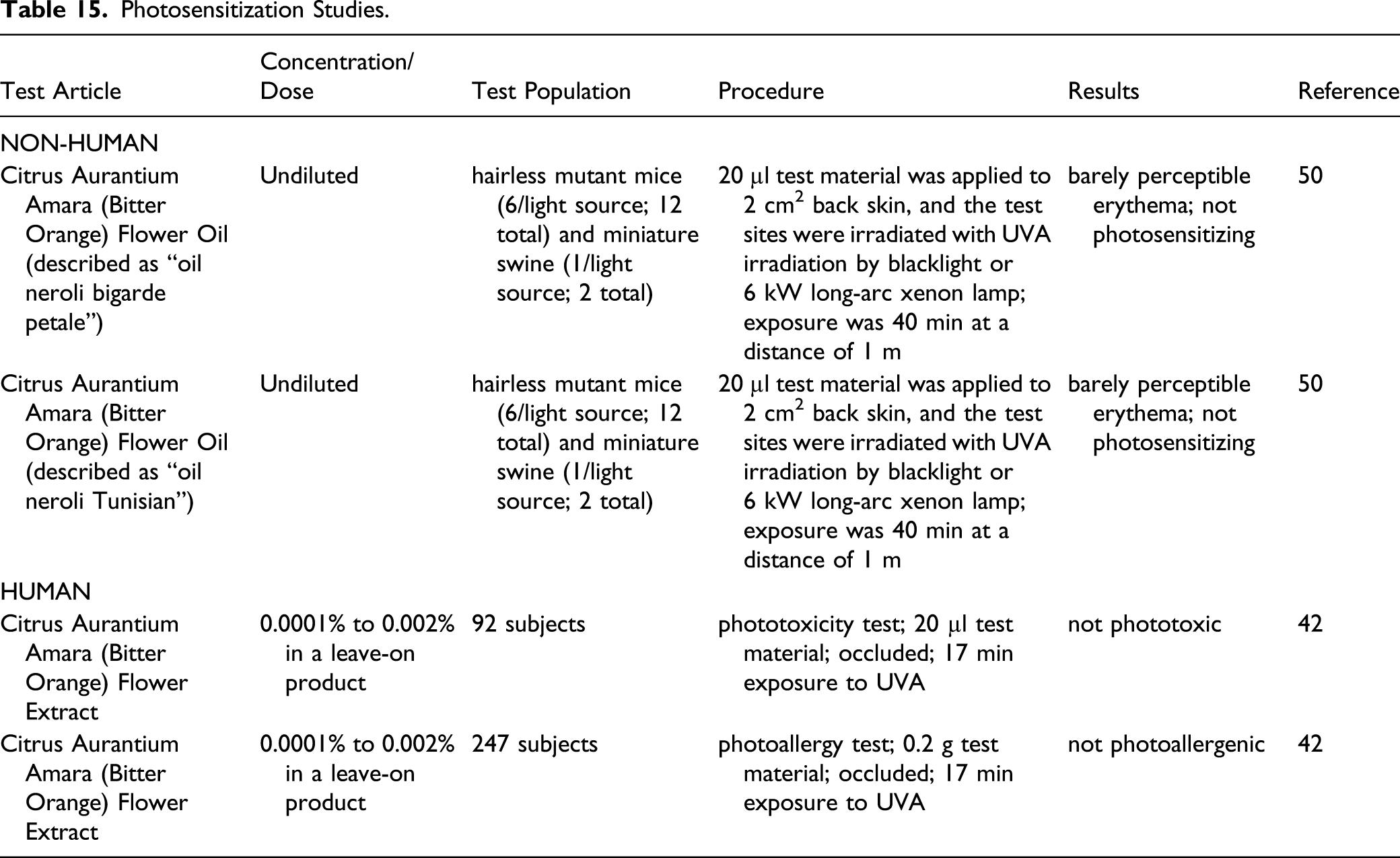
Photosensitization Studies.
Clinical Studies
No relevant published clinical studies on Citrus flower- and leaf-derived ingredients were identified in a literature search for these ingredients and no unpublished data were submitted.
Summary
The 33 Citrus flower- and leaf-derived ingredients described in this report function primarily as fragrances and/or skin-conditioning agents. Botanicals such as Citrus are composed of hundreds of constituents, some of which have the potential to be toxic. CIR reviewed the information available for each Citrus flower- and leaf-derived ingredient as a whole, complex substance; CIR did not review the potential toxicity information on the individual constituents of which the Citrus flower- and leaf-derived ingredients are composed.
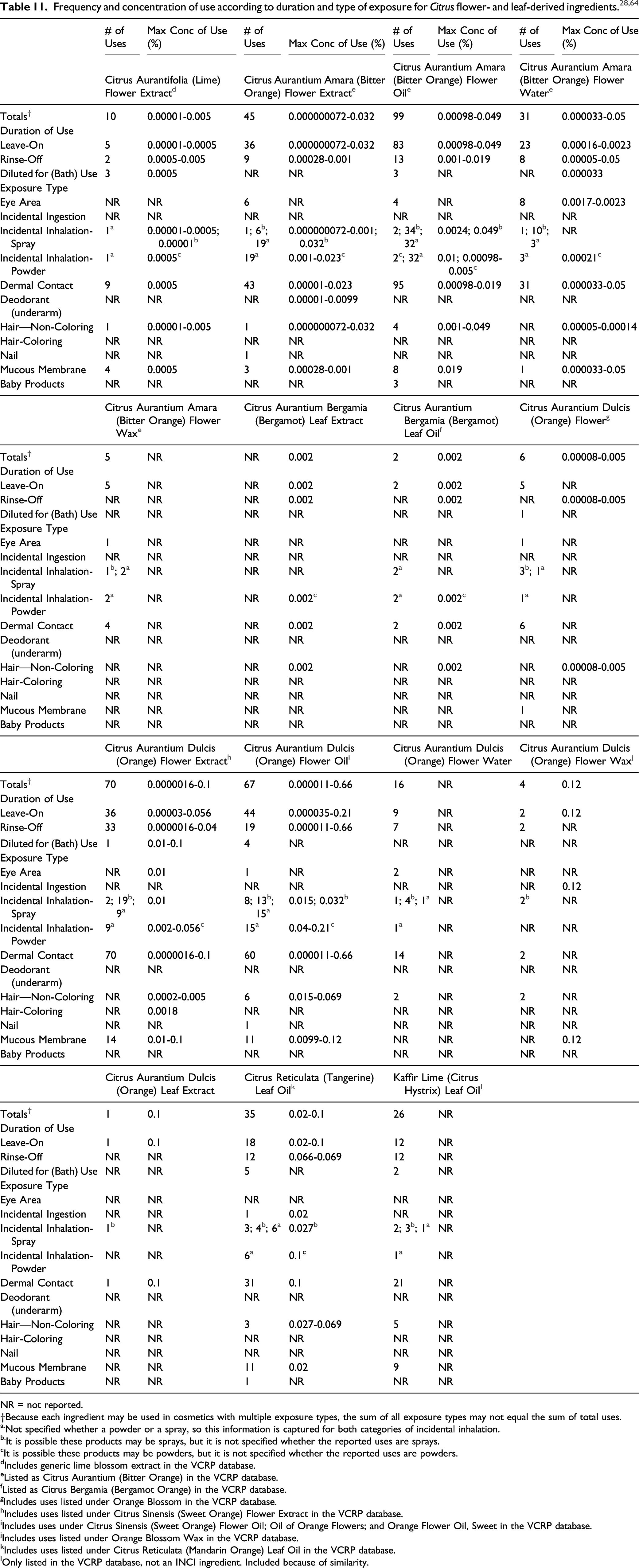
Citrus Aurantium Amara (Bitter Orange) Flower Oil has the most reported uses of the cosmetic ingredients in this report, with a total of 99; the majority of the uses are in leave-on skin care preparations. Citrus Aurantium Dulcis (Orange) Flower Extract has the second greatest number of overall uses reported, with a total of 70; a majority of the uses are in rinse-off and leave-on skin care preparations. The results of the concentration of use survey conducted by the Council indicate that Citrus Aurantium Dulcis (Orange) Flower Oil has the highest reported maximum concentration of use; it is used at up to 0.66% in a depilatory. Citrus Aurantium Dulcis (Orange) Flower Wax had the second highest reported maximum concentration of use; it is used at up to 0.12% in a lipstick.
The Citrus flower- and leaf-derived ingredients in this safety assessment are not restricted from use under the rules governing cosmetic products in the European Union.
Some of the Citrus ingredients in this assessment are found in foods, and daily exposures from food use would result in much larger systemic exposures than those resulting from use in cosmetic products. Essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from some Citrus sources (specifically Citrus aurantium (bitter orange; the flowers), Citrus sinensis (orange; the leaf and flowers), Citrus limon (lemon; leaves), and Citrus reticulata (mandarin or tangerine; leaves)) are GRAS for their intended use in foods for human and animal consumption according to the FDA. While not specifically listed as GRAS, Citrus hystrix (kaffir lime) leaves are commonly used in Southeast Asian cuisine.
In rabbit studies, moderate, reversible erythema reactions were observed with undiluted Citrus Aurantium Amara (Bitter Orange) Flower Wax. Citrus Hystrix Leaf Extract was not irritating in rabbits at up to 10%. In human subjects, no irritation was observed after topical exposure to Citrus Natsudaidai Flower Oil (1% in jojoba seed oil).
A raw material product containing >98% Citrus Aurantium Amara (Bitter Orange) Flower Water was considered practically not irritating in a HET-CAM assay but was considered slightly irritating to the eyes in a CFIO test. Essentially no cytotoxicity was observed in an in vitro eye tolerance study of Citrus Aurantium Amara (Bitter Orange) Flower Wax (>50%) using the SIRC cell strain.
Citrus Hystrix Leaf Extract was not sensitizing in guinea pigs at up to 10%. In human studies, Citrus Aurantium Amara (Bitter Orange) Flower Extract was considered not sensitizing in formulations at up to 0.0225%, and Citrus Aurantium Amara (Bitter Orange) Flower Oil was not sensitizing at 4% in petrolatum. Citrus Aurantium Dulcis (Orange) Flower Oil was not irritating or sensitizing at 0.4% in a face and neck product, and Citrus Reticulata (Tangerine) Leaf Oil was not irritating or sensitizing at 0.1% in a face and neck product.
No published studies on toxicokinetics, repeated dose toxicity, reproductive and development toxicity, genotoxicity, carcinogenicity, or clinical studies of Citrus flower- and leaf-derived ingredients were discovered and no unpublished data were submitted to address these topics.
Discussion
The Citrus ingredients in this assessment are found in foods, and daily exposures from the consumption of foods can be expected to yield much larger systemic exposures to these ingredients than those from use in cosmetic products. Essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some Citrus flowers and leaves are GRAS in foods and animal feeds. Consequently, the primary focus of this safety assessment is on the potential for irritation and sensitization from topical exposures to the Citrus ingredients.
The Panel noted that, because botanical ingredients are complex mixtures, there is concern that multiple botanical ingredients in one formulation may each contribute to the final concentration of a single constituent. Therefore, when formulating products, manufacturers should avoid reaching levels in final formulation of botanical constituents that may cause sensitization or other adverse effects. Specific examples of constituents that could induce adverse effects include the hydroperoxides of limonene and linalool.
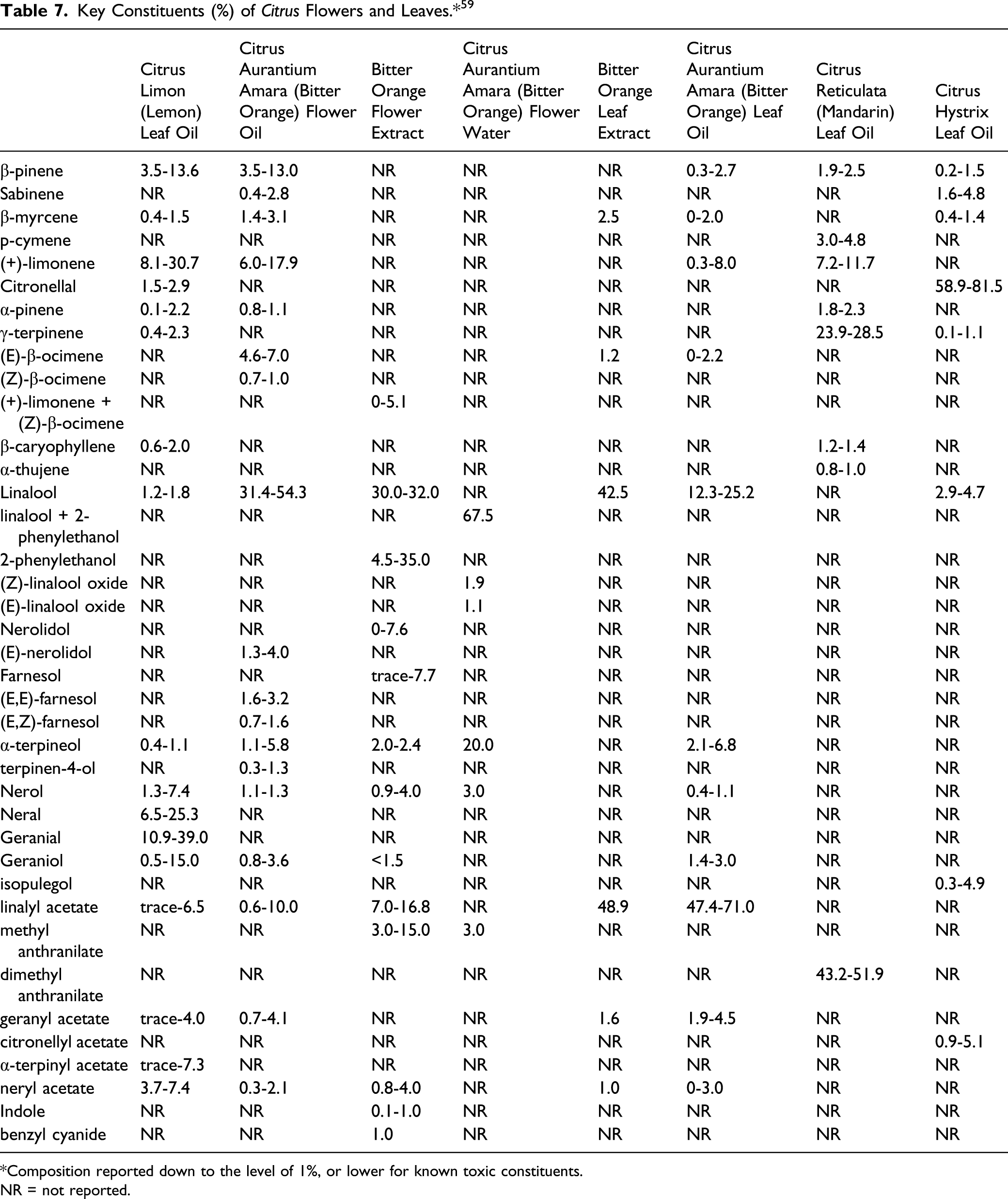
The Panel also noted that the compositions of the ingredients included in this report are highly variable, even when derived from plants within a single cultivar of a species. For example, leaf oil from some cultivars of Citrus reticulata may contain up to about 50% methyl-N-methylanthranilate (also known as dimethyl anthranilate), which is phototoxic. IFRA and the European Union SCCS have issued a 0.01% limit on this constituent in leave-on products. The maximum reported concentration of use for Citrus Reticulata (Tangerine) Leaf Oil in a leave-on product was 0.1%. Because the concentration of methyl-N-methylanthranilate in this ingredient would be below the limit established by IFRA and the SCCS, the Panel determined that there are no safety concerns about Citrus Reticulata (Tangerine) Leaf Oil as used in cosmetics.
The Panel recognized that constituents such as methyl-N-methylanthranilate may be prone to N-nitrosation. However, the use concentrations of these ingredients, of which such constituents are a small part, are so low that the Panel had no safety concerns about the possibility of N-nitrosation of these Citrus-derived ingredients.
The Panel discussed the issue of incidental inhalation exposure from hair sprays, fragrance preparations, and face powders. There were no inhalation toxicity data available. The Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel also expressed concern about pesticide residues, heavy metals, and other plant species that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Conclusion

*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Christina L. Burnett contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript.Wilma F. Bergfeld, Donald V. Belsito, Ronald A. Hill, Curtis D. Klaassen, Daniel C. Liebler, James G. Marks, Ronald C. Shank, Thomas J. Slaga, Paul W. Snyder, and Lillian J. Gill contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript.Bart Heldreth contributed to design, contributed to analysis and interpretation, and critically revised manuscript.All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.