Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 80 Citrus fruit-derived ingredients, which are most frequently reported to function in cosmetics as fragrances and/or skin-conditioning agents. The Panel reviewed the available data to determine the safety of these ingredients. Because final product formulations may contain multiple botanicals, each containing similar constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. Industry should use good manufacturing practices to limit impurities that could be present in botanical ingredients. The Panel concluded that these ingredients are safe for use in both rinse-off and leave-on cosmetic products when formulated to be non-sensitizing and non-irritating, provided that leave-on products do not contain more than 0.0015% (15 ppm) 5-methoxypsoralen (5-MOP).
Keywords
Introduction

Definitions and Functions of Citrus-Derived Ingredients.

The Panel has reviewed previously the safety of Citrus-derived peel oils and concluded that 14 Citrus-derived peel oil ingredients are safe for use in both rinse-off and leave-on cosmetic products when formulated to be non-sensitizing and non-irritating, provided that leave-on products do not contain more than 0.0015% (15 ppm) 5-methoxypsoralen (5-MOP). 2
The Citrus fruits that are used to derive the ingredients described in this safety assessment are used as food. The U.S. Food and Drug Administration (FDA) determined that the use of Citrus fruits as direct food substances is generally recognized as safe (GRAS). Additionally, essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some Citrus fruits are GRAS for their intended use in foods for human and animal consumption. Daily consumption of these GRAS foods would result in much larger systemic exposures than what is expected from use in cosmetic products, even if there was 100% absorption. Thus, the systemic toxicity potential of Citrus fruit-derived ingredients via oral exposure is not addressed further in this report. The primary focus of the safety assessment is the review of safety based on topical exposure.
Citrus Ingredients That Potentially Function Solely as Fragrance Ingredients.

Botanicals, such as Citrus ingredients, contain hundreds of constituents, some of which have the potential to cause toxic effects. For example, bergapten (also known as 5-methoxypsoralen or 5-MOP) is a naturally occurring phototoxic furanocoumarin (psoralen) in Citrus fruits. In this assessment, the Panel is reviewing the potential toxicity of each Citrus fruit-derived ingredient as a whole, complex substance. Except for specific constituents of concern that have been identified, the Panel is not reviewing the potential toxicity of the individual constituents of the Citrus fruits from which the ingredients in this report are derived.
Review of Citrus Genus Species Names. 3

Chemistry
The definitions and functions of the Citrus fruit-derived ingredients included in this report are provided in Table 1. In some cases, the definition provides insight on the method(s) of manufacture. It should be noted that the term “water” in Citrus waters refers to the water soluble fraction of the condensate from steam distillation of the plant parts of interest. Essential oils are the hydrophobic, liquid, volatile aroma compounds in the insoluble condensate fraction. The essential oils are typically small molecules, but their chemical structures can vary rather widely. Fixed oils, on the other hand are hydrophobic, non-volatile, fatty compounds from plants, animals, or algae. These are primarily composed of glycerides, and to some extent, free fatty acids. Constituents of these Citrus-derived ingredients may include both oil types. The volatile nature of essential oils makes them more likely to be useful as fragrances, but that does not necessitate that fragrance is their only function.
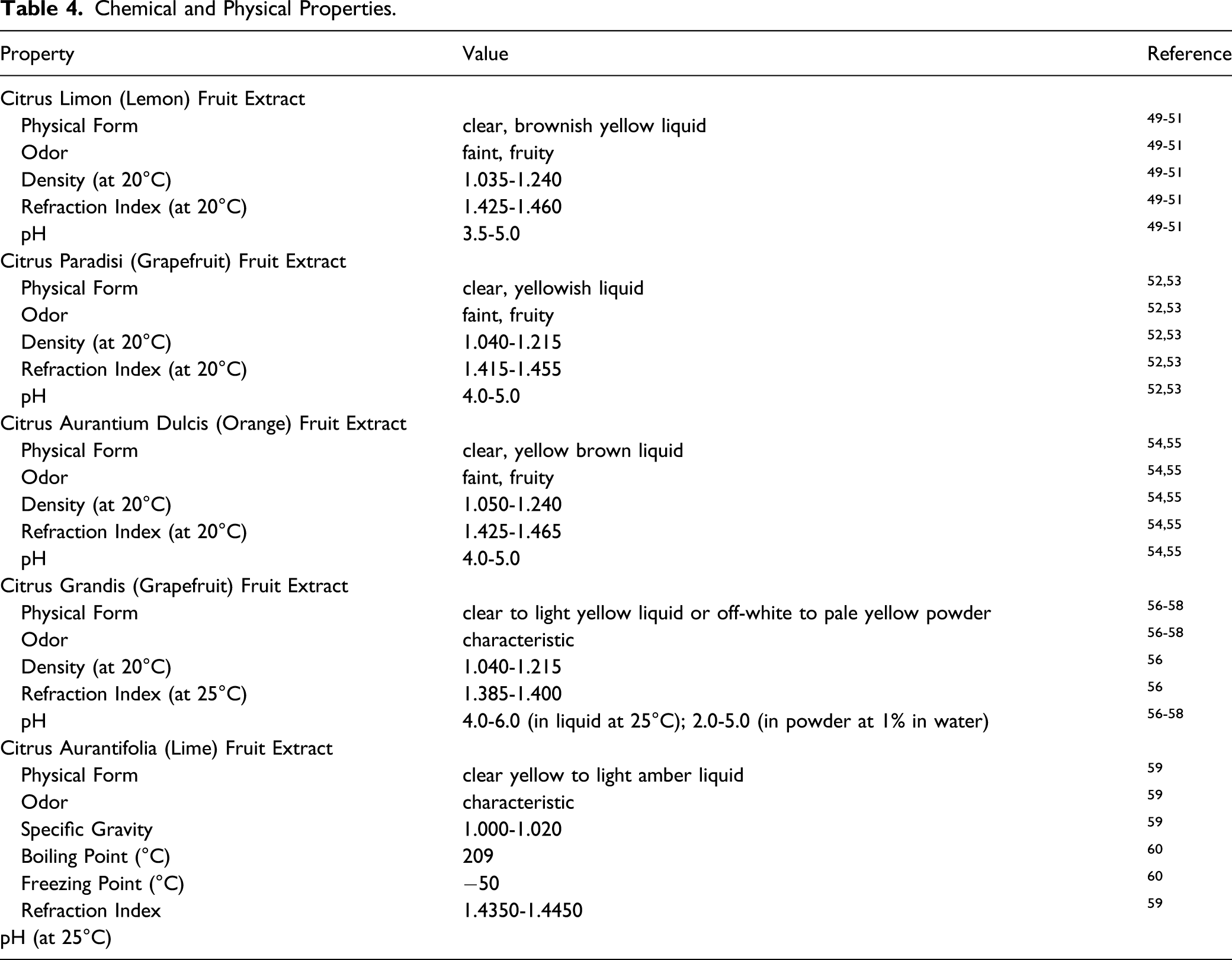
Chemical and Physical Properties
Chemical and Physical Properties.
Method of Manufacturing
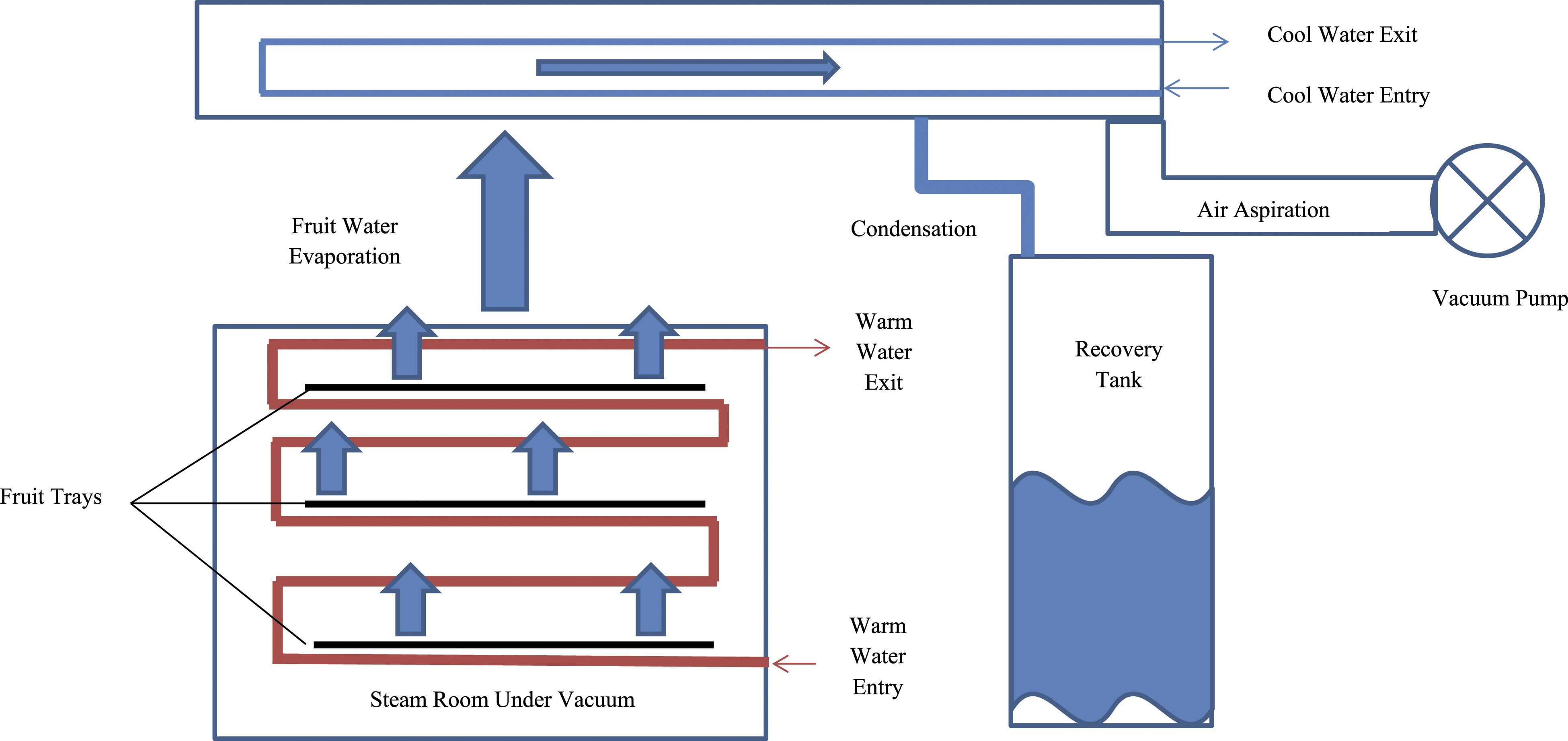
Figure 1 is a generic representation of the method of manufacturing for Citrus fruit waters. Figures 2 and 3 are the manufacturing flow charts provided by suppliers of Citrus Nobilis (Mandarin Orange) Fruit Extract and Citrus Grandis (Grapefruit) Fruit Extract. Method of manufacturing of fruit waters.
46
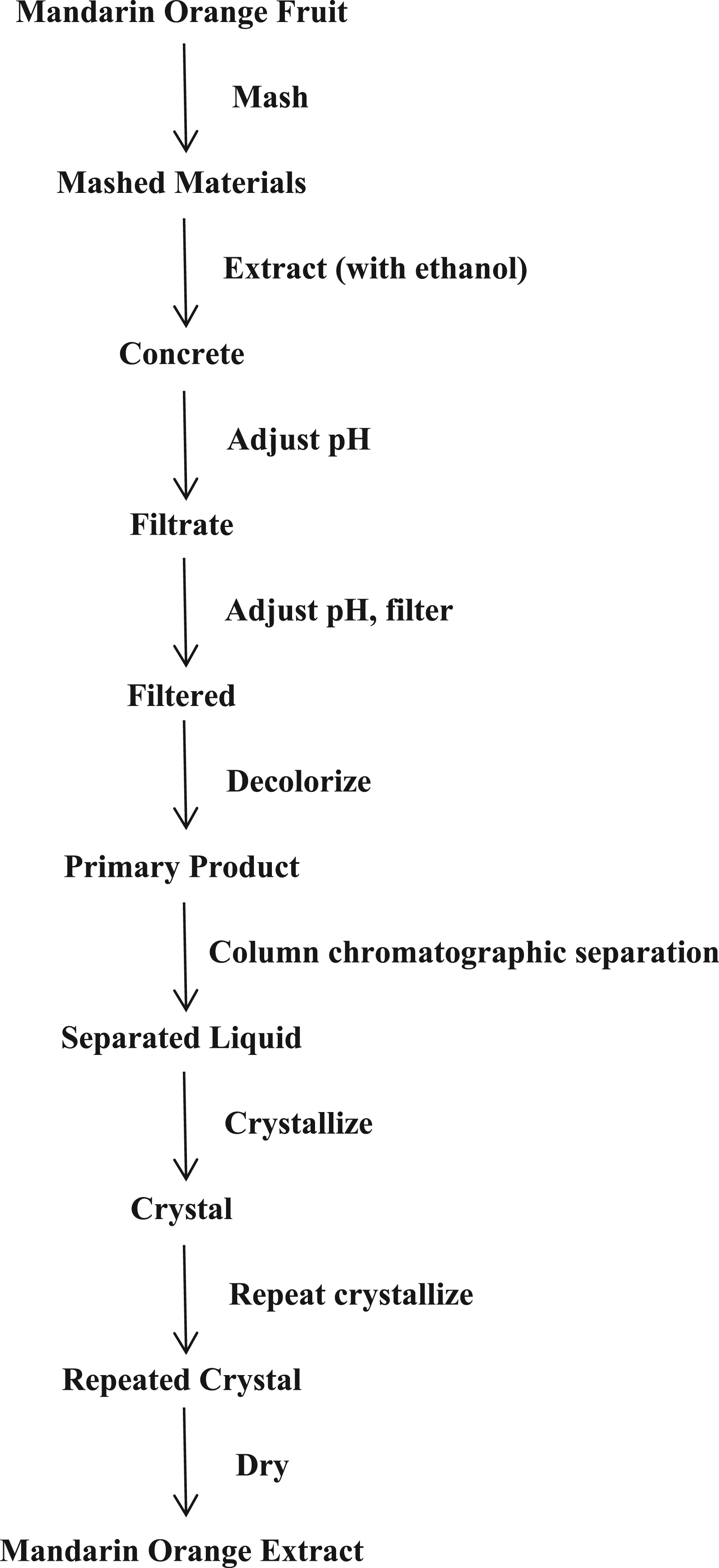
Manufacturing flow chart for Citrus Nobilis (Mandarin Orange) Fruit Extract.
47
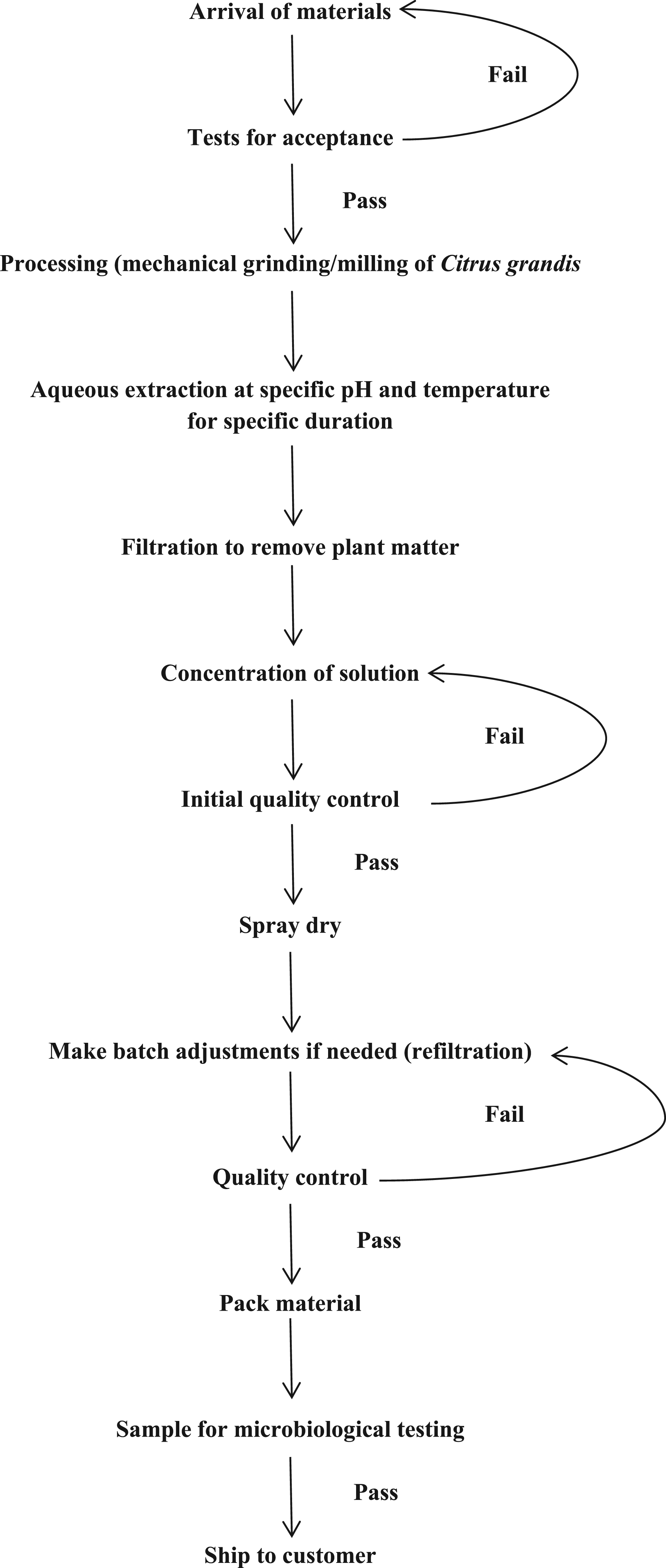
Manufacturing flow chart for Citrus Grandis (Grapefruit) Fruit Extract—Powder Form.
48
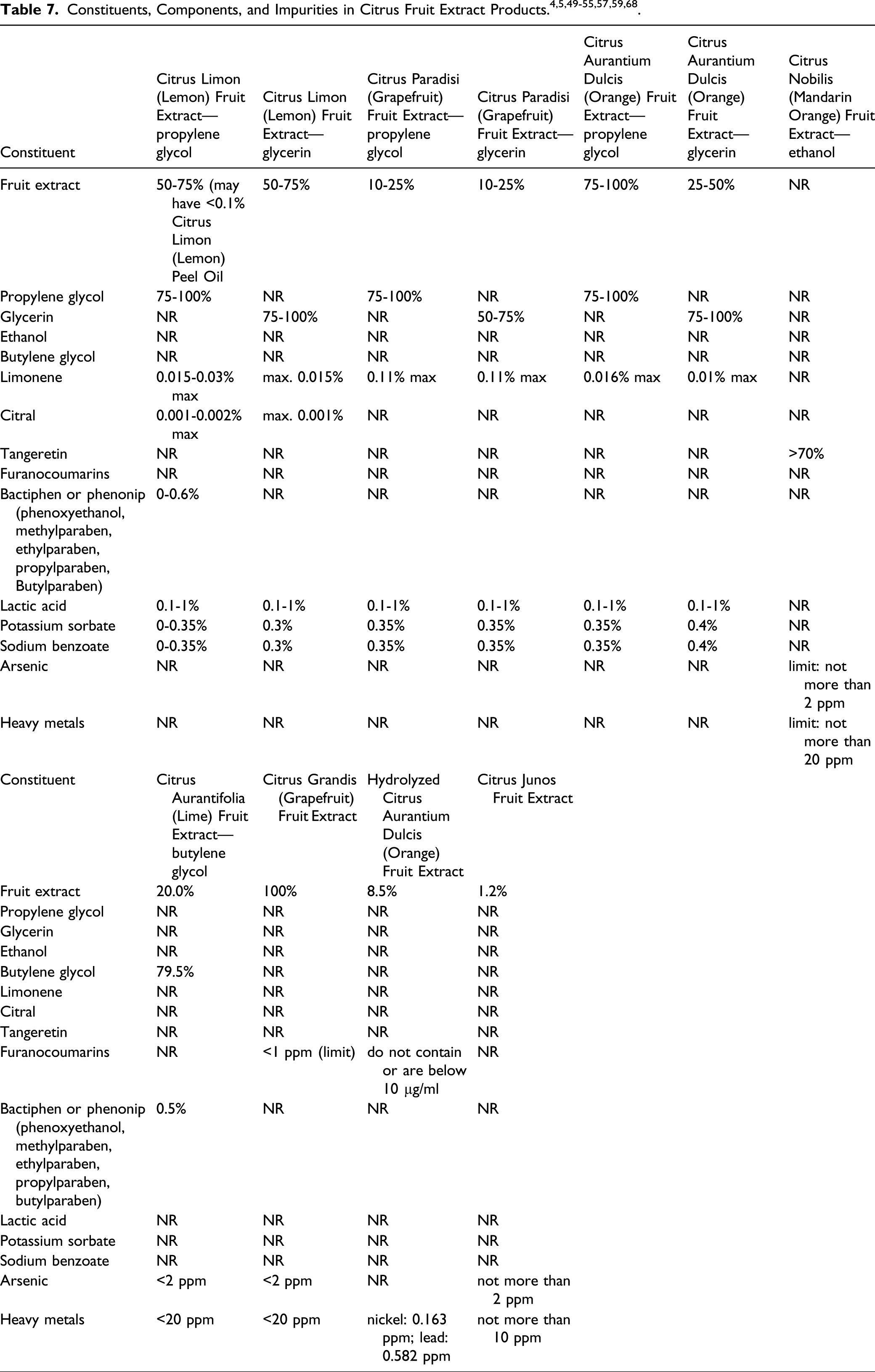
A supplier reports that Hydrolyzed Citrus Aurantium Dulcis (Orange) Fruit is produced by solubilization of Citrus aurantium dulcis fruit in water, followed by enzymatic hydrolysis, separation of soluble and insoluble phases, filtration, and sterilizing filtration. 4 Another supplier reports that Citrus Junos Fruit Extract is produced by extracting the dried raw fruit with 50% vol. ethanolic solution, filtering, allowing sedimentation before adjusting the product, and packaging. 5
Constituents/Composition
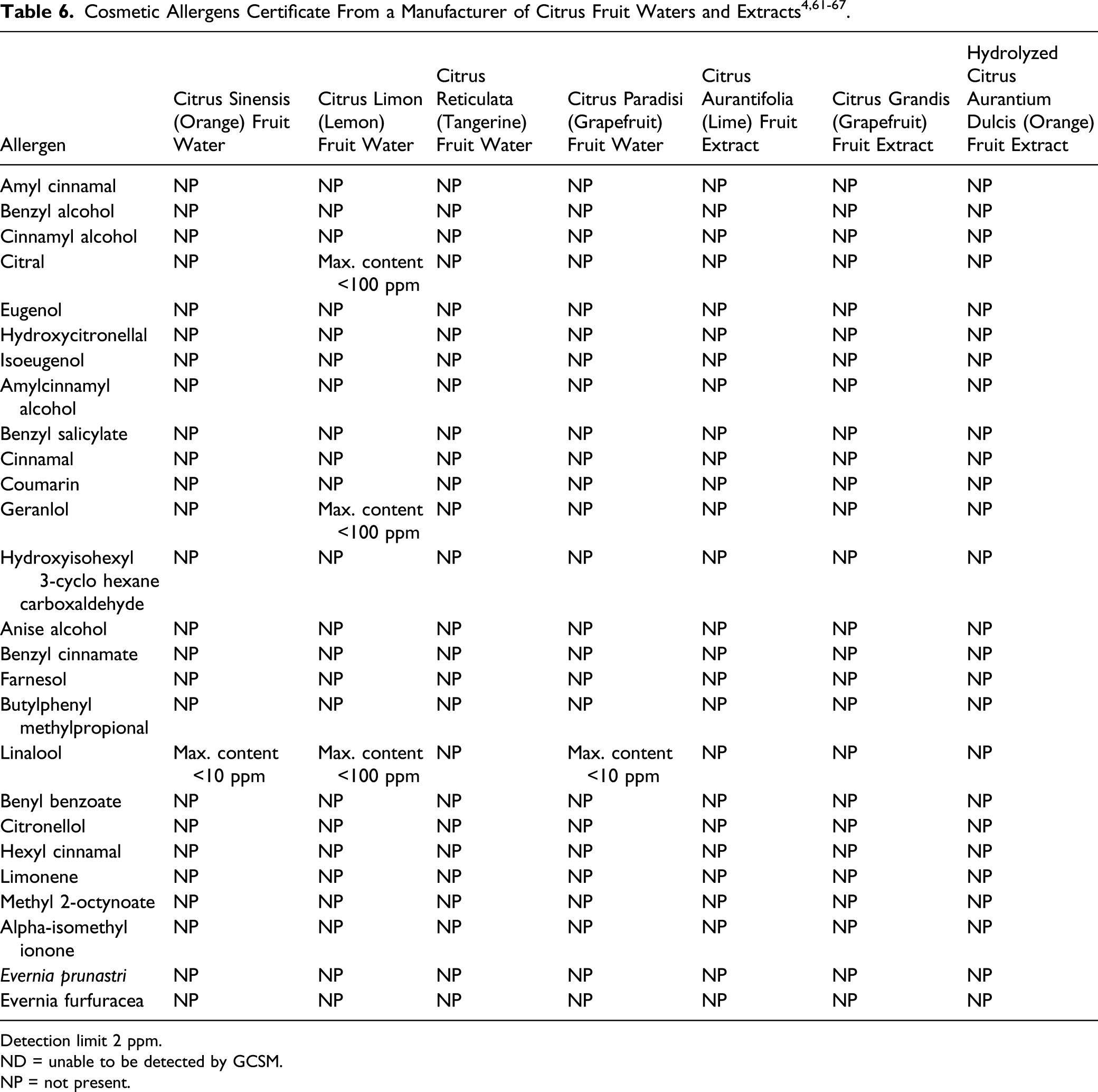
Potential Constituents That Are Established Contact Allergens in Humans, According to the Scientific Committee on Consumer Safety.
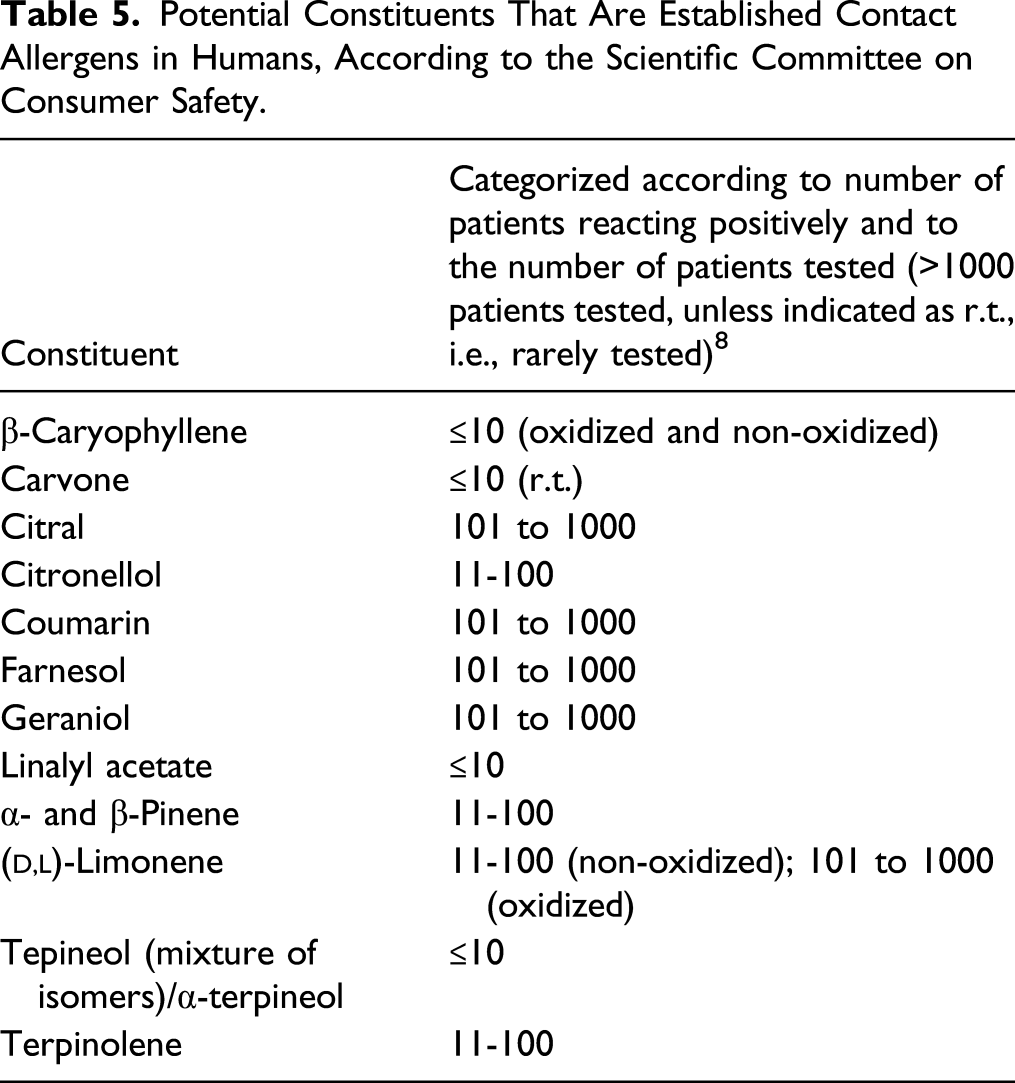
Detection limit 2 ppm.
ND = unable to be detected by GCSM.
NP = not present.
Constituents of Citrus aurantium (Bitter Orange). 69

Additional Constituent Data From Various Citrus Fruits. 70

Use
Cosmetic
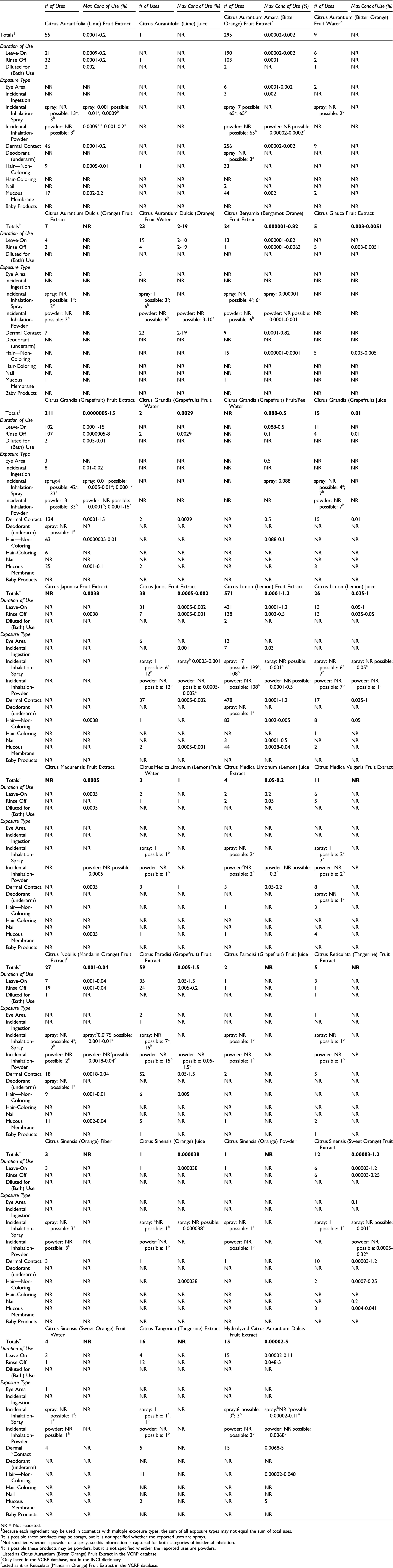
The safety of these cosmetic ingredients is evaluated on the basis of the expected use in cosmetics. The Panel utilizes data received from the FDA and the cosmetics industry in determining safety. The data received from the FDA are those it collects from manufacturers on the use of individual ingredients in cosmetics by cosmetic product category in its VCRP, and those from the cosmetic industry are submitted in response to a survey of the maximum reported use concentrations by category conducted by the Council.
NR = Not reported.
†Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
aIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
bNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
cIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
dListed as Citrus Aurantium (Bitter Orange) Fruit Extract in the VCRP database.
eOnly listed in the VCRP database, not in the INCI dictionary.
fListed as itrus Reticulata (Mandarin Orange) Fruit Extract in the VCRP database.
In some cases, reports of uses were received from the VCRP, but no concentration of use data were provided. For example, Citrus Medica Vulgaris Fruit Extract is reported to be used in 11 formulations, but no use concentration data were available. In other cases, no reported uses were reported to the VCRP, but a maximum use concentration was provided in the industry survey. For example, Citrus Japonica Fruit Extract was not reported in the VCRP database to be in use, but the industry survey indicated that it is used in non-coloring hair conditioners at up to 0.0038%. It should be presumed that Citrus Japonica Fruit Extract is used in at least one cosmetic formulation.
Ingredients That are Not Reported to Be in Use.

Some of these ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes. For example, Citrus Limon (Lemon) Fruit Extract is used at 0.03% in lipstick and Citrus Aurantifolia (Lime) Fruit Extract is used at 0.2% in personal cleanliness products. Additionally, some of these ingredients were reported to be used in hair sprays and body and hand sprays and could possibly be inhaled. For example, Citrus Nobilis (Mandarin Orange) Fruit Extract was reported to be used in body and hand sprays at a maximum concentration of 0.0075%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.11-14 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (i.e., they would not enter the lungs) to any appreciable amount.12,13
Under the European Commission’s regulations concerning cosmetic products Annex II List of Substances Prohibited in Cosmetic Products, furocoumarins (including 5-MOP) are prohibited in cosmetics except for normal content in natural essences and must be below 1 mg/kg in sun-protection products and in bronzing products. 15 The International Fragrance Association (IFRA) has issued standards for Citrus oils and other furocoumarin-containing essential oils. 16 Finished products that are applied to the skin, excluding rinse-off products like bath preparations and soaps, must not contain more than 0.0015% or 15 ppm 5-MOP. This equates to a level of 0.0075% or 75 ppm in a fragrance compound when used at 20% in a consumer product that is applied to the skin. If the level of 5-MOP has not been determined, IFRA limits specified for individual oils should be observed, and when such oils are used in combination with other phototoxic constituent containing ingredients, the potential for an additive effect should be considered and use levels should be reduced accordingly. 16
Non-Cosmetic
The Citrus fruits that are used to derive the ingredients described in this safety assessment are used as food. Per 21CFR§170.30(c) (1) and (2) and §170.30(c): “… food ingredients of natural biological origin that has been widely consumed for its nutrient properties in the United States prior to January 1, 1958, without known detrimental effects, which is subject only to conventional processing as practiced prior to January 1, 1958, and for which no known safety hazard exists, will ordinarily be regarded as generally recognized as safe (GRAS) without specific inclusion in 21CFR§182, §184, or §186.1.”
Further…“A substance used in food prior to January 1, 1958, may be GRAS through experience based on its common use in food when that use occurred exclusively or primarily outside of the United States if the information about the experience establishes that the use of the substance is safe within the meaning of the act (see §170.3(i)). Common use in food prior to January 1, 1958, that occurred outside of the United States shall be documented by published or other information and shall be corroborated by information from a second, independent source that confirms the history and circumstances of use of the substance. The information used to document and to corroborate the history and circumstances of use of the substance must be generally available; that is, it must be widely available in the country in which the history of use has occurred and readily available to interested qualified experts in this country. Persons claiming GRAS status for a substance based on its common use in food outside of the United States should obtain U.S. Food and Drug Administration concurrence that the use of the substance is GRAS.”
The essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from the following Citrus fruits are GRAS for their intended use in foods for human consumption: Citrus aurantifolia (lime); Citrus aurantium (bergamot); Citrus aurantium (bitter orange; the flowers and peel); Citrus limon (lemon); Citrus paradisi (grapefruit); Citrus reticulata (tangerine); Citrus reticulata blanco (mandarin); Citrus sinensis (orange; the leaf, flowers, and peel); and Citrus peels (species not specified) (21CFR§182.20). These essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of these Citrus fruits are GRAS for their intended use in animal drugs, feeds, and related products (21CFR§582.20).
Essential (or volatile) oils of limes, lemons, grapefruits, bitter oranges, oranges, and tangerines are described as flavoring agents in the Food Chemicals Codex, a compendium of internationally recognized standards published by the United States Pharmacopeia (USP) for the purity and identity of food ingredients. 17
Citrus aurantium amara (bitter orange) and extracts of its dried fruit and peel have been used in traditional Western medicines and in Chinese and Japanese herbal medicines. 18
Toxicological Studies
As noted earlier, the Citrus ingredients in this assessment are found in foods, and daily exposures from food use would result in a much larger systemic exposure than those from use in cosmetic products. Essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some Citrus fruits are GRAS for their intended use in foods for human and animal consumption according to the FDA. Volatile oils of limes, lemons, grapefruits, bitter oranges, oranges, and tangerines are described as flavoring agents in the USP Food Chemicals Codex. Because the safety of these ingredients has been established for foods, and the systemic exposure from foods would be much greater than exposure from use of these ingredients in cosmetics, the systemic toxicity potential of these ingredients is not addressed further in this report. The primary focus of this safety assessment is on the potential for irritation and sensitization from topical exposure to these Citrus ingredients as used in cosmetic products.
Genotoxicity
In Vitro
Citrus Nobilis (Mandarin Orange) Fruit Extract in dimethyl sulfoxide was analyzed for mutagenic potential in an Ames assay using Salmonella typhimurium strains TA97a, TA98, TA100, TA102, and TA1535, with and without S9 metabolic activation. 19 Concentrations tested were 0.05, 0.1, 0.5, 1.0, and 5.0 mg/plate. Positive and negative controls yielded expected results. No sign of mutagenicity was observed with or without S9. The test material was cytotoxic at 5.0 mg/plate. It was concluded that Citrus Nobilis (Mandarin Orange) Fruit Extract was not mutagenic, with or without metabolic activation.
Citrus Junos Fruit Extract (1.2% w/v; ethanol extract) was not mutagenic in an Ames assay using S typhimurium strains TA98, TA100, TA1535, or TA1537 and Escherichia coli strain WP2uvrA. 5 No further details were provided.
Irritation and Sensitization
Ocular Irritation
Citrus Grandis (Grapefruit) Fruit Extract (100% in powder form) was not an ocular irritant in an EpiOcular eye irritation test. 20 Citrus Nobilis (Mandarin Orange) Fruit Extract was not an ocular irritant when tested up to 100% in a hen egg chorioallantoic membrane assay (HET-CAM). 21 Undiluted Citrus Sinensis (Orange) Fruit Water (0.3 g) also was not an ocular irritant in a HET-CAM assay. 22 Hydrolyzed Citrus Aurantium Dulcis (Orange) Fruit Extract (8.5%) was slightly irritating when tested in 3 New Zealand rabbit eyes according to the method 405 of the Organisation for Economic Co-operation and Development (OECD). 4
Dermal Irritation and Sensitization
Dermal Irritation and Sensitization Studies.

Phototoxicity and Photosensitization
Photosensitization and Phototoxicity Studies.

Case Reports
Epidemiology
The risk of cutaneous malignant melanoma associated with psoralen-rich citrus consumption was analyzed in a prospective study using data from the Nurses’ Health Study (63 810 women from 1984 to 2010) and the Health Professional Follow-Up Study (41 622 men from 1986 to 2010). 45 Dietary information was repeatedly assessed every 2 to 4 years during follow-up of the participants. Incident melanoma cases were identified through self-reporting and confirmed by pathologic records. In the 24–26 years of follow-up, 1840 cases of melanoma were documented. After adjusting for other risk factors, the pooled multivariable hazard ratios for melanoma were 1.00 for overall citrus consumption that was less than twice per week (reference), 1.10 (95% confidence interval (CI) 0.94-1.30) for consumption 2 to 4 times per week, 1.26 (95% CI 1.08-1.47) for consumption 5 to 6 times per week, 1.27 (95% CI 1.09-1.49) for consumption once to 1.5 times per day, and 1.36 (95% CI 1.14-1.63) for consumption greater than or equal to1.6 times per day (P < 0.001). Citrus analyzed for consumption included grapefruit (half), orange (one whole), and grapefruit and orange juice (6 ounce serving). Of the citrus products consumed, grapefruit showed the most apparent association with risk of melanoma, which was independent of other lifestyle and dietary factors. The pooled multivariable hazard ratio for melanoma comparing the extreme consumption categories of grapefruit (greater than or equal to 3 times/week vs never) was 1.41 (95% CI 1.10 to 1.82, P < 0.001). The authors of the study concluded that citrus consumption was associated with an increased risk of malignant melanoma in 2 cohorts of men and women, but stated that further studies are needed to confirm the findings.
Summary
The 80 Citrus fruit-derived ingredients described in this report function primarily as skin-conditioning agents-miscellaneous and fragrance. Botanicals such as Citrus are composed of hundreds of constituents, some of which have the potential to cause toxic effects; for example, bergapten (also known as 5-methoxypsoralen or 5-MOP) is a naturally occurring, phototoxic furanocoumarin (psoralen) in Citrus fruits. Presently, the Panel reviewed the information available for each Citrus fruit-derived ingredient as a whole, complex substance; the Panel did not review the potential toxicity information on the individual constituents of which the Citrus fruit-derived ingredients are comprised.
Citrus Limon (Lemon) Fruit Extract has the most reported uses in cosmetic products, with a total of 571; more than half of the uses are in leave-on skin care preparations. The maximum use concentration range for Citrus Limon (Lemon) Fruit Extract is 0.0001% to 1.2%, with 1.2% reported in night skin care products. Citrus Aurantium Amara (Bitter Orange) Fruit Extract has the second greatest number of overall uses reported, with a total of 295; more than half of those uses are in leave-on skin care preparations. Citrus Aurantium Amara (Bitter Orange) Fruit Extract had a maximum use concentration range of 0.00002% to 0.002%, with 0.002% reported in lipstick and eye makeup preparations. Most of the other use concentrations that were reported had similar ranges. However, maximum use concentrations as high as 19% were reported for Citrus Aurantium Dulcis (Orange) Fruit Water in paste masks and mud packs and as high as 15% for Citrus Grandis (Grapefruit) Fruit Extract in face and neck products.
Under the European Commission’s regulations concerning cosmetic products, furocoumarins (including 5-MOP) are prohibited in cosmetics except for normal content in natural essences and must be below 1 mg/kg in sun-protection products and in bronzing products. IFRA also has issued standards for Citrus oils and other furocoumarin-containing essential oils. Finished products that are applied to the skin, excluding rinse-off products like bath preparations and soaps, must not contain more than 0.0015% or 15 ppm 5-MOP. If the level of 5-MOP has not been determined, limits specified for individual oils should be observed, and when such oils are used in combination with other phototoxic ingredients, the potential additive effect should be taken into consideration and use levels should be reduced accordingly.
The Citrus fruit-derived ingredients in this assessment are found in foods, and the daily exposure from food use would result in a much larger systemic dose than that resulting from use in cosmetic products. Essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from some Citrus fruits are GRAS for their intended use in foods for human and animal consumption according to the FDA.
Citrus Grandis (Grapefruit) Fruit Extract (100% in powder form) and Citrus Nobilis (Mandarin Orange) Fruit Extract (up to 100%) were not ocular irritants in in vitro studies.
Citrus Grandis (Grapefruit) Fruit Extract (100% in powder form) was not a dermal irritant in an vitro study. Fruit extracts of Citrus Aurantium Bergamia (Bergamot) (0.081525%), Citrus Aurantium Dulcis (Orange) (1.2%), Citrus Limon (Lemon) (1.2%), Citrus Nobilis (Mandarin Orange) (1%), and Citrus Grandis (Grapefruit) (0.16%) and fruit waters of Citrus Aurantium Dulcis (Orange) (38%) and Citrus Limon (Lemon) (1%) were not sensitizing in HRIPTs.
Phytophotodermatitis was not observed in rats following exposure to undiluted lemon fruit juice and natural sunlight. No reactions were observed in human subjects exposed to 0.081525% Citrus Aurantium Bergamia (Bergamot) Fruit Extract and UVA/UVB light or undiluted pure or extracts of orange mesocarp or fruit and UVA light. Phytophotodermatitis was noted in numerous patients exposed to the juices of lemons or limes.
A prospective study of men and women health professionals has found an association between citrus consumption and malignant melanoma.
Discussion
The Citrus ingredients in this assessment are found in foods, and daily exposures from the consumption of foods can be expected to yield much larger systemic exposures to these ingredients than those from the use of cosmetic products. Essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some Citrus fruits are GRAS in foods and animal feeds. Additionally, volatile oils of limes, lemons, grapefruits, bitter oranges, oranges, and tangerines are used as flavoring agents. Consequently, the primary focus of this safety assessment is on the potential for skin irritation and sensitization from topical exposures to the Citrus ingredients.
The Panel expressed concern about the potential for constituents in Citrus fruit-derived ingredients, including the furocoumarin 5-MOP, to cause phototoxicity. IFRA has issued standards for Citrus oils and other furocoumarin-containing essential oils, and the Panel agreed that adherence to the IFRA standards for such constituents will prevent phototoxicity. According to these standards, finished products that are applied to the skin, excluding rinse-off products, must not contain more than 0.0015%, or 15 ppm, 5-MOP.
The Panel reviewed the findings of the prospective study that reported that there may be an increased risk for malignant melanoma from the consumption of citrus fruits and juices. The Panel noted that the hazard ratios are low in this study, with some confidence intervals including the background value 1.0, which indicates that the results are barely statistically significant. Additionally, any risk that is associated with the consumption of the citrus fruit and juice may be associated with the psoralen content, for which the Panel has advised a limit in cosmetics. Finally, this study lacked some controls, including a geographic control for consumption in areas where citrus fruits are grown and more readily available to the public (e.g., the southern United States). The Panel also noted the lack of evidence for mutagenicity of constituents of these citrus ingredients and their favorable safety profile. Thus, the Panel did not find the results of this study to be significant with regard to cosmetics.
The Panel noted that, because botanical ingredients are complex mixtures, there is concern that multiple botanical ingredients may each contribute to the final concentration of a single constituent. Therefore, when formulating products, manufacturers should avoid reaching levels in final formulation of botanical constituents that may cause sensitization or other adverse effects. Specific examples of constituents that could induce adverse effects are synephrine, limonene, citral and other monoterpenes, and furocoumarins (such as 5- MOP).
Additionally, during the assessment of safety for the Citrus-derived peel oils, the Panel was concerned with findings of a rodent carcinogenicity study in which tumor promotion activity may have been caused by repeated skin irritation and resultant proliferation of DMBA-treated basal cells. The Panel concluded that Citrus-derived peel oils could potentially act as tumor-promoters if formulated to produce irritation. While no significant skin irritation was reported following the use of Citrus fruit-derived ingredients, the Panel felt that these botanical ingredients must be formulated to be non-irritating.
The Panel discussed the issue of incidental inhalation exposure from hair sprays and body and hand sprays. There were no inhalation toxicity data available. The Panel considered pertinent data indicating that incidental inhalation exposures to Citrus ingredients in such cosmetic products would not cause adverse health effects, including data characterizing the potential for Citrus ingredients to cause ocular or dermal irritation or sensitization. The Panel noted that 95%–99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at the Cosmetic Ingredient Review (CIR) website, http://www.cir-safety.org/cir-findings.
Finally, the Panel expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Conclusion

*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.