Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 5 acyl sarcosines and 9 sarcosinate salts as used in cosmetics; all of these ingredients are reported to function in cosmetics as hair conditioning agents and most also can function as surfactants—cleansing agents. The ingredients reviewed in this assessment are composed of an amide comprising a fatty acyl residue and sarcosine and are either free acids or simple salts thereof. The Panel relied on relevant new data, including concentration of use, and considered data from the previous Panel report, such as the reaction of sarcosine with oxidizing materials possibly resulting in nitrosation and the formation of N-nitrososarcosine. The Panel concluded that these ingredients are safe as used in cosmetics when formulated to be non-irritating, but these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed.
Introduction

Concentration of use data was not provided at the time of the original safety assessment; because those values were not available, the concentration limit of 5% was established for leave-on products based upon the highest concentration tested in human repeat-insult patch tests. Concentration of use data is now available, and additional new relevant data have been discovered; therefore, the Panel re-opened the 2001 safety assessment to reassess the original conclusion.

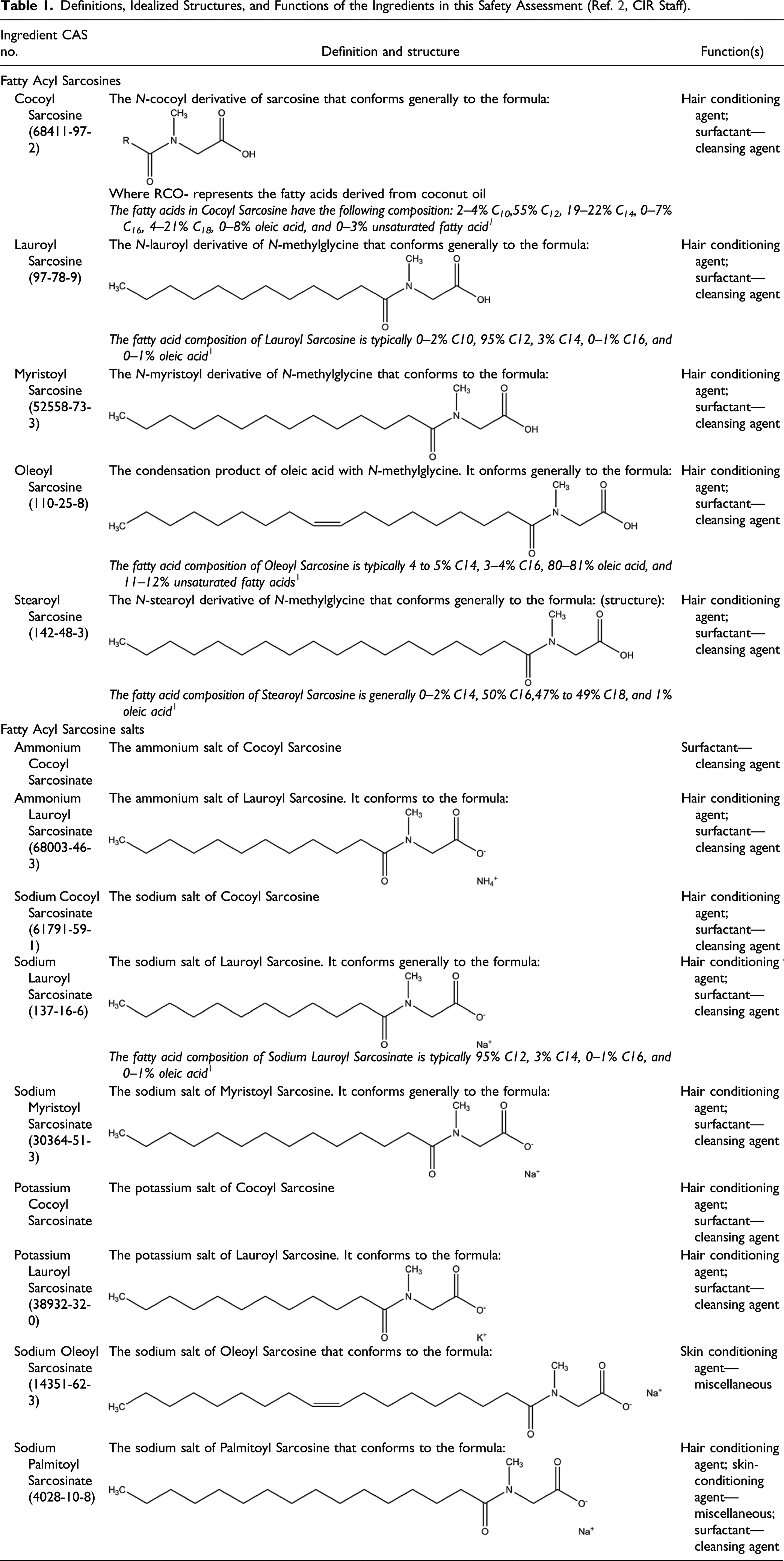
Definitions, Idealized Structures, and Functions of the Ingredients in this Safety Assessment (Ref. 2, CIR Staff).
Excerpts from the summary of the 2001 report are disseminated throughout the text of this re-review document, as appropriate. (This information is not included in the summary section.)
Several previous Cosmetic Ingredient Review (CIR) safety assessments are relevant to this safety assessment because they discuss the safety of components of the acyl sarcosines and sarcosinate salts. In 2011, the Panel concluded that Cocos Nucifera (Coconut) Oil and Elaeis Guineensis (Palm) Oil are safe in the present practices of use and concentration. 3 In 1987, the Panel published a report with the conclusion that Oleic, Lauric, Palmitic, Myristic, and Stearic Acids are safe in present practices of use and concentration in cosmetics 4 ; this conclusion was reaffirmed in 2006. 5
This safety assessment includes relevant published and unpublished data that are available for each endpoint that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the endpoints that the Panel typically evaluates, is provided on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Much of the new toxicity data included in this safety assessment was found on the European Chemicals Agency (ECHA) website. 6 The ECHA website provides summaries of information generated by industry, and it is those summary data that are reported in this safety assessment when ECHA is cited.
Chemistry
Definition and Structure
Sarcosine, also known as N-methylglycine or N-methylaminoacetic acid, is derived from the decomposition of creatine or caffeine.
1
Sarcosine is also a naturally occurring amino acid found in marine animals. It conforms generally to the formula shown in Figure 1. Sarcosine.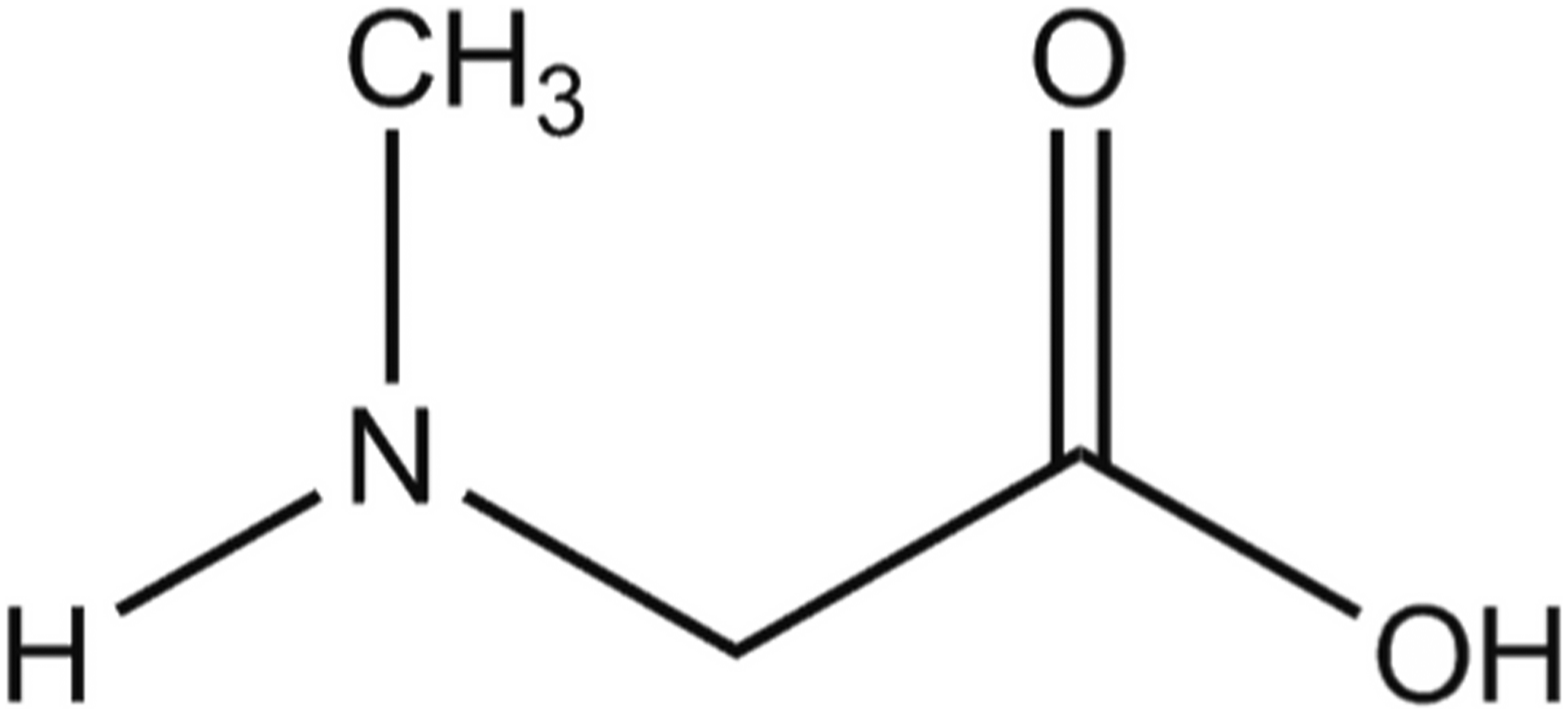
The ingredients in this report are each an amide comprising a fatty acyl residue and sarcosine, with connectivity occurring via the nitrogen atom of sarcosine and the carbonyl of the fatty acyl residue. These ingredients are either free acids (the carboxylic functional group of the sarcosine residue), or are simple salts thereof Figure 2. The salts in this report recite the term “sarcosinate” in the name and were referred to in the previous report as “sarcosinates” or “sarcosinates amides.” Since these previously utilized terms could erroneously be interpreted to mean esters or amides with connectivity through the carbonyl of sarcosine, these salts are hereto referred to simply as fatty acyl sarcosine salts. Myristoyl Sarcosine and Sodium Myristoyl Sarcosinate, a representative fatty acyl sarcosine and salt, respectively.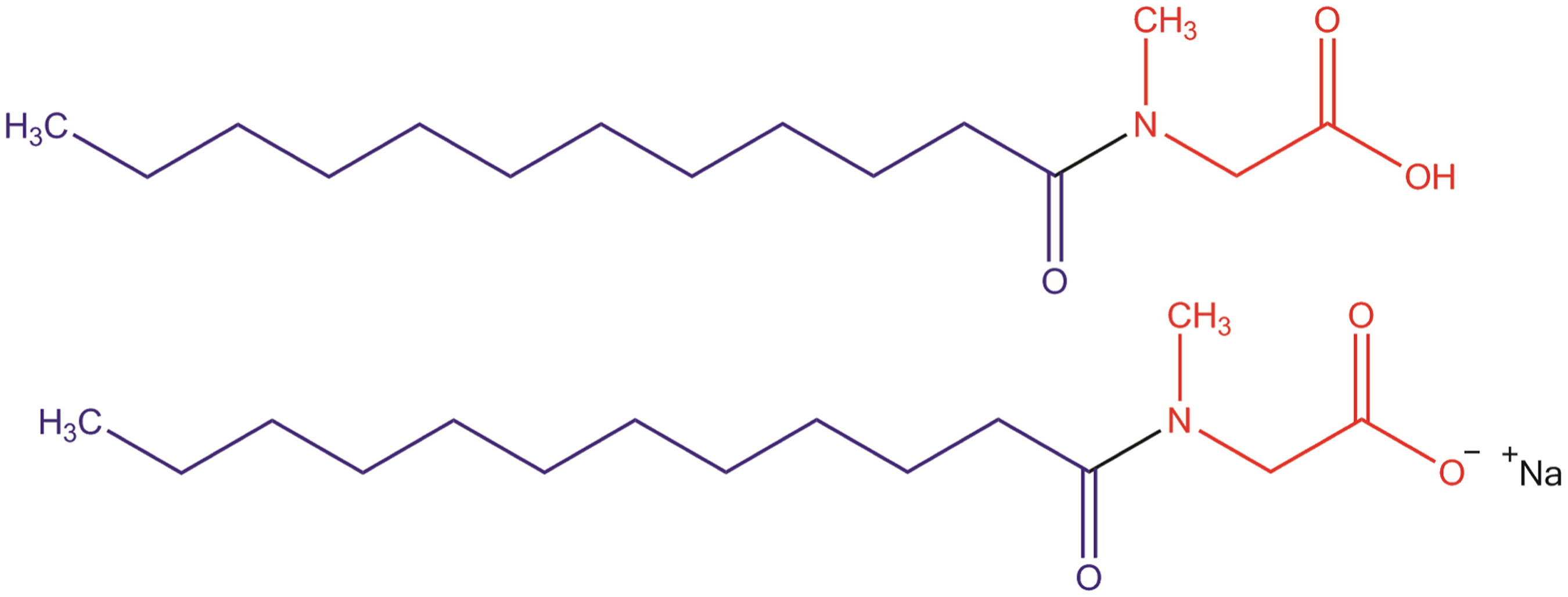
Physical and Chemical Properties
Physical and Chemical Properties.
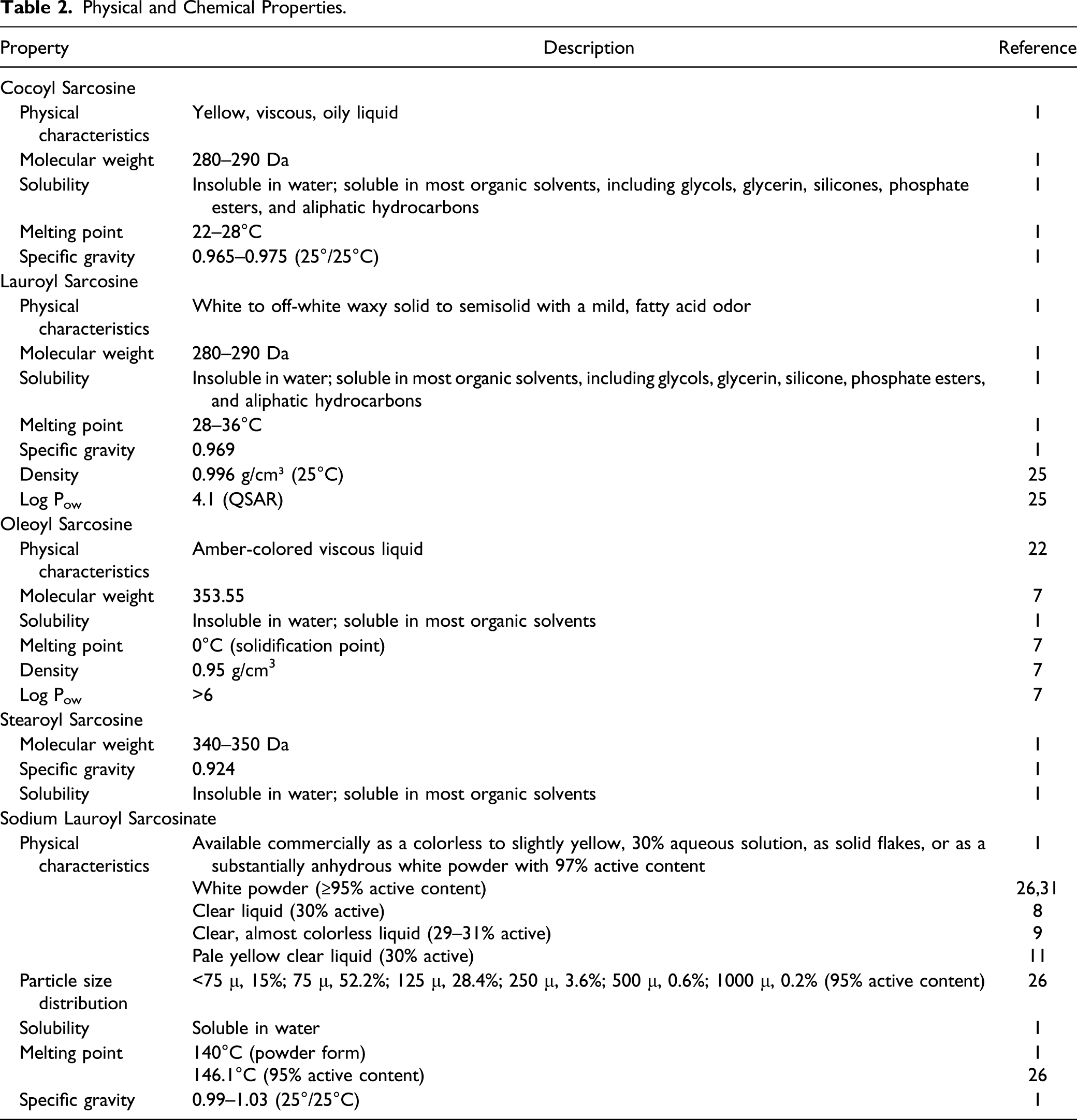
The modification of the hydrocarbon chain imparts greater solubility and crystallinity to the molecule. 1 Acyl sarcosines are somewhat stronger acids than the parent fatty acids, and they form salts in the neutral and mildly acidic pH range. The salts are similar physically and chemically to fatty acid soaps; the fatty acyl sarcosine salts are, however, more soluble in water and less affected by water hardness than are common soaps.
Method of Manufacture
The acyl sarcosines are the condensation products of sarcosine with natural fatty acids and are produced commercially by the reaction of sodium sarcosine and the parent fatty acid chlorides. 1 The acyl sarcosines can then be neutralized to form the sodium or ammonium salts.
The acyl sarcosinates are often supplied as 30% or 95% aqueous solutions. 1 According to a manufacturer, only internally prepared sodium sarcosinate is used as a starting material. The sodium sarcosinate is then reacted directly with the acyl chloride, which has been prepared from the free fatty acid by treatment with phosphorus trichloride.
Impurities/Composition
Thirty percent aqueous solutions of Lauroyl Sarcosine and Sodium Lauroyl Sarcosinate were analyzed for nitrosamines (test method unavailable). 1 The detection limits were 65 ppb for N-nitrososarcosine in Lauroyl Sarcosine and 15 ppb in Sodium Lauroyl Sarcosinate, respectively; no nitrosamines were detected. The synthesis reaction is kept in a closed system for up to several days prior to the succeeding reaction to prevent contamination with nitrite precursors. The reaction conditions are not conducive to the formation of nitrosamines as contaminants, and neither nitrates nor nitrites are used in the manufacturing process.
Precursors necessary for the “hypothetical formation” for polynuclear aromatic hydrocarbons are also absent from the synthesis reactions and none of the starting materials are prepared or provided in a hydrocarbon solvent. 1 Similarly, the presence of dioxins was considered “exceedingly improbable,” as no phenolic compounds were present in any of the synthesis reactions.
Oleoyl Sarcosine
Oleoyl Sarcosine is reported to be 97% pure. 7 It may contain 2% free fatty acids.
Sodium Lauroyl Sarcosinate
According to several suppliers, sodium Lauroyl Sarcosinate (30% active) contains 1–1.5% (max.) Sodium Laurate, 2.5% (max.) free fatty acid, 0.2–0.5% (max.) inorganic salt, and 0.35% (max.) chloride.8-11
Nitrosation
Sarcosine can react with oxidizing materials and can be nitrosated to form N-nitrososarcosine, 1 a compound that is a liver carcinogen. 12 N-nitrososarcosine has been formed by the reaction of sarcosine with sodium nitrite in an acid solution and by passing nitrous acid fumes through a sarcosine solution. 1 N-nitrososarcosine can also be produced by nitrosating N-methylsarcosine hydrochloride or by treating creatine in an acid medium with an aqueous solution of sodium nitrite. Primary routes of potential human exposure to N-nitrososarcosine are inhalation, ingestion, and dermal contact. N-nitrososarcosine has been detected in foodstuffs, particularly meat, at concentrations of 2–56 μg/kg of sample. It can be produced by various reactions in air, water, soil, food, and animal systems.
When 50 mg of Sodium Lauroyl Sarcosinate was incubated with 100 mg of sodium nitrite in 10% hydrochloric acid, investigators detected sarcosine, Lauroyl Sarcosine, and N-nitrososarcosine using thin-layer chromatography. 1 The yield of N-nitrososarcosine was 6.0%.
Use
Cosmetic
The safety of the cosmetic ingredients included in this safety assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Current and Historical Frequency and Concentration of Use According to Duration and Exposure.
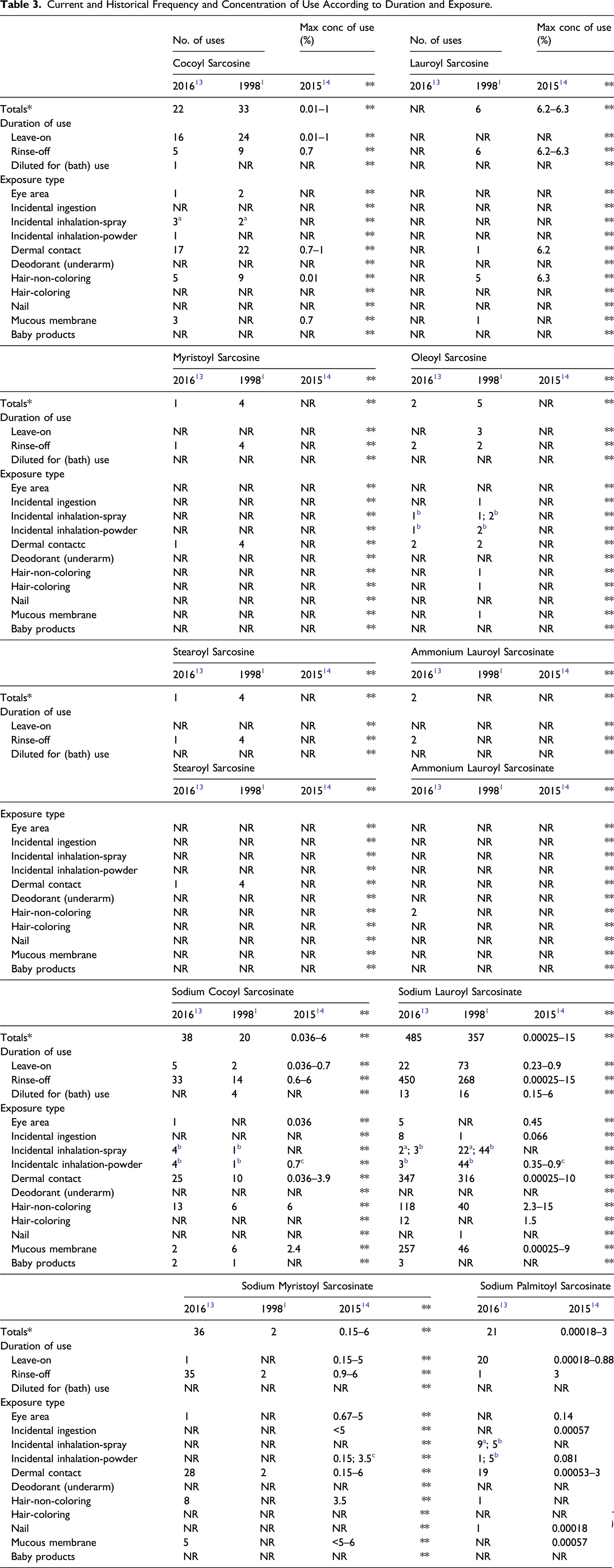
NR, not reported.
*Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
**Concentration of use data was not available at the time of the original assessment.
aIncludes products that can be sprays, but it is not known whether the reported uses are sprays.
bNot specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation.
cIncludes products that can be powders, but it is not known whether the reported uses are powders.
All but one of the in-use ingredients has been reviewed previously by the Panel, and the frequencies of use of these ingredients have not changed significantly. Concentration of use data was not provided at the time of the original safety assessment; therefore, it is not apparent whether the concentration of use has changed. (Because those values were not available, a concentration limit of 5% was established for leave-on products, based upon the highest concentration tested in human repeat-insult patch tests. 1 )
Ingredients currently not reported to be used.

#Not previously reviewed.
Several of the ingredients included in this assessment are used in products that may be ingested (eg, <5% Sodium Myristoyl Sarcosinate in lipstick), are used near the eye (eg, 5% Sodium Myristoyl Sarcosinate in eye shadow), or come in contact with mucous membranes (eg, ≤9% Sodium Lauroyl Sarcosinate in bath soaps and detergents). Additionally, some of the fatty acyl sarcosines and salts are listed in the VCRP in product types that can be sprays, but it is not known whether or not the reported uses are in sprays. In practice, 95–99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles <10 μm compared with pump sprays.15,16 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.17,18 Sodium Myristoyl Sarcosinate and Sodium Palmitoyl Sarcosinate were reported to be used in face powders at concentrations of 0.15% and 0.081%, respectively. Conservative estimates of inhalation exposures to respirable particles during the use of loose-powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.19-21
All of the fatty acyl sarcosines and salts named in the report are listed in the European Union inventory of cosmetic ingredients, and none of the listed ingredients are restricted from use in any way under the rules governing cosmetic products in the European Union.
Non-Cosmetic
Several of the fatty acyl sarcosines and salts are approved for the following indirect food additive uses: • N-Acyl sarcosines, where the acyl group is lauroyl, oleoyl, or derived from the combined fatty acids of coconut oil, are approved as antistatic and/or antifogging agents at levels not to exceed a total of 0.15% by weight of polyolefin film and ethylene-vinyl acetate copolymer film for which average thickness of the copolymer films shall not exceed 0.003 inches. [21CFR178.3130] • In polymers (specifically, cellophane), N-acyl sarcosines, where the acyl group is lauroyl or stearoyl, are approved for use only as release agents in coatings at levels not to exceed a total of 0.3% by weight of the finished packaging cellophane, and Sodium Lauroyl Sarcosinate is approved for use at 0.35% only in vinylidene chloride copolymer coatings. [21CFR177.1200] • Oleoyl Sarcosine is approved for use as a corrosion inhibitor in lubricants with incidental food contact at levels not to exceed 0.5% by weight of the lubricant. [21CFR178.3570] • Sodium Lauroyl Sarcosinate is approved in adhesives without limitations. [21CFR175.105]
Oleoyl Sarcosine is used in lubricants and greases, metal working fluids, washing and cleaning products, hydraulic fluids, textile treatment products and dyes, metal surface treatment products, and leather treatment products. 22 It is used in the formulation of mixtures and/or re-packaging, building and construction work and agriculture, forestry, and fishing. Also, Oleoyl Sarcosine is used for the manufacture of plastic products, mineral products (eg plasters and cement), fabricated metal products, machinery and vehicles, furniture and textiles, and leather or fur.
Toxicokinetics
Dermal Penetration
The amount of transdermal penetration from 1% Lauroyl Sarcosine (0.5 g) in an ointment was ∼1660 μg over 24 hours in Wistar rat, as determined using high-performance liquid chromatography; addition of 30% vitamin E or 10% squalene enhanced Lauroyl Sarcosine penetration. 1
Penetration Enhancement
Lauroyl Sarcosine (30%) increased the penetration of isosorbide dinitrate through the skin of the rat; the addition of 30% vitamin E or 10% squalene maintained or enhanced the effect of Lauroyl Sarcosine. 1 In a study of the effects of surfactants on epidermal permeability, 30% Sodium Lauroyl Sarcosine did not increase permeability.
Lauroyl Sarcosine
The effect of Lauroyl Sarcosine (98% pure) on transdermal fluorescein delivery across the epidermis of human cadaver skin was determined using Franz cells. 23 The vehicles were phosphate buffered solution (PBS; in which Lauroyl Sarcosine was generally insoluble) and aq. ethanol solution. A 0.7 cm2 skin surface was exposed to 0.3 mL of test solution. Lauroyl Sarcosine only did not significantly enhance transdermal flux. With ethanol, skin permeability increased with increasing Lauroyl Sarcosine concentrations (1–3%) in 25–50% ethanol solution, and transdermal delivery of fluorescein was increased by 47-fold using formulations containing 3% Lauroyl Sarcosine in aq. 50% ethanol solutions. The effects of higher concentrations of ethanol (ie, 75% or 100%) as the vehicle resulted in weaker enhancement effects. Lauroyl Sarcosine and ethanol synergistically increased skin permeability, and the researchers concluded that permeability was increased due to a mechanism that involved synergistic lipid-fluidization activity in the stratum corneum.
Sodium Lauroyl Sarcosinate
In the study described above, the researchers also examined the effect of Sodium Lauroyl Sarcosinate on transdermal fluorescein delivery across human cadaver skin epidermis. 23 Sodium Lauroyl Sarcosinate was completely dissolved in PBS. Only a “very small increase in transdermal flux” (0.061 ± 0.013 μg) was observed.
Absorption, Distribution, Metabolism, and Excretion
When [14C]Sodium Lauroyl Sarcosinate was administered to rats (route of administration not available) during a metabolism study, 82–89% of the 50 mg/kg dose was excreted in the urine and feces within 24 hours. 1 For the next 24 hours, 1–2% was excreted. Nearly all of the excreted material was found in the urine. In a study in which [14C]Sodium Lauroyl Sarcosinate was applied to the teeth, oral mucosa, and tongue of rats, the mean distribution of the radiolabel was 1.12% in the teeth, 2.22% in the oral mucosa, and 2.95% in the tongue immediately after dosing. At 24 hours, the mean distribution was 0.79% in the teeth, 0.92% in the oral mucosa, 0.57% in the tongue, 1.6% in the liver, 0.8% in the kidneys, 1.8% in the feces, and 42.2% in the urine. The data indicated that Sodium Lauroyl Sarcosinate was not absorbed by the tissues of the mouth but was swallowed and absorbed into the blood. Approximately 34% of the radioactivity was excreted in the urine over a period of 4 hours, and 42% was excreted within 24 hours.
Sarcosine is a natural amino acid found in muscles and other body tissues; it is found naturally as an intermediate in the metabolism of choline to glycine. 24 Oleoyl Sarcosine is a normal metabolite in man. 7
Toxicological Studies
The fatty acyl sarcosines and sarcosinate salts have low oral toxicity. 1 The oral LD50 values of Sodium Lauroyl Sarcosinate, Cocoyl Sarcosine, and Sodium Cocoyl Sarcosinate were 4.2 to 6.0 g/kg in rats. The oral LD50 of Cocoyl Sarcosine in mice was 2.1 g/kg. Ten male Yale Sherman Wistar rats per group were given a single dose (gavage) of 2.5% aqueous Sodium Lauroyl Sarcosine; no deaths occurred in groups given up to 1000 mg/kg, 1 rat each died in the 1200- and 1500-mg/kg groups, 2 died in the 1750 mg/kg group, 4 died after treatment with 2000 mg/kg, 7 died in the 2250 mg/kg group, and all 10 rats died in the group given 2500 g/kg. Weanling rats fed 0.5–2% Sodium Lauroyl Sarcosinate for up to 6 months had no signs of toxicity. During a 2-year feeding study using Wistar rats, the no-observed-effect level of Sodium Lauroyl Sarcosinate was 1000 mg/kg/day.
Acute Toxicity Studies
Dermal
Sodium Myristoyl Sarcosinate
A dose of 2000 mg/kg Sodium Myristoyl Sarcosinate in arachis oil was applied for 24 hours to the backs and flanks of 5 male and 5 female RCC Han:WIST rats using semi-occlusive patches. 25 Approximately 10% of the body was covered. Observations were made 0.5, 1, 2, and 4 hours after dosing and then once daily for 14 days. All animals survived until study termination. Very slight erythema, which was observed in 7/10 animals, was fully reversible within 5 days. The dermal LD50 of Sodium Myristoyl Sarcosinate was >2000 mg/kg in male and female rats.
Oral
Oleoyl Sarcosine
One study reported that the oral LD50 of Oleoyl Sarcosine was >5000 mg/kg, by gavage, in male and female Sprague-Dawley rats, 22 and another reported it as 9200 mg/kg in the rat. 25 (Details were not provided.)
Sodium Lauroyl Sarcosinate
Male and female Sprague-Dawley rats were given a single dose by gavage of 5000 mg/kg aq. Sodium Lauroyl Sarcosinate. 26 One female died on day 2; clinical signs were not observed in any of the remaining animals. The oral LD50 was >5000 mg/kg.
Sodium Oleoyl Sarcosinate
The oral LD50 of Sodium Oleoyl Sarcosinate in rats was 6000 mg/kg. 1
Inhalation
Oleoyl Sarcosine
Ten male and 10 female Sprague-Dawley rats were exposed nose/head only to Oleoyl Sarcosine in 10% ethanol for 4 hours, according to Organisation for Economic Development (OECD) Guideline 403 (acute inhalation study). 22 The LC50 for male and female rats was >1.01–1.85 mg/L air. No additional details were provided.
Sodium Lauroyl Sarcosinate
Groups of 5 male and 5 female Wistar:Han rats were exposed nose/head only to aq. 0.5, 1, or 5 mg/L air Sodium Lauroyl Sarcosinate (34.5% pure) for 4 hours. 26 The mass median aerodynamic diameters (MMADs) of the aerosol particles at target concentrations of 0.5, 1, and 5 mg/L were 2.6–3.1 μm, 2.0–2.7 μm, and 2.5–4.5 μm, respectively; the researchers stated that at 5 mg/L, the MMAD measurements showed an abnormal distribution, which may have been caused by the high test substance concentration in relation with high relative humidity, but the results were sufficient to conclude that the droplets size was suitable to warrant a correct exposure with sufficient distribution over the lungs. Two females and 3 males of the 5 mg/L group were found dead immediately after exposure, and the remaining animals were killed within 1 hour after exposure for humane reasons; death was attributed to acute respiratory tract irritancy. No mortality occurred in the 0.5 or 1.0 mg/L groups; shallow respiration was noted in all animals of these 2 groups. Some treatment-related gross and microscopic lesions were observed in the lungs of some animals at all test concentrations. The LC50 of Sodium Lauroyl Sarcosinate (34.5% pure) was between 1 and 5% for male and female rats.
The acute inhalation toxicity of Sodium Lauroyl Sarcosinate (96.2% pure) was evaluated in Wistar rats following a 4-hour nose-only exposure; the test was performed according to US Environmental Protection Agency (EPA) OPPTS 870.1300 guideline for acute inhalation toxicity. 25 Groups of 5 males were exposed to 0.05 or 0.5 mg/L, and 5 males and 5 females were exposed to 1 or 5 mg/L. The MMAD of the aerosol particles at target concentrations of 0.05, 0.5, 1, and 5 mg/L were 4.1–4.6 μm, 2.5–3.2 μm, 3.5–3.8 μm, and 5.8–6.2 μm, respectively. The 10 animals exposed to 5 mg/L died within 1–2 hours of dosing, and the 10 animals exposed to 1 mg/mL and 4/5 of the animals exposed to 0.5 mg/L died within 1–2 days after dosing; none of the 5 animals exposed to 0.05 mg/L died during the study. During exposure, labored respiration was only observed in the 1 mg/L group only. After exposure, no clinical signs were noted in the low or high dose groups: lethargy, flat/hunched posture, labored respiration, piloerection, and red discoloration of the mouth and nose among the males of the 0.5 mg/L group and most females (but not males) in the 1 mg/L group. At necropsy, red foci were noted on the lungs in animals of all groups except the lowest dose group. The LC50 of Sodium Lauroyl Sarcosinate in rats was 0.05–0.5 mg/L air following the 4-hour exposure.
Short-Term Toxicity Studies
Oral
Sodium Lauroyl Sarcosinate
Groups of 15 male and 15 female Sprague-Dawley albino rats were dosed orally by gavage daily with 0, 30, 100, and 250 mg/kg Sodium Lauroyl Sarcosinate in distilled water for 91 or 92 days. 25 Body weight gains were decreased in males of the 100 and 250 mg/kg groups; the decrease was statistically significant compared to controls for 5 of 13 weeks in the mid-dose group and 8 of 13 weeks in the high doses group. Absolute stomach weights (in males), stomach-to-body weight ratios, and stomach-to-brain-weight ratios (in males and females) were statistically significantly increased in the 100 and 250 mg/kg dose groups. There was an increase in stomach wall thickness and yellow discoloration of non-glandular gastric mucosa, and histopathology revealed an increase in incidence and severity of squamous cell hyperplasia, hyperkeratosis/parakeratosis, inflammation, and edema of the non-glandular mucosa in both male and females of these groups. Weights of several other organs that were statistically significantly different from control values were not considered toxicologically significant. There were no toxicologically significant changes in hematology or clinical chemistry parameters. No test material-related mortality was reported. The no-observed effect level (NOEL), lowest observable adverse effect level (LOAEL; local effects), and no-observable adverse effect level (NOAEL; systemic effects) for male and female animals were 30, 100, and 250 mg/kg/day, respectively.
Inhalation
Oleoyl Sarcosine
In a 28-day inhalation study performed according to OECD Guideline 412 (Repeated Dose Inhalation Toxicity: 28/14-day), groups of 3 male and 3 female Fischer 344 rats were exposed nose-/head-only to 0, 0.006, 0.02, or 0.06 mg/L Oleoyl Sarcosine in <10% ethanol. 22 The daily exposure time was not specified; however, according to OECD Guideline 412, daily exposure is 6 hours in this type of study. The MMAD of the aerosol particles were 1.11, 1.15, and 1.22 μm for the low, mid, and high concentrations, respectively. All test concentrations caused effects at several sites of the respiratory system with indications for a local irritation, squamous metaplasia and epithelium proliferation and submucous acute inflammation at the base of the epiglottis; these changes may be explained by the amounts of inert material deposited within the respiratory system. In the lungs and bronchi, the most prominent finding was a focal early stage of fibrosis. The researchers stated that due to the high amount of test substance deposits in the lungs, especially in the 0.02 and 0.06 mg/L groups, these changes may be explained as an overloading of the tissue and do not necessarily imply an intrinsic toxicity of the test material; an intrinsic toxicity is unlikely because the test material is insoluble and the shape of the particles is not fibrous. There was an effect on testes, thymus, brain, lung, and kidneys weights, but details were not provided. The NOEL was <0.006 mg/L air in males and females; the basis for the effect level was local irritation. The no-observed adverse effect concentration (NOAEC) was >0.06 mg/L air in males, and the basis for that effect level was an effect on organ weight.
Developmental and Reproductive Toxicity
The feeding of up to 1000 mg/kg/day Sodium Lauroyl Sarcosinate did not adversely affect fertility of albino Sherman Wistar rats during a 2-year oral toxicity study. 1
Sodium Lauroyl Sarcosinate
A prenatal developmental toxicity study (OECD Guideline 414) was conducted for Sodium Lauroyl Sarcosinate (95% pure) in Sprague-Dawley rats. 26 Groups of 24 gravid female rats were dosed once daily by gavage with 0, 30, 100, and 250 mg/kg/day of the test article in distilled water on days 5–10 of gestation, and the animals were killed on day 20 of gestation. Sodium Lauroyl Sarcosinate was not embryotoxic or teratogenic. Maternal body weight gains (adjusted) in the mid- and high-dose group were decreased during gestation as compared to the controls. Feed consumption was decreased in the high dose group; the decrease was statistically significant between days 8–11 and days 14–17 of gestation. Two high-dose dams died during the study, one on day 10 and one on day 18 of gestation. The dam that died on day 18 of gestation had sloughing on the non-glandular region of the stomach, 7 dead fetuses had sloughing in the right uterine horn, and 5 dead fetuses had sloughing in the left uterine horn, and the high-dose females killed at study termination all had sloughing on the non-glandular region of the stomach; this effect was not observed in the low- or mid-dose groups. The NOAEL (maternal toxicity), LOAEL (maternal toxicity), and NOEL (developmental toxicity) were 30, 100, and ≥250 mg/kg/day Sodium Lauroyl Sarcosinate, respectively.
Genotoxicity
In Vitro
Sodium Lauroyl Sarcosinate was not considered mutagenic in five strains of Salmonella typhimurium during plate incorporation assays and spot tests. 1 In addition, Sodium Lauroyl Sarcosinate did not induce double-strand DNA breaks in the comet assay using human white blood cells and V79 Chinese Hamster cells, but the compound was cytotoxic.
Oleoyl Sarcosine
Oleoyl Sarcosine in dimethyl sulfoxide at concentrations of ≥5000 μg/plate, with or without metabolic activation, was not mutagenic in an Ames test with S. typhimurium TA1535, TA1537, TA100, and TA9825 or in an Ames test with S. typhimurium TA97a, TA98, TA100, TA102, or TA1535. 22 Positive and vehicle controls gave expected results.
Sodium Lauroyl Sarcosinate
The genotoxic potential of Sodium Lauroyl Sarcosinate (96.2% pure) was evaluated in an in vitro mammalian chromosomal aberration assay in lymphocytes. 26 Cells were treated with 22.5–360 μg/mL for 4 hours with or without metabolic activation and with 22.5–270 μg/mL for 24 hours without metabolic activation. Minimal essential media served as the vehicle. Sodium Lauroyl Sarcosinate was not genotoxic. Solvent and positive controls gave expected results.
Carcinogenicity
Carcinogenicity data of the fatty acyl sarcosines and their salts were not available; however, the ingredients were not considered likely carcinogens as they and their metabolites “do not belong to any class of compounds that contains a significant number of mutagens or oncogens.” 1
Dermal Irritation and Sensitization
Sodium Lauroyl Sarcosinate was non-irritating to rabbits when administered as a 20–30% solution, at a concentration of 2% in formulation, or as the pure powder. 1 A formulation containing 30% Sodium Myristoyl Sarcosinate was not a primary skin irritant in rabbits, and 0.01% aq. Sodium Lauroyl Sarcosinate was non-sensitizing to the skin of guinea pigs.
During a clinical study using 27 subjects, cocobetaine (it was unclear whether the cocobetaine tested was Cocoyl Sarcosine or the related, cocoyl N-dimethyl glycine derivative) markedly influenced skin water vapor loss and caused erythema, scaling, and fissuring of the skin of the volar forearm. 1 In another study, Cocoyl Sarcosine and Sodium Lauroyl Sarcosinate retarded moisture loss from the skin via the formation of a hydrophobic protective layer on the epidermal surface; in an epicutaneous patch test using highly dermatitic subjects, “practically no reaction” was observed. In other clinical studies, Sodium Lauroyl Sarcosinate (2–5%) was non-irritating and non-sensitizing.
Dermal Irritation
In Vitro
Sodium Lauroyl Sarcosinate
The irritation potential of Sodium Lauroyl Sarcosinate was evaluated in an In Vitro Skin Corrosion Human Skin Model Test (OECD Guideline 431) using reconstructed human epidermis. 26 Twenty mg of the test material in 0.9% sodium chloride solution was applied to the tissue for 3, 60, or 240 minutes, and tissue viability was measured using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) uptake. Sodium Lauroyl Sarcosinate was non-corrosive to reconstructed human epidermis. Appropriate negative and positive controls gave valid results.
Animal
Oleoyl Sarcosine
Oleoyl Sarcosine, 0.5 mL, was applied neat to the shaved intact and abraded skin of 3 male and 3 female New Zealand White rabbits under an occlusive patch for 24 hours. 22 The test sites were scored upon patch removal and over a 7-day period. The mean erythema score at 24 and 72 hours was 2.5–2.8 and 2.7–3, respectively. Additionally, at 72 hours, the treated areas developed slight necrosis and the skin hardened. Oleoyl Sarcosine was classified as irritating.
Sodium Lauroyl Sarcosinate
The dermal irritation potential of Sodium Lauroyl Sarcosinate was evaluated in 6 female New Zealand White rabbits. 25 The test material was diluted 1:3 in water to give 10% active material, and occlusive patches containing 0.5 mL were applied for 24 hours to shaved intact and abraded skin of each animal. Well-defined erythema was observed at both intact and abraded treatment sites of all 6 animals following the 24 hours dosing period, and slight edema was observed at 4 intact and 2 abraded treatment sites. After 72 hours, well-defined erythema remained at both the intact and abraded sites of 4 animals, and very slight erythema was observed at both sites in one animal. Slight edema was observed at the abraded site of one animal, and very slight edema was observed at 3 abraded sites and 3 intact sites. Test sites were scored at 24 and 72 hours, and the mean scores for erythema and edema were 1.83/4 and 1.06/4, respectively; erythema and edema were not fully reversible within 72 hours.
Sensitization
Animal
Oleoyl Sarcosine
In a guinea pig maximization test (GPMT) using groups of 10 male and 10 female Pirbright White guinea pigs, the intradermal induction consisted of 3 pairs of injections of a 1:1 mixture of Freund’s Complete Adjuvant (FCA) and saline; 5% Oleoyl Sarcosine in saline; and a mixture of Oleoyl Sarcosine with FCA/saline.22,25 The epicutaneous induction concentration was 30% Oleoyl Sarcosine in petrolatum. The challenge was performed on day 20 and consisted of a 24-hour patch at a concentration of 3% in petrolatum. The test site was evaluated after 48 hours; 3 animals had very slight erythema and 2 had well-defined erythema. The researchers classified the test substance as not sensitizing.
Photosensitization
Animal
Oleoyl Sarcosine
A photosensitization study of Oleoyl Sarcosine was conducted using groups of 10 male and 10 female Pirbright White guinea pigs. 27 Induction consisted of open applications of 0.1 mL of a 0.1% suspension of the test substance in 80% DAE (40% dimethylacetamide, 30% acetone, and 30% ethanol) and 20% physiological saline that were applied topically to the shaved skin on the necks of the animals 4 times/wk for 3 weeks. One hour after each application, the animals were irradiated for 10 minutes; during week 1, the animals were exposed to ultraviolet A (UVA) and visible light using a Schott WG 335, 3 mm filter, and during weeks 2 and 3, they were exposed to UVA, UVB, and visible light using a Schott WG 280, 3 mm filter. The test sites were scored 24 hours after each induction application during induction week 1. The sites were not scored during induction weeks 2 and 3; during this time, a total of four adjuvant injections of 0.1 mL FCA/saline were made to the 4 corners of the application site on Monday and Wednesday of both weeks.
The first challenge was performed 16 days after the last induction irradiation; open applications of 0.1 mL of the test substance was applied to the dorsal skin of the animals for 3 days, and the sites were irradiated 1 hour after the application with a suberythematogenic dose of UVA, UVB, and visible light. The second challenge was performed after a 14-day non-treatment period; the test material was applied in the same manner as the first challenge, but this time the application was followed with 10 minutes irradiation with a suberythematogenic dose of UVA and visible light. The test sites were evaluated 24 hours after each challenge application. Any animal in which the irritation score after challenge was 1+ point above the score from week 1 of induction was considered to be photosensitized. The test animals were compared to the control group that was treated with the vehicle alone.
Three control animals and 2 test animals died during the study; the deaths were not related to dosing. Three of 17 control animals had slight erythema during the first challenge. However, 17 and 15 of the 18 test animals had positive results after the first and second challenges, respectively. Oleoyl Sarcosine was considered to possess a photocontact-allergenic potential in guinea pigs.
Ocular Irritation
Sodium Cocoyl Sarcosinate (10%) at neutral or slightly acid pH caused slight, temporary ocular irritation but no corneal damage in rabbits according to the procedures of the Draize-Woodard test. 1 In another ocular irritation study using rabbits, a 5% aqueous solution of Sodium Lauroyl Sarcosinate caused minimal conjunctival irritation and no apparent damage to the cornea.
In Vitro
Sodium Myristoyl Sarcosinate
The ocular irritation potential of 20% Sodium Myristoyl Sarcosinate (92.1% pure) in physiological saline was evaluated in the Bovine Corneal Opacity and Permeability (BCOP) test. 25 The test article was considered to be an ocular corrosive or severe irritant in the BCOP test.
Animal
Oleoyl Sarcosine
Oleoyl Sarcosine was instilled into one eye of rabbits according to EPA guidelines; the eyes of half the rabbits were rinsed after 30 seconds. 7 Details on the dose and number of animals were not provided. Draize scores of 47 and 40 were reported for unrinsed and rinse eyes, respectively. Oleoyl Sarcosine was classified as moderately irritating to rabbit eyes.
In another study, detachment and clouding of the cornea was seen in rabbits treated with either Oleoyl Sarcosine or its sodium salt. 7 After treatment with the sodium salt, the effects on the cornea had worsened after 1 week; this change was not reversible after 15 days.
Sodium Cocoyl Sarcosinate and Sodium Lauroyl Sarcosinate
Sodium Cocoyl Sarcosinate (10%) at neutral or slightly acid pH caused slight, temporary ocular irritation but no corneal damage in rabbits according to the procedures of the Draize-Woodard test. 1 In another ocular irritation study using rabbits, a 5% aqueous solution of Sodium Lauroyl Sarcosinate caused minimal conjunctival irritation and no apparent damage to the cornea.
Sodium Myristoyl Sarcosinate
One-tenth milliliter of a mixture of Sodium Myristoyl Sarcosinate and sodium myristate was instilled neat into the conjunctival sac of the right eye of 6 rabbits, and the contralateral eye served as the control. 28 All animals had a positive response to the test article, and the maximum average eye irritation score was 55.3 at 24 hours after instillation. The mixture of Sodium Myristoyl Sarcosinate and sodium myristate was extremely irritating to rabbit eyes and considered a primary eye irritant.
Mucosal Irritation
Sodium Lauroyl Sarcosinate (20% aq. solution, 2% in formulation, powder) was non-irritating to the gums and oral mucosa of rabbits. 1
Clinical Reports
Case Reports
Sodium Lauroyl Sarcosinate
A female patient developed an acute severe eczematous reaction on her hands, face, and neck, and the reaction was related to use of a hand soap. 29 After open and unoccluded patch test resulted in a +3 bullous reaction to the product, patch testing with some of the individual constituents was performed. A +3 bullous reaction to a 30% aq. solution of Sodium Lauroyl Sarcosinate in sterile water was reported. In 2 subjects patch tested with the soap and Sodium Lauroyl Sarcosinate, negative results were obtained.
In another report, a female patient with recurrent hand dermatitis had a positive reaction to semi-open application of a liquid cleanser that contained Sodium Lauroyl Sarcosinate. 30 Positive reactions were observed in follow-up patch testing with 0.1, 0.5, and 1% aq. Sodium Lauroyl Sarcosinate; at 98 hours, the scores were “–,” “+/−,” and “+” at these concentrations, respectively.
Summary
In 2001, the Panel published a safety assessment with the conclusion that 5 fatty acyl sarcosines and 5 sarcosinate salts are safe as used in rinse-off products, safe for use in leave-on products at concentrations of ≤5%, and the data are insufficient to determine the safety for use in products where the fatty acyl sarcosines and salts are likely to be inhaled. These ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed. This assessment is a re-review of those original ingredients, as well as 4 additional salts.
Sarcosine (which is also known as N-methylglycine or N-methylaminoacetic acid) is a natural amino acid found in muscles and other body tissues, and it is found naturally as an intermediate in the metabolism of choline to glycine. Oleoyl Sarcosine is also a normal metabolite in man.
Ten of the 14 ingredients included in this safety assessment are currently in use. Sodium Lauroyl Sarcosinate has the highest frequency of use, with 485 reported uses; the majority of these uses are in rinse-off formulations, primarily bath soaps and detergents (230 uses) and shampoos (113 uses). Sodium Lauroyl Sarcosinate also has the highest concentration of use, with maximum use concentrations up to 15% in rinse-off products. The highest reported leave-on concentration is 5% Sodium Myristoyl Sarcosinate in eye shadow formulations.
Lauroyl Sarcosine and ethanol synergistically increased skin permeability, as demonstrated by up to a 47-fold increase in transdermal delivery of fluorescein across human cadaver epidermis using 3% lauroyl sarcosine in aq. 50% ethanol. Lauroyl Sarcosine and Sodium Lauroyl Sarcosinate alone (in PBS) did not significantly affect penetration.
Sodium Myristoyl Sarcosinate had a dermal LD50 of >2000 mg/kg in male and female rats. In acute oral studies in rats, oleoyl sarcosine had an LD50 of 9200 mg/kg, Sodium Lauroyl Sarcosinate had an LD50 of >5000 mg/kg, and Sodium Oleoyl Sarcosinate had an LD50 of 6000 mg/kg. In a 3-month gavage study of Sodium Lauroyl Sarcosinate in rats, the NOEL, LOAEL (local effects), and NOAEL (systemic effects) were 30, 100, and 250 mg/kg/day, respectively.
Acute inhalation studies were performed in rats; with a 4-hour exposure, Oleoyl Sarcosine had a LC50 of >1.01–1.85 mg/L air, Sodium Lauroyl Sarcosinate (34.5% pure) had an LC50 between 1 and 5%, and Sodium Lauroyl Sarcosinate (96.2% pure) had an LC50 of 0.05–0.5 mg/L air. A 28-day inhalation study was performed in rats with Oleoyl Sarcosinate; the NOEL was <0.006 mg/L air, and the NOAEC was 0.06 mg/L air.
No embryotoxicity or teratogenicity was observed in a prenatal developmental toxicity study in which gravid rats were dosed by gavage with up to 250 mg/kg/day Sodium Lauroyl Sarcosinate on days 5–10 of gestation. The NOAEL and LOAEL for maternal toxicity were 30 and 100 mg/kg/day.
Oleoyl Sarcosine was not mutagenic in an Ames test (≥5000 μg/plate, with or without metabolic activation), and Sodium Lauroyl Sarcosinate (22.5–360 μg/mL for 4 hours with or without metabolic activation; 22.5–270 μg/mL for 24 hours without metabolic activation) was not genotoxic in an in vitro mammalian chromosomal aberration assay in lymphocytes.
Sodium Lauroyl Sarcosinate was non-corrosive to reconstructed human epidermis in an In Vitro Skin Corrosion Human Skin Model Test. Undiluted Oleoyl Sarcosine was irritating to rabbit skin, and Oleoyl Sarcosine was classified as not sensitizing in a GPMT in which 3 and 2/20 guinea pigs had very slight and well-defined erythema, respectively, 48 hours after challenge with 3% Oleoyl Sarcosine in petrolatum. Oleoyl Sarcosine was also considered to possess photocontact-allergenic potential in guinea pigs. A single 24-hour application of Sodium Lauroyl Sarcosinate (10% active material) produced mean erythema and edema scores of 1.83/4 and 1.06/4 in rabbits, and the effects were not fully reversible within 72 hours.
Sodium Myristoyl Sarcosinate, 20%, was considered to be an ocular corrosive or severe irritant in vitro in the BCOP test, and a mixture of Sodium Myristoyl Sarcosinate and sodium myristate was extremely irritating to rabbit eyes and considered a primary eye irritant. Oleoyl sarcosine was classified as moderately irritating to rabbit eyes.
Discussion
A safety assessment of 5 fatty acyl sarcosines and 5 fatty acyl sarcosine salts was published in 2001 with the conclusions that these ingredients are safe as used in rinse-off products, safe for use in leave-on products at concentrations of ≤5%, and the data are insufficient to determine the safety for use in products where the fatty acyl sarcosines and their salts are likely to be inhaled. Also, the conclusion stated that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed. Concentration of use data was not provided at the time of the original safety assessment; because those values were not available, the concentration limit of 5% was established for leave-on products based upon the highest concentration tested in human repeat-insult patch tests. Concentration of use data is now available, and because sensitization is not observed in studies at the highest concentration currently reported to be used, the Panel re-opened the safety assessment to remove the 5% concentration limit for leave-on products.
The Panel determined 4 previously unreviewed fatty acyl sarcosine salts used as cosmetic ingredients are structurally similar to the ingredients reviewed in the original assessment, and that the data from the original safety assessment, together with the new data presented in this report, support their safety. Therefore, these 4 ingredients are included in this review.
Some of the ingredients included in this report, particularly Lauroyl Sarcosine, can potentially enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data, or when dermal absorption was a concern.
Sarcosine, a starting material in the manufacture of the acyl sarcosines and sarcosinates, can react with oxidizing materials and can be nitrosated to form N-nitrososarcosine, a known animal liver carcinogen. As a result, the Panel concluded that fatty acyl sarcosines and salts should not be used in cosmetic products in which N-nitroso compounds can be formed.
The Panel was concerned that the potential exists for dermal irritation with the use of products formulated using fatty acyl sarcosines and sarcosinate salts. The Panel specified that products containing these ingredients must be formulated to be non-irritating.
A photosensitization study indicated that Oleoyl Sarcosine may possess photocontact-allergenic potential in guinea pigs. The Panel noted that the chemical structure of Oleoyl Sarcosine does not have a chromophore, so there are no structural alerts for photosensitization. Additionally, the study did not indicate that an unirradiated control was used. The Panel stated that the allergenic response observed in the study was not to Oleoyl Sarcosine and was most probably due to a contaminant.
The Panel acknowledged that some of the fatty acyl sarcosines and sarcosinate salts may contain cocoyl fatty acyl substituents and expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit impurities.
Additionally, the Panel discussed the issue of incidental inhalation exposure of fatty acyl sarcosines and their salts. Some of these ingredients are listed in the VCRP in product types that can be sprays, but it is not known whether or not the reported uses are in sprays. However, Sodium Myristoyl Sarcosinate and Sodium Palmitoyl Sarcosinate are reported to be used in face powders at concentrations of 0.15% and 0.081%, respectively, and these products may become airborne. Single dose, 4-hour inhalation studies of 10% Oleoyl Sarcosine and Sodium Lauroyl Sarcosinate (96.2% pure) reported LC50 value of >1.01–1.85 mg/L air, and 0.05–0.5 mg/L, respectively; a 28-day inhalation study of oleoyl sarcosine in rats found that an intrinsic toxicity is unlikely because the test material is insoluble and the shape of the particles is not fibrous. The Panel also noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion

*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
