Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of inorganic hydroxides, which function in cosmetics primarily as pH adjusters. Representatives from the cosmetic industry have indicated these ingredients are used in depilating and hair waving/straightening formulations to raise pH values. The Panel considered relevant data related to these ingredients. The Panel concluded that these inorganic hydroxides are safe in hair straighteners and depilatories under conditions of recommended use; users should minimize skin contact. These ingredients are safe for all other present practices of use and concentration described in this safety assessment when formulated to be nonirritating.
Introduction
This report addresses the safety of the inorganic hydroxides, Calcium Hydroxide (also known as calcium hydrate or slaked lime), Magnesium Hydroxide, Potassium Hydroxide (potassium hydrate or potash), and Sodium Hydroxide (sodium hydrate, lye, or caustic soda). These ingredients are all alkaline salts and are reported in the International Cosmetic Ingredient Dictionary and Handbook (Dictionary) to function as pH adjusters in cosmetics; however, representatives from the cosmetics industry and formulary references have stated that the inorganic hydroxides are also used at very high pH values in hair straighteners and depilatories.1-5 Additionally, Sodium Hydroxide has been reacted with fats to form soap for millennia.
The inorganic hydroxides in this report, except for Magnesium Hydroxide, are well-known caustic agents that can cause severe burns and corrosion with acute exposures. Sodium Hydroxide is commonly used as a positive control in efficacy studies of skin protective creams and in other studies of irritant contact dermatitis. 6
Some chemical and toxicological data on the inorganic hydroxides included in this safety assessment were obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) by companies as part of the REACH chemical registration process. These data summaries are available on the ECHA website.7-10
Chemistry
Definition
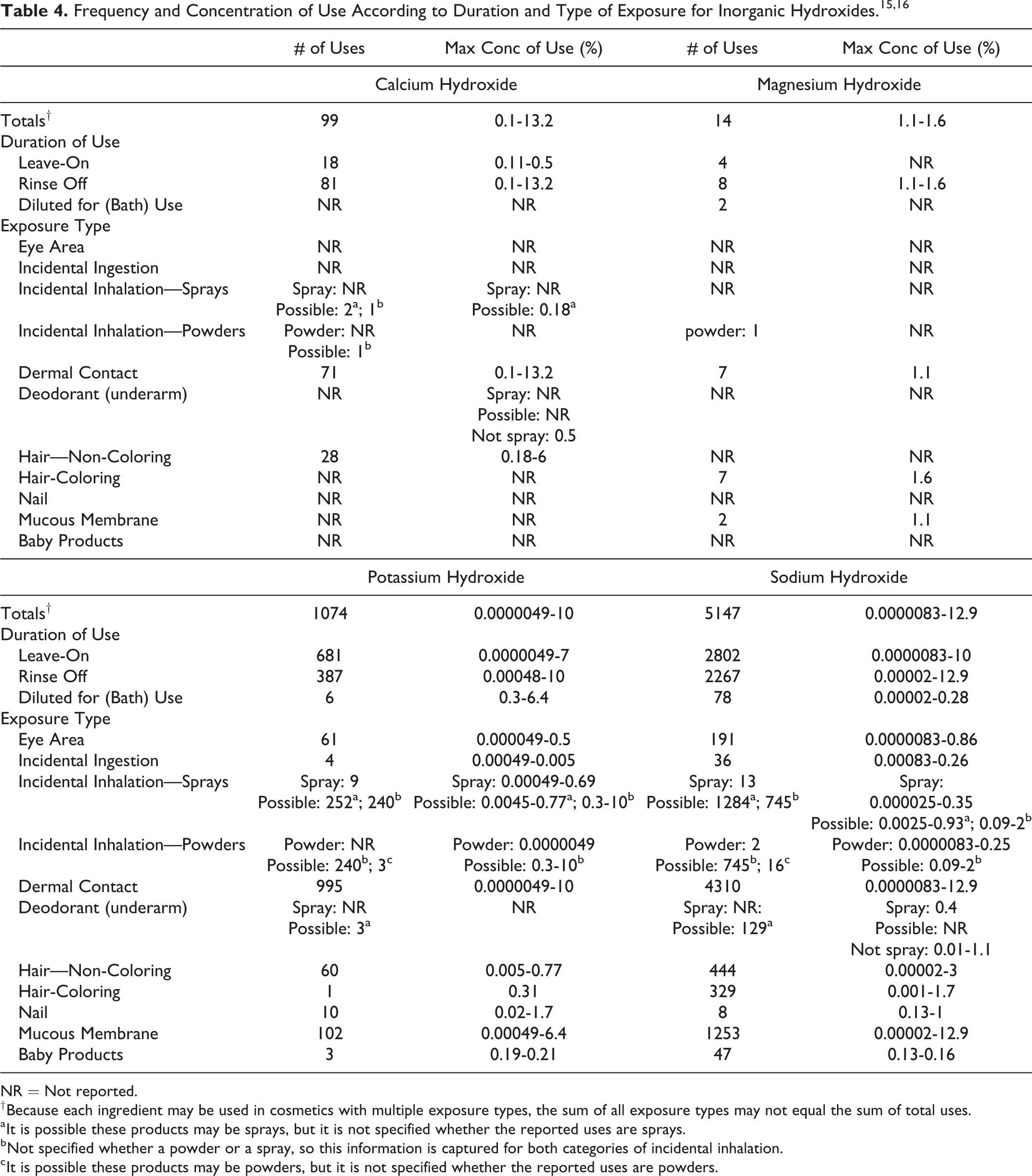
Inorganic hydroxides are alkaline salts formed by treating oxides with water or via decomposing salts by adding other soluble hydroxides to a solution thereof (eg, adding Sodium Hydroxide to magnesium sulfate will produce Magnesium Hydroxide). The formation of an inorganic hydroxide, such as specifically lime or Calcium Hydroxide, by reaction of an oxide with water is known as slaking. 11 The resulting highly water soluble ingredients only vary structurally by the metal cation. These variations result in different degrees of basicity across these 4 ingredients, ranging in pKb values from 0.2 to 4.0. Used primarily as pH adjusters (to increase the pH of an otherwise acidic formulation), the caustic nature of these ingredients is unlikely to be observable in typical, final cosmetic formulations, except when used in depilatory or hair straightener product types.
The definitions, structures, and functions of the inorganic hydroxides included in this report are provided Figure 1 and in Table 1.
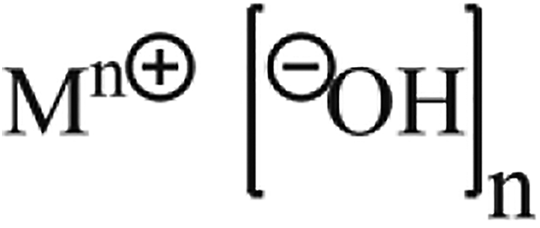
Inorganic hydroxides (wherein “M” is a group I or II metal).
Definitions, Structures, and Reported Functions of the Ingredients in This Safety Assessment. 1

Chemical Properties
The inorganic hydroxides are all highly water-soluble, white solids with specific gravities around 2. Chemical properties of the inorganic hydroxides in this report are provided in Table 2.
Chemical Properties of Inorganic Hydroxides.

Method of Manufacturing
Calcium Hydroxide
Calcium Hydroxide may be formed by the hydration of lime or by treating an aqueous solution of a calcium salt with alkali. 12
Magnesium Hydroxide
Magnesium Hydroxide may be formed by reacting magnesium chloride or sulfate with Sodium Hydroxide. 12 Most commercial-grade Magnesium Hydroxide is obtained from seawater or brine using lime or dolomitic lime. 11
Potassium Hydroxide
Potassium Hydroxide may be produced by treating oxides with water, a process known as brine electrolysis.11,12
Sodium Hydroxide
Sodium Hydroxide is formed by brine electrolysis. 11
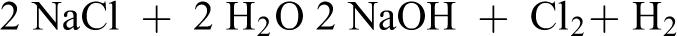
Process 1. Brine Electrolysis
Sodium Hydroxide may also be formed by reacting lime with soda ash. 11
Process 2. Slaking
Impurities
The US Pharmacopeia and Food Chemicals Codex provides specifications for acceptable levels of impurities for the inorganic hydroxides named in this report.13,14 These specifications are provided in Table 3.
* NMT = no more than.
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated on the basis of the reported use in cosmetics using the data received from the US Food and Drug Administration (FDA) and the cosmetics industry. The data received from the FDA are those it collects from manufacturers on the use of individual ingredients in cosmetics by cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP), and those from the cosmetic industry are submitted in response to a survey of the maximum reported use concentrations by category conducted by the Personal Care Products Council (Council).
According to the 2015 VCRP data, Sodium Hydroxide has the most reported uses in cosmetic formulations of the ingredients listed in this safety assessment, with a total of 5147; about half of the uses are in leave-on skin care formulations (Table 4). 15 Potassium Hydroxide has the second greatest number of overall uses reported, with a total of 1074; the majority of the uses also are in leave-on skin care formulations. The results of the concentration of use survey conducted in 2014 by the Council indicate Calcium Hydroxide has the highest reported maximum concentration of use; it is used at up to 13.2% in rinse-off shaving preparations; however, it is only used at up to 0.5% in leave-on formulations (deodorants). 16 Sodium Hydroxide is used at up to 10% in an “other” skin care preparation, which may or may not be a leave-on formulation. The next highest concentration of use reported for Sodium Hydroxide in a leave-on formulation is 6.9% in a face or neck formulation. Potassium Hydroxide is used at up to 7% in a leave-on body and hand formulation. Examples of depilatory and hair straightening formulations containing inorganic hydroxides are found in Tables 5 and 6.
NR = Not reported.
† Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
a It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
c It is possible these products may be powders, but it is not specified whether the reported uses are powders.
Example Depilatory Formulations. 5
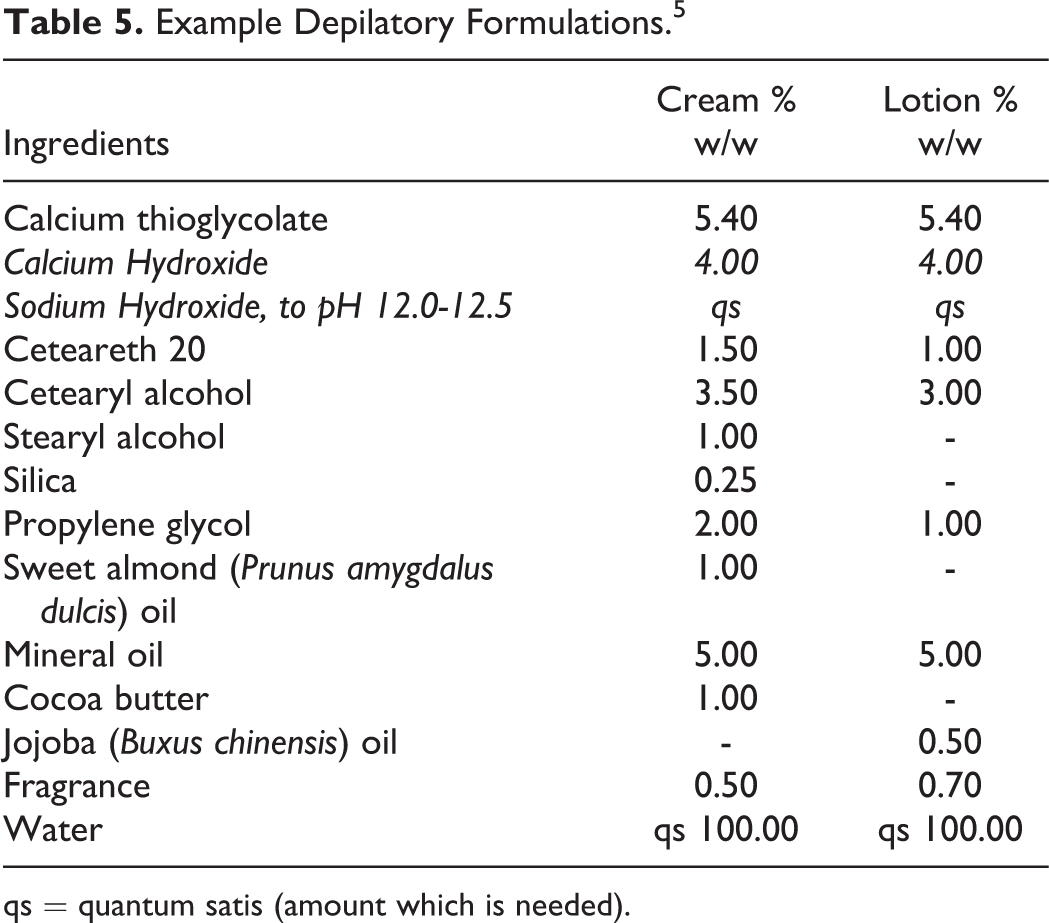
qs = quantum satis (amount which is needed).
Example Hair Relaxer/Straightener Formulations. 3
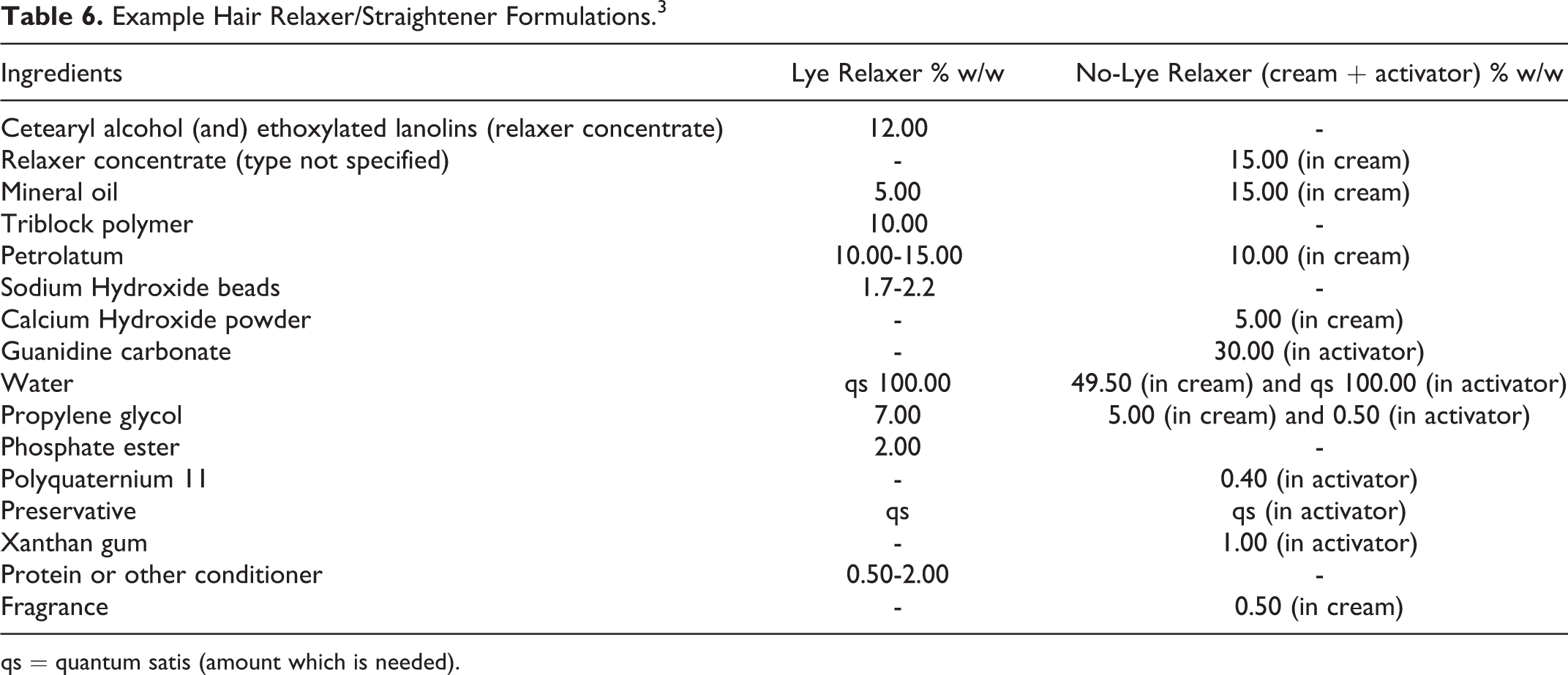
qs = quantum satis (amount which is needed).
Some of these ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes. For example, Sodium Hydroxide is used in lipstick (at least one use at up to 0.26%) and in bath soaps and detergents (860 uses at up to 12.9%). Additionally, some of these ingredients were reported to be used in hair sprays and body and hand sprays and could possibly be inhaled. For example, Potassium Hydroxide was reported to be used in hair sprays at a maximum concentration of 0.69%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.17-20 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.18,19
The European Union Scientific Committee on Consumer Safety opined that Potassium Hydroxide is safe for use as a callosity softener/remover with a concentration of up to 1.5%. 21 A proposed change to the European Commission’s regulation under Annex III List of Substances Which Cosmetic Products Must Not Contain Except Subject to the Restrictions Laid Down has been sent to the World Trade Organization for consideration. Currently, Sodium Hydroxide, Potassium Hydroxide, and Calcium Hydroxide are listed on Annex III with the restrictions listed here. 22 The uses of Sodium Hydroxide and Potassium Hydroxide may not exceed 5% in nail cuticle solvents; 2% for general use and 4.5% in professional use of hair straighteners; must have a pH below 12.7 when used as a pH adjuster in depilatories; and must have pH below 11 in other uses. The use of Calcium Hydroxide may not exceed 7% in hair straighteners containing Calcium Hydroxide and a guanidine salt, must have a pH below 12.7 when used as a pH adjuster in depilatories, and must have a pH below 11 in all other uses.
Magnesium Hydroxide is not restricted from use in any way under the rules governing cosmetic products in the European Union. 22
Non-Cosmetic
According to US FDA, the inorganic hydroxides in this report are generally recognized as safe (GRAS) as direct food substances based upon following current good manufacturing practice conditions of use (21CFR§184). Additionally, these are GRAS as feed additives for animals (21CFR§582). The FDA has also separately reviewed Calcium Hydroxide and Magnesium Hydroxide for use as active ingredients in over-the-counter (OTC) drugs. Calcium Hydroxide is listed for antidiarrheal products and Magnesium Hydroxide is listed for digestive aid products (21CFR §310).
Calcium Hydroxide is used in mortar, plaster, cement, and other building and paving materials. 12 It is also used in lubricants, drilling fluids, pesticides, fireproofing coatings, water paints, as egg preservative, in the manufacture of paper pulp, in rubber vulcanization in water treatment, in dehairing hides, and as an absorbent for carbon dioxide. Therapeutically, it is used as an astringent.
Magnesium Hydroxide may be used therapeutically as an antacid, cathartic, or laxative. 12 It is an approved OTC active ingredient (21 CFR§ 331.11).
Non-cosmetic uses of Potassium Hydroxide include as a mordant for wood, for mercerizing cotton, for absorbing carbon dioxide, for removing paint and varnish, for electroplating, for photoengraving and lithography, in printing inks, for debudding calves’ horns, and for dissolving scales and hair in skin scrapings. 12
Sodium Hydroxide is a strong base and is extremely corrosive. Sodium Hydroxide solutions are used to neutralize acids and to make sodium salts (eg, in petroleum refining to remove sulfuric and organic acids), to treat cellulose during viscose rayon and cellophane production, to reclaim rubber, in plastics manufacturing, and in dehorning calves.11,12
Toxicokinetics
No relevant published toxicokinetics studies on inorganic hydroxides were identified in a literature search for these ingredients and no unpublished data were submitted. Data on the kinetics of the metal ions of these ingredients are abundant in the published literature, but these data are not useful in assessing the safety of these ingredients as they are used in cosmetics.
Toxicological Studies
Acute Toxicity
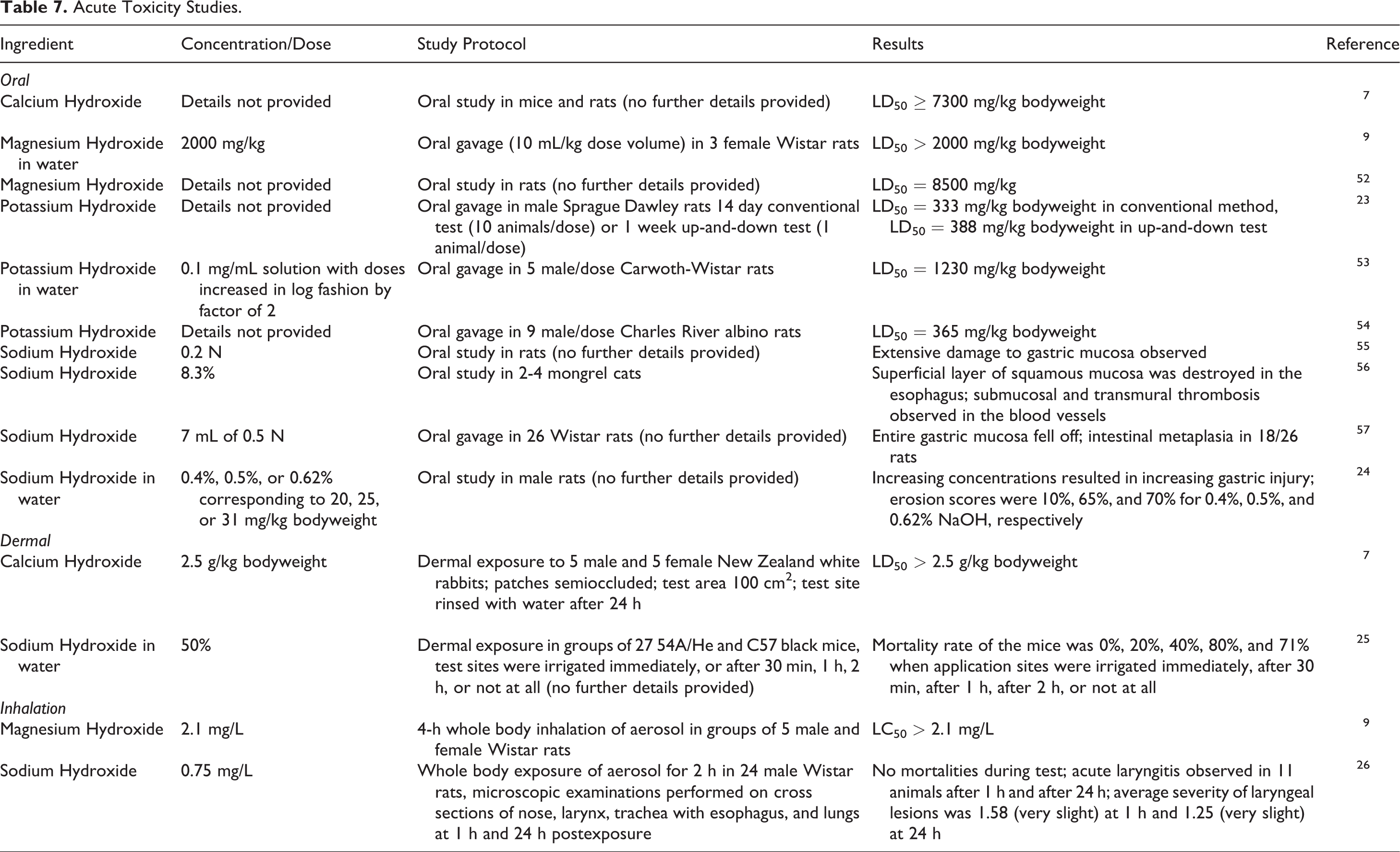
Animal acute toxicity studies are presented in Table 7.7-10,23-26 In oral toxicity studies, Calcium Hydroxide had an LD50 > 7300 mg/kg bodyweight in rats and mice and Magnesium Hydroxide had an LD50 > 2000 mg/kg bodyweight in rats. An LD50 of 1230 mg/kg bodyweight was observed in rats that received Potassium Hydroxide at doses that increased in log fashion by a factor of 2 starting at 0.1 mg/mL solution. Other oral studies of Potassium Hydroxide in rats have LD50 results of 333 to 388 mg/kg bodyweight. Oral studies of Sodium Hydroxide led to extensive gastric damages in the animal tested. In dermal toxicity studies, Calcium Hydroxide had an LD50 > 2.5 g/kg bodyweight in rabbits, and mice treated with 50% Sodium Hydroxide had better survival rates when the test compound was washed off within an hour of application. In inhalation studies in rats, the LC50s for Magnesium Hydroxide and Sodium Hydroxide were > 2.1 mg/L (4-h exposure) and > 0.75 mg/L (2-h exposure), respectively.
Acute Toxicity Studies.
Repeated Dose Toxicity
A combined repeated dose toxicity study and reproductive and developmental toxicity screening test was performed with Magnesium Hydroxide in accordance with Organisation for Economic Co-operation and Development (OECD) guideline 422. 9 These results are described in the Reproductive and Developmental Toxicity section of this report. No other relevant published repeated dose toxicity studies on inorganic hydroxides were identified in a literature search for these ingredients and no unpublished data were submitted.
Reproductive and Developmental Toxicity
Magnesium Hydroxide
The reproductive toxicity of Magnesium Hydroxide (pH = 10) was studied in rats that received the test material via gavage. 9 The study followed OECD guideline 422. Groups of 10 male and 10 female Wistar rats received 0, 110, 330, or 1000 mg/kg bw/d Magnesium Hydroxide in water daily. Males were exposed for 29 days (ie, 2 weeks prior to mating, during mating, and up until treatment end) and females were exposed for 41 to 45 days (ie, 2 weeks prior to mating, during mating, during gestation, and during at least 4 days of lactation). No treatment-related effects were observed on clinical signs, bodyweight or weight gain, feed consumption, or hematology. In parental males, lower total protein levels (330 and 1000 mg/kg dose groups), lower albumin levels (1000 mg/kg dose group), and lower calcium levels (330 and 1000 mg/kg dose groups) in the blood, and lower sodium and potassium excretion (1000 mg/kg dose group) and higher calcium concentration in urine (1000 mg/kg dose group) were observed; however, these changes only just exceeded or remained within normal ranges and there were no corresponding histopathological changes. No toxicologically relevant changes from the test material were observed in parental organ weights or in gross pathology. There were no treatment-related effects on offspring development. The no observed adverse effect level (NOAEL) for parental systemic effects, parental reproductive effects, and offspring effects in this 1 generation rat study of Magnesium Hydroxide was 1000 mg/kg bw/d.
Genotoxicity
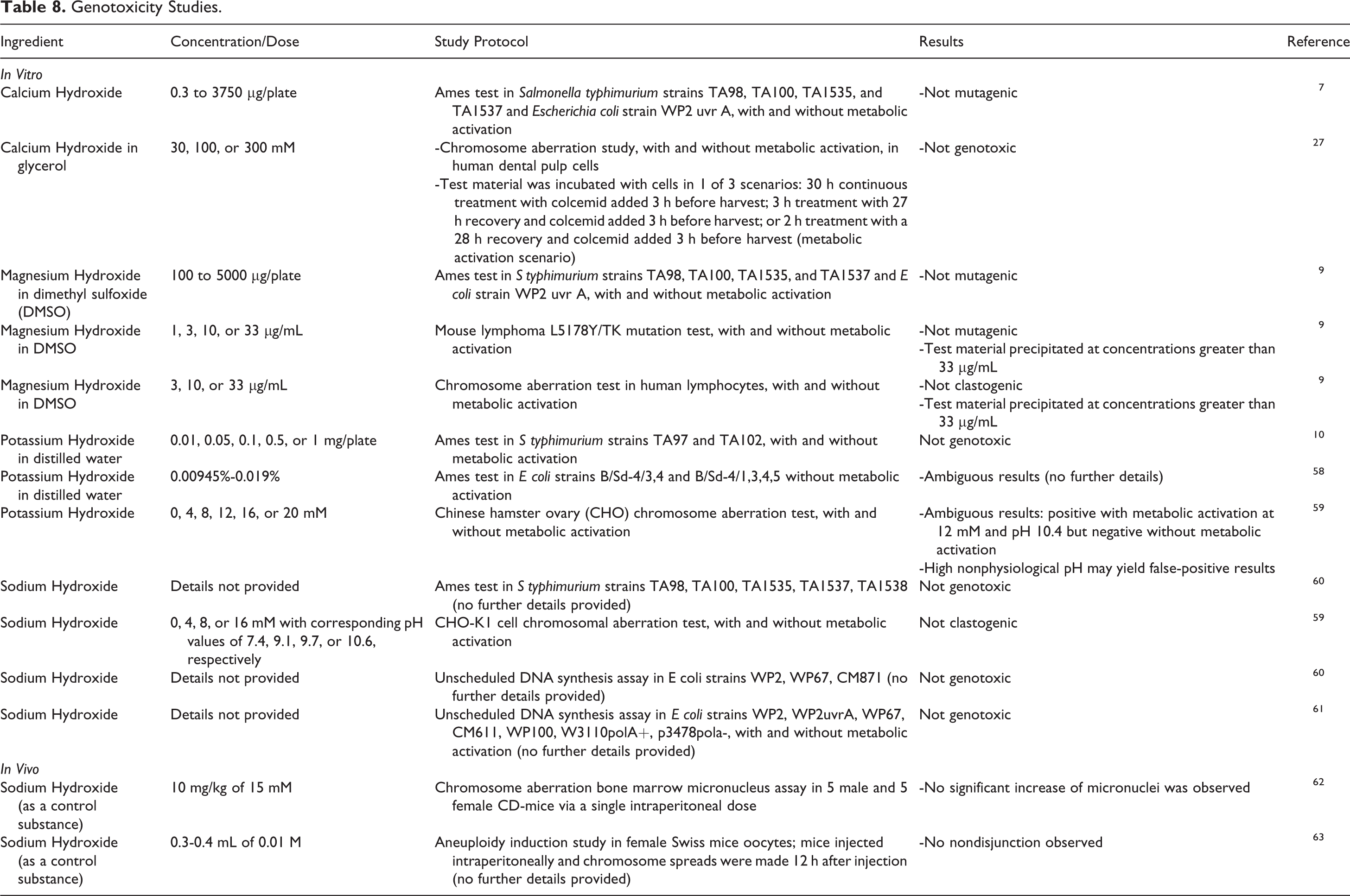
Genotoxicity studies are presented in Table 8.7-10,27 Calcium Hydroxide, Magnesium Hydroxide, and Sodium Hydroxide were not genotoxic in several different in vitro assays. Potassium Hydroxide was not genotoxic in 1e Ames test, but results were ambiguous in another Ames test and a chromosome aberration test. Sodium Hydroxide was not genotoxic in an in vivo mouse oocyte aneuploidy induction study at up to 0.015 M. High, nonphysiological pH may yield false-positive results in genotoxicity studies.
Genotoxicity Studies.
Carcinogenicity
No relevant published carcinogenicity data on inorganic hydroxides were identified in a literature search for these ingredients and no unpublished data were submitted.
Irritation and Sensitization
Dermal Irritation
Sodium Hydroxide is a corrosive material that can produce immediate coagulative necrosis resulting in considerable tissue damage with ulceration and sloughing. 28 Toxicity is a function of pH, with greater toxicity associated with increasing pH values. High pH (strong alkalinity) can cause deep burns and readily denatures keratin. Following exposure, the chemical must be removed quickly and completely in order to avoid further damage to the skin or possible systemic injury.
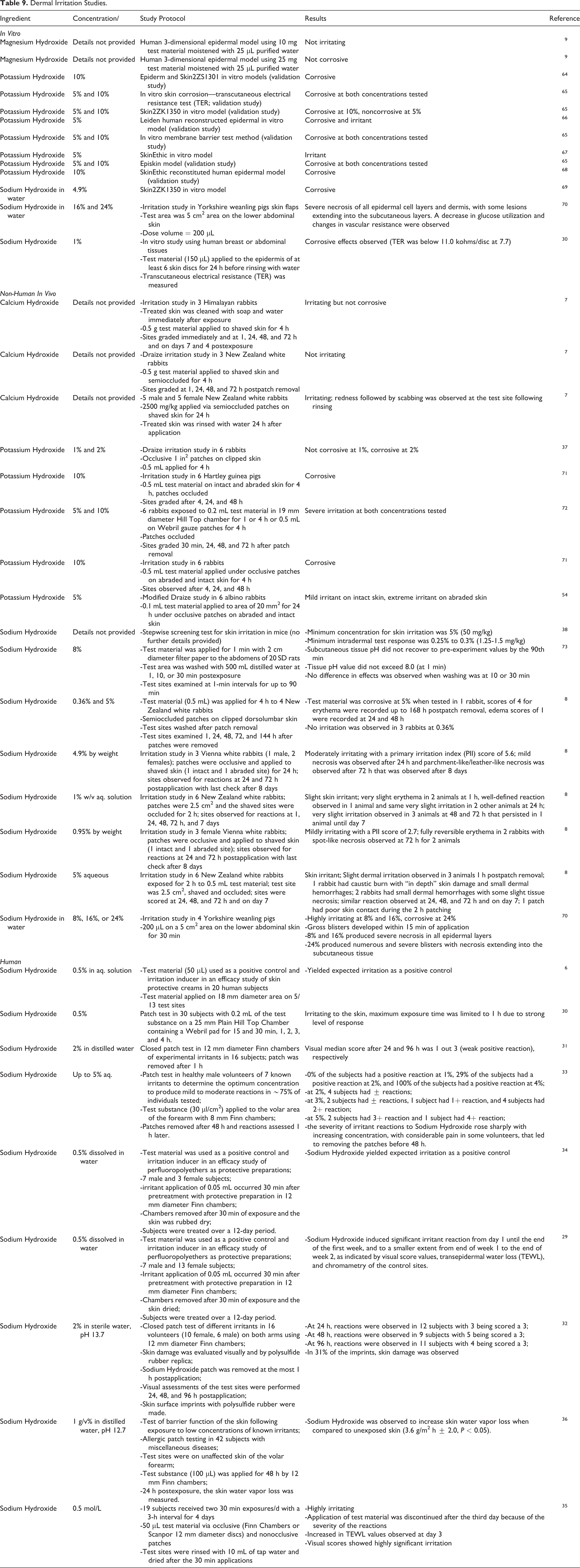
A representative sampling of dermal irritation studies are presented in Table 9.6-10,29-38 Magnesium Hydroxide was not irritating or corrosive when tested in vitro (concentrations not reported); however, Potassium Hydroxide and Sodium Hydroxide were corrosive at concentrations as low as 1%. Calcium Hydroxide was generally irritating but not corrosive in rabbit dermal studies (concentrations not reported). Potassium Hydroxide was irritating and/or corrosive in rabbit (at 2% or greater) and guinea pig (at 10%) studies. Sodium Hydroxide was irritating/corrosive in a concentration-dependent manner in rat, rabbit, and pig studies. In humans, Sodium Hydroxide was irritating at concentrations as low as 0.5%. Because of the large number of studies that include Sodium Hydroxide as a positive control, only a sampling has been presented in this safety assessment.
Dermal Irritation Studies.
Ocular Irritation
Caustic chemicals like Sodium Hydroxide can rapidly penetrate ocular tissues. 28 Toxicity is a function of pH, with greater toxicity associated with increasing pH values. The concentration of the solution and duration of contact with the eye are important determinants of the eventual clinical outcome.
A representative sampling of ocular irritation studies are presented in Table 10.7-10,38-42 Calcium Hydroxide was predicted to be irritating in hen’s egg test-chorioallantoic membrane (HET-CAM) in vitro tests while Magnesium Hydroxide was predicted not to be irritating in a bovine corneal opacity and permeability (BCOP) in vitro test. In rabbit studies, Calcium Hydroxide was severely irritating at a concentration as low as 10% and pH of 9. Potassium Hydroxide and Sodium Hydroxide were severely irritating and/or corrosive in a concentration-dependent manner in rodents. Magnesium Hydroxide was not irritating in a rabbit study.
Ocular Irritation Studies.
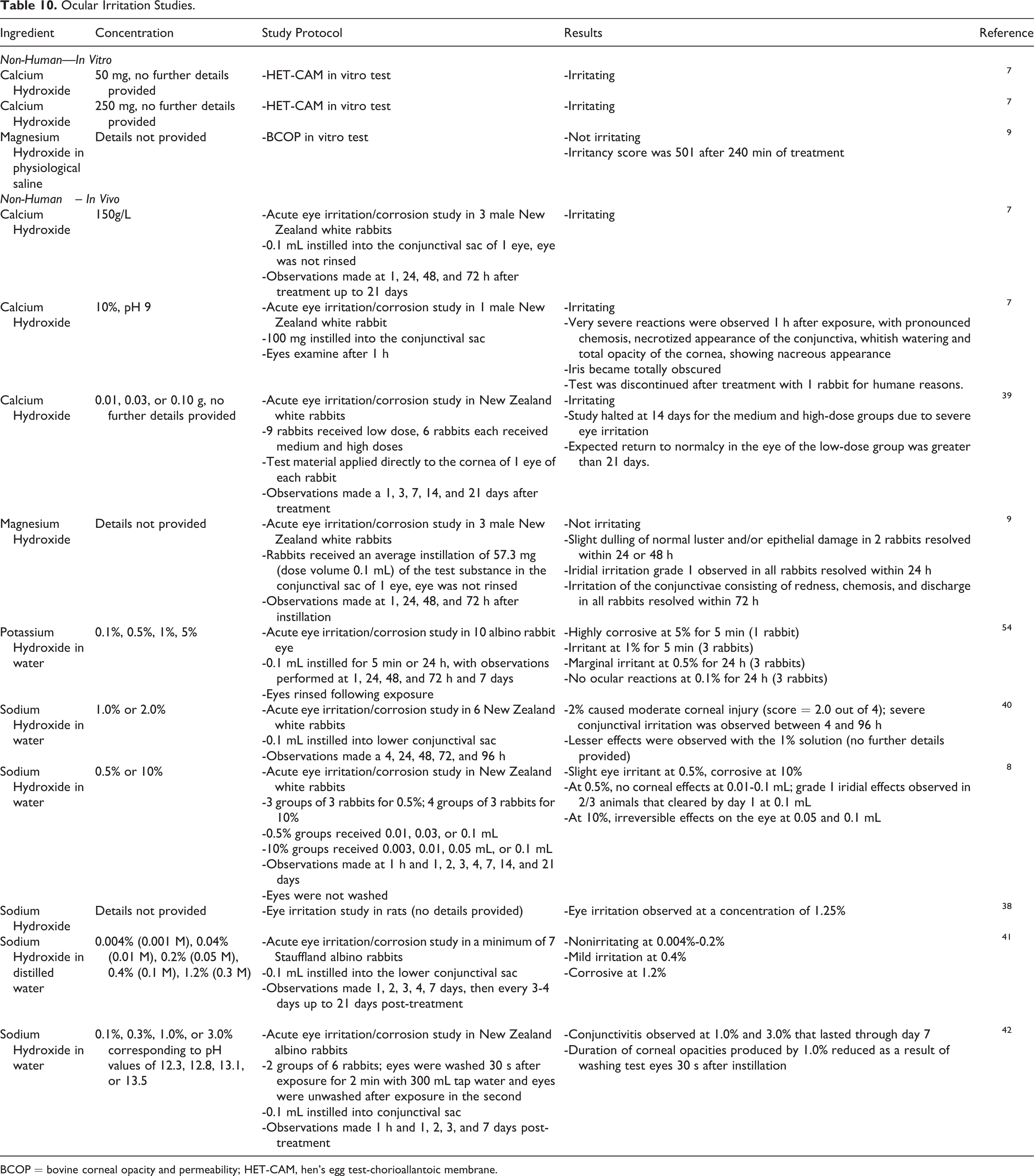
BCOP = bovine corneal opacity and permeability; HET-CAM, hen’s egg test-chorioallantoic membrane.
Dermal Sensitization
Dermal sensitization studies are summarized in Table 11.8-10 Potassium Hydroxide (0.1%) was not sensitizing in a guinea pig study, whereas Magnesium Hydroxide in propylene glycol was sensitizing in a local lymph node assay (LLNA) when tested at up to 50%. In a human repeat insult patch test (HRIPT), Sodium Hydroxide was not sensitizing when induced at up to 1.0% and challenged at 0.125%, but irritation was observed.
Sensitization Studies.

HRIPT = human repeat insult patch test.
Case Reports
There were no case reports discovered in the published literature indicating adverse events that could be associated specifically with the presence of inorganic hydroxide ingredients in cosmetic products. However, there are numerous case reports suggesting an association between the use of depilatory and hair straightening products that contain active ingredients like thioglycolates (Table 5) as well as inorganic hydroxides.43-50
Summary
The inorganic hydroxides, Calcium Hydroxide, Magnesium Hydroxide, Potassium Hydroxide, and Sodium Hydroxide, are all alkaline salts and are reported in the Dictionary to function most commonly as pH adjusters in cosmetics; however, representatives from the cosmetics industry have stated that the inorganic hydroxides are added to depilating and hair waving/straightening formulations to raise pH values. Inorganic hydroxides, except for Magnesium Hydroxide, are well known caustic agents that can cause severe burns and corrosion in acute exposures. Sodium Hydroxide is commonly used as a positive control in efficacy studies of skin protective creams and in other studies of irritant contact dermatitis.
According to the 2015 VCRP data, Sodium Hydroxide has the most reported uses of the ingredients listed in this safety assessment in cosmetic products, with a total of 5147; about half of the uses are in leave-on skin care products. Potassium Hydroxide has the second greatest number of overall uses reported, with a total of 1074; the majority of the uses also are in leave-on skin care products. The results of the concentration of use survey conducted in 2014 by the Council indicate Calcium Hydroxide has the highest reported maximum concentration of use; it is used at up to 13.2% in rinse-off shaving preparations. However, it is only used up to 0.5% in leave-on products (deodorants). Sodium Hydroxide is used at up to 10% in an “other” skin care preparation, which may or may not be a leave-on product. The next highest concentration of use for a leave-on product for Sodium Hydroxide is 6.9% in a face or neck product.
The inorganic hydroxides in this report are GRAS as direct food substances and as feed additives for animals. The FDA has also reviewed Calcium Hydroxide and Magnesium Hydroxide for use as an active ingredient in OTC drugs. Inorganic hydroxides have numerous non-cosmetic uses.
In oral toxicity studies, Calcium Hydroxide had an LD50 > 7300 mg/kg bodyweight in rats and mice and Magnesium Hydroxide had an LD50 > 2000 mg/kg bodyweight in rats. An LD50 of 1230 mg/kg bodyweight was observed in rats that received Potassium Hydroxide at doses that increased in log fashion by a factor of 2 starting at 0.1 mg/mL solution. Other oral studies of Potassium Hydroxide in rats have LD50 results of 333 to 388 mg/kg bodyweight. Oral studies of Sodium Hydroxide led to extensive gastric damages in the animal tested. In dermal toxicity studies, Calcium Hydroxide had an LD50 > 2.5 g/kg bodyweight in rabbits, and mice treated with 50% Sodium Hydroxide had better survival rates when the test compound was washed off within an hour of application. In inhalation studies in rats, the LC50s for Magnesium Hydroxide and Sodium Hydroxide were > 2.1 mg/L (4-h exposure) and >750 µg/L (2-h exposure), respectively.
The NOAEL for parental and offspring effects following oral exposure to Magnesium Hydroxide (pH = 10) was 1000 mg/kg bw/d. No treatment-related effects were observed on clinical signs, bodyweight or weight gain, feed consumption, or hematology. No toxicologically relevant changes from the test material were observed in parental organ weights or in gross pathology. There were no treatment-related effects on offspring development.
Calcium Hydroxide, Magnesium Hydroxide, and Sodium Hydroxide were not genotoxic in several different in vitro assays. Potassium Hydroxide was not genotoxic in 1 Ames test, but results were ambiguous in another Ames test and a chromosome aberration test. Sodium Hydroxide was not genotoxic in mice studies (intraperitoneal injection) at up to 0.015 M.
Magnesium Hydroxide was not irritating or corrosive in in vitro tests; however, Potassium Hydroxide and Sodium Hydroxide were corrosive at concentrations as low as 5%. Calcium Hydroxide was irritating but not corrosive in dermal rabbit studies. Potassium Hydroxide was irritating and/or corrosive in rabbit and guinea pig studies at concentrations of 2% or greater. Sodium Hydroxide was irritating and/or corrosive in a concentration-dependent manner in rat, rabbit, and pig studies. In humans, Sodium Hydroxide was irritating at concentrations as low as 0.5%. Because of the large number of studies that include Sodium Hydroxide as a positive control, only a sampling has been presented in this safety assessment.
In ocular studies, Calcium Hydroxide was irritating in HET-CAM in vitro tests, whereas Magnesium Hydroxide was not irritating in a BCOP in vitro test. In rabbit studies, Calcium Hydroxide was severely irritating at a concentration of 10% and pH of 9. Potassium Hydroxide and Sodium Hydroxide were severely irritating and/or corrosive in a concentration-dependent manner. Magnesium Hydroxide was not irritating in a rabbit study.
Potassium Hydroxide (0.1%) was not sensitizing in a guinea pig study while Magnesium Hydroxide in propylene glycol was sensitizing in an LLNA when tested at up to 50%. In an HRIPT, Sodium Hydroxide was not sensitizing when induced at up to 1.0% and challenged at 0.125%, but irritation was observed.
There were no case reports discovered in the published literature indicating adverse events that could be associated specifically with the presence of inorganic hydroxide ingredients in cosmetic products. However, there are numerous case reports suggesting an association between the use of depilatory and hair straightening products that contain active ingredients like thioglycolates, as well as inorganic hydroxides.
Discussion
The Panel reviewed studies performed to assess the dermal and ocular irritation and sensitization potential of the inorganic hydroxide ingredients. The Panel recognized that these ingredients can cause dermal and/or ocular irritation when used to adjust the pH of highly alkaline cosmetic formulations, specifically depilatory and hair straightening products. However, most of the alkalinity of the inorganic hydroxides in other types of cosmetic products will be neutralized by other ingredients in the formulation. Thus, neither local nor systemic toxicity is expected to be a concern for the use of the inorganic hydroxide ingredients in such formulations.
The safety of inorganic hydroxide ingredients as pH adjusters should not be based on the concentration of use, but on the concentration of free hydroxide ions that remain in a formulation. In general, the concentration of free hydroxide ion in a formulation depends on the acidity of the other ingredients in the formulation. The concentration of free hydroxide ions is expected to be low in cosmetic formulations, except in some depilatory and hair-straightening formulations.
The inorganic hydroxide ingredients are not listed in the Dictionary as depilating or hair waving/straightening agents. However, inorganic hydroxides are added to some depilating and waving/straightening products to enable or enhance the depilatory and waving/straightening action of these products by increasing the pH of the formulations to high values. If the inorganic hydroxides are used in hair waving/straightening products, use concentrations should be limited and adequate instructions should be provided to users to prevent skin contact on the hands (such as by wearing gloves) and to minimize skin exposure (such as by limiting the frequency of product use) to ensure that irritation is not a concern. The Panel noted, for example, that hair dressers should use adequate measures to protect their skin before repeatedly applying hair straighteners containing inorganic hydroxides to multiple clients over a short period. In addition, users should avoid prolonged skin exposures on hands and scalp. The Panel emphasized that following manufacturers’ instructions on the proper application and use are especially important if these products are used on children.
The Panel further discussed the potential for skin irritation and sensitization specifically from the use of inorganic hydroxides in depilatories. The Panel recognized that nearly all methods of hair removal may cause some irritation. However, clinically significant adverse reactions to the ingredients in depilatories are not commonly seen in the experience of the Panel. This indicates that current products are formulated to be practically nonirritating under conditions of recommended use.
No case reports were discovered indicating that topical exposures to inorganic hydroxides in cosmetic products are generally associated with adverse health effects. However, there are numerous case reports of adverse events associated with the use of depilatory or hair-straightening products containing inorganic hydroxides. These events were not clearly attributable to the inorganic hydroxides rather than to other ingredients, such as thioglycolates, in such products.
These ingredients are also reported to function as absorbents (Magnesium Hydroxide) and denaturants (Sodium Hydroxide) in cosmetic products. The Panel had no concerns about the safe use of these ingredients when used for these purposes in cosmetic products that are formulated to be nonirritating.
No carcinogenicity data were discovered. However, the Panel agreed that the cations of the hydroxides used in cosmetics are not expected to cause cancer or other local or systemic toxicity.
The Panel discussed the issue of incidental inhalation exposure from hair sprays and body and hand sprays, hair color sprays, fragrance preparations, and foot powders. Limited data are available from acute inhalation toxicity studies on Magnesium Hydroxide and Sodium Hydroxide. There were no inhalation toxicity data available on the remaining ingredients. These ingredients are reportedly used at concentrations up to 0.69% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for inorganic hydroxides to cause irritation and sensitization and, as noted above, recognize that these potentially irritating substances would be neutralized in formulation and are unlikely to cause local effects in the respiratory tract. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Calcium Hydroxide, Magnesium Hydroxide, Potassium Hydroxide, and Sodium Hydroxide are safe in hair straighteners and depilatories under conditions of recommended use; users should minimize skin contact. These ingredients are also safe for all other present practices of use and concentration described in this safety assessment when formulated to be nonirritating.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Burnett, C. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript; Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.