Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reopened the safety assessment of Acid Violet 43, a cosmetic ingredient that is an anthraquinone dye reported to function in cosmetics as a colorant. This colorant has the same chemical structure as Ext. D&C Violet No. 2, which is a certified colorant; however, Acid Violet 43 is not a certified color and it could have impurities that are not allowed in the certified color. The Panel reviewed relevant new data related to this ingredient and concluded that Acid Violet 43 is safe in the present practices of use and concentration for use in hair dye formulations. This conclusion supersedes the previous conclusion for Acid Violet 43 that included impurity specifications indicated for the certified color.
Keywords
Introduction
In 2001, the Expert Panel for Cosmetic Ingredient Safety (Panel) published a safety assessment with the conclusion that Acid Violet 43 is safe for use in hair dye formulations when free of impurities, except for the following: ≤18% volatile matter (at 135 °C) and chlorides and sulfates (calculated as sodium salts); ≤0.4% water-insoluble matter; ≤0.2% 1-hydroxy-9,10-anthracenedione; ≤0.2% 1,4-dihydroxy-9,10-anthracenedione; ≤0.1% p-toluidine; ≤0.2% p-toluidine sulfonic acids, sodium salts; ≤1% subsidiary colors; ≤20 ppm lead (as Pb); ≤3 ppm arsenic (as As); ≤1 ppm mercury as (Hg); and ≥80% total color. 1 These restrictions refer to the specifications for Ext. D&C Violet No. 2. Acid Violet 43 has the same chemical structure as Ext. D&C Violet No. 2, but it is not a certified color and it could have impurities that are not allowed in the certified color. These specifications were included in the Panel’s original conclusion on Acid Violet 43 because the safety test data included in the original assessment pertained to Ext. D&C Violet No. 2.
In accordance with the Cosmetic Ingredient Review (CIR) Procedures, the Panel evaluates the conclusions of previously issued reports every 15 years to determine whether the conclusion should be reaffirmed or revised. During the literature search conducted for the re-review, an updated European Commission (EC) Scientific Committee on Consumer Safety (SCCS) opinion was discovered. Because this opinion included new data on Acid Violet 43, the Panel conducted a re-review. 2 According to the SCCS opinion, Acid Violet 43 is used as a nonoxidative (ie, direct or semipermanent) hair dye. One source 3 indicates that it can also be used in oxidative hair coloring products, but no other sources corroborated that type of use; therefore, this safety assessment pertains to the use of Acid Violet 43 as a nonoxidative hair dye.
This safety assessment includes relevant published and unpublished data that are available for each end point that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the end points that the Panel typically evaluates, are provided on the CIR websites (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Excerpts from the summary of the 2001 report are disseminated throughout the text of this re-review document, as appropriate. As stated above, the toxicity data from that original assessment pertain to the certified dye.
Chemistry
Definition and Structure
Acid Violet 43 (CAS No. 4430-18-6; technical name benzenesulfonic acid, 2-[(9,10-dihydro-4-hydroxy-9,10-dioxo-1-anthracenyl)amino]-5-methyl-, monosodium salt) is reported to function in cosmetics, according to the International Cosmetic Ingredient Dictionary and Handbook (Dictionary), as a colorant and is classed chemically as an anthraquinone color. 4 It conforms to the formula in Figure 1:
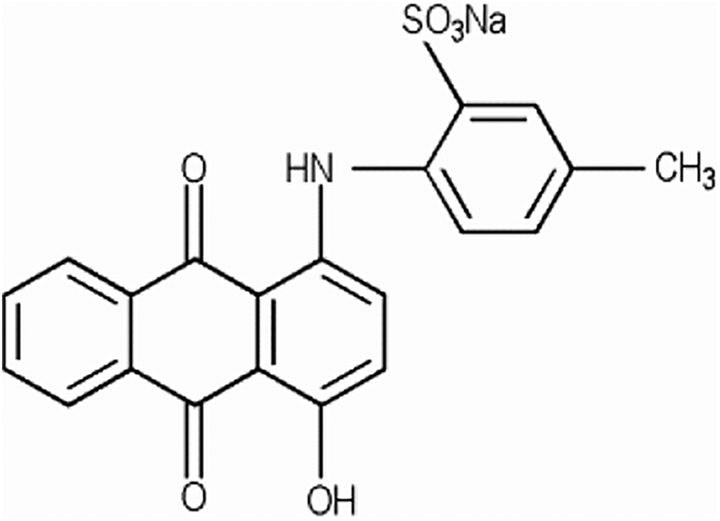
Acid Violet 43.
Acid Violet 43 is the Dictionary name for batches of this colorant that have not been certified. 4 To identify the certified colorant for labeling purposes in the United States, the Dictionary name Ext. Violet 2 must be used. In the European Union (EU) and Japan, the Dictionary names CI 60730 and Murasaki401, respectively, must be used for identification of this colorant.
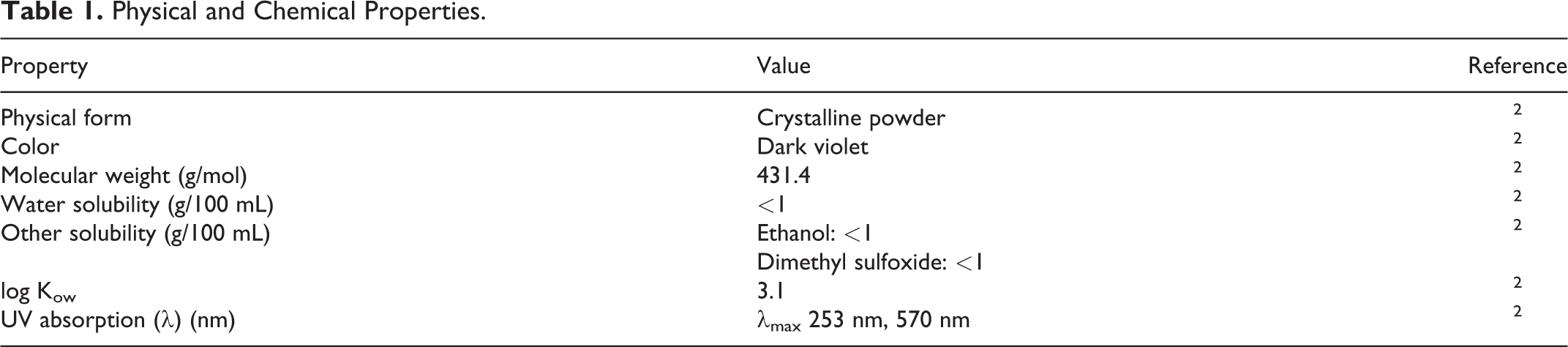
Physical and Chemical Properties
Acid Violet 43 is a dark violet crystalline powder with a molecular weight of 431.4 Da2 (Table 1). It absorbs in the UV–vis spectrum at 253 and 570 nm.
Physical and Chemical Properties.
Method of Manufacture
Acid Violet 43 can be prepared by sulfonating the anthraquinone color, CI 60725 (Figure 2), and converting it to the sodium salt. 1
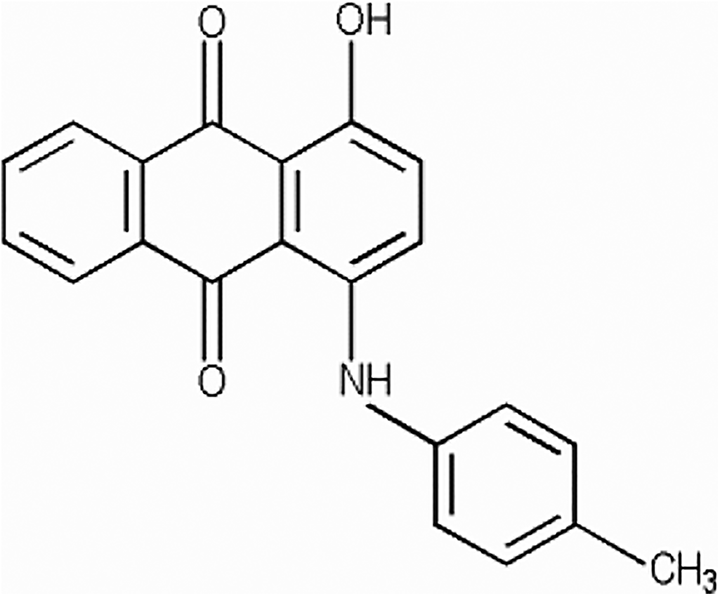
CI 60725. 4
Impurities/Composition
Acid Violet 43, as a noncertified color, is commercially available in different forms with different analytical specifications and impurity profiles. It tends to have more impurities than the certified color, Ext. D&C Violet No. 2, which contains the same color chemically but is of higher purity. The studies summarized in the SCCS Opinion were performed with both Acid Violet 43 and Ext. D&C Violet No. 2. Three batches of each compound were analyzed; the purity of Acid Violet 43, as tested in the studies reported in the SCCS opinion, ranged from 54.4% to 60.5% and that of Ext. D&C Violet No. 2 ranged from 84% to 93.8% (Table 2). 2
Composition of Test Compounds Studied in the SCCS Opinion: Acid Violet 43 and Ext. D&C Violet No. 22.
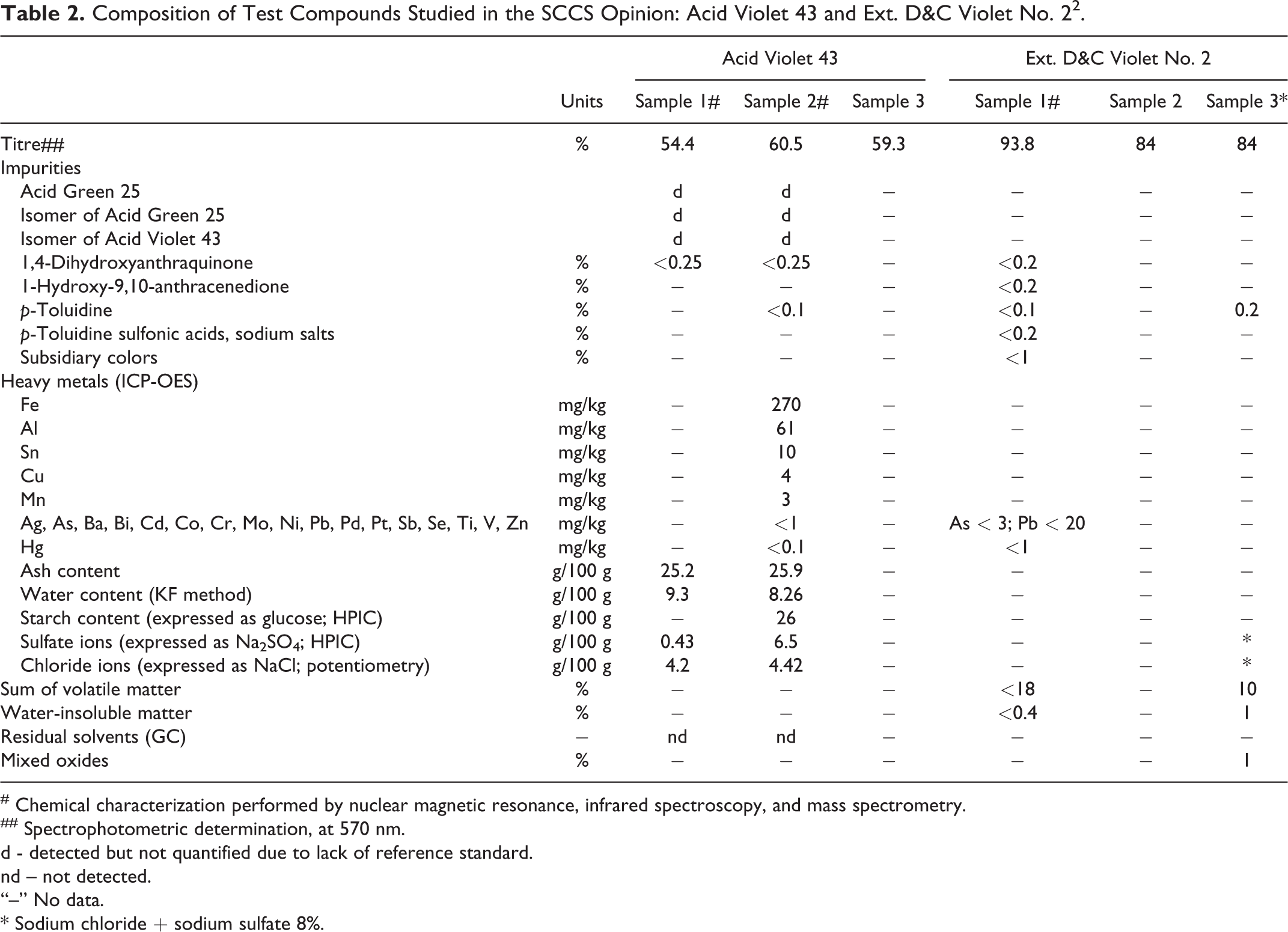
# Chemical characterization performed by nuclear magnetic resonance, infrared spectroscopy, and mass spectrometry.
## Spectrophotometric determination, at 570 nm.
d - detected but not quantified due to lack of reference standard.
nd – not detected.
“–” No data.
* Sodium chloride + sodium sulfate 8%.
The specifications for Ext. D&C Violet No. 2, as described in the conclusion of the Panel safety assessment published in 2001, have not changed [21CFR74.2602a].
Use
Cosmetic
The safety of the cosmetic ingredient addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
Acid Violet 43 is a nonoxidative (ie, direct) hair dye, but it also reported that it might be used in oxidative hair coloring products. 3 Based on 2016 VCRP data, Acid Violet 43 is reported to be used in 48 hair coloring preparations; 23 of the reported uses are in coloring hair rinses. 5 According to the results of the Council survey, in which data were collected in 2015 and 2016, the maximum reported concentrations of use of Acid Violet 43 range from 0.13% to 0.35%; the highest reported maximum concentration of use is 0.35% in hair tints. 6 In the original safety assessment, Acid Violet 43 was reported to be used in only 2 hair coloring formulations in 1998; at that time, concentration of used data was not available. 1 However, historical FDA data included in that report indicated that, in 1984, Acid Violet 43 was reported to be used in 30 coloring hair formulations at concentrations ≤0.1% and in 1 coloring shampoo at 0.1% to 1%.
Acid Violet 43 is considered a coal tar hair dye for which regulations require caution statements and instructions regarding patch tests in order to be exempt from certain adulteration and color additive provisions of the US Federal Food, Drug, and Cosmetic Act. In order to be exempt, the following caution statement must be displayed on all coal tar hair dye products:
Caution: This product contains ingredients that may cause skin irritation on certain individuals and a preliminary test according to accompanying directions should be made. This product must not be used for dying the eyelashes or eyebrows; to do so may cause blindness.
Product labels shall also bear patch test instructions for determining whether the product causes skin irritation. The Panel recommends that an open patch test be applied and evaluated by the beautician and/or consumer for sensitization 48 hours after application of the test material and prior to the use of a hair dye formulation.
In 2012, a report was published regarding such self-testing for contact sensitization to hair dyes. 7 The authors of that report concluded that, in its present form, the hair dye self-test has severe limitations. The authors issued the warning that, if the use of a hair dye self-test to predict contact sensitization becomes widespread, there is severe risk that a tool has been marketed that may cause morbidity in European consumers. An accompanying editorial written on behalf of the European Society of Contact Dermatitis (ESCD) asserted that industry is focusing on predicting the risks from exposure to hair dyes by having millions of European consumers perform a self-test prior to each hair dying and stated that it is the opinion of the ESCD that attention must be given to reducing the risks of serious allergic reactions by improving the safety of the products themselves. 8
In 2013, the SCCS concluded that the use of Acid Violet 43 (as related to batch Ext. D&C Violet No.2 0609RA, purity of 94%) as a nonoxidative hair dye with a maximum on head concentration of 0.5% does not pose a risk to the health of the consumer. 2 The SCCS also stated that a sensitizing potential cannot be excluded. The conclusion of the SCCS opinion stated that “Acid Violet 43 is also used as a colorant, but that use was not assessed in this opinion.”
In the EU, sodium, 4-[(9,10-dihydro-4-hydroxy-9,10-dioxo-1-anthryl)amino]toluene-3-sulphonate (ie, Acid Violet 43) is listed in Annex III (ie, List of Substances Which Cosmetic Products Must Not Contain Except Subject to the Restrictions Laid Down; III/291) of the Cosmetic Regulations (Regulation EU No. 1190/2015). It is allowed as a hair dye substance in nonoxidative hair dye products at a maximum concentration in a ready-for-use preparation of 0.5%. 9 Sodium 4-[(9,10-dihydro-4-hydroxy-9,10-dioxo-1-anthryl)amino]toluene-3-sulphonate, as CI 60730, is listed in Annex IV (ie, List of Colorants Allowed in Cosmetics; IV/90) of the Cosmetic Regulations (Regulation EC No. 1223/2009), with the restriction that is not to be used in products applied to mucous membranes. 10
In the United States, Ext. Violet 2 may be safely used for coloring externally applied cosmetics in amounts consistent with good manufacturing practice [21CFR74.2602a]. Externally applied cosmetics may not be used in products that are applied to the lips or any body surface covered by mucous membrane. 11
Toxicokinetic Studies
Dermal Penetration
In vitro
The percutaneous absorption of Acid Violet 43 (59.3% pure) was evaluated using human skin samples. 2 The skin samples (thickness 380 ± 25 µm) were mounted in diffusion cells and exposed for 30 minutes to 0.12% Acid Violet 43 in a semipermanent hair dye formulation (20 mg/cm2, corresponding to 25.4 µg dye/cm2). The skin surface was washed after exposure, and percutaneous absorption was determined 24 hours after application by measuring the concentration of Acid Violet 43 in the washing, stratum corneum (isolated via tape stripping), the epidermis + dermis, and the receptor fluid (0.9% saline). Most of the test substance applied on the skin surface was removed by the washing procedure (∼104% of the applied dose), and the total calculated recovery was ∼105%. No Acid Violet 43 was measured in the receptor fluid. The mean absorbed amount of Acid Violet 43 (estimated as the sum of amounts measured in epidermis, dermis, and receptor fluid, and assuming concentrations at the detection limit in the receptor fluid of 40 ng) was 0.11 ± 0.06 µg dye/cm2 (0.53% ± 0.33% of the applied dose).
Two experiments were performed using porcine ear skin mounted in glass flow-through diffusion chambers to determine the percutaneous absorption of Ext. D&C Violet No. 2 (95% pure). 2 The skin sample thickness was 100 to 450 µm in experiment 1 and 800 to 900 µm in experiment 2. In the first experiment, 2 mg/mL of a 20% ethanol in a water solution was tested, and in the second experiment, it was 0.5% Ext. D&C Violet No. 2 in a semipermanent hair dye formulation. The per surface area dose of the formulation in both studies was 1000 mg/cm2. The amount of test substance placed in the donor chamber was 1 mL in the first experiment and 1.25 mL of the hair dye formulation (corresponding to 5 mg/cm2 test substance) in the second experiment. For both studies, the exposure time was 30 minutes, and the receptor fluid was 20% ethanol in water. The remaining test material was removed at the end of the exposure period, and the donor chambers were filled with 1 mL of receptor fluid; samples were collected from the receptor chamber at several intervals during the exposure period for up to 24 hours. Because the stratum corneum was not separated from the epidermal and dermal compartments, the amounts of test material in receptor solution plus that in skin extracts were considered to be absorbed. After 24 hours of sampling, the mean total recovery of test material was 88.8% ± 4.08% in experiment 1 and 90.6% ± 4.67% in experiment 2. The test substance could not be detected in the receptor fluid at any sampling time or in skin extracts of experiment 1 (the limit of detection was 500 ng/mL). Maximal flux rates were calculated by assuming the detection limit as the concentration of the test material in the receptor fluid samples. Absorption was estimated by adding skin extract values to the calculated fluxes. Based on the highest values calculated for the flux (19.3 µg or 19.1 μg/cm2 in experiment 1 and 19.0 µg or 24.8 μg/cm2 in experiment 2) and values for skin extracts (1.85 ± 0.01 µg or 1.83 ± 0.01 μg/cm2 in experiment 1, based on the detection limit of 500 ng/mL, and 5.82 ± 2.67 µg or 5.75 ± 2.6 μg/cm2 in experiment 2), the highest rate for skin absorption was estimated as 20.93 μg/cm2 (∼1%) in experiment 1 and 30.55 μg/cm2 (∼0.6%) in experiment 2.
Absorption, Distribution, Metabolism, and Excretion
A toxicokinetic study of Acid Violet 43 (59.8% pure) was performed as part of a bone marrow micronucleus test (described in section “Genotoxicity”). 2 In the toxicokinetic study, 3 mice/sex/sampling time were given a single dosage by gavage of 0 or 2000 mg dye/kg bw (∼3348 mg dye/kg bw) in water (20 mL/kg bw). Blood samples were taken 15, 30, or 60 minutes after dosing (single sample per animal). There were no clinical signs of toxicity and no mortality. Oral bioavailability was demonstrated by “high blood levels of Acid Violet 43 observed in all animals (range 5.7-34.6 µg/mL).”
Toxicological Studies
Acute Toxicity Studies
The oral LD50 of Ext. D&C Violet No. 2 was >4640 mg/kg for male rats and >2000 mg/kg for mongrel dogs. 1
Short-Term Toxicity Studies
In a short-term toxicity study using guinea pigs in which dermal applications of a US Pharmacopeia (USP) hydrophilic ointment containing 0.1% or 1.0% Ext. D&C Violet No. 2 were made 5 d/wk for 3 weeks, test article-related gross and microscopic lesions were not observed. 1
Subchronic Toxicity Studies
Dermal
In a subchronic toxicity study using rabbits in which dermal applications of a USP hydrophilic ointment containing 0.1% or 1.0% Ext. D&C Violet No. 2 were made 5 d/wk for a total of 65 applications, test article-related gross and microscopic lesions were not observed. 1
Oral
Groups of 10 male and 10 female Sprague-Dawley rats were dosed once daily by gavage for 13 weeks with 0, 50, 200, or 800 mg/kg bw/d Acid Violet 43 (54.4% pure) in 5 mL/kg water. 2 The doses corresponded to 0, 27, 109, or 435 mg dye/kg bw/d, respectively. The animals were observed daily, body weights, and feed consumption were measured weekly, and hematology, blood clinical chemistry, and urinalysis parameters were determined at study termination. At necropsy, gross and macroscopic examinations were performed, and organ weights, including those of the ovaries and testes, were measured.
No test article-related mortality was reported. Dose-dependent increases in salivation were observed at all dose levels. Colored urine, feces, hair, and extremities were observed at all doses and were attributed to the staining properties of the test article. No other clinical signs, no ocular findings, and no changes in body weights and feed consumption were reported. Statistically significant changes in blood clinical chemistry parameters were considered to be of no toxicological importance. Several statistically significant changes in hematological parameters were observed; only an increased activated partial thromboplastin time (APTT) in high-dose males was considered to be treatment related. There were no significant differences in organ weights among treated and control groups. Dose-related greenish contents or greenish colorations of the mucosa of the digestive tract were observed at necropsy. No treatment-related microscopic changes were observed. The no-observed adverse effect level (NOAEL) was 200 mg/kg bw/d Acid Violet 43 (109 mg dye/kg bw/d), based on the increased APTT in the high-dose males.
A 13-week study following the same protocol was performed with Ext. D&C Violet No. 2 (93.8% pure). 2 Groups of 10 male and 10 female Wistar rats were dosed daily for 13 weeks by gavage with 0, 100, 300, and 1000 mg/kg bw/d Ext. D&C Violet No. 2 (0, 94, 282, or 940 mg dye/kg bw/d, respectively) in 1% aqueous carboxymethylcellulose. None of the animals died during the study. No adverse clinical signs were reported, and there were no changes in body weights or feed consumption. Dark blue feces were observed at all doses and were attributed to staining properties of the test substance. Decreased locomotor activity in high-dose males (observed after 15 minutes) and in mid- and high-dose females (after 45 minutes, and persisting in high-dose females) and changes in grip strength were considered unlikely to be test article related. A statistically significant increase in prothrombin time was reported in low- and high-dose males, and an increase in APTT was reported in high-dose males. Changes observed in blood clinical chemistry laboratory parameters and in urinalysis were considered by the study authors to be of no toxicological significance. Statistically significant changes in some organ weights were considered incidental. According to the researchers, the NOAEL was 1000 mg/kg bw/d Ext. D&C Violet No. 2 (940 mg dye/kg bw/d). However, the SCCS considered the increased APTT in high-dose males an adverse effect, and the NOAEL to therefore be 300 mg/kg bw/d Ext. D&C Violet No. 2 (282 mg dye/kg bw/d).
Developmental and Reproductive Toxicity Studies
Groups of 25 female Sprague-Dawley rats were administered Acid Violet 43 (54.4% pure) in water by gavage at a dose of 0, 50, 200, or 800 mg/kg bw/d (equivalent to 0, 27, 109, or 435 mg dye/kg bw/d) on days 6 to 15 of gestation. 2 The animals were killed on day 20 of gestation. There were 18, 23, 21, and 21 gravid animals in the 0, 50, 200, or 800 mg/kg bw/d groups, respectively. No mortality was reported. Increased salivation was observed in the 800 mg/kg bw/d group. Discolored feces were observed in all groups, except in the low-dose group. Discoloration of placenta was also observed in the high-dose group. No effects on litter parameters or fetal weight were reported. According to the researcher, there were no external soft-tissue or skeletal anomalies that could be attributed to treatment with the test substance. The NOAEL was 800 mg/kg bw/d (435 mg dye/kg bw/d) for teratogenicity and maternal toxicity.
Groups of 22 female Wistar rats were administered 1% Ext. D&C Violet No. 2 (93.8% pure) in carboxymethylcellulose by gavage at doses of 0, 100, 300, and 1000 mg/kg bw/d (equivalent to 0, 94, 282, or 940 mg dye/kg bw/d, respectively) on days 6 to 17 of gestation. 2 The animals were killed on day 21 of gestation. Twenty to 22 females per group were gravid. No mortality was reported. Discolored feces were observed in the high-dose group. One low-dose and 1 high-dose animal had only embryonic resorptions, and 1 high-dose animal had only empty implantation sites; the researcher considered these to be incidental findings (ie, not likely to be test article related) due to the lack of an observed dose-dependent response. Fetal and litter incidences of external, soft–tissue, and skeletal anomalies were similar for control and treated groups. The NOAEL for teratogenicity and maternal toxicity was 1000 mg/kg bw/d (940 mg dye/kg bw/d).
Genotoxicity Studies
In Vitro
Ext. D&C Violet No. 2 was not mutagenic in a spot and/or plate test of a Salmonella/mammalian microsome test with or without metabolic activation. 1
Acid Violet 43 (54.4% pure) and Ext. D&C Violet No. 2 (95% pure) were not mutagenic in the Ames test when tested at concentrations ≥2720 µg/plate in water or ≥4750 µg dye/plate in dimethyl sulfoxide (DMSO), respectively (Table 3). In chromosomal aberration assays of Acid Violet 43 (54.4% pure), significant increases in aberrant cell frequency were observed with a 2-hour exposure at the highest test concentration (408 µg/mL) with metabolic activation, and in the 48-hour treatment group at the highest concentration (272 μg/mL) without metabolic activation in human lymphocytes; and structural aberrations were statistically significantly increased in Chinese hamster ovary (CHO) cells at concentrations ≥102 µg dye/mL. Ext. D&C Violet No. 2, at concentrations ≤342 µg dye/mL in DMSO, was not genotoxic in a mammalian cell mutation assay in mouse lymphoma cells.
Genotoxicity studies.
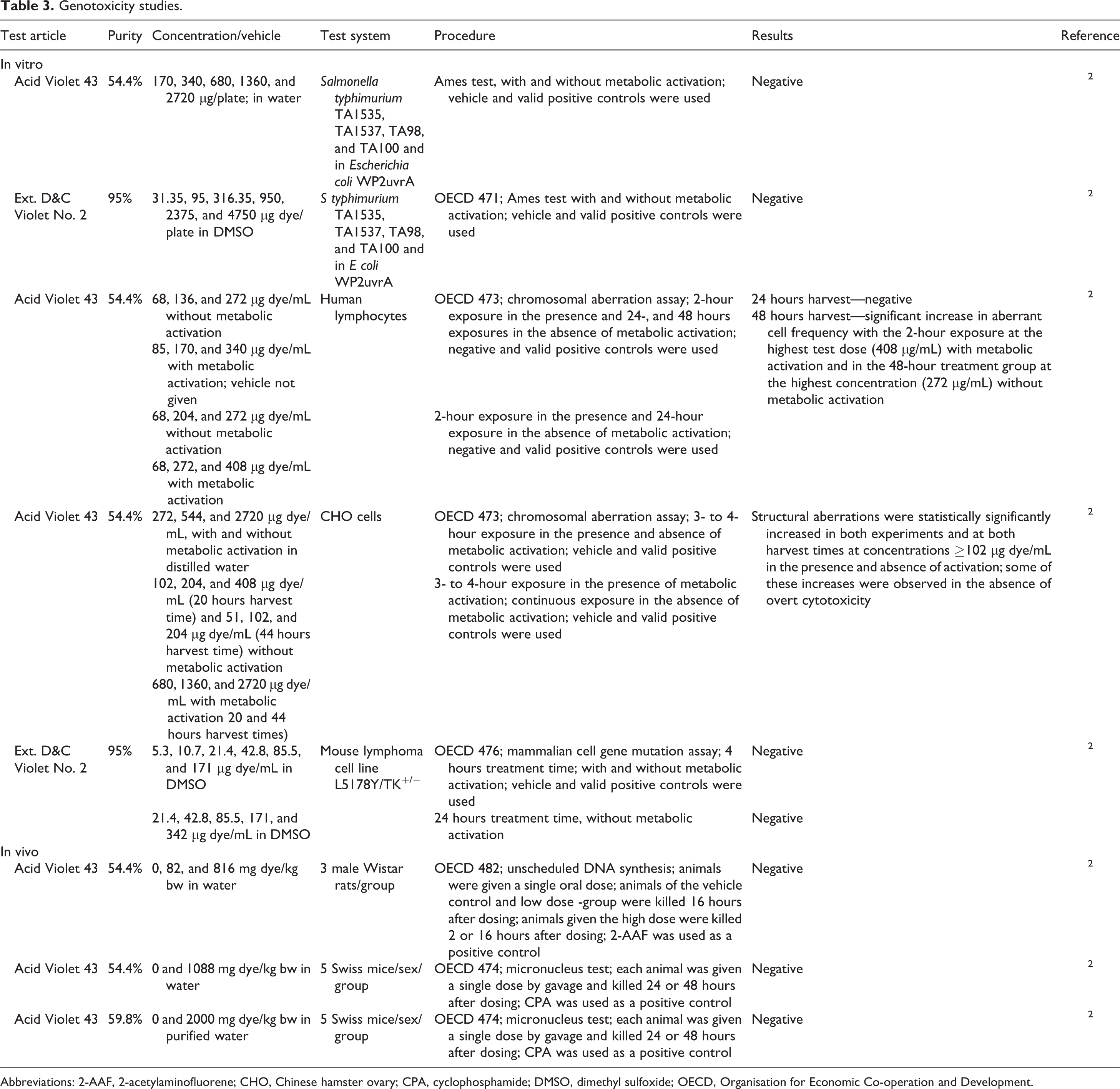
Abbreviations: 2-AAF, 2-acetylaminofluorene; CHO, Chinese hamster ovary; CPA, cyclophosphamide; DMSO, dimethyl sulfoxide; OECD, Organisation for Economic Co-operation and Development.
In Vivo
Acid Violet 43 (54.4% pure) was not genotoxic in an unscheduled DNA synthesis assay in which rats were given a single oral dose of up to 816 mg dye/kg bw in water. Acid Violet 43 (purity 54.4% or 58.8%) was not genotoxic in micronucleus tests in which mice were given a single oral dose of 1088 or 2000 mg dye/kg bw, respectively, in water.
Carcinogenicity
Dermal application of Ext. D&C Violet No. 2 was not carcinogenic to Swiss-Millerton mice. 1 The majority of the doses (>95) were applied as 1% dispersions in propylene glycol; a total of 103 applications were made.
Dermal Irritation and Sensitization Studies
Sensitization
Animal
The sensitization potential of Acid Violet 43 (60.5% pure) was evaluated in a local lymph node assay (LLNA) performed in accordance with the Organisation for Economic Co-operation and Development (OECD) test guideline 429. 2 During induction, 25 µL of the test compound was applied to each ear of female CBA/J mice (4 per group) at concentrations of 0.6, 1.5, 3, 6, and 15% dye (w/v) in acetone/olive oil (AOO, 4/1 [v/v]; % w/w of “pure” dye based on purity of 60.5%) once daily for 3 days. A vehicle control group and a positive control (25% hexylcinnamaldehyde in AOO) group were treated in the same manner. Lymphocyte proliferation was determined after a 2-day nontreatment period; no lymphocyte proliferation was observed at any test concentration. The highest stimulation index (SI) for the test article was 1.55 at the 15% test concentration. (An SI of ≥3 is regarded as positive.) No cutaneous reactions and no increases in ear thickness were observed in any of the animals treated with the test substance. However, a black coloration of the skin of the ears was noted in all treated animals from day 2 up to day 6; this coloration could have concealed possible erythema. The SCCS commented that the highest concentrations of Acid Violet 43 tested (15% of a material that was 60.5% pure—ie, 9.075%) were too low to exclude a sensitizing potential.
The potential for Ext. D&C Violet No. 2 to induce hypersensitivity was evaluated in a modified guinea pig testing protocol, the adjuvant, and patch test. 12 As supplied, the commercial-grade test substance contained 990 ppm 1,4-dihydroxy-9,10-anthracenedione (aka quinizarin); upon purification by precipitation from aqueous solution with ethyl alcohol, after pretreatment of the solution with activated carbon, the 1,4-dihydroxy-9,10-anthracenedione content was <1 ppm. Groups of 14 female Hartley albino guinea pigs were used. Test animals were sensitized with 0.2% 1,4-dihydroxy-9,10-anthracenedione; control groups were not sensitized. One-tenth milliliter of a water-in-oil emulsion of distilled water: Freund complete adjuvant (1:1) was injected intradermally into the 4 corners of a 2 cm × 4 cm shaved area of the animals. Grid-like scratches were made at the injection sites, and occlusive patches of 0.1 mL 1% commercial or purified Ext. D&C Violet No. 2 was applied to the injection sites for 24 hours. Abrasions and patch application was repeated on the following 2 days. One week after the initial applications, 10% sodium lauryl sulfate in petrolatum was applied to the intradermal injection sites and, on the following day, an occlusive patch of the test material was applied to the same sites for 48 hours. After a 2-week nontreatment period, a test sample was applied to a shaved area of the back of the animals; excess test material was removed after 24 hours by washing with acetone. The test sites were scored 1, 24, and 48 hours after washing. One animal of the sensitized group had a positive reaction to the commercial-grade substance. None of the animals patch tested with purified Ext. D&C Violet No. 2 and none of the control animals exposed to either grade of the test material showed any positive reactions.
Quantitative Structure–Activity Relationship
A quantitative structure–activity relationship (QSAR) model was utilized to predict the sensitization potential of all hair dye ingredients registered in Europe (229 substances as of 2004). 3 The model predicted Acid Violet 43 to be a strong/moderate sensitizer. The QSAR analysis involved calculating TOPological Substructural MOlecular DEsign descriptors and correlating them to unspecified sensitization data from LLNA that were available in July 2003.
Ocular Irritation Studies
One-tenth milliliter of 1% (dye) Acid Violet 43 (60.5% pure) in water was instilled into the conjunctival sac of the left eye of 3 male New Zealand White rabbits. 2 Slight chemosis was observed in 1 animal on days 1 and 2, and slight conjunctival erythema was observed in all 3 animals from day 1 up to day 2 or 3. A clear discharge was also noted in 1 animal on day 2. All reactions were reversible by day 4. It was not known if the “impurities’ in the dye contributed to the irritation.
Clinical Studies
Multicenter Studies
A total of 736 patients with eczema from 3 dermatology clinics were patch tested with 1% Acid Violet 43 (purity not specified) in petrolatum during a 6-month period in 2007 to 2008. 13 Twenty milligram of the test substance was applied to the upper back of each patient using 8-mm Finn chambers on Scanpor tape. The test sites were scored on days 2, 3, 4, and 7. None of the patients had a positive reaction to the patch test, and there were no irritant reactions. Doubtful reactions were noted in 70 (9.5%) patients; these reactions were all recorded at one test center that considered discoloration of the skin as a doubtful reaction.
Risk Assessment
Margin of Safety
The margin of safety (MOS) for the use of Ext. D&C Violet No. 2 (purity 94%) under nonoxidative conditions (maximum on head concentration of 0.5% dye) was calculated in accordance with OECD test guideline 408, using 50% bioavailability in accordance with the SCCS’s Notes of Guidance for the testing of cosmetic ingredients and their safety evaluation. 2
Absorption through the skin (A) = 0.92 µg/cm2
Skin Area surface (SAS) = 580 cm2
Dermal absorption per treatment (SAS × A × 0.001) = 0.534 mg
Typical body weight of human = 60 kg
Systemic exposure dose (SED; SAS × A × 0.001/60) = 0.009 mg/kg bw/d
NOAEL (from 90-day, oral, rat) = 282 mg/kg bw/d
50% bioavailability = 141 mg/kg bw/d
Epidemiological Studies
Acid Violet 43 is used in nonoxidative hair dyes. Although the safety of individual hair dye ingredients is not addressed in epidemiology studies that seek to determine links, if any, between hair dye use and disease, such studies do provide broad information. Currently available epidemiology studies provided insufficient evidence to support a causal association between personal hair dye use and a variety of tumors and cancers.
A detailed summary of the available hair dye epidemiology data is available at http://www.cir-safety.org/cir-findings.
Summary
In 2001, the Panel published a safety assessment with the conclusion that Acid Violet 43 is safe for use in hair dye formulations when free of impurities, except for the following: ≤18% volatile matter (at 135 °C) and chlorides and sulfates (calculated as sodium salts); ≤0.4% water-insoluble matter; ≤0.2% 1-hydroxy-9,10-anthracenedione; ≤0.2% 1,4-dihydroxy-9,10-anthracenedione; ≤0.1% p-toluidine; ≤0.2% p-toluidine sulfonic acids, sodium salts; ≤1% subsidiary colors, ≤20 ppm lead (as Pb); ≤3 ppm arsenic (as As); ≤1 ppm mercury as (Hg); and ≥80% total color. Acid Violet 43, an anthraquinone color, is a noncertified color; the certified colorant is identified as Ext. Violet 2 (aka Ext. D&C Violet 2).
Based on 2016 VCRP data, Acid Violet 43 is reported to be used in 48 hair coloring preparations at a maximum reported concentration of use of 0.35% (in hair tints). In the original safety assessment, Acid Violet 43 was reported to be used in only 2 hair coloring formulations in 1998; at that time, concentration of use data was not available. However, according to historical FDA data that were included in that report, in 1984, Acid Violet 43 was reported to be used in 30 coloring hair formulations at concentrations of ≤0.1% and in 1 coloring shampoo at a concentration of 0.1% to 1%. In Europe, the SCCS concluded that the use of Acid Violet 43 (as related to batch Ext. D&C Violet No. 2, 94% pure) as a nonoxidative hair dye with a maximum on head concentration of 0.5% dye does not pose a risk to the health of the consumer, but a sensitizing potential could not be excluded.
In a study examining the percutaneous absorption of Acid Violet 43 (59.3% pure) through human skin samples, the mean absorbed amount of Acid Violet 43 (estimated as the sum of amounts measured in epidermis, dermis, and receptor fluid when assuming concentrations at the detection limit in the receptor fluid of 40 ng) was 0.11 ± 0.06 µg dye/cm2 (0.53% ± 0.33% of the applied dose). In studies using porcine ear skin, the highest rates for skin absorption of Ext. D&C Violet No. 2 (95% pure) were estimated as 20.93 μg/cm2 (∼1%) and 30.55 μg/cm2 (∼0.6%) in 2 experiments.
In a toxicokinetic study of Acid Violet 43 (59.8% pure) that was performed as part of a bone marrow micronucleus test in mice, oral bioavailability was demonstrated by high blood levels of Acid Violet 43 observed in all animals.
In a subchronic toxicity study in which Sprague-Dawley rats were dosed by gavage once daily for 13 weeks with up to 800 mg/kg bw/d Acid Violet 43 (54.4% pure), the NOAEL was 200 mg/kg bw/d Acid Violet 43 (109 mg dye/kg bw/d), based on increased APTT in high-dose males; no clinical signs of toxicity were reported. The NOAEL in a 13-week study of Ext. D&C Violet No. 2 (93.8% pure) in rats following the same protocol, with a maximum dose of 1000 mg/kg bw/d, was 300 mg/kg bw/d Ext. D&C Violet No. 2 (282 mg dye/kg bw/d); again, the NOAEL was based on an increased in APTT in high-dose males.
Gravid female Sprague-Dawley rats were administered Acid Violet 43 (54.4% pure) in water by gavage at doses up to 800 mg/kg bw/d (ie, up to 435 mg dye/kg bw/d) on days 6 to 15 of gestation. No external soft-tissue or skeletal anomalies were attributed to treatment with the test substance, and the NOAEL was 800 mg/kg bw/d for teratogenicity and maternal toxicity. In a study in which female Wistar rats were administered 1% Ext. D&C Violet No. 2 (93.8% pure) in carboxymethylcellulose by gavage at doses up to 1000 mg/kg bw/d (ie, up to 940 mg dye/kg bw/d) on days 6 to 17 of gestation, the NOAEL for teratogenicity and maternal toxicity was 1000 mg/kg bw/d.
Acid Violet 43 (54.4% pure) and Ext. D&C Violet No. 2 (95% pure) were not mutagenic in the Ames test when tested at concentrations of ≥2720 µg/plate in water or ≥4750 µg dye/plate in DMSO, respectively. In chromosomal aberration assays of Acid Violet 43 (54.4% pure), significant increases in aberrant cell frequency were observed with a 2-hour exposure at the highest test dose (408 g/mL) with metabolic activation and in the 48-hour treatment group at the highest concentration (272 μg/mL) without metabolic activation in human lymphocytes, and structural aberrations were statistically significantly increased in CHO cells at concentrations ≥102 µg dye/mL. Ext. D&C Violet No. 2, at concentrations ≤342 µg dye/mL in DMSO, was not genotoxic in a mammalian cell mutation assay in mouse lymphoma cells. In in vivo studies, Acid Violet 43 (54.4% pure) was not genotoxic in an unscheduled DNA synthesis assay in which rats were given a single oral dose of up to 816 mg dye/kg bw in water, or in micronucleus tests in which mice were given a single oral dose of 1088 mg dye/kg bw (54.4% pure) or 2000 mg dye/kg bw in water.
Concentrations of up to 15% Acid Violet 43 (60.5% pure) dye (w/v) in AOO did not produce positive results in a mouse LLNA; however, the SCCS commented that the highest concentrations of Acid Violet 43 tested were too low to exclude a sensitizing potential. An adjuvant and patch test for hypersensitivity potential of commercial-grade and purified Ext. D&C Violet No. 2 (containing 990 ppm and <1 ppm 1,4-dihydroxy-9,10-anthracenedione, respectively) was conducted in female Hartley albino guinea pigs that were sensitized with 0.2% 1,4-dihydroxy-9,10-anthracenedione and in control (nonsensitized) guinea pigs. One animal of the sensitized group had a positive reaction to the commercial-grade substance. None of the animals patch tested with purified Ext. D&C Violet No. 2 and none of the control animals exposed to either grade of the test material showed any positive reactions. A QSAR model predicted Acid Violet 43 to be a strong/moderate sensitizer.
Instillation of 1% (dye) Acid Violet 43 (60.5% pure) into rabbit eyes resulted in slight chemosis in one-third of animals and slight conjunctival erythema in all animals. All reactions were reversible by day 4.
A total of 736 patients with eczema from 3 dermatology clinics were patch tested with 1% Acid Violet 43 (purity not specified) in petrolatum during a 6-month period. No positive reactions were noted.
The MOS for the use of Ext. D&C Violet No. 2 (94% purity) under nonoxidative conditions (maximum on head concentration of 0.5% dye), assuming 50% bioavailability, was calculated as 15667.
Discussion
In 2001, the Panel published a safety assessment of Acid Violet 43 (a noncertified colorant) that utilized safety test data on Ext. D&C Violet No. 2 (a certified color). Acid Violet 43 has the same structure as Ext. D&C Violet No. 2, but because it is not a certified color, it may contain impurities at different levels or that are not allowed in the certified color. In the 2001 assessment, the Panel concluded that Acid Violet 43 was safe for use in hair dye formulations when it met the impurity specifications indicated for the certified color.
In accordance with its CIR Procedures, the Panel evaluates the conclusions of previously issued reports every 15 years to determine whether or not the conclusion remains valid. In the time since the original safety assessment was published, test data on Acid Violet 43 have become available; the compounds tested were 54.4% to 60.5% pure. Because safety data are now available on Acid Violet 43 (the noncertified colorant), and because the results of the studies on Acid Violet 43 were similar to those obtained in studies of Ext. D&C Violet No. 2 (the certified color), the Panel reopened the report to remove the impurity specifications from the conclusion. The Panel did state that the composition of Acid Violet should be the same or similar to that identified for the test substance that was assessed in this report.
Amended Conclusion
The Panel concluded Acid Violet 43 is safe in the present practices of use and concentration for use in hair dye formulations.
Footnotes
Authors’ Note
Fiume, M. and Heldreth, B. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript; Bergfeld, W., Belisto, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to conception and design, analysis, and interpretation and critically revised manuscript; Gill, Lillian J. contributed to conception and design; contributed to analysis; and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
