Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of Phosphoric Acid and its salts (31 ingredients), which are reported to function as buffering agents, corrosion inhibitors, chelating agents, and pH adjusters in cosmetic products. The Panel reviewed data relating to the safety of these ingredients and concluded that Phosphoric Acid and its salts are safe in the present practices of use and concentration in cosmetics when formulated to be nonirritating.
Introduction
The safety of the following 31 ingredients, as used in cosmetics, is reviewed in this safety assessment:

According to the web based-International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), the functions of these ingredients in cosmetic products include buffering agents, corrosion inhibitors, chelating agents, and pH adjusters. 1
Three of the phosphate salt ingredients included in this safety assessment, that is, Sodium Metaphosphate, Sodium Trimetaphosphate, and Sodium Hexametaphosphate, have been previously reviewed by the Expert Panel for Cosmetic Ingredient Safety (Panel). 2 In 2001, the Panel concluded that these ingredients are safe for use in cosmetics when formulated to avoid skin irritation.
Chemistry
Definition and Structure
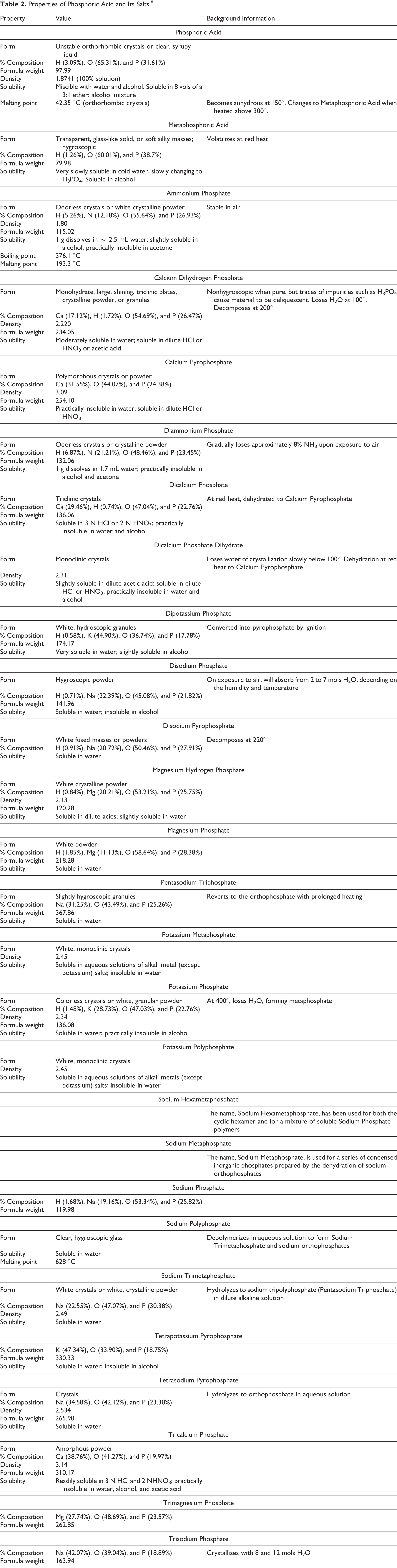
The definitions, structures, and functions in cosmetics of Phosphoric Acid and its salts are presented in Table 1.
Phosphoric Acid and its salts all share the same phosphate core in common. Except for Phosphoric Acid and Metaphosphoric Acid, the ingredients in this report are either alkaline earth metal (Periodic Table column I or II) salts or ammonium salts of a phosphoric acid. These ingredients are related to each other as inorganic phosphates, with varying cation identity and degree of protonation (Figure 1). This group comprises phosphate salts for which property differences are attributable primarily to having different cation(s). Characterizing these differences in one report that addresses all of these ingredients is more informative than attempting to assess the safety of these salts in separate reports that each addresses only one ingredient.
Phosphoric Acid is a polyprotic acid which is deprotonated to mono-, di-, and triphosphates with rising pH.
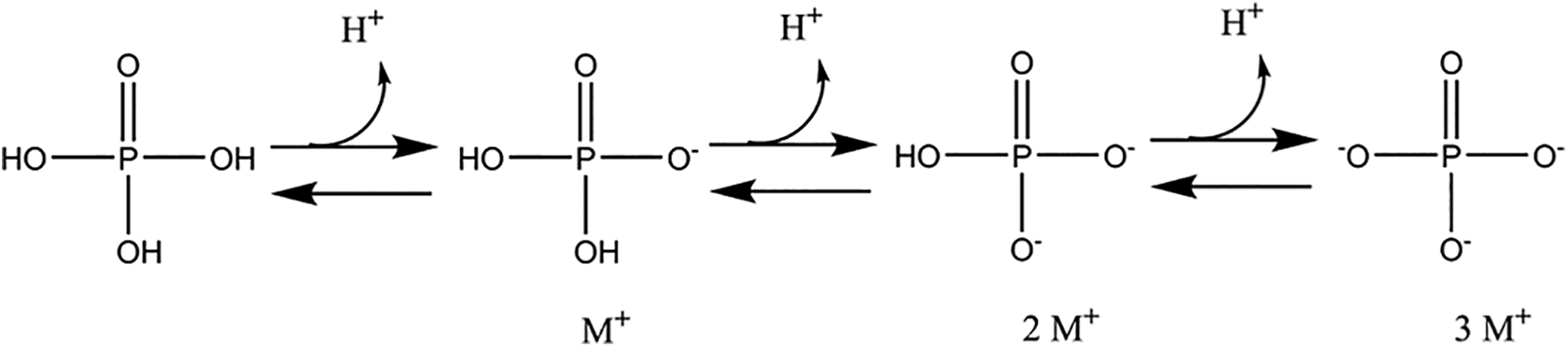
Phosphoric Acid and the orthophosphates (dihydrogen phosphate, hydrogen phosphate, and phosphate).
However, Phosphoric Acid and phosphate salts also exist as dimers and trimers of phosphate, pyro- and meta- respectively. Accordingly, these ingredients vary by the identity of associated cations, degree of protonation, and in the number of phosphate repeat units (ie, 1 repeat is ortho-, 2 repeats is pyro-, and 3 repeats is meta-; Figure 2).
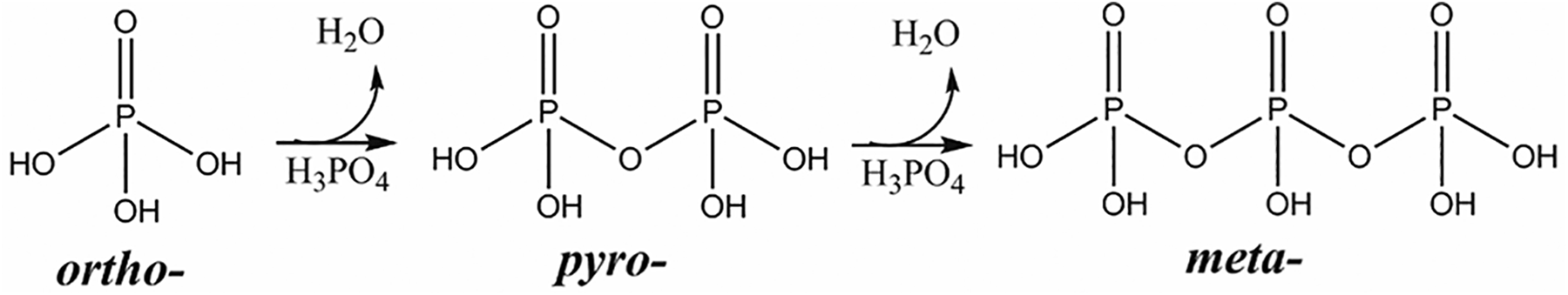
Dehydration of phosphoric acids, from ortho- to pyro- to meta-phosphoric acid.
As some of the Dictionary names for these ingredients vary from the customary names and may be confusing, systematic or common names have been added to Table 1. However, elsewhere in this report only the Dictionary ingredient name is used.
Chemical and Physical Properties
These ingredients range from colorless crystalline solids to white amorphous powders. The water solubilities of these ingredients are pH dependent (Table 2).
Properties of Phosphoric Acid and Its Salts. 6
Method of Manufacture
Acids
Phosphoric Acid
Phosphoric Acid is manufactured by the wet process or the furnace (thermal) process. In the wet process, Phosphoric Acid is produced directly from phosphate ores and is said to be of low purity. 3 This process is used mostly for the production of fertilizers. In the thermal or furnace process, phosphoric acid is produced from elemental phosphorus. This process is used in the production of phosphoric acid for uses other than fertilizer production, such as metal treatment, refractories, catalysts, and use in food and beverages.
Ammonium Salts
Ammonium Phosphate
In the process for manufacturing Ammonium Phosphate, a one-to-one ratio of ammonia (NH3) and Phosphoric Acid (H3PO4) is reacted, and the resulting slurry of Ammonium Phosphate is solidified in a granulator. 4
Diammonium Phosphate
In the manufacture of Diammonium Phosphate, each stoichiometric equivalent of Phosphoric Acid is neutralized by approximately 2 equivalents of ammonia. 5
Sodium Salts
Disodium Phosphate
Disodium Phosphate is prepared by the ignition of Dicalcium Phosphate. 6
Sodium Metaphosphate
Sodium Metaphosphate is prepared by dehydration of sodium orthophosphates. 6
Sodium Polyphosphate
Sodium phosphate monobasic hydrate was used to prepare Sodium Polyphosphate with a degree of polymerization (Dp) lower than ≈ 500. 7 Sodium phosphate monobasic hydrate was heated to 700 °C for 1, 3, or 9 hours, and the melt was then quenched on a copper plate. To fraction the Sodium Polyphosphate glass, the frit was ground and dissolved in deionized water to yield a 10% (w/v) Sodium Polyphosphate solution. The solution was stirred, fractioned by serial dilution with acetone, and then centrifuged to collect the precipitate. Sodium Polyphosphate with a Dp > 500 was obtained from an ion-exchange process on a potassium polyphosphate crystalline phase.
Tetrasodium Pyrophosphate
Tetrasodium Pyrophosphate is produced by molecular dehydration of dibasic Sodium Phosphate at 500 °C. 6
Pentasodium Triphosphate
Pentasodium Triphosphate is prepared by the molecular dehydration of mono- and disodium phosphates. 6
Potassium Salts
Potassium Metaphosphate
Potassium Metaphosphate is obtained by the fusion of monopotassium phosphates. 8 It is also prepared by dehydration of Potassium Phosphate. 6
Potassium Phosphate
Food-grade potassium phosphates have been prepared by the neutralization of Phosphoric Acid with potassium hydroxide at 50 °C to 60 °C. 9
Potassium Polyphosphate
Potassium Polyphosphate can be obtained by heating monopotassium orthophosphate to any temperature above 150 °C. 10
Calcium Salts
Calcium Pyrophosphate
Calcium Pyrophosphate can be obtained by a solid state reaction (870 °C and normal atmosphere) from a mixture of Tricalcium Phosphate and Phosphoric Acid. 11 It can also be prepared by ignition of Dicalcium Phosphate. 6
Dicalcium Phosphate
Commercial Dicalcium Phosphate is not a chemically discrete entity but is a mixture of varying amounts of dicalcium and monocalcium phosphates, Phosphoric Acid, calcium carbonate, and impurities, depending on the origin of the raw material and procedures employed in its industrial production. 12
Tricalcium Phosphate
Tricalcium Phosphate has been produced by a calcination process (at high temperatures of 1500 °C-1600 °C) that is preceded by the grinding and mixing of phosphate rock and sodium carbonate and the addition of Phosphoric Acid to the reaction mixture. 13
Magnesium Salts
Magnesium Phosphate
Magnesium Phosphates have been prepared by adding a magnesium nitrate solution into mixed solutions of potassium hydroxide and Phosphoric Acid at temperatures of 29 °C to 95 °C. 14
Composition/Impurities
Phosphoric Acid
According to the Food Chemicals Codex specification for this chemical, the following limits for inorganic impurities in Phosphoric Acid have been established: arsenic (≤3 mg/kg), cadmium (≤3 mg/kg), fluoride (≤10 mg/kg), and lead (≤3 mg/kg). 15
Ammonium Salts
Ammonium Phosphate
According to the Food Chemicals Codex specification for Ammonium Phosphate, the acceptance criteria for this chemical indicate that it contains not less than 96% Ammonium Phosphate and not more than 100% Ammonium Phosphate. The following limits for inorganic impurities in Ammonium Phosphate have been established: arsenic (≤3 mg/kg), fluoride (≤10 mg/kg), and lead (≤4 mg/kg). 15 According to another source, iron and aluminum have been mentioned as Ammonium Phosphate impurities. 4
Diammonium Phosphate
According to the Food Chemicals Codex specification for Diammonium Phosphate, the acceptance criteria for this chemical indicate that it contains not less than 96% Diammonium Phosphate and not more than 100% Diammonium Phosphate. The following limits for inorganic impurities in Diammonium Phosphate have been established: arsenic (≤3 mg/kg), fluoride (≤10 mg/kg), and lead (≤ 4 mg/kg). 15
Sodium Salts
Sodium Hexametaphosphate
Sodium Hexametaphosphate contains 10 to 12 repeating pyrophosphate subunits. 16
Sodium Phosphate
According to the Food Chemicals Codex specification for Sodium Phosphate, the acceptance criteria for this chemical are not less than 98% Sodium Phosphate and not more than 103% Sodium Phosphate on the dried basis, and the following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤0.005%), and lead (≤4 mg/kg). 15
Sodium Polyphosphate
According to the Food Chemicals Codex specification for Sodium Polyphosphate, the acceptance criteria for phosphorus pentoxide content range from 60% to 71%, and the following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤0.005%), and lead (≤4 mg/kg). 15
Trisodium Phosphate
According to the Food Chemicals Codex specification for Trisodium Phosphate, the acceptance criteria for this chemical are not less than 97% Trisodium Phosphate (anhydrous and monohydrate forms), calculated on the ignited basis, and not less than 90% Trisodium Phosphate (dodecahydrate), calculated on the ignited basis. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤0.005%), and lead (≤4 mg/kg). 15
Potassium Salts
Dipotassium Phosphate
According to the Food Chemicals Codex specification for Dipotassium Phosphate, the acceptance criteria for this chemical indicate that it contains not less than 98% Dipotassium Phosphate, on the dried basis. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤10 mg/kg), and lead (≤2 mg/kg). 15 According to another source, heavy metal (as lead, 0.0006%) and arsenic (0.00005%) impurities have been reported for Dipotassium Phosphate. 17
Potassium Metaphosphate
According to the Food Chemicals Codex specification for Potassium Metaphosphate, the acceptance criteria for this chemical are not less than 59% phosphorus pentoxide and not more than 61% phosphorus pentoxide. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤10 mg/kg), and lead (≤2 mg/kg). 15
Potassium Phosphate
According to the Food Chemicals Codex specification for Potassium Phosphate, the acceptance criteria for this chemical indicate that it contains not less than 98% Potassium Phosphate, on the dried basis. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤10 mg/kg), and lead (≤2 mg/kg). 15
Potassium Pyrophosphate
According to the Food Chemicals Codex specification for Potassium Pyrophosphate, the acceptance criteria for this chemical indicate that it contains not less than 95% Potassium Pyrophosphate. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤10 mg/kg), and lead (≤2 mg/kg). 15
Calcium Salts
Calcium Dihydrogen Phosphate
According to another source, Calcium Dihydrogen Phosphate may contain a trace amount of Phosphoric Acid as an impurity. 6
Calcium Phosphate
Calcium Phosphate is approximately 96% pure, usually containing an excess of calcium oxide. 6
Dicalcium Phosphate
Commercial Dicalcium Phosphate is not a chemically discrete entity but is a mixture of varying amounts of dicalcium and monocalcium phosphates, Phosphoric Acid, calcium carbonate, and impurities, depending on the origin of the raw material and procedures employed in its industrial production. 12
According to the Food Chemicals Codex specification for Dicalcium Phosphate, the acceptance criteria for this chemical indicate that it contains not less than 97% Dicalcium Phosphate and not more than 105% Dicalcium Phosphate (anhydrous or dehydrate form). The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤0.005%), and lead (≤2 mg/kg). 15
Tricalcium Phosphate
According to the Food Chemicals Codex specification for Tricalcium Phosphate, the acceptance criteria for this chemical indicate that it contains not less than 34% calcium and not more than 40% calcium. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤0.0075%), and lead (≤2 mg/kg). 15
Magnesium Salts
Magnesium Hydrogen Phosphate
According to the Food Chemicals Codex specification for Magnesium Hydrogen Phosphate, the acceptance criteria for this chemical indicate that it is not less than 96% pure, on the ignited basis. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤25 mg/kg), and lead (≤2 mg/kg). 15
Trimagnesium Phosphate
According to the Food Chemicals Codex specification for Trimagnesium Phosphate, the acceptance criteria for this chemical indicate that it is not less than 98% pure. The following limits for inorganic impurities have been established: arsenic (≤3 mg/kg), fluoride (≤25 mg/kg), and lead (≤2 mg/kg). 15
Use
Cosmetic
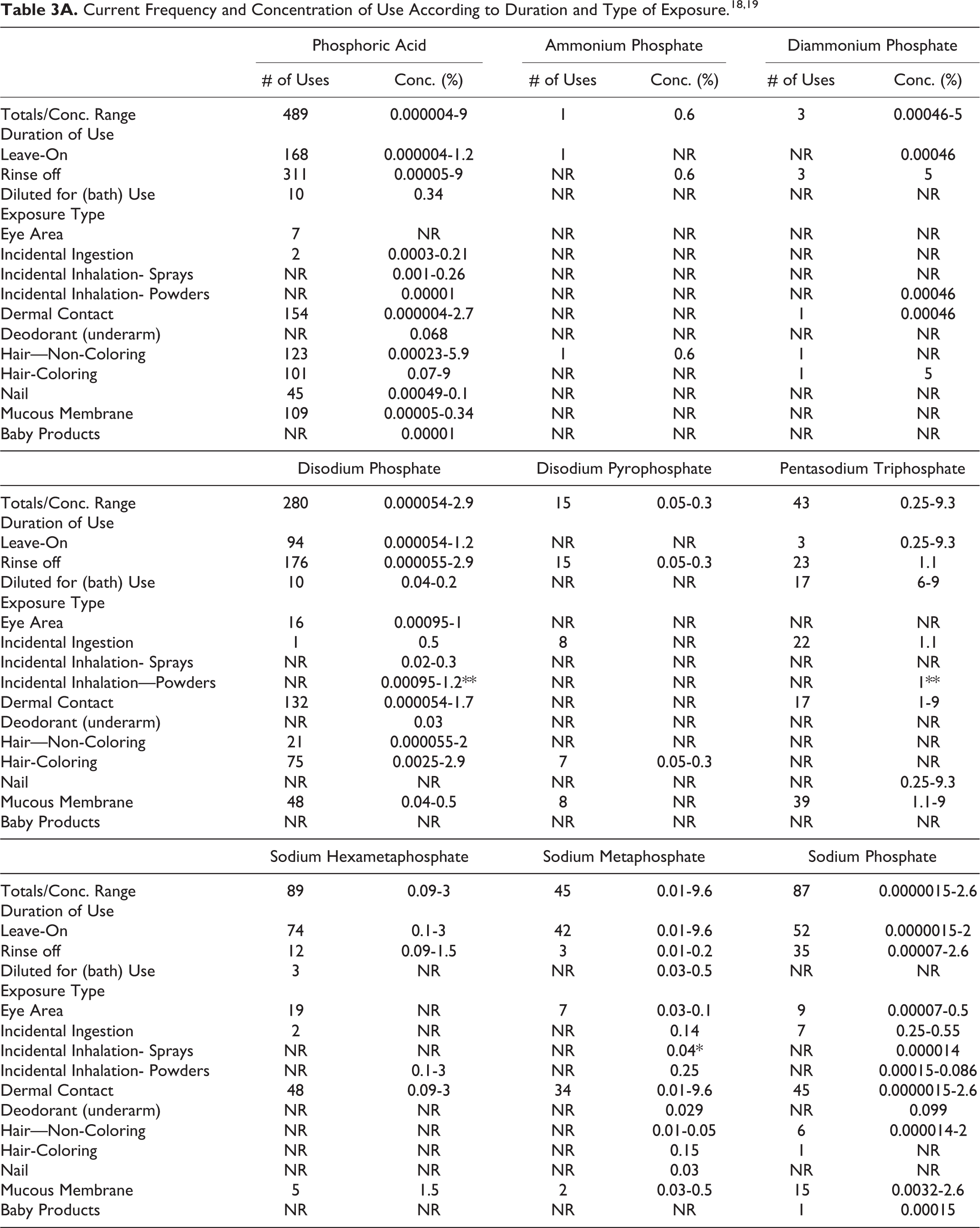
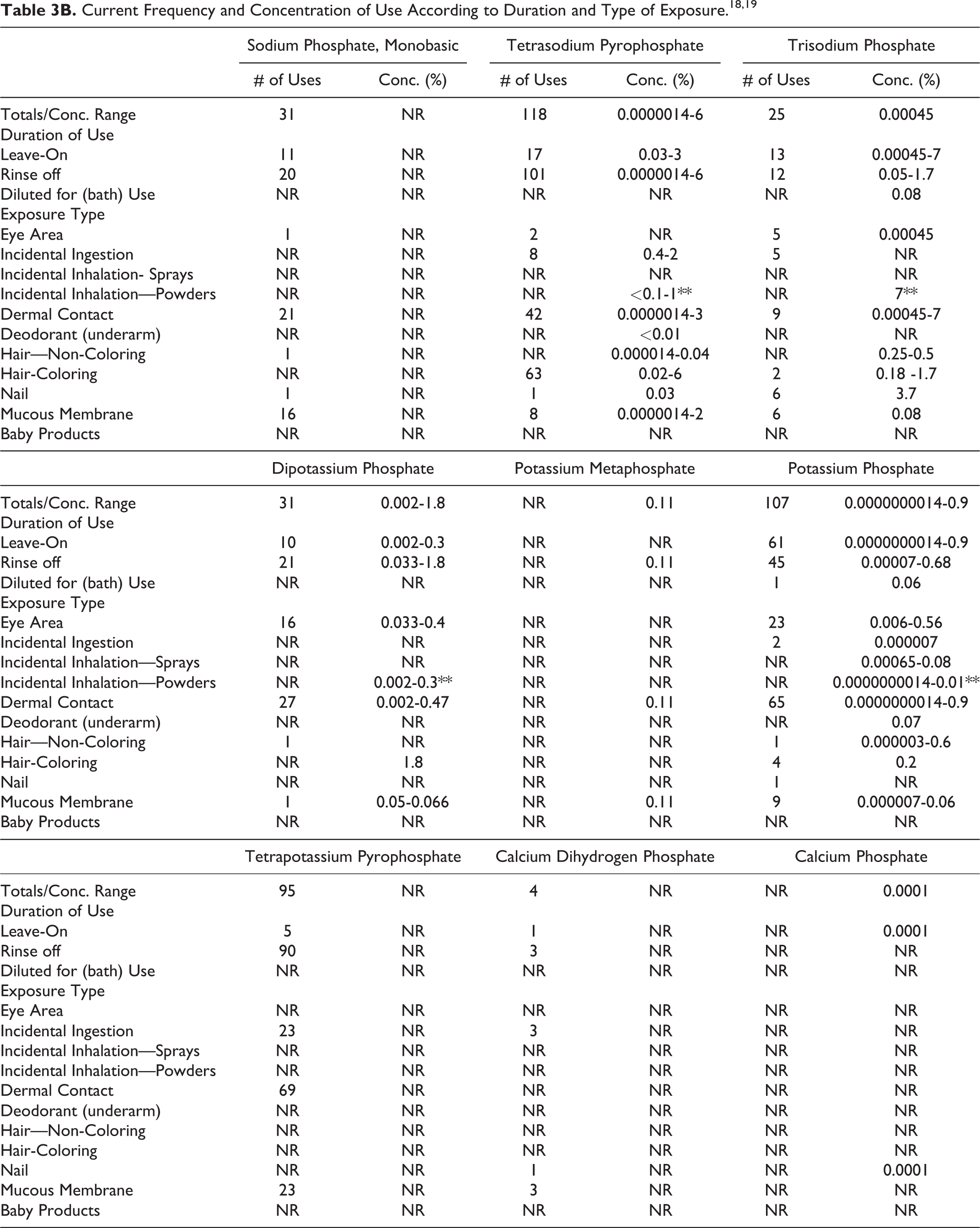
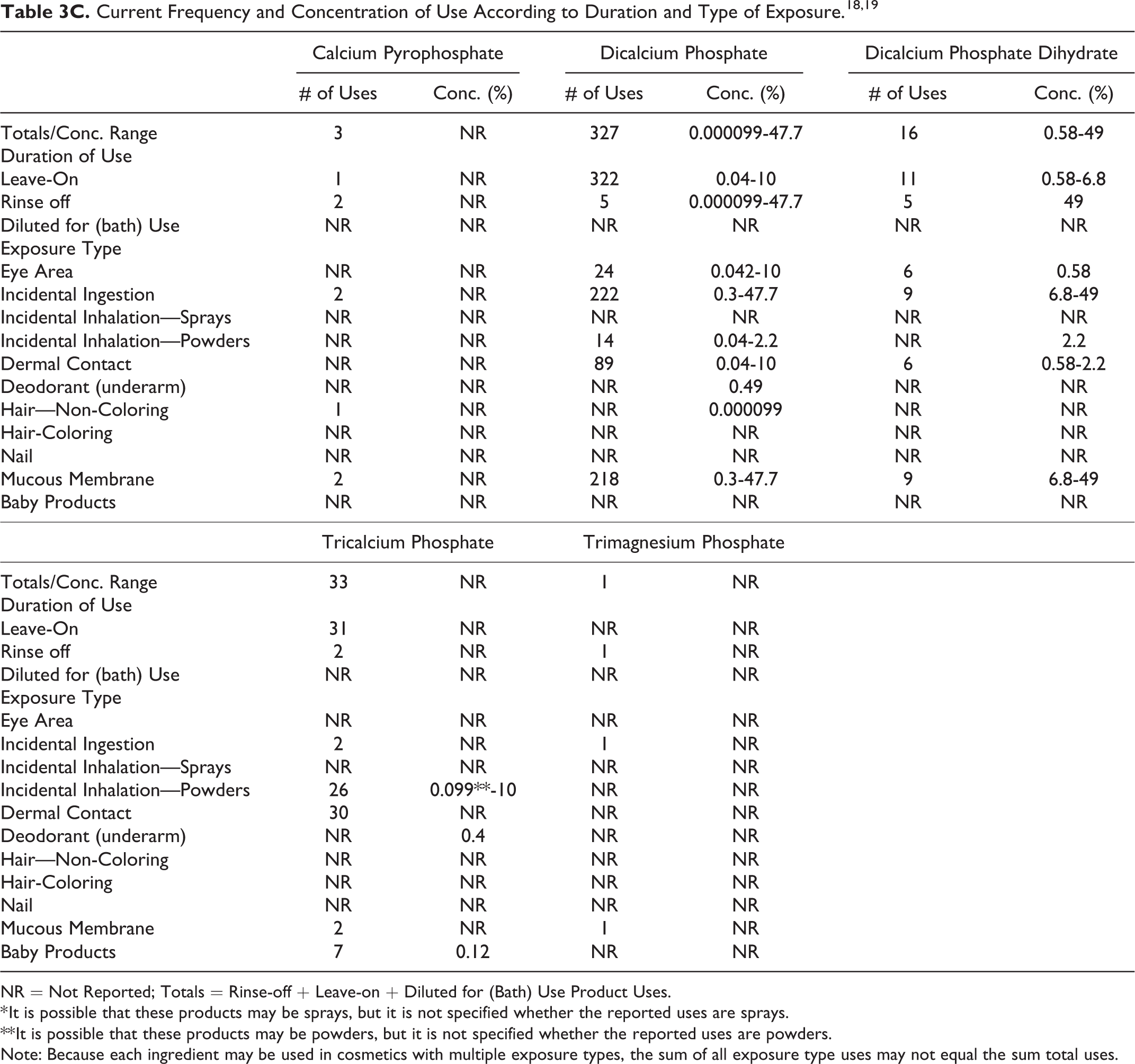
The safety of Phosphoric Acid and its salts included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys conducted by the Personal Care Products Council (Council) of maximum reported use concentrations, by product category. Collectively, the use frequency and use concentration data indicate that 22 of the 31 ingredients in this safety assessment are currently being used in cosmetic products (see Tables 3A-C). According to these data, the following 9 ingredients are not reported as being used in cosmetics:
NR = Not Reported; Totals = Rinse-off + Leave-on + Diluted for (Bath) Use Product Uses.
* It is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
** It is possible that these products may be powders, but it is not specified whether the reported uses are powders.
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.

According to 2016 VCRP data, the greatest reported use frequency is for Phosphoric Acid (489 formulations, mostly rinse-off products), followed by Dicalcium Phosphate (327 formulations, mostly leave-on products; Tables 3A-C). 18 The results of a concentration of use survey provided in 2015 indicate that Dicalcium Phosphate Dihydrate has the highest maximum concentration of use; it is used at concentrations up to 49% in rinse-off products (dentifrices; Tables 3A-C). 19
The highest maximum ingredient use concentration in leave-on products (10% in eye shadow) is being reported for Dicalcium Phosphate. In some cases, reported uses appear in the VCRP database, but concentrations of use data were not provided; the opposite is also true. For example, according to the VCRP, Tetrapotassium Pyrophosphate and Calcium Pyrophosphate are being used in 95 and 3 cosmetic products, respectively; however, use concentration data on these ingredients were not provided in the concentration of use survey. Furthermore, use concentration data on Calcium Phosphate were provided in the concentration of use survey; however, use frequency data were not reported in the VCRP data.
Cosmetic products containing Phosphoric Acid or its salts may be applied to the skin and hair or, incidentally, may come in contact with the eyes (eg, Dicalcium Phosphate at maximum use concentrations up to 10% in eye area cosmetics) and mucous membranes (eg, Dicalcium Phosphate Dihydrate at maximum use concentrations up to 49% in dentifrices). Additionally, some of these ingredients are being used in products that may result in incidental ingestion. For example, Dicalcium Phosphate Dihydrate is being used in dentifrices at maximum use concentrations up to 49%, and Dicalcium Phosphate is being used in lipstick at maximum use concentrations up to 10%. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Phosphoric Acid is used in aerosol hair sprays at concentrations of <0.01% and in pump hair sprays at concentrations up to 0.26%. The following other ingredients are also used in hair sprays: Potassium Phosphate (pump hair sprays up to 0.09%) and Sodium Phosphate (pump hair sprays up to 0.000014%). The following ingredients are used in face powders: Dicalcium Phosphate (up to 2.2%), Diammonium Phosphate (up to 0.00046%), Dicalcium Phosphate Dihydrate (up to 2.2%), Sodium Metaphosphate (up to 0.25%), and Sodium Phosphate (up to 0.086%). In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays.20-23 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.20,21 Additionally, Phosphoric Acid is used in dusting and talcum powders at concentrations up to 0.00001%, and Tricalcium Phosphate is used in dusting and talcum powders at concentrations up to 10%. Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.24-26
Non-Cosmetic
Phosphoric Acid and Phosphates
The FDA has determined that the following 20 ingredients included in this report are direct food additives that are generally recognized as safe (GRAS): 27

Additionally, the FDA has determined that potassium polymetaphosphate, chemically similar to one or more ingredients on the preceding list, is a GRAS direct food additive.
Acids
Phosphoric Acid
Phosphoric Acid is used in the manufacture of the following: phosphate salts, superphosphate fertilizers, detergents, activated carbon, animal feed, ceramics, dental cement, pharmaceuticals, soft drinks, gelatin, rust inhibitors, wax, and rubber latex. 3 Use in the following other processes has also been reported: electropolishing, engraving, photoengraving, lithograving, metal cleaning, sugar refining, and water treatment.
Metaphosphoric Acid
In dentistry, Metaphosphoric Acid is used to make zinc oxyphosphate cement. 6 It is also used as a reagent in chemical analysis.
Ammonium Salts
Ammonium Phosphate
In agriculture, Ammonium Phosphate has been an important granular fertilizer for many years. 4 Ammonium Phosphate is also used in dry chemical fire extinguishers, which are commonly found in offices, schools, and homes. The extinguisher spray disperses finely powdered Ammonium Phosphate, which coats the fuel and rapidly smothers the flame.
Diammonium Phosphate
Diammonium Phosphate is a complex fertilizer that contains 2 major nutrients, nitrogen and phosphorus. 28 Additionally, Diammonium Phosphate is used in fireproofing textiles, paper, wood, vegetable fibers, and dentifrices. 6
Sodium Salts
Disodium Phosphate
Disodium Phosphate is used as an emulsifier and buffer in foods, and in the manufacture of enamels, ceramics, detergents, and boiler compounds. 6
Disodium Pyrophosphate
Disodium Pyrophosphate is used chiefly in baking powders. 6
Pentasodium Triphosphate
Pentasodium Triphosphate is used as a preservative, sequestrant, and texturizer in foods, and as whitening agent in toothpaste; it is also used in water softeners and detergents. 6
Sodium Hexametaphosphate
Sodium Hexametaphosphate is an antitartar ingredient in toothpaste and is known to remove stains. 16
Sodium Phosphate
Sodium phosphate products have been used for bowel cleansing prior to medical procedures such as colonoscopy. The FDA is aware of reports of acute phosphate nephropathy that are associated with such usages. 29 Acute phosphate nephropathy is a form of acute kidney injury that is associated with deposits of calcium phosphate crystals in the renal tubules, which may result in permanent renal function impairment. In response, FDA requires that the manufacturer of 2 oral sodium phosphate products (prescription only) for bowel cleansing add a Boxed Warning to the labeling for these products. The FDA has also stated that, in light of the risk of acute phosphate nephropathy, over-the-counter (OTC) laxative oral sodium phosphate products should not be used for bowel cleansing.
Sodium Phosphate is also used in baking powders and as dry acidulant and sequestrant for foods. 6
Sodium Polyphosphate, Sodium Trimetaphosphate, and Tetrasodium Pyrophosphate
Blended phosphates (usually ortho and glassy polyphosphates) are used in municipal water treatment as part of scale-control and corrosion-control programs in the United States, because these compounds bind calcium carbonate, iron, magnesium, and manganese. 30 Sodium Polyphosphate, Sodium Trimetaphosphate, and Tetrasodium Pyrophosphate are some of the chemicals that are found in the phosphate blends. Sodium Trimetaphosphate is also used in detergent processing and as a crosslinking agent for starch in foods and pharmaceuticals. 6
Tetrasodium Pyrophosphate is also used in processed meat products, as an emulsifier in cheese, and as a color preservative in soybean paste. 31 Other uses include: sequestrant, dispersant, deflocculant, detergent builder, and component of solid or liquid fertilizers. 32 Tetrasodium Pyrophosphate is one of the anticalculus components of most tartar control dentifrices that are marketed. 33
The US Environmental Protection Agency (EPA) has established an exemption from the requirement of a tolerance for residues of Tetrasodium Pyrophosphate when used as an inert ingredient in pesticide formulations applied to growing crops only. 34
Trisodium Phosphate
Trisodium Phosphate is used in photographic developers, in detergent mixtures, and in the manufacture of paper. 6
Potassium Salts
Dipotassium Phosphate
Dipotassium Phosphate is used as a buffering agent in antifreeze, nutrient in the culturing of antibiotics, ingredient of instant fertilizers, and as a sequestrant in the preparation of nondairy powdered coffee creams. 6
Potassium Phosphate
Potassium Phosphate is used as a buffering agent in pharmaceuticals. 6
Tetrapotassium Pyrophosphate
Blended phosphates (usually ortho and glassy polyphosphates) are used in municipal water treatment as part of scale-control and corrosion-control programs in the United States, because these compounds bind calcium carbonate, iron, magnesium, and manganese. 30 Sodium Polyphosphate, Sodium Trimetaphosphate, and Tetrasodium Pyrophosphate are some of the chemicals that are found in the phosphate blends. Sodium Trimetaphosphate is also used in detergent processing and as a crosslinking agent for starch in foods and pharmaceuticals. 6
Calcium Salts
Calcium Phosphate
Calcium Phosphate has been used as an adjuvant (ie, a material that can increase the humoral or cellular immune response to an antigen) for simultaneous immunizations with diphtheria, tetanus, polio, Bacillus Calmette-Guérin, yellow fever, measles and hepatitis B vaccines, with hepatitis B and DTP-polio vaccines, and immunization with allergens. 35 It has also been used in the manufacture of fertilizers, Phosphoric Acid, P compounds, milk-glass, polishing and dental powders, porcelains, and pottery. 6
Calcium Phosphate is an active ingredient in antacid OTC drug products that are GRAS and effective. 36
Calcium Pyrophosphate
One form of Calcium Pyrophosphate has been used clinically as a bone-graft extender, because it bonds with host bone. 37 It is also used in dentifrices and in the production of ceramic ware and glass. 6
Dicalcium Phosphate
Dicalcium Phosphate is used chiefly in animal feeds and is also used as a mineral supplement in cereals and other foods. 6
Dicalcium Phosphate Dihydrate
Dicalcium Phosphate Dihydrate is a cleaning and polishing agent that is specifically used in dentifrices that contain monofluorophosphate. 38 As an abrasive, this ingredient assists in the removal of dental stains and deposits that form on tooth surfaces.
The FDA has determined that there are inadequate data to establish general recognition of the safety and effectiveness of Dicalcium Phosphate Dihydrate as an active ingredient in anticaries OTC drug products. 36
Tricalcium Phosphate
Tricalcium Phosphate, described as a porous ceramic material, is used in bone transplantation surgery. 39 It acts as a scaffold for bone ingrowth, undergoing progressive degradation, and replacement by bone. Most often, it is used in granule or powder form during surgery.
Tricalcium Phosphate is an active ingredient in antacid OTC drug products, and FDA has established a maximum daily dosage limit of 24 grams for Tricalcium Phosphate in these products. 40
Magnesium Salts
Magnesium Hydrogen Phosphate and Trimagnesium Phosphate
The FDA has determined that Magnesium Hydrogen Phosphate and Trimagnesium Phosphate are GRAS as a direct human food ingredients. 41
Toxicokinetics
Phosphorus (as phosphate) is an essential constituent of all known protoplasms, and its content is uniform across most plant and animal tissues. 42 According to the 1994 US Department of Agriculture survey of food intake of individuals, values for the mean daily phosphorus intake from food were 1495 mg (males, ≥ 9 years) and 1024 mg (females, ≥ 9 years). In both sexes, intakes decreased at age ≥ 51 years.
Structurally, phosphorus occurs as phospholipids, which constitute a major component of most biological membranes, and as components as nucleotides and nucleic acids. The total phosphorus concentration in whole blood is 13 mmol/L (40 mg/dL), most of which is in the phospholipids of red blood cells and plasma lipoproteins. Approximately 1 mmol/L (3.1 mg/dL) is present as inorganic phosphate (P i ), which is a tiny fraction of body phosphorus (<0.1%). In adults, P i makes up approximately 15 mmol (465 mg) of body phosphorus and is located mainly in the blood and extracellular fluid. Phosphate enters the P i pool during absorption from the diet and resorption from bone and is the primary source from which cells of all tissues derive both structural and high-energy phosphate. 42 Furthermore, most of the urinary phosphorus and hydroxyapatite mineral phosphorus are derived from the P i .
Phosphates are absorbed from the gastrointestinal tract, and the transport of phosphate from the lumen is an active, energy-dependent process; vitamin D stimulates phosphate absorption. 43 At physiologic pH (7.4), extracellular phosphate is present primarily as the Disodium Phosphate and Sodium Phosphate (4:1). Once absorbed, phosphate combines with calcium to form Dicalcium Phosphate in bones and teeth. 30 Free orthophosphate is the primary form by which dietary P i is absorbed. When phosphate ion is ingested in very large amounts, most of the phosphate ion uptake from the gut is eliminated in the feces. 44 According to another source, approximately two-thirds of the ingested phosphate is absorbed from the gastrointestinal tract in adults, and absorbed phosphate is almost entirely excreted in the urine. 43
Animal
Phosphoric Acid
Phosphoric Acid dissociates and is then absorbed as phosphate and hydronium ions through mucous membranes. 45
Sodium Salts
Sodium Hexametaphosphate
Sodium Hexametaphosphate is converted to Sodium Phosphate in the stomach. 46
After hexametaphosphate was administered to rats and rabbits by stomach tube, no more than trace amounts of labile phosphate were found in the urine.8,47
Sodium Polyphosphate
Ingested polyphosphates are degraded by phosphatase enzymes to monophosphates. 30 The short- and long-chain polyphosphates are absorbed intact only to a very limited extent, if at all, and the larger molecules are hydrolyzed by phosphatases (present in the gut) to monophosphates. 48
In an animal study (number and species not stated), 10% to 30% of administered Sodium Polyphosphate was absorbed as monophosphate, and small amounts of oligophosphates were found in the urine. 8 In another experiment in which labeled Sodium Polyphosphate was administered to rats, the chemical was not absorbed as such, but was taken up, after hydrolysis, as monophosphate and diphosphate. In 18 hours, 40% of the dose was hydrolyzed and absorbed.8,49
Potassium Salts
Potassium Metaphosphate
In an animal study (species and number not stated), 10% to 30% of administered Potassium Metaphosphate was absorbed as monophosphate, and small amounts of oligophosphates were found in the urine. 50 Study details were not provided.
When radiolabeled (radiolabel not specified), Potassium Metaphosphate was administered orally to rats, approximately half of the radioactivity was recovered from the feces, mainly as polymeric phosphate. Only a small percentage of the dose was found in the urine, in the form of monophosphate. 50
Human
Sodium Salts
Sodium Phosphate
In a pharmacokinetic analysis, 45 mL of a laxative containing 30 g of Sodium Phosphate was administered to 13 normal volunteers.51-53 The subjects were divided into the following 2 groups: group 1 (median weight = 60 kg) and group 2 (median weight = 119.2 kg). Serum and urine electrolytes were measured for 12 hours. Hydration was maintained by monitoring the weight, fluid intake, and total body water. Markedly elevated serum phosphate levels were observed in group 1 compared to group 2. The normalized area under the phosphate versus time curve was much higher in group 1 (1120 ± 190 mg/dL·min) than in group 2 (685 ± 136 mg/dL· min); P < 0.001was reported for this comparison. The urinary excretion of calcium was significantly lower in group 1 (mean = 16.4 ± 7.6 mg), compared to group 2 (mean = 39.2 ± 7.8 mg); P < 0.001 was reported for this comparison. The results of this study demonstrated that lower body-weight individuals develop prolonged high serum phosphate levels after ingesting Sodium Phosphate. The authors noted that individuals of lower body weight are at risk for acute phosphate nephropathy when they use colonoscopy preparations containing Sodium Phosphate.
Calcium Salts
Tricalcium Phosphate
The absorption of ingested Tricalcium Phosphate was evaluated in 10 women. The subjects ingested Tricalcium Phosphate (1200 mg) after fasting for 12 hours.54,55 Calcium and phosphorus absorption were determined by the post-load rise in urinary calcium and phosphate, respectively, above baseline. A statistically significant increase in urinary calcium excretion (P < 0.001) was observed during the 2 to 4 hours post-load period, and a statistically significant increase in serum calcium (P < 0.02) was observed at 4 hours post-load. Statistically significant increases in urinary phosphate excretion (P < 0.001) and serum phosphorus (P < 0.001) were also reported.
Toxicology
Calcium Phosphate
The English abstract of a Japanese publication on the safety of a Calcium Phosphate bone paste was available. 56 The following series of tests was performed: acute toxicity, pyrogenicity, hemolysis, intracutaneous reactivity, sensitization, genotoxicity, and cytotoxicity. The authors noted that there was no evidence of abnormal or toxic effects in any of these tests. The abstract does not include pertinent details relating to study results.
Single Dose (Acute) Toxicity
Animal
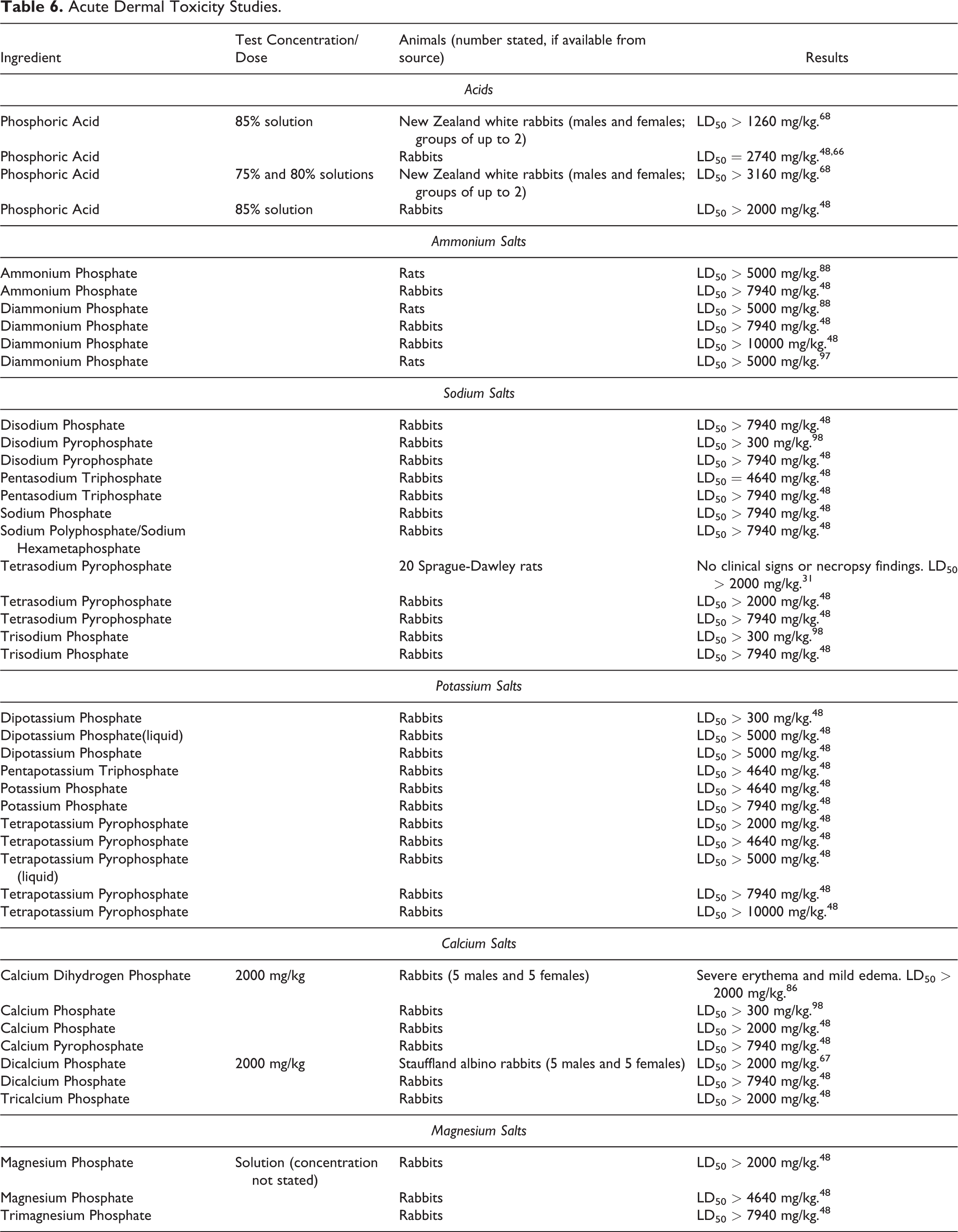
Dermal
Phosphoric Acid and Salts: Results of acute dermal studies for Phosphoric Acid and its salts are presented in Table 6. In studies involving rabbits, an LD50 of 2740 mg/kg and an LD50 > 3160 mg/kg were reported for Phosphoric Acid. For ammonium salts of phosphoric acid, the reported LD50 was > 5000 mg/kg (rats) and ranged from > 7940 mg/kg to > 10000 mg/kg (rabbits). LD50 values ranging from > 300 mg/kg to > 7940 mg/kg (rabbits) were reported for sodium salts of phosphoric acid. The dermal administration of potassium salts of phosphoric acid to rabbits resulted in reported LD50 values ranging from > 300 mg/kg to > 10000 mg/kg. LD50 values ranging from > 300 mg/kg to > 7940 mg/kg were reported for calcium salts of phosphoric acid. Reported LD50 values ranging from > 2000 mg/kg to > 7940 mg/kg were reported for magnesium salts of phosphoric acid.
Oral
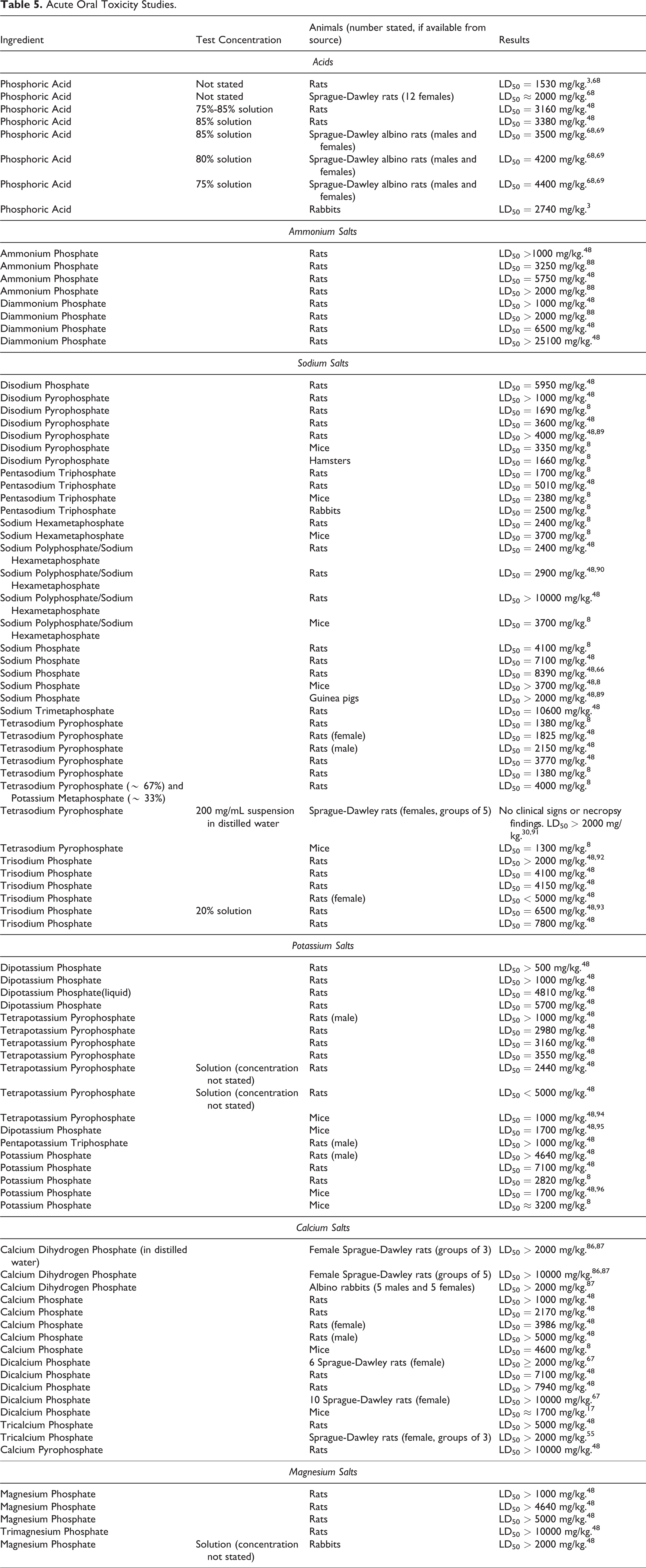
Phosphoric Acid and Salts: Acute oral LD50 values for Phosphoric Acid and its salts are presented in Table 5. In studies involving rats, the LD50 for Phosphoric Acid ranged from 1530 mg/kg to 4400 mg/kg. The LD50 for Phosphoric Acid in rabbits was 2740 mg/kg. The oral LD50 for the ammonium salts of phosphoric acid in studies involving rats ranged from 3250 mg/kg (Ammonium Phosphate) to > 25100 mg/kg (Diammonium Phosphate). Sodium salts of phosphoric acid were administered to rats, mice, hamsters, and guinea pigs in acute oral toxicity studies, and the LD50 ranged from 1300 mg/kg (Tetrasodium Pyrophosphate [mice]) to 10600 mg/kg (Sodium Trimetaphosphate [rats]). For potassium salts of phosphoric acid administered orally in studies involving rats or mice, the acute oral LD50 ranged from 1000 mg/kg (Tetrapotassium Pyrophosphate [mice]) to 7100 mg/kg (Potassium Phosphate [rats]). In acute oral toxicity studies on calcium salts of phosphoric acid involving rats or mice, the reported LD50 ranged from 2170 mg/kg (Calcium Phosphate [rats]) to > 10000 mg/kg (Calcium Pyrophosphate [rats]). The reported LD50 for Magnesium Phosphate in studies involving rats ranged from > 1000 mg/kg (Magnesium Phosphate) to > 10000 mg/kg (Trimagnesium Phosphate).
Inhalation
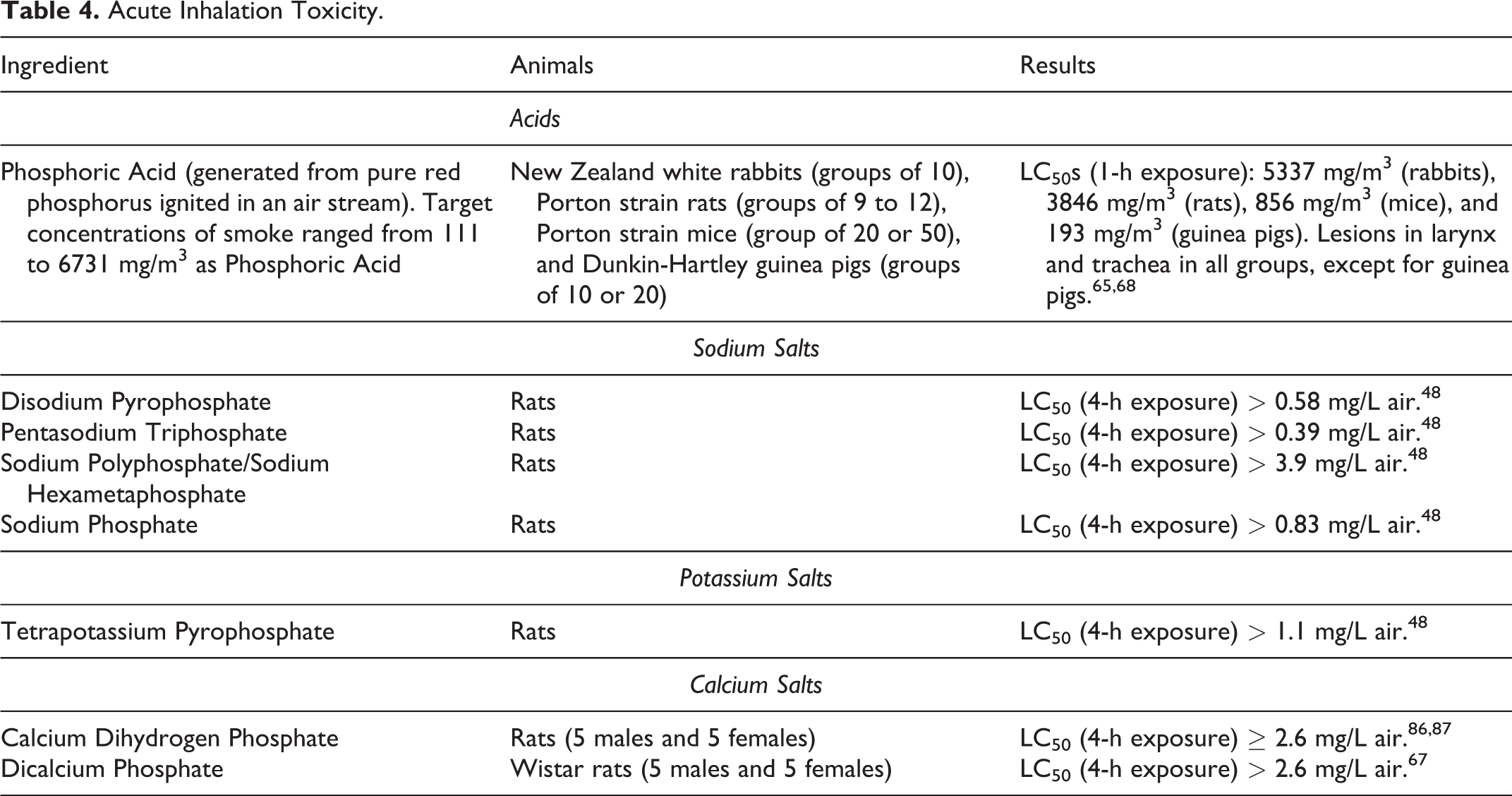
Phosphoric Acid and Salts: Acute inhalation toxicity data on Phosphoric Acid and its sodium, potassium, and calcium salts are presented in Table 4. At the highest lethal concentrations tested, Phosphoric Acid caused tracheal lesions in rabbits, rats, and mice, but not in guinea pigs. Due to its hygroscopic nature, Phosphoric Acid aerosols will combine with water molecules in the air within the human tracheobronchial tree, which increases the aerodynamic diameter of the particles of the aerosol. This effect is known as hygroscopic growth. As a result, the deposition characteristics of these aerosols change as they pass through the respiratory tract, which will affect the total deliverable dose in the lungs after inhalation. 45 Overall, the data suggest that the sodium, potassium, and calcium salts of Phosphoric Acid exhibit a low potential for inhalation toxicity.
Acute Inhalation Toxicity.
Acute Oral Toxicity Studies.
Acute Dermal Toxicity Studies.
According to one publication, Phosphoric Acid caused moderate to acute inhalation toxicity in mice. 57 Pertinent details were not included in this BIBRA Toxicity Profile abstract on phosphoric acid and common inorganic phosphates.
Short-Term, Subchronic, and Chronic Toxicity Studies
Oral
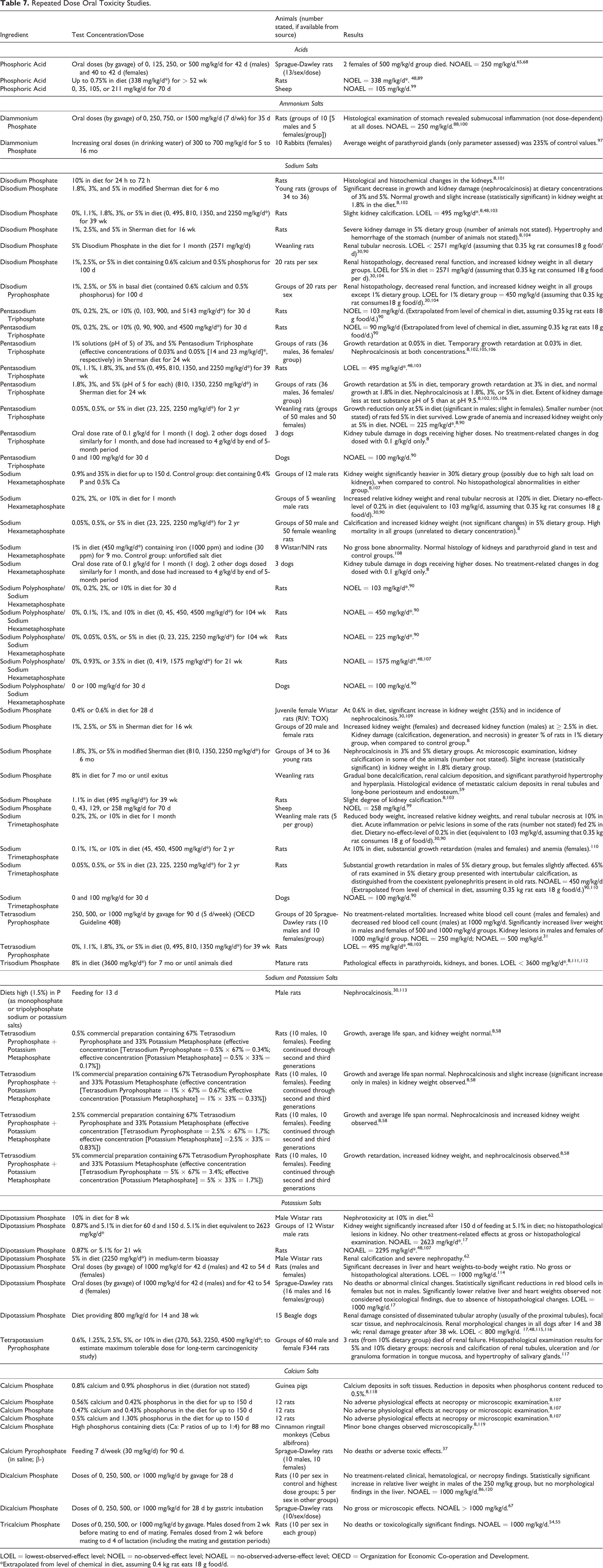
The results of short-term, subchronic, and chronic oral toxicity studies on Phosphoric Acid and its salts are summarized in Table 7. In the longest duration feeding study on Phosphoric Acid, a no-observed-effect level (NOEL) of 338 mg/kg/d was reported for rats that received concentrations up to 0.75% in the diet for 1 year. The average weight of the parathyroid glands (only parameter assessed) was 235% of control values in rabbits that received oral doses of Diammonium Phosphate up to 700 mg/kg/d for up to 16 months. Kidney damage (nephrocalcinosis) was a common pathological finding in repeated oral dose toxicity studies involving sodium and potassium salts of Phosphoric Acid. There were basically no adverse effects in rats/monkeys fed calcium salts of Phosphoric Acid in the diet.
Repeated Dose Oral Toxicity Studies.
LOEL = lowest-observed-effect level; NOEL = no-observed-effect level; NOAEL = no-observed-adverse-effect level; OECD = Organization for Economic Co-operation and Development.
* Extrapolated from level of chemical in diet, assuming 0.4 kg rat eats 18 g food/d.
Developmental and Reproductive Toxicity
Reproductive and developmental toxicity data on ammonium, sodium, potassium, and calcium salts of Phosphoric Acid are presented in Table 8. Teratogenicity was assessed primarily using rats and mice; however, rabbits and hamsters were also used. These salts did not produce teratogenic effects in vivo, and the highest dose tested was 1500 mg/kg/d Diammonium Phosphate (in rats) for 35 days. However, the following salts of Phosphoric Acid were teratogenic to chick embryos: Tetrasodium Pyrophosphate (injection of 5 mg/egg), Sodium Hexametaphosphate (injection of 0.5-10 mg/egg), Sodium Phosphate (injection of 0.5-10 mg/egg), Potassium Phosphate (injection of 10 mg/egg), Calcium Phosphate (injection of 2.5 mg/egg), and Tricalcium Phosphate (injection of 2.5 mg/egg). Information relating to whether or not pH was measured or controlled in the eggs was not found.
Reproductive and Developmental Toxicity Studies.

NOEL = no-observed-effect level.
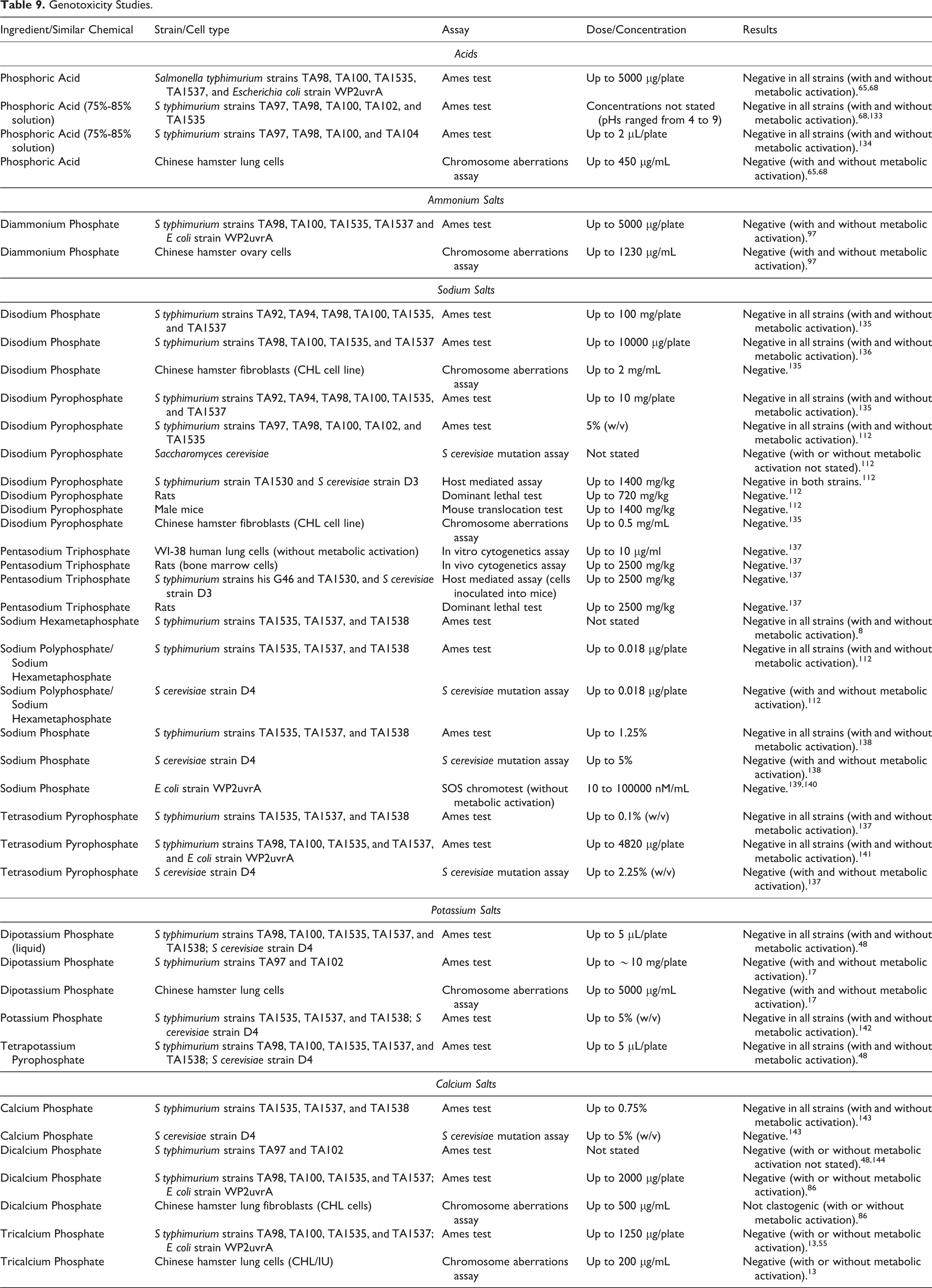
Genotoxicity
The in vitro and in vivo genotoxicity data on Phosphoric Acid and its ammonium, sodium, potassium, and calcium salts are presented in Table 9. The in vitro tests included the Ames/Salmonella mutagenicity assay (with and without metabolic activation), the Saccharomyces cerevisiae mutagenicity assay (with and without metabolic activation), the chromosome aberrations assay (Chinese hamster fibroblasts), and the in vitro cytogenetics assay (human lung cells). The in vivo tests included the dominant lethal test (rats), host-mediated assay (mice), and the mouse translocation test. Phosphoric Acid and its ammonium, sodium, potassium, and calcium salts did not produce positive responses in in vitro or in vivo genotoxicity assays.
Genotoxicity Studies.
Carcinogenicity
Animal
Acids
Phosphoric Acid
According to one source, no carcinogenic potential was demonstrated in limited feeding studies involving rats treated with Phosphoric Acid or several of its salts. However, in rodents treated orally, several phosphates have been shown to promote the effects of known carcinogenicity. 57 Pertinent details were not included in this BIBRA Toxicity Profile abstract on phosphoric acid and common inorganic phosphates.
Sodium Salts
Disodium Phosphate and Tetrasodium Pyrophosphate
An oral feeding study involving groups of 10 male and 10 female rats fed various concentrations of a mixed preparation (33% Potassium Metaphosphate + 67% Tetrasodium Pyrophosphate [in Sherman diet]) was conducted.8,58 The following diets were fed to the rats: 0.5% commercial preparation (effective concentration [Potassium Metaphosphate] = 0.5% × 33% = 0.17%; effective concentration [Tetrasodium Pyrophosphate] = 0.5% × 67% = 0.34%) 1% commercial preparation (effective concentration [Potassium Metaphosphate] = 1% × 33% = 0.33%; effective concentration [Tetrasodium Pyrophosphate] = 1% × 67% = 0.67%) 5% commercial preparation (effective concentration [Potassium Metaphosphate] = 5% × 33% = 1.7%; effective concentration [Tetrasodium Pyrophosphate] = 5% × 67% = 3.4%)
From each dietary group, second and third generations were produced and feeding was continued. For all dietary groups, the tumor incidence was not greater than that observed in control animals. Additional study details were not provided.
Pentasodium Triphosphate
Groups of weanling rats of the Rochester strain (number not stated) were maintained on a diet supplemented with 0.05%, 0.5%, or 5% Pentasodium Triphosphate for 2 years. 59 The carcinogenesis indexes were similar to the frequencies expected for aging rats and did not vary among dietary groups.
Sodium Hexametaphosphate
Groups of weanling rats (males and females; number and strain not stated) were fed a diet containing 0.05%, 0.5%, or 5% Sodium Hexametaphosphate for 2 years. 59 There was no correlation between the dietary level of Sodium Hexametaphosphate and tumor incidence.
Sodium Trimetaphosphate
A diet containing 0.1%, 1%, or 10% Sodium Trimetaphosphate was fed to groups of weanling rats (number and strain not stated) for 2 years. There was no correlation between the dietary level of Sodium Trimetaphosphate and tumor incidence. 59
Sodium Metaphosphate
Calcium sodium metaphosphate (CSM) fiber is a man-made inorganic fiber composed of condensed polyphosphate chains in a specific crystal lattice. 60 Male and female Fischer 344 rats (80/sex/group) were exposed (inhalation) to CSM fiber 6 h/d, 5 d/wk at target-exposure levels of 0, 1, 5, or 25 mg/m3 (corresponding to 0, 27, 80, and 513 fibers/mL, respectively) for 24 months. At 3 and 12 months, 10 rats/sex/group were killed and, at 18 and 24 months, 5 rats/sex/group were killed. Additionally, 5 rats/sex/group were removed from exposure at 18 months and maintained for a 6-month recovery period. No increase in tumors (benign or malignant) was observed in this study.
Tumor Promotion
Potassium Salts
Dipotassium Phosphate
In a tumor promotion study involving groups of 20 uninephrectomized male rats, the following diets were used17,61: Group I: 1000 ppm N-ethyl-N-hydroxyethylnitrosamine (EHEN) diet (2 weeks) + 50000 ppm Dipotassium Phosphate diet (18 weeks) Group II: Basal diet (2 weeks) + 50000 ppm Dipotassium Phosphate (18 weeks) Group III: 1000 ppm EHEN diet (2 weeks) + the basal diet (18 weeks) Group IV: Basal diet (20 weeks)
The rats were fed EHEN (1000 ppm) in the diet for 2 weeks, and the left kidney was removed at week 3. After nephrectomy, the rats were fed Dipotassium Phosphate (50000 ppm) in the diet for 18 weeks (from week 3 to week 20). A control group of 20 rats received basal diet only after EHEN administration and nephrectomy. The mean relative kidney weight per body weight in group I was significantly greater when compared to group III. Additionally, the mean kidney weight in group II was significantly greater when compared to group IV. The numbers of simple hyperplastic foci and adenomatous hyperplastic foci in group I animals were statistically significantly greater (P < 0.05) when compared to group III. The incidence of renal cell tumors was 30% in group I. Nephropathy, lymphocyte accumulation, hyaline droplets in proximal convoluted tubular cells, and dilatation of the proximal convoluted tubular cells were observed in the cortex of group I and group II animals. Calcification was observed in the renal medulla and cortex of groups I and II. It was concluded that Dipotassium Phosphate promoted the development of renal tubular cell tumors. The authors noted that the results documented in this study clearly suggest a link between toxicity-dependent proliferation and promoting ability.
In a medium-term bioassay for renal tumorigenesis, the feeding of male Wistar rats with 5% potassium dibasic phosphate in the diet promoted the development of preneoplastic lesions. 62 These study results were obtained from the limited details found in the English abstract of a Japanese publication.
Phosphate
A study was performed to elucidate the potential effects of high dietary phosphate (Pi) on the development of lung cancer. 63 The first experiment involved 2 groups of male K-rasLA1 mice (9 per group). One group received an AIN93-based diet containing 0.5% Pi (normal Pi), and the other group received the same diet fortified with 1% Pi (high Pi). Both diets were fed to the mice for 4 weeks, after which the animals were killed. Blood samples were obtained and necropsy was performed. Tumor lesions of lung surfaces were counted and the diameter of each lesion was measured. A lobe of the left lung was prepared for histopathological examination and immunohistochemistry. The diet containing 1% Pi increased lung tumor progression and growth, when compared with the diet containing 0.5% Pi. Histopathological examination results showed that pulmonary tumor progression was markedly stimulated by 1% Pi in the diet. The number and size (at least 1.5 mm in diameter) of lung surface tumor lesions (adenomas) increased significantly (P < 0.05). Pi (1%) in the diet also had the following effects: (1) increased the sodium-dependent inorganic phosphate transporter-2b protein levels in the lungs; (2) stimulated pulmonary protein kinase B (Akt; known to regulate cell cycle progression) activity, while suppressing (a) the protein levels of tumor suppressor phosphatase and tensin homolog deleted on chromosome 10 and (b) the Akt binding partner carboxyl-terminal modulator protein, resulting in facilitated cap-dependent protein translation; and (3) significantly (P < 0.05) stimulated cell proliferation in the lungs of K-rasLA1 mice.
In a second study (urethane-induced lung cancer model), A/J mice were injected intraperitoneally with urethane (1 mg/g body weight) in saline. At 4-week postinjection, the mice were divided into 2 groups (7 mice per group) and fed 1% Pi and 0.5% Pi in the diet, respectively, for 4 weeks. The effect of high dietary Pi on lung tumorigenesis was confirmed in this experiment. Pi (1%) in the diet statistically significantly increased (P < 0.05) tumor development. Both the mean number of tumors and the mean tumor diameter (at least 1 mm in diameter) increased statistically significantly (P < 0.05). Histopathological examination results also showed that pulmonary tumor progression was stimulated. The authors noted that the results of this study indicate that high dietary Pi strongly activated Akt signaling and increased lung tumorigenesis. 63
Other Relevant Studies
Cytotoxicity
Calcium Phosphate and Dicalcium Phosphate Dihydrate
The cytotoxicity of the following mixture was evaluated using a mouse L-929 cell suspension: Tricalcium Phosphate (90%; α-) and Dicalcium Phosphate Dihydrate (10%) in a solution containing chondroitin sulfate (5%) and sodium succinate (12%). 64 Cell morphology was evaluated at 24 hours; the affected area of the cell layer was determined using microscopy. Contracted cells, rounded cells with dark nuclei, and broken cells were considered damaged cells. A very low degree of cytotoxicity (mild cytotoxicity) was observed.
Calcium Pyrophosphate
The cytotoxicity of Calcium Pyrophosphate was studied using Chinese hamster ovary K-1 cells. 11 Cytotoxicity potential was determined quantitatively by cytolethality (expressed as the cytotoxicity index [IC50%]) using a colony suppression assay. The IC50% is defined as the concentration that is necessary to kill half of the cell population or the concentration that suppresses colony formation to 50% of the control value. Phenol solution (0.02%) and alumina extracts served as positive and negative controls, respectively. Calcium Pyrophosphate was not cytotoxic (IC50% = 100). The positive and negative controls performed as expected.
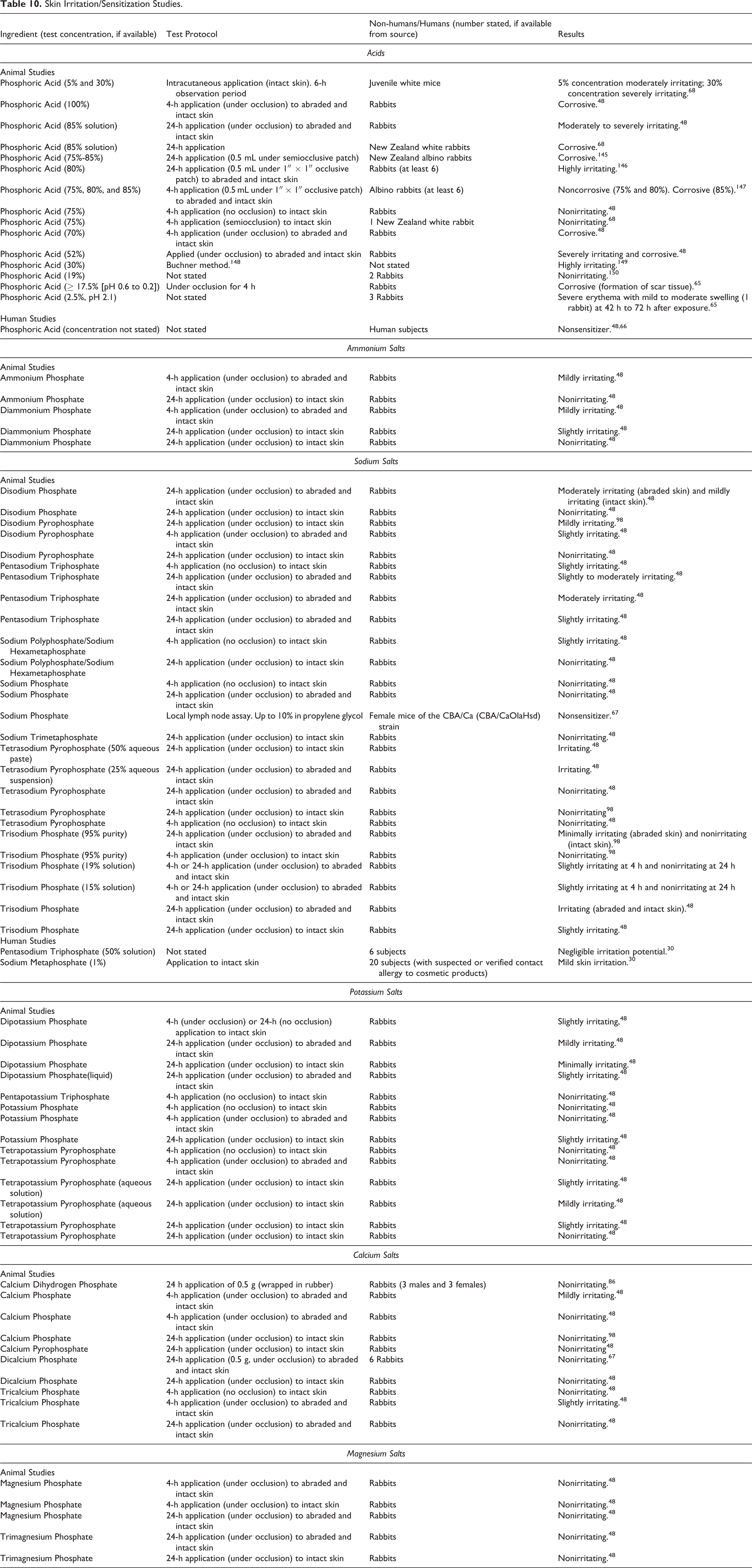
Dermal Irritation and Sensitization Studies
Skin irritation and sensitization data on Phosphoric Acid and its ammonium, sodium, potassium, calcium, and magnesium salts are presented in Table 10. A broad range of reactions (from irritation/no irritation [Phosphoric acid and salts] to irritating/corrosive [Phosphoric Acid] effects) reported for phosphoric acid or its salts at concentrations within and outside of the range of those used in cosmetic products. Phosphoric acid was an irritant at concentrations as low as 2.5%; however, the pH of the test substance was low, pH of 2.1.67 The corrosive effect of Phosphoric Acid was observed at concentrations ranging from 17.5% (pH of 0.6-0.2) to 100%.48,65 The sodium salts were nonirritating (Sodium Phosphate)50 to moderately irritating (Disodium Phosphate), 48 and the potassium and calcium salts were nonirritating (Potassium Phosphate and Dicalcium Phosphate) 48 to mildly irritating (Dipotassium Phosphate and Calcium Phosphate)50 to rabbit skin. The ammonium salts (Ammonium Phosphate and Diammonium Phosphate) were nonirritating to mildly irritating to rabbit skin. 48 The magnesium salts of Phosphoric Acid (Magnesium Phosphate and Trimagnesium Phosphate) were nonirritating to the skin of rabbits. 48 Pentasodium Triphosphate (50% solution) and Sodium Metaphosphate (1% solution) were mildly irritating to the skin of human subjects. 30 Phosphoric Acid was not sensitizing in human subjects,48,66 and Sodium Phosphate (10% in propylene glycol) was not sensitizing in the local lymph node assay. 67
Skin Irritation/Sensitization Studies.
Ocular Irritation
Animal
Ocular irritation data on phosphoric acid and its ammonium, sodium, potassium, calcium, and magnesium salts are presented in Table 11. Phosphoric Acid was corrosive to the eyes of rabbits at concentrations in the 70% to 85% range48,68,69 but was nonirritating at concentrations of 10% and 17%.65,68 None of the salts of Phosphoric Acid was found to be corrosive to the eyes of rabbits. However, ocular irritation was observed; for example, Tetrasodium Pyrophosphate was irritating at a concentration of 10% and Trisodium Phosphate was irritating at concentrations of 10% and 15%.30,48
Ocular Irritation/Toxicity Studies.
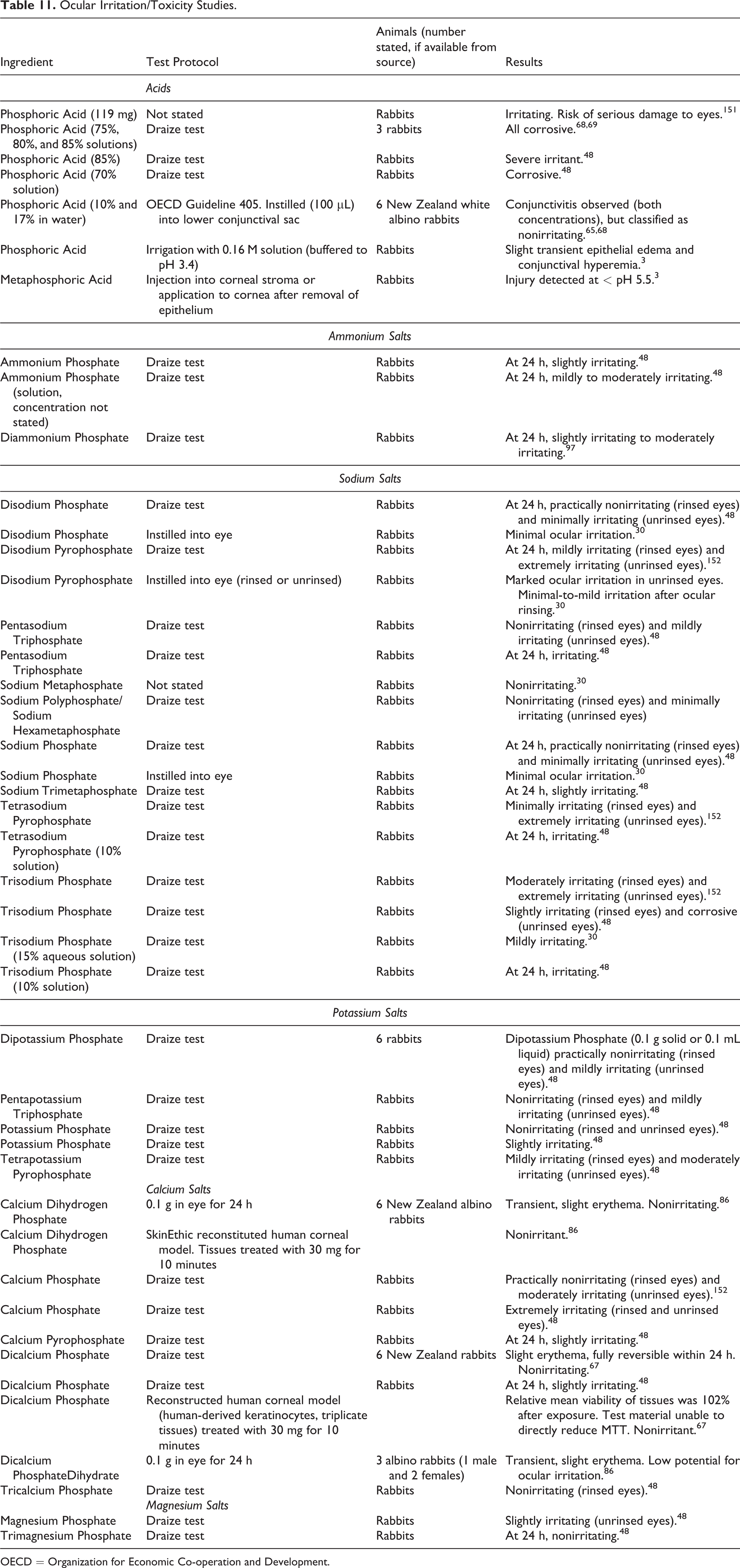
OECD = Organization for Economic Co-operation and Development.
Mucosal Irritation
Human
Phosphoric Acid
Phosphoric Acid (50%) was applied to the gingival tissue and teeth of 26 orthodontic patients. 3 The 90-s contact period for the acid was followed by rinsing. No demonstrable test substance-related effect on treated tissues was observed during the 7-day observation period.
Tetrasodium Pyrophosphate and Tetrapotassium Pyrophosphate
Some nonprescription dentifrices, particularly pyrophosphate-based tartar control toothpastes, may be irritating to oral tissues. 33 The following clinical observations were made in patients (number not stated) at a dental clinic who frequently uses tartar control toothpastes containing Tetrasodium Pyrophosphate and/or Tetrapotassium Pyrophosphate: pale gingival tissues, mucosal sloughing, small blisters, dryness of oral tissues, and/or free-gingival-margin erythema. Subjective symptoms included a painful, burning sensation of oral tissues (most commonly gingival mucosa); a generalized, nonspecific sensitivity or odd feeling to teeth and/or soft tissues; and sensations of ″itchy″ oral tissues. Patient complaints averaged approximately 5 per week over a 2-year period. Amelioration of the patients’ chief symptoms occurred rapidly upon switching to a nontartar control toothpaste.
Clinical Reports
Calcium Pyrophosphate
The articular deposition of Calcium Pyrophosphate (Calcium Pyrophosphate deposition disease) is a common age-related phenomenon. Frequently, this disease is asymptomatic and unassociated with structural joint damage.70,71 Acute attacks of synovitis, resulting in pseudogout, are observed. 72 These attacks are often provoked by local trauma or surgery and commonly involve the knee, and, less often, the wrist, hip, shoulder, and elbow.
Sodium Phosphate
A systematic review of adverse event reports relating to oral Sodium Phosphate (used for bowel cleansing prior to colonoscopy) was performed. 73 Fifty-eight publications of significant events in 109 patients who used Sodium Phosphate were identified. Between January of 2006 and December of 2007, the most commonly reported findings were electrolyte disturbances, renal failure, and colonic ulceration. The number of cases of renal failure reported to FDA during this period was 171.
A retrospective study of renal adverse event reports was performed using the FDA Adverse Event Reporting System, a voluntary reporting system available for public access. 74 A total of 2097223 files (year 2004-2008 and the first 9 months of 2009) from FDA’s website were examined. Of the 178 patients (71% women) on sodium phosphate tablets identified, an increasing number of renal adverse drug reactions (ADRs) associated with tablet preparations were reported each year. In 2006, 9 of 74 (12%) renal ADRs were reported to be from ingesting tablets; results for subsequent year were as follows: 40 of 181 (22%; 2007), 46 of 148 (31%; 2008), and 60 of 795 (7.55%; 2009). The mean weight for women with renal complications from tablet preparations was 68.57 ± 1.78 kg, statistically significantly lower than the national average weight of 74 ± 0.5 kg for the same age-group (P = 0.003) in the National Health and Nutrition Examination Survey. It was concluded that renal ADRs from sodium phosphate tablets were more common in women with a mean body weight lower than the national average weight.
In more recent studies, 10 adult cases of acute phosphate nephropathy, associated with acute renal failure, following administration of a Sodium Phosphate preparation before colonoscopy, and a case series of 3 children with severe hyperphosphatemia and hypocalcemia after the use of Sodium Phosphate-containing laxatives were reported.75,76 Acute renal failure due to phosphate nephropathy following bowel cleansing with an oral Sodium Phosphate solution was reported in another patient. 77 Electron microscopy of a kidney biopsy sample revealed membranous glomerulonephritis and Calcium Phosphate deposits were observed in tubular cells and in tubules. Phosphate remained elevated for 11 days; other electrolyte levels were normal. A biopsy taken only 2 months before the acute kidney disease showed no sign of the Calcium Phosphate deposits in the second biopsy. It was concluded that the phosphate load given to the patient was responsible for the biopsy findings.
Epidemiology
Acids
Phosphoric acid
In the 1980s, a large population-based case-control study in Montreal was performed to explore the possible associations among hundreds of occupational substances and multiple cancer sites, 78 and an analysis of the occupational information collected in this study (focusing on renal cell cancer) was subsequently performed. 78 In this study, the following individuals were interviewed: 142 male patients with pathologically confirmed renal carcinoma; 1900 controls with cancer at other sites; and 533 population-based controls. Logistic regression results for exposure to selected substances were presented, including the following 2 sets of odds ratios (ORs): (1) OR1 (95% CI): ORs (adjusted for respondent status, age, smoking, and body mass index [BMI]) and 95% CI; (2) OR2 (95% CI): ORs (adjusted for respondent status, age, smoking, BMI, and occupational confounders) and 95% CI. The authors concluded that there was evidence of excess risk for renal cell carcinoma following workplace exposure to Phosphoric Acid, as indicated by the following ORs: The OR1 value reported for phosphoric acid was 3.4 (1.3-9.2), and an OR2 value of 2.4 (0.8-7.0) was reported. 78
In the International Agency for Research on Cancer (IARC) monograph on occupational exposures to mists and vapors from sulfuric acid and other inorganic acids (including Phosphoric Acid), several questionable epidemiological studies in the phosphate fertilizer manufacturing industry showed excess lung cancer; but, IARC did not classify Phosphoric Acid as carcinogenic. 79 However, IARC did conclude that occupational exposure to strong inorganic acid mists containing sulfuric acid is carcinogenic to humans.
Phosphates
Cancer morbidity and mortality were studied in a population of employees of phosphate ore mining and processing operations in Central Florida. 80 The workers involved in the study were employed by participating phosphates companies between 1950 and 1979, and the study population consisted of 3541 male employees who had worked for 6 months or more. Based upon an industrial hygiene analysis, only drying/shipping, chemical/fertilizer, and maintenance job categories were found to have the potential for exposure to high levels of dust, chemical fumes, or radiation. Cancer incidence was traced using questionnaires confirmed by medical records and by tumor registry searches. Standardized incidence ratios were calculated. To estimate the study population’s risk in relation to general rates in the United States, standardized mortality ratios (SMRs) adjusted for age and calendar time were calculated. The SMRs were tested for statistical significance at the 0.05 level using the Poisson distribution. Statistically significant elevations in lung cancer (SMR = 1.62) and emphysema were observed when compared to rates in the United States. For workers employed over a period of 20 years, there was a dose-response trend of increasing lung cancer risk with increasing duration of employment (SMR = 2.48, with 20 years of employment). There was no evidence of excess lung cancer risk among employees who were hired after 1960. The authors noted that multivariate analyses and internal comparisons of risk by job type were consistent with a hypothesis of occupationally related lung cancer, but that the small numbers prevented firm conclusions.
Risk Assessment
Phosphates, Diphosphates, and Polyphosphates
Oral
Phosphates, diphosphates (ie, pyrophosphates), and polyphosphates (eg, metaphosphates) were evaluated by the Joint Food and Agriculture Organization (FAO)/World Health Organization (WHO) Expert Committee on Food Additives. 81 A maximum tolerable daily intake (MTDI) of 70 mg/kg was determined, based on the lowest concentration of phosphorus (6600 mg/d) that caused nephrocalcinosis in rats. ″The MTDI is expressed as phosphorus and applies to the sum of phosphates naturally present in food and the phosphates derived from use of these food additives.″ The FAO/WHO Expert Committee considered establishing an average daily intake to be inappropriate because phosphorus (as phosphates) is an essential nutrient and an unavoidable constituent of food. The Federation of American Societies for Experimental Biology estimate of MTDI of phosphates in man is also 70 mg/kg. 82
Inhalation
Phosphoric Acid
The EPA calculated an inhalation reference concentration (RfC) of 1 × 10−2 mg/m3 for Phosphoric Acid (the critical effect is bronchiolar fibrosis). 83 Development of an inhalation RfC involves evaluating toxic effects inside the respiratory system (port-of-entry effects) and outside the respiratory system (extrarespiratory effects). In general, the RfC is an estimate (with uncertainty spanning perhaps an order of magnitude) of a daily inhalation exposure of the human population (including sensitive subgroups) that is likely to be without an appreciable risk of deleterious effects over a lifetime of exposure. The calculated RfC for Phosphoric Acid is based on inhalation toxicity data summarized in the Repeated Dose Toxicity Section of this safety assessment. 84 Based on the histologic lesions in the tracheobronchiolar region, 180 mg/m3 was the LOAEL, and 50 mg/m3 was the NOAEL in this study.
Summary
The safety of 31 ingredients, Phosphoric Acid and its salts, as used in cosmetics is reviewed in this safety assessment. The functions of these ingredients in cosmetic products frequently include buffering agents, corrosion inhibitors, chelating agents, and pH adjusters.
According to the 2016 VCRP data, the greatest reported use frequency is for Phosphoric Acid (489 formulations, mostly rinse-off), followed by Dicalcium Phosphate (327 formulations, mostly leave-on). Lower use frequencies were reported for the remaining salts. The results of a concentration of use survey provided in 2015 indicate that Dicalcium Phosphate Dihydrate has the highest maximum concentration of use; it is used at concentrations up to 49% in rinse-off products (dentifrices).
Phosphoric Acid ionizes and is then absorbed as phosphate and hydronium ions through mucous membranes. Some of the phosphate and hydronium ions are conjugated in the liver and then excreted in the urine. Following the absorption of phosphates from the gastrointestinal tract, phosphate combines with calcium to form calcium hydrogen orthophosphate in bones and teeth. Free orthophosphate is the primary form by which dietary Pi is absorbed. In general, approximately two-thirds of the ingested phosphate is absorbed from the gastrointestinal tract in adults, and absorbed phosphate is almost entirely excreted in the urine.
In acute inhalation toxicity studies, at the highest lethal concentrations, Phosphoric Acid caused tracheal lesions in rabbits, rats, and mice, but not in guinea pigs. Overall, the data suggest that the sodium, potassium, and calcium salts exhibit a low potential for inhalation toxicity. The EPA has calculated an inhalation RfC of 0.01 mg/m3 for Phosphoric Acid, based on the results from 2 parallel 13-week inhalation toxicity studies involving rats. In general, the RfC is an estimate of a daily inhalation exposure of the human population that is likely to be without an appreciable risk of deleterious effects during a lifetime.
In acute oral toxicity studies involving rats, the LD50 for Phosphoric Acid ranged from 1530 mg/kg to 4400 mg/kg. The oral LD50 for Phosphoric Acid in rabbits was 2740 mg/kg. The oral LD50 for the ammonium salts of Phosphoric Acid in studies involving rats ranged from 5750 mg/kg (Ammonium Phosphate) to > 25100 mg/kg (Diammonium Phosphate). Sodium salts of Phosphoric Acid were administered to rats, mice, hamsters, and guinea pigs in acute oral toxicity studies, and LD50 values ranged from 1300 mg/kg (Tetrasodium Pyrophosphate [mice]) to 10600 mg/kg (Sodium Trimetaphosphate [rats]). For potassium salts of Phosphoric Acid administered orally in studies involving rats or mice, acute oral LD50 values ranged from 1000 mg/kg (Tetrapotassium Pyrophosphate, involving mice) to 7100 mg/kg (Potassium Phosphate [rats]). In acute oral toxicity studies on calcium salts of Phosphoric Acid involving rats or mice, LD50 values ranged from 2170 mg/kg (Calcium Phosphate [rats]) to > 10000 mg/kg (Calcium Pyrophosphate [rats]). LD50 values for Magnesium Phosphate in studies involving rats ranged from > 1000 mg/kg (Magnesium Phosphate) to > 10000 mg/kg (Trimagnesium Phosphate).
The feeding of Phosphoric Acid at concentrations up to 0.75% in the diet of rats for 52 weeks yielded an NOEL of 338 mg/kg/d. An NOAEL of 105 mg/kg/d was reported in a study in which sheep received doses of Phosphoric Acid up to 211 mg/kg/d for 70 days. An NOAEL of 250 mg/kg/d was reported for groups of rats that received Diammonium Phosphate at doses up to 1500 mg/kg/d for 35 days. The average weight of the parathyroid glands (only parameter assessed) was 235% of control values in rabbits that received oral doses of Diammonium Phosphate up to 700 mg/kg/d for up to 16 months.
A study of rats fed Disodium Phosphate or Disodium Pyrophosphate (up to 5% in the diet) for 100 days resulted in an LOEL (renal histopathology) of < 2571 mg/kg/d (Disodium Phosphate) and an LOEL (renal histopathology) = 450 mg/kg/d (Disodium Pyrophosphate). When Disodium Phosphate, Pentasodium Triphosphate, or Tetrasodium Pyrophosphate was administered to rats at concentrations up to 5% in the diet for 39 weeks, an LOEL of 495 mg/kg/d was reported. Of the NOELs determined in rat studies, the highest NOEL (338 mg/kg/d) was reported in a study in which rats were fed Phosphoric Acid at concentrations up to 0.75% in the diet daily for > 52 weeks. The highest NOAEL (2623 mg/kg/d) was reported in a study in which rats were fed Dipotassium Phosphate at concentrations up to 5.1% in the diet daily for 150 days. In studies involving dogs, an NOAEL of 100 mg/kg/d was reported for the following sodium salts, each of which was administered orally at a dose of 100 mg/kg/d for 30 days: Pentasodium Triphosphate, Sodium Polyphosphate/Sodium Hexametaphosphate, and Sodium Trimetaphosphate. Kidney damage (nephrocalcinosis) was a common finding in repeated dose oral toxicity studies involving sodium salts of Phosphoric Acid. The feeding of commercial preparations, to rats, containing effective concentrations of up to 3.4% Tetrasodium Pyrophosphate and 1.7% Potassium Metaphosphate also resulted in nephrocalcinosis.
When potassium salts of Phosphoric Acid were fed in the diet of rats at concentrations ranging from 0.6% to 10%, nephrocalcinosis/nephrotoxicity was observed at concentrations of 5% (Tetrapotassium Pyrophosphate [daily doses; number of days not stated]) and 10% (Tetrapotassium Pyrophosphate [daily doses; number of days not stated] or Dipotassium Phosphate [8 weeks]). Nephrocalcinosis was also observed in dogs that were fed a diet providing Dipotassium Phosphate at a dose of 800 mg/kg/d. There were basically no adverse findings in rats/monkeys fed calcium salts of Phosphoric Acid in the diet (up to 0.8% calcium and 1.30% phosphorus). The same was true for rats that received Dicalcium Phosphate or Tricalcium Phosphate at doses up to 1000 mg/kg/d.
In acute dermal toxicity studies involving rabbits, an LD50 of 2740 mg/kg and an LD50 > 3160 mg/kg were reported for Phosphoric Acid. For ammonium salts of Phosphoric Acid, the dermal LD50 for rats was > 5000 mg/kg (rats) and ranged from > 7940 mg/kg to > 10000 mg/kg for rabbits. Dermal LD50 values ranging from > 300 mg/kg to > 7940 mg/kg (rabbits) were reported for sodium salts of Phosphoric Acid. The dermal administration of potassium salts of Phosphoric Acid to rabbits resulted in dermal LD50 values ranging from > 300 mg/kg to > 10000 mg/kg. Dermal LD50 values ranging from > 300 mg/kg to > 7940 mg/kg were reported for calcium salts of Phosphoric Acid. LD50 values ranging from > 2000 mg/kg to > 7940 mg/kg were reported for magnesium salts of Phosphoric Acid.
The teratogenicity of ammonium, sodium, potassium, and calcium salts of Phosphoric Acid was assessed primarily using rats and mice; however, rabbits and hamsters were also used. These salts did not produce teratogenic effects in vivo, and the highest dose tested was Diammonium Phosphate at 1500 mg/kg/d for 35 days. However, the following salts of Phosphoric Acid were teratogenic to chick embryos: Tetrasodium Pyrophosphate (injection of 5 mg/egg), Sodium Hexametaphosphate (injection of 0.5-10 mg/egg), Sodium Phosphate (injection of 0.5-10 mg/egg), Potassium Phosphate (injection of 10 mg/egg), Calcium Phosphate (injection of 2.5 mg/egg), and Tricalcium Phosphate (injection of 2.5 mg/egg).
Both in vitro and in vivo genotoxicity data on Phosphoric Acid and its ammonium, sodium, potassium, and calcium salts are available. The in vitro tests included the Ames/Salmonella mutagenicity assay (with and without metabolic activation), the S cerevisiae mutagenicity assay (with and without metabolic activation), the chromosome aberrations assay (Chinese hamster fibroblasts), and the in vitro cytogenetics assay (human lung cells). The in vivo tests included the dominant lethal test (rats), host-mediated assay (mice), and the mouse translocation test. Phosphoric Acid and its ammonium, sodium, potassium, and calcium salts did not produce positive responses in in vitro or in vivo genotoxicity assays.
In an oral carcinogenicity study, rats were fed mixtures containing up to 1.7% Potassium Metaphosphate and up to 5% Tetrasodium Pyrophosphate. Feeding was continued through the second and third generations produced. For all dietary groups, the tumor incidence was similar to control animals. When groups of rats were fed Pentasodium Triphosphate or Sodium Hexametaphosphate at concentrations up to 5% for 2 years, there was no correlation between concentration in the diet and tumor incidence. The same was true for rats fed a diet containing up to 10% Sodium Trimetaphosphate.
The results of a study on high dietary Pi intake and the development of lung cancer in mice indicated that high dietary Pi strongly activated Akt signaling and increased lung tumorigenesis
In a population-based case-control study, workplace exposure to Phosphoric Acid produced some evidence of excess risk of renal cell carcinoma. Furthermore, in an IARC monograph on occupational exposure to Phosphoric Acid and other inorganic acids, there were several questionable epidemiological studies of the phosphate fertilizer manufacturing industry that showed excess lung cancer. However, IARC did not classify Phosphoric Acid as carcinogenic. Dipotassium Phosphate, given in the diet (containing the carcinogen, EHEN) of male rats, promoted the development of renal tumors.
Skin irritation and sensitization data on Phosphoric Acid and its ammonium, sodium, potassium, calcium, and magnesium salts are available, and a broad range of reactions (nonirritating to corrosive) have been reported. Phosphoric Acid was classified as nonirritating or corrosive. Phosphoric acid was an irritant at concentrations as low as 2.5%; however, the pH of the test substance was low, pH of 2.1. The corrosive effect of Phosphoric Acid was observed at concentrations ranging from 17.5% (pH of 0.6-0.2) to 100%, but 19% Phosphoric Acid was nonirritating. The sodium salts were nonirritating to moderately irritating, and the potassium and calcium salts were nonirritating to mildly irritating to rabbit skin. The magnesium salts of Phosphoric Acid were nonirritating to the skin of rabbits. Pentasodium Triphosphate and Sodium Metaphosphate were mildly irritating to the skin of human subjects. Phosphoric Acid was a nonsensitizer in human subjects, and Sodium Phosphate was a nonsensitizer in the local lymph node assay.
Phosphoric Acid was corrosive to the eyes of rabbits at concentrations in the 70% to 85% range, but was nonirritating at concentrations of 10% and 17%. None of the salts of Phosphoric Acid was found to be corrosive to the eyes of rabbits. However, ocular irritation was observed; for example, Tetrasodium Pyrophosphate was irritating at a concentration of 10% and Trisodium Phosphate was irritating at concentrations of 10% and 15%.
Renal failure has resulted from the use of sodium phosphate-containing colonoscopy preparations. Other case reports have indicated that some nonprescription dentifrices, particularly pyrophosphate-based tartar control toothpastes, may be irritating (erythema, burning, and mucosal sloughing) to oral tissues. The clinical findings relate to tartar control toothpastes containing Tetrasodium Pyrophosphate and/or Tetrapotassium Pyrophosphate.
Discussion
The Panel noted the broad range of results (from irritation/no irritation to irritating/corrosive effects) reported for Phosphoric Acid or its salts at concentrations within and outside of the range of those used in cosmetic products. The results of a concentration of use survey provided by the Council in 2015 indicate that Dicalcium Phosphate Dihydrate has the highest maximum concentration of use; it is used at concentrations up to 49% in rinse-off products (dentifrices). Phosphoric acid was an irritant at concentrations as low as 2.5%; however, the pH of the test substance was low, pH of 2.1. The corrosive effect of Phosphoric Acid was observed at concentrations ranging from 17.5% (pH of 0.6-0.2) to 100%. For salts of Phosphoric Acid, skin irritation was observed at concentrations ranging from 1% to 50% and ocular irritation was observed at concentrations as low as 10% and 15%.
The Panel noted that test animals fed high concentrations of Phosphoric Acid in the diet exhibited renal damage and evidence of the tumor-promoting potential of Phosphoric Acid. The oral exposures to Potassium Phosphate in one of these studies promoted the development of kidney tumors initiated by treatment with a potent renal carcinogen. The Panel also discussed animal studies on Potassium Phosphate indicating that this salt was not associated with renal damage or cancer, and one epidemiological study suggesting an association between occupational exposures to Phosphoric Acid and kidney and lung cancer. The Panel concluded that renal toxicity and tumor promotion would not be expected from exposures to cosmetic products containing phosphoric acid or its salts, because such exposures can reasonably be anticipated to be substantially lower than those associated with adverse effects in these studies.
Concern about heavy metals that may be present in salts of Phosphoric Acid was expressed by the Panel. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit impurities in the ingredient before blending into cosmetic formulations.
The Panel discussed the issue of incidental inhalation exposure from propellant and pump hair sprays and face powders. The Panel considered inhalation toxicity data and pertinent data indicating that incidental inhalation exposures to these ingredients in such cosmetic products would not cause adverse health effects, including acute inhalation toxicity data on Phosphoric Acid and its salts and data characterizing the potential for these ingredients to cause acute and repeated dose oral toxicity, and ocular or dermal irritation or sensitization. The Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that the following 31 ingredients are safe in the present practices of use and concentration in cosmetics as described in this safety assessment, when formulated to be nonirritating.

*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author Contribution
Johnson, W. contributed to conception and design; contributed to acquisition, analysis, and interpretation; and drafted the manuscript. Heldreth, B. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Boyer, I., Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception and design; contributed to analysis and interpretation; and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.