Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) assessed the safety of 8 dialkyl sulfosuccinate salts for use in cosmetics, finding that these ingredients are safe in cosmetics in the present practices of use and concentration when formulated to be nonirritating. The dialkyl sulfosuccinate salts primarily function as surfactants in cosmetics. The Panel reviewed the new and existing available animal and clinical data in making its determination of safety. The Panel found it appropriate to extrapolate the data on diethylhexyl sodium sulfosuccinate to assess the safety of the entire group because all of the diesters are of a similar alkyl chain length, all are symmetrically substituted, and all have similar functions in cosmetic formulations.
Introduction
Diethylhexyl sodium sulfosuccinate (previously named dioctyl sodium sulfosuccinate) was reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel (Panel) in 1994, and a safe concentration limit of 0.42% was established. A petition to open the report to review new clinical data was received, and in 1998, the Panel amended the report to conclude that this ingredient is safe as used in cosmetic formulations. 1 In the discussion, the Panel stressed that care should be taken to avoid irritancy, especially in those products intended for prolonged contact with the skin.
The International Cosmetic Ingredient Dictionary and Handbook lists 7 additional dialkyl sulfosuccinate salts.
2
All the dialkyl sulfosuccinate salts are anionic surfactants, and the Panel determined that the data on diethylhexyl sodium sulfosuccinate can be extrapolated to support the safety of the following 7 salts: Ammonium dinonyl sulfosuccinate Diamyl sodium sulfosuccinate Dicapryl sodium sulfosuccinate Diheptyl sodium sulfosuccinate Dihexyl sodium sulfosuccinate Diisobutyl sodium sulfosuccinate Ditridecyl sodium sulfosuccinate
New data, including published literature that has become available since the CIR safety assessment was issued in 1998, are presented in this review. Data from 1998 report on diethylhexyl sodium sulfosuccinate are summarized in Table 1.
Data From the Previous Review of Diethylhexyl Sodium Sulfosuccinate. 1

Abbreviations: AF, acanthosis factor; bw, body weight; HRIPT, human repeated insult patch test; IV, intravenous; LD50, median lethal dose; PII, primary irritation index.
The CIR has not reviewed and concluded on the safety of all of the individual alcohol constituents that make up the sulfosuccinate salts. However, data on caprylic, isobutyl, and ethylhexyl alcohols, which are constituents of a few of the dialkyl sulfosuccinate salts, have been summarized in previous CIR reviews. 3 Accordingly, these data are provided in Table 2.
Data on Constituent Alcohols. 3

Abbreviations: bw, body weight; DNA, deoxyribonucleic acid.
Chemistry
Definition and Structure
The ingredients included in this review are the salts of diesters of 2-sulfosuccinic acid. The ingredients all share a sulfo-substituted succinic acid core; accordingly, these salts are sulfosuccinates. For example, diheptyl sodium sulfosuccinate consists of a 7-carbon alkyl chain (heptyl), bonded to the sulfosuccinate core via an ester linkage, and followed by an ester linkage to an additional 7-carbon alkyl chain (Figure 1).
Diheptyl sodium sulfosuccinate.
Due to the ester linkage, these sulfosuccinate ingredients are theoretically sensitive to hydrolysis, especially under acidic conditions.
The dialkyl sulfosuccinate salts included in this assessment are defined in Table 3, and the structures are depicted as follows:
Definitions and Functions.

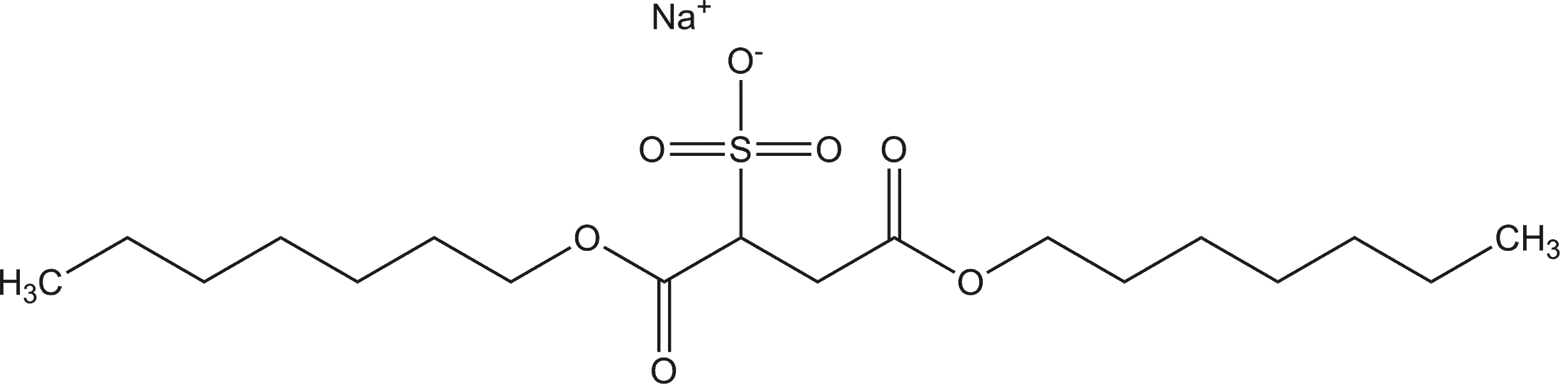
Ammonium dinonyl sulfosuccinate 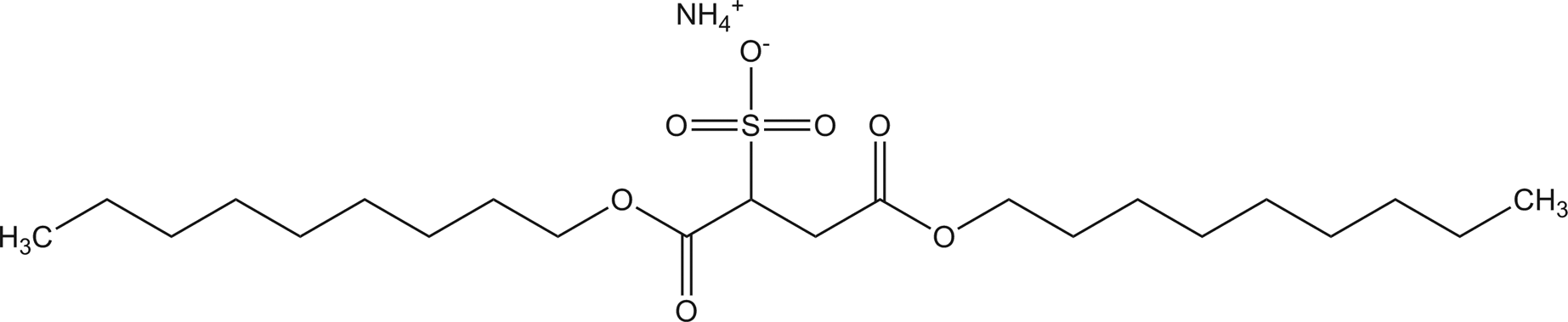
Diamyl sodium sulfosuccinate 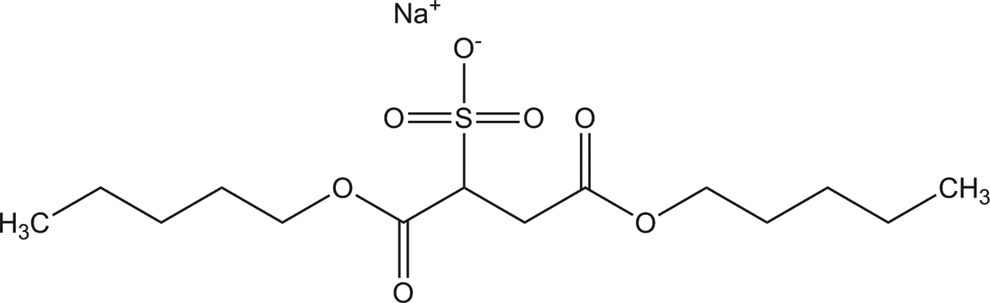
Dicapryl sodium sulfosuccinate 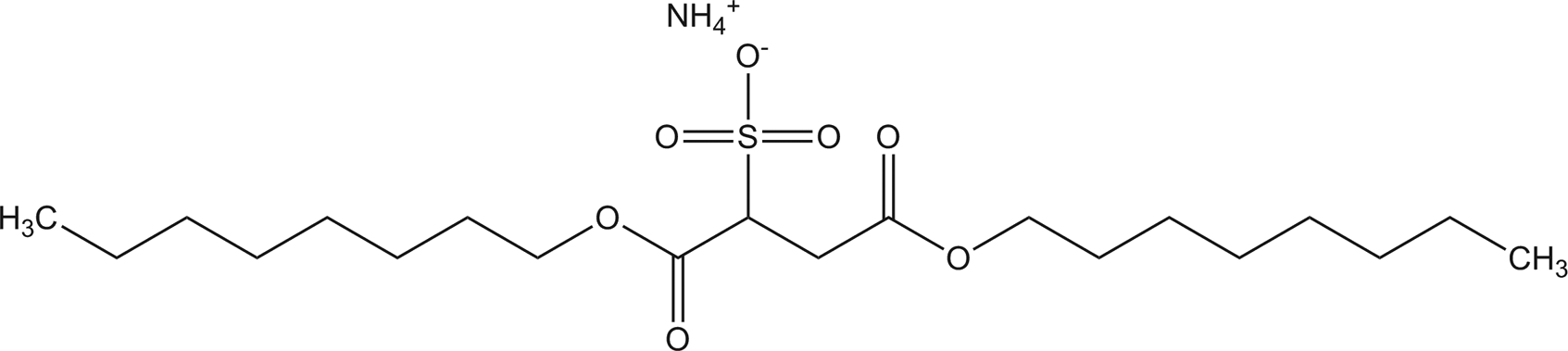
Diethylhexyl sodium sulfosuccinate 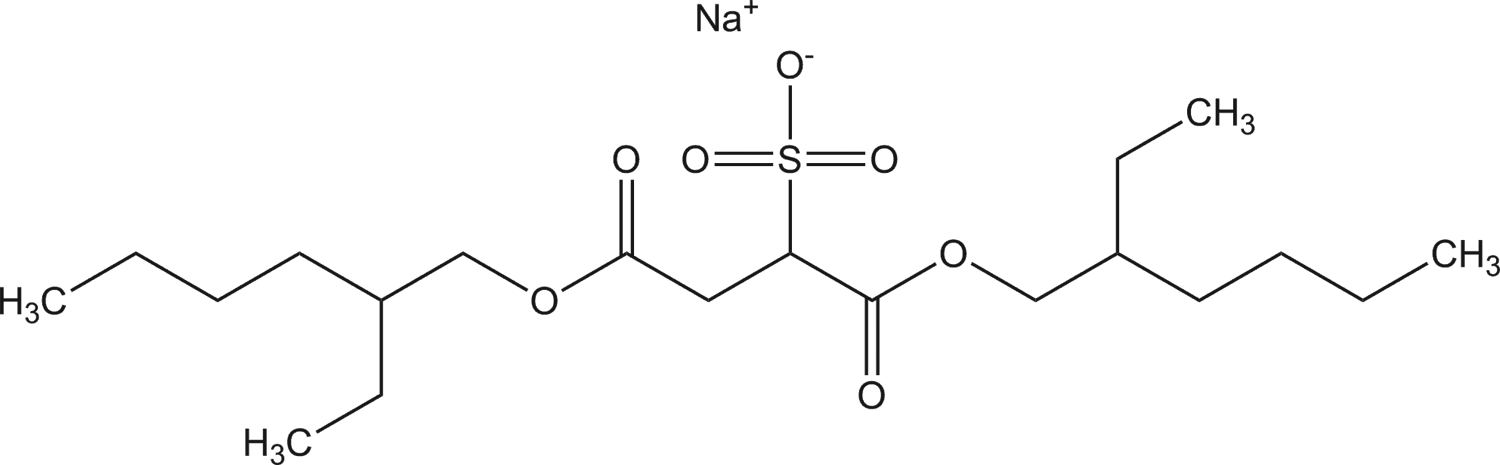
Diheptyl sodium sulfosuccinate 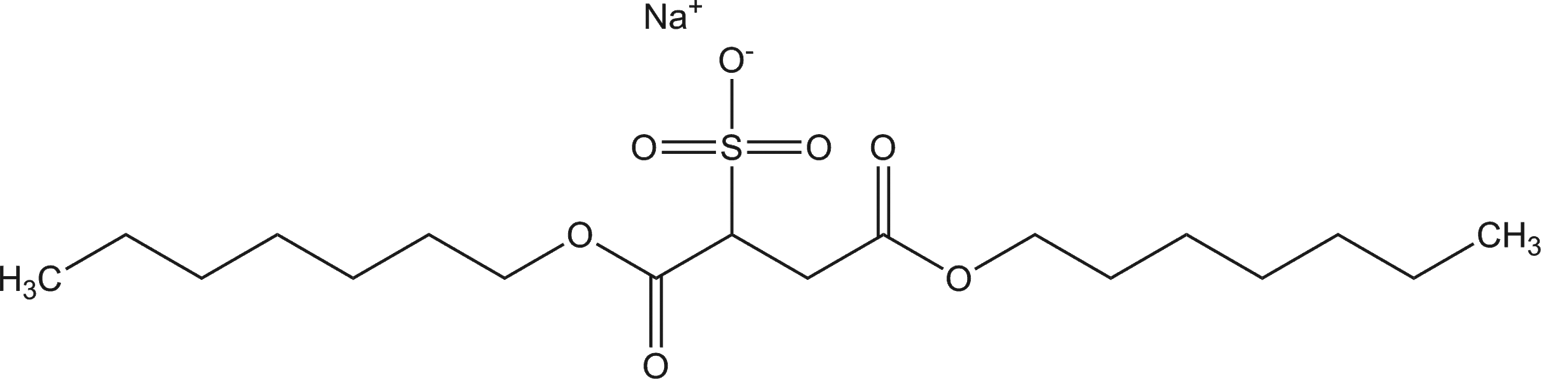
Dihexyl sodium sulfosuccinate 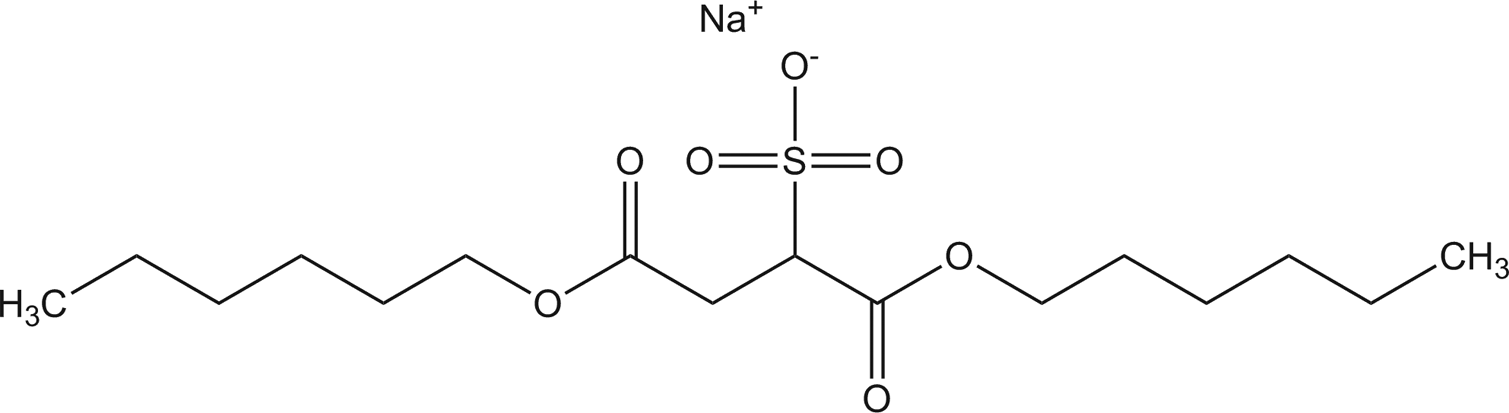
Diisobutyl sodium sulfosuccinate 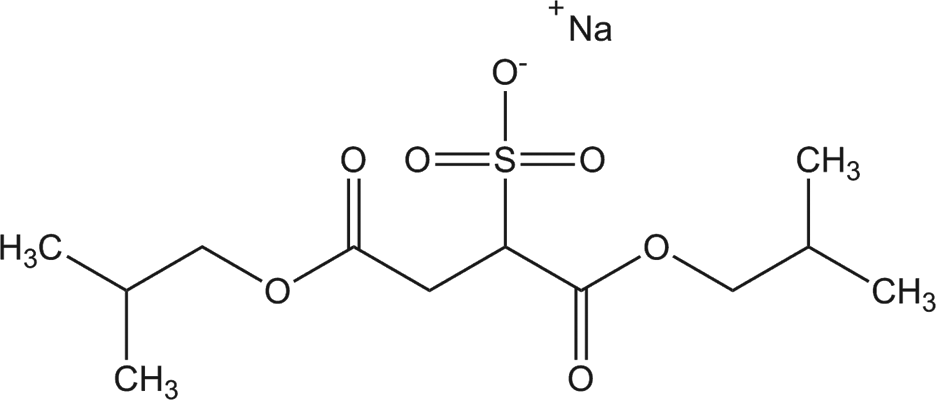
Ditridecyl sodium sulfosuccinate 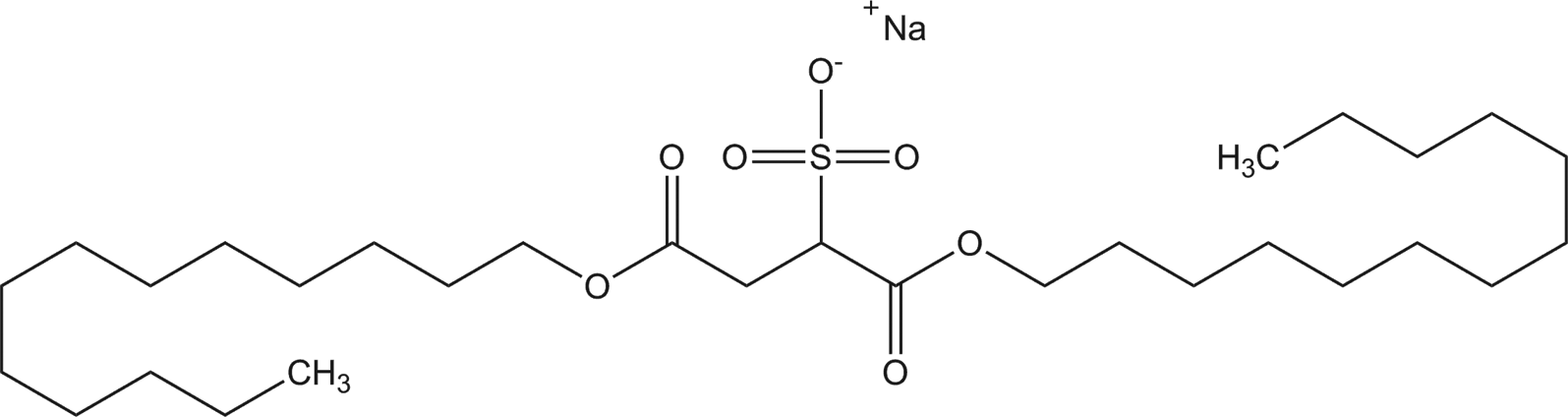
Physical and Chemical Properties
Little published data on physical and chemical properties were found. The data that were available are provided in Table 4.
Physical and Chemical Properties.

Method of Manufacture
Diethylhexyl sodium sulfonate
Data available from a previous CIR safety assessment on the method and manufacture of diethylhexyl sodium sulfosuccinate are included in Table 1.
In the production of diethylhexyl sodium sulfosuccinate, malic acid and 2-ethylhexanol are reacted to form the diester, which is sulfonated using sodium metabisulfite. 4 The reaction takes place in a closed system that is opened only for the addition of the reactants.
Dialkyl sodium sulfosuccinate
The dialkyl sodium sulfosuccinates are prepared by the action of the appropriate alcohols on maleic anhydride followed by the addition of sodium bisulfite. 5 –7
Impurities
Diethylhexyl sodium sulfosuccinate
The Food Chemicals Codex has the following acceptance criteria for diethylhexyl sodium sulfosuccinate: not less than (NLT) 98.5% C20H37NaO7S, not more than (NMT) 2 mg/kg lead, NMT 0.2% bis(2-ethylhexyl)maleate, NMT 2.0% loss on drying, and 15.5% to 16.2% residue on ignition. 8 The United States Pharmacopeia acceptance criteria are NLT 99.0% and NMT 100.5% C20H37NaO7S calculated on an anhydrous basis, NMT 2.0% water, NMT 0.001% heavy metals, NMT 0.4% bis(2-ethylhexyl)maleate, and 15.5% to 16.5% residue on ignition, calculated on an anhydrous basis. 9
Use
Cosmetic
The dialkyl sulfosuccinate salts are reported to function in cosmetics as surfactants (Table 3). 2 The Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2013, 10 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council), 11 indicate that diethylhexyl sodium sulfosuccinate is the only dialkyl sulfosuccinate salt in use.
The current and historical frequency and concentration of use data for diethylhexyl sodium sulfosuccinate are provided in Table 5. The frequency of use increased from use in 38 cosmetic formulations (1995 data) 1 to use in 62 cosmetic formulations (2013 data). 10 The use concentration appears to not have changed. According to a survey conducted by the Council in 2013, the maximum concentration of use reported for diethylhexyl sodium sulfosuccinate is 4.4% in eyebrow pencil formulations 11 ; the 1998 safety assessment stated that although concentration of use data were no longer reported to the FDA, 1984 data indicated that diethylhexyl sodium sulfosuccinate was used in a variety of product types at concentrations of ≤5%.
Current and Historical Frequency and Concentration of Use of Diethylhexyl Sodium Sulfosuccinate According to Duration and Exposure.
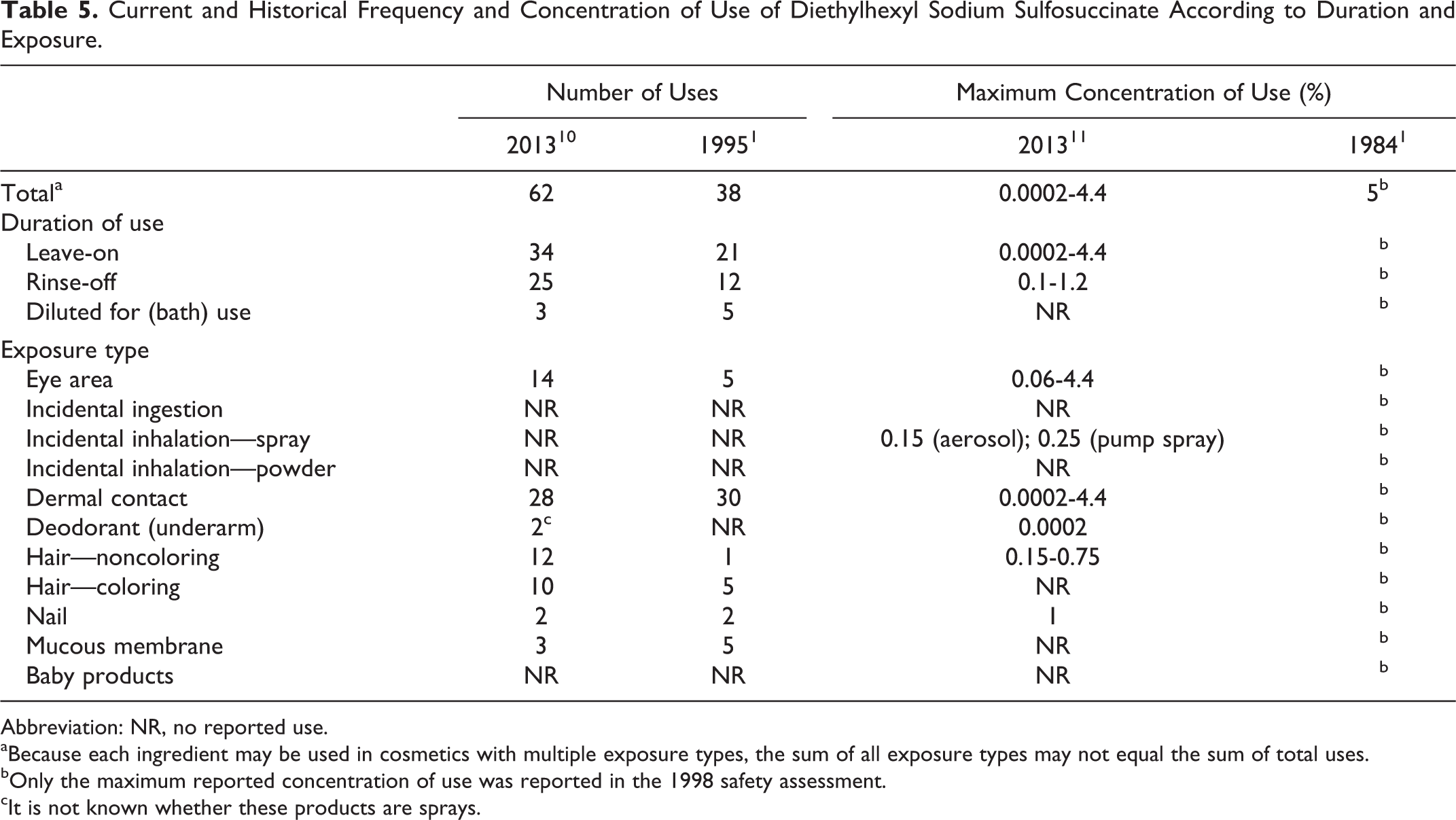
Abbreviation: NR, no reported use.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bOnly the maximum reported concentration of use was reported in the 1998 safety assessment.
cIt is not known whether these products are sprays.
Diethylhexyl sodium sulfosuccinate is used in hair spray formulations at a concentration of 0.15% in an aerosol and at 0.25% in pump spray formulations. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. 12,13 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 14,15
All of the dialkyl sulfosuccinate salts named in this report appear in the European Commission database with information on cosmetic ingredients and substances (CosIng) inventory. 16 Listing in the inventory does not indicate the ingredients are actually used in cosmetic products or approved for such use.
Noncosmetic
Sodium 1,4-dialkyl sulfosuccinates are exempt from the requirement of a tolerance for residues when used as an inert ingredient in pesticide formulations for preharvest and postharvest uses as well as for application to animals under 40 Code of Federal Regulations (CFR) 180.910 and 40 CFR 180.930, respectively. This regulation eliminates the need to establish a maximum permissible level for residues of the sodium 1,4-dialkyl sulfosuccinates.
Diethylhexyl sodium sulfosuccinate
Diethylhexyl sodium sulfosuccinate is generally recognized as safe and effective as a laxative drug product for over-the-counter use (58 FR 46589, September 2, 1993).
Diethylhexyl sodium sulfosuccinate is included in the Listing of Color Additives Exempt From Certification; it is used as a diluent in color additive mixtures for food use exempt from certification and has a limitation of <9 parts per million (ppm, 21CFR 73.1). It is approved as the direct food additive “cocoa with dioctyl sodium sulfosuccinate for manufacturing,” whereby the amount of diethylhexyl sodium sulfosuccinate does not exceed 75 ppm of the finished beverage (21CFR 172.520). Diethylhexyl sodium sulfosuccinate is also allowed as a multipurpose food additive when it meets the specifications of the Food Chemicals Codex (21CFR172.810). With use as an emulsifier, the Joint FAO/WHO Expert Committee on Food Additives has established an acceptable daily intake of 0 to 0.1 mg/kg of body weight (bw). 17
Diethylhexyl sodium sulfosuccinate is approved for the following that are used as an indirect food additive: in adhesives (21CFR175.105); in resinous and polymeric coatings (21CFR175.300); in resinous and polymeric coatings for polyolefin films (21 CFR 175.320); as a component of paper and paperboard in contact with aqueous and fatty foods (21 CFR 176.170); in defoaming agents used in the manufacture of paper and paperboards (21 CFR 176.210); in cellophane (21 CFR 177.1200); in polymers in textile and textile fibers (21 CFR 177.2800); in sanitizing solutions for use on food-contact articles (21 CFR 178.1010); and in emulsifiers and/or surface-active agents in adjuvants, production aids, and sanitizers (21 CFR 178.3400).
Diamyl, dihexyl, and diisobutyl sodium sulfosuccinate
Diamyl, dihexyl, and diisobutyl sodium sulfosuccinate are used as wetting agents, and diamyl sodium sulfosuccinate is used as an emulsifier in emulsion polymerization. 5 –7
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
The metabolism and excretion of diethylhexyl sodium sulfosuccinate was determined in rats in several studies; limited details were available. Albino rats were given a single oral dose of 50 mg/kg bw [35S]diethylhexyl sodium sulfosuccinate in an alcohol and water (1:1) solution. 18 More than 85% of the diethylhexyl sodium sulfosuccinate was excreted within 24 to 48 hours after dosing, and all were excreted within 96 to 120 hours. The majority of the radioactivity, 66%, was excreted in the feces. Only 25% to 35% of the dose was excreted in the urine, and that was within 24 to 48 hours after dosing. At 96 to 168 hours after dosing, only trace amounts of radioactivity were found in the tissues.
However, in other studies, the feces were not the primary route of excretion. In a study in which 2 rats were given a single oral dose of 5 or 10 mg diethylhexyl sodium sulfosuccinate in water, and 2 rats were given a single intravenous (IV) dose of 10 mg diethylhexyl sodium sulfosuccinate, the animals dosed orally with 5 and 10 mg excreted 18.6% and 15.5% of the total dose and the animals dosed IV excreted 12.3% to 15.5% of the dose in the urine in 24 hours. 18 The rats dosed orally excreted 0.9% and 8.7% of the dose in the feces in this time period; however, the animals that were dosed intravenously did not excrete any of the dose in the feces. The 24- to 48-hour urine samples were analyzed for 2-ethylhexanol and no detectable levels were found (use of radiolabel was not specified).
In a study in which a male rat was dosed by gavage with 10 mg/kg bw [14C]diethylhexyl sodium sulfosuccinate, 64.1% of the radioactivity was excreted in the urine and 37.4% in the feces in the first 24 hours, and then only approximately 1% in the urine and 0.9% in the feces in the next 24 hours. 18 The researchers stated that diethylhexyl sodium sulfosuccinate must undergo extensive metabolism in the rat because no unchanged diethylhexyl sodium sulfosuccinate was found in the urine, and only a small amount was present in the feces.
Metabolism and excretion was also determined in rabbits and dogs; as with the rat studies, limited details were available. One female rabbit and 1 male beagle dog were each given a single oral dose, and 1 of each species was given a single IV dose of 4 mg [14C]diethylhexyl sodium sulfosuccinate. 18 In the rabbits, within 24 hours, 87% and 69.7% of the radioactivity was excreted in the urine following oral and IV dosing, respectively, and similar patterns of metabolites were found with both routes of administration.
In the dogs, similar excretion patterns and metabolic profiles were observed for both routes of dosing. Approximately 21% of the radioactivity was excreted in the urine in the first 24 hours. The majority of the radioactivity, approximately 70%, was excreted in the feces at 24 to 48 hours postdosing. Blood samples were analyzed for 2-ethylhexanol compounds; with IV administration, the blood levels fell rapidly during the first hour, and none was found after 8 hours. Similarly, following oral administration, small amounts of 2-ethylhexanol was found in the blood after 1 hour, and none was found after 8 hours.
Penetration Enhancement
Surfactants can enhance the permeation rate of various compounds, inducing a concentration-dependent biphasic action with respect to altering skin permeability. 19 Surfactant molecules must diffuse through the lipid region of the stratum corneum in order to interact with the deeper protein-rich areas. Anionic surfactants can solubilize the less-soluble protein, or they can remain on the skin due to the formation of chemical compounds with skin keratin, and they can interact strongly with both keratin and lipids. If exposure time is short, permeation through the stratum corneum by anionic materials is generally poor; however, permeation increases with a longer exposure time.
The effect of a diethylhexyl sodium sulfosuccinate microemulsion on the distribution of the polyphenols curcumin and resveratrol between the epidermis and dermis was examined in excised guinea pig and Yucatan micropig (YMP) skin. 20 The microemulsion consisted of 150 mM saline solution, isopropyl palmitate, diethylhexyl sodium sulfosuccinate, and ethanol, with a weight ratio of 20.2:31.3:33.3:15.2, and the mean particle size was 16.6 ± 1.8 nm. Franz-type diffusion cells were used, and 0.5 mL (guinea pig skin) or 1 mL (YMP skin) of the vehicle containing each polyphenol was added to the donor compartment as saturated concentration; the available diffusion area was approximately 0.62 cm2. Vehicles consisting of a Tween 80 microemulsion or isopropyl myristate were also evaluated. Treatment time was 20 hours for guinea pig skin and 40 hours for YMP skin. The accumulation of the polyphenols in guinea pig and YMP skin was statistically significantly increased using diethylhexyl sodium sulfosuccinate microemulsion as the vehicle, as compared to that found with the Tween 80 microemulsion or isopropyl myristate. Approximately 1.7% curcumin and 2.2% resveratrol added to donor compartments were incorporated into the skin by the diethylhexyl sodium sulfosuccinate microemulsion. Skin accumulation of curcumin in the diethylhexyl sodium sulfosuccinate microemulsion was approximately 1.9 µmol/g skin in guinea pig skin and approximately 0.24 µmol/g skin in YMP skin; in the isopropyl myristate vehicle, almost no curcumin accumulated in either skin type. Skin accumulation of resveratrol in the microemulsion was approximately 12 µmol/g skin in guinea pig skin and approximately 3 µmol/g skin in YMP skin; in the isopropyl myristate vehicle, approximately 1 µmol/g skin accumulated in guinea pig skin and 0.1 µmol/g accumulated in YMP skin. In determining the distribution in guinea pig and YMP skin, it was found that diethylhexyl sodium sulfosuccinate, curcumin, and resveratrol penetrated deep into the skin. In YMP skin, the distribution ratio of the polyphenols between the dermis and epidermis decreased with increased molecular weight.
Toxicological Studies
Single-Dose (Acute) Toxicity
Dermal
The dermal median lethal dose (LD50) of undiluted diethylhexyl sodium sulfosuccinate in rabbits was >10g/kg. 4 Occlusive patches of 10 g/kg of the test material were applied to the clipped, unabraded, skin of 5 male New Zealand white rabbits. Skin fissuring, desquamation, and coriaceousness were observed.
Oral
Data from the original safety assessment on single-dose oral toxicity of diethylhexyl sodium sulfosuccinate are available in Table 1.
The oral LD50 of diethylhexyl sodium sulfosuccinate in 4% acacia was 2.64 g/kg bw in male albino ARS/ICR mice. 21 In guinea pigs, the oral LD50 was approximately 0.65 g/kg bw aqueous (aq.) diethylhexyl sodium sulfosuccinate. 22
Repeated-Dose Toxicity
Dermal
Repeated-dose dermal toxicity data from the original safety assessment on diethylhexyl sodium sulfosuccinate are available in Table 1.
Oral
Repeated-dose oral toxicity data from the original safety assessment on diethylhexyl sodium sulfosuccinate are available in Table 1.
A group of 20 male and 20 female albino rats were fed a diet containing 1% diethylhexyl sodium sulfosuccinate (100% pure) for 90 days, and controls were given untreated feed. 4 All animals survived until study termination. There were no clinical signs of toxicity, and no dosing-related macroscopic or microscopic findings. Differences in body weights or organ weights compared to controls were not statistically significant.
Twelve rats/group were fed a diet containing 0%, 0.5%, 1.04%, or 1.5% diethylhexyl sodium sulfosuccinate for 26 weeks. 23 Body weight gains of females of the 1.04% and 1.5% dose groups were decreased during week 3. Two control animals and 4 animals of the 1.5% group died during the study; 2 of the 4 animals of the 1.5% group had hemorrhagic gastroenteritis. No other effects were noted. The no-observable adverse effect level (NOAEL) was 0.5%, and the lowest-observable adverse effect level was 1.04%.
Groups of 4 male and 4 female beagle dogs were dosed orally with tablets containing 30 mg/kg bw diethylhexyl sodium sulfosuccinate, 10 mg/kg bw diethylhexyl sodium sulfosuccinate + 5 mg/kg bw 1,8-dihydroxyanthraquinone, or 30 mg/kg bw diethylhexyl sodium sulfosuccinate, and 15 mg/kg bw 1,8-dihydroxyanthraquinone, daily, for 1 year. 21 A control group was given a placebo tablet. Urinalysis was performed, and hematological and clinical chemistry parameters were measured at various intervals. No signs of toxicity were observed in any of the groups. Diethylhexyl sodium sulfosuccinate, alone and in combination with 1,8-dihydroxyanthraquinone, did not have any adverse effects on urinalysis, hematological or clinical parameters, or body weights, and it did not induce any gross or microscopic lesions. The NOAEL was >30 mg/kg bw.
Inhalation
Repeated-dose inhalation toxicity data from the original safety assessment on diethylhexyl sodium sulfosuccinate are available in Table 1.
Fluorescent latex particles, 0.63-µm diameter, were administered in aerosol form to 30 rabbits. 24 Six rabbits were killed immediately after administration of the fluorescent particles (baseline group); 12 rabbits were given a diethylhexyl sodium sulfosuccinate aerosol prepared as a 2% solution in equal volumes of ethanol and physiological saline (detergent group) and 12 were given vehicle aerosol (control group). The detergent and control aerosols were administered as 200 pressure-controlled breaths at a frequency of 40/min, resulting in deposition of approximately 10 µL of fluid in the lungs; aerosol administration was repeated after 90 minutes. Groups of 6 animals from the detergent and control groups were then exposed to large tidal volume ventilation (LTVV) or conventional ventilation for 3 hours. The total number of particles in the alveoli and ducts were similar for all groups, except for a statistically significant decrease in the control LTVV group. All test groups had reduced number of single particles in the alveoli as compared to the baseline group. The number of clustered particles was statistically significantly increased in the alveoli + ducts in the detergent LTVV group, as compared to the baseline group.
Rabbits were administered [99mTc]diethylene triamine pentaacetate (99mTc-DTPA) using a nebulizer and the effect of diethylhexyl sodium sulfosuccinate on the absorption of this compound from the lungs was examined. 25 The alveolocapillary transfer of 99mTc-DTPA was measured for 30 minutes, and the rabbits were then nebulized with 0.2% solution of diethylhexyl sodium sulfosuccinate for 5 minutes. Thirty minutes later, the rabbits were nebulized with a 2% diethylhexyl sodium sulfosuccinate solution for 5 minutes. Diethylhexyl sodium sulfosuccinate greatly enhanced the alveolar absorption of 99mTc-DTPA.
Ocular Irritation
Refer to Table 1 for a summary of ocular irritation data from the original safety assessment on diethylhexyl sodium sulfosuccinate. Diethylhexyl sodium sulfosuccinate, 0.1 g, was instilled into the conjunctival sac of the eyes of 6 rabbits. 4 The eyes were scored for irritation after 24, 48, and 72 hours, and the following scores were reported: 11.66, 12.50, and 4.16, respectively (cornea); 1.66 at all 3 times (iris); and 5.33, 4.33, and 1.66, respectively (conjunctivae). No destruction or irreversible changes of the tissue in 24 hours were reported.
Diethylhexyl sodium sulfosuccinate, 10% (vehicle not specified), was used as a positive control in a Draize eye irritancy test. 26 One-tenth micro liter of the test substance was instilled into the conjunctival sac of 1 eye of each of 3 rabbits for 2 seconds; the eyes were rinsed. Diethylhexyl sodium sulfosuccinate, 10%, was severely irritating to rabbit eyes, inducing perforated damages. Diisobutyl sodium sulfosuccinate was irritating to eyes and mucous membranes 7 (details were not provided).
Reproductive and Developmental Toxicity
A summary of reproductive developmental toxicity data from the original safety assessment on diethylhexyl sodium sulfosuccinate are available in Table 1. In developmental toxicity studies, groups of 20 gravid female mice and 20 gravid female rats were dosed by gavage with 0, 16, 80, or 400 mg/kg bw of a test substance containing 0.4% (w/v) diethylhexyl sodium sulfosuccinate. 4 The mice were dosed on days 6 to 15 and killed on day 17 of gestation and the rats were dosed on days 5 to 19 of gestation and killed on day 20 of gestation. The NOAEL for maternal toxicity and teratogenic effects for both mice and rats was 400 mg/kg bw of the test substance containing 0.4% (w/v) diethylhexyl sodium sulfosuccinate.
Groups of 20 to 39 gravid female Sprague Dawley rats were fed a diet containing 0%, 1%, or 2% diethylhexyl sodium sulfosuccinate (equivalent to 0, 1,074, and 1,983 mg/kg bw, respectively) on days 6 to 15 of gestation, and the dams were killed on day 21 of gestation. 4 No adverse effects on maternal or fetal parameters were observed in the 1% test group. In the 2% test group, significant incidences of resorptions and gross abnormalities, primarily exencephaly and, at times, spina bifida, anophthalmia, and associated skeletal defects, were reported. The NOAEL for maternal toxicity and teratogenic effects was 1%.
Groups of 30 female rats were dosed by gavage with 0, 16, 80, or 400 mg/kg bw of a test substance containing 0.4% (w/v) diethylhexyl sodium sulfosuccinate once daily for 14 days prior to mating with untreated males; one-half of the animals in each group were dosed until day 13 of gestation, at which time the animals were killed, and the remaining animals were dosed until parturition and were not killed. 4 No effects on reproductive parameters, fertility, or pup weight and condition were observed. The parental NOAEL was 400 mg/kg bw of the test substance containing 0.4% (w/v) diethylhexyl sodium sulfosuccinate.
A 3-generation study was performed in which male and female CFE rats were continuously fed a diet containing 0.5% and 1% of a test substance containing 50% diethylhexyl sodium sulfosuccinate in aq. beverage-grade ethanol; the control group was untreated. 4 The number of animals per group was not stated. Dosing was initiated at weaning of rats of the F0 generation; these rats were mated twice to produce the F1a and F1b generation. Rats of the F1b generation were mated to produce the F2 generation, and the F2 generation was mated twice to produce the F3a and F3b offspring. F1a and F3b offspring were the only pups weaned directly to the test diets. Because of a high incidence of pup mortality, all other dams were given a control diet on the last expected day of gestation. Necropsy and microscopic examination were performed only on pups from the first mating of the F2 animals that died or were killed at weaning.
Until the F2 generation, body weights in parental males were 6% to 10% lower than control body weights. There were no significant treatment-related effects on mean litter size and the mean number of viable pups in each litter or on fertility or gestational indices. For all pups of the F1a generation, including controls, the number of pups weaned and the average body weight of those pups at weaning was reduced; however, greater reductions were seen in the test groups than in the control group. The viability indices of the F3b pups receiving the test diet were reduced. The researchers stated the most remarkable result of the study was the reduced number of offspring surviving from day 5 until weaning; it was hypothesized that pups stopped nursing because they could taste the test article. A no-observed effect level (NOEL) for parental toxicity and effects on pups was not established; the NOEL for reproduction was 1%.
Genotoxicity
Summary genotoxicity data from the original safety assessment on diethylhexyl sodium sulfosuccinate are available in Table 1.
Carcinogenicity
Effect on Colorectal Carcinogenesis
A group of 84 inbred male F344 rats was fed a diet containing 1% diethylhexyl sodium sulfosuccinate, and the control group was fed untreated feed. 27 As part of a rodent model for colon carcinogenesis, rats of both groups were given a subcutaneous injection of 20 mg/kg bw of 1,2-dimethylhydrazine, once weekly for 20 weeks. Twenty rats per group were killed after 3, 4, 5, and 6 months. The test group tolerated the diethylhexyl sodium sulfosuccinate feed well. There was no statistically significant difference between the test and control group in the percentage of rats bearing tumors, and the number of tumors per rat increased progressively throughout the study. However, at 5 and 6 months, each rat in the test group had fewer tumors of all histologic types (combined), at all organ sites, compared to controls; this difference was statistically significant for the duodenum, colon, rectum, and total number of gastrointestinal tumors at 5 months.
Irritation and Sensitization
Dermal Irritation and Sensitization
Nonhuman dermal irritation and sensitization data and human dermal irritation and sensitization data from the original safety assessment on diethylhexyl sodium sulfosuccinate are available in Table 1.
Nonhuman
Occlusive patches containing 0.5 mL diethylhexyl sodium sulfosuccinate were applied to intact and abraded skin of 6 rabbits; the duration of exposure was not stated. 4 For intact skin, the mean Draize scores for erythema and edema were 2.33 and 2.50, respectively, after 24 hours and 1.66 and 1.0, respectively, after 72 hours. For abraded skin the mean scores for erythema and edema were 2.50 and 2.50, respectively, after 24 hours and 1.66 and 1.60, respectively, after 72 hours.
Human
Diethylhexyl sodium sulfosuccinate produced irritation, but it was not a sensitizer. 4 For induction, a 15-mm occlusive patch containing 0.30 g of 2.5% ethylhexyl sodium sulfosuccinate in petrolatum was applied to the backs or forearms of 100 participants; the patches were applied for 10 alternate 24-hour periods. Challenge patches containing 0.30 g diethylhexyl sodium sulfosuccinate were applied to a previously untreated site on the back or forearm following a 7-day nontreatment period. The challenge sites were scored upon patch removal and 24 hours later. During induction, the following observations were made: mild erythema in 11 participants on days 3 to 10 and in 1 participant on days 3 to 7; mild erythema on all days except day 7 and intense erythema on day 7 in 1 participant; mild erythema on days 3 to 6/7 followed by intense erythema on days 6/7 to 10 in 6 participants. No reactions were observed at challenge sites.
In a case report, a female participant had allergic contact dermatitis from diethylhexyl sodium sulfosuccinate that was an ingredient in a topical corticosteroid. 28 In patch testing, the patient had a +++ reaction to 1% aq. diethylhexyl sodium sulfosuccinate on days 2 and 4. The researchers noted that this was a rare reaction.
Phototoxicity/Photoallergenicity
Human
Human photo toxicity data from the original safety assessment on diethylhexyl sodium sulfosuccinate are available in Table 1.
Summary
Diethylhexyl sodium sulfosuccinate (previously named dioctyl sodium sulfosuccinate), an anionic surfactant, was reviewed by the CIR Expert Panel in 1994, and the report was amended in 1998. In 1998, the Panel concluded that diethylhexyl sodium sulfosuccinate is safe as used in cosmetic formulations. Since the 1998 report was issued, the number of reported uses in cosmetic formulations has increased from 35 to 62 uses. However, the concentration of use has not changed. The data that were available for the 1998 report indicated that diethylhexyl sodium sulfosuccinate was used in a variety of product-types at concentrations of ≤5%; current information report that the maximum use concentration is 4.4% in eyebrow pencil formulations.
The Panel has determined that the data included in the original safety assessment, as well as in this rereview document, support the safety of an additional 7 dialkyl sulfosuccinate salts. These salts, which are diesters of 2-sulfosuccinic acid, all share a sulfo-substituted succinic acid core; all contain 2 ester linkages, and are theoretically sensitive to hydrolysis, especially under acidic conditions; and all are anionic surfactants.
Metabolism and excretion studies have given mixed results on the primary route of excretion of diethylhexyl sodium sulfosuccinate; it does appear that diethylhexyl sodium sulfosuccinate is metabolized prior to excretion, and most of the dose is excreted within 24 hours of dosing. In 1 oral study in rats, 66% of the radioactivity (labeled with [35S]) was excreted in the feces and only 25% to 35% in urine, within 24 to 48 hours after dosing. In other rat studies, with oral and IV administration, the majority of the radioactivity (radiolabel not specified) was excreted in the urine, rather than in the feces. Studies were also performed in rabbits and dogs in which diethylhexyl sodium sulfosuccinate was labeled with [14C], and again conflicting results were obtained. In rabbits, 87% and 69.7% of the radioactivity was excreted in the urine following oral and IV dosing, respectively; in dogs, approximately 70% of the radioactivity was excreted in the feces at 24 to 48 hours after oral and IV dosing.
Diethylhexyl sodium sulfosuccinate increased the penetration of curcumin and resveratrol, in vitro, through excised guinea pig and YMP skin. The dermal LD50 of undiluted diethylhexyl sodium sulfosuccinate in rabbits was >10 g/kg; skin irritation was observed following the single dermal dose of 10 g/kg test material. The oral LD50 was 2.64 g/kg bw in male albino ARS/ICR mice and approximately 0.65 g/kg bw in guinea pigs.
In repeated-dose oral studies in which rats were given a feed containing 1% diethylhexyl sodium sulfosuccinate for 90 days or up to 1.5% for 26 weeks, and in studies in which beagle dogs were given tablets containing 30 mg/kg bw/d diethylhexyl sodium sulfosuccinate for 1 year, no remarkable toxic effects were reported. In an inhalation study in rabbits, a 5-minute exposure to 0.2% diethylhexyl sodium sulfosuccinate, followed 30 minutes later by a 5-minute exposure to 2% diethylhexyl sodium sulfosuccinate, greatly enhanced the alveolar absorption of 99mTc-DTPA. Diethylhexyl sodium sulfosuccinate was used as a positive control in a Draize ocular irritation study; 10% diethylhexyl sodium sulfosuccinate was severely irritating to rabbit eyes, inducing perforated damages.
Numerous studies examining the effect of the oral administration of diethylhexyl sodium sulfosuccinate, both dietary and by gavage, on the reproductive and developmental toxicity in rats were performed; 1 study was performed in mice. In a developmental study in mice and rats of a test substance containing 0.4% (w/v) diethylhexyl sodium sulfosuccinate, the NOAEL for maternal toxicity and teratogenic effects for both mice and rats was 400 mg/kg bw. In another developmental toxicity study in rats, the parental NOAEL was 400 mg/kg bw for a test substance containing 0.4% (w/v) diethylhexyl sodium sulfosuccinate. In a study in which gravid female Sprague Dawley rats were fed a diet containing up to 2% diethylhexyl sodium sulfosuccinate, no adverse effects on maternal or fetal parameters were observed in the 1% test group, but in the 2% test group, significant incidences of resorptions and gross abnormalities, primarily exencephaly and, at times, spina bifida, anophthalmia, and associated skeletal defects, were reported. The NOAEL for maternal toxicity and teratogenic effects was 1%. In a 3-generation study in which rats were fed a diet containing up to 1% of a test substance containing 50% diethylhexyl sodium sulfosuccinate in aq. beverage-grade ethanol, a NOEL for parental toxicity and effects on pups was not established because of reduced body weight gains in the parents and reduced viability indices in the pups, but the NOEL for reproduction was 1%; the reduced viability index most likely was attributed to the pups discontinuing nursing because they could taste the test article.
In rats, a diet containing 1% diethylhexyl sodium sulfosuccinate did not have an effect on 1,2-dimethylhydrazine-induced colorectal carcinogenesis. In clinical studies, 2.5% diethylhexyl sodium sulfosuccinate was an irritant, but not a sensitizer.
Discussion
The Expert Panel determined that the existing safety assessment on diethylhexyl sodium sulfosuccinate should be expanded to include the 7 dialkyl sulfosuccinate salts that are listed in the International Cosmetic Ingredient Dictionary and Handbook. Although data were not available on most of these additional ingredients, the Panel found the existing data on diethylhexyl sodium sulfosuccinate are sufficient to support the safety of this entire family of ingredients, stating that diethylhexyl sodium sulfosuccinate is a reasonable representative of all of the diesters. All of the diesters are of a similar alkyl chain length, all are symmetrically substituted, and all have similar functions in cosmetic formulations. Additionally, these esters are not expected to be absorbed through the skin to any significant extent, and the reproductive effects observed in test animals orally exposed to diethylhexyl sodium sulfosuccinate are not likely effects of topical application of cosmetics containing these ingredients. Furthermore, there were no uses reported by which incidental ingestion would occur. Consistent with this view, the Panel noted that acute dermal toxicity of undiluted diethylhexyl sodium sulfosuccinate was quite low, with a dermal LD50 of >10 g/kg in rabbits.
The Panel recognized that the dialkyl sulfosuccinate salts may enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients, whose safety was based on their lack of dermal absorption data or when dermal absorption was a concern.
In addition, the Panel confirmed its original discussion, acknowledging that under the exaggerated exposure conditions of the 2 repeated insult patch tests (continuous occlusive patch testing) presented in the original safety assessment of sodium diethylhexyl sulfosuccinate, the ingredient is a cumulative irritant, though not a sensitizer. The Panel recognized that a surfactant would most likely produce irritation under such conditions, and therefore, specified that products containing dialkyl sulfosuccinate salts must be formulated to be nonirritating.
Finally, the Panel discussed the issue of incidental inhalation exposure from hair sprays. The limited data available from short-term pharmaceutical studies in test animals exposed to diethylhexyl sodium sulfosuccinate aerosols suggest little potential for respiratory effects. This ingredient is reportedly used at concentrations up to 0.25% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract in these small amounts present no toxicological concerns based on the chemical properties and biological properties of this ingredient. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for the dialkyl sulfosuccinate salts to cause systemic toxicity, irritation, sensitization, reproductive and developmental toxicity, genotoxicity, and carcinogenicity. They noted the lack of systemic toxicity in several acute and subchronic oral exposure studies, little or no irritation or sensitization in tests of dermal and ocular exposure, the absence of genotoxicity in Ames tests, and the lack of carcinogenicity in a subchronic oral exposure study. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that the following 8 dialkyl sulfosuccinate salts are safe in the present practices of use and concentration in cosmetics described in this safety assessment when formulated to be nonirritating. Ammonium dinonyl sulfosuccinate* Diamyl sodium sulfosuccinate* Dicapryl sodium sulfosuccinate* Diethylhexyl sodium sulfosuccinate Diheptyl sodium sulfosuccinate* Dihexyl sodium sulfosuccinate* Diisobutyl sodium sulfosuccinate* Ditridecyl sodium sulfosuccinate*
*Ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Washington, DC 20036, USA.
Author Contributions
M. Fiume contributed to conception and design, acquisition, analysis, and interpretation, and drafted the manuscript. B. Heldreth contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript, and critically revised manuscript. L. Gill, F. Alan Andersen, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, analysis and interpretation, and critically revised manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
