Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) rereviewed the safety of 12 isethionate salts as used in cosmetics and concluded that these ingredients are safe in the present practices of use and concentration, when formulated to be nonirritating. These isethionate salts are reported to function mostly as surfactants and cleansing agents in cosmetic products. The Panel reviewed the available animal and clinical data as well as information from previous CIR reports. Although there are data gaps, the shared chemical core structure, expected similarities in physicochemical properties, and similar functions and concentrations in cosmetics enabled grouping these ingredients and reading across the available toxicological data to support the safety assessment of each ingredient.
Keywords
Introduction
As given in the International Cosmetic Ingredient Dictionary and Handbook, these 12 ingredients function mostly as surfactants and cleansing agents in cosmetic products.
1
The ingredients included in this report are as follows: Sodium cocoyl isethionate Ammonium cocoyl isethionate Sodium hydrogenated cocoyl methyl isethionate Sodium isethionate Sodium lauroyl isethionate Sodium lauroyl methyl isethionate Sodium methyl isethionate Sodium myristoyl isethionate Sodium oleoyl isethionate Sodium oleyl methyl isethionate Sodium palm kerneloyl isethionate Sodium stearoyl methyl isethionate
In 1993, Cosmetic Ingredient Review (CIR) published the safety assessment of sodium cocoyl isethionate, with the conclusion “safe for use in cosmetic formulations at 50% in rinse-off products and at 17% in leave-on products.” 2(p. 477) These concentration limits were based on the maximum concentrations reported in safety test data at the time. Sodium cocoyl isethionate functions primarily as a surfactant-cleansing agent and the majority of the uses reported are in coloring and noncoloring hair products. 1,3
Since the original review, a few new studies were published relating to general toxicokinetics and clinical assessment of safety. These new data have been incorporated in this amended safety assessment.
In addition to the original ingredient, sodium cocoyl isethionate, the ingredients ammonium cocoyl isethionate, sodium hydrogenated cocoyl methyl isethionate, sodium isethionate, sodium lauroyl isethionate, sodium lauroyl methyl isethionate, sodium methyl isethionate, sodium myristoyl isethionate, sodium oleoyl isethionate, sodium oleyl methyl isethionate, sodium palm kerneloyl isethionate, and sodium stearoyl methyl isethionate have been added to this safety assessment.
The shared core chemical structure, similar functions and concentrations in cosmetics, and the expected similarities in physicochemical properties enabled grouping these ingredients and reading across the toxicological data to support the safety assessment of each compound in the group. This shared core is expected to carry greater potential to cause any toxic effects that might be associated with exposures to these ingredients, as used in cosmetics, than the alkyl chains of varying lengths and the cations (ie, ammonium or sodium), which are not likely to contribute significantly to toxicity. These cosmetic ingredients include components that have been reviewed previously and were determined by the CIR Expert Panel (Panel) to be safe for use. The conclusions, summary of the findings, and published citations for these previously reviewed ingredients are presented in Table 1.
Constituent Acids With Cosmetic Ingredient Review (CIR) Conclusions.

Toxicological data on sodium isethionate (synonym: sodium 2-hydroxyethanesulphonate) in this safety assessment were obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) by private companies as part of the REACH chemical registration process. These data are available on ECHA’s Web site. 4
Chemistry
Definition and Structure
The definitions, structures, and cosmetic functions of the ingredients addressed in this report are presented in Table 2. These ingredients share a common 2-hydroxyethanesulfonic acid structural core (Figure 1), which has an alcohol moiety at 1 end of a 2-carbon alkyl chain, and a sulfonic acid at the other end (that is in an acid salt form in these ingredients). Sodium isethionate is the cosmetic ingredient name for the sodium salt of 2 hydroxyethanesulfonic acid; the other ingredients are fatty acyl esters formed with 2 hydroxyethanesulfonic acid. These chemicals have the typical structural components of surfactants, with a hydrophobic alkyl tail and a hydrophilic sulfonate anion at the opposite end.
Definitions, Structures, and Functions of Isethionate Salts. 1,a
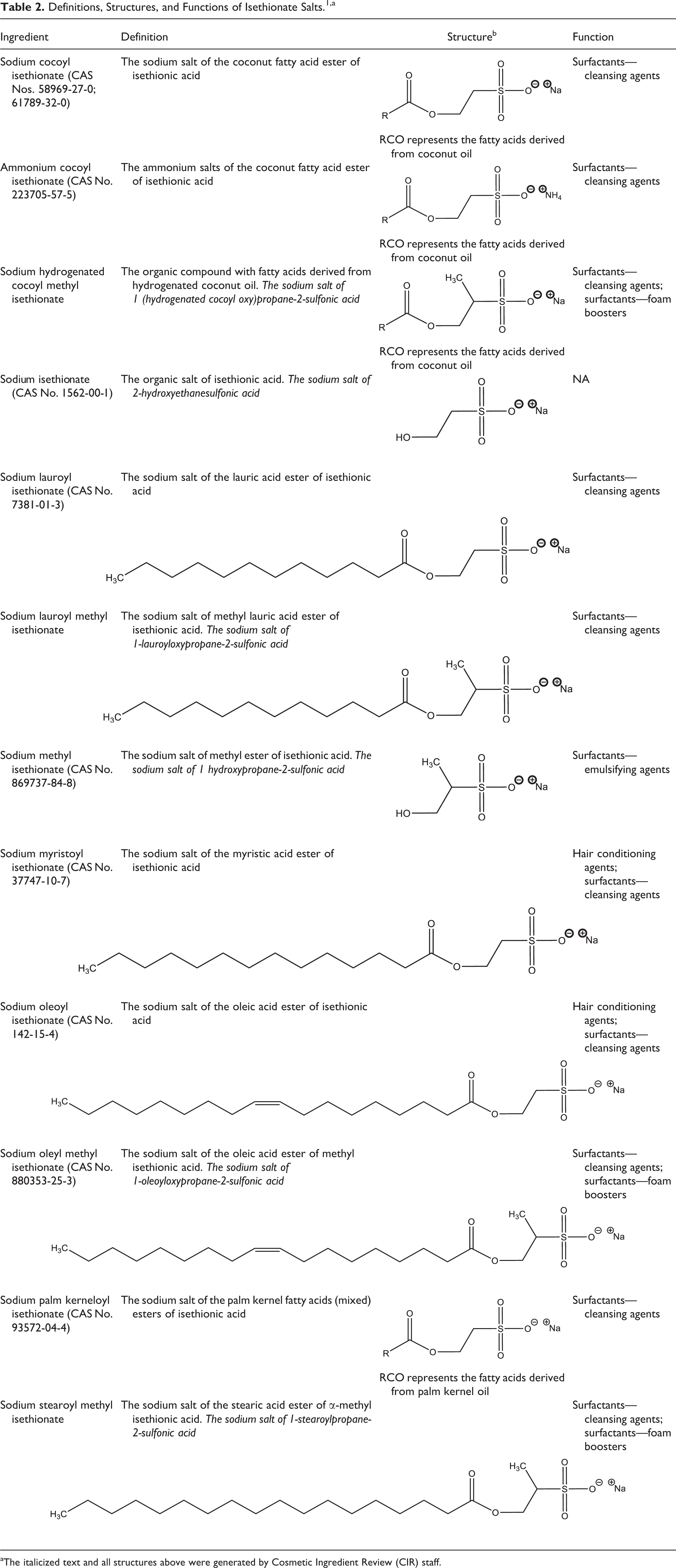
aThe italicized text and all structures above were generated by Cosmetic Ingredient Review (CIR) staff.
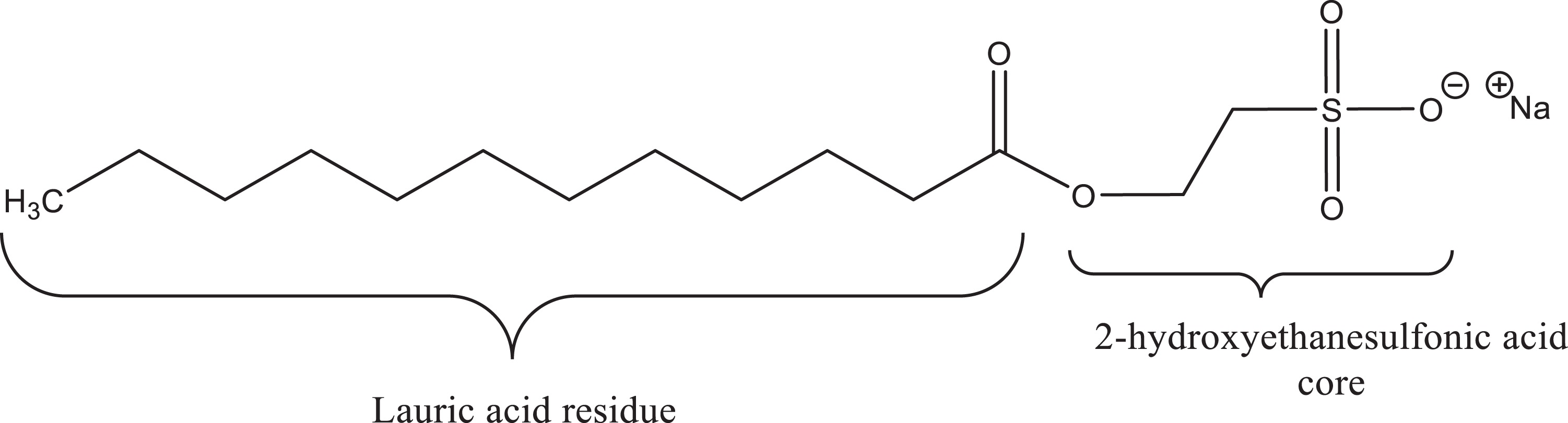
Sodium lauroyl isethionate.
Physical and Chemical Properties
Physical and chemical properties of sodium cocoyl isethionate and sodium isethionate are provided in Table 3. As noted, sodium cocoyl isethionate has limited solubility in water (0.01% by weight at 25°C). Zwitterionic detergents (betaines), alkylamphoacetates, and nonionic sugar surfactants of alkyl glucose esters, aldobionamides, gluconamides, glyceramides, glyceroglycolipids, polyhydroxy fatty acid amides, and alkyl polyglycosides have been used in liquid detergents to increase the solubility of sodium cocoyl isethionate. 5
Physical and Chemical Properties.

Abbreviation: UV, ultraviolet.
Impurities
As reported in the 1993 safety assessment, sodium cocoyl isethionate may contain the following impurities: arsenic (3 ppm max), iron (25 ppm max), lead (20 ppm max), sodium chloride (0.8% max), free fatty matter (10% max), sodium isethionate (5%), free fatty acid (18%), and sodium soap (3%). 2
Use
Sodium cocoyl isethionate is reported to be a surfactant ingredient in mild synthetic detergent (syndet) cleansing bars. 6
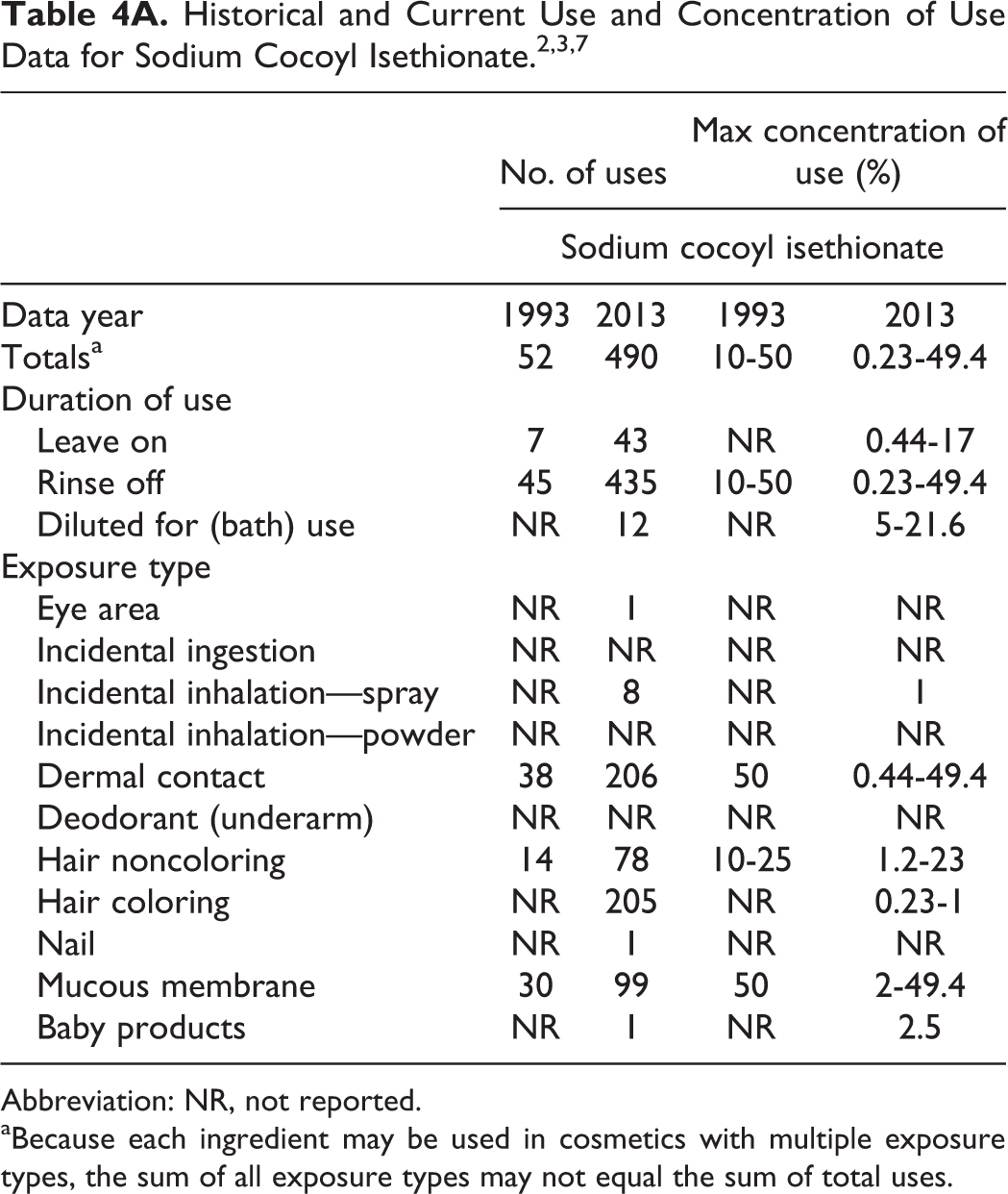
Table 4A presents the available product formulation data for sodium cocoyl isethionate. According to information supplied to the Food and Drug Administration by industry as part of the Voluntary Cosmetic Ingredient Reporting Program (VCRP), sodium cocoyl isethionate was used in a total of 52 cosmetic products at the time of the original safety assessment. Use concentrations ranged from 10% to 50%. 2 Current VCRP data indicate that sodium cocoyl isethionate is now used in at least 490 cosmetic products, with almost half of the uses reported to be in hair dyes and colors. 3 A survey of use concentrations conducted by the Personal Care Products Council in 2013 reported a maximum concentration of use range from 0.23% to 49.4%. 7
Abbreviation: NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
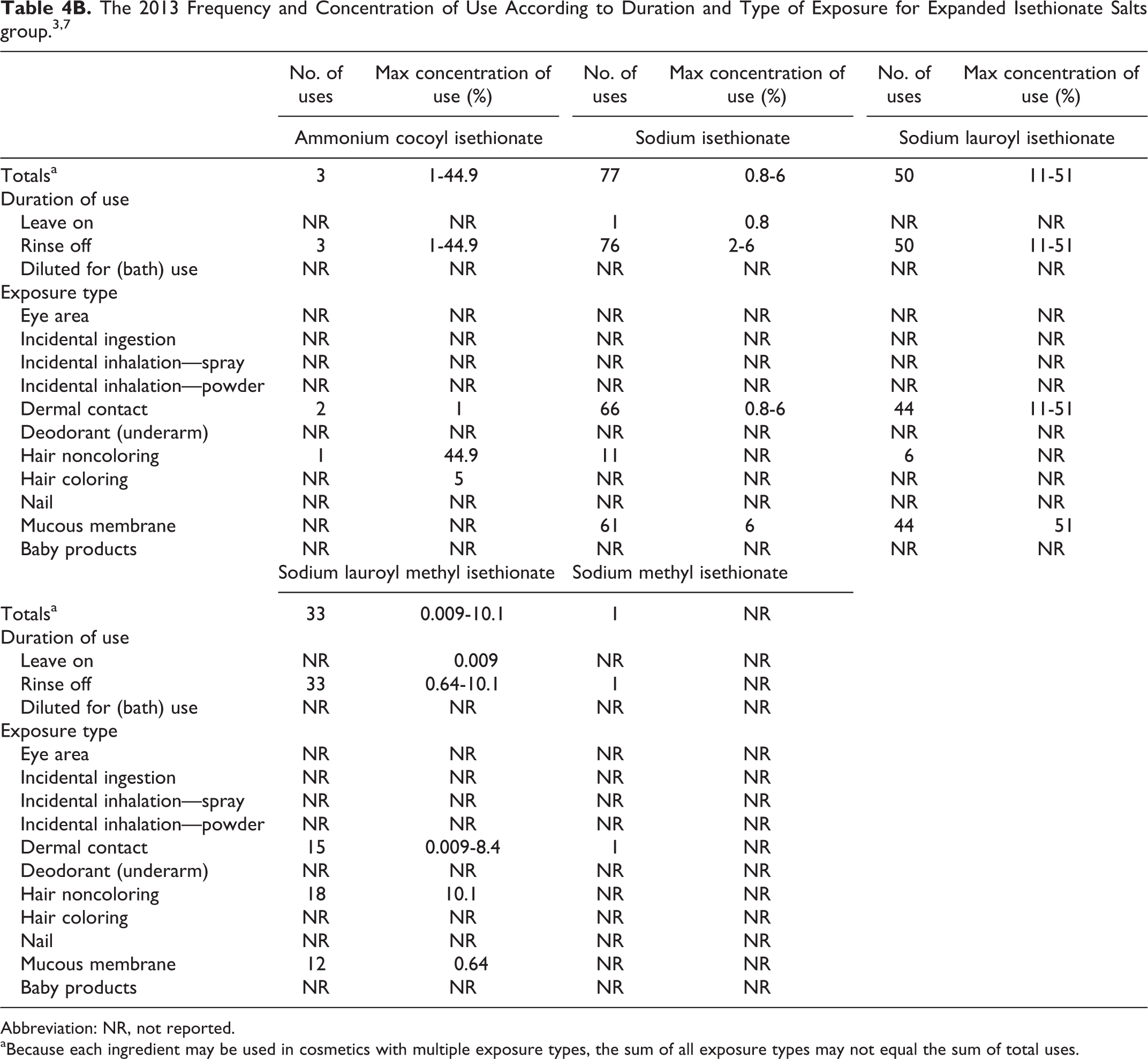
Table 4B presents the 2013 VCRP data and the 2008 use concentration data for the cosmetic ingredients that were added to the sodium cocoyl isethionate safety assessment. Currently, the VCRP database indicates that, of the additional ingredients, sodium isethionate has the most uses (77), with the majority in bath soaps and detergents. 3 The maximum use concentration range for sodium isethionate was 0.8% to 6%, with the 6% reported in bath soaps and detergents. 7
Abbreviation: NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
Those ingredients with no reported uses or use concentrations are listed in Table 4C. Sodium cocoyl isethionate was reported to be used in indoor tanning preparations that may be aerosolized and could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 8 –11 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 9,10 The isethionate salts are not restricted from use in any way under the rules governing cosmetic products in the European Union. 12
Not Reported in Use.

Toxicokinetics
Sodium Cocoyl Isethionate
An in vitro study of the effects of the size of sodium cocoyl isethionate micelles relative to the aqueous pores in the stratum corneum through mannitol skin permeability and average skin electrical resistivity measurements was conducted using female Yorkshire pig skin. 13 A sodium cocoyl isethionate solution (0.2-200 mM) was applied to the skin in vertical Franz diffusion cells for 5 hours. The exposure was in the context of a hindered-transport aqueous pore pathway model of the stratum corneum.
Sodium cocoyl isethionate micelles and the aqueous pores of the stratum corneum had average radii of 33.5
The authors also performed an in vitro quantitative skin radioactivity assay using radiolabeled sodium cocoyl isethionate and pig full-thickness skin. Skin penetration of sodium cocoyl isethionate was concentration dependent in a manner consistent with the effects of micelle formation. This finding further supported the authors’ conclusion that sodium cocoyl isethionate micelles cannot penetrate through the smaller aqueous pores of the stratum corneum and thus cannot induce skin barrier perturbation. 13
The ability of sodium cocoyl isethionate, sodium dodecyl sulfate (SDS) with and without glycerol, glycerol, and the control, and phosphate-buffered saline (PBS) to affect the skin barrier was studied using two-photon fluorescence microscopy (TPM). 6 Sodium cocoyl isethionate was prepared for visualization as a 1% by weight solution with sulforhodamine B (SRB) and applied to harvested female Yorkshire pig skin in Franz diffusion cells for 5 hours. After the application period, the skin samples were rinsed 4 times with PBS, exposed to an aqueous SRB fluorescent probe solution in the diffusion cells for an additional 24 hours, and then rinsed again 4 times with PBS and blotted to remove excess SRB. The skin samples then underwent TPM imaging.
When compared to SDS, sodium cocoyl isethionate had a weaker skin barrier interaction, especially in the corneocyte envelopes and the corneocyte keratins. Sodium cocoyl isethionate did not induce the formation of localized transport regions in the skin barrier, and sodium cocoyl isethionate promoted SRB penetration into the intercellular lipid bilayers of the stratum corneum, although this effect is lower than that observed in SDS. Sodium cocoyl isethionate did not induce significantly deeper penetration of SRB and had significantly smaller SRB-skin partition coefficients and SRB-skin penetration depths, all when compared to SDS. This study indicates that sodium cocoyl isethionate is a mild surfactant relative to SDS because it decreases skin penetration of an irritant by reducing porosity to tortuosity ratio without reducing average pore radius. 6
Toxicological Studies
Oral and dermal acute toxicity data were available from earlier CIR safety assessments, supporting the safety of isethionate salts. Sodium cocoyl isethionate was slightly to practically nontoxic in rats, and a dermal application of 1.0% to 36.0% wt/wt aqueous sodium cocoyl isethionate to rats did not result in significant toxic effects. Erythema was observed at times during the study. 1
Acute Toxicity
Oral—nonhuman
Sodium isethionate
In an acute oral toxicity study, 5 male and 5 female Wistar rats received 5,000 mg/kg body weight sodium isethionate in water (50% wt/vol). 4 One female rat died after administration of the test substance. The death was not treatment related. No clinical signs of toxicity were observed in any of the rats. Decreased body weight was observed in 1 female rat. There were no macroscopic findings at necropsy. The LD50 value was greater than 5,000 mg/kg body weight.
Repeated Dose Toxicity
Oral—nonhuman
Sodium isethionate
The toxicity of sodium isethionate was evaluated in groups of 10 animals of each sex, except for the control group and the high-dose group, which consisted of 15 animals of each sex. Male and female Wistar rats received oral doses of sodium isethionate at 50, 200, or 1,000 mg/kg body weight/day in bi-distilled water (10 mL/kg body weight) for 91/92 days via gavage. 4 The study was performed according to Organization for Economic Cooperation and Development (OECD) guideline 408. All animals were killed at study end, and gross pathology and histopathology examinations were performed.
All rats survived until study end. No clinical signs of toxicity were observed during daily or weekly observations, and no toxicologically relevant ophthalmoscopic changes, differences in the mean feed consumption, changes in hematology parameters at 50 mg/kg/d or 200 mg/kg/d, or changes in urinalysis parameters at 50 mg/kg/d were observed. Statistically significant differences were noted in the mean hind limb grip strength of males treated with 1,000 mg/kg/d, but these were considered to be secondary to decreased body weights. Slightly decreased mean absolute and relative body weights were observed in 1,000 mg/kg/d males. Changes in the hematology parameters of 1,000 mg/kg/d group included decreased mean corpuscular hemoglobin concentration, increased mean absolute and relative reticulocyte counts, and a “left-shift” in the reticulocyte maturity indices indicative of increased reticulocyte turnover and decreased hemoglobin distribution width in females only. In 1,000 mg/kg/d rats, the clinical biochemistry parameters included decreased glucose levels, increased total bilirubin levels, increased cholesterol and phospholipid levels, and increased aspartate or alanine aminotransferase activities. Increased sodium levels in all 3 dose groups, decreased potassium levels in all 3 dose groups, increased calcium levels at 1,000 mg/kg/d, increased phosphorus in females at 1,000 mg/kg/d, and increased chloride levels in males at 200 mg/kg/d were also observed. Gross pathology and histopathology findings included increased spleen weights in rats at 1,000 mg/kg/d, macroscopic changes in the liver (increased incidence of tan foci in the liver of males and females treated with 1,000 mg/kg/d), microscopic changes in the liver (degeneration and necrosis [focal or single hepatocytes]), bile duct hyperplasia, focal hepatocytic hyperplasia, peribiliary fibrosis, and an increased incidence and severity of mixed inflammatory cell infiltration in the parenchyma and spleen (increased hemopoiesis) with complete postrecovery reversibility. The authors concluded that the no observed adverse effect level (NOAEL) for sodium isethionate was 200 mg/kg body weight/day. 4
Reproductive and Developmental Effects
Sodium Isethionate
The teratogenic potential of sodium isethionate was studied in Wistar rats. 4 Groups of 4 females received once daily oral treatments of 0, 50, 200, or 1,000 mg/kg body weight sodium isethionate in highly purified water (dose volume = 10 mL/kg) from day 0 to day 20 postcoitum. During the treatment period, the dams were observed for clinical signs of toxicity, and feed consumption and body weights were measured. All dams were killed on day 21 postcoitum for necropsy and the fetuses were removed by cesarean delivery for examination. All dams survived until the scheduled necropsy and no clinical signs of toxicity were observed. Feed consumption was marginally decreased when compared to the controls in the high-dose group, but the body weight gains were within normal parameters and this observation was not considered toxicologically relevant. Feed consumption and body weight gains were within normal parameters in the remaining dose groups. Preimplantation and postimplantation loss and the mean number of fetuses per dam were not affected by treatment with sodium isethionate at any dose level. No macroscopic findings were noted during necropsy. In the fetuses, no test material–related effects on sex ratios or body weights were observed. Also, no test material–related abnormalities were noted during the visceral examination or during the examination of fetal skeletons and cartilages. It was concluded that sodium isethionate was not teratogenic at the doses tested in this study and the maternal and fetal NOAEL was considered to be 1,000 mg/kg body weight/day.
Genotoxicity
Genotoxicity data were available from an earlier CIR safety assessment supporting the safety of sodium cocoyl isethionate. 1
In Vitro
Sodium isethionate
The genotoxic potential of sodium isethionate was studied in an Ames test for mutagenicity with Salmonella typhimurium strains TA 98, TA100, TA 1535, TA 1537, and TA 1538 and Escherichia coli WP2uvrA. The test was conducted with and without metabolic activation with concentrations up to 10,000 µg/plate. 4 Sodium isethionate was not toxic to the bacterial strains. No dose-dependent increase in the number of revertants was observed in any of the bacterial strains with and without metabolic activation. Sodium isethionate was not mutagenic in this Ames test.
The potential of sodium isethionate to induce mutations was studied using the mouse lymphoma thymidine kinase locus L5178Y assay according to the OECD guideline 476. 4 Two parallel experiments were performed: the first had a 4-hour treatment period with and without metabolic activation, and the second had a 24-hour treatment period without metabolic activation and a 4-hour treatment period with metabolic activation. A range-finding experiment preceded the main testing. Sodium isethionate in deionized water was tested at concentrations up to 1,500 μg/mL. Positive controls were methyl methane sulfonate and cyclophosphamide. No substantial and reproducible dose-dependent increase in mutant colony numbers was observed in both main experiments. No relevant shift in the ratio of small versus large colonies was observed up to 1,500 μg/mL. The positive controls yielded expected results. In this mouse lymphoma thymidine kinase locus L5178Y assay, sodium isethionate did not induce mutations with or without metabolic activation.
The potential for sodium isethionate up to 1,500 µg/mL to induce micronuclei in human lymphocytes was assessed according to the OECD guideline 487. 4 Two parallel experiments were performed—in the first, the exposure period to sodium isethionate in deionized water was 4 hours with and without metabolic activation, and in the second, the exposure period to the test material was 24 hours without metabolic activation mix and 4 hours with metabolic activation. The chromosomes were prepared 32 hours (experiment 1) and 52 hours (experiment 2) after the start of treatment with the test material. No visible precipitation of the test item in the culture medium was observed. No relevant cytotoxicity, indicated by reduced cytochalasin blocked proliferation index and described as cytostasis could be observed in this study up to 1,500 µg/mL. In both experiments, with and without metabolic activation, no biologically relevant increase in the number of cells carrying micronuclei was observed.
Carcinogenicity
No relevant published carcinogenicity studies on isethionate salts were discovered, and no unpublished data were submitted.
Irritation and Sensitization
In a previous safety assessment, the irritation and sensitization potential of sodium cocoyl isethionate
1
was evaluated in ocular irritation, dermal, and phototoxicity studies in rabbits. Sodium cocoyl isethionate was a mild to a primary ocular irritant at 2.5% to 49%; an ocular irritant at
In human irritation studies, an 8% aqueous solution of sodium cocoyl isethionate produced minimal irritation in 5 modified soap chamber tests while testing was discontinued in the sixth study due to the resulting irritation. A 4% aqueous solution of a formulation containing 15% sodium cocoyl isethionate was nonirritating. Solutions containing 0.10% to 1.0% sodium cocoyl isethionate were mildly irritating, whereas a 4% to 6% solution of a formulation containing 15% sodium cocoyl isethionate was a moderate to severe irritant. An RIPT was performed using a formulation containing 49.87% sodium cocoyl isethionate at 0.1% to 0.5% under a closed patch and at 4.0% to 8.0% under open conditions. The test article did not produce a sensitization reaction. In 2 RIPTs, one using a formulation containing 17% sodium cocoyl isethionate and the other using a 2% solution of a formulation containing 47.5% sodium cocoyl isethionate, the test article was not clinically irritating and did not induce allergic contact dermatitis. In a human study using a modified Draize procedure, a formulation containing 15% sodium cocoyl isethionate did not produce an allergic reaction. 1
Irritation
Dermal—nonhuman
Sodium isethionate
The skin irritation potential of sodium isethionate was tested according to OECD guideline 404 in 3 New Zealand White rabbits. 4 Approximately 500 mg of sodium isethionate in 0.1 mL of isotonic saline was applied to shaved skin and semioccluded for 4 hours before being rinsed off. The skin did not show any sign of erythema or edema up to 3 days after application. Mean scores on all observation time points after application were 0 for the 3 animals. The test substance was classified as not irritating.
Ocular—nonhuman
Sodium isethionate
The eye irritation potential of sodium isethionate was tested according to the OECD guideline 405 in 3 New Zealand White rabbits. 4 Approximately 100 mg of the test substance (undiluted) was instilled for 24 hours. Swelling of the lids and redness of the conjunctiva and iris was observed in the eyes 1 hour after application. The mean scores for the 3 animals on days 1, 2, and 3 for chemosis and redness of the conjunctiva were 0.2 and 0.7, respectively. These symptoms were fully reversible by 48 hours. The test substance was not considered irritating.
Sensitization
Dermal—nonhuman
Sodium isethionate
The sensitization potential of sodium isethionate was investigated by an LLNA test according to the OECD guideline 429. 4 Female CBA mice (5 animals/dose) received the test materials at concentrations of 10%, 25%, or 50% in ethanol–deionized water (30:70) according to the study protocol. No deaths were observed during the study period. No symptoms of local toxicity on the ears of the mice, and no signs of systemic toxicity were observed during the study. The body weights were within normal ranges. The positive control, hexyl cinnamic aldehyde, yielded expected results. The stimulation indices were determined to be 0.46, 0.48, and 0.56 for sodium isethionate at 10%, 25%, and 50%, respectively. An EC3 value could not be calculated. It was concluded that sodium isethionate was not a skin sensitizer in this LLNA test.
Clinical Assessment of Safety
Sodium Cocoyl Isethionate
Sodium cocoyl isethionate (2.9%), as well as sodium lauryl sulfate (SLS), disodium lauryl 3-ethoxysulfosuccinate (SUC), and a sodium soap of fatty acids derived from palm oil and coconut oil (SOAP) were used to evaluate the outcome of different irritancy testing methods in 25 volunteers.
14
In visual scoring of 1-time occlusive tests, the irritancy rank order for the anionic detergents was SOAP
The different aspects of irritant reactions and skin barrier recovery was studied in 8 surfactants, including 5% sodium cocoyl isethionate. 15 The substances were diluted in a citrate buffer and then applied with Finn chambers to the forearms of 12 volunteers for 48 hours. Irritancy was evaluated by clinical assessment, an evaporimeter, a laser Doppler flowmeter, and a corneometer on the day the patches were removed (day 1) and again on days 2, 5, 9, and 14. Sodium cocoyl isethionate produced visual erythema in 42%, 31%, 23%, 13%, and 10% of total on days 1, 2, 5, 9, and 14, respectively. Scaling was observed on day 2 in 3% of total and increased to 22% by day 14. The TEWL was elevated on days 1 and 2 with median values at approximately 37 and 31 g/m2/h, respectively. Cutaneous blood flow was elevated on day 2. Among the 8 surfactants tested, SLS was the most irritating, with sodium cocoyl isethionate the next most irritating.
Summary
Note that the summary only includes information available since the original safety assessment was published. The original safety assessment should be consulted for details on the studies that support the original conclusion.
Sodium cocoyl isethionate functions primarily as a surfactant-cleansing agent, and the majority of the uses reported are in coloring and noncoloring hair products. In 1993, CIR published a safety assessment on this ingredient with the conclusion “safe for use in cosmetic formulations at 50% in rinse-off products and at 17% in leave-on products.” Because of a shared core chemical structure and similar functions, the cosmetic ingredients ammonium cocoyl isethionate, sodium hydrogenated cocoyl methyl isethionate, sodium isethionate, sodium lauroyl isethionate, sodium lauroyl methyl isethionate, sodium methyl isethionate, sodium myristoyl isethionate, sodium oleoyl isethionate, sodium oleyl methyl isethionate, sodium palm kerneloyl isethionate, and sodium stearoyl methyl isethionate have been added to this safety assessment.
Sodium cocoyl isethionate was reported to be used in a total of 52 cosmetic products at the time of the original safety assessment. Use concentrations ranged from 10% to 50%. Current VCRP data indicate that sodium cocoyl isethionate is used in 490 cosmetic products, with almost half of the uses reported to be in hair dyes and colors. A survey of use concentrations conducted by the Personal Care Products Council in 2013 reported a range from 0.23% to 53%. Among the ingredients added to this amended safety assessment, sodium isethionate has the most uses (77) with the majority in bath soaps and detergents. The maximum use concentration range for sodium isethionate was 0.8% to 6%, with the 6% reported in bath soaps and detergents.
Toxicokinetics studies have found that sodium cocoyl isethionate micelles cannot contribute to sodium cocoyl isethionate skin penetration and associated skin barrier perturbation. The LD50 value was greater than 5,000 mg/kg body weight in an acute oral toxicity study in Wistar rats that received 5,000 mg/kg body weight sodium isethionate in water (50% wt/vol). In a repeated oral dose toxicity study in Wistar rats that received sodium isethionate at doses of 50, 200, or 1,000 mg/kg body weight/day in bi-distilled, the NOAEL was 200 mg/kg/d.
Sodium isethionate was not teratogenic in Wistar rat dams that received daily oral treatments of 0, 50, 200, or 1,000 mg/kg sodium isethionate in highly purified water on days 0 through 20 of gestation. The maternal and fetal NOAEL were considered to be 1,000 mg/kg body weight/day.
Sodium isethionate was not mutagenic in an Ames test at concentrations up to 10,000 µg/plate. This ingredient at concentrations up to 1,500 µg/mL also did not induce mutations in a mouse lymphoma thymidine kinase locus L5178Y assay, nor did it induce micronuclei in a human lymphocyte assay.
In New Zealand White rabbits, sodium isethionate was not a dermal irritant, nor was it an ocular irritant. When tested at concentrations up to 50% in ethanol–deionized water, sodium isethionate was not a skin sensitizer in an LLNA test.
Clinical testing of sodium cocoyl isethionate (2.9%) to compare irritancy potential to other surfactants found that sodium cocoyl isethionate was irritating but less irritating than SLS.
Discussion
A safety assessment for sodium cocoyl isethionate was published by CIR in 1993 with the conclusion of safe for use in cosmetic formulations at 50% in rinse-off products and at 17% in leave-on products. These concentration limits were based on the maximum concentrations reported in safety test data at the time. The CIR Expert Panel reopened the final report on sodium cocoyl isethionate based on new data and determined that the report should also address the safety of 11 additional isethionate salts.
The Panel considered that the available data on isethionate salts and noted the lack of systemic toxicity at high doses in single dose and repeated dose oral animal studies, no teratogenic effects in animal studies, and little or no irritation or sensitization in multiple tests of dermal and ocular exposure. The Panel acknowledged the absence of carcinogenicity data but considered the data demonstrating that sodium cocoyl isethionate and sodium isethionate were not genotoxic in Ames tests and mammalian assays adequate to support the safety of these ingredients.
Although there are data gaps, the shared core chemical structure, similar functions and concentrations in cosmetics, and the expected similarities in physicochemical properties enabled grouping these ingredients and reading across the available toxicological data to support the safety assessment of each individual compound in the entire group.
The Panel looked at changes in the patterns of use and concentrations of use since the original safety assessment of sodium cocoyl isethionate and noted that the earlier safety assessment had specified use concentrations of up to 50% in rinse-off products and up to 17% in leave-on products as safe. The most recently reported concentration of use of sodium cocoyl isethionate in rinse-off products is 49.4%. The Panel noted that most surfactants exhibit some irritancy, as was the case with sodium cocoyl isethionate at 2.9%. Thus, the Panel stated that products that include these ingredients should be formulated to be nonirritating.
The Panel discussed the issue of incidental inhalation exposure from suntan preparations. There were no inhalation toxicity data available. Sodium cocoyl isethionate is reportedly used at concentrations up to 1% in cosmetic products that may be aerosolized. The Panel believes that the sizes of a substantial majority of the particles of these ingredients, as manufactured, are larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that the 12 isethionate salts listed below are safe in the present practices of use and concentration in cosmetics, when formulated to be nonirritating. This conclusion supersedes the earlier conclusion issued by the Expert Panel in 1993. Sodium cocoyl isethionate Ammonium cocoyl isethionate Sodium hydrogenated cocoyl methyl isethionate* Sodium isethionate Sodium lauroyl isethionate Sodium lauroyl methyl isethionate Sodium methyl isethionate Sodium myristoyl isethionate* Sodium oleoyl isethionate* Sodium oleyl methyl isethionate* Sodium palm kerneloyl isethionate* Sodium stearoyl methyl isethionate*
*Ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author Contributions
C. Burnett contributed to conception and design; contributed to acquisition, analysis, and interpretation; and drafted manuscript; B. Heldreth contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript; L. Gill, F. Alan Andersen, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the director, Cosmetic Ingredient Review, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.