Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 8 keratin-derived ingredients, which function mainly as skin and hair conditioning agents in personal care products. The Panel reviewed relevant data provided in this safety assessment and concluded that the 8 keratin-derived ingredients are safe in the present practices of use and concentration described in this safety assessment.
Introduction
The keratin-derived ingredients detailed in this report are described by the Hydrolyzed Keratin Hydrolyzed Hair Keratin Hydrolyzed Oxidized Keratin Hydrolyzed Sulfonated Keratin Keratin Oxidized Keratin Soluble Keratin Sulfonated Keratin
This is the first Cosmetic Ingredient Review report on ingredients derived from the keratin family of proteins. However, the safety of several hydrolyzed proteins as used in cosmetics has been reviewed by the Panel in several previous assessments. The Panel concluded that Hydrolyzed Collagen, Hydrolyzed Soy Protein, Hydrolyzed Silk, Hydrolyzed Rice Protein, and Hydrolyzed Corn Protein are safe for use in cosmetics.2-7 Additionally, the Panel concluded that Hydrolyzed Wheat Gluten and Hydrolyzed Wheat Protein are safe for use in cosmetics when formulated to restrict peptides to a weight-average molecular weight (MW) of 3500 Da or less. 8
Keratin occurs naturally in epithelial cells and is essential for normal tissue structure and function. Much of the available published literature evaluated the ability of cosmetic or pharmaceutical ingredients to have a desired effect on naturally occurring keratin in skin, hair, or other tissues. These studies were not considered relevant for assessing the safety of the keratin-derived ingredients as used in cosmetics and are not included in this assessment.
The main sources of Keratin ingredients are sheep wool and bovine hoof (nail) or horn. 9 Goat (cashmere) wool and bird (chicken) feathers may also be used. Human hair was once a major source of Keratin, but its use is now limited because of the European ban on human-sourced materials for cosmetics. These differing sources could potentially produce or result in keratin with unique properties, which may result in varying compositions and impurities within a single ingredient (eg, Keratin from human hair may have some impurities that are different from Keratin obtained from bird feathers).
When the specific cosmetic ingredient names are discussed in this report, Keratin and the related ingredients will be capitalized, which is how they are presented in the
Chemistry
Definition
The definition, structures, and functions of the keratin-derived ingredients in this report are provided in Table 1.
Definitions, Structures, and Functions of the Ingredients in this Safety Assessment. 1

The general term, keratin, refers to a broad category of proteins that result in intermediate filaments that form the bulk of cytoplasmic epithelia and epidermal appendage structures (ie, hair, wool, horn, nails and similar tissues in animals). 10 Keratins can be classified into 2 distinct groups (“hard” and “soft”) based on their structure, function, and regulation.
“Hard” keratin filaments form ordered arrays and are the primary contributors to the tough structure of epidermal appendages. Hard keratins (derived from hair, wool, horn, nails, or other similar tissues in animals) are utilized as cosmetic ingredients, rather than soft (derived, eg, from cytoplasmic epithelial keratins). These hard keratin proteins contain a much higher content of cysteine residues than the soft keratins in their nonhelical domains, and, thus form tougher and more durable structures via intra/intermolecular disulfide bond formation. These hard keratins may be modified/extracted (ie, hydrolyzed, oxidized, sulfonated, etc) to produce other cosmetic ingredients (eg, Hydrolyzed Oxidized Keratin). The structural subunits of these keratins comprise 2 chains, which differ by MW and amino acid residue sequence (designated types I and II), that each contain nonhelical end terminal domains, and a highly conserved, central α-helical domain.
Chemical and Physical Properties
Keratin is insoluble in water, and many keratin-derived materials have MWs between 9 and 60 kDa.11,12 Keratin proteins extracted from hair are classified into 3 broad groups: α, β, and γ. 13 α-Keratin (found in hair fiber cortex) is low in sulfur content and has an average MW of 60 to 80 kDa. β-Keratin is protective and forms the majority of the hair cuticle; this type of keratin is difficult to extract. γ-Keratin is globular, high in sulfur content, and has a MW of ∼15 kDa.
Molecular weights for Hydrolyzed Hair Keratin are reported to be around 400 Da. 14 The MWs for Hydrolyzed Keratin have been reported to be as low as 150 Da, but may be around 1000 to 3000 Da.14-16
Method of Manufacturing
Keratin, Oxidized Keratin, and Sulfonated Keratin
Keratins are difficult to solubilize compared to other proteins. After mechanical means of processing, such as grinding of materials like animal horn, one of the first known processes for extracting keratins involved harsh (caustic) conditions using lime. Eventually, a number of oxidative and reductive methods were developed for extracting keratin. Production of many of the ingredients in this assessment involves extraction of keratin via acid- or enzymatic hydrolysis. 17
Keratin may be produced by nonchemical and chemical methods. Using nonchemical methods, Keratin may be dissolved and converted from wool via steam explosion or from feathers via superheated water. 11 Under conditions of steam explosion, wool is heated under pressure at a high temperature (with steam ∼220 °C) for several minutes (∼10 minutes), followed by explosive decompression. The wool is disrupted into solid and liquid phases consisting of oligopeptides, water soluble peptides, and free amino acids. Feather barbs are treated with superheated water (liquid ∼220 °C) for 2 hours and then cooled. The products are oligopeptides with MWs of 1.0 to 1.8 kDa.
For the chemical methods, wool Keratin is extracted by reduction or oxidation of disulfide bonds. 18 Reducing agents include thioglycolic acid, dithiothreitol, or 2-mercaptoethanol, and oxidizing agents include peracetic acid or performic acid. These agents must work in combination with a protein denaturing agent, like urea, to break disulfide bonds and disrupt hydrogen bonding. These chemical extraction methods can directly result in some of these modified ingredients, such as Oxidized Keratin or Sulfonated Keratin. Resultant keratins may have low or high sulfur content (low sulfur MW between 45 and 60 kDa, high sulfur MW between 11 and 28 kDa) or high glycine and tyrosine content (MW between 9 and 12 kDa). However, for a fully sulfonated keratin, an oxidant such as molecular oxygen is required for the complete sulfonation of protein thiol (S-H) groups by sulfite. 19 It has been reported that Keratin may be extracted from chicken feathers with reducing agents. 20
Hydrolyzed Keratin
Hydrolyzed Keratin may be prepared from sheep wool. 21 The wool is first washed to remove soil and debris and then boiled to remove residual oils. Next, the wool is enzyme hydrolyzed under mild conditions for 4 to 6 hours. When the target MW is reached, the pH is adjusted to neutralize the enzyme. The resultant solution is a mixture of Hydrolyzed Keratin fractions with a MW of ∼1000 Da. The solution may be diluted to produce a 30% active material.
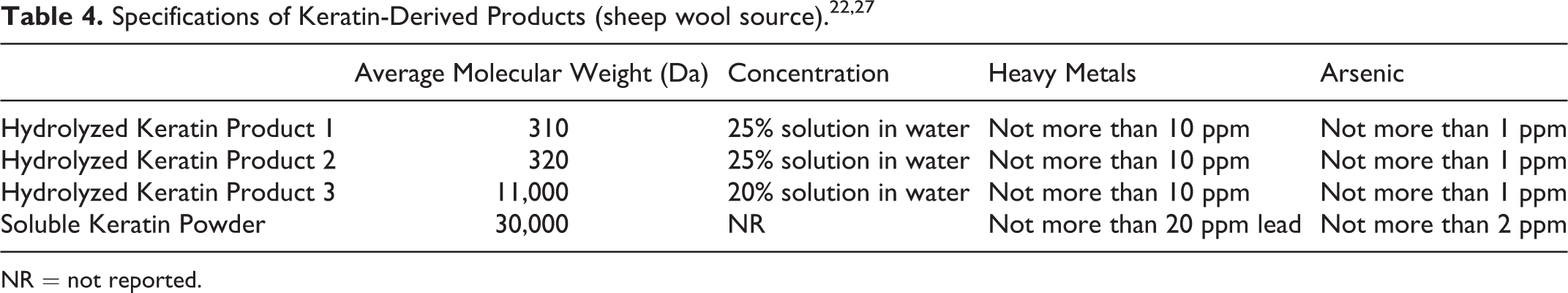
A supplier reported that 3 Hydrolyzed Keratin products (MW = 310, 320, and 11,000 Da, respectively) are prepared by acidic, alkaline, and/or enzymatic hydrolysis of sheep wool until the MW reaches the target range. 22 Further properties and specifications of these products are described in Table 2.
Another supplier certified that their powdered and liquid Hydrolyzed Keratin products are derived from sheep or goat wool.23-25
Hydrolyzed Keratin and Hydrolyzed Hair Keratin
A supplier reported that Hydrolyzed Hair Keratin and Hydrolyzed Keratin are obtained through acid hydrolysis. 15 Another supplier reported that Hydrolyzed Keratin is manufactured by enzymatic hydrolysis for a specific duration of time and at an elevated temperature (details not provided). 26 The resultant hydrolyzed proteins have MWs in the 2000 to 4000 Da range and all contain di- and tripeptides.
Soluble Keratin
A supplier reported that a Soluble Keratin powder product (MW = 30,000 Da) is prepared by extraction from sheep wool. 27
Composition
Keratins ubiquitously consist of central α-helical rod domains that are flanked by non-α-helical head and tail domains. 28 However, the amino sequences are not highly conserved across various source species (eg, equine versus human) or even among tissue-specific function types within 1 species (eg, hair vs nail versus skin). Indeed, the differences in amino acid sequences among keratins can be rather striking (eg, many cysteine residues in hair keratin vs a very small number in epidermal keratin, to no cysteine residues in other types). Accordingly, the species and tissues from which Keratin is derived can significantly impact the composition of the cosmetic ingredient. The amino acid sequences of some keratin proteins have been elucidated. However, the definition of Keratin in the Dictionary is “the protein derived from hair, wool, horn, nails or other similar tissues in animals.” Thus, the composition of a keratin-derived ingredient cannot be known without composition data from raw material suppliers.
Keratin
Cysteine residues in Keratin protein molecules make up 7% to 20% of the total amino acid residues. 11
Hydrolyzed Keratin
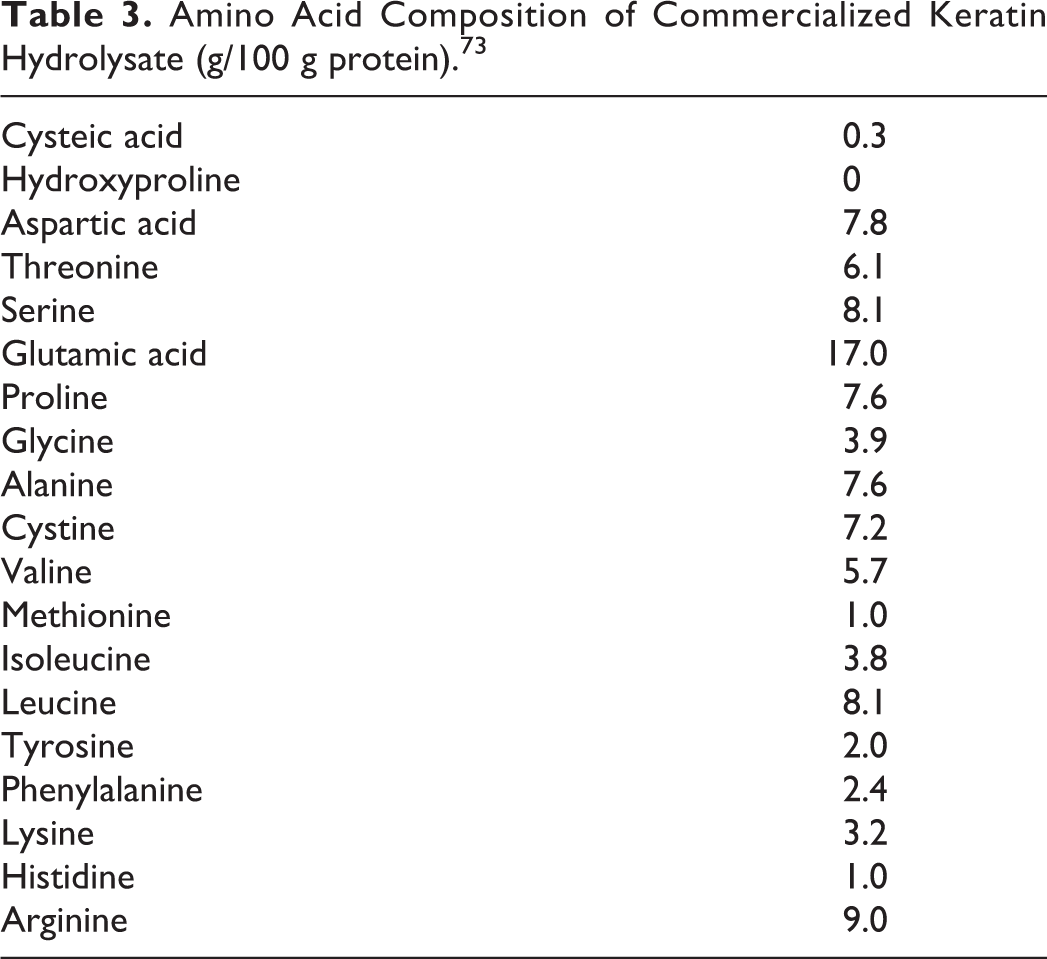
Amino acid composition data on Hydrolyzed Keratin is described in Table 3.
Amino Acid Composition of Commercialized Keratin Hydrolysate (g/100 g protein). 73
Impurities
Product specifications for Hydrolyzed Keratin and Soluble Keratin are presented in Table 4.
As with most nonsynthetic raw materials, numerous environmental factors may profoundly affect the presence and concentration of impurities, dependent on the Keratin source. Accordingly, manufacturers should use best practices to ensure the lowest possible values of any impurities.
Hydrolyzed Keratin
A supplier has certified that their Hydrolyzed Keratin products are free of bovine spongiform encephalopathy (BSE) infectivity, or the prions that cause BSE.23-25
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
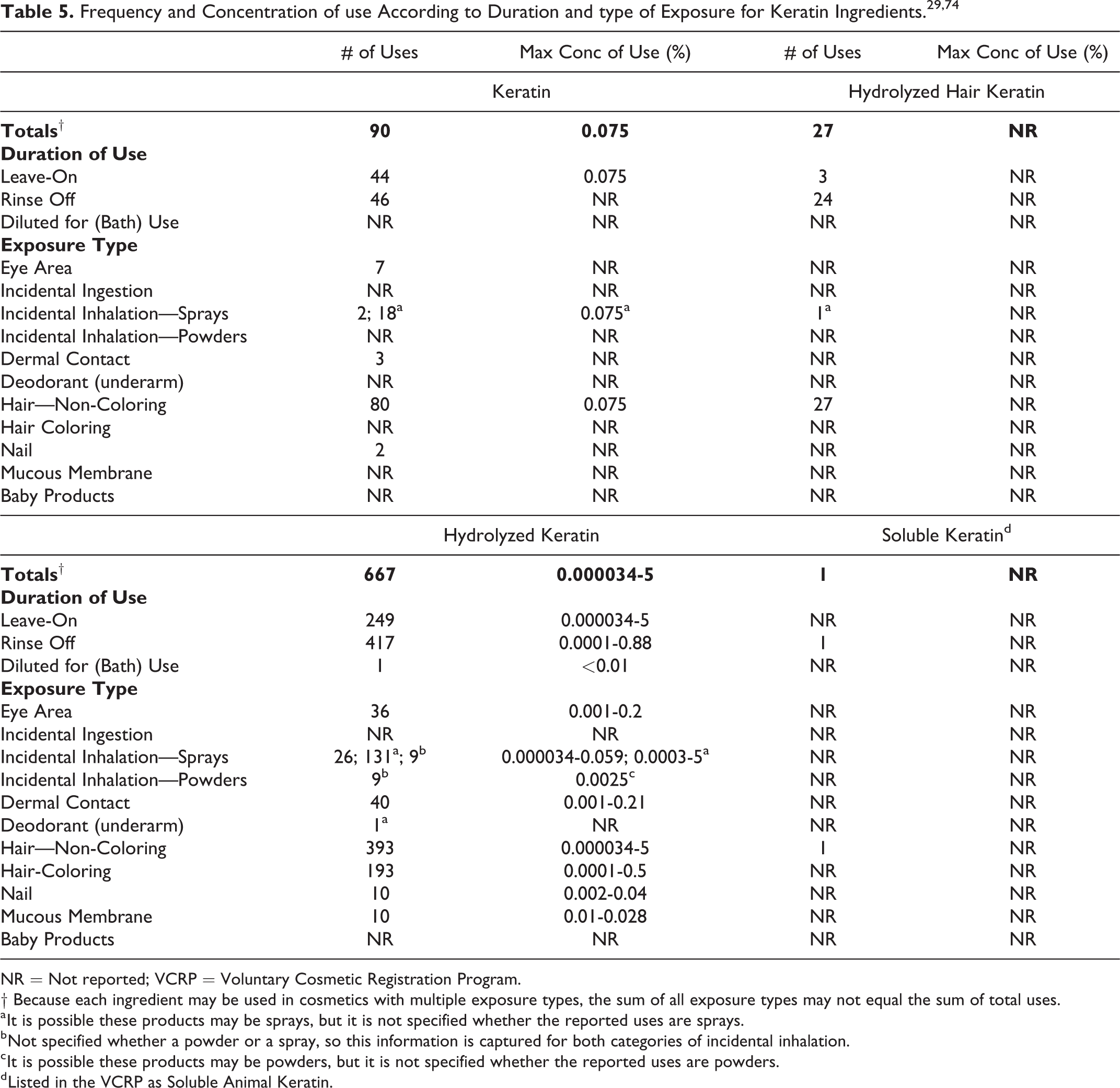
According to 2016 VCRP data, Hydrolyzed Keratin has the most reported uses of the ingredients listed in this safety assessment in cosmetic products, with a total of 667; more than half of the uses are in rinse-off non-coloring hair products (Table 5). 29 Keratin has the second greatest number of overall uses reported, with a total of 90; the majority of the uses are in non-coloring hair products. The results of the concentration of use survey conducted in 2015 by the Council indicate Hydrolyzed Keratin has the highest reported maximum concentration of use; it is used at up to 5% in hair tonics, dressings, and other hair grooming aids. Keratin is used at up to 0.075% in hair tonics, dressings, and other hair grooming aids.
Frequency and Concentration of use According to Duration and type of Exposure for Keratin Ingredients.29,74
NR = Not reported; VCRP = Voluntary Cosmetic Registration Program.
† Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
a It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
c It is possible these products may be powders, but it is not specified whether the reported uses are powders.
d Listed in the VCRP as Soluble Animal Keratin.
Based on the VCRP data and the results of the Council’s concentration of use survey, Hydrolyzed Oxidized Keratin, Hydrolyzed Sulfonated Keratin, Oxidized Keratin, and Sulfonated Keratin are not in use.
Some of these ingredients may be used in products that can come into contact with the eye or mucous membranes. For example, Hydrolyzed Keratin is used in mascara at up to 0.2% and in bath soaps and detergents at up to 0.028%. Additionally, some of these ingredients were reported to be used in hair sprays and could possibly be inhaled. For example, Hydrolyzed Keratin was reported to be used in hair sprays at a maximum concentration of 0.059%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.30-33 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.30,32
The FDA has recently issued a new final rule on use of materials derived from cattle in cosmetics (21 CFR §700.27). Prohibited materials did not include hoofs or horns or the keratin that may be derived from these substances.
The Keratin ingredients described in this safety assessment, with the exception of Hydrolyzed Hair Keratin, are not restricted from use in any way under the rules governing cosmetic products in the European Union (EU). 34 Hydrolyzed Hair Keratin is a substance prohibited in cosmetic products in the EU due to its human origin; however, the Scientific Committee on Consumer Products (SCCP) concluded that “the resulting risk, based on current scientific data, of the use [of amino acids obtained by hydrolysis of human hair] in cosmetic products for topical application…is negligible” when the human hair has undergone hydrolysis with concentrated HCl (> 20%) for 6 hours at 100 °C followed by activated carbon filtration, crystallization, and drying. 35
Noncosmetic
Noncosmetic uses of Keratin include use as a biopolymer in nanomaterials and in biomedical applications such as wound dressings, drug delivery, tissue engineering, and trauma and medical devices.10-13,36,37
Toxicokinetics
Absorption, Distribution, Metabolism, Excretion
Keratin
The tissue distribution of enzymatically and chemically fragmented Keratin (MW ∼ 8000 Da and 33,000 Da, respectively) was studied in male ddY mice. 37 The fragmented Keratin was radiolabeled with Na[125I]. The radiolabeled test materials were then injected via the tail vein at a dose of 2 mg/kg. The mice were killed at varying times following injection. Tissues were excised and weighed, and the radioactivity measured. The fragmented Keratin was found to be quickly eliminated from the plasma, taken up into the kidney, and gradually excreted in urine. Chemically fragmented Keratin was also observed to be taken up into the liver.
Dermal Penetration
Hydrolyzed keratin
A study of the efficacy of Hydrolyzed Keratin derived from wool stated that Hydrolyzed Keratin peptide can penetrate into the skin and increase moisturization. 38 No further details regarding the penetration properties of the Hydrolyzed Keratin were provided.
Toxicological Studies
Acute Toxicity
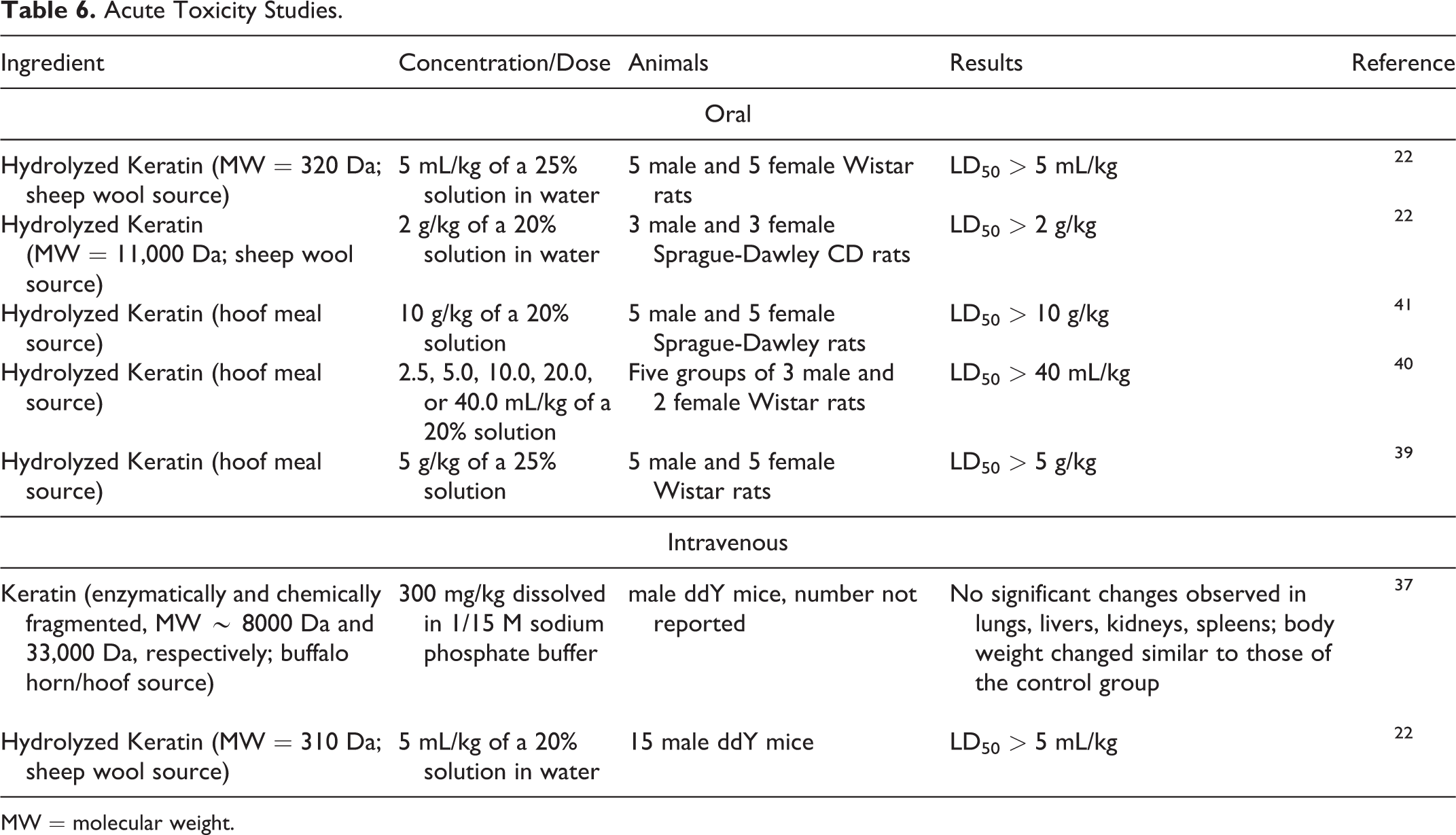
Acute toxicity studies are presented in Table 6.22,37,39-41 Hydrolyzed Keratin sourced from sheep wool and hoof meal was nontoxic in oral studies in rats (ie, the LD50 was greater than 40 mL/kg in a 20% solution). Keratin (MW = 8000 Da and 33,000 Da; buffalo horn/hoof source) and Hydrolyzed Keratin (MW = 310 Da; sheep wool source) were nontoxic in intravenous studies in mice.
Acute Toxicity Studies.
MW = molecular weight.
Dose Toxicity
No relevant published repeated dose toxicity studies on Keratin ingredients were identified in a literature search for these ingredients, and no unpublished data were submitted.
Reproductive and Developmental Toxicity
No relevant published reproductive and developmental toxicity studies on Keratin ingredients were identified in a literature search for these ingredients and no unpublished data were submitted.
Genotoxicity
In vitro genotoxicity studies are presented in Table 7.22,27,42-47 Hydrolyzed Keratin (MW ranging from 320-11,000 Da), Soluble Keratin (MW = 30,000 Da), and Keratin (MW not specified) that were mainly sourced from sheep wool were not mutagenic in Ames studies. In a comet assay, a Keratin peptide (MW = 1600 Da; synthesized from human hair) was not genotoxic in aqueous solutions, but genotoxicity was observed in organic solvent solutions. However, the negative controls also displayed genotoxicity in these solutions.
In Vitro Genotoxicity Studies.

h = hours; MW = molecular weight.
Carcinogenicity
No relevant published carcinogenicity studies on Keratin ingredients were identified in a literature search for these ingredients and no unpublished data were submitted.
Irritation and Sensitization
Dermal Irritation
In vitro, animal, and human dermal irritation studies are presented in Table 8.20,22,26,27,38,39,47-58 No irritation was predicted in in vitro studies of sheep wool-sourced Hydrolyzed Keratin tested at 100% and Keratin tested at 5%. Keratin (concentration not reported; chicken feather source) and Hydrolyzed Keratin (tested neat; source not provided) were mainly not irritating in rabbit studies. Hydrolyzed Keratin was not irritating in human studies at concentrations up to 25% (sheep wool source), nor was Soluble Keratin at 10% (sheep wool source).
Dermal Irritation Studies.
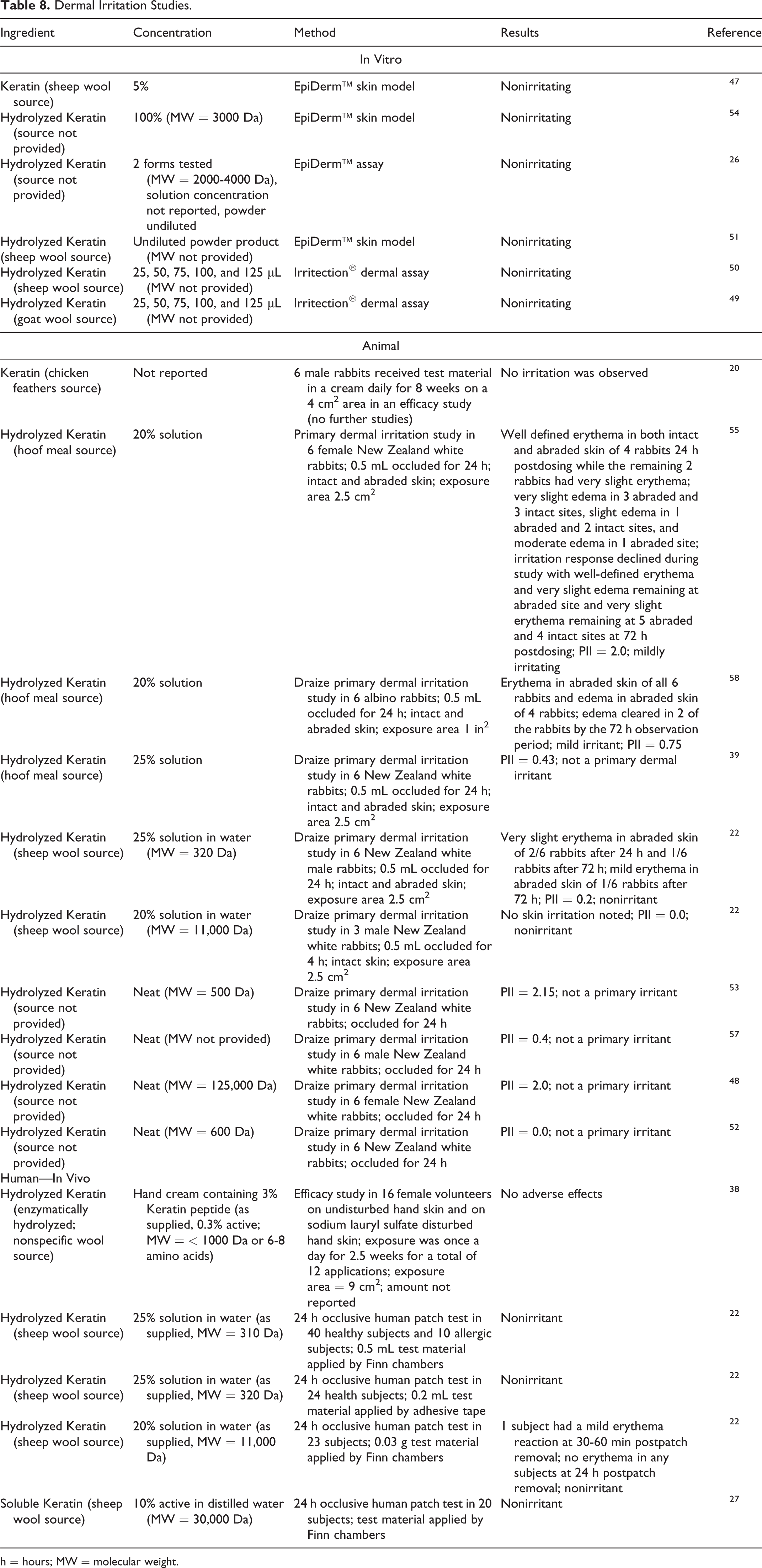
h = hours; MW = molecular weight.
Ocular Irritation
Ocular irritation studies are presented in Table 9.22,26,27,39,47,49-53,56,59-62 No irritation was predicted in in vitro studies of Keratin (5%), Hydrolyzed Keratin (undiluted), or Soluble Keratin (5%), which were mainly sourced from sheep wool. Hydrolyzed Keratin was minimally to nonirritating in Draize ocular rabbit studies when tested neat (source not provided).
Ocular Irritation Studies.

h = hours; HET-CAM Egg Test-Chorioallantoic Membrane; MW = molecular weight.
Dermal Sensitization
Dermal sensitization studies are presented in Table 10.22,27,47,63-68 No sensitization was predicted in in vitro studies of Hydrolyzed Keratin (sheep and goat wool sources). No sensitization to Hydrolyzed Keratin was observed when tested up to 25% in a guinea pig maximization study (sheep wool source) or up to 5% in human repeat insult patch tests (HRIPTs; bovine source). No sensitization was observed in HRIPTs to Keratin (5%; sheep wool source) or Soluble Keratin (10%; sheep wool source).
Dermal Sensitization Studies.
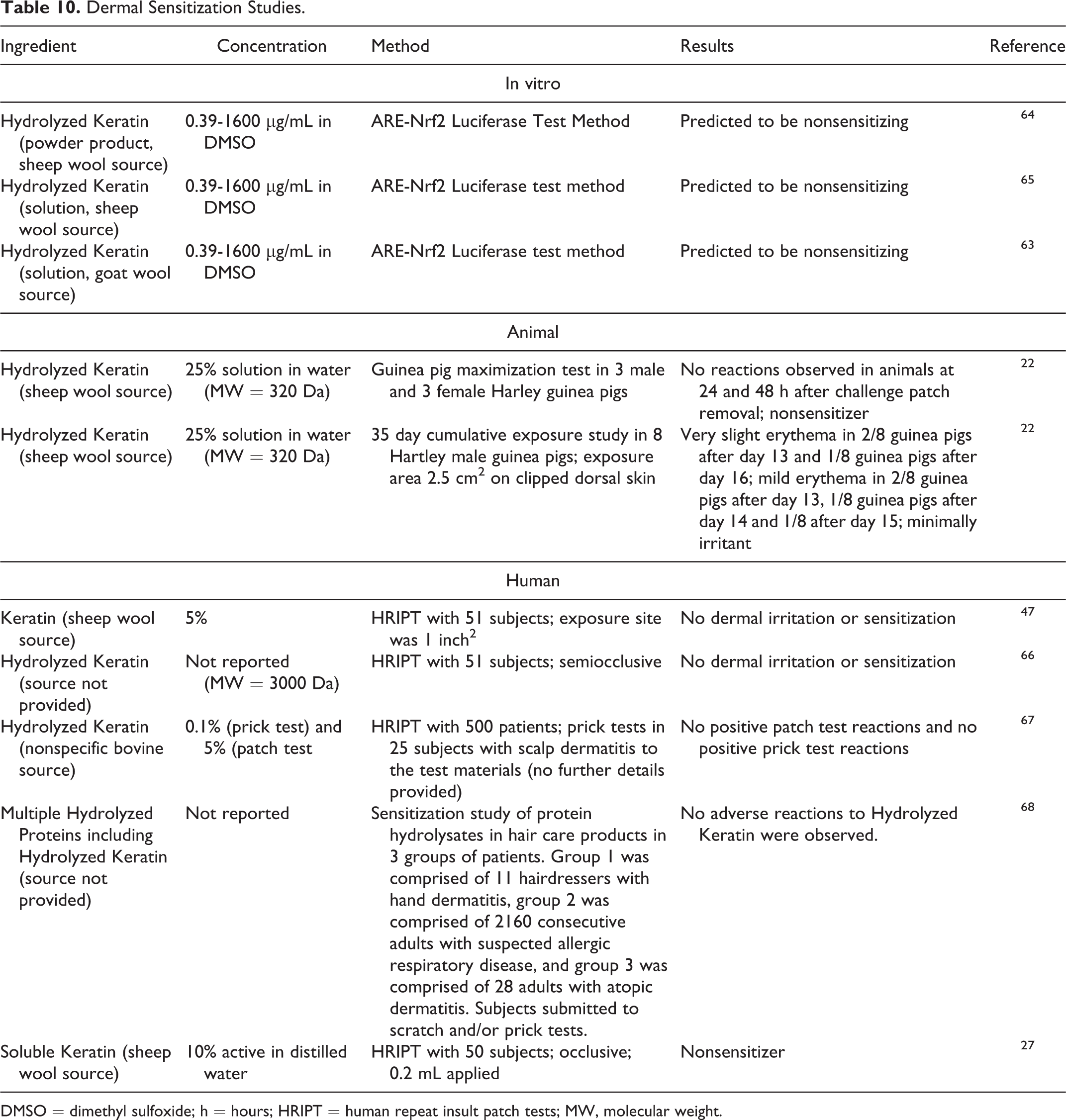
DMSO = dimethyl sulfoxide; h = hours; HRIPT = human repeat insult patch tests; MW, molecular weight.
Phototoxicity and Photosensitization
The potential for phototoxicity of Hydrolyzed Keratin (MW = 320 Da; sheep wool source) was studied in 6 male Hartley guinea pigs. 22 A 25% solution in water was applied to 2 clipped dorsal skin sites (2 cm2). Each site received 0.05 mL test material. One site was covered and the other site was irradiated for 2 hours. The sites were evaluated at 24, 28, and 72 hours postirradiation. No further details were provided. No erythema or edema was observed with or without irradiation.
The potential for photosensitization of Hydrolyzed Keratin (MW = 320 Da; sheep wool source) was studied in 6 male Hartley guinea pigs. 22 The guinea pigs were induced on 2 clipped dorsal skin sites (2 cm2) with 0.05 mL of a 25% solution in water. One site was covered and the other irradiated for 2 hours daily, 5 times per week for 2 weeks. Following a 2-week rest period, the test sites received the test material with the same site covered and the other site irradiated for 2 hours. Sites were evaluated at 24, 48, and 72 hours postirradiation. No further details were provided. No evidence of phototoxicity or photosensitization was observed.
Clinical Studies
Case Reports
Hydrolyzed keratin
A 22-year-old woman was reported to have a severe allergic reaction that included marked periorbital edema and swollen, sore, and itchy eyes and hands following use of a hair conditioner. 69 Prick testing elicited a strong positive (10 mm) wheal-and-flare response to the hair conditioner, which contained steartrimonium hydrolyzed animal protein (also known as Steartrimonium Hydroxyethyl Hydrolyzed Collagen). Additional prick testing showed further reactions to the quaternary hydrolyzed protein as well as to shampoos and conditioners that contained gelatin keratin amino acids, Hydrolyzed Keratin, and/or hydrolyzed collagen. The allergic reaction to Hydrolyzed Keratin was only observed in the prick test. Patch tests using the European standard series and a series of 15 common bases of medicines and cosmetics were negative.
Summary
The keratin-derived ingredients detailed in this report function mainly as skin and hair conditioning agents in personal care products. Keratin occurs naturally in epithelial cells and is essential for normal tissue structure and function.
According to 2016 VCRP data, Hydrolyzed Keratin has the most reported uses of the ingredients listed in this safety assessment in cosmetic formulations, with a total of 667; more than half of the uses are in rinse-off non-coloring hair formulations. Keratin has the second greatest number of overall uses reported, with a total of 90; the majority of the uses are in non-coloring hair formulations. The results of the concentration of use survey conducted in 2014 by the Council indicate Hydrolyzed Keratin has the highest reported maximum concentration of use; it is used at up to 5% in hair tonics, dressings, and other hair grooming aids. Keratin is used at up to 0.075% in hair tonics, dressings, and other hair grooming aids.
Noncosmetic uses of Keratin include use as a biopolymer in nanomaterials and in biomedical applications such as wound dressings, drug delivery, tissue engineering, and trauma and medical devices.
In an intravenous tissue distribution study of enzymatically and chemically fragmented Keratin, fragmented Keratin was found to be quickly eliminated from the plasma, taken up into the kidney, and gradually excreted in urine. Chemically fragmented Keratin was also observed to be taken up into the liver.
Hydrolyzed Keratin peptide derived from wool may penetrate into the skin.
Hydrolyzed Keratin was nontoxic in oral studies in rats (ie, the LD50 was greater than 5 mL/kg in a product with MW = 320 Da). Keratin (MW = 8000 Da and 33,000 Da) and Hydrolyzed Keratin (MW = 310 Da) were nontoxic in intravenous studies in mice.
Hydrolyzed Keratin (MW ranging from 320 to 11,000 Da), Soluble Keratin (MW = 30,000 Da), and Keratin (MW not specified) were not mutagenic in Ames studies. In a comet assay, a Keratin peptide (MW = 1600 Da) was not genotoxic in aqueous solutions, but genotoxicity was observed in organic solvent formulations. However, the negative controls also displayed genotoxicity in these formulations.
No dermal irritation was predicted in in vitro studies of Hydrolyzed Keratin tested at 100% and Keratin tested at 5%. Keratin (concentration not reported) and Hydrolyzed Keratin (tested neat) were not irritating in rabbit studies. Hydrolyzed Keratin was not irritating in human studies at concentrations up to 25%, nor was Soluble Keratin at 10%.
No ocular irritation was predicted in in vitro studies of Keratin (5%), Hydrolyzed Keratin (undiluted), or Soluble Keratin (5%). Hydrolyzed Keratin was minimally to nonirritating in Draize ocular rabbit studies when tested neat.
No sensitization was predicted in in vitro studies of Hydrolyzed Keratin. No sensitization to Hydrolyzed Keratin was observed when tested up to 25% in a guinea pig maximization study or up to 5% in HRIPTs. No sensitization was observed in HRIPTs to Keratin (5%) or Soluble Keratin (10%).
Hydrolyzed Keratin in a 25% solution was not phototoxic or photosensitizing in guinea pigs.
No relevant published repeated dose toxicity, reproductive and developmental toxicity, or carcinogenicity studies on Keratin ingredients were identified in a literature search for these ingredients and no unpublished data were submitted.
Discussion
Keratin is a protein that occurs naturally in epithelial cells and is essential for normal tissue structure and function. The main sources for Keratin and Hydrolyzed Keratin are sheep wool and bovine hoof or horn. Goat wool, bird feathers, and human hair may also be used. Keratin ingredients derived from different sources are likely to have different compositions and impurities, which may result in varying compositions and impurities within a single ingredient (eg, Keratin from human hair may have some impurities that are different from Keratin obtained from bird feathers).
Pesticide residues and heavy metals may be present in Keratin source materials. The Panel stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredients before blending into cosmetic formulations.
The Panel was also concerned about the inherent risks of using animal- and human-derived ingredients in cosmetic products, namely the potential for transmission of infectious agents. The Panel stressed that these ingredients must be free of detectible infectious pathogens (eg, BSE, HIV, and Creutzfeld-Jacob disease). Raw material suppliers and formulators of these ingredients must assure that these ingredients are free from pathogenic viruses and other infectious agents.
The Panel discussed the issue of incidental inhalation exposure in hair sprays. There were no inhalation toxicity data available. The Panel considered other pertinent data indicating that incidental inhalation exposures to keratin-derived ingredients in such cosmetic products would not cause adverse health effects, including data characterizing the potential for keratin-derived ingredients to cause ocular or dermal irritation or sensitization, and other effects. These ingredients are reportedly used at concentrations up to 0.059% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded the following keratin-derived ingredients are safe in cosmetics in the present practices of use and concentration as described in this safety assessment. Hydrolyzed Keratin Hydrolyzed Hair Keratin Hydrolyzed Oxidized Keratin* Hydrolyzed Sulfonated Keratin* Keratin Oxidized Keratin* Soluble Keratin Sulfonated Keratin*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Burnett, C. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.