Abstract
Our experience indicates that extrapolation of doses from the maximum tolerated doses (MTD) derived from 4-week dose range finding (DRF) studies conducted in CByB6F1 may overpredict tolerability and undermine utility of the high-dose groups in 26-week carcinogenicity studies conducted in Tg.rasH2. In the 26-week carcinogenicity studies conducted in Tg.rasH2 mice, we analyzed the initial body weights, food consumption (FC), terminal body weights, body weight gain (BWG), mortality, and tumor incidence for vehicle and test article–treated dose groups for 26 studies conducted from 2014 to 2018. Although not statistically significant compared to the control dose group, the % BWG decreased in male mice of mid- and high-dose groups by >10%, whereas in females there were no differences. The mortality increased in a statistically significant manner for medium and high doses of males. In female mice, the mortality increased in the high-dose group but not in a statistically significant manner. When the cause of death (COD) was analyzed in all dose groups of both sexes, the COD due to tumors was highest in the control groups, whereas it was lowest in high-dose groups of both sexes. At the same time, the COD due to undetermined causes, which is possible indication of test article–induced toxicity, was highest in high-dose groups of both sexes. These findings together indicate that MTD derived from earlier DRF studies was exceeded when applied to 26-week carcinogenicity studies and did not serve any purpose in the outcome of these studies.
Introduction
Data used in this paper is derived from all studies conducted at MilliporeSigma (previously known as BioReliance). Henceforth, throughout the paper, we will use the term BioReliance. Over the last decade, 26-week transgenic Tg.rasH2 mouse studies have replaced at least 75% of all 2-year mouse carcinogenicity studies and the number continues to rise.1-5 The Tg.rasH2 model is reputed to predict neoplastic findings relevant to human cancer risk assessment, produces fewer nonbiologically significant neoplastic outcomes, and is thus often preferable to a 2-year rodent study. 6 Prior to the establishment of 26-week Tg.rasH2 studies, carcinogenicity studies were conducted in rats and mice for a duration of 2 years. These 2-year studies have been conducted for over 30 years. As per the current practices, there is usually a control dose group and 3 test article–treated dose groups (low, mid, and high dose). The highest dose group is usually set at the maximum tolerated dose (MTD) that is derived from earlier dose range finding (DRF) studies. The low-dose group should not show signs of toxicity and should be close to or a little more than the human clinical exposure, and the mid-dose group should fall proportionately somewhere between the low- and high-dose groups.7,8 If exposure is proportional to dose, doses spaced at a factor of 10, for example, 1, 10, and 100, are not advised as the high dose may be too high, and the low dose may be too low.9,10 The MTD derived from the results of the earlier DRF studies is actually an estimated MTD (EMTD) because of predictivity aspects involved in the process.11,12 When the EMTD is determined, certain expectations or assumptions are made based on the analysis of data available from DRF studies. The assumptions are that the EMTD will not cause >10% decrease in the BWs of the high-dose group, minimal toxic effects are expected, shortening of the animal’s normal longevity will not occur, and the normal well-being of the animal will not be unduly compromised except for the effects of carcinogenicity. However, retrospective analyses of 2-year rodent carcinogenicity studies have shown that these assumptions have often failed resulting in overestimation of the MTD.1,8,11-17 As such, the appropriateness of this method for selecting the highest dose group at MTD in the 2-year rodent carcinogenicity studies has been questioned. 8 Some believe that every bioassay would be positive at MTD due to chronic toxicity, if sample size and duration of the assay were increased. 8 Thus, administering chemicals at MTD in the 2-year bioassays is considered nondiscriminating because they do not really differentiate between carcinogens and noncarcinogens. 8 As per the cancer evaluation criteria established by the National Toxicology Program (NTP), positive results demonstrate that a chemical is carcinogenic for laboratory animals under the conditions of the study and indicate that exposure to the chemical has the potential for hazard to humans. 18 Apart from these problems associated with MTD, there have been several other factors that have been identified in the 2-year rodent studies that can complicate the interpretation of these studies. These factors that are interlinked with each other include increasing initial body weights (IBWs), high mortality, and high incidence of spontaneous tumors.13,14,19-32 But based on our research and publications, such problems do not exist in Tg.rasH2 studies.4,5,33-36
In a previous publication, we presented the findings of retrospective analyses of 29 studies conducted from 2004 to 2013. 35 In the 2015 paper, we evaluated parameters such as IBW, terminal body weight (TBW), FC, body weight gain (BWG), mortality, and cause of death (COD). The COD was further classified as death due to spontaneous tumors, non-tumor causes, undetermined causes, and dosing accidents. Based on this analysis, it was found that the COD due to tumors in the control groups was higher than that in the high-dose groups which may be related to a >10% decrease in % BWG in the high dose. Undetermined COD likely reflects test article–related toxicity that was not predicted in range finding studies. In the current paper, we analyze an additional 26 studies performed between 2014 and 2018 to further evaluate the relationship between decreases in BWG, mortality, and tumor incidence. These findings together (2015 paper and this paper) suggest that MTD derived from earlier 28-day DRF studies conducted in CByB6F1 mice was exceeded when applied to 26-week carcinogenicity studies. Therefore, a proposal for new strategies in dose selection for 26-week Tg.rasH2 studies is introduced.
Material and Methods
Animals
All studies included in this paper were conducted at BioReliance. CByB6F1-Tg(HRAS)2Jic (+ hemizygous c-Ha-ras) mice, obtained from Taconic Farms, were used in all studies. The knock-in Tg element (human prototype c Ha-ras gene with its own promoter/enhancer) was injected into C57BL/6_ BALB/c F2 zygotes, which were crossed back to C57BL/6J forming C57BL/6JJic-Tg(HRAS)2Jic. The CByB6F1-Tg(HRAS)2Jic (+ hemizygous c-Ha-ras) is the offspring from a cross of the C57BL/6JJic-Tg(HRAS)2Jic hemizygous male mice with the BALB/cByJJic female mice. Each mouse was genotyped by Taconic to verify the presence of the transgene before being placed on study. Animals were randomized by body weight into groups using a computer program. On the first day of treatment, animals were 6 to 8 weeks of age and weighed at least 20g or 15g (males and females, respectively). Individual body weights for each dose group of each sex were within 20% of the mean at the start of the study.
Housing and Environmental Conditions
Housing and environmental conditions were similar in all studies. Animals were single-housed in polycarbonate cages with hardwood bedding chips in environmentally controlled rooms. Animals were verified to be free of illness prior to being placed on a study. All animals had ad libitum access to water and powdered feed (Harlan TEKLAD Global Diet).
Regulatory Requirements
The numbers of animals, procedures, and experimental design for each study were reviewed and approved by the BioReliance Institutional Animal Care and Use Committee. All procedures followed the specifications recommended in The Guide for the Care and Use of Laboratory Animals and were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited facility. All procedures involving but not limited to quarantine and acclimation, randomization, application of unique identification system, housing, provision of food and water, administration of test article, recording of clinical signs, necropsy, and tissue processing were followed in strict accordance with the Good Laboratory Practice Regulations, Standard Operating Procedures and protocol for each study.
Retrospective Analysis
The database was constructed based on Tg.rasH2 mice assigned to 26 studies between 2014 and 2018, conducted at the BioReliance facility, following the same study design. Animals in 22 of these studies were dosed by gavage, 3 were dosed subcutaneously, and the remaining one was dosed intravenously. The first of these studies was completed in 2014, and the last study was completed in 2018. Data were analyzed from 5,770 mice that included 2,995 male and 2,775 female mice. The control groups contained 905 male and 850 female mice, the low-dose groups contained 730 male and 650 female, the mid-dose groups contained and 680 male and 650 female mice, and the high-dose groups contained 680 male and 625 female mice. One study was conducted only in males. Variations in the number of animals are due to differences in the design of each of the study protocols, particularly in control groups, where additional dose groups were frequently added. Initial body weights were collected on day 1, and mice were weighed weekly thereafter for the next 13 consecutive weeks. After 13 weeks, the body weights were collected biweekly until study termination at 26 weeks (TBWs). The BWGs were calculated by subtracting the IBW from the TBW. The FC was recorded weekly for individual animals from day 1 until the termination. Upon the death or moribund sacrifice of an animal and at the termination of the study, all animals in each study were humanely sacrificed by CO2 overdose when applicable and subjected to complete necropsy. Protocol-required tissues were saved in 10% neutral buffered formalin, processed routinely, stained with hematoxylin and eosin (H&E), and evaluated microscopically. Each study was peer reviewed. For all studies in each of the 4 groups (control, low, mid, and high dose), the mean + SD for each of the parameters, IBW, average daily FC for the entire study, mean % BWG, % mortality, and % tumor incidence was calculated. Further, COD for each dead or moribund sacrificed animal was categorized as follows: (a) tumor: any tumor that was considered to be responsible for the COD; (b) nontumor: any nontumor lesion, test article induced or not, that was considered to be the COD; (c) undetermined: any animal in which the COD could not be determined based on the gross or microscopic examination of protocol required tissues; and (d) accidental: any animal in which an accident or gavage error was considered to be the COD. Further, based on greater than 10% decline in BWG or increased mortality (not caused by tumors) as criteria for exceeding MTD, the number of studies that had exceeded MTD in the high- and mid-dose groups of both sexes were determined. We further compared the percentage of tumors in the corresponding vehicle and treated dose groups of both sexes in those studies in which the MTD was exceeded and those studies where MTD was not exceeded. Statistical analysis was performed by using nonparametric Dunn test for the IBW, FC, % BWG, and % tumor incidence. 37 Fisher exact test was applied for analysis of % mortality. Linear treatment–related trend analysis was performed for % BWG, % mortality, and % tumor incidence. The COD was analyzed by w2 test for independence. The statistical analysis for the Fisher exact, trend test, and w2 test were performed by SAS1 Proprietary Software, Version 9.2. 38 For each sex, the comparison for each parameter was made between the control and the test article–treated groups, such as low, mid, and high.
Results
The summary data and the results of the statistical analyses for IBW, FC, % BWG, % mortality, and % tumor incidence for low-, mid-, and high-dose groups, compared to control, are presented in Tables 1 and 2 and Figures 1 and 2, for male and female mice, respectively. Results from the linear treatment–related trend analysis for % BWG, % mortality, and % tumor incidence are presented in Tables 3 and 4 for males and females, respectively. The data pertaining to the COD are presented in Tables 5 and 6 and Figures 3 and 4 for male and female mice, respectively. The data pertaining to studies in which the MTD was exceeded or MTD was not exceeded, and the percentages of tumors associated with these studies are presented in Tables 7 and 8. The statistical significance, whenever noted, is demonstrated by an asterisk in each table. The results for the males and females are summarized subsequently.
Male Mice Summary Data and Statistical Analysis for IBW, Fc, % BWG, % Mortality, and %Tumor.a
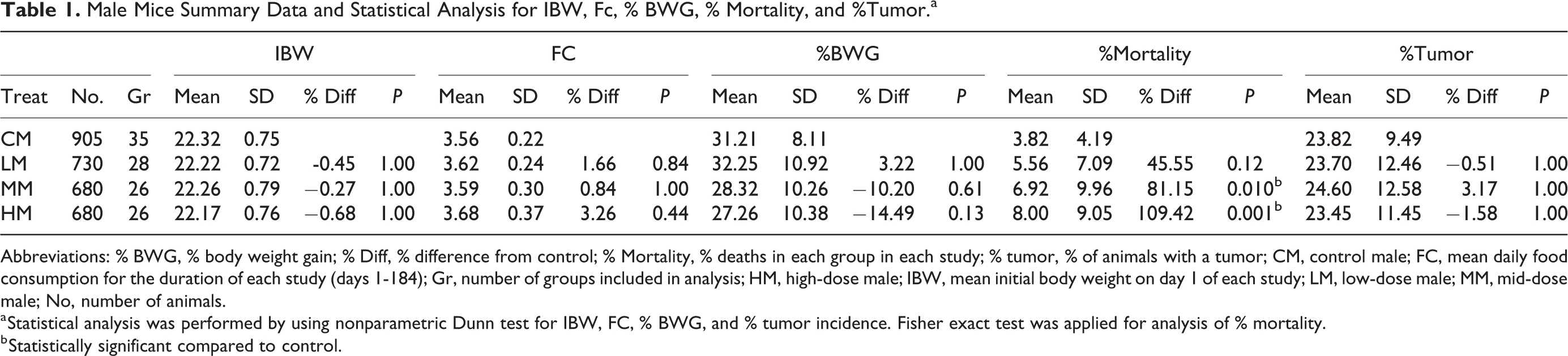
Abbreviations: % BWG, % body weight gain; % Diff, % difference from control; % Mortality, % deaths in each group in each study; % tumor, % of animals with a tumor; CM, control male; FC, mean daily food consumption for the duration of each study (days 1-184); Gr, number of groups included in analysis; HM, high-dose male; IBW, mean initial body weight on day 1 of each study; LM, low-dose male; MM, mid-dose male; No, number of animals.
a Statistical analysis was performed by using nonparametric Dunn test for IBW, FC, % BWG, and % tumor incidence. Fisher exact test was applied for analysis of % mortality.
b Statistically significant compared to control.
Female Mice Summary Data and Statistical Analysis for IBW, Fc, % BWG, % Mortality, and % tumor.a
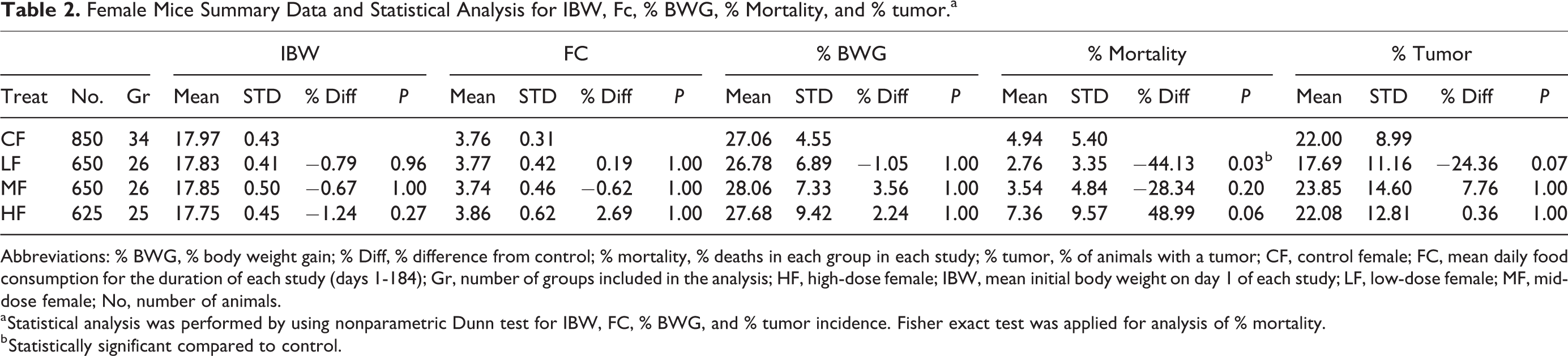
Abbreviations: % BWG, % body weight gain; % Diff, % difference from control; % mortality, % deaths in each group in each study; % tumor, % of animals with a tumor; CF, control female; FC, mean daily food consumption for the duration of each study (days 1-184); Gr, number of groups included in the analysis; HF, high-dose female; IBW, mean initial body weight on day 1 of each study; LF, low-dose female; MF, mid-dose female; No, number of animals.
a Statistical analysis was performed by using nonparametric Dunn test for IBW, FC, % BWG, and % tumor incidence. Fisher exact test was applied for analysis of % mortality.
b Statistically significant compared to control.
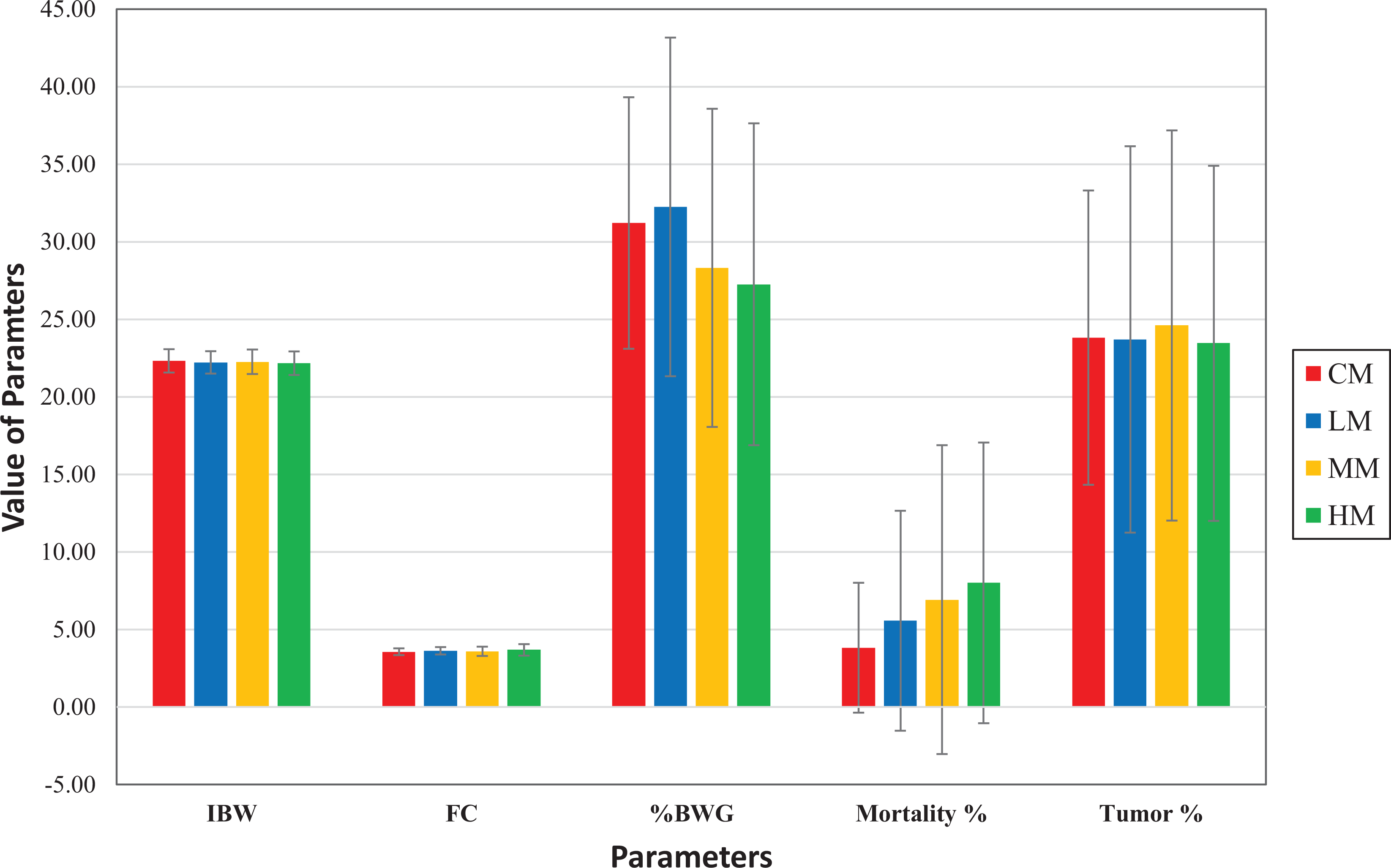
Comparison of mean (± SD) of initial body weights (IBW), food consumption (FC), % body weight gain (% BWG), % mortality, and % tumor incidence in control males (CM), low-dose males (LM), medium-dose males (MM), and high-dose males (HM).
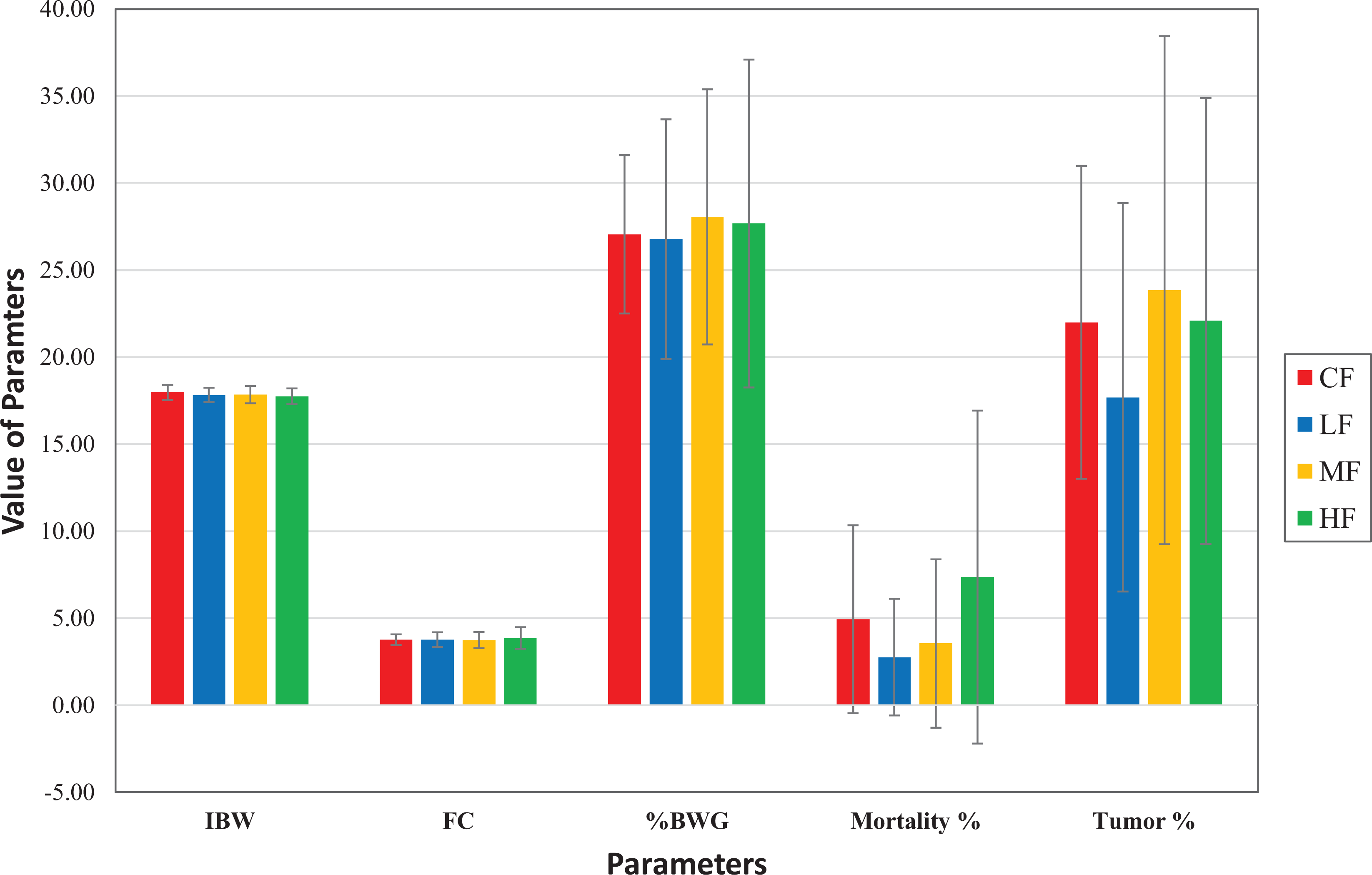
Comparison of mean (± SD) of initial body weights (IBW), food consumption (FC), % body weight gain (% BWG), % mortality, and % tumor incidence in control females (CF), low-dose females (LF), medium-dose females (MF), and high-dose females (HF).
Male Mice Linear Treatment–Related Trend Analysis.
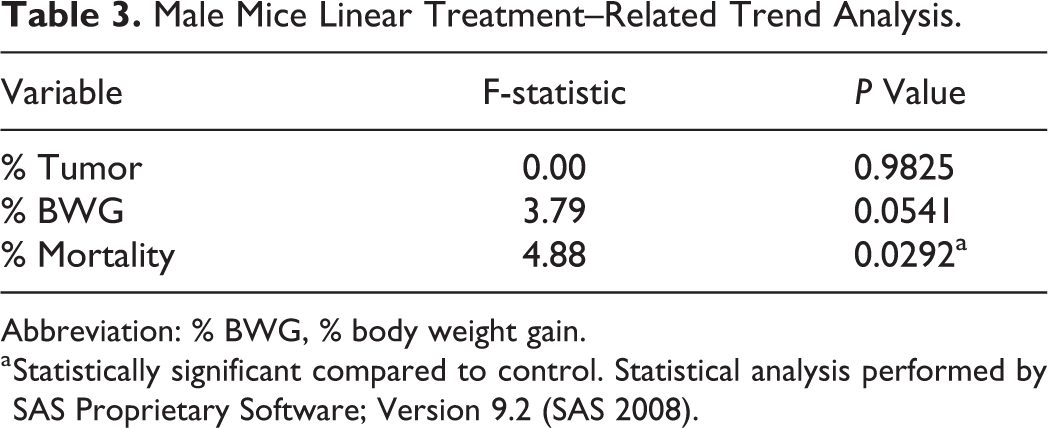
Abbreviation: % BWG, % body weight gain.
a Statistically significant compared to control. Statistical analysis performed by SAS Proprietary Software; Version 9.2 (SAS 2008).
Female Mice Linear Treatment–Related Trend Analysis.
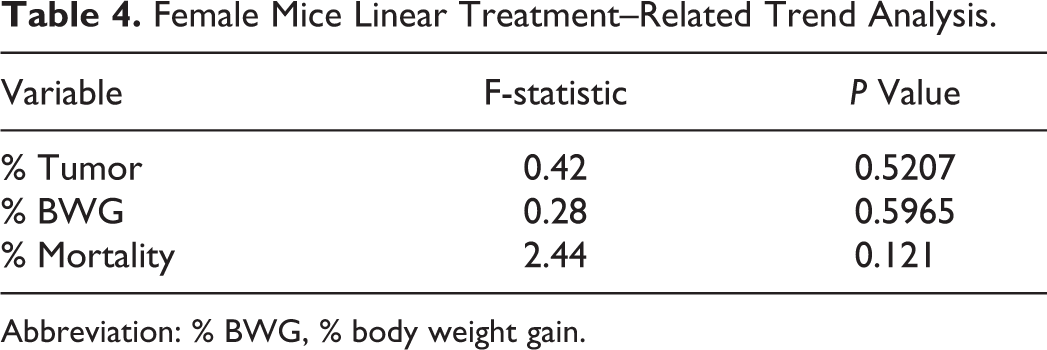
Abbreviation: % BWG, % body weight gain.
Male Mice Cod Statistical Analysis.
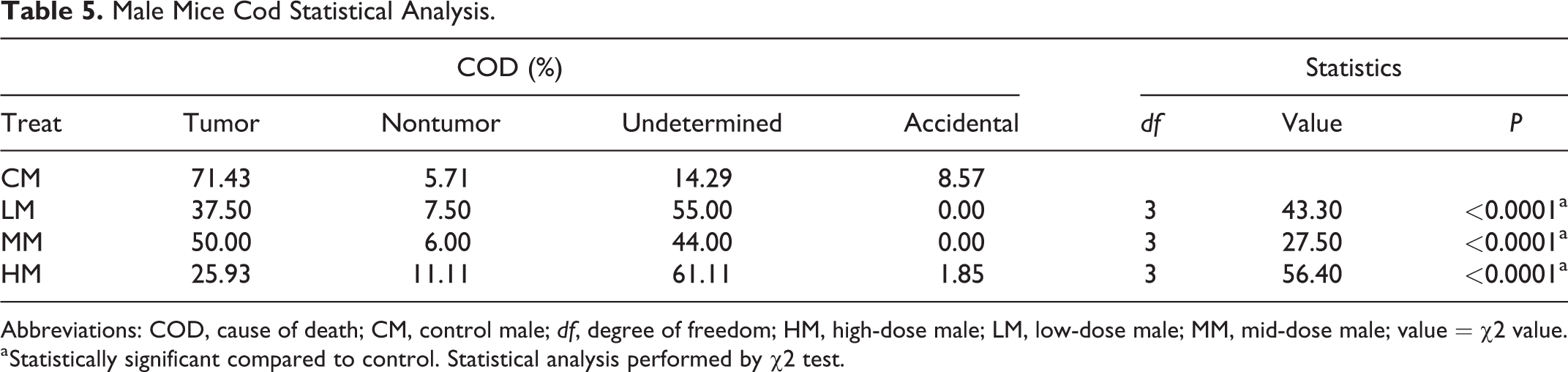
Abbreviations: COD, cause of death; CM, control male; df, degree of freedom; HM, high-dose male; LM, low-dose male; MM, mid-dose male; value = χ2 value.
a Statistically significant compared to control. Statistical analysis performed by χ2 test.
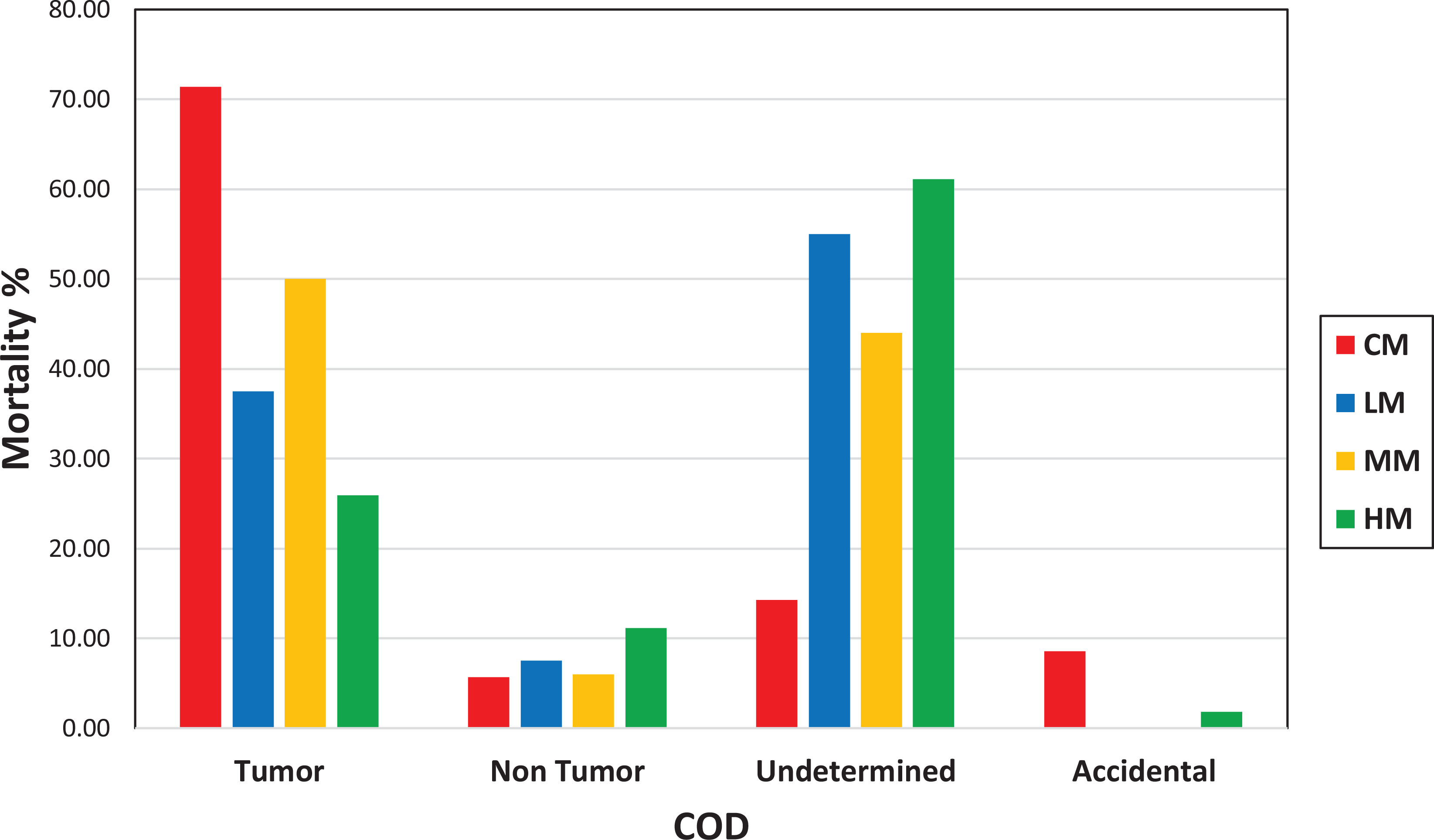
Comparison of cause of death (tumor, nontumor, undetermined, and accidental) between control males (CM), low-dose males (LM), medium-dose males (MM), and high-dose males (HM).
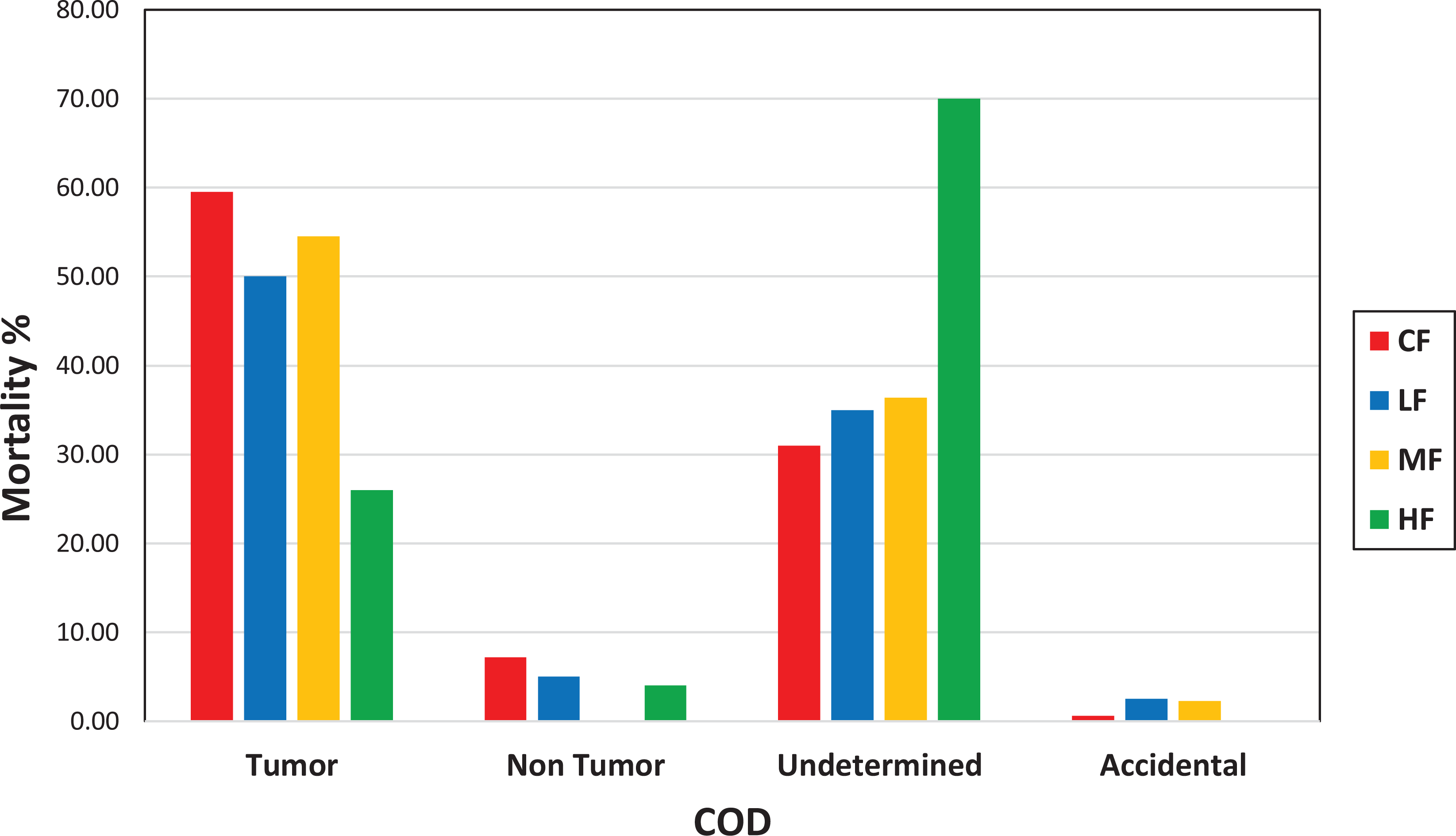
Comparison of cause of death (tumor, nontumor, undetermined, and accidental) between control females (CF), low-dose females (LF), medium-dose females (MF), and high-dose females (HF).
Female Mice Cod Statistical Analysis.
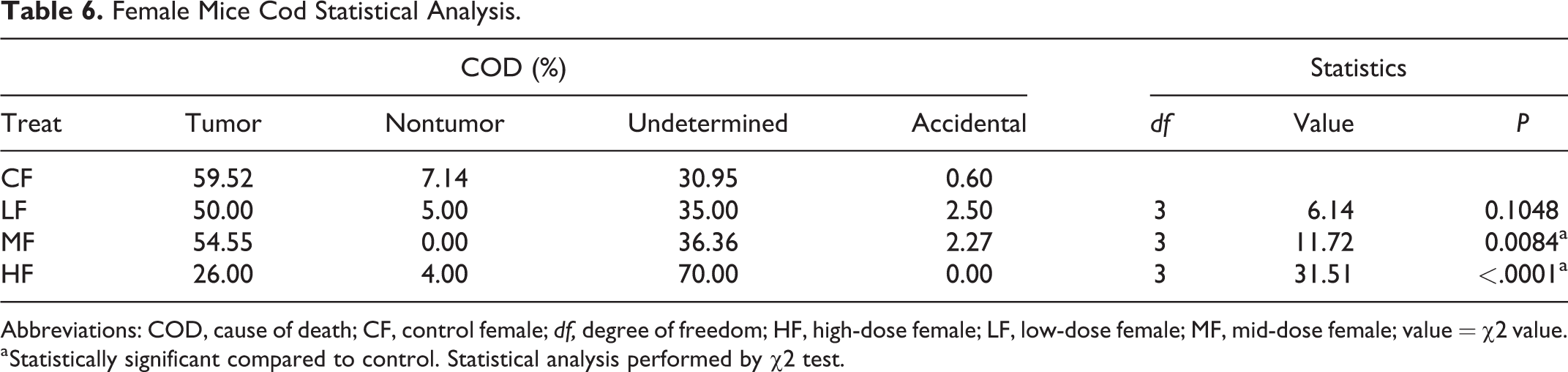
Abbreviations: COD, cause of death; CF, control female; df, degree of freedom; HF, high-dose female; LF, low-dose female; MF, mid-dose female; value = χ2 value.
a Statistically significant compared to control. Statistical analysis performed by χ2 test.
Female Mice Cod Statistical Analysis.
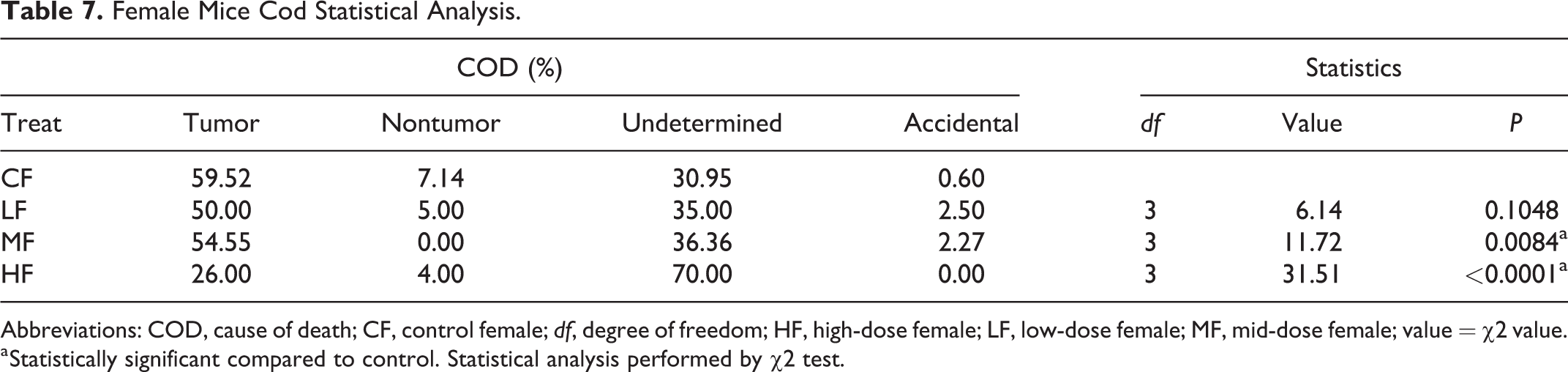
Abbreviations: COD, cause of death; CF, control female; df, degree of freedom; HF, high-dose female; LF, low-dose female; MF, mid-dose female; value = χ2 value.
a Statistically significant compared to control. Statistical analysis performed by χ2 test.
Studies With High- and Mid-Dose Groups (Males) or High-Dose Groups (Females) That Did Not Exceed the MTD.
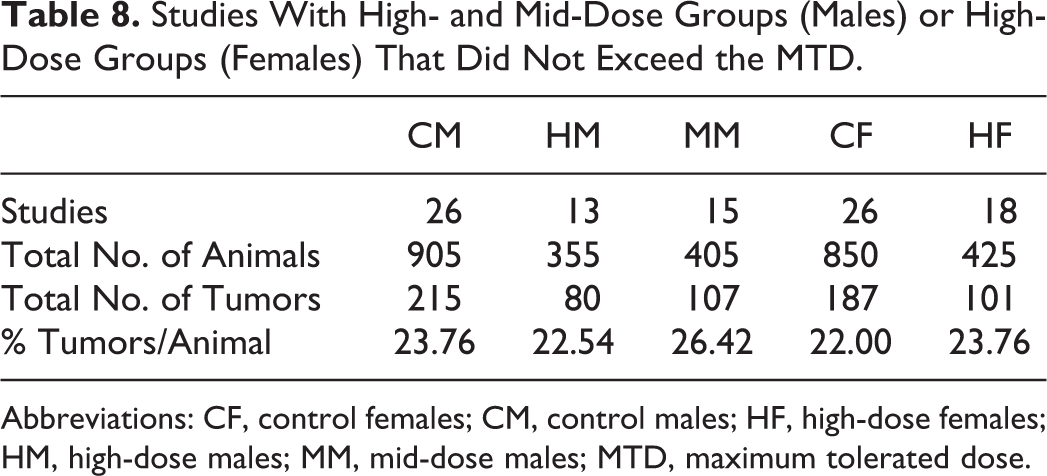
Abbreviations: CF, control females; CM, control males; HF, high-dose females; HM, high-dose males; MM, mid-dose males; MTD, maximum tolerated dose.
IBW, FC, % BWG, % Mortality, and % Tumor Incidence With Trend Analysis
The IBW of control, low-, mid-, and high-dose groups were similar across all groups, and there were no statistically significant differences between control and test article–treated groups in either sex. Since animals were randomized prior to the start of each study, and all animals were within 20% of the mean body weight at study initiation, it was expected that the IBW would be similar across all groups. Food consumption was also similar across all groups in both males and females. However, FC was slightly higher than controls in the high-dose males and females, although the differences were not statistically significant. In males, there was a dose-dependent decrease in the % BWG in the test article–treated mid- and high-dose groups compared to the controls, but these differences were not statistically significant. The % BWG differences in males were −10.20%, and −14.49% in mid- and high-dose male groups, respectively, compared to the control groups. There was a consistent, dose-dependent increase in mortality in male mice of test article–treated groups, and these differences were statistically significant in the mid- and high-dose groups. In females, there were no statistically significant differences in % BWG or mortality. Compared to the control dose groups in the males and females, % of tumors in test article–treated groups remained similar, and there were no statistically significant differences.
In males, % mortality was significant for treatment-related trend (Table 3) and P value for the difference in BWG at the high dose was 0.0541. Thus, increasing dose levels were associated with increases in % mortality and lower BWG. In females, no such trends were observed (Table 4).
Cause of Death
We analyzed the COD of all animals that died prior to study termination in each of the studies (Tables 5 and 6). Mortality due to tumors was highest in the control groups of both males and females and remained lower in low-, mid-, and high-dose groups of both sexes compared to controls. Tumor as a COD was lowest in high-dose groups of both sexes. In both sexes, nontumor CODs were sporadic and not noteworthy. Undetermined as the COD was lowest in the control groups of both sexes, but increased in low, mid, and high doses compared to controls, in all treated groups; this was dose-dependent in females but not in males. However, in the high-dose groups of both sexes, the undetermined causes were much higher compared to the controls (Figures 3 and 4). Undetermined causes simply meant that there were no significant gross or microscopic observations that could be related to COD. This increase in undetermined COD could be an indication of overwhelming toxicity and cause of early deaths. Statistical analysis of the CODs in each of the groups revealed a significant difference in the COD between controls and low-, mid-, and high-dose males, and mid- and high-dose females (Tables 5 and 6).
Comparison of Tumor Incidence in Studies That Did or Did Not Exceed MTD
As per Tables 7 and 8, in 13 studies, MTD was exceeded (>10% decrease in BW) in high-dose males and in 11 studies in mid-dose males. In high-dose females, MTD exceeded (> 10% decrease in BW) in 8 studies. In those studies that exceeded the MTD, the % of tumors was lower in mid dose of males and high dose of females. However, there were no statistically significant differences in % of tumors in any of the male and female groups.
Discussion and Conclusions
The purpose of the 26-week studies conducted in Tg.rasH2 is to evaluate the carcinogenic potential of a test article. Usually, 5- and 28-day DRF studies are conducted in CByB6F1 mice, the wild-type littermates of Tg.rasH2 mice, before the 26-week Tg.rasH2 carcinogenicity study. The purpose of the 5-day study is to estimate the doses for the 28-day study, and the purpose of the 28-day study is to estimate the doses for the 26-week study. Following a 28-day study, the dose selection, and particularly the MTD, is determined based on criteria similar to the ones used in 2-year rodent studies. Parameters that are taken into account but are not limited to include mortality, BWGs, gross and histopathology findings, clinical findings, and clinical pathology findings. A draft protocol for the 26-week study is then submitted to the Carcinogenicity Assessment Committee for the approval of the dose levels and the study design. Based on the earlier 28-day DRF studies, the MTD was determined for the 26-week studies. When this MTD was selected and applied as a high dose in the carcinogenicity studies, the high-dose groups at MTD were considered to be 100%. Compared to the high dose at MTD the mid-doses were generally at 40% of the dose at MTD and the low doses were generally at 20% of the dose at MTD in both sexes.
The most well-accepted definition of MTD is the highest dose that will elicit minimal signs of toxicity, should not cause >10% decrease in the BWGs compared to concurrent controls, should not shorten the animal’s normal longevity, or unduly compromise normal well-being of the animal, except for the effects of carcinogenicity. 16 While the high dose should elicit some signs of toxicity and biological effects, doses that produce excessive mortality apart from chemically induced carcinogenicity are undesirable.15,39 Decreased survival does not automatically imply that the MTD was exceeded if the mortality was due to test article–induced carcinogenicity.11,12 Greater than 10% decrease in BWG is considered as an accepted standard in determining MTD in 2-year and 26-week carcinogenicity studies. Retrospective analysis of the data presented here shows that in the mid and high doses of males there was decrease in % BWG greater than 10%, and the decrease was not due to decreased FC. There was in fact an increase in FC in the mid and high groups of males. The % mortality in the mid- and high-dose groups of males was increased in a statistically significant manner. The % tumor in males did not vary in a statistically significant manner. On the other hand, retrospective analysis of the data in females showed that none of the parameters IBW, FC, % BWG, % mortality, and % tumor varied in a statistically significant manner.
If the MTD is estimated correctly, then the carcinogenic response observed at the low, mid, and high doses should proportionately increase in a dose-dependent manner.10,17,40 However, that did not happen in the data presented here, and the % tumor more or less remained constant across all dose groups in both sexes. In fact, % tumor in control and high doses of each sexes remained almost identical, proving that the high doses in these studies did not give any additional information that could contribute to the outcome of the studies.
When the mortality was broken down by COD in our retrospective analysis, tumors as the COD was highest in the control-dose groups of both sexes. However, these tumors decreased in the test article–treated dose groups of both sexes compared to the control groups, and this decrease was highest in the high-dose groups of both sexes. On the other hand, undetermined as COD was substantially higher in the high-dose groups of both sexes but lowest in the control dose groups of both sexes. The undetermined as COD is assigned when there are no gross or microscopic lesions in a given animal that are considered to be responsible for death. However, this does not rule out that these deaths were simply due to toxicity, but morphological alterations, whether neoplastic or nonneoplastic, were simply not present. In fact, the higher incidence of undetermined COD for the unscheduled/early deaths means that these mice did not get the same opportunity to develop tumors as other animals in the study that survived to the terminal sacrifice. This also means that the doses were so high that the toxicity overwhelmed, and carcinogenic response was severely suppressed. Based on these factors, the high-dose groups of both sexes were an overestimation of the MTD and did not contribute to the assessment of the carcinogenic response. The control groups of both sexes had the highest number of deaths due to tumors, and the high-dose groups of both sexes had the lowest number of deaths due to tumors as shown in Figures 3 and 4. Now if you look at only low- and mid-dose groups in Figures 3 and 4, it shows that there is a dose-dependent increase in mortality due to tumors in the low- and mid-dose groups.
Thus, the data suggest that MTD in the high-dose groups of both sexes was overestimated, and these high-dose groups did not provide any information on the carcinogenic effects of the test article but rather defeated the purpose for which these carcinogenicity studies are conducted.1,8,11-17
A statistically significant increase in tumors in a dose-dependent manner has never been seen in any of studies reviewed so far. However, in a limited number of studies, we have seen a statistically significant increase in a particular tumor in a single-dose group, mostly in the low- or mid-dose groups, and infrequently in the high-dose group. Generally, there was no increase in tumors in any group that had a lower % BWG, compared to the control group. Therefore, it is concluded that a drop in % BWG plays a major role in reducing the incidence of tumors in Tg.rasH2 mice. We feel that the criteria for selection of high-dose group at MTD should not exceed >5% decrease in BWG, in future Tg.rasH2 studies. Apart from an overestimation of the MTD in the 2-year rodent carcinogenicity bioassays, several other problems have been identified that can further complicate the interpretation of these 2-year studies. These problems that are interlinked with each other include increasing IBWs, % BWG, high mortality, and high incidence of spontaneous tumors.13,14,19-32 But based on our research and publications, such problems do not exist in Tg.rasH2 studies.4,5,33-36
The body weights of the Tg.rasH2 mice are 85% to 90% of their wild-type counterparts—namely, C57BL/6 and the BALB/c mice.33,34,41,42 The IBWs of CD-1 and B6C3F1 mice when placed on study at 6 to 8 weeks of age are about 20% to 25% more than the Tg.rasH2 mice.43,44 Thus, the smaller size of the Tg.rasH2 mice compared to conventional mice may make these mice more susceptible to the decrease in body weights caused by toxicity, eventually leading to decreased incidence of tumors in the high-dose groups set at MTD. Therefore, the rule of thumb of no greater than 10% decline in the body weights applied to conventional rodents may not apply to the Tg.rasH2 mice, and this decline in the body weights needs to be set at a lower percentage, so that the true carcinogenic potentials of the test articles can be explored. Based on our analysis, we feel that no greater than 5% drop in BWG in the range finding studies would be more appropriate percentages in determining the EMTD. From retrospective analysis of MTD in 2-year studies as well as in 26-week Tg.rasH2 studies, it appears that due consideration is not given to the extended duration of the toxic effects of the drug compared to the DRF studies. The original NTP/National Cancer Institute (NCI) 2-year carcinogenicity studies had included only 2 test article–treated dose groups, set at MTD and one-half MTD and these original NTP/NCI studies were designed to provide information on carcinogenicity of a test article at MTD. But NTP started adding a third lower test article–treated dose group to nullify the effects of overt toxicity and excess mortality in the high-dose groups and to provide a safety margin against overestimation of MTD. The third-dose group was also added because having only 2 test article–treated dose groups did not provide information on dose response relationship or trends and no observed effect levels or no observed adverse effect levels.10-13,45-47 Since our findings clearly demonstrate that the high-dose group at MTD could not detect a carcinogenic effect while the mid and low doses were informative in detecting the true carcinogenic potential of the assay, a high dose set at MTD is not needed. If the high dose at MTD is taken out of the equation, then the cushion against excessively decreased body weights and high mortality is not required and doses set lower than the MTD, for example, one-half MTD and one-fourth MTD, will fully serve the purpose of the assay. Based on the earlier discussion, it is clear that the problems that significantly contribute to the interpretation of 2-year rodent carcinogenicity studies, mainly the variations in the body weight parameters, high mortality, and high incidence of spontaneous tumors, are not as profound in 26-week Tg.rasH2 studies.4,5,33,34 However, similar to the 2-year rodent studies, in the 26-week Tg.rasH2 studies, the estimation of MTD was not accurate and our findings clearly demonstrate that the high-dose group at MTD could not detect a carcinogenic effect.
In order to investigate why the dose selection process failed in the 26-week carcinogenicity studies, the IBW, TBW, % BWG, FC, and mortality from the first 4 weeks of 26-week studies with Tg.rasH2 mice were compared with 28-day DRF studies conducted with CByB6F1 mice.
36
Both the 26-week and the earlier 28-day studies were conducted with the exact same vehicle, test article, and similar dose levels. The analysis of these results further emphasizes that the EMTD and subsequent lower doses, determined on the basis of the 28-day studies in CByB6F1 mice, may not be an accurate strategy for selecting appropriate dose levels for the 26-week carcinogenicity studies in Tg.rasH2 mice,
36
and it was proposed that the Tg.rasH2 mice and not the CByB6F1 mice should be used in future DRF studies. The Tg.rasH2 mice demonstrate more toxicity than the CByB6F1 mice, possibly because of their smaller size compared to CByB6F1 mice. The Tg.rasH2 mice are at least 10% to 15% smaller when they are put on the study compared to the wild type. Thus, the EMTD derived from the CByB6F1 mice may be too toxic for the Tg.rasH2. Data from a previous publication
35
and the current publication show that in a total of 55 studies, the mortality caused by tumors was highest in the control groups of both sexes and lowest in the high-dose groups. On the other hand, the mortality caused by undetermined causes was lowest in the control-dose groups and highest in the high-dose groups. Thus, this clearly suggests that the doses applied in Tg.rasH2 studies were too high and caused increased toxicity which resulted in suppression of carcinogenicity, clearly defeating the purpose of the assay. Based on data presented here and from previous publications, the following changes are proposed: The criterion of not >10% decrease in % BWG does not appear to be appropriate for Tg.rasH2 mice and the cutoff point should be at 5%. The body weight criterion for high-dose selection for 26-week Tg.rasH2 studies should be changed because it failed to detect any possible tumorigenic response, defeating the purpose of the assay. However, if the studies are to be continued as per current practices, we know that the EMTD derived from earlier DRF studies is generally too high because of higher body weights of the CByB6F1 mice. So, when applying these doses to the Tg.rasH2 mice, they must be lowered considering the lower weight of Tg.rasH2 and longer duration of the studies. We propose that the doses should be ½ (high), ¼ (mid) and ⅛ (low) of the EMTD. Alternatively, the low dose could be close to or slightly higher than the clinical dose. In conforming with the 3Rs of animal research, the lowering of the doses will certainly cause less stress and pain, lower undue toxicity, and reduce the mortality due to overwhelming toxicity and possibly increase the specificity and sensitivity of the assay, the sole purpose of which is to detect the carcinogenic potential.
Thus, it is time to reconsider the doses applied in the Tg.rasH2 carcinogenicity studies, so that these assays will serve the desired purpose of the studies, which is to detect the carcinogenicity effects. We feel that the cutoff point of no greater than 5% decrease in the body weights, at the estimation of MTD should lower subsequent doses and improve utilization of every single animal on the study and provide more sensitive and specific results. Considering all parameters analyzed, decrease in BWG by 10% played a major role in overestimation of MTD, along with increased mortality, lower mortality due to tumors in treated groups, and increased mortality due to undetermined as a COD in high-dose groups of both sexes. Based on our analysis of 55 studies, a proposal for new strategies in dose selection process for 26-week Tg.rasH2 studies is introduced.
Footnotes
Acknowledgments
Author Contributions
Paranjpe, M. substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation, and drafted the manuscript; Vidmar, T. contributed to acquisition, analysis, or interpretation and critically revised the manuscript for important intellectual content; Elbekai, R. substantially contributed to conception or design and critically revised the manuscript for important intellectual content; Mann, P. contributed to acquisition, analysis, or interpretation and critically revised the manuscript for important intellectual content; Denton, M. contributed to acquisition, analysis, or interpretation and critically revised the manuscript for important intellectual content; McKeon, M. contributed to acquisition, analysis, or interpretation and critically revised the manuscript for important intellectual content; Brown, C. contributed to acquisition, analysis, or interpretation and critically revised the manuscript for important intellectual content; Martinek, M. contributed to acquisition, analysis, or interpretation and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
