Abstract
Melanin is a group of natural pigments that determines the human skin color and provides fundamental protection against the harmful impacts of physical and chemical stimuli. The aim of this study was to establish the regulatory role of aryl hydrocarbon receptor (AhR) in α-melanocyte-stimulating hormone (α-MSH) induced melanogenesis. In the present study, following knockdown of AhR, murine B16F10 cells were treated with α-MSH (200 nM) and tyrosinase activities, cellular melanin content, mRNA levels of several important genes involved in melanogenesis including AhR, CTNNB1, TYR2, and microphthalmia-associated transcription factor (MITF) were measured as endpoints. Exposure to α-MSH led to elevated expression of AhR, CTNNB1, MITF, and TYR in accordance with increased tyrosinase enzyme activity as well as a significant rise in the total melanin content. Our results suggest that AhR plays a regulatory role in α-MSH-stimulated melanogenesis.
Introduction
Melanin as a natural pigment synthesized by melanocytes via a process called melanogenesis 1 accumulates in the specialized intracellular organelles, “melanosomes” 2 to protect human and animal skin against harmful effects of ultraviolet radiation and also against various environmental toxins. 2 As a matter of fact, melanin abnormal production including hyperpigmentation and hypopigmentation and their genetic associated alteration were found to be linked with a number of serious dermatological disorders such as melasma, areolar hyperpigmentation, solar lentigines, vitiligo, mycosis fungoides, and leprosy. 3,4 There are 2 kinds of melanin in the melanosomes: eumelanin, which is found in the dark skin and black hair, and pheomelanin, which is found in the red hair/freckled skin type. 5
Melanogenesis is a complex process regulated by 3 important enzymes, namely tyrosinase (TYR), TYR-related protein-1 (TRP-1), and TRP-2.
6
In particular, TYR is a crucial regulatory enzyme that catalyzes the initial and rate-limiting steps of melanogenesis.
7
Both eumelanin and pheomelanin are synthesized by oxidation of
It is well known that human skin is constantly exposed to a various environmental pollutants that activate AhR, leading to some skin conditions such as chloracne and hyperpigmentation. 9,10 Aryl hydrocarbon receptor is a sensor of physical and chemical stimuli that belongs to the family of PER-ARNT-SIM basic helix–loop–helix transcription factors, 11 which are expressed in most tissues. Increasing evidence suggests that the AhR signaling pathway can affect the process of melanin synthesis in melanocyte cells. Luecke et al also reported that normal human melanocytes produce melanin in an AhR-dependent manner. 12 Nakamura et al showed AhR-dependent melanogenic effects in melanocytes by tobacco smoke extract. 13 In addition, epidermal TYR activation in the AhR-deficient mice has been shown to be compromised. 14
Many questions remain unanswered regarding both the AhR-dependent regulation of the melanocytes homeostasis and the association between AhR and other signaling pathways involved in these processes. In this study, we evaluated the regulatory role of AhR in α-MSH-induced melanogenesis.
Materials and Methods
Chemicals
The chemicals used in this study were obtained from the following suppliers: α-MSH,
Cell Culture and Treatments
The B16-F10 melanoma cells were obtained from the National Cell Bank of Iran. B16F10 mouse melanoma cells were grown in Dulbecco’s Modified Eagle Medium supplemented with 10% fetal bovine serum and 1% penicillin–streptomycin in a humidified atmosphere containing 5% CO2 at 37 °C throughout the experiment. Murine B16F10 cells (2 × 105 cells/well) were seeded in 6-well culture plates and were allowed to attach for 24 hours and subsequently were treated with α-MSH (200 nM) and XAV939 (3,5,7,8-Tetrahydro-2-[4-(trifluoromethyl)phenyl]-4H-thiopyrano[4,3-d]pyrimidin-4-one), a WNT pathway inhibitor (1 µM) when they reached 70% confluence.
Transfection With siRNA Against AhR
For siRNA transfection, the cells were seeded into 6-well culture dishes allowed to reach 50% to 60% confluence. Then, a total of 20 nM AhR siRNA (or scrambled siRNA) was mixed with 3-µL siRNA transfection reagent (sc-29528) in 1,000 µL siRNA transfection medium (sc-368668) and was added to each well. At 15-hour post-transfection, the siRNA transfection medium was removed and the cells were incubated with α-MSH (200 nM) for 24 hour in complete culture medium. The effectiveness of AhR knockdown was determined by 7-ethoxy-resorufin-O-deethylase (EROD) activity in response to a high affinity AhR agonist, 6-formylindolo[3,2-b]carbazole (FICZ).
7-ethoxy-resorufin-O-deethylase Activity
7-ethoxy-resorufin-O-deethylase assays were performed on murine B16F10 cells as reported previously. 15 Cells were placed in 96-well dishes at a density of 10,000 cells per well and were allowed to adhere for 24 hours. Thereafter, the cells were preincubated with siRNA or scrambled siRNA for 24 hours, and then treated with FICZ (10 nM) or DMSO for 15 hours. After treatment, the medium was removed and the cells were washed twice with phosphate-buffered saline (PBS). Then, 200 µL of 2 µM 7-ethoxyresorufin in the sodium phosphate buffer (50 mM pH 8.0) was added to each well. After 20 minutes of incubation at 37 °C, the formation of resorufin was measured with the excitation/emission wavelengths of 535/590 nm; data were normalized to the amount protein using the Bradford method.
Quantitative Reverse Transcription Polymerase Chain Reaction
Total RNA was extracted from the cells using the Trizol reagent (Bio Basic) according to the manufacturer’s instructions. Synthesis of complementary DNA (cDNA) was performed with 1 µg of total RNA using a cDNA synthesis kit (Takara) as described by the manufacturer. Polymerase chain reactions were performed to evaluate the primer design, and the resultant products were analyzed by electrophoresis on agarose gels. For real-time reverse transcription polymerase chain reaction (RT-PCR), the samples were assayed in triplicate by aliquots 1 µL of cDNA in a reaction mixture (25 µL) containing SYBR Green (12 µL), nuclease-free water (10 µL) and 1 µL of forward and reverse primers (10 µM; Table 1). All real-time assays were carried out under the following conditions: 1 cycle of predenaturation for 15 minutes at 95 °C, 40 cycles of denaturation at 95 °C for 15 seconds, annealing and extension at 58 °C and 72 °C for 30 seconds, respectively. Melt curve analysis was performed to confirm the specificity of the amplified products. Gene expression levels were calculated using the 2−ΔΔCT method which was normalized against the β-actin threshold cycle values. The data were presented as the fold change in the normalized gene expression relative to the untreated control.
The Specific Primer Sequences for Real-Time RT-PCR.
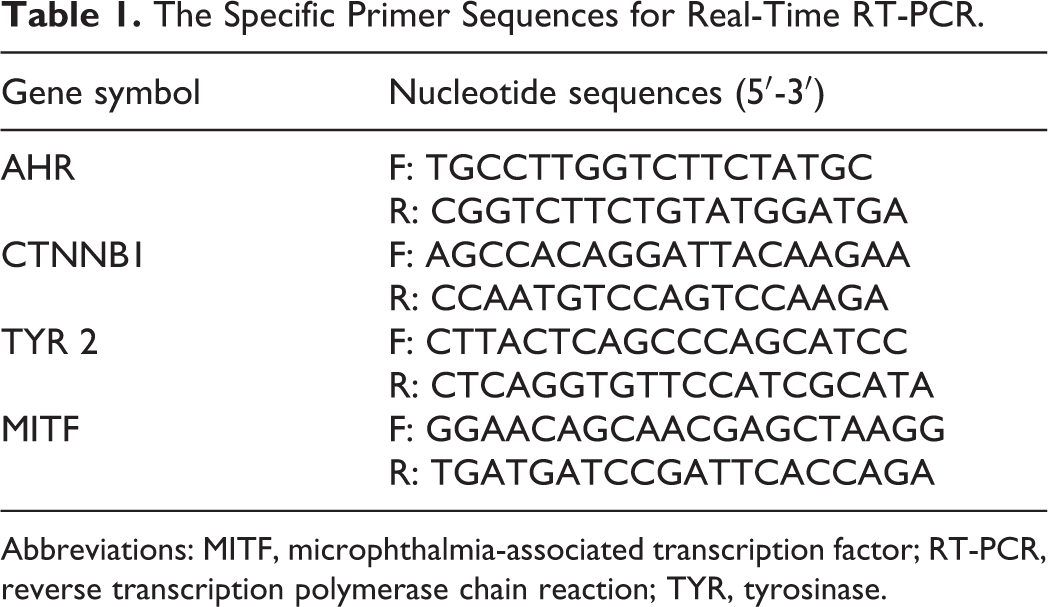
Abbreviations: MITF, microphthalmia-associated transcription factor; RT-PCR, reverse transcription polymerase chain reaction; TYR, tyrosinase.
Tyrosinase Activity
Tyrosinase activity was measured as described previously, with a slight modification.
16
Briefly, murine B16F10 cells were first treated with α-MSH for 24 hours and were then washed with cold PBS and lysed at 4 °C in the lysis buffer (10 mM Tris-HCl, PH 7.4, 150 mM NaCl, 1 mM EDTA, 1% Nonidet P40, 0.1% sodium deoxycholate, and 0.1% sodium dodecyl sulfate). The cell lysates were centrifuged at 13,000 × g at 4 °C for 20 minutes. Then, 100 µL of the cell extract was transferred to a 96-well plate containing 100 µL of
Melanin Content Determination
The melanin contents of the cultured B16F10 cells were measured as described previously with a slight modification. 14 After treatments, cells were washed twice with ice-cold PBS and were lysed with the RIPA buffer (50 mM Tris-HCL, PH 7.4, 150 mM NaCl, 1% Nonidet P40, and 0.1% sodium dodecyl sulfate) for 10 minutes. The lysates were centrifuged at 10,000 g for 10 minutes. The protein concentration was analyzed using supernatants; pellets which contained intracellular melanin were dissolved in 1 N NaOH containing 10% DMSO at 80 °C for 1 hour. Then, the absorbance was measured at 405 nm using a plate reader.
Statistical Analysis
All the experiments were performed at least in 3 independent triplicates. All data were expressed as mean ± SD to compare experimental groups; the 1-way analysis of variance test followed by the Tukey test was used. Probability P values <0.05 were considered significant statistically.
Result
Efficacy of AhR Silencing
To investigate the effect of α-MSH on AhR pathway, efficacy of AhR silencing was examined by the quantification of the EROD activity in murine B16F10 cells. As shown in Figure 1, the EROD activity in the transfected cells (with siRNA against AhR and treated with FICZ 10 nM) was 14% compared to the scrambled siRNA.
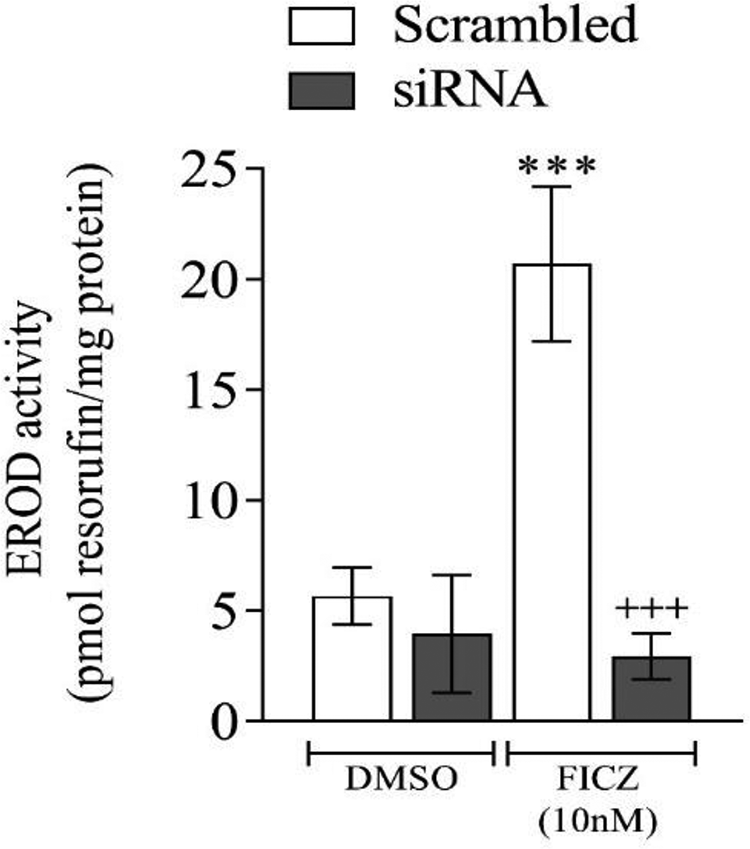
The silencing of aryl hydrocarbon receptor (AhR) pathway in murine B-16F10 cells: the cells were transfected with AhR small interfering RNA (siRNA) or scrambled non-targeting RNA for 15 hours. After the transfection period, the cells were treated with a vehicle (dimethyl sulfoxide, DMSO) or FICZ (10 nM) for 24 hours, and 7-ethoxy-resorufin-O-deethylase (EROD) activity was measured. (+++) indicates the significant differences between the cells treated with AhR siRNA and those treated with scrambled RNA. (***) indicates the significant differences between the cells treated with FICZ or DMSO.
Effects of α-MSH on Genes Expression Involved in Melanogenesis
Next, the effect of α-MSH on the mRNA levels of AhR, CTNNB1, MITF, and TYR was measured after AhR silencing. The results showed that α-MSH treatment resulted in an increase in the mRNA levels of AhR, CTNNB1, MITF, and TYR (Figure 2A-D). In AhR siRNA-treated groups, the levels of AhR, MITF, and TYR genes were significantly lower compared to the scrambled group treated with α-MSH (Figure 2A-D). Next, the effect of XAV939 (1 µM) on the mRNA levels of AhR and MITF was assessed after treatment with α-MSH. The results showed that XAV939 treatment which inhibits Wnt/CTNNB1 mediated transcription, resulted in a decrease in the mRNA levels of AhR (Figure 3A). In α-MSH-treated cells, the expression of AhR was significantly reduced by XAV939 (Figure 3A). Although XAV939 exposure reduces the expression MITF, the reduction was not significant (Figure 3B).
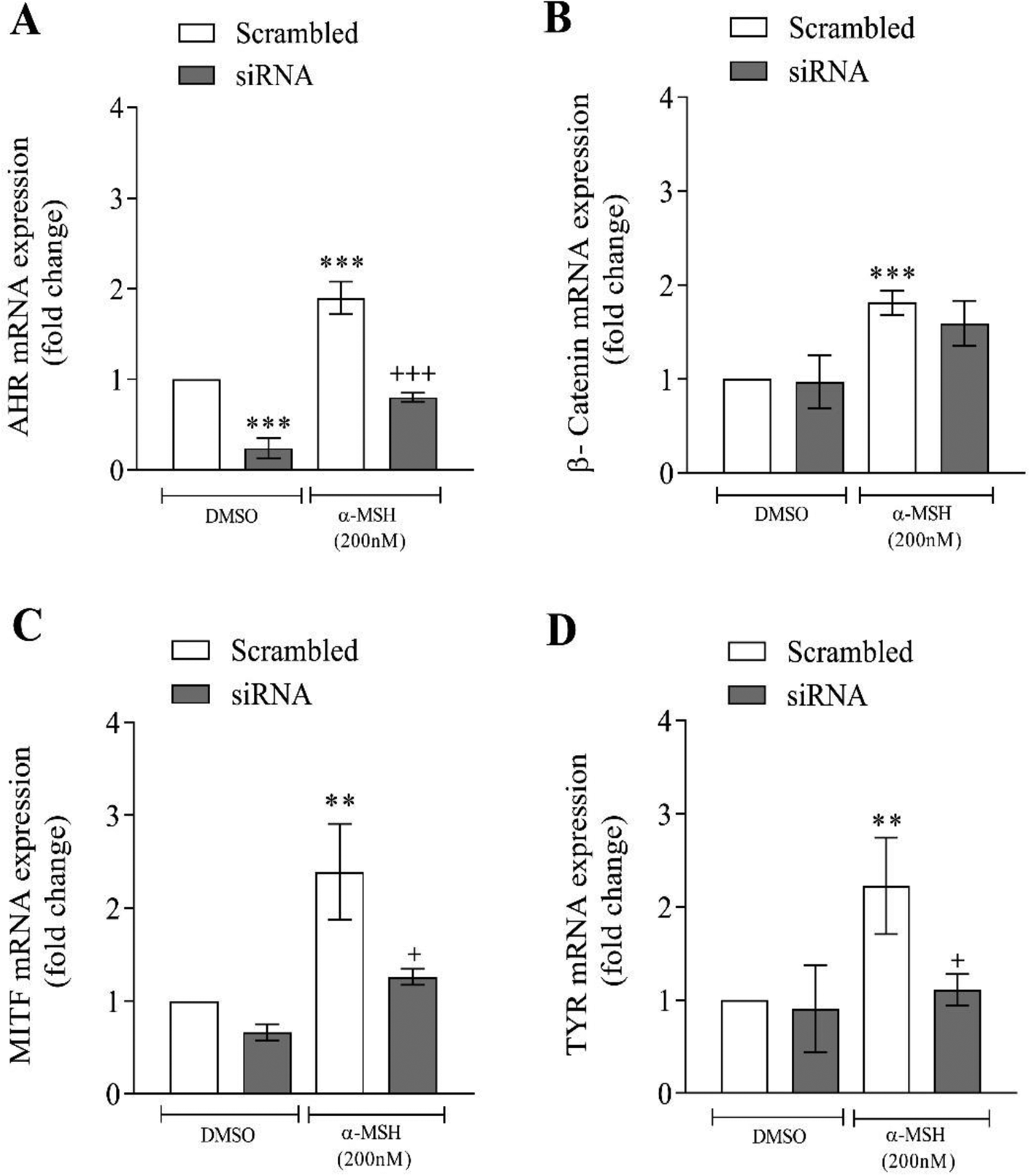
Effects α-melanocyte-stimulating hormone (α-MSH) on the mRNA levels of aryl hydrocarbon receptor (AhR), CTNNB1, microphthalmia-associated transcription factor (MITF), and tyrosinase (TYR) after silencing of AhR. Murine B-16 cells were transfected with AhR small interfering RNA (siRNA) or scrambled nontargeting RNA for 15 hours. After the transfection period, the cells were treated with a vehicle (dimethyl sulfoxide, DMSO) or α-MSH for 24 hours. Real-time reverse transcription polymerase chain reaction (RT-PCR) of the mentioned genes was evaluated. Values are expressed as means ± SD; asterisks denote significant differences (**P < 0.01 and ***P < 0.001) between the control and other treated groups and significant differences (+P < 0.05 and +++P < 0.001) between siRNA and the cells treated with scrambled RNA.
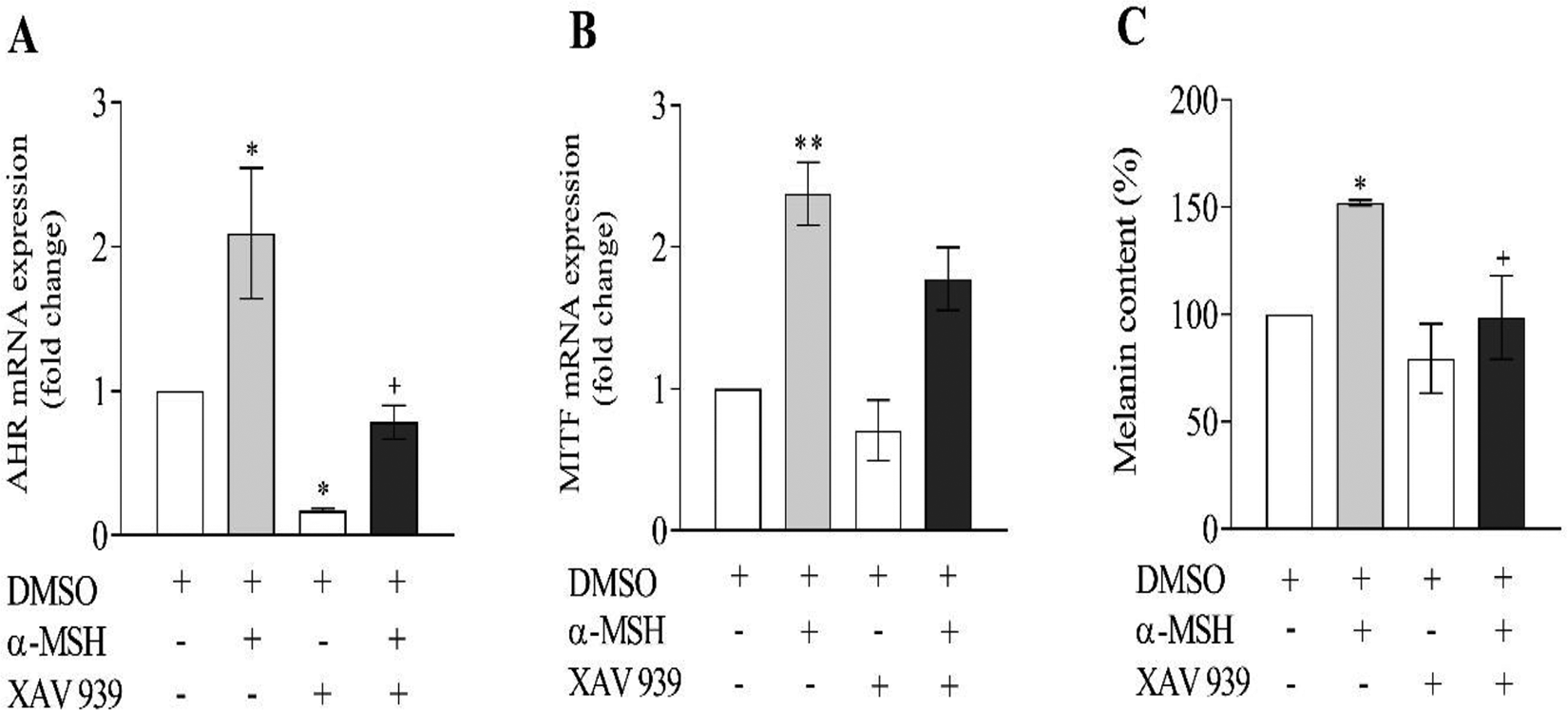
Effects of α-melanocyte-stimulating hormone (α-MSH) and XAV939 on the mRNA levels of (A) aryl hydrocarbon receptor (AhR) and (B) microphthalmia-associated transcription factor (MITF). The cells were treated with a vehicle (dimethyl sulfoxide, DMSO) or XAV939 or α-MSH for 24 hours. Reverse transcription polymerase chain reaction (RT-PCR) of the mentioned genes was performed. Values are expressed as means ± SD; asterisks denote significant differences (*P < 0.05 and **P < 0.01) between the control and other treated groups and significant differences (+P < 0.05) between the cells treated with α-MSH and α-MSH+XAV939. (C) Murine B16F10 cells were treated with α-MSH and XAV939 either alone or together and cellular melanin content was determined. Asterisks denote significant differences (*P < 0.05) between the control and other treated groups and significant differences (+P < 0.05) between the cells treated with α-MSH and α-MSH+XAV939.
Effects of α-MSH on the TYR Activity and Melanin Content of Murine B16F10 Cells
Murine B16F10 cells were incubated in media containing various concentrations of α-MSH (0.1, 0.2, and 0.4 µM) for 24 hours. The TYR activity and cellular melanin content were, respectively, 128%, 143%, 149% and 121%, 152%, 166% of the controls (Figure 4A and 4C). As shown in Figure 4B and D, the cellular TYR activity and intracellular melanin contents were significantly reduced in the groups treated with siRNA targeting AhR compared with the scramble groups. Finally, the effect of XAV939, an inhibitor Wnt/CTNNB1-mediated transcription, on the cellular melanin contents was measured after treatment with α-MSH. In α-MSH+ XAV939-treated cells, cellular melanin contents declined significantly than the α-MSH group (Figure 3C). There was a reduction in melanin level in the XAV939 group, but this reduction was not statistically significant compared to the control group (Figure 3C).
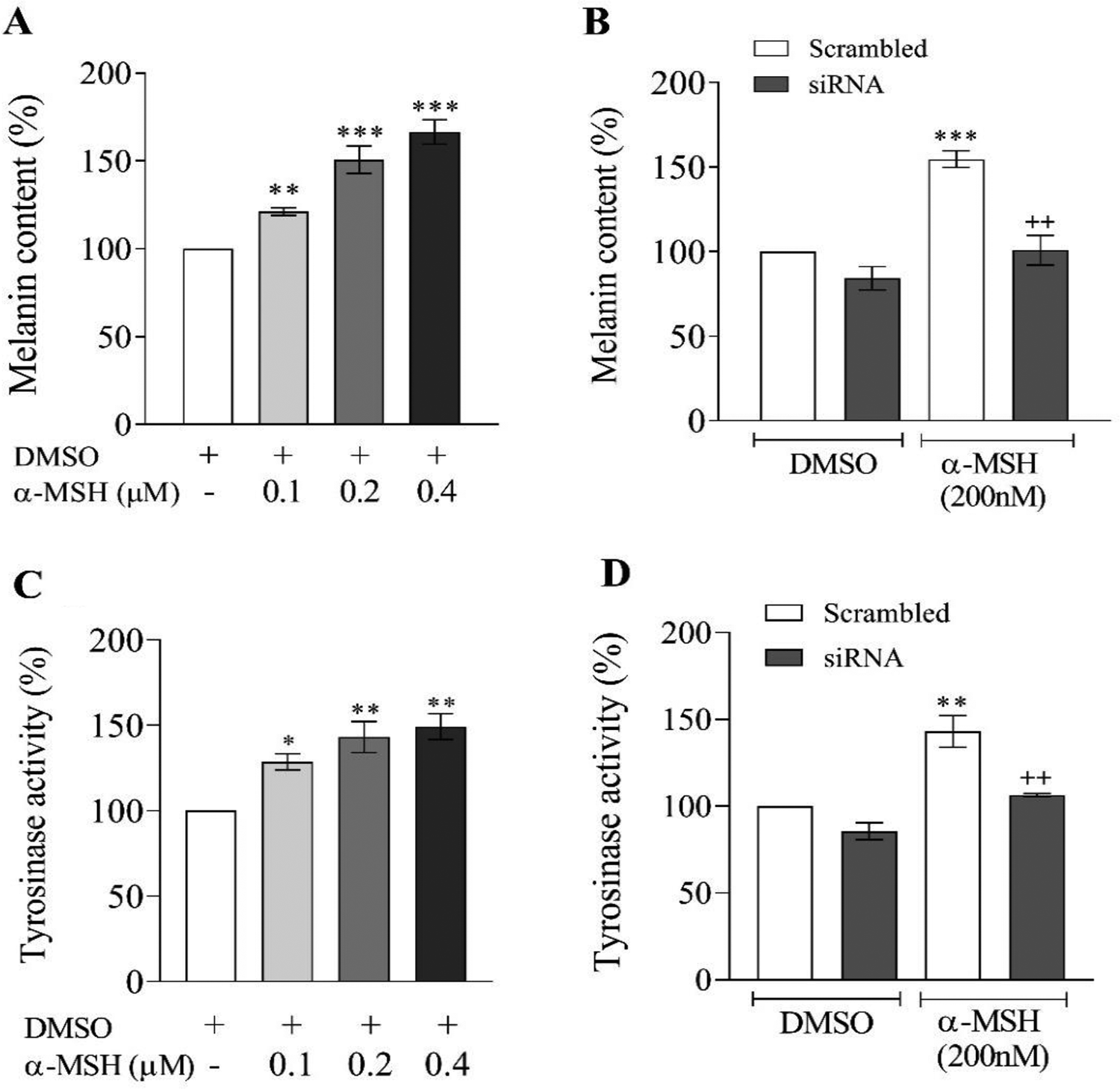
Effects of α-melanocyte-stimulating hormone (α-MSH) on the melanin levels and tyrosinase activity in murine B16F10 cells. Murine B16F10 cells were treated for 24 hours with the indicated concentrations of α-MSH. (A and C) The melanin levels and tyrosinase activity were determined. (B and D) Cells were transfected with aryl hydrocarbon receptor (AhR) small interfering RNA (siRNA) or scrambled nontargeting RNA for 15 hours. After the transfection period, the cells were treated with a vehicle (dimethyl sulfoxide, DMSO) or α-MSH for 24 hours. The melanin level and tyrosinase activity were determined. Asterisks denote significant differences (*P < 0.05, **P < 0.01, and ***P < 0.001) between the control and other treated groups and significant differences (++P < 0.01) between AhR small interfering RNA (siRNA) and the cells treated with scrambled RNA.
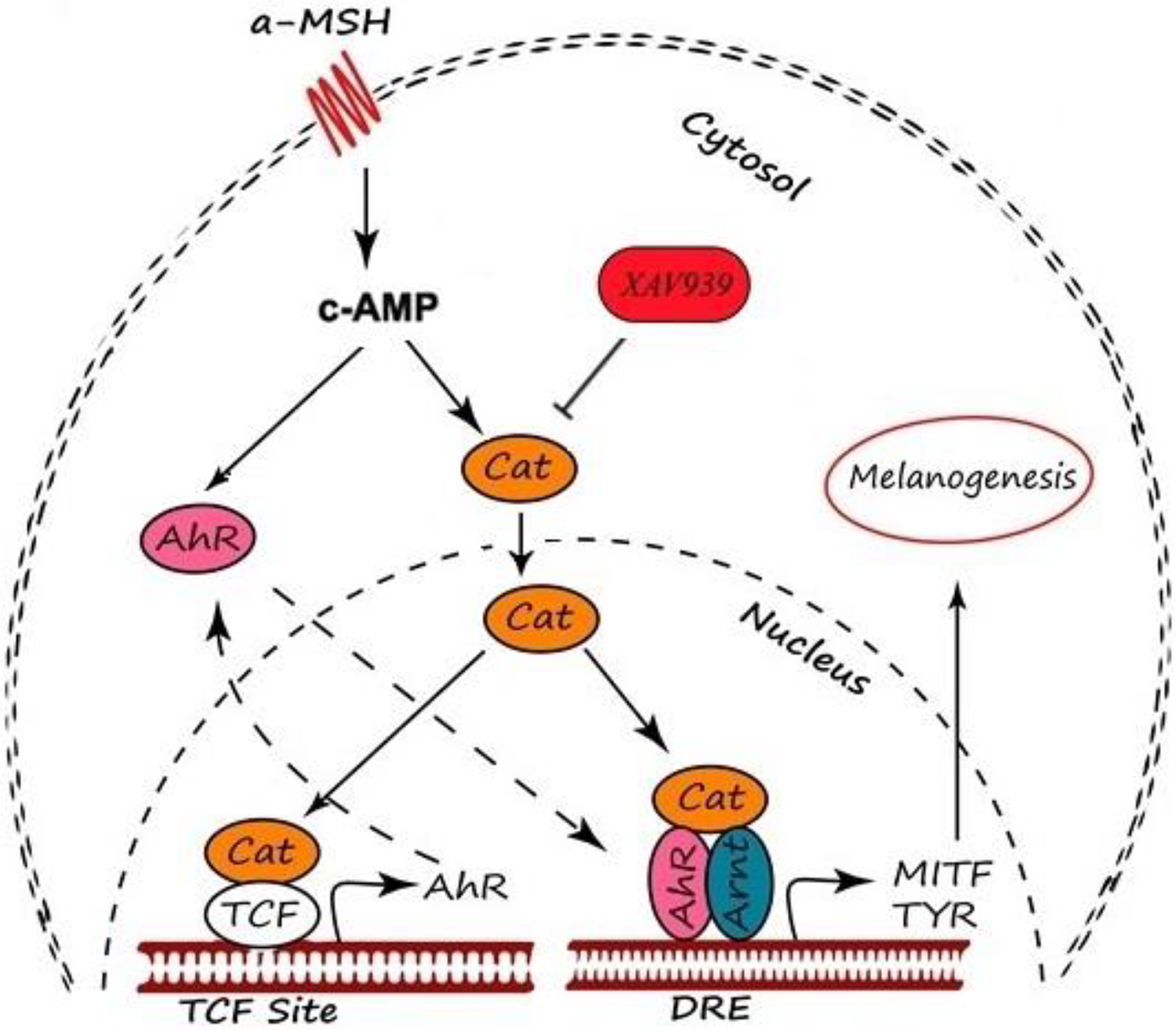
Schematic diagram showing effects of α-MSH in aryl hydrocarbon receptor (AhR)–induced melanogenesis. α-MSH increase melanin production by upregulating the expression of CTNNB1, AhR, microphthalmia-associated transcription factor (MITF), and tyrosinase (TYR) at the transcriptional level (Figure 5). AhR indicates aryl hydrocarbon receptor; Arnt, aryl hydrocarbon receptor nuclear translocator; α-MSH, alpha-Melanocyte-stimulating hormone; CTNNB1, catenin beta 1; DRE, dioxin response element; TCF, T cell factor.
Discussion
Increasing evidence suggests that AhR transcription factor could affect many critical aspects of the physiology of skin cells. 17 In the current study, we demonstrated that α-MSH can induce melanogenesis in an AhR-dependent manner. Previous studies showed that tanning responses and TYR activity in AhR-deficient mice were significantly compromised. 14 Data obtained from the normal human melanocyte cells also indicated that AHR ligands induce melanogenesis. 12,13
In α-MSH-treated B16 cells, the expression levels of MITF and consequently TYR were higher than the control group. This was in line with the results of previous studies demonstrating that MITF and TYR gene expressions were elevated after being exposed to α-MSH. 18,19 In AhR silencing experiments, MITF and TYR expressions decreased significantly. These results suggested that MITF and TYR expression was mediated by the AhR pathway. These results were also supported by the Nakamura et al who observed that MITF was significantly inhibited by silencing the AhR gene. 13 The effect of AhR silencing on the melanin and TYR activity levels in the presence of α-MSH was tested. The results obtained from our experiments showed that in α-MSH treated cells, levels of melanin and TYR activity were significantly reduced by silencing the AhR. These results suggest a role of AHR in α-MSH-induced melanogenesis.
Interestingly, the expression of AhR and CTNNB1 increased significantly in the presence of α-MSH. Furthermore, changes in CTNNB1 gene expression by α-MSH were not significant in either scramble or AhR siRNA groups. The cross talk between the AhR and WNT/CTNNB1 pathway has been vigorously studied, but the complexities in this interaction are still vague. Previous reports indicated that CTNNB1 expression was strongly induced in melanocytes and astrocytes after exposure to AhR ligands. 13,20 In contrast, a continual AhR activation by TCDD caused inhibition of CTNNB1 21 and expression of Wnt/CTNNB1 catenin target genes. 22 Previous studies have shown that α-MSH stimulates melanogenesis via CTNNB1 protein. 18,23 It was also observed that treating of liver and prostate cells with a medium enriched with a physiological ligand of CTNNB1 signaling, Wnt3A, resulted in a significant rise in the AhR protein and mRNA levels. 22,24 Therefore, an increase in the AhR expression by α-MSH in our study might be due to the increase in either synthesis or stabilization of the CTNNB1 protein. It is noteworthy to mention that AhR null mice melanocytes express low levels of stem cell factor-1 (SCF) and c-kit receptor compared to those of wild types. 14 Binding SCF to the extracellular domain of the c-kit receptor leads to the activation of PI3K/Akt signaling pathway and the accumulation of CTNNB1 protein in the melanocytes by inhibiting glycogen synthase kinase 3β. 25,26
After that, the effects of CTNNB1 on AhR and MITF gene expression stimulated by α-MSH was measured using a Wnt/CTNNB1 signaling pathway inhibitor (XAV939). XAV939 increases the protein levels of the axin-GSK3β complex and promotes the degradation of CTNNB1 by inhibiting the tankyrase 1 (a poly-ADP-ribosyltransferase) and 2 activity. 27 The results of this study demonstrated that XAV939 suppressed α-MSH-stimulated AhR expression significantly. These results suggested that α-MSH-stimulated AhR expression was mediated by the CTNNB1 pathway. Further, in cell culture, specific silencing of CTNNB1 expression lessens the levels of melanocyte differentiation-associated markers such as melanin synthesis and TYR activity. 23 In addition, exposure to the combinations of α-MSH and XAV939 did not show a significant alteration in the MITF expression compared to that of the α-MSH group. This finding is not unexpected because α-MSH can stimulate melanogenesis through different pathways. 8 Then, the effect of XAV939 on the melanin levels in the presence of α-MSH was examined. Increased melanin content in murine B16F10 cells by α-MSH was inhibited by XAV939 in this study. These results suggest that the CTNNB1 pathway might be involved in the α-MSH-stimulated melanogenesis.
The increased expression of CTNNB1 gene could not prevent the reduction in MITF and TYR levels in AhR-silenced experiments. Therefore, an increase in the MITF and TYR expression by α-MSH in these studies might be due to interaction of CTNNB1 and AhR at DNA binding sites. Likewise, interaction of CTNNB1 and AhR on CYP1A1 gene expression can be mediated by their physical interaction at DNA binding sites. 21 Therefore, the simultaneous presence of CTNNB1 and AhR is likely to be required for the induction of α-MSH-stimulated melanogenesis.
Ligand-independent nuclear translocation of AhR is another aspect of AhR physiology that can be considered. Although toxic compounds and some endogenous chemicals can directly activate the AhR, cyclic adenosine monophosphate (c-AMP) can also function as an endogenous activator of the AhR and cause its translocation to the nucleus in the absence of any exogenously added ligand. 28 It is also known that activation of melanocortin receptor by α-MSH can lead to transient increases in cAMP synthesis. This in turn activates protein kinase A (PKA), which then phosphorylates c-AMP response element binding protein (CREB) transcription factor to bind to the c-AMP response elements in the promoter regions of c-AMP responsive genes, including MITF. 5 Blockage of c-AMP synthesis has also been shown to inhibit melanogenesis in B16 cells. 28 In addition, activation of AhR by forskolin increases melanin synthesis via cAMP production, PKA activation, and CREB phosphorylation. 29 Furthermore, c-AMP is known to activate Wnt/CTNNB1 signaling 30 and it is known that WNT/CTNNB1 pathway is a transcriptional regulator of AhR target genes. 31 Therefore, the increased expression of α-MSH-induced AhR in our study may be due to the influence of c-AMP directly or indirectly via the Wnt/CTNNB1 pathway or both pathways.
Together, our data suggest the possibility that AhR occupies a crucial position in the signal transduction cascade leading to melanogenesis in response to both normal physiological and toxic stimuli.
Footnotes
Authors’ Contribution
A.G.B. and A.M-.B. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. A.J., M.K., M-.R.A., H.M. contributed to design; contributed to analysis; drafted manuscript; and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgments
The authors of this manuscript wish to express their gratitude to Shiraz University of Medical Sciences, Shiraz, Iran.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant of Shiraz University of Medical Sciences for the accomplishment of the PhD thesis of Ali Ghaffarian Bahraman [Grant number: 95-01-05-12553].
