Abstract
The present study was aimed to evaluate the chemopreventive potential of galangin against benzo(a)pyrene (BaP)-induced stomach carcinogenesis in Swiss albino mice. Stomach cancer was induced in experimental mice using BaP oral administration. The mice were treated with galangin (10 mg/kg b.wt.) before and during BaP administration. Oral administration of galangin at a dose of 10 mg/kg b.wt. significantly (p < 0.05) prevented the tumor incidence, tumor volume in the experimental animals. Further, galangin pretreatment prevents BaP-induced lipid peroxidation and restores BaP-mediated loss of cellular antioxidants status. It has also been found that galangin prevents BaP-induced activation of phase I detoxification enzymes. Furthermore, galangin pretreatment prevented the BaP-induced overexpression of cytochrome P450s isoform genes (CYP1A1, CYP1B1), aryl hydrocarbon receptor system (AhR, ARNT), transcriptional activators (CBP/p300, NF-kB), tumor growth factors, proto-oncogenes, invasion markers (TGFB, SRC-1, MYC, iNOS, MMP2, MMP9) and Phase II metabolic isoenzyme genes (GST) in the stomach tissue homogenate when compared to the control groups. The western blot results confirm that galangin (10 mg/kg. b.wt.) treatment significantly prevented the BaP-mediated expression of ArR, ARNT, and CYP1A1 proteins in the mouse stomach tissue. Therefore, the present results confirm that galangin prevents BaP-induced stomach carcinogenesis probably through modulating ArR and ARNT expression in the experimental mice.
Introduction
Stomach cancer, also called gastric cancer, ranks fifth in worldwide cancer incidence. It ranks the third position in the case of cancer mortality. 1 About 42% of stomach cancer incidence was reported in China alone. Despite the development of modern diagnostic and therapeutic tools, the global stomach cancer burden remains high. It affects mainly the proximal stomach, particularly near the junction of the esophagus. Adenocarcinoma is the major cancer sup-type, and it comprises 90% of gastric malignancies. 2 The adenocarcinoma of the stomach has been linked with exposure to polycyclic aromatic hydrocarbons (PAHs). 3
Benzo(a)pyrene (BaP), a prominent polyaromatic hydrocarbon (PAH), is a product formed during repeated cooking of oils, grilled/broiled foods, incomplete combustion of crude oils, coal tars, and cigarette smoke. 4 The International Agency for Research on Cancer listed BaP as a group I carcinogen. 5 The human system has absorbed the BaP by inhalation, ingestion, and skin exposure. The BaP inhalation and ingestion mainly affect the small intestine, stomach, liver, and esophagus. 6 The BaP has been activated by phase I and phase II metabolic enzymes that resulted in highly reactive epoxide metabolites. Further, the cytochrome P450 (CYP1A1 and CYP1B1) has also been significantly increased in the stomach and liver of experimental rats. 7 This BaP generated 7,8-diol-9,10-epoxide metabolites readily binds with DNA forms BaP-DNA adducts. These DNA adducts interfere with the vital cellular process like DNA replication that resulted in mutagenesis and eventually linked with carcinogenesis. The dose-dependent carcinogenic potential of BaP in experimental animals has well been established. 8 Further, the BaP-mediated carcinogenesis was mediated via the aryl hydrocarbon receptor (AhR) and the ARNT signaling pathway. 9 The AhR activation BaP modulates the overexpression of inflammatory signaling events that are involved in carcinogenesis. It has been reported that the BaP promotes carcinogenesis in stomach cells through the upregulation of matrix metalloproteinase-2 and -9 (MMP-2 and -9) and c-MYC expression. Further, reports also illustrate that the epigenetic activation of Bcl6b promotes gastric cancer through the amplification of the gastric inflammatory response. 10
Lipid peroxides play a major role in the activation of BaP. The lipid peroxidation products such as malonaldehyde directly interact with BaP and form highly reactive BaP-quinone products even in the absence of cytochrome P450 oxidase system. 11 The BaP-mediated reactive metabolites induce oxidative damages in the stomach tissues. Antioxidant enzymes are the major defense mechanism against BaP-induced oxidative damages. 12 Antioxidant enzymes such as superoxide dismutase (SOD), catalase, and glutathione peroxidase (GPx), and non-enzymatic antioxidants like reduced glutathione (GSH), protects the cellular milieu from oxidative damages. Since BaP induces oxidative stress-mediated carcinogenesis, the antioxidant phytochemicals gain importance as potential chemopreventive agents. Further, dietary phytochemicals have been reported as an inhibitor of CYP1A1 through binding with AhR. 13 Many flavonoids are reported as AhR ligands.14–16 Galangin is a dietary flavonol found in Alpinia officinarum, Helichrysum aureonitens, and in the rhizome of Alpinia galanga and propolis. 17 Galangin has been reported to possess several interesting pharmacological properties. It inhibits alpha-glucosidase activity, thereby prevents the formation of glycation products. 18 It prevents airway remodeling by inhibition of ROS generation and modulating MAPK signaling pathway. 19 Further, galangin treatment induces apoptotic cell death in gastric cancer cell lines 20 and suppresses the formation of epithelial-mesenchymal transition in renal cell carcinoma. 21 Although galangin is reported as a major functional food component, 22 its preventive effect against BaP-induced stomach carcinogenesis has not been reported. Therefore, in this study, we studied the chemopreventive effect of galangin against BaP-induced carcinogenesis through modulating AhR and CYPA1 in Swiss albino mice.
Materials and methods
Animals
Swiss albino mice aged 6–7 weeks were employed for stomach carcinogenesis studies. The experimental mice were housed at room temperature (24° ± 3°C) with a 12 h light:12 h dark cycle. Each mouse was maintained using separate polypropylene cages. Standard mice feed and tap water were provided ad libidum. The diet and water consumption by the control and treated mice were recorded. Animal experiments were conducted as per the procedures complied with the Declaration of Helsinki, as revised in 1996. The experiments were conducted after approval by the Institutional Animal Ethical Committee (IAEC). Animals were sacrificed by cervical dislocation after the experimental period. The small intestine was excised for performing various biochemical estimations, and part of the tissue was stored at −80°C deep freezer.
Carcinogenesis and experimental design
We followed the standard chemical carcinogenesis protocol to induce stomach tumorigenesis. The BaP-induced multistage carcinogenesis in mice was performed according to the method of Goyal et al.
23
For this purpose, 6–8 weeks-old mice were grouped (n = 8) as given below: Group I: The mice received 100 µl sesame oil using oral gavages two times per week for 4 consecutive weeks. Group II: Galangin alone administered: Experimental mice were given galangin at a concentration of 10 mg/kg/b.wt. using oral gavages daily throughout the experimental periods. The optimum therapeutic dose was selected by a separate acute toxicity study (Supplementary Table 1). Group III: Carcinogen (BaP) alone treated: The experimental mice of this group were given 1 mg/kg b.wt. of BaP in 100 µl sesame oil using oral gavages two times per week for 4 consecutive weeks; totally mice receive eight administrations of BaP. Experimental mice were sacrificed 14 weeks after the last administration of BaP. Group IV: Galangin + BaP-treated: The mice of this group were received 10 mg/kg/b.wt of galangin using oral gavages daily for 2 weeks (pretreatment). This group’s animals were administered with 1 mg/kg b.wt. of BaP in 100 µl sesame oil using oral gavages twice a week for 4 consecutive weeks. The galangin (10 mg/kg b.wt.) treatment was continued throughout the BaP experimental periods (co-treatment).
All the group of experimental mice was given sterile drinking tap water throughout the experimental duration.
Tumor incidence
The body weights of the experimental mice were noted before and after the experimental period. The number of tumors, % tumor incidence and tumor volume (mm3/mice) were calculated and tabulated. A digital caliper was used to measure the tumor volume (width and length). The tumor volume was calculated as per the formula: Tumor volume (mm3) = (l × w2)/2; where l was length and w was width.
Antioxidants studies
The whole stomach tissue was taken out from the experimental mice for the estimations of antioxidants parameters. The collected stomach tissue was washed once in PBS. Stomach tissue was homogenized in a Potter-Elvehjem homogenizer using a Teflon pestle for 3 min in Tris HCl buffer (0.1 M). The resulting tissue homogenate was centrifuged for 10 min, and the supernatant was used to analyze various antioxidants assays.
The status of lipid peroxidation and antioxidant enzyme activities were measured colorimetrically in the stomach tissue homogenate as per the previous methods. The TBARS and LOOH were the lipid peroxidation products formed during BaP administration in the stomach tissue. The stomach tissue levels of TBARS were estimated using the method of Ohkawa et al. 24 The concentration of stomach tissue lipid hydroperoxide (LOOH) was estimated using method of Jiang et al. 25 The SOD is the antioxidant enzyme that dismutates superoxide anion radicals to water and hydrogen peroxide. The stomach tissue SOD activity was determined by the method of Kakkar et al. 26 Catalase is the major antioxidant enzyme found in all the tissues that degrade hydrogen peroxide into water and molecular oxygen. The stomach tissue catalase activity was estimated by the method of Hadwan. 27 The GPX is another antioxidant enzyme that degrades lipid hydroperoxides and hydrogen peroxide to water. The stomach tissue GPX activity in stomach tissue homogenate was determined by the method of Chow and Tappel. 28 The reduced GSH is an endogenous non-enzymatic antioxidant that scavenges oxygen-derived free radicals. The stomach tissue reduced GSH levels were estimated by the method of Davidson and Hird. 29
Estimation of detoxification enzymes
Phase 1 detoxification enzymes activate the toxic compounds for detoxification through the P-oxidases cytochrome (CYP) enzymes. Cytochrome P450 and cytochrome b5 are the Phase I enzymes involved in detoxification. The level of these detoxification enzymes was measured by Omura and Takesue 30 and Mihara and Sato, 31 respectively. Cytochrome P450 was estimated by CO difference spectrum. Cytochrome P450 combines with CO yield a colored product which was measured at 450 nm. The cytochrome b5 was estimated based on the difference between the absorption spectrum of the reduced and oxidized form of cytochrome b5.
Stomach carcinogenesis related gene expression analysis by qRT-PCR
The total mRNA population from stomach tissue was isolated from the stomach tissue of experimental animals using an RNeasy mini kit and used for qRT-PCR arrays. The RNA quality was determined by spectrophotometer and agarose gel electrophoresis. Then, cDNA was prepared using a reverse transcriptase enzyme kit (Qiagen, USA). The custom-based qRT-PCR array analyzed the relative expression pattern of Cytochrome P450s isoform genes (CYP1A1, CYP1B1, Nqo1) aryl hydrocarbon receptor system (AhR, ARNT), transcriptional coactivators (CBP/p300, NF-kB), proto-oncogenes (SRC-1, MYC) tumor growth factors (TGFB, iNOS, MMP2, MMP9), Phase II metabolic isoenzyme genes (GST) and proto-oncogenes (SRC-a, MYC). The gene expression fold changes were calculated using 2−ΔΔCt were plotted as cluster grams. The expression-based heat maps were constructed using http://www.heatmapper.ca/ online tool. GAPDH was used as the internal standard.
Western blot analysis
About 50 mg of tumor-bearing stomach tissue was used for protein isolation using sample lysis buffer (Tris-SDS-mercaptoethanol-glycerol-bromophenol blue). Protein samples were extracted from the stomach tissue of galangin and/or BaP treated experimental animals using RIPA buffer that consists of the protease cocktail inhibitor (1 µg/1 µl). The total protein concentration in the tissues was measured by the Bradford method. 32 We measured the expression pattern of AhR, ARNT, and CYP1A1 in the total protein lysate. The amounts of protein concentration were measured by using the Nanodrop Spectrophotometer (Thermo Scientific, Austria). We followed the methodology for western blot as described by Johnson. 33 The protein bands were spotted by using a chemiluminescence detection method. β-actin was used as a loading control, and the protein expression was normalized to β-actin expression.
Statistical methods
The data were generally given as mean ± standard deviation (SD) unless otherwise stated. Analysis of variance (One way ANOVA) followed by Duncan Multiple Range Test (DMRT) was employed to understand the differences between BaP treated and/or galangin treated groups. The data were considered significant if the p values were found less than 0.05. The statistical analysis was performed using SPSS 16 package.
Results
Acute toxicity of galangin in Swiss albino mice
Dose-Probing Tests were conducted to determine the optimum experimental concentration of galangin (https://www.fda.gov/media/72257/download). The administration of different concentrations of galangin (2.5–10 mg/kg b.wt) has not induced mortality during the 14 days observation period. However, 16.6% of animals died when the galangin dose was raised to 12.5 mg/kg b.wt. A further increase in the dose of galangin (15 mg/kg.b.wt.) resulted in 33.3% mortality. About 50% reduction in the survival of mice was observed at 17.5 mg/kg.b.wt (LD50/14 days). Finally, 25 and 50 mg/kg.b.wt. galangin treatment caused 100% (Table 1). Therefore, we used 10 mg/kg b.wt galangin for the chemoprevention experiments (Supplementary Table 1).
Effect of galangin (10 mg/kg b.wt.) on BaP-induced tumor incidence, total number of tumors and tumor volume (mm3/animal) in experimental rats.

Percentage tumor incidence was calculated based on the number of tumors formed during BaP treatment. Tumor volume was recorded using a digital caliper. The differences between the experimental groups were compared by One way ANOVA followed by DMRT.
Values not sharing a uniform superscript (a,b,c) differ significantly at p ≤ 0.05.
Effect of galangin on tumor incidence in BaP experimental mice
We observed 100% tumor incidence in BaP treated mice. Totally eight tumors were observed in eight BaP administered mice. The tumor volume was found to be 8.85 ± 2.94 mm3/animal in BaP alone treated groups. Whereas oral administration of galangin at a dose of 10 mg/kg b.wt. significantly (p < 0.05) prevented the tumor incidence (37.5%) and tumor volume (3.24 ± 1.98), when compared to BaP alone, treated groups (Table 1). We observed no tumor formation in the sham control, and galangin alone treated mice at the end of the experimental periods.
Effect of galangin on stomach tissue antioxidant status
Antioxidant enzymes are the primary defense system that protects the cellular milieu from xenobiotics induced free radicals. SODS, CAT, GPx were the major antioxidant enzymes, and their activities in the stomach tissue were measured. The activities of SOD, CAT, GPx were found to be attenuated in the BaP alone treated mice stomach homogenate. Oral administration of galangin (10 mg/kg b.wt.) significantly (p < 0.05) restored the activities of SOD, CAT, GPx, when compared with BaP alone treated mice. The reduced GSH is the primary non-enzymatic antioxidant in the cellular milieu. We observed attenuated levels of reduced GSH levels in BaP alone treated mice stomach tissue homogenate. Whereas, galangin pretreatment before BaP treatment significantly improved the reduced GSH levels in the mice stomach tissue homogenate. However, there was no significant difference (p < 0.05) in the antioxidants status in the mice administered with galangin alone when compared to the sham control group (Table 2).
Effect of galangin administration (10 mg/kg b.wt) on the activities of enzymatic antioxidants (SOD, catalase and GPx) and reduced GSH levels in experimental animals.

* The SOD activity was expressed as the amount of enzyme required to inhibit 50% NBT reduction/min, **the catalase activity was expressed as μmol of hydrogen peroxide utilized/min, and ***the GPx activity was expressed as µg of reduced glutathione consumed/min. The experimental data were expressed as mean ± SD. The differences between the experimental groups were analyzed by One way ANOVA followed by DMRT. Data not sharing a uniform superscript (a, b, c, etc.) differ significantly at p < 0.05 (DMRT).
Effect of galangin on lipid peroxidation status
Lipid peroxidation status has been considered as the major biomarker of tumorigenesis. We observed increased levels of TBARS and LOOH in BaP alone treated mice stomach tissue homogenate as an indication of oxidative tissue damages. Whereas oral administration of galangin prevented BaP-induced TBARS and LOOH levels in the stomach tissue (p < 0.05). However, there was no significant difference in the lipid peroxidation status in the mice administered with galangin alone than the sham control group (Table 3).
Effect of galangin and/or BaP on TBARS and LOOH levels in the tumor-bearing stomach tissue homogenate.

The levels of TBARS and LOOH are measured as mmol/100 g wet tissue. The experimental values are expressed as mean ± SD. The statistical differences between the groups are compared by one way ANOVA followed by DMRT. Values not sharing a uniform superscript (a, b, c,) differ significantly at p < 0.05.
Effect of galangin on phase I detoxification enzymes
Both cytochrome P450 and b5 were significantly increased in BaP treated tumor-bearing mice stomach homogenate than sham control mice. However, the oral administration of galangin prevented BaP-mediated activation of cytochrome P450 and b5 compared to the Bap alone treated group (p < 0.05). Galangin alone treatment showed no significant difference in cytochrome P450 and b5 status compared to sham control mice (Table 4).
Effect of galangin (10 mg/kg b.wt) and/or BaP on cytochrome P450 and cytochrome b5 activities.

The experimental data was expressed as mean ± SD. Values not sharing a uniform superscript (a, b, c…) differ significantly at p < 0.05. (DMRT). *Micromoles of cytochrome P450 formed; **micromoles of cytochrome b5 formed.
BaP induced carcinogenesis related gene expression pattern
The relative expression pattern of Cytochrome P450s isoform genes (CYP1A1, CYP1B1, aryl hydrocarbon receptor system (AhR, ARNT), transcriptional coactivators (CBP/p300, NF-kB), tumor growth factors (TGFB) and Phase II metabolic isoenzyme genes (GST) were found to be overexpressed in the stomach tumor tissue homogenate. Furthermore, proto-oncogenes (SRC-a, MYC) was found to be upregulated in the BaP-induced stomach cancer tissues. Conversely, the galangin pretreatment prevented the overexpression of cytochrome P450s, aryl hydrocarbon receptor system, transcription activators, and Phase II metabolic enzymes related gene expression toward a normal level (Figure 1).

(a) Effect of galangin (10 mg/kg b.wt) on BaP-induced tumorigenesis related gene expression pattern. The relative gene expressions were calculated using 2−ΔΔCt. GADPH was used as an internal standard. Heatmap was constructed using an online tool http://www.heatmapper.ca/expression/. Average linkage was achieved between the experimental groups. Eucledean distance measurement method was achieved. (b) Bar diagram show the differences between fold changes of relative gene expressions between the experimental groups..
Galangin prevents BaP-induced AhR, ARNT, and CYP1A1 expression in the stomach tissue
Galangin on BaP-induced ArR and CYP1A1 expressions was analyzed by western blot analysis. BaP treated mouse stomach tissue clearly shows increased expression ArR and CYP1B1 when compared to the control mouse. Galangin (10 mg/kg. b.wt.) treatment prevented (p < 0.05) the BaP-mediated expression of ArR and CYP1A1 proteins in the mouse stomach tissue (Figure 2).
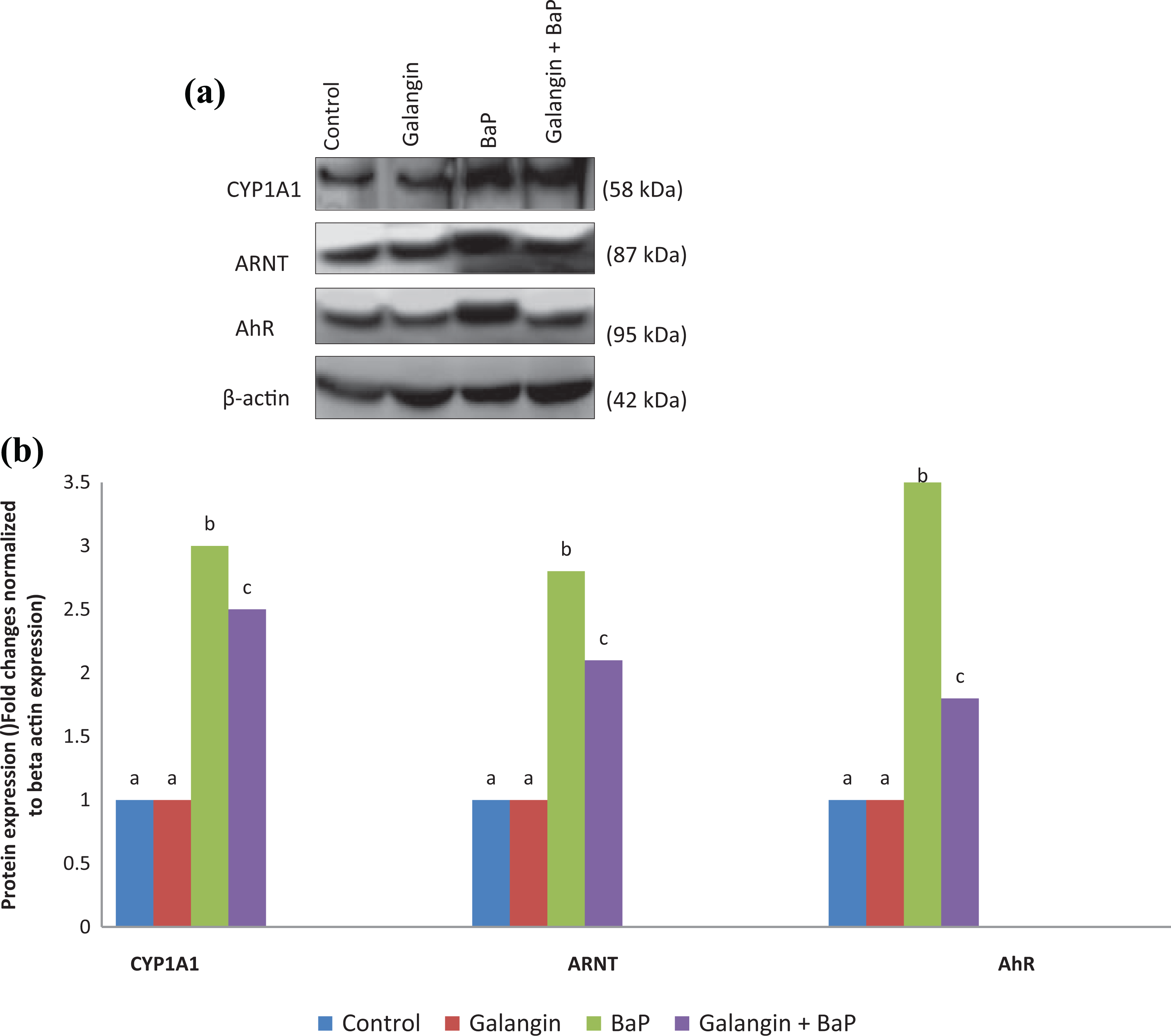
(a) Effect of galangin (10 mg/kg b.wt) on the protein expression pattern of AhR, ARNT and CYP1A1 in the stomach cancer tissue homogenate. Western blot images show the expression pattern of AhR, ARNT and CYP1A1 in BaP and/or galangin + BaP treated stomach cancer tissues. β-actin expression was used to confirm equal loading of protein samples. The bands were developed using chemiluminescence detection kit. (b) The western blot band intensities were analyzed by image “J” software and normalized to β-actin expression. Values not sharing a uniform superscript (a, b, c…) differ significantly at p < 0.05. (DMRT).
Discussion
The gastric cancers detected are mostly adenocarcinomas with a 5-year survival rate of 5–10%. The severe adverse effects of chemotherapy are the major concern in cancer clinics. Ginger has widely been used to treat various gastric disorders. Galangin is the major active bioflavonoid present in ginger has endowed with various pharmacological properties. Galangin exhibits excellent free radical scavenging properties and thereby safeguard human cells from oxidative damages mediated disorders. 34 In this study, we found that galangin prevents BaP-induced gastric carcinogenesis in Swiss albino mice. Galangin possesses hydroxyl substituents on carbon 3 and/or 5. The structure-activity relationship studies illustrated that such hydroxyl groups are responsible for terminating free radical reactions and subsequently for its anticancer potential. 35 In this study, the non-toxic concentration of galangin (10 mg/kg b.wt.) effectively inhibits BaP-induced stomach tumor formation in the mice. The BaP has been classified as a potential pro-carcinogen, and it readily generates several reactive oxygen intermediates. BaP-mediated oxidative damages for solid tumor formation in the stomach have well been established. 8 The EPR studies illustrate the formation of oxygenated radicals from BaP under various reaction conditions. 36 The 6-oxybenzo(a)pyrene radical formed from BaP induces severe oxidative damages in the cellular milieu. 37 We found that a severe oxidative tumor environment in the BaP treated mice. The results showed that increased tumor % of incidence and tumor volume in the BaP alone treated animals. Conversely, galangin pretreatment prevented BaP-mediated tumor incidence and tumor volume. Previous results show that galangin mediates anticancer property through a mitochondrial pathway against hepatocellular carcinoma. 38 Further, recent studies show that galangin regulates several carcinogenic signaling elements in experimental models.21,39 The BaP generates many reactive radicals that make nucleophilic attach to the membrane lipids. We found that increased TBARS and LOOH as a result of BaP-mediated lipid peroxidation. The present results illustrate that galangin treatment significantly inhibited BaP-induced lipid peroxidation. Increased lipid peroxidation markers in the serum of BaP treated mice indicate the severe oxidative damages of membrane lipids due to BaP administration. Administration of galangin before GaP treatment decreased the TBARS levels to those of normal levels. The anti-lipid peroxidative effect of galangin has well been documented. 40 In an earlier study, galangin decreased oxidative status and increased antioxidant status. 41 Thus, the antioxidative action of galangin may have excellent properties to scavenge free radicals.
The BaP-mediated reactive radicals affect the cellular endogenous antioxidants system. The activities of enzymatic antioxidants like SOD, CAT, and GPx and the levels of reduced GSH maintain the cellular redox balance. 42 These first-line cellular antioxidant systems maintain redox homeostasis and prevent several events for BaP-induced multistage carcinogenesis. These antioxidants are further reported to inhibit carcinogen activation in the cellular milieu. 43 In this study, the enzymatic (SOD, CAT, and GPx) and non-enzymatic (reduced GSH) antioxidants were decreased in the serum of BaP administered mice compared with normal healthy mice. These decreased antioxidants might be due to higher utilization by the gastric tissues to combat the oxidative stress induced by BaP administration. Whereas galangin pretreatment significantly prevented the GaP-mediated loss of antioxidant levels in the tumor-bearing stomach tissue. Thus, galangin pretreatment maintains cellular antioxidant status, thereby might prevent BaP-mediated oxidative tumor environment. Galangin possesses the ability to scavenge free radicals by providing hydrogen atoms from their hydroxyl group. 44 The increase in antioxidant enzymes by galangin reflects that it inhibits the process of oxidative stress-induced carcinogenesis. The present study reveals that galangin protects systemic antioxidant status from BaP-induced oxidative stress. Previously, it has been reported that galangin protects against oxidative damages and restores the antioxidants enzyme system in high fructose-fed rats. 45 Further, galangin maintains the antioxidant enzyme system in diabetic rats through modulating mitochondrial function. 46
X-ray crystallographic and NMR studies illustrate the BaP binding distorts the double-helical structure of DNA. 47 This disturbs the normal cellular replication and transcription process, which resulted in carcinogenesis. The BaP has been metabolically activated by cytochrome p450 enzymes, particularly P450A1 (CYP1A1) resulted in the formation of 7,8-diol-9,10-epoxide (BPDE). This activation has been found responsible for the formation of DNA adducts. 48 The BaP induces the expression of CYP1A1 through binding with the aryl hydrocarbon receptor (AhR) in the cytoplasm. The transformed receptor translocates to the nucleus, where it dimerizes with the aryl hydrocarbon receptor nuclear translocator (ARNT). This activates the xenobiotic response elements (XRE). The ARNT heterodimer binds specific DNA sequences located in the promoter regions of XRE (5′-TA/TGCGTG-3′) in the DNA, which resulted in the overexpression of CYP1A1.49,50 In this study, the western blot results showed that BaP administration activates the overexpression of CYP1A1 and AhR in the gastric tissue. Further, galangin pretreatment significantly prevents the BaP-mediated activation of AhR in the mice gastric tissue. Similarly, galangin has been reported to prevent AhR activation during dioxin induced carcinogenesis. 51 Galangin, like flavonoids, possesses structural features that show AhR agonists/antagonists properties. 52 Galangin has previously been proved as a suppressor of the genotoxicity of carcinogenic chemicals. 53 Further, galangin appears to be a potential candidate for the chemoprevention of ovarian cancer and hepatocellular carcinoma in humans.54,55
Identification of potential molecular targets of BaP-induced carcinogenesis is essential for the prevention of carcinogenesis steps. Overexpressed mRNA pattern of several BaP linked XRE a downstream gene was observed in this study (Figure 1(a) and (b)). We noticed the overexpression of both AhR and ARNT in BaP-treated stomach tissues. The AhR-ARNT modulates the target genes such as CBP/p300 (cAMP response element-binding protein), 56 SRC-1 (steroid receptor coactivator 1), p/CIP (p300/CBP/Co-integrator-associated Protein). 57 Further, MYC has been activated during BaP-induced carcinogenesis, and natural phytochemicals prevent this molecular event in experimental models. 58 The AhR ligands like BaP induce the expression of pro-invasion molecules. The overexpression of iNOS, TGFB, MMP2, MMP9 has been well established during BaP-induced carcinogenesis.59,60 The xenobiotic detoxification phase II enzymes like Nqo1 and GST were overexpressed during BaP-mediated carcinogenesis. 61 Interestingly, galangin pretreatment prevents the BaP-mediated activation of these carcinogenic molecules. This indicates that galangin is not only acted as an antioxidant to scavenge BaP-induced free radicals, but it also acts at the molecular level, thereby prevents tumorigenesis events. Therefore, our present findings illustrate the chemopreventive effect of galangin against BaP-induced stomach carcinogenesis in experimental animals.
Conclusion
Thus, galangin behave as a potent chemopreventive agent against BaP-induced stomach tumorigenesis. The galangin pretreatment prevented BaP-mediated tumor formation through modulation of oxidative damage markers in the mice stomach tissue. Further, the galangin treatment modulates the molecules involved in BaP-induced AhR signaling, invasion, and angiogenesis. Hence, galangin may be considered as a potential chemopreventive agent.
Supplemental material
Supplemental Material, sj-pdf-1-het-10.1177_0960327121997979 - Chemopreventive effect of galangin against benzo(a)pyrene-induced stomach tumorigenesis through modulating aryl hydrocarbon receptor in Swiss albino mice
Supplemental Material, sj-pdf-1-het-10.1177_0960327121997979 for Chemopreventive effect of galangin against benzo(a)pyrene-induced stomach tumorigenesis through modulating aryl hydrocarbon receptor in Swiss albino mice by L Wang, J Xue, F Wei, G Zheng, M Cheng and S Liu in Human & Experimental Toxicology
Footnotes
Author contributions
SL conceptualized the work and study plan. LW conducted animal experiments. JX, FW, GZ, MF helped in the analytical part of the work. SL and LW edited the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
