Abstract
Novel urinary protein biomarkers have recently been identified and qualified in rats for the early detection of renal injury in drug development studies. However, there are few reports on the utility of these renal biomarkers in mice, another important and widely used preclinical animal species for drug development studies. The purpose of this study was to assess the value of these recently qualified biomarkers for the early detection of drug-induced kidney injury (DIKI) in different strains of mice using multiple assay panels. To this end, we evaluated biomarker response to kidney injury induced by several nephrotoxic agents including amphotericin B, compound X, and compound Y. Several of the biomarkers were shown to be sensitive to DIKI in mice. When measured, urinary albumin and neutrophil gelatinase-associated lipocalin were highly sensitive to renal tubular injury, regardless of the assay platforms, mouse strain, and nephrotoxic agents. Depending on the type of renal tubular injury, kidney injury molecule-1 was also highly sensitive, regardless of the assay platforms and mouse strain. Osteopontin and cystatin C were modestly to highly sensitive to renal tubular injury, but the assay type and/or the mouse strain should be considered before using these biomarkers. Calbindin D28 was highly sensitive to injury to the distal nephron in mice. To our knowledge, this is the first report that demonstrates the utility of novel urinary biomarkers evaluated across multiple assay platforms and nephrotoxicants in different mice strains with DIKI. These results will help drug developers make informed decisions when selecting urinary biomarkers for monitoring DIKI in mice for toxicology studies.
Introduction
Given the lack of sensitivity of traditional renal biomarkers—blood urea nitrogen (BUN) and serum creatinine (sCr)—drug-induced kidney injury (DIKI) in preclinical toxicology studies is usually identified through histological examination, which may or may not be accompanied by corresponding changes in traditional renal biomarkers. However, histopathology cannot be used to monitor for DIKI during clinical studies. As such, the Predictive Safety Testing Consortium Nephrotoxicity Working Group has recently identified novel urinary biomarkers in preclinical animal species for the early detection of renal injury.
1
-8
Most of these urinary biomarkers (total protein [TP], albumin [ALB], kidney injury molecule-1 [KIM-1], osteopontin [OPN], neutrophil gelatinase-associated lipocalin [NGAL], N-acetyl-β-
As previously mentioned, these novel biomarkers have already been qualified for use in rats, and they are currently being evaluated in other preclinical animal species including dogs and cynomolgus monkeys. That said, there are few reports on the utility of these biomarkers in mice, but these reports only highlight the utility of only some of these renal biomarkers. 10,11 This information is critical to the use of these biomarkers, given that mice are a widely used preclinical animal species for drug development studies.
The purpose of this study was to assess the value of these biomarkers for the early detection of DIKI in mice. We evaluated the response of these biomarkers in mice to DIKI induced by several nephrotoxic agents including amphotericin B, compound X, and compound Y. A collective data set measured with several recently developed multiplex platforms was utilized for these studies. Amphotericin B is an antifungal drug 12,13 known to cause nephrotoxicity through different mechanisms including changes in cell permeability, the same mechanism that is responsible for its antifungal activity, and through oxidative and ionophoric damage to the renal tubule cell membrane. 14 -17 Amphotericin B also causes direct renal tubular damage through renal vasoconstriction due to its direct effect on smooth muscle cells and consequent reduction in renal blood flow and glomerular filtration rate. 18 Compounds X and Y are investigational molecules with nephrotoxic effects from Sanofi and Merck Sharp & Dohme (MSD), respectively. Along with the traditional identification and monitoring tools of DIKI, we evaluated the urinary protein concentrations of these renal biomarkers in response to DIKI. The results described in this report fill a significant gap in the field and will help inform drug developers of the utility of renal biomarkers in mice for toxicology studies.
Materials and Methods
Amphotericin B Study
Ethical statement
All in vivo animal procedures were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) accredited facility under an Institutional Animal Care and Use Committee approved protocol at Janssen Research and Development, LLC, San Diego, California, USA. Except where noted, standard procedures and conditions for animal care, housing, access to water and food, environment, and room maintenance were used. All other procedures were performed in accordance with laboratory standard operating procedures and/or established laboratory best practices.
Animals, test article administration, biofluid collections, and histopathology
Male C57BL/6 mice, approximately 7 weeks old, were purchased from Charles River Laboratories. Naïve animals (4 per group) were randomly assigned to either a control group or a test article group. There were 3 control and 3 test article groups assigned for terminal sample collection and necropsy on either day (D) 1, 3, or 5 of the study. Amphotericin B USP grade powder (X-Gen Pharmaceuticals, Inc) was formulated in vehicle (5% dextrose solution for injection, USP, Baxter). On study D1 to D4, mice were administered via lateral tail vein, a slow intravenous (IV) infusion (1 mL/min) of either a single dose of amphotericin B (2 mg/kg/d) or vehicle (5% dextrose). Animals designated to the D1 group were fasted for 8 hours postdose, whereas the animals in the D3 and D5 groups were fasted overnight (∼16 hours) prior to sample collections.
Whole-blood (serum) samples were collected on D1 (8 hours after the first dose), D3 (∼24 hours after the second dose), or D5 (∼24 hours after the fourth dose) from the designated control and test article groups. For the D1 animals, urine samples were collected for 8 hours on wet ice after dosing, while urine samples were collected overnight (∼16 hours) on wet ice from the D3 and D5 animals. Urine samples aliquoted for downstream biomarker analysis were centrifuged at 2,900 rpm at 4 °C for 10 minutes and stored at 80 °C until time of analysis.
One control group and 1 test article group were euthanized on D1, D3, or D5 of the study by CO2 inhalation and exsanguination via vena cava blood collection into serum separation tubes. Following euthanasia, the kidneys were fixed in 10% neutral-buffered formalin and embedded in paraffin wax, sectioned at 4 μm, stained with hematoxylin and eosin (H&E), and examined microscopically by the study pathologist. Injury severity grades were assigned using a 0 to 5 grading scale: 0 (no observable pathology), 1 (minimal), 2 (mild), 3 (moderate), 4 (marked), and 5 (severe).
Biomarker measurement
Siemens Advia 1800 automated chemistry system and reagents (Siemens Corp) were used to measure serum concentrations of traditional DIKI biomarkers sCr and BUN, novel biomarkers including serum CysC, and urinary concentrations of creatinine (uCr), NAG, TP, and ALB). Both MilliporeSigma Milliplex Map Mouse Kidney Injury Magnetic Bead Panels 1 and 2 (MKI1MAG-94K and MKI2MAG-94K) and FirePLex Immunoassay Panel from Abcam (ab235661 | FirePLex Mouse Kidney Toxicity) were used to measure urine biomarkers including OPN, NGAL, CLU, KIM-1, and CysC. All assays were performed according to manufacturer’s instructions. All samples were measured in triplicates, and the uCr-normalized average of the readouts was recorded.
Compound X Study
Ethical statement
This study was conducted at Covance Laboratories Ltd, Harrogate, UK, in accordance with the requirements of the Animals (Scientific Procedures) Act 1986, and a local ethical review was maintained.
Animals, test article administration, biofluid collections, and histopathology
CD1 mice, approximately 9 weeks old, were obtained from Charles River Laboratories. Animals (n = 10 mice/sex/group/necropsy time point) were given a solution of sterile saline buffered with sodium acetate (vehicle control group) or compound X in vehicle at 30, 100, 300 mg/kg/d in daily 2-hour IV infusion using the tail cuff method of cannulation for 14 days. For both groups, continuous infusion was performed with a dosing volume of 7.5 mL/kg/h. All animals were infused with saline at a dose rate of 7.5 mL/kg/h during the other 22 hours of the day.
Food was not removed (a few pellets of food was provided per animal) at the end of the dosing on D15, and blood samples were collected at necropsy and 16-hour urine samples were collected overnight on wet ice. Urine samples were frozen and stored at −70 °C for urinary biomarker analyses. Each animal was euthanized using an intraperitoneal overdose of sodium pentobarbitone. Immediately upon confirmation of death, the animals were exsanguinated via vena cava blood collection into serum separation tubes. The kidneys were fixed in 10% neutral-buffered formalin and embedded in paraffin wax, sectioned at 5 μm, stained with H&E, and examined microscopically by the study pathologist. Injury severity grades were assigned using a 0 to 5 grading scale: 0 (no observable pathology), 1 (minimal), 2 (mild), 3 (moderate), 4 (marked), and 5 (severe).
Biomarker measurement
Plasma concentrations of BUN and Cr along with urinary concentrations of TP and Cr were measured using standard clinical chemistry method with a Beckman Chemistry Analyzer. Urinary NAG activity was measured by enzymatic method, and urinary ALB concentration was measured by immuno-turbidimetric assay using a Roche Modular P800 Analyzer. Urinary calbindin D28 (CALB), CLU, and CysC concentrations were determined by using a rat-specific multianalyte profile technology that cross-reacted with mice. This technology is a Luminex bead–based multiplex immunoassay that was developed by Rules-Based Medicine (RBM). Urinary KIM-1 was concomitantly measured by RBM’s newly developed mouse-specific ELISA singleplex assay. All assays were performed per manufacturer’s instructions. All samples were measured in triplicates, and the uCr-normalized average of the readouts was recorded.
Compound Y Study
Ethical statement
All studies were approved by the institutional animal care and use committee of MSD Corp, a subsidiary of Merck & Co, Inc, Kenilworth, New Jersey, USA, and were conducted in an AAALAC-accredited facility in compliance with the NIH Guide for the Care and Use of Laboratory Animals and the Animal Welfare Act.
Animals, test article administration, biofluid collections, and histopathology
CD1 mice, approximately 6 weeks of age, were obtained from Charles River Laboratories. A total of 36 mice were included in the study; 18 male and 18 female mice were divided into 4 dosing groups, with 3 mice per sex in the vehicle control group and 5 mice per sex in each dosed group. Male mice dosing groups were 0, 2, 20, and 100 mg/kg, while female mice dosing groups were 0, 20, 100, and 250 mg/kg. The route of drug administration was by oral gavage for a single dose per animal with 0.4% (wt/vol) aqueous hydroxypropyl methylcellulose as vehicle and the dosing volume was 5 mL/kg. All animals were fasted overnight (∼16 hours) after dosing. Urine was collected postdosing overnight (16 hours) on wet ice, and the collected urine was frozen and stored at −70 °C for urinary biomarker analyses. Twenty-four hours after dosing, fasted animals were euthanized by CO2 inhalation and exsanguination via vena cava blood collection into serum separation tubes. Serum chemistry was only performed for females. Following euthanasia, the kidneys were fixed in 10% neutral-buffered formalin and embedded in paraffin wax, sectioned at 4 μm, stained with H&E, and examined microscopically by the study pathologist. Injury severity grades were assigned using a 0 to 5 grading scale: 0 (no observable pathology), 1 (minimal), 2 (mild), 3 (moderate), 4 (marked), and 5 (severe).
Biomarker measurement
Serum concentrations of BUN and Cr were measured using standard clinical chemistry method with a Beckman Chemistry Analyzer. Urinary concentrations of mouse kidney injury biomarkers KIM-1 and CLU were determined using R&D Systems ELISA kits DY1817 and DY2747, respectively. Performance of a set of urine biomarkers including OPN, NGAL, CLU, KIM-1, CysC, B2M, vascular endothelial growth factor (VEGF), and epidermal growth factor (EGF) was determined using MilliporeSigma Milliplex Map Mouse Kidney Injury Magnetic Bead Panels 1 and 2 (MKI1MAG-94K and MKI2MAG-94K). All assays were performed per manufacturer’s instructions. All samples were measured in triplicates, and the uCr-normalized average of the readouts was recorded.
Data Analysis
The concentrations of urine-based biomarkers were normalized to concurrent uCr, and the average of the normalized concentrations of the biomarkers was determined. We analyzed fold changes across different dose groups relative to the concurrent control for each of the urine biomarkers. In general, 2 comparisons were performed. For the amphotericin B study, the fold change between control and dosed mice was compared for 3 distinct time points: 8 hours after the first dose, 24 hours after the second dose, and 24 hours after the fourth dose. For the compound X and Y studies, the comparison visualized the fold change differences in mice based on dose groupings of control mice versus low-, mid-, and high-dose groups. For all studies, a fold change was calculated as the average normalized urine biomarker value of the dosed group divided by the average normalized urine biomarker value of the control group. Where baseline urine biomarkers were undetectable, a fold change was calculated as the measured lower limit of quantitation value divided by the average uCr value of the control group. All statistical analyses and figures were generated in the statistical computing language R. 19
Results
Histopathology Showed Time-Dependent Kidney Injury by Amphotericin B (2 mg/kg/d) in C57BL/6 Mice
As previously mentioned, serial necropsy was performed in this study, and no renal gross lesions were observed. Microscopically, in the renal cortex, tubular injury incidence increased over time, while in the renal medulla, the severity of tubular injury increased over time.
On D1 of dosing (8 hours after the first dose), minimal-to-mild, multifocal, tubular dilation with intraluminal eosinophilic granular materials or hyaline casts were observed in the renal cortex. In the renal medulla, minimal-to-moderate, multifocal-diffuse, tubular (loop of Henle) epithelial cell necrosis was observed along with minimal-to-mild, multifocal, intraluminal eosinophilic granular material and hyaline casts (Table 1). On D3 (∼24 hours after the second dose), mild, focal, tubular dilation with intraluminal eosinophilic granular materials and hyaline casts were observed in the renal cortex. In the renal medulla, minimal-to-moderate, multifocal, tubular (loop of Henle) epithelial cell necrosis was observed along with minimal-to-mild, multifocal, intraluminal eosinophilic granular material or hyaline casts (Table 1).
Group Incidence and Severity of Microscopic Findings in Kidney of Male C57BL/6 Mice Dosed With Amphotericin B.
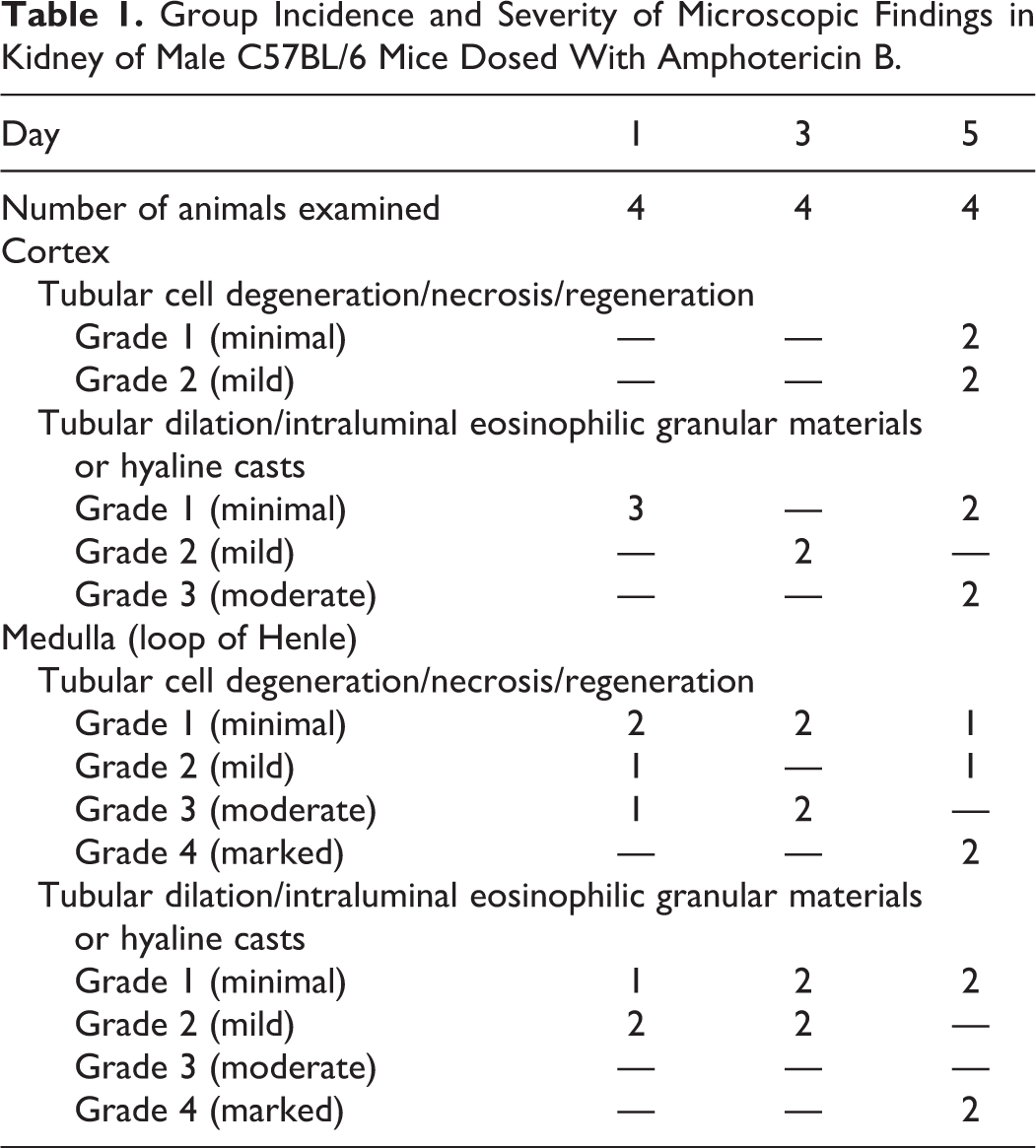
On D5 (∼24 hours after the fourth dose), minimal-to-mild, multifocal, tubular degeneration/necrosis and minimal, multifocal, basophilic tubules were observed along with minimal-to-moderate, multifocal tubular dilatation with intraluminal eosinophilic granular materials or hyaline casts in the renal cortex. In the renal medulla, minimal to marked diffuse, tubular basophilia with prominent mitotic figures and fibroplasia (regenerative responses) were observed along with minimal, interstitial inflammatory cell infiltration (mononuclear and polymorphonuclear cells). Moreover, minimal to marked, multifocal to diffuse, tubular dilatation with intraluminal eosinophilic granular material and hyaline casts were observed along with mild multifocal tubular (loop of Henle) epithelial cell degeneration/necrosis (Table 1).
ALB, KIM-1, NGAL, and OPN Showed Significant Changes in Response to Amphotericin B-Induced Kidney Injury
Urine and whole-blood (serum) samples were collected on D1, D3, or D5 from the designated control and test article groups. Two assay platforms were used for analyzing the novel urinary biomarkers for the evaluation of amphotericin B-induced renal injury in C57BL/6 mice (Table 2). The MilliporeSigma and FirePLex assay platforms were used for measuring NGAL, CLU, KIM-1, and OPN. Cystatin C was analyzed by the FirePLex immunoassay panel only. The traditional or standard clinical chemistry method (see description in Materials and Methods) was used for measuring urinary ALB, NAG, TP, sCr, sCysC, and BUN.
Biomarker Performance in the Amphotericin B Study.a
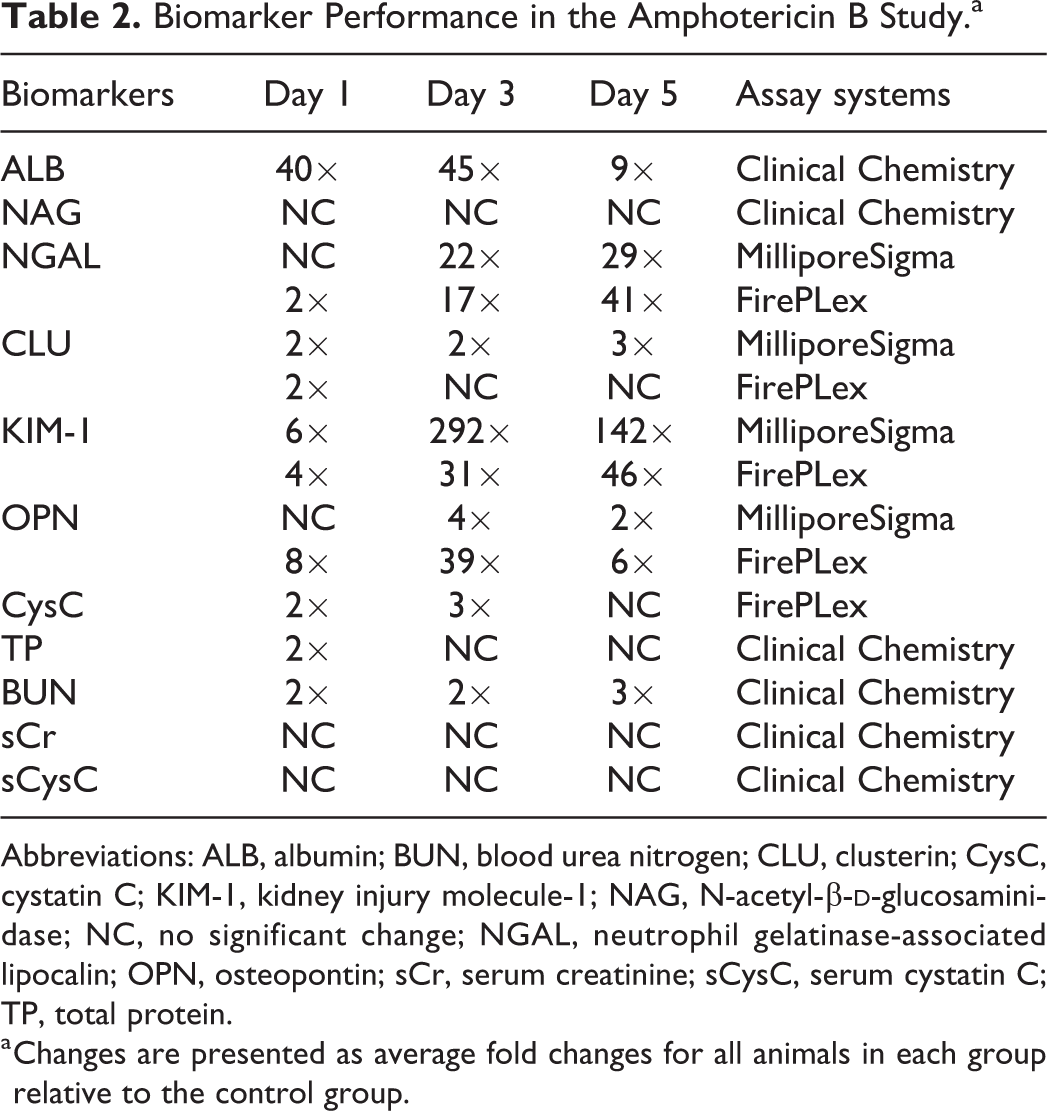
Abbreviations: ALB, albumin; BUN, blood urea nitrogen; CLU, clusterin; CysC, cystatin C; KIM-1, kidney injury molecule-1; NAG, N-acetyl-β-
a Changes are presented as average fold changes for all animals in each group relative to the control group.
The results showed that ALB, NGAL, KIM-1, and OPN were markedly elevated in response to renal tubular injury (Figures 1 and 2, Table 2 and Supplemental Table 1). Both ALB and KIM-1 showed significant increases to early renal tubular injury on D1. Regardless of the assay platforms, NGAL, ALB, and KIM-1 showed significant increases on D3 and D5. The FirePLex immunoassay panel appeared to be slightly more sensitive for NGAL detection of early renal tubular injury on D1 relative to the MilliporeSigma assay panel. Osteopontin showed significant increases in response to renal tubular injury on D1, D3, and D5 in the FirePLex immunoassay panel, while slight increase in OPN was observed only on D3 in the MilliporeSigma assay platform (Figures 2C and D, Table 2). Clusterin showed slight increase only on D1 in the FirePLex immunoassay panel, while the MilliporeSigma assay platform detected similar fold changes in CLU on D1, D3, and D5. Both CysC (D1 and D3) and TP (D1 only) showed slight increases on different days, while NAG, sCr, and sCysC did not show any change(s) in response to renal tubular injury at any time point (Table 2). Blood urea nitrogen showed a 2-fold change, and this was considered a modest change of biological significance in mice (Table 2). Therefore, BUN was considered a better sensitive traditional serum biomarker for detecting renal tubular injury, relative to CLU, CysC, TP, sCr, and sCysC in this study.
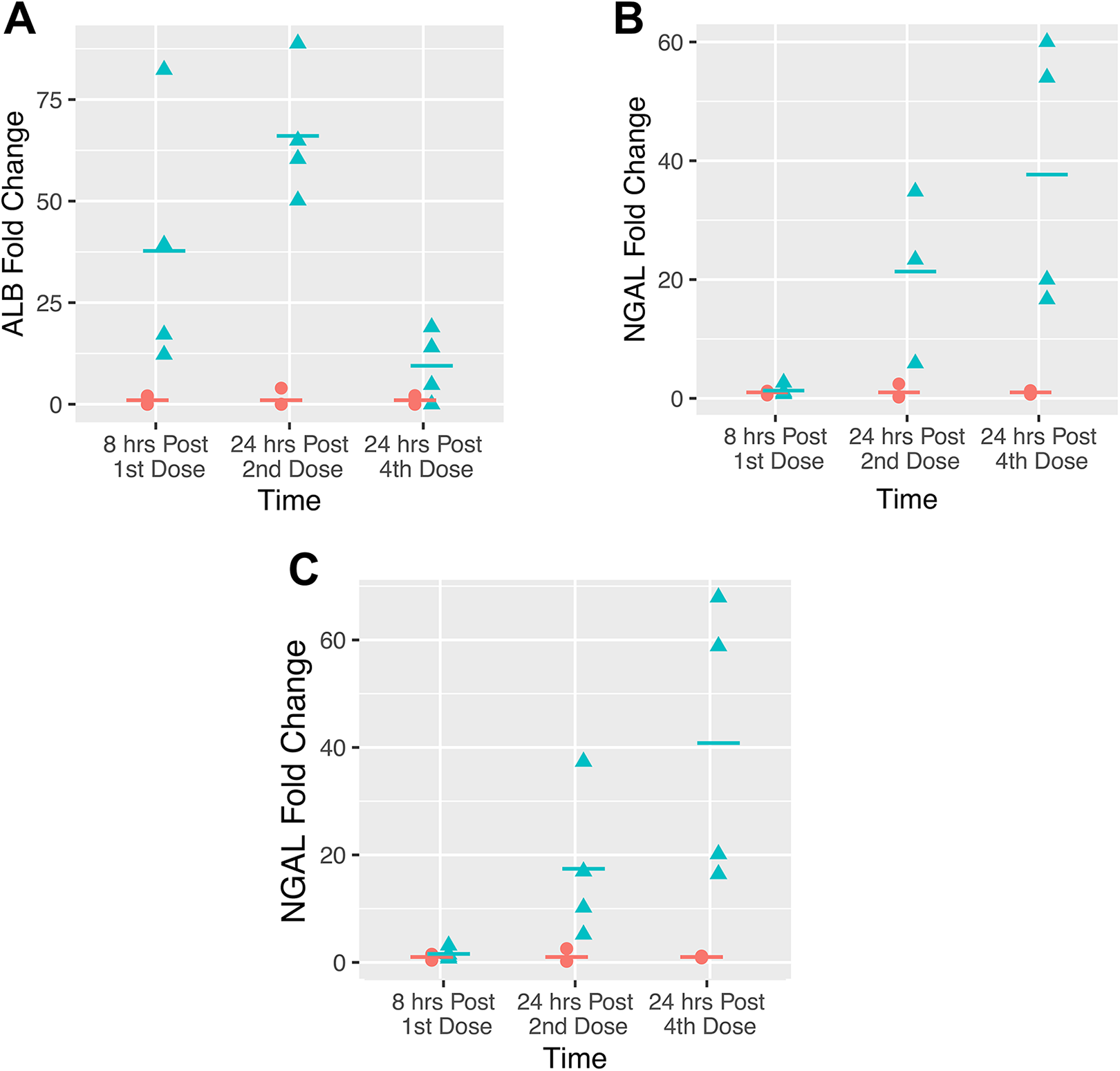
Fold changes of urinary (A) ALB, (B) NGAL-MilliporeSigma, and (C) NGAL-FirePLex between the control (peach circle) and the dosed C57BL/6 mice (cyan triangle) over time in the amphotericin B study. ALB indicates albumin; NGAL, neutrophil gelatinase-associated lipocalin.
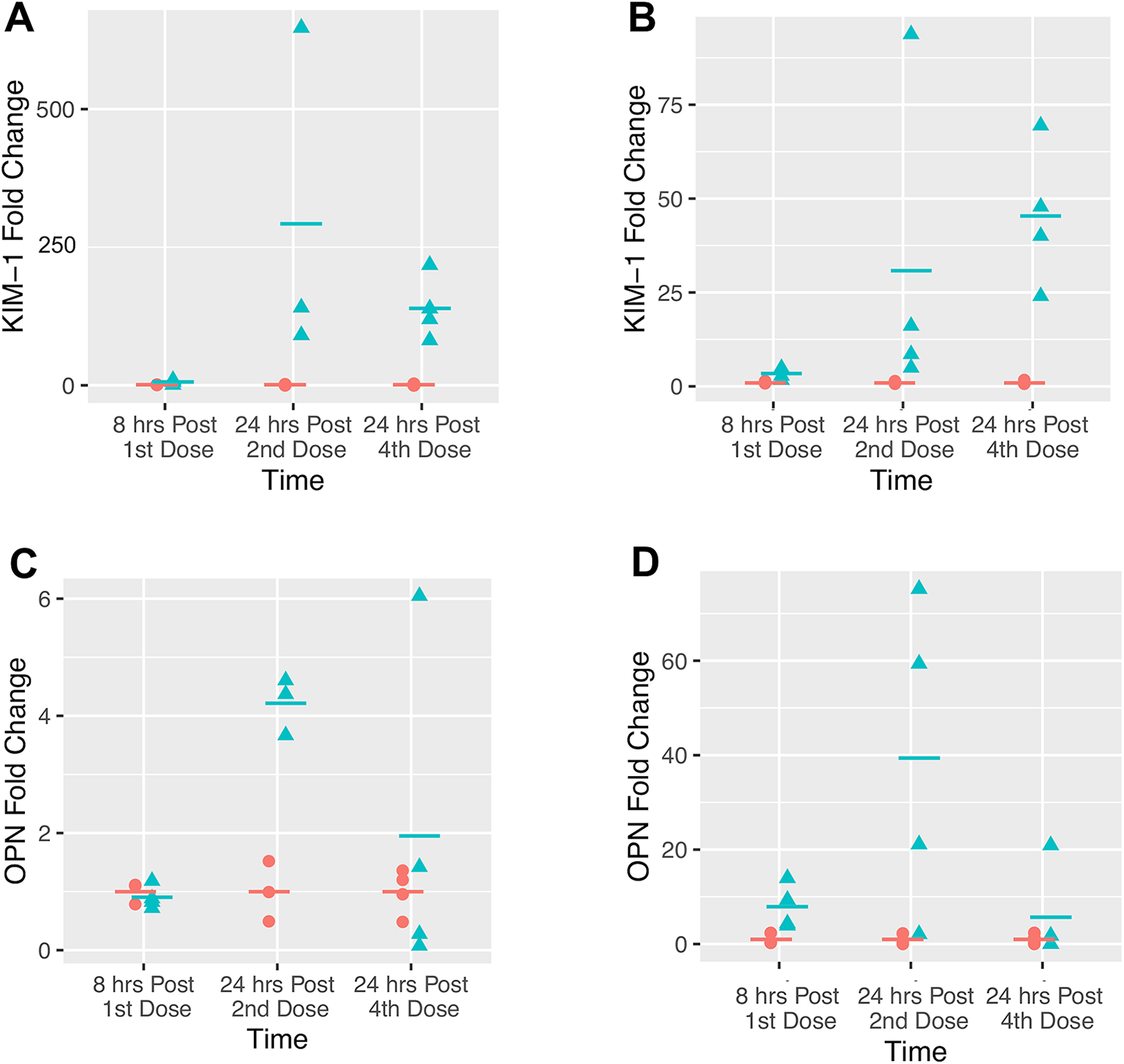
Fold changes of urinary (A) KIM-1-MilliporeSigma, (B) KIM-1-FirePLex, (C) OPN-MilliporeSigma, and (D) OPN-FirePLex between the control (peach circle) and the dosed C57BL/6 mice (cyan triangle) over time in the amphotericin B study. KIM-1 indicates kidney injury molecule-1; OPN, osteopontin.
Overall, ALB, NGAL, and KIM-1 were considered the most sensitive markers of renal tubular injury in this study, while OPN and BUN were considered to be of moderate significance.
Histopathology Showed DOSE-Dependent Kidney Injury by Compound X (30, 100, 300 mg/kg/d) in CD1 Mice
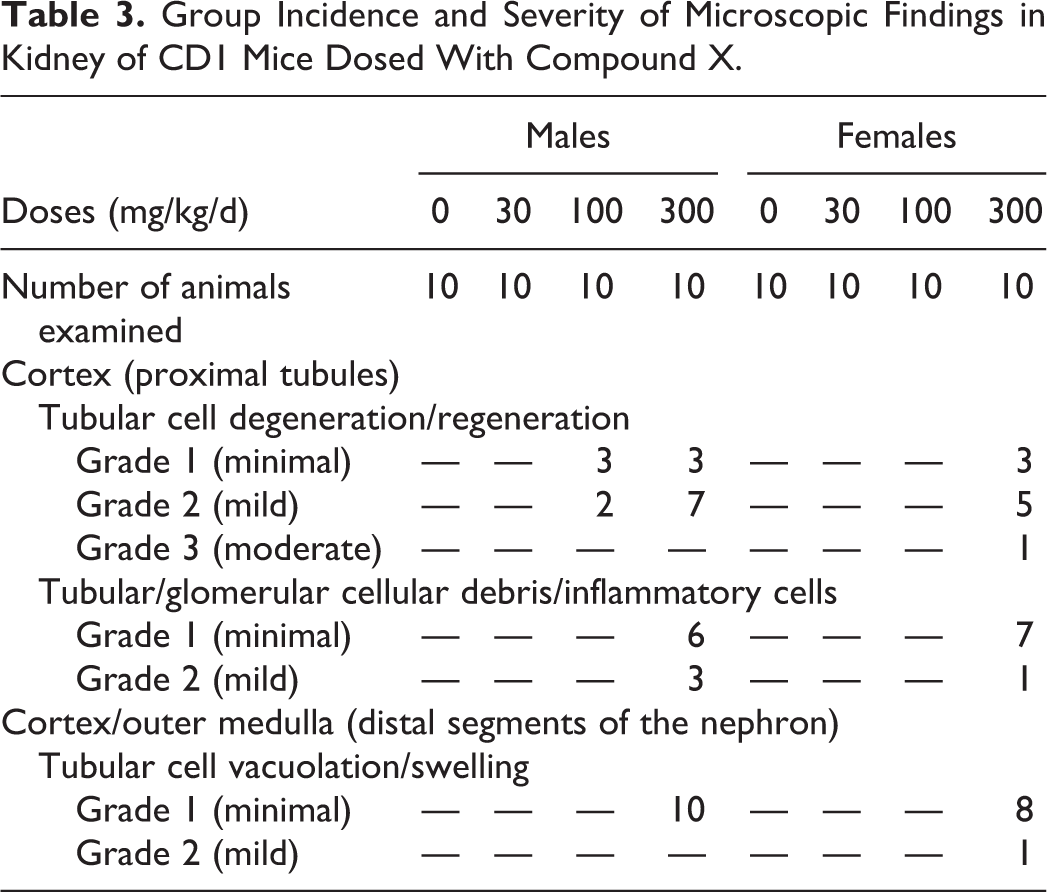
As previously mentioned in Materials and Methods, mice were given either the vehicle, 30, 100, or 300 mg/kg/d of compound X for 14 days. Necropsy was performed on D15 of the study, and no renal gross lesions were observed. In the 30 mg/kg/d dose group, no lesions were observed. In the 100 mg/kg/d dose group, only males were affected. In the renal cortex, microscopic findings observed in this group consisted of minimal-to-mild tubular cell degeneration/regeneration in the proximal tubules. In the 300 mg/kg/d dose group, almost all animals from both sexes were affected. In the renal cortex, minimal-to-moderate tubular cell degeneration/regeneration was observed in the proximal tubules, along with minimal-to-mild tubular/glomerular cellular debris/inflammatory cells. Moreover, in the cortex and outer stripe of the outer medulla, minimal-to-mild tubular cell vacuolation/swelling affecting the distal segment of the nephron was observed (Table 3).
Group Incidence and Severity of Microscopic Findings in Kidney of CD1 Mice Dosed With Compound X.
ALB, KIM-1, CysC, and CALB Showed Significant Changes in Response to Compound X-Induced Kidney Injury
Blood samples were collected at necropsy and 16-hour urine samples were collected overnight on wet ice prior to necropsy on D15. One assay platform was used for analyzing the novel urinary biomarkers for the evaluation of compound X-induced renal injury in CD1 mice (Table 4). The RBM-Rat panel was used for measuring CLU, CysC, and CALB, while KIM-1 was measured by RBM with a newly developed mouse-specific ELISA singleplex assay. The traditional or standard clinical chemistry method (see description in Materials and Methods for compound X) was used for measuring urinary ALB, NAG, TP, plasma creatinine (pCr), and BUN.
Biomarker Performance in the Compound X Study.a
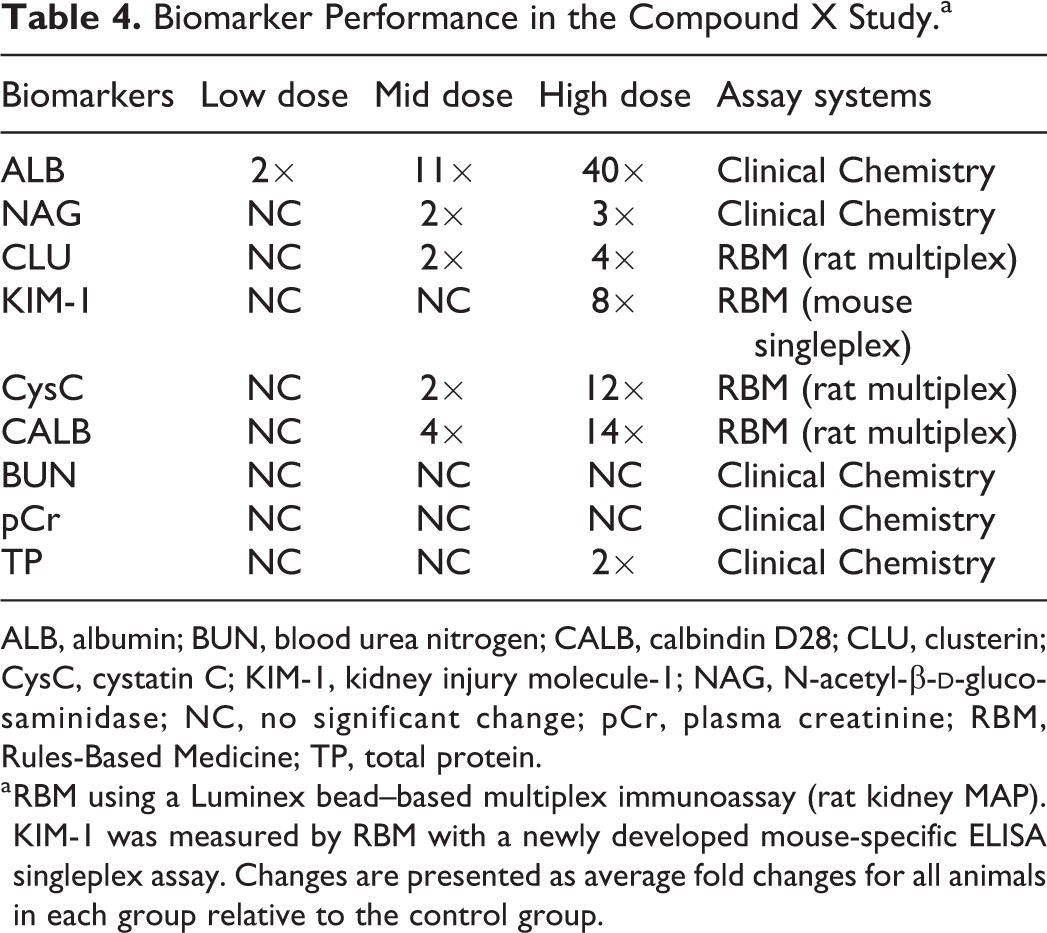
ALB, albumin; BUN, blood urea nitrogen; CALB, calbindin D28; CLU, clusterin; CysC, cystatin C; KIM-1, kidney injury molecule-1; NAG, N-acetyl-β-
a RBM using a Luminex bead–based multiplex immunoassay (rat kidney MAP). KIM-1 was measured by RBM with a newly developed mouse-specific ELISA singleplex assay. Changes are presented as average fold changes for all animals in each group relative to the control group.
The most significant changes were increases in ALB, CysC, KIM-1, and CALB in the 300 mg/kg/d dose group, regardless of the sex (Figures 3 and 4, Table 4 and Supplemental Table 2). Both ALB and CALB were highly sensitive to renal injury even at the early stages of the tubular injury in the ≤100 mg/kg/d dose groups (Figure 3A and C, Table 4). And, CLU, NAG, and TP were considered to have slight increases in response to renal injury, while BUN and pCr did not exhibit any significant changes as shown in Table 4.
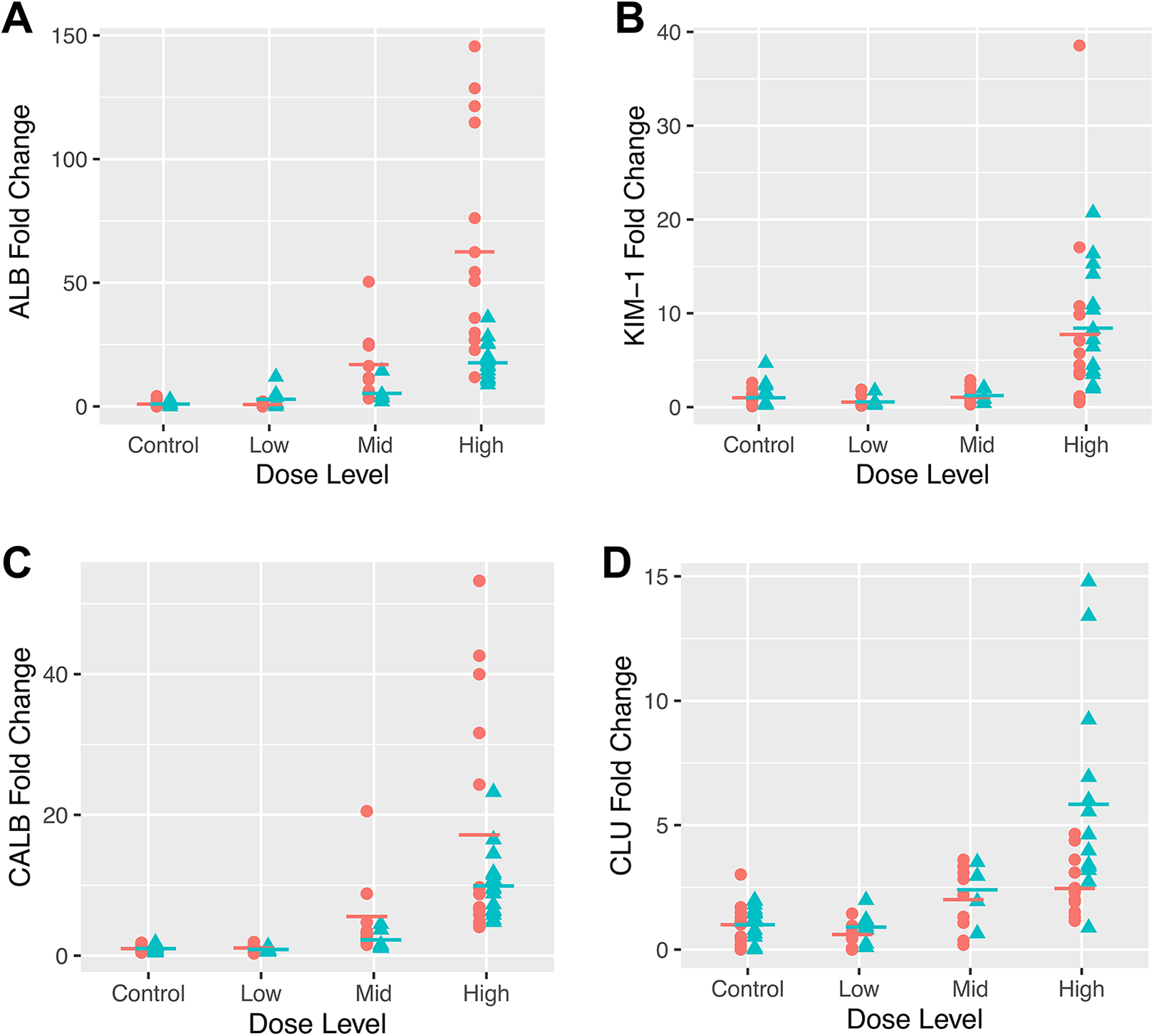
Fold changes of urinary (A) ALB, (B) KIM-1, (C) CALB, and (D) CLU between the male (cyan triangle) and female (peach circle) CD1 mice from different dose groups in the compound X study. ALB indicates albumin; CALB, calbindin D28; CLU, clusterin; KIM-1, kidney injury molecule-1.
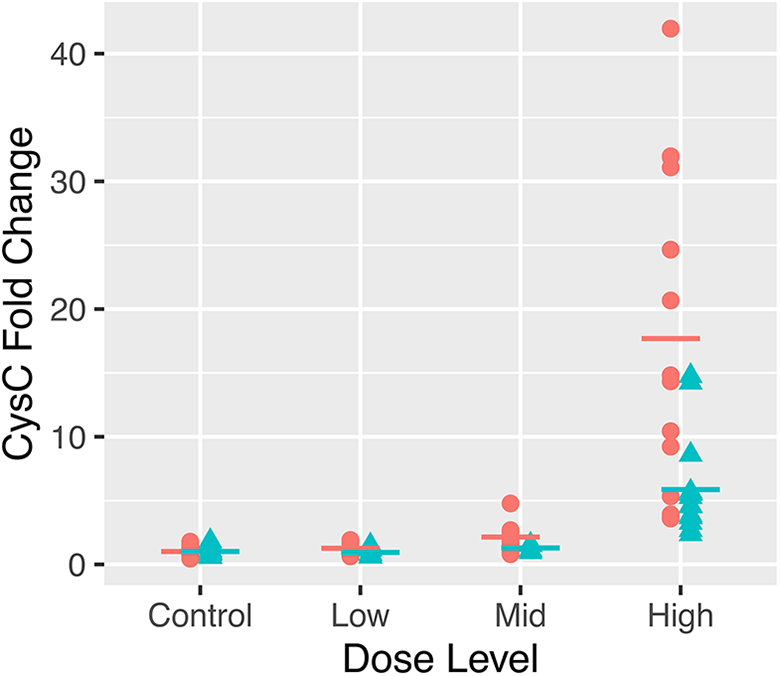
Fold changes of urinary cystatin C between the male (cyan triangle) and female (peach circle) CD1 mice from different dose groups in the compound X study.
Overall, ALB, KIM-1, CysC, and CALB were the most sensitive markers to renal injury in this study. Other than KIM-1, it should be noted that the RBM assay platform was a rat-specific assay panel that identified mouse urinary proteins with no observable sensitivity problems.
Histopathology Showed Dose-Dependent Kidney Injury by Compound Y in CD1 Mice (2, 20, 100 mg/kg/d, Males; 20, 100, 250 mg/kg/d, Females)
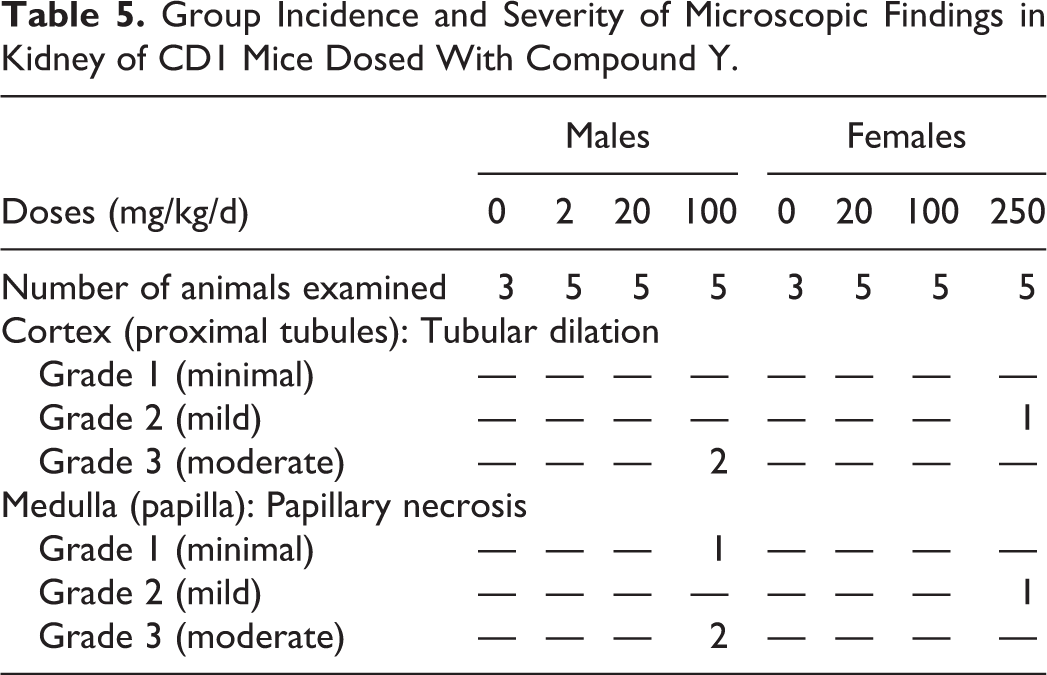
For this study, animals were only dosed once, and necropsy was performed 24 hours after dosing, and no renal gross lesions were observed. No lesions were observed in the low- and mid-dose groups in either male and female mice. Histopathologic changes were observed in 5 of 10 animals in the high-dose groups (3 males, 2 females), with overall higher severity in the males. These changes included minimal-to-moderate papillary necrosis in the medulla and mild-to-moderate dilation of the proximal tubules in the cortex (Table 5).
Group Incidence and Severity of Microscopic Findings in Kidney of CD1 Mice Dosed With Compound Y.
CLU, NGAL, BUN, and sCr Showed Significant Changes in Response to Compound Y-Induced Kidney Injury
Blood samples were collected at necropsy and 16-hour urine samples were collected overnight on wet ice prior to necropsy 24 hours postdose. Two assay platforms were used to analyze the novel urinary biomarkers for the evaluation of compound Y-induced renal injury in CD1 mice (Table 6). The R&D assay system was used to measure CLU and KIM-1, and the MilliporeSigma assay platform was used to measure OPN, NGAL, CLU, KIM-1, CysC, B2M, VEGF, and EGF. The traditional or standard clinical chemistry method was used for measuring BUN and sCr.
Biomarker Performance in the Compound Y Study.c
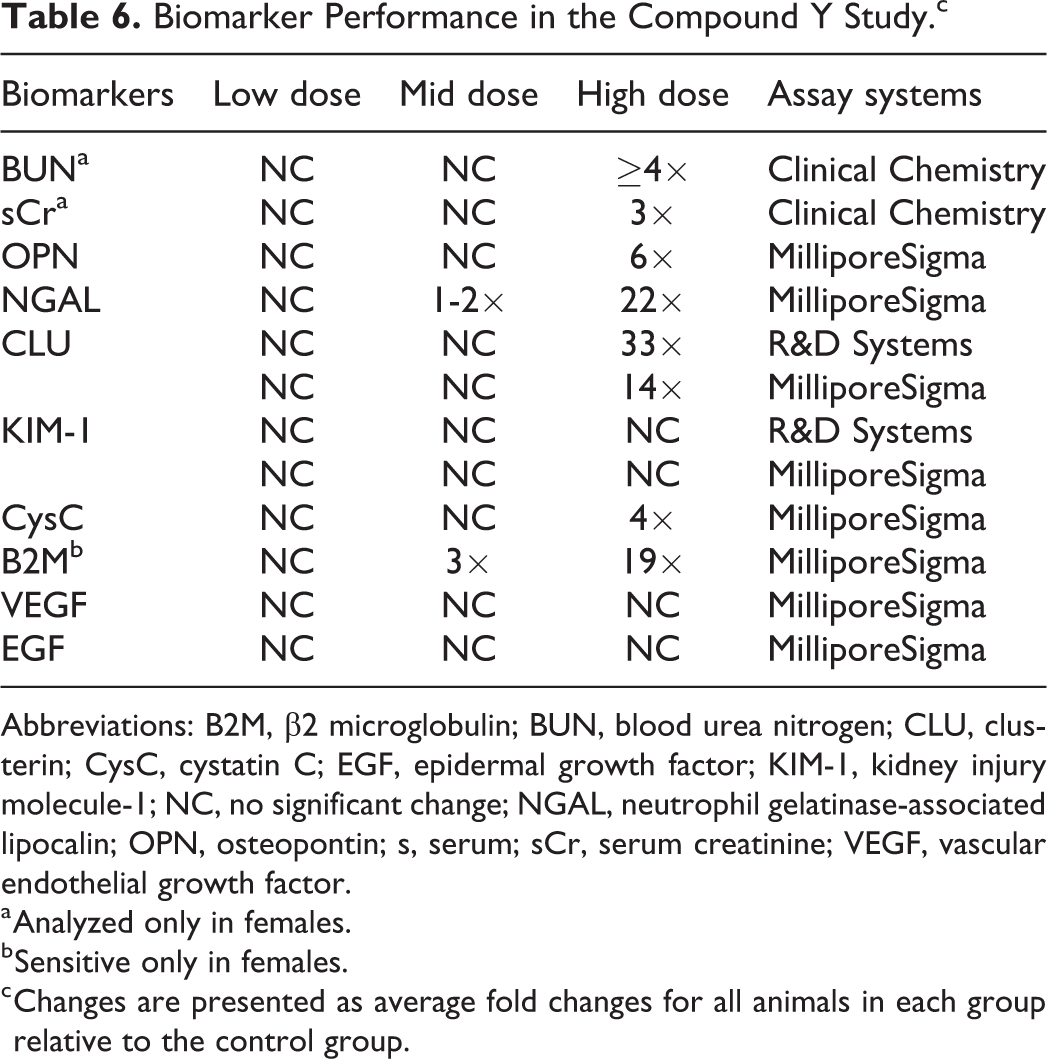
Abbreviations: B2M, β2 microglobulin; BUN, blood urea nitrogen; CLU, clusterin; CysC, cystatin C; EGF, epidermal growth factor; KIM-1, kidney injury molecule-1; NC, no significant change; NGAL, neutrophil gelatinase-associated lipocalin; OPN, osteopontin; s, serum; sCr, serum creatinine; VEGF, vascular endothelial growth factor.
a Analyzed only in females.
b Sensitive only in females.
c Changes are presented as average fold changes for all animals in each group relative to the control group.
Significant changes were observed with increases in CLU, NGAL, BUN, and sCr in the high-dose group (Figure 5, Table 6 and Supplemental Table 3). Both BUN and sCr were only measured in the female mice. Both BUN and sCr were markedly elevated in the high-dose group (250 mg/kg/d) in response to compound Y-induced kidney injury (Table 6 and Supplemental Table 3). Both CLU and KIM-1 were measured in both R&D and MilliporeSigma assay platforms, and the results were consistent between the 2 platforms. The levels of CLU were markedly elevated in the high-dose group with no change in the low- and mid-dose groups (Figure 5C and D), while KIM-1 did not show any change. Both OPN and CysC had slight to moderate increases in response to renal injury, while B2M showed significant increases only in females with no change in males despite the presence of higher severity of DIKI in males (Figure 5B, Tables 5 and 6 and Supplemental Table 3). In addition, KIM-1, EGF, and VEGF were not sensitive to compound Y-induced kidney injury in either sex in this study (Table 6).
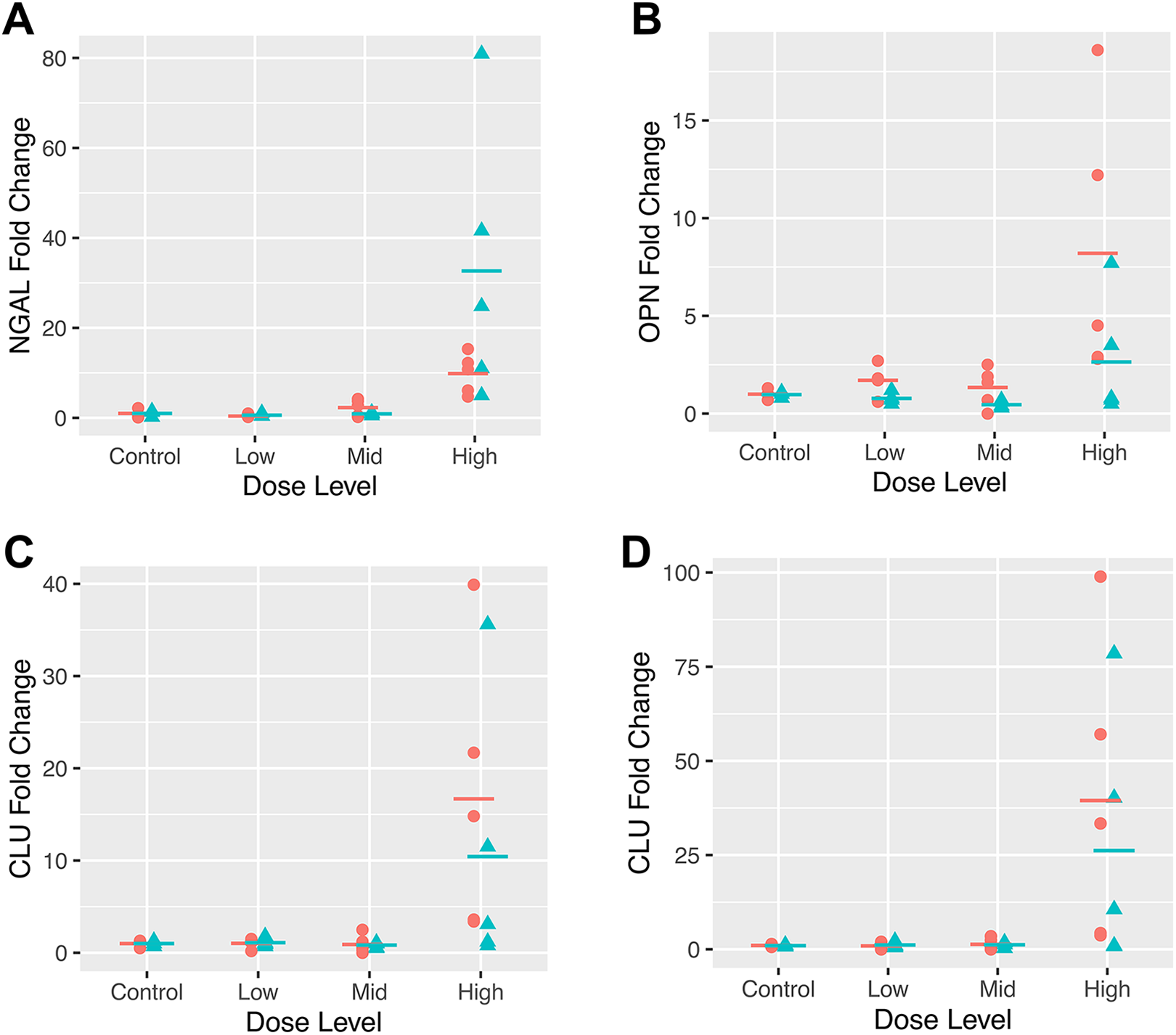
Fold changes of urinary (A) NGAL, (B) OPN, (C) CLU-MilliporeSigma, and (D) CLU-R&D system between the male (cyan triangle) and female (peach circle) CD1 mice from different dose groups in the compound Y study. CLU indicates clusterin; NGAL, neutrophil gelatinase-associated lipocalin; OPN, osteopontin.
Overall, CLU, NGAL, BUN, and sCr were considered to be the most sensitive markers to renal injury in this study. Also, CysC and OPN were considered to be slightly to moderately sensitive, while KIM-1, VEGF, and EGF did not show any significant change.
Discussion
One of the most important observation in these studies was the marked increases of urinary ALB in response to acute tubular injury, regardless of the assay type and mouse strain, as the magnitude of ALB increase was comparable in the amphotericin B and compound X studies. Although NGAL was only measured in 2 of the 3 studies, it also showed comparable significant increases in the 2 assay platforms used in the amphotericin B study and the MilliporeSigma assay system in the compound Y study. This suggests that NGAL is a very sensitive marker for renal injury in mice, regardless of the mouse strain and assay platform. Despite the fact that the histopathologic changes on D1 and D3 were similar in severity, the magnitude of NGAL increases between D3 and D5 was considered highly remarkable in the amphotericin B study, further attesting to the sensitivity of this marker in early-stage renal tubular injury. These results were comparable to the sensitivities of ALB and NGAL in rats with DIKI in previous reports. 1,6,7,20,21
The KIM-1 showed no response to the renal injury observed in the compound Y study. This might be attributed to the fact that the primary tubular injury in that study was mostly limited to the papillary region of the kidney comprising the distal portion of the nephron, whereas KIM-1 is mainly expressed at the proximal tubule. 22 -27 The KIM-1 is still considered a sensitive marker to proximal renal tubular injury in mice, given the magnitude of change in the amphotericin B study assay platforms despite the minimal-to-mild nature of the histopathological renal tubular injury on D1 and D3. In the MilliporeSigma assay panel, the higher fold changes of KIM-1 on D3 relative to D5 were attributed to intra-assay variability, as one sample in the concurrent control group on D5 had a much higher baseline KIM-1 concentration relative to other samples in the control group. This higher concurrent control value reduced the average fold change for the D5 dosed animals relative to D3 (Supplemental Table 1). Nevertheless, these results were comparable to previous reports in rats with DIKI, where KIM-1 was also considered to be a highly sensitive biomarker for DIKI. 1,6,7,20,21,28,29
Regardless of the mouse strain, OPN was considered to have comparable increases in both the amphotericin B and the compound Y studies. That said, OPN showed a much better response in the FirePLex immunoassay panel relative to the MilliporeSigma platforms in the amphotericin B and the compound Y studies. These differences were considered to be due to inter-assay variability.
Another interesting observation in this report was the disparity in CysC and CLU responses across studies. Cystatin C showed remarkable increases in the compound X study, a slight increase in the compound Y study, and insignificant changes in the amphotericin B study. Despite the fact that there was only proximal tubular dilation with no necrosis or degeneration in the compound Y study, CysC appeared to be more sensitive to renal injury in the compound Y study versus tubular degeneration and necrosis in the amphotericin B study. In the same vein, CLU showed consistent significant increases in both platforms used in the compound Y study and a relatively lower increase in the amphotericin B and compound X studies. Although the causes of the differences were uncertain, we could not rule out the effect of mouse strain and assay platform differences, along with variations in histopathology findings. That said, the magnitude of CLU changes observed in the compound Y study was more comparable to what was observed in rats and dogs with DIKI, 30,31 and the lackluster performance observed for CysC in the amphotericin B and compound X studies was more comparable to that reported for rats by Vlasakova et al. 7
Calbindins belong to a family of high-affinity calcium-binding proteins that contain the EF-hand structural motif required for calcium binding. Many types of calbindins have been identified including calbindin-D9k, which is mainly expressed in the intestine and kidney, and calbindin-D28k (CALB), which is mainly expressed in the neurons and the distal segment of the nephron in the kidney, and the subject of investigation in this study. 32 -37 In this report, although CALB was only measured in one assay, its response was considered remarkable in the compound X study, as this was only in reaction to the distal tubular cell vacuolation/swelling without necrosis or degeneration. That said, the results obtained from this study were in contrast to what was reported in another study where the authors observed decreased CALB messenger RNA and protein expression in humans and rats with distal nephron injury. 38 We attribute this disparity to the difference in the morphological changes in the studies, along with the mechanisms for these changes. In the previous study, the histopathology revealed renal distal tubular dilation that is often due to injury that causes functional impairment or could be secondary to lower urinary tract obstruction versus this current study with renal tubular swelling and vacuolation that is due to direct injury to the renal epithelium that may eventually progress to degeneration and necrosis over time. Therefore, CALB is considered a sensitive marker for detecting injury to the distal segment of the nephron, but the data interpretation should be made within the context of the histopathologic changes observed.
The primary reason for the need for novel renal biomarkers was due to the inconsistency and poor sensitivity of the traditional renal biomarkers to early-stage renal injury. As shown in this report, the inconsistencies and variable sensitivities associated with these traditional biomarkers were observed across studies; BUN and sCr showed remarkable increases in the compound Y study, and no changes were observed in the amphotericin B or compound X studies.
In conclusion, based on the results from all these studies, when measured, urinary ALB and NGAL were considered the best markers for renal tubular injury in mice, regardless of the assay platforms, mouse strain, and nephrotoxic agents, as they showed better response to DIKI than the traditional biomarkers. Depending on the type of renal tubular injury, KIM-1 was also highly sensitive, regardless of the assay platforms and mouse strain. Both OPN and CysC also showed modest to significant increases, in response to proximal tubular injury, but the assay type and/or the mouse strain should be considered before using these biomarkers; and CALB was considered to be highly sensitive to injury to the distal portion of the nephron in mice.
Supplemental Material
Supplemental_Tables_1-3_revised - The Utility of Novel Urinary Biomarkers in Mice for Drug Development Studies
Supplemental_Tables_1-3_revised for The Utility of Novel Urinary Biomarkers in Mice for Drug Development Studies by Adeyemi O. Adedeji, Yi-Zhong Gu, Tony Pourmohamad, Justin Kanerva, Yafei Chen, Elnaz Atabakhsh, Michael R. Tackett, Feifei Chen, Bhavana Bhatt, Thierry Gury, Olivier Dorchies, Manisha Sonee, Michelle Morgan, Jennifer Burkey, Jean-Charles Gautier and James E. McDuffie in International Journal of Toxicology
Footnotes
Acknowledgments
This material is based upon work supported by Critical Path Institute’s (CPath) Predictive Safety Testing Consortium (PSTC). The authors thank the members of PSTC for their scientific, financial, and in-kind contributions that supported these research activities, as well as the input from FDA and EMA scientists who serve as advisors. The authors also thank Amy C. Porter for her critical review of the manuscript.
Author Contributions
Adedeji, A. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Gu, Y.-Z. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Pourmohamad, T. contributed to analysis and drafted manuscript; Kanerva, J. contributed to acquisition and critically revised manuscript; Chen, Y. contributed to analysis and critically revised manuscript; Atabakhsh, E. contributed to analysis and critically revised manuscript; Tackett, M. contributed to analysis and critically revised manuscript; Feifei, C. contributed to analysis and critically revised manuscript; Bhatt, B. contributed to analysis and critically revised manuscript; Gury, T. contributed to analysis and critically revised manuscript; Dorchies, O. contributed to analysis and interpretation and critically revised manuscript; Sonee, M. contributed to conception and design, contributed to acquisition, and critically revised manuscript; Morgan, M. contributed to conception and design, contributed to acquisition and critically revised manuscript; Burkey, J. contributed to conception and design, contributed to acquisition, and critically revised manuscript; Gautier, J. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; McDuffie, J. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All work was supported by Janssen Research & Development, LLC, Merck & Co., Inc, Sanofi R&D, and Genentech, a member of the Roche group. Critical Path Institute is supported, in part, by grant U18 FD005320 from the US Food and Drug Administration.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
