Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of Hydrolyzed Silk and 9 other silk protein ingredients, which function primarily as skin and hair conditioning agents and bulking agents in cosmetic products. The Panel reviewed relevant data relating to the safety of these ingredients and concluded that 8 ingredients are safe in the present practices of use and concentration in cosmetics, as described in this safety assessment, but that the available data are insufficient for determining the safety of 2 silk protein ingredients in cosmetic products, MEA-Hydrolyzed Silk and Silkworm Cocoon Extract.
Keywords
Introduction
The safety of the following 10 Silk protein ingredients as used in cosmetics is reviewed in this safety assessment: Fibroin Hydrolyzed Fibroin Hydrolyzed Sericin Hydrolyzed Silk MEA-Hydrolyzed Silk Sericin Silk Silk Extract Silk Powder Silkworm Cocoon Extract
According to the International Cosmetic Ingredient Dictionary and Handbook (Dictionary), these ingredients are reported to function as skin and hair conditioning agents and bulking agents in cosmetic products. 1
Silk is a fibrous protein that is a product of the silkworm, Bombyx mori, and spiders (Nephila clavipes and Araneus diadematus). Silk proteins are usually produced within specialized glands. These proteins are biosynthesized in epithelial cells and secreted into the lumen of these glands, where the proteins are stored prior to being spun into silk fibers. 2 The silkworm (B mori) is the source of silk from which cosmetic ingredients are derived. 1
MEA-Hydrolyzed Silk is evaluated in this safety assessment, and it should be noted that the Expert Panel for Cosmetic Ingredient Safety (Panel) has previously issued a final amended report on other MEA-ingredients with the conclusion that the following ingredients are safe in the present practices of use and concentration described in the report (rinse-off products only) when formulated to be nonirritating 3 : ethanolamine MEA, ethanolamine HCl, MEA-benzoate, MEA-cocoate, MEA-laureth-6 carboxylate, MEA-laureth sulfate, MEA-lauryl sulfate, MEA-PPG-6 laureth-7 carboxylate, MEA-PPG laureth-6 carboxylate, MEA-PPG-8 steareth-7 carboxylate, MEA-salicylate, MEA-sulfite, MEA-tallowate, and MEA-undecylenate. The Panel also cautioned that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed.
Data on skin depigmentation effects are included in this safety assessment; however, skin depigmentation is considered a drug, and not a cosmetic, effect in the United States, and is not within the Panel’s purview.
Chemistry
Definition and Structure
The Silkworm, B mori, produces silk proteins during the final stage of larval development. Two silk proteins, Fibroin and Sericin, have been distinguished as major components of silk cocoons. 4 All of the ingredients in this report are related because they are derived from the silk fibers produced by B mori. The definitions and functions of Fibroin, Sericin, and other silk protein ingredients reviewed in this safety assessment are presented in Table 1. 1
Definitions and Reported Functions of the Ingredients in This Safety Assessment. 1

Fibroin
Bombyx mori silk Fibroin was determined via nuclear magnetic resonance to exist as a repeated type II β-turn structure, wherein the conformation of one chain enables the formation of intramolecular hydrogen bonds with 2 juxtaposed (adjacent) chains. 5
Sericin
Circular dichroism and infrared absorption spectra show that the molecular configuration of Sericin is mainly random crimp. 6,7 X-ray diffraction analysis and differential thermal analysis indicate that the assembled structure or Sericin powder is an amorphous structure 7 ; however, the amorphous Sericin transforms into a β-structure in the presence of water. 8
Chemical and Physical Properties
Properties of Fibroin, Sericin, and other silk protein ingredients are summarized in Table 2. Polarization microscopy shows that, in Silk, Sericin forms 3 layers surrounding a Fibroin fiber. 5
Chemical Properties of Silk Proteins.

Method of Manufacture
Silk
Fibroin (main protein of Silk) and Sericin (another Silk protein) are secreted by insect silk glands. Fibroin, in aqueous solution, is converted into silk fibers by a process that is called spinning. 10,11 According to another source, in the process of manufacturing lustrous Silk from the dried cocoons of silkworms, Fibroin is separated from Sericin, the other major component of the cocoon, by a degumming process, and the Sericin is mostly discarded in the wastewater. 9
Several methods have been reported for removing Sericin in the degumming process of cocoons. Practically all industrial removal processes involve extraction with soaps and detergents. Heat and acid extraction are other methods. Sericin extracted by different methods can yield different amino acid compositions. 9 Additional information indicates that some commercial Silk is prepared from natural Silk by removing Sericin and that the purified aqueous Fibroin is dried and pulverized into a powder. 12
Hydrolyzed Silk
Hydrolyzed Silk has been reported to be prepared from the cocoon of the silkworm moth (B mori). 13 The silk thread is isolated from the cocoon and the fibers are cleaned and degummed. The individual silk fiber is then wound with other silk fibers to create one long thread. The threads are then combed to remove noils, which are short fibers considered to be by-products of the textile industry. The noils are used in the production of Hydrolyzed Silk proteins through carefully controlled hydrolysis. The resultant material is a 5% solution of a water-soluble silk protein.
It has been reported that Hydrolyzed Silk (average molecular weight[MW] = 300 Da) may be prepared by acid, alkaline, or enzyme-catalyzed hydrolysis; Hydrolyzed Silk protein (average MW = 650 Da) may be prepared by alkaline or enzymatic hydrolysis. 14,15 These processes occur for several hours until the desired MW is reached. The final product is a 20% water solution of Hydrolyzed Silk protein (MW = 300 Da) or a 6.5% water solution of Hydrolyzed Silk protein (MW = 650 Da). Furthermore, another supplier has reported that Hydrolyzed Silk is prepared by acid and enzyme hydrolysis until the MW reaches the target range. 16
According to other sources, Hydrolyzed Silk is produced according to the following procedures 17 –19 —Procedure 1: (1) hydrolysis, (2) inactivation of hydrolytic agent, (3) filtration, (4) treatment, (5) concentration, and (6) sterilization. Procedure 2: (1) proteins hydrolyzed in water at specific pH and temperature for specific duration, (2) filtration to isolate desired components, (3) addition of quaternium-15, EDTA, and methylparaben, or just EDTA and methylparaben, and (4) make batch adjustments if needed (refiltration).
Composition/Impurities
Silk
Silk contains nitrogen (13%-20%) and, for material from one supplier, the reported or specified maximum concentration of heavy metals is 20 ppm. 12
Hydrolyzed Silk
Data on the composition of Hydrolyzed Silk are presented in Table 3. 20,21 Hydrolyzed Silk is marketed as an amino peptide concentrate that is rich in the 2 proteins that comprise natural Silk, Sericin and Fibroin. 22 It consists of ∼19% Hydrolyzed Silk and also contains the preservatives phenoxyethanol (0.4%) and potassium sorbate (0.2%). 23 Other preservatives in Hydrolyzed Silk include quaternium-15, EDTA, and methylparaben. 18,19
Abbreviation: NR, not reported.
Another source indicates that Hydrolyzed Silk (MW = 300 Da) is marketed as a 20% water solution and that Hydrolyzed Silk protein (MW = 650 Da) is marketed as a 6.5% water solution. 16 Hydrolyzed Silk protein (MW = 300 Da) from one source was reported to contain heavy metals and arsenic at levels of ≤4 ppm and 0.4 ppm, respectively. 14 Hydrolyzed Silk protein from another source (MW = 650 Da) was reported to contain heavy metals and arsenic at ≤10 ppm and 1 ppm, respectively. 15
According to another supplier, their Hydrolyzed Silk ingredients are marketed as aqueous solutions, 2 of which are 20% to 30% Hydrolyzed Silk and 27% to 32% Hydrolyzed Silk. 24
Fibroin
Silk derived from the silkworm B mori contains 2 major proteins, Fibroin and Sericin. Fibroin is a fibrous protein, present as a delicate twin thread in which the 2 strands are linked by disulfide bonds and enveloped by successive sticky layers of Sericin. 9 Individual filaments are large molecules (3700 amino acids). 25 Fibroin has also been described as a glycoprotein composed of 2 comparably composed protein subunits covalently linked by disulfide bonds. Fibroin filaments have both crystalline and amorphous domains. 2 The amorphous domains are characterized by the presence of amino acids with bulkier side chains, 26 whereas the crystalline domains are characterized by high percentages of alanine, glycine, and serine. 2 Fibroin is a highly insoluble protein containing, as a whole, up to 90% of the amino acids glycine, alanine, and serine. 27 According to another source, Fibroin contains 46% glycine, 29% alanine, and 12% serine. 25
More detailed information on the composition of Fibroin, from the cocoon of the B mori caterpillar, indicates that it consists of 2 polypeptide chains or, more specifically, heavy and light chains of 391 kDaa and 25 kDaa, respectively; a disulfide bridge links the heavy chain to the light chain. 28 The heavy chains contain 5263 residues, composed of 45.9% glycine, 30.3% alanine, 12.1% serine, 5.3% tyrosine, 1.8% valine, and only 4.7% of the other 15 amino acid types.
Sericin
Sericin, also referred to as silk glue, is a globular protein that constitutes 25% to 30% of silk proteins. It contains 18 amino acids, most of which have highly polar side-chains, containing hydroxyl, carboxyl, or amino groups. The highly hydrophilic nature of Sericin is due to the high content of serine and aspartic acid, approximately 33.4% and 16.7% of Sericin, respectively. 9 The predominant amino acids comprising Sericin are serine, glycine, and glutamic acid, and side-chain hydroxyl, carboxyl, and amino groups enable easy cross-linking, copolymerization and blending with other natural or synthetic polymers. 29 According to another source, Sericin contains 37% serine, 17% glycine, and 16% aspartate. 25
Depending on the solubility, Sericin can be separated into 3 fractions 2 : A, B, and C. Sericin A (17.2% nitrogen) comprises the outermost layer and is insoluble in hot water. Sericin B (16.8% nitrogen) is the middle layer and, on acid hydrolysis, yields the same amino acids as Sericin A and, additionally, tryptophan. Sericin C is the innermost layer, positioned adjacent to the Fibroin strands.
Because the method of production of Sericin involves extraction from cocoons using soaps and detergents, alkali soaps and detergents are typically present as impurities. 9
USE
Cosmetic
The safety of the silk protein ingredients included in this safety assessment is evaluated based on the expected use of these ingredients in cosmetics. The Panel uses data received from the US Food and Drug Administration (FDA) and the cosmetics industry to determine expected cosmetic use. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys of maximum reported use concentrations, by product category, which are conducted by the Personal Care Products Council (Council). Collectively, the use frequency and use concentration data indicate that 7 of the 10 Silk protein ingredients are currently being used in cosmetic products. According to these data, the following 3 Silk protein ingredients are not being used in cosmetics: Fibroin MEA-Hydrolyzed Silk Silkworm Cocoon Extract
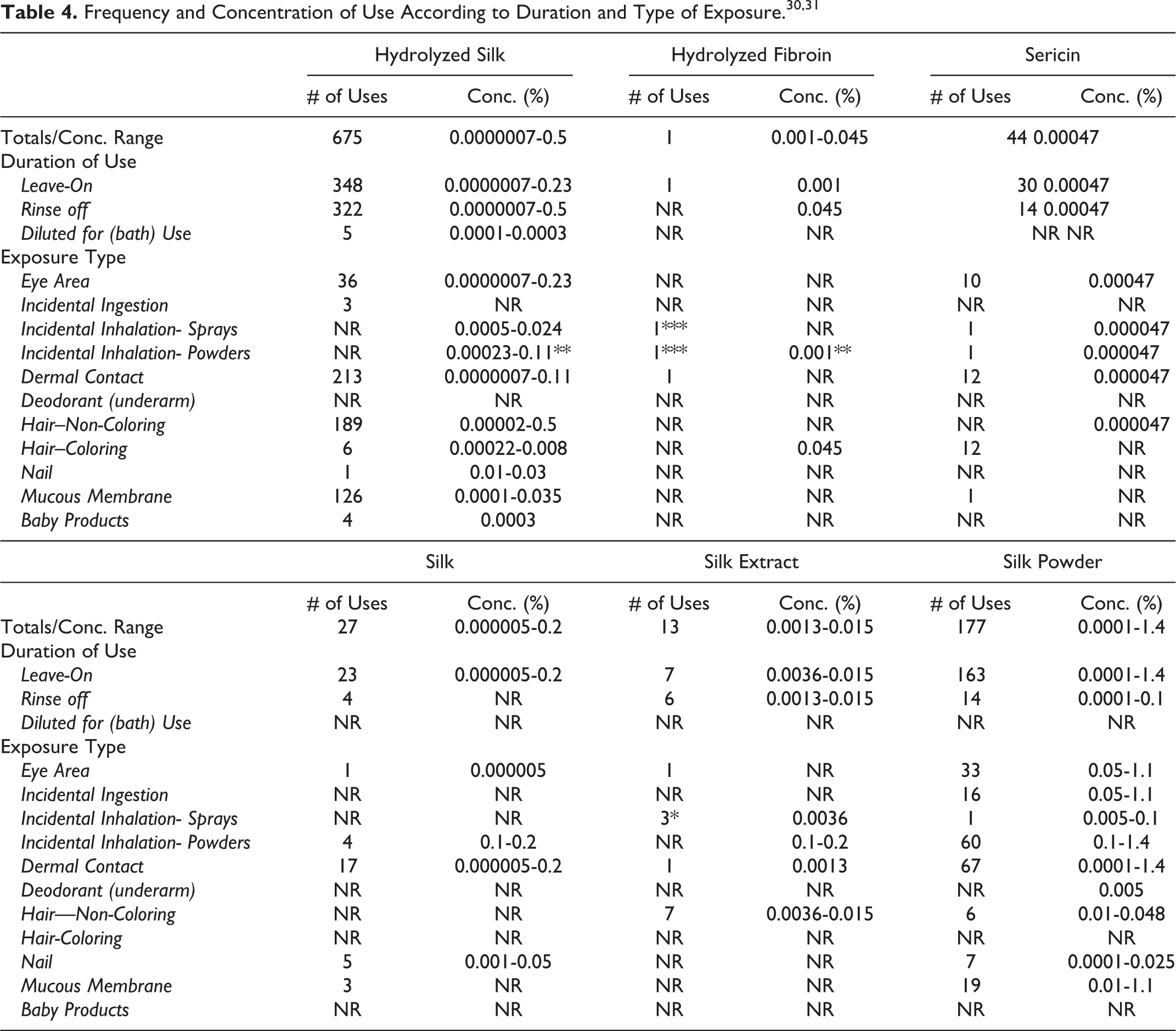
According to 2015 VCRP data, the greatest reported use frequency is for Hydrolyzed Silk (675 formulations, mostly rinse-off), followed by Silk Powder (177 formulations, mostly leave-on) (Table 4). 30 Lower use frequencies are reported for the remaining silk ingredients. The results of a concentration-of-use survey conducted in 2014 indicated that Silk Powder had the highest maximum concentration of use; it was used at concentrations up to 1.4% in leave-on products (face powders; Table 4). 31 In some cases, reported uses appear in the VCRP database, but concentrations-of-use data were not provided. For example, Hydrolyzed Sericin is reported as used in 4 cosmetic formulations, but use concentration data were not submitted.
Abbreviation: NR, not reported.
Totals = Rinse-off + Leave-on + Diluted for (Bath) Use Product Uses.
* It is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
** It is possible that these products may be powders, but it is not specified whether the reported uses are powders.
*** Not specified whether a powder or spray, so this information is captured for both categories of incidental inhalation.
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Cosmetic products containing Silk proteins may be applied to the skin and hair or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Hydrolyzed Silk and Silk Extract are used in hairspray at maximum concentrations up to 0.024% and 0.0036%, respectively. Silk Powder is also used in hairspray (maximum concentration 0.02%). Hydrolyzed Fibroin and Silk Powder are used in perfume at maximum concentrations up to 0.000047% and 0.1%, respectively. Maximum use concentrations for the following ingredients in face powders are reported: Sericin (0.00047%), Silk (0.1%-0.2%), and Silk Powder (0.1%-1.4%). In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays. 32 –35 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 32,33
The silk proteins reviewed in this safety assessment do not appear on the list of ingredients prohibited from use in cosmetic products marketed within the European Union (Annex II of the Council of the European Communities Council Directive) or on the list of ingredients with use restrictions in products marketed within the European Union (Annex III). 36
Noncosmetic
Fibroin
Silk fibers made from ibroin have many uses in textiles (medical and industrial applications) mainly because of the unique properties of Fibroin, such as water absorbency, dying affinity, thermo-tolerance, luster, and insulation properties. Fibroin is also a raw material for producing precious fabrics, parachutes, tire lining materials, artificial blood vessels, and surgical sutures. 9 Natural, nonabsorbable Silk surgical suture containing the organic protein, Fibroin, is an FDA-approved medical device. 37
Toxicokinetics
Toxicokinetics studies of the silk proteins reviewed in this safety assessment were not found in the published literature, and unpublished data were not submitted.
Toxicology
Single Dose (Acute) Toxicity
Oral
Hydrolyzed Silk
The acute oral toxicity of Hydrolyzed Silk (MW ∼300 Da; acid and enzyme hydrolysis product) was evaluated using rats (5 males, 5 females; strain not stated). 38 A single dose of 10 g/kg was administered orally to each animal. No signs of toxicity were observed during the 14-day observation period after dosing.
In another study, the acute oral toxicity of Hydrolyzed Silk protein (MW ∼1000 Da; produced via alkali hydrolysis) was evaluated using albino rats (5 males, 5 females). 39 A single dose of the test material (5 g/kg body weight) was administered using an intragastric feeding needle. Signs of toxicity were not observed during the study and none of the animals died. The median lethal dose (LD50) was >5 g/kg.
The acute oral toxicity of Hydrolyzed Silk protein (15%-25% in water; specific gravity = 1.10) was studied using 10 albino rats (5 males, 5 females). 39 The test substance was administered orally at a dose of 5 g/kg, and dosing was followed by a 14-day observation period. Gross necropsy was performed on all animals. Only 1 animal (male) died, and thoracic cavity filled with fibrous tissue was noted at necropsy. Whether or not this death was due to experimental error was not stated. Gross changes were not observed in the remaining animals. The test substance was classified as nontoxic.
Silk
Ten male Sprague-Dawley rats were dosed orally (16 g/kg) with Silk. 40 The form of Silk administered, test concentration, vehicle, and dosing method were not stated. Dosing was followed by a 14-day observation period. None of the animals died, and except for slight lethargy, there were no signs of toxicity during the observation period. The oral LD50 was >16 g/kg, and Silk was considered nontoxic in this study.
Silk Powder
A 30% solution of Silk Powder in distilled water was administered orally (feeding tube) to 12 female DD-strain mice. 41 The animals received doses up to 12 g/kg body weight (dose volume = 40 mL/kg). Dosing was followed by a 7-day observation period. Toxic signs were not observed during the study, and the test substance was classified as practically nontoxic (LD50 >12 g/kg body weight).
Repeated Dose Toxicity
Dermal
Hydrolyzed Silk
In a cumulative skin irritation study involving 8 Hartley guinea pigs, a 6.5% aqueous solution of Hydrolyzed Silk protein (MW = 650 Da) was applied to the back once daily for 35 days. 42 The animals were killed and necropsied at the end of the dosing period. Body weight gain was normal, and no abnormalities were noted at necropsy. Other study results relating to skin irritation potential are included in Table 5.
Skin Irritation/Sensitization Potential of Silk Ingredients.

Cytotoxicity
Sericin obtained via urea extraction was slightly toxic to mouse fibroblasts in vitro at concentrations as low as 60 µg/mL, and toxicity was substantial (ie, severely harmful) at concentrations greater than 100 µg/mL. When using other extraction methods (heat, acid, or alkaline), Sericin yielded less toxicity, as measured by the percentage of viable fibroblasts. 44
Skin Depigmentation
Sericin
Sericin was formulated as an 8% cream and applied to one side of the extremity (arm and leg) of renal patients who normally experienced dry and itchy skin. 45 A cream base was applied to the other extremity and served as the control. From 47 subjects who completed the study, the skin hydration of the patients’ extremities increased after receiving both Sericin cream and the cream base, but the changes in skin hydration were much greater on the side receiving the Sericin cream than on the side receiving the cream base. Additionally, at the end of the study, the skin pigmentation level was significantly reduced on both the arms (P = 0.032) and legs (P = 0.021) of the Sericin-treated side compared with the side treated with cream base.
The depigmentation effect was presumed to result from tyrosinase inhibition. The degree of inhibition of tyrosinase (the rate-limiting enzyme for melanin production) activity by Sericin is known to depend upon the extraction method and silk strain source. 46 For example, colored silk cocoons, which contain flavonoids and carotenoids, exhibit higher anti-tyrosinase activity than white-shelled cocoons.
Reproductive and Developmental Toxicity
Reproductive and developmental toxicity studies of the silk proteins reviewed in this safety assessment were not found in the published literature, and unpublished data were not submitted.
Genotoxicity
Hydrolyzed Silk
The genotoxicity of Hydrolyzed Silk protein (10% aqueous) was evaluated in the Ames test using the following Salmonella typhimurium strains, with and without metabolic activation: TA98, TA100, TA1535, TA1537, and TA1538. 47 Results were negative in all bacterial strains. The positive and negative controls performed as expected in this assay.
Carcinogenicity
Cell Proliferation
Sericin
The effect of Sericin on the rat insulinoma cell line (seeded on ASF104 culture medium [serum-free medium containing insulin and transferrin]) was evaluated. Bovine serum albumin (BSA) served as the control protein. The RIN-5F cell cultures were identified as follows: ASF104 (1 mL), ASF104 with 0.1% Sericin, and ASF104 with 0.1% BSA. The cells were cultured for 22 hours. Viable and nonviable cell numbers were determined using the trypan blue exclusion method. The cells in the control culture failed to proliferate. However, RIN-5F cells propagated significantly in the presence of Sericin (P < 0.05) or BSA (P < 0.01). Therefore, Sericin and BSA were efficient inducers of RIN-5F cell proliferation. 48
Anticarcinogenicity
Sericin
A study was performed to evaluate the protective effect of Sericin on tumor promotion in the 7,12 dimethylbenz[a]anthracene (DMBA)-initiated and 12-O-tetradecanoylphorbol-13-acetate (TPA)-promoted mouse skin tumorigenesis model. 49 In the first experiment, Sericin was applied topically to DMBA (190 nM)-initiated, female CD-1 (ICR): Crj mice (groups of 16) at doses of 2.5 mg and 5 mg twice weekly for 16 weeks. Sericin was applied 30 minutes prior to each promotion treatment with TPA (3.2 nM). The tumor necrosis factor α (TNF-α, pro-inflammatory cytokine) and 4-hydroxynonenal (4-HNE) staining method was used, whereby the affinity-purified goat polyclonal TNF-α antibody and monoclonal antibody were placed on the specimens. Sericin caused a significant reduction in both the tumor incidence and tumor multiplicity at doses of 2.5 mg and 5 mg per application, compared to the control group that was not treated with Sericin. The expression of TNF-α protein and the level of 4-HNE in normal epidermis were significantly reduced in both Sericin treatment groups.
In the second experiment (groups of 5 ICR mice), Sericin (5 mg) was applied topically to dorsal skin 30 minutes prior to application of TPA. The same doses of TPA and Sericin (used in first experiment) were applied twice at an interval of 24 hours. Treatment with Sericin inhibited double TPA treatment-induced morphological changes; these were indicative of an inflammatory response, that is leukocyte infiltration, hyperplasia, and cell proliferation. Treatment with Sericin also significantly suppressed the elevation of 4-HNE levels and elevated expressions of c-fos, c-myc, and cyclooxygenase-2 (COX-2) in normal epidermis that were induced by double application of TPA. The results of this study suggest that Sericin has a protective effect against tumor promotion in mouse skin by suppressing oxidative stress, inflammatory responses, and TNF-α. 49
In another study, male CD-1 (ICR): Crj mice (2 groups of 11 and 12, respectively) were fed diets supplemented with 1.5% Sericin (3 g/kg/d) or 3% Sericin (6 g/kg/d) for 5 weeks. 50 The animals also received weekly injections of 1,2 dimethylhydrazine (DMH) during the initial 3 weeks of the study. The feeding of Sericin in the diet resulted in a dose-dependent decrease in the development of colonic aberrant crypt foci. In a second experiment, mice were fed a diet supplemented with 3% Sericin for 115 days. The animals were also injected with DMH weekly during the initial 10 weeks. Both the incidence and number of colon tumors were suppressed by Sericin consumption.
A study was performed to assess the protective effect of Sericin on mid-wavelength ultraviolet light (UVB)-induced acute damage and tumor promotion in HR-1 hairless mouse skin. 51 Three groups of 10 mice were treated dermally with Sericin, BSA, and vehicle (ethanol), respectively, in the first experiment. One group of mice was treated with 180 mJ/cm2 UVB light once daily for 7 days, after which red sunburn lesions of the skin were observed. Both the area and the intensity of the redness of these lesions were reduced by the topical application of 5 mg Sericin immediately after UVB treatment. The differences (area and intensity of the redness) between the vehicle and Sericin groups were statistically significant (P < 0.01). This was not true when the group treated with BSA (5 mg), rather than Sericin, was compared to the vehicle control. The results of immunohistochemical analyses indicated that the application of Sericin suppressed UVB-induced elevations in 4-HNE, expression of COX-2 protein, and proliferating cell nuclear antigen–labeling index in the UVB-exposed epidermis.
Three groups of 15 mice of the same strain were treated with Sericin, BSA, and vehicle (ethanol), respectively, in the second experiment. One group of mice was treated (dermal application) with 200 nmol DMBA, followed by a 1-week nontreatment period. 7,12 dimethylbenz[a]anthracene–treated skin was then irradiated with 180 mJ/cm2 of UVB twice weekly, and each irradiation was followed by topical treatment with Sericin (5 mg). Another group of mice was treated similarly with BSA (5 mg), rather than Sericin. Treatments (UVB dosing, followed by topical treatment) were repeated for 22 weeks. A statistically significant reduction in both tumor incidence and multiplicity was noted at a dose of 5 mg, indicative of a suppressive effect of Sericin. When compared to all of the animals in the vehicle and BSA groups having skin tumors 22 weeks after the topical application of DMBA, only 6% of the DMBA-exposed mice in the Sericin-treated group exhibited skin tumors, indicating 94% (P < 0.001) reduction in tumor incidence. Similarly, when the tumor data were evaluated for tumor multiplicity (ie, the number of tumors per mouse), from the first tumor appearance to the termination of the experiment, Sericin produced statistically significant (P < 0.05) protection against UVB-induced tumor promotion in DMBA-exposed mouse skin. The results of this study (including the first and second experiments) suggest that Sericin possesses a photoprotective effect against UVB-induced acute damage and tumor promotion by reducing oxidative stress, COX-2, and cell proliferation in mouse skin. 51
Irritation and Sensitization
Skin Irritation and Sensitization
Skin irritation and sensitization studies on silk protein ingredients are summarized in Table 5. These ingredients are, at most, mild skin irritants and lack skin sensitization potential. In the only available human skin irritation/sensitization tests in which test concentrations were reported, 20% aqueous Hydrolyzed Silk was a nonirritant and a non-sensitizer and 6.5% aqueous Hydrolyzed Silk was a nonirritant (skin irritation only evaluated).
Allergenicity
Human
Silk
The relationship between Silk sensitization and asthma incidence was evaluated in 871 children living in China. 52 Skin testing was performed using a slightly modified version of the semiquantitative puncture method. The results of multivariate analyses of asthma incidence and skin test reactivity to aeroallergens were presented. Individual skin test results were not provided. Children who were sensitized to Silk had 2.6 times higher odds of having asthma than did nonreactors, after adjustment for age, gender, familial correlations, and skin test reactivity to other aeroallergens using generalized estimating equations. This association between sensitization to Silk and asthma yielded lower statistical P values when the eosinophil counts of the participants were included as either a categorical variable or a linear term in the multivariate model.
Sixty-four (<15 years old; males and females) children with Silk-induced asthma in China were studied. 53 The diagnosis was based on a history of wheezing, positive skin tests to Silk, positive nasal or conjunctival provocation tests, or serum immunoglobulin E (IgE)-Silk waste (serum antibodies against Silk waste [severely broken Silk threads, used only as filling for bed quilts or clothes and mattresses]). The average age of asthma onset was 4 years 2 months. Conjunctival provocation tests were performed on 80% of the cases. The first symptom was observed an average of 10 months after initial exposure to Silk. Asthma was accompanied by allergic rhinitis in 61% of the patients and was accompanied by conjunctivitis in 14% of the cases. In most cases, asthma occurred during the winter, due to the seasonal use of bed quilts or clothes filled with Silk. The average mean wheal diameter elicited by Silk in prick tests was greater than the diameters measured from 2 histamine equivalent prick tests per Silk-sensitive subject.
In relation to the preceding study, it should be noted that allergenic proteins have been extracted from 1 Silk batch that was imported to be used as filling material for bed mattresses and rugs. 54 Immunoglobulin E and immunoglobulin G (IgG) antibodies to the extracted Silk proteins were measured by radioallergosorbent in sera of 9 Silk-sensitive subjects and in sera of healthy control subjects. Immunoglobulin E and IgG antibodies to the individual Silk polypeptides (40 kDa, 45 kDa, and 70 kDa) were detected using the immunoblot technique; the 6 kDa Silk polypeptide bound only IgG. The sera of Silk-sensitive subjects contained high titers of IgE and low titers of IgG antibodies to the separated Silk polypeptides. Low IgG antibody titers to a limited number of these polypeptides were detected in sera of control subjects. In another study involving 10 patients with a positive skin prick test to Silkworm crude extract, arginine kinase (42-kDa protein) was identified as a major allergen in this crude extract. 55
Phototoxicity
Hydrolyzed Silk
The phototoxicity of 6.5% aqueous Hydrolyzed Silk protein (MW = 650 Da; produced by alkaline and enzyme hydrolysis) was evaluated using groups of 6 Hartley guinea pigs. 56 The 3 groups were identified as test, positive control, and negative control groups. 8-Methoxypsoralen (1%) served as the positive control. The negative control was not stated. The test material was applied topically (dose = 0.05 mL/2 × 2 cm) to 2 sites on dorsal skin. One site was irradiated once with a FL-40 S lamp and BLP lamp (wavelength range not stated), and the other site was covered. The sites were examined macroscopically at 24, 48, and 72 hours. Phototoxicity was evaluated based on the difference in severity of skin reactions between the irradiated and non-irradiated sites. Hydrolyzed Silk protein was classified as non-phototoxic. Positive responses were observed in all guinea pigs treated with 8-methoxypsoralen + light.
Silk Powder
Silk Powder (0.1 g) was applied to the back (2 sites (2 areas per site) of each of 6 female guinea pigs of the Hartley strain. 41 The test site (cm2 area not stated) was covered with a patch plaster for 4 hours, after which the sites were irradiated with UV (minimal erythema dose, 15-minute exposure) from a 20-W lamp. The test sites were evaluated for erythema and eschar formation after 24 and 48 hours. Positive reactions were not observed in this study.
Photoallergenicity
Hydrolyzed Silk
The photoallergenicity of 6.5% aqueous Hydrolyzed Silk protein (MW = 650 Da; produced by alkaline and enzyme hydrolysis) was evaluated using groups of 6 Hartley guinea pigs. 57 The 3 groups were identified as test, positive control, and negative control groups; the positive control was 3,5,4’-tribromosalicylanilide (2% in 85% dimethyl sulfoxide), but the negative control was not stated. The test material was applied transdermally (0.05 mL/2 × 2 cm), with or without UV, 5 times per week (2 hours per day) for a total of 10 applications. Applications were made on both sides of the dorsal area, symmetrically. One side was irradiated for 2 hours, and the other side was covered. The photo challenge phase was initiated after a 2-week nontreatment period. The test material was applied to 2 sites. One side was irradiated for 2 hours, and the other side was covered. The application site was examined macroscopically after the challenge and 24, 48, and 72 hours later. No effects were observed in negative controls or in guinea pigs treated with the test material (with or without exposure to light). Hydrolyzed Silk protein was considered non-photosensitizing in this study. The expected results were achieved with the positive control.
Case Reports
According to one case report, recurrent granulomas with remarkable infiltration of eosinophils may have resulted from an IgE-mediated hypersensitivity reaction to silk Fibroin, a component of the braided Silk suture used. 58 In this report, a lateral skin flap technique had been performed to correct tracheostomal stenosis, using Silk sutures, after a total laryngectomy.
Adverse reactions to virgin Silk sutures in 12 cataract surgery patients have also been reported. 59 Nodular episcleritis, peripheral corneal ulceration, and wound necrosis with dehiscence were observed, sometimes resulting in endophthalmus or epithelial downgrowth. Conjunctival and scleral histopathologic studies in 4 eyes showed acute and chronic inflammation with multinucleated giant cells. Type I allergic responses and upregulated levels of specific IgE were reported to occur in patients after repeated surgical procedures. 58,60
A female patient with a history of severe atopic dermatitis and various allergies, and a family history of atopic eczema and asthma, presented with exacerbation of eczema on her hands and wrists and urticarial papules on the flexor aspects of both forearms. 61 The same types of lesions had been observed on both arms after wearing the same Silk shirt but quickly disappeared after the shirt was taken off. Radioallergosorbent tests yielded positive results for Silk waste (k73) of 2.08 universal arbitrary units (Ua)/mL and positive results for Silk (k74) of 3.62 Ua/mL. The total IgE count was >5000 kilo units (kU)/L. These results confirmed the diagnosis of immunological contact urticaria caused by Silk.
Ocular Irritation
Animal
Hydrolyzed Silk
The ocular irritation potential of Hydrolyzed Silk protein (15%-25% in water) was evaluated in the Draize test using 6 New Zealand white rabbits. 62 The test substance (0.1 mL) was instilled into 1 eye of each animal, and eyes were not rinsed. Contralateral eyes served as controls. Observations for ocular reactions were made up to 72 hours post-instillation. The test substance was practically nonirritating to the eyes of rabbits.
The ocular irritation potential of 6.5% aqueous Hydrolyzed Silk (MW ∼300 Da) was studied using 6 New Zealand white rabbits. 63 The test material (0.1 mL) was instilled into the right eye of each animal, and the left eye served as the untreated control. Reactions were scored at 24, 48, and 72 hours post-instillation. Slight conjunctival redness, the only reaction reported, was observed in 1 rabbit. It was concluded that it is not likely that Hydrolyzed Silk would be classified as an ocular irritant, according to the definitions of the US Federal Hazardous Substances Act. The ocular irritation potential of a higher MW Hydrolyzed Silk (MW = 650 Da; test concentration not stated) was evaluated in New Zealand white rabbits according to the same test procedure, and the results were negative. 64
Hydrolyzed Silk (MW ∼ 1000 Da; produced by alkali hydrolysis) was placed (0.1 mL) in the right eye of each of 6 New Zealand white rabbits. 39 Observations for any signs of corneal opacity, iritis, or conjunctivitis were made at 24, 48, and 72 hours post-instillation. The authors concluded that Hydrolyzed Silk was practically nonirritating to the eyes of rabbits.
In a cumulative ocular irritation test, 6.5% aqueous Hydrolyzed Silk protein (MW = 650 Da; 0.1 mL; produced by acid, alkaline, and enzyme hydrolysis), was instilled into the eyes of 6 New Zealand white rabbits 3 times per day for 4 days continuously. 65 Conjunctival redness was observed in 5 of 6 animals at 3 or 4 days. Reactions in the cornea or iris were not observed. The authors concluded that Hydrolyzed Silk protein was practically nonirritating to the eyes of rabbits.
Silk
Silk (0.1 g) was instilled into 1 eye of each of 9 adult albino rabbits. 40 The eyes of 3 rabbits were rinsed immediately after instillation. Untreated eyes served as controls. The eyes were examined at 24, 48, and 72 hours post-instillation. Transient conjunctival redness (unrinsed eyes) was observed in 5 of 6 rabbits. However, no effects on the cornea or iris were observed. Ocular irritation was not observed in the 3 rabbits subjected to ocular rinsing. Silk was classified as a nonirritant in this study.
Silk Powder
The ocular irritation potential of a Silk Powder solution (10% in saline) and the supernatant fluid from this solution (filtered after 24 hours) was evaluated using a total of 6 white rabbits. 41 Four rabbits received the 10% in saline solution and 6 rabbits received the supernatant. Either test substance (0.1 mL) was instilled into the right eye, and eyes were rinsed. Untreated left eyes served as controls. The eyes were examined for reactions for up to 168 hours post-instillation. The Silk Powder solution (10% in saline) was classified as slightly positive. The supernatant from this solution was not an ocular irritant.
In Vitro
Hydrolyzed Silk
The ocular irritation potential of Hydrolyzed Silk protein (MW = 300 Da; 2% active solution) was evaluated in the in vitro hen’s egg test on the chorioallantoic membrane (HET-CAM). 14,66 The material (0.3 mL) was tested on the chorioallantoic membrane of fertilized Leghorn hens’ eggs that had been incubated for 10 days. Results were negative (score = 0.3).
Hydrolyzed Silk protein (MW = 300 Da; 10% active solution) was tested for ocular irritation potential in the in vitro red blood cell aggregation test, which evaluates effects on the cytoplasmic membrane. 14,67 A total irritation classification was obtained by determining the hemolysis/denaturation (L/D) ratio. A substance with an L/D of >100 was classified as a nonirritating. Hydrolyzed Silk caused neither hemolysis nor denaturation and was classified as nonirritating.
The Irritection assay was used to evaluate the ocular irritation potential of Hydrolyzed Silk. 68 The test material was applied to the Irritection system at dose volumes of 25, 50, 75, 100, and 125 µL. The samples remained at room temperature for 24 hours and were then analyzed by spectrophotometry. Over the range of dose volumes tested, ocular Irritection scores for Hydrolyzed Silk ranged from 2.5 to 3.5. Scores in this range corresponded to a classification of minimally irritating.
The ocular irritation potential of Hydrolyzed Silk was studied using the EpiOcular model assay. 69 The test material was applied to a reconstructed human corneal epithelial model for 30 minutes, and cell viability was measured by dehydrogenase, present in the cell mitochondria, reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), into blue, MTT formazan salt. The irritation potential of the test material is dictated by the reduction in tissue viability of exposed tissues, compared to the negative control (sterile deionized water). Methyl acetate served as the positive control. Hydrolyzed Silk was classified as a nonirritant. The negative and positive controls were nonirritating and irritating, respectively.
Other Effects
Immunological Responses
Sericin and fibroin
In Vitro
Soluble Sericin proteins (2 µg of Sericin per well tissue culture plate) extracted from native Silk fibers did not induce significant macrophage activation. 70 Macrophages exposed in vitro to the Silk preparations failed to respond with consistently elevated levels of TNF in either short- or long-term cultures. However, the suspension of the crystalline particles prepared by enzymatic digestion of Silk Fibroin was the only Silk preparation that yielded significant TNF release, which was probably a nonspecific response to insoluble physical particulates, rather than a specific, chemically induced response to Silk. Whether or not the statistical significance of this finding was determined was not stated. However, the average TNF release (corrected for volume and expressed as total release from specified cell count) and standard error of the mean were determined.
A study was designed to investigate the inflammatory mediators induced by Sericin. 71 Sericin was analyzed in an in vitro macrophage (mouse alveolar macrophage cell line) and monocyte (mouse monocyte cell line) assay to assess cytokine activation. These cell lines were used for monitoring levels of interleukin (IL)-1β and TNF-α generated after activation by Sericin at concentrations of 0.2 to 1.0 mg/mL. Inflammatory mediators activated by Sericin were also investigated in vivo, using a rat wound-healing model. At the concentrations tested, silk Sericin increased the amounts of inflammatory mediators and pro-inflammatory cytokines, TNF-α and IL-1β (involved in the modulation of skin growth, repair, and scarring during inflammation). However, the maximum levels of TNF-α and IL-1β released from monocytes and macrophage cells after silk Sericin exposure were 500 and 350 pg/mL, respectively. It was noted that these levels of cytokines would not be sufficient to cause an inflammatory response or prevent cellular proliferation.
The suppression of inflammation by Sericin has been reported. 72 Sericin solution (0.004-0.080 mg/mL) applied topically to the top of the hind paw of rats prior to a carrageenan subcutaneous injection under the plantar surface of the hind paw exhibited anti-inflammatory activity, similar to the effect of indomethacin (a nonsteroidal anti-inflammatory drug used as a control). The amount of mast cells in rat tissue treated with Sericin or indomethacin was much lower compared to the number of cells found in tissue treated with water (control). Further investigation indicated that Sericin did not cause a hypersensitivity reaction. On the contrary, it inhibited COX-2 and inducible nitric oxide synthase (iNOS) production (monitored by total RNA and real-time polymerase chain reaction) in fibroblast cell culture, resulting in lowering the inflammation of the carrageenan induction.
Wound Healing
Nonhuman
Sericin
The effect of a Sericin cream on wound healing was evaluated using 18 male Sprague-Dawley rats (8 weeks old). 73 The composition of the cream was described as follows: 8% Sericin, white petrolatum, mineral oil, lanolin, glycerin, bisabolol, propylparaben, and methylparaben. Except for Sericin, the concentration of each cream component was not stated. The cream was applied topically to full-thickness skin wounds on the dorsum of each animal, and wound surfaces were observed for 15 days post-application. Cream base without Sericin served as the control. Histological examination of wounds after 15 days of treatment with 8% Sericin cream revealed complete healing, no ulceration, and an increase in collagen, as compared to treatment with the control cream. Wounds treated with the control cream had some ulceration and acute inflammatory exudative materials.
In a similar study involving 45 Sprague-Dawley rats (8 weeks old), 8% Sericin cream was applied to full-thickness wounds on the dorsum of each animal. Cream base without Sericin served as the control. Excised rat tissue was prepared for cytokine determination. Interleukin-1β and TNF-α are pro-inflammatory cytokines that are involved in a variety of immunological functions. Wounds treated with Sericin cream did not yield significantly high levels of IL-1β and TNF-α on day 7, which suggests that the cream did not induce an inflammatory or immunological response. 74
In Vitro
Sericin
Human skin fibroblasts were incubated with Sericin in vitro for 72 hours. 75 The cell count in treated cultures after 72 hours was enhanced to 250% of the untreated (ie, no-Sericin) control cultures. In another study, replacing the culture medium with Sericin solution in the mouse L929 fibroblastic cell line in culture increased the percentage of cell proliferation significantly, especially at a high Sericin concentration (1.0 mg/mL). 71 The amount of NF-1α and IL-1β released from alveolar macrophage NR8383 and mouse J774.2 monocyte cell lines after the addition of Sericin (0.2-1.0 mg/mL) to the culture media was negligible, indicating that Sericin did not cause severe damage to the cells.
Summary
The safety of the following 10 silk protein ingredients in cosmetics is reviewed in this safety assessment: Fibroin, Hydrolyzed Fibroin, Hydrolyzed Sericin, Hydrolyzed Silk, MEA-Hydrolyzed Silk, Sericin, Silk, Silk Extract, Silk Powder, and Silkworm Cocoon Extract. These ingredients are reported to function as skin and hair conditioning agents and bulking agents in cosmetic products. Frequency of use data from the FDA’s VCRP and the results of an industry survey indicate that 7 of the 10 Silk protein ingredients are being used in cosmetic products. Silk Powder has the highest reported maximum concentration of use; it is used at concentrations up to 1.4% in leave-on products (face powders).
The silkworm, B mori, produces Silk proteins during the final stage of larval development, and 2 Silk proteins, Fibroin and Sericin, have been distinguished as major components of Silk cocoons. In the process of manufacturing Silk, Fibroin is separated from Sericin by a degumming process. There are several methods for removing Sericin in the degumming process of cocoons. Because the method of production of Sericin involves extraction from cocoons using soaps and detergents, detergents and alkali soaps and are typically present as impurities in Sericin.
Hydrolyzed Silk is prepared from the cocoon of B mori. Hydrolyzed Silk protein (MW = 300 Da) may be prepared by acid, alkaline, or enzyme hydrolysis; Hydrolyzed Silk protein (650 Da) may be prepared by alkaline and enzyme hydrolysis.
In acute oral toxicity studies involving rats, LD50 values of >10 g/kg body weight (Hydrolyzed Silk protein, MW ∼300 Da), >5 g/kg (Hydrolyzed Silk protein, MW ∼1000 Da), >16 g/kg (Silk), and >12 g/kg (30% aqueous Silk Powder) were reported. Signs of toxicity were not observed.
In a repeated insult patch test (RIPT, occlusive patches) involving dermal applications of Silk Powder (50% in sterile water) to 20 guinea pigs over a 3-week period, 2 animals died. However, necropsy results were not indicative of a test substance–related effect. Similarly, no abnormalities were noted at necropsy in a cumulative skin irritation in which 6.5% aqueous Hydrolyzed Silk (MW = 650 Da) was applied to 8 Hartley guinea pigs once daily for 35 days.
Sericin obtained via urea extraction was toxic to mouse fibroblasts in vitro at concentrations as low as 60 µg/mL. When compared to urea extraction, the extraction of Sericin by other methods resulted in reduced toxicity to mouse fibroblasts in vitro.
In the Ames test, results for 10% aqueous Hydrolyzed Silk protein were negative. Sericin and BSA were efficient inducers of rat RIN-5F cell proliferation in a rat insulinoma cell line. The results of another study involving CD-1 (ICR): Crj mice suggested that Sericin had a protective effect against tumor promotion in mouse skin by suppressing oxidative stress, inflammatory responses, and TNF-α.
The results of a study in mice suggested that Sericin possesses a photoprotective effect against UVB-induced damage and tumor promotion by reducing oxidative stress and cell proliferation in mouse skin. In another study involving mice, the feeding of diets supplemented with 1.5% or 3% Sericin for 5 weeks resulted in a dose-dependent decrease in the development of colonic aberrant crypt foci.
Undiluted Hydrolyzed Silk protein (MW ∼300 Da; dose = 0.5 mL/2.5 cm2) caused reactions ranging from very slight to well-defined erythema on intact and abraded skin of rabbits. However, a primary irritation index (PII) of 1.1 was reported, and the test material was not classified as a primary skin irritant. Hydrolyzed Silk protein (MW ∼1000 Da; dose = 0.5 mL/2.5 cm2; PII = 0.65) and Hydrolyzed Silk protein (MW = 650 Da; dose = 0.5 mL/2.5 cm2; PII = 0.05) were also classified as nonirritating to the skin of rabbits. Hydrolyzed Silk protein (15%-25% aqueous) was nonirritating to the skin of rabbits. In a cumulative skin irritation study involving guinea pigs, Hydrolyzed Silk protein (MW = 650 Da) was considered nonirritating to the skin. Hydrolyzed Silk protein (MW = 650) did not induce skin sensitization in a study involving guinea pigs. Silk Powder (0.5 g) was nonirritating to abraded or intact skin of rabbits, and Silk Powder (0.1 g) was nonirritating and non-sensitizing to the skin of guinea pigs.
There was no evidence of skin irritation in 6 rabbits after the application of Silk (0.5 g) under a 2 cm2 patch for 24 hours. Silk Powder (0.5% wt/vol in distilled water) did not induce sensitization in an RIPT involving 20 guinea pigs. Also, the results of a preliminary skin irritation test at concentrations up to 10% in distilled water were negative. Similarly, Silk Powder (50% in sterile water) did not induce sensitization in an RIPT involving 20 guinea pigs. Furthermore, the results of a preliminary skin irritation test indicated that 75% Silk Powder was reasonably tolerated by the 5 guinea pigs tested.
Negative results were reported for Hydrolyzed Silk protein (MW = 300 Da) in a skin irritation study involving 20 subjects and for Hydrolyzed Silk protein (MW = 650) in a study involving 24 subjects. In 2 RIPTs involving 57 subjects and 49 subjects, respectively, Hydrolyzed Silk protein (MW ∼1000 Da; dose ∼0.2 mL/1” × 3/4” area and dose ∼2 mL/4 cm2 area, respectively) was not classified as a skin irritant or sensitizer. In a human repeated insult patch test involving 48 subjects, the results relating to the skin irritation and sensitization potential of Hydrolyzed Silk protein (MW ∼300 Da; dose = 20 µL/40 mm2 Finn chamber) were negative. Silk Powder (0.05 g) was nonirritating to the skin of 30 subjects.
Results for Hydrolyzed Silk were negative for skin irritation potential in the Irritection (dose volumes up to 125 µL) and Epiderm (aqueous solution containing 27%-32% Hydrolyzed Silk) in vitro assays. The mouse local lymph node assay yielded negative results relating to the sensitization potential of 20% aqueous Hydrolyzed Silk protein (MW = 300 Da).
A 6.5% aqueous solution of Hydrolyzed Silk protein (MW = 650 Da; dose = 0.05 mL/2 cm2) was neither phototoxic nor photoallergenic to the skin of guinea pigs. Silk Powder (0.1 g) also was not phototoxic when applied to the skin of guinea pigs.
Hydrolyzed Silk protein of an average MW of ∼300 Da, 650 Da, or ∼ 1000 Da did not induce ocular irritation when instilled into the eyes of rabbits. The tests involving Hydrolyzed Silk protein (MW ∼300 Da or ∼1000 Da) were single-instillation tests, whereas Hydrolyzed Silk protein (MW = 650 Da) was instilled 3 times per day for 4 days. In another test, Hydrolyzed Silk protein (15%-25% aqueous, single instillation) was nonirritating to the eyes of rabbits. Hydrolyzed Silk protein (MW = 300 Da; 2% active solution) was also negative for ocular irritation potential in the HeT-CAM, Irritection, and Epiderm in vitro assays. Silk (0.1 g) was also classified as a nonirritant when instilled into the eyes of rabbits, and the results for 10% aqueous Silk Powder were slightly positive when instilled into the eyes of rabbits.
The suppression of inflammation by Sericin applied topically was reported in a study on rats. A hypersensitivity reaction was not observed.
An association between sensitization to Silk and asthma incidence was found in a study of 871 children. In another study of 64 children, the average mean wheal diameter elicited by ilk in prick tests was greater than 2 histamine equivalent prick tests. Allergenic proteins have been extracted from a Silk batch imported for use as filling material for bed mattresses and rugs. Immunoglobulin E and IgG antibodies to the extracted Silk proteins were measured in the sera of 9 Silk-sensitive subjects, and these sera were found to contain high titers of IgE and low titers of IgG antibodies to the separated silk polypeptides. In a study involving 10 patients with a positive skin prick test to silkworm crude extract, arginine kinase was identified as a major allergen in this crude extract.
A female patient with a history of severe atopic dermatitis and various allergies, and a family history of atopic eczema and asthma, presented with exacerbation of eczema on her hands and wrists and urticarial papules on the flexor aspects of both forearms. 61 Radioallergosorbent tests yielded positive results for silk waste (k73) of 2.08 universal arbitrary units (Ua)/mL and positive results for Silk (k74) of 3.62 Ua/mL. The total IgE count was >5000 kilo units (kU)/L. These results confirmed the diagnosis of immunological contact urticarial caused by Silk.
In a case report, recurrent granulomas with remarkable infiltration of eosinophils may have resulted from an IgE-mediated hypersensitivity reaction to silk Fibroin. Additionally, type I allergic responses and upregulated levels of specific IgE have been reported in patients after repeated surgical procedures that involved the use of Silk sutures. In another case report, the diagnosis of immunological contact urticarial caused by Silk was confirmed.
Skin depigmentation has been observed in renal patients after application of an 8% Sericin cream for treatment of dry and itchy skin. Histological examination of wounds in rats after 15 days of treatment with 8% Sericin cream revealed complete healing, no ulceration, and an increase in collagen, as compared to treatment with the control cream.
Discussion
After reviewing the available data, concerns pertaining to skin depigmentation, allergenicity, inhalation exposure, and the insufficiency of data on certain silk protein ingredients were discussed by the Panel. The Panel reviewed a study in which an 8% Sericin cream applied to the skin of renal patients caused depigmentation, but agreed that a review of ingredients for drug effects (ie, noncosmetic effects, such as depigmentation) is not within the Panel’s purview. However, the Panel noted that an effect on cutaneous pigmentation would not be expected at the use concentrations of Silk protein ingredients in cosmetic products. Sericin is used at a maximum concentration of 0.00047% in cosmetic products.
Studies on patients inhalationally exposed to Silk waste via inhalation, which is distinct from the Silk protein ingredients that are being evaluated in this safety assessment, showed an association between asthma and dermal allergies to Silk in children in China. The Panel determined that the results of these studies do not support a cause-and-effect relationship of Silk protein ingredients in this safety assessment and the development of asthma.
Concern about pesticide residues and heavy metals that may be present in ingredients was expressed by the Panel. They stressed that the cosmetics industry should continue to use current good manufacturing practices (cGMPs) to limit impurities.
Some of the Silk protein ingredients are used in products that could be incidentally inhaled. For example, Hydrolyzed Silk and Silk Extract are used in hairspray at maximum concentrations up to 0.024% and 0.0036%, respectively, and the highest maximum use concentration for ingredient use in face powders is being reported for Silk Powder (1.4%). However, the Panel did not express concern over the use of these ingredients in formulations that might be inhaled. The Panel also noted that in aerosol products, 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients and their low use concentrations. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
The Panel determined that the available data are insufficient for evaluating the safety of MEA-Hydrolyzed Silk and Silkworm Cocoon Extract in cosmetic products and that the following data on these ingredients are needed:
Method of manufacture and impurities
Concentration of use
28-day dermal toxicity study; if absorbed, genotoxicity and reproductive and developmental toxicity data may be needed
Skin irritation and sensitization data
Conclusion
The Panel concluded that the following 8 ingredients are safe in the present practices of use and concentration in cosmetics, as described in this safety assessment. Fibroin* Hydrolyzed Fibroin Hydrolyzed Sericin Hydrolyzed Silk Sericin Silk Silk Extract Silk Powder
*Not reported to be in current use. Were this ingredient not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
The Panel also concluded that the available data are insufficient for to determine the safety of 2 silk protein ingredients in cosmetic products, MEA-Hydrolyzed Silk and Silkworm Cocoon Extract. Neither of these ingredients is reported to be in use.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Johnson contributed to conception and design; acquisition, analysis, and interpretation; and drafted manuscript. Heldreth contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Bergfeld, Belsito, Hill, Klaassen, Liebler, Marks, Shank, Slaga, Snyder, and Gill contributed to conception and design; acquisition and interpretation; and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.