Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of ceramides, which function in cosmetics primarily as hair-conditioning agents and skin-conditioning agents—miscellaneous. The Panel considered relevant data related to these ingredients. The Panel concluded that ceramides were safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Introduction
Ceramide ingredients are reported to function primarily as hair-conditioning agents and skin-conditioning agents—miscellaneous in cosmetics.
1
The 23 ingredients reviewed in this safety assessment are listed below: Ceramide 1 [retired] Ceramide 2 [retired] Ceramide 3 [retired] Ceramide 4 [retired] Ceramide 5 [retired] Ceramide 1A [retired] Ceramide 6 II [retired] Ceramide AP Ceramide EOP Ceramide EOS Ceramide NP Ceramide NG Ceramide NS Ceramide AS Ceramide NS dilaurate Caprooyl Phytosphingosine Caprooyl Sphingosine Hydroxypalmitoyl Sphinganine 2-Oleamido-1,3-Octadecanediol Caproyl Sphingosine Hydroxylauroyl Phytosphingosine Hydroxycapryloyl Phytosphingosine Hydroxycaproyl Phytosphingosine
Many of the reports found in the published literature presented efficacy studies on the named cosmetic ingredients, efficacy studies of other cosmetic ingredients or pharmaceuticals in which naturally occurring ceramide levels in the skin were evaluated, and data on pseudo-ceramides (such as that found in an approved medical device; the chemical structures of which were determined by the Expert Panel for Cosmetic Ingredient Safety (Panel) to be significantly different from those of the cosmetic ingredients addressed in this report). However, these studies were not relevant for assessing the safety of the ceramide ingredients included in this assessment. Additionally, a published paper that presented data from toxicology studies of several ceramides (1, 3, 3A, 3B, 6) and a phytosphingosine was reviewed, but the data from this paper were not incorporated into this report because some data points appeared to be merely cumulative to those of unpublished studies that were submitted by the Personal Care Products Council (Council) to the Cosmetic Ingredient Review (CIR), and the information presented in the paper is too incomplete to advance the development of a proper safety assessment. 2
Many published reports address the essential nature of extracellular ceramides as components of the epidermal permeability barrier. These ceramides are clearly segregated to the extracellular spaces of the stratum corneum and other upper layers of the epidermis. 3,4 The family of ceramides that serve this function comprise about 50% of the lipid weight, and 5% of the total weight, of the stratum corneum. 4 Many other reports address the central role of ceramides in sphingolipid metabolism and the mediation of antiproliferative and proapoptotic functions inside cells, including keratinocytes. However, the extracellular barrier-forming ceramides are partly O-acylated molecules with long-chain fatty acids, in contrast to the signal-transducing ceramides. 3 Further, naturally occurring ceramides are nearly cell impermeant, and metabolic pathways can suppress intracellular ceramide accumulation to protect cells from ceramide-induced apoptosis and other effects. Thus, much of the extensive literature on the signal-transducing properties of ceramides does not appear to be relevant and was not incorporated into this safety assessment report.
The names of ceramide ingredients were changed during the development of this safety assessment. For instance the International Nomenclature Cosmetic Ingredient (INCI) name, Ceramide 1, which was originally assigned in 1997, has been retired. For an interim period, trade name assignments formerly published with the INCI name Ceramide 1 will be retained in the retired monograph and also published with the new name assignment, Ceramide EOP. This means that, during the “interim period,” products on the market may be labeled with either name, Ceramide 1 or Ceramide EOP, although both names refer to the same ingredient. Reported use data are associated primarily with the retired names. Likewise, most ingredient-specific data received for these ingredients may be associated with the retired INCI names. Accordingly, throughout this safety assessment report, the retired INCI names are used consistently to refer to the ingredients listed under either nomenclature. However, the data and the conclusions of the Panel will apply to these ingredients under both the new and the retired nomenclature.
The other name changes include (further explained in Table 1): Ceramide 2 will be replaced by 2 names, Ceramide NS (limited to sphingosine-based ceramides) and Ceramide NG (limited to sphinganine-based ceramides); Ceramide 3 will be replaced by Ceramide NP; Ceramide 4 and Ceramide 5 will both be replaced by Ceramide AS; and Ceramide 6 II will be replaced by Ceramide AP.
Definitions and Idealized Structures of the Ingredients in This Safety Assessment. 1

Information on manufacturing methods for several ceramide ingredients submitted to CIR by the Council indicates that these ingredients are produced synthetically. Although some ceramides are plentiful in bovine central nervous system tissues (eg, brain and spinal cord), the US Food and Drug Administration (FDA) prohibits the use of ingredients derived from such tissues in cosmetic products because of the risk of transmitting infectious agents, such as bovine spongiform encephalitis (BSE; 21 CFR 700.27). Some ceramide ingredients may be derived from plant sources (eg, those designated as phytosphingosines in the INCI names or definitions), which do not pose the risks associated with ingredients derived from the bovine central nervous system. 5
Chemistry
Definition and Characterization
Generally, a ceramide is the amidation reaction product of a sphingoid base and a fatty acid (Figure 1).
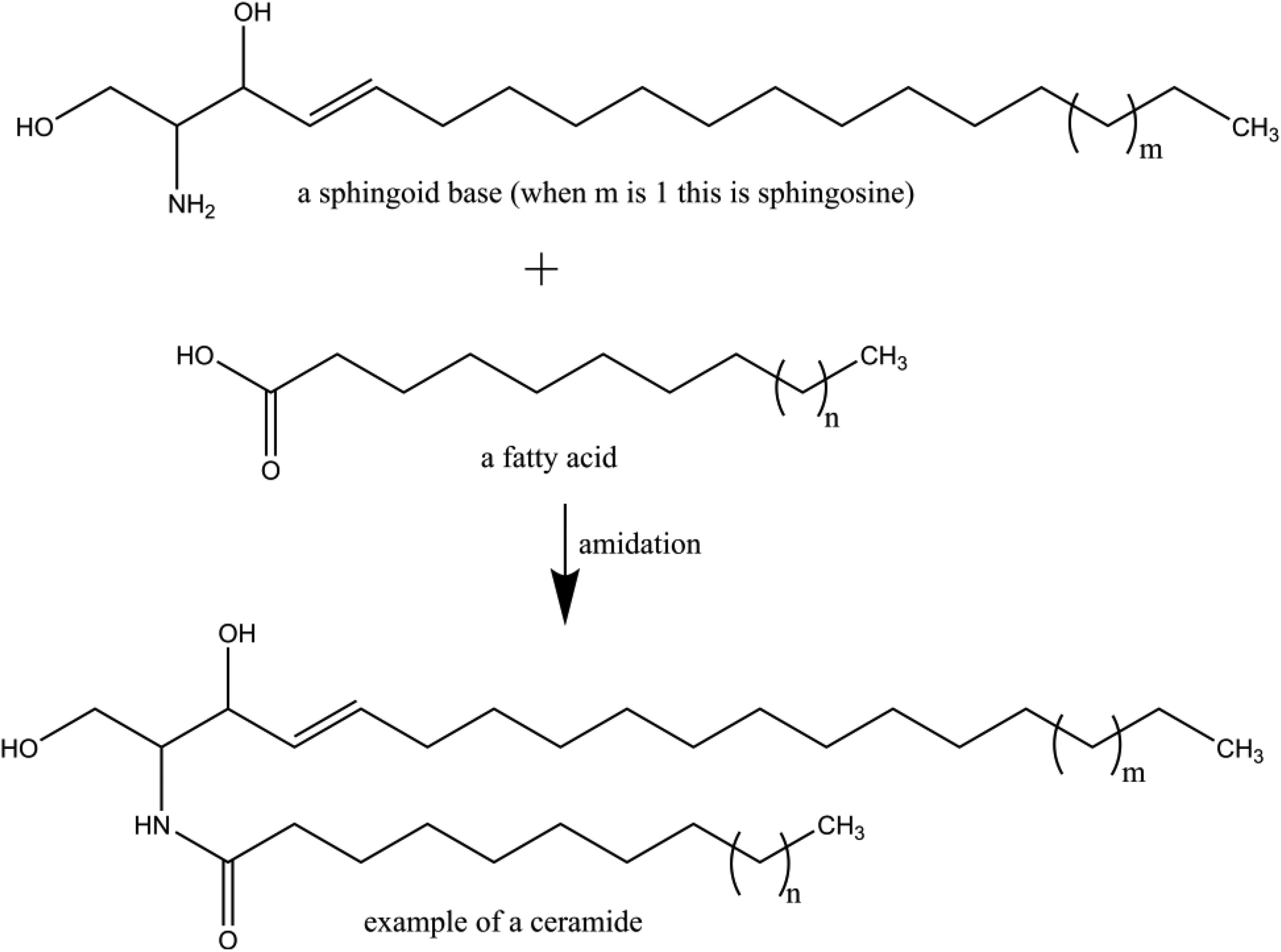
Example of a ceramide structure.
The ceramide ingredients described herein vary principally in the chain lengths of the sphingoid and fatty acid residues and in the degree of unsaturation in the chains. The ceramide ingredients are also defined as having a certain stereochemistry related to the 2 stereocenters of the sphingoid base. Specifically, all of the ceramide ingredients, like those ceramides found in human skin, are defined as
Chemical Properties of Ceramide 2.

However, some of the ingredients in this report are not traditional ceramides and the stereochemistries therein are not defined. For example, the stereochemistries of Hydroxypalmitoyl Sphinganine and 2-Oleamido-1,3-Octadecanediol are not recited in the respective monographs.
Method of Manufacturing
In biological systems, ceramides are synthesized by de novo synthesis or sphingomyelin hydrolysis or through a salvage pathway. 6 Ceramide manufacture could be accomplished by a variety of synthetic methods, but most methods involve amidation of a fatty acid with a sphingoid base. 7 This can be accomplished by reaction of the sphingoid base with an acyl chloride, but the results are not selective and esterification and amidation occur concurrently. However, mild alkaline hydrolysis can selectively remove the esters. Alternatively, activating the fatty acid with a carbodiimide enables ceramide synthesis without concurrent esterification.
The unpublished data on method of manufacturing detailed below were received from suppliers of Ceramide 2, Ceramide 5, 2-Oleamido-1,3-Octadecanediol, and Hydroxypalmitoyl Sphinganine.
Ceramide 2
Ceramide 2 is produced synthetically via amide formation (ie, reaction of [2S,3R]-sphinganine with methyl stearate to produce an amide), and other constituents/impurities are the isomers (Figure 2).
8
Formation of ceramide 2.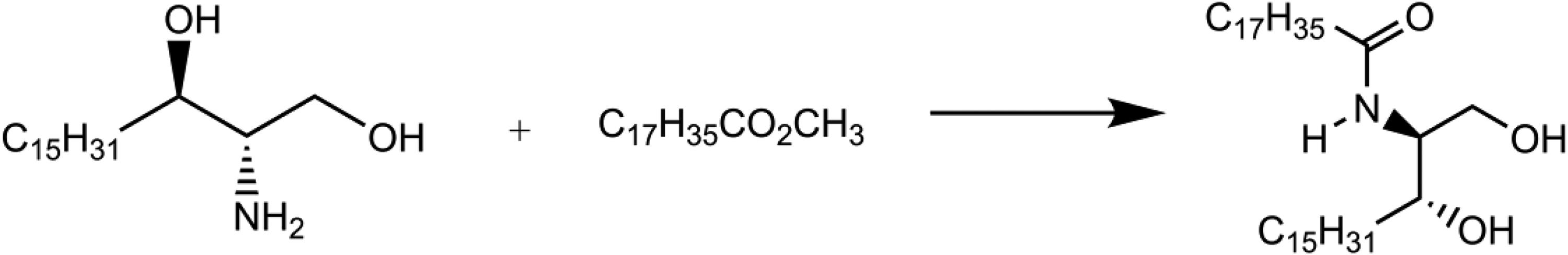
It has been reported that Ceramide 2 is a pure substance obtained by reacting a glycine ester derivative and activated palmitic acid before further reacting with stearoyl chloride.
9
Ceramide 2 is a mixture of
Ceramide 5
Ceramide 5 is produced synthetically via amide formation (ie, reaction of [2S,3R]-sphinganine) with methyl 2-hydroxyhexadecanoate to produce an amide (Figure 3).
10
Formation of ceramide 5.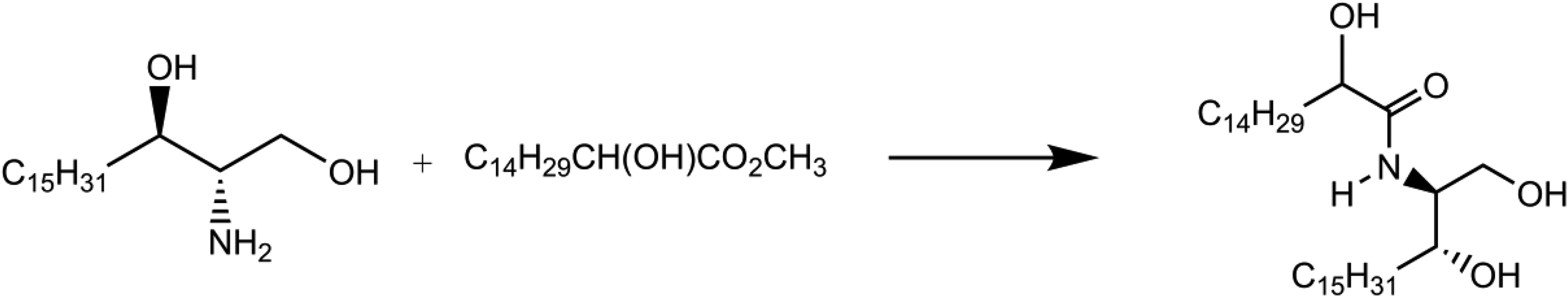
2-Oleamido-1,3-Octadecanediol
2-Oleamido-1,3-Octadecanediol is obtained by chemical reaction between dihydrosphingosine and an oleic acid derivative. 11
Hydroxypalmitoyl Sphinganine
Hydroxypalmitoyl Sphinganine is obtained by chemical reaction between dihydrosphingosine and a 2-bromohexadecanoic acid derivative, followed by the indirect substitution of bromine by hydroxyl group. 11
Impurities
Ceramide 2
In a high-performance liquid chromatography analysis of Ceramide 2, only 1 peak was detected. No residual solvent was detected and the water content was less than 0.5% (no further details provided). 9
A heavy metals analysis performed on Ceramide 2 (reported under the new name Ceramide NG) yielded the following results: lead < 10 ppm, arsenic < 3 ppm, mercury < 1 ppm, cadmium < 1 ppm, nickel < 1 ppm, and palladium < 1 ppm. 12
Ceramide 5
The residue-on-ignition value for Ceramide 5 was reported to be less than 0.5%. 10
Ultraviolet/Visible Absorption
Ceramide 2
The ultraviolet and visible absorption spectra of solutions of Ceramide 2 were measured with a Perkin Elmer Lambda 15 spectrophotometer.
13
Ceramide 2 was tested at concentrations of 0.001%, 0.01%, 0.1%, and 1% in 95% ethanol or 1,4-dioxane. Negligible absorption was observed (
Use
Cosmetic
The safety of the cosmetic ingredients included in this safety assessment is evaluated on the basis of the expected use in cosmetics. The Panel utilizes data received from the FDA and the cosmetics industry in determining the expected cosmetic use. The data received from the FDA are those it collects from manufacturers on the use of individual ingredients in cosmetics by cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP), and those from the cosmetic industry are submitted in response to a survey of the maximum reported use concentrations by category conducted by the Council.
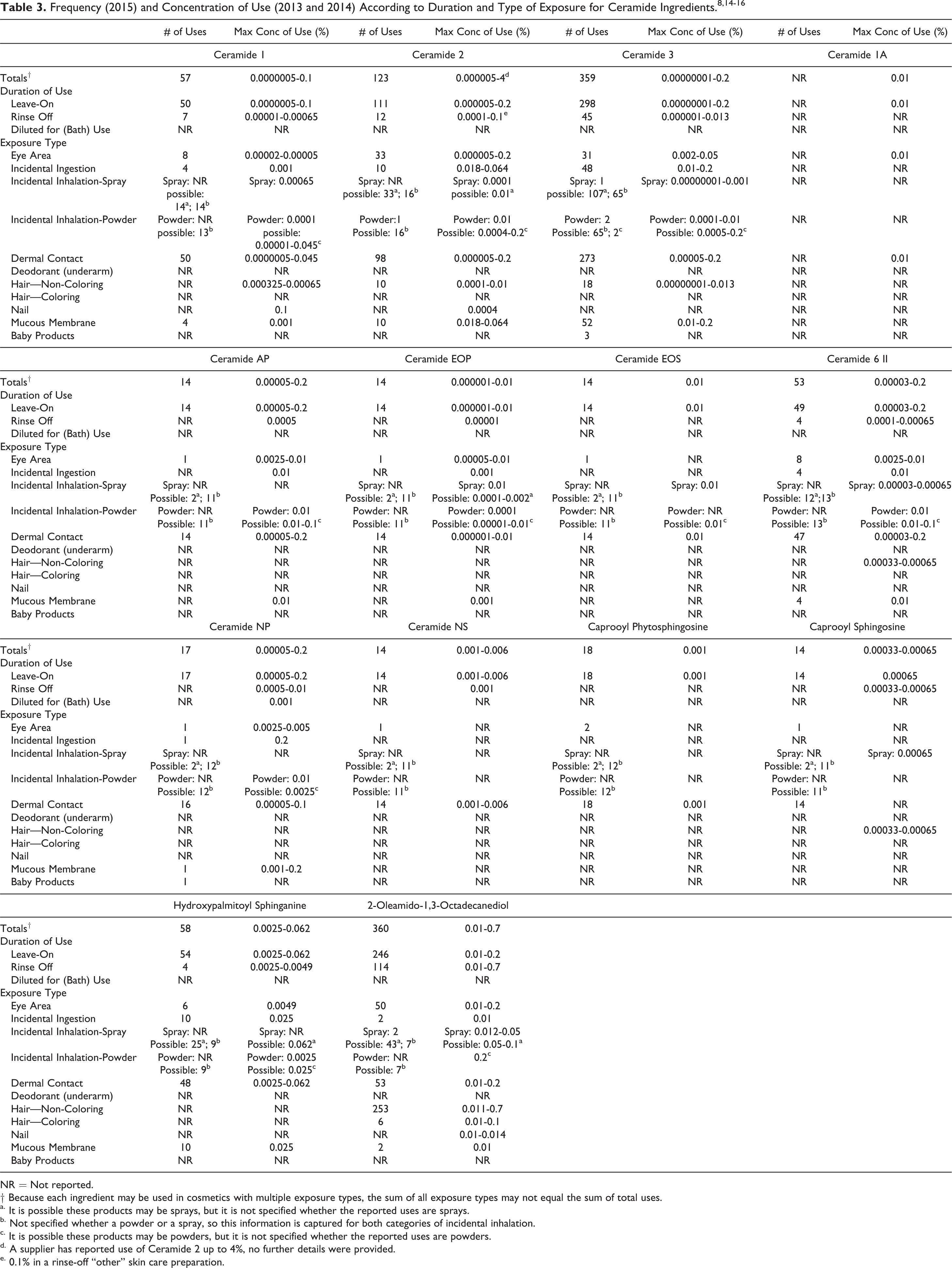
According to the 2015 VCRP survey data, 2-Oleamido-1,3-Octadecanediol is reported to be used in 360 formulations (Table 3). 14 Ceramide 3 is reported to be used in 359 formulations. The majority of the uses for all ceramide ingredients are in leave-on skin care preparations. The results of the concentration of use survey conducted by the Council in 2013 and 2014 indicate 2-Oleamido-1,3-Octadecanediol has the highest reported maximum concentration of use; it is used at up to 0.7% in hair conditioners. 15,16 The highest maximum concentration of use reported for products resulting in leave-on dermal exposure is 0.2% for Ceramide 2, Ceramide 3, Ceramide 6 II, Ceramide AP, Ceramide NP, and 2-Oleamido-1,3-Octadecanediol in skin care preparations. Suppliers reported that Ceramide 2 and Ceramide 5 are used at concentrations up to 4% and 2%, respectively, in cosmetic products (no further details were provided). 8,10 Table 4 lists the 8 ceramide ingredients not indicated to be in use based on the VCRP data and the results of the Council’s concentration of use survey.
NR = Not reported.
† Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
a. It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b. Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
c. It is possible these products may be powders, but it is not specified whether the reported uses are powders.
d. A supplier has reported use of Ceramide 2 up to 4%, no further details were provided.
e. 0.1% in a rinse-off “other” skin care preparation.
Ingredients That Are Not Reported to Be in Use in the VCRP or the Council Survey.

* A supplier has reported use of Ceramide 5 up to 2%, no further details were provided. 10
In some cases, no reported uses were received from the VCRP, but a maximum use concentration was provided in the industry survey. For example, Ceramide 1A was not reported in the VCRP, but the industry survey indicated that it is used in eye lotions at concentrations up to 0.01%. It should be presumed that Ceramide 1A is used in at least one cosmetic formulation.
Some of these ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes. For example, Ceramide 3 is used at 0.2% in lipstick and Ceramide 2 is used at 0.2% in eye lotion. Additionally, some of these ingredients were reported to be used in hair sprays and body and hand or moisturizing sprays and could possibly be inhaled. For example, Ceramide 3 was reported to be used in body and hand sprays at a maximum concentration of 0.001%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 17 –20 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 18,19 The ceramide ingredients in this report are not restricted from use in any way under the rules governing cosmetic products in the European Union. 21
Toxicokinetics
Ceramides are among the lipids that make up sphingomyelin, which is a major component of the lipid bilayer of the cell membranes of keratinocytes in the epidermis. 8,10 Ceramides are lipophilic and are likely to be readily absorbed into the skin. However, they are expected to remain in the stratum corneum and not penetrate any deeper.
The absorption, distribution, and excretion of an analogous radiolabeled ceramide ([3-
3
H]-palmitoyl
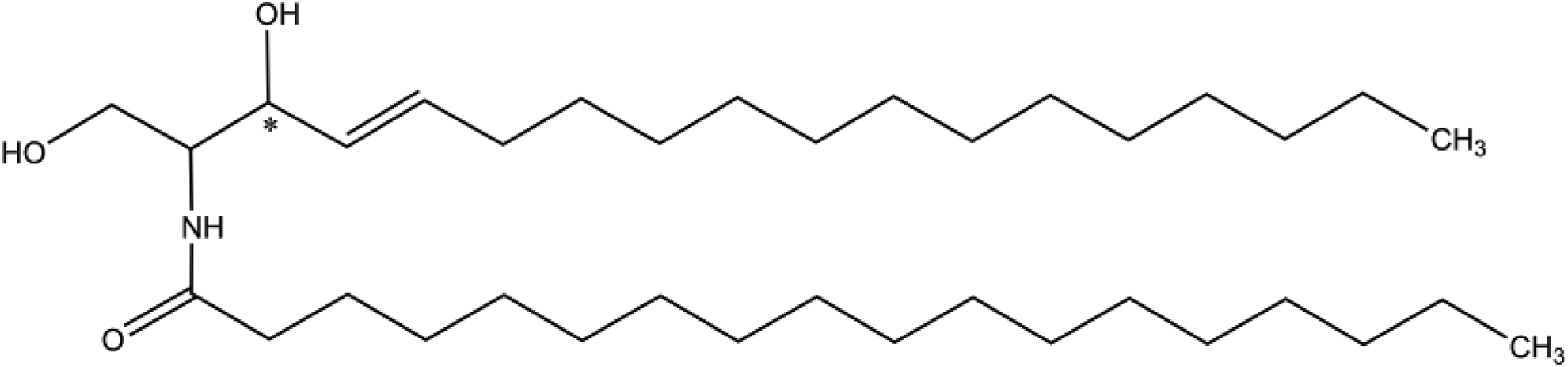
Ceramide analog.
An unreported number of rats received a single oral administration (300 kBq/30 µg/kg) of [ 3 H]-ceramide. Blood samples were serially collected from the subclavian vein at 0.5, 1, 2, 4, 8, 12, 24, 48, 72, 96, and 144 hours and the concentration of total radioactivity in the plasma was determined. Radioactivity was also measured in urine and feces collected up to 96 hours posttreatment and in excised abdominal skin and carcass. Distribution of radioactivity was measured in selected organs and tissues up to 168 hours posttreatment. The mean plasma concentration of radioactivity reached a maximum at approximately 10.67 hours and then decreased with a half-life of 67.12 hours. The mean cumulative excretion of radioactivity in urine and feces was approximately 4.79% and 87.44% of the dose, respectively. At 96 hours after dosing, 1.67% and 3.67%, respectively, of the dose were still present in the skin and carcass. The radioactivity in the skin at 12 hours was lower than that in plasma and the ratio of skin to plasma concentration was 0.7. However, at 120 hours after dosing, the ratio of skin to plasma concentration increased to 4. An analysis of the distribution of radioactivity in a section of skin found radioactivity in the dermis and epidermis. At 72 and 168 hours, the radioactivity in the epidermis was 5.6% and 8.0%, respectively, of the radioactivity in skin, while at these same observation periods, the radioactivity in the dermis was 94.4% and 92.0%, respectively, of the radioactivity in skin. This study found that, following oral exposure, radiolabeled ceramide is distributed gradually in the dermis and then transferred to the epidermis.
Toxicological Studies
Acute Toxicity
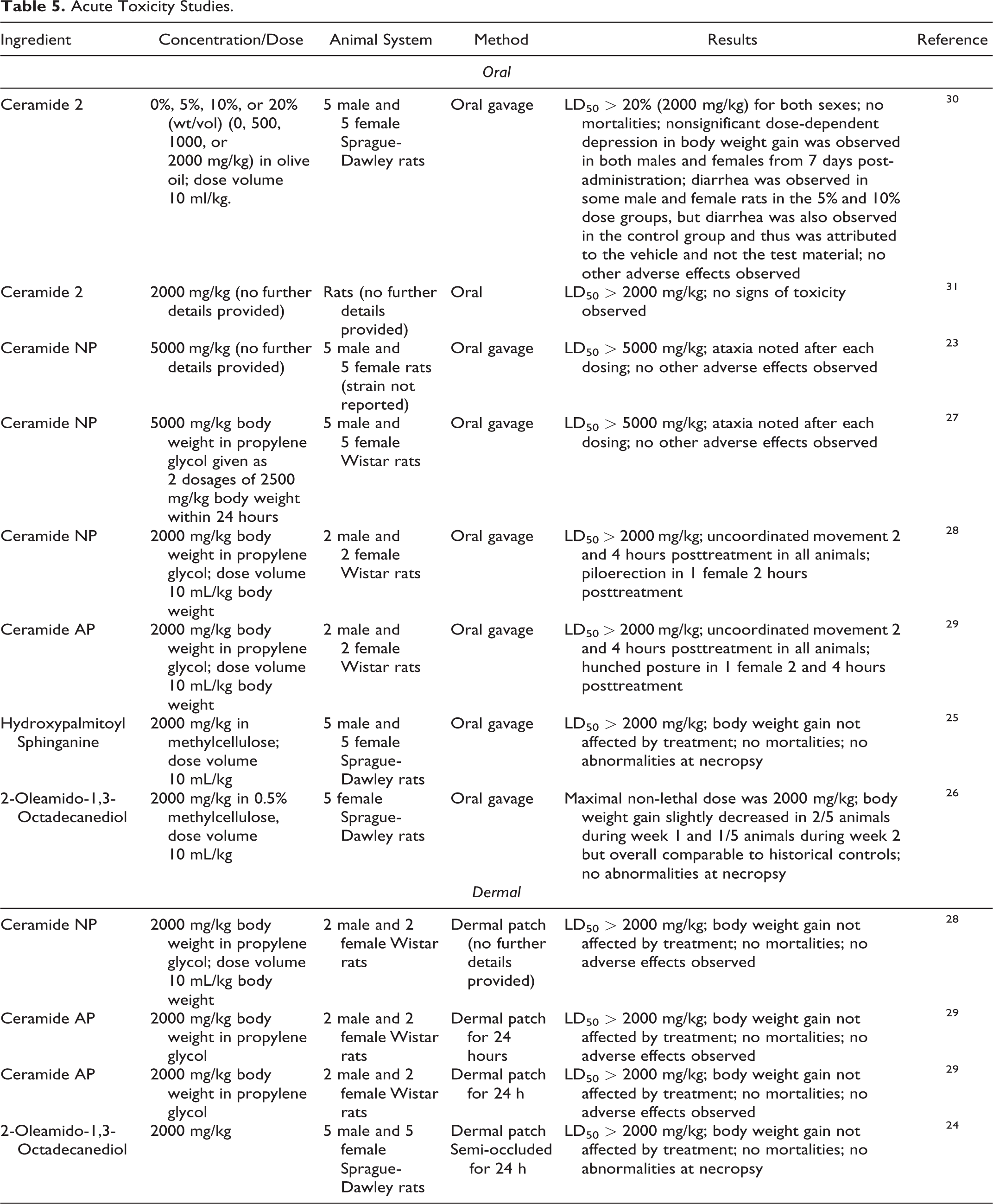
Acute oral and dermal toxicity studies are summarized in Table 5. 23 –31 The median lethal oral dose (LD50) was greater than 2000 mg/kg in rat studies of Ceramide 2, Ceramide AP, Hydroxypalmitoyl Sphinganine, and 2 Oleamido-1,3-Octadecanediol, and greater than 5000 mg/kg in rat studies of Ceramide NP. In dermal studies on rats, the LD50 s were greater than 2000 mg/kg for Ceramide NP, Ceramide AP, and 2-Oleamido-1,3-Octadecanediol.
Acute Toxicity Studies.
Repeated Dose Toxicity
Oral—Non-Human
2-Oleamido-1,3-Octadecanediol
The toxicity of 2-Oleamido-1,3-Octadecanediol was assessed in a 28-day oral study in groups of 5 male and 5 female Sprague-Dawley rats. 32 The rats received 10, 30, or 100 mg/kg of the test material in carboxymethylcellulose daily via gavage. An additional group of 5 males and 5 females received the vehicle alone as a control. The rats were observed weekly for clinical signs of toxicity and feed and water consumption. The rats were weighed twice weekly. Hematology, blood clinical chemistry, and urinalysis were also performed. Macroscopic and histologic examinations were performed at study end.
No mortalities were observed during the study. With the exception of the 30 mg/kg dose females, behavior, body weight gain, and feed consumption of the treated animals were comparable to those of the control animals. In the 30 mg/kg females, a slight decrease in body weight gain was observed. Decreased water consumption was observed during week 4 in the 10 mg/kg dose males and during week 2 to week 4 in the 30 mg/kg dose males. In the females, increased water consumption was observed in week 2 and week 3 in the 10 mg/kg dose group and during week 1 and week 2 in the 30 and 100 mg/kg dose groups. Mean white cell count in the 10 mg/kg dose females, mean neutrophils count in the 10 and 100 mg/kg dose males, and mean lymphocyte count in the 10 mg/kg dose females were statistically significantly lower than those of the controls, but individual results were within physiological ranges. Alanine transaminase and aspartate transaminase activities were high in one 100 mg/kg dose female, and a histopathological examination of the liver of this animal revealed moderate single cell necrosis with inflammatory cell infiltration. The mean urea level in the blood in the 30 mg/kg dose females was significantly lower than that of the controls, but individual results were within physiological ranges. No other statistically significant differences were observed in other hematologic and clinical chemistry parameters. Macroscopic examination found mean absolute weight of the heart in the 30 mg/kg dose males and mean relative weight of the heart in the 30 and 100 mg/kg dose males statistically significantly lower than those of the controls, but individual results were within normal physiological ranges. At 100 mg/kg in male rats, a statistically significant increase in mean absolute and relative weight of the thymus was observed. Based on the findings in the thymus, the no observed effect level (NOEL) for 2-Oleamido-1,3-Octadecanediol was 30 mg/kg/d in rats. 32
Dermal—Nonhuman
2-Oleamido-1,3-Octadecanediol
The cutaneous toxicity of 2-Oleamido-1,3-Octadecanediol was tested in 3 male and 3 female Sprague-Dawley rats. 33 The animals received 1 g/kg body weight of test material in powder form applied to 40 cm2 of the costal cutaneous area once daily for 14 consecutive days. Test sites were occluded for 6 hours and then washed. An additional group of 3 male and 3 female rats that did not receive the powder served as control. The rats were observed daily for clinical signs of toxicity, mortality, and cutaneous reactions. The rats were weighed on days 7 and 14. Macroscopic and histologic examinations were performed at study end. No mortalities were observed during the study. Behavior and mean body weight gains of the treated animals were comparable to those of the control animals. No adverse reactions were observed in the skin. No macroscopic abnormalities of the skin or main abdominal and thoracic organs were observed. No treatment-related skin irritation was noted in the histological evaluation. It was concluded that 1 g/kg 2-Oleamido-1,3-Octadecanediol did not cause cutaneous toxicity in rats.
Reproductive and Developmental Toxicity
2-Oleamido-1,3-Octadecanediol
The effects of 2-Oleamido-1,3-Octadecanediol on reproduction and development were studied in groups of 10 rats/sex/dose by oral gavage. 34 Dose levels tested were 0, 100, 300, and 1000 mg/kg body weight/d at a dose volume of 5 mL/kg body weight. The vehicle was a 0.5% aqueous solution of methylcellulose in purified water. Parental males were exposed to the test material 2 weeks prior to mating, during mating, and about 2 weeks postmating (approximately 6 weeks total). Parental females were exposed 2 weeks prior to mating, during mating, during gestation, and during at least 4 days of lactation.
In the 1000 mg/kg dose group, one pregnant rat was observed with poor clinical condition and body weight loss toward the end of gestation and did not deliver by day 24 postmating. Fibrinous and necrotic inflammation of the pericardium was observed microscopically. Another female in the high-dose group was also observed with poor clinical condition from the end of gestation until day 2 postpartum. A normal delivery was observed in this rat, though, and no abnormal findings were observed at necropsy. The findings in these 2 high-dose females were not considered treatment-related. No treatment-related clinical effects were noted in the other animals in any dose group. No treatment-related effects were observed with mean body weights, mean feed consumption, or mating or fertility parameters (including mean numbers of corpora lutea, implantation sites per litter, pups delivered, and live pups).
Further, no increased incidences of mortality, adverse effects on body weight, influence on sex ratio, or treatment-related gross malformations were noted in the pups. In the 1000 mg/kg/d dose group, one parental male had several findings in the urinary tract, including urinary lithiasis, thickened and red urinary bladder mucosa, and dilated right ureter and pelvic cavity of the right kidney. The researchers could not determine whether these findings were treatment-related or a random event. No other treatment-related effects were noted in parental animals at necropsy.
Based on the results of this study on 2-Oleamido-1,3-Octadecanediol, the researchers determined the maternal no observed adverse effect level (NOAEL) to be 1000 mg/kg body weight/d, and the NOEL for mating and fertility to be 1000 mg/kg body weight/d. 34
Genotoxicity
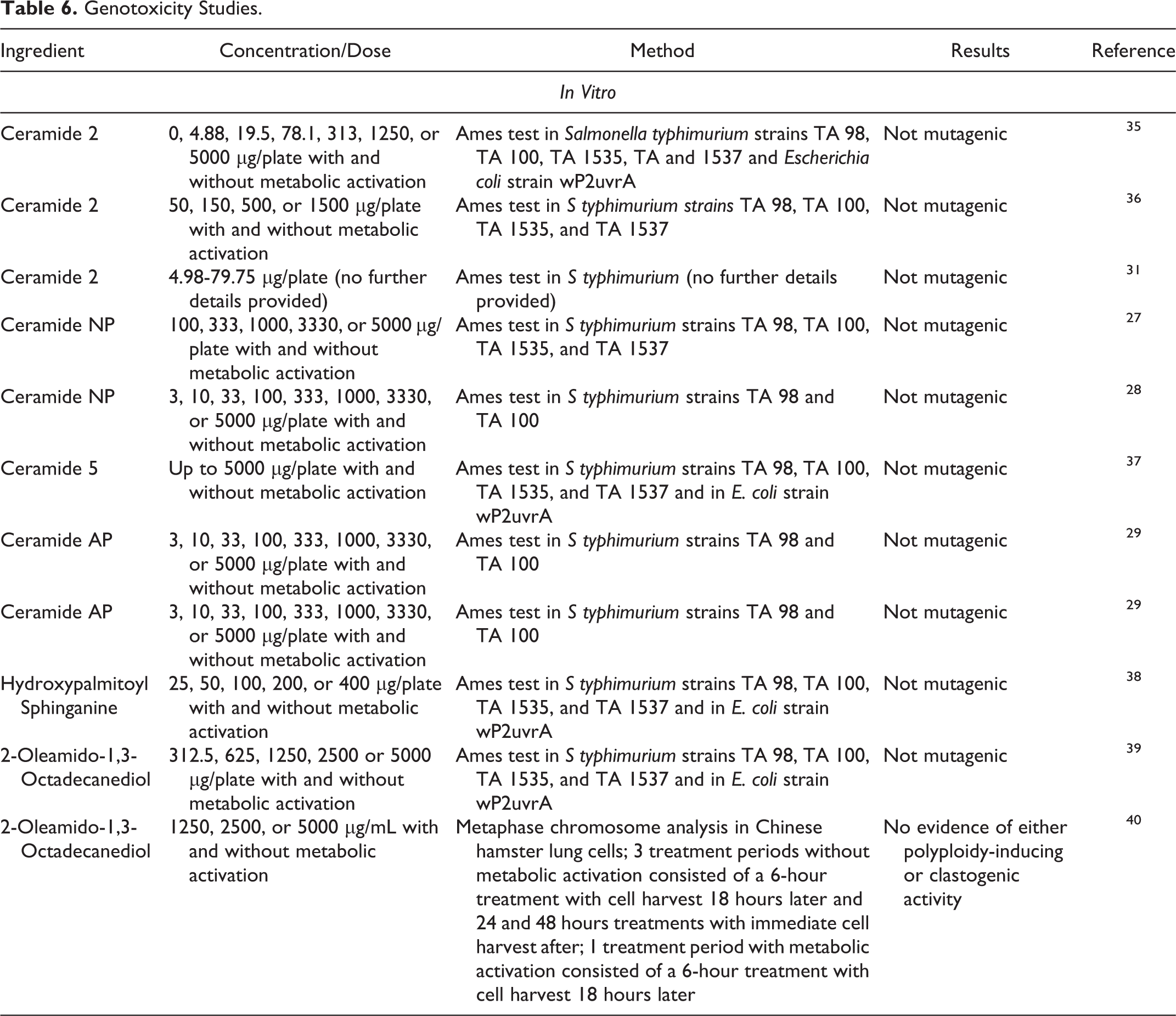
In vitro genotoxicity studies are summarized in Table 6. 27 –29,31,35 –40 Ceramide 2, Ceramide NP, Ceramide 5, Ceramide AP, Hydroxypalmitoyl Sphinganine, and 2-Oleamido-1,3-Octadecanediol were not mutagenic in Ames assays. Additionally, 2-Oleamido-1,3-Octadecanediol was not polyploidy-inducing or clastogenic in a metaphase chromosome analysis in Chinese hamster lung cells at a concentration up to 5000 μg/mL with and without metabolic activation.
Genotoxicity Studies.
Carcinogenicity
Data on carcinogenicity were not found in the published literature for ceramides nor were unpublished data provided.
Irritation and Sensitization
Irritation
Ocular
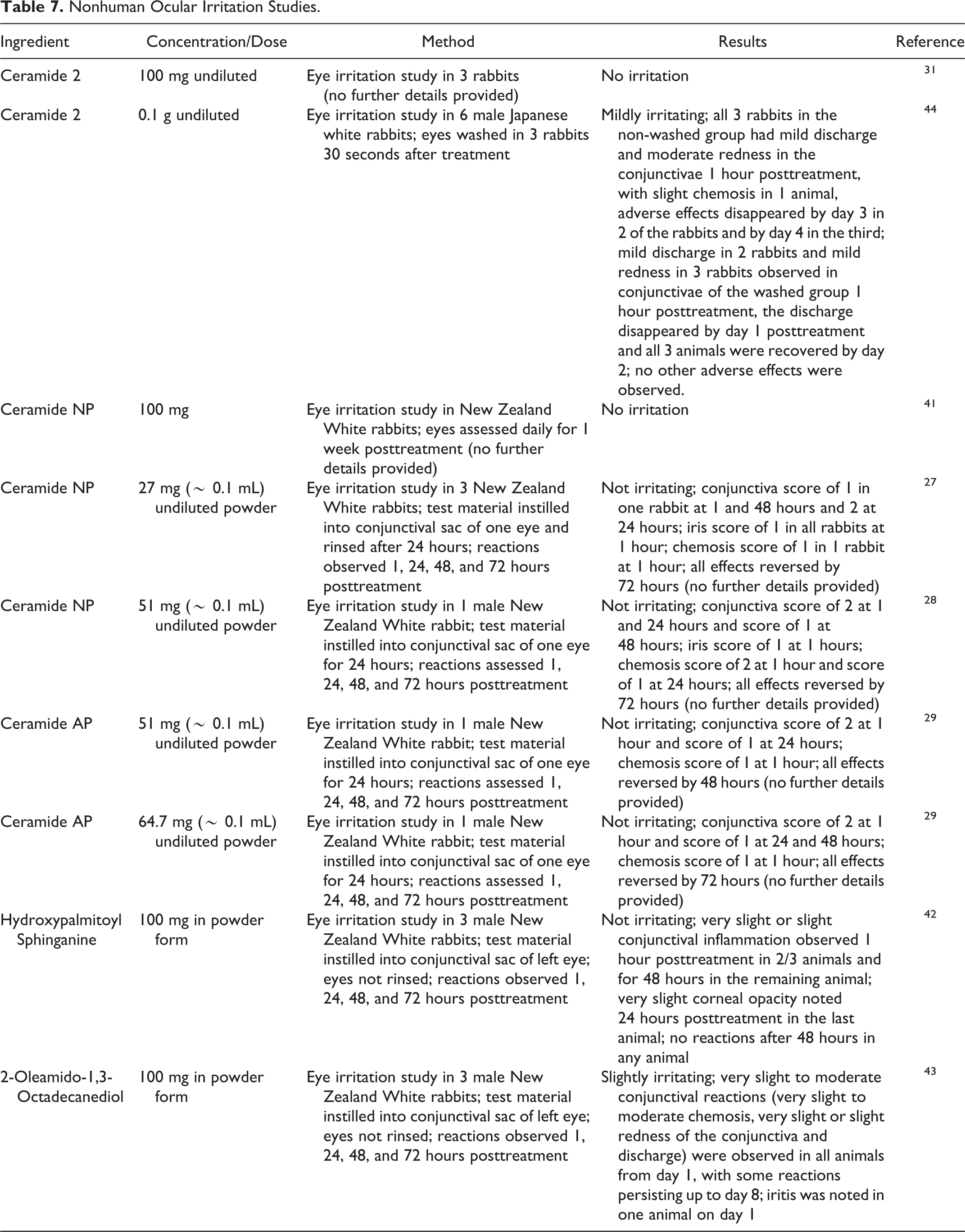
Ocular irritation studies are summarized in Table 7. 27 –29,31,41 –44 Undiluted Ceramide 2 was mildly irritating to rabbit eyes in one study but not irritating in another. Ceramide NP, Ceramide AP, and Hydroxypalmitoyl Sphinganine were also not irritating in rabbit eye studies when tested neat. 2-Oleamido-1,3-octadecanediol was slightly irritating when tested neat in rabbit eyes.
Nonhuman Ocular Irritation Studies.
Dermal
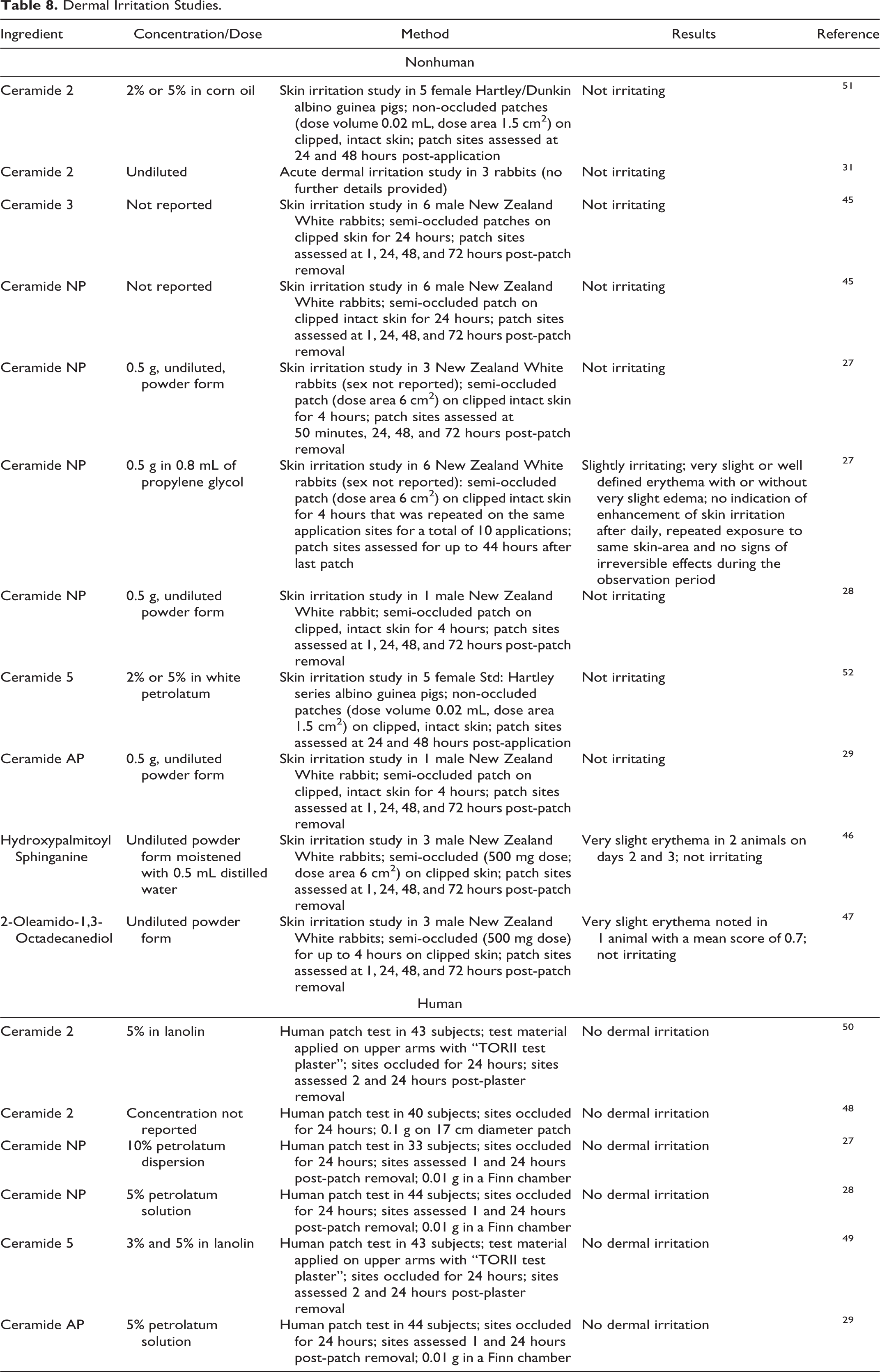
Dermal irritation studies are summarized in Table 8. 27 –29,31,45 –52 In dermal studies, Ceramide 2 (up to 5% in corn oil and undiluted), Ceramide 3 (concentration not reported), Ceramide 5 (up to 5% in white petrolatum), Ceramide AP (undiluted), Hydroxypalmitoyl Sphinganine (undiluted), and 2-Oleamido-1,3-Octadecanediol (undiluted) were not irritating to rabbit or guinea pig skin. Slight irritation was observed in a dermal irritation study in rabbits with Ceramide NP in propylene glycol, but no irritation was observed in other studies of the undiluted form. No dermal irritation was observed in human patch tests of Ceramide 2 (up to 5% in lanolin), Ceramide NP (up to 10% dispersion in petrolatum), Ceramide 5 (up to 5% in lanolin), and Ceramide AP (5% dispersion in petrolatum).
Dermal Irritation Studies.
Sensitization
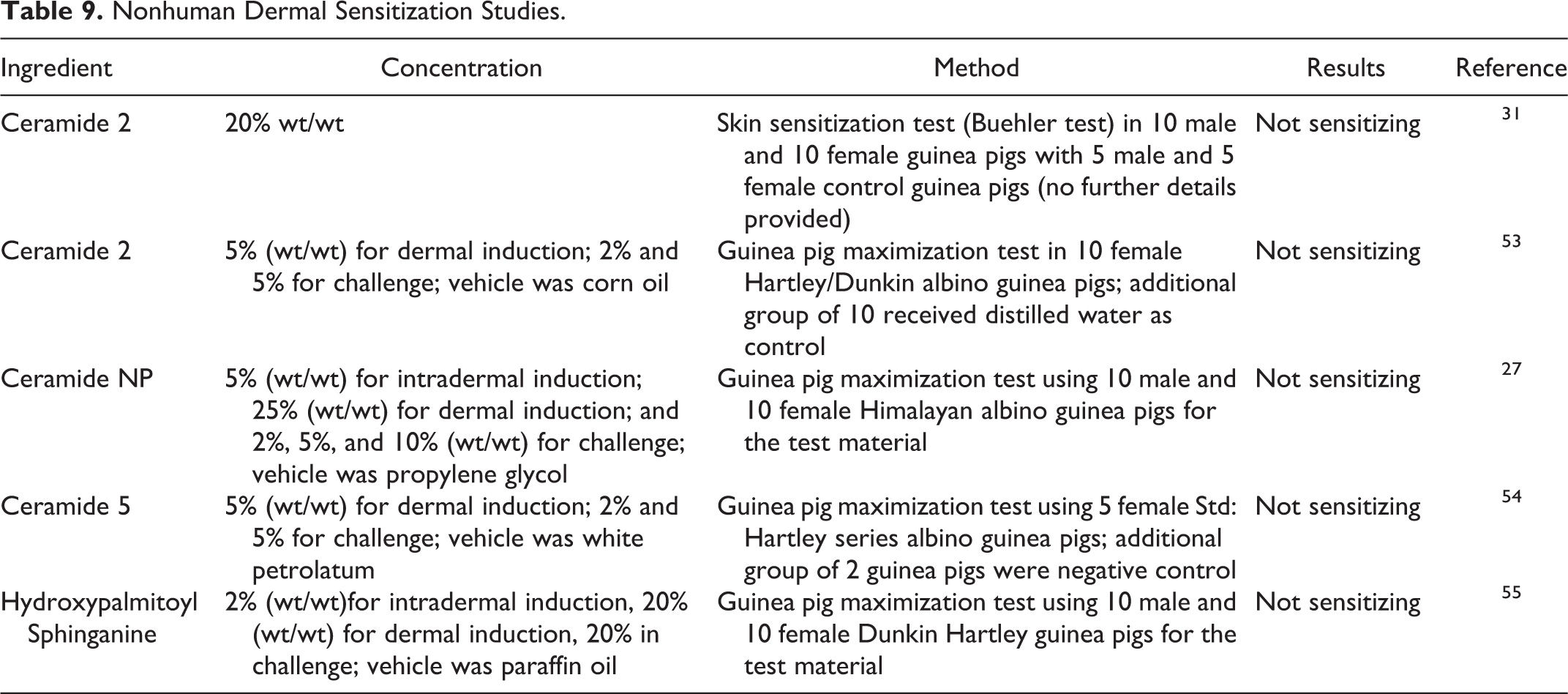
Dermal sensitization studies are summarized in Table 9. 27,31,53 –55 Ceramide 2 (up to 20%), Ceramide NP (up to 10%), Ceramide 5 (up to 5%), and Hydroxypalmitoyl Sphinganine (up to 20%) were not sensitizing in guinea pig maximization tests.
Nonhuman Dermal Sensitization Studies.
Summary
Ceramides function primarily as hair conditioning agents and skin-conditioning agents—miscellaneous in personal care products. Naturally occurring ceramides are normal constituents of the skin and are essential components of the epidermal permeability barrier. The names of ceramide ingredients changed during the development of this safety assessment. Reported use data are associated primarily with the retired names. Likewise, most ingredient-specific data received for these ingredients may be associated with the retired INCI names. Accordingly, the retired INCI names are used consistently to refer to the ingredients listed under either nomenclature. However, the data and the conclusions of the Panel will apply to these ingredients under both the new and the retired nomenclature.
2-Oleamido-1,3-Octadecanediol has the most reported uses in cosmetics, with a total of 360; the majority of the uses are in leave-on skin care preparations. Ceramide 3 has the second greatest number of overall uses reported, with a total of 359; the majority of those uses are also in leave-on skin care preparations. In the Council’s use-concentration survey, 2-Oleamido-1,3-Octadecanediol had a maximum use concentration of 0.7% reported in hair conditioners. The highest maximum concentration of use reported for products resulting in leave-on dermal exposure is 0.2% for Ceramide 2, Ceramide 3, Ceramide 6 II, Ceramide AP, Ceramide NP, and 2-Oleamido-1,3-Octadecanediol in skin care preparations.
Ceramides are among the lipids that make up sphingomyelin, which is a major component of the lipid bilayer that forms cell membranes of cells in the stratum corneum. Thus, ceramides are lipophilic and likely to be absorbed into the skin. However, they are expected to remain in the stratum corneum and not penetrate any deeper. An absorption, distribution and excretion study of an analogous radiolabeled ceramide in male rats found that, following oral exposure, the ceramide was distributed gradually to the dermis and then transferred to the epidermis.
The oral LD50 was greater than 2000 mg/kg in rat studies of Ceramide 2, Ceramide AP, Hydroxypalmitoyl Sphinganine, and 2-Oleamido-1,3-Octadecanediol, and greater than 5000 mg/kg in rat studies of Ceramide NP. In dermal studies, the LD50 was greater than 2000 mg/kg in rats exposed to Ceramide NP, Ceramide AP, or 2 Oleamido-1,3-Octadecanediol.
In an oral repeated dose toxicity study, the NOEL for 2-Oleamido-1,3-Octadecanediol was 30 mg/kg/d in rats. At 100 mg/kg in male rats, a statistically significant increase in mean absolute and relative weights of the thymus was observed. In a dermal repeated dose toxicity study, the cutaneous tolerance of 1 g/kg 2-Oleamido-1,3-Octadecanediol in rats was good. No skin reactions or systemic effects were observed,
The maternal NOAEL and mating and fertility NOEL were both 1000 mg/kg body weight/d, respectively, in a rat reproduction and developmental study of 2-Oleamido-1,3-Octadecanediol. In the1000 mg/kg/d dose group, one male had several lesions in the urinary tract, including urinary lithiasis, thickened and red urinary bladder mucosa, and dilated right ureter and pelvic cavity of the right kidney. The researchers could not determine whether this finding was treatment-related or a random event. No other treatment-related effects were noted in parental animals. No increased incidences of mortality, adverse effects on body weight, influence on sex ratio, or treatment-related gross malformations were noted in the pups.
Ceramide 2, Ceramide NP, Ceramide 5, Ceramide AP, Hydroxypalmitoyl Sphinganine, and 2-Oleamido-1,3-Octadecanediol were not mutagenic in Ames assays. Additionally, 2-Oleamido-1,3-Octadecanediol was not polyploidy-inducing or clastogenic in a metaphase chromosome analysis in Chinese hamster lung cells at concentrations up to 5000 μg/mL with and without metabolic activation.
Data on carcinogenicity were not found for ceramides in the published literature. Unpublished data were not provided. Undiluted Ceramide 2 was mildly irritating to rabbit eyes in one study, but not irritating in another. Ceramide NP, Ceramide AP, and Hydroxypalmitoyl Sphinganine were also not irritating in rabbit eye studies when tested neat. 2 Oleamido-1,3-octadecanediol was slightly irritating when tested neat in rabbit eyes. In dermal studies, Ceramide 2 (up to 5% in corn oil and undiluted), Ceramide 3 (concentration not reported), Ceramide 5 (up to 5% in white petrolatum), Ceramide AP (undiluted), Hydroxypalmitoyl Sphinganine (undiluted), and 2-Oleamido-1,3-Octadecanediol (undiluted) were not irritating to rabbit or guinea pig skin. Slight irritation was observed in a dermal irritation study in rabbits with Ceramide NP in propylene glycol, but no irritation was observed in other dermal studies of the undiluted ingredient. No dermal irritation was observed in human patch tests of Ceramide 2 (up to 5% in lanolin), Ceramide NP (up to 10% petrolatum dispersion), Ceramide 5 (up to 5% in lanolin), and Ceramide AP (5% petrolatum dispersion). Ceramide 2 (up to 20%), Ceramide NP (up to 10%), Ceramide 5 (up to 5%), and Hydroxypalmitoyl Sphinganine (up to 20%) were not sensitizing in guinea pig maximization tests.
Discussion
The Panel considered the available data on ceramides and noted that there were no substantive data on reproductive and developmental toxicity or carcinogenicity. However, concerns were mitigated in light of the negative results of a reproductive and developmental toxicity study of 2-Oleamido-1,3-Octadecanediol in rats and of in vitro genotoxicity assays as well as the findings of no systemic toxicity at high doses in single and repeated oral dose animal studies, little to no irritation in ocular and dermal animal studies, no dermal irritation in human studies, and no dermal sensitization in multiple animal studies. The Panel noted that ceramides with structures that were identical or very similar to the structures of these cosmetic ingredients, exist naturally in the stratum corneum, and commented that the ceramides that are cosmetic ingredients would not be readily absorbed through the skin.
The Panel discussed the issue of incidental inhalation exposure from hair sprays and body and hand or moisturizing sprays. There were no inhalation toxicity data available. The Panel considered pertinent data indicating that incidental inhalation exposures to ceramides in such cosmetic products would not cause adverse health effects, including data characterizing the potential for ceramides to cause systemic toxicity, ocular or dermal irritation or sensitization, and other effects. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that the following ceramide ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment: Ceramide 1 [retired] Ceramide 2 [retired] Ceramide 3 [retired] Ceramide 4* [retired] Ceramide 5 [retired] Ceramide 1A [retired] Ceramide 6 II [retired] Ceramide AP Ceramide EOP Ceramide EOS Ceramide NP Ceramide NG* Ceramide NS Ceramide AS Ceramide NS dilaurate* Caprooyl Phytosphingosine Caprooyl Sphingosine Hydroxypalmitoyl Sphinganine 2-Oleamido-1,3-Octadecanediol Caproyl Sphingosine* Hydroxylauroyl Phytosphingosine* Hydroxycapryloyl Phytosphingosine* Hydroxycaproyl Phytosphingosine*
*Not reported to be in current use. Where ingredients in this group not in current use to be used in the future, the expectation is that those would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Burnett contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Boyer contributed to analysis and interpretation. Bergfeld, Belsito, Hill, Klaassen, Liebler, Marks, Shank, Slaga, and Snyder contributed to conception and design; analysis and interpretation; and critically revised manuscript. Gill contributed to analysis and interpretation and critically revised manuscript. Heldreth contributed to conception and design; analysis and interpretation; and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.