Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 6 trimellitic anhydride copolymers as used in cosmetics. These ingredients are related as copolymers in that they all share trimellitic anhydride (ie, 1,2,4-benzenetricarboxylic acid anhydride) as a monomer, are reported to function as film formers in cosmetics, and are reported to be primarily used in nail products. Very limited safety data were available or submitted. The Panel concluded that Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer and Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer are safe in nail product formulations in the present practices of use and concentration, but the data are insufficient to make a determination of safety on the use of these 2 ingredients in all other types of cosmetic formulations. The Panel also concluded that the available data are insufficient to make a determination that the remaining trimellitic anhydride copolymers are safe for use in cosmetic formulations.
Introduction
This assessment pertains to the safety of the following 6 trimellitic anhydride copolymers as used in cosmetic formulations: Adipic Acid/CHDM/MA/Neopentyl Glycol/Trimellitic Anhydride Copolymer Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer Isostearoyl Trimellitic Anhydride/Trimethylolpropane Copolymer Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer Propylene Glycol/Sebacic Acid/Trimellitic Anhydride Copolymer Trimethylpentanediol/Isophthalic Acid/Trimellitic Anhydride Copolymer
According to the International Cosmetic Ingredient Dictionary and Handbook (Dictionary), most of the trimellitic anhydride copolymers are reported to function as film formers in cosmetic formulations 1 (Table 1).
Definitions and Reported Functions 1, CIR Staff .
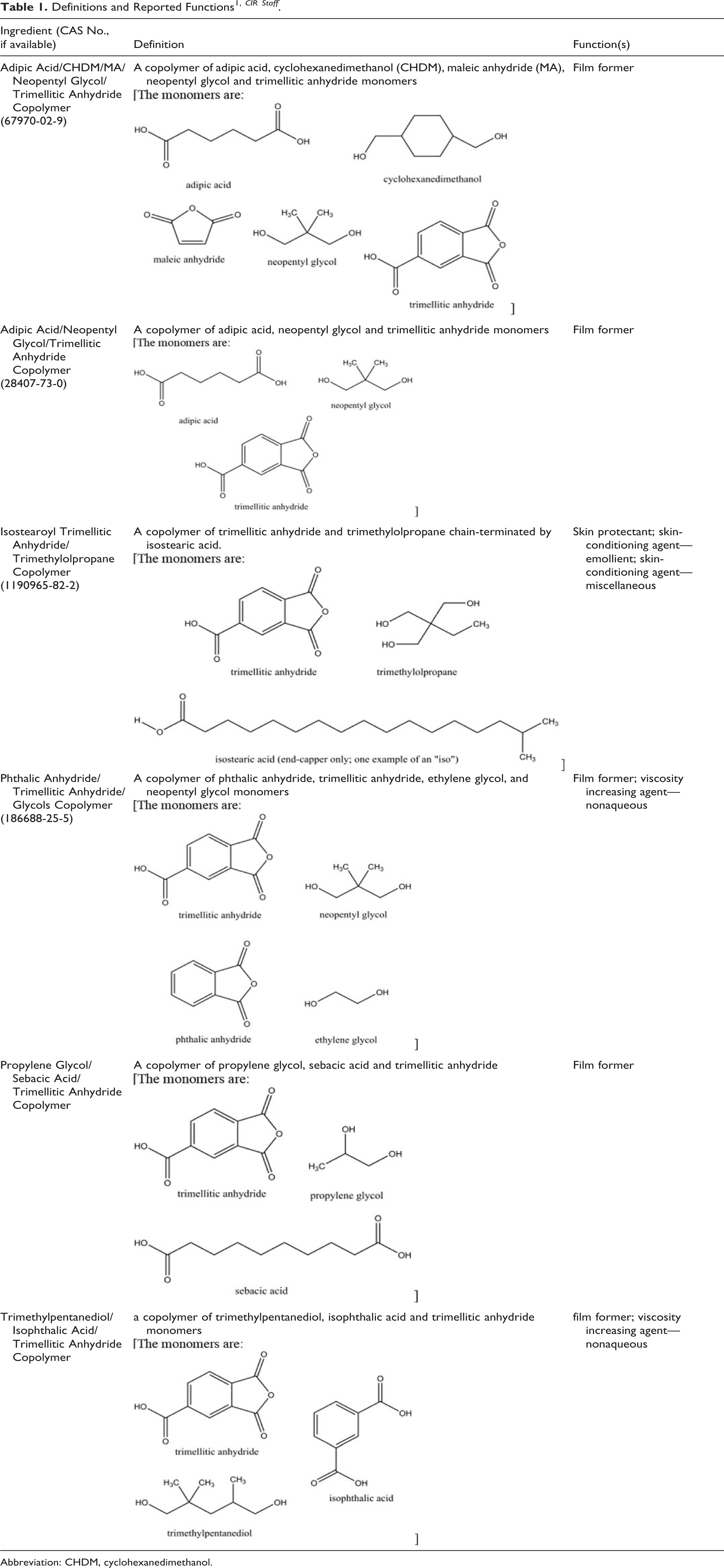
Abbreviation: CHDM, cyclohexanedimethanol.
The ingredients in this report are related as copolymers in that they all share trimellitic anhydride (ie, 1,2,4-benzenetricarboxylic acid anhydride) as a monomer. Each copolymer is also composed of 1 to 4 of the following additional monomers: adipic acid, cyclohexanedimethanol (CHDM), ethylene glycol, isophthalic acid, maleic anhydride, neopentyl glycol, phthalic anhydride, propylene glycol, sebacic acid, trimethylpentanediol, or trimethylolpropane (chain-terminated by isostearic acid). 1 Relevant information on the toxicity of these monomers is presented in Table 2. 2 –31 This information is not intended to be exhaustive or complete, but purely summary information intended to provide insight as to any possible toxicity concerns for these monomers.
Monomer Safety Data.

Abbreviations: DMSO – dimethyl sulfoxide; EPA – Environmental Protection Agency; CHO – Chinese hamster ovary; GPMT – guinea pig maximization test; HGPRT - hypoxanthine-guanine phosphoribosyltransferase; LLNA – local lymph node assay; LOAEL – lowest observable adverse effect level; NIOSH – National Institute for Occupational Safety and Health; NOAEL – no-observable adverse effect level; OECD – Organisation for Economic Co-operation and Development; OEHHA - Office of Environmental Health Hazard Assessment (California); OET – open epicutaneous test; NIOSH REL – recommended exposure limit; OEHHA REL – reference exposure level; RfC – reference concentration; RfD – reference dose; RIPT – repeated insult patch test; SCE – sister chromatid exchange; SIOPT – single insult occlusive patch test; TMA – trimellitic anhydride; TWA – time-weighted average; VCRP – Voluntary Cosmetic Registration Program.
The safety of several of the non-anhydride monomers has previously been reviewed by the Expert Panel for Cosmetic Ingredient Safety (Panel). In 2012, the Panel concluded that adipic acid and sebacic acid are safe in the present practices of use and concentration 8 and that propylene glycol is safe as used in cosmetic formulations at the present practices of use and concentration when formulated to be nonirritating. 26 In 1999, the Panel published a special report on the reproductive and developmental toxicity of ethylene glycol and its ethers, concluding that the metabolites of ethylene glycol monoalkyl ethers are reproductive and developmental toxins, but in general, these metabolites of concern are not expected to be present in cosmetic formulations that contain polymers of ethylene glycol. 11
Chemistry
Definition and Structure
The ingredients in this report are copolymers that share in common trimellitic anhydride as a monomer. They are interconnected via ester bonds to form highly branched polymeric (polyester) networks. For example, Propylene Glycol/Sebacic Acid/Trimellitic Anhydride Copolymer is the result of the polymerization of propylene glycol, sebacic acid, and trimellitic anhydride (Figure 1). In this case, there are 2 acid monomers (sebacic acid and trimellitic anhydride) and one alcohol monomer (propylene glycol). This means that to form a polyester copolymer, every other repeat unit must be propylene glycol. Whether the repeat unit on either end of propylene glycol is the residue of sebacic acid or the residue of trimellitic acid is dependent on the polymerization conditions. Since trimellitic anhydride serves as a rigid, nonlinear, trifunctional (trivalent) monomer, these polymers are branched, if not highly branched, in a manner similar to dendrimers. The definitions and structures of the ingredients included in this review are provided in Table 1.
Propylene Glycol/Sebacic Acid/Trimellitic Anhydride Copolymer—the connectivity between alcohols (propylene glycol, in this case) and which acids (sebacic acid and trimellitic anhydride, in this case) can vary; this is just one theoretical example of a fragment within a polymeric network.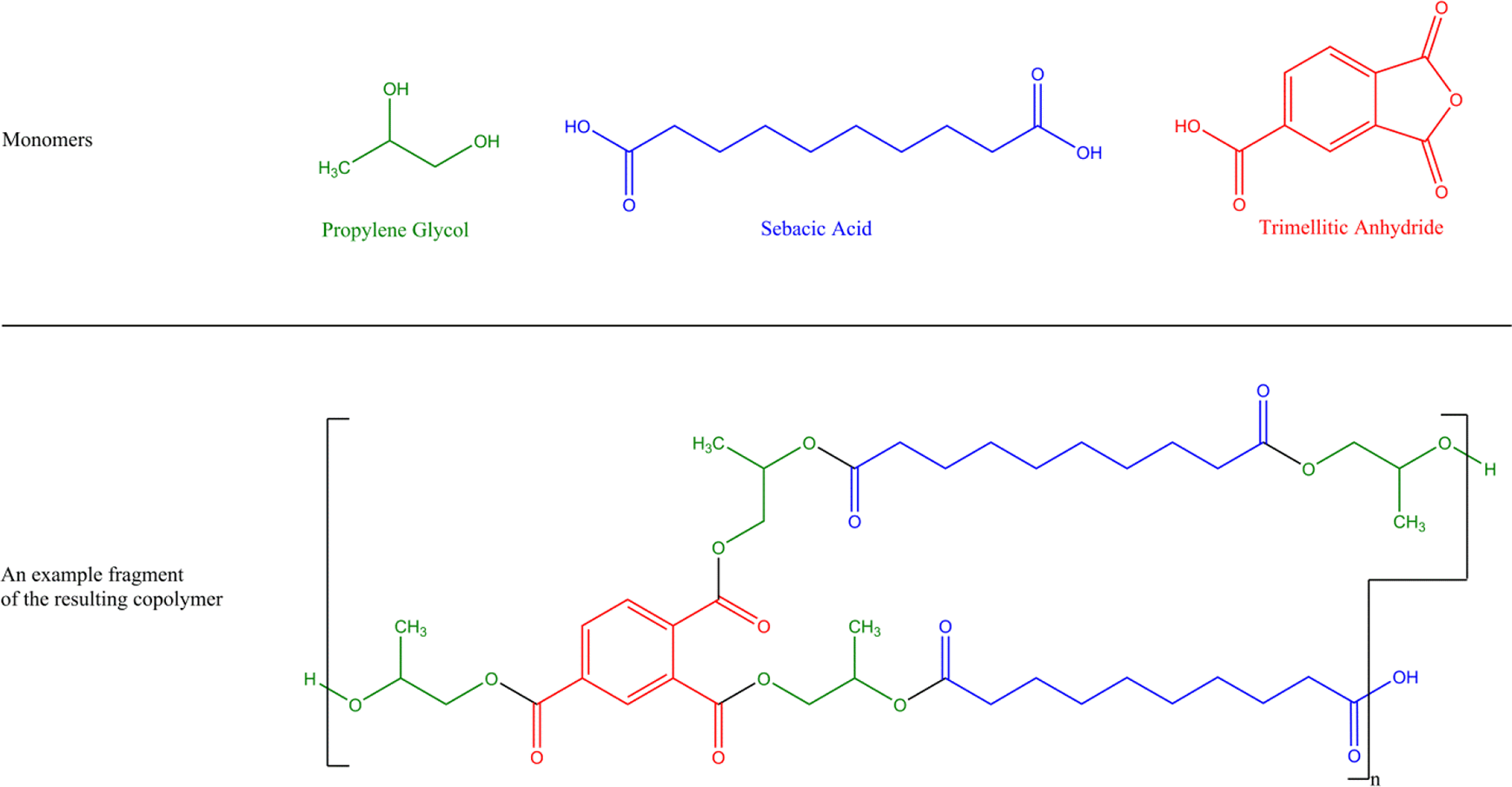
Chemical Properties
Limited chemical properties data on the trimellitic anhydride copolymers as cosmetic ingredients were found in the published literature, and unpublished data were not submitted. The information that was identified is presented in Table 3. 32 –34
Chemical Properties.
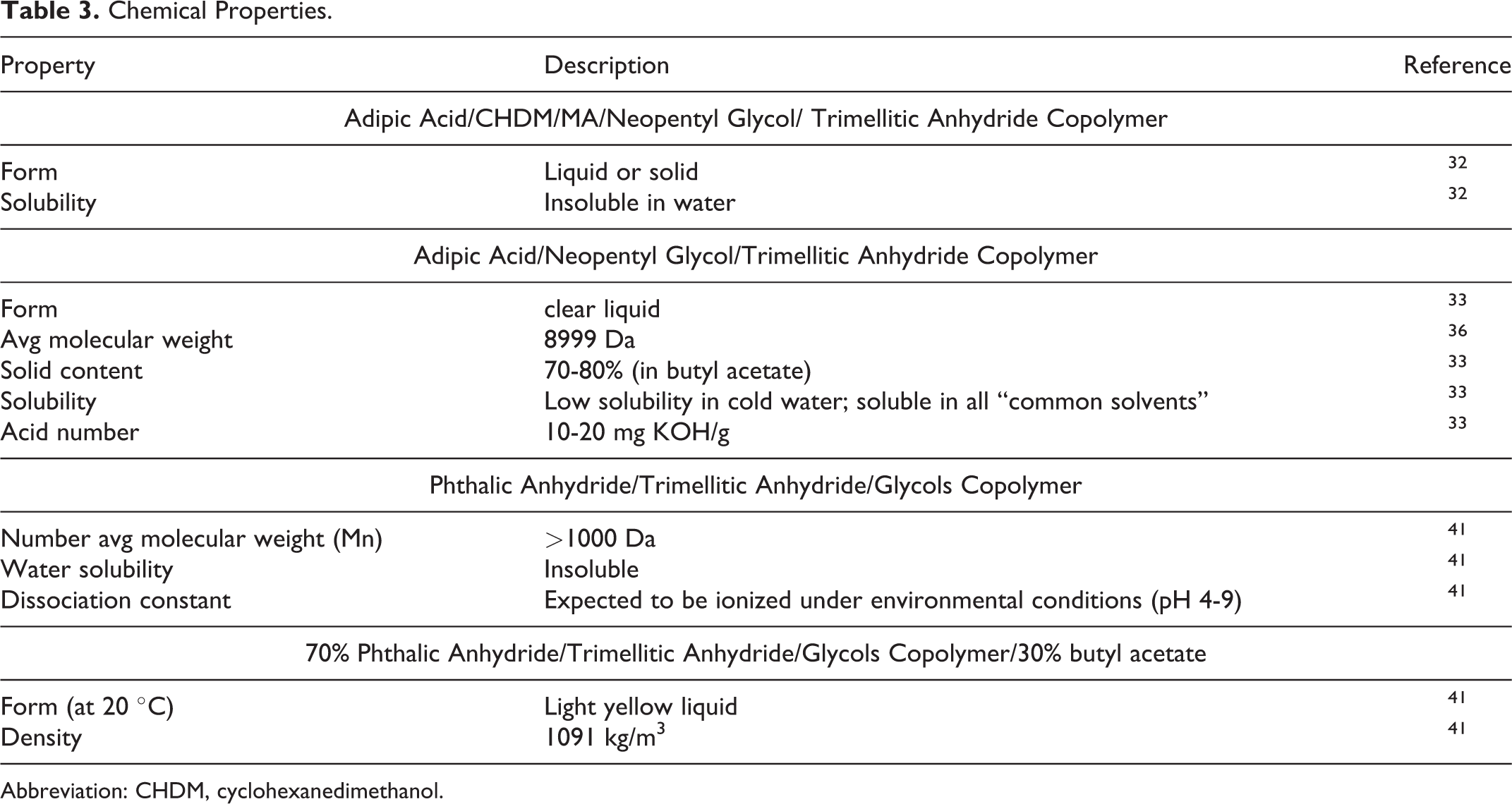
Abbreviation: CHDM, cyclohexanedimethanol.
Method of Manufacture
Common, general polyester synthesis techniques can be described for the manufacture of trimellitic anhydride copolymers. 35 The manufacture of each of these trimellitic anhydride copolymers begins with trimellitic anhydride (an activated form of trimellitic acid). For example, synthesis would start by dissolving trimellitic anhydride in a dry solvent, such as dimethylformamide, under an inert gas, such as nitrogen. Next, the appropriate polyol monomer (eg, propylene glycol, neopentyl glycol, cyclohexanedimethanol, trimethylolpropane, ethylene glycol, or trimethylpentanediol) would be added to the solution and distilled under reduced pressure. In a second step, the resultant terminal alcohol groups may be further esterified with appropriate mono- or multifunctional carboxylic acids (eg, sebacic acid, adipic acid, maleic anhydride, isostearic acid, phthalic anhydride, sebacic acid, or isophthalic acid), and, likely, a catalyst. This 2-step methodology would result in dendrimer-like copolymers, with trimellitic acid as the central core in each. Alternatively, trimellitic anhydride, a polyol, and an additional carboxylic acid of choice could be reacted together in 1 step, under similar conditions, to form more randomly structured copolymers. Due to the trivalent nature of trimellitic acid, however, the resultant polymers from either of these 2 methodologies would be nonlinear.
Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer
In the synthesis of Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer, the trifunctional glycol(s) and the di- and tribasic acids are charged into a clean and dry reaction vessel. 36 Heat is applied to drive the polyester reaction, and water is generated as a by-product of the reaction; the rate of reaction is controlled by the heat applied. The reactant mixture is heated gradually to 200 °C, and the in-process wet properties are measured. The reaction mixture is cooled and dissolved in the solvent; the final product is cooled further and packaged.
Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer
Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer results from condensation of phthalic anhydride, trimellitic anhydride, ethylene glycol, and neopentyl glycol monomers. 37
Impurities/Composition
Limited impurities data on the trimellitic anhydride copolymers as cosmetic ingredients were found in the published literature, and unpublished data were not submitted. The reactions used to manufacture the copolymers can be designed to result in little to no residual monomer, but information on the exact manufacturing process was not available for most of the polymers. Without having a precise manufacturing method, the remaining residual amount of monomer present in the copolymer is unknown. Therefore, it is possible that residual amounts of the following monomers (used in the production of these copolymers) could be present in these ingredients: adipic acid, cyclohexanedimethanol, ethylene glycol, isophthalic acid, maleic anhydride (or maleic acid), neopentyl glycol, phthalic anhydride (or phthalic acid), trimellitic anhydride (or trimellitic acid), trimethylpentanediol, trimethylolpropane, and isostearic acid.
Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer
Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer is synthesized from adipic acid, neopentyl glycol, and trimellitic anhydride. 36 A quantitative [1 H] nuclear magnetic resonance study reported that approximately 1.5% residual glycol is present. Residual trimellitic anhydride and adipic acid concentrations were below the limit of detection (ie, 50 ppm or 0.005%).
One supplier has reported that Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer is sold in butyl acetate (70%-80% solids) and does not contain formaldehyde, toluene, or xylene. 33
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
According to 2016 VCRP data, Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer is reported to be used in 471 cosmetic formulations, 470 of which are nail formulations, and Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer is reported to be used in 85 cosmetic formulations, 83 of which are nail formulations; the remaining uses reported to the VCRP indicated dermal exposure 38 (Table 4). However, the results of the concentration of use survey conducted by the Council in 2015 only report use in nail formulations. According to the Council survey, the highest maximum concentration of use for both of these ingredients is in nail polish and enamel; Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer is reported to be used at up to 32.8% and Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer at up to 12%. 39
Frequency and Concentration of Use According to Duration and Type of Exposure.
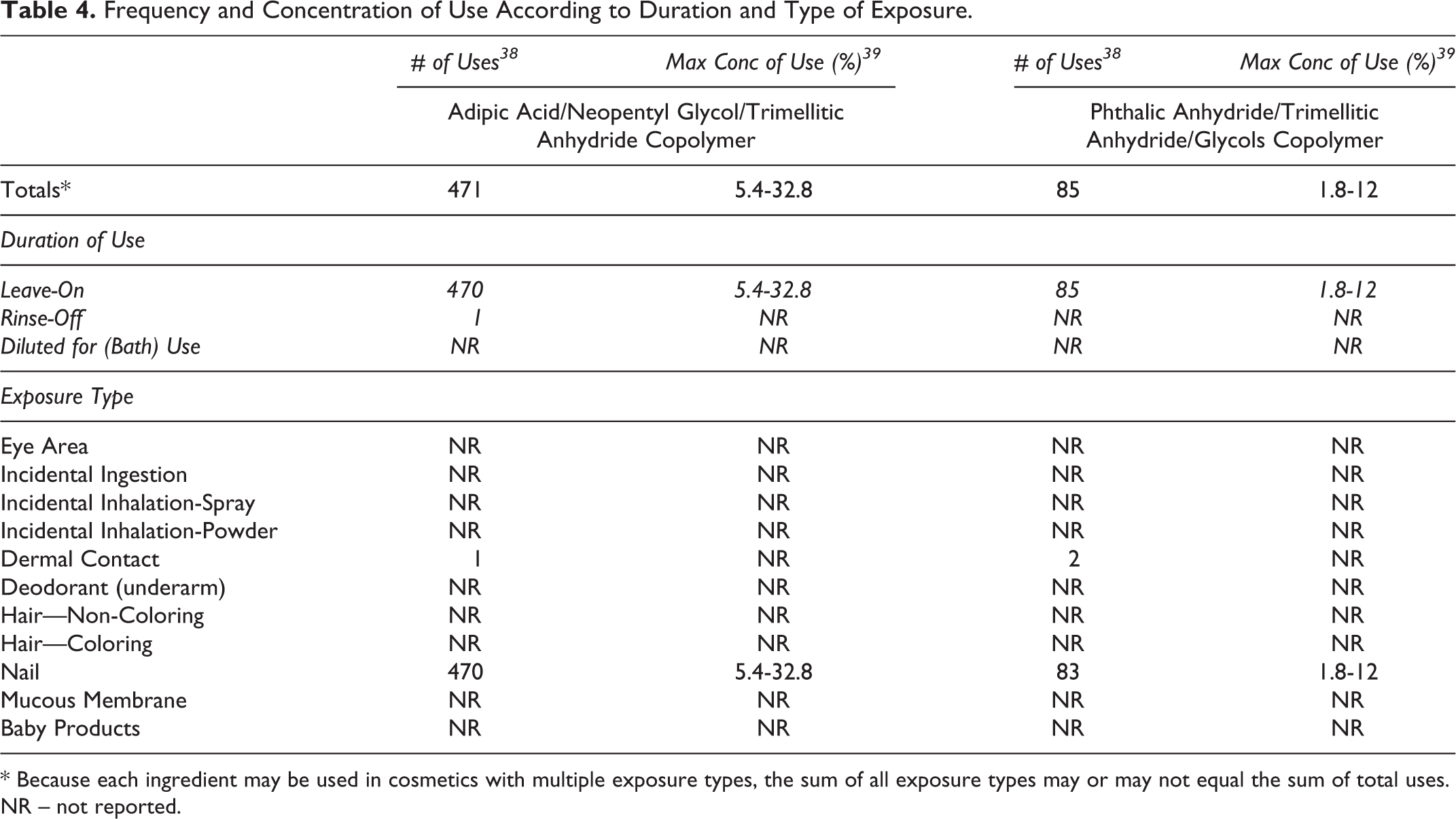
* Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may or may not equal the sum of total uses.
NR – not reported.
The remaining trimellitic anhydride copolymers (ie, Adipic Acid/CHDM/MA/Neopentyl Glycol/Trimellitic Anhydride Copolymer; Isostearoyl Trimellitic Anhydride/Trimethylolpropane Copolymer; Propylene Glycol/Sebacic Acid/Trimellitic Anhydride Copolymer; or Trimethylpentanediol/Isophthalic Acid/Trimellitic Anhydride Copolymer) are not reported to be in use.
None of the trimellitic anhydride copolymers named in the report are restricted from use in any way under the rules governing cosmetic products in the European Union. 40
In Australia, according to a National Industrial Chemicals Notification and Assessment Scheme (NICNAS), Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer was determined to be a polymer of low concern (PLC), having met criteria for molecular weight requirements, functional group equivalent weight requirements, low charge density, approved elements only, stability under normal conditions, it is not water absorbing, and it is not a hazardous substance or dangerous good. 41 Therefore, it is not considered to pose an unreasonable risk to the health of workers and the public when used as a component of cosmetic nail products.
Non-Cosmetic
Adipic Acid/CHDM/MA/Neopentyl Glycol/Trimellitic Anhydride Copolymer is used in preparation of glass fiber reinforced plastic. 32 Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer is used in the manufacture of dyes, pharmaceuticals, insecticides, and as a hardener for resins. 42
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Toxicokinetics studies on the trimellitic anhydride copolymers were not found in the published literature, and unpublished data were not submitted.
Toxicological studies
Toxicological studies on the trimellitic anhydride copolymers were not found in the published literature, and unpublished data were not submitted.
Developmental and Reproductive Toxicity Studies
Developmental and reproductive toxicity studies on the trimellitic anhydride copolymers were not found in the published literature, and unpublished data were not submitted.
Genotoxicity Studies
Genotoxicity studies on the trimellitic anhydride copolymers were not found in the published literature, and unpublished data were not submitted.
Carcinogenicity Studies
Carcinogenicity studies on the trimellitic anhydride copolymers were not found in the published literature, and unpublished data were not submitted.
Dermal Irritation and Sensitization Studies
Human repeated insult patch tests (HRIPTs) were conducted with nail polish formulations containing up to 15% Adipic Acid/ Neopentyl Glycol/Trimellitic Anhydride Copolymer 43 –48 and up to 7.5% Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer 49,50 (Table 5). None of the formulations were sensitizers or irritants.
Human Repeated Insult Patch Tests of Nail Polish Formulations.
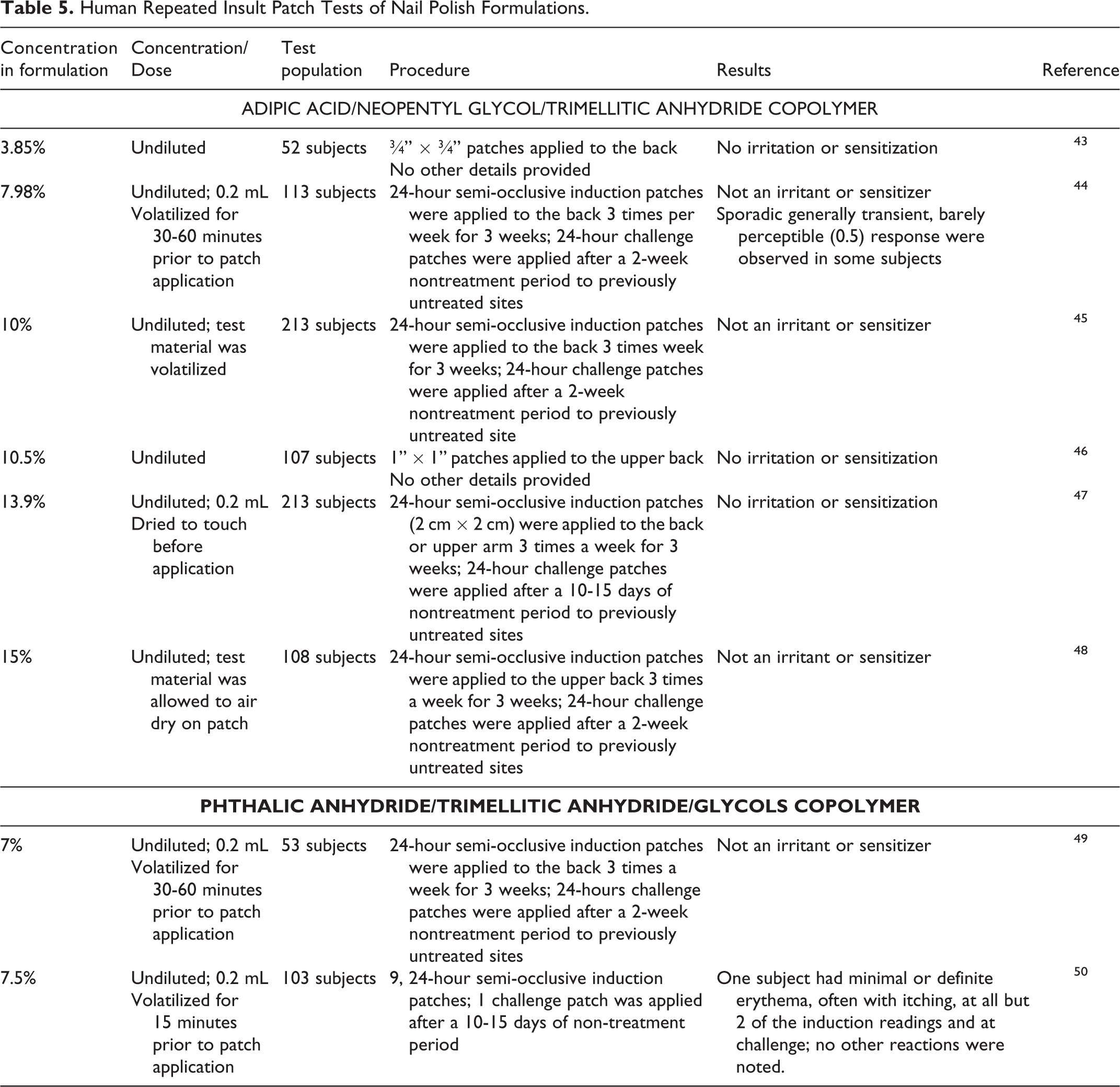
According to an NICNAS report, there is uncertainty regarding the potential sensitization of Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer, but the ingredient cannot be classified for sensitization. 41
Ocular Irritation Studies
Ocular irritation studies on the trimellitic anhydride copolymers were not found in the published literature, and unpublished data were not submitted.
Clinical Reports
Case Reports
Several case reports describing allergic reactions to Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer in nail polish were available. 37,42,51 Details from these case reports are summarized in Table 6.
Case Reports With Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer in Nail Polish

Abbreviations: BCDSB - British Contact Dermatitis Society Standard Battery; ICDRG - International Contact Dermatitis Research Group.
Summary
This report addresses the safety of 6 trimellitic anhydride copolymers as used in cosmetics. According to the Dictionary, these ingredients are reported to function as film formers. The trimellitic anhydride copolymers are related as they all share a common monomer, that is, trimellitic anhydride; each copolymer is also composed of 1 to 4 different monomers. The monomers that comprise these copolymers are interconnected via ester bonds to form highly branched polymeric (polyester) networks. Limited information on the amount of residual monomer in these copolymers was submitted.
Voluntary Cosmetic Registration Program data obtained from the FDA and data received in response to a survey of the maximum reported use concentration by product category conducted by the Council indicate that 2 of the 6 ingredients included in this safety assessment are used in cosmetic formulations. According to the VCRP, Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer is reported to be used in 471 cosmetic formulations, 470 of which are nail formulations, and Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer is reported to be used in 85 cosmetic formulations, 83 of which are nail formulations. Only uses in nail products were reported in response to the Council survey, and the highest maximum concentration of use for both of these ingredients is in nail polish and enamel; Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer is reported to be used at up to 32.8%, and Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer is reported to be used at up to 12%.
Human repeated insult patch tests were conducted with nail polish formulations containing up to 15% Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer and up to 7.5% Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer. None of the formulations were sensitizers or irritants. Several case reports have, however, described allergic reactions to Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer in nail polish. Toxicokinetics, toxicological, genotoxicity, and ocular irritation data were not found in the published literature, and unpublished data were not submitted.
Discussion
The Panel considered this safety assessment of 6 trimellitic anhydride copolymers as used in cosmetics; these ingredients are related in that they all include trimellitic anhydride (aka 1,2,4-benzenetricarboxylic acid anhydride) as a monomer. Because very limited data were available for use in determining the safety of these ingredients, the Panel issued an Insufficient Data Announcement (IDA) at its September 2015 meeting, notifying the public that the data on the trimellitic anhydride copolymers were insufficient to determine whether the ingredients, under each relevant condition of use as indicated in this assessment, are either safe or unsafe. Both, the FDA VCRP data and the Council concentration of use survey, report that these ingredients are used (or are likely to be used) in nail products but only the VCRP data indicate that any of these ingredients are used in products that are intended to come into contact with the skin.
Dermal irritation and sensitization data on nail polish formulations were submitted to the Cosmetic Ingredient Review (CIR) in response to the IDA; the test data were not at the maximum concentrations of use indicated in this assessment. The Panel remained concerned that the copolymers were essentially chemically undefined. In response to this concern, method of manufacture and composition data were received for Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer; these data were sufficient for the Panel to conclude on the safety of Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer in nail, but not in non-nail, formulations.
The Panel considered the NICNAS determination that Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer is a PLC and is not considered to pose an unreasonable risk to public health of workers and the public when used as a component of cosmetic nail products. The PLC criteria that were met include molecular weight requirements, functional group equivalent weight requirements, low charge density, approved elements only, stability under normal conditions, not water absorbing, and not a hazardous substance or dangerous good. The fact that Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer met these criteria mitigated concern regarding the safe use of this ingredient in nail formulations; it did not address information needs for use in non-nail cosmetic formulations.
In order to determine safety of the use of Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer and Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer in cosmetics other than nail products, irritation and sensitization data at maximum leave-on concentrations of use are needed.
The data remain insufficient to determine the cosmetic safety of the remaining 4 ingredients included in this safety assessment; these ingredients are not reported to be in use. The following are needed to evaluate the safety of these ingredients for use in nail products: Method of manufacture; and Composition data.
In addition, the following data are needed to evaluate the safety of these 4 ingredients in non-nail cosmetic formulations: Molecular weight; Impurities, specifically, the amount of residual monomer in each copolymer; Metabolism, specifically, whether these ingredients are metabolized in the skin; Dermal absorption; if absorbed, then genotoxicity and reproductive toxicity data are needed; and Dermal irritation and sensitization data at maximum leave-on concentrations of use.
The Panel acknowledged that anhydride monomers (eg, trimellitic anhydride) can be respiratory sensitizers, and exposures in the workplace to the anhydrides that are used in the production of these copolymers have resulted in numerous adverse effects. However, because the levels of residual monomer in nail products are expected to be minimal, exposure to trimellitic anhydride is not expected with cosmetic use and is not of concern.
Conclusion
The Panel concluded that Adipic Acid/Neopentyl Glycol/Trimellitic Anhydride Copolymer and Phthalic Anhydride/Trimellitic Anhydride/Glycols Copolymer are safe in nail product formulations in the present practices of use and concentration described in this safety assessment; the data are insufficient to make a determination of safety on the use of these ingredients in all other types of cosmetic formulations.
The Expert Panel for Cosmetic Ingredient Safety also concluded that the available data are insufficient to make a determination that the following 4 trimellitic anhydride copolymers are safe under the intended conditions of use in cosmetic formulations: Adipic Acid/CHDM/MA/Neopentyl Glycol/Trimellitic Anhydride Copolymer* Isostearoyl Trimellitic Anhydride/Trimethylolpropane Copolymer* Propylene Glycol/Sebacic Acid/Trimellitic Anhydride Copolymer* Trimethylpentanediol/Isophthalic Acid/Trimellitic Anhydride Copolymer*
* Not reported to be in current use.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Fiume and Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Bergfeld, Belsito, Hill, Klaassen, Liebler, Marks, Shank, Slaga, and Snyder, P. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
