Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of Polyaminopropyl Biguanide (polyhexamethylene biguanide hydrochloride), which functions as a preservative in cosmetic products. The Panel reviewed relevant data relating to the safety of this ingredient and concluded that Polyaminopropyl Biguanide is safe in cosmetics in the present practices of use and concentration described in the safety assessment, when formulated to be nonirritating and nonsensitizing, which may be based on a quantitative risk assessment or other accepted methodologies. The Panel also concluded that the data are insufficient to determine the safety of Polyaminopropyl Biguanide in products that may be incidentally inhaled.
Introduction
The safety of the cosmetic ingredient identified by the International Nomenclature of Cosmetic Ingredients (INCI) name Polyaminopropyl Biguanide is reviewed in this assessment. 1 Polyaminopropyl Biguanide is reported to be used as a preservative in cosmetics, according to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary). The chemical name that corresponds to the cosmetic ingredient is polyhexamethylene biguanide hydrochloride (PHMB HCl), and it is the hydrochloride salt of an amino polymer comprising hexyl biguanide repeat units; it has a 6-carbon chain in each monomeric repeat unit, and is always supplied as the hydrochloride salt. International Nomenclature of Cosmetic Ingredients nomenclature often differs from standard chemical naming conventions; therefore, it should be noted that the substance identified by the chemical name polyaminopropyl biguanide is not a cosmetic ingredient.
In Cosmetic Ingredient Review (CIR) safety assessments, it is standard procedure to capitalize INCI names, but to use lower case for standard chemical names. Accordingly, throughout this report, when the INCI name Polyaminopropyl Biguanide is used (with appropriate capitalization), it is to be understood that it is referring to the chemical polyhexamethylene biguanide hydrochloride, and this is the ingredient with reported uses in cosmetics. Furthermore, most of the safety test data included in this report are on the chemical PHMB HCl, as indicated by the use of the INCI name. The only exception to the exclusive use of the INCI name Polyaminopropyl Biguanide in this safety assessment relates to the summary of a cytotoxicity study, in which results for PHMB and polyaminopropyl biguanide are compared.
This safety assessment includes relevant published and unpublished data for each end point that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A list of the typical search engines and websites used, sources explored, and end points that CIR evaluates, is available on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition and General Characterization
Polyaminopropyl Biguanide (CAS Numbers: 32289-58-0 [PHMB HCl]; 27083-27-8 [PHMB HCl]; 28757-47-3 [PHMB]) is the hydrochloride salt of an amino polymer comprising hexyl biguanide repeat units (PHMB HCl). According to the Dictionary, it is the organic compound that conforms to the formula depicted in Figure 1. 1
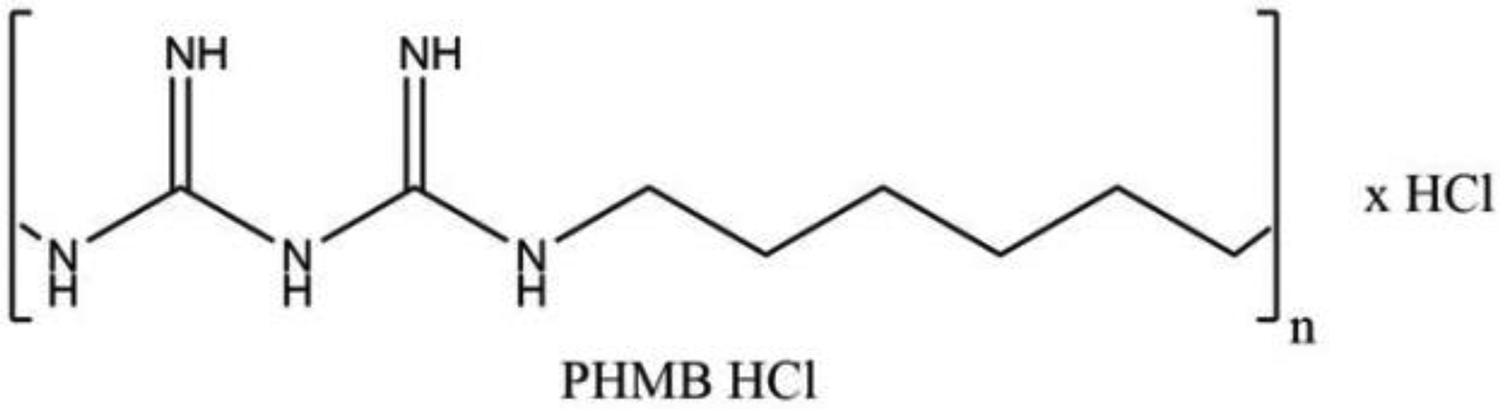
Polyaminopropyl biguanide (PHMB HCl).
Comments on the identity of Polyaminopropyl Biguanide were received from a chemical supplier, which stated that, effectively, all Polyaminopropyl Biguanide is PHMB HCl (ie, C6 alkyl chains linked together by biguanide groups), and no propyl biguanide groups are present. 2 The INCI name is an artifact of arbitrarily choosing the middle of the C6 alkyl chains to identify the polymer repeating units of the ingredient.
Chemical and Physical Properties
Polyaminopropyl Biguanide is a polymer that, in its neat form (as hydrochloride salt), is a solid/powder with purity >94.2%. 3 It is often marketed as an approximately 20% aqueous, preformulation solution. Chemical and physical properties are summarized in Table 1.
Chemical Properties of Polyaminopropyl Biguanide.

Method of Manufacture
One of the current methods for manufacturing Polyaminopropyl Biguanide is through the polycondensation of sodium dicyanamide and hexamethylenediamine (Figure 2). 4

Polyaminopropyl Biguanide (PHMB HCl) versus PHMG phosphate.
Impurities
The following chemicals have been reported as possible impurities of Polyaminopropyl Biguanide: N-(6-aminohexyl)-N′-(6-(6-guanidinohexyl)guanidine, N-cyano-N′-(6-N-cyanoaminohexyl)guanidine, N-cyano-N′-(6-aminohexyl)guanidine), N-cyano-N′-6-(6-guanidinohexyl)guanidine hydrochloride, and 1,6-diguanidinohexane dihydrochloride. 3
The trace metals content (in ppm, wt/wt) of 5 different batches of technical grade Polyaminopropyl Biguanide (solid) has been reported as follows: cadmium (<0.25), chromium (<0.25-0.7), cobalt (<0.25), iron (14-40), lead (<2), zinc (370-540), arsenic (<2), and mercury (<0.2). 3
Use
Cosmetic
The safety of Polyaminopropyl Biguanide is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. 5 Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Council, of maximum reported use concentrations by product category. 6
According to 2019 VCRP data, Polyaminopropyl Biguanide is being used in 147 cosmetic formulations, which are mostly leave-on products (Table 2). 5 The results of a concentration of use survey provided in 2017 indicate that Polyaminopropyl Biguanide is used at concentrations up to 0.2% in leave-on products (eye lotions), and used in baby lotions, oils, and creams (leave-on products) at concentrations up to 0.1%. 6 Polyaminopropyl Biguanide is also used at concentrations up to 0.1% in rinse-off products (ie, hair dyes and colors, and in skin cleansing products).
Frequency (2019) and Concentration (2017) of Use According to Duration and Type of Exposure.

Abbreviation: NR, not reported.
* Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may or may not equal the sum of total uses.
b Not specified these products are sprays or powders, but it is possible the use can be as a spray or powder, therefore the information is captured in both categories.
c It is possible these products are powders, but it is not specified whether the reported uses are powders.
Cosmetic products containing Polyaminopropyl Biguanide may be applied to the skin and hair or may come in incidental contact with the eyes (at maximum use concentrations up to 0.2% in eye lotions) and mucous membranes (0.006% in “other” personal cleanliness products). Polyaminopropyl Biguanide is used in a lipstick product, the application of which may result in incidental ingestion; no concentration data were reported for this use. It is also used in baby lotions, oils, or creams at maximum use concentrations up to 0.1%. Products containing Polyaminopropyl Biguanide may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
According to FDA VCRP data, Polyaminopropyl Biguanide is used in a fragrance preparation, which may result in incidental inhalation exposure; concentration data were not reported for this use. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays. 7 –10 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 7,11
Polyaminopropyl Biguanide is listed in Annex V (entry 28) of the European Commission Regulation No. 1223/2009 (Cosmetic Regulation) as a preservative to be used in all cosmetic products at a maximum concentration of 0.3%. 12 Additionally, Polyaminopropyl Biguanide is classified as CMR 2 (Carc. 2) according to the Commission Regulation (EU) No. 944/2013. CMR substances are classified as carcinogenic, mutagenic, or toxic for reproduction. A substance is placed in carcinogen Category 2 (Carc. 2, suspected human carcinogens) when the evidence obtained from human and/or animal studies is not sufficiently convincing to place the substance in Category 1A (substances known to have carcinogenic potential for humans) or Category 1B (substances presumed to have carcinogenic potential for humans). The Carc. 2 classification was effective as of January 1, 2015, and, according to Article 15 (1) of the Cosmetics Regulation, the use of Polyaminopropyl Biguanide as a cosmetic ingredient is considered to be prohibited as of this date. 3 However, Article 15 (1) of the Cosmetics Regulation also states that a substance classified in Category 2 may be used in cosmetic products if the substance has been evaluated by the Scientific Committee on Consumer Safety (SCCS) and found safe for use in cosmetic products. Accordingly, in 2017, the SCCS issued a final opinion stating that “the use of Polyaminopropyl Biguanide as a preservative in all cosmetic products at concentrations up to 0.1% is safe and that its use in sprayable formulations is not advised.”
Noncosmetic
Polyaminopropyl Biguanide is reported to be the most frequently used antiseptic in traumatic and orthopedic surgery. 13 According to another source, Polyaminopropyl Biguanide has the following uses: fungicide, algicide, sanitizer in swimming pools, preservative for cut flowers, materials preservative, bacteriostat in industrial processes and water systems, and hard surface disinfectant (food and nonfood contact surfaces). 14
Polyaminopropyl Biguanide is a broad-spectrum antimicrobial agent used in a variety of products, including contact lens cleaning solutions, skin disinfectant solutions, and wound dressings. 15 Solid wound dressings are composed of various synthetic or naturally derived materials, and typically contain added antimicrobials, such as silver, bismuth, chlorhexidine, bacitracin, or Polyaminopropyl Biguanide. Wound dressings are regulated by the US FDA as Class 1 medical devices (ie, the device is exempt from premarket notification procedures). However, this classification does not apply to wound dressings that contain added drugs, such as antimicrobial agents. 16
Additionally, Polyaminopropyl Biguanide has been reviewed by the US Environmental Protection Agency (EPA). The EPA concluded that its use as a pesticide has very low aggregate risk of adverse health effects to the public or environment. 14
In Australia, Polyaminopropyl Biguanide is listed in the Poisons Standard—the Standard for the Uniform Scheduling of Medicines and Poisons in Schedule 6. 17 Schedule 6 chemicals are described as “substances with a moderate potential for causing harm, the extent of which can be reduced through the use of distinctive packaging with strong warnings and safety directions on the label.” Schedule 6 chemicals are labeled “Poison.” According to this standard, Polyaminopropyl Biguanide can be used in preparations containing concentrations of 5% or less and when packed and labeled for therapeutic use.
Toxicokinetics Studies
Dermal Penetration
The dermal penetration studies summarized below are presented in Table 3.
Dermal Penetration Studies.

Abbreviation: SCCS, Scientific Committee on Consumer Safety.
In vitro
In one study, skin penetration experiments were performed using both rat (skin disks in solutions; 5-day equilibration phase) and human skin (receptor fluid in diffusion cell collected up to 15 days) in vitro. 18 At 0.4%, 1.4%, 5%, and 20% concentrations of Polyaminopropyl Biguanide, absorption rates through human epidermis were 8.13, 22.8, 350, and 1005 ng/cm2/h, respectively. At 0.4%, 20% (early phase), and 20% (late phase) [14C]-Polyaminopropyl Biguanide, absorption rates in rat whole skin were 131, 3695, and 11,940 ng/cm2/h, respectively. Another study involved the application of Polyaminopropyl Biguanide (5% solution) to rat skin biopsies from newborn hairless rats and human epidermal skin in diffusion chambers. In rat skin, no absorption was detected up to day 5 of exposure. In human epidermal skin biopsies, a low rate of penetration (∼ 0.09%) was noted after 24 hours. Polyaminopropyl Biguanide solutions (0.1% aqueous micellar solution, 0.1% oil-in-water emulsion, 0.3% aqueous micellar solution, and 0.3% oil-in-water emulsion) were applied to human split-thickness skin in a 2-part dermal penetration study. 3 In Part 1, penetration of the 0.1% aqueous micellar solution and 0.1% in oil-in-water emulsion was determined directly after the 24-hour exposure period. In Part 2, 24-hour exposure to the 0.3% aqueous micellar solution and to 0.3% in an oil-in-water emulsion was followed by an additional 72-hour period to determine whether the test compound that was absorbed into the skin during the previous 24-hour period would move from the skin into the receptor fluid after the washout. Skin absorption was found to be 1.56% (dermis contained 1.56% of applied dose) + 0.03% (absorbed dose = 0.03% of applied dose found in the receptor fluid). Based on SCCS Notes of Guidance, one standard deviation (2.5%) was added to absorbed amount, yielding a calculated dermal absorption value of 4.09% (1.56% + 0.03% + 2.5% = 4.09%). After [14C]-Polyaminopropyl Biguanide (tested at 0.3% wt/wt in a cosmetic formulation) was applied (occluded) to human split-thickness skin a diffusion cell, the mean absorbed dose was 0.17% at 24 hours. 18 When [14C]-Polyaminopropyl Biguanide (20.2% aqueous) was applied (occluded) to human skin epidermal membranes in a diffusion cell at ∼ 200 g active ingredient/L, the mean absorption percentage was 0.001% over a 24-hour period. 18 After the same test substance was warmed to 40 °C and the applied (occluded) to human skin epidermal membranes at 200 g active ingredient/L, the mean absorption percentage was 0.007% over a 24-hour period. 18
Absorption, Distribution, Metabolism, and Excretion
The toxicokinetics studies (oral exposure) summarized below are presented in Table 4.
Toxicokinetics Studies—Oral Exposure.

Animal
Oral
In rats, radiolabeled Polyaminopropyl Biguanide dosed orally was excreted principally in the feces. 3,18 In one study, rats were dosed orally with 20 mg/kg/d for 10 days and elimination after dosing was described as follows: 5.6% ± 0.35% in urine, 93.1% ± 1.58% in feces, and 0.2% exhaled. 18,19 In another animal study (male Alderley Park rats) of the distribution of radioactivity after dosing (in diet), the greatest amounts of radioactivity were detected in adipose tissue, followed by the kidneys and liver. 18,19 No radioactivity was detected in brain. Small amounts of Polyaminopropyl Biguanide oligomers with 2-cyanoguanidino end groups were found in the urine, together with trace constituents, 3,3-dicyano-1,1-hexamethylenediguanidine and a compound considered to be 1-(6-aminohexyl)-3-cyanoguanidine. Absorption was not detected in a study in which mice received a single oral dose (2 mL) of [14C]-Polyaminopropyl Biguanide. 18 In contrast, the results from a study in which groups of Wistar Han rats received [14C]-Polyaminopropyl Biguanide in drinking water, or in the diet for 7 days, indicated that most was absorbed, and that excretion was primarily via the urine. 20
Toxicological Studies
Acute Toxicity Studies
Animal
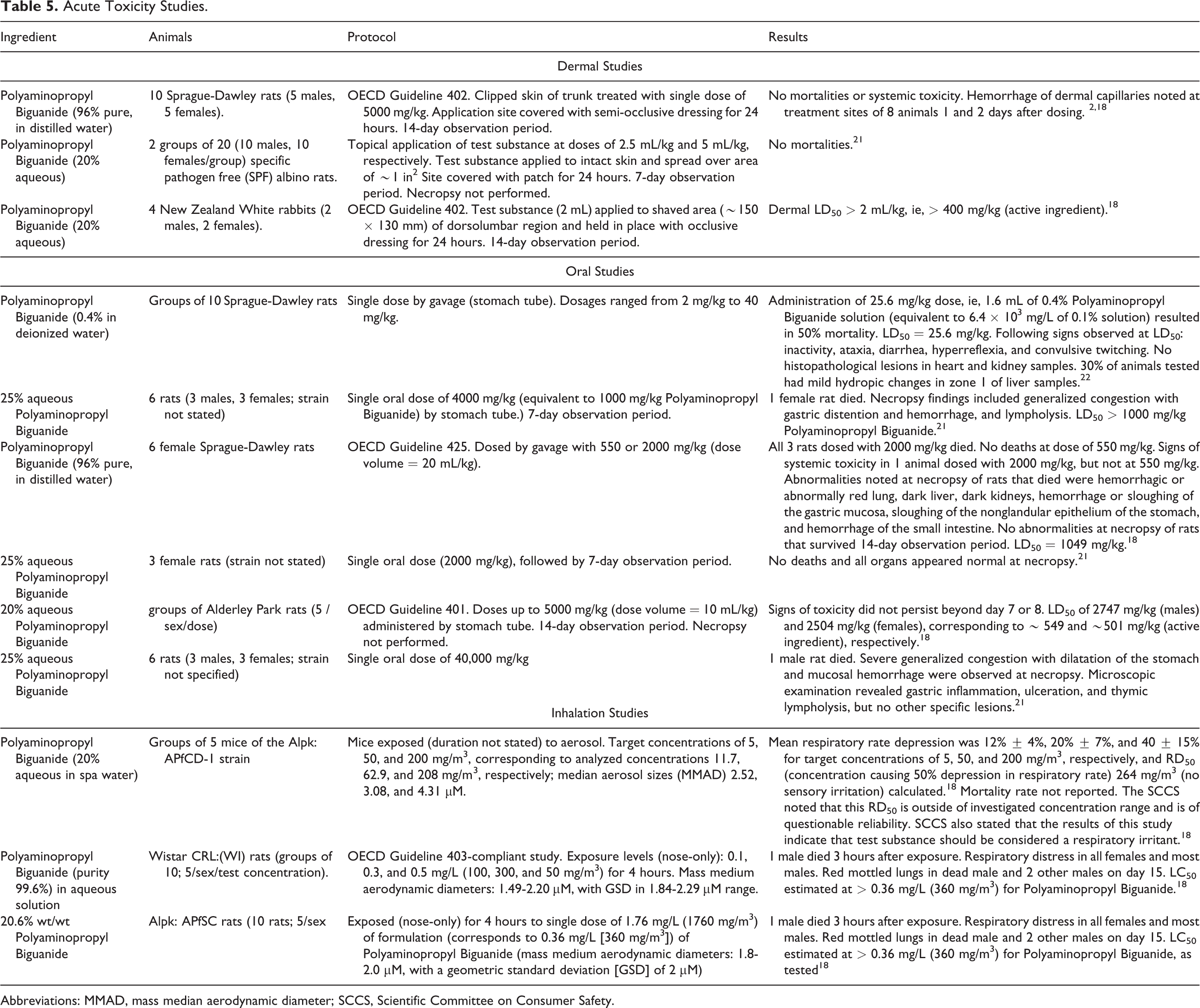
The acute dermal, oral, and inhalation toxicity studies in animals summarized below are presented in Table 5.
Acute Toxicity Studies.
Abbreviations: MMAD, mass median aerodynamic diameter; SCCS, Scientific Committee on Consumer Safety.
Dermal
There was no mortality or other signs of systemic toxicity in rats that received a single dermal dosage of 5000 mg/kg aqueous Polyaminopropyl Biguanide (96% pure, in distilled water), but hemorrhage of dermal capillaries at the application site was observed. 2,18 In another acute dermal toxicity study involving rats that received a topical application of 20% aqueous Polyaminopropyl Biguanide, none of the animals died. 21 In an acute dermal toxicity study of 20% aqueous Polyaminopropyl Biguanide involving rabbits, the LD50 was reported to be > 400 mg/kg. 18
Oral
An LD50 of 1040 mg/kg was reported in a study in which rats were dosed orally with Polyaminopropyl Biguanide (concentration not stated) in distilled water. 18 LD50 values of > 1000 mg/kg were reported for rats dosed orally with aqueous solutions of up to 25% Polyaminopropyl Biguanide. 18,21 A median lethal dosage of 25.6 mg/kg was reported for rats dosed orally with a 0.4% Polyaminopropyl Biguanide solution. 22
Inhalation
LC50 s of > 0.36 mg/L and equal to 0.37 mg/L were reported in acute inhalation toxicity studies in which rats were exposed for 4 hours to Polyaminopropyl Biguanide (99.6%) solutions at concentrations of 360 mg/m3 in air and up to 300 mg/m3 in air, respectively. 18 Dark/red lungs were observed at necropsy. A concentration-related depression of respiratory rate was reported in a study in which mice were exposed to Polyaminopropyl Biguanide (20% aqueous) at concentrations up to 208 mg/m3. 18
Human
Risk assessment—oral
The EPA conducted a screening-level acute dietary human health risk assessment for Polyaminopropyl Biguanide in food. 14 Risk estimates were calculated for females 13 to 50 years old, the only population subgroup with an acute toxicity end point (not stated) that was of concern. “Risk estimates for the use with the highest exposures were 9% of the acute Population Adjusted Dose (aPAD = 0.2 mg/kg/day) and, therefore, were not of concern.” The EPA defines an aPAD as a dose at which an individual could be exposed on any given day and no adverse health effects would be expected.
Short-Term Toxicity Studies
The short-term dermal, oral, and inhalation toxicity studies summarized below are presented in Table 6.
Short-Term Toxicity Studies.

Abbreviations: LOAEL, lowest observed adverse effect level; MMAD, mass median aerodynamic diameter; NOAEC, no observed adverse effect concentrations; NOAEL, no observed adverse effect level; SPF, specific pathogen free.
Dermal
No specific systemic effects were observed after 25% aqueous Polyaminopropyl Biguanide was applied to the skin of rats for 3 alternating 24-hour periods. 21 There was also no evidence of systemic toxicity in rats that received 6 24-hour dermal applications of 20% Polyaminopropyl Biguanide (diluted with water to 0.04% active ingredient). There were no mortalities or signs of systemic toxicity in rats that received dermal applications of 20.2% aqueous Polyaminopropyl Biguanide at dosages up to 200 mg/kg daily over a 30-day period (21 applications total; no-observed adverse effect level [NOAEL] = 200 mg/kg/d). 18 In a 21-day dermal toxicity study involving rabbits, there was no evidence of toxic effects on the skin after 20% aqueous Polyaminopropyl Biguanide (12,000 ppm solution [1 mL]) was applied daily. 18
Oral
Gastrointestinal inflammation was observed in rats dosed orally with 25% aqueous Polyaminopropyl Biguanide (in distilled water; initially at 1 g/kg and subsequently at 0.5 g/kg) for 21 days. 21 A lowest observed adverse effect level (LOAEL) of 0.1 mg/mL for 20% aqueous Polyaminopropyl Biguanide (in drinking water) was reported in 28-day oral toxicity studies involving rats and mice. 18 Rats (groups of 10) that received Polyaminopropyl Biguanide (in drinking water, up to 150 mg/kg) for 4 weeks experienced dehydration, clinical signs of rough coat and hunched posture, and body weight loss (all classified as severe). 20 Across the 3 dose groups, 10 rats had to be terminated due to severe weight loss, whereas, the remaining rats eventually adapted and began to gain weight. Absolute liver weights in all dose groups were similar to the control group. Mild centrilobular hypertrophy in the liver was observed in some of the rats (all dose groups). In the same study, Polyaminopropyl Biguanide administered (in the diet, 4000 mg/kg) to rats for 4 weeks caused a statistically significant decrease in body weight and absolute liver weight. In this dietary group, there was no evidence of centrilobular hypertrophy in the liver. Also, there was no evidence of necrosis or inflammatory lesions in the liver when Polyaminopropyl Biguanide was administered in drinking water or in the diet. In a 60-day gavage study on Polyaminopropyl Biguanide involving rats, mild toxicity in the liver or kidneys was observed (by microscopic examination) at 2 mg/kg/d (dose equivalent to 0.2 mg/L of 0.4% solution of test substance), 8 mg/kg/d (dose equivalent to 0.4 mg/L of 0.4% solution of test substance), and 32 mg/kg/d (highest dose, equivalent to 1.2 mg/L of 0.4% solution of test substance). 22 None of the animals died.
Inhalation
In 21-day and 28-day inhalation toxicity studies on Polyaminopropyl Biguanide involving rats, no observed adverse effect concentrations (NOAECs) of 0.025 mg/m3 and 0.0239 mg/m3 were reported, respectively. 18 In the 21-day study, the animals were exposed (nose-only, concentrations up to 26 mg/m3) to the test substance 5 days per week, 6 hours/day. Slightly-to-moderately severe pneumonitis was observed at histopathological examination in rats exposed to 0.25 mg/m3. Moderate to severe pneumonitis was observed in rats exposed to 2.75 mg/m3, and severe nasal irritation and dyspnea were observed at a concentration of 12.5 mg/m3. Additionally, all rats of the 12.5 and 26 mg/m3 groups died. In the 28-day study (nose-only, concentrations up to 2.5 mg/m3, 6 hours/day, 5 days per week), squamous metaplasia was observed in the larynx of males and females exposed to 0.25 mg/m3 and 2.5 mg/m3, and tracheal inflammation was observed in males and females exposed to 2.5 mg/m3. Pneumonitis and bronchitis were observed in the lungs of males and females exposed to 2.5 mg/m3.
Subchronic Toxicity Studies
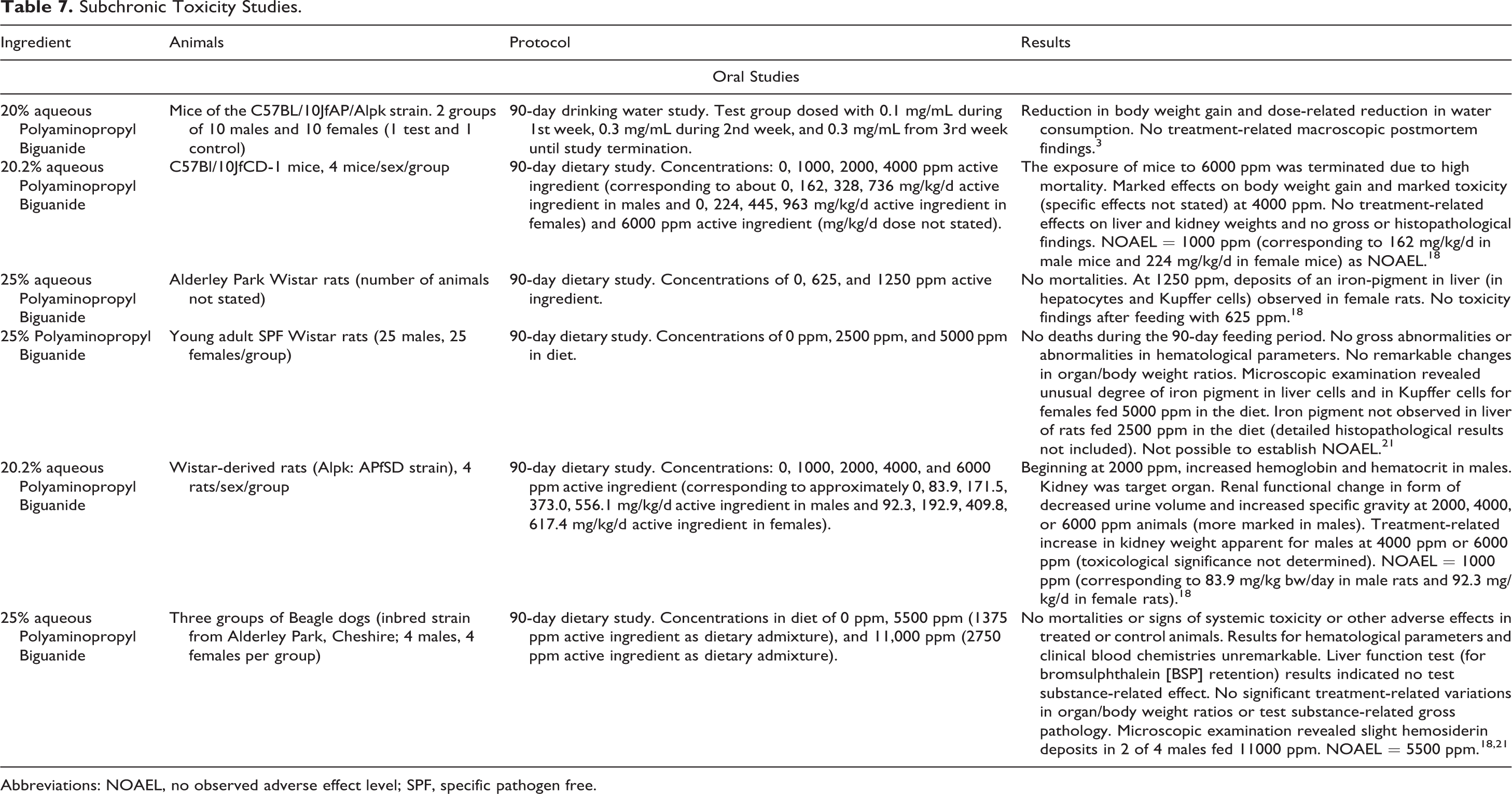
The subchronic oral toxicity studies summarized below are presented in Table 7.
Subchronic Toxicity Studies.
Abbreviations: NOAEL, no observed adverse effect level; SPF, specific pathogen free.
Oral
There were no treatment-related macroscopic postmortem findings in mice in a 90-day drinking water study of 20% aqueous Polyaminopropyl Biguanide (concentrations up to 0.3 mg/mL in drinking water), 3 and a NOAEL of 1000 ppm was reported for this ingredient in a 90-day feeding study on 20.2% aqueous Polyaminopropyl Biguanide in which mice received concentrations up to 4000 ppm in the diet. 18 The following results were reported in 90-day oral toxicity studies on Polyaminopropyl Biguanide involving rats: no mortalities, but iron pigment/deposits were observed in Kupffer cells at 1250 ppm (in one study on 25% aqueous Polyaminopropyl Biguanide) and 5000 ppm (in another study on 25% aqueous Polyaminopropyl Biguanide) in diet; and a NOAEL of 1000 ppm (in a third study on 20.2% aqueous Polyaminopropyl Biguanide). 18,21 An NOAEL of 5500 ppm was reported for Beagle dogs fed Polyaminopropyl Biguanide at concentrations up to 11,000 ppm in the diet for 90 days. 18,21
Chronic Toxicity Studies
Animal
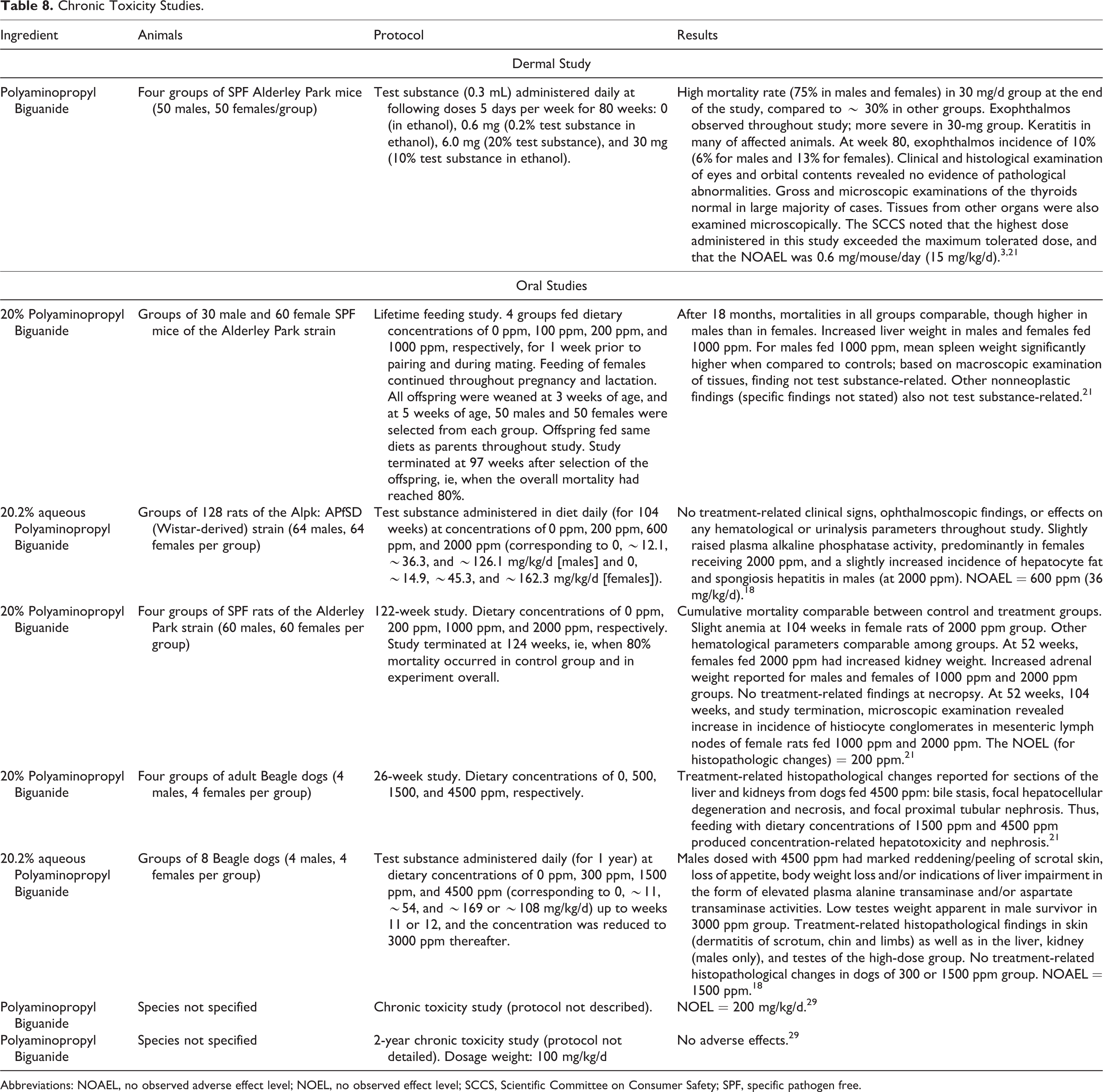
The chronic dermal and oral toxicity studies summarized below are presented in Table 8.
Chronic Toxicity Studies.
Abbreviations: NOAEL, no observed adverse effect level; NOEL, no observed effect level; SCCS, Scientific Committee on Consumer Safety; SPF, specific pathogen free.
Dermal
In an 80-week chronic toxicity study involving mice (dermal applications 5 days/week), a mortality rate of 75% was reported for the highest dose group (10% Polyaminopropyl Biguanide; 30 mg dose). 3,21 The exophthalmos observed throughout the study was more severe in this group, compared with the other groups, but the results of histological examination of the eyes and gross and microscopic examination of the thyroids were negative. An NOAEL of 0.6 mg/mouse/day (15 mg/kg/d) was reported.
Oral
Mice were fed 20% Polyaminopropyl Biguanide in the diet (concentrations up to 1000 ppm; feeding 1 week prior to mating and during mating [males and females] and continuation of feeding throughout pregnancy and lactation [females]). 21 The offspring of these mice were also fed 20% Polyaminopropyl Biguanide at concentrations up to 1000 ppm in the diet for 97 weeks. For parents and their offspring, feeding with the test substance did not cause macroscopic changes in the spleen or liver. 18 In a 104-week oral toxicity study involving rats, an NOAEL of 2000 ppm (highest concentration tested in diet) was reported for 20.2% Polyaminopropyl Biguanide. 18 This concentration corresponded to ∼ 126 mg/kg/d (in male rats) and 162.3 mg/kg/d (in female rats). A no observed effect level (NOEL) of 200 ppm for histopathologic changes was reported in a 122-week oral toxicity study involving rats fed 20% Polyaminopropyl Biguanide at concentrations up to 2000 ppm in the diet. 21 Increased adrenal weight was reported for males and females at concentrations of 1000 ppm and 2000 ppm in the diet. In a 26-week feeding study involving dogs, dietary concentrations of 1500 ppm and 4500 ppm 20% Polyaminopropyl Biguanide produced concentration-related hepatotoxicity and nephrosis. 21 An NOAEL of 1500 ppm for 20.2% Polyaminopropyl Biguanide was reported in a 1-year feeding study involving dogs; treatment-related histopathological findings in the liver and kidneys were reported in the high-dose group. 18 In this study, groups of animals were fed test-substance concentrations of 300 ppm, 1500 ppm, and 4500 ppm for up to weeks 11/12. The 4500 ppm concentration was reduced to 3000 ppm for the remainder of the study because high dose males exhibited unexpected signs of toxicity, including marked reddening/peeling of scrotal skin, loss of appetite, body weight loss, and/or indications of liver impairment in the form of elevated plasma alanine transaminase and/or aspartate transaminase activities.
Human
Risk assessment—dermal
In a chronic oral study, rats received diet containing 0, 200, 600, and 2000 ppm Polyaminopropyl Biguanide (corresponding to about 0, 12.1, 36.3, and 126.1 mg/kg bw/day in males; 0, 14.9, 45.3, and 162.3 mg/kg bw/day in females) for 104 weeks (summarized in Table 8). 18 There were no treatment related clinical signs, ophthalmoscopic findings, or effects on any hematological or urinalysis parameters throughout the study. The pathological examination showed no nonneoplastic and no neoplastic findings at any dose level in either sex. However, at 2000 ppm, slightly raised plasma alkaline phosphatase activity, predominantly in females, and a slightly increased incidence of hepatocyte fat and spongiosis hepatis in males were identified. Though an increased incidence of hemangiosarcoma in females was identified (it gave positive results in the trend test, but not statistically significant in the Fisher Exact Test), a subsequent review conducted by a Pathology Working Group concluded that evidence from this rat study did not support a clear treatment-related effect with respect to vascular tumors. Based on these findings, NOAELs of 36 and 45 mg/kg bw/day were derived for male and female rats, respectively. The SCCS further performed a margin of safety (MOS) calculation by using an NOAEL of 36 mg/kg bw/day on the basis of the following additional assumptions: 3 (1) all cosmetics contain 0.1% Polyaminopropyl Biguanide; (2) the NOAEL is 3.1 mg/kg/d, based on oral absorption of 8.5%; and (3) dermal penetration is 4.09%. Dermal penetration was determined using the results of a dermal penetration study summarized earlier in the report in which 1.56% of applied dose was found in the dermis, 0.03% of the absorbed dose was recovered in the receptor fluid, and one standard deviation (2.5%) was added to the absorbed amount. 3,18 The MOS values are 258 (based on cosmetic exposure estimate) and 227 (based on cosmetic exposure estimate + noncosmetic exposure estimate). Thus, the MOS is lower when additional exposure from noncosmetic use is incorporated.
Risk assessment—inhalation
The Council survey of maximum reported use concentrations by product category provided to CIR on July 18, 2017, indicates that Polyaminopropyl Biguanide is not used in pump or propellant hair sprays. 23 However, products categorized as Tonics, Dressings, and Other Hair Grooming Aids that contain Polyaminopropyl Biguanide at maximum use concentrations of up to 0.1% are reported in the survey, and it is possible that products included in this category are sprays. Furthermore, 2019 FDA VCRP data indicate that Polyaminopropyl Biguanide is used in the Other Fragrance Preparations product category (use concentration data were not provided by industry for this use). Given the potential for inhalation exposure, CIR performed a risk assessment using the ConsExpo Web Spray Model (Consumer Exposure Model, Web version 1.0.1). 8,24 –27 The maximum concentrations of use (0.0004% in propellant hair sprays and 0.053% in pump hair sprays) included in this risk assessment to estimate the inhalation exposure concentrations of Polyaminopropyl Biguanide during the use of cosmetic spray products are based on survey results that were originally submitted by the Council to CIR on April 11, 2017. 6
The parameters used in this risk assessment are presented in Table 9. Conservative default values published by Rijksinstituut voor Volksgezondheid en Milieu (RIVM—the Dutch National Institute for Health and Environment) were used in all of the calculations. 8 One exception is that the room ventilation rate was assumed to be 0.2 room-air exchanges per hour, which is the default value specified in European Union’s Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) guidance, rather than the 2 exchanges per hour indicated by RIVM guidance for bathrooms. 25 The more conservative value (0.2/h) appears to be more appropriate to represent low-end air-exchange rates in homes in the United States, in which ventilation fans may not be used routinely. No default values were available specifically for pump hair spray products. Thus, the spray duration assumed for propellant hair sprays (14.4 s) and default values for pump toilet-water sprays were used in the calculations for pump hair sprays.
Exposure Concentrations and Margins of Safety (MOSs) for Hair Spray Products Calculated Using the Cons Expo Web Model (version 1.0.1). 27
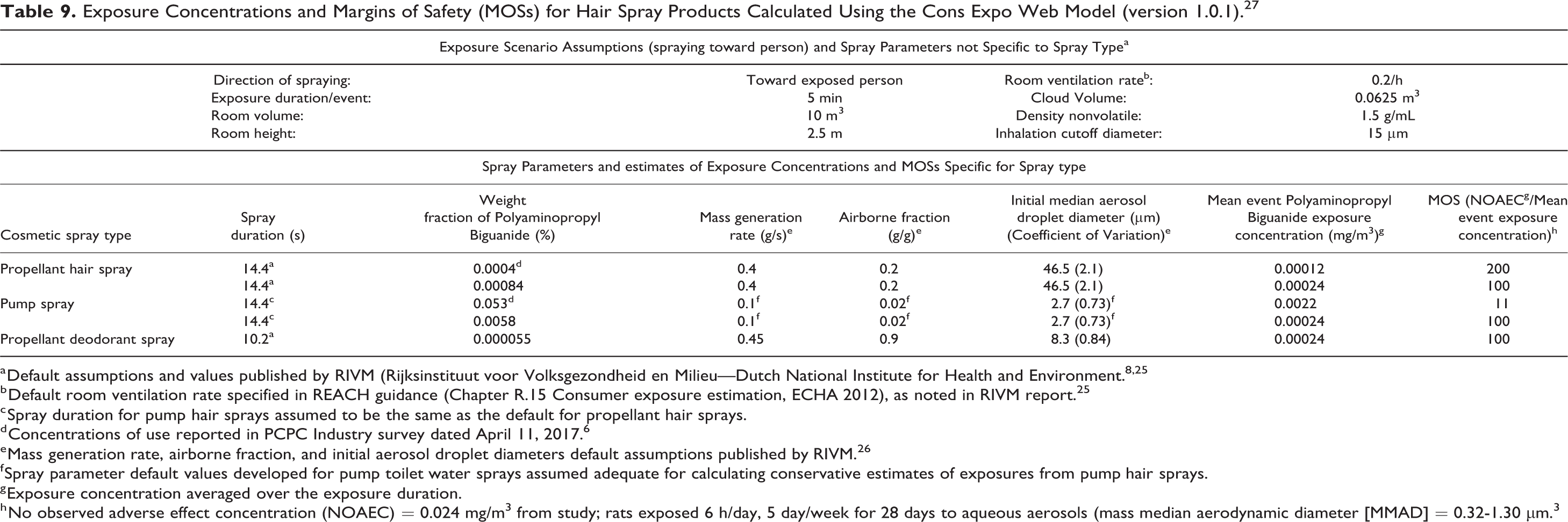
a Default assumptions and values published by RIVM (Rijksinstituut voor Volksgezondheid en Milieu—Dutch National Institute for Health and Environment. 8,25
b Default room ventilation rate specified in REACH guidance (Chapter R.15 Consumer exposure estimation, ECHA 2012), as noted in RIVM report. 25
c Spray duration for pump hair sprays assumed to be the same as the default for propellant hair sprays.
d Concentrations of use reported in PCPC Industry survey dated April 11, 2017. 6
e Mass generation rate, airborne fraction, and initial aerosol droplet diameters default assumptions published by RIVM. 26
f Spray parameter default values developed for pump toilet water sprays assumed adequate for calculating conservative estimates of exposures from pump hair sprays.
g Exposure concentration averaged over the exposure duration.
h No observed adverse effect concentration (NOAEC) = 0.024 mg/m3 from study; rats exposed 6 h/day, 5 day/week for 28 days to aqueous aerosols (mass median aerodynamic diameter [MMAD] = 0.32-1.30 μm.3
The use of conservative default values for multiple exposure parameters ensures that high-end, “reasonable worst-case” exposures are calculated. 8,25 Generally, the exposure concentrations predicted by the ConsExpo Model increase with increasing spray durations and decrease with increasing exposure durations/event (ie, the time over which the exposure concentrations are averaged after each spraying event).
The average Polyaminopropyl Biguanide inhalation exposure concentrations over the 5-minute default exposure duration/event were 0.00012 mg/m3 for propellant hair sprays and 0.0022 mg/m3 for pump hair sprays.
The NOAEC was approximately 0.024 mg/m3 in a 28-day inhalation study in which rats were exposed, nose only, to Polyaminopropyl Biguanide in an aerosolized water solution, 6 h/day, 5 days/week. 3 Margin of safety values were calculated by dividing the NOAEC by the average inhalation exposure concentrations/event estimated using the ConsExpo model. The MOS values were 200 for propellant hair sprays and 11 for pump hair sprays.
An MOS of 100 may be considered to be adequate to allow for the uncertainties associated with using the NOAEC from a short-term rat study to evaluate potential chronic human exposures (ie, 10 for short-term to long-term exposure extrapolation × 10 for interspecies extrapolation = 100). Accordingly, the ConsExpo Web model was used to calculate concentrations of use that would yield an MOS of 100 for Polyaminopropyl Biguanide in pump and propellant hair spray products and propellant deodorant products. The results indicate that use concentrations of 0.0058% in pump hair sprays, 0.00084% in propellant hair sprays, and 0.000055% in propellant deodorant sprays would each be associated with an MOS of 100.
The daily exposure duration in the rat study (6 hours) from which the NOAEC was derived (ie, 6 h/day or 360 min/day) is 72 times greater than the exposure duration of a person using a hair spray once a day (1 event/day × 5 min/event = 5 min/day), 5 days per week, and 24 times greater than the exposure duration of a person using a hair spray 3 times a day 5 days/week.
The daily exposure duration in the rat study is about 7 times greater than the exposure duration would be for a beautician applying hair spray to customers, an average of 10 times a day, 5 days/week. The beautician’s occupational exposure may be reduced by workplace ventilation systems and larger room volumes, as well as the direction of the spraying (ie, away from the beautician).
Risk assessment—various exposure routes
The US EPA assessed the human health risks associated with residential-handler and post-application pesticide exposure scenarios (including pesticides containing Polyaminopropyl Biguanide) using surrogate exposure data, maximum application rates (specified on the product labels), and standard assumptions. 14 Residential handler exposures may occur when individuals mix, load, or apply a pesticide. Post-application exposures may occur to bystanders affected during or after application by others. The agency determined that all margins of exposure (MOEs) from dermal and inhalation exposure for residential handlers are above the protective target of at least 100 and, therefore, were not concerning. For post-application dermal and incidental ingestion (oral exposures) scenarios, MOEs calculated based on an oral NOAEL of 20 mg/kg/d were also above the EPA’s level of concern.
Chronic dietary risk estimates were provided for the general US population and all population subgroups. 14 The chronic Population Adjusted Dose (cPAD) is the level of exposure (mg/kg/d) that the EPA determines should not be exceeded. These estimates were below EPA’s level of concern for the general US population (ie, < 10% of the cPAD) and all population subgroups (ie, < 37% of the cPAD for children). Therefore, the chronic dietary risk is not of concern.
1The aggregate risk assessment integrates the assessments that were conducted for dietary and residential exposure. Aggregate calculations were performed for adults and children using the Aggregate Risk Index (ARI) method. Aggregate Risk Index values were greater than 1.2 for children and greater than 5.4 for adults, and these risks were determined not to warrant the EPA’s concern. 14 As a general rule, an ARI of ≥ 1 is of little concern, but an ARI of < 1 suggests a risk that is of concern.
Developmental and Reproductive Toxicity Studies
The developmental and reproductive toxicity studies summarized below are presented in Table 10.
Developmental and Reproductive Toxicity Studies.

Abbreviations: NOAEC, no observed adverse effect concentrations; NOAEL, no observed adverse effect level; SPF, specific pathogen free.
An NOAEL of 10 mg/kg/d for developmental toxicity was reported in an oral dosing (by gavage on gestation days 6 through 15) study on 20% aqueous Polyaminopropyl Biguanide involving mice. 18,28 In oral reproductive and developmental toxicity studies on 20% aqueous Polyaminopropyl Biguanide in rats, NOAECs of 1000 ppm (after feeding in diet on gestation days 1 through 20) 18 and 1300 ppm (after feeding in diet during a 9-day premating period and until the 3rd generation) 18,21 have been reported. An NOAEL of 2000 ppm for reproductive and development effects was reported in a study in which rats were fed (through 2 successive generations) with 20.2% aqueous Polyaminopropyl Biguanide at dietary concentrations up to 2000 ppm. 18 Polyaminopropyl Biguanide has been classified as embryotoxic at a dosage rate of 100 mg/kg/d (rats; protocol not stated). 29 An NOAEL of 40 mg/kg/d for developmental toxicity has been reported in an oral dosing (20.2% aqueous Polyaminopropyl Biguanide by gavage on gestation days 8 through 20) study involving rabbits. 18 Polyaminopropyl Biguanide (0.04% in polyethylene glycol [PEG]) has been classified as embryotoxic in rabbits at an oral dosage rate of 32 mg/kg/d (animal strain and dosing protocol not stated). 29 Polyaminopropyl Biguanide (concentration not stated) has been classified as teratogenic in rats at an intraperitoneal dosage rate of 10 mg/kg/d (dosing protocol not stated). 29 In an inhalation study on 20% aqueous Polyaminopropyl Biguanide, degeneration of seminiferous tubules in the testis of 1 male rat was observed after exposure to 0.25 mg/m3 (6 h/day, 5 days/week for 3 weeks), but this was not observed in any other group, including the group exposed to the highest concentration (26 mg/m3). 18
Genotoxicity Studies
The genotoxicity studies (in vitro and in vivo) summarized below are presented in Table 11.
Genotoxicity Studies.
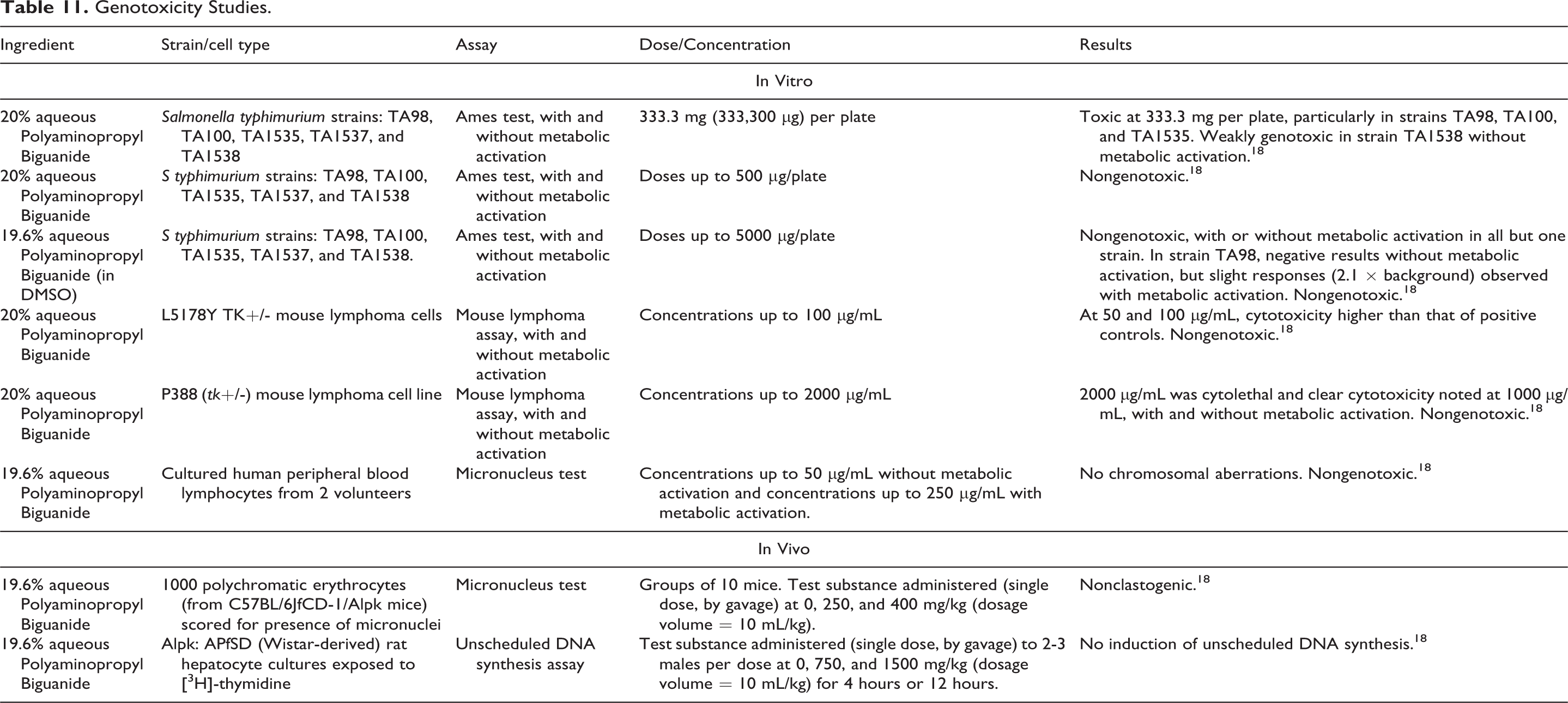
In Ames tests, ∼20% Polyaminopropyl Biguanide was nongenotoxic at doses up to 5000 µg/plate, with and without metabolic activation. 18 At the highest dose evaluated (333,300 µg/plate) in the Ames test, Polyaminopropyl Biguanide was weakly genotoxic in Salmonella typhimurium strain TA 1538 without metabolic activation. Polyaminopropyl Biguanide was nongenotoxic in a mouse lymphoma assays at concentrations up to 2000 µg/mL with and without metabolic activation, or in an in vitro micronucleus test (cultured human peripheral blood lymphocytes) at concentrations up to 50 µg/mL (without metabolic activation) and up to 250 µg/mL (with metabolic activation). In an in vivo micronucleus test, Polyaminopropyl Biguanide was nonclastogenic in polychromatic erythrocytes from mice that received single oral dosages up to 400 mg/kg. In an in vivo unscheduled DNA synthesis assay, there was no induction of unscheduled DNA synthesis in hepatocytes from rats that received single oral doses up to 1500 mg/kg.
Carcinogenicity Studies
The carcinogenicity studies (in vitro, dermal, and oral) summarized below are presented in Table 12.
Carcinogenicity Studies.

Abbreviations: ADME, Absorption, Distribution, Metabolism, and Excretion; NOAEL, no observed adverse effect level; SCCS, Scientific Committee on Consumer Safety; SPF, specific pathogen free.
In Vitro
Polyaminopropyl Biguanide (20%) was evaluated at concentrations up to 3000 µg/mL in the cell transformation assay (using baby hamster kidney fibroblasts); there was no difference in the number of transformed cell colonies between test and negative control cultures. 18 In another assay, RAW 264.7 mouse macrophages (a macrophage-like, Abelson leukemia virus transformed cell line derived from BALB/c mice) were cocultured with SVEC-10 mouse liver endothelial cells in various experimental conditions: preactivation of macrophages with Polyaminopropyl Biguanide or lipopolysaccharide (LPS) and/or coculture in presence of Polyaminopropyl Biguanide. 3 Polyaminopropyl Biguanide, tested at concentrations up to 1 ppm, had no direct effect on liver cell proliferation and did not potentiate cell proliferation induced by activated macrophages.
Animal
Dermal
Polyaminopropyl Biguanide was classified as a hepatocarcinogen in mice at the highest dose tested in a study in which Polyaminopropyl Biguanide in ethanol was applied to the skin daily at doses up to 750 mg/kg/d (5 days/week) for 80 weeks. 21 The NOAEL was 0.6 mg/mouse/day (15 mg/kg/d). A variety of inflammatory hepatic changes was observed in all groups, including the controls. However, at 750 mg/kg/d, severe hepatitis was observed in some of the animals. These hepatic changes appeared to have been mainly responsible for causing increased numbers of deaths in the high-dose group. (Additional study results are included in the 80-week chronic dermal toxicity study that is summarized earlier in this safety assessment.) A scientific advisory panel advising the SCCS indicated that the hepatitis observed in this study may be attributable to the Helicobacter hepaticus infections, which may also be responsible for the increased incidence of hepatocellular neoplasms in these animals.
Oral
Mice were fed 20.2% Polyaminopropyl Biguanide in the diet (up to 1000 ppm active ingredient; feeding 1 week prior to mating and during mating [males and females]). 18 The offspring of these mice were also fed 20.2% Polyaminopropyl Biguanide at concentrations up to 1000 ppm active ingredient in the diet for 97 weeks. Except for vascular tumors, there were no treatment-related (nonneoplastic or neoplastic) increases in histopathologic findings. Vascular tumors (hemangiosarcomas or hemangiomas) in the liver or other sites and a high mortality incidence (80%) were reported by week 97. A dose-related increase in the liver vascular tumor incidence was reported. After reviewing the results of this study, the SCCS concluded that the data are considered to be of low reliability due to the high mortality rate that was reported. When mice were fed Polyaminopropyl Biguanide at dietary concentrations up to 4000 ppm for up to 28 days, increased cell proliferation in a concentration-related manner was noted at 1200 ppm and 4000 ppm. 3 In another study, a statistically significant increase in the incidence of hemangiosarcomas and hemangiomas was reported in male mice (C57B1/10J/CD-1 strain) that received Polyaminopropyl Biguanide at a dietary concentration of 4000 ppm daily for 2 years. 18 A low incidence of hemangioma (2 of 64 males; 2 of 64 females) and hemangiosarcoma (1 of 64 females) was reported in a study in which rats were fed Polyaminopropyl Biguanide at a dietary concentration of 2000 ppm for 2 years. 30 In another 2-year study, Polyaminopropyl Biguanide was administered (in drinking water or in diet) to groups of rats. 20 Hepatocellular tumors were induced at concentrations (in water) of 1000 mg/L and 1500 mg/L, but not at a concentration of 500 mg/L (in water). Administration of Polyaminopropyl Biguanide in diet did not cause increase in hepatocellular tumors. The hypothesized mode of action (MOA) for liver tumors induced by Polyaminopropyl Biguanide in drinking water involves increased hepatocyte proliferation and induction of hepatocellular foci and tumors. 20 Polyaminopropyl Biguanide was classified as noncarcinogenic in rats fed dietary concentrations up to 2000 ppm for 122 weeks; mortality was 80%. 21 In a feeding study in which rats were fed Polyaminopropyl Biguanide at concentrations up to 2000 ppm, 80% mortality was reported at 124 weeks. 18 A low incidence of hemangiomas or hemangiosarcomas (mostly in lymph nodes) was observed in the groups of remaining animals (7 groups, with 8 to 21 rats/group; 1 animal with a hemangioma or hemangiosarcoma per group).
Other Relevant Studies
Effect on Lung Cells
A study was performed to characterize the inflammatory responses, including the mechanism of action, induced in lung cells exposed to Polyaminopropyl Biguanide. 31 A549 cells that were exposed to Polyaminopropyl Biguanide showed concentration-dependent (0 to 80 μg/mL) decreased viability, significant reactive oxygen species (ROS) generation (at 20 μg/mL), inflammatory cytokine secretion (statistically significant increase in tumor necrosis factor alpha [TNF-α] release at 20 μg/mL), and nuclear factor kappa B (NF-κB) activation (expression of IκB-α protein significantly degraded at concentrations > 10 μg/mL). Statistically significant cytotoxicity to A549 cells was observed at concentrations >10 μg/mL. Polyaminopropyl Biguanide triggered inflammatory cytokine secretion and NF-κB activation by modulating the degradation of IκB-α and through the accumulation of nuclear p65. It was noted that TNF-α plays important roles in interleukin 8 (IL-8) expression as well as in NF-κB activation. Interleukin 8 production induced by Polyaminopropyl Biguanide was completely suppressed by an NF-κB inhibitor, but not by an ROS scavenger. The authors suggested that Polyaminopropyl Biguanide induces inflammatory responses via the NF-κB signaling pathway.
Other Cellular Effects and Antimicrobial Activity
Polyaminopropyl Biguanide (PHMB; C6) was compared to the (structurally) closely related polyaminopropyl biguanide (C3) with respect to antiseptic efficacy and cytotoxicity in vitro. 32 Antimicrobial efficacy tests were performed via determination of the minimum bactericidal concentration. Polyaminopropyl Biguanide (PHMB; C6) exhibited high antimicrobial activity against Staphylococcus aureus and Escherichia coli (minimal bactericidal concentration ≤ 0.05 mg/mL [0.005%]), whereas polyaminopropyl biguanide (C3) proved to be ineffective in bacterial eradication. These results suggest that even small differences in the chemical structure of related agents, such as Polyaminopropyl Biguanide (PHMB; C6) and polyaminopropyl biguanide (C3), can substantially affect their efficacy.
Cytotoxicity was evaluated in human keratinocytes (HaCaTs) and murine fibroblasts (L929). 32 In fibroblast or keratinocyte cultures, concentrations for both test substances ranged from 0.005% to 1% vol/vol. Polyaminopropyl biguanide (C3) was also tested at concentrations ranging from 0.25% to 3% vol/vol. Cultures were incubated for up to 72 hours. For all tested concentrations, Polyaminopropyl Biguanide (PHMB; C6) was highly cytotoxic to human HaCaT and L929 murine fibroblast cell after 24 and 72 hours of incubation, never exceeding a survival rate of 27%. Polyaminopropyl biguanide (C3) displayed significantly lower cytotoxicity at concentrations ranging from 0.005% to 0.1% vol/vol. At concentrations up to 0.1%, no cytotoxic effect could be detected in L929 cells after 24 hours, whereas, for HaCaT cells, moderate and high cytotoxicity was evident at 0.05% and 0.1% polyaminopropyl biguanide (C3). After 72 hours, only a weak cytotoxic effect on L929 cell at 0.05% and 0.1% polyaminopropyl biguanide (C3) could be observed, while, for HaCaT cells, concentrations up to 0.1% were classified as noncytotoxic. However, concentrations ≥ 0.25% polyaminopropyl biguanide (C3) were highly cytotoxic to cells of both cell lines after 24 hours of incubation. When compared directly, polyaminopropyl biguanide (C3) consistently resulted in a significantly higher cell survival rate than Polyaminopropyl Biguanide (PHMB; C6), irrespective of concentration and incubation time (P ≤ 0.0006).
It has been hypothesized that exposures to Polyaminopropyl Biguanide may have epigenetic effects, including nongenotoxic DNA base modifications (eg, changes in DNA-base methylation) and altered mitogenic cytokine production. 33 These effects have been assessed in vitro using 3 cell types: Caco-2 cells (from a human colon adenocarcinoma) with a p53 nonfunctional gene (Δp53: mut p53), Neuro-2A (mouse neuroblastoma cells), and HepG2 cells (human hepatocellular carcinoma) with functional p53 genes. The studies focused mainly on Polyaminopropyl Biguanide effects on the liver, but also examined the gut and brain since these are also the target organs. At Polyaminopropyl Biguanide concentrations of 1μg/mL to 20 μg/mL, neither a growth stimulatory effect nor a growth inhibitory effect was observed. Viability testing using neutral red resulted in an IC50 of 20 to 25 μg/mL after treatment with Polyaminopropyl Biguanide for 3 hours, whereas the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cell viability test led to IC50 of 80 μg/mL, 160 μg/mL, and 160 μg/mL for HepG2 cells, Neuro-2A cells, and Caco-2 cells, respectively. The neutral red test showed that the cell lines had similar sensitivity to Polyaminopropyl Biguanide at much lower concentrations than with the MTT assay, indicating that the cellular target is the membrane. (The principle of neutral red test is based on the integrity of the cellular, lysosomal and endosomal membrane, while the MTT test is an indicator of metabolic activity in living cells.) Polyaminopropyl Biguanide does not induce significant oxidative stress (as determined by measuring production of malondialdehyde or lipoperoxidation, nor does it induce hydroxylation of DNA (8-hydroxy-2′-deoxyguanosine) and/or its hypermethylation (5-methylcytosine [m5dC] content), the latter being strongly implicated in DNA replication and regulation and cell division.
Additional results from this study indicated that Polyaminopropyl Biguanide did not induce significant production of mitogenic cytokines, such as TNF-α (tumor necrosis factor-alpha), interleukins (IL-1 alpha), and NF-κB, which can cause either apoptosis or stimulate the growth of transformed cells or tumors. Instead, concentrations of 20 to 100 μg/mL Polyaminopropyl Biguanide killed cells of all types in less than 3 hours. The expression of genes involved in the mechanisms of cell death induced by Polyaminopropyl Biguanide, including p53, the pro apoptotic gene bax and others, and the anti-apoptotic bcl-2 and caspase-3 genes, has been evaluated using reverse transcription polymerase chain reaction methodology. Results indicated that it does not appear that Polyaminopropyl Biguanide-induced cell death is the result of apoptosis, but, rather, is cytotoxic at the cell membrane level, resulting in necrotic cell death. Finally, there was no apparent inhibition of GAP-junctions (ie, gap junctional intercellular communication) in the presence of Polyaminopropyl Biguanide. Taken together, the data indicate that Polyaminopropyl Biguanide did not exhibit clear or remarkable epigenetic effects, except for a slight increase in the levels of some cytokines and a transcription factor at concentrations that cause rapid cell lysis.
Dermal Irritation and Sensitization Studies
The skin irritation, sensitization, and phototoxicity/photosensitization studies summarized below are presented in Table 13.
Dermal Irritation and Sensitization Studies.

Abbreviations: HRIPT, human repeated insult patch test; SPF, specific pathogen free.
Irritation
In a study involving mice, the highest dose of Polyaminopropyl Biguanide (10% concentration in ethanol, 30 mg dose) caused hyperkeratosis and, occasionally, ulceration extending into the dermis when applied repeatedly for 80 weeks. 21 Polyaminopropyl Biguanide (0.04%) was classified as a nonirritant when applied to the skin of rats for 24 hours. Repeated applications of 20.2% aqueous Polyaminopropyl Biguanide to rats for 21 days resulted in slight skin irritation (at 60 mg/kg/d) and moderate irritation (at 200 mg/kg/d). 18 Severe skin irritation was observed in all rats that received a single 24-hour application of 25% aqueous Polyaminopropyl Biguanide at dosages of 2.5 mL/kg and 5 mL/kg. 21 Focal ulceration and edema were observed in rats after 25% aqueous Polyaminopropyl Biguanide was applied repeatedly to the skin. Repeated 23-hour applications of Polyaminopropyl Biguanide (12,000 ppm; 1 mL per application) to the skin of rabbits for 21 days were not irritating. Single applications (24 hours) of 20% aqueous Polyaminopropyl Biguanide to rabbits indicates that this compound is noncorrosive, but moderately irritating, to intact skin, and severely irritating to abraded skin. 18 Polyaminopropyl Biguanide (20% aqueous) induced erythema at abraded, but not intact, skin sites in rabbits, and was classified as a noncorrosive material. Moderate erythema was observed after 20% aqueous Polyaminopropyl Biguanide was applied for 24 hours to the skin of rabbits. Mild skin irritation resulted after Polyaminopropyl Biguanide (96%, as powder) was applied to the skin of rabbits for up to 4 hours. 3 Polyaminopropyl Biguanide (0.5 g, moistened with water; single 4-hour application) was also classified as a mild skin irritant in rabbits. 18 Slight to moderate erythema was observed in guinea pigs that received repeated applications of 25% aqueous Polyaminopropyl Biguanide for 3 days. 18
Polyaminopropyl Biguanide (up to 1.5% active) was not classified as a primary skin irritant when applied for 24 hours to the skin of human subjects (17 males and 28 females). 18 A bacterial nonocclusive dressing loaded with 1% wt/vol sericin and 0.3% wt/vol Polyaminopropyl Biguanide was classified as nonirritating after application to the skin of 105 subjects for 3 days. 34
Sensitization
Results of a local lymph node assay (LLNA) were positive with Polyaminopropyl Biguanide. 35,36 In maximization tests on Polyaminopropyl Biguanide, moderate skin sensitization was observed in guinea pigs induced with 0.06% active ingredient (intradermal injection) and 20.2% active ingredient (occlusive application) and challenged with Polyaminopropyl Biguanide (20.2% active ingredient) and a 30% solution of the ingredient (6% active ingredient) in deionized water, and moderate to strong sensitization was observed in guinea pigs induced with 0.2% active ingredient (intradermal injection) and 20.2% active ingredient (topical application) and challenged with Polyaminopropyl Biguanide (20.2% active ingredient). 18 In another guinea pig maximization test, sensitization was not observed in guinea pigs induced with 0.15% (intradermal injection) and 20% Polyaminopropyl Biguanide (topical application) and challenged with 10% or 20% Polyaminopropyl Biguanide. In one Buehler test on Polyaminopropyl Biguanide, guinea pigs were induced with 2% active ingredient (topical application), challenged with 2% active ingredient, and rechallenged with 0.2%, 2%, and 4% active ingredient. The initial challenge with 2% active ingredient and rechallenge with 2% and 4% active ingredient resulted in faint erythema; rechallenge with 0.2% active ingredient produced negative results. Polyaminopropyl Biguanide (2% active ingredient) was classified as a moderate sensitizer. In another Buehler test, it was determined that the threshold for eliciting sensitization in guinea pigs was ∼ 1%. Induction concentrations ranged from 0.3% to 5% and challenge concentrations ranged from 0.075% to 15%. Results from a study evaluating the possible cross-reactivity of Polyaminopropyl Biguanide (challenge with 20%) with chlorhexidine (challenge with up to 4% chlorhexidine gluconate) in guinea pigs were negative.
In a human repeated insult patch test (HRIPT; 191 subjects), it was determined that 20% aqueous Polyaminopropyl Biguanide (2% active ingredient; effective concentration = 0.4%) was not capable of causing primary skin irritation, but was capable of causing sensitization. 3 When a leave-on product containing 20% Polyaminopropyl Biguanide (tested at 0.5%, effective concentration 0.1% Polyaminopropyl Biguanide) was evaluated in an HRIPT involving 207 subjects, it was concluded that the product did not induce dermal sensitization. 37 In another HRIPT (115 subjects; any ethnicity, provided that their degree of skin pigmentation did not significantly interfere with evaluations) on a neck cream containing 0.2% Polyaminopropyl Biguanide, the product did not cause clinically meaningful irritation or sensitization. 38 In another study, the skin sensitization potential of 20% Polyaminopropyl Biguanide (diluted with distilled water to 1% vol/vol prior to testing; 750 µg/cm2) was evaluated using semi-occlusive patches in an HRIPT involving 108 subjects (Asian [∼2%], Biracial [∼3%], Black [∼ 23%], Caucasian [∼ 33%], and Hispanic [∼39%]; Fitzpatrick skin types not stated). 39 The authors concluded that Polyaminopropyl Biguanide did not induce dermal sensitization in the subjects tested, and a no expected sensitization induction level (NESIL, determined based on data from this HRIPT) of 750 µg/cm2 was used to calculate quantitative risk assessments (QRA) on various product types.
Risk Assessment
A series of QRAs was performed by industry in response to the Panel’s concerns about sensitization potential. 40 The QRA for contact dermatitis with Polyaminopropyl Biguanide in cosmetics utilized a NESIL of 1000 µg/cm2, which supports the use of this ingredient at concentrations of ≤ 0.1%. Among the human data that were used to derive the NESIL was a phototoxicity/photosensitization study (described below) involving 26 subjects tested with 1% Polyaminopropyl Biguanide at a dose of 1000 µg/cm2, the highest nonsensitizing dose in relation to all of the HRIPT data that were considered. The NESIL of 1000 µg/cm2 was used to determine whether estimated exposure, using maximum use concentrations from a Council survey, could be considered safe. The ratio of AEL (Acceptable Exposure Level)/CEL (Consumer Exposure Level) was > 1, except for the product that contained 0.2% Polyaminopropyl Biguanide (Table 14).
Quantitative Risk Assessment (QRA) Worksheet, Utilizing a NESIL of 1000 µg/cm2. 40
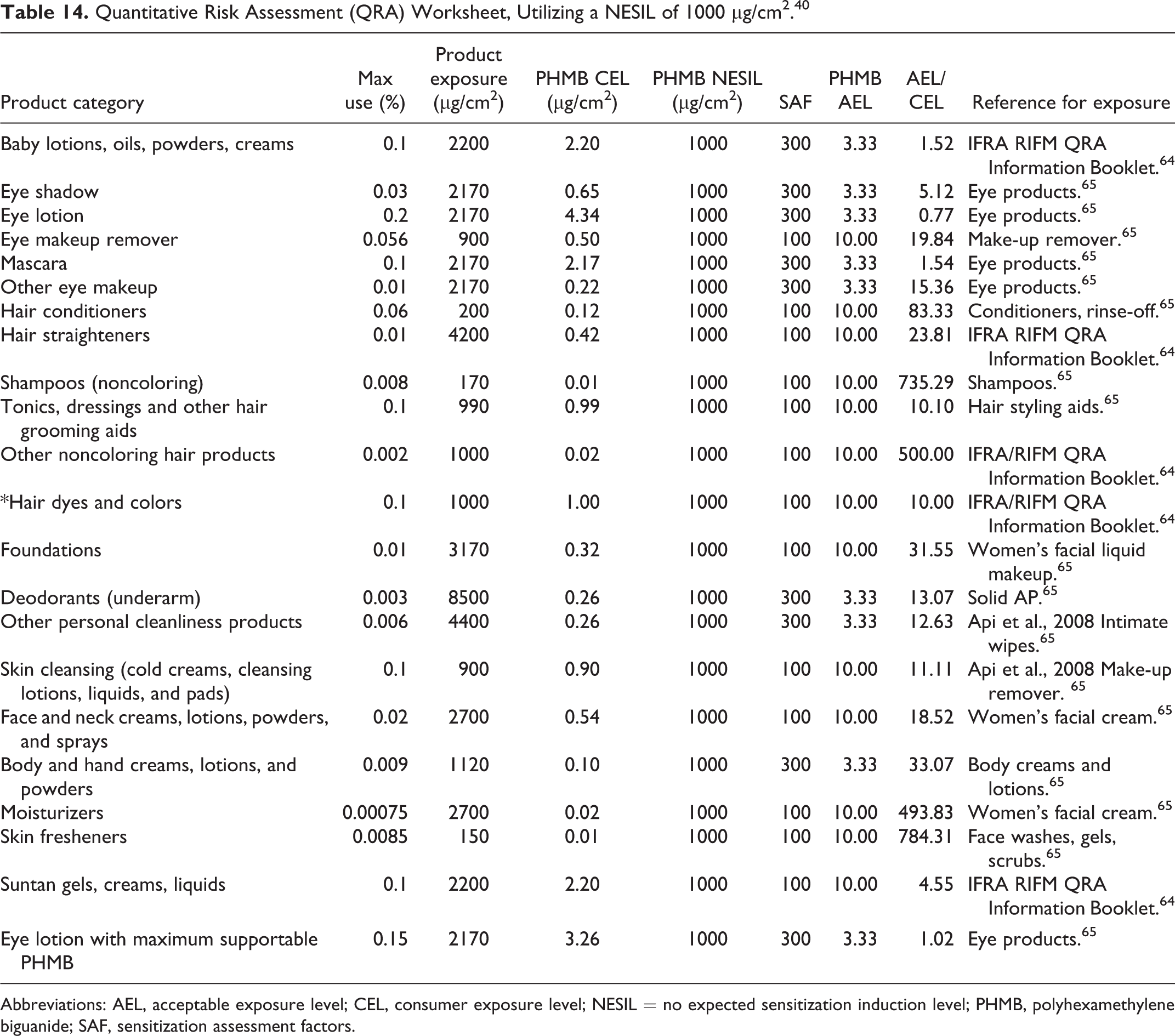
Abbreviations: AEL, acceptable exposure level; CEL, consumer exposure level; NESIL = no expected sensitization induction level; PHMB, polyhexamethylene biguanide; SAF, sensitization assessment factors.
A second QRA, performed by industry, utilized a NESIL of 750 µg/cm2. The human data that were used to derive the NESIL are from an HRIPT involving 108 subjects (Asian [∼2%], Bi-racial [∼3%], Black [∼ 23%], Caucasian [∼ 33%], and Hispanic [∼39%]) tested with 1% vol/vol Polyaminopropyl Biguanide, that is summarized earlier in this report. 39 This QRA resulted in similar ratios of AEL/CEL values >1, and the same exception for the product that contained 0.2% Polyaminopropyl Biguanide (Table 15). 41
Quantitative Risk Assessment (QRA) Worksheet, Utilizing a NESIL of 750 µg/cm2. 41
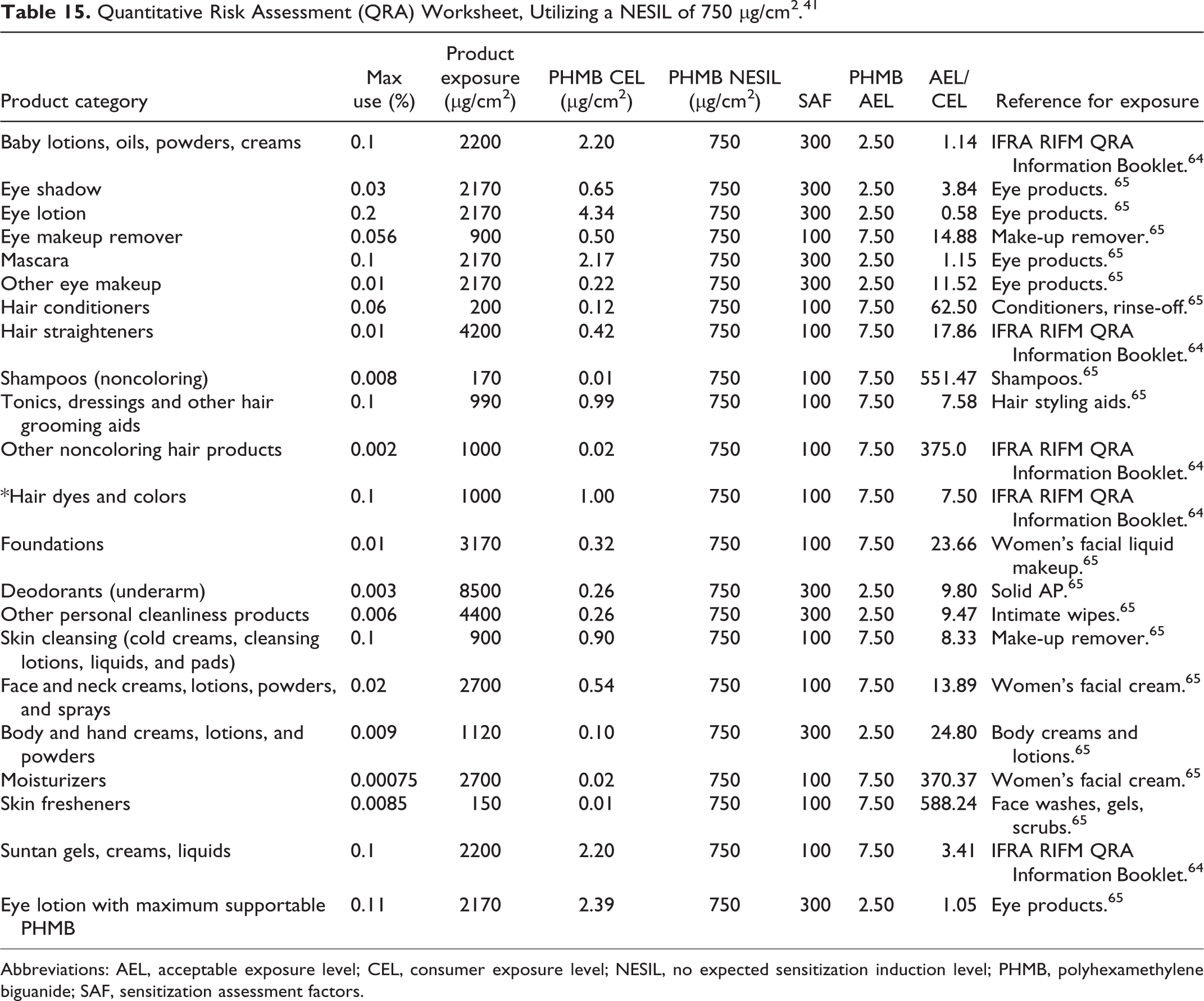
Abbreviations: AEL, acceptable exposure level; CEL, consumer exposure level; NESIL, no expected sensitization induction level; PHMB, polyhexamethylene biguanide; SAF, sensitization assessment factors.
Photosensitization/Phototoxicity
Animal
The photoirritation potential of 20% aqueous Polyaminopropyl Biguanide was studied using 10 male rats. 21 The following two concentrations of the test substance (in distilled water) were evaluated: 10% (effective concentration = 2%) and 25% (effective concentration = 5%). Each test concentration (0.1 mL) was applied to dorsal skin once daily for 4 days. The test site was irradiated with short-wave ultraviolet light (UVC; black lamp) for 3 hours daily. Very strong irritation potential, but no significant photoirritancy, was reported in a study in which male rats were tested with Polyaminopropyl Biguanide at concentrations of 2% and 5%.
Human
A phototoxicity/photoallergenicity study on 20% aqueous Polyaminopropyl Biguanide was performed using 26 male and female subjects. 21 The test substance was diluted (1:20 in water; effective concentration = 1%) prior to application. Patches (20 mm × 20 mm Webril pad affixed to a 40 mm × 40 mm adhesive square) were moistened with 0.4 mL of the test substance (dose = 1 mg/cm2) 42 and applied to the upper arm for 24 hours, 3 times per week for 4 successive weeks. Immediately after patch removal, the sites were exposed to direct rays of mid-day sun for 1 hour. The challenge application occurred at week 6. Polyaminopropyl Biguanide was essentially nonirritating and did not induce sensitization, phototoxicity, or photoallergenicity.
Ocular Irritation Studies
The ocular irritation studies summarized below are presented in Table 16.
Ocular Irritation Studies.
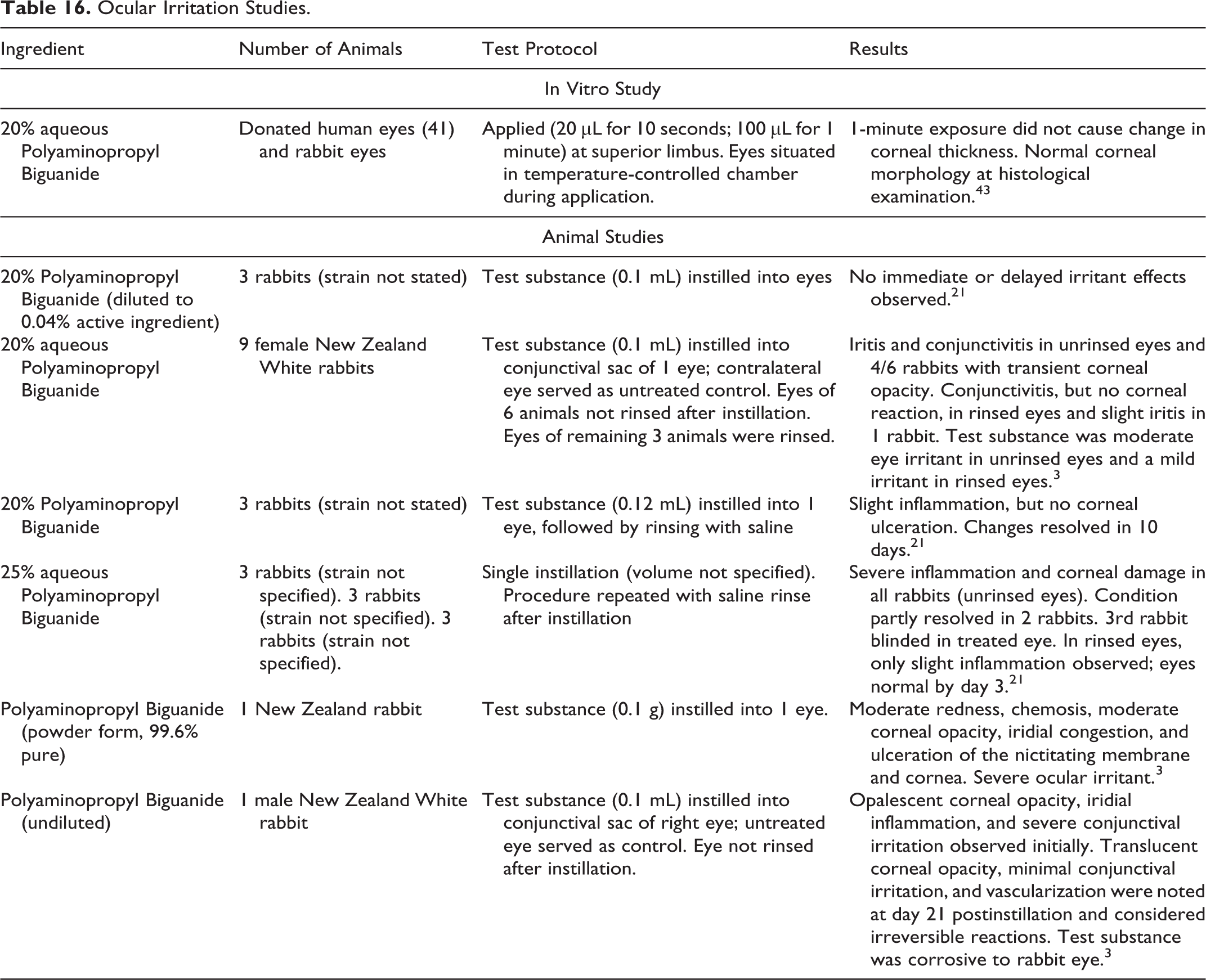
Undiluted Polyaminopropyl Biguanide was a severe ocular irritant/corrosive agent when instilled into rabbit eyes. 3 The instillation of 25% aqueous Polyaminopropyl Biguanide into the eyes of rabbits resulted in severe inflammation and corneal damage in unrinsed eyes and slight inflammation in rinsed eyes. 21 Moderate and mild ocular irritation were observed in unrinsed and rinsed rabbit eyes, respectively, after 20% aqueous Polyaminopropyl Biguanide was instilled. 3 In another study involving rabbits, the instillation of Polyaminopropyl Biguanide (25% aqueous) into the eyes induced slight inflammation, but no corneal ulceration. 21 Ocular irritation was not observed when Polyaminopropyl Biguanide (0.04% active ingredient) was instilled into the eyes of rabbits. 21 In a study in which 20% aqueous Polyaminopropyl Biguanide (100 µL) was instilled into human eyes (from cadavers) and the eyes of rabbits in a temperature-controlled chamber (32 °C-36 °C), normal corneal morphology was observed at histological examination. 43
Clinical Studies
The patient multicenter studies summarized below are presented in the Human Sensitization Studies section of Table 13. In another type of clinical study, no adverse effects were noted following the exposure of 29 patients to a preoperative antiseptic for cataract surgery that contained 0.2% Polyaminopropyl Biguanide. 44
Retrospective and Multicenter Studies
In a multicenter study involving 374 patients (United Kingdom study) patch tested with 2.5% aqueous Polyaminopropyl Biguanide, 2 sensitization reactions were reported. 45,46 In a second multicenter study (German study) involving 1554 patients, sensitization reactions were observed in 6 patients patch tested with 0.5% Polyaminopropyl Biguanide. 47 It was noted that this initial series of data suggested that the baseline frequency of Polyaminopropyl Biguanide sensitization was very low (0.5% and 0.4% in the United Kingdom and German studies, respectively). The majority of positive reactions were considered weak. It was noted that these data suggested that Polyaminopropyl Biguanide may not be a relevant contact allergen.
In a subsequent German multicenter study involving 1975 patients, 10 patients had sensitization reactions to 0.5% Polyaminopropyl Biguanide (20% aqueous Polyaminopropyl Biguanide tested at 2.5% concentration) and 16 patients had sensitization reactions to 1% Polyaminopropyl Biguanide. 48 The majority of the positive reactions were considered weak. When results of the 3 studies were considered together, it was noted that the frequency of sensitization reactions to Polyaminopropyl Biguanide remained low and stable, in spite of the use of Polyaminopropyl Biguanide in underarm deodorants.
Contact Urticaria
A female patient experienced grade III anaphylaxis (immunoglobulin E [IgE]-mediated mechanism confirmed) with palmar pruritus, flush, swelling of lips, swallowing difficulties, hypotension, and loss of consciousness while using a new brand of wet toilet paper containing Polyaminopropyl Biguanide as a disinfectant. 17,49 The detailed allergy history of the patient indicated 3 prior anaphylactic episodes (grade II) during wound care of a leg ulcer. One of the episodes occurred after the use of a wound dressing that contained Polyaminopropyl Biguanide. The other 2 episodes occurred after wound cleansing with 2 different Polyaminopropyl Biguanide disinfectants, one of which contained Polyaminopropyl Biguanide, PEG 4000, and no other additives. The composition of the other disinfectant that contained Polyaminopropyl Biguanide was not detailed. However, according to another publication, the composition of that disinfectant (liquid and gel) is as follows: 0.1% Polyaminopropyl Biguanide, 0.1% undecylenamidopropyl betaine, and water; the gel also contains glycerol and hydroxyethyl cellulose. 50 The patient had no known allergies or atopic diseases. Skin prick tests were positive for the disinfectant of known composition, which was tested in a 1:10 dilution, corresponding to 20 µg/mL Polyaminopropyl Biguanide. Positive skin prick test results were also reported for chlorhexidine in different commercial preparations. Skin prick test results for PEG 4000 were negative, and the same was true for the 5 healthy volunteers who were prick tested with the disinfectant of known composition. Whether or not the other disinfectant containing Polyaminopropyl Biguanide was evaluated in prick tests was not mentioned. Other results reported in this case report indicated that there was limited in vitro cross-reactivity between Polyaminopropyl Biguanide and chlorhexidine. The author noted that patients with known chlorhexidine allergy could be at risk for anaphylactic reactions to Polyaminopropyl Biguanide.
A male patient (atopic and diabetic) had a history of angioedema and pruritus after using wet wipes. 13 Patch test results for an ingredient of the wipes, Polyaminopropyl Biguanide (tested at 1:10 in water), and the wipe itself were negative. However, prick tests resulted in strong positive reactions to the wipe and this ingredient after 15 minutes, and the reactions continued to increase in intensity during the following 2 hours.
The prick test (protocol and test concentration not specified) was used to diagnose immediate contact urticarial reactions in 44 patients with eczematous dermatitis. A positive reaction to Polyaminopropyl Biguanide was observed in 1 patient. 51
Two cases of severe anaphylaxis were reported following contact of a surgical wound with a hospital disinfectant containing 0.2% Polyaminopropyl Biguanide. 52 Immediate-type hypersensitivity to Polyaminopropyl Biguanide was suggested by positive skin prick tests in both patients and by negative skin tests in control individuals. Skin tests involving chlorhexidine were negative.
The case of a 77-year-old female patient who suffered from severe anaphylaxis during wound (leg ulcer) care was presented. 53 The results of an allergologic evaluation indicated specific IgE antibodies to chlorhexidine (a biguanide antiseptic), but anaphylaxis to chlorhexidine was not congruent with the patient history and dermal provocation tests. However, skin prick tests were indicative of sensitization to Polyaminopropyl Biguanide. These results were supported by the detection of specific IgE antibodies to Polyaminopropyl Biguanide, the results of basophil activation tests, and IgE inhibition analysis. In an assay to assess cross-reactivity, varying concentrations of Polyaminopropyl Biguanide and chlorhexidine (0.1 to 100 µg/mL) were added to the patient’s serum. The results of this assay suggested a cross-reaction between Polyaminopropyl Biguanide and chlorhexidine. The authors presumed cross-reactive IgE antibodies binding to both biguanide antiseptics and identified Polyaminopropyl Biguanide as the likely cause of the anaphylactic reaction. Polyaminopropyl Biguanide was recognized as an emerging allergen that has to be considered as a cause of anaphylaxis.
Case Reports
An itchy rash on the hand was observed over a 2-year period in a nonatopic patient with a history of retinal detachment surgery. 54 The patient had regularly used a rinse-off contact lens cleaning solution containing 0.001% Polyaminopropyl Biguanide twice daily. A patch test chamber containing the undiluted contact lens cleaning solution was applied to the skin for 2 days, and doubtful results were reported on day 4. A patch test chamber containing a 10% dilution of the product (0.0001% Polyaminopropyl Biguanide tested) was subsequently applied to the skin, and positive results (+ reaction) were observed on day 7. Additionally, semi-open tests of the undiluted product yielded a weak positive reaction on day 7. In other tests, the individual ingredients (obtained from the manufacturer) of the contact lens cleaning solution were diluted to different concentrations in water. There were no reactions to 2% aqueous Polyaminopropyl Biguanide, but a weak, late reaction (1+ reaction) to 5% aqueous Polyaminopropyl Biguanide was observed on day 7. However, stronger and earlier reactions were observed after the application of 10% aqueous Polyaminopropyl Biguanide (+? reaction on day 2; 2+ reaction on days 5 and 7) and 20% aqueous Polyaminopropyl Biguanide (2+ reaction on day 2; 3+ reaction on days 5 and 7). Patch test results for 20% aqueous Polyaminopropyl Biguanide in 10 control subjects were negative.
In a case report on a nonatopic patient with a history of bilateral leg ulcers and multiple contact allergies, mild hand dermatitis was observed after repeated use of a wound irrigation solution that contained Polyaminopropyl Biguanide and a wound gel containing the same disinfectant. 50 The composition of the disinfectant (liquid and gel) was as follows: 0.1% Polyaminopropyl Biguanide, 0.1% undecylenamidopropyl betaine, and water; the gel also contained glycerol and hydroxyethyl cellulose. In a repeated open application test, a positive reaction was observed after the gel was applied twice daily (in elbow fold) for 10 days. The patient was also patch tested (patch test chamber) with 5% aqueous Polyaminopropyl Biguanide (a dilution of a 20% aqueous solution). The solution was applied to the upper arm for 2 days; reactions, scored according to International Contact Dermatitis Research Group (ICDRG) guidelines were negative on day 2, but were positive on day 4. The patch test (same procedure) was repeated at concentrations of 2.5% and 5% aqueous Polyaminopropyl Biguanide. Positive reactions to the 5% concentration were observed on day 2 (+) and day 4 (++, with partially pustular morphology). Results for the gel and liquid were negative in patch tests.
A chronic, recurrent and itchy dermatitis was observed in a male patient who used wet wipes. 55 Polyaminopropyl Biguanide, an ingredient of the product, was tested at different concentrations (20%, 2%, and 0.2% aqueous). Scoring was performed in accordance with ICDRG guidelines. On day 2 and day 4, respectively, + and ++ reactions to 20% Polyaminopropyl Biguanide (with a papulovesicular reaction, extending outside of the test chamber) were observed; +? and + reactions to 2% Polyaminopropyl Biguanide were observed on days 2 and 4, respectively. No reactions to 0.2% Polyaminopropyl Biguanide were observed.
A nonatopic patient with a history of Crohn disease presented with a dermatitis eruption in the area around where the gastrostomy tube had been inserted. Polyaminopropyl Biguanide was a component of the antimicrobial foam dressing that was used. The patient was patch tested with Polyaminopropyl Biguanide (5% aqueous) at 2 separate sites on the upper back. At 96 hours, + reactions were observed at both sites. Negative results were reported for the 10 control subjects who were patch tested. 56
Other Clinical Reports
Based on medical surveillance information obtained between 2004 and 2007 on employees who came in contact with Polyaminopropyl Biguanide in the workplace, no cases of skin sensitization to this chemical were reported. 18 All manufacturing and laboratory employees were offered complete medical evaluations on a regular basis depending on their age. These were conducted every 1 to 2 years.
In a clinical trial (106 dialysis patients) in which patients were treated for infections, Polyaminopropyl Biguanide was well-tolerated and there were only 2 cases of transient local skin erythema. 57 Four of 28 patients were excluded from a cohort study because of adverse effects related to a Polyaminopropyl Biguanide dressing. 58 Reportedly, the application of very high doses (doses not stated) of Polyaminopropyl Biguanide can trigger fever and a generalized exanthema. 29
Polyhexamethylene Guanidine Phosphate
Beginning in 2006, epidemics of a fatal lung injury were observed in Korea every spring. 59 It was subsequently demonstrated that this type of children’s interstitial lung disease (chILD), characterized by rapid progression and high mortality, was associated with humidifier disinfectant use. These disinfectants contain oligo (2- (2-ethoxy)ethoxyethyl) guanidium chloride, polyhexamethylene guanidine (PHMG), 5-chloro-2-methylisothiazol-3(2H)-one/2-methylisothiazol-3-(2H)-one, and didecyldimethylammonium chloride. Polyhexamethylene guanidine (not the ingredient that is under review in this safety assessment) has some chemical similarity with Polyaminopropyl Biguanide. The 2 chemical structures are presented below. Polyhexamethylene guanidine contains guanidine as part of its chemical structure, whereas Polyaminopropyl Biguanide contains biguanide.
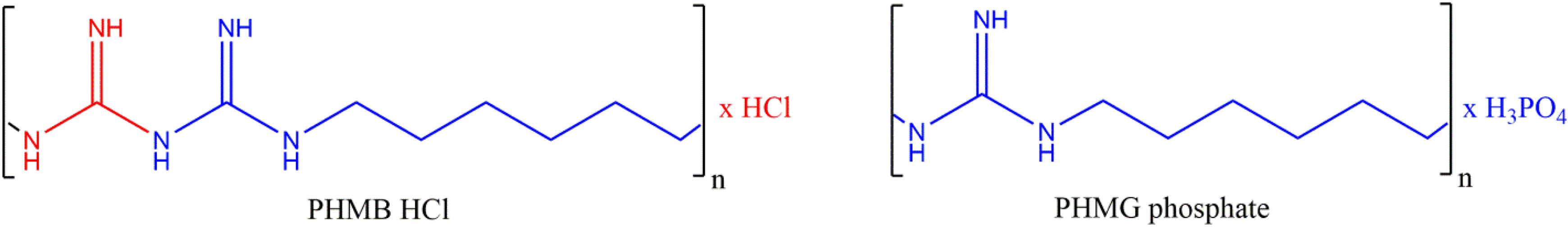
Synthesis of Polyaminopropyl Biguanide via the polycondensation of hexamethylenediamine and dicyanamide.
The clinical characteristics of suspected cases between 2006 and 2011 were determined by a nationwide retrospective epidemiological study. The potential causal relationship with humidifier disinfectants was examined by a prospective surveillance study after humidifier disinfectant sales were suspended. One-hundred thirty-eight children (average age = 30.4 months) were diagnosed with chILD. The annual incidence increased in 2011 and then decreased to zero in 2012. At the time of hospital admission, the most frequent symptoms were cough and dyspnea. Disease progression resulted in spontaneous air leak and 80 children (58%) died. No new cases were found 2 years after the sale of humidifier disinfectants was suspended. The authors noted that the results of this study suggest that humidifier disinfectant inhalation causes an idiopathic type of chILD that is characterized by spontaneous air leak, rapid progression, lack of response to treatment, and high mortality.
A case–control study, with community-dwelling controls, was performed to validate the preceding study’s findings and to confirm the exposure-response relationship between humidifier disinfectant and lung injury. 60 This study was based on reexamination of lung CAT scans and medical records at a hospital in Korea where many of the cases appeared. The purpose of the reexamination was to identify all cases of lung injury that fit certain criteria (ie, criteria for the type of lung injury that was associated with the use of humidifier disinfectants in the previous studies). Each case of lung injury was matched with 4 community-dwelling controls, according to age (± 3 years), sex, residence, and history of childbirth since 2006 (for women). Using a questionnaire, environmental risk factors, which included the humidifier (type and use) and the humidifier disinfectant, were investigated in August of 2011. Exposure to the humidifier disinfectant was calculated for both cases and controls, and the corresponding risks of lung injury were compared. Sixteen patients who were among the 28 eligible cases agreed to participate. Sixty matched controls (selected from the community that the hospital serves) were considered eligible for participation in the study.
Study results indicated a statistically significant, exposure-response relationship between humidifier disinfectant exposure and lung injury. The cases were significantly more likely to have been exposed to humidifier disinfectants, compared to controls (odds ratio [OR]: 116.1; 95% confidence interval [CI]: 6.5-2063.7). The OR for an association between use of a humidifier disinfectant in which the active ingredient was specifically PHMG and lung injury was even greater (OR: 203.8; 95% CI: 11.1-3724.1) suggesting that the lung injuries observed in people who used humidifier disinfectants were attributable to the use of humidifier disinfectants containing PHMG. All cases used several liquid humidifier disinfectant formulations that contained the same proportion of PHMG phosphate. The concentration of PHMG phosphate in the humidifier mist was not stated. Further examination of associations between exposure (number of bottles of disinfectant used per month × duration of exposure as number of months used × volume per bottle of disinfectant/days/ month) and lung injury indicated a clear relationship between the magnitude of daily exposure to disinfectants containing PHMG and the magnitude of the ORs. There was no association between lung injury and use of humidifier disinfectants in which the active ingredient was a combination of isothiazolinone derivatives (5-chloro-2-methyl-4-isothiazolin-3-one/2-methyl-4-isothiazolin-3-one [MCI/MI]) or a guanidinium derivative (oligo(2-(2-ethoxy)ethoxyethyl guanidinium chloride [PHG]). 60
An analysis of patients and fatalities attributed to inhalation exposure to PHMG indicates that this chemical mainly causes lung diseases, such as pulmonary fibrosis. 61 Of the known main components of the humidifier disinfectants, PHMG has been identified as the chemical substance that caused the most deaths. In surveys conducted to identify victims of the humidifier disinfectant, 22% of the research participants answered that they had used the humidifier disinfectant, and 21% complained of side effects. According to another source, hundreds of individuals died from fatal lung injuries associated with use of humidifier disinfectants in Korea from 2002 through 2015. 62 Humidifier disinfectants containing PHMG were the most frequently used among confirmed humidifier-associated lung injury patients (n = 123, 55.7%). The development of humidifier-associated lung injury was found, clinically, to be associated with the use of several humidifier disinfectant products containing PHMG or other humidifier disinfectants.
In a refined risk assessment, 63 the time-weighted average PHMG concentration in the bedroom air was 0.06 mg/m3 (calculated value, which is 250 times greater than the exposure concentration of Polyaminopropyl Biguanide at 0.00024 mg/m3, which is derived from a MOS of 100) for this scenario, averaged over 8 hours. (The 28-day inhalation study on Polyaminopropyl Biguanide, summarized earlier in this safety assessment, was used as a comparison for the PHMG humidifier exposures. 18 ) This concentration in air is 27 times greater than the 0.0022 mg/m3 (the exposure concentration of Polyaminopropyl Biguanide that yields a MOS of 11) inhalation exposure concentration of Polyaminopropyl Biguanide estimated for the use of a pump hair spray containing the highest maximum reported concentration of use (0.053%) Polyaminopropyl Biguanide (see Table 9 in the safety assessment report). Further, the exposure duration of 8 hours for PHMG in the humidifier use scenario is 96 times greater than the conservative 5-minute exposure duration/event assumed for Polyaminopropyl Biguanide in the consumer spray scenarios evaluated in the safety assessment.
Summary
The safety of Polyaminopropyl Biguanide, which is used as a preservative in cosmetics, is reviewed in this assessment. Polyaminopropyl Biguanide is an INCI name; it refers to the hydrochloride salt of an amino polymer comprising hexyl biguanide repeat units (chemical name, polyhexamethylene biguanide hydrochloride [PHMB HCl]). It is not synonymous with the substance identified by the chemical name polyaminopropyl biguanide.
Polyaminopropyl Biguanide, in its neat form, represents a solid/powder of > 94.2% purity, and is usually marketed as an approximately 20% aqueous, preformulation solution. One method for manufacturing Polyaminopropyl Biguanide is via the polycondensation of sodium dicyanamide and hexamethylenediamine.
The following chemicals have been reported as possible impurities of Polyaminopropyl Biguanide: N-(6-aminohexyl)-N′-(6-(6-guanidinohexyl)guanidine, N-cyano-N′-(6-N-cyanoaminohexyl)guanidine, N-cyano-N′-(6-amnohexyl)guanidine), N-cyano-N′-6-(6-guanidinohexyl)guanidine hydrochloride, and 1,6-diguanidinohexane dihydrochloride.
According to 2019 VCRP data, Polyaminopropyl Biguanide is being used in 147 cosmetic formulations, which are mostly leave-on products. The results of a concentration of use survey provided in 2017 indicate that Polyaminopropyl Biguanide is being used at concentrations up to 0.2% in leave-on products (eye lotions), and use in baby lotions, oils, and creams (leave-on products) at concentrations up to 0.1% is also being reported. The highest maximum use concentration that is being reported for Polyaminopropyl Biguanide in rinse-off products is 0.1% in hair dyes and colors and in skin cleansing products. Polyaminopropyl Biguanide in reported to be used in aerosolized products according to VCRP data, but this type of use was not reported in the concentration of use survey.
In 2017, the SCCS issued a final opinion stating that the use of Polyaminopropyl Biguanide as a preservative in all cosmetic products at concentrations up to 0.1% is safe and that its use in sprayable formulations is not advised.
The safety of Polyaminopropyl Biguanide has been reviewed by the US EPA. The Agency concluded that this pesticide has very low aggregate risk of adverse health effects to the public or environment.
The results of a dermal penetration study on Polyaminopropyl Biguanide indicated that absorption through the skin equaled 1.56% (dermis contained 1.56% of applied dose) + 0.03% (receptor fluid contained 0.03% of applied dose). Based on SCCS Notes of Guidance, one standard deviation (2.5%) was added to the absorbed amount, yielding a calculated dermal absorption value of 4.09% (1.56% + 0.03% + 2.5% = 4.09%).
The principal route of excretion of radioactivity from orally administered (radiolabeled) Polyaminopropyl Biguanide was in the feces in the majority of the rat studies. However, in one rat study, most of the [14C] Polyaminopropyl Biguanide administered in drinking water or in the diet was absorbed; and the majority was excreted in the urine, with lesser amounts excreted in the feces. The following components have been detected in the urine of rats fed Polyaminopropyl Biguanide in the diet: oligomers with 2 cyanoguanidino end groups, as well as the trace constituents, 3,3-dicyano-1,1-hexamethylene-diguanidine and 1-(6-aminohexyl)-3-cyanoguanidine.
There was no incidence of mortality or systemic toxicity in rats that received a single dermal dose of 5000 mg/kg aqueous Polyaminopropyl Biguanide; but, hemorrhage of dermal capillaries at the application site was observed. In an acute dermal toxicity study on 20% aqueous Polyaminopropyl Biguanide involving rabbits, an LD50 > 400 mg/kg was reported.
An LD50 of 1040 mg/kg was reported in a study in which rats were dosed orally with Polyaminopropyl Biguanide (concentration not stated) in distilled water. LD50 values of > 1000 mg/kg were reported for rats dosed orally with aqueous solutions of up to 25% Polyaminopropyl Biguanide. A median lethal dosage of 25.6 mg/kg was reported for rats dosed orally with a 0.4% Polyaminopropyl Biguanide solution.
LC50 s of > 0.36 mg/L and equal to 0.37 mg/L were reported in acute inhalation toxicity studies in which rats were exposed for 4 hours to Polyaminopropyl Biguanide (99.6%) solutions at concentrations of 360 mg/m3 in air and up to 300 mg/m3 in air, respectively. Dark/red lungs were observed at necropsy. A concentration-related depression of respiratory rate was reported in a study in which mice were exposed to Polyaminopropyl Biguanide (20% aqueous) at concentrations up to 208 mg/m3.
No specific systemic effects were observed after 25% aqueous Polyaminopropyl Biguanide was applied to the skin of rats for 3 alternating 24-hour periods. There was also no evidence of systemic toxicity in rats that received 6, 24 hours dermal applications of 20% Polyaminopropyl Biguanide (diluted with water to 0.04% active ingredient). There were no mortalities or signs of systemic toxicity in rats that received dermal applications of 20.2% aqueous Polyaminopropyl Biguanide at dosages up to 200 mg/kg daily over a 30-day period (21 applications total; NOAEL = 200 mg/kg/d). In a 21-day dermal toxicity study involving rabbits, there was no evidence of toxic effects on the skin after 20% aqueous Polyaminopropyl Biguanide (12,000 ppm solution (1 mL) was applied daily.
Gastrointestinal inflammation was observed in rats dosed orally with 25% aqueous Polyaminopropyl Biguanide (in distilled water; initially at 1 g/kg and subsequently at 0.5 g/kg) for 21 days. 21 A LOAEL of 0.1 mg/mL for 20% aqueous Polyaminopropyl Biguanide (in drinking water) was reported in 28-day oral toxicity studies involving rats and mice.
Rats (groups of 10) that received Polyaminopropyl Biguanide (in drinking water, up to 150 mg/kg) for 4 weeks experienced dehydration, clinical signs of rough coat and hunched posture, and body weight loss (all classified as severe). Across the 3 dose groups, 10 rats had to be terminated due to severe weight loss, whereas, the remaining rats eventually adapted and began to gain weight. Absolute liver weights in all dose groups were similar to the control group. Mild centrilobular hypertrophy in the liver was observed in some of the rats (all dose groups). In the same study, Polyaminopropyl Biguanide administered (in the diet, 4000 mg/kg) to rats for 4 weeks caused a statistically significant decrease in body weight and absolute liver weight. In this dietary group, there was no evidence of centrilobular hypertrophy in the liver. Also, there was no evidence of necrosis or inflammatory lesions in the liver when Polyaminopropyl Biguanide was administered in drinking water or in the diet. In a 60-day gavage study on Polyaminopropyl Biguanide involving rats, mild toxicity in the liver or kidneys was observed (by microscopic examination) at 2 mg/kg/d (dose equivalent to 0.2 mg/L of 0.4% solution of test substance), 8 mg/kg/d (dose equivalent to 0.4 mg/L of 0.4% solution of test substance), and 32 mg/kg/d (highest dose, equivalent to 1.2 mg/L of 0.4% solution of test substance). None of the animals died.
In 21-day and 28-day inhalation toxicity studies on Polyaminopropyl Biguanide involving rats, NOAECs of 0.025 mg/m3 and 0.0239 mg/m3 were reported, respectively. In the 21-day study, the animals were exposed (nose-only, concentrations up to 26 mg/m3) to the test substance 5 days per week, 6 h/day. Slightly-to-moderately severe pneumonitis was observed at histopathological examination in rats exposed to 0.25 mg/m3. Moderate to severe pneumonitis was observed in rats exposed to 2.75 mg/m3, and severe nasal irritation and dyspnea were observed at a concentration of 12.5 mg/m3. Additionally, all rats of the 12.5 and 26 mg/m3 groups died. In the 28-day study (nose-only, concentrations up to 2.5 mg/m3, 6 h/day, 5 days per week), squamous metaplasia was observed in the larynx of males and females exposed to 0.25 mg/m3 and 2.5 mg/m3, and tracheal inflammation was observed in males and females exposed to 2.5 mg/m3. Pneumonitis and bronchitis were observed in the lungs of males and females exposed to 2.5 mg/m3.
There were no treatment-related macroscopic postmortem findings in mice in a 90-day drinking water study of 20% aqueous Polyaminopropyl Biguanide (concentrations up to 0.3 mg/mL in drinking water), 3 and a NOAEL of 1000 ppm was reported for this ingredient in a 90-day feeding study on 20.2% aqueous Polyaminopropyl Biguanide in which mice received concentrations up to 4000 ppm in the diet. The following results were reported in 90-day oral toxicity studies on Polyaminopropyl Biguanide involving rats: no mortalities, but iron pigment/deposits were observed in Kupffer cells (at 1250 ppm [in one study on 25% aqueous Polyaminopropyl Biguanide] and 5000 ppm [in another study on 25% aqueous Polyaminopropyl Biguanide] in diet; and an NOAEL of 1000 ppm [in a third study on 20.2% aqueous Polyaminopropyl Biguanide]). An NOAEL of 5500 ppm was reported for Beagle dogs fed Polyaminopropyl Biguanide at concentrations up to 11,000 ppm in the diet for 90 days.
In an 80-week chronic toxicity study involving mice (dermal applications 5 days/week), a mortality rate of 75% was reported for the highest dose group (10% Polyaminopropyl Biguanide; 30 mg dose). The exophthalmos observed throughout the study was more severe in this group, compared with the other groups, but the results of histological examination of the eyes and gross and microscopic examination of the thyroids were negative. An NOAEL of 0.6 mg/mouse/day (15 mg/kg/d) was reported.
Mice were fed 20% Polyaminopropyl Biguanide in the diet (concentrations up to 1000 ppm; feeding 1 week prior to mating and during mating [males and females] and continuation of feeding throughout pregnancy and lactation [females]). The offspring of these mice were also fed 20% Polyaminopropyl Biguanide at concentrations up to 1000 ppm in the diet for 97 weeks. For parents and their offspring, feeding with the test substance did not cause macroscopic changes in the spleen or liver. In a 104-week oral toxicity study on 20.2% Polyaminopropyl Biguanide involving rats, an NOAEL of 600 ppm (36 mg/kg/d) was reported. The SCCS used this NOAEL to calculate a MOS. This calculation was performed, assuming that all cosmetics contain 0.1% Polyaminopropyl Biguanide and a dermal absorption value of 4.09%, and using the NOAEL of 600 ppm (36 mg/kg/d; corrected to 3.1 mg/kg/d, based on 8.5% oral absorption) and a SED of 0.012 mg/kg/d; MOS values of 258 (based on cosmetic exposure estimate) and 227 (based on cosmetic exposure estimate + noncosmetic exposure estimate) were determined. An NOEL of 200 ppm for histopathologic changes was reported in a 122-week oral toxicity study involving rats fed 20% Polyaminopropyl Biguanide at concentrations up to 2000 ppm in the diet. Increased adrenal weight was reported for males and females at concentrations of 1000 ppm and 2000 ppm in the diet.
In a 26-week feeding study involving dogs, dietary concentrations of 1500 ppm and 4500 ppm 20% Polyaminopropyl Biguanide produced concentration-related hepatotoxicity and nephrosis. An NOAEL of 1500 ppm for 20.2% Polyaminopropyl Biguanide was reported in a 1-year feeding study involving dogs; treatment-related histopathological findings in the liver and kidneys were reported in the high-dose group. In this study, groups of animals were fed test-substance concentrations of 300 ppm, 1500 ppm, and 4500 ppm for up to weeks 11/12. The 4500 ppm concentration was reduced to 3000 ppm for the remainder of the study because high dose males exhibited unexpected signs of toxicity, including marked reddening/peeling of scrotal skin, loss of appetite, body weight loss, and/or indications of liver impairment in the form of elevated plasma alanine transaminase and/or aspartate transaminase activities.
An NOAEL of 10 mg/kg/d for developmental toxicity was reported in an oral dosing (by gavage on gestation days 6 through 15) study on 20% aqueous Polyaminopropyl Biguanide involving mice. In oral reproductive and developmental toxicity studies on 20% aqueous Polyaminopropyl Biguanide in rats, NOAECs of 1000 ppm (after feeding in diet on gestation days 1 through 20) and 1300 ppm (after feeding in diet during a 9-day premating period and until the 3rd generation) have been reported. An NOAEL of 2000 ppm for reproductive and development effects was reported in a study in which rats were fed (through 2 successive generations) with 20.2% aqueous Polyaminopropyl Biguanide at dietary concentrations up to 2000 ppm. Polyaminopropyl Biguanide has been classified as embryotoxic at a dosage rate of 100 mg/kg/d (rats; protocol not stated).
An NOAEL of 40 mg/kg/d for developmental toxicity has been reported in an oral dosing (20.2% aqueous Polyaminopropyl Biguanide by gavage on gestation days 8 through 20) study involving rabbits. Polyaminopropyl Biguanide (0.04% in PEG) has been classified as embryotoxic in rabbits at an oral dosage rate of 32 mg/kg/d (animal strain and dosing protocol not stated). Polyaminopropyl Biguanide (concentration not stated) has been classified as teratogenic in rats at an intraperitoneal dosage rate of 10 mg/kg/d (dosing protocol not stated). 29 In an inhalation study on 20% aqueous Polyaminopropyl Biguanide, degeneration of seminiferous tubules in the testis of 1 male rat was observed after exposure to 0.25 mg/m3 (6 h/day, 5 days/week for 3 weeks), but this was not observed in any other group, including the group exposed to the highest concentration (26 mg/m3).
In Ames tests, ∼20% Polyaminopropyl Biguanide was nongenotoxic at doses up to 5000 µg/plate, with and without metabolic activation. At the highest dose evaluated (333,300 µg/plate) in the Ames test, Polyaminopropyl Biguanide was weakly genotoxic in Salmonella typhimurium strain TA 1538 without metabolic activation. Polyaminopropyl Biguanide was nongenotoxic in a mouse lymphoma assays at concentrations up to 2000 µg/mL with and without metabolic activation, or in an in vitro micronucleus test (cultured human peripheral blood lymphocytes) at concentrations up to 50 µg/mL (without metabolic activation) and up to 250 µg/mL (with metabolic activation). In an in vivo micronucleus test, Polyaminopropyl Biguanide was nonclastogenic in polychromatic erythrocytes from mice that received single oral dosages up to 400 mg/kg. In an in vivo unscheduled DNA synthesis assay, there was no induction of unscheduled DNA synthesis in hepatocytes from rats that received single oral doses up to 1500 mg/kg.
Polyaminopropyl Biguanide (20%) was evaluated at concentrations up to 3000 µg/mL in the cell transformation assay (using baby hamster kidney fibroblasts); there was no difference in the number of transformed cell colonies between test and negative control cultures. In another assay, RAW 264.7 mouse macrophages (a macrophage-like, Abelson leukemia virus transformed cell line derived from BALB/c mice) were cocultured with SVEC-10 mouse liver endothelial cells in various experimental conditions: preactivation of macrophages with Polyaminopropyl Biguanide or LPS and/or coculture in presence of Polyaminopropyl Biguanide. Polyaminopropyl Biguanide, tested at concentrations up to 1 ppm, had no direct effect on liver cell proliferation and did not potentiate cell proliferation induced by activated macrophages.
Polyaminopropyl Biguanide was classified as a hepatocarcinogen in mice at the highest dose tested in a study in which Polyaminopropyl Biguanide in ethanol was applied to the skin daily at doses up to 750 mg/kg/d (5 days/week) for 80 weeks. The NOAEL was 0.6 mg/mouse/day (15 mg/kg/d). A variety of inflammatory hepatic changes was observed in all groups, including the controls. However, at 750 mg/kg/d, severe hepatitis was observed in some of the animals. These hepatic changes appeared to have been mainly responsible for causing increased numbers of deaths in the high-dose group. A scientific advisory panel advising the SCCS indicated that the hepatitis observed in this study may be attributable to the Helicobacter hepaticus infections, which may also be responsible for the increased incidence of hepatocellular neoplasms in these animals.
Mice were fed 20.2% Polyaminopropyl Biguanide in the diet (up to 1000 ppm active ingredient; feeding 1 week prior to mating and during mating [males and females]). The offspring of these mice were also fed 20.2% Polyaminopropyl Biguanide at concentrations up to 1000 ppm active ingredient in the diet for 97 weeks. Except for vascular tumors, there were no treatment-related (nonneoplastic or neoplastic) increases in histopathologic findings. Vascular tumors (hemangiosarcomas or hemangiomas) in the liver or other sites and a high mortality incidence (80%) were reported by week 97. A dose-related increase in the liver vascular tumor incidence was reported. After reviewing the results of this study, the SCCS concluded that the data are considered to be of low reliability due to the high mortality rate that was reported. When mice were fed Polyaminopropyl Biguanide at dietary concentrations up to 4000 ppm for up to 28 days, increased cell proliferation in a concentration-related manner was noted at 1200 ppm and 4000 ppm. In another study, a statistically significant increase in the incidence of hemangiosarcomas and hemangiomas was reported in male mice (C57B1/10J/CD-1 strain) that received Polyaminopropyl Biguanide at a dietary concentration of 4000 ppm daily for 2 years. A low incidence of hemangioma (2 of 64 males; 2 of 64 females) and hemangiosarcoma (1 of 64 females) was reported in a study in which rats were fed Polyaminopropyl Biguanide at a dietary concentration of 2000 ppm for 2 years.
In another 2-year study, Polyaminopropyl Biguanide was administered (in drinking water or in diet) to groups of rats. Hepatocellular tumors were induced at concentrations (in water) of 1000 mg/L and 1500 mg/L, but not at a concentration of 500 mg/L (in water). Administration of Polyaminopropyl Biguanide in diet did not cause increase in hepatocellular tumors. The hypothesized MOA for liver tumors induced by Polyaminopropyl Biguanide in drinking water involves increased hepatocyte proliferation and induction of hepatocellular foci and tumors. Polyaminopropyl Biguanide was classified as noncarcinogenic in rats fed dietary concentrations up to 2000 ppm for 122 weeks; mortality was 80%. In a feeding study in which rats were fed Polyaminopropyl Biguanide at concentrations up to 2000 ppm, 80% mortality was reported at 124 weeks. A low incidence of hemangiomas or hemangiosarcomas (mostly in lymph nodes) was observed in the groups of remaining animals (7 groups, with 8 to 21 rats/group; 1 animal with a hemangioma or hemangiosarcoma per group).
In a study involving A549 lung cells in vitro, it was noted that Polyaminopropyl Biguanide induces inflammatory responses via the NF-κB signaling pathway. Except for a slight increase in some cytokines and transcription factor at concentrations at which cell lysis occurs rapidly, Polyaminopropyl Biguanide did not exhibit clear and remarkable epigenetic properties at 20 to 100 μg/mL.
Polyaminopropyl Biguanide (PHMB) exhibited high antimicrobial activity against Staphylococcus aureus and Escherichia coli, whereas, though chemically closely related, polyaminopropyl biguanide proved to be ineffective in bacterial eradication. When compared to Polyaminopropyl Biguanide (PHMB), polyaminopropyl biguanide displayed significantly lower cytotoxicity at concentrations ranging from 0.005% to 0.1% vol/vol; both chemicals were cytotoxic.
In a study involving mice, the highest dose of Polyaminopropyl Biguanide (10% concentration in ethanol, 30 mg dose) caused hyperkeratosis and, occasionally, ulceration extending into the dermis when applied repeatedly for 80 weeks. 21 Polyaminopropyl Biguanide (0.04%) was classified as a nonirritant when applied to the skin of rats for 24 hours. Repeated applications of 20.2% aqueous Polyaminopropyl Biguanide to rats for 21 days resulted in slight skin irritation (at 60 mg/kg/d) and moderate irritation (at 200 mg/kg/d). Severe skin irritation was observed in all rats that received a single 24-hour application of 25% aqueous Polyaminopropyl Biguanide at dosages of 2.5 mL/kg and 5 mL/kg. Focal ulceration and edema were observed in rats after 25% aqueous Polyaminopropyl Biguanide was applied repeatedly to the skin.
Repeated 23-hour applications of Polyaminopropyl Biguanide (12,000 ppm; 1 mL per application) to the skin of rabbits for 21 days were not irritating. Single applications (24 hours) of 20% aqueous Polyaminopropyl Biguanide to rabbits indicates that this compound is noncorrosive, but moderately irritating, to intact skin, and severely irritating to abraded skin. Polyaminopropyl Biguanide (20% aqueous) induced erythema at abraded, but not intact, skin sites in rabbits, and was classified as a noncorrosive material. Moderate erythema was observed after 20% aqueous Polyaminopropyl Biguanide was applied for 24 hours to the skin of rabbits. Mild skin irritation resulted after Polyaminopropyl Biguanide (96%, as powder) was applied to the skin of rabbits for up to 4 hours. Polyaminopropyl Biguanide (0.5 g, moistened with water; single 4-hour application) was also classified as a mild skin irritant in rabbits. Slight to moderate erythema was observed in guinea pigs that received repeated applications of 25% aqueous Polyaminopropyl Biguanide for 3 days.
Polyaminopropyl Biguanide (up to 1.5% active) was not classified as a primary skin irritant when applied for 24 hours to the skin of human subjects (17 males and 28 females). 18 A bacterial, nonocclusive dressing loaded with 1% wt/vol sericin and 0.3% wt/vol Polyaminopropyl Biguanide was classified as nonirritating after application to the skin of 105 subjects for 3 days. 34
In a guinea pig maximization test on Polyaminopropyl Biguanide, moderate skin sensitization was observed in guinea pigs challenged with Polyaminopropyl Biguanide (20.2% active ingredient) and a 30% solution of the ingredient (6% active ingredient) in deionized water. The challenge phase was preceded by induction with 0.06% active ingredient (intradermal injection) and 20.2% active ingredient (occlusive application). Moderate to strong sensitization was observed in guinea pigs challenged with Polyaminopropyl Biguanide (20.2% active ingredient). The challenge phase was preceded by induction with 0.2% active ingredient (intradermal injection) and 20.2% active ingredient (topical application). In another guinea pig maximization test, sensitization was not observed in guinea pigs induced with 0.15% (intradermal injection) and 20% Polyaminopropyl Biguanide (topical application), followed by challenge with 10% or 20% Polyaminopropyl Biguanide.
In one Buehler test on Polyaminopropyl Biguanide, guinea pigs were induced with 2% active ingredient (topical application), challenged with 2% active ingredient, and rechallenged with 0.2%, 2%, and 4% active ingredient. The initial challenge with 2% active ingredient and rechallenge with 2% and 4% active ingredient resulted in faint erythema; rechallenge with 0.2% active ingredient produced negative results. Polyaminopropyl Biguanide (2% active ingredient) was classified as a moderate sensitizer. In another Buehler test, it was determined that the threshold for eliciting sensitization in guinea pigs was ∼ 1%. Induction concentrations ranged from 0.3% to 5% and challenge concentrations ranged from 0.075% to 15%. Results from a study evaluating the possible cross-reactivity of Polyaminopropyl Biguanide (challenge with 20%) with chlorhexidine (challenge with up to 4% chlorhexidine gluconate) in guinea pigs were negative.
In an HRIPT (191 subjects) 20% aqueous Polyaminopropyl Biguanide (2% active ingredient; effective concentration = 0.4%) did not cause primary skin irritation, but did result in sensitization. When a leave-on product containing 20% Polyaminopropyl Biguanide (tested at 0.5%, effective concentration 0.1%) was evaluated in an HRIPT involving 207 subjects, it was concluded that the product did not induce dermal sensitization. In another HRIPT (115 subjects; any ethnicity, provided that their degree of skin pigmentation did not significantly interfere with evaluations) on a neck cream containing 0.2% Polyaminopropyl Biguanide, the product did not cause clinically meaningful irritation or sensitization.
The skin sensitization potential of 20% Polyaminopropyl Biguanide (diluted with distilled water to 1% vol/vol prior to testing; 750 µg/cm2) was evaluated using semi-occlusive patches in an HRIPT involving 108 subjects (Asian [∼2%], Bi-racial [∼3%], Black [∼ 23%], Caucasian [∼ 33%], and Hispanic [∼39%]; Fitzpatrick skin types not stated). The authors concluded that Polyaminopropyl Biguanide did not induce dermal sensitization in the subjects tested, and a NESIL (determined based on data from this HRIPT) of 750 µg/cm2 was used to calculate a QRA on various product types. The authors concluded that Polyaminopropyl Biguanide did not induce dermal sensitization in the subjects tested. Using the results from this study, a NESIL of 750 µg/cm2 was chosen and used to perform QRAs on products of various use types at maximum reported use concentrations.
Very strong irritation potential, but no significant photoirritancy, was reported in a study in which male rats were tested (dermal application) with Polyaminopropyl Biguanide at concentrations of 2% and 5%. When tested at a concentration of 1%, in 26 subjects, Polyaminopropyl Biguanide was essentially nonirritating and did not induce sensitization, phototoxicity, or photoallergenicity.
Case reports with sensitization reactions to Polyaminopropyl Biguanide (reported as an ingredient of wet wipes, contact lens cleansing solutions, wound irrigation solutions, preoperative antiseptics, and antimicrobial foam dressings) have been reported. The prick test was used to diagnose immediate contact urticarial reactions in 44 patients with eczematous dermatitis. A positive reaction was observed in 1 patient.
Undiluted and 25% aqueous Polyaminopropyl Biguanide were severe ocular irritants when instilled into unrinsed rabbit eyes. Polyaminopropyl Biguanide (20% aqueous) induced slight inflammation, and Polyaminopropyl Biguanide (0.04% active ingredient) was nonirritating to the eyes of rabbits. In a study in which 20% aqueous Polyaminopropyl Biguanide was instilled into human eyes and the eyes of rabbits in a temperature-controlled chamber, normal corneal morphology was observed at histological examination.
Discussion
The safety of Polyaminopropyl Biguanide for use as a preservative in cosmetics is reviewed in this safety assessment. Polyaminopropyl Biguanide is an INCI name; it refers to the hydrochloride salt of an amino polymer comprising hexyl biguanide repeat units (PHMB HCl). This ingredient does not actually contain the chemical polyaminopropyl biguanide, which has a 3-carbon chain in each monomeric repeat unit. Rather, the INCI name, Polyaminopropyl Biguanide, applies exclusively to chemical PHMB, which has a 6 carbon chain in each monomeric repeat unit, and is always supplied as the hydrochloride salt. The chemical polyaminopropyl biguanide is not a cosmetic ingredient.
There was no evidence of systemic toxicity following dermal exposure to 0.4% Polyaminopropyl Biguanide, which is greater than the 0.2% maximum reported cosmetic use concentration of this ingredient. Furthermore, the Panel noted that the dermal penetration of Polyaminopropyl Biguanide is minimal, considering that most of the compound remains in the epidermis and its distribution systemically is not a concern.
Overall, the available in vivo and in vitro genotoxicity data on Polyaminopropyl Biguanide in bacterial and mammalian cells are negative. The Panel noted that in vitro genotoxicity assays are difficult to interpret for microbial toxins such as the cytotoxic preservative Polyaminopropyl Biguanide. However, after reviewing the available data, the Panel determined that genotoxicity is not a concern. A low incidence of hemangiomas and hemangiosarcomas was reported in a study in which rats were fed Polyaminopropyl Biguanide at a dietary concentration of 2000 ppm for 2 years. The Panel noted that the vascular tumors observed in rats and mice were likely attributable to sustained hepatotoxicity (ie, a nongenotoxic mechanism) from high exposures (ie, doses above the maximum tolerated dose) that the Panel considered not toxicologically relevant to cosmetic use. Furthermore, the carcinogenicity study results reviewed are equivocal.
Results were classified as positive for Polyaminopropyl Biguanide in the LLNA. However, interpreting the study results is hampered by the absence of a reported EC3. Additionally, the Panel noted that Polyaminopropyl Biguanide is a sensitizer at 2%, and that elicitation occurs at a much lower concentration (0.2%) in animal studies. Based on the results of these studies, the Panel expressed concerns about sensitization potential. In response, industry performed a QRA.
A NESIL of 1000 µg/cm2 was chosen and used to form the QRA for contact dermatitis with Polyaminopropyl Biguanide in cosmetics. This NESIL supports the use of this ingredient at concentrations of ≤ 0.1%. Among the human data that were used to derive the NESIL was an HRIPT involving 26 subjects tested with 1% Polyaminopropyl Biguanide at a dose of 1000 µg/cm2, the highest nonsensitizing dose in relation to all of the HRIPT data that were considered. However, the Panel noted the small subject population in this HRIPT (a test population of ≥ 100 subjects is usually preferred). Furthermore, in an HRIPT on a neck cream containing 0.2% Polyaminopropyl Biguanide (dose = 100 µg/cm2) that involved more than 100 subjects, faint and pink reactions were observed at various times during challenge or during induction in some subjects, but the skin types evaluated were not sufficiently diverse. Based on these observations, the Panel suggested that the NESIL of 1000 µg/cm2 may not be correct and determined that an HRIPT (with at least 100 subjects with a range of Fitzpatrick skin types) on Polyaminopropyl Biguanide at doses of 500 and 1000 µg/cm2 is needed. In response to this data need, an HRIPT on 1% aqueous Polyaminopropyl Biguanide (dose = 750 µg/cm2) involving 108 subjects (Asian [∼2%], biracial [∼3%], Black [∼ 23%], Caucasian [∼ 33%], and Hispanic [∼39%]; Fitzpatrick skin types not stated) was provided by the Council. Polyaminopropyl Biguanide (1%) did not induce dermal sensitization in any subjects tested. Using the results from this study, a NESIL of 750 µg/cm2 was chosen and used to perform QRAs on products of various use types at maximum reported use concentrations. However, other data included in this CIR safety assessment indicate the potential for sensitization to Polyaminopropyl Biguanide, specifically in an LLNA, HRIPT, and in guinea pig maximization tests. Acknowledging the positive sensitization data on Polyaminopropyl Biguanide, the Panel noted that the potential for induction of skin sensitization varies depending on a number of factors, including the area of product application and final formulation; thus, formulators should assess the potential for final formulations to induce sensitization using a QRA or other accepted methodologies.
In addition to concerns relating to sensitization potential, the Panel also expressed concern over the existence of case reports of contact urticaria attributable to the use of Polyaminopropyl Biguanide in wound dressings. However, it was determined that contact urticaria would not be an issue in relation to cosmetic product applications after considering that this reaction was observed under the conditions of burn dressings on severely damaged skin. The Panel also determined that skin irritation potential at cosmetic use concentrations is not a concern, considering that Polyaminopropyl Biguanide (up to 1.5% active) was not classified as a primary skin irritant when applied for 24 hours to the skin of human subjects.
The Panel noted clinical studies relating to child deaths in South Korea that were associated with inhalation exposure from humidifiers that had been disinfected with a humidifier disinfectant containing polyhexamethylene guanidine phosphate (often referred to as polyhexamethylene guanidine; PHMG). Polyhexamethylene guanidine is not the same chemical as the cosmetic ingredient Polyaminopropyl Biguanide. However, in an abundance of caution, the Panel requested MOS calculations for Polyaminopropyl Biguanide inhalation exposure from cosmetic products that are sprayed.
A Council survey of maximum reported use concentrations by product category indicates that Polyaminopropyl Biguanide is no longer used in pump or propellant hair sprays. However, products categorized as “Tonics, Dressings, and Other Hair Grooming” that contain Polyaminopropyl Biguanide at maximum use concentrations of up to 0.1% are reported in the survey, and it is possible that products included in this category are sprays. Furthermore, 2019 FDA VCRP data indicate that Polyaminopropyl Biguanide is used in the Other Fragrance Preparations product category (use concentration data unavailable). Given the potential for incidental inhalation exposure, CIR performed a risk assessment using the ConsExpo Web Spray Model (Consumer Exposure Model, Web version 1.0.1). The maximum concentrations of use (0.0004% in propellant hair sprays and 0.053% in pump hair sprays) included in this risk assessment to estimate the inhalation exposure concentrations of Polyaminopropyl Biguanide during the use of cosmetic spray products were based on results initially submitted to the Council in response to a use survey (submitted to CIR on April 11, 2017). The ConsExpo Web Spray Model and an NOAEC (from a 28-day inhalation study in which rats were exposed, nose only, to Polyaminopropyl Biguanide in an aerosolized water solution for 6 h/day, 5 days/week) were used in the MOS calculations for inhalation exposure. Margin of safety values for pump hair sprays (MOS = 11) and propellant hair sprays (MOS = 200) were calculated. Exposure concentrations that would yield an MOS of 100 for propellant and pump hair sprays were also calculated.
After reviewing this risk assessment, the Panel noted that the exposure scenario in the 28-day inhalation study is not representative of consumer pump and propellant hair spray product use, and determined that data more relevant to consumer use are needed. The Panel also noted that there are potential safety issues relating to chronic ingredient inhalation exposure, potentially experienced by hairdressers, but acknowledged that evaluation of occupational safety is not within the purview of the Panel.
The Panel determined that the following data are yet needed in order to evaluate the safety of Polyaminopropyl Biguanide in cosmetic products which may be incidentally inhaled: Consumer use data on pump and propellant hair sprays, for use in determining the extent of exposure to Polyaminopropyl Biguanide during product use. As part of this data insufficiency, use concentration data on this ingredient in aerosolized products and the particle size that is associated with the spray product are needed if Polyaminopropyl Biguanide is used in products that could be inhaled (ie, in products which may result in incidental inhalation).
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Polyaminopropyl Biguanide is safe in cosmetics in the present practices of use and concentration described in the safety assessment, when formulated to be nonirritating and nonsensitizing, which may be based on a QRA or other accepted methodologies. The Panel also concluded that the data are insufficient to determine the safety of Polyaminopropyl Biguanide in products that may be incidentally inhaled.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L St, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Johnson, W. contributed to conception, design, acquisition, analysis, interpretation, and drafted the manuscript; Heldreth, B. contributed to conception, design, acquisition, analysis, interpretation, drafted the manuscript, and critically revised the manuscript; Boyer, I. contributed to analysis and interpretation and critically revised the manuscript; Zhu, J. contributed to conception, design, analysis, interpretation, drafted the manuscript, and critically revised the manuscript; Bergfeld, W. F., Belsito, D. V., Hill, R. A., Klaassen, C. D., Liebler, D. C., Marks, J. G., Shank, R. C., Slaga, T. J., and Snyder, P. W. contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
