Abstract
The phosphoglycerides considered in this safety assessment are reported to function primarily as skin and hair conditioning agents, emulsifying agents, and surfactants in cosmetic products and are used up to a maximum reported concentration of 50%. Although phospholipids exert physiologic effects, these are not reproduced by application of phospholipid ingredients to the skin. Given the possibility that Lecithin may be derived from animal sources, it should be noted that the Food and Drug Administration does not permit the use of ingredients made from bovine specified risk materials in cosmetic products. The Expert Panel for Cosmetic Ingredient Safety concluded that the 17 phosphoglycerides are safe in the present practices of use and concentration in cosmetics, as described in this safety assessment.
Introduction
The safety of Lecithin and other phosphoglycerides, listed below, in cosmetics is reviewed in this safety assessment. These 17 ingredients are reported to function primarily as skin and hair conditioning agents, emulsifying agents, and surfactants in cosmetic products. Lecithin Hydrogenated Lecithin Lysolecithin Hydrogenated Lysolecithin Phospholipids Hydrolyzed Phospholipids Phosphatidic Acid Lysophosphatidic Acid Phosphatidylglycerol Lysophosphatidylglycerol Phosphatidylserine Ammonium Phosphatidyl Rapeseedate Phosphatidylcholine (PC) Hydrogenated PC Hydrogenated Lysophosphatidylcholine Lysophosphatidylethanolamine Phosphatidylinositol
The Expert Panel for Cosmetic Ingredient Safety (Panel) had previously evaluated the safety of Lecithin and Hydrogenated Lecithin in cosmetics and issued a final report (published in 2001) with the following conclusion: Lecithin and Hydrogenated Lecithin are safe as used in rinse-off products, safe for use in leave-on products at concentrations of ≤ 15%, and the data are insufficient to determine the safety of use in cosmetic products where Lecithin and Hydrogenated Lecithin are likely to be inhaled; Lecithin and Hydrogenated Lecithin should not be used in cosmetic products in which N-nitroso compounds may be formed. 1 The qualification relating to the formation of N-nitroso compounds was based on concern over N-nitrosation of a potential bacterial metabolite. The 15% concentration limit and the qualification relating to the formation of N-nitroso compounds are, however, no longer applicable, and the available data addressing cosmetics that may be inhaled are sufficient.
The Panel considered that phospholipids are the ubiquitous components of cell membranes and play a role in physiological processes. Because phospholipids have a physiologic role only when they are found in certain configurations in cell membranes, dermal application of these cosmetic ingredients does not result in the same physiologic effect that result from these chemicals as incorporated into cell membranes.
If it is known that the substance discussed is specifically a cosmetic ingredient, the Ingredient Nomenclature Committee (INC) nomenclature (eg, “Phospholipids”) will be used; capitalization of the first letter of each word is used in INC names. However, in cases where a term is used for its general, customary meaning (eg, “phospholipids” as in the paragraph above), standard naming will be used (ie, no capitalization); for chemicals discussed that are not cosmetic ingredients in this assessment (eg, phosphatidylethanolamine), INC-type capitalization will not be used.
Chemistry
Definition and Structure
The ingredients in this report are glycerides of fatty acids, linked to phosphoric acid or to a phosphoric ester. Lecithin, for example, is a complex mixture of phosphatides, consisting chiefly of PC, phosphatidylethanolamine, Phosphatidylserine, and Phosphatidylinositol, with varying amounts of triglycerides, fatty acids, and carbohydrates isolated from animal or vegetable sources. 1 In naturally occurring lecithins, the phosphoric acid is attached to the glycerol at the α-position. However, the phosphoric acid moiety can also be attached in the β-position of glycerin, as a by-product of synthesis. 2 A structural scheme that is representative of the systematic nature of the phosphoglycerides family is presented in Figure 1.
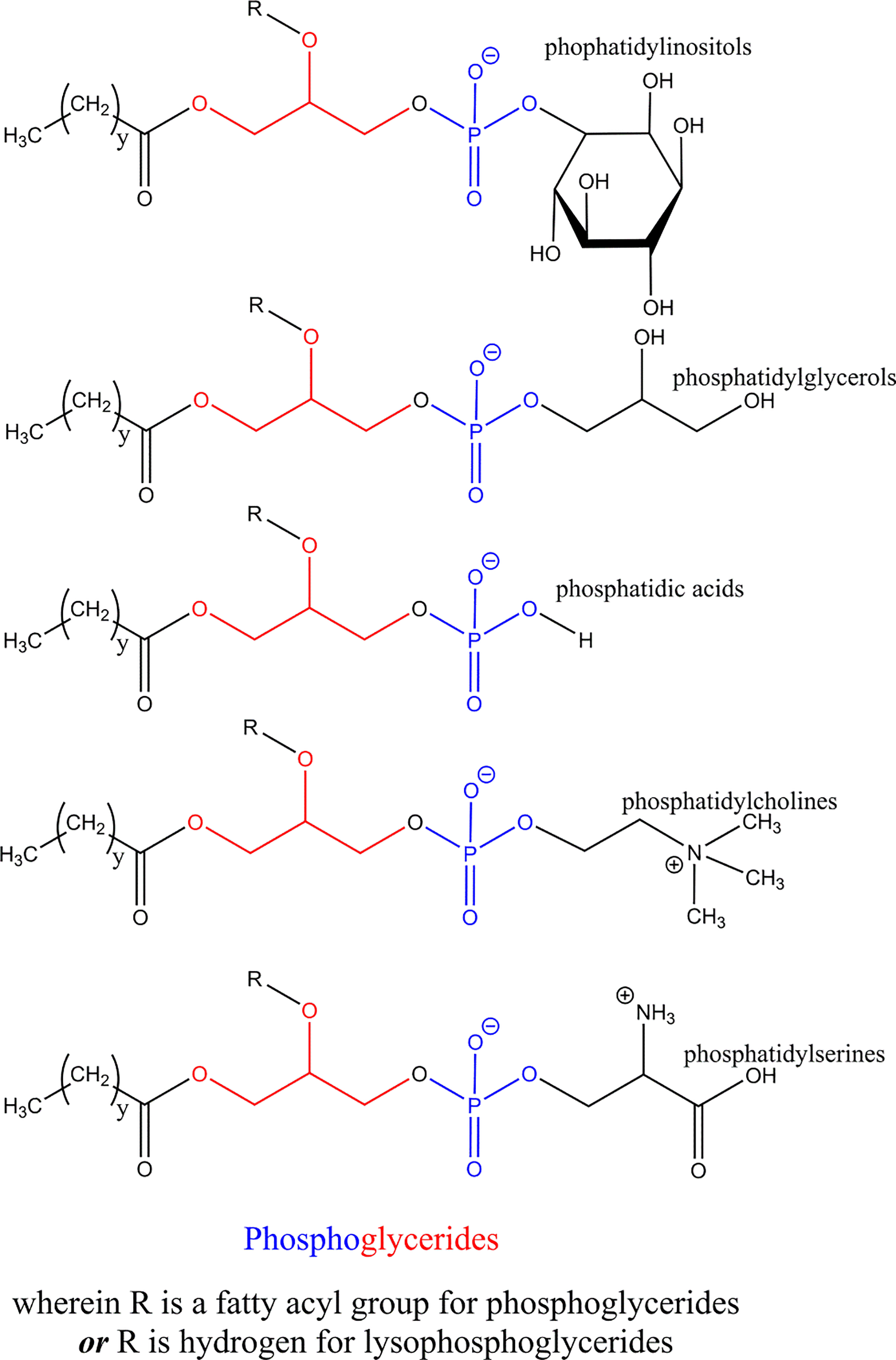
Phosphoglycerides.
Phosphatidylserine is one example of a phosphoglyceride substituted with a fatty acid ester at the secondary alcohol residue of glycerin (ie, is not a lysophosphoglyceride) and an amino acid (ie, serine) attached through the phosphate group, as depicted in Figure 2.
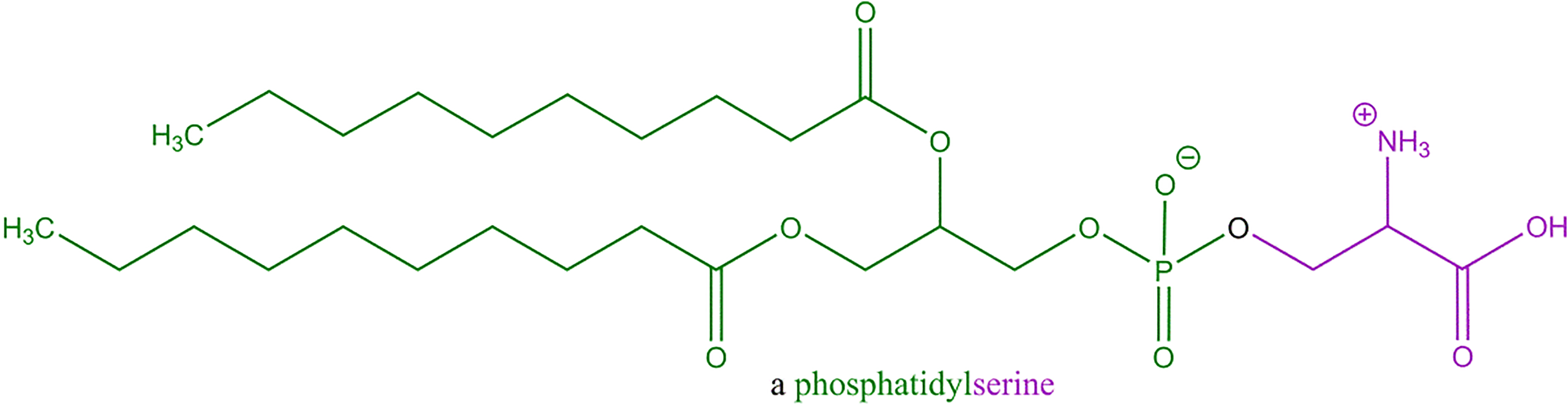
One example of a phosphoglyceride, Phosphatidylserine (depicted is just 1 example of the possible fatty acyl chain lengths).
Hydrogenated Lecithin (CAS No. 92128-87-5) is the end product of the controlled hydrogenation of Lecithin. 1 Despite the ionic, or even zwitterionic, natures of these ingredients, these ingredients are mostly insoluble in water. 3 These ingredients typically are waxy, hygroscopic substances that swell in contact with water to form, dependent on their molecular composition and structure, liposomes, micelles, or mixed micelles.
The main sources of naturally occurring phosphoglycerides, such as Lecithin, as used in personal care products, are maize, egg yolk, and soybean.
4
Phospholipids constitute 0.3% to 0.6% of soybean seed, or 1.5% to 3.0% of crude soybean oil. The composition of phospholipids in soybeans has been reported as follows
5
: PC (12%-46%) Phosphatidylethanolamine (8%-34%) Phosphatidylinositol (1.7%-21%) Phosphatidic Acid (0.2%-14%) Phosphatidylserine (0.2%-6.3%) Lysophosphatidylcholine (1.5%-8.5%) Lysophosphatidylinositol (0.4%-1.8%) Lysophosphatidylserine (1%) Lysophosphatidic Acid (1%)
The definitions, structures, and functions of the phosphoglycerides reviewed in this safety assessment are included in Table 1. 6 There is some overlap among the ingredients in this report. While Phosphatidylserine and PC are listed as separate ingredients, they are likely components of the ingredient named Phospholipids.
Names, CAS Registry Numbers, Structures, and Definitions of the Phosphoglyceride Ingredients. 6
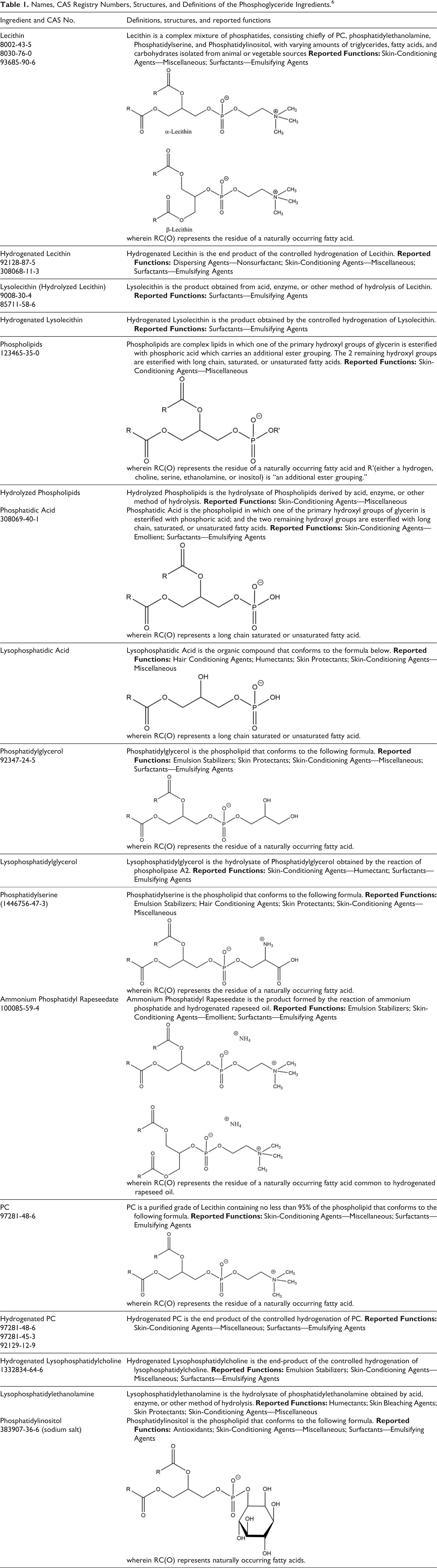
The ingredients in this report form a systematic, logical grouping that interrelates on numerous levels. All of these ingredients: (1) are glycerides of fatty acids, linked to phosphoric acid, or to a phosphoric ester; (2) conform to Figure 1; (3) are ionic and mostly insoluble in water; (4) are typically waxy, hygroscopic substances that swell, when in contact with water, to form liposomes; and (5) either come from sources such as maize, egg yolk, and soybean, or are synthesized via transphosphatidylation of PC (which itself is sourced from soy).
Chemical Properties
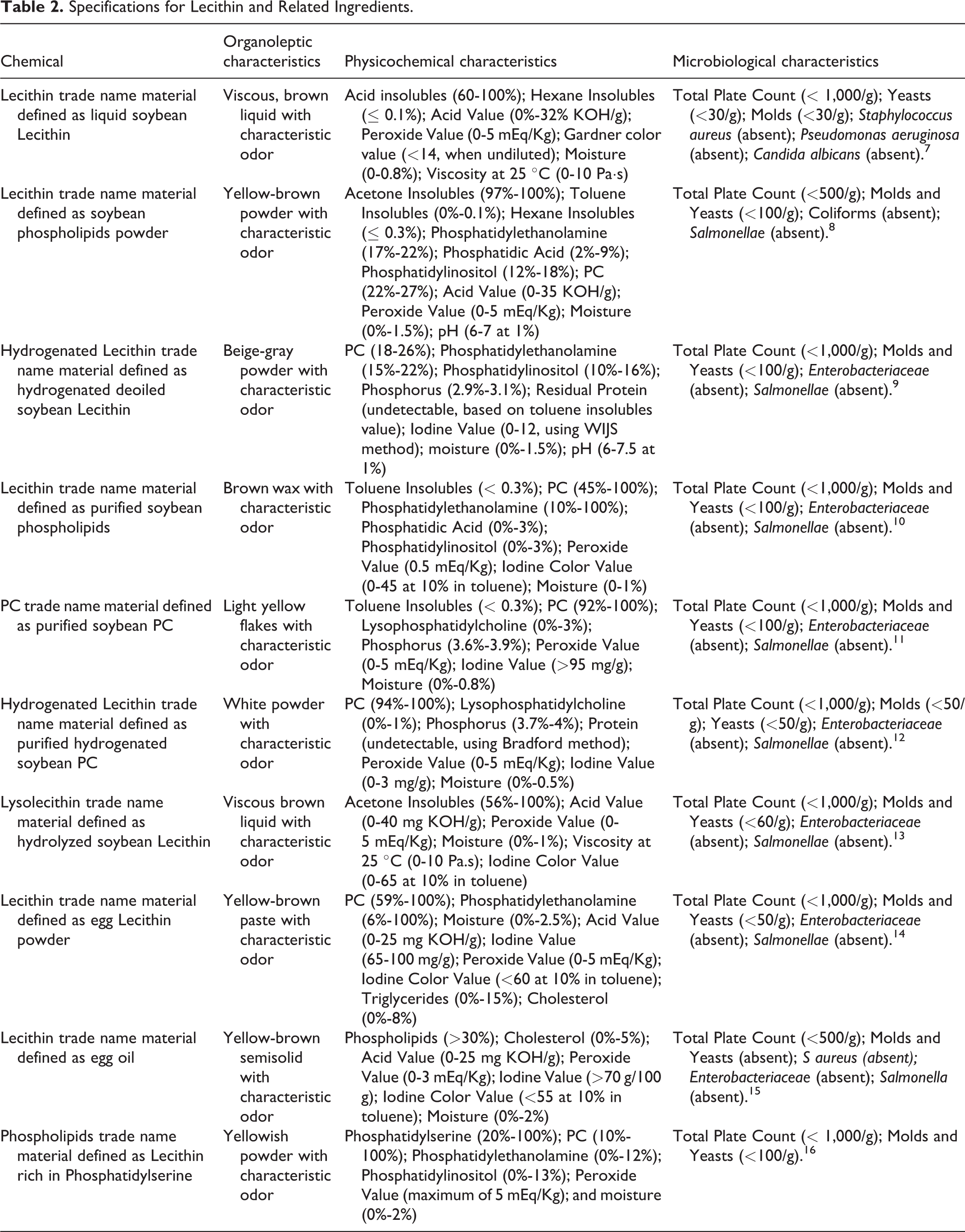
Specifications for Lecithin and related ingredients are presented in Table 2. 7 -16 Included are chemical properties data and microbiological specifications.
Specifications for Lecithin and Related Ingredients.
Method of Manufacture
Lecithin and Lecithin (Enzyme-Modified)
Commercial Lecithin is isolated as a gum following hydration of solvent-extracted soy, safflower, or corn oils. 17 Lecithin is bleached, if desired, by hydrogen peroxide and benzoyl peroxide, and dried by heating. During the manufacture of Lecithin derived from soy, most, if not all, of the soy protein is removed. If present, soy allergens would be found in the protein fraction. 18 According to another source, soy Lecithin is usually produced from the hexane extract of soybean. 19
In addition to the commercial Lecithin mentioned above, it should be noted that another form of Lecithin, enzyme-modified Lecithin (ie, Lysolecithin), is prepared by treating Lecithin with either phospholipase A2 or pancreatin. 20
Phosphoglycerides
Synthetic phosphoglycerides can be produced via phospholipase
Phosphatidylglycerol
Phosphatidylcholine (minimum purity of 90%) in the presence of enzyme and glycerin yields Phosphatidylglycerol (minimum purity of 85%) and choline. 22
Phosphatidylserine
Soy-derived Phosphatidylserine (phosphatidylserine complex derived from soy lecithin) consists of serine-substituted soy lecithin phospholipids and other phospholipids occurring naturally in lecithin. 23 Production of such soy lecithin phosphatidylserine complex involves the enzymatic transphosphatidylation of PC and phosphatidylethanolamine from soy lecithin (via cabbage-derived phospholipase in the presence of exogenous serine) to Phosphatidylserine. The production of the phosphatidylserine-enriched complex proceeds without the use of solvents during the manufacturing process. Thus, the final soy lecithin phosphatidylserine complex is solvent-free.
In addition to the preceding methods for manufacturing phospholipids, it should be noted that various other methods have been described in detail. 24
Composition
Lecithin and Lecithin (enzyme-modified)
Composition data on various phosphoglyceride tradename materials are included in Table 2. Commercial Lecithin is a naturally occurring mixture of the phosphatides of choline, ethanolamine, and inositol, with smaller amounts of other lipids. 17 Practically all of the Lecithin in commerce is derived from soybeans. Phosphoglycerides are the major constituents of Lecithin, and commercial Lecithin may contain up to 35% triglycerides. 25
Impurities
As US Food and Drug Administration (FDA) approved direct food additive for human food consumption, Lecithin (enzyme-modified) meets the following specifications
20
: Acetone-insoluble matter (phosphatides), not less than 50% Acid value, not more than 40 Lead, not more than 1.0 part per million, as determined by atomic absorption spectroscopy Heavy metals (as Pb), not more than 20 ppm Hexane-insoluble matter, not more than 0.3% Peroxide value, not more than 20 Water, not more than 4% Lysolecithin, 50 to 80 mole% of total phosphatides
The Food Chemicals Codex 26 stipulates that food grade Lecithin must not contain more than 0.3% hexane-insoluble matter. Because the protein fraction of Lecithin would reside in this insoluble material, this specification limits the amount of protein in food grade Lecithin to 0.3% or 300 mg/100 g of Lecithin.
The United States Pharmacopeia (USP) stipulates that sunflower Lecithin in food contain not more than 1% hexane-insoluble matter. 27 It is also stipulated in the USP that soy Lecithin contain not more than 20 ppm heavy metals and not more than 10 ppm lead. Potential impurities are included in the specifications for various phosphoglyceride tradename materials that are summarized in Table 2. 7 -16
Nitrosamine Formation
Lecithin
Dimethyl nitrosamine (DMNA) was reportedly formed in a model system in which 22.8 mmol sodium nitrite in 15 mL of water was added to a buffered solution, pH 5.6, containing 4.56 mmol of Lecithin and stirred at 78 °C for 4 hours. 28 The amount of DMNA formed (mg DMNA/kg of compound), confirmed by mass spectrometry, with various lecithins was as follows: soy Lecithin (edible), 2.05 ppm; soy Lecithin (commercial), 0.70 ppm; vegetable Lecithin, 1.02 ppm; egg Lecithin, 5.40 ppm; bovine Lecithin (purified), 1.66 ppm; bovine Lecithin (60%), 30.76 ppm; and synthetic Lecithin, 319.7 ppm.
Lecithin is metabolized to choline by bacterial phospholipases and the released choline is dealkylated to dimethylamine, which is N-nitrosatable in the presence of nitrate. 29 Of concern in cosmetics is the conversion (nitrosation) of nitrogen-bearing ingredients into N nitroso chemicals that may be carcinogenic. Of the approximately 209 nitrosamines tested, 85% have been shown to produce cancer in laboratory animals. 30 Nitrosation can occur under physiologic conditions. 31 Depending on the nitrosating agent and the substrate, nitrosation can occur under acidic, neutral, or alkaline conditions. Atmospheric NO2 may also participate in nitrosation in aqueous solution. 32
Use
Cosmetic
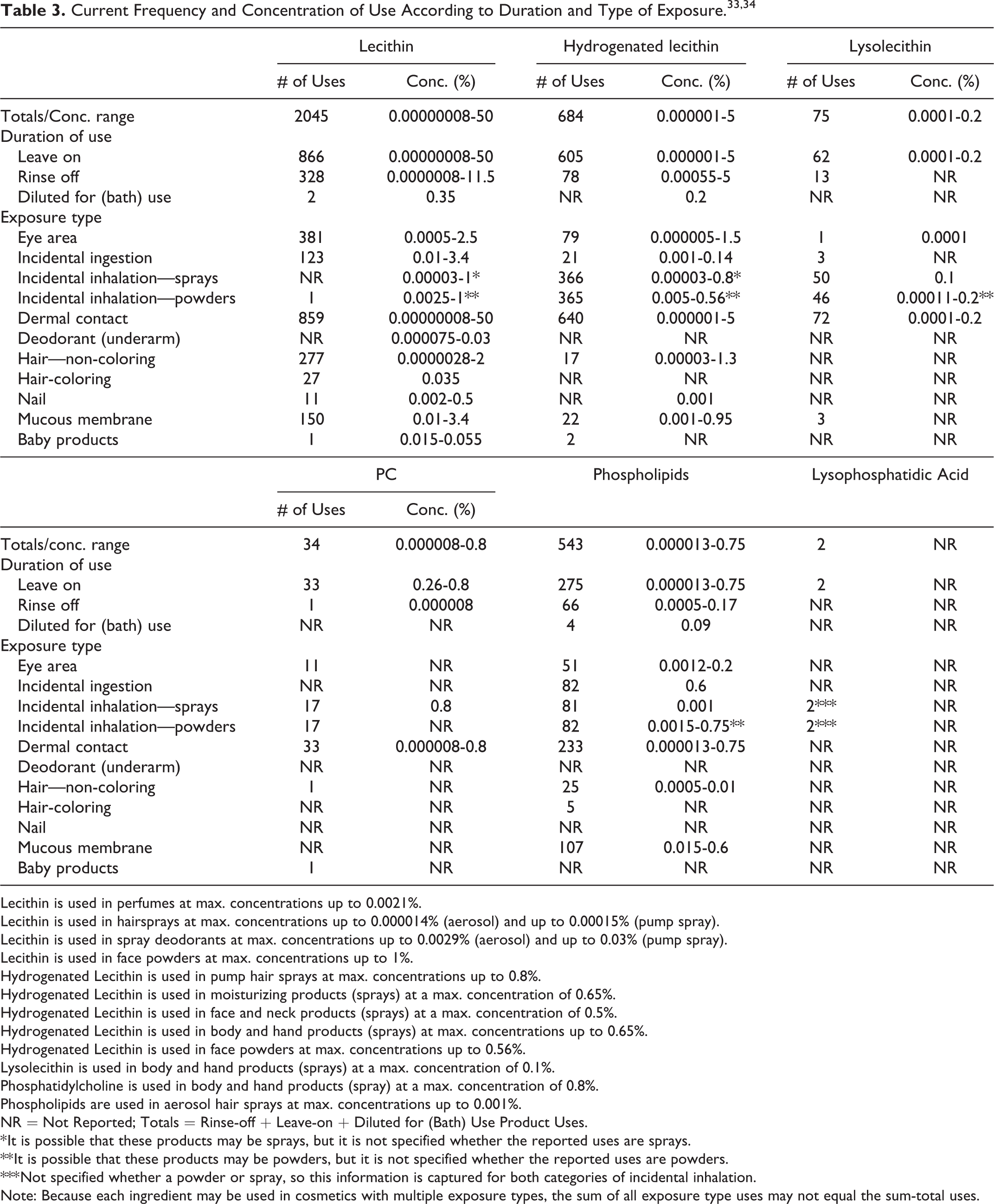
The ingredients reviewed in this safety assessment function mainly as skin and hair conditioning agents, emulsifying agents, and surfactants in cosmetic products. 6 According to information supplied to the US FDA by industry as part of the Voluntary Cosmetic Registration Program, and the results from a survey of ingredient use concentrations conducted by the Personal Care Products Council (Council), the following phosphoglycerides are used in cosmetic products at maximum concentrations ranging, for different product categories, from 0.00000008% (Lecithin in skin cleansing products and face products) to 50% (Lecithin in foot products [leave-on]) 33,34 : Lecithin, Hydrogenated Lecithin, Lysolecithin, Lysophosphatidic Acid, PC, and Phospholipids. The reported highest maximum use concentrations for other ingredients evaluated in this safety assessment are as follows, all relating to use in leave-on products: Hydrogenated Lecithin (5%, face and neck products [not spray]), Lysolecithin (0.2%, in face and neck products [not spray]), PC (0.8%, in body and hand products [not spray]), and Phospholipids (0.75%, in face and neck products [not spray]). Use frequency and concentration of use data are presented in Table 3.
Lecithin is used in perfumes at max. concentrations up to 0.0021%.
Lecithin is used in hairsprays at max. concentrations up to 0.000014% (aerosol) and up to 0.00015% (pump spray).
Lecithin is used in spray deodorants at max. concentrations up to 0.0029% (aerosol) and up to 0.03% (pump spray).
Lecithin is used in face powders at max. concentrations up to 1%.
Hydrogenated Lecithin is used in pump hair sprays at max. concentrations up to 0.8%.
Hydrogenated Lecithin is used in moisturizing products (sprays) at a max. concentration of 0.65%.
Hydrogenated Lecithin is used in face and neck products (sprays) at a max. concentration of 0.5%.
Hydrogenated Lecithin is used in body and hand products (sprays) at max. concentrations up to 0.65%.
Hydrogenated Lecithin is used in face powders at max. concentrations up to 0.56%.
Lysolecithin is used in body and hand products (sprays) at a max. concentration of 0.1%.
Phosphatidylcholine is used in body and hand products (spray) at a max. concentration of 0.8%.
Phospholipids are used in aerosol hair sprays at max. concentrations up to 0.001%.
NR = Not Reported; Totals = Rinse-off + Leave-on + Diluted for (Bath) Use Product Uses.
* It is possible that these products may be sprays, but it is not specified whether the reported uses are sprays.
** It is possible that these products may be powders, but it is not specified whether the reported uses are powders.
*** Not specified whether a powder or spray, so this information is captured for both categories of incidental inhalation.
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum-total uses.
According to the Panel final safety assessment on Lecithin and Hydrogenated Lecithin published in 2001, data received from FDA in 1984 indicated that the maximum reported use concentration of Lecithin was in the 25% to 50% concentration range; use concentration data on Hydrogenated Lecithin were not included. 1 Concentration of use data provided by the Council in 1996 indicated that 65% Lecithin was used at concentrations of 0.1% to 3%; use concentration data on Hydrogenated Lecithin were not provided. 1
Cosmetic products containing phosphoglycerides may be applied to the skin and hair or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Lecithin is used in perfumes at maximum concentrations up to 0.0021% and in hairspray formulations at maximum concentrations up to 0.000014% (aerosol) and up to 0.00015% (pump spray); Hydrogenated Lecithin is used in pump hair spray formulations at maximum concentrations up to 0.8%. Lecithin is also used in spray deodorants at maximum concentrations up to 0.0029% (aerosol) and up to 0.03% (pump spray). Phospholipids are used in aerosol hair spray at maximum concentrations up to 0.8%, and Lysolecithin, PC, and Hydrogenated Lecithin are used in body and hand sprays at maximum concentrations up to 0.1%, 0.8%, and 0.65%, respectively. Hydrogenated Lecithin is used in moisturizing sprays and face and neck sprays at maximum concentrations of 0.65% and 0.5%, respectively. Ingredient use in face powders is also reported for Lecithin (up to 1%) and Hydrogenated Lecithin (up to 0.56%). Because phosphoglycerides are used in products that are sprayed or in powder form, these could be incidentally inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays. 35 -38 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 35,36 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 36 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Because of concerns about potential transmission of bovine spongiform encephalopathy, cosmetic products are not permitted to contain ingredients made from bovine specified risk materials, which includes the central nervous system. 39
Non Cosmetic
Commercial Lecithin, defined as a naturally occurring mixture of the phosphatides of choline, ethanolamine, and inositol, is a direct food substance affirmed as generally recognized as safe (GRAS). 17 Additionally, enzyme-modified Lecithin is listed among the substances added directly to human food affirmed as GRAS. 20 Food uses of Lecithin include emulsifier, stabilizer, dispersing aid, and release agent for baked goods. 18 Lecithin is also used in topical medicaments.
The Food Allergen Labeling and Consumer Protection act (FALCPA) of 2004 altered the way in which Lecithin derived from soy must be declared on food labels. Whether intended to have a technical or functional effect in the finished food or to be used as an incidental additive (such as a release agent), Lecithin that is derived from soy must be declared as an ingredient, using its common or usual name, and with the food source (“soy,” “soya,” or “soybeans”) declared as required by section 403(w) of the Act. 18
Lecithin (source not stated) is listed as an inactive ingredient in inhaled (aerosol, metered; maximum potency = 0.0002%) drug products that have been approved by FDA, and the same is true for soybean Lecithin (aerosol, metered; maximum potency = 0.1%). Additionally, the approval of soy Hydrogenated Lecithin as an inactive ingredient in inhaled (aerosol, metered; maximum potency = 0.28%) drug products by FDA was pending at the time of this review. 40
Phosphatidylcholine is the most abundant phospholipid in mammalian cellular membranes, bile, and lipoproteins. 41 The injection of a PC and deoxycholic acid preparation is widely used as an alternative to liposuction for the reduction of subcutaneous fat. 42
Toxicokinetics
Nonhuman
Lecithin and Lysolecithin
The distribution of intravenously (IV) injected 1-[14C] palmitoyl-[32P]-lysolecithin was studied using male albino rats (number not stated). 43 The animals were injected IV with 1 mL of rat serum containing endogenously labeled [32P] Lysolecithin or with 1 mL of rat serum containing endogenously labeled [32P]-phospholipids. The amount of Lysolecithin incorporated into 1 mL of serum ranged between 550 and 850 µg. In some of the experiments, Lysolecithin labeled with both [32P] and [14C] (in fatty acid moiety) was used. At 20 and 60 minutes postinjection, 45% and 77% of injected radioactive material was removed from the blood, respectively. There was an uptake of labeled phospholipids by heart and skeletal muscle. The percentage of [32P]-Lysolecithin in these organs was much higher than in the injected material. The authors noted that the high percentage of labeled Lysolecithin in the skeletal and heart muscle indicated that Lysolecithin might leave the vascular compartment more rapidly than Lecithin and be metabolized by the tissues. The percent composition of labeled phospholipids found in the liver resembled that of the injected serum, with Lecithin as the major labeled component. However, the percentage of Lysolecithin was lower than that in the starting material. The authors noted that, based on these results, it seems more likely that the liver takes up phospholipids indiscriminately and that Lysolecithin is rapidly converted to Lecithin.
In subsequent experiments in which rats (number not stated) were injected with serum containing [32P] Lysolecithin, the labeled Lysolecithin disappeared from the bloodstream very rapidly (t 1/2 ∼ 2 minutes). Considerable amounts of Lysolecithin were recovered in the liver and skeletal muscle at a time when the serum radioactivity decreased to negligible levels. The conversion of Lysolecithin to Lecithin was observed in all the tissues examined, and this reaction was most rapid in the liver. Lysolecithin taken up by the liver was converted to Lecithin by an acylation reaction. 43
Lysophosphatidic Acid
Lysophosphatidic Acid was degraded to glycerophosphate and orthophosphate by phosphatidases and phosphatases, respectively, in an enzyme preparation, that is, cytoplasmic particulate fraction of guinea pig brain or liver. 44
Phosphatidylserine
Following IV administration to rats and mice, Phosphatidylserine was eliminated from plasma in a biphasic manner and largely distributed to several major organs, including the liver spleen and brain. 45 -49 Conversely, orally administered Phosphatidylserine was extensively hydrolyzed by phospholipase A2 to lysophosphatidylserine in the gastrointestinal tract prior to absorption, as is the case for all other dietary phospholipids. 45,50,51 In rats, approximately 60% of an orally administered dose of Phosphatidylserine (20 mg/kg body weight) was recovered in the feces, of which 50% was identified as lysophosphatidylserine. Approximately 10% of the orally administered dose was detected in the urine. 48
Studies in which animals were injected IV with radiolabled Phosphatidylserine also indicate that phospholipids undergo hydrolytic cleavage to the monoacyl derivative lysophosphatidylserine in the plasma as well as decarboxylation of the serine moiety to phosphatidylethanolamine in circulating blood cells. 46,47,49 Lysophosphatidylserine and phosphatidylethanolamine were also detected in the liver and brain after IV administration. However, in all organs, the majority of radioactivity (∼90%) was consistently accounted for as unmetabolized Phosphatidylserine. 48,49 Conversely, at 60 minutes post oral dosing of radiolabled Phosphatidylserine (20 mg/kg) in rats, the majority of the circulating plasma radioactivity consisted of Phosphatidylserine; the radioactivity at 24 hours was attributed primarily to Phosphatidylserine degradation products. 48 Furthermore, less than 20% of the administered dose recovered in the mesenteric lymph of rats after oral administration of Phosphatidylserine (560 mg/kg body weight of [3 H]-glycerol-labeled, brain-derived Phosphatidylserine) was liposoluble, with phospholipids comprising 11% of the liposoluble fraction. 51 The majority of the radioactivity was recovered as triglycerides and, to a smaller extent, diacylglycerol, indicating complete degradation of orally administered Phosphatidylserine.
In the mitochondria of mammalian cells, Phosphatidylserine may undergo decarboxylation to phosphatidylethanolamine, which is followed by potential reformation of Phosphatidylserine through exchange of the ethanolamine moiety with serine in the endoplasmic reticulum or mitochondria-associated membrane. 52 Thus, in the intestinal absorptive cells, lysophosphatidylserine may be reacylated to yield Phosphatidylserine and ultimately converted to phosphatidylethanolamine. 51 Re-esterified phospholipids are subsequently incorporated into intestinal lipoproteins (ie, chylomicrons) or directly transported as lysophospholipids via the portal system to the liver. 53,54 As the chylomicrons circulate in the blood, their components, including phospholipids, are degraded via lipoprotein lipase hydrolytic activity. 54 Ultimately, the Phosphatidylserine degradation products (ie, free fatty acids, serine, glycerol, and phosphorus-containing substances) enter common physiological pathways of amino acid and lipid metabolism. In turn, intact phospholipids are excreted in the bile and thus may be subject to enterohepatic circulation.
Pharmacokinetic studies indicate exogenous Phosphatidylserine crosses the blood–brain barrier, where it appears to have an affinity for the hypothalamus. 55 Oral administration results in peak levels in 1 to 4 hours.
Human
In 8 human subjects, the oral consumption of 500 mg Phosphatidylserine (as soy lecithin phosphatidylserine capsules) resulted in peak plasma Phosphatidylserine levels of 3.95% of the total phospholipid plasma concentration, compared to background Phosphatidylserine levels of 1.8% to 2.2% of total plasma phospholipids. 23
Skin Penetration Enhancement
The effects of PC and Hydrogenated PC on the permeation of indomethacin through hairless rat skin were studied using liquid paraffin and a gel prepared with liquid paraffin and hydrogenated soybean phospholipid. 56 Indomethacin (1%) was mixed with liquid paraffin and PC or Hydrogenated PC and heated at 95 °C for 30 minutes. The mixture was then cooled to room temperature and allowed to stand for 1 day. Skin permeation was measured using a modified Franz-type diffusion cell apparatus. Permeation rates for indomethacin from the liquid paraffin suspension with PC or Hydrogenated PC were determined. For liquid paraffin without PC or Hydrogenated PC, permeation of indomethacin was observed only after 10 hours. However, within 10 minutes, indomethacin permeated at rates of ∼ 10 µg/cm2 and 5 µg/cm2 from liquid paraffin with PC and Hydrogenated PC, respectively.
The effect of hydrophilic groups of phospholipids on the percutaneous penetration of indomethacin in vitro was examined in a Franz-type diffusion chamber, using dorsal skin from guinea pigs. 57 The following phospholipids were evaluated for enhancement of indomethacin skin penetration: PC, phosphatidylethanolamine, Phosphatidylinositol, Phosphatidylserine, Phosphatidylglycerol, Phosphatidic Acid, and sphingomyelin. Phospholipid-induced enhancement of indomethacin percutaneous penetration was in the following order: Phosphatidylglycerol > phosphatidylethanolamine > PC > Phosphatidylserine > Phosphatidic Acid > Phosphatidylinositol > control > sphingomyelin.
Toxicology
Single-Dose (Acute) Toxicity
Oral
Phosphatidylserine
The oral administration of a purified phospholipid preparation obtained from bovine cerebral cortex (in phosphate buffer suspension) to Sprague-Dawley rats (CD strain; number not stated) indicated that the LD50 is > 5 g/kg body weight. 58
Intravenous
Phosphatidylserine
Following the IV dosing of Sprague-Dawley rats (CD strain; number not stated) with Phosphatidylserine derived specifically from bovine cerebral cortex (in phosphate buffer suspension), an LD50 of 236 mg/kg body weight was reported. 58
Subcutaneous
Phosphatidylserine
When Sprague-Dawley rats (CD strain; number not stated) were dosed subcutaneously (SC) with Phosphatidylserine derived specifically from bovine cerebral cortex (in phosphate buffer suspension), the LD50 was reported to be > 5 g/kg body weight. 58
Repeated Dose Toxicity
Inhalation
Phosphatidylcholine
The effects of chronic exposure to liposome aerosols on lung histology and alveolar macrophage function were studied. 59 Liposomes were made from hydrogenated soy PC (50 mg/mL). Groups of 30 (strain not stated) mice were placed in a nose-only exposure module and exposed to liposome (20-mL total volume, 50-mg lipid/mL phosphate-buffered saline) or saline aerosols 1 hour per day, 5 days per week, for 4 weeks. Five mice of both the experimental and control groups were removed weekly and their lungs examined. The animals were killed and bronchoalveolar lavage (BAL) was performed through a tracheostomy. In vivo uptake of liposomes by alveolar macrophages was documented by fluorescence microscopy and flow cytometry of BAL. A consistent amount of 1 to 3 µg of lipid inhaled per dosing per mouse was estimated from fluorescence measurements. No histologic changes of the lungs or untoward effects on general health or survival of animals were noted. Alveolar macrophage phagocytic function was not affected. Transmission electron microscopy and morphometry showed no treatment-related alterations.
Oral
Nonhuman
Lecithin
A group of 48 male and 48 female SPF Wistar rats was fed 4% (soya) Lecithin for 2 years, while a control group was fed commercial diet only. 60 Feed consumption and body weights were determined prior to dosing, at intervals up to week 95, at week 102, and at study termination. The mean Lecithin intake was 1,470 and 2,280 mg/kg/d for males and females, respectively. No statistically significant differences were observed in mortality, feed consumption, or body weight between the treated and control groups, but it was noted that feed consumption and body weight were sometimes greater in the treated group when compared to controls. Hematology values of animals of the treated group were similar to those of control animals, as were organ weights and gross and microscopic alterations. Increased parathyroid gland hyperplasia, particularly in the males, was attributed to an increased phosphate intake. The incidence of tumor formation was similar in the treated and control groups. A slightly increased incidence of myocardial fibrosis was associated with parathyroid gland hyperplasia.
Lecithin, Phosphatidylserine, PC, Phosphatidylethanolamine, Phosphatidylinositol, and Phosphatidic Acid
A 90-day feeding study was performed to evaluate the safety of dietary soy lecithin transphosphatidylated phosphatidylserine (soybean-derived Phosphatidylserine [SB-PS]), with or without fish oil-derived long-chain polyunsaturated fatty acids (LC-PUFA) mixed or conjugated to the glyceride backbone.
61
One hundred two male Wistar rats (wild type, pathogen free) were randomly assigned to 6 groups. The 5 groups consumed 100 mg chow containing each of the following components, respectively, incorporated in 1 mL of milk-based supplement matrix: medium-chain triglycerides (MCT group) fish oil diluted with MCT to yield 30% (wt/wt) of omega-3 LC-PUFA (omega-3 group) soybean 78% powdered SB-PS (final concentration of 20% SB-PS [wt/wt]) emulsified with 13% PC, 2% Phosphatidylethanolamine, 1% Phosphatidylinositol, 4% Phosphatidic Acid, and further diluted with MCT (SB-PS group) fish oil mixed with soybean 78% powdered SB-PS and diluted with MCT to yield a final concentration of 20% SB-PS (wt/wt) and 30% (wt/wt) of omega-3 LC-PUFA (omega-PS group) 20% Phosphatidylserine (wt/wt) consisting largely of molecular species of palmitic acid (16:0) and docosahexaenoic acid (DHA) (22:6) or eicosapentaenoic acid (20:5), resulting in 30% (wt/wt) of omega-3 LC-PUFA (PS-DHA group).
The control group consumed normal chow. Blood samples were drawn, and hematological parameters evaluated. Signs of toxicity were not observed during the feeding period. At the end of the study, gross examination of organs was performed. The following mortalities were reported: 1 rat (control group), 2 rats (MCT group), 1 rat (omega-3 group), and 1 rat (PS-DHA group). Pathological examinations did not reveal a specific cause of death; however, the authors concluded that the deaths were not treatment-related. Hematological parameters were normal in all treatment groups. At gross pathological examination, there were mild signs of liver enlargement in 5 of 102 rats, but these were considered unrelated to treatment. Possible early signs of lung metastasis (pale color nodes and different tissue consistency) were observed in 4 of 102 rats, but these findings were considered typical and abundant in rats of this age (15 months old). It was noted that none of these pathological findings occurred in PS-fed rats. It was concluded that no adverse effects were associated with diets fed in this study.
Phosphatidylserine
The repeated dose toxicity of Phosphatidylserine derived specifically from bovine cerebral cortex (in phosphate buffer suspension) was studied using 3 groups of Sprague-Dawley rats of the CD strain (20 males, 20 females/group). 58 The 3 groups received doses of 10, 100, and 1,000 mg/kg/d, respectively, by gavage for 26 weeks. The control group was dosed with phosphate buffer only. Body weight gain and food consumption in all dose groups were comparable to the control group. No significant hematological changes were observed. At week 13, slightly elevated alkaline phosphatase levels in male and female rats and slightly lowered serum albumin levels in males was observed in the 1,000 mg/kg/d dose group. Elevated potassium and lower sodium values were reported for males at week 13. Terminal studies indicated no major problems, and there were no significant morphological changes. It was concluded that Phosphatidylserine derived specifically from bovine cerebral cortex did not cause significant toxicity in this study.
Another repeated dose toxicity study involved groups of 40 beagle dogs (20 males, 20 females). 58 Three groups received Phosphatidylserine derived specifically from bovine cerebral cortex (in corn oil) orally at doses of 10, 100, and 1,000 mg/kg/d (dose volume = 5 mL/kg), respectively, for 1 year. The control group was dosed with corn oil. None of the animals died. At the highest dose administered, blood glucose and cholesterol levels were significantly lowered. There were no significant macroscopic findings, and organ weights were within normal range. Histopathological examination of tissues did not indicate treatment-related changes. It was concluded that Phosphatidylserine derived specifically from bovine cerebral cortex did not cause significant toxicity in this study.
Human
Phosphatidylserine
Human subjects (120) received soy lecithin-derived Phosphatidylserine (300 or 600 mg orally) daily in a 12-week study. 62 There were no clinically significant variations in blood chemistry or hematology. Additionally, there were no differences in the occurrence of side effects between test and placebo groups.
Intravenous
Phosphatidylserine
Phosphatidylserine derived specifically from bovine cerebral cortex (in phosphate buffer suspension) was administered IV to groups of Sprague-Dawley rats of the CD strain (10 males, 10 females/group). 58 The 3 groups received doses of 5, 20, and 80 mg/kg/d (dose volume = 2.5 mL/kg), respectively, for 4 weeks. The control group was dosed with vehicle only. Except for females of the 5 mg/kg/d group, reddening and swelling of the paws and around the muscle region were observed in all dose groups. The following hematological changes were observed in male and female rats of the 80 mg/kg/d dose group: significant lowering of the erythrocyte count, hemoglobin concentration, and packed cell volume and increased neutrophil and lymphocyte counts. An increase in spleen weight in males and females of the 80 mg/kg dose group and males of the 20 mg/kg/d dose group was reported. Kidney weights of males dosed with 80 or 20 mg/kg/d were also increased when compared to controls. Adrenal weights of males and females of the 80 mg/kg dose group were marginally increased. Results at microscopic examination revealed an injection site thrombosis in some rats in all dose groups, with an increase in severity in rats dosed with 20 mg/kg/d or 80 mg/kg/d. Whether or not this finding was reported for the control group was not stated. An increase in the incidence of extramedullary hematopoiesis was observed in the 80 mg/kg/d dose group. It was concluded that Phosphatidylserine derived specifically from bovine cerebral cortex did not cause significant toxicity in this study.
The IV toxicity of Phosphatidylserine derived specifically from bovine cerebral cortex (in phosphate buffer suspension) was evaluated using groups of 24 (12 males, 12 females) Beagle dogs. 58 Three groups were injected IV with Phosphatidylserine derived specifically from bovine cerebral cortex at doses of 5, 15, and 40 mg/kg/d, respectively, for 4 weeks. A fourth group was dosed with vehicle only. Generalized tremors of body muscles were observed in animals of the 15 or 40 mg/kg/d group. A significant increase in total white cell count and a reduction in total serum protein values were reported for dogs dosed with 40 mg/kg/d. At gross examination, hemorrhage was observed around the injection sites. There were no significant group differences in organ weights. Microscopic examination of the liver revealed centrilobular and periportal sinusoidal aggregations of polymorphonuclear leukocytes in 1 animal of the 15 mg/kg/d dose group and in 4 animals of the 40 mg/kg/d dose group. It was concluded that Phosphatidylserine derived specifically from bovine cerebral cortex did not cause significant toxicity in this study.
Intramuscular
Phosphatidylserine
The intramuscular toxicity of Phosphatidylserine derived specifically from bovine cerebral cortex (in phosphate buffer suspension) was evaluated using groups of 32 (16 males, 16 females) Beagle dogs. 58 Three groups were injected intramuscularly with Phosphatidylserine derived specifically from bovine cerebral cortex at doses of 5, 10, and 15 mg/kg/d, respectively, for 6 weeks. A fourth group served as the vehicle control. None of the animals died. Subcutaneous hardening and/or swelling of injection sites was observed in the 10 and 15 mg/kg/d dose groups. Hematological analyses indicated elevation of the erythrocyte sedimentation rate and an increase in total white blood cell count in the 15 mg/kg dose group. At gross examination, subcutaneous hemorrhage and adhesion between the skin and muscles (at injection site) was reported for all groups, including the control group. This finding was considered dose-related. Organ weights were within normal ranges. Muscle degeneration and subcutaneous and intramuscular acute inflammatory cell infiltration and necrosis were also observed at injection sites. It was concluded that Phosphatidylserine derived specifically from bovine cerebral cortex did not cause significant toxicity, that is, there were no significant signs of systemic toxicity.
Cytotoxicity
Lysolecithin
Lysolecithin has been described as a powerful hemolytic and cytolytic phosphoglyceride. 63,64 Furthermore, the toxic effect of many snake venoms is attributable to their content of phosphatidase A, an enzyme capable of converting plasma phosphatides into lysophosphatides, one of which is Lysolecithin.
Reproductive and Developmental Toxicity
Phosphatidylserine
In a teratogenicity study, Phosphatidylserine derived from bovine cerebral cortex was administered by gavage to pregnant Sprague-Dawley rats (CD strain; number not stated) at doses of 0, 10, 100, and 200 mg/kg/d on days 6 through 18 of gestation. 58 The animals were killed on day 20 of gestation, and litters were examined for skeletal and visceral abnormalities. At terminal necropsy, there were no treatment-related gross changes. The following litter values were not affected by treatment with Phosphatidylserine: litter size, postimplantation loss, litter and mean fetal weights, and embryonic and fetal development.
Phosphatidylserine derived from bovine cerebral cortex was also administered by gavage to pregnant New Zealand White rabbits (number not stated) at doses of 0, 50, 150, and 450 mg/kg/d on days 6 through 18 of gestation. 58 On gestation day 29, the animals were killed and litters subjected to gross examination. Fetuses were examined externally and internally for evidence of visceral and skeletal malformations. There was no evidence of systemic effect, and neither pregnancy nor mortality was affected by treatment. At the highest dose, mean fetal weights were slightly lower when compared to control values, but the difference was not statistically significant. There were no treatment-related effects on embryonic and fetal development.
Lysophosphatidic Acid
Lysophosphatidic acid and sphingosine-1-phosphate are both lysophospholipids. 65 Because Lysophosphatidic Acid promotes prostaglandin synthesis, mediators in the lysophosphatidic acid pathway may also play a significant role in implantation and parturition. Sphingosine-1-phosphate signaling is thought to be essential in vascular formation within the uteroplacental unit and in fetomaternal immunologic interactions. Derangements in either one of these lysophospholipid signaling pathways could result in pregnancy complications that may include implantation failure, preeclampsia, and preterm labor.
Immature germinal vesicle stage oocytes from 5- to 6-week-old female BDF-1 mice were incubated for 17 to 18 hours in in vitro maturation (IVM) medium containing 0-, 1-, 10-, or 30-µM Lysophosphatidic Acid and then either fertilized in vitro with epididymal sperm or assessed for spindle morphology or mitochondrial membrane potential. 66 Chromosomal aneuploidy in the resultant blastocysts and the production of normal pups were not assessed. The fertilized embryos were grown in vitro to assess blastocyst formation rates, differential cell counts and apoptosis. The supplementation of IVM with 30-µM Lysophosphatidic Acid enhanced the maturation and developmental competence of mouse oocytes. Rates of maturation, fertilization, and blastocyst formation and hatching were significantly higher in the 30-µM Lysophosphatidic Acid-supplemented group (94.3%, 96.3%, 79.1%, and 51.3%, respectively) than in the unsupplemented control (0 mM) group (80.5%, 87.5%, 61.3%, and 37.8%, respectively), and more comparable to that of the in vivo matured oocytes (100%, 96.5%, 95.3%, and 92.9%, respectively). Lysophosphatidic acid did not adversely affect mitochondrial activity, spindle integrity, or blastocyst cell number. The results of this study imply that the supplementation of IVM medium with 30-µM Lysophosphatidic Acid may enhance the developmental competence of mouse oocytes without affecting apoptosis, spindle normalcy or mitochondrial integrity.
Genotoxicity
In Vitro
Hydrogenated Lecithin
The genotoxicity of liposome-fullerene (Lpsm-Flln) or liposome solution (Lpsm; 313-5,000 μg/plate) was examined, with and without metabolic activation, using Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2uvrA (pKM101). 67 Neither liposome was genotoxic in this assay, with or without metabolic activation. The following 5 positive controls were genotoxic: sodium azide, N-ethyl-N’-nitro-N-nitrosoguanidine, 2-nitrofluorene, 9-aminoacridine, and 2 aminoanthracene.
Phosphatidylserine
Human lymphocyte cultures were incubated with Phosphatidylserine derived from bovine cerebral cortex concentrations up to 165.6 µg/mL, with and without metabolic activation. 58 Cyclophosphamide served as the positive control. Phosphatidylserine derived specifically from bovine cerebral cortex was not genotoxic with or without metabolic activation.
In the mouse lymphoma assay, Phosphatidylserine derived from bovine cerebral cortex was not genotoxic to mouse lymphoma L5178Y cells with or without metabolic activation. 58 Test concentrations were not stated.
Phosphatidylserine derived from bovine cerebral cortex was evaluated in a DNA repair assay involving human epithelioid cells (HeLa S3 cells), with and without metabolic activation. 58 Test concentrations were not stated. Increases in the number of silver grains found in autoradiographic film over cell nuclei served as indicators of repair synthesis. There was no evidence of DNA repair synthesis, with or without metabolic activation.
In Vivo
In the micronucleus test, Phosphatidylserine derived from bovine cerebral cortex was administered orally (by gavage) to mice at doses up to 300 mg/kg body weight. 58 Two equal doses were administered, separated by a 24-hour interval. Mitomycin C served as the positive control. Bone marrow smears were examined for the presence of micronuclei in 1,000 polychromatic erythrocytes per mouse and for the ratio of normochromatic to polychromatic erythrocytes. Phosphatidylserine derived from bovine cerebral cortex was neither cytotoxic nor genotoxic to bone marrow cells.
Modulation of Gene Expression
Phospholipids
The capacity of a formulation of grape seed extract and soy Phospholipids (formulation identified as SBD.5HC) to trigger a regenerative response in the dermis and epidermis through a selective action on the hypodermis was investigated using human skin (from breast reduction surgeries). 68 SBD.5HC was prepared by combining grape seed extract (95% proanthocyanidins grade) and soy Lecithin (95%-98% phospholipids grade) at a ratio of 1:3 wt/wt. After 5 days of culture under control conditions, full-thickness human skin biopsies showed marked degradation, characterized by pyknotic nuclei in fibroblasts and basal keratinocytes as well as intercellular gaps in spiny and granular layers of the epidermis. The inclusion of SBD.5HC (100 μg/mL) in the medium bathing the hypodermal layer of the biopsies resulted in an improved overall morphology. Treated skin samples had mostly normal, elongated fibroblasts, fewer dying basal keratinocytes at the dermal–epidermal junction, less gaping spaces in the stratum spinosum, and better-preserved granulosum and stratum corneum. Thus, study results suggested that the application of SBD.5HC to the hypodermal layer of skin triggered modulation of gene expression in the upper layers of skin and resulted in morphological changes in the dermis and epidermis.
Carcinogenicity
Lecithin
TM strain mice were fed 5- to 10-mg Lecithin mixed with sugar (for palatability), and a second group was fed Lecithin (5-10 mg) and cholesterol (4-5 mg). 69 The mice were bred and their offspring dosed following the same procedures; dosing continued until all mice became moribund or had died. A control group was given laboratory feed ad libitum. The total number of mice fed Lecithin, Lecithin and cholesterol, or control feed was 166, 212, and 360, respectively. Animals were killed and brain necropsies performed. It was noted that the brains of moribund animals or animals found dead were removed and necropsied, but necropsy results were not reported. Brain nerve cell tumors (2-5 mm) were found in 18 of 73 examined animals fed Lecithin and in 27 of 88 examined animals fed Lecithin and cholesterol, whereas no brain nerve cell tumors were found in 188 control animals.
Groups of female dd mice were dosed SC as follows: 50 mice were given 0.1 mL of a 0.25% mixture of 4 nitroquinoline-1-oxide (in 10% aqueous Lecithin) in a single injection until the total dose was 2.5 mg. 70 The injections were repeated weekly, each time in a different site on the back. Thirty mice were dosed (10 times) with a Lecithin water mixture at the same total dose as in the previous group. Twenty mice were not dosed and served as controls. The mice were killed after 221 to 296 days. Animals dosed with 4-nitroquinoline-1-oxide/Lecithin that survived more than 221 days after dose initiation (36/50) had pulmonary neoplasms; skin neoplasia at the injection site (1 animal) and leukemia (1 animal) were also observed in this group. No surviving mice dosed with Lecithin water or untreated control mice had pulmonary or any other type of neoplasia. However, 3/28 animals of the Lecithin water group and 3/18 control animals had lung adenomas; these were considered spontaneous.
In the same study, groups of female Buffalo rats were dosed SC as follows: 25 rats were given 0.2 mL of a 0.25% mixture of 4-nitroquinoline 1-oxide (in 10% aqueous Lecithin) in a single injection until the dose reached 10 mg; the injections were repeated weekly. Fifteen rats were dosed (20 times) with a Lecithin water mixture, having received the same total dose. The rats were killed after 264 to 329 days. Nineteen of the 25 animals dosed with 4-nitroquinoline 1-oxide/Lecithin that survived more than 264 days after dose initiation had pulmonary neoplasms, with 11 SC sarcomas and 2 endometrial sarcomas also reported. No neoplasms were found in any of the 13/15 surviving rats dosed with Lecithin water. 70
Irritation and Sensitization
Ocular Irritation
Lecithin
Lecithin 65% (solution of 65% Lecithin) and products containing 2.25% or 3.0% Lecithin 65% were non- to minimally irritating to unrinsed rabbit eyes. A soap containing 0.83% Lecithin powder (tested at 25%) was moderately irritating, and Lecithin-containing liposomes were practically nonirritating in a Draize test. 1
Skin Irritation and Skin Sensitization
Nonhuman
Lecithin and Hydrogenated Lecithin
In single-insult occlusive patch tests (rabbits), Lecithin 65% (solution of 65% Lecithin) was minimally irritating, products containing 3% Lecithin 65% were practically non- to mildly irritating, and a product containing 2.25% Lecithin 65% was non-irritating to the skin of rabbits. In a guinea pig immersion study, 0.5% of a soap containing 0.83% Lecithin powder was practically nonirritating. Hydrogenated Lecithin was not a primary dermal irritant in rabbits. 1
Human
Lecithin and Hydrogenated Lecithin
In clinical irritation studies, cosmetic formulations containing 0.3% or 3% Lecithin 65% (solution of 65% Lecithin), a soap containing 0.83% Lecithin powder (tested at 0.5%), and Lecithin liposomes were generally nonirritating. Barely perceptible erythema was the most severe reaction observed. Hydrogenated Lecithin also was not an irritant, and Hydrogenated Lecithin (15% in petrolatum) was not a sensitizer. Additionally, a tanning oil containing 3% Lecithin 65%, a mascara containing 0.1% Lecithin 65%, and a foundation containing 0.3% Lecithin 65% were non-sensitizing. 1
Lysolecithin
The intracutaneous injection of 0.04-µM to 0.25-µM Lysolecithin, derived from beef serum, human serum, or beef brain Lecithin, caused typical wheal and erythema reactions in the 3 subjects tested. 71 Lysolecithin (0.125 and 0.17 µM) produced wheal and erythema reactions that were roughly equivalent to that produced by the injection of histamine (0.5 µg). These reactions consisted of a pale, elevated central swelling (occasionally with small pseudopods), surrounded by a bright red zone of erythema. The lower concentrations of Lysolecithin (0.085 and 0.043 µM) caused minor reactions that were smaller than those obtained with 0.3-µg histamine, but slightly greater than those caused by 0.1-µg histamine (slight threshold reaction). A faint, but definite, reaction was observed at concentrations as low as 0.013 µM in another experiment.
Allergenicity
Lecithin
The antigenicity of soy Lecithin was studied using 30 soybean-sensitive patients and 22 controls. 19 One control group (11 subjects) consisted of nonatopic individuals, and the other control group (11 subjects) consisted of allergic patients with negative IgE to soybean (radioallergosorbent test [RAST] score = 0). The IgE- and IgG4-binding activities of the soy Lecithin proteins were evaluated by immunoblotting with sera obtained from the patients, 7 of whom had a positive challenge test. In 100 grams of sample, the soy Lecithin contained 2.8 mg of proteins. The proteins present in soy Lecithin were analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis. For the soy Lecithin, the detection rate of only one protein (molecular weight: 31 kDa) by the serum IgE of patients was statistically significantly different when compared to serum from the 2 control groups combined (detection rates: 40% [patient sera] and 4.5% [control sera]). Proteins in the molecular weight range of 58 to 67 kDa were rarely bound to serum IgE. Only one of the patients with a positive challenge test had IgE antibodies to soy Lecithin proteins. The presence of IgG4-binding proteins in soy Lecithin was described as rare. It was concluded that the proteins present in soy Lecithin have little antigenicity with respect to soybean allergy.
Photocytotoxicity
Nonhuman
Hydrogenated Lecithin
The photocytotoxicity of Lpsm-Flln, 0.2% aqueous, was studied using Balb/3T3 fibroblastic cells. 67 Bacterial assay results for this liposome are included in the Genotoxicity section of this report. Lpsm-Flln had the following composition: Hydrogenated Lecithin, glycine soja (soybean) sterols, and fullerene C60 (C60) in the weight ratio of 89.7:10:0.3 (ie, contains 89.7% Hydrogenated Lecithin). Results were compared with that of a 0.2% Lpsm that did not contain C60, described as follows: Hydrogenated Lecithin and glycine soja (soybean) sterols in the weight ratio of 90:10 (ie, contains 90% Hydrogenated Lecithin). The fibroblasts (in Lpsm-Flln or Lpsm at doses of 0.49 to 1,000 µg/mL) were exposed to sham-irradiation or long-wavelength ultraviolet light (UVA; 5 J/cm2; 320-400 nm; λmax = 360 nm) for 50 minutes. Cell viability of Balb/3T3 fibroblastic cells in Lpsm-Flln was 96.3% to 158.5% for the UVA group and 94.5% to 149.6% for the sham group and did not decrease dose-dependently. Also, cell viability in Lpsm was like that in Lpsm-Flln. These results show that Lpsm-Flln (89.7% Hydrogenated Lecithin) or Lpsm (90% Hydrogenated Lecithin) at a concentration of 0.2% was not photocytotoxic to Balb/3T3 fibroblasts.
Phototoxicity/Photosensitization
Lecithin and Hydrogenated Lecithin
A foundation containing 0.3% Lecithin 65% (solution of 65% Lecithin) was not a photosensitizer in human subjects. The subjects were exposed for 1 minute to a UV light source (360 nm peak output), at 12 inches, after removal of the first, fourth, seventh, and 10th induction patches and challenge patches. Photosensitization reactions were determined 48 hours after exposure. Lecithin and Hydrogenated Lecithin (both at 15% in petrolatum) were not phototoxic or photosensitizing in human subjects. On days 1, 4, and 7 of induction, patches were removed, and test sites were irradiated with a dose of 3 MED of UVA. A fourth induction patch was also applied, followed by irradiation of the test site. Challenge patch sites were exposed to a dose of 9.5 MED and 0.5 MED of simulated solar light (UVA and mid-wavelength UV [UVB]). 1
Case Report
Lecithin
A 3-year-old boy with a history of asthma and peanut allergy was treated for asthma that developed after an upper respiratory tract infection. 72 He developed respiratory distress and generalized urticaria within an hour after receiving the second of 2 inhalations of an ipratropium bromide inhaler. All signs regressed within 48 hours of withdrawal of the drug. Soy Lecithin, an excipient in the metered dose inhaler, was strongly suspected of causing the adverse events.
Other Studies
Phospholipid Signaling
The organization of biological systems involves communication between cells, and in multicellular organisms, these interactions are mediated through cell–cell contacts. 73 In order for a cell to react, a signal must be detected and converted across the physical boundary that is defined by the cell membrane. Membrane lipids have important roles in signaling reactions. These ligands can function as signaling molecules in various ways, and the binding of a ligand to a cell surface receptor is an initiating event of cellular signaling. Transmembrane signaling frequently includes the activation of enzymes that act on metabolizing lipids in the vicinity of the respective receptor, leading to the generation of membrane-bound and diffusible metabolites. Receptor stimulation is often accompanied by the activation of downstream factors that are regulated by products of lipid metabolism. Some of the phospholipids involved in signaling include Phosphatidylinositol, PC, Lysophosphatidic Acid, and Phosphatidylserine. Signaling effects relating to these phospholipids are summarized below.
Minor products of inositol phospholipid metabolism, such as phosphatidylinositol-3,4,5-triphosphate, serve as key intermediates in cell signaling. 74 Furthermore, phosphoinositides, a family of lipid molecules derived from the phosphorylation of Phosphatidylinositol, control important cellular processes, including cell proliferation, apoptosis, metabolism, and migration. 75 Phosphoinositides make up only a small fraction of cellular phospholipids, yet they control almost all aspects of a cell’s life and death. 76 The specific interaction of phosphoinositides with proteins is critical for a plethora of cellular processes, including cytoskeleton remodeling, mitogenic signaling, ion channel regulation, and membrane trafficking. 77 Phosphoinositide homeostasis is tightly regulated by a large number of inositol kinases and phosphatases that have been implicated in regulating membrane trafficking, and the dysregulation of these enzymes has been linked to a number of human diseases, ranging from cancer and diabetes to neurological disorders and asthma.
Cancer cells display sensitivity to ablation of fatty acid synthesis, possibly as a result of the diminished capacity to synthesize complex lipids involved in signaling or growth pathways. 78 Evidence has accrued that PC, the major phospholipid component of eukaryotic membranes, as well as choline metabolites derived from its synthesis and catabolism, contributes to both proliferative growth and programmed cell death. Coordinated changes in substrate availability, gene expression, and enzyme activity lead to altered PC synthesis in cancer.
Lysophosphatidic acid is capable of stimulating a plethora of different cellular responses through the activation of its family of cognate G protein-coupled receptors. 79 It mediates a wide range of biological effects in many tissue types, including vasculogenesis, angiogenesis, and vascular maturation, and has also been implicated in the regulation of pathophysiologic vascular responses. For example, Lysophosphatidic Acid was found to signal through G protein α q subunit (Gαq) to promote the growth and migration of vascular smooth muscle cells, which is essential for the development of intimal hyperplasia after vascular injury.
Phosphatidylserine-specific binding is important in the function of A-, B-, and C-Raf kinases, which are important regulators of many signal transduction pathways. Raf kinases are generally downstream from the Ras GTPases and transmit information to activate mitogen-activated protein kinase signaling. 80 The activation of protein kinase B, Raf-1, and protein kinase C signaling, which supports neuronal survival and differentiation, requires the interaction of these proteins with Phosphatidylserine. 81 Phosphatidylserine, exposed extracellularly, is instrumental in triggering blood clotting and also serves as a signal for the clearance of apoptotic cells. 80
Skin Composition
Lecithin, Lysolecithin, Phosphatidylethanolamine, and Phosphatidylserine
Lecithin, phosphatidylethanolamine, and Phosphatidylserine comprise the major phospholipid components of skin from young adult female albino rabbits. 82 Polyglycerolphosphatides, Lysolecithin, and sphingomyelin are also present.
In a study in which the total lipid concentration, distribution of all major lipid species, and the fatty acid composition in human stratum corneum were assessed, the following lipids were found: phospholipids (phosphatidylethanolamine), cholesterol sulfate, neutral lipids (free sterols, free fatty acids, triglycerides, sterol and wax esters, squalene, and n-alkanes), and sphingolipids. 83 The neutral lipids contributed the greatest proportion to the stratum corneum lipids. Values for the phospholipid composition (lipid weight %) at the following 4 skin sites were: abdomen (4.9 ± 1.6), leg (5.2 ± 1.1), face (3.3 ± 0.3), and plantar (3.2 ± 0.89).
Summary
The safety of the following 17 ingredients in cosmetics is reviewed in this safety assessment: Lecithin, Hydrogenated Lecithin, Lysolecithin, Hydrogenated Lysolecithin, Phospholipids, Hydrolyzed Phospholipids, Phosphatidic Acid, Lysophosphatidic Acid, Phosphatidylglycerol, Lysophosphatidylglycerol, Phosphatidylserine, Ammonium Phosphatidyl Rapeseedate, PC, Hydrogenated PC, Hydrogenated Lysophosphatidylcholine, Lysophosphatidylethanolamine, and Phosphatidylinositol. These ingredients are reported to function primarily as skin and hair conditioning agents, emulsifying agents, and surfactants in cosmetic products. Frequency of use data from FDA and the results of an industry survey indicate that the following ingredients are being used in cosmetic products: Lecithin, Hydrogenated Lecithin, Lysolecithin, Lysophosphatidic Acid, PC, and Phospholipids. Of these ingredients, the highest maximum concentration of use is 50% Lecithin in a leave-on foot product.
The fate of IV-injected 1-[14C]-palmitoyl-[32P]-Lysolecithin was studied using male albino rats. A high percentage of labeled Lysolecithin was detected in skeletal and heart muscle, and it is likely that that Lysolecithin is rapidly converted to Lecithin in the liver. Following IV administration to rats and mice, Phosphatidylserine was eliminated from plasma in a biphasic manner and largely distributed to several major organs, including the liver spleen and brain tissue. In rats, approximately 60% of an orally administered dose of Phosphatidylserine (20 mg/kg body weight) was recovered in the feces of which 50% was identified as lysophosphatidylserine. Approximately 10% of this orally administered dose was detected in the urine. In humans, the oral consumption of soy lecithin Phosphatidylserine capsules (total of 500 mg Phosphatidylserine) resulted in peak plasma Phosphatidylserine levels of 3.95% of the total phospholipid plasma concentration, when compared to background Phosphatidylserine levels of 1.8% to 2.2% of total plasma phospholipids.
The effect of the following phospholipids on the percutaneous penetration of indomethacin was evaluated in vitro using dorsal skin from guinea pigs: PC, phosphatidylethanolamine, Phosphatidylinositol, Phosphatidylserine, Phosphatidylglycerol, Phosphatidic Acid, and sphingomyelin. Phospholipid-induced enhancement of indomethacin percutaneous penetration was in the following order: Phosphatidylglycerol > phosphatidylethanolamine > PC > Phosphatidylserine > Phosphatidic Acid > Phosphatidylinositol > control > sphingomyelin.
In a study in which a purified phospholipid preparation obtained from bovine brain (Phosphatidylserine derived specifically from bovine cerebral cortex, in phosphate buffer suspension) was administered orally to Sprague-Dawley rats, the LD50 was > 5 g/kg body weight.
In a repeated dose inhalation toxicity study involving mice exposed to PC liposomes, no histologic changes of the lungs or untoward effects on general health or survival of animals were noted. In a 2-year feeding study on 4% Lecithin involving rats, no significant differences were observed for mortality, feed consumption, or body weight between the treated and control groups. Additionally, there were no differences in gross or microscopic findings when the groups were compared.
In a 12-week study in which human subjects received soy lecithin-derived Phosphatidylserine daily, there were no clinically significant variations in blood chemistry or hematology. Additionally, there were no differences in the occurrence of side effects between test and placebo groups.
Lecithin 65% (solution of 65% Lecithin) and products containing 2.25% or 3.0% Lecithin 65% were non- to minimally irritating to unrinsed rabbit eyes. In single-insult occlusive patch tests (rabbits), Lecithin 65% was minimally irritating, products containing 3% Lecithin 65% were practically non- to mildly irritating, and a product containing 2.25% Lecithin 65% was nonirritating to the skin of rabbits.
The photocytotoxicity of Lpsm-Flln (0.2% aqueous) was studied using Balb/3T3 fibroblastic cells; results were negative. A foundation containing 0.3% Lecithin 65% (solution of 65% Lecithin) was not a photosensitizer. Lecithin and Hydrogenated Lecithin (both at 15% in petrolatum) were not phototoxic or photosensitizing.
In oral teratogenicity studies on Phosphatidylserine derived specifically from bovine cerebral cortex involving rats and rabbits, there were no treatment-related effects on embryonic and fetal development. Lysophosphatidic acid (30 µM) enhanced the maturation and developmental competence of BDF-1 mouse oocytes in vitro.
Hydrogenated Lecithin was not genotoxic to Salmonella typhimurium or E coli bacterial strains with or without metabolic activation. The results for Phosphatidylserine in mammalian cell assays (ie, mouse lymphoma, DNA repair [HeLa cells], micronucleus assays) were also negative.
TM strain mice were fed 5- to 10-mg Lecithin mixed with sugar, and a second group was fed Lecithin and 4- to 5-mg cholesterol. Brain nerve cell tumors (2-5 mm) were found in 18 of 73 examined animals fed Lecithin and in 27 of 88 examined animals fed Lecithin and cholesterol; brain nerve cell tumors were not found in 188 control animals. In another study, groups of female dd mice were dosed SC with a 0.25% mixture of 4-nitroquinoline1-oxide (in 10% aqueous Lecithin). No surviving mice dosed with Lecithin water or untreated control mice had pulmonary or any other type of neoplasia. However, 3/28 animals of the Lecithin water group and 3/18 control animals had adenomas, which were considered spontaneous.
Membrane lipids, that is, phospholipids, have important roles in signaling reactions. However, these effects are not relevant to the use of phospholipids as cosmetic ingredients.
Discussion
The Panel acknowledged their previous conclusion, published in 2001, that Lecithin and Hydrogenated Lecithin are safe as used in rinse-off products and safe for use in leave-on products at concentrations of ≤ 15% and that the data are insufficient to determine the safety cosmetic products where Lecithin and Hydrogenated Lecithin are likely to be inhaled; and Lecithin and Hydrogenated Lecithin should not be used in cosmetic products in which N-nitroso compounds may be formed. This 15% concentration limit was the highest concentration evaluated in tests for skin irritation, sensitization, phototoxicity, and photosensitization potential in human subjects, all of which were negative. The Panel also noted that the highest maximum use concentration of Lecithin reported in 2014 was 50% in leave-on cosmetic products. The Panel agreed that there is little sensitization potential at this concentration, based on extensive clinical experience indicating no problems associated with the application of Lecithin to the skin. Thus, the Panel determined that the concentrations of Lecithin, Hydrogenated Lecithin, and other phosphoglycerides reviewed in this safety assessment need not be limited to 15% in cosmetic products. This decision, based in part, on clinical experience with Lecithin, is applicable across the phosphoglycerides reviewed in this safety assessment because Lecithin is a complex mixture consisting primarily of PC, phosphatidylethanolamine, Phosphatidylserine, and Phosphatidylinositol, with varying amounts of triglycerides, fatty acids, and carbohydrates from vegetable or animal sources.
The Panel discussed the potential for incidental inhalation exposures to phosphoglycerides in products that are sprayed or in powder form and agreed that, based on the results of the repeated dose inhalation toxicity study, likely airborne particle size distributions and concentrations in the breathing zone and ingredient use, incidental inhalation would not lead to local respiratory effects or systemic effects. The Panel also considered the safe use of Lecithin as an inactive ingredient in FDA-approved aerosolized drug products. Thus, it was agreed that the previous conclusion should be amended, acknowledging that the data are no longer insufficient to determine the safety of cosmetic products where Lecithin and Hydrogenated Lecithin are likely to be inhaled.
Additionally, in the previous safety assessment, concerns about the formation of N-nitroso compounds in cosmetic products containing Lecithin and Hydrogenated Lecithin were based on experimental conditions that do not represent plausible cosmetic use conditions. For example, Lecithin has been reported to be metabolized to choline by bacterial phospholipases in a model system, and the released choline can be dealkylated to dimethylamine, which is N-nitrosatable in the presence of nitrate. The Panel determined that these experimental conditions do not reflect ingredient use in cosmetic products and thus agreed that the previous conclusion should be amended, removing the restriction that Lecithin and Hydrogenated Lecithin should not be used in cosmetic products in which N-nitroso compounds may be formed.
The Panel initially expressed concern about animal tissue as a potential source of phosphoglycerides, particularly bovine brain as a source of Phosphatidylserine and Lysolecithin. However, the Panel determined that these phosphoglycerides are safe as used, noting that ingredients derived from bovine central nervous system tissues are not permitted for use in cosmetic products. Concern about pesticide residues and heavy metals that may be present in botanical ingredients was also expressed. The Panel stressed that the cosmetics industry should continue to use current good manufacturing practices to limit such impurities.
Phosphoglycerides are known to enhance the dermal penetration of some drugs. The Panel noted that formulators should be aware of the potential for enhancing the dermal penetration of other ingredients in cosmetic formulations that contain the ingredients that are evaluated in this safety assessment, especially in products intended for use on infants.
Acknowledging the involvement of cell-membrane lipids in cellular signaling cascades, the Panel noted that these signaling effects are not relevant to the use of phosphoglycerides as cosmetic ingredients. The Panel also acknowledged that derangements in phosphoglyceride metabolism can be associated with prostate, breast, or ovarian cancer but noted that these changes are artifacts of cancer and are not relevant for assessing the safety of cosmetic ingredients. Furthermore, concern over systemic toxicity is mitigated due to, among other reasons, phospholipids are the ubiquitous components of cell membranes and are GRAS for human consumption.
Conclusion
The Panel concluded that the following 17 ingredients are safe in the present practices of use and concentration in cosmetics, as described in this safety assessment. Lecithin Hydrogenated Lecithin Lysolecithin Hydrogenated Lysolecithin* Phospholipids Hydrolyzed Phospholipids* Phosphatidic Acid* Lysophosphatidic Acid Phosphatidylglycerol* Lysophosphatidylglycerol* Phosphatidylserine* Ammonium Phosphatidyl Rapeseedate* PC Hydrogenated PC* Hydrogenated Lysophosphatidylcholine* Lysophosphatidylethanolamine* Phosphatidylinositol*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that those ingredients would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors' Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Johnson, W. contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Heldreth, B. contributed to conception and design; acquisition, analysis, and interpretation; and critically revised manuscript. Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to conception and design, analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.