Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 60 PEGylated alkyl glycerides. PEGylated alkyl glycerides are mono-, di-, and/or triglycerides that have been modified with ethylene glycol repeat units (in the starting material form as epoxide). Most of the PEGylated alkyl glycerides are reported to function as skin-conditioning agents or surfactants. The Panel reviewed the available animal and clinical data as well as data from the 1999 report for the 5 polyethylene glycol (PEG) glyceryl cocoates and the 2012 report of PEGylated oils, to determine the safety of these ingredients. The Panel concluded these ingredients are safe in the current practices of use and concentration when formulated to be nonirritating; this conclusion supersedes the 1999 conclusion issued on 5 PEG glyceryl cocoate ingredients.
Introduction
This report assesses the safety of the following 60 PEGylated alkyl glycerides as used in cosmetic formulations: Polyethylene glycol (PEG)-6 Almond Glycerides PEG-20 Almond Glycerides PEG-35 Almond Glycerides PEG-60 Almond Glycerides PEG-192 Apricot Kernel Glycerides PEG-11 Avocado Glycerides PEG-14 Avocado Glycerides PEG-11 Babassu Glycerides PEG-42 Babassu Glycerides PEG-4 Caprylic/Capric Glycerides PEG-6 Caprylic/Capric Glycerides PEG-7 Caprylic/Capric Glycerides PEG-8 Caprylic/Capric Glycerides PEG-11 Cocoa Butter Glycerides PEG-75 Cocoa Butter Glycerides PEG-7 Cocoglycerides PEG-9 Cocoglycerides PEG-20 Corn Glycerides PEG-60 Corn Glycerides PEG-20 Evening Primrose Glycerides PEG-60 Evening Primrose Glycerides PEG-3 Glyceryl Cocoate PEG-7 Glyceryl Cocoate* PEG-30 Glyceryl Cocoate* PEG-40 Glyceryl Cocoate* PEG-78 Glyceryl Cocoate* PEG-80 Glyceryl Cocoate* PEG-5 Hydrogenated Corn Glycerides PEG-8 Hydrogenated Fish Glycerides PEG-20 Hydrogenated Palm Glycerides PEG-6 Hydrogenated Palm/Palm Kernel Glyceride PEG-16 Macadamia Glycerides PEG-70 Mango Glycerides PEG-13 Mink Glycerides PEG-25 Moringa Glycerides PEG-42 Mushroom Glycerides PEG-2 Olive Glycerides PEG-6 Olive Glycerides PEG-7 Olive Glycerides PEG-10 Olive Glycerides PEG-40 Olive Glycerides PEG-18 Palm Glycerides PEG-12 Palm Kernel Glycerides PEG-45 Palm Kernel Glycerides PEG-60 Passiflora Edulis Seed Glycerides PEG-60 Passiflora Incarnata Seed Glycerides PEG-45 Safflower Glycerides PEG-60 Shea Butter Glycerides PEG-75 Shea Butter Glycerides PEG-75 Shorea Butter Glycerides PEG-35 Soy Glycerides PEG-75 Soy Glycerides PEG-2 Sunflower Glycerides PEG-7 Sunflower Glycerides PEG-10 Sunflower Glycerides PEG-13 Sunflower Glycerides PEG-5 Tsubakiate Glycerides PEG-10 Tsubakiate Glycerides PEG-20 Tsubakiate Glycerides PEG-60 Tsubakiate Glycerides *originally reviewed in 1999
PEGylated alkyl glycerides are mono-, di-, and/or triglycerides that have been modified with ethylene glycol repeat units (in the starting material form as epoxide). The number of units (eg, “40” in PEG-40 Glyceryl Cocoate) is representative of an average number of ethylene glycol repeat units (ie, PEG-40 Glyceryl Cocoate contains some PEG-39 and PEG-41 glyceryl cocoates, in addition to PEG-40). Most of the PEGylated alkyl glycerides are reported to function as skin-conditioning agents or surfactants 1 (Table 1). As above, ingredient names follow the International Nomenclature Committee conventions for cosmetic labeling (ie, capitalization of the first letter of each word, and no italicization); such nomenclature conventions are utilized throughout this report when denoting specific cosmetic ingredients.
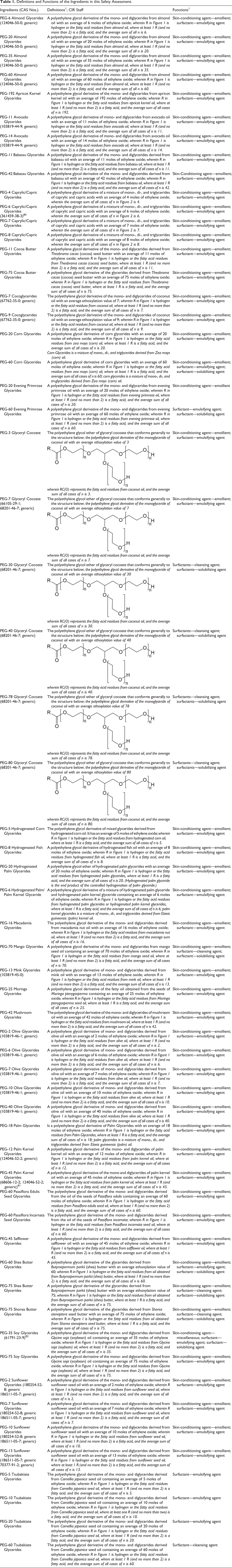
Definitions and Functions of the Ingredients in this Safety Assessment.
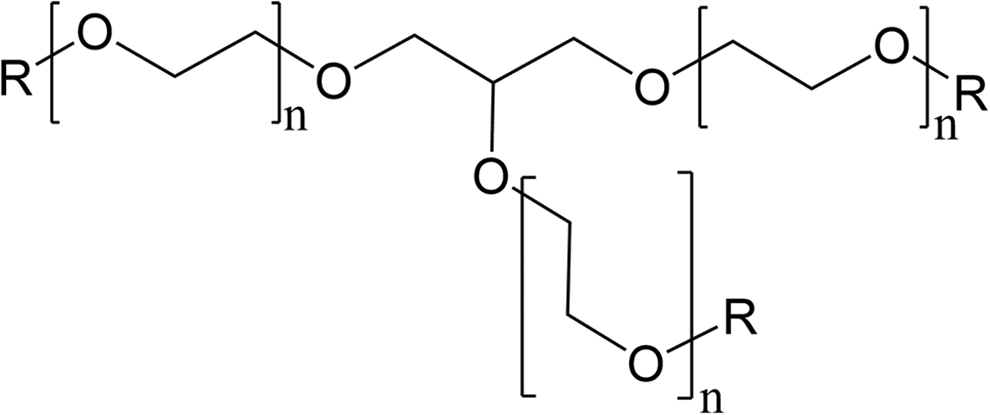
Generic PEGylated alkyl glyceride structure, wherein R is hydrogen or the fatty acids declared in the name or found in the source material, and the sum of all cases of n is “X” in PEG-”X.”
Five PEG glyceryl cocoate ingredients, indicated above, have been reviewed previously, and in 1999, the Expert Panel for Cosmetic Ingredient Safety (Panel) concluded those ingredients were safe as used in rinse-off products and safe at up to 10% in leave-on products. According to the Discussion in that report, the basis of the concentration limit for leave-on products was derived from the ocular and dermal sensitization data. 2 At the September 2014 meeting, the Panel determined that it is appropriate to include those ingredients in this report, and where appropriate, information from the 1999 report is included in this safety assessment.
In 2012, the Panel reviewed a very similar family of ingredients, the PEGylated oils, and concluded the PEGylated oils are safe as used in cosmetics when formulated to be nonirritating. 3 PEGylated oils are complex mixtures of the etherification and transesterification products of fatty acid glycerides and fatty acids derived from identified plant sources with the necessary equivalents of ethylene oxide to achieve the desired polyethylene glycol (PEG) length. The only difference between the PEGylated oils and the PEGylated alkyl glycerides is that prior to the PEGylation step in the production of PEGylated alkyl glycerides, a further refinement of the oil has been performed to retain only the glycerides and in some cases refined (or partially de-esterified) to only the mono-, di-, or triglycerides. In other words, PEGylated alkyl glycerides are a subfraction of PEGylated oils. PEGylated oils mostly contain PEGylated triglycerides and some PEGylated fatty acids (ie, not attached to a molecule of glycerin), while PEGylated alkyl glycerides contain only specific PEGylated glycerides and no (or virtually no) PEGylated fatty acids.
As an example, the PEGylated oil, almond oil PEG-6 esters, consists mostly of triglycerides, wherein the 6 stoichiometric equivalents of ethylene oxide have been inserted between the glycerin core and the attached almond fatty acids. Free almond fatty acids that have been PEGylated to some degree may also be present. In contrast, the PEGylated alkyl glyceride, PEG-6 Almond Glycerides, consists mostly of mono- and diglycerides, wherein 6 stoichiometric equivalents of ethylene oxide have (1) been inserted between the glycerin core and the attached almond fatty acids, (2) etherified the free alcohol(s) of the glyceride, or (3) both. No appreciable free almond fatty acids should be present.
The materials that are used in the manufacture of the PEGylated alkyl glycerides, that is, ethylene oxide (and any incidentally generated free PEG chains) and the applicable glycerides (and the oils from which they are derived), may be present as impurities in the final ingredient. The Cosmetic Ingredient Review (CIR) has reviewed the safety of the majority of the component ingredients, and although the information from those safety assessments may not be relevant for read-across, the reports have some relevance because starting materials may be present as residual impurities. Therefore, summary information and the conclusion of each relevant report are provided (Table 2). 4 -11 Previous Panel reports are available on all component oils except hydrogenated corn oil (although a report on corn oil was available), hydrogenated fish oil, mushroom oil, and Passiflora incarnata oil. Additionally, the conclusion and a data summary from the PEGylated oils 12 report are provided (Table 2).
Summaries of Previous CIR Safety Assessments.

Chemistry
Definition and Structure
PEGylated alkyl glycerides are mono-, di-, and/or triglycerides that have been modified with ethylene glycol repeat units (in the starting material form as epoxide).
Just as oils and other PEGylated materials are mixtures, the PEGylated alkyl glycerides are mixtures. PEGylation of glycerides occurs not only as an etherification of the free alcohol groups of the glycerides with ethylene oxide groups but also as a transesterification which results in net insertion of PEG groups between the glyceryl and fatty acid components of the glyceride. 13,14 For example, the glycerin core of PEG-6 Caprylic/Capric Glycerides is ethoxylated (Figure 2), wherein the sum of all n is equal to the number of ethylene oxide repeat units and need not be the same at all places of the molecule.
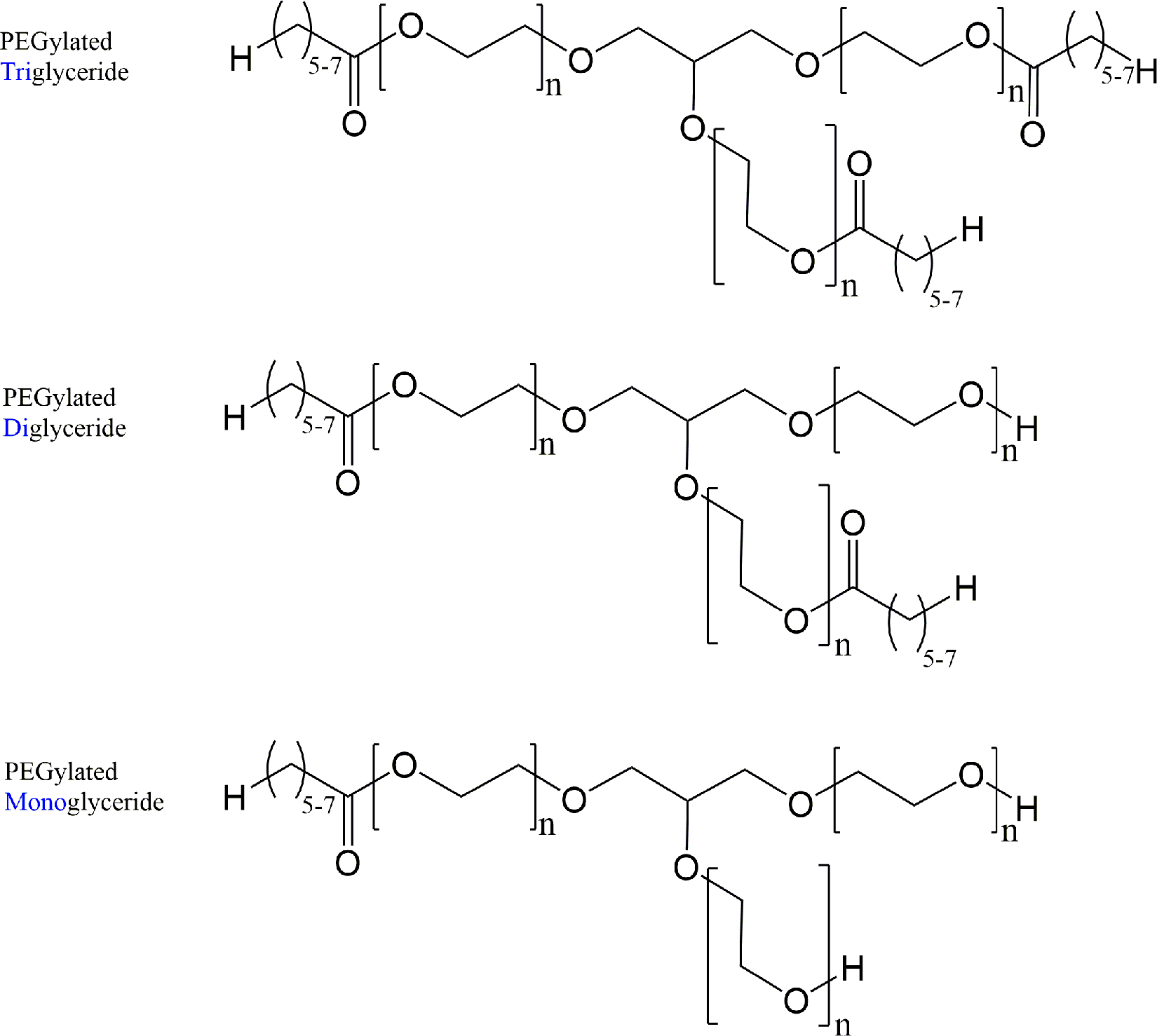
PEG-6 Caprylic/Capric Glycerides, wherein the sum of all instances of “n” is representative of an average number of ethylene glycol repeat units (ie, “PEG-6”).
It should be stated that n is not equal to X. In other words, wherein the value of X in PEG-X is equal to 6 (eg, PEG-6 Caprylic/Capric Glycerides), n is not equal to 6. Instead, X represents the number of stoichiometric equivalents of ethylene oxide that were added to one stoichiometric equivalent of caprylic/capric glycerides. Therefore, the sum of all of the different n values in the mixture may be no more than X. Indeed, when one mole of ethylene oxide is reacted with one mole of fatty alcohol, adducts having no added ethylene oxide are the predominate material in the mixture. 13 Furthermore, wherein ethylene oxide reacts with mono- or diglyceride, it is approximately twice as likely that it will react at an ester site versus an alcohol site. Moreover, a percentage (13% in one specific case) of the ethylene oxide simply reacts with other molecules of ethylene oxide, resulting in some polyethylene glycols, unattached to glycerides.
Most of the ingredients in this report share in common a specific nomenclature, generically PEG-X “source”/alkyl-chain-common-name glycerides (eg, PEG-6 Caprylic/Capric Glycerides). The PEG glyceryl cocoates have a slightly different order of nomenclature, but they are not structurally distinct from the other ingredients in this report.
Chemical Properties
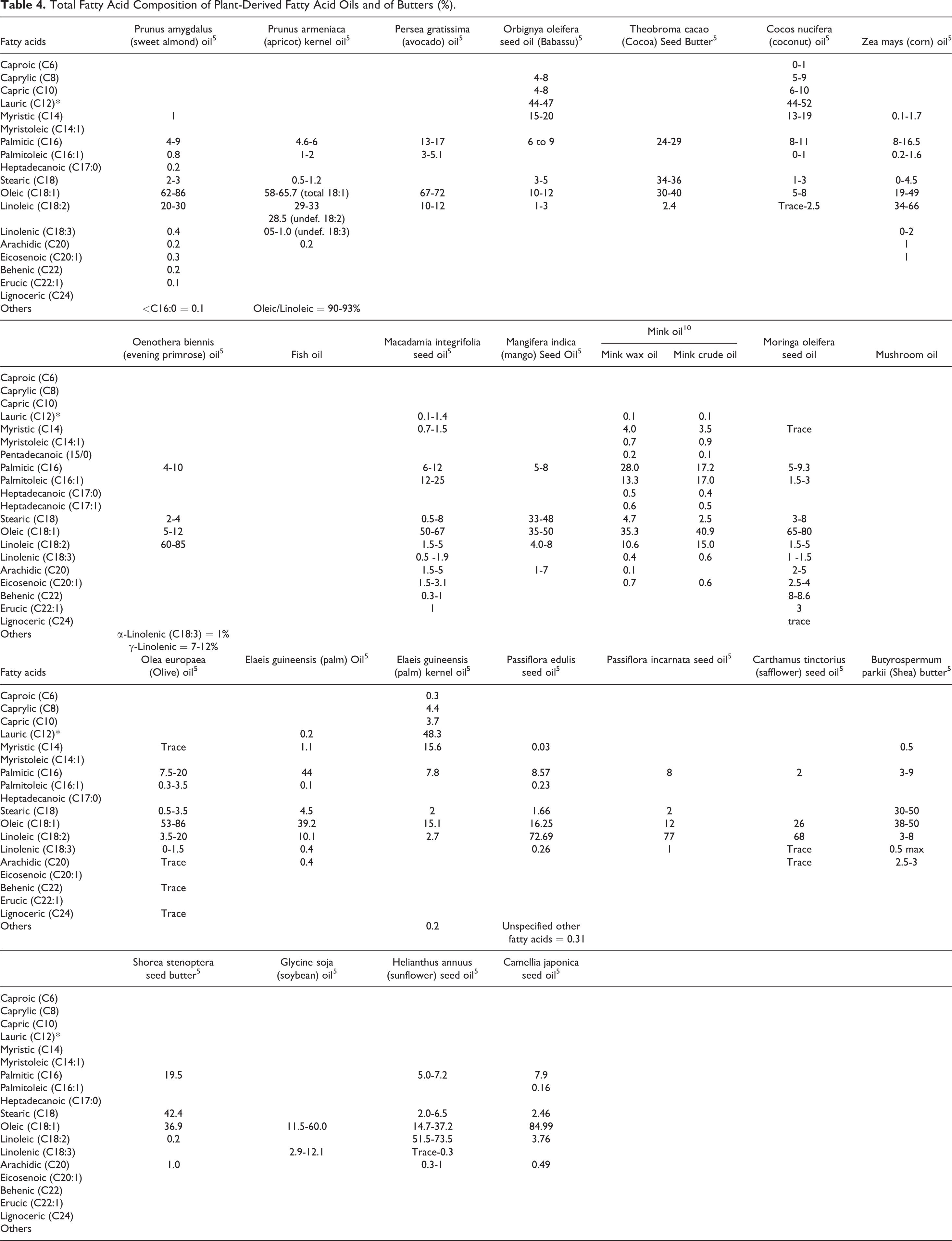
Much like the oils these ingredients are derived from, the PEGylated alkyl glycerides range from oily liquids to waxy solids, depending on alkyl chain length and degree of PEGylation (Table 3). Owing to the combination of polar and nonpolar functional groups, most of these ingredients are soluble in some organic solvents and are at least dispersible in water, if not soluble. The fatty acid compositions of the oils that comprise the alkylated glycerides are provided in Table 4.
Chemical Properties.

Total Fatty Acid Composition of Plant-Derived Fatty Acid Oils and of Butters (%).
Methods of Manufacture
PEG-11 Avocado Glycerides, 15 PEG-9 Cocoglycerides, 16 and PEG-10 Olive Glycerides 17 are produced by the reaction of their respective oil with ethylene oxide. The PEG-6 Caprylic/Capric Glycerides is derived completely from a vegetable/synthetic source, and palm kernel oil and/or rapeseed oil are the starting materials. 18
One source states that PEG-6 Caprylic/Capric Glycerides is manufactured in a 2-step procedure starting with the preparation of a mono/diglyceride of a C8/C10 fatty acid blend; the C8:C10 ratio is approximately 60:40. 19 The raw materials are charged with an excess of glycerin, the blend is purged with nitrogen and heated under pressure, and the mixture is then cooled, treated with Fuller earth, and filtered. Following filtration, step 2 is initiated and includes the addition of an alkaline catalyst in a solvent, heating, and the removal of the solvent under pressure. The pressure in the reactor is then normalized with nitrogen, and ethylene oxide is added until the desired saponification value is reached; the mixture is then stirred until the ethylene oxide value is ≤ 1 ppm. After cooling, the product is neutralized with lactic or acetic acid. Finally, the raw ethoxylate is treated with steam to remove the volatile impurities, and if necessary, the product may be bleached with Fuller earth.
PEG-8 Caprylic/Capric Glycerides can be obtained by partial transesterification of medium-chain triglycerides using PEG, by esterification of glycerol and PEG with caprylic acid and capric acid, or as a mixture of glycerol esters and condensates of ethylene oxide with caprylic acid and capric acid. 20,21 Methods of manufacture of the other PEGylated alkyl glycerides were not found in the published literature, nor were unpublished data provided.
Composition and Impurities
Residual PEG may be present in the PEGylated alkyl glycerides. The PEGs are the condensation products of ethylene oxide and water, with the chain length controlled by number of moles of ethylene oxide that are polymerized. The PEGs may contain trace amounts of 1,4-dioxane, a by-product of ethoxylation; 1,4 dioxane is a known animal carcinogen. 22 The US Food and Drug Administration (FDA) has been periodically monitoring the levels of 1,4-dioxane in cosmetic products, and the cosmetic industry reported that it is aware that 1,4-dioxane may be an impurity in PEG-containing ingredients and, thus, uses additional purification steps to limit it in the ingredient before blending into cosmetic formulations. 23,24
Raw material information sheets for PEG-11 Avocado Glycerides, 15 PEG-9 Cocoglycerides, 16 and PEG-10 Olive Glycerides 17 state that these compounds contain < 5 ppm 1,4-dioxane, < 1 ppm ethylene oxide, and < 1 ppm residual monomers. Solvents are not added to these materials. The PEG-6 Caprylic/Capric Glycerides contains ≤ 5 ppm dioxane and ≤ 1 ppm ethylene oxide, and it meets the valid regulatory requirements for limits on agricultural pesticides. 18,25 Residual solvents, monomers, free amines, and nitrosamines are not by-products of the production process.
Nuclear magnetic resonance spectroscopy indicated PEG-8 Caprylic/Capric Glycerides consists of 30% mono-, di-, and triglycerides of C8 and C10 fatty acids, 50% of mono- and diesters of PEG-8 (ie, esters of capric or caprylic acid with PEG 8; not glycerides), and 20% free PEG-8. 26 It is reported to contain ≤ 10 ppm 1,4-dioxane, < 1 ppm ethylene oxide, and < 10 ppm heavy metals (lead). 27,28
Use
Cosmetic
Most of the PEGylated alkyl glycerides are reported to function as a skin conditioning agent—emollient and as a surfactant—emulsifying agent (Table 1). 1 Emollients function by their ability to remain on the skin surface or in the stratum corneum to act as lubricants, to reduce flaking, and to improve the skin’s appearance. For the surfactant—emulsifying agent function, the efficacy of an emulsifying agent depends on its ability to reduce surface tension, to form complex films on the surface of emulsified droplets, and to create a repulsive barrier on emulsified droplets to prevent their coalescence. A few of the PEGylated alkyl glycerides also are reported to act as surfactant—solubilizing agents or surfactant—cleansing agents.
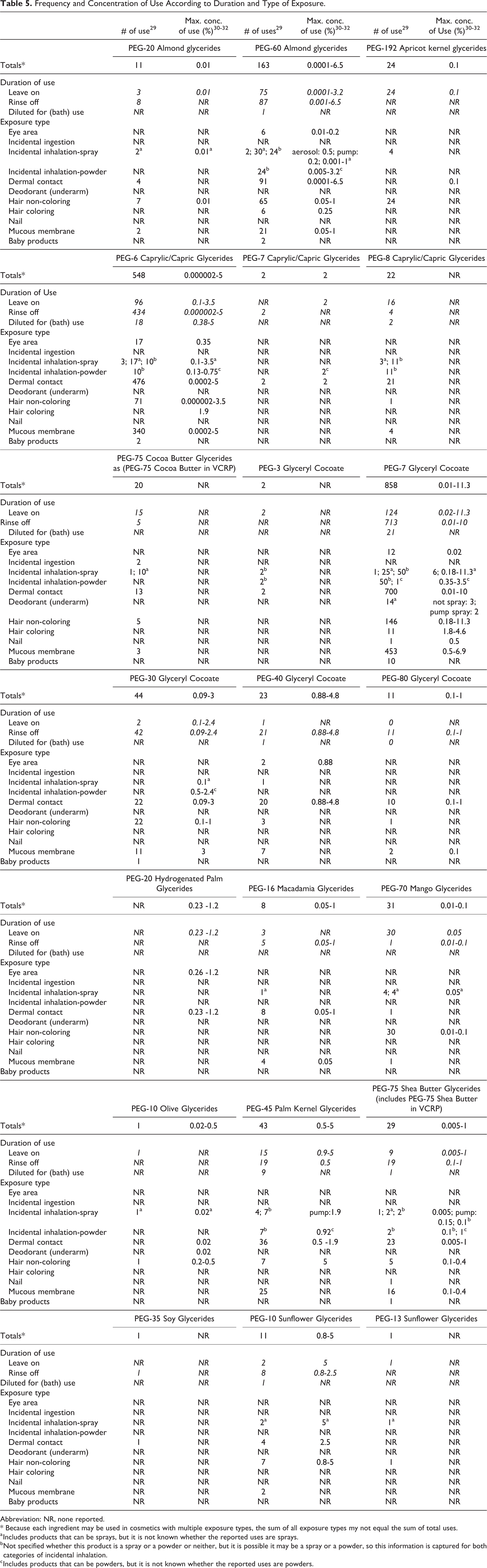
The FDA collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). In 2014, VCRP data obtained from the FDA 29 and data received in response to surveys of the maximum reported use concentration by category that were conducted by the Personal Care Products Council (Council) 30 -32 indicate that 21 of the 60 ingredients included in this safety assessment are used in cosmetic formulations (Table 5). (The ingredients not currently reported to be used are listed in Table 6.)
Frequency and Concentration of Use According to Duration and Type of Exposure.
Abbreviation: NR, none reported.
* Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types my not equal the sum of total uses.
a Includes products that can be sprays, but it is not known whether the reported uses are sprays.
b Not specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation.
c Includes products that can be powders, but it is not known whether the reported uses are powders.
Ingredients Not Reported to be Used.

According to 2014 VCRP data, PEG-7 Glyceryl Cocoate has the most reported uses, that is, 858; the majority of these uses (713) are in rinse-off formulations. 29 PEG-6 Caprylic/Capric Glycerides is reported to be used in 548 formulations, the majority of which are rinse-off formulations, and PEG-60 Almond Glycerides is reported to be used in 163 cosmetic formulations. All other in-use PEGylated alkyl glycerides are reported to be used in less than 45 formulations. The PEG Glyceryl Cocoates have increased in use. In 1996, PEG-7 Glyceryl Cocoate was reported to be used in 173 formulations, and it is now reported to be used in 858 formulations; however, both then and now, PEG-7 Glyceryl Cocoate is used mostly in rinse-off formulations. 2,29 The frequency of use also has increased for PEG-30 Glyceryl Cocoate (from 10 to 44 uses), PEG 40 Glyceryl Cocoate (from 5 to 23 uses), and PEG-80 Glyceryl Cocoate (from 2 to 11 uses).
The results of the concentration of use survey conducted by the Council indicate the highest leave-on concentrations reported are 11.3% and 6% for PEG-7 Glyceryl Cocoate in tonics, dressings, and other hair grooming aids and “other” fragrance preparations. 30 PEG-7 Glyceryl Cocoate also has the highest rinse-off concentration of use reported, that is, 10% in skin cleansing products.
At the time of the original safety assessment, concentration of use data were not available from the FDA. However, based on ocular and sensitization data, a concentration limit of 10% was placed on leave-on products containing PEG glyceryl cocoates. The current reported concentration of use in leave-on products is slightly higher, that is, 11.3% in tonics, dressings, and other hair grooming aids.
One ingredient is used in products that could be incidentally ingested (ie, PEG-75 Cocoa Butter Glycerides is reported to be used in 2 lipstick formulations) and a few of the ingredients are used near the eye (eg, PEG-20 Hydrogenated Palm Glycerides is used at 1.2% in an eyebrow pencil) or mucous membranes (PEG-7 Glyceryl Cocoate is used at up to 6.9% in other personal cleanliness products). Five PEGylated alkyl glycerides were reported to the VCRP as used in baby products, but concentration of use data were not submitted by industry for these uses. Additionally, according to the VCRP, several ingredients are used in products that can be incidentally inhaled, and results of the Council survey reported concentrations of up to 6% PEG-7 Glyceryl Cocoate in “other” fragrance preparations, 2% PEG-7 Glyceryl Cocoate in a pump spray deodorant, and up to 1.9% PEG-45 Palm Kernel Glycerides in a pump spray suntan product. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm. 33,34 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 35,36 The PEGylated alkyl glycerides named in this safety assessment are listed in the European Union inventory of cosmetic ingredients. 37
Non-Cosmetic
PEG-8 Caprylic/Capric Glycerides has use as a pharmaceutical excipient. 38 It has a maximum potency of 70 mg in oral soft gelatin capsules and 6.12% in oral solutions. 39 A mixture of mono-, di-, and triglycerides and PEG mono- and diesters of fatty acids (polyglycides) of hydrogenated vegetable oils may be safely used as an excipient in dietary supplement tablets, capsules, and liquid formulations that are intended for ingestion in daily quantities measured in drops or similar small units of measure when the total ester content is > 90%, the acid value is < 2, the hydroxyl value is < 56, the lead content is < 0.1 mg/kg, and < 10 mg/kg 1,4-dioxane and < 1 mg/kg ethylene oxide is present (21CFR172.736).
Fats, oils, fatty acids, and fatty alcohols derived from coconut, palm, and soybean oils and reacted with 400-3,000 Da PEG are approved for use as indirect food additives in polymers for use as components of articles intended for repeated use in textiles and textile fibers (21CFR177.2800). Also, the fatty acids and alcohols derived from coconut, corn, palm, and soybean oils reacted with 200-4,600 Da PEG, with or without dehydration, to form an ester are approved for use as an indirect food additive in defoaming agents used in the manufacture of paper and paperboard components (21CFR176.210).
Toxicokinetics
Penetration Enhancement
Surfactants can enhance penetration of some compounds through the skin. The ability of PEG-8 Caprylic/Capric Glycerides to be a dermal penetration enhancer has been investigated in a number of studies; a few examples of those studies are provided. In one study in which transdermal delivery of lidocaine (lipophilic) and prilocaine hydrochloride (hydrophilic) was measured in vitro in Franz-type diffusion cells, several microemulsion systems that contained 11.5% to 53% PEG-8 Caprylic/Capric Glycerides, 3% to 70% isostearyl isostearate, 8% to 35% polyglyceryl-6 isostearate, and 7% to 65% water increased delivery of lidocaine up to 4 times compared to an oil/water emulsion vehicle, and increased the delivery of prilocaine hydrochloride almost 10 times compared to a hydrogel. 26 The increase in delivery was attributed to increased solubility of the drug and appeared to be dependent on drug mobility in the vehicle.
Another example is the effect of PEG-8 Caprylic/Capric Glycerides on the transdermal flux of carvedilol, a nonselective β adrenergic antagonist, through porcine skin. The flux of carvedilol from a PEG-8 Caprylic/Capric Glycerides vehicle (ie, 14.28 µg/cm2/h) was 8.6 times greater than that obtained with the control vehicle (ie, 1.66 µg/cm2/h) composed of phosphate buffered saline containing 40% vol/vol PEG 400. 40 However, when placed in the donor compartment, flux was only 4.7 times greater with 5% PEG-8 Caprylic/Capric Glycerides compared to controls. In another study, when used as a vehicle, PEG-8 Caprylic/Capric Glycerides was only a weak penetration enhancer of cyclosporin A, a nonpolar cyclic oligopeptide, through excised rat skin. 41
A formulation containing 40% PEG-8 Caprylic/Capric Glycerides, 39.75% ethoxydiglycol, 10% ethanol, 5% ethyl oleate, and 5% sorbitan oleate improved the skin delivery of kahalalide F, a lipophilic cyclic molecule with a high molecular weight; however, the amount of kahalalide F that penetrated and permeated through the skin was much greater with dimethyl sulfoxide. 41 Additionally, a formulation containing 39.75% PEG-8 Caprylic/Capric Glycerides, 35% propylene glycol, 10% ethanol, 10% n-methyl-2-pyrrolidone, and 5% isopropyl myristate did not improve skin delivery of kahalalide F.
Toxicological Studies
Single Dose (Acute) Toxicity
Oral
PEG-6 Caprylic/Capric Glycerides has an oral LD50 of > 5 g/kg in rats; 5 males and 5 females were given a single dose of 5.5 g/kg, and none of the animals died. 42 PEG-8 Caprylic/Capric Glycerides has an oral LD50 of 22 g/kg in rats. 43 The oral LD50 of PEG-7 Glyceryl Cocoate in rats was > 19.9 mg/g, the highest dose administered. 2
Repeated Dose Toxicity
Oral
PEG-8 Caprylic/Capric Glycerides
The oral toxicity of a blend of 3 solvents was evaluated in Wistar rats; the blend consisted of 40% PEG-8 Caprylic/Capric Glycerides, 40% apricot kernel oil PEG-6 esters, and 20% ethoxydiglycol. 44 (In the published paper, the blend was described using trade names; trade name and other information suggests that the second portion of the blend is apricot kernel oil PEG-6 esters.) Groups of 10 male and 10 female rats were given by gavage a volume of 5, 10, or 20 mL/kg/d test article for 4 weeks. A negative control group of 10 rats/sex received 20 mL/kg/d 1% (wt/vol) hydroxyethyl cellulose in purified water. The animals were examined daily for signs of toxicity, and hematology, clinical chemistry, and urinalysis parameters were measured at study termination; at necropsy, organs were examined grossly and microscopically, and hepatic cytochrome P450 (CYP450) content and related activities were evaluated.
The test article was relatively well tolerated at 5 mL/kg/d, and this volume was considered the no-observed adverse effect level (NOAEL). Adverse effects were observed with the greater doses, and the main effects were observed in the liver, kidneys, and adrenal glands. One female of the 20 mL/kg/d group died on day 28; the death was considered test article–related because the animal had worsening clinical condition prior to death and necropsy results included enlarged and pale kidneys, a dilated stomach with multiple dark red areas on the glandular mucosa, a dilated caecum, and several whitish areas on the liver. In the surviving 20 mL/kg/d animals, there was an increase in clinical signs of toxicity, including ptyaliasm, partial blepharoptosis, and piloerection, and a statistically significant decrease in feed consumption and water intake in males and females and in body weights in males, when compared to control animals. At study termination, most of the high-dose animals had dilated caecums containing soft brownish content, and 12 of the animals had pale adrenal glands; 5 of the 10 males and 3 of the 9 surviving females had enlarged kidneys. Absolute and relative organ-to-body weights were also statistically significantly affected; increases were reported in absolute and relative kidney weights in males and females, relative liver weights in males, and absolute and relative liver weights in females, and decreases were reported in absolute and relative spleen weights in males and females, absolute thymus weights in males, and absolute and relative thymus weights in females. Microscopic findings at this dose included mild centrilobular hepatocellular hypertrophy, mostly mild bilateral diffuse tubular dilation and bilateral multifocal tubular epithelium degeneration and necrosis in the kidney, and vacuolation of adrenal gland cortex. Changes in some hematological parameters were also reported. A volume-related statistically significant increase in induction of hepatic CYP450 was reported. Some similar effects were reported in animals given 10 mL/kg/d but not at the same rate of incidence. No adverse effects were observed in dogs that were dosed orally for 13 weeks with 1.0 g/kg/d PEG-8 Caprylic/Capric Glycerides. 43 Further details were not provided.
PEG-7 Glyceryl Cocoate
Groups of 10 male rats were fed a diet containing 0%, 2.5%, 5.0%, or 7.5% of a formulation containing 0.8% PEG-7 Glyceryl Cocoate for 28 days. 45 All animals survived until study termination. For all groups, select tissues were weighed at necropsy. Select tissues of animals in the control and high-dose group were examined microscopically. Spleen weights were significantly decreased in high-dose animals; although there were no associated microscopic changes, the researchers stated the change could be associated with dosing. Soft feces, a distended cecum, and enlarged mesenteric lymph nodes were observed at necropsy in the mid- and high-dose animals. The no-observable effect level (NOEL) was 2.5% of the formulation containing 0.8% PEG-7 Glyceryl Cocoate.
Reproductive and Developmental Toxicity
A segment II developmental toxicity test was performed in rats with PEG-8 Caprylic/Capric Glycerides. 46 The animals were dosed with 0, 1,000, 2,000, or 3,000 mg/kg/d by gavage on days 6 through 17 of gestation. The maternal NOAEL was 2,000 mg/kg/d; effects on body weigh were reported at 3,000 mg/kg/d. The embryo/fetal NOAEL was 3,000 mg/kg/d. No signs of embryotoxicity, fetotoxicity, or teratogenicity were noted at any dose level. No other details were provided.
Genotoxicity
PEG-6 Caprylic/Capric Glycerides, PEG-7 Glyceryl Cocoate, and PEG-10 Olive Glycerides were not mutagenic, with or without metabolic activation. Salmonella typhimurium strains TA98, TA100, TA1535, TA1537, and TA102 were exposed to concentrations of 50-5,000 µg/plate PEG-6 Caprylic/Capric Glycerides in one study, 42 and Salmonella typhimurium strains TA98, TA100, TA1535, TA1537, and TA1538 were exposed to 0.04% PEG-6 Caprylic/Capric Glycerides in 0.9% saline in another. 47 Salmonella typhimurium strains TA98, TA100, TA1535, TA1537, and TA1538 also were exposed to 0.625% PEG-7 Glyceryl Cocoate in 0.9% saline. 48 Details of testing with PEG-10 Olive Glycerides, including strains used and concentrations tested, were not provided. 17 Genotoxicity studies of other PEGylated alkyl glycerides were not found in the published literature nor were additional unpublished data provided.
Carcinogenicity
Carcinogenicity data on PEGylated alkyl glycerides were not found in the published literature nor were unpublished data provided.
Irritation and Sensitization
Dermal
The irritation and sensitization potentials of several PEGylated alkyl glycerides were evaluated in alternative, 26,49 -55 nonhuman, 2,17,42,56 -62 and human studies, 2,17,42,63 -72 and these ingredients generally were not irritants or sensitizers in the alternative and human studies (Table 7). Mixed results were observed in the nonhuman studies; specifically, undiluted PEG-20 Almond Glycerides, 61 PEG-60 Almond Glycerides, 60 PEG-12 Palm Kernel Glycerides, 59 and PEG-45 Palm Kernel Glycerides 58 were irritating to rabbit skin in primary skin irritation studies, and PEG-7 Glyceryl Cocoate (concentration not specified) was mildly irritating to rabbit skin. 2 The only reaction reported in clinical testing was PEG-10 Olive Glycerides (2% active matter) was nonirritating to mildly irritating. 17 In other clinical studies, up to 20% PEG-60 Almond Glycerides, 64,69 undiluted PEG-6 Caprylic/Capric Glycerides, 42,63,66 undiluted PEG-7 Glyceryl Cocoate, 2,65,70 -72 and a formulation containing 1% PEG-75 Shea Butter Glycerides 67,68 were not irritants or sensitizers.
Dermal Irritation and Sensitization.
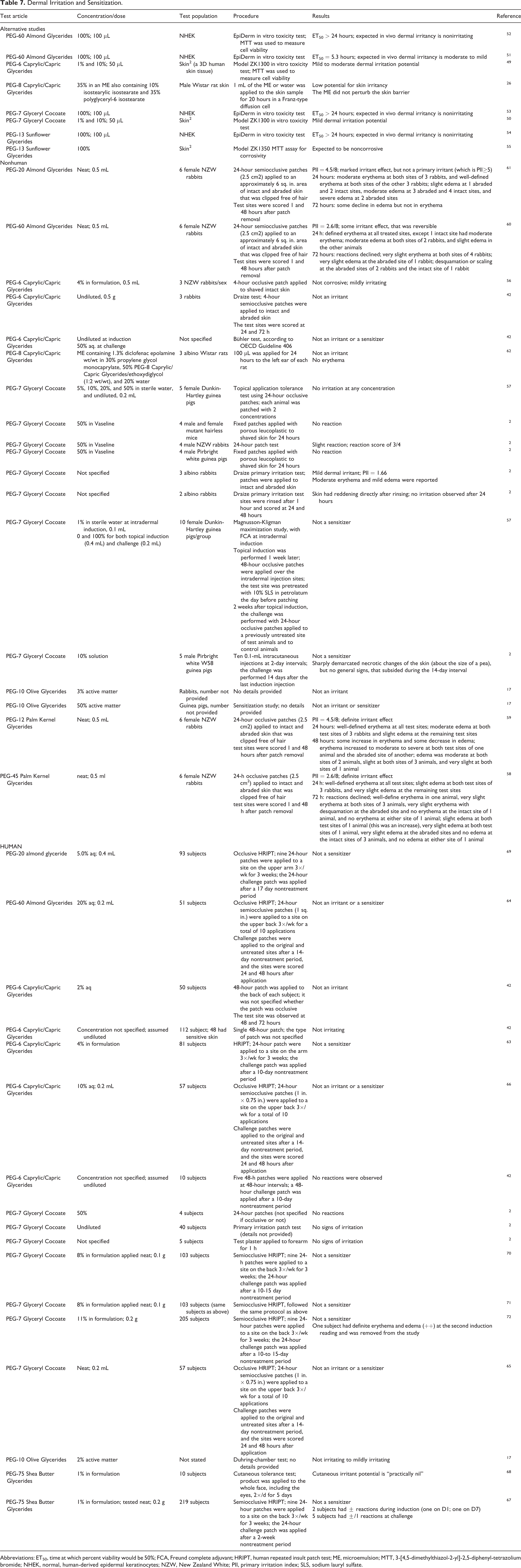
Abbreviations: ET50, time at which percent viability would be 50%; FCA, Freund complete adjuvant; HRIPT, human repeated insult patch test; ME, microemulsion; MTT, 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl-tetrazolium bromide; NHEK, normal, human-derived epidermal keratinocytes; NZW, New Zealand White; PII, primary irritation index; SLS, sodium lauryl sulfate.
Phototoxicity
PEG-7 Glyceryl Cocoate was not phototoxic in mice at a concentration of 50%. 2 Formulations containing 0.3% PEG-7 Glyceryl Cocoate were not photosensitizers in clinical testing.
Ocular
In a hen’s egg test utilizing the chorioallantoic membrane (HET-CAM), a formulation containing 11% PEG-7 Glyceryl Cocoate had practically no irritation potential, 73 and concentrations of 10, 50, and 100% PEG-7 Glyceryl Cocoate were slightly irritating; 74 in a neutral red uptake (NRU) assay, the ocular irritation potential of PEG-7 Glyceryl Cocoate was not classified 75 (Table 8). The PEG-10 sunflower glyceride was a nonirritant in a chorioallantoic membrane vascular assay (CAMVA). 76 In rabbits, 10% aq. The PEG-7 Glyceryl Cocoate 2 and up to 3% PEG-8 Caprylic/Capric Glycerides were not ocular irritants, 77 and 5% PEG-8 Caprylic/Capric Glycerides was a slight irritant to rabbit eyes. 77 Undiluted PEG-6 Caprylic/Capric Glycerides was a strong ocular irritant; however, aq. solutions of up to 5% 42 and formulations containing 4% PEG-6 Caprylic/Capric Glycerides 78 were not ocular irritants in rabbits. In clinical testing, a formulation containing 1% PEG-75 Shea Butter Glycerides did not induce ocular irritation in a tolerance test. 68
Ocular Irritation Studies.

Abbreviations: CAMVA, chorioallantoic membrane vascular assay; HET-CAM, hen’s egg test utilizing the chorioallantoic membrane; NRU, neutral red uptake; NZW, New Zealand White; OECD, Organisation for Economic Co-operation and Development; RC50, theoretical concentration producing a positive reaction in 50% of treated eggs.
Mucous Membrane
PEG-10 Olive Glycerides, 3% active matter, did not irritate the mucous membrane. 17 (No details were provided.) A female hygiene product containing 0.1% PEG-75 Shea Butter Glycerides did not induce any evidence of vaginovulvar irritation in a use study. 79 Twenty-one female subjects bathed in a bubble bath using a “quarter-size amount” of the test material for 10 to 20 minute daily for 1 week.
Summary
This report addresses the safety of 60 PEGylated alkyl glycerides as used in cosmetics. PEGylated alkyl glycerides are mono-, di-, and/or triglycerides that have been modified with ethylene glycol repeat units (in the starting material form as epoxide). Most of the PEGylated alkyl glycerides are reported to function as skin conditioning agents or surfactants.
VCRP data obtained from the FDA, and data received in response to surveys of the maximum reported use concentration by category that were conducted by the Council, indicate that 21 of the 60 ingredients included in this safety assessment are used in cosmetic formulations. The PEG-7 Glyceryl Cocoate has the most reported uses, that is, 858, followed by PEG-6 Caprylic/Capric Glycerides with 548 reported uses; the majority of uses for both of these ingredients are in rinse-off formulations. The highest concentrations of use reported for products resulting in leave-on dermal exposure are 11.3% and 6% PEG-7 Glyceryl Cocoate in tonics, dressings, and other hair grooming aids and other fragrance preparations. The PEG-7 Glyceryl Cocoate also has the highest rinse-off concentration of use reported, tha is, 10% in skin cleansing products.
Since the original safety assessment was published, the frequency of use of PEG-7 Glyceryl Cocoate increased from 173 reported uses in 1996 to 858 reported uses in 2014. At the time of the original safety assessment, concentration of use data were not available from the FDA. However, based on ocular and sensitization data, a concentration limit of 10% was placed on leave-on products containing PEG Glyceryl Cocoates. The current reported concentration of use in leave-on products is slightly higher, that is, 11.3% PEG-7 Glyceryl Cocoate in tonics, dressings, and other hair grooming aids.
PEG-8 Caprylic/Capric Glycerides can be a dermal penetration enhancer. In rats, PEG-6 Caprylic/Capric Glycerides had an oral LD50 of > 5 g/kg, PEG-8 Caprylic/Capric Glycerides had an oral LD50 of 22 g/kg, and PEG-7 Glyceryl Cocoate has an oral LD50 of > 19.9 mg/g. In a 4-week study in rats, the NOAEL for a blend of 40% PEG-8 Caprylic/Capric Glycerides, 40% apricot kernel oil PEG-6 esters, and 20% ethoxydiglycol was 5 mL/kg/d. Test-article related effects were reported in the kidneys, livers, and gastrointestinal systems of animals dosed with 20 mL/g/d; effects were also observed in animals given 10 mL/kg/d, but they did not occur at the same rate of incidence. In a 4-week dietary study in rats, the NOEL of a formulation containing 0.8% PEG-7 Glyceryl Cocoate was 2.5%; at higher concentrations, soft feces, a distended cecum, and enlarged mesenteric lymph nodes were observed, and a decrease in spleen weights was reported. No adverse effects were observed in dogs that were dosed orally for 13 weeks with 1.0 g/kg/d PEG-8 Caprylic/Capric Glycerides.
PEG-8 Caprylic/Capric Glycerides, up to 3,000 mg/kg in purified water, did not produce embryotoxicity, fetotoxicity, or teratogenicity. The maternal embryo/fetal NOAELs were 2,000 and 3,000 mg/kg/d, respectively. The PEG-6 Caprylic/Capric Glycerides, PEG-7 Glyceryl Cocoate, and PEG-10 Olive Glycerides were not mutagenic in the Ames test.
The irritation and sensitization potentials of several PEGylated alkyl glycerides were evaluated in alternative, nonhuman, and human studies, and these ingredients generally were not irritants or sensitizers in the alternative and human studies. Mixed results were observed in the nonhuman studies; specifically, undiluted PEG-20 Almond Glycerides, PEG-60 Almond Glycerides, PEG-12 Palm Kernel Glycerides, and PEG-45 Palm Kernel Glycerides were irritating to rabbit skin in primary skin irritation studies, and PEG-7 Glyceryl Cocoate was mildly irritating to rabbit skin in one study. The only reactions reported in clinical testing was PEG-10 Olive Glycerides (2% active matter) was nonirritating to mildly irritating. In other clinical studies, up to 20% PEG-60 Almond Glycerides, undiluted PEG-6 Caprylic/Capric Glycerides, undiluted PEG-7 Glyceryl Cocoate, and a formulation containing 1% PEG-75 Shea Butter Glycerides were not irritants or sensitizers.
In a HET-CAM, a formulation containing 11% PEG-7 Glyceryl Cocoate had practically no ocular irritation potential, and concentrations of 10%, 50%, and 100% PEG-7 Glyceryl Cocoate were slightly irritating. The PEG-10 Sunflower Glycerides was classified as a nonirritant in a CAMVA. In rabbits, formulations containing 4% PEG-6 Caprylic/Capric Glycerides and up to 3% PEG-8 Caprylic/Capric Glycerides were not ocular irritants and 5% PEG-8 Caprylic/Capric Glycerides was a slight irritant. Undiluted PEG-6 Caprylic/Capric Glycerides was a strong ocular irritant; however, aq. solutions of up to 5%, and formulations containing 4% PEG-6 Caprylic/Capric Glycerides were not ocular irritants in rabbits. In clinical testing, a formulation containing 1% PEG-75 Shea Butter Glycerides did not induce ocular irritation in a tolerance test.
PEG-10 Olive Glycerides, 3% active matter, did not irritate the mucous membrane. In a use study, a female hygiene product containing 0.1% PEG-75 Shea Butter Glycerides did not induce any evidence of vaginovulvar irritation.
Data on the carcinogenicity of PEGylated alkyl glycerides were not found in the published literature nor were unpublished data provided.
Discussion
Five of the ingredients included in this review, that is, PEG-7 Glyceryl Cocoate, PEG-30 Glyceryl Cocoate, PEG-40 Glyceryl Cocoate, PEG-78 Glyceryl Cocoate, and PEG-80 Glyceryl Cocoate were reviewed previously; in 1999, the Panel concluded these 5 ingredients are safe as used in rinse-off products and safe at up to 10% in leave-on products. The safety assessment of these ingredients was scheduled for rereview in 2014; however, the Panel found it appropriate to include the PEG Glyceryl Cocoates in this report, rather than conduct a separate rereview.
The Panel used the 1999 PEG Glyceryl Cocoates repor as well as the 2012 safety assessment of the PEGylated oils and the findings on skin irritation and sensitization included in this current report to inform the safety of these PEGylated alkyl glycerides. There are no data on skin penetration by the PEGylated alkyl glycerides, but many of the PEGylated alkyl glycerides are approved for use in dietary supplements and as indirect food additives, and PEG-8 Caprylic/Capric Glycerides is reported to be used as a pharmaceutical excipient. Data on carcinogenicity were not found, and these ingredients were not mutagenic and lack structural features of concern. Thus, the Panel focused its review on the safety of topical application.
Some ingredients demonstrated irritation, but at concentrations above those used in cosmetic formulations; however, because the potential exists for dermal irritation with the use of products containing PEGylated alky glycerides, the Panel specified that products containing these ingredients must be formulated to be nonirritating. It should be noted that although the conclusion of the 1999 report of PEG glyceryl cocoates limited their use to 10% in leave-on products, negative HRIPT data at and above the reported concentration of use, and the inclusion of the specification that these ingredients be used in products when formulated to be nonirritating, alleviated the need for that concentration limit in the current report.
The Panel recognized that these ingredients can enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data or when dermal absorption was a concern.
The Panel noted that some of the PEGylated alkyl glycerides are used in products that could be incidentally inhaled. Although reported concentrations of use included 6% PEG-7 Glyceryl Cocoate in “other” fragrance preparations and 2% PEG 7 Glyceryl Cocoate in a pump spray deodorant, and there were no inhalation data available, the Panel was not concerned with the use of these ingredients in such formulations. The Panel noted that in aerosol products, 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Finally, because these ingredients are obtained from plant sources, the Panel expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. The Panel stressed that the cosmetics industry should continue to use current good manufacturing practices to limit these impurities in the ingredient before blending into cosmetic formulation.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following 60 PEGylated alkyl glycerides are safe in cosmetics in the present practices of use and concentration when formulated to be nonirritating: PEG-6 Almond Glycerides* PEG-20 Almond Glycerides PEG-35 Almond Glycerides* PEG-60 Almond Glycerides PEG-192 Apricot Kernel Glycerides PEG-11 Avocado Glycerides* PEG-14 Avocado Glycerides* PEG-11 Babassu Glycerides* PEG-42 Babassu Glycerides* PEG-4 Caprylic/Capric Glycerides* PEG-6 Caprylic/Capric Glycerides PEG-7 Caprylic/Capric Glycerides PEG-8 Caprylic/Capric Glycerides PEG-11 Cocoa Butter Glycerides* PEG-75 Cocoa Butter Glycerides PEG-7 Cocoglycerides* PEG-9 Cocoglycerides* PEG-20 Corn Glycerides* PEG-60 Corn Glycerides* PEG-20 Evening Primrose Glycerides* PEG-60 Evening Primrose Glycerides* PEG-3 Glyceryl Cocoate PEG-7 Glyceryl Cocoate PEG-30 Glyceryl Cocoate PEG-40 Glyceryl Cocoate PEG-78 Glyceryl Cocoate* PEG-80 Glyceryl Cocoate PEG-5 Hydrogenated Corn Glycerides* PEG-8 Hydrogenated Fish Glycerides* PEG-20 Hydrogenated Palm Glycerides PEG-6 Hydrogenated Palm/Palm Kernel Glyceride* PEG-16 Macadamia Glycerides PEG-70 Mango Glycerides PEG-13 Mink Glycerides* PEG-25 Moringa Glycerides* PEG-42 Mushroom Glycerides* PEG-2 Olive Glycerides* PEG-6 Olive Glycerides* PEG-7 Olive Glycerides* PEG-10 Olive Glycerides PEG-40 Olive Glycerides* PEG-18 Palm Glycerides* PEG-12 Palm Kernel Glycerides* PEG-45 Palm Kernel Glycerides PEG-60 Passiflora Edulis Seed Glycerides* PEG-60 Passiflora Incarnata Seed Glycerides* PEG-45 Safflower Glycerides* PEG-60 Shea Butter Glycerides PEG-75 Shea Butter Glycerides PEG-75 Shorea Butter Glycerides* PEG-35 Soy Glycerides PEG-75 Soy Glycerides* PEG-2 Sunflower Glycerides* PEG-7 Sunflower Glycerides* PEG-10 Sunflower Glycerides PEG-13 Sunflower Glycerides PEG-5 Tsubakiate Glycerides* PEG-10 Tsubakiate Glycerides* PEG-20 Tsubakiate Glycerides* PEG-60 Tsubakiate Glycerides*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that those would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author Contributions
Fiume, M. contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception and design, analysis and interpretation, and critically revised manuscript. Heldreth, B. contributed to analysis and interpretation and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors' Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA. The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
