Abstract
Intrathecal (IT) dosing (ie, injection into the subarachnoidal space at the lumbar region) is a common route of administration in cynomolgus monkey preclinical safety studies conducted for antisense oligonucleotides (ASO) that target central nervous system diseases. Herein we report on neurological signs that have been observed in 28 IT studies conducted in 1,016 cynomolgus monkeys. Neurological signs were classified into 5 groups: (1) A nonadverse transient absence of lower spinal reflexes. This observation occurred at low incidence in nontreated animals and in those that were injected artificial cerebrospinal fluid. The incidence increased in animals that were injected an ASO. Reflexes were present again at 24 hours or 48 hours after dosing. The incidence appeared to increase with dose. (2) Test-article-related adverse muscle tremor or muscle spasticity occurring during the injection procedure or immediately thereafter. In one-third of animals this finding responded to treatment with diazepam, in two-third it required euthanasia. (3) Neurological findings occurring between 30 minutes and 4 hours after dosing were characterized by any combination of ataxia, paresis, nystagmus, urinary incontinence, or muscle tremor. Those conditions either spontaneously resolved or they slowly worsened, eventually resulting in a poor general condition. (4) Neurological findings due to spinal cord injury were characterized by rapidly progressing paralysis of hind limbs. Magnetic resonance imaging revealed a focal hyperintense lesion, indicative of spinal cord necrosis. (5) Test-article-related adverse hind limb paresis or paralysis that occurred between 2 and 18 days after dosing. Those findings were rare and resulted in a poor general condition requiring euthanasia.
Introduction
Many potential drugs against neurodegenerative diseases, particularly antisense oligonucleotides (ASO), are administered via lumbar intrathecal (IT) injection, as they do not cross the blood–brain barrier. 1 This route administers the test article into the subarachnoidal space and is well established in cynomolgus monkeys that are commonly used for preclinical safety assessment when other nonrodent species such as dog or minipig cannot be used. Based on the experience with 28 preclinical safety studies investigating 16 different ASO compounds, this publication describes the range of clinical neurological signs that were observed in these studies.
Materials and Methods
This article refers to data obtained from 28 preclinical safety studies that were conducted between 2012 and 2018 at the test facility Covance Preclinical Services GmbH in Münster, Germany. All studies were authorized by the state authority (Landesamt für Umwelt, Natur und Verbraucherschutz, Recklinghausen, Germany) and by an institutional animal care and use committee. All in vivo studies were conducted to support clinical trials. Cynomolgus monkeys of Asian or Mauritian origin were used for these experiments.
The 28 studies investigated pharmacokinetics and pharmacodynamics, screened multiple batches of ASOs with the same target for tolerability after repeated dosing, or were pivotal repeat dose toxicity studies up to 52 weeks duration. Test articles were administered by lumbar puncture and injection, dosed in intervals, typically with a 2-week interval in the first month and once per month dosing thereafter.
Parameters such as body weight, food consumption, cardiovascular examinations, ophthalmology examinations, bone mineral content or bone mineral density, femur length, learning ability, clinical pathology, blood gas analysis, complement activation, T-cell dependent antibody reactions, and anatomic pathology were evaluated in the scope of these studies but are not within the scope of this publication.
Test System
The 28 studies comprised a total of 1,016 animals. Out of these approximately 180 animals were injected artificial cerebrospinal fluid (aCSF) and the remaining were injected a test article. Animals originated from breeding colonies in Asia or Mauritius, and they were between 11 months and 9 years of age.
Test Articles
Test articles were 16 different ASO developed by various sponsors and for various central nervous system indications. Test articles were mostly diluted in aCSF (R and D Systems) and had concentrations between 0.4 and 66.7 mg/mL. Administered dose levels ranged from 0.3 to 60 mg per dose. The vehicle used for test article dilution and to dose control group animals was aCSF.
Intrathecal Administration Procedure
Overall >4,500 IT punctures and injections were conducted, each with a dose volume of 0.75 to 2.0 mL. Approximately, 1,100 of those 4,500 injections have been performed with aCSF.
Following sedation by intramuscular injection of ketamine (2.5 mg/ kg) and medetomidine (0.06 mg/kg), animals were placed in abdominal recumbency on a curved metal grid (Figure 1). The L6 spinous process was identified by an imaginary line drawn from one iliac crest to the other. From this line, the next cranial intervertebral space was considered to be L5/L6. For animals of 15 months or older, lumbal puncture was performed at L5/ L6 (or L4 /L5 if L5/L6 was too narrow). For younger animals (between 9 and 15 months) lumbal puncture was performed at L4/L5 (or L3/L4 if L4/L5 was too narrow).
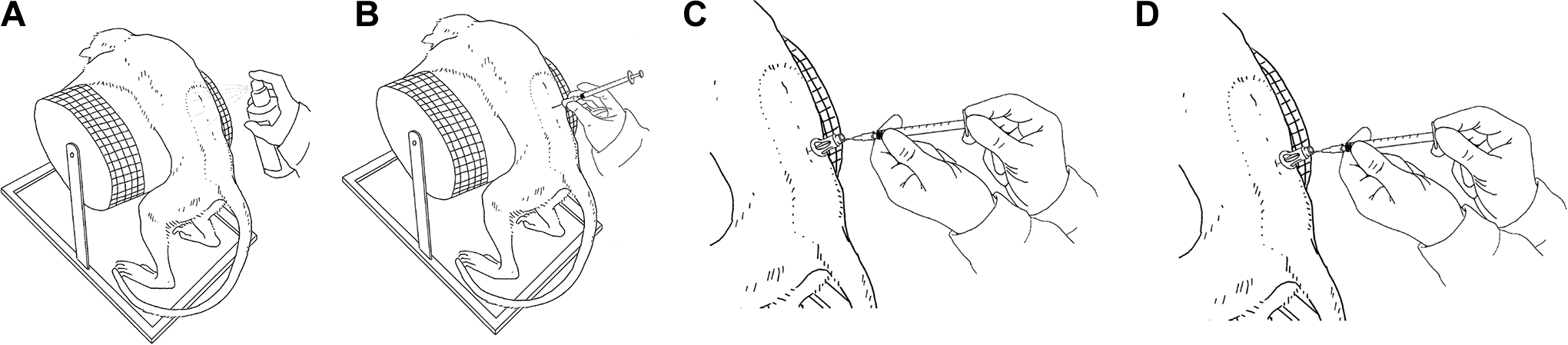
Schematic drawing of the lumbar intrathecal dosing procedure. (A) Skin disinfection; (B) insertion of blunt spinal needle through previously created skin incision; (C) CSF withdrawal; (D) dose administration.
The area of lumbal puncture was shaved, and a micro incision of the skin was conducted using a 20G needle. Thereafter, a Pencan Paed pencil-point needle (25 G, B. Braun) was inserted with the needle opening pointing cranially. When CSF flow was present, 0.5 mL of CSF were withdrawn. Then, the test article was injected through the same needle over approximately 1 minute. Dose volumes were between 0.75 and 2.0 mL, always followed by a 0.25 mL flush with aCSF.
After the procedure, the animal was transferred into a transport box where it was placed in lateral recumbency for approximately 15 minutes. Thereafter, medetomidine was antagonized with atipamezole (0.3mg/kg) and the animal was returned to its home cage.
Clinical and Neurological Postdose Examination
Before dosing and at 4 hours after dosing, animals were clinically examined outside of their home cage. The animal was placed on a 3 step metal ladder, and its arms and head were manually restrained by an assistant. The examination was conducted by a veterinarian and comprised a general inspection of the animal (including auscultation of heart and lungs, palpation of abdomen and axillary lymph nodes, and measurement of rectal body temperature). A detailed neurological examination comprised evaluation of behavior, gait, posture, neuromotor condition, and reflexes (Table 1). Reflexes were scored as “present”, “absent,” or “not evaluable.”
Reflexes That Were Examined in All Animals and Description of the Examination Procedure.

Results
The clinical and neurological observations can generally be classified into 5 groups:
Transient Absence of Reflexes
This observation is characterized by a lack of the pupillary reflex, the patellar reflex, the anal reflex, or the foot grip reflex, or any combination thereof. The lack of these reflexes is not associated with any abnormality in gait or with any impairment in the ability to climb or move. The incidence of this observation was assessed in detail in a subset of the 28 studies that were evaluated; this subset comprised 5 studies with 300 dosed animals (40 controls, 260 test tem treated).
The absence of reflexes is not necessarily a test-article-related effect. It was noted in 5% of the animals that were examined before receiving the first IT dose (Figure 2A). In animals that were intrathecally injected with aCSF, the incidence raised to 13% at 4 hours post injection (referring to the first dosing event). This indicates that the IT dosing procedure per se has an effect on the presence/absence of reflexes. All reflexes were present again at 24 hours after dosing. In subsequent dosing events, an increased absence of reflexes was not observed, since the incidence was in the same range as predose observations (Figure 2A).
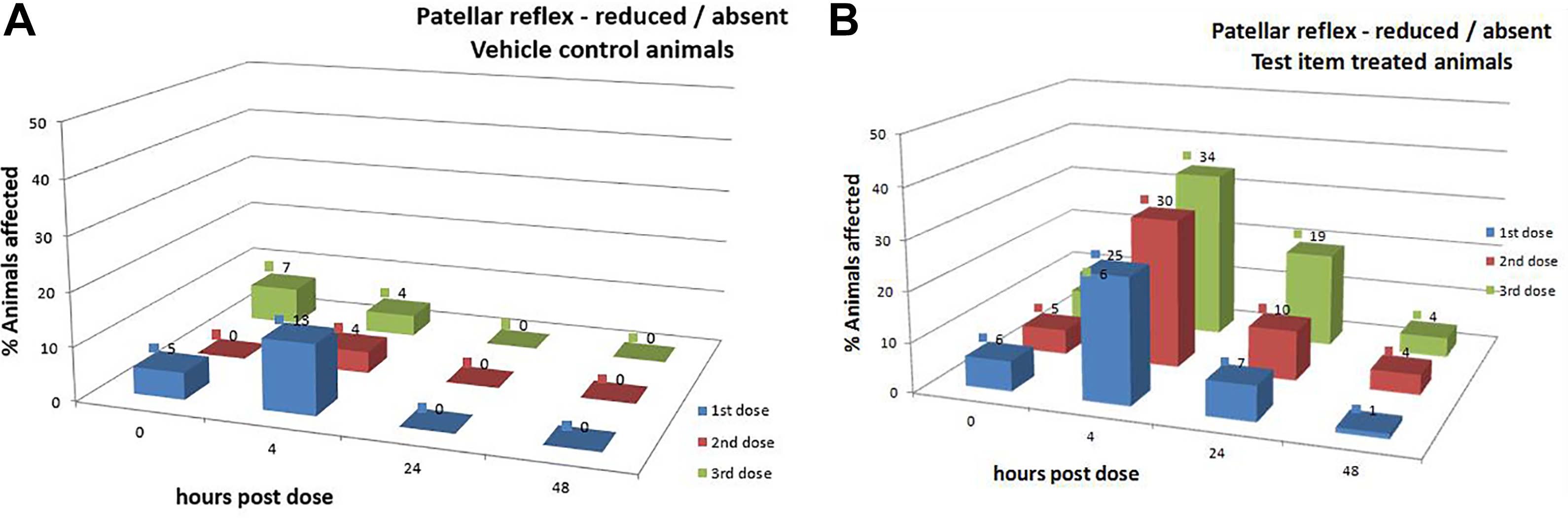
A, Percentage of animals affected by absence of the patellar reflex according to the lumbar intrathecal injection (dosing event) and the time after dosing. All animals (n = 40) were injected aCSF. Dosing was performed at 14-day intervals. B, Percentage of animals affected by absence of the patellar reflex according to the lumbar intrathecal injection (dosing event) and the time after dosing. All animals (n = 260) were injected an ASO (different ASOs and different dose levels were combined). Dosing was performed at 14-day intervals. ASO indicates antisense oligonucleotides.
In ASO-dosed animals, the absence of reflexes occurs at higher incidence (25%) compared to aCSF-treated controls (13%). In addition, the absence of reflexes occurs consistently throughout following dosing events, with an apparent increase over the number of doses (Figure 2B). Interestingly, the absence of reflexes does not always resolve within 24 hours. In 7% to 19% of animals, reflexes were still absent at 24 hours post injection. At 48 hours post injection, the incidence of absent reflexes was similar to predose values, that is, had fully recovered (Figure 2B). When dose groups were compared, there was a higher incidence in high-dose groups compared to low-dose groups (data not shown).
Clinical Findings Occurring Under Dosing or Immediately After Dosing
In 3 out of 28 studies and in 6 out of 1,016 animals (0.6%), severe muscle tremor (rhythmic contraction and relaxation) or muscle spasticity (tightness) was observed under injection of the test article or immediately thereafter (ie, within 30 minutes during the wake-up phase). Those muscle contractions concerned the muscles on the back and the tail and eventually affected both hind limbs. All animals with this observation were dosed with a test article and were allocated to a high-dose group. Notably, 3 out of these 6 animals were juvenile with a body weight between 1.2 and 1.8 kg. The described clinical findings occurring under dosing were not observed in any animal receiving aCSF.
In all 6 cases, intravenous treatment with diazepam was initiated, which improved the symptoms in 2 animals. The 4 animals in which diazepam did not improve the symptoms were euthanized on the day of dosing, because muscle tremor resulted in paralysis (loss of muscle function) and poor general condition.
Neurological Findings Occurring Between 30 Minutes and 4 Hours After Dosing
In 4 out of 28 studies and in 6 out of 1,016 animals (0.6%), one or more of the following findings occurred between 30 minutes (after narcosis) and 4 hours after dosing: Any combination of nystagmus (eye movement), tremor of hind limb muscles (rhythmic contraction and relaxation), hind limb paresis (impaired movement), ataxia (gait abnormality), or urinary incontinence.
All 6 animals were dosed with a test article and were allocated to different dose levels. Such observations were never seen in control animals (administered aCSF) or in animals before the first dose (predose phase). In two animals, the condition spontaneously resolved within 7 or 29 days, whereas in the remaining 4 animals, the condition slowly worsened over 2 to 7 days and resulted in hind limb paralysis (loss of muscle function), often associated with evidence for impaired health such as rough fur, reduced feed intake, hypothermia, or dehydration. These animals were preterminally euthanized.
Postdose Neurological Findings Associated With Spinal Cord Injury
Three animals (allocated to 3 different studies) exhibited hind limb paresis (impaired movement) and ataxia (gait abnormality) after dosing. In these 3 animals, nystagmus (eye movement) or muscle tremor were not observed. In addition, the condition rapidly progressed to hind limb paralysis within 4 hours. Although those observations show some overlap with the test-article-related ones summarized above, they were distinct in as far as paralysis of both hind limbs was present after 4 hours. In these 3 animals, the lumbar spinal cord was examined by magnetic resonance imaging (MRI) which revealed a focal hyperintense lesion, indicative of spinal cord necrosis. Consequently, these 3 cases were considered to represent a traumatic spinal cord injury that was induced by the dosing procedure. Lumbar spinal cord necrosis is a very rare event with a prevalence of less than 0.1% according to our database.
Neurological Findings Occurring Later Than 2 Days After Dosing
In 1 out of 28 studies and in 25 (2.5%) out of 1,016 animals, hind limb paralysis occurred later than 2 days after dosing. The longest time between dosing and onset of paralysis was 18 days. All affected animals had been dosed with a test article. Paralysis was never seen in control animals (administered aCSF) or in animals before the first dose (predose phase). While at the onset of paralysis the animals were still able to move within their cage by the use of their arms, the general condition rapidly worsened and animals needed to be euthanized within 1 or 2 days after onset.
Discussion
Based on the experience with 28 studies that all comprised the IT administration of ASOs, we conclude that a detailed clinical and neurological postdose examination is important to detect test-article-related abnormalities. These can be differentiated according to the temporal relationship of the occurrence relative to dosing of the test article.
Neurological findings that occur under dosing (ie, within the minute of dosing or immediately thereafter) are primarily characterized by intense muscle tremor/tension. Immediate treatment with diazepam is recommended although it has been helpful only in 33% of cases. Animals not responding to the therapy need to be euthanized, as these symptoms do not spontaneously resolve and may result in severe distress.
Neurological findings that occur between 30 minutes and 4 hours after dosing can be further differentiated into 3 groups: An increased incidence in the transient loss of one or more reflexes. This is a consistent observation in IT ASO studies (occurring in 100% of studies). It is test-article-related but nonadverse, because it is not associated with any deficit in gait or behaviour (Engelhardt; personal communication). Muscle tremor (less severe compared to those occurring within dosing) affecting the hind limbs, associated with nystagmus (rapid uncoordinated eye movement), urinary incontinence, or unsteady gait (ataxia). This may slowly progress to paresis or paralysis of hind limbs and may require that animals be euthanized. These neurological findings are rare, but they are test-article-related and are adverse. Rapidly progressing loss of hind limb muscle function resulting in paralysis of both hind limbs. This neurological finding is rare and is associated with a focal hyper-intense lesions in MRI, representing focal spinal cord necrosis. This finding is not test-article-related but is due to local injury associated with the IT administration technique.
Neurological findings that may occur later than 48 hours after dosing are characterized by sudden onset of paresis or paralysis of one or both hind limbs, which typically progresses rapidly and does not resolve spontaneously. It typically requires that animals are euthanized. This finding is test-article-related and adverse.
Conclusion
The lumbar IT injection of ASO might be associated with test-article-related observations, which can be classified according to their character and their time of occurrence in reference to the dosing procedure. The suggested classification, which is based on 28 studies in a total of 1,016 cynomolgus monkeys, allows to discriminate nonadverse test-article-related observations and injury-related observations from those that are test-article-related and adverse.
Footnotes
Author Contributions
Korte, S. substantially contributed to conception or design and drafted the manuscript; Runge, F. substantially contributed to conception or design and drafted the manuscript; Wozniak, M. M. substantially contributed to conception or design and drafted the manuscript; Ludwig, F. T. substantially contributed to conception or design and critically revised the manuscript for important intellectual content; Smieja, D. contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content; Korytko, P. substantially contributed to conception or design and drafted the manuscript; Mecklenburg, L. contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
