Abstract
Direct delivery of therapeutics to the central nervous system (CNS) greatly expands opportunities to treat neurological diseases but is technically challenging. This opinion outlines principal technical aspects of direct CNS delivery via intracerebroventricular (ICV) or intrathecal (IT) injection to common nonclinical test species (rodents, dogs, and nonhuman primates) and describes procedure-related clinical and histopathological effects that confound interpretation of test article-related effects. Direct dosing is by ICV injection in mice due to their small body size, while other species are dosed IT in the lumbar cistern. The most frequent procedure-related functional effects are transient absence of lower spinal reflexes after IT injection or death soon after ICV dosing. Common procedure-related microscopic findings in all species include leukocyte infiltrates in CNS meninges or perivascular (Virchow-Robin) spaces; nerve fiber degeneration in the spinal cord white matter (especially dorsal and lateral tracts compressed by dosing needles or indwelling catheters), spinal nerve roots, and sciatic nerve; meningeal fibrosis at or near IT injection sites; hemorrhage; and gliosis. Findings typically are minimal to occasionally mild. Findings tend to be more severe and/or have a higher incidence in the spinal cord segments and spinal nerve roots at or close to the site of administration.
Keywords
This is an opinion article submitted to the Toxicologic Pathology Forum. This perspective represents the views of the authors. It does not constitute an official position of the Society of Toxicologic Pathology (STP), British Society of Toxicological Pathology (BSTP), or European Society of Toxicologic Pathology (ESTP), and the views expressed do not reflect the policies, positions, or opinions of these Societies, the authors’ respective organizations and employers, or regulatory agencies. The Toxicologic Pathology Forum is designed to stimulate discussion of topics relevant to regulatory issues in toxicologic pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these opinion articles or ideas for new discussion topics to
Introduction
Direct delivery of therapeutics into the central nervous system (CNS) has several advantages over systemic administration in treating neurological diseases. These benefits include the ability to bypass the blood-brain barrier (BBB) and allow for accumulation of higher peak drug levels within CNS tissues while limiting systemic exposure, all of which can improve the overall safety profile. However, the disadvantages of direct CNS delivery are also notable and include the invasive nature of routinely used administration procedures that can potentially result in undesirable side effects related to the procedure (e.g., injection site pain and neural tissue trauma) and the requirement that drug delivery be performed at a specialty medical facility. In nonclinical species, clinical observations and histopathology findings associated with direct CNS delivery procedures may confound interpretation of potential test article-related effects.
In this opinion piece, we outline our experience with technical aspects of direct CNS delivery in common nonclinical species (rodents, dogs, and nonhuman primates [NHPs]) and highlight the common procedure-related effects (clinical and neurological observations as well as histopathological findings) that we have observed in vehicle control groups. These data have been gathered from multiple studies testing the nonclinical safety of antisense oligonucleotides (ASO) intended to treat CNS diseases. All studies were sponsored by Ionis Pharmaceuticals (Carlsbad, CA, USA) and performed at either Ionis Pharmaceuticals or at a select number of contract research organizations (including Labcorp, Münster, Germany) who have a wealth of nonclinical expertise in direct CNS delivery. The constellation of studies compiled for this opinion piece include both non-Good Laboratory Practice (GLP) investigative and GLP-compliant nonclinical toxicity studies in mice, rats, dogs, and NHPs (Table 1). All studies were reviewed and approved in advance by relevant Institutional Animal Care and Use Committees. All animal care and use procedures were conducted in alignment with animal welfare regulatory requirements at Association for Assessment and Accreditation of Laboratory Animal Care accredited animal programs and in accordance with then-existing local and national laws. For all studies, materials were administered into the cerebrospinal fluid (CSF) of test species via intracerebroventricular (ICV) injection (mice) or intrathecal (IT) injection (rats, dogs, and NHPs). Except for two of the rat studies that were single-dose, all studies had a repeat-dose design. All studies were at least 2 weeks in duration but were usually at least 6 weeks in duration. In all studies, artificial CSF (aCSF) or ASO diluted in aCSF was administered once weekly or less frequently. The aCSF vehicle was a water-based solution with a pH of approximately 7.2 and contained sodium, potassium, calcium, magnesium, chloride, and phosphorus. Thus, it closely approximated the pH and electrolyte composition of mammalian CSF. All administered solutions were delivered at approximately room temperature (20°C–22°C).
Direct CNS delivery experience through Ionis-sponsored nonclinical studies.
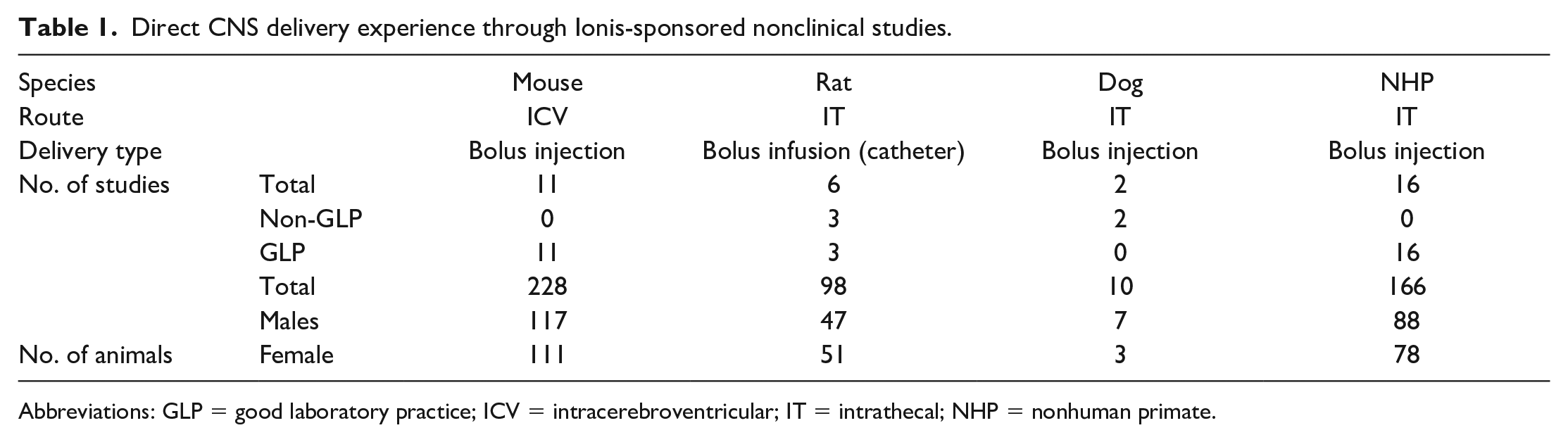
Abbreviations: GLP = good laboratory practice; ICV = intracerebroventricular; IT = intrathecal; NHP = nonhuman primate.
The bulk of our collective direct CNS delivery experience comes from studies of bolus injections in mice and NHPs. Therefore, much of the information we share will focus on these species. Studies with rats have typically been limited to delivery by catheter infusion during either single-dose or repeat-dose exploratory and tolerability screening studies. We have only recently evaluated dogs as the large animal species for direct CNS delivery studies and to date have performed only a few studies in this species. Dogs have been utilized in non-GLP pilot studies to establish the IT delivery procedure and to define the ASO toxicity profile after IT injection in this species.
Technical Considerations for Direct CNS Delivery
Procedures used for direct CNS delivery are conceptually similar: a CSF-filled space somewhere along the neuraxis must be penetrated aseptically to permit instillation of a dosing solution. However, methodological details vary with the species and the targeted injection site. Consideration must be given to species-specific anatomical differences. For example, the rat, dog, and NHP only have one or two intervertebral spaces through which the lumbar cistern can be accessed while in the human, the large cauda equina space and dorsal spinal anatomy is best for lumbar puncture or lumbar catheterization. 28 This section briefly recapitulates species-specific elements of the direct CNS delivery process for common species used for nonclinical toxicity testing of ASO test articles.
For nonclinical safety testing, ASO can be administered by bolus infusion or bolus injection. Bolus infusion typically employs an indwelling polyurethane catheter (outer diameter 0.6 mm [18 FR] for the rat) to introduce the test article over one to two minutes. More commonly, bolus injection delivers the test article using a 22- to 25-gauge rigid metal needle (22-gauge in the mouse; 25-gauge, 2 or 3.5 inches long in the NHP and dog, respectively). Most of our nonclinical safety studies evaluating direct CNS delivery of ASO test articles in mice and NHPs have been performed by bolus injection using needles. Studies in rats have been conducted primarily by bolus infusion using indwelling catheters, and studies in dogs have utilized either bolus injection or bolus infusion via indwelling catheters.
Mice—Intracerebroventricular Injection
Mice are the preferred rodent species for Ionis-sponsored GLP-compliant toxicity studies of ASO test articles and are also used in non-GLP tolerability studies designed to select the best candidates to advance to studies in nonrodents (generally NHPs). This is because brain and spinal cord ASO exposure after repeated ICV bolus injection in the mouse more closely models ASO distribution in the CNS of NHPs after IT lumbar puncture bolus administration versus IT bolus catheter infusion in rats. 16
Although IT injection is the intended route of administration for neuroactive ASOs in humans, direct CNS delivery in mice is via ICV injection because the lateral ventricle of the brain offers a more reliable target in this species compared with the very small size of the lumbar cistern. While the cisterna magna offers another comparatively large-volume chamber into which solutions may be injected, we do not use intra cisterna magna (ICM) injection in mice for our repeat-dose studies due to the likelihood of penetrating the brainstem, which would disrupt key structures that are necessary to maintain life. 2 Similarly, repeat-dose IT injections are technically difficult in mice due to the small volume of the vertebral canal and correspondingly reduced size of the lumbar cistern. While we have some experience using repeat-dose ASO delivery by ICV injection in larger species (rats, dogs, and NHPs), IT injection is used more often in the latter because: (1) direct IT delivery is the relevant route of administration in humans; and (2) the larger volume of the lumbar cistern in these species readily supports IT injection.
We perform ICV bolus injections into a lateral ventricle of anesthetized mice using a rigid 22-gauge needle to deliver a volume of up to 10 µL over a time of approximately 10 seconds. 13 Less frequently, we instill slow infusions into the mouse lateral ventricle using a rigid cannula connected to a subcutaneously implanted osmotic minipump. 13 Typically, ICV bolus injections are simpler and more cost-effective in mice, especially in smaller juveniles. Correct placement of the injection device is determined using a stereotactic apparatus to introduce the device to a depth of approximately 3 mm from the surface of the skull in adult animals. ICV injection in mice is usually limited to 5 or fewer injections to prevent morbidity and/or mortality due to cumulative local tissue injury. Such injury may be minimized by inserting the needle along the same track for each injection.
Rats—Intrathecal Injection
While rats are not generally used in Ionis-sponsored GLP-compliant candidate selection and chronic toxicity studies for neuroactive ASOs, they are used in Ionis investigative toxicity studies. Using rats rather than mice for exploratory and tolerability screening allows for chronic dosing into the lumbar cistern, which more closely approximates the route of administration in human patients than does dosing via ICV injection.
Direct CNS delivery in Ionis rat studies is undertaken via IT lumbar catheterization.11,20 The simple surgical procedure we developed uses no specialized equipment, can be completed in less than 25 minutes per animal, and has a success rate >90% (as determined by appropriate target gene knockdown [unpublished] data) when performed by properly trained and sufficiently experienced technical staff. Briefly, anesthetized rats are positioned in sternal recumbency, and a small incision is made through the skin and underlying muscle overlying vertebrae L4-L5. Using a modified 19-gauge × ½ inch needle as a guide cannula, a 23-gauge × 0.75-inch needle is used to puncture the intrathecal sac, and a semi-rigid catheter (0.025-inch outer diameter) is inserted into the lumbar cistern and is extended cranially in the subarachnoid space over the spinal cord for approximately 2 cm. The standard dosing volume, regardless of rat weight, is 30 µL followed by a 40 µL vehicle flush, both delivered by manual pressure over approximately 30 seconds. After dosing, the catheter is heat-sealed to prevent retrograde leakage of the dosing solution and CSF. The protruding catheter end is then inserted into a pocket made in the subcutis before the skin incision is closed. A major advantage of this surgical procedure is that the IT catheter can be left in place for up to 13 weeks during which time repeated doses can be easily administered at the desired frequency by re-anesthetizing the animals, opening the catheter, and then repeating the dosing and wound closure steps.
IT bolus injection via lumbar puncture using a rigid needle (25- to 30-gauge) is less frequently performed in Ionis rat studies. Our aversion to repeat dosing using needles in this species reflects: (1) the lower success rate in terms of proper placement of the delivery device (and thus target knockdown) across injection tries; (2) frequent leakage of dosing solution into the subcutis from the needle hole created in the meninges; and (3) limited distribution of the dosing solution, mainly to the lower spinal cord and nerve roots such that only variable amounts of the test article reaches the brain. 16
Dogs—Intrathecal Injection
Although infrequently used to assess attributes of ASO test articles at Ionis, dog studies by IT bolus injection using a needle (25-gauge) or IT bolus infusion using a catheter (27-gauge × 4 inch) may be suitable in some circumstances. In our experience, both IT administration procedures have similar success rates in the dog as determined by ASO distribution and target gene knockdown within the CNS (unpublished data). Administration by IT bolus injection is technically more straightforward since it involves a ventrally directed needle penetration into the lumbar cistern over the cauda equina. In contrast, IT catheterization involves introducing a semi-rigid catheter 2 to 3 cm cranially into the subarachnoid space overlying the lumbar spinal cord, thus risking trauma to the spinal nerve roots and spinal cord during insertion. Moreover, the catheter protruding from the vertebral canal must be secured in place to avoid displacement over time, which can further damage the cord parenchyma. We believe this potential to iatrogenically damage the delicate cord tissue is the reason for an increased incidence of adverse procedure-related effects that persist after dosing by IT catheterization when compared with IT bolus injection (whether single or repeated dosing). In some cases, adverse procedure-related effects necessitated removal of the animal from the study and even euthanasia. Importantly, in Ionis nonrodent studies, dosing by IT bolus injection is preferred over IT catheter bolus infusion for nonclinical toxicity testing because it more accurately models the ASO administration procedure (intermittent lumbar puncture) in human patients.
When performing IT bolus injections, anesthetized dogs are positioned in either sternal or lateral recumbency with hind legs pulled cranially and secured. This orientation flexes the vertebral column and opens the intervertebral spaces, especially in the lumbar region. The hair over vertebrae L5 to S1 is clipped, and the skin is aseptically prepared. A micro-incision of the skin over either L5-L6 or L7-S1 is made using a 20-gauge needle, after which the 25-gauge dosing needle is introduced and advanced through the soft tissue, threaded between the vertebrae, and inserted through the dura mater. Prior to ASO administration, 2 mL of CSF is withdrawn via the dosing needle to ensure that CSF flow is present and that the needle is correctly positioned. Dose volumes of up to 2.0 mL followed by a 0.5-mL aCSF flush are administered over approximately 1 minute. After dosing, the needle is withdrawn, and gentle manual pressure is applied to the site for at least 30 seconds to permit the soft tissue edges to anneal and heal without further treatment.
Nonhuman Primates—Intrathecal Injection
NHPs are the most utilized nonrodent species for nonclinical toxicity assessment of ASOs. The IT bolus injection procedure in NHPs has a high success rate (>99%) as indicated by target gene knockdown, and adverse procedure-related neurobehavioral findings are rare (<0.1%).17,18 Interpretation of ASO-related neurobehavioral effects in NHPs is relatively straightforward as test article-related and procedure-related effects can be effectively differentiated by considering such factors as the presence or absence of a dose-response relationship as well as the incidence, timing, and correlative findings (e.g., blood contamination of CSF) during dosing. For example, while ASO class-related effects are typically transient (usually persisting for 4 to no more than 24 hours after dosing), adverse procedure-related effects are more likely to persist beyond 24 hours after dosing.17,18
The IT bolus injection procedure for NHPs has been previously described17,18 and is similar to the details described above for the dog. Briefly, sedated animals are placed in sternal recumbency on a curved metal grid with the arms fixed with slings to prevent movement (Figure 1A-D). The hair over vertebrae L3-L5 is shaved, the skin surface is prepared aseptically, and a micro-incision is made in the skin using a 20-gauge needle before introduction of the 25-gauge dosing needle. As with dog IT injections, it is essential that CSF flow is present to confirm correct placement of the needle. After withdrawal of 0.6 mL of CSF, dose volumes ranging from 0.75 to 2.0 mL (typically 1.0 mL) followed by a 0.25-mL aCSF flush are administered over approximately 1 minute.
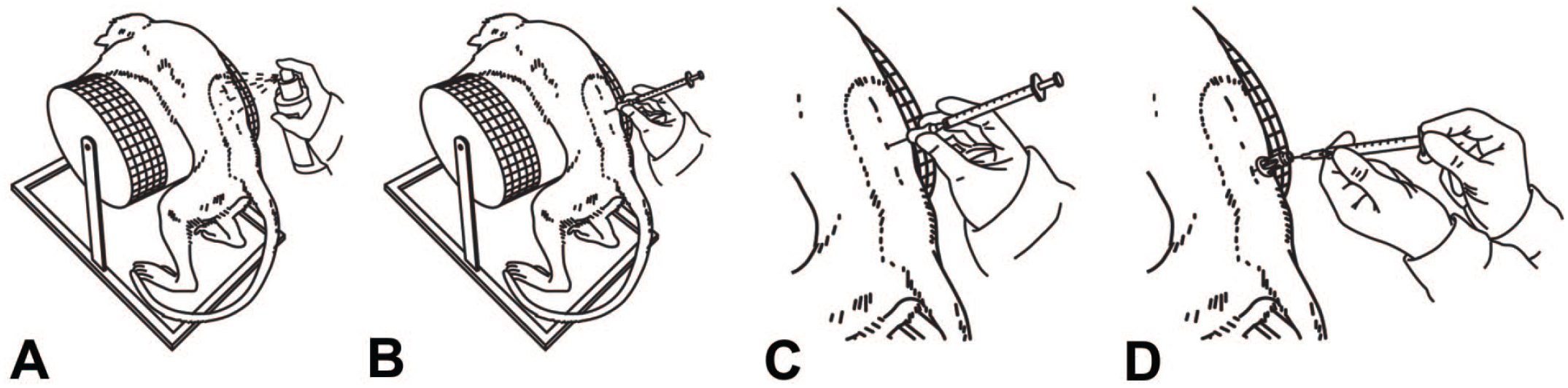
Schematic diagrams demonstrating intrathecal injection of a test article into the lumbar cistern of a nonhuman primate. The animal is positioned in sternal recumbency over a cylinder to flex the vertebral column and widen the intervertebral spaces. (A) Skin disinfection; (B) insertion of a blunt 25-gauge dosing needle through a previously created skin micro-incision; (C) cerebrospinal fluid (CSF) withdrawal in preparation for dosing; (D) dose administration (Modified from Korte et al.17,18).
Procedure-Related Functional Effects of Direct CNS Delivery
In nonclinical toxicity studies involving test article administration by direct CNS delivery, both clinical and neurological observations may be altered. In all species, clinical observations (mobility, posture, etc.) are generally documented twice daily during the dosing phase while neurological examinations (behaviors, reflexes, etc.) are generally performed prior to each dose and at 4 hours post-dose, potentially followed by a functional observational battery (FOB)14,15,19,22 or modified Irwin test15,19 (i.e., multi-assay batteries that assess core autonomic, behavioral, neuromuscular, and sensorimotor functions) in the rare case that severe clinical and/or neurological abnormalities, such as severe ataxia, are observed. If abnormalities are seen in screening, additional neurological examinations may be performed to follow the course of resolution.
The kinds of functional abnormalities evident following IT administration depend on the species. In ICV studies in mice, the most common procedure-related functional finding is death within a few hours after dosing. In IT studies in rats and NHPs, neurological abnormalities encountered after dosing encompass a range of immediate (minutes to hours) and intermediate-term (2 or more days), usually transient findings that can be divided into four main categories (as detailed for NHPs). 18 First, lower spinal reflexes (i.e., those involving reflex arcs mediated by cell populations near the injection site) may be transiently absent. Second, muscle cramping and/or tremors mainly in the hind limbs may occur during or immediately after dosing. Third, a plethora of transient neurological findings (e.g., nystagmus, hind limb paresis, ataxia, urinary incontinence) may be seen from 30 minutes to 4 hours after dosing. Finally, some neurological findings (generally paresis or paralysis of the hind limbs) may be delayed until 2 or more days after dosing. Of these 4 classes of neurological findings, transient absence of spinal reflexes is the most common, and is the only category that is routinely observed in control (aCSF-injected) animals affecting up to 13% of aCSF-injected NHPs. 18 The latter 3 categories of neurological abnormalities have not been observed in control NHPs except in the rare cases in which demonstrable dosing errors yielding visible tissue damage can be confirmed (by macroscopic and/or microscopic evaluation) as the cause of spinal cord injury.17,18
Our experience with IT studies in dogs is limited. Therefore, a detailed summary of what procedure-related clinical and neurological abnormalities may be expected in this species is lacking. The range of reported procedure-related findings in our dog toxicity studies includes transient and minor alterations such as loss of reflexes and gait abnormalities with infrequent instances of paralysis related to catheter penetration of the spinal cord (confirmed during necropsy).
Procedure-Related Histopathologic Effects of Direct CNS Delivery
In addition to a thorough knowledge of the range of spontaneous microscopic findings in the CNS of the species and stock / strain / breed examined, the primary challenge for the study pathologist involved in the review and interpretation of neural tissues collected from animals from direct CNS delivery studies is differentiating between effects related to the injection procedure versus those associated with the test article. This discrimination is particularly difficult in direct CNS delivery studies because the findings observed in vehicle control and ASO-treated animals are often similar, the main differences being the incidence and severity of various microscopic findings. In addition, the low numbers of animals per group in nonclinical studies using nonrodents magnifies the challenge of differentiating procedure-related from ASO-related findings. Historical control data (HCD) is useful when available, particularly when findings are present only in ASO-treated animals but are of low incidence and/or lack a dose-response relationship. In these situations, confirming the mere presence, and ideally the incidence of a given microscopic finding in control animals in other similar studies of direct CNS delivery, can provide a scientific rationale for concluding that a finding is not related to test article exposure. Such HCD must be compiled for the route of administration (e.g., ICV vs IT) and the delivery method (e.g., bolus injection by rigid needle vs bolus infusion by semi-rigid catheter) to be relevant for data interpretation. However, review of species-specific HCD related to other routes of test article administration (e.g., subcutaneous) can also be helpful in differentiating between procedure-related findings and spontaneous background changes.
Regardless of the species, some steps in the pathology evaluation are common to all nonclinical studies designed as screens for potential procedural effects (and ASO toxicity) following direct CNS delivery. A spectrum of neural tissues is examined, including the brain and spinal cord and the injection site for IT, but not ICV studies. In IT studies, selected dorsal root ganglia (DRG) with attached spinal nerve roots and at least one nerve (usually the sciatic but sometimes the tibial trunk, where in both motor and sensory nerve fibers are intermingled) are evaluated as well. Samples are fixed by immersion in 10% neutral buffered formalin (NBF) (available commercially as a solution containing approximately 1% methanol as a stabilizing agent) for 24 to 48 hours, followed by routine processing into paraffin, and sectioning at 4 to 5 µm. For general screening, sections are stained with hematoxylin and eosin (H&E). If warranted, special neurohistological methods, including FluoroJade (B or C), may be applied to serial sections of selected CNS regions or structures (typically brain and/or spinal cord) to better characterize any tissue responses to test article administration. 6 In this regard, various glial markers (e.g., immunohistochemistry to detect the glial fibrillary acidic protein [GFAP] in astrocytes or ionized calcium-binding adaptor molecule 1 [Iba1] in microglia and macrophages) or methods to demonstrate structural integrity (e.g., Bielschowsky’s silver for axons, Luxol fast blue [LFB] for myelin) are useful techniques for defining patterns of structural changes within neural tissues.6,7
Microscopic findings related to IT catheterization in the lumbar cistern have been described in control rats, dogs, sheep, and NHPs. 9 Leukocyte infiltrates (i.e., cell accumulation without any damage to the involved neural parenchyma) and localized inflammation (i.e., cell aggregation with damage to the parenchyma) as well as nerve fiber degeneration in the spinal nerve roots and/or the spinal cord at the catheterization site are common in all species. Gliosis in the spinal cord is also sometimes observed. The nerve fiber degeneration and gliosis are typically asymmetric in severity and likely represent trauma to the superficial white matter layers of the dorsal funiculi associated with sustained compression by the indwelling delivery device. We have observed similar findings at the catheterization site in our internal Ionis IT studies in rats (unpublished data).
The character of procedure-related findings will be influenced by the approaches to dose administration, neural sampling, and tissue processing. For our ASO studies, such findings vary by species due to subtle differences in the regional anatomy at the injection site. 3 Recommended approaches to sampling and processing the CNS5,6 and peripheral nervous system (PNS) 7 permit some degree of standardization across rodent and nonrodent species while permitting sufficient flexibility to adjust procedures to optimize the pathology evaluation to meet program- or test article-specific study objectives.4,24-26
Mice—Intracerebroventricular Injection
The mouse is the obvious outlier in terms of the detected spectrum of procedural effects. The differences relate to both the ICV injection site in this species, which is displaced substantially in space from the IT (lumbar cistern) route used in other species, and because the approaches to neural sampling and processing are particular to this species. In our mouse ICV studies, brain is sectioned longitudinally, after which the right hemisphere (which received the ICV injection(s)) is typically used for evaluating ASO tissue concentration while the left half (not injected) is fixed by immersion for microscopic evaluation. Spinal cord is removed from the vertebral column and immersion fixed, then trimmed in cross and longitudinal sections at the cervical, thoracic, and lumbar levels. The DRG and spinal nerve roots are not specifically examined microscopically in our mouse ICV studies, but spinal nerve roots are sometimes present in routine spinal cord sections. Accordingly, procedure-related microscopic findings in the brain and spinal cord are uncommon in mouse ICV studies.
When present, procedure-related microscopic findings within the brain of ICV-injected mice differ depending on the area(s) available for evaluation (Table 2 and Figure 2A-D). In the noninjected hemisphere, common findings include scattered, mononuclear cell infiltrates of minimal severity within the meninges and perivascular spaces along penetrating blood vessels; a constellation of one or more parenchymal changes indicative of neural tissue trauma (e.g., acute hemorrhage, deposition of hemosiderin pigment, neuropil vacuolation [i.e., edema], reactive gliosis, and/or tissue loss); and occasionally ventricular enlargement. If brain tissue from the injection site is evaluated, procedure-related microscopic findings observed locally (primarily in the meninges) include adhesions between the various meningeal layers and/or the leptomeninges to the skull, meningeal fibroplasia or fibrosis, foreign material (usually bone or hair shaft fragments) with or without granuloma formation, skull fractures sometimes with callus formation bridging the break, hemorrhage, mononuclear cell or mixed cell infiltrates, mixed cell inflammation, and/or macrophage infiltrates. The incidences of procedure-related findings observed for control mice in ICV studies are given in Table 2, and examples are provided in Figure 2A-D.
Incidence of common procedure-related microscopic findings in control mice after repeated intracerebroventricular (ICV) injection.
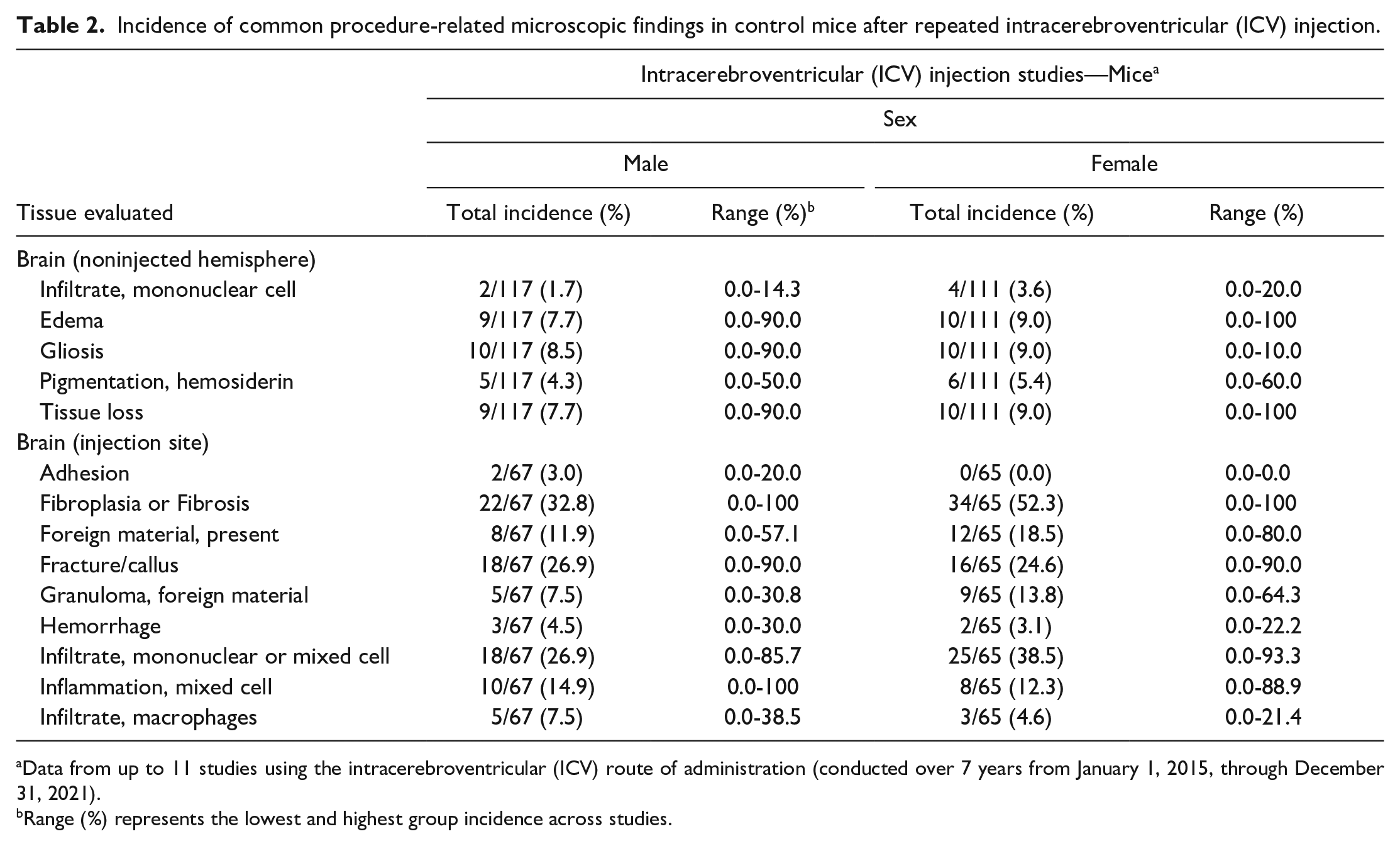
Data from up to 11 studies using the intracerebroventricular (ICV) route of administration (conducted over 7 years from January 1, 2015, through December 31, 2021).
Range (%) represents the lowest and highest group incidence across studies.
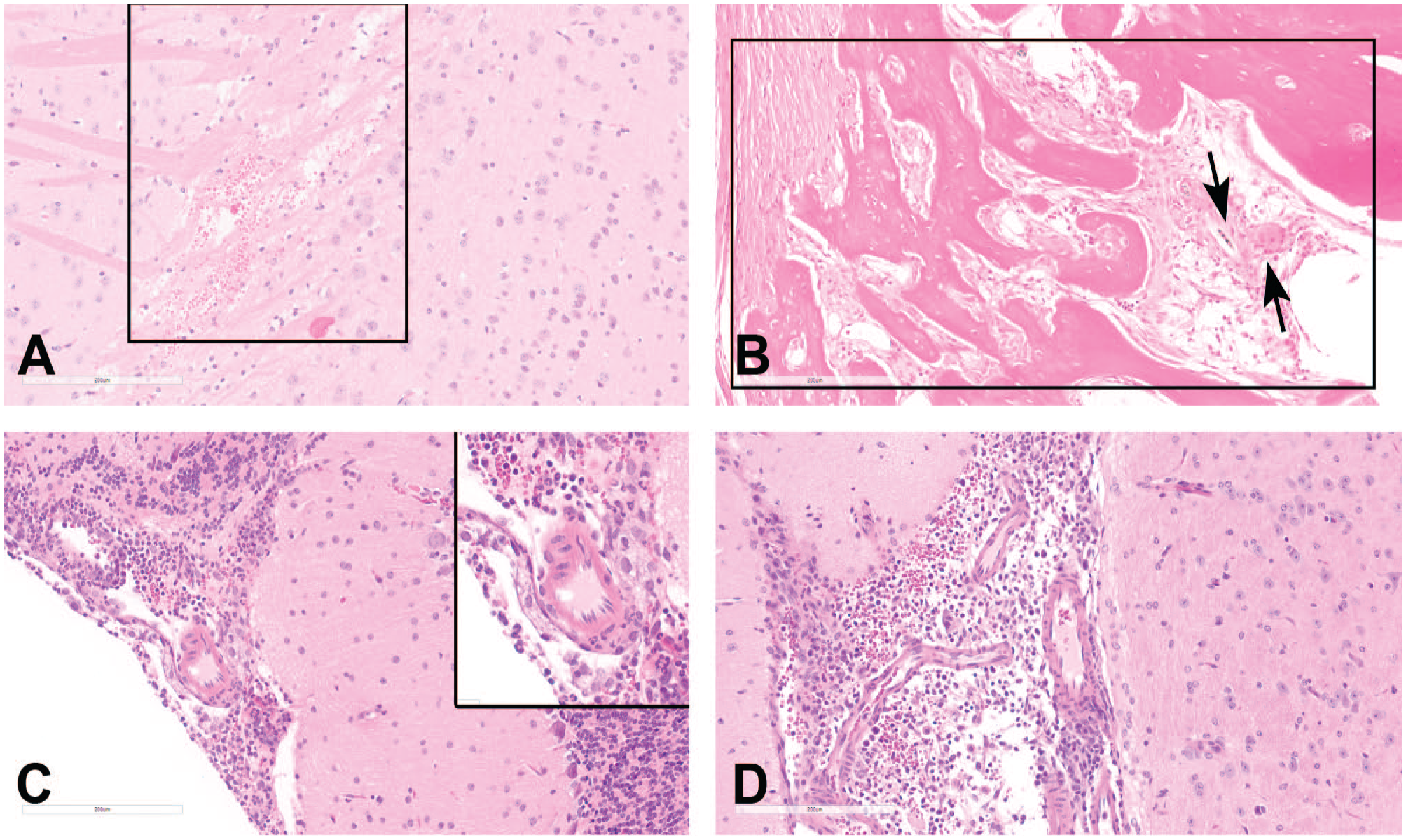
Findings related to intracerebroventricular (ICV) injection in the mouse. (A) Minimal periventricular edema and hemorrhage (boxed area) in the cerebral cortex. (B) Fibro-osseous proliferation in the meninges at the injection site. Note the deposition of new woven bone and fibrous connective tissue (boxed area) and a nearby free hair shaft surrounded by mononuclear cells, macrophages, and a multinucleated giant cell (black arrows). (C) and (D) Mixed cell inflammation and hemorrhage within the meninges of the cerebellum (2C and inset) and in the lateral ventricle (2D). H&E indicates hematoxylin and eosin.
Rats—Intrathecal Injection
In our rat IT studies, the brain is sectioned in coronal (cross) section. For spinal cord, transverse and longitudinal oblique sections of the cervical, thoracic, lumbar, and sacral (with cauda equina) segments as well as the IT injection site (close to the sacral segment) are collected. The DRG (with spinal nerve roots) from the cervical, thoracic, and lumbar segments as well as the injection site are also harvested. The sciatic nerve is retained unilaterally.
Procedure-related findings in CNS tissues of rats dosed IT (Table 3 and Figure 3A-D) are similar to those of mice dosed ICV (Table 2) in terms of types of findings, but the distributions of findings differ due to the disparate location of the injection sites. In the brain of rats dosed IT, the most common procedure-related microscopic findings in control rats are minimal mononuclear or mixed cell infiltrates in the meninges and/or perivascular spaces and foci of acute parenchymal and/or meningeal hemorrhage. Less often, nerve fiber degeneration of minimal severity within white matter tracts and randomly distributed foci of gliosis are noted. In the spinal cord of rats dosed IT, the most prevalent procedure-related finding in our studies has been minimal to occasionally mild, usually asymmetric, nerve fiber degeneration affecting the dorsal and/or lateral funiculi of the segments that are closest to the indwelling catheter (i.e., at the injection site itself as well as the nearby lumbar and sacral [cauda equina] levels). This change occurs at a lower incidence and reduced severity in more cranial segments and occasionally involves the cervical spinal cord. Other microscopic changes in the control rat spinal cord after repeat IT dosing include minimal mononuclear or mixed cell infiltrates at the lumbar and sacral segments, localized fibrosis of the meninges and connective tissue adjacent to the lumbar spinal cord, foreign body granulomas (characterized by macrophages and variable numbers of multinucleated giant cells) at the injection site, meningeal and/or parenchymal hemorrhage, and gliosis.
Incidence of common procedure-related microscopic findings in control rats after repeated intrathecal (IT) infusion.
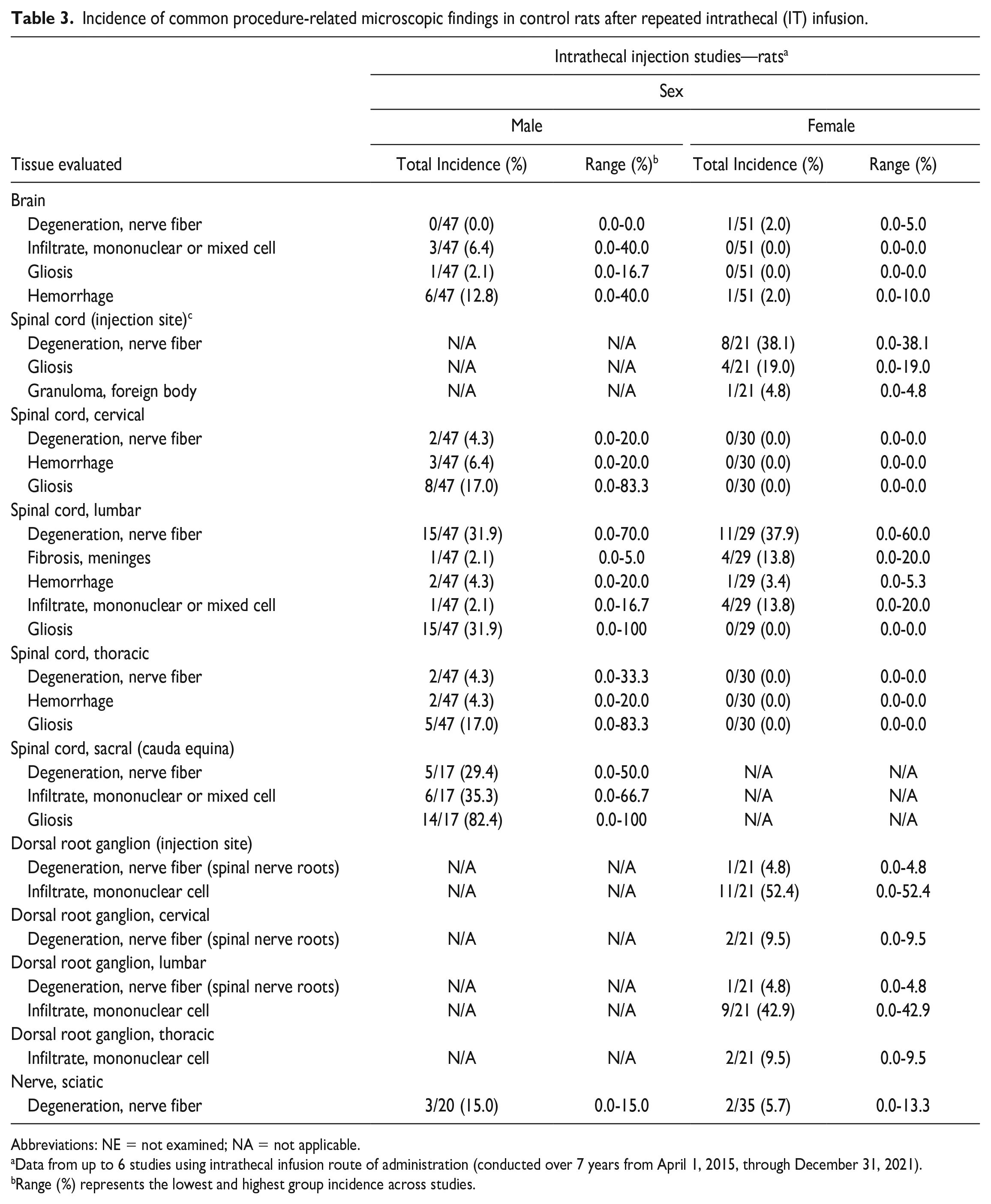
Abbreviations: NE = not examined; NA = not applicable.
Data from up to 6 studies using intrathecal infusion route of administration (conducted over 7 years from April 1, 2015, through December 31, 2021).
Range (%) represents the lowest and highest group incidence across studies.
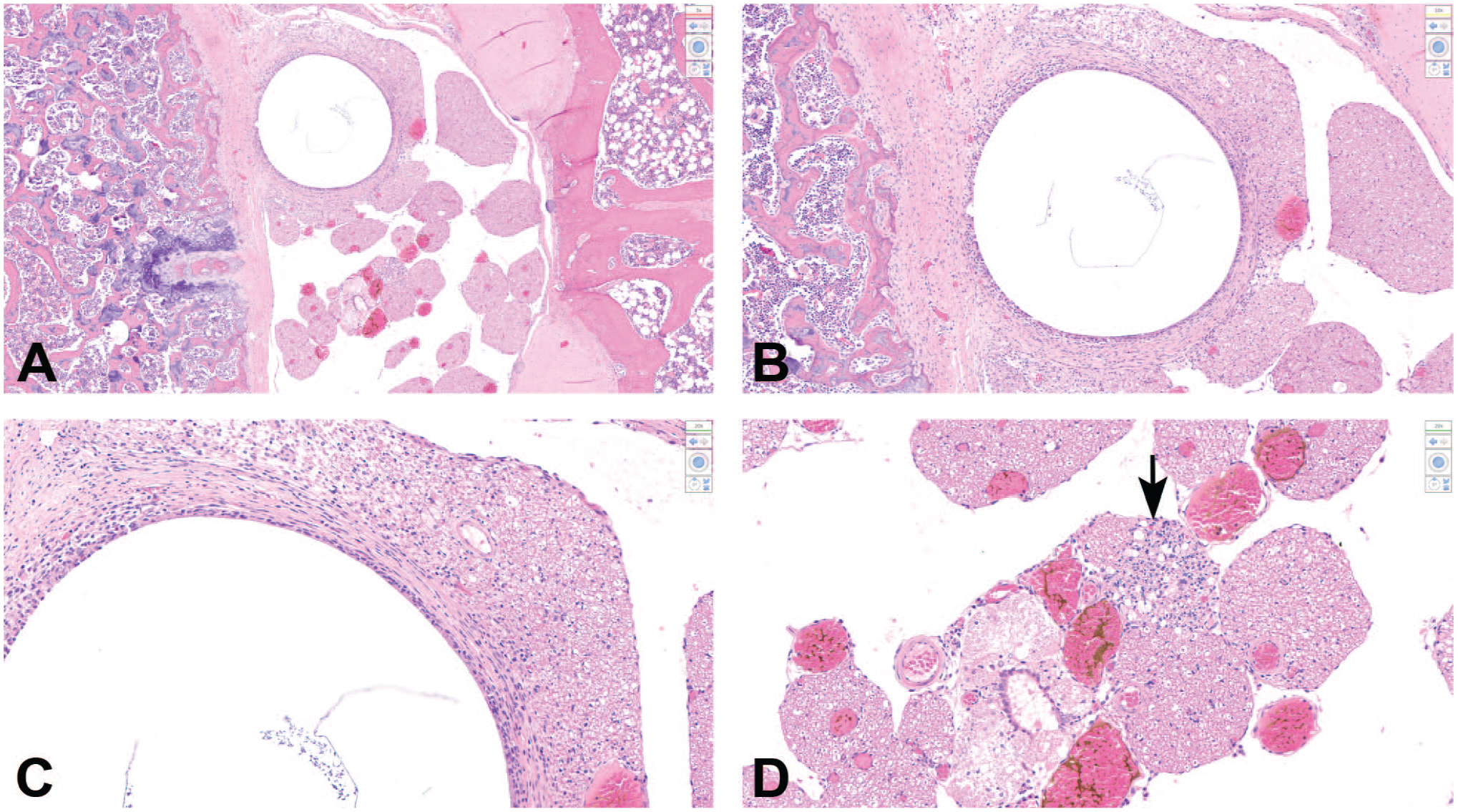
Findings related to intrathecal catheter insertion adjacent to the lumbosacral spinal cord of a rat. (A) A circular clear area representing the site where the intrathecal catheter was located is visible adjacent to multiple spinal nerve roots. (B) and (C) At higher magnification, the catheter site is surrounded by a ring of fibrosis representing a localized chronic reaction to the catheter. The dense fibrous tissue is continuous with the meninges and is compressing the adjacent nerve roots. (D) Minimal nerve fiber degeneration and glial (Schwann cell) proliferation (arrow) are apparent in the affected nerve roots; many regional capillaries are engorged with blood. H&E indicates hematoxylin and eosin.
Procedure-related findings in PNS tissues of rats dosed IT in the lumbar cistern (Table 3) are comparable with those in CNS tissues (Table 3). The most common microscopic findings in DRG are nerve fiber degeneration localized to the spinal nerve roots and mononuclear cell infiltrates scattered among the DRG neuron fields and/or in connective tissue surrounding ganglia. In general, the nerve fiber degeneration is not associated with concomitant DRG neuronal degeneration or necrosis. Nerve fiber degeneration may develop within the sciatic nerve and is usually of minimal severity.
Dogs—Intrathecal Injection
For IT studies in the dog, brain is trimmed in 7 coronal hemi-sections per current best practice recommendations 6 while spinal cord and DRG (including spinal nerve roots) are collected and sectioned as described above for the rat. The sampled nerve is usually the sciatic trunk.
Procedure-related findings in neural tissues of dogs dosed IT in the lumbar cistern (Table 4 and Figure 4A-C) are comparable with those identified in rats dosed IT (Table 3). In the dog brain, minimal mononuclear cell infiltrates have been observed in the meninges and/or perivascular spaces in control animals, which suggests a relationship to the procedure itself; however, these infiltrates do occur in dogs as a background finding,21,27 so the possibility that these foci are incidental cannot be ruled out. In spinal cord, procedure-related microscopic findings in dogs include minimal to occasionally mild nerve fiber degeneration in the dorsal and/or lateral funiculi, meningeal and/or parenchymal hemorrhage, fibrosis (affecting the meninges focally), and minimal mononuclear cell infiltrates within the meninges. These findings are typically limited to the cord segments closest to the site where the needle was inserted (i.e., at the injection site itself, the lumbar cord, and/or the sacral cord [cauda equina]), but they may extend as far forward as the thoracic spinal cord. In the DRG and associated spinal nerve roots, the most common findings are nerve fiber degeneration and glial (Schwann cell) hyperplasia in the nerve roots and mononuclear cell infiltrates in the DRG proper and rarely in the nerve roots. Importantly, the nerve fiber degeneration in spinal nerve roots tends to be restricted to the dorsal nerve roots of the lumbar and sacral segments while mononuclear cell infiltrates have been noted as far cranially as the cervical DRG.
Incidence of common procedure-related microscopic findings in control dogs after repeated intrathecal (IT) injection.
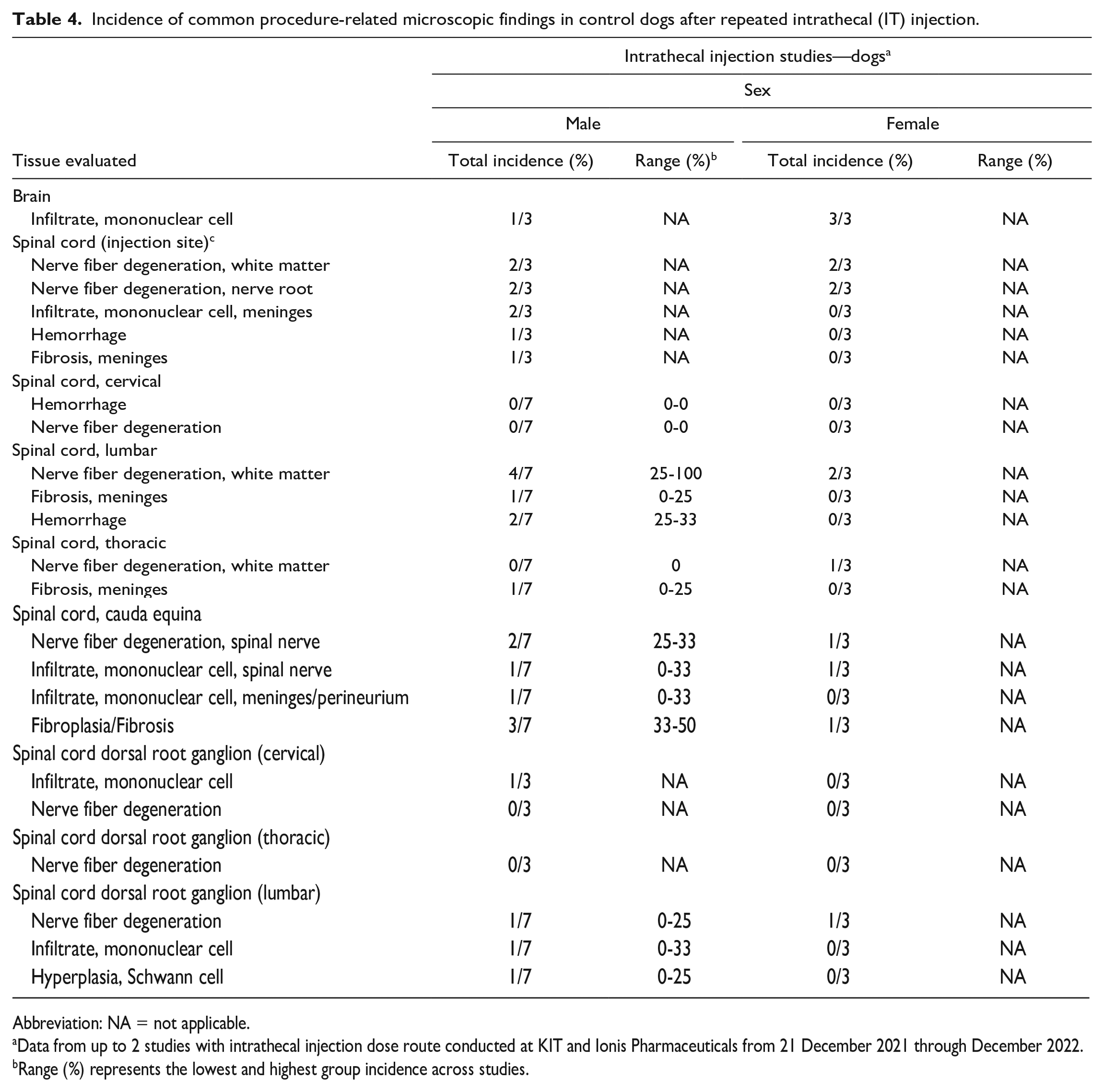
Abbreviation: NA = not applicable.
Data from up to 2 studies with intrathecal injection dose route conducted at KIT and Ionis Pharmaceuticals from 21 December 2021 through December 2022.
Range (%) represents the lowest and highest group incidence across studies.
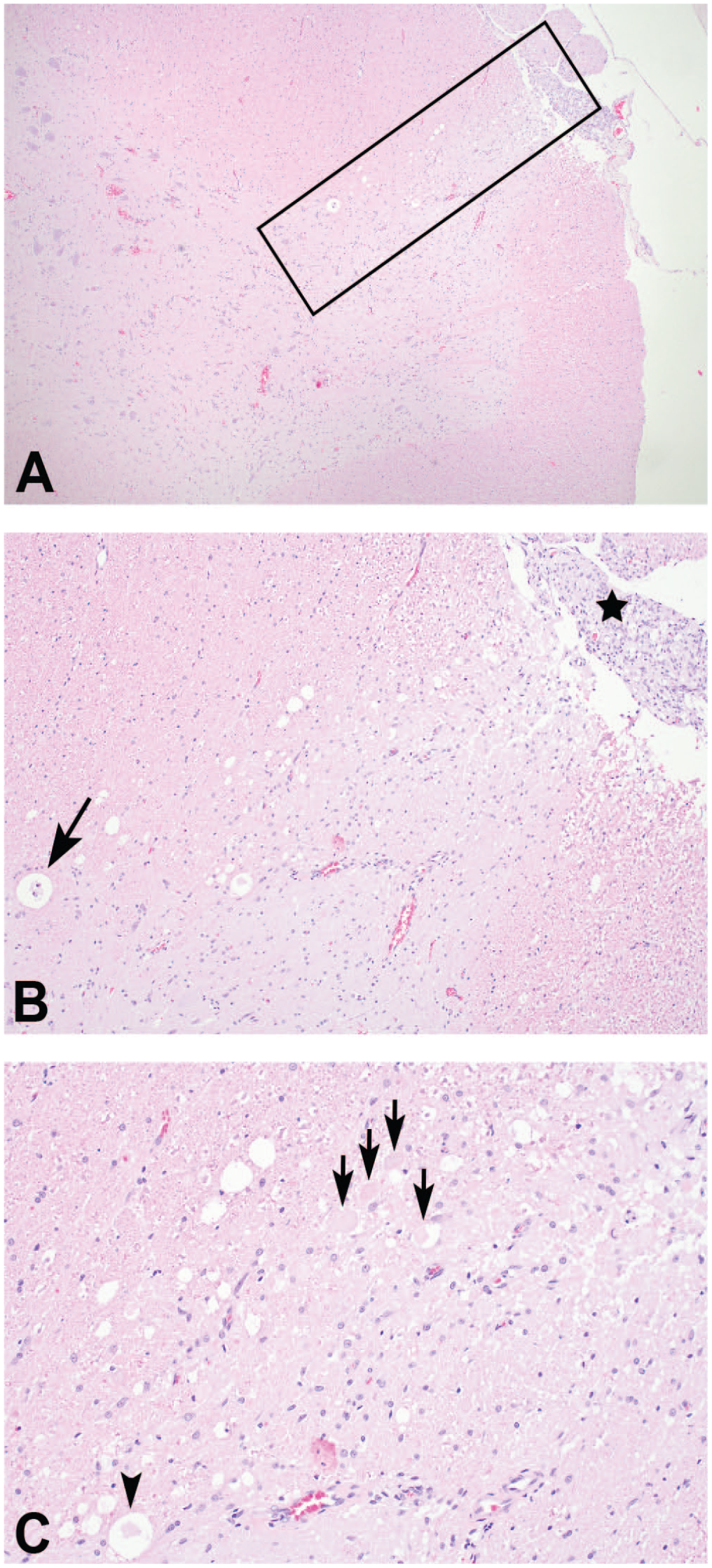
Nerve fiber degeneration in the lateral funiculus white matter of the lumbar spinal cord of a beagle dog given intermittent intrathecal injections in the lumbar cistern. (A) At low magnification, a distinct linear area of white matter vacuolation is noted in the lateral funiculus (boxed area). (B) At higher magnification, enlarged nerve fiber sheaths are observed with digestion chamber formation (arrow). Schwann cell proliferation (hyperplasia), as manifested by an increase in cellularity (star), is apparent in an associated spinal nerve root. (C) Swollen and/or fragmented hypereosinophilic axons (black and cyan arrows) are prominent within the area of nerve fiber degeneration and are accompanied by gliosis. H&E indicates hematoxylin and eosin.
Nonhuman Primates—Intrathecal Injection
For IT lumbar puncture bolus studies in NHPs, the brain is trimmed in 7 coronal hemi-sections consistent with current best practice recommendations. 6 The spinal cord and DRG (including spinal nerve roots) are collected and sectioned as described above for the rat and dog. In addition to the sciatic nerve trunk, the femoral, sural, and/or tibial nerves may be sampled as well.
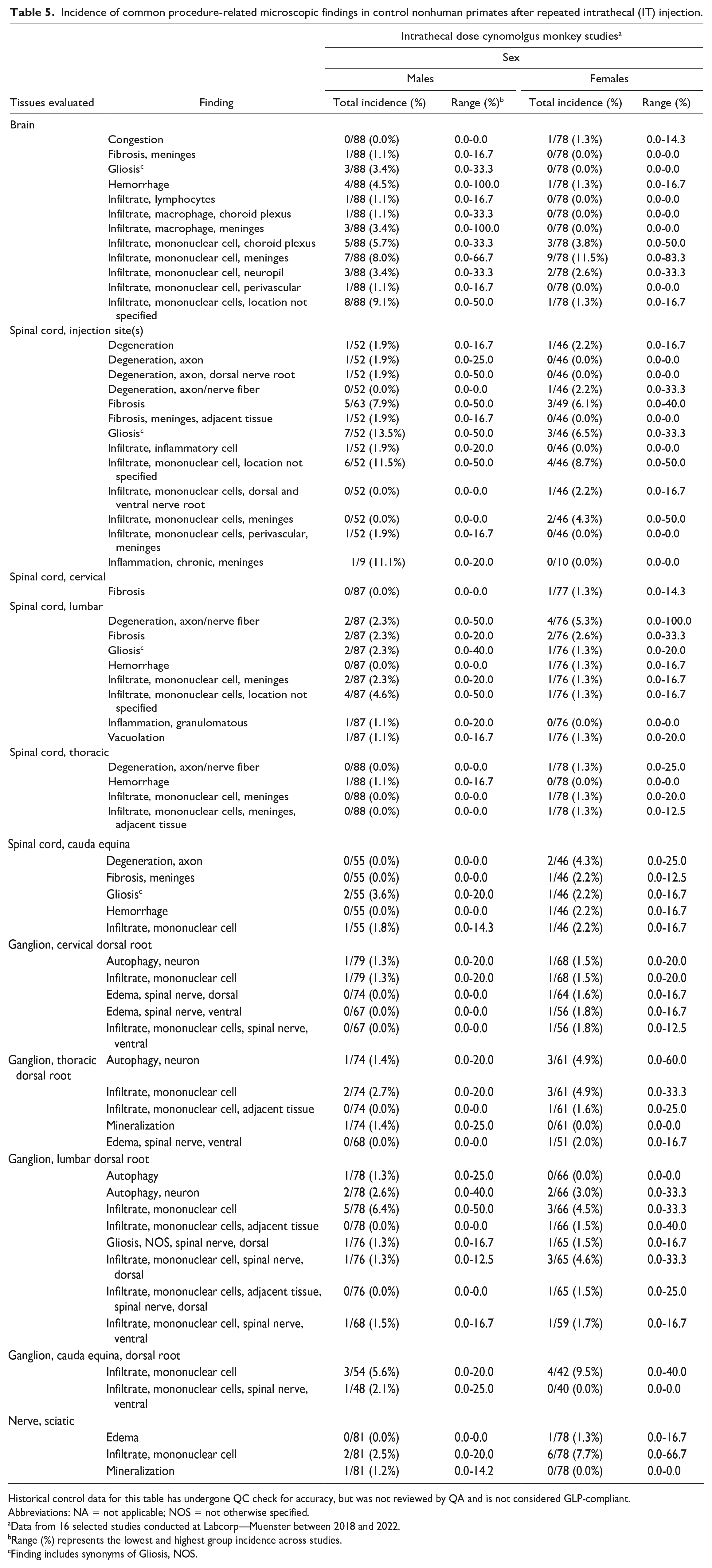
Procedure-related findings in neural tissues of NHPs dosed IT in the lumbar cistern are listed in Table 5, and examples are provided in Figure 5A-D. In the brain, leukocyte infiltrates within the meninges, choroid plexus, neuropil, and/or perivascular space are commonly noted. In our database, these infiltrates are most often classified as the “mononuclear cell” type (with lymphocyte- and/or macrophage-dominance specified in some cases). In our experience, such infiltrates may also be identified in brains from control NHPs from non-IT studies.10,21 Therefore, these minimal leukocyte infiltrates may represent, at least in part, a background finding instead of a purely procedure-related one. In contrast, changes not seen in our HCD for subcutaneously administered test articles and thus classified as procedure-related findings in the brain include meningeal and/or parenchymal hemorrhage, gliosis, congestion, and meningeal fibrosis. In the spinal cord, procedure-related changes include: mononuclear cell infiltrates in the meninges, perivascular spaces, the neuropil, and/or the associated spinal nerve roots; nerve fiber degeneration (sometimes listed as “axon degeneration”) in the white matter of the cord itself or in the associated nerve roots; gliosis; meningeal and/or parenchymal hemorrhage; and fibrosis (most often of the meninges), and chronic inflammation (at times described as “granulomatous”) in the meninges.
Incidence of common procedure-related microscopic findings in control nonhuman primates after repeated intrathecal (IT) injection.
Historical control data for this table has undergone QC check for accuracy, but was not reviewed by QA and is not considered GLP-compliant.
Abbreviations: NA = not applicable; NOS = not otherwise specified.
Data from 16 selected studies conducted at Labcorp—Muenster between 2018 and 2022.
Range (%) represents the lowest and highest group incidence across studies.
Finding includes synonyms of Gliosis, NOS.
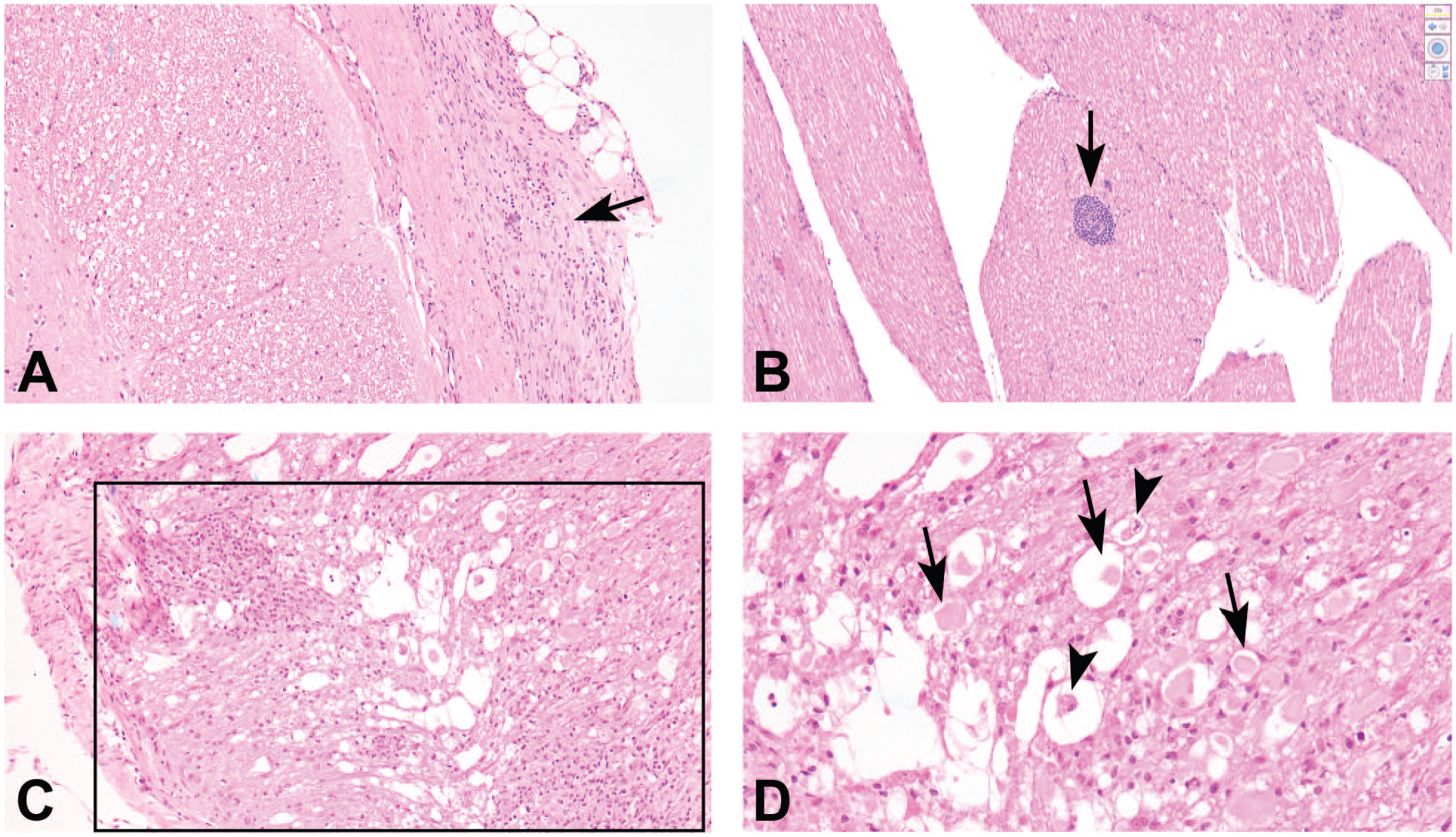
Procedure-related findings located at the intrathecal injection site (lumbar cistern) of a cynomolgus macaque. (A) Fibrosis associated with a mixed cell infiltrate (arrow) in the meninges. (B) Perivascular mononuclear cell infiltrate (arrow) in a nerve root of the cauda equina. (C) and (D) Nerve fiber degeneration with associated gliosis and mononuclear cell infiltrate (boxed area) in the spinal cord white matter. Swollen and/or fragmented hypereosinophilic axons (black arrows) and digestion chambers (black arrowheads) are present within the area of nerve fiber degeneration. H&E indicates hematoxylin and eosin.
As in the dog, these findings in NHPs are typically most prominent in and/or limited to the cord segments closest to the site where the needle was inserted (i.e., at the injection site itself, the lumbar cord, and/or the sacral cord [cauda equina]), although nerve fiber degeneration, hemorrhage, and mononuclear cell infiltrates in the meninges and/or adjacent connective tissue have been observed as far cranially as the thoracic cord. With more than 4500 IT bolus injections in more than 1000 cynomolgus monkeys, only 3 cases of trauma-related spinal cord necrosis have been observed. Therefore, lumbar spinal cord necrosis is a very rare effect and is presumed to be a mechanical complication of IT dosing in cynomolgus monkeys. 17
In DRG, neuronal autophagy, mononuclear cell infiltrates in the ganglia proper or the adjacent connective tissue, and mineralization have been noted. Autophagy has been well described as a common finding in dorsal root ganglia of NHPs8,12 and is considered a spontaneous incidental change rather than a procedure-related effect. Mononuclear cell infiltrates in the spinal nerve roots or adjacent connective tissue, edema, and gliosis have been described in the spinal nerve roots. In the sciatic nerve, we have noted mononuclear cell infiltrates, edema, and mineralization in our NHP studies. Similar changes have also been observed in the femoral, sural, and/or tibial nerves (unpublished data).
Conclusion
Direct CNS delivery has dramatically expanded the number of neurological diseases that can be effectively treated, including repeated administration of therapeutics that require intermittent administration (e.g., ASO technology). Patients have and will continue to benefit from treatments introduced via this route. Given the challenges of this delivery method, however, much effort has been directed toward innovation in ASO chemistry that will allow for less frequent dosing in the clinic 1 as well as conjugation strategies which would allow systemically administered ASOs to cross the BBB. 23
Direct CNS delivery to nonclinical species is generally well-tolerated, but the procedure itself can induce a number of in-life functional and histopathologic effects that must be well understood by nonclinical safety scientists so that interpretation of study findings as procedure- or test article-related can be determined with a high degree of confidence. The most common procedure-related functional effects are death in mice and transient absence of spinal reflexes in rats, dogs, and NHPs. In the mouse, procedure-related histopathologic changes are limited to the brain (i.e., the ICV injection site) and, as expected, are concentrated in the injected rather than the noninjected hemisphere. Procedure-related histopathologic findings in the rat, dog, and NHP can be seen in the brain, spinal cord, DRG and associated spinal nerve roots, and the sciatic nerve and tend to be more severe and/or have a higher incidence in the spinal cord segments, DRGs, and spinal nerve roots from or near the IT injection site. These procedure-related findings are unavoidable and represent a key confounder for study pathologists tasked with assessing the safety of test articles delivered directly into the CNS.
Footnotes
Acknowledgements
The authors acknowledge Dr. Scott Henry for sharing his expertise and for critical review of this manuscript and Ms. Tracy Reigle for her assistance in preparing the figures for this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
