Abstract
Gold nanoparticles (AuNPs) have been widely used in many applications. Their usage as drug delivery vehicles has also gained considerable attention due to their chemical and optical properties as well as their good biocompatibility. The present study was conducted to evaluate the efficiency of AuNPs in enhancing the cytotoxic and apoptotic induction activity of lantadene A (LA), separated from
Keywords
Introduction
Cancer is one of the most challenging diseases worldwide. Regardless of the progress made in the discovery and development of cancer therapeutics, cancer remains the second cause of death worldwide, after cardiovascular disease. 1 Chemotherapy is the most common treatment prescribed for cancer aside from other limited treatments, such as radiation, immunotherapy, and surgery. 2 However, these methods cause numerous negative side effects, which in some cases can be fatal.. 3 Apart from the chemotherapy effects on hair follicles, oral mucosal cells, and bone marrow, the treatment has gastrointestinal, cardiovascular, and neurological toxicity. 4 Another obstacle facing chemotherapy is the development of cancer drug resistance that limits the use of these therapeutics. 5 Further, a major disadvantage to chemotherapeutics is the absence of specificity in targeting cancerous cells, which means normal cells are also affected. 6 As such, there is an urgent need for new, effective, and better-tolerated agents with chemotherapeutic activity that deliver these agents specifically to tumor cells.
Tumors, by the nature of their rapid replication and high production of proangiogenic growth factors, have blood vessels that differ from normal cells due to large gaps in their epithelium. 7 These gaps give rise to the enhanced permeability and retention effect (EPR). 8 Nanotechnology used this feature, along with the defective lymphatic function in tumors, to deliver nanoscale structures and drug carriers specifically to tumor cells and allow their accumulation in the tumor interstitium, while avoiding normal cells to ensure minimum possible side effects. 9
Nanotechnology is one of the most important technologies utilized in different areas of biology, pharmaceuticals, medicine, and technology. 10 When the size of materials is reduced to nanoscale, particles gain distinctive properties that set them apart from their bulk equivalents. 11 Gold nanoparticles (AuNPs) have shown notable advantages over other nanoscale counterparts in drug delivery, gene therapy, and other biomedical applications due to their unique physical and chemical attributes such as their small size, high surface area to volume ratio, stability, high dispersity, biocompatibility, and unique thermal and optical properties. In addition, various strategies were used for preparation of AuNPs within nanoscale range (5-90 nm) like using plant extracts as reducing agents, thermosensitive, γ-irradiation, and others. 12,13 Many chemical and physical methods have been utilized for the synthesis of nanoparticles, which involve complex and expensive procedures and employ toxic chemicals incompatible with medical applications. 14 Green synthesis of nanoparticles, where plant extracts act as stabilizers, has proven to be of extreme significance as a simple, inexpensive, and eco-friendly process. This process synthesizes nanoparticles with a certain degree of control over their size, shape, and absorbance through factors of temperature, pH, and plant extract concentration. 15
For centuries, medicinal plants have been the source of a large number of biologically active compounds and have been used extensively to treat different diseases, including cancer.
16,17
Lantadenes (lantadene A, B, C, and D) are pentacyclic triterpenoids found naturally in different parts of
Methods
Materials
Chemicals including gold (III) chloride trihydrate (99.9%; HAuCl4.3H2O), RPMI-1640, trypsin, DNase I, and fetal bovine serum were purchased from Sigma-Aldrich. Silica gel was obtained from Thomas Baker, while activated charcoal was sourced from M and B. Rehmannic acid (Lantadene A Standard) was ordered from ChemFaces. The cell cytotoxicity MTT kit was purchased from Intron Biotech, and FlowTACS apoptosis detection kit was purchased from Trevigen, Inc. Kits used for real-time polymerase chain reaction (RT-PCR), TRIzol Plus RNA purification kit and RevertAid RT kit, were obtained from ThermoScientific. The designed primer used for quantitative RT-PCR was supplied from BioNeer.
Cell Line and Cell Culture
A human breast tumor cell line MCF-7 (Passage No. 23) was thankfully provided by the Al-Nahrain Biotechnology Center of Al-Nharin University. Cells were subcultured using RPMI-1640 media containing 10% fetal bovine serum and supplemented with 103 IU 100−1 mL penicillin G. Streptomycin at 0.001 g 100−1 mL (Ajanta Pharm) was used to prevent microbial contamination. Cells were incubated at 37 °C in a humidified incubator containing 5% CO2. For growth initiation, MCF-7 cells were seeded into T 25-cm2 tissue culture flasks (ThermoScientific) at density 2 × 104 cells mL−1. When reached exponential growing phase (36-48 hours), the cells were harvested after brief trypsinization (50 mg.mL−1 of trypsin) and were seeded at desired concentration. 25
Extraction of LA From L Camara
Leaves of
Purification of LA
Fifty grams of silica gel (60-120 mesh) was activated in an oven at 110 °C for 1 hour. The slurry was prepared by mixing the activated silica gel with a methanol–water mixture (1:7 vol/vol) and packed in a glass column (2.6 cm × 50 cm). The column was equilibrated and washed for 24 hours with methanol–water mixture. The partially purified lantadene (1.7 g) was dissolved in 40 mL of the mixture and concentrated in oven at 55 °C to reach a final volume of 8 mL. The lantadene solution was applied to the column and eluted (flow rate 5 mL 4 min−1) with 250 mL of methanol–water mixture. 26
Detection of LA by Thin-Layer Chromatography and High-Performance Liquid Chromatography
Fractions obtained from column were monitored by a procedure described by Sharma and Dawra, 21 with a minor modification. Ready to use silica gel G plates (200 × 200 mm) were activated at 110 °C for 1 hour before starting the experiment. Aliquots of 10 µL of both LA standard and the sample were spotted on the chromatograms and were incubated in a thin-layer chromatography (TLC) glass jar (250 × 250 × 120 mm) containing a modified ratio of the solvent system (Hexane 60 mL, methanol 35 mL, and ethyl acetate 5 mL). After the solvent reached the top of the chromatograms, the plates were taken out and left to air dry. Liebermann-Burchard reagent was sprayed over the plates and left to air dry for 15 minutes. The sprayed plates were incubated at 110 °C for 10 minutes. Fractions positive for LA were pooled up and dried in oven at 55 °C to obtain pure LA. Final purity was checked using high-performance liquid chromatography (HPLC) system. The analysis was carried out using a Nova-Pak C-18 (250 × 4.6 mm) column supplied with a 490E multichannel detector, a Millennium data processor (Shimadzu), and a Rheodyne injector with 20-µL loop. The mobile phase, methanol–acetonitrile–water–acetic acid (68:20:12:0.01 vol/vol), was used at a flow rate of l mL min−1. Aliquots of 100 µL of both sample and LA standard were injected, and peaks were detected at wavelength 210 nm.
Synthesis of Lantadene A-AuNPs
The purified LA (50 mg) was dissolved in 50 mL deionized water with stirring using a magnetic stirrer to obtain a yellowish stock solution. An aliquot of 0.5 mL HAuCl4 solution (3 mg·mL-1) was added drop wise to the LA solution with constant stirring at 100 °C for 30 minutes. 27
Characterization of LA-AuNPs
Ultraviolet–Visible absorption spectrophotometry
The bioreduction of the Au3+ to Au0 was monitored by a ultraviolet–visible (UV–Vis) absorption spectrophotometry (V670 JASCO). The absorption spectrum was acquired in the range of 400 to 900 nm for both LA and LA-AuNPs. The absorption spectrum for deionized water was used as the background reference.
Atomic force microscope
Surface morphology and average particle size (20 measurements/sample) of the nanoparticles were analyzed using NTEGRA (NT-MDT) device. Samples were prepared by casting drops of LA-AuNPs solution onto glass slides and were left to air dry at room temperature before analysis.
Scanning electron microscope, particle size, and zeta potential
Scanning electron microscope (SEM) was used to examine the morphology of LA and LA-AuNPs. The SEM images were acquired using JSM-7600F Schottky Field Emission Scanning Electron Microscope (Jeol), while particle size, size distribution, and zeta potential were measured by using ZetaPLUS analyzer (Brookhaven Instruments). Samples were first diluted in ddH2O, filtered using 0.22-µm Millipore filter unit, and then samples were placed in ultrasonic bath and subjected to ultrasonic force for 5 minutes and finally analyzed at room temperature.
Cytotoxicity Test (MTT Assay)
Gold NPs, LA, and LA-AuNPs were examined for their cytotoxic effect against MCF-7 cells using MTT colorimetric assay. MCF-7 cells (1 × 104 to 1 × 106) were grown in 96 flat well microtiter plates in a final volume of 200 µL culture medium per well at 37 °C, 5% CO2 for 24 hours. After incubation, the medium was removed and 2-fold serial dilutions of the desired compound (25, 50, 100, 200, and 400 µg·mL−1) were added to the wells. Triplicates were used per concentration as well as the controls (cells treated with serum-free medium). Plates were incubated at 37 °C, 5% CO2 for 24 hours. Later, 10 µL of MTT solution was added to each well and the plates were further incubated at 37 °C, 5% CO2 for 4 hours. The media was carefully removed, and 100 µL of solubilization solution was added into each well and incubated for 5 minutes. Formazan formation was determined by measuring the absorbance at 575 nm using an enzyme-linked immunosorbent assay microplate reader (Bio-Rad).
Apoptosis Assay (TUNEL)
A flow cytometric detection of DNA fragmentation and cell apoptosis was examined using a streptavidin–FITC conjugate FlowTACS kit to compare the apoptotic effect of purified LA and LA-AuNPs against MCF-7 cells. Cells were seeded at density 2 × 105 in T 75-cm2 flasks (ThermoScientific). The flasks were incubated at 37 °C and 5% CO2 for 24 hours. After incubation, the medium was removed, and the cells were treated with AuNPs, LA, and LA-AuNPs (100 µg·mL−1 for each treatment) and doxorubicin (1 µg·mL−1) for 48 hours. After incubation, the cells (1 × 106) were centrifuged at 1,000 ×
RNA Extraction
Following MCF-7 cells subculture and treatment with AuNPs, LA, LA-AuNPs (both at 100 µg·mL−1), and doxorubicin (1 µg·mL−1) for 48 hours, total cellular RNA was extracted using TRIzol Plus kit by adopting the manufacturer’s protocol. RNA molecules were eluted in RNase-free water, and DNA contaminants were removed with DNase I treatment at 37 °C. The purity and concentration of the extracted RNA were determined by measuring the absorbance at 260/280 nm using Nano-drop spectrophotometer (ThermoFisher). RNA samples were stored at −80 °C until use.
Complementary DNA Synthesis, qRT-PCR Protocol, and Data Analysis
Reverse transcription of total RNA to complementary DNA (cDNA) was achieved using RevertAid RT kit following the manufacturer’s step-by-step procedure. The resulting cDNA was used to detect the expression of
Primers Used for qRT-PCR.

Statistical Analysis
All statistical analyses were performed using Graph Pad Prism version 6 (Graph Pad Software Inc). A one-way analysis of variance (Tukey post hoc test) was performed to test if differences between the groups were significant or not. Statistical significance was defined as *
Results
Isolation and Purification of LA
Out of 100 g of
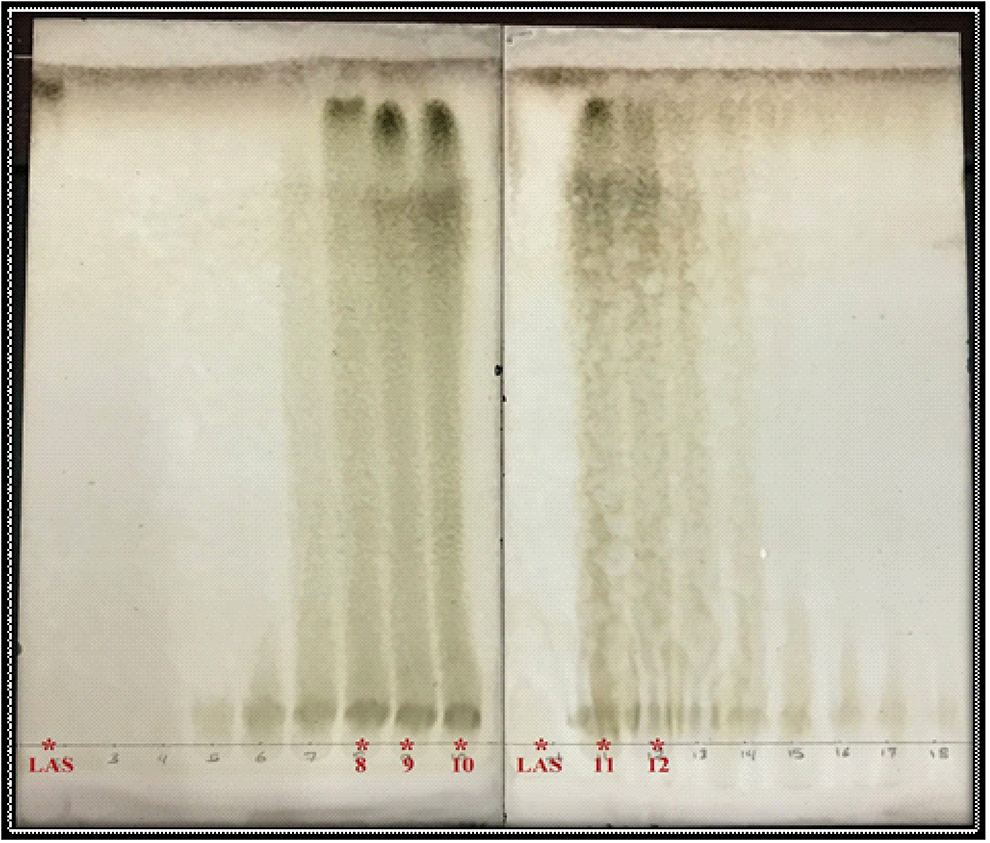
Thin-layer chromatography (TLC) profile of fractions 8, 9, 10, 11, and 12 on silica gel G plates (200 × 200 mm) as stationary phase and hexane: methanol: ethyl acetate (60:35:5 vol/vol) as a mobile phase. Fractions were compared with LA standard (LAS).
The purity and concentration of LA was detected by HPLC. Figure 2 and Table 2 show 3 minor peaks and 1 major peak. The major peak was detected at a retention time of 10.543 minutes with a peak area of 21,497.522 mAU*s. Based on the results, the major peak from the unknown sample was identified as LA by comparing the retention time of the standard with the retention time of the major peak. The calculated concentration of purified LA was 1.8 mg·g−1 with an overall area percentage of 93% depending on equation of Argekar and Powar. 28
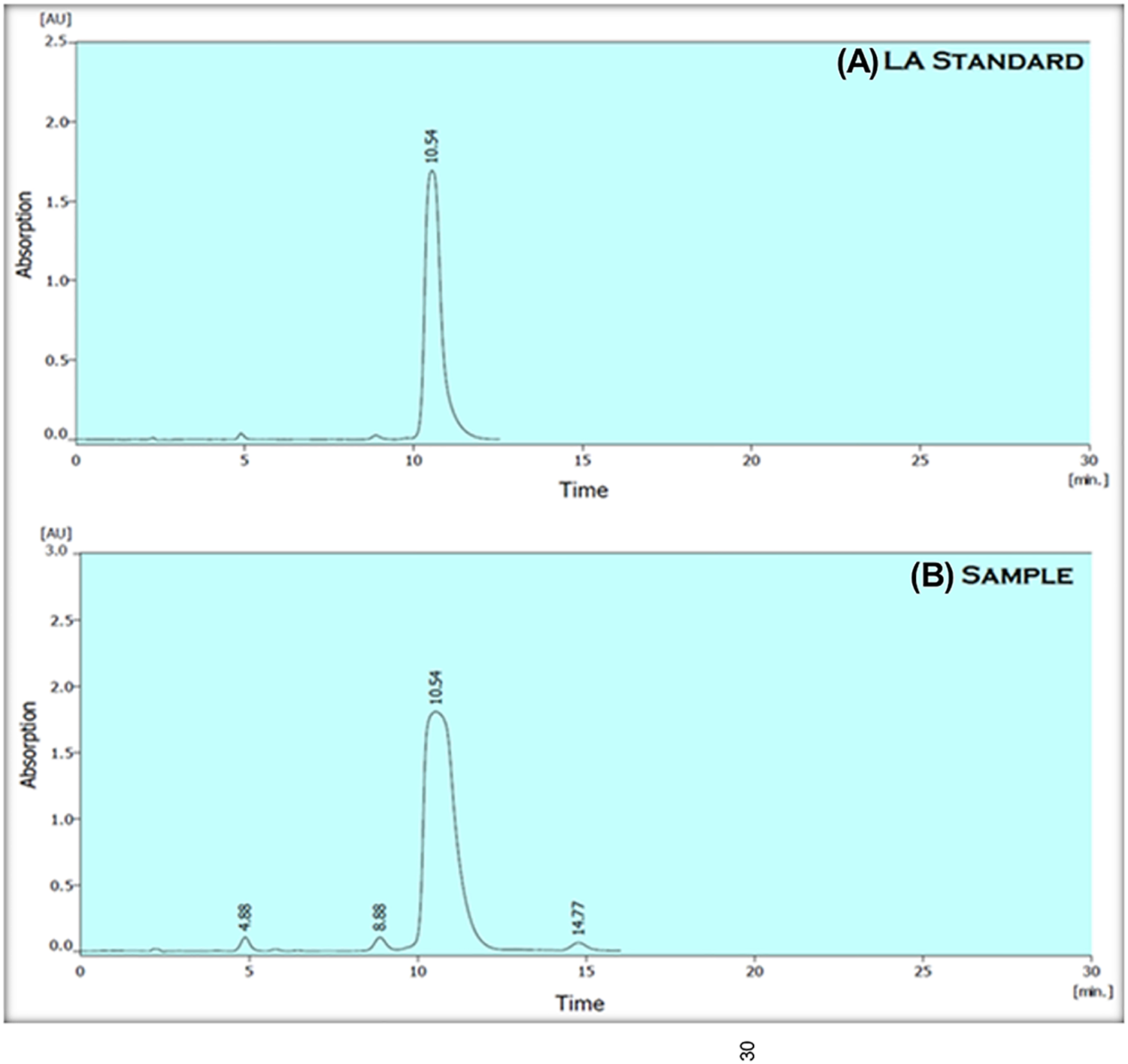
High-performance liquid chromatography (HPLC) chromatogram at 210 nm for (A) LA standard and (B) the test sample.
Quantitative Analysis of Peaks Obtained From Purified LA Eluted From HPLC and Its Respective LA Standard.
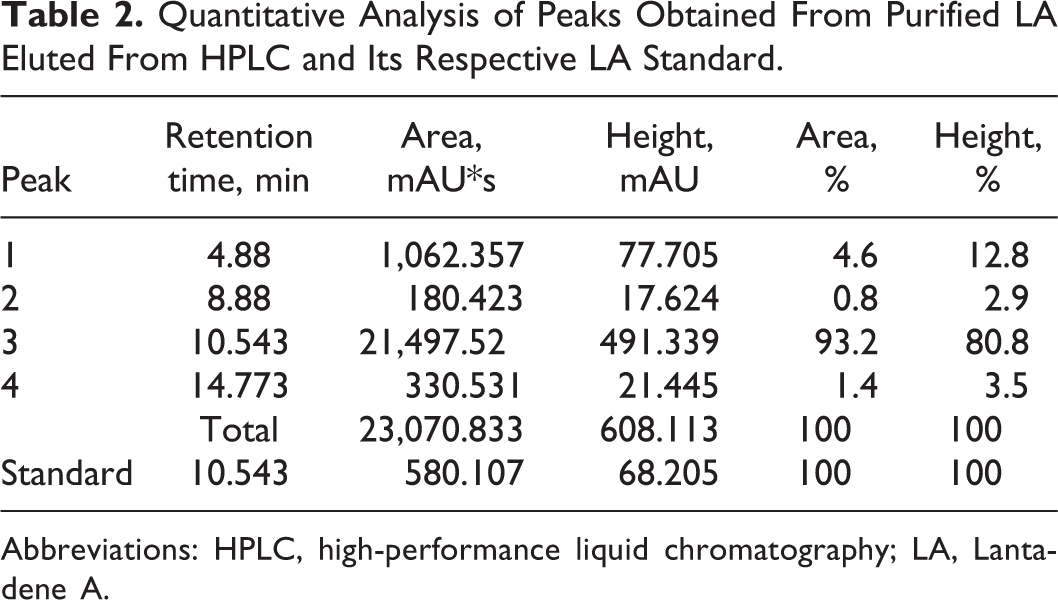
Abbreviations: HPLC, high-performance liquid chromatography; LA, Lantadene A.
Synthesis and Characterization of LA-AuNPs
Following the drop wise addition of Au3+ solution to 50 mg of purified LA solution, the color was transformed from pale yellow to bluish gray (Figure 3). Such a change was considered an indicator for the formation of AuNPs. The color of LA solution began changing seconds after the addition of Au3+ solution.

Synthesis of Lantadene A gold nanoparticles (LA-AuNPs). (A) LA before adding HAuCl4. (B) LA-AuNPs after the completion of the reaction.
The UV–Vis spectroscopy was used to evaluate the successful reduction of Au3+ to Au0 to detect the optical properties of LA-AuNPs and to identify the position of the localized surface plasmon resonance (LSPR) band. This band is known to have a characteristic maximum for Au within the range of 520 to 580 nm. An LSPR absorption band was not noticed for LA (blue line), while the absorption spectrum for LA-AuNPs (red line) included a wide band with 2 detected peaks (Figure 4). One peak at 539 nm represented the LSPR band, a characteristic of spherical nanoparticles, and was associated with a single plasmon resonance frequency. The second peak, at 604 nm, was an indicative of 2 plasmon resonance frequencies (539 nm and 604 nm), which are representative of AuNPs aggregation 29 or the formation of different shapes of nanoparticles, such as triangles or rods. 27
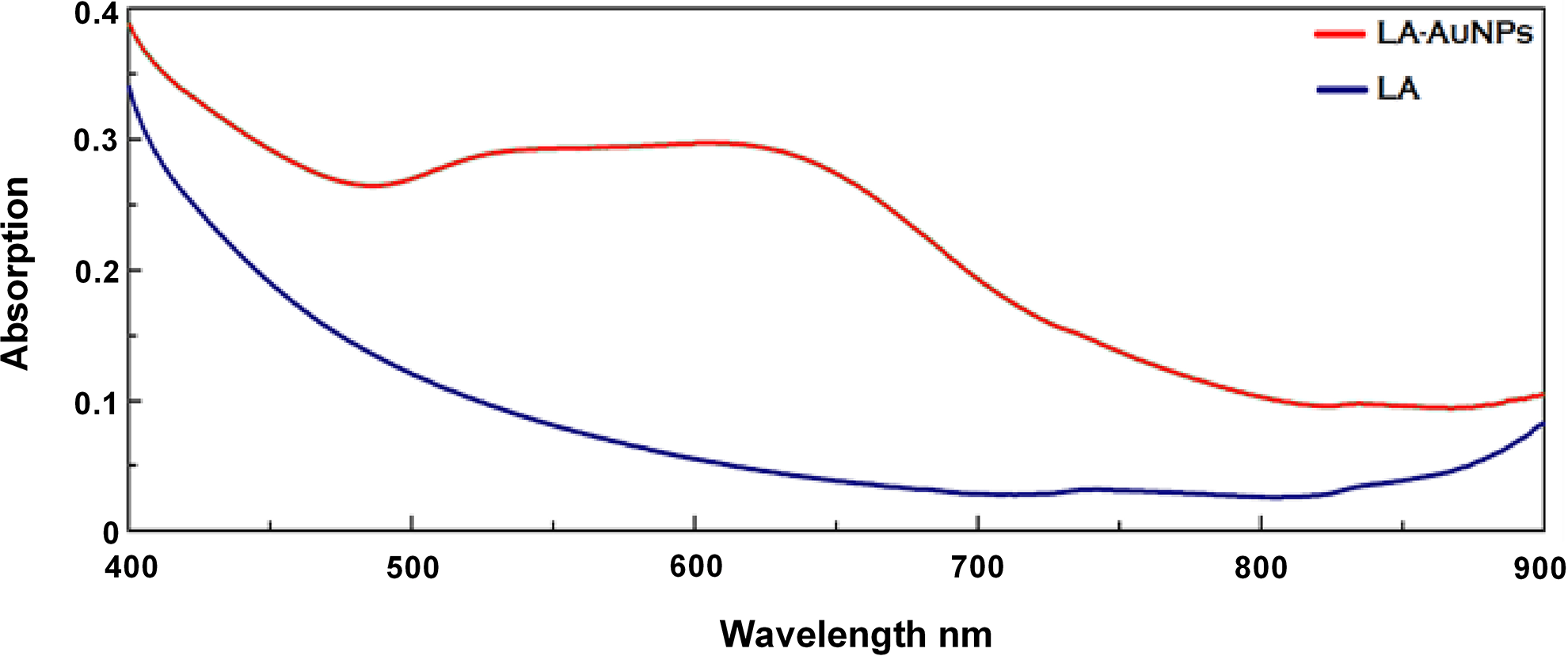
Ultraviolet–visible (UV–Vis) spectrograph of Lantadene A (LA; blue line) and Lantadene A gold nanoparticles (LA-AuNPs; red line) measured 24 hours after the completion of the reaction. Lantadene A-AuNPs were produced by mixing 0.5 mL of 3,000 mg·L−1 of HAuCl4 solution and aqueous LA stock solution (50 mg/50 mL) at 100 °C.
According to atomic force microscope (AFM) images (Figure 5), the average particle size of LA-AuNPs was 32.78 ± 5.29 nm as well as some aggregations, which could be the cause of the second plasmonic band. On the other hand, SEM was conducted to determine the surface morphology of LA and LA-AuNPs. In Figure 6A, LA particles showed an ill-defined morphology and large particle size, while the image for LA-AuNPs (Figure 6B) clearly exhibited the presence of small, mostly spherical particles of different sizes within the nanometer scale with an average nanoparticle size of 55.88 ± 14.95 nm. Finally, the mean particle size and distribution was analyzed, and LA-AuNPs had an average particle size of 35.62 ± 4.71 nm with a polydispersity of 0.145 ± 0.039; on the other hand, the zeta potential of LA-AuNPs exhibited a surface-negative charge with value of −33.04 ± 2.39 mV.
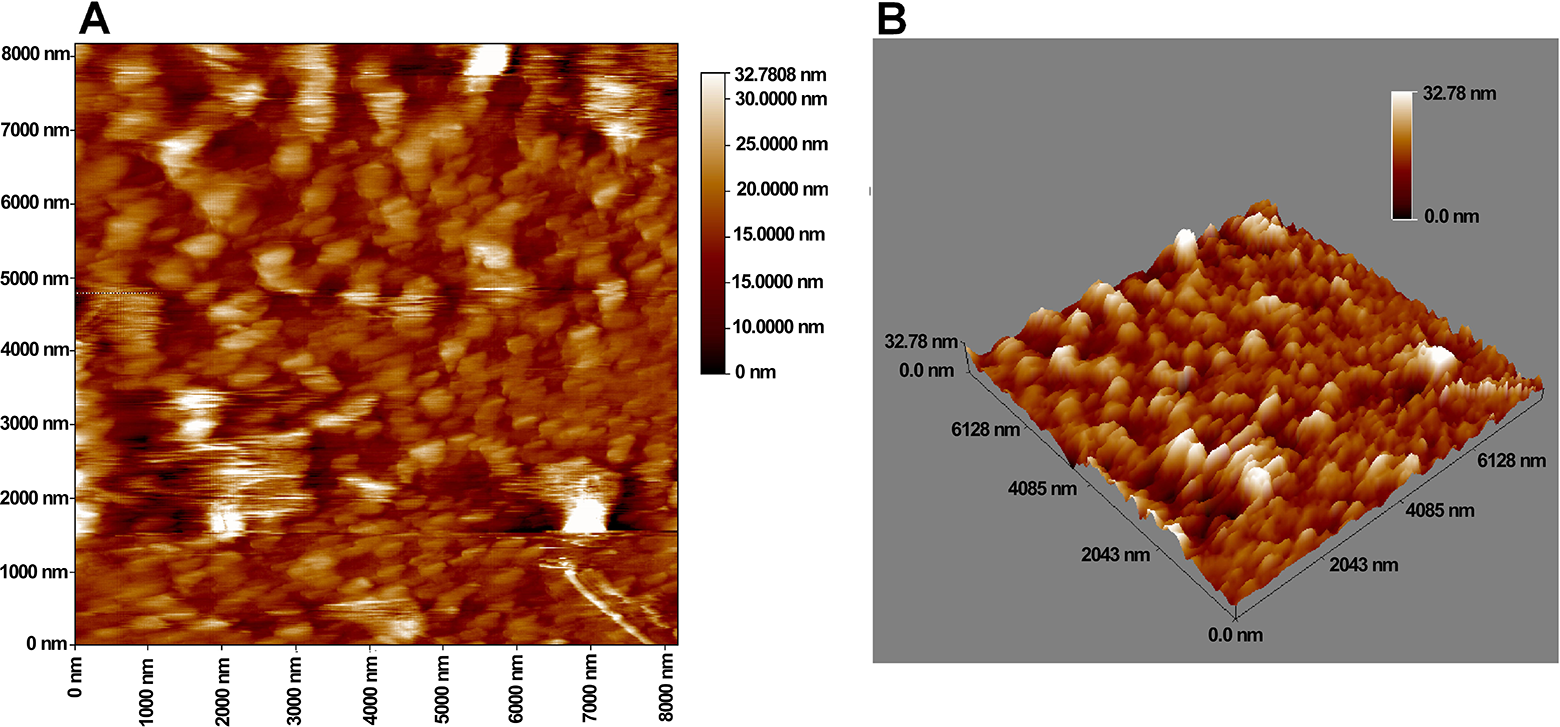
Atomic force microscope (AFM) images for Lantadene A gold nanoparticles (LA-AuNPs) in (A) 2 dimensions display and (B) 3 dimensions display at 2-µm magnification in air at room temperature.

Scanning electron microscopy (SEM) image for (A) Lantadene A (LA) and (B) Lantadene A gold nanoparticles (LA-AuNPs).
Cytotoxicity of LA and LA-AuNPs Against MCF-7 Cell Lines In Vitro
MTT experiments (Figure 7) revealed that the growth inhibition of MCF-7 cells after treatment with LA-AuNPs was found to be in the range of 6.78% ± 0.68% to 77.69% ± 4.28%. Moreover, both AuNPs and LA exhibited an inhibition range of 4.66% ± 1.40% to 47.19% ± 2.24% and 7.40% ± 2.17% to 52.40% ± 3.64%, respectively. All compounds reduced cell viability in a dose-dependent manner. However, the cytotoxic effect of LA-AuNPs was significantly higher than that of free AuNPs and LA with an IC50 of 64.39, 222.3, and 158.20µg·mL−1, respectively.
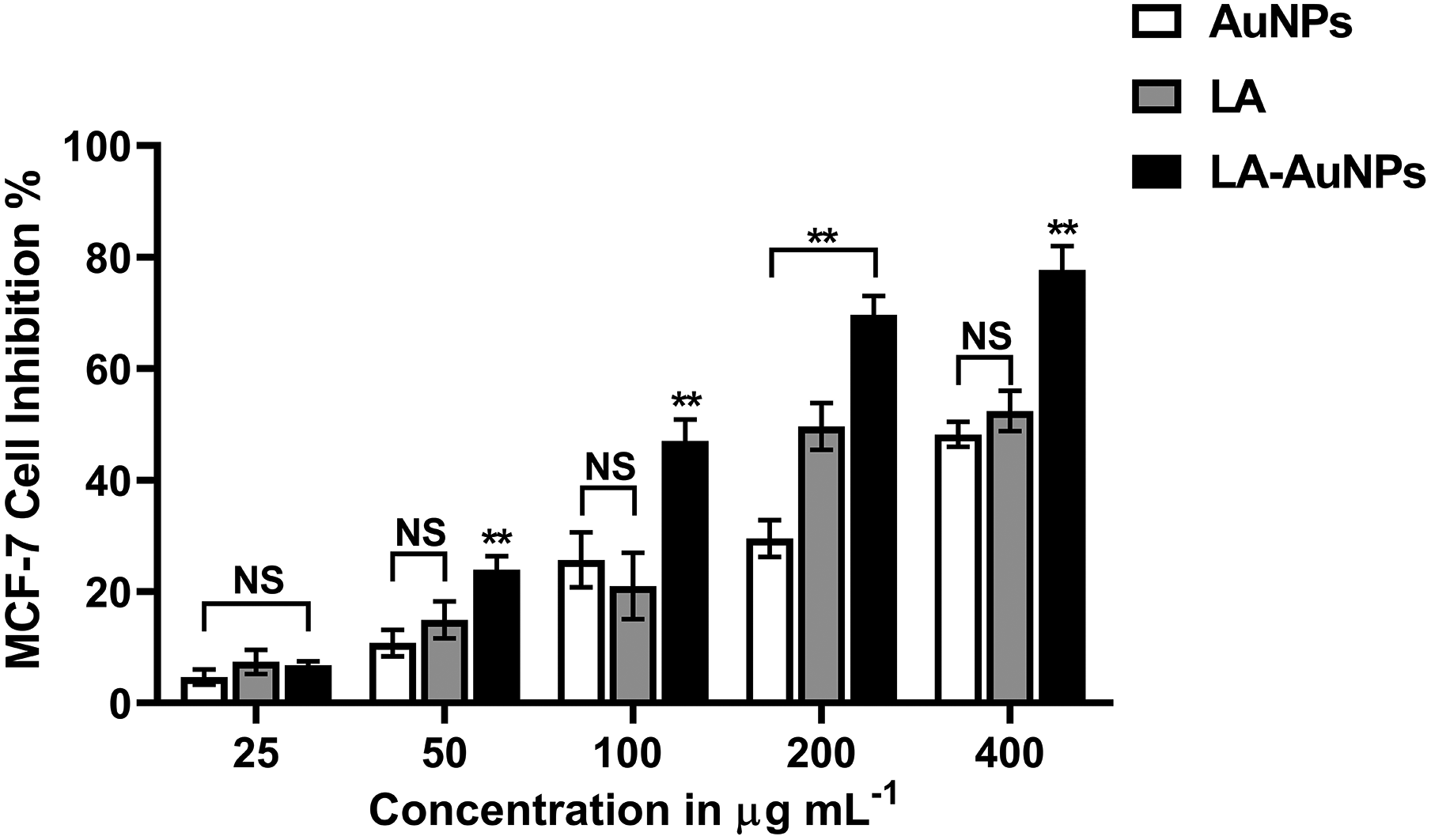
Dose-dependent cytotoxic effect of AuNPs, Lantadene A (LA), and Lantadene A gold nanoparticles (LA-AuNPs) against MCF-7 cell line. NS indicates nonsignificant, **
DNA Fragmentation Assay (TUNEL Assay)
TUNEL assay results for LA and LA-AuNPs are demonstrated in Figure 8. When MCF-7 cells were cultivated with 100 µg·mL−1 LA-AuNPs for 48 hours, TUNEL-positive cells significantly (
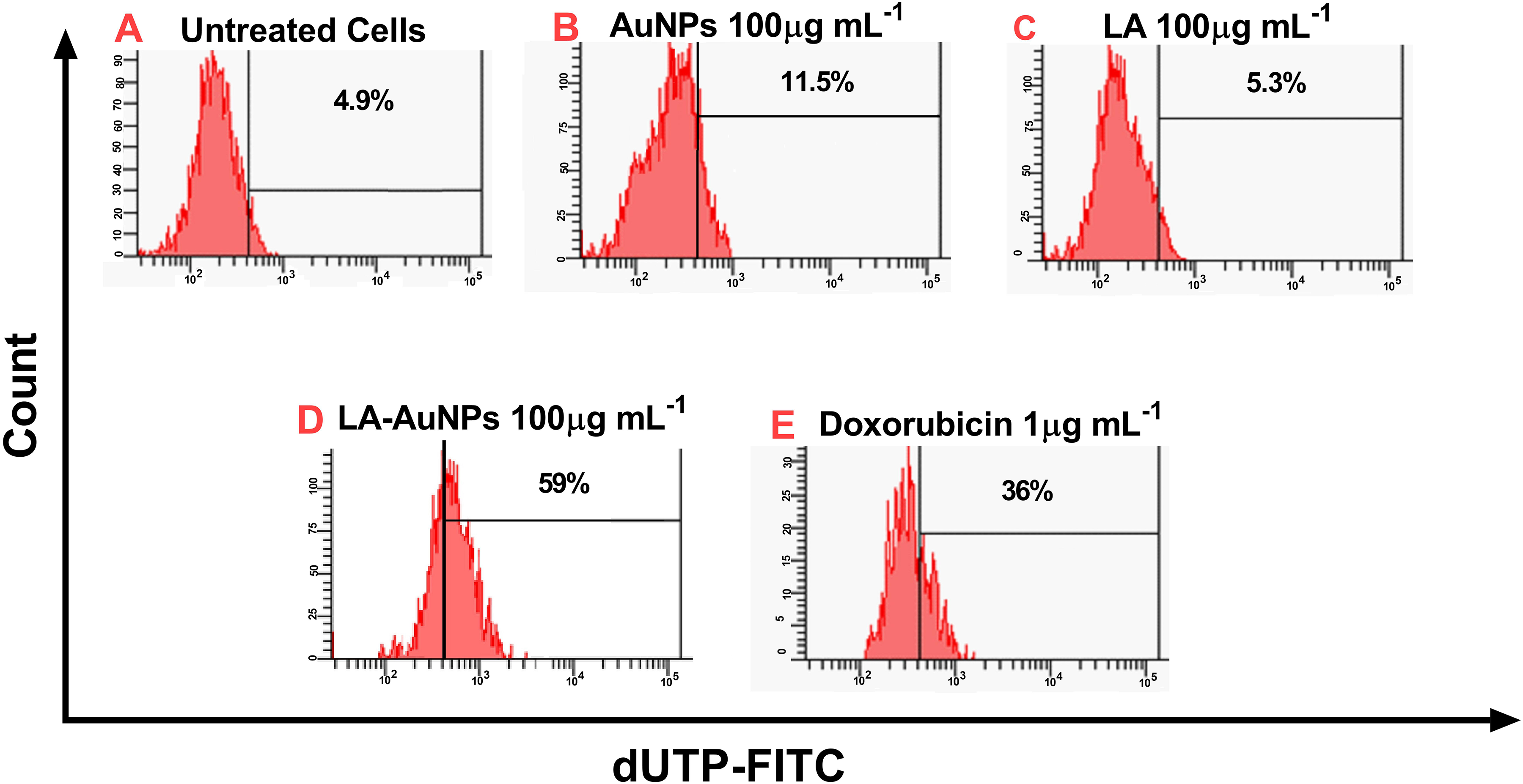
DNA fragmentation TUNEL assay of (A) untreated MCF-7 cells and MCF-7 cells treated with (B) gold nanoparticles (AuNPs; 100 µg·mL−1), (C) Lantadene A (LA; 100 µg·mL−1), (D) Lantadene A gold nanoparticles (LA-AuNPs;100 µg·mL−1), and (E) doxorubicin (1 µg·mL−1) for 48 hours. Results represent the percentage of cells with DNA damage. The shifting toward the right is an indicative of the increase in TUNEL positive cells (
Apoptotic Gene Expression Analyses
By using qRT-PCR, the expression levels of
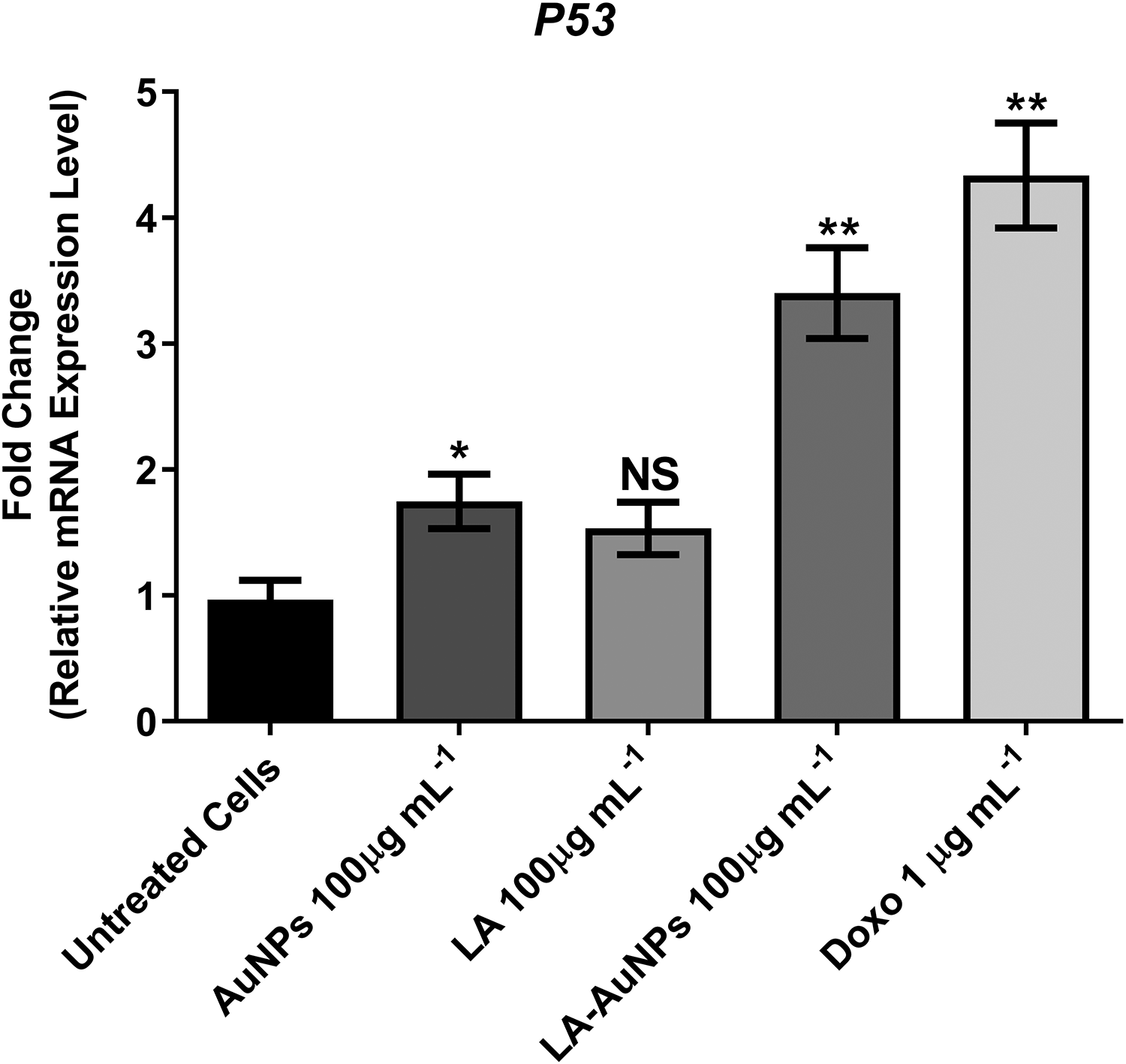
Effect of treating MCF-7 cells with AuNPs, Lantadene A (LA), and Lantadene A gold nanoparticles (LA-AuNPs; 100 µg·mL−1) and doxorubicin (1 µg·mL−1) for 24 hours on
Gold nanoparticles, LA, and LA-AuNPs had a significant effect on the regulation of
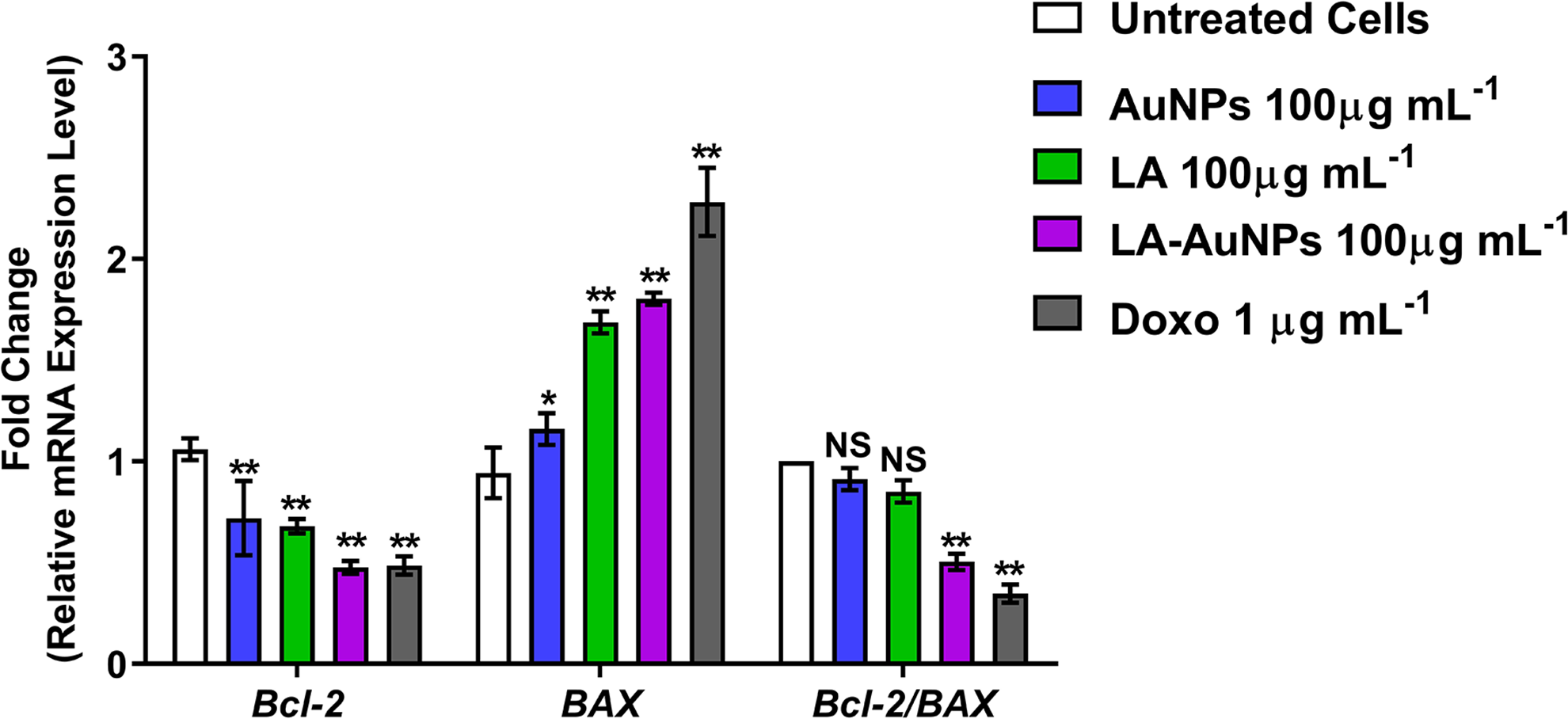
Gene expression results of
Discussions
The plant
Results of HPLC provided evidence for the presence of LA in the extracted sample with 93% purity. This percentage testifies to the efficiency of the extraction and purification method and to the accuracy of the analytical system used in HPLC analysis. 32 The peak detected at 4.880 minutes was assigned to the HPLC mobile phase, while the other 2 peaks at 8.880 and 14.773 minutes were not identified through the course of this study.
Gold nanoparticles were chosen to act as LA-carriers primarily because of their efficiency, benign nature, and their unsophisticated synthesis procedure. It was reported that inorganic nanoparticles (specifically AuNPs) uphold the second highest efficiency in drug delivery after dendrimers. 33 However, dendrimers have failed in most clinical trials due to their slow, complex, and expensive synthesis procedures, their high toxicity, unknown biocompatibility, unpredictable behavior in living organisms, and the difficulty in controlling therapeutic dose selection. 34 On the other hand, AuNPs are the least toxic among metallic nanoparticles, and studies have revealed that prolonged exposure of AuNPs did not harm the cells even at high concentrations. 13 Gold NPs can be easily functionalized and conjugated with a variety of molecules simultaneously; they can pack various different sizes and types of dendrimers and several different types of ligands along with the chosen drug so as to effectively combat different types of cancers to put AuNPs at the top as the best drug-carrier candidates. 35
Considerable interest has been granted to the development of several delivery nanosystems with different types of pentacyclic triterpenoids to enhance their well-known phytopharmaceutical and anticancer properties
36
; for example, ursolic acid was loaded on AuNPs with the addition of PLGA and was shown to cause cervical cancer cell death by inducing apoptosis through targeting apoptosis-specific proteins such as caspases and p53 and downregulating BCL-2 and cIAP both
Based on Mie’s theory, LSPR band red shifts with increasing particle size of the nanoparticles,
38
and the position of the LSPR band for LA-AuNPs, at 539 nm, postulate to have a relatively small average particle size. The UV–Vis spectra demonstrated that the LSPR band blue shifted as the temperature increased suggesting that smaller AuNPs can be obtained with a higher synthesis temperature.
39
Equivalently, the concentration of Au3+ affects the size of AuNPs formed. Gold NPs were synthesized using the aqueous leaf extracts of
Atomic force microscope analysis showed that the average particle size of LA-AuNPs was 32.78 nm, making these nanoparticles suitable candidates for delivering LA to tumor cells to boost its cytotoxic effects. Results of AFM provide another agreement with the results elucidated by UV–Vis that highlighted the small particle average size based on the LSPR band position at 539 nm. It has been determined that for the nanoparticle to be an effective drug carrier, it should have a diameter within 10 to 150 nm to guarantee a longer circulation time, increased accumulation in the tumor interstitium, and to avoid clearance by the renal system and mononuclear phagocytic system. 6
The increased cytotoxicity of LA-AuNPs, compared to free AuNPs and LA, on MCF-7 cells was most likely due to the enhanced solubility of LA-AuNPs and the effect of conjugated AuNPs in enhancing the penetration and accumulation of LA into the tumor tissue and promoting the availability of LA within the tumor; this induced the greatest cytotoxic effect with the lowest possible IC50 and hence caused insignificant toxicity on normal cells.
40
No such cytotoxicity was observed when the all compounds were applied to the normal WRL-68 cell line with the same range of concentrations (data not shown). In TUNEL assay, results have shown that the cooperation of LA and AuNPs induced DNA fragmentation at a ratio of 59%, indicating that the activity of LA-AuNPs in promoting apoptosis in MCF-7 cells could be attributed to the capability of AuNPs in enhancing the penetration and accumulation of LA into breast cancer cells and eventually boosting its cytotoxicity. Gold NPs were investigated for their cytotoxicity up to 200 µg·mL−1 (31% inhibition rate) in a dose-dependent pattern and apoptosis induction via
Conclusions
To summarize this study, AuNPs were synthesized through the reduction of HAuCl4 by LA extracted and purified from
Footnotes
Authors’ Note
All data and results supporting the conclusions of this research are available in the article.
Acknowledgments
The authors are grateful to the College of Biotechnology, College of Science and Biotechnology Research Centre at Al-Naharin University for offering funding, space, and utilities to accomplish the project.
Author Contributions
Jaafar, Noor D. substantially contributed to conception or design and drafted the manuscript; Al-Saffar, Ali Z. and Yousif, Emad A. contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by the College of Biotechnology, Al-Nahrain University, Baghdad—Iraq (Grant No. COB 4279).
