Abstract
Thrombin generation assay (TGA) is a sensitive method for the assessment of the global clotting potential of plasma. This kinetic assay can detect both hypocoagulable and hypercoagulable conditions: delayed or reduced thrombin generation leading to a prolonged clotting time, or induced thrombin activity, shifting the coagulation cascade toward thrombosis. The purpose of this study is to qualify the TGA in nonhuman primates (NHP) and rats for its use during nonclinical in vivo and in vitro studies. Blood was drawn from nonanesthetized animals, and platelet-poor plasma was obtained after double centrifugation; coefficients of variation were <10% for all derived parameters of thrombin generation assessed with 5 pM of tissue factor. Thrombin generation was evaluated in vitro in rat and NHP plasmas with ascending doses of unfractionated heparin (UFH), recombinant tissue factor, and anticoagulant compounds. Thrombin generation was decreased with UFH and anticoagulant compounds, but was increased in the presence of tissue factor, in a dose-dependent manner. In a rat model of inflammation, animals were administered a low dose of lipopolysaccharides. Thrombin generation measurements were decreased 3 hours post-LPS administration with a nadir at 24 hours, while thrombin–antithrombin complexes reached a peak at 8 hours, supporting an earlier production of thrombin. In conclusion, these data demonstrated that TGA can be performed in vitro for screening of compounds expected to have effects on coagulation cascade, and thrombin generation can be measured at interim time points during nonclinical in vivo studies in rats and NHP.
Keywords
Introduction
Evaluation of compound-related alterations on hemostasis is done during nonclinical studies by the evaluation of coagulation times: prothrombin time (PT), activated partial thromboplastin time (aPTT), and platelet counts. Regulatory guidelines recommend to not only evaluate these end points at a minimum but also advocate for a flexible approach depending on the species, the observed or expected effects of a given substance, and state that additional testing should be considered if necessary for the investigation of toxic effects. 1 -3 Coagulation times PT and aPTT assays consist of measuring coagulation in platelet-poor plasma (PPP) of the extrinsic and intrinsic pathways, respectively, as well as the common pathway. The prolongation of these coagulation times indicates a deficiency, dysfunction, or inhibition of one or more coagulation factors, but their capability to detect subtle changes remains limited because the clot formation occurs when only a small portion of the thrombin potentially present in plasma is produced. 4 These assays are also not designed to detect increases in coagulation factor activities or compound-induced hypercoagulability in nonclinical studies, which remains a major cause of compound withdrawal. 5 Thrombin generation assay (TGA) detects delayed thrombin generation, leading to a prolonged clotting time, and also increased thrombin activity, leading to an increased risk of thrombosis.
Thrombin plays a critical role in hemostasis, as it is the terminal enzyme of the coagulation cascade that transforms fibrinogen into fibrin. The peak of thrombin influences the rate of fibrin formation and the strength of the fibrin clot meshwork. It is also a potent platelet agonist and a major positive and negative feedback regulator of coagulation via interactions with thrombomodulin and the activated protein C pathway (Figure 1). Thrombin generation is a kinetic assay that is gaining interest in nonclinical studies, as it can detect both hypocoagulable and hypercoagulable conditions. The assay is designed to assess the plasmatic elements that participate in the formation of thrombin. It involves measuring the formation of thrombin in PPP after initiation of the coagulation pathway by addition of tissue factor. It can be performed on previously frozen plasma, which offers the possibility to investigate abnormal hemostasis findings observed during a nonclinical study, and it can be validated in all laboratory animal species as the processes of thrombin generation and fibrin clot formation are similar among all.
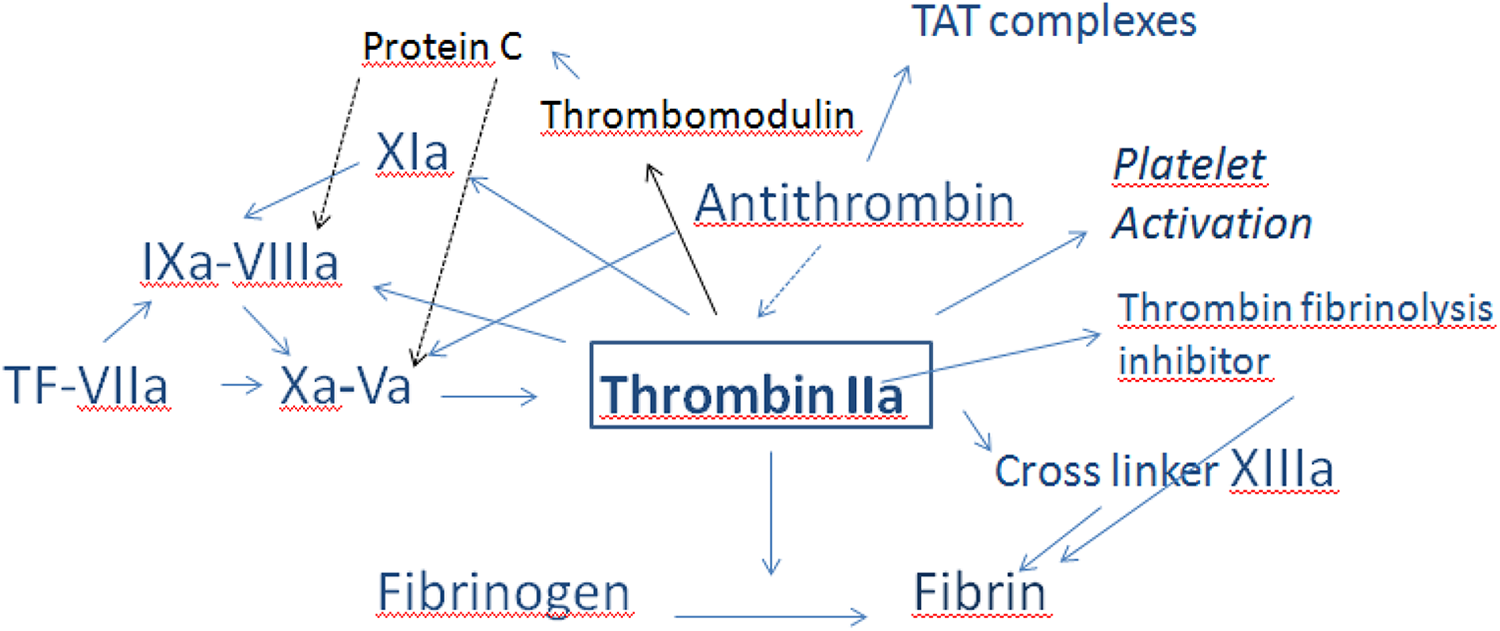
The role of thrombin in hemostasis. Thrombin has a central role in hemostasis. It acts as a procoagulant, activates platelets, cleaves fibrinogen to fibrin, and activates fibrin cross linker factor XIII and thrombin activatable fibrin inhibitor. Thrombin acts as an anticoagulant by the activation of protein C system via thrombomodulin. Thrombin inhibited by antithrombin form thrombin–antithrombin complexes. (Modified from Smith 17 )
The purpose of this study is to test the assay in nonhuman primates and rats for its use during nonclinical in vivo and in vitro studies. In an in vitro assay, the influence of heparin, recombinant tissue factor, and anticoagulant compounds on thrombin generation was evaluated. In a rat model of inflammation, the kinetics of the TGA over time was also performed.
Material and Methods
Animals
Adult Sprague-Dawley rats (males and females) ranging between 230 and 650 g body weight, originating from Charles River-Research Models and Services (St. Constant, Canada), were bled for in vitro assays or enrolled in the in vivo lipopolysaccharides (LPS) study.
Nonhuman primates included cynomolgus and rhesus monkeys. Cynomolgus monkeys (Macaca fascicularis) were of Chinese origin, male and female individuals, aged between 2.5 and 8 years, originated from an approved vendor (Charles River-Research Model and Services), and were part of the Charles River Montréal-ULC (Unlimited Liability Corp) training colony.
Rhesus monkeys (Macaca mulatta) were of Chinese origin, male and female individuals, aged between 2 and 11 years, originated from an approved vendor (World Wide Primates, Miami, Florida), and were part of the Charles River Nevada-ULC training colony.
Animals were housed in climate-controlled rooms, had access to drinking water, and were fed with a commercial diet. All animals were healthy, did not show any clinical signs, and had a normal physical evaluation. Before beginning the study, complete blood counts, serum biochemistry, and coagulation times were evaluated in NHP. All values were within the expected ranges. All procedures for animal use in these experiments were reviewed and approved by the Institutional Animal Care and Use Committee at Charles River-Montréal and Charles River-Nevada and were in compliance with the Guide for the Care and Use of Laboratory Animals 6 and in compliance with national and local animal use regulations.
Blood Samples
Animals were fasted overnight before blood sampling but had access to drinking water. Blood from rats was collected from the jugular vein using a 23- to 25-gauge needle attached to a syringe, and blood was immediately transferred to 3.2% sodium-citrated tubes after blood collection, with a final blood to anticoagulant ratio of 9:1. Blood from NHP was collected similarly with a 21-gauge needle attached to a syringe from the femoral vein of cynomolgus monkeys and with a 21-gauge vacutainer needle and tube from the femoral or brachial vein of rhesus monkeys. Blood volume was determined by body weight and study requirements and varied between 0.9 and 2.7 mL in rats, and between 1.8 and 15.3 mL in NHP.
Samples were inspected and any tubes containing visible clots were discarded. Tubes were centrifuged for 10 minutes at 4°C within 1 hour postcollection, at 2,400 g. Plasma was transferred into plain plastic tubes and centrifuged a second time at 2,400 g for 10 minutes at 4°C. Plasma was aliquoted and transferred into plain plastic tubes and to a freezer set to maintain −80°C prior thrombin generation analysis.
Coagulation Assays
Coagulation assays consisted of aPTT and PT and were performed on aliquots of frozen PPP from rats and rhesus monkeys. Plasma was thawed at room temperature (RT) and analyzed using an automated coagulation analyzer (StaCompact, Diagnostica Stago, Canada) and commercial reagents: aPTT-Synthasil for rats (Instrument Laboratory), aPTT-CK-Prest for NHP, and PT-Thromboplastin for all species (Diagnostica Stago).
Thrombin Generation Assay
Thrombin generation assays were performed on a Calibrated Automated Thrombogram instrument (CAT, Diagnostica Stago, Asnières sur Seine, France). The CAT assay uses a fluorogenic thrombin substrate and measures thrombin generation by comparing the fluorescence in plasma during clotting after tissue factor activation to the fluorescence of a reference thrombin calibrator. Samples were thawed at RT for 30 minutes and mixed by inversion or vortex. PPP Reagent (80 µL; Thrombinoscope Diagnostica Stago, Canada) containing a mixture of phospholipids, tissue factor, and thrombomodulin was loaded in triplicate in clotting wells of the 96-well plate. Thrombin calibrator (20 µL) was loaded in triplicate in calibrator wells. Platelet-poor plasma (80 µL) was added to all wells for each sample. For sample analysis, the prepared 96-well plate was placed on the CAT instrument. After the addition of plasma, the instrument dispenses 20 µL of a mixture of fluorogenic substrate (Z-Gly-Gly-Arg-AMC) and fluo-buffer containing calcium (FluCa-Kit, Diagnostica Stago, Canada) to start the coagulation in each well with a final concentration of tissue factor of 5 pM. Upon thrombin activation, the fluorogenic substrate releases a fluorescent marker, which is measured by a 390-nm-excitation and a 460-nm-emission filter set. All analyses were performed in triplicate, as recommended by the manufacturer, in most cases. Analyses were performed in duplicate when plasma volume was insufficient. With the thrombin calibrator as a reference, the molar concentration of thrombin was calculated during the measurement using the Thrombinoscope software. The Thrombinoscope software provides a tracing representing the mean of the triplicated reactions and all the calculated parameters of the thrombogram: endogenous thrombin potential (ETP; nM/min), peak (nM), lag time (minutes), time to peak (minutes), and start tail (minutes) values (Figure 2).
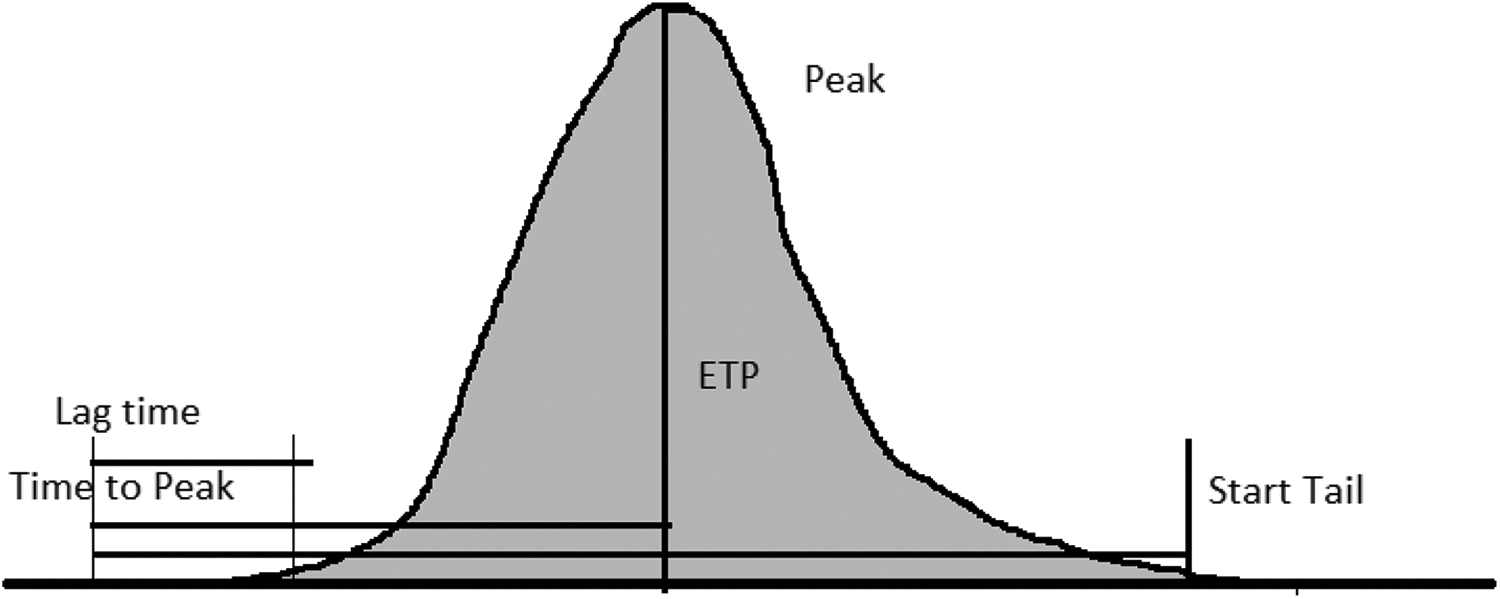
Thrombin generation assay variables. The tracing represents a mean of the triplicate reactions. The lag time represents the time for initial thrombin formation (minutes), peak indicated the maximal thrombin concentration obtained (nM), the time to peak represents its time, the endogenous thrombin potential (ETP) represents the area under the thrombin curve, and the start tail represents the time of the end of reaction.
Method Qualification
Intra- and interassay variability and stability over time were evaluated using individual plasmas for NHP and pooled plasmas for rats. Intra-assay variability was evaluated on 7 repeats processed in the same run. Interassay variability was based on 9 repeats performed in different batches. For stability, all plasma samples were collected and frozen as soon as possible for early time points.
In Vitro Heparin, Tissue Factor, and Anticoagulants Sensitivity Assays
To evaluate the sensitivity of the assay to anticoagulants, thrombin generation was measured in rat or NHP pooled plasma (males and females) spiked with serial dilutions of unfractionated heparin (UFH; Sandoz) in deionized water (0.0, 0.01, 0.05, 0.1, 0.2, and 0.4 U/mL).
Two other anticoagulants were also spiked in rat pooled plasmas in order to evaluate their effect on thrombin generation. Factor Xa (FXa) inhibitor, rivaroxaban (Cayman chemical), or a FIIa inhibitor, dabigatran (Cayman chemical), were diluted in 100% dimethyl sulfoxide to obtain dosing solutions of 30 μg/mL and were spiked in rat whole blood. All samples were incubated 1 hour at 37°C before being centrifuged twice for plasma separation and frozen at −80°C before TGA analysis. Samples spiked with anticoagulants were also evaluated for aPTT and PT measurements.
Similarly, to evaluate the sensitivity of the assay to procoagulants, thrombin generation was measured in rat or NHP pooled plasma spiked with thromboplastin containing recombinant human tissue factor (rh-TF). Two dilutions (1:100 and 1:1,000) of rh-TF (Dade Innovin, Siemens Medical System, Ontario, Canada) prepared in a phosphate-buffered saline solution (pH 7.4) containing 4% bovine serum albumin were used. Prior to analysis, 35 µL of either the buffer solution alone or the rh-TF dilutions (1:100 and 1:1,000) were added to 665 µL of pooled plasma.
In Vivo Evaluation of Thrombin Generation in Rats
Administration of LPS is known to trigger pro-inflammatory cytokines and to upregulate the expression of tissue factor, leading to the activation of the coagulation system. Low-dose LPS treatment in rats provides a time-course model of inflammation to develop biomarker profiles reflecting procoagulant imbalance and rebalance under inflammatory conditions. 7
A total of 8 male and 8 female rats were administered once by intraperitoneal injection at a low dose of LPS (Escherichia coli serotype 055: B5): 1 mg/kg. Rat blood samples were collected from the jugular vein, prior to LPS administration and 3, 8, 24, and 48 hours postdose. Blood samples were placed in citrated tubes for the measurement of TGA, and in ethylenediaminetetraacetic acid tubes for evaluation of thrombin–antithrombin complexes concentration by an ELISA method (Rat Thrombin-AntiThrombin Complex ELISA kit, Elabscience, Colorado). 8
Statistical Analysis
Numerical data collected for the different variables were analyzed according to the following description. Descriptive statistics: means and standard deviations or % coefficient of variation (% CV) were reported whenever possible. For the in vivo phase of the study, inferential statistics were performed but excluded any group with less than 3 observations. Statistical analysis was conducted using Levene test to assess the homogeneity of group variances. The groups were compared using an overall 1-way analysis of variance, F test if Levene test was not significant, or the Kruskal-Wallis test if it was significant. If the overall F test or Kruskal-Wallis test was found to be significant, then pairwise comparisons were conducted using Dunnett or Dunn test, respectively. Data sets with 2 groups were compared using a Dunnett test or Dunn test.
Pairwise comparisons were as follows: 3 hours postdose versus predose, 8 hours postdose versus predose, 24 hours postdose versus predose, and 48 hours postdose versus predose.
Results
Qualification of TGA in Rat and Cynomolgus and Rhesus Monkey PPP
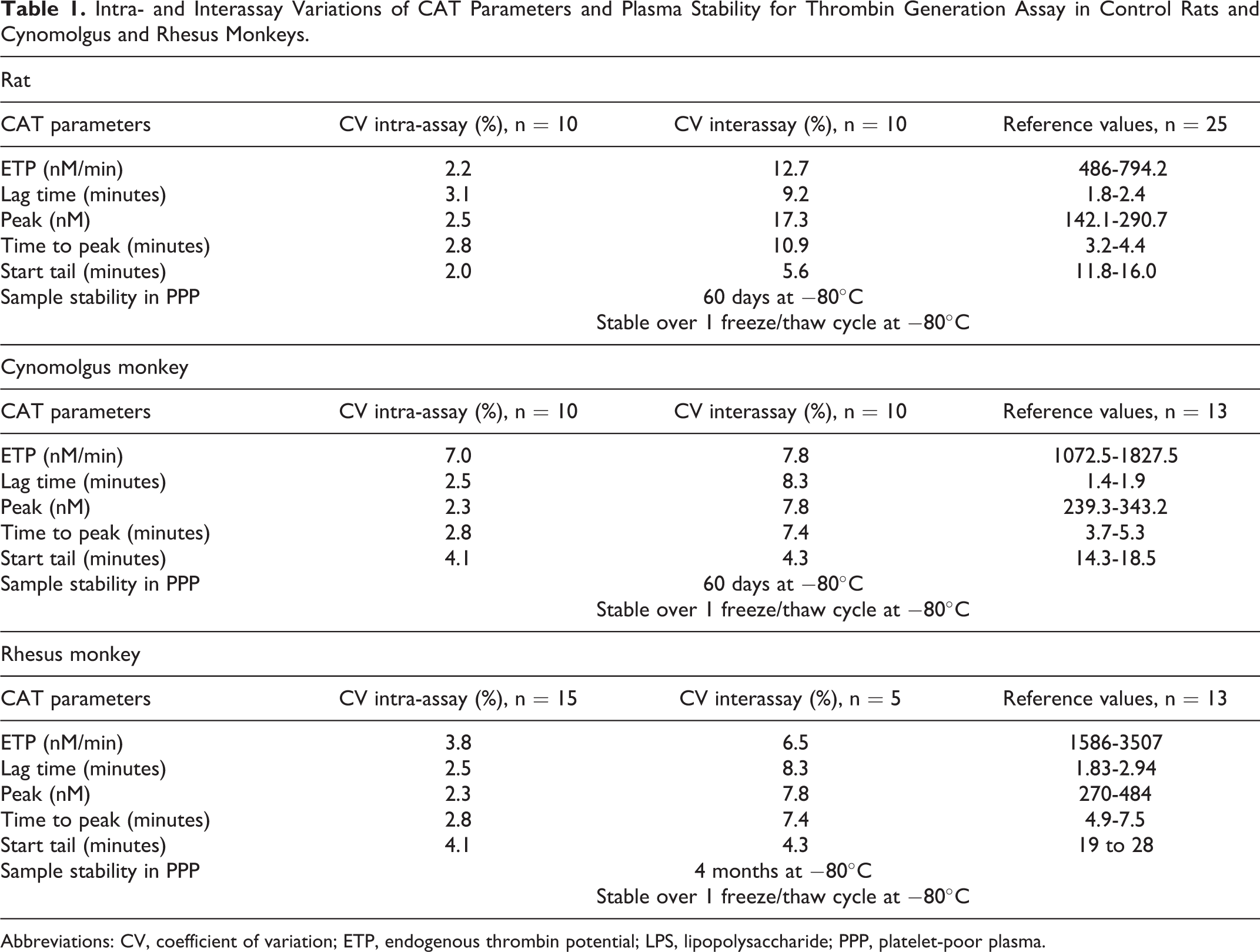
Table 1 displays the intra- and interassays coefficient of variation for TGA performed on frozen–thawed pooled rat PPP and individual NHP PPP.
Intra- and Interassay Variations of CAT Parameters and Plasma Stability for Thrombin Generation Assay in Control Rats and Cynomolgus and Rhesus Monkeys.
Abbreviations: CV, coefficient of variation; ETP, endogenous thrombin potential; LPS, lipopolysaccharide; PPP, platelet-poor plasma.
% CV intra- and interassays
The variability of TGA parameters between- and within-pooled plasmas was low for all parameters and for all species tested. Intra-assay % CV for ETP, lag time, peak, time to peak, and start tail ranged between 2.2% and 3.1% for rat plasma, 2.3% and 7.0% for cynomolgus monkey plasma, and 2.3% and 4.1% for rhesus monkey plasma. Interassay % CV range was 9.2% and 17.3% for rat plasma, 4.3% and 8.3% for cynomolgus monkey plasma, and 4.3% and 8.3% for rhesus monkey plasma. The variability of CAT parameters was low and acceptable (≤20%) for all parameters, the maximum variability was observed in the peak values.
Stability
Storage of PPP did not affect TGA results: at −80°C for up to 15 days in rats, up to 60 days in cynomolgus monkeys, and up to 4 months in rhesus monkeys. Samples were stable over 1 freeze/thaw cycle in a freezer set to maintain at −80°C.
Reference values were established independently of the sex of the animals because of the limited number of females available in the different colonies (Table 1). A large range of individual values was noted within each species, it was independent of the assay.
In Vitro Sensitivity to UFH in Rat and Rhesus Monkey PPP lipopolysaccharides
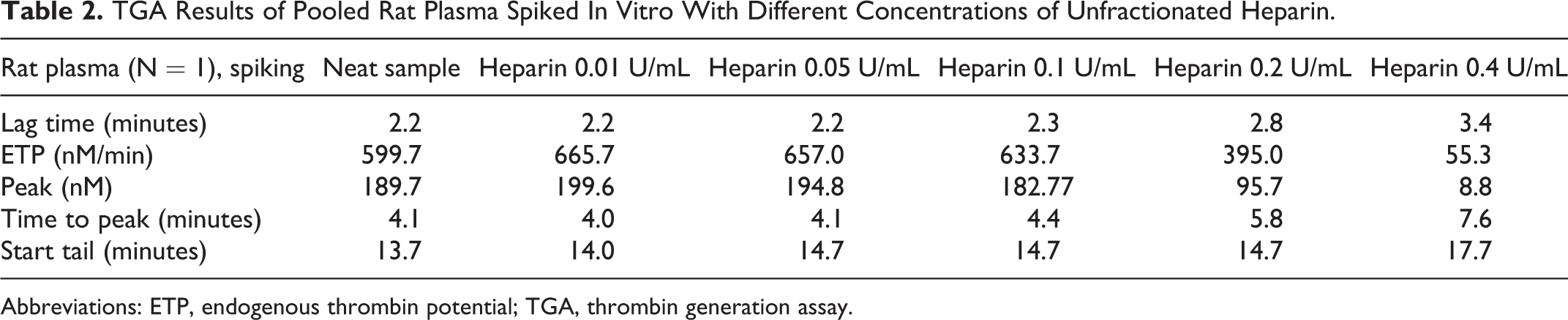
Pooled rat PPP was spiked with 0, 0.01, 0.05, 0.1, 0.2, and 0.4 U/mL of UFH (Table 2). Compared to nonspiked samples (baseline), heparin decreased the generation of thrombin in a dose-dependent manner: ETP and thrombin peak were decreased due a dose-dependent lower thrombin production, starting at 0.1 U/mL heparin, with marked decreases at heparin doses ≥0.2 U/mL. A dose-dependent increased time of reaction was also noted: The time of study start (lag time), time to peak, and time of the end of reaction (start tail) were higher. With low doses of heparin at 0.01 or 0.05 U/mL, there was no change in the shape of the thrombin generation curves, ETP and peak were unchanged or slightly increased, but the thrombin generation was delayed as start tail and time to peak tended to increase (Figure 3).
TGA Results of Pooled Rat Plasma Spiked In Vitro With Different Concentrations of Unfractionated Heparin.
Abbreviations: ETP, endogenous thrombin potential; TGA, thrombin generation assay.
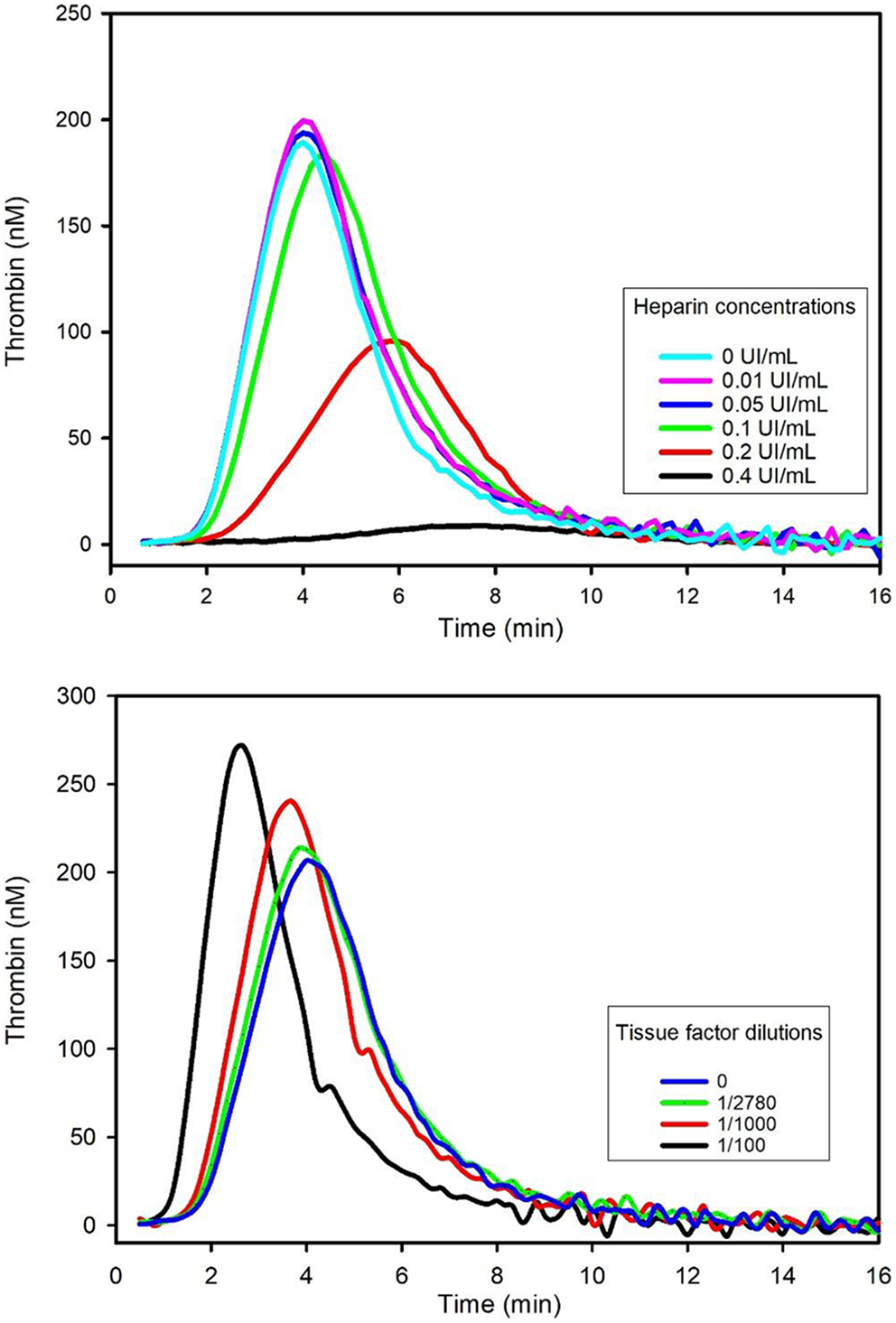
In vitro sensibility to heparin and recombinant tissue factor in rat plasma. Pooled rat pooled PPP was spiked in vitro with different concentrations of unfractionated heparin. Compared to nonspiked samples, heparin decreased ETP and thrombin peak in a dose-dependent manner, starting at 0.1 U/mL heparin, and marked decreases were noted at heparin doses ≥0.2 U/mL. Pooled rat PPP sample was spiked with a recombinant human thromboplastin reagent (Innovin) and showed increased generation of thrombin in a dose-dependent manner: Peak was higher and the time of reaction was faster. ETP indicates endogenous thrombin potential; PPP, platelet-poor plasma.
Individual rhesus monkey PPP was spiked with 0.2 U/mL UFH (Table 3). Compared to nonspiked sample (baseline), heparin moderately to markedly decreased the generation of thrombin in a dose-dependent manner: Mean thrombin peak and ETP were decreased (−71% and −69%, respective mean values compared to baseline). The time of reaction was prolonged: lag time, time to peak, and start tail were increased (+54%, +45%, and +13%, respective mean values compared to baseline).
TGA Results of Rhesus Monkey Plasma Spiked In Vitro With Unfractionated Heparin or Recombinant Human Thromboplastin (Innovin).
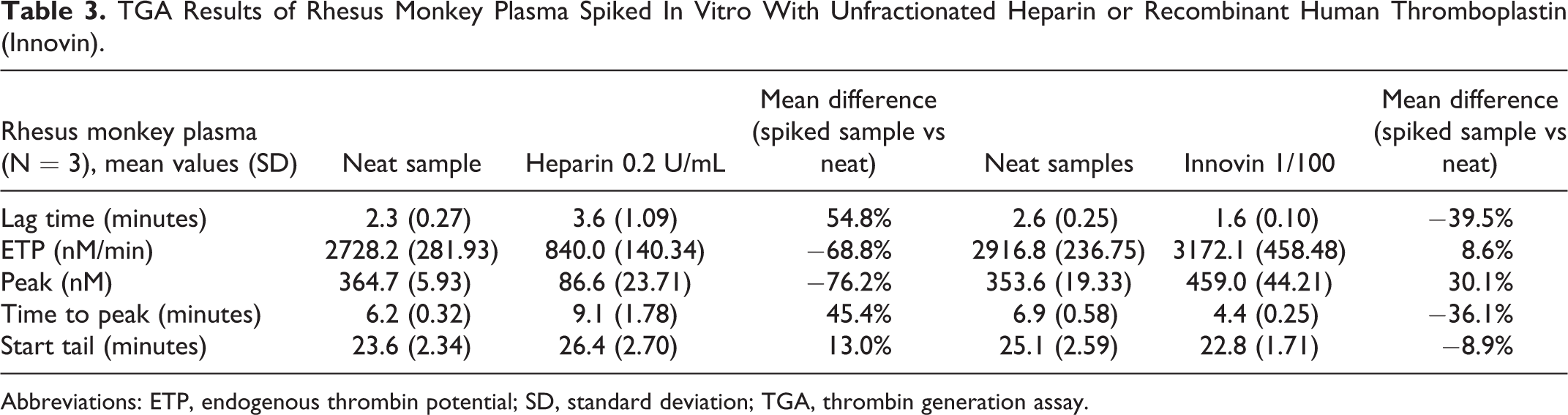
Abbreviations: ETP, endogenous thrombin potential; SD, standard deviation; TGA, thrombin generation assay.
In Vitro Sensitivity to Recombinant Tissue Factor in Rat and Rhesus Monkey PPP
Pooled rat PPP samples spiked with a recombinant human thromboplastin reagent (Dade Innovin, Siemens Medical System) containing rh-TF was compared to nonspiked samples (baseline; Table 4). Tissue factor increased the generation of thrombin in a dose-dependent manner: Thrombin peak was increased at all dilutions, starting at 1/2,780. The time of reaction was faster with increased doses of thromboplastin starting at 1/1,000: lag time, time to peak, and/or start tail were decreased. Endogenous thrombin potential was slightly increased with low doses of thromboplastin (1/2,780 and 1/1,000) and decreased with low dilutions of thromboplastin (1/100 and 1/10). These discrepant results were due to the shape of the curve that was sharper with higher concentrations of thromboplastin (Figure 3).
TGA Results of Rat Plasma Spiked In Vitro With a Human Thromboplastin Reagent (Innovin) Containing Tissue Factor.
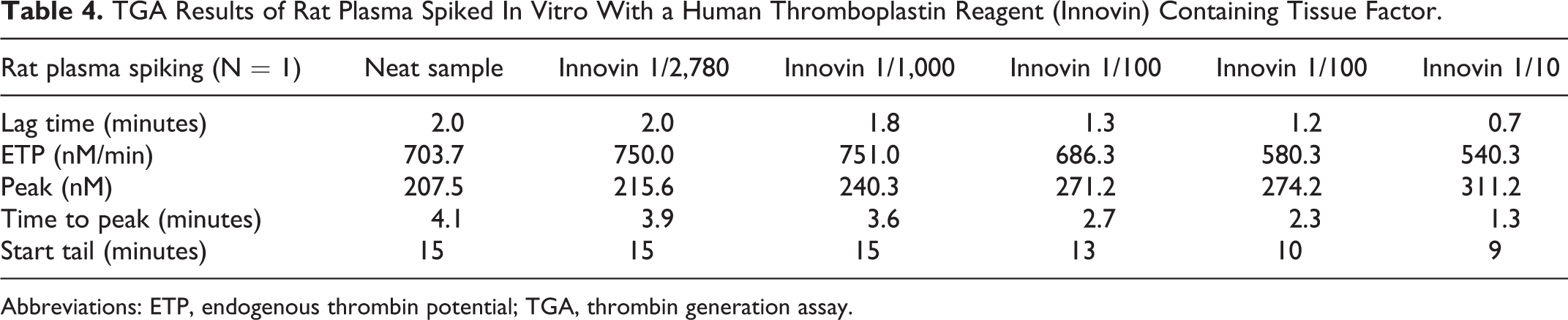
Abbreviations: ETP, endogenous thrombin potential; TGA, thrombin generation assay.
Individual rhesus monkey PPPs were spiked with a recombinant human thromboplastin reagent (Table 4). Compared to nonspiked sample (baseline), thromboplastin moderately increased the generation of thrombin in a dose-dependent manner: Mean thrombin peak and ETP were increased (+30% and +9%, respective mean values compared to baseline). The time of reaction was decreased: Mean lag time, time to peak, and start tail were decreased (−40%, −36%, and −9%, respective mean values compared to baseline).
In Vitro Sensibility to Dabigatran (FIIa Inhibitor) or Rivaroxaban (FXa Inhibitor) in Rat PPP
Individual rat plasmas were spiked in vitro with dabigatran (FIIa inhibitor) or rivaroxaban (FXa inhibitor; Table 5). In vitro spiking with dabigatran 0.3 μg/mL, a thrombin (FIIa) inhibitor, completely inhibited thrombin generation, as there was no curve observed.
Mean TGA Results of Individual Rat Plasma Spiked In Vitro With Dabigatran (FIIa Inhibitor) or Rivaroxaban (FXa Inhibitor) in Rat Plasma.
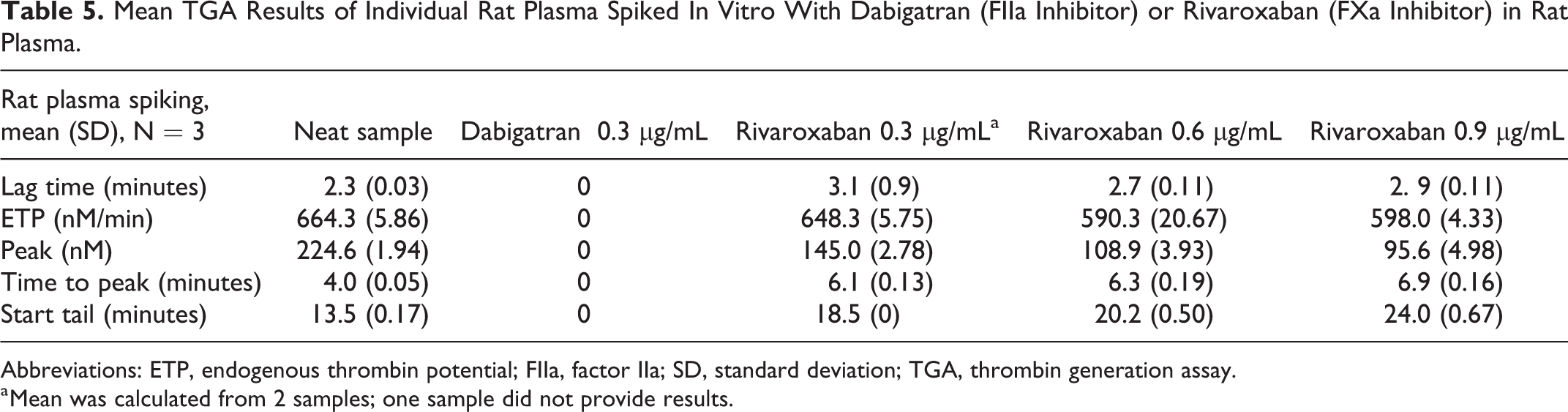
Abbreviations: ETP, endogenous thrombin potential; FIIa, factor IIa; SD, standard deviation; TGA, thrombin generation assay.
a Mean was calculated from 2 samples; one sample did not provide results.
In vitro spiking of rat plasma samples with rivaroxaban at 0.3, 0.6, and 0.9 µg/mL, a direct FXa inhibitor, showed a dose-related decrease in thrombin generation: Mean ETP and thrombin peak were decreased due to a lower thrombin production. The time of reaction was prolonged: Time to peak and start tail were increased, while the lag time was unchanged.
Activated partial thromboplastin time was measured on spiked samples and showed concurrent increases (2- to 3.5-fold changes, individual values compared to baseline; data not shown).
In Vivo Evaluation of Thrombin Generation in Rats
Thrombin generation was evaluated in rats administered a low dose of LPS by intraperitoneal administration. Coagulation times (PT and aPTT) showed no relevant changes over time of administration, but differences were observed in thrombin generation parameters.
Mean rat PPP thrombin generation variables measured at 3, 8, 24, and 48 hours postdose were compared to predose respective values for male and female groups (Table 6). Compared to predose values, mean lag time was decreased at 3 and 8 hours (−11% to −17%), and time to peak was decreased at 3 and 8 hours (−9% to −10%). Both ETP and peak showed moderate decreases at 3 hours postdose, with a nadir at 24 hours (−28% to −44%), and a mild rebound at 48 hours (Figure 4). Start tail was unchanged. Increases in TAT concentration were observed at 3 hours, reached a peak at 8 hours (up to 6 fold), and gradually decreased at subsequent time points (Figure 5).
TGA Results of Rat Administered LPS In Vivo (N = 8 per Group).
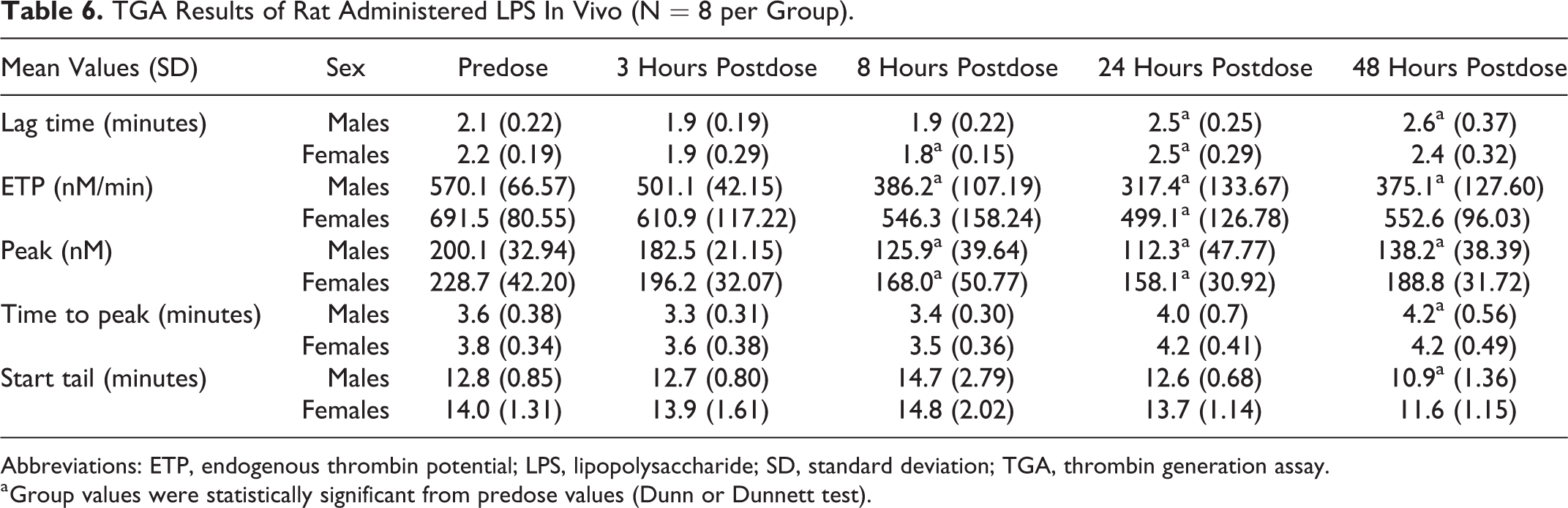
Abbreviations: ETP, endogenous thrombin potential; LPS, lipopolysaccharide; SD, standard deviation; TGA, thrombin generation assay.
a Group values were statistically significant from predose values (Dunn or Dunnett test).
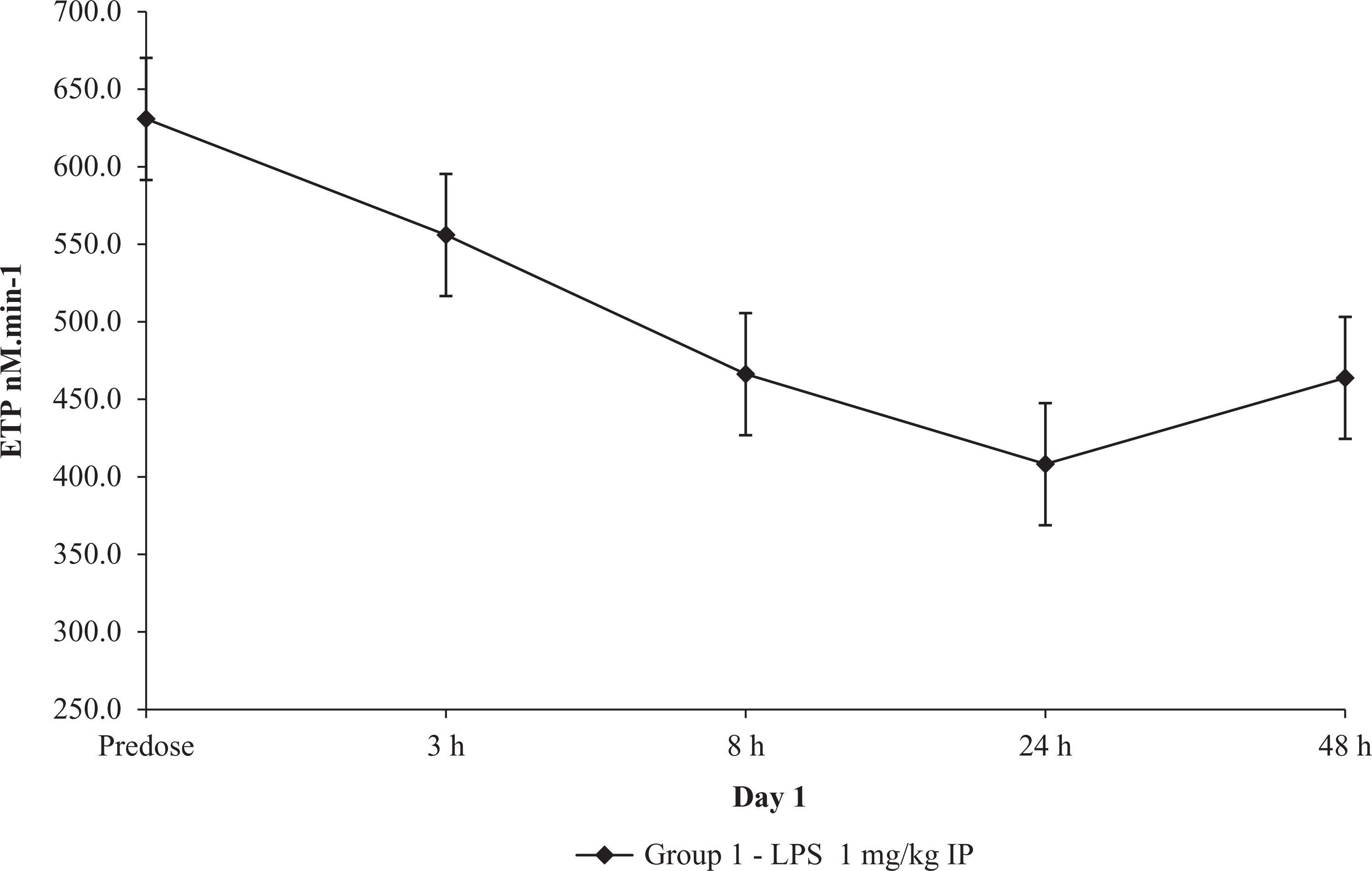
Thrombin generation after in vivo low-dose LPS administration in rats. Mean ± standard error ETP results (N = 10). ETP indicates endogenous thrombin potential; LPS, lipopolysaccharide.
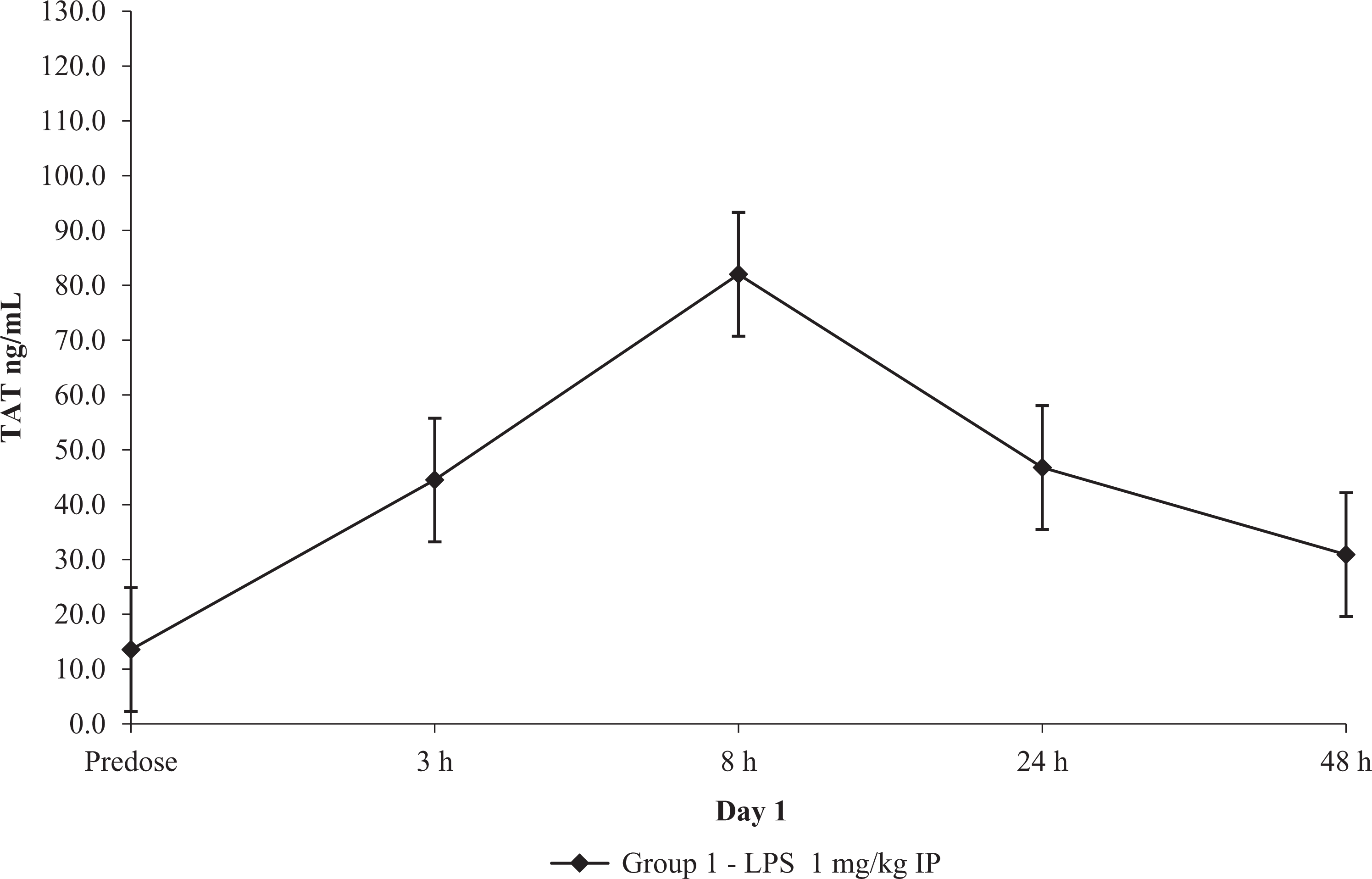
TAT concentration after in vivo low-dose LPS administration in rats. Mean ± standard error ETP results (N = 10). ETP indicates endogenous thrombin potential; LPS, lipopolysaccharides.
Discussion
Our results demonstrated that thrombin generation can easily be measured in rats and NHP during nonclinical studies. We followed previously published recommendations for blood collection. Skilled technicians collected the blood by clean draws on quiet nonanesthetized animals with only minimal restraint, limiting the activation of the coagulation pathway by the release of tissue factor during venous puncture. 4,9 Platelet-poor plasma was obtained from blood, rapidly after collection by double centrifugation in order to avoid any contamination by residual platelets debris or microparticles that may influence thrombin generation. Indeed, platelets and microparticles provide phospholipid membranes that support the assembly of coagulation complexes that catalyze thrombin generation. 4 Application of this standardized procedure resulted in acceptable validation results with CVs below 10% for all derived thrombin generation parameters. Important intra- and interassay variability of TGA results obtained during previous studies had raised questions about the interest of this assay as they resulted in conflicting results. 10 It is probable that TGA using low concentrations of tissue factor reagents are more prone to preanalytical variability, while sample processing may have less influence on TGA triggered by high concentration of tissue factor reagent. In this study, we used a PPP reagent that contained a moderate concentration of tissue factor of 5 pM.
Discarding the first tube of collection is often recommended for specialized coagulation assays, as tissue factor is released during endothelial puncture. In NHP, we choose to keep the first tube collected for the evaluation of coagulation times and evaluated thrombin generation on the second tube collected, but we kept the first tube collected from rats for TGA because of their limited blood volume. Results were consistent in rats, although they were obtained from the first blood tube collected from the jugular vein. This method of collection is ideal for obtaining quality blood samples for interim evaluations during rat preclinical studies. We used small gauge needles fixed on syringes or vacutainer tubes; the use of a butterfly needle was not recommended for blood collection as contact activation and hemolysis may be induced by the long tubing of the butterfly device and may increase thrombin generation. 11 Blood was collected on citrated plastic tubes or vacutainer-citrated plastic tubes for the different species, and there was no resulting difference noted between these plastic containers.
In order to measure thrombin activity in animal plasmas that have much faster thrombin generation than in humans, the instrument was set for shorter measurement intervals to 10 seconds in order to obtain smooth and continuous curves. Also, we used only the half of the 96-well plates to limit the delay between the start of the reaction and the reading start of the plates, as the reading of the plates can start only when the FluCa-kit has been dispensed to all measuring wells.
Stability of the PPP samples was limited in rats. Samples were not stable at RT or at 4°C after 24 hours. This finding was not surprising, as we have previously observed limited stability of PPP for aPTT coagulation time in rats in our laboratory: 6 hours at RT, while stability of 24 to 48 hours was observed for other species. We elected to freeze the samples immediately at −80°C after their preparation to minimize any variability. Stability was established on frozen samples and tested over a freeze and thaw cycle. For practical issues, we did not extend the stability assay to longer times, but there was no indication that longer periods of stabilities could not be proven. All samples were tested in batch. To eliminate interassay variation, pooled plasmas were analyzed on the same plate for each spiking experiment. Similarly, for the LPS experiment, all the plasmas collected from a single animal were analyzed on the same plate. The PPP samples were thawed at RT and preheated 10 minutes at 37°C in the fluorometer. Assay reproducibility and stability were satisfying in rats and NHP. However, a large range of individual values was noted, which was independent of analytical variability but showed that it is necessary to evaluate individuals compared to their respective pretreatment values.
In vitro assays showed the effects of heparin and tissue factor in a predictable manner on the generation of thrombin in rat and rhesus monkey samples. A dose-related effect was observed with both compounds, demonstrating decreased thrombin generation and hypocoagulability with heparin spiking, and increased thrombin kinetics and hypercoagulability with tissue factor spiking. Spiking of dabigatran, a thrombin inhibitor, on rat plasma showed as expected a complete inhibition of thrombin generation. Spiking of riboxavaran, a FXa inhibitor, on rat plasma showed a dose-related inhibition of thrombin generation. These assays demonstrated the interest of TGA for the screening of compounds having an expected effect on coagulation factors as the assay is more sensitive than coagulation times, or to understand unexpected off-target effects of compounds on coagulation times during nonclinical studies. When evaluating the different changes observed in such assay, it is important to note that all parameters derived from the thrombin generation curves do not vary in the same direction. Parameters representing the time of reaction (start tail, time to peak, or lag time) tend to vary with low concentrations of compounds, whereas parameters representing a change in the curve and in thrombin concentration (peak and ETP) will vary with higher concentrations of compounds.
An in vivo rat study was initiated to demonstrate the interest of TGA for the detection of prothrombotic states. Administration of LPS is known to trigger an inflammatory process, increasing plasma cytokines and tissue factor bearing microparticles that tend to activate the coagulation cascade and unbalance the coagulation equilibrium toward hypercoagulability. 7 After administration of LPS in rats, the first blood collection was done at 3-hour postdose, and we observed minimal decreases in lag time and time to peak reflecting a slight acceleration of the generation of thrombin, and moderate decreases in peak and ETP reflecting a decrease in production of thrombin, while TAT complexes were increased. Thrombin generation continued to decrease at 8 and 24 hours and slightly reversed at 24 hours. TAT increases reached a peak at 8 hours and gradually decreased at subsequent time points. The increases in TAT were indicative of an increased production of thrombin, which may have started earlier than 3 hours postdose, as we did not observe any increases in thrombin generation at 3 hours. Decreases in TGA showed a reduced production of thrombin probably secondary to a previous increase, but it was not detected at the 3 hours postdose time point. That result was unexpected. With LPS-induced inflammation, we expected an increase in TGA, resulting in an increase in TAT. Indeed, when thrombin is generated, thrombin–antithrombin complexes are formed, resulting in thrombin inactivation. However, TGA was decreased at 3 hours postdose, meaning that the full potential of the elevation of thrombin had occurred before 3 hours, as indicated by increased level of TAT complexes. A rapid elevation of TAT had previously been reported to peak at 4 hours postdose after intravenous administration of thomboplastin, and we did not anticipate such a rapid change in TGA after an intraperitoneal LPS administration. 12 This assay demonstrated the interest of the measurement of combined TAT and TGA during in vivo studies as markers of hypercoagulable or prothrombotic conditions in rats and also the necessity to set up early time points. Concurrent TGA and TAT results indicated an excessive generation of thrombin consistent with hypercoagulability, with a gradual return to baseline concentrations. After administration of low dose of LPS, early time points at 15 minutes, 30 minutes, and/or 1 hour postdose should be considered to detect the changes in thrombin. Coagulation times showed no relevant changes over time of administration in this study, demonstrating the higher sensitivity of TGA and TAT measurement to increased or decreased coagulation factor activity.
There are multiple applications of the TGA. It was used in a recent study to characterize the coagulant status of antithrombin inhibitors on hemophilic NHP and mouse models and to demonstrate a rebalance of the coagulation equilibrium after administration of the compounds. 13 Thrombin generation assay was also considered a standard assay to assess nanoparticles procoagulant activity in human plasma, as it is a fast and accurate method for toxicological preclinical screening of the potential impact of nanoparticles on coagulation. 14,15 Assessment of thrombin generation in combination with platelet aggregation is a very interesting tool for the evaluation of potential medications for the prevention of cardiovascular events that use dual therapies combining anticoagulants and antiplatelet compounds, as a persistent excess of thrombin generation is recognized as a risk of myocardial infarction. 16
In conclusion, we have shown that with minimal precautions, it is possible to obtain adequate thrombin generation results and to monitor these at interim time points during a nonclinical study. Thrombin generation assay can be performed on frozen plasma and can be added as a second tier in nonclinical studies in order to investigate unexpected hemostasis. It is a global assay, similar to thromboelastography, that evaluates the contribution of all plasmatic elements to the formation of the fibrin clot. It is a more sensitive assay than coagulation times to increased or decreased coagulation factor activity. It has also the advantage to be applicable to any species in nonclinical studies and in human in clinical studies. In vitro TGA is also of interest for the screening of compounds expected to have effect on coagulation factors.
Footnotes
Author Contribution
F. Poitout-Belissent contributed to design, contributed to acquisition and interpretation, drafted the manuscript, and critically revised the manuscript; D. Culang contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript; D. Poulin contributed to design, contributed to acquisition and analysis, and critically revised the manuscript; S. Cotton contributed to analysis and critically revised the manuscript; R. Samadfan contributed to design, contributed to analysis, and critically revised the manuscript; C. Bedard contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. D. Culang performed thrombin generation experiments. S. Cotton performed thrombin-–antithrombin measurements. D. Poulin and R. Samadfan performed the in vivo study. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Charles River Laboratories Canada.
