Abstract
Aflatoxin (AF) B1 is a potent hepatotoxic, mutagenic, teratogenic mycotoxin and may cause immune suppression/dysregulation in humans and animals. Toxic effects of AFB1 on key mammalian immune cells (ie, leukocytes) needs to be mechanistically elucidated. In this study, along with the determination of AFB1’s LC50 for certain leukocytes, we analyzed the effect of naturally occurring levels of AFB1 on apoptosis/necrosis of neutrophils, lymphocytes, and monocytes from healthy young humans (20- to 25-year-old male), dogs (1- to 2-year-old Persian/herd breed), and cattle (1- to 2-year-old cattle). Leukocytes were incubated for approximately 24 hours with naturally occurring levels of AFB1 (10 ng/mL). Intracellular adenosine triphosphate (ATP) depletion and caspase-3/7 activity were then determined by luciferase-dependent bioluminescence (BL). Furthermore, the necrotic leukocytes were measured using propidium iodide (PI)-related flow cytometry. A significant decrease (24%-45%, 33.2% ± 2.7%) in intracellular ATP content was observed in AFB1-treated neutrophils, lymphocytes, and monocytes in all studied mammals. Also, with such a low level (10 ng/mL) of AFB1, BL-based caspase-3/7 activity (BL intensity) in all 3 tested mammalian leukocyte lineages was noticeably increased (∼>2-fold). Flow cytometry-based PI staining (for viability assay) of the AFB1-treated leukocytes showed slightly/insignificantly more increase of necrotic (PI+) neutrophils, lymphocytes, and monocytes in human, dogs, and cattle. Even though in vitro LC50s for AFB1’ (∼20,000-40,000 ng/mL) were approximately 2,000 to 4,000 times higher than background, these studies demonstrate leukocytes from human and farm/companion animals are sensitive to naturally occurring levels of AFB1. The observed in vitro ATP depletion and caspase activation in AFB1-exposed leukocytes can partially explain the underlying mechanisms of AFB1-induced immune disorders in mammals.
Introduction
Human/animals’ exposure to aflatoxin (AF) B1 poses a high risk of various infectious and noninfectious diseases. Aflatoxin B1 produced mainly by the Aspergillus flavus is a potent carcinogen/toxin for various cells and organs and a cause of immunosuppression/dysregulation in human and animals. 1 -7 Aflatoxigenic fungi, particularly A flavus and Aspergillus parasiticus, grow in a wide range of humid, warm, and dry weather, are a common contaminate in raw food and feed, and can predominantly produce carcinogenic AFs. 1,8 -10 Nowadays, worldwide environmental changes, global warming, high temperatures, high moisture, and many other controversial issues contribute to fungi proliferation and possibly mycotoxin contamination, endangering feed/food safety during pre- and postharvesting activities. 11 The Food and Agricultural Organization of the United Nations has estimated that up to 25% of the world’s food crops are highly contaminated with mycotoxins. 12 Aflatoxin B1 is the most predominant and frequently occurring mycotoxin/aflatoxin. 9,13 Based on the recent research reports, carcinogenicity, immunosuppressive effects, acute hepatic necrosis, bile duct proliferation, edema, and lethargy are some of the pathologic conditions caused by AFs in human and animals. 8,14,15 Since 1993, the International Agency for Research on Cancer has classified AFB1 and mixtures of AFs as group 1 carcinogens. 16 Several metabolites of AFs that are produced through epoxidation and hydroxylation pathways (AFB1-8,9-epoxide, aflatoxicol and AFM1) are highly toxic. 17
Apoptosis or programed cell death is a significant form of cell death to maintain homeostasis, which is physiologically and genetically programmed and considered an active cellular response in both pathologic and physiologic conditions. 9,18,19 Excessive apoptosis in circulating leukocytes can cause severe immunopathologic conditions such as autoimmune diseases and cancer. 19,20 Apoptosis is featured with a list of morphological and enzymatic changes, including, caspase activation, cell contraction, extensive plasma membrane budding, chromatin condensation, fragmented nucleus and formation of macromolecules, enzymatic hydrolysis, and apoptosome. 18,19,21,22 The 3 highly regulated main pathways of cell death activation are referred to as the mitochondria-associated apoptosome formation (intrinsic), the binding of extracellular death ligand (extrinsic), and the cytotoxic lymphocytes-initiated granzyme B with several caspases- and adenosine triphosphate (ATP)-related physiopathological pathways. 18 -25
Aflatoxin B1 is an external toxin and has documented pathologic effects on apoptosis in the liver, kidney, thymus, spleen, and gastrointestinal tract immune system. 26 Based on the dose and time of effect, one report indicates AFB1 may reduce the number of B and T lymphocytes and disturb cytolysis activity in neutrophils. 27 Research has been confirmed that AFB1 can induce apoptosis and potential ATP depletion in various cells, 28 -30 which might occur through caspase-3/7 activation and ATP depletion.
Reports have been published on the effects of AFB1 on leukocytes/immune cells, 2,3,31 -34 but little research has ever been conducted on the molecular mechanisms of AFB1 in various species, specifically with the high-throughput bioluminescence (BL)-based assays. In comparison with the existing routine labor-intensive tests (eg, flow cytometery), here we used a novel, less labor-intensive, elegant, sensitive, and high-throughput assay based on firefly luciferase to detect the exact quantity of intracellular ATP and caspase-3/7 in various leukocytes of human, dog (worldwide popular pet/companion animal), and cattle (a considerable food supplier), which were exposed to an environmentally relevant level (∼10 ng/mL) of AFB1. Thus, this study is an effort to analyze somehow the comparative effects of AFB1 on neutrophils, lymphocytes, and monocytes of the 3 mammalian species.
Materials and Methods
Blood Sampling and Leukocyte Culture
Blood samples were aseptically collected from 3 sample groups consisting of six 20- to 25-year-old male volunteers, six 1- to 2-year-old dogs with similar breed (Persian/herd; from Mehr Animal Shelter), and six 1- to 2-year-old Holstein dairy cows (from Ferdowsi University of Mashhad’s dairy farm). Human, dog, and cattle’s heparinized blood samples were immediately checked for paraclinical/hematological (complete blood count and leukocyte differentiation), isolation of various immune cells, flow cytometry and BL-based ATP, and caspase assays/tests. Isolation of human, dog, and cattle leukocytes from peripheral blood were performed using hypotonic lysis of erythrocytes.
3,4,27,31,32
Briefly to isolate the peripheral blood mononuclear cells (PBMCs), the human, dog, and cattle blood samples were separately diluted 4-fold with phosphate-buffer saline (PBS)/culture medium and were gently poured/added onto the same volume of Ficoll and then centrifuged (800 × g, 18°C, 30 minutes). The PBMCs layer was collected and washed twice with PBS/culture medium (350×g, 4°C, 10 minutes). Lastly, 3 mL of 107 PBMCs/mL of complete RPMI medium (with >98% viability) was seeded in a 3-cm diameter culture plate and was incubated for 2 hours at 37°C (incubator Memmert, Buechenbach, Germany) with 95% humidity and 5% CO2. Suspended leukocytes (>95% lymphocytes) and adherent cells (>95% monocytes) were resuspended in the complete RPMI medium containing 10% fetal bovine serum, 3 mmol/L
Aflatoxin B1 Treatment
Aflatoxin B1 powder was purchased from Sigma-Aldrich Chemie (A6636; Sigma-Aldrich, St. Louis, Missouri); it was Aspergillus derived, >98% pure, and free of lipopolysaccharide and other chemicals. The AFB1 was separately dissolved, as a stock, in 96% ethanol (0.1 mg/mL; Sigma-Aldrich) as described previously. 3,4 Further dilutions (∼105-fold) were made with PBS, making the concentrations of ethanol in the working solution very negligible. Aflatoxin B1 was added to the medium containing isolated immune cells at final concentrations of 10 ng/mL. The test group samples were cultured for approximately 24 hours in a 24-well plate containing 10 ng/mL AFB1 at 37°C with 95% humidity and 5% CO2 (incubator Memmert). For the control groups, the same volume of medium/PBS without AFB1 was used.
Bioluminescence-Based ATP and Caspase Assays
As ATP depletion is a major result of active process of apoptosis, 23,28,36 intracellular ATP was measured by luciferin-luciferase reaction-based BL assay. Human, dog, and cattle neutrophils, lymphocytes, and monocytes of all groups were transferred individually into microtubes and stored at −80°C until assay was performed. Then, they were gently lysed with lysis buffer on smashed ice. The intracellular ATP levels of the AFB1 treated and control leukocytes were quantified by measuring the BL intensity (Berthold LB 9501 luminometer) generated in an ATP-dependent luciferin-luciferase BL assay (Bioluminescence Somatic Cell Assay System; Sigma-aldrich, Munich, Germany). A >6-point standard curve of ATP (0-0.5 mM) was considered in each series of assay and then the ATP content was quantified according to the standard curve, 30 ensuring the notion that the cathodoluminescence intensity is an exact equivalent to the cells’ ATP content. To determine ATP levels in immune cells, after treatment of the cells, activity of luciferase is simply measured. Note: For the benefit of the readers, the detailed methods and results from data from the ATP standard curve are also provided in supplemental (Figure S1).
To ensure the cytotoxic effects of AFB1, the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) tetrazolium reduction assay was used to the LC50 of AFB1 on all studied immune cells. 36 Note: For the benefit of the readers, the detailed methods and results from the MTT are also provided in supplemental (Figure S2).
Human, dog, and cattle neutrophils, lymphocytes, and monocytes in both AFB1-treated and control groups were also assayed for caspase-3/7 activity using a carboxyfluorescein FLICA apoptosis detection kit (Immunochemistry Technologies, LLC) by a BL microplate reader (Biotek, Bad Friedrichshall, Germany). Every cell pellet in microtubes was placed on smashed ice for BL-related caspases assays. Then, 100 µL lysis buffer (100 g of lysis buffer consisting of EDTA: 37 g, KH2PO4: 231 g, K2HPO4: 3.18 g, 7 mm2-mercaptoethanol: 48 L, 7 mm2-mercaptoethanol: 48 L, 1% [vol/vol] Triton X-100: 1 mL, glycerol: 10 mL) was added to every tube and mixed well (20-30 times pipetting for complete lysis). Supernatants were centrifuged (12,000×g, 4°C, 5 minutes) and used for ATP and caspase assays. It is worth mentioning that after assaying and analyses of both ATP and caspase activity, the cumulative RLU/s was finally converted to the natural or Naperian logarithmic (LN or log2) scale for better readability of the results.
Flow Cytometry Assay for Necrosis Detection
After 18 to 24 hours of incubation of human, dog, and cattle neutrophils, lymphocytes, and monocytes, 1 mL of cells from every plate (control and AFB1-treated) was stained with propidium iodide (PI; 5 mL of a dilution of 5 µg in 1 mL) and was incubated for 5 to 15 minutes on ice before analyzing them on flow cytometer, as previously described. 32,34 Then, samples were analyzed in 605 nm (BD Accuri C6 Co) to detect the percentage of necrotic neutrophils, lymphocytes, and monocytes.
Statistical Analysis
Collected data were analyzed using SPSS software (version 21), and Wilcoxon signed-ranks test (P value < 0.5) were used. SPSS software (version 21) was used to perform the statistical analyses. Means and standard errors of the mean (SEM) were calculated, and data were presented as means ± SEM. To determine the statistically different groups, hypothesis testing was performed at 5% significance level.
Results
One key indicator of cells undergoing apoptosis is intracellular levels of ATP. In this study, ATP changes in treated leukocytes were evaluated mainly due to its role in the regulation of caspase activity and apoptosome formation. The BL intensity (equal to the content of ATP) in leukocytes was measured 24 hours after exposure to a single concentration of AFB1 using firefly luciferase assay. Twenty-four hours after AFB1 treatment, BL intensity/intracellular ATP content substantially decreased in leukocytes, that is, herein in human, dog, and cattle neutrophils, lymphocytes, and monocytes (Figure 1).
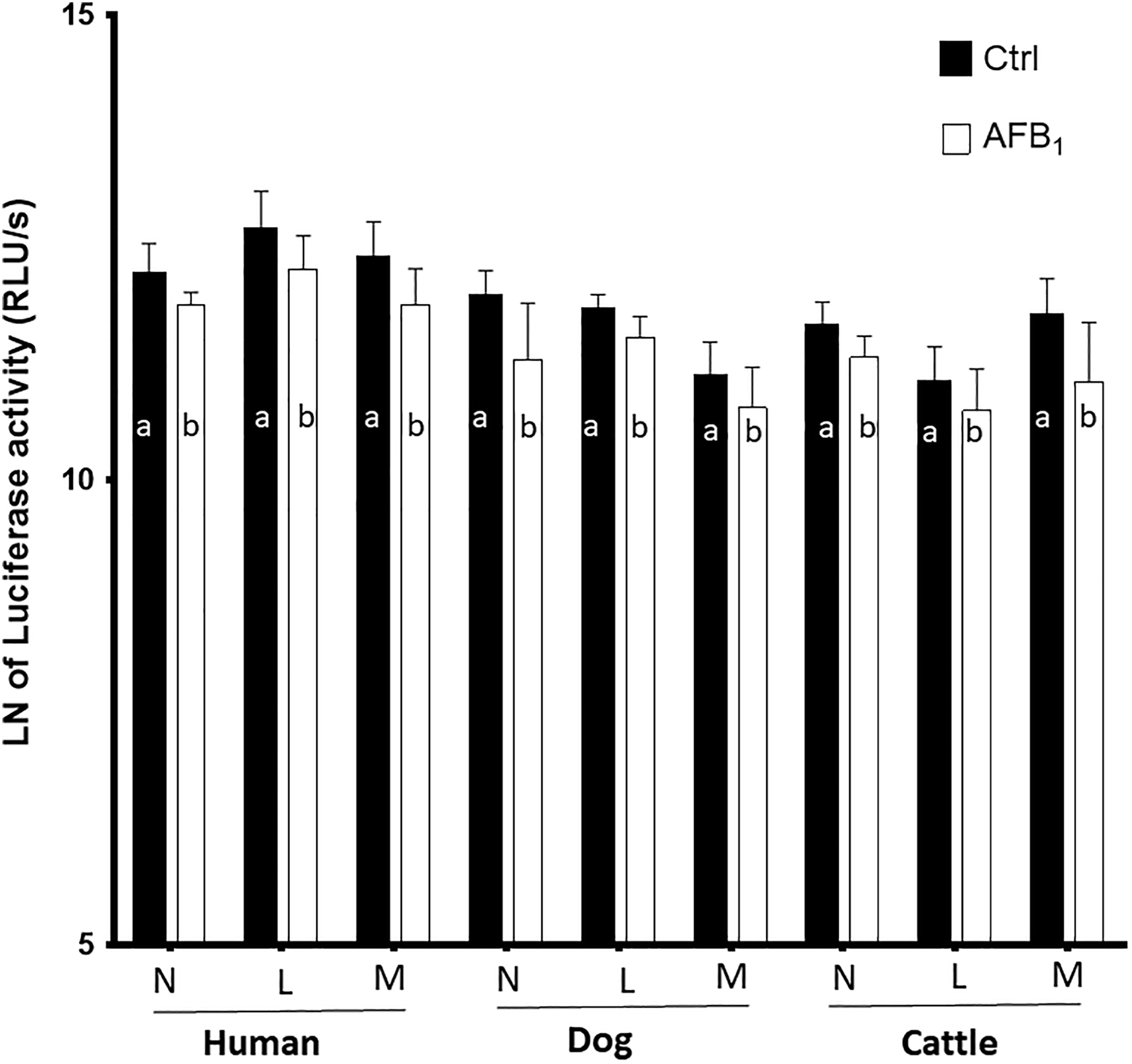
Changes in adenosine triphosphate (ATP) quantity of human, dogs, and cattle neutrophils (N), lymphocytes (L), and monocytes (M) caused by naturally occurring level of aflatoxin (AF) B1 exposure. As it can be seen from the charts, ATP quantities significantly decreased in AFB1-treated groups. Different letters indicate significance (P < 0.05) from their respective control (nontreated ones). Values are mean ± standard error of the mean of 12 replicates. The cumulative RLU/s or bioluminescence intensity was converted to the natural or Naperian logarithmic (LN or log2) scale for better readability.
Conversely, the caspase-7/3 activity substantially increased in post-AFB1-exposed neutrophils, lymphocytes, and monocytes of all 3 species (Figure 2). In this study, we did not compare the values of BL intensity (either ATP or caspases) among human, dog, and cattle samples. Furthermore, the flow cytometric diagrams were comparatively shown in Figure 3A. In the majority of groups, percent of necrotic leukocytes in AFB1-treated groups tended to increase compared to the non-AFB1-treated/control groups (see Figure 3B). The in vitro LC50 s of AFB1 (∼20,000-40,000 ng/mL) in various cells revealed the notion that they were approximately 2,000 to 4,000 times higher than background (∼10 ng/mL; see supplemental Figure S2).
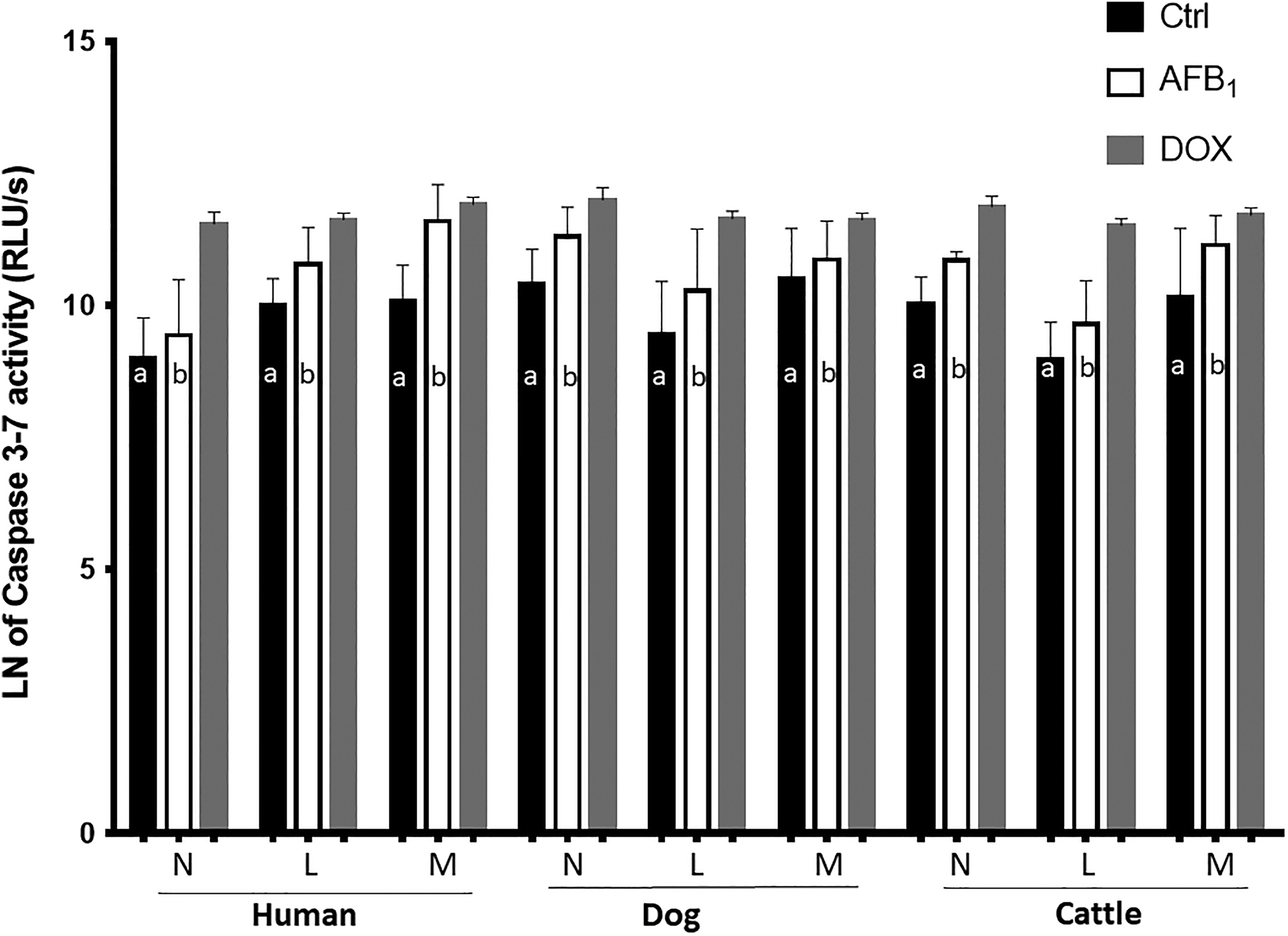
Changes in caspase-3/7 activity of human, dogs, and cattle neutrophils, lymphocytes, and monocytes caused by naturally occurring level of aflatoxin (AF) B1 exposure. In all 3 species, lymphocytes show an increase of 100% to 120% in caspase-3/7 activity after exposure to AFB1; also, human monocytes show a remarkable increase (∼4-fold) in caspase activity after treating with AFB1. Caspase activity in monocytes and other cells of human, dogs, and cattle also revealed increased activity after AFB1 affection. Different letters indicate significance (P < 0.05) from their respective control (nontreated ones). Values are mean ± standard error of the mean of 12 replicates. Doxorubicin was used only as positive control for apoptosis/caspase activity and were not statistically compared from other groups in the experimental design. The cumulative RLU/s or bioluminescence intensity was converted to the natural or Naperian logarithmic (LN or log2) scale for better readability.
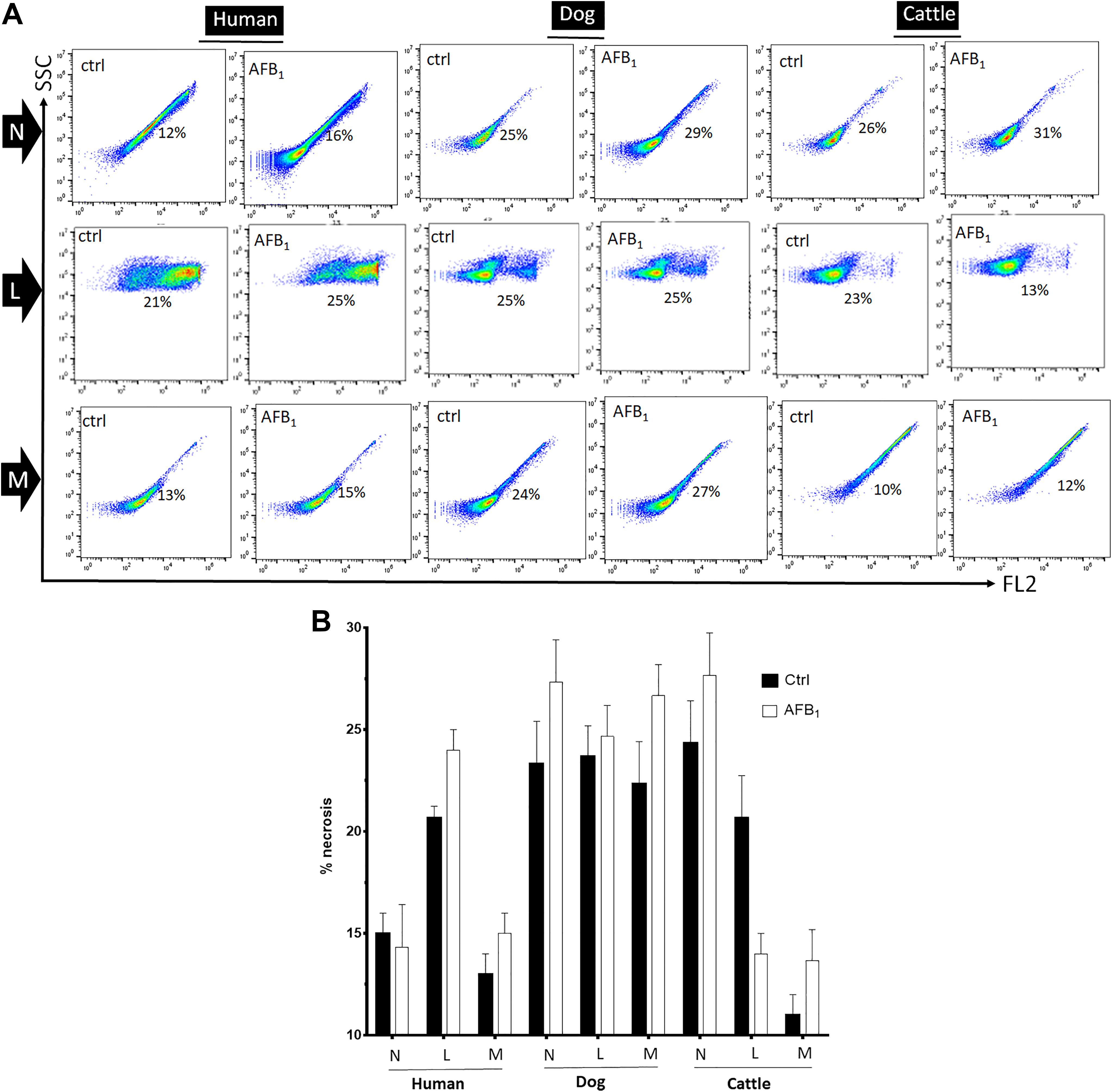
A, Representative results of leukocytes population displayed in the scatter plot showing the changes of necrotic cells in all groups of aflatoxin (AF) B1-treated leukocytes. The percentage of necrotic, upper right, neutrophils (N), lymphocytes (L), and monocytes (M) is shown in the representative histograms. B, Histogram showing overall differences between respective N, L, and M groups; the augmentation of necrotic leukocytes in AFB1-treated groups tended to slightly increase, though insignificant. Values are mean ± standard error of the mean of 6 replicates.
Discussion
Herein, along with the determination of in vitro AFB1’s LC50 s for certain leukocytes, which was extremely high (see supplemental Figure S2), we described the effect of naturally occurring level of AFB1 on the induction of apoptosis in immune cells (neutrophils, lymphocytes, and monocytes) isolated from healthy human, dogs, and cattle sources. We measured the ATP level and particularly caspase-3/7 along with PI staining as indicator of cells injury (ie, apoptosis and necrosis). It is also worth mentioning that here, accordingly, we used environmentally relevant range of AFB1 (∼10 ng/mL), unquestionably making the dose rationale in our study relevant, worldwide. 1,3,34,37 Indeed, the used amount of AFB1 routinely circulates in agricultural commodity and thus food-producing animals and human body, which potentially disturbs immunity; such a detectable amount of AFB1 could even exist in agricultural commodity/animals and human body in western/northern Europe 3,37,38 and poses a serious environmental problem, worldwide, potentially invisibly/chronically harming mammals’/humans’ key immune cells. 2 -4,30 -34,37
Importance of AFB1 toxicity in human and animal’s food, air, water, and environment with its carcinogenic impacts is undeniable. 32 -34 In addition, a rising concern is the development of a globally more suitable condition for the growth and proliferation of fungi species. Aflatoxin B1 in very low doses can lead to immunological deficiency, DNA mutation, and oxidative imbalance. 4,32,34,39 These can stimulate inflammation and increase the risk of infectious and noninfectious diseases, especially cancer, 40,41 via adversely impacting on the function or availability of immune cells.
In spite of research on the effects of AFB1 on cancer, 41 immunity, necrosis, and apoptosis, 30,32,34,41 the mechanism of action of AFB1 remains to be understood. In addition, there is no information regarding to the BL-based approaches for detection of AFB1 effects. We quantified the effects of AFB1 on leukocytes in human, dogs, and cattle using BL-based approach and concluded that AFB1–leukocytes interactions could mechanistically trigger immunotoxicity in mammals. Although observed ATP depletion and caspase activity along with slight decrease in cell viability herein (they would be, but not always, one of the hallmarks of cell toxicity/damage/apoptosis), nonetheless other confirming assays such as TUNEL stain, Annexin staining, cytochrome c release from mitochondria with Western blot, and morphologic confirmation of apoptosis are worth considering.
Based on the previous reports, after the entrance through cell membrane (here leukocytes), AFB1 is bioactivated by cytochrome P450 (CYP) enzymes/isoforms and glutathione S-transferase/GSH families to generate the activated highly toxic metabolites of AFB1 (ie, AFB1-endo/exo-8, 9-epoxide, and aflatoxicole) and excludable nontoxic AFB1-GSH, respectively. 3,13,31 -34 These potent secondary metabolites (AFB1-epoxide and aflatoxicole) can cause severe oxidative stress in immune cells and disturb leukocytes’ antioxidant activity that lead to disorders in immune system. 4,31 -34 Aflatoxin B1-epoxide and aflatoxicole can simply attach to cell components, especially to proteins and DNA, easily affecting many physiologic, immunologic, and signaling pathways, 20,39 of which the extent of disruption remains elusive. Induction of apoptosis in immune cells, increase in total free radicals, reactive oxygen species system impairment, and the decreased concentration of Abs caused by AFB1 are parameters, which need further investigation.
In the present study, for the first time, we analyzed the damaging effects of AFB1 on mammalian neutrophils, lymphocytes, and monocytes by high-throughput BL assay. Previous studies 22,24,31 -34 represented the direct effect of AFB1 and other mycotoxins on human and animal leukocytes. But our novel work herein is that AFB1’s special effect on apoptosis of predominant mammalian’s various types of leukocytes measured by luciferin-luciferase-based BL technique.
Necrosis and apoptosis are 2 major forms of cell death, which differ in morphology, mechanism, and incidence. Typical features of necrosis are membrane rupture, respiratory poisons and hypoxia causing ATP depletion, metabolic collapse, cell swelling, and rupture leading to inflammation. Necrosis can be described as cellular metabolic collapse and occurs when a cell loses its ionic homeostasis. 21
Apart from intracellular ATP depletion, apoptosis itself further harms leukocyte respiration. Via decreased ATP, observed herein, AFB1 also causes impaired cellular respiration. Changes in caspase-3 and caspase-7 activity and ATP in different immune cells and different mammalian species followed a similar pattern. According to the results, it is unquestionable that the total intracellular enzymes involved in AFB1 metabolism vary in different animals, which causes differences in the level of activity of caspases (eg, in comparison with human and dog monocytes); we have recently found that the level of AFB1 metabolizing enzyme in monocytes was higher than lymphocytes. 31,32,34 Nevertheless, it is more acceptable to say that human monocytes are more sensitive than other immune cells. 31,32 However, our study hardly supports the point of higher sensitivity of human species to AFB1, and we did not tested the related enzymes or metabolic differences across species. Indeed, the enzymes involved in detoxification of the AFB1 in different species might vary depending on various extracellular/intracellular molecules/enzymes and even the permeability of different cell membranes might be involved, which is beyond the scope of this study. Since we measured the difference within the cells and the metabolism inside the cells, we did not compare any membranes permeability either, which is worth studying.
We can generalize that the total intracellular enzymes involved in AFB1 metabolism vary in different animals, which causes differences in the level of activity of caspases (eg, in comparison with human and dog monocytes, the level and functions of the AFB1-metabolizing enzymes in monocytes might be higher than lymphocytes). 31,32,34
Based on the result of current study, human monocytes were shown to be more sensitive than other cells to AFB1 and its effects on apoptosis pathway. To expand our knowledge on the effects of AFB1 on mammalian leukocytes, it is suggested to analyze cell membrane transparency/permeabilization to AFB1 in different species. Our study proves the fact that AFB1 increase the caspase-3/7, but whether this effect is directly or through mitochondrial mediation remains to be seen. Nevertheless, we previously found that AFB1 increases cytochrome c in other cells like murine astrocytes. 2,30 But still the gap exists, and AFB1 could simply induce leukocytes apoptosis via caspase-3 and caspase-7 in different leukocytes and species; as such, deactivation of caspase-3 and caspase-7 can be as important and reliable indicators of apoptosis. Indeed, apoptosis is an ATP-consuming phenomenon, which is accompanied with an amplification in both primary or intrinsic (caspase-9) and executioner or extrinsic (caspase-3/7) caspases (eg, 10-fold and 2-fold increases in caspase-3/7 and caspase-9 activity, respectively). 42 As such, in majority of cells, it has been shown that upon apoptosis induction, content of cellular ATP hugely decreases, though the extent of the decrease varies (20%-40%) and largely depends on the cell type, type, and concentration of apoptosis inducer. 43
In conclusion, the results indicated that a biologically relevant concentration of AFB1 (10 ng/mL) induces apoptosis in mammalian neutrophils, lymphocytes, and monocytes through ATP depletion and caspases activation. Though there is no clear evidence of what cellular levels of ATP or caspases activities result in apoptosis in specific immune cells, nonetheless a decreased intracellular level of free ATP by approximately 20% leads to apoptosis in cell lines. 44,45 Further, at late stage of apoptosis, caspase activity is maximal. 46 Such in vitro effects of very tiny amount (>2,000 time lower than LC50, see Figure S2) of AFB1 on leukocytes at their organelles and protein levels opens new doors to understanding the biological behavior of AFB1 as well as molecular mechanisms that might translate to in vivo immune system, infection, and cancer in mammals caused by chronic exposure to AFB1 (see Figure 4).
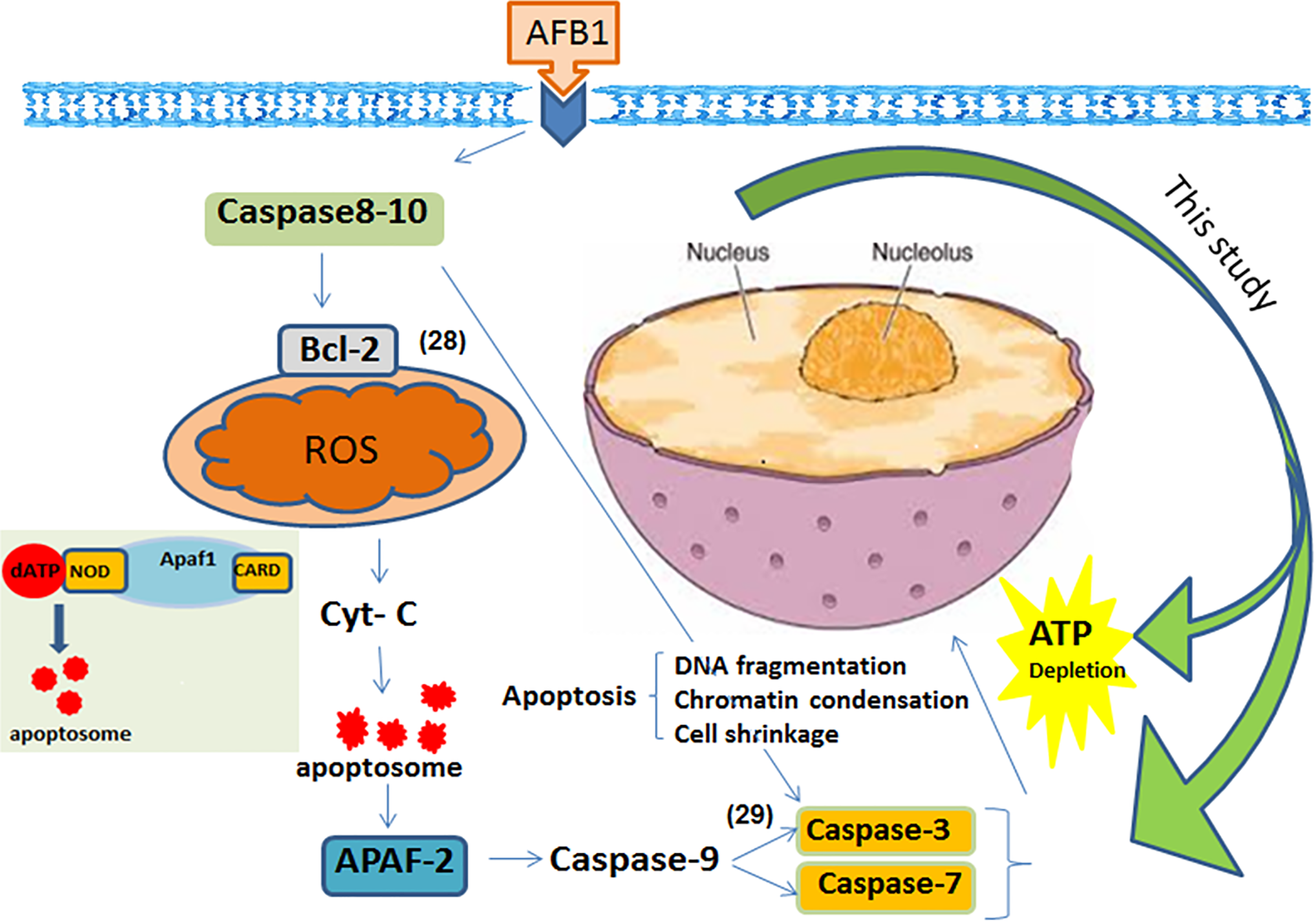
Aflatoxin (AF) B1 can induce apoptosis via caspase-3/7 activation and ATP depletion. This might be occurred through the death-receptor-mediated, free radicals, cell signaling, and mitochondrial pathways of cell apoptosis. 2,3,4,28 -30 In the intrinsic pathways, various apoptotic stimuli (eg, ROS or other microenvironmental stresses) mediate the permeabilization of the mitochondrial outer membrane and trigger signaling pathways. Within the cytosol, cytochrome c along with Apaf-1 and dATP forms the apoptosome complex to which the initiator procaspase-9 is recruited and activated. Caspase-9-catalyzed activation of the effector caspase-3 executes the final steps of apoptosis. This schematic picture highlights what are detected in this study as apoptosis indicators (caspase-3/7 and intracellular ATP depletion); such an apoptosis detection approach should be implemented to understanding the biological behavior of AFB1 as well as molecular mechanisms that might translate in vivo in human and animals. ATP indicates adenosine triphosphate; ROS, reactive oxygen species.
Supplemental Material
Supplemental Material, IJT892613_Figure_S1 - Naturally Occurring Level of Aflatoxin B1 Injures Human, Canine and Bovine Leukocytes Through ATP Depletion and Caspase Activation
Supplemental Material, IJT892613_Figure_S1 for Naturally Occurring Level of Aflatoxin B1 Injures Human, Canine and Bovine Leukocytes Through ATP Depletion and Caspase Activation by Jalil Mehrzad, Fatemeh Fazel, Nazaninzeynam Pouyamehr, Saman Hosseinkhani and Hesam Dehghani in International Journal of Toxicology
Supplemental Material
Supplemental Material, IJT892613_Figure_S2 - Naturally Occurring Level of Aflatoxin B1 Injures Human, Canine and Bovine Leukocytes Through ATP Depletion and Caspase Activation
Supplemental Material, IJT892613_Figure_S2 for Naturally Occurring Level of Aflatoxin B1 Injures Human, Canine and Bovine Leukocytes Through ATP Depletion and Caspase Activation by Jalil Mehrzad, Fatemeh Fazel, Nazaninzeynam Pouyamehr, Saman Hosseinkhani and Hesam Dehghani in International Journal of Toxicology
Footnotes
Acknowledgments
The authors gratefully acknowledge the supports of Ferdowsi University of Mashhad.
Author’ Contributions
Mehrzad J. and Fazel F. are equal first authors. Mehrzad J. contributed to conception and design, contributed to acquisition, drafted the manuscript, and critically revised the manuscript; Fazel F. contributed to design, contributed to acquisition and analysis, drafted the manuscript, and critically revised the manuscript; Pouyamehr N. contributed to design and acquisition; Hosseinkhani S. contributed to acquisition and analysis; Dehghani H. contributed to design and analysis, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
