Abstract
To investigate the underlying mechanism of neurotoxicity of cadmium, we examined the effects of intraperitoneal injection of cadmium on messenger RNA (mRNA) expression of
Introduction
Cadmium is one of the most toxic heavy metals that is distributed in our environment. 1 The main sources of cadmium intake are food, water and smoking cigarette. Cadmium can reach the brain by the olfactory route. 2 –5 Cadmium has been linked to health problems including osteoporosis, damage to liver and kidneys, increased mortality and cardiovascular system dysfunction, 1,6 and it is classified as a group I human carcinogen by the International Agency for Research on Cancer. 7
Several studies have shown cadmium toxicity in humans and animals. Furthermore, the different tissues increased the antioxidant molecules to prevent the oxidative damage caused by cadmium. 8 Blood–brain barrier (BBB) has high levels of antioxidant enzymes that protect from oxidative stress. Exposure to cadmium has been linked to antioxidant defenses in the brain’s microvasculature and leads to BBB dysfunction, resulting in more metal entering the brain. 9 Cadmium can directly damage the choroids plexus and increase BBB permeability in rats, 10 leading to brain intracellular accumulation, cellular dysfunction and cerebral oedema. 11 Khan and Parvez showed that the highly deleterious capacity of cadmium to cross the BBB induces neurotoxicity. 12
Previous studies have shown that the brain is a target for cadmium, 13,14 wherein cadmium can induce neurotoxicity, 15 changes in the neurochemistry of the brain 16 and genotoxicity in endothelial cells. 17 Amara et al. showed that due to cadmium exposure the antioxidant enzyme activity reduced in rat hippocampus and cortex. 18
The mechanism of toxic and neurotoxic effects of cadmium could be due to oxidative stress, 19 free radicals generation, mitochondria membrane depolarization 20 and of reactive oxygen species (ROS) generation, 9 altering genes expression and eventually inducing apoptosis. 21 ROS is a regulator of cell death. ROS can activate phosphatases and protein kinases, activate or inactivate transcription factors, altering gene expression in cells and apoptotic cell death. 22 However, the mechanism of action of cadmium-induced apoptosis is still unclear.
Apoptotic cell death occurs in response to environmental stimuli. Regulation of apoptosis is important for treatment of cancer, normal growth and also for development and embryogenesis.
23
The studies of human brain tissue and experimental animal models have been shown that the
Memory dysfunction and learning ability destruction occur in the hippocampus and cortex due to neuronal apoptosis. 26 In particular, stress induced neuronal death, possibly through apoptosis, 27 and apoptosis-associated molecules can be involved in neurodegenerative diseases of the central nervous system such as Alzheimer’s disease and Parkinson’s disease. 28 An acute intraperitoneal injection of cadmium causes behavioural, biochemical and neurochemical dysfunctions in a dose-dependent manner. 29
Thus, in this study, the exposure to cadmium was examined to assess messenger RNA (mRNA) expression of
Materials and methods
Animals and experimental groups
Experiments were conducted on 28 male Wistar rats weighing 200–250 g at 8 weeks of age, and the rats were procured from Veterinary Medicine of Tehran University (Iran). Animals were housed at 22°C ± 3°C and 12-h/12-h light/dark cycle in the animal house of Parand Islamic Azad University and fed rodent chow and water. After 2 weeks of adaptation to the new environment, they were randomly allocated into four groups of seven each: a control and three experimental groups. All experiments conformed to guidelines of the ethical committee of Parand Islamic Azad University.
Cadmium nitrate administration
Cd(NO3)2 (cadmium nitrate) solution was purchased from Kimia Pars, Inc. (Merck, Germany). The dose of cadmium was chosen according to previous research. 30 –33 Injections were performed intraperitoneally with a final volume of 1 cc for each dose. The control group received saline (vehicle of cadmium) and the experimental groups were administrated cadmium concentrations of 1, 2 and 4 mg/kg body weight for 15 consecutive days. One day after the last injection, the rats were deeply anaesthetized with chloroform and rapidly decapitalized. The brains were dissected and placed on an ice-cold cutting board. After removal of the meninges, hippocampus and frontal cortex were extracted, snap frozen in liquid nitrogen and stored at −70°C until further tests.
RNA extraction and complementary DNA synthesis
Total RNA of hippocampus and cortex tissue were isolated using the RNX-TM plus (CinnaGen Inc., Tehran, Iran). The quantity and purity of the extracted RNA was determined using a spectrophotometer (NanoDrop ND-2000, NanoDrop Technologies; Wilmington, Delaware, US), and only the extracted RNAs with an A260/A280 ratio ranging from 1.8 to 2.0 were used for complementary DNA (cDNA) synthesis. Real-time transcription was performed with 1 µg of RNA and a first strand cDNA synthesis kit (Fermentas; Thermo Scientific, Waltham, MA, USA), according to the manufacturer’s instructions.
Real-time quantitative PCR using SYBER Green
Real-time PCR was used to evaluate the quantitative expression of mRNA for
Primer sequences used for real-time PCR.
Caspase-3/7 activity assay
The apoptosis assessments in hippocampal and cortex cells were carried out using the caspase-Glo 3/7 luminescent assay system (Promega). The cell lysates were prepared by cytosolic fractionation method. 35 Briefly, Caspase-Glo® 3/7 reagent (5 µL) was added to equal total protein concentrations of control group cells and experimental groups’ cells of hippocampal and cortex, followed by gentle mixing for 30 s. Incubation was followed for 30 min. The luminescence was measured by a luminometer for each sample (Berthold Detection System, Germany). 35
Data analysis
The data collected from the experiment was recorded and analyzed using SPSS 22 statistical software package. The results are presented as the mean ± standard deviation. Statistical significance of differences throughout this study was assessed using one-way analysis of variance (Tukey’s test). A
Results
Melting curve analysis for real-time PCR products obtained with the specific primer pairs for

Melting curve analysis of real-time PCR for
Figure 2 shows the effect of cadmium on the expression of

Effect of cadmium at doses 1, 2 and 4 mg/kg (body weight) on expression of
Figure 3 shows the effect of cadmium on the expression of
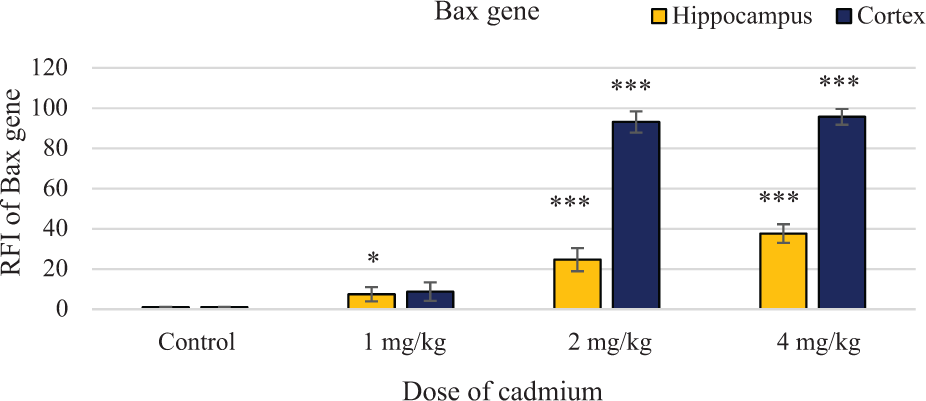
Effect of cadmium at doses 1, 2 and 4 mg/kg on expression of
As Table 2 shows, in hippocampus,
The ratio of

mRNA: messenger RNA.
a
b
In apoptosis, the critical caspases are caspase-3/7, the activities of which have been detected in different cells. In this study, caspase-3/7 activation capability was evaluated in hippocampus and cortex cells (Figure 4). In rat hippocampus, cadmium increased the activity of caspase-3/7 at doses of 1, 2 and 4 mg/kg by nearly 3.9, 2.43 and 2.43 times, respectively, compared to the control group. In rat cortex, cadmium increased caspase-3/7 activity at doses of 1, 2, and 4 mg/kg by nearly 1.43, 1.02 and 1.86 times, respectively, compared to the control groups. As illustrated, in hippocampus cells, cadmium causes a statistically significant increase in caspase-3/7 activity at doses of 1, 2 and 4 mg/kg (
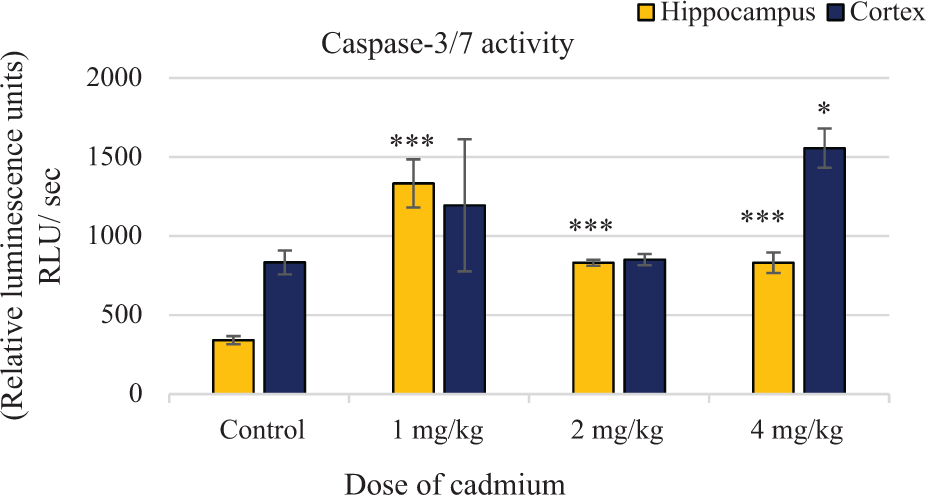
Cadmium-induced activation of caspase-3/7 in the rat hippocampus and cortex cells. Apoptosis was assessed by measuring caspase-3/7 activity using the Promega caspase-3/7 assay kit. Data are expressed as mean ± SD of ratio of treated rats to sham controls (
Discussion
Despite several studies investigating the neurotoxic effects of cadmium, the data on underlying mechanism remain not completely understood. The purpose of this study was to evaluate the neurotoxic effects of cadmium on rat hippocampus and cortex through assessment caspase-3/7 activity and the expression of genes involved in apoptosis. The present study showed that cadmium exposure leads to decreases in mRNA expression of anti-apoptotic
We used quantitative real-time PCR to determine changes in the mRNA levels apoptotic genes of
One of the targets of cadmium is the brain. 13,14 Several mechanisms are involved in cadmium-induced toxicity in the brain. One of the mechanisms is changing permeability of vascular endothelium in nerve cells. 36 Cadmium crosses the BBC by disruption in BBC. 9,12 Cadmium increases the permeability of the BBB in rats 11,32 and accumulates in the brain of developing and adult rats. 37,38 Moreover, cadmium decreased enzymatic antioxidants levels in the frontal cortex tissue. 32 In various organs, cadmium induces heavy metal-binding proteins such as metallothionein (MT). MT is a cysteine-rich protein and high metal affinity. MT has an important role in the metabolism of cadmium, which is expressed in the brain. A protective mechanism against cadmium-induced neurotoxicity is MT. Cadmium exposure induces MT deficiency and Alzheimer’s disease, 39 and it also causes behavioural and neurochemical dysfunctions. 31
Based on multiple studies, the possible mechanisms of cadmium-related toxicity include change in gene expression, repress of expression of cell cycle-regulated protein, 40 and alter of the activity of antioxidant enzymes, 18 stimulation of oxidative stress and production of free radical and oxidative damages of lipids, proteins and DNA in humans and animal. 41 Moreover, cadmium changes pro-apoptotic gene expression and DNA repair-related genes. 40
Several studies have shown mechanisms of cadmium-induced toxicity in production of free radicals and generation of ROS that result in damage of mitochondrial and apoptosis induction. 42 Moreover, damage of mitochondria can produce ROS and apoptosis induction. 11 On the other hand, heavy metals such as cadmium affect the mitochondrial function 43 and increases ROS production.
In neural tissues, cadmium as a neurotoxic metal induces histopathological damage. 36 In experimental and clinical conditions, cadmium is demonstrated to induce oxidative stress which initiates cell damage and neurodegenerative processes in the brain. 44 Cadmium has neurotoxic effects on the hippocampus, parietal cortex, striatum and cerebellum of rats. 18,45 In the brain, free radicals can potentially cause damage to neurons. 11 On the other hand, oxidative stress observed especially in the frontal cortex and hippocampus of rats exposed to cadmium may result from the increased production of free radicals. 18
Previous studies have shown that cadmium-induced apoptosis is inhibited by overexpression of the anti-apoptotic protein Bcl-2 in mammalian cells. 46,47 The Bcl-2 protein family induces programmed cell death by mitochondrial pathway of apoptosis which is also known as intrinsic. In response to various cytotoxic stresses, pro-apoptotic proteins begin to release apopotogenic factors such as cytochrome c into the cytosol and promote caspase activation in the cytosol which are are known in both initiation and execution of apoptosis. 48
It has been known that overexpression of
In this study, we demonstrated that cadmium decreases the expression of
As we report here, increase in caspase-3/7 activity and alteration in the ratio of
Conclusion
In conclusion, the results of the current study showed that intraperitoneal administration of cadmium increases the expression of pro-apoptotic
Footnotes
Author’s Note
The part of the results presented in this article was based on a student’s thesis work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
