Abstract
The paucity of specific feline antibodies for flow cytometry (FC) is an ongoing challenge. Flow cytometrists must extrapolate information from relatively few markers. We evaluated the expression pattern of the panleukocyte markers CD18 and CD44 on leukocyte (white blood cell, WBC) subclasses in the peripheral blood (PB) of 14 healthy cats. The degree of expression of CD18 and CD44 was calculated as the ratio between the median fluorescence intensity (MFI) value of antibody-stained cells and autofluorescence. All samples were acquired with the same cytometer with constant photomultiplier setting and compensation matrices. Both molecules were expressed at higher levels on monocytes, intermediate levels on polymorphonuclear cells (PMNs), and lower levels on lymphocytes. CD18-MFI discriminated well among the 3 populations, whereas CD44-MFI mostly overlapped between monocytes and PMNs. However, CD44-MFI had a lower intra-population variability. Evaluation of CD18 and CD44, together with morphologic parameters, was useful for discriminating among WBC subclasses in healthy cats. This information may be helpful for future studies given that an increase in CD18-MFI may indicate reactive changes, whereas fluctuations in CD44-MFI may suggest neoplasia.
Flow cytometry (FC) in feline veterinary medicine is decades behind human and canine medicine, although our research group has demonstrated that most feline samples are suitable for FC analysis. 9 The diagnostic accuracy of FC in discriminating between neoplastic and non-neoplastic lymphoproliferative disorders has been demonstrated in cats. 8 However, feline FC still suffers from a paucity of commercial antibodies that cross-react in this species. Thus, flow cytometrists attempt to gain as much information as possible by analyzing only a few molecules. The degree of expression of panleukocyte markers helps in identifying the white blood cell (WBC) subpopulations in the peripheral blood (PB) from healthy dogs, and to characterize hematologic neoplasms in dogs.4,5,7
We describe herein the pattern of expression of 2 different panleukocyte markers, CD18 and CD44, on WBC subpopulations in the PB of healthy cats, to provide basic data useful for future studies on diseased animals. We selected these 2 antigens for our study because of their potential diagnostic utility.
CD18 is a component of β2 integrins, which are adhesion molecules involved in leukocyte extravasation. CD18 is expressed on the cell surface of all WBC subclasses, with variable levels of expression according to cellular activation and differentiation status, and is primarily involved in leukocytes rolling on the endothelium and their subsequent diapedesis. 12 Accordingly, analyses of the degree of expression may prove useful in reactive conditions.
CD44 is a hyaluronan receptor that is expressed ubiquitously on the cell surface and is involved in many processes requiring interaction with the extracellular matrix. This molecule is considered a cancer stem cell marker and has been studied extensively because of its role in tumorigenesis and development of metastasis. 11 Thus, analyzing its degree of expression may prove useful in neoplastic conditions.
We analyzed PB samples collected into EDTA tubes from 14 healthy cats. All samples were delivered to the laboratory and processed within 2 h of sampling. All cats were privately owned and sampled as part of routine health examinations at the Veterinary Teaching Hospital of the University of Milan, Italy. As per policy at this institution, specific approval from the Ethical Committee is not required for research use of leftover diagnostic specimens (EC decision 29 October 2012, renewed with protocol 02-2016).
A complete blood count was performed with an automated hematology analyzer (XT 2000-iV; Sysmex), and sample processing for FC was performed according to protocols published previously. 9 One tube served as negative control (unstained cells); 3 other tubes were investigated by means of the following antibody (Ab) cocktails: CD5/CD21/CD18, CD4/CD8/CD18, and CD44/CD18. To assess cell viability, propidium iodide (PI; GeneTex) was included in the third tube. Ab clones and fluorochromes (Table 1) have already been documented to stain feline samples by FC.3,9,10 All antibodies, as well as PI, had been titered before use to determine the best working dilutions. All samples were evaluated immediately after staining by means of a flow cytometer (BriCyte E6; Mindray) equipped with 2 lasers and detectors for up to 4 fluorescence channels. The cytometer status was checked and, if needed, calibrated at the beginning of each laboratory session by means of specific controls (SPHERO Supra rainbow fluorescent particles mid-range; Spherotech). Photomultiplier (PMT) voltages and compensation matrices were kept constant during the entire experiment. For each tube, 10 × 103 nucleated cells were evaluated.
Antibodies used for flow cytometric analysis of peripheral blood samples from 14 healthy cats.
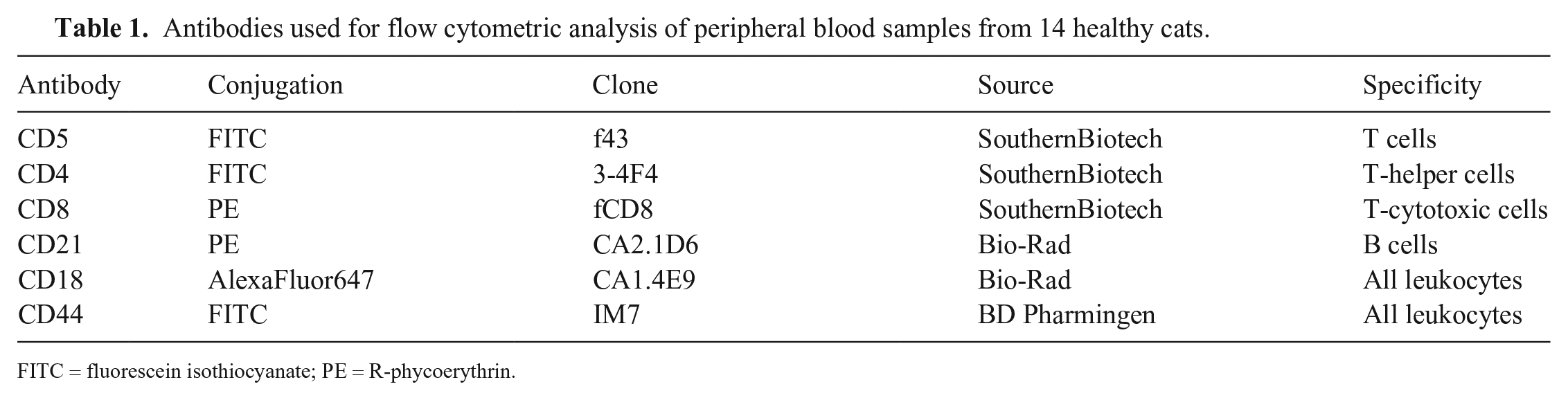
FITC = fluorescein isothiocyanate; PE = R-phycoerythrin.
Analyses were performed by means of specific software (MR Flow; Mindray) by a single operator (V. Martini) in a single session. The percentage of PI-positive (necrotic) cells was regarded as an index of sample viability. Outlier samples with an excessively high percentage of PI-positive cells were excluded from the study. The Dixon method was used to make this determination. 6
For each sample, the median fluorescence intensity (MFI) of unstained cells (FL-1 and FL-4 channels), CD18, and CD44 was recorded separately for polymorphonuclear cells (PMNs), monocytes, and lymphocytes, via a back-gating strategy based on cellular morphologic properties (forward scatter–height [FSC-H] vs. side scatter–height [SSC-H]). PMNs were considered as a whole, without subgrouping into neutrophils, eosinophils, and basophils, because morphologic or phenotypic properties were not available to distinguish the 3 subpopulations by FC. CD18-MFI and CD44-MFI were calculated for each population by dividing the MFI value of Ab-stained cells by the MFI value of unstained cells in the corresponding fluorescence channel.1,7 Statistical analyses performed included a Shapiro–Wilk test to assess normal distribution of data, a Friedman test to assess possible differences among WBC subclasses in CD18- and CD44-MFI, and a Wilcoxon signed-rank test for post-hoc analyses. Significance was set at p ≤ 0.05.
Both CD18- and CD44-MFI varied significantly among WBC subclasses (p < 0.001 for both analyses; Table 2). Monocytes had the highest level of expression of both CD18 and CD44; lymphocytes had the lowest expression for both antigens. CD18-MFI was 5-fold higher in monocytes than in PMNs (mean CD18-MFI ratio between monocytes and PMNs = 5.6 ± 3.7; p = 0.001), 8-fold higher in PMNs than in lymphocytes (mean CD18-MFI ratio between PMNs and lymphocytes = 8.4 ± 7.7; p = 0.001), and 34-fold higher in monocytes than in lymphocytes (mean CD18-MFI ratio between monocytes and lymphocytes = 34.1 ± 30.5; p = 0.001). CD44-MFI did not differ between monocytes and PMN (mean CD44-MFI ratio = 1.1 ± 0.3; p = 0.196) and was 2-fold higher in PMNs and in monocytes than in lymphocytes (mean CD44-MFI ratio = 2.7 ± 1.2 and 2.9 ± 0.9, respectively; p = 0.001 for both analyses).
Expression of CD18 and CD44 antigens on the cell surface of circulating leukocyte subclasses from 14 healthy cats, as determined by fluorescence analyses on flow cytometry. CD18-MFI and CD44-MFI were calculated for each population by dividing the median fluorescence intensity (MFI) value of antibody-stained cells by the MFI value of unstained cells, in the corresponding fluorescence channel.
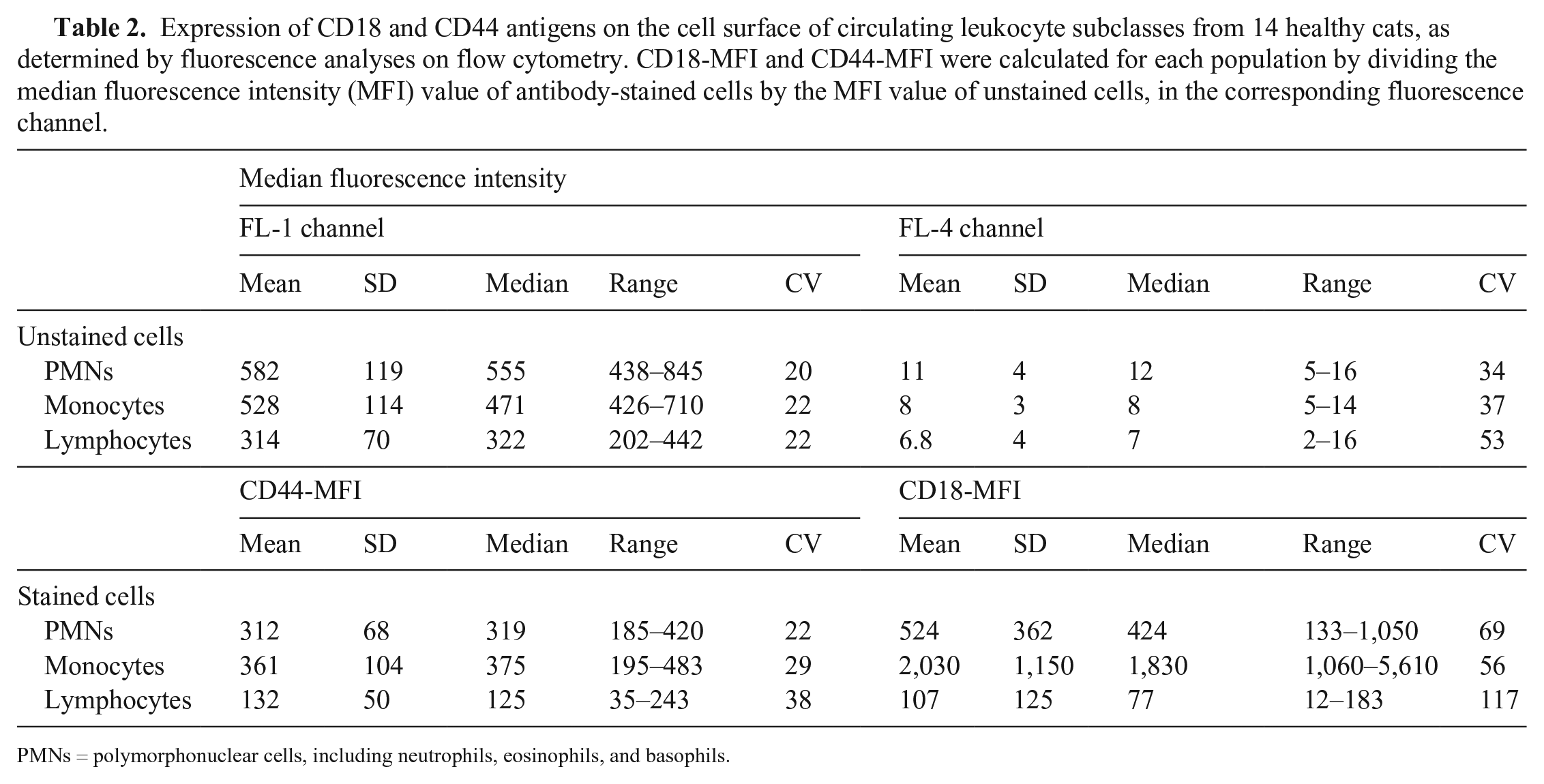
PMNs = polymorphonuclear cells, including neutrophils, eosinophils, and basophils.
Although both markers stained all leukocytes, analysis of the level of expression in the different cell populations allowed differentiation among the leukocyte groups. Both proteins are expressed at higher levels on monocytes than on PMNs and lymphocytes. However, CD18-MFI allows better discrimination than CD44-MFI among the 3 subclasses, as documented by the higher ratios obtained when coupling CD18-MFI on monocytes with either CD18-MFI on PMNs or lymphocytes. Despite the higher mean and median CD44-MFI shown by monocytes compared with PMNs, differences were not significant, thus complicating the discrimination between these 2 classes based on fluorescence level. As a result, lymphocytes are easily identified in a dot plot coupling the intensity of fluorescence of CD18 and CD44 as a discrete population with low intensity of fluorescence of both antigens. Conversely, monocytes are located at the edge of a smear with homogeneous CD44-MFI, without a clear separation from the PMN cloud (Fig. 1). Monocytes and PMNs are more easily discriminated by coupling CD18-MFI with a complexity index (SSC-H); this type of scattergram seems to be the most appropriate to distinguish among WBC subclasses in cats. Unfortunately, CD18-MFI suffers from a great variability within each WBC subclass, as documented by our results; monocytes had the lowest CV for CD18-MFI (>50%), and a peak of >100% was reached for lymphocytes, whereas the CV for CD44-MFI was consistently <40%.
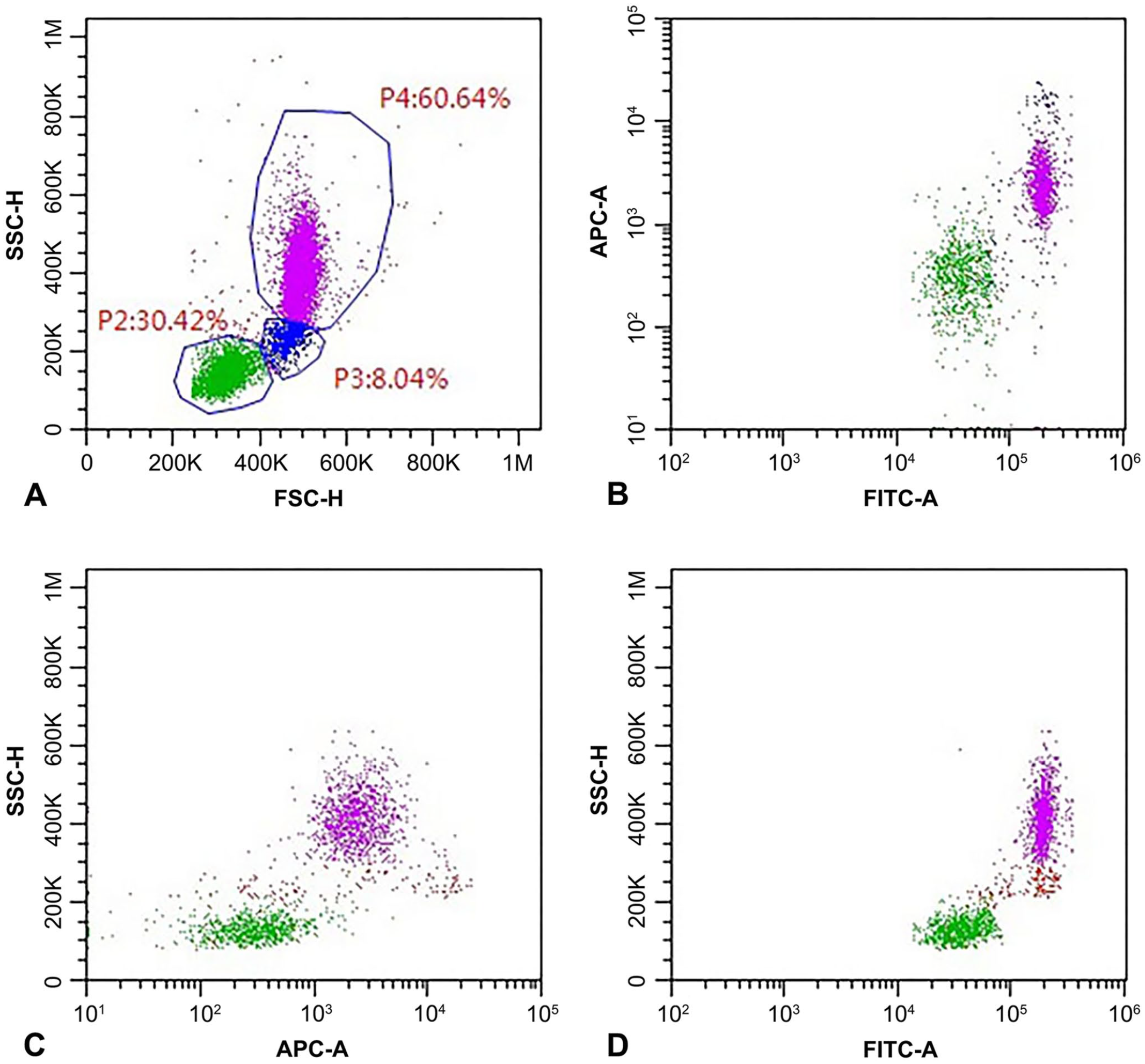
Flow cytometric analysis of a peripheral blood sample from a healthy cat. A gate (P1) was set to exclude platelets and debris in a morphologic scattergram (forward scatter–height [FSC-H] vs. side scatter–height [SSC-H]; Suppl. Fig. 1).
Unfortunately, we were not able to assess possible differences in panleukocyte marker expression among different lymphoid subclasses, because of conflicting combinations of Abs and fluorochromes (CD44 was FITC labeled, as well as CD5 and CD4, thus preventing concomitant assessment of these molecules) and because of the low number of cells within each lymphoid subclass. Interestingly, CD expression on lymphocytes was normally distributed, with a single peak on histogram (Suppl. Fig. 1); different levels of expression between B and T cells is therefore unlikely because 2 different peaks would have resulted.
Limitations of our study are the small number of samples analyzed and the lack of isotypic controls. Most studies have relied on isotypic controls to set fluorescence background; however, this approach has been demonstrated to be misleading. 2
Supplemental Material
Supplemental_material – Supplemental material for Flow cytometry expression pattern of CD44 and CD18 markers on feline leukocytes
Supplemental material, Supplemental_material for Flow cytometry expression pattern of CD44 and CD18 markers on feline leukocytes by Valeria Martini, Serena Bernardi, Alessia Giordano and Stefano Comazzi in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the staff of the Veterinary Teaching Hospital for providing samples for our study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Flow cytometry diagnostic service income was used to pay for laboratory materials, reagents, and cytometer use.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
