Abstract
Cosmetic ingredients derived from Camellia sinensis (tea) plant parts function as antioxidants and skin conditioning agents—humectant and miscellaneous. The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) reviewed relevant animal and human data related to these ingredients. The use of the leaf ingredients in beverages results in much larger systemic exposures than those possible from cosmetic use. Accordingly, concern over the systemic toxicity potential of leaf-derived ingredients is mitigated. Because product formulations may contain multiple botanical ingredients, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may lead to sensitization or other toxic effects. The Panel concluded that the C sinensis leaf-derived ingredients are safe in the present practices of use and concentration described in this safety assessment when formulated to be nonsensitizing. However, the available data are insufficient to determine whether the non-leaf-derived ingredients are safe for use in cosmetics.
Introduction
This is a safety assessment of cosmetic ingredients derived from Camellia sinensis (tea) plants. The reported functions of these ingredients include antifungal agent, antimicrobial agent, antioxidant, cosmetic astringent, fragrance ingredient, light stabilizer, oral care agent, skin protectant, skin conditioning agent—emollient, skin conditioning agent—humectant, and skin conditioning agent—miscellaneous (Table 1). 1 The 14 ingredients in this report are as follows:

Definitions and Functions of Camellia sinensis–Derived Ingredients in This Report. 1

Camellia sinensis seed oil was included in a 2011 Cosmetic Ingredient Review (CIR) safety assessment of plant-based oils with the conclusion that it was safe in the present practices of use and concentration. 2
Some of the C sinensis–derived ingredients in this safety assessment are used to make tea. Exposures to the leaf-derived ingredients in beverages results in much larger systemic oral exposures than would be possible from cosmetic use. Therefore, though oral data are included in this report, the systemic toxicity potential of the leaf-derived cosmetic ingredients is mitigated and thus not a primary focus of this report. While data on the potential for reproductive toxicity, genotoxicity, and carcinogenicity are presented, the primary focus of this report is on the potential for irritation and sensitization for leaf-derived ingredients.
Due to the nature of plant-derived ingredients, there is variation in the constituent content of these botanical ingredients. For example, the definition of C sinensis catechins does not provide any limitation on which catechins or what quantities of catechins are in the ingredient. Therefore, all studies of catechins are listed under the ingredient Camellia Sinensis Catechins.
Chemistry
Definition and Description
The definitions and functions of C sinensis-derived ingredients are provided in Table 1.
Camellia sinensis
There are 4 varieties of the C sinensis plant: sinensis, assamica, pubilimba, and dehungensis. The first 2 are most commonly used to prepare tea for human consumption. The type of tea (ie, white, green, oolong, black) depends on time of year harvested, age of leaves when harvested, location/soil/climate, and processing after harvest. The processing of tea for a beverage is referred to as fermentation because it was originally believed that the leaves were fermented, but the process actually involves an enzymatic oxidation. 3,4
The C sinensis plant is native to East, South, and Southeast Asia. 4 -6 However, it is also cultivated in other tropical and subtropical regions. The leaves of this evergreen shrub can be lanceolate to obovate, up to 30 cm long (usually 4-15 cm) and 2- to 5-cm broad, pubescent, and sometimes become glabrous, serrate, acute, or acuminate. The plant has a strong taproot. The 3- to 5-cm, yellow/white flowers are globular and have a delicate fragrance. These plants are neither the source of nor are they related to tea tree oil, which is derived from Melaleuca alternifolia.
Constituents
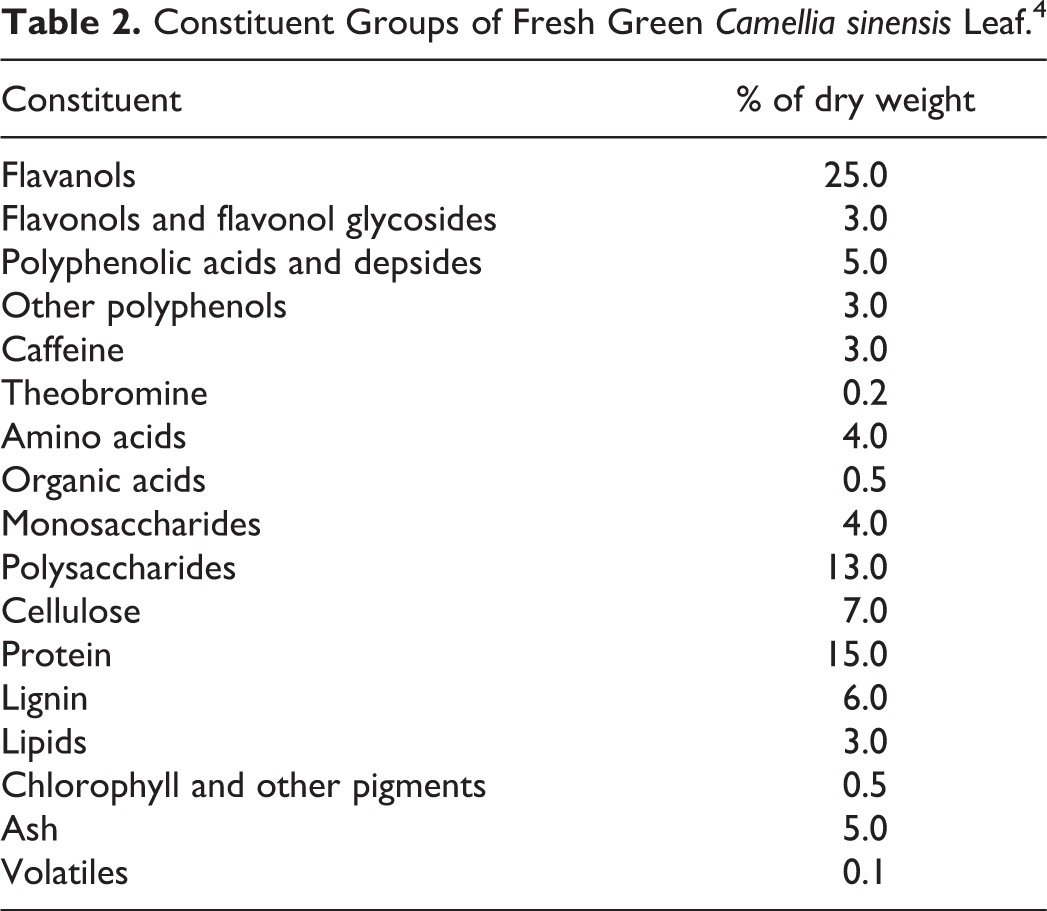
The constituent groups of fresh green leaf C sinensis are provided in Table 2. The constituent group having the highest concentrations is the flavanols (25.0% dry weight), which is followed by proteins (15.0%) and polysaccharides (13.0%). 4
Constituent Groups of Fresh Green Camellia sinensis Leaf. 4
Constituent groups found in C sinensis plant parts include: Amino acids—The most abundant amino acid is one not typically found in proteins, theanine (5 N ethylglutamine).
4,7
Carotenoids—These are present in low levels in the leaves. They include neoxanthin, violaxanthin, lutein, chlorophylls a and b, and β-carotene.
4,8,9
Seventy-nine pigments, 41 chlorophylls, and 38 other carotenoids have been detected.
10
Enzymes—Fresh C sinensis leaves contain high levels of the enzyme polyphenol oxidase. Methylxanthines—Theobromine can range from 0.16% to 0.2% of a dry-weight leaf.
4,11,12
Dried leaves contain not less than 2% caffeine (dried weight). Increased use of nitrogen fertilizer can increase caffeine content by up to 40%. Theophylline is present at <0.04% dry leaf weight. Flavonoids—These include flavonols, flavanols, and glycosides. Flavanols include catechins, which are present in small amounts, and may occur as flavonols and glycosides.
4,13,14
Flavonols reported to be in leaf extract are kaempferol, quercetin, and myricetin.
4,15
Catechins—These polyphenolic molecules are a subgroup belonging to the flavanol family.
4,16
-18
They typically make up 20% to 30% of the weight of tea leaves. Catechins are especially concentrated in the leaves of green tea, wherein they account for 30% to 40% of the dry weight of the leaves. The most abundant type of catechin in green tea is epigallocatechin gallate (EGCG; 12%). The other catechins are catechin (C), epicatechin (EC), gallocatechin (GC), epigallocatechin (EGC), catechin gallate (CG), gallocatechin gallate (GCG), and epicatechin gallate (ECG; Figure 1). Minerals elements—Potassium is the most abundant mineral element, present at 40% of the total mineral element content of dry matter of fresh leaves. The leaves are rich in fluoride and they also accumulate aluminum and manganese.
4,19,20
Other elements present in mineral form include calcium, magnesium, sodium, phosphorus, and sulfur. Minor elements include boron, barium, chromium, copper, iron, molybdenum, nickel, rubidium, strontium, and zinc.
19
Trace elements include silver, arsenic, beryllium, bismuth, cadmium, cerium, cobalt, cesium, mercury, indium, lithium, lead, rare earth elements, antimony, selenium, tin, tellurium, thallium, uranium, vanadium, ytterbium, and zirconium. Volatiles—There are a large number of volatile constituents in fresh leaves; trans-2-hexenal and cis-3-hexenol are present in the greatest amounts.
4,14,21,22
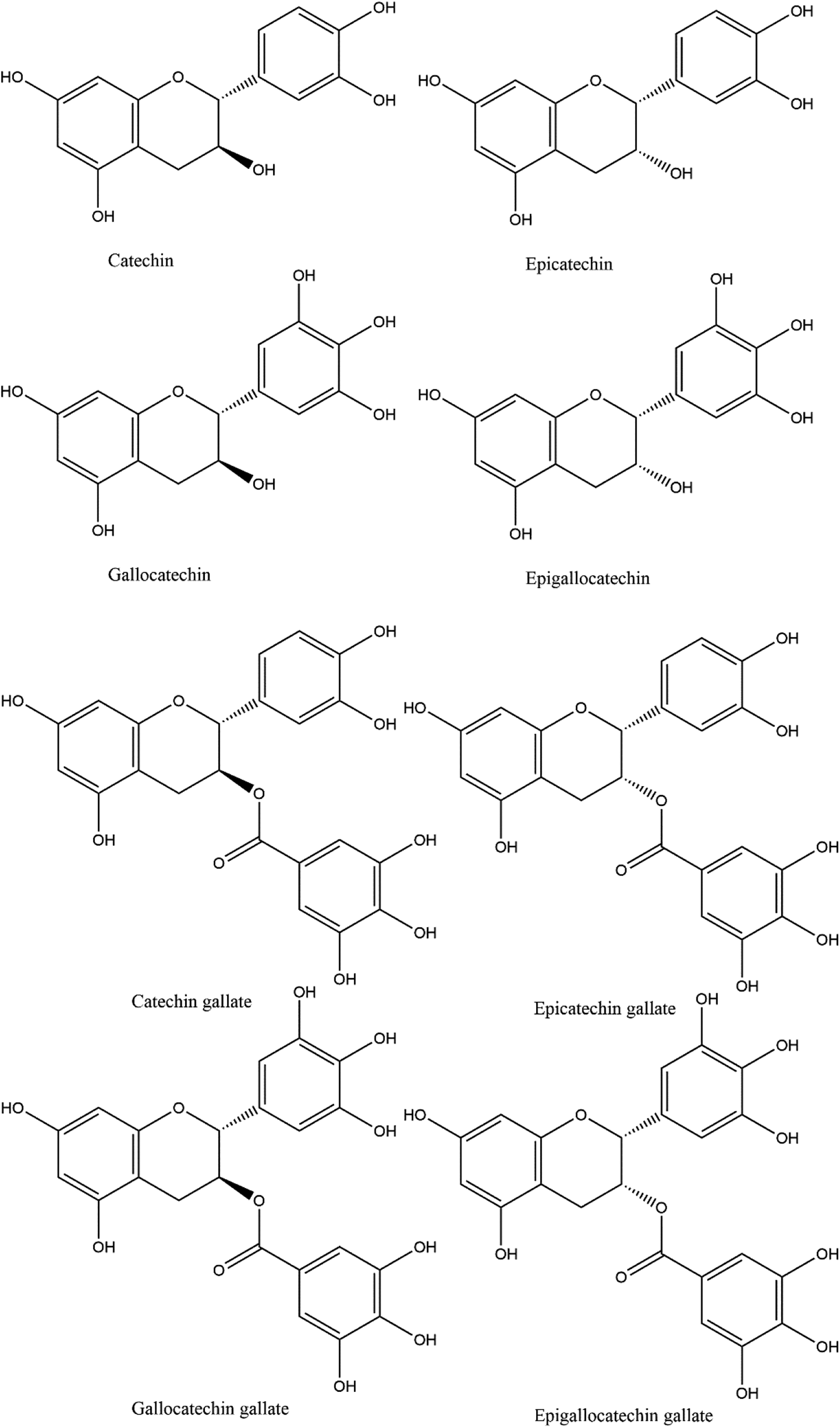
Catechins from Camellia sinensis. 32
Constituents reported to be predominately in the seeds of C sinensis include caffeine, glucothea saponin, stearic acid, theasaponin, and theobromine. 23
Climatic conditions during cultivation may affect the content of theanine, common proteinaceous α-amino acids (ie, isoleucine, leucine, valine, alanine, threonine, and glutamine), quinic acid, EC, EGC, EGCG, and caffeine levels in Camellia Sinensis Leaf Extract (as green tea). 24,25 Soil conditions and cultivation methods may markedly affect mineral levels. 19
The presence of minerals and elements in an extract depends on the extent of entrapment in the organic matrix, the degree of solubility/choice of solvent, the duration of extraction, temperature, pH, and agitation. 19 Most elements, especially the metals, are complexed with the flavonols, catechols, tannins, and polyphenols.
Constituents of Concern
Linalool and several compounds containing the linalool moiety (ie, R-linalool, linalool-oxide-(cis-furanoid), linalool-oxide-(cis-pyranoid), linalool-oxide-(trans-pyranoid), linalool-β-
Constituents of Concern in Camellia sinensis.

Sample Analysis
Constituents in a Camellia Sinensis Extract include methylxanthines, flavanols (10%-25%), flavonols, flavones, phenolic acids, amino acids (including theanine, 3%), terpene saponins, polysaccharides, proanthocyanidins, vitamins, and minerals (Table 4). 16,27 -31
Analyses of 3 lots of Camellia Sinensis Catechins (each prepared as a dietary supplement) indicated 27.6% to 62.3% total catechin monomers and 30.6% to 67.4% polyphenols. 32 Analysis for other components showed: caffeine (≤7%), organic acids (≤10%), protein and amino acids (≤10%), saccharide (≤12%), fiber (≤1%), fat (≤1%), and ash (≤5%).
Characterization
As herbal supplements, extracts are characterized by the drug/extract ratio (DER), which is the ratio of the quantity of herbal substance used in the manufacture of an herbal preparation (given as a range) to the quantity of the herbal preparation obtained in the finished product. 33 The specifications for Camellia Sinensis Folium as a herbal supplement in the European Union for the dry extract, purified (DER 45-56:1, extraction solvent:water) corresponds to 55% to 72% () epigallocatechin-3-O-gallate. 27,33 The decaffeinated dry extract (DER 6:1-10:1, solvents such as alcohol, methanol, acetone, or water or mixtures of these solvents) contains not less than 60% of polyphenols, calculated as () epigallocatechin 3-O-gallate, not less than 40% of (−)-epigallocatechin-3-O-gallate and not more than 0.1% of caffeine, calculated on the anhydrous basis.
Impurities
No published data on impurities of these cosmetic ingredients were discovered, and no unpublished data were submitted. The information below applies to impurities found in C sinensis as a food or as food ingredients.
In analyses of 12 Camellia Sinensis Catechins lots extracted as food ingredients, arsenic (<0.2 ppm), cadmium (<0.1 ppm), lead (0.4 ppm), and tin (<150 ppm) were below levels of detection. 32 Three lots of Camellia Sinensis Catechins were analyzed and no microbial contamination was detected.
Ten commercial C sinensis teas for drinking were analyzed for metals. 34 The ranges for metal content were: zinc 1.05 to 3.21 mg/kg; iron, 5.47 to 8.41 mg/kg; manganese, 1.27 to 2.73 mg/kg; copper, 0.01 to 0.93 mg/kg; nickel, 0.01 to 0.64 mg/kg; lead, 0.26 to 1.25 mg/kg; and cadmium, 0.01 to 0.05 mg/kg. The authors asserted that differences in content of the samples were attributable to differences in geographic region of cultivation.
Aflatoxigenic molds and aflatoxins have been reported to be present on C sinensis teas for drinking. 35 In a sampling of 27 commercial black teas (7 branded, 20 nonbranded), aflatoxigenic molds were detected in 1 branded and 6 unbranded (25.9%) tea samples. Only 1 of the samples (nonbranded) had detectable aflatoxins (19.2 µg/kg). In black teas that had been spiked with aflatoxins, most of the aflatoxin residue was still present in the leaves after boiling in water; 30.6% was present in the final beverage.
It was reported that levels of 712 to 1,530 µg/g, 166 to 280 µg/g, 1.7 to 7.5 µg/g, and 1.51 to 2.63 µg/g tea of aluminum, iron, chromium, and lead, respectively, were found in commercial tea samples (n = 2) using electrothermal atomic absorption spectrometry and inductively coupled plasma atomic emission spectrometry (Table 5). 36 These samples came in powder or granular forms. For 2 types of green tea, the ranges were 605 to 620 µg/g, 1,486 to 1,550 µg/g, 4.5 to 4.7 µg/g, and 2.20 to 2.34 µg/g, respectively. For the infusions of these branded and unbranded teas (exact extraction method not provided), the levels for aluminum, iron, copper, and zinc were 149 to 367 µg/g, 7.6 to 11.0 µg/g, 0.7 to 3.2 µg/g, and 36 to 43 µg/g, respectively. For green tea infusions, these values were 124 to 127 µg/g, 22 to 23 µg/g, 0.2 to 0.5 µg/g, and 31 to 32 µg/g, respectively.
Trace Elements in Commercial Teas and Their Infusions. 36,a
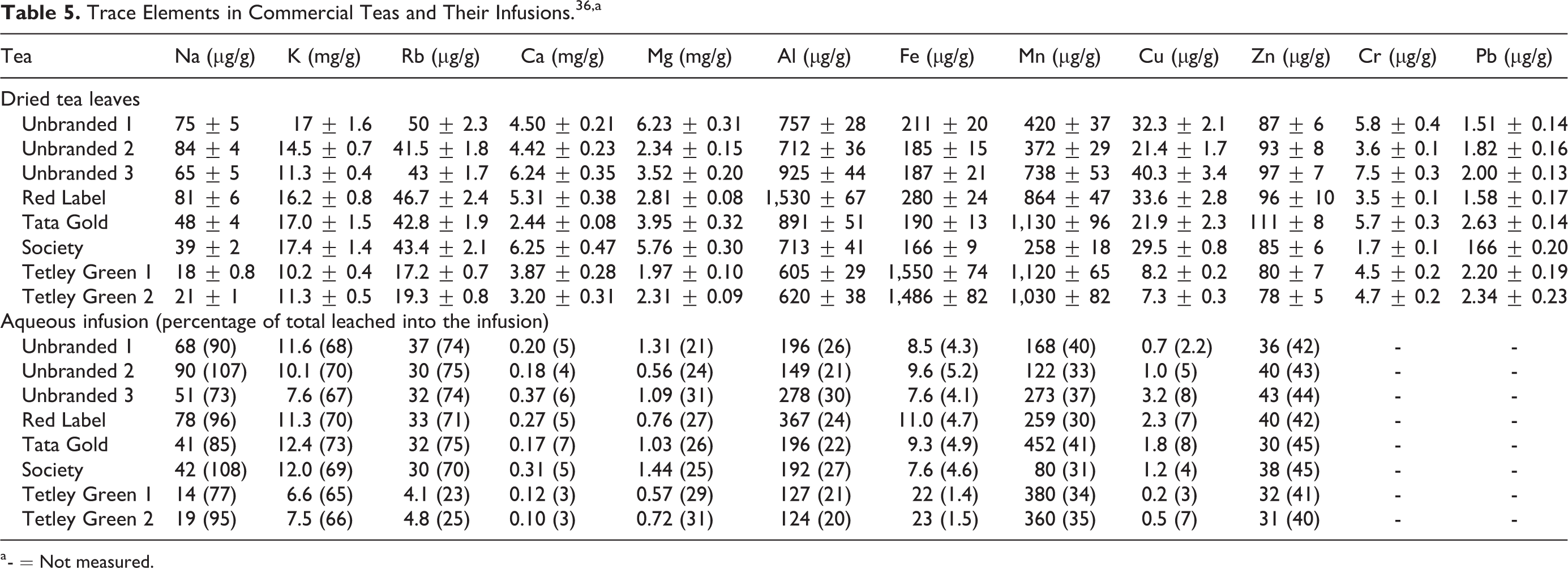
a - = Not measured.
Physical and Chemical Properties
Camellia Sinensis Catechins have an astringent taste and are soluble in water. 4,16 Three lots of oolong tea with Camellia Sinensis Catechins were stable for 18 months in unopened packaging at −20°C. 32 After 6 months of storage at 25°C, total catechin monomers were decreased from 100% to 97% in polyethylene terephthalate bottles and to 98% in steel cans. At 37°C for 2 months, catechin content was decreased to 96% in both types of containers. A sunscreen with various percentages of C sinensis (2%-5%), in the form of green tea extract, was stable for up to 6 months. 37
Method of Manufacture
The most widely used method for preparing essential oils from plants is associated with steam distillation. 1 The condensate from steam distillation produces 2 distinct fractions that contain the volatile ingredients from the plant. The water-insoluble fraction contains the “oil.” The water-soluble fraction contains ingredients from the plant that are water soluble. The water-insoluble fraction from steam distilled plant materials is identified as “oil” in the International Nomenclature of Cosmetic Ingredients (INCI) name. The water-soluble fraction from the steam distilled plant material is identified as “water” in the INCI name.
No information on the method of manufacture for C sinensis–derived cosmetic ingredients was discovered or submitted. The methods below are general to the processing of C sinensis for food or food ingredients. The makeup of the Camellia Sinensis Extract will differ with the manufacturing process.
Camellia Sinensis Leaf in the form of green tea consists of whole or cut young, unfermented, rapidly heat-dried leaves. 17,27 The fresh leaves are processed by a method designed to prevent the enzymatic oxidation of catechins. The enzymes are inactivated by heat (pan frying or steaming).
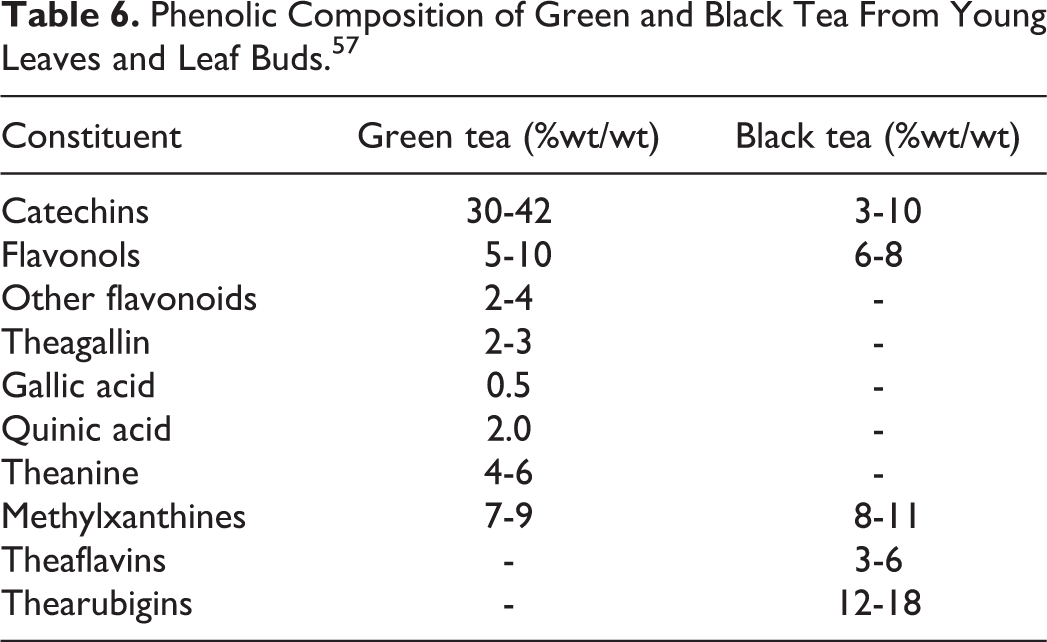
There are different harvesting and manufacturing processes for white, green, black, and oolong teas used for drinking. 17,19,27,38 White tea is made from very young leaves and leaf buds. Green tea is made from new, fully formed leaves. These 2 types of tea are minimally processed, steamed, and dried. Black tea and oolong tea are made from older, fully formed leaves. Oolong tea is withered and rolled during “fermentation,” then fired and dried. Black tea is withered, crushed, and rolled during “fermentation,” then fired and dried. Phenolic content typically differs substantially between green and black teas (Table 6).
Phenolic Composition of Green and Black Tea From Young Leaves and Leaf Buds. 57
Catechins are isolated through an initial hot water extraction with ethyl acetate and then separation by chromatography, followed by spray drying. 39 The spray-dried catechins may be recrystallized. Two other processes for the extraction of catechins from C sinensis leaves are conducted with or without enzymatic treatment with tannase. The initial extract is further extracted with water and ethanol and then filtered through multiple media. The product of the process without tannase is sterilized above 100°C, whereas the product obtained with the tannase treatment is sterilized below 100°C.
Use
Cosmetic
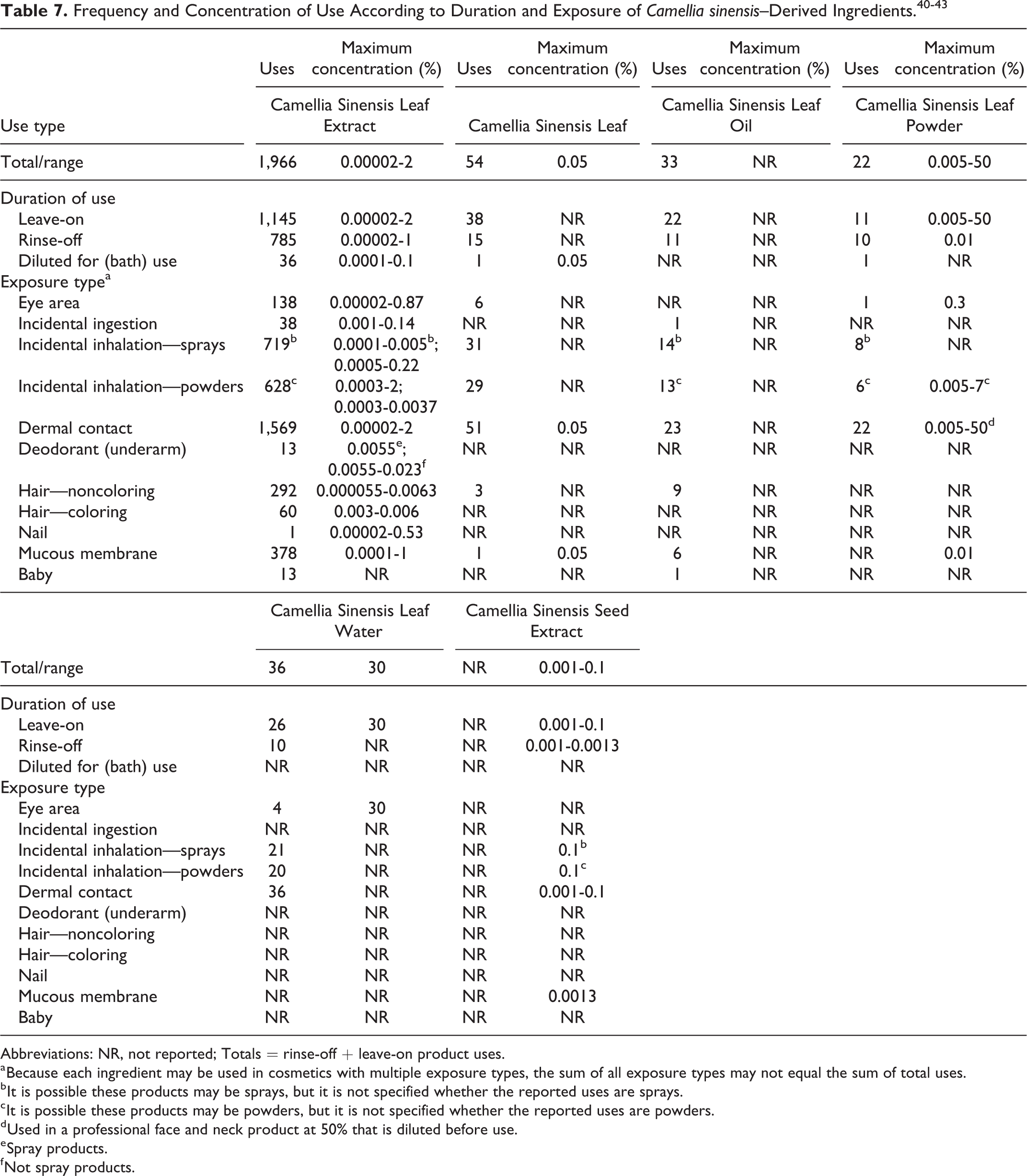
Data on ingredient usage are provided to the US Food and Drug Administration (FDA) Voluntary Cosmetic Registration Program (VCRP; Table 7).
40
A survey was conducted by the Personal Care Products Council (Council) of the maximum use concentrations for C sinensis–derived ingredients.
41,42
Data were available from both the VCRP and the Council for the following ingredients: Camellia Sinensis Leaf Extract was reported to be used in 1,145 leave-on, 785 rinse-off, and 36 bath cosmetic products.
40
There are reported uses in every exposure type (Table 7). Usage of cosmetic ingredients called “green tea” and “green tea extract” were also reported in the VCRP. Since these are technical names for Camellia Sinensis leaf extract, the VCRP numbers for these 3 listings were combined. Camellia Sinensis Leaf Extract was reported to be used up to 2% in leave-on products (the highest concentrations in face and neck products) and up to 1% in rinse-off products (the highest concentration in bath soaps and detergents).
41
It is also reported to be used in products diluted in the bath at up to 0.1% (the highest concentration in bubble baths). It is reported to be used up to 0.14% in an ingestible oral hygiene product. Camellia Sinensis Leaf was reported to be used in 38 leave-on, 15 rinse-off, and 1 bath product.
40
Camellia Sinensis Leaf was reported to be used up to 0.05% in bubble baths.
41
A previously reported product of tea bags for the eyes at 97% is no longer sold.
41,43
Camellia Sinensis Leaf Powder was reported to be used in 11 leave-on, 10 rinse-off products, and 1 bath product.
40
Camellia sinensis Leaf Powder was reported to be used in body and hand products up to 7% and up to 0.01% in rinse-off products (highest concentration in bath soaps and detergents).
41
It is also reported to be used in a professional face and neck product at 50% that is diluted before use. Camellia Sinensis Leaf Water was reported to be used in 26 leave-on and 10 rinse-off products.
40
This ingredient was reported to be used up to 30% in mascara.
41
Abbreviations: NR, not reported; Totals = rinse-off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
c It is possible these products may be powders, but it is not specified whether the reported uses are powders.
d Used in a professional face and neck product at 50% that is diluted before use.
e Spray products.
f Not spray products.
Data were available only on the frequency of use (VCRP) for Camellia Sinensis Leaf Oil. It was reported to be used in 22 leave-on products and 11 rinse-off products, including 1 baby product and 1 lipstick. 40 No concentration of use data were reported.
Data were available only on concentration of use for Camellia Sinensis Seed Extract. It was reported to be used in leave-on products up to 0.1% (highest concentration in moisturizing creams and lotions) and in rinse-off products up to 0.0013% (highest concentration in bath soaps and detergents). 41 There were no uses reported in the VCRP.
There were no frequency of use or concentration of use data reported for: Camellia Sinensis Catechins Camellia Sinensis Flower Extract Camellia Sinensis Flower/Leaf/Stem Juice Camellia Sinensis Root Extract Camellia Sinensis Seed Coat Powder Camellia Sinensis Seed Powder Hydrolyzed Camellia Sinensis Leaf Hydrolyzed Camellia Sinensis Seed Extract
Camellia Sinensis Leaf Extract was reported to be used in pump and aerosol sprays. Camellia Sinensis Leaf Extract is reported to be used up to 0.0005% in pump hair sprays, up to 0.0055% in pump deodorant sprays, up to 0.0005% in body and hand sprays, up to 0.22% in foot spray, and up to 0.07% in pump suntan product. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 44 -47 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 44,46 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 44 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Noncosmetic
The essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of tea, under the previous name Thea sinensis, are generally recognized as safe (GRAS) by the US FDA [21 CFR 582.20]. In Europe, C sinensis preparations are used to treat asthenia and as an adjuvant treatment in weight loss/control. 27 Preparations are also used in cutaneous treatment of external genital and perianal warts (Condylomata acuminata) in immune-compromised patients. 27,48
In the United States, green tea products are used as dietary supplements, primarily for purported weight loss and antioxidant properties. 17,18,49 -58 Other health benefits attributed to green tea include prevention and/or control of atherosclerosis, hypertension, coronary heart disease, diabetes, metabolic syndrome, obesity, and cancer, as well as antibacterial, antiviral, antifungal, and neuroprotective effects.
Annual tea consumption varies from country to country, ranging from negligible to approximately 3 kg per person. 59 Worldwide average consumption is approximately 0.5 kg per person.
In 2012, over 79 billion servings of tea were consumed in the United States (over 3.60 billion gallons). 60 Of this, Americans consumed approximately 84% black tea, 15% green tea, and the rest oolong and white tea.
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Dermal/percutaneous
Camellia Sinensis Catechins
When Camellia sinensis Leaf Extract (as green tea) was administered to full-thickness pig ear skin using a Franz cell, there was a dose-dependent transdermal penetration of the catechins EGCG, EGC, and EC (Table 8). 61
The Permeation of Constituents of Camellia Sinensis Leaf Extract (as Green Tea) Through Full-Thickness Pig Ear Skin Using Different Vehicles Using a Franz Cell. 61
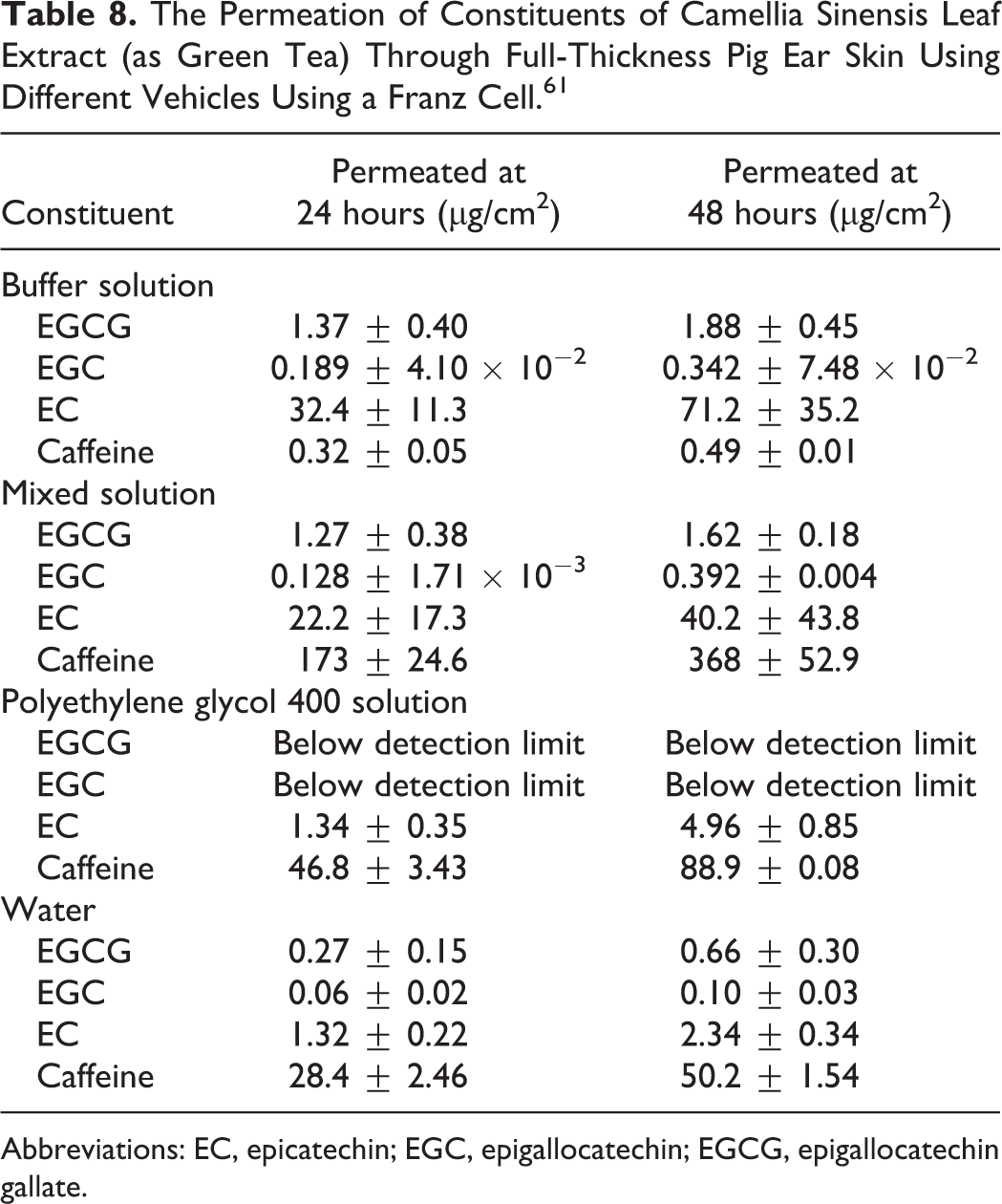
Abbreviations: EC, epicatechin; EGC, epigallocatechin; EGCG, epigallocatechin gallate.
Saturated solutions of C sinensis were formulated using water, polyethylene glycol 400, citrate/phosphate buffer (pH 5.5), and a 50:50 mixture of polyethylene glycol 400 and the buffer. The solutions were applied to drug-in-adhesive transdermal patches under occlusion in methanol and applied to the pig skin. The receptor cell was sampled periodically for 48 hours. Penetration by the catechins was fastest in the buffer solution and slowest in polyethylene glycol 400 solution. 61
When EGCG was dermally applied in a transdermal gel (50 mg/kg; 28.6 μg/cm2) to female SKH-1 mice (n = 4, 5, or 6), EGCG was detected in the skin, plasma, liver, small intestines, and colon for at least 24 hours. 62 The test material was administered once. Over the next 24 hours, blood was collected under anesthesia and dorsal skin was removed, fractioned into epidermis and dermis, and analyzed. Liver, small intestine, and colon tissues were removed and analyzed.
In the total plasma, the maximum plasma concentration (Cmax) at 6 hours was 44.5 ± 8.4 ng/mL, the half-life (t1/2) was 94.4 ± 13.2 hours, and the area under the concentration–time curve (AUC0-24) was 881.5 ± 123.4 ng/mL/h. The Cmax at 1 hour for the epidermis and dermis were 1,365.7 ± 613.8 ng/mL and 411.2 ± 21.5 ng/mL, respectively; the AUC0-24 was 5,978.3 ± 2,779.9 ng/g/h and 1,729.5 ± 259.4 ng/g/h, respectively. The t1/2 was 9.3 ± 4.3 hours and 10.9 ± 1.6 hours, respectively.
The Cmax of EGCG in the liver at 2 hours was 164.8 ± 83.0 ng/g with a t1/2 of 74.6 ± 20.1 hours and an AUC0-24 of 2,494.8 ± 673.6 ng/g/h. The Cmax in the small intestine at 2 hours was 203.1 ± 64.0 ng/g with a t1/2 of 26.8 ± 5.6 hours and an AUC0→24 of 2,802.8 ± 588.5 ng/g/h. The Cmax in the colon at 1 hour was 77.0 ± 22.4 ng/g with a t1/2 of 21.3 ± 3.2 hours and an AUC0-24 of 715.0 ± 107.3 ng/g/h. The Cmax, t1/2 (no time provided), and AUC0-24 for EGCG in the urine were 177 ng/mL, 70.0 hours, and 3,427.9 ng/mL, respectively. 62
Toxicological Studies
The C sinensis–derived ingredients in this safety assessment are used to make tea and exposure to these ingredients in beverages would result in much greater systemic exposures than are possible from the use of cosmetic products. Consequently, systemic toxicity potential is mitigated for leaf-derived ingredients. Although data are presented on the potential for reproductive toxicity, genotoxicity, and carcinogenicity, the focus for these ingredients is primarily on the potential for local effects, such as irritation and sensitization.
Acute Toxicity
Oral—Nonhuman
Camellia Sinensis Leaf Extract
When Camellia sinensis Leaf Extract (2 g/kg; 1.94 mL/kg) was administered by gavage to Sprague Dawley (SPF) rats (n = 5/sex), the minimum lethal dose was >2 g/kg. 63 The test substance was administered after 16 hours on a hydric diet. After administration, the rats were observed for 6 hours for clinical signs and then followed for 14 days. There were no effects on weight gains and there were no mortalities. Necropsy was unremarkable. The test substance was a cold extract of green tea using water/propylene glycol (10% dry plant material) and prepared in the same manner as that used to prepare tea for drinking
The above experiment was repeated with an extract of black tea (2 g) provided to the laboratory as a brown powder with the same conclusion. 64 Decreased motility and ptosis of the eyelids was observed in all rats 1 hour after administration. Necropsies were unremarkable.
The oral LD50 of a Chinese tea extract (0.85% solids) and an oolong tea extract (1.0% solids) was >2 g/kg for mice, and the oral LD50 of a green tea extract (1.6% solids) was >2 mL/kg for rats. 65
Dermal—Nonhuman
Camellia Sinensis Catechins
The dermal LD50 of EGCG (2,000 mg/kg extract; 1,860 mg EGCG/kg; 4 mL/kg) was >1,860 mg/kg for HanBrl: WIST (SPF) rats (n = 5/sex). 39 The acute dermal toxicity test was conducted in accordance with the Organization for Economic Co-operation and Development (OECD) test guideline (TG) 402 under semi occlusion. The day before the study, the backs of the rats were clipped with an electric clipper exposing approximately 10% of the total body surface. The dressing covering the test site was removed at 24 hours and flushed with lukewarm tap water and dried. The rats were observed twice daily for 15 days. Macroscopic examination of all animals was performed at day 15.
There were no signs of systemic toxicity in any of the rats. Slight to moderate erythema was observed in all treated rats after removal of the dressing, which persisted for up to 5 days. No abnormal macroscopic findings were observed at necropsy. 39
Repeated Dose Toxicity
Inhalation—Human
Camellia Sinensis Catechins
There were no adverse effects observed when subjects (n = 36) suffering from cerebrovascular diseases inhaled catechins (3.7 mg/mL in saline; 2 mL; 43% of catechins composed of EGCG) or the vehicle (n = 33). 66 The test substance was delivered by a handheld nebulizer 3 times/day for 7 days. The sputum of the subjects all tested positive for methicillin-resistant Staphylococcus aureus (MRSA). Total catechin content was 73.0% (31% (-)-EGCG, 21% (-)-EGC, 8.6% (-)-EC, 8.6% () ECG, 2.9% (-)-GCG, and 0.8% (-)-CG)
No adverse effects were observed when tea catechins extract (10 or 20 mg/mL in saline; 2 mL; ≥30% tea polyphenol and ≥10% EGCG; assumed to be C sinensis) was inhaled using a nebulizer by subjects (n = 26) being treated for MRSA 3 times per day for 79 days. 67
No adverse effects were observed when tea catechins extract (10 or 20 mg/mL in saline; 2 mL; ≥38% tea polyphenol and ≥14% EGCG; assumed to be C sinensis) was inhaled using a nebulizer by subjects (n = 26) being treated for MRSA 3 times per day for 79 days. 68
Cytotoxicity and Cellular Effects
Camellia Sinensis Extract
A Camellia Sinensis Extract (10, 50, and 100 μg/mL) was not cytotoxic to rat pheochromocytoma (PC12) cells when exposed in vitro for 24 hours. 38 However, at higher concentrations (250, 500, 1,000 μg/mL), the extract was cytotoxic with <40% viability at the 2 highest concentrations. When the cells were incubated with the extract and hydrogen peroxide (250 μM), hydrogen peroxide poisoning was mitigated by the extract at 50, 100, and 250 μM.
A Camellia Sinensis Water Extract (as Korean green tea) had a 50% inhibitory dose (ID50) of 2.05% (0.28 mg/mL dry matter) in the inhibition of protein synthesis in Sprague Dawley rat hepatic cells. 69 The 25% effective dose (ED25) for lactate dehydrogenase (LDH) release was 1.84% (0.25 mg/mL). Another Camellia Sinensis Extract (in the form of black tea) had an ID50 of 2.50% (0.46 mg/mL) for protein synthesis and an ED25 for LDH release of 5.11% (0.94 mg/mL).
Camellia Sinensis Leaf Extract
In a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide test (n = 6), Camellia Sinensis Leaf Extract (0%, 0.00013%, 0.0006%, 0.0032%, 0.016%, 0.08%, 0.4%, 2%, and 10%) was cytotoxic at 2% and 10% to human keratinocytes. 70 Morphological modifications of the cells were observed at 0.4%. The test substance was a cold extract of green tea using water/propylene glycol (10% dry plant material).
When human keratinocytes were incubated in Camellia Sinensis Leaf Extract (0.05% and 0.1%), there was a 43% protection against oxidation when the cells were exposed to UV radiation (312 nm; 160 mJ/cm2) for 45 minutes. 70
Epigallocatechin gallate induced apoptosis at 400 and 800 μM in neonatal human dermal fibroblasts.
71
At 200 μM EGCG, a decrease in the proportion of cells in the S and G2/M phases of the cell cycle and an increase in the proportion of cells in the G0/G1 phase were observed. Regulation of the expression of phosphorylatednuclear factor-KB (pNF-
Reproductive and Developmental Toxicity
Camellia Sinensis Leaf Extract
There were no adverse effects when pregnant Wistar rats (n = 6) were administered Camellia Sinensis Leaf Extract (0, 84, 167, 501, and 1,336 mg/mL/d; in the form of black tea) by gastric intubation. 72 The caffeine content was 4.14% (865 mg for the highest dose). The test material was administered either on gestation days 1 to 7, 8 to 14, or 15 to 21. Internal examinations of the dams were conducted by laparoscopy under anesthesia on gestation day 10 (early dose groups) or 15 (mid dose groups). Pups were examined daily until the appearance of hair and the opening of the eyelids. The test doses were calculated to be equivalent to 1.5, 3, 9, and 24 cups of tea. There were no mortalities. There were no differences in number of pregnancies, number of uterine implants, number of viable implants, implantation index, preimplantation loss, postimplantation loss, gestation index, number of pups born, litter index, live birth index, and viability index compared to controls. There were no differences in length of the implants/fetus, gestation duration, cranial length, cranial diameter, and tail length of pups. There were no differences in time taken to open eyes, eruption of incisors, and appearance of fur. There were no gross morphological developmental abnormalities observed.
Camellia Sinensis Catechins
Unpublished studies were submitted to the FDA for the approval of a topical ointment as a drug that contains up to 15% Camellia Sinensis Catechins to treat warts (Table 9). 73 In oral studies, there were increased resorptions at 1,000 mg/kg/d ointment when administered on gestation days 6 to 15 in rats and decreased maternal body weights when administered on gestation days 6 to 18. Rabbit dams had decreased body weights when 1,000 mg/kg ointment was orally administered. In subcutaneous studies using rabbits, the test substance was not well tolerated; subcutaneous lesions with necrosis developed. There were spontaneous abortions, increased resorptions, and increased fetal malformations at doses as low as 4 mg/kg/d. Intravaginal administrations up to 0.15 mL/d yielded no adverse effects in rats.
Reproductive and Developmental Studies Submitted to the FDA for the Approval of an Ointment Containing 15% Catechins From Camellia sinensis. 73

Abbreviation: EGCG, epigallocatechin gallate.
When Camellia Sinensis Catechins (1,400, 4,200, and 14,000 ppm in feed; EGCG 90%, ECG ≤3.01%, GCG ≤0.12%, and other catechins ≤0.54%) were administered to pregnant Wistar (SPF) rats (n = 25) on gestation days 6 to 20, there were no adverse effects observed. 74 All rats survived treatment and there were no clinical signs. There was a transient reduction in feed consumption in the high-dose group and an increase in water consumption in the mid- and high-dose groups. There were no treatment-related macroscopic findings in the dams. There was no effect to embryo/fetal survival, fetal weights, or sex ratios.
In a 2-generation study of Camellia Sinensis Catechins (1,200, 3,600, 12,000 ppm in feed) using Sprague Dawley rats (n = 30/sex), there were no adverse effects in either generation. The rats were treated for 10 weeks and then paired for mating. The diet continued through gestation until after weaning. The dams were killed and necropsied after weaning. The pups were culled to 25/sex and the above treatment repeated with mating taking place after 8 weeks.
The offspring of the high-dose group had decreased growth rates, and there was an increase in pup loss. Reduced growth rates were observed among pups at 3,600 ppm, but only in the second generation. Both sexes of the F1 generation in the high-dose group showed decreased absolute kidney and liver weights. The F1 males had decreased spleen and prostate weights, but the females’ spleens were normal. Histological examination revealed no abnormalities. The lowest dose was considered the overall no observed adverse effects level (NOAEL). The authors derived an NOAEL of 200 mg/kg body weight per day EGCG preparation. Because dams consumed twice the amount of feed during the crucial lactation period, during which effects occurred, twice the lowest dose (ie, 2 × 100 mg/kg/d) was estimated to be the NOAEL. 74
Genotoxicity
Camellia Sinensis Catechins
Camellia Sinensis Catechins were not genotoxic in multiple in vitro and in vivo assays including Ames tests (up to 5,000 µg/plate), mouse micronucleus assays (up to 12,600 mg/kg), and other micronucleus assays in rats (up to 8,500 mg/kg; Table 10). A polyphenol mixture was lethal at 2,000 mg/kg/d to mice. In mouse lymphoma assays at concentrations >100 µg/mL, 3 assays of catechin mixtures were negative; an assay of EGCG was mutagenic at concentrations ≥125 µg/mL with metabolic activation. 73,75,76
Genotoxicity Studies of Camellia Sinensis Extracts and Constituents.
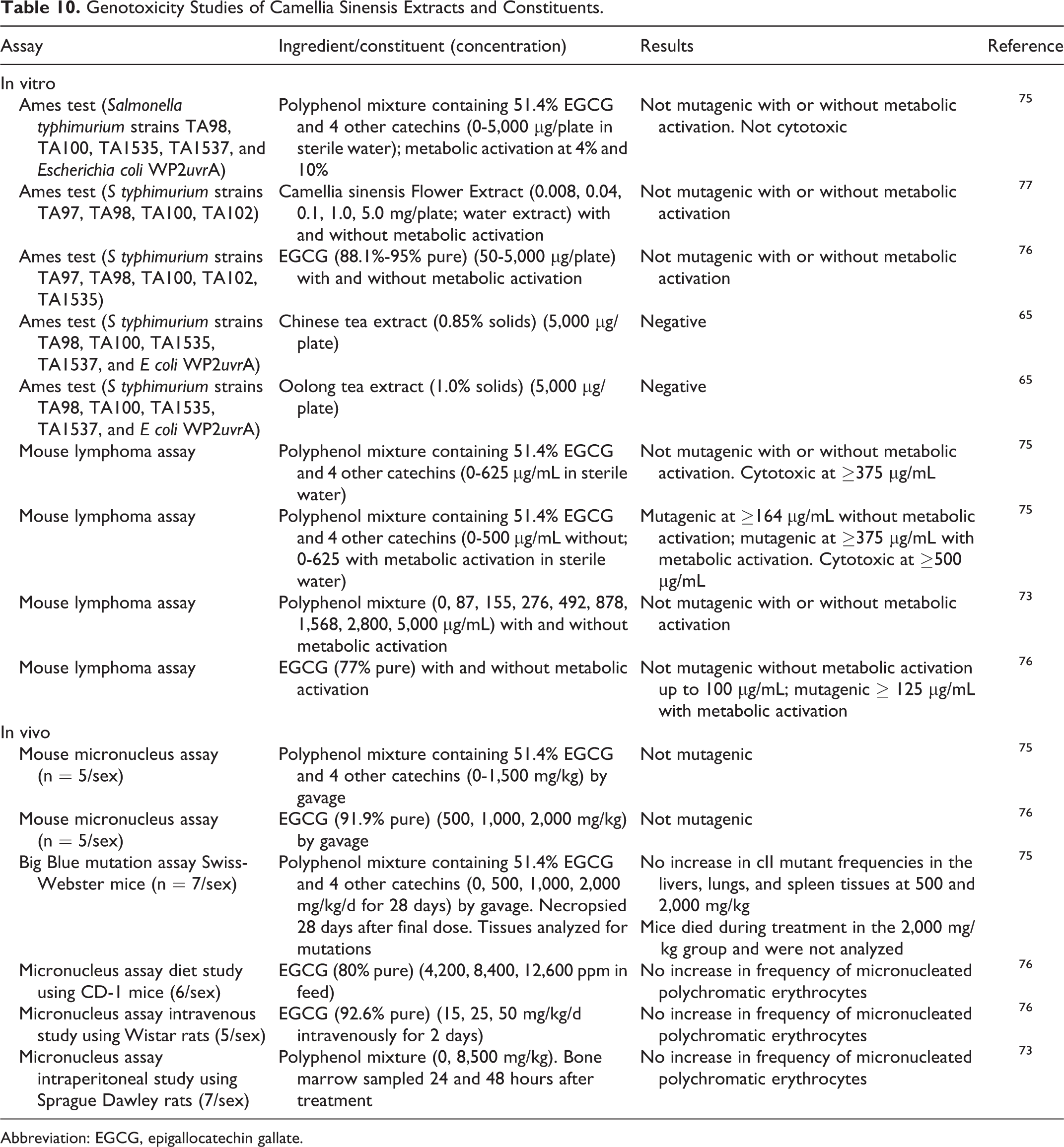
Abbreviation: EGCG, epigallocatechin gallate.
Camellia Sinensis Flower Extract
Camellia Sinensis Flower Extract was not genotoxic in an Ames test up to 5.0 mg/plate, with or without metabolic activation. 77
Camellia Sinensis Leaf Extract
Camellia Sinensis Leaf Extract was not genotoxic in 2 Ames tests up to 5,000 µg/plate. 65
Carcinogenicity
In 1997, the International Agency of Research on Cancer listed green tea in group 3, meaning that it is not classifiable according to its carcinogenicity to humans. 59
The National Toxicology Program (NTP) has completed a 2-year bioassay in rats and mice of C sinensis as green tea extract with the draft conclusion of no evidence of carcinogenic activity of green tea extract in male and female Wistar Han rats and in male B6C3F1/N mice and recommend the conclusion of no evidence of carcinogenic activity of green tea extract in female B6C3F1/N mice because the NTP panel disagreed with the NTP conclusion that occurrences of squamous cell neoplasms (squamous cell papilloma or squamous cell carcinoma [SCC]) of the tongue may have been related to treatment. 78,79 The NTP report has not been finalized at the time of this safety assessment.
A Camellia Sinensis Extract (125, 250, and 500 mg/kg/d; as green tea; 85%-95% catechins wt/wt, 55% EGCG) did not increase the incidence of neoplastic or non-neoplastic lesions in the organs and tissues of p53 transgenic heterozygous mice (n = 25). 73 The mice were treated by gavage daily for 26 weeks. p-Cresidine and water served as controls.
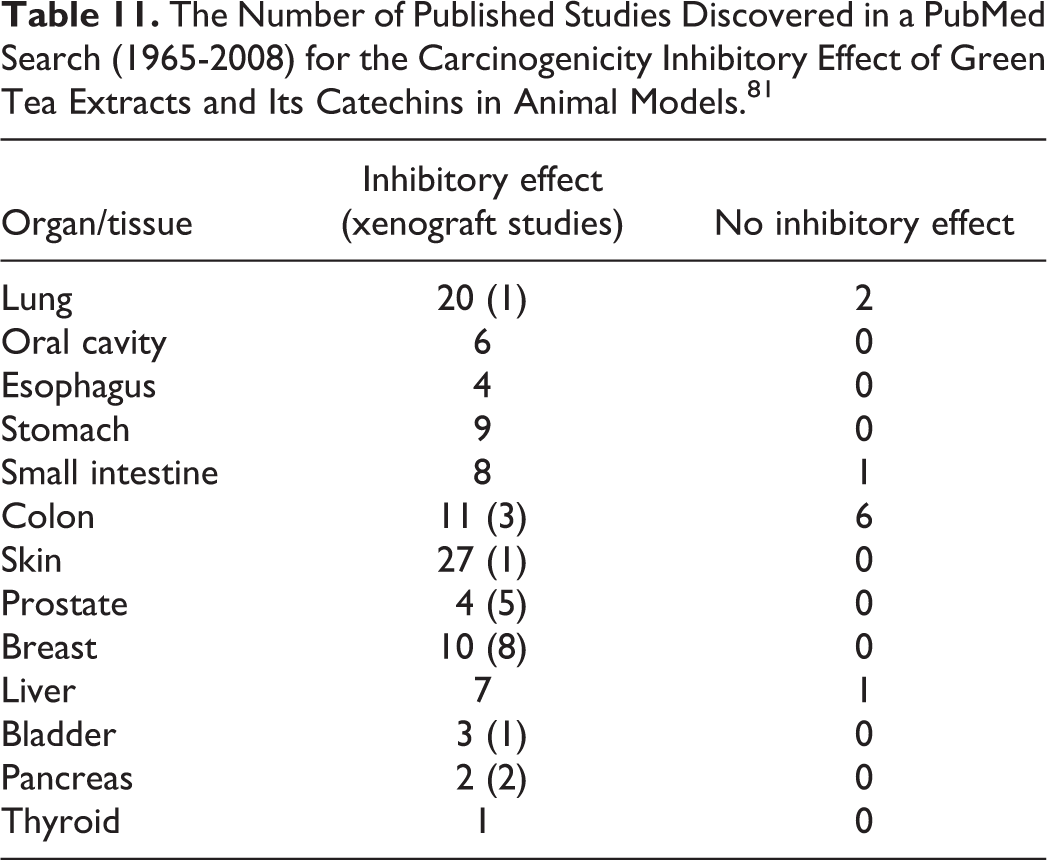
The catechins in C sinensis, especially EGCG, have been shown to have preventive and treatment effects in cancer cell lines related to cancers of the prostate, lung, skin, pancreas, breast, and ovaries. 18 There are several reviews regarding the protective effects of green tea extracts and its catechins, especially EGCG, against chemical carcinogens. 51,80 -83 According to one review, 81 there are more than 133 studies published from 1991 to 2008 on the effectiveness of C sinensis on cancers (Table 11). Inhibitory effects of tea and/or tea constituents on lung, oral, stomach, intestine, dermal, prostate, breast, liver, bladder, pancreas, and thyroid cancers were found.
The Number of Published Studies Discovered in a PubMed Search (1965-2008) for the Carcinogenicity Inhibitory Effect of Green Tea Extracts and Its Catechins in Animal Models. 81
In a population-based case–control study of residents of southern Arizona (n = 238 males, 166 females; mean age 66.6 ± 10 years), subjects who consumed black tea within the last year had fewer instances of SCC of the skin (odds ratio: 0.60) than a control group consisting of residents of Tucson (n = 226 males, 165 females; average age 66.2 ± 11.1 years). 84 Arizona was chosen because it has one of the highest risks of skin SCC worldwide. Variables were controlled for tanning ability, antioxidant intake, education, gender, smoking, and average sun exposure.
When female SKH-l mice (n = 28 or 29) were orally administered C sinensis as lyophilized green tea (0.3%, 0.9%; 3, 9 mg of tea solids/mL) in place of drinking water and exposed to mid-length ultraviolet light (UVB; 30 mJ/cm2 for 25-30 seconds) twice per week for 35 weeks, there was a decrease in the number of tumors per mouse by 35% and 94%, respectively, compared to controls exposed to UVB without C sinensis treatment. 85 The tumor volume per mouse was decreased by 49% and 97%, respectively. The composition of the green tea polyphenol fraction was: EGCG (49.5%), EGC (11.5%), ECG (11.4%), caffeine (7.6%), EC (6.1%), C (0.5%), and gallic acid (0.4%).
When female SKH-1 mice (n = 29) were orally administered C sinensis (as green tea for their drinking water; 1.25 g steeped in 100 mL hot water; approximately 4 mg tea solids/mL) UVB-induced complete carcinogenesis was inhibited. This was not the case with decaffeinated green tea. The Camellia Sinensis Extract was administered for 2 weeks before and concurrently twice per week treatment with enhanced UVB (280-320 nm; 75%-80% total energy; 30 mJ/cm2 for 25-30 seconds) exposure. There were increases in apoptosis in the epidermis observed, but no effect in non-UVB-treated normal epidermis. The authors concluded that administration of green tea and caffeine may inhibit UVB-induced carcinogenesis, at least in part, by enhancing UVB-induced apoptosis.
Oral administration of C sinensis (1.25% as green or black tea leaf extract; 1.25 g of tea leaf steeped in 100 mL water; 4.0 or 4.4 mg tea solids/mL) as the drinking water to the UVB-treated mice decreased the number of tumors per mouse by 51% and 41%, respectively. Tumor volume/mouse was decreased by 79% and 70%, respectively. The mice were treated with gradually increasing doses of the test substances for 2 weeks before the start of the twice/week treatment with UVB for 40 weeks. The mice were killed 4 weeks after the end of the UVB administration. Decaffeinated green or black Camellia Sinensis Leaf Extracts (1.25%) containing 3.6 or 3.9 mg of tea solids/mL, respectively, were less effective than regular green or black tea extracts, and decaffeinated black tea was less effective than decaffeinated green tea at inhibiting the formation of skin tumors. Adding 0.36 mg of caffeine/mL to the decaffeinated extracts either fully or partially restored the inhibitory effects on UVB-induced tumorigenesis. 85
Irritation and Sensitization
Irritation
Dermal—Nonhuman
Camellia Sinensis Leaf Extract
When Camellia Sinensis Leaf Extract (100%; 0.5 mL) was dermally administered to the clipped skin of albino New Zealand rabbits (n = 3), there were no signs of irritation. 86 The test substance was a cold extract of green tea using water/propylene glycol (10% dry plant material). It was provided to the laboratory as a brown liquid. The test substance was applied to a 2.5 cm2 gauze pad, which was then kept in contact with the skin for 24 hours using hypoallergenic adhesive tape. The test site was examined within 1 hour of removal and at 24 and 72 hours after removal.
The above experiment was repeated as stated with an extract of black tea (0.5 g), provided to the laboratory as a brown powder, with a conclusion that the test substance was a slight irritant. 87 A slight to definite erythema was observed on all treated rabbits. Cutaneous dryness and a slight decrease in skin suppleness were observed. The test sites were observed at 1, 25, and 72 hours after removing the pad.
Chinese tea extract (10%, 100%; 0.85% solids), green tea extract (100%; 1.6% solids), and oolong tea extract (10%, 100%; 1.0% solids) were not irritating to rabbit skin. 65 No further details were provided.
Camellia Sinensis Catechins
There were no signs of irritation when EGCG was administered to the clipped flanks of male New Zealand White rabbits (n = 3) for 4 hours under semi-occluded patch. 39 The tests were conducted according to the EC Commission Directive 92/69/EEC, B.4, “Acute Toxicity—Skin Irritation” and OECD TG 404 (1992). The dorsal fur of the rabbits was removed with electric clippers 24 hours before the administration of the test material. Each rabbit was treated with 0.5 g of EGCG preparation (93.4% EGCG) dissolved in 0.3 mL distilled water and applied to the skin of 1 flank using a semi-occlusive patch. After removing the patch, the skin was cleaned with water. Skin reactions and irritation effects were assessed at approximately 1, 24, 48, and 72 hours after patch removal. Adjacent areas of untreated skin from each animal served as controls.
In a preliminary study for a guinea pig maximization test, an intradermal injection of 0.09% EGCG was found to be the greatest tolerable dose. 39 A grade 3 erythema was produced, but not necrosis. At 48 hours of dermal exposure, there was no reaction in the preliminary test at concentrations up to 50%.
Dermal—Human
When C sinensis preparations (DER ranging from 0.1% to 10%) were used in dermal applications of ointments (compositions not provided) to treat genital warts, the following were among the adverse effects: erythema, pruritus, irritation/burning, pain, ulcer, edema, induration, and vesicles. 27 A full list of reported effects is provided in Table 12.
Dermal Reactions to Camellia Sinensis Leaf (Aqueous Extract or Dried Leaves) Application in Ointments for Dermal Treatment of Genital and Perianal Warts. 27

Camellia Sinensis Leaf Extract
A black tea extract (100%; 1.0% solids) was negative in a patch test (n = 100). No further details were provided. 65
Camellia Sinensis Catechins
In a trial (n = 502) of an ointment containing Camellia Sinensis Catechins (10% and 15%) for the treatment of anogenital warts, there was no irritation or other adverse effects reported. 88 The ointment was administered 3 times per day for up to 16 weeks. Observations were made during treatment and during the 12-week follow-up period, and the ointment was reported to be well tolerated.
Camellia Sinensis Leaf Water
In a patch test (n = 10) of a mascara containing Camellia Sinensis Leaf Water (30%), there were no signs of irritation at 30 minutes and 24 and 48 hours after the removing the patch. 89 The test substance was administered to the inner side of the upper arm for 24 hours using a “pin” chamber.
Mucosal—Nonhuman
Camellia Sinensis Catechins
Intravaginal administration of an ointment containing Camellia Sinensis Catechins (15%) to pregnant SD rats (n = 25) from gestation day 6 to the end of lactation caused ulceration and erosion of the vaginal mucosa with inflammation for the duration of treatment. 73 The control group (no catechins) did not exhibit damage to the vaginal mucosa. The effects resolved when treatment stopped.
Ocular
Camellia Sinensis Leaf Extract
Camellia Sinensis Leaf Extract (100%; 0.1 mL) administered to the lower conjunctival sac of the right eye of albino New Zealand rabbits (n = 3) was a slight ocular irritant. 90 There was slight irritation of the conjunctiva at 1 hour; there were no iris lesions. Two rabbits had a very slight superficial epithelial attack of the cornea. All signs of irritation were resolved within 24 hours. The test substance was a cold extract of green tea using water/propylene glycol (10% dry plant material). It was provided to the laboratory as a brown liquid. The eyes were examined 1 hour after instillation and 1, 2, and 3 days later.
The above experiment was repeated with an extract of black tea (0.1 g), provided to the laboratory as a brown powder, with the same conclusion. 91 There was a slight irritation of the conjunctiva observed at 1 hour; there were no lesions of the iris. All rabbits had a slight epithelial attack of the cornea. All signs of irritation were resolved within 48 hours.
Green tea extract (100%; 1.6% solids) was not irritating to the eyes of rabbits. No further information was provided.
Camellia Sinensis Catechins
The administration of EGCG preparation (0.093 g EGCG; 0.1 g total) into the eye of a single female New Zealand white rabbit resulted in moderate to severe irritation including reddened conjunctivae and sclera, discharge, and chemosis. 39 A slight to moderate corneal opacity affecting the whole area of the cornea was observed up to 72 hours after administration of the test material. No damage to the iris and no corrosion or staining of the eye by EGCG was observed throughout this study. The test was done in compliance with OECD TG 405. Both eyes of the rabbits were examined at the beginning of the study. The lids were briefly held together after administration; the eyes were not rinsed. The animal was observed for ocular irritancy for 17 days. Because EGCG was suspected to be an ocular irritant, a single animal was treated first and observed to recovery. Based on the results from this preliminary study, no additional rabbits were tested.
Sensitization
Dermal—Nonhuman
Camellia Sinensis Leaf Extract
Oolong tea extract (1.0% solids) was not sensitizing to guinea pigs (n not provided). 65 First induction was at 50%; second induction was at 25%. Challenge was at 5% and 10%. No further details were provided.
The IFRA reported that in a local lymph node assay, the amount of a chemical that is required to elicit a 3-fold increase in lymph node cell proliferative activity (EC3) was >1,250 µg/cm2 for Camellia sinensis Leaf Extract (as tea leaf absolute). 92 Irritation was observed at higher concentrations (not provided), so the actual EC3 could not be calculated.
Camellia Sinensis Catechins
In a sensitization assay using female GOHI (SPF) guinea pigs (n = 6), Camellia Sinensis Catechins (5%, 10%, and 30% in ethanol; 100 µL/8 cm2; 4%, 8%, and 24% EGCG) was sensitizing at challenge (1%, 3%, 5%, and 10%) as well as at a second challenge (0.1%, 0.5%, 1%, 3%, 5%, and 10%) 2 weeks later. 39 The skin sensitization assay was performed using a procedure adopted from OECD TG 406. During the induction phase of the assay, an EGCG preparation (80% EGCG) was applied to the shaved right flanks of the guinea pigs 5 days/week for 4 weeks. Control animals were treated with ethanol. Treatment sites were left open between applications. During induction, new treatment sites were chosen whenever the irritation became considerable. Immediately following the induction period, the guinea pigs were challenged with EGCG (25 µL/2 cm2 on the left flank). During the induction period, the guinea pigs were observed for signs of erythema and edema on each test site. Challenge reactions were assessed at 24 and 48 hours after application.
Irritation responses increased throughout the induction period starting with the fifth application; the 30% group had the greatest frequency of reactions. Erythema became evident in the 10% and 5% groups after the 13th and 16th administrations, respectively. In the 10% group, a slight erythema was observed in 2 of 6 guinea pigs after the 13th application, with all guinea pigs showing similar signs by the 16th application. For the 5% group, erythema was observed only for 3 days in 1 of 6 guinea pigs. Both EGCG preparations elicited positive effects in the test groups during the challenges.
Control animals showed no response after the first challenge; 1 or 2 of the 6 control guinea pigs had slight or well-defined erythema after the second challenge with 0.8% or higher EGCG. Although there was a positive dose–response effect for the challenge, it did not clearly correlate with the induction doses. There were a greater number of reactions in the 5% induction group (6 at 24 hours, 5 at 48 hours) than in the 30% induction group (2 at 24 hours, 1 at 48 hours). No mortalities or symptoms of systemic toxicity were observed in any of the guinea pigs, and body weights of the test animals were in the same range as those of the controls during the study period. 39
In a maximization test using female Himalayan strain albino guinea pigs (n = 10; control n = 5), Camellia Sinensis Catechins (0.1% in distilled water; 0.1 mL; 90% EGCG) was a sensitizer. 39 All guinea pigs had grade 3 or 4 erythema following challenge by dermal administration of Camellia Sinensis Catechins (50% in distilled water; 0.15 mL). Grade 1 erythema was observed following the first test challenge in 3 of 10 in the test group and 0 of 5 in the control group. In a second challenge 1 week later, 9 of 10 in the test group showed stronger (grade 2) erythema. No mortalities or signs of systemic toxicity were observed in any of the guinea pigs and body weights of the test group were in the same range as those of the controls during the study period.
Dermal—Human
Camellia Sinensis Leaf Extract
A facial line filler treatment product (150 µL) containing Camellia Sinensis Leaf Extract (0.86%; black tea) was not irritating or sensitizing in a human repeated insult patch test (HRIPT). 93,94 The test substance was administered 9 times on a 2 × 2 cm absorbent pad under occlusion. No reactions were observed in any of the 106 to complete the induction phase. No reactions were observed in any of the 101 that completed the challenge phase.
An eye cream containing Camellia Sinensis Leaf Extract (0.86%; black tea) was not irritating or sensitizing in an HRIPT (n = 638). The test substance was administered under occlusion. 94,95
A black tea extract (100%; 1.0% solids) was negative in an HRIPT (n = 100). No further details were provided. 65
Camellia Sinensis Leaf Extract (as tea leaf absolute) was reported to have a no observed effect level of 480 µg/cm2 in an HRIPT. 92
Camellia Sinensis Leaf Water
In an HRIPT (n = 110) of a mascara containing Camellia Sinensis Leaf Water (30%), there were no signs of irritation or sensitization. 96
Phototoxicity
Camellia Sinensis Leaf Extract
There were no signs of erythema on treated sites on the forearms of subjects (n = 6) treated with Camellia Sinensis Leaf Extracts (10%; in the form of green or black tea) and exposed to long-, mid-, and short-wavelength ultraviolet light (UVA, B, and C). 97 Freeze-dried green and black tea extracts were used to make gels with 1% carbomer solution and sodium hydroxide. These were administered to a 4 cm2 area. The controls were an untreated area and an area treated with just the gel. The arms were then exposed to UVA/UVB/UVC (UVA = 4,550 µW/cm2; UVB = 2,800 µW/cm2; UVC = 500 µW/cm2) for 2.5 minutes. Erythema was observed in the control and carbomer treated sites, but not the treatment sites.
Clinical Use
Case Studies
A 51-year-old man was diagnosed with hypersensitivity pneumonitis after undergoing catechin inhalation therapy for 1 month. 98 The diagnosis was based on the clinical course, bronchoscopy, and a challenge test. The subject was being treated for tuberculosis and had been administered the catechin inhalation therapy when MRSA was observed in his sputum. He was administered catechin-rich green tea extract solution (2 mL) dissolved in distilled water (50 mg/mL) once or twice daily using a handheld nebulizer. There were no initial symptoms, but the subject later noticed that he coughed frequently during and after inhalation of the extract.
Miscellaneous Studies
Antimicrobial Activity
Camellia Sinensis Leaf Extract
The decaffeinated methanolic extract of the leaves of C sinensis exhibited in vitro antimicrobial properties against 111 bacteria comprising 2 genera of Gram-positive and 7 genera of Gram-negative bacteria. 99 The extract was active in the range of 10 to 50 μg/mL. A few strains were sensitive at lower concentrations (5 μg/mL). In decreasing order of sensitivity, the bacterial groups were Staphylococcus aureus, Vibrio cholerae, Escherichia coli, Shigella spp., Salmonella spp., Bacillus spp., Klebsiella spp., and Pseudomonas aeruginosa.
When the above extract (30, 60 μg/mouse) was intraperitoneally administered to a Swiss strain of white mice (n = 20; control = 60), the mice were protected from a challenge of a median lethal dose injection of S typhimurium. 99 In the control group (no C sinensis), 48 mice died. In the low-dose group, 4 mice died and no mice died in the high-dose group.
Anti-Inflammatory Effects
Camellia Sinensis Extract
When saponins (0, 50, 100, and 200 mg/kg) extracted from Camellia Sinensis Leaf were orally administered to rats prior to a subcutaneous injection of carrageenan (1%; 0.5 mL) in a rat hind paw edema assay, edema in response to carrageenan was mitigated in a dose-dependent manner. 100
Other Assessments
An IFRA standard for Camellia Sinensis Leaf Extract had the following restrictions for use: lip products, 0.01%; deodorants/antiperspirants, 0.02%; hydroalcoholics for shaved skin, 0.07%; hydroalcoholics for unshaved skin, 0.2%; hand cream, 0.1%; mouthwash, 0.3%; intimate wipes, 0.04%; hair styling aids, 0.5%; and rinse-off hair conditioners, 2.4%. 92 Based on animal data and using the classification system defined by European Centre for Ecotoxicology and Toxicology of Chemicals, 101 green tea absolute was determined to be a moderate sensitizer. These limits were derived from the application of the exposure-based quantitative risk assessment (QRA) approach for fragrance ingredients; a dose of 480 µg/cm2 was the weight-of-evidence no expected sensitization level (NESIL) used to develop the IFRA standard based on a QRA for sensitization.
In a safety assessment of C sinensis as green tea used in dietary supplement products, the US Pharmacopeia Dietary Supplement Information Expert Committee concluded that when supplements containing concentrated green tea extracts are used and formulated appropriately, there are no significant safety issues with the caveat that a caution statement be included in the labeling section. 102 The caution statement warns of the potential of liver damage when concentrated green tea supplements are consumed on an empty stomach. This does not apply to C sinensis as a beverage.
Summary
This is a safety assessment of C sinensis (tea)–derived cosmetic ingredients. These ingredients are reported to function mostly as antioxidants and skin conditioning agents—miscellaneous. Although oral data are included in this report for leaf-derived ingredients, because tea leaves are ingested in food and drink, the systemic toxicity potential of the leaf-derived cosmetic ingredients is mitigated. Thus, the primary focus herein for these ingredients is on the potential for local effects, such as irritation and sensitization.
The constituents of C sinensis include amino acids, carotenoids, catechins, enzymes, flavonoids (including flavanols and flavonols), and glycosides. The concentrations of these constituents in plant parts are influenced by growing conditions, geographical location, soil conditions, and processing.
Linalool and several compounds containing the linalool moiety have been reported in the leaves ranging from 6 to 1,984 ppm and in the leaf essential oil ranging from 31,800 to 198,400 ppm in C sinensis plants. Oxidation products of linalool are dermal sensitizers. Quercetin and several compounds containing quercetin have been reported in the leaf, plant, and shoot ranging from 760 to 10,000 ppm. A positive genotoxic effect in an Ames assay has been reported, and genotoxicity was observed in in vitro tests and in some in vivo studies with intraperitoneal (IP) exposures, but results were consistently nongenotoxic in oral exposure studies using mice and rats.
Camellia Sinensis Leaf Extract was reported to be used in 1,145 leave-on, 785 rinse-off, and 36 bath cosmetic products; it was used up to 2% in leave-on products, 1% in rinse-off products, and up to 0.1% in bath products. Camellia Sinensis Leaf was reported to be used in 38 leave-on, 15 rinse-off, and 1 bath product; it was used up to 0.05% in bubble baths. Camellia Sinensis Leaf Powder was reported to be used in 11 leave-on and 10 rinse-off products; it was used up to 7% in leave-on products, up to 50% in a professional product that is diluted before use, and up to 0.01% in rinse-off products. Camellia Sinensis Leaf Water was reported to be used in 26 leave-on and 10 rinse-off products; it was used up to 30% in mascara. Camellia Sinensis Leaf Oil was reported to be used in 22 leave-on products and 11 rinse-off products. Camellia Sinensis Seed Extract was reported to be used in leave-on products up to 0.1% and in rinse-off products up to 0.0013%. There are no reported uses or concentrations of use for the rest of the ingredients.
The FDA considers C sinensis to be GRAS for use as a food additive. Catechins from Camellia Sinensis Leaf Extract penetrated pig ear skin as did caffeine in in vitro studies. Epigallocatechin gallate penetrated mouse skin in in vivo studies.
The oral LD50 for rats was >2 g/kg for Camellia Sinensis Leaf Extract as both green and black tea. The dermal LD50 of EGCG was >1,860 mg/kg for rats. There was slight to moderate erythema observed.
There were no adverse effects when up to 20 mg/mL tea catechins (assumed to be C sinensis) were inhaled for up to 79 days by human subjects.
Camellia Sinensis Extract was not cytotoxic to rat pheochromocytoma cells up to 100 μg/mL but induced apoptosis to neonatal human dermal fibroblasts at 400 and 800 μmol/L.
There were multiple studies of an ointment that contained up to 15% Camellia Sinensis Catechins. In oral studies, there were increased resorptions at 1,000 mg/kg/d ointment when administered on gestation days 6 to 15 in rats or decreased maternal body weights when administered on gestation days 6 to 18. Rabbit dams had decreased body weights when 1,000 mg/kg ointment was orally administered. In subcutaneous studies, the test substance was not well tolerated; subcutaneous lesions with necrosis developed. There were spontaneous abortions, increased resorptions, and increased fetal malformations at doses as low as 4 mg/kg/d. Intravaginal administrations up to 0.15 mL/d yielded no adverse effects in rats.
Camellia Sinensis Extract had no adverse effects when orally administered to pregnant rats up to 1,336 mg/mL/d in drinking water. There were no adverse effects observed when Camellia Sinensis Catechins, up to 14,000 ppm in feed, were administered to rats on gestation days 6 to 20.
In a 2-generation study, Camellia Sinensis Catechins up to 3,600 ppm in feed caused no clinical signs and no effects to embryo/fetal survival, fetal weights, or sex ratios. The offspring of the 12,000 ppm group had decreased growth rates, and there was an increase in pup loss. While there were some decreased organ weights, histological examination revealed no abnormalities. The NOAEL was 200 mg/kg/d EGCG.
Catechins were not genotoxic in multiple in vitro and in vivo assays including Ames tests up to 5,000 µg/plate, mouse micronucleus assays up to 12,600 mg/kg, and other micronucleus assays in rats up to 8,500 mg/kg. A polyphenol mixture was lethal at 2,000 mg/kg/d to mice. In mouse lymphoma assays at concentrations >100 µg/mL, 3 assays of catechin mixtures were negative; an assay of EGCG was mutagenic at concentrations ≥125 µg/mL with metabolic activation.
Camellia Sinensis Extract at 500 mg/kg/d was not carcinogenic to p53 mice after 26 weeks. The catechins in C sinensis, especially EGCG, have been shown to have preventive and treatment effects in cancer cell lines related to cancers of the prostate, lung, skin, pancreas, breast, and ovaries.
Camellia Sinensis Leaf Extracts, that contained 10% dry green or black tea, were not dermally irritating to rabbits. Camellia Sinensis Catechins were not irritating to rabbits with intact skin with a content of 93.4% EGCG.
In a 16-week trial of an ointment containing Camellia Sinensis Catechins at 10% and 15% for the treatment of anogenital warts, there was no irritation or other adverse effects reported. Camellia sinensis preparations with >10% plant material in ointments for dermal treatment of genital and perianal warts caused erythema, pruritus, irritation/burning, pain, ulcer, edema, induration, and vesicles in human trials.
There were no adverse effects in a human patch test of mascara containing Camellia Sinensis Leaf Water at 30%. The intravaginal administration of an ointment containing Camellia Sinensis Catechins at 15% caused ulceration and erosion of the vaginal mucosa with inflammation for 4 weeks in rats.
Camellia Sinensis Leaf Extracts from green or black tea were slight ocular irritants. The administration of a preparation containing 0.093% EGCG into the eye of a single rabbit resulted in moderate to severe irritation including reddened conjunctivae and sclera, discharge, and chemosis.
Camellia Sinensis Catechins were sensitizing to guinea pigs at 5%. In another guinea pig test, Camellia Sinensis Catechins was a sensitizer at 0.1%.
Camellia Sinensis Leaf Extract was not irritating or sensitizing in 2 HIRPTs conducted on 2 cosmetic products that contain this ingredient at 0.86%. A black tea extract was negative in an HRIPT at 100% (1% solids). In an HRIPT of a mascara product containing Camellia Sinensis Leaf Water at 30%, there were no signs of irritation or sensitization.
There was no sign of erythema at treatment sites on the forearms of subjects treated with 10% Camellia Sinensis Leaf Extract in the form of green or black tea, then exposed to UVA, UVB, and UVC. Topical treatment with green tea polyphenols at 3 mg/2.5 cm2 to human skin decreased the UVB induction of cyclobutane pyrimidine dimer formation and erythema in a dose-dependent manner. Metalloproteinase activity in cultured fibroblasts and keratinocytes decreased when incubated in EGCG at 0.01 and 0.1 μM for 24 hours before exposure to UVA radiation. Multiple in vitro and in vivo studies demonstrated UV-protective effects of Camellia Sinensis Catechins.
Camellia Sinensis Leaf Extract exhibited antimicrobial properties toward multiple bacterial species and wound-healing properties. Camellia Sinensis Leaf Extract at 100% caused no adverse effect to the skin of burned rabbits.
An IFRA standard reported tea leaf absolute (aka Camellia Sinensis Leaf Extract) to be a moderate sensitizer based on animal data. The NESIL was 480 µg/cm2.
Discussion
The essential oils, oleoresins, and natural extractives (including distillates) of tea are considered GRAS by the FDA for use as food additives. The C sinensis–derived ingredients in this safety assessment are from plants that are used extensively in the human diet. The Expert Panel (Panel) agreed that exposures to these ingredients in beverages result in much larger systemic exposures than are possible from cosmetic uses; thus, systemic toxicity cosmetic exposures to leaf-derived ingredients is not a primary concern. Reproductive toxicity, genotoxicity, and carcinogenicity data are presented in the safety assessment for these ingredients; however, the primary focus of the assessment is on the potential for local effects, such as irritation and sensitization.
The Panel acknowledged the ongoing evaluation of C sinensis-derived green tea by NTP and decided that the current data are sufficient for determining the safety of these ingredients for cosmetic use.
Oxidation products of linalool are dermal sensitizers. Linalool and several compounds containing the linalool moiety have been reported in the leaves ranging from 6 to 1,984 ppm and in the leaf essential oil ranging from 31,800 to 198,400 ppm in C sinensis plants. Also, quercetin and several compounds containing quercetin have been reported in the leaf, plant, and shoot ranging from 760 to 10,000 ppm. A positive genotoxic effect in an Ames assay has been reported, and genotoxicity was observed in in vitro tests and in some in vivo studies of IP exposures, but results were consistently nongenotoxic in oral exposure studies using mice and rats. The Panel noted that the linalool and quercetin found in C sinensis leaves and essential oil, depending on growing conditions and methods of manufacture, may or may not be found in cosmetic ingredients. Therefore, when formulating products, manufacturers should avoid reaching levels of these plant constituents, and any other constituent, that may cause sensitization or other adverse health effects. Furthermore, because product formulations may contain multiple botanical ingredients, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may lead to sensitization or other toxic effects.
The Panel recognized that every leaf extract would likely be somewhat different and that the compositions of the plant-derived ingredients addressed in this safety assessment are characterized by broad variation. Nonetheless, the available composition data represent what would be found commonly in ingredients prepared in the manner described. The Panel assumes that the manufacturing process is the same in products prepared for oral consumption and for cosmetic uses. The Panel emphasized that the conclusion of this safety assessment applies only to ingredients prepared in a manner that produces a chemical profile similar to that described in this report. Extracts not prepared in a manner that produces similar chemical profiles could be considered safe only if they have similar safety test profiles.
The Panel expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients and they stressed that the cosmetics industry should continue to use current good manufacturing practices to limit such impurities.
Aflatoxins have been detected in dried C sinensis leaves for drinking. The Panel believes that aflatoxins will not be present at levels of toxicological concern in C sinensis–derived ingredients. The Panel recognizes the US Department of Agriculture designation of ≤15 ppb as corresponding to “negative” aflatoxin content.
The Panel noted that HRIPTs using leaf-derived ingredients in test formulations at concentrations up to 100% demonstrated no signs of dermal irritation or sensitization. Based on several studies that showed photoprotective effects of C sinensis–derived ingredients, the Panel is not concerned that phototoxicity is a problem.
Because final product formulations may contain multiple botanicals, each possibly containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. Therefore, when formulating products, manufacturers should avoid reaching levels of plant constituents that may cause sensitization or other adverse health effects.
The Panel discussed the issue of incidental inhalation exposure from pump spray deodorants, suntan, and hair sprays and aerosol body and hand and foot sprays. The limited data available from inhalation studies, including short-term and chronic exposure data, suggest little potential for respiratory effects at relevant doses. The Panel believes that the sizes of a substantial majority of the particles of these ingredients, as manufactured, are larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation. Thus, the adverse effects reported using high doses of respirable particles in the inhalation studies do not indicate risks posed by use in cosmetics. These ingredients are reportedly used at concentrations up to 0.22% in cosmetic products that may be aerosolized and up to 0.0055% in other products that may become airborne. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, these ingredients are not likely to cause any direct toxic effects in the upper respiratory tract, based on the properties of C sinensis–derived ingredients and on data that show that these ingredients are not irritants.
Nevertheless, there are insufficient data to come to a conclusion of safety for the ingredients that are not derived from C sinensis leaves. To make a determination of safety for these ingredients, the data needs are: Method of manufacture Characterization of the constituents of these ingredients Concentration of use in cosmetics
Should this data be provided, and if the data are sufficient to change the conclusion, the safety assessment will be reopened and a new safety assessment developed.
Conclusion
The CIR Panel concluded that Camellia Sinensis Leaf–derived ingredients are safe as used in cosmetic products when formulated to be nonsensitizing. These are:

*Not is use. Were the ingredients in this group not in current use to be used in the future, the expectation is that each would be used in product categories and at concentrations comparable to others in this group.
The Panel also concluded that the available data or information are insufficient to make a determination that the following non-leaf-derived C sinensis ingredients are safe for use in cosmetics:

Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
L. Becker contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, P. Snyder, and L. Gill contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. B. Heldreth contributed to analysis and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.